Abstract
The lateral hypothalamic area (LHA) is essential for motivated ingestive and locomotor behaviors that impact body weight, yet it remains unclear how the neurochemically defined subpopulations of LHA neurons contribute to energy balance. In particular, the role of the large population of LHA neurotensin (Nts) neurons has remained ambiguous due to the lack of methods to easily visualize and modulate these neurons. Because LHA Nts neurons are activated by leptin and other anorectic cues and they modulate dopamine or local LHA orexin neurons implicated in energy balance, they may have important, unappreciated roles for coordinating behaviors necessary for proper body weight. In this study, we genetically ablated or chemogenetically inhibited LHA Nts neurons in adult mice to determine their necessity for control of motivated behaviors and body weight. Genetic ablation of LHA Nts neurons resulted in profoundly increased adiposity compared with mice with intact LHA Nts neurons, as well as diminished locomotor activity, energy expenditure, and water intake. Complete loss of LHA Nts neurons also led to downregulation of orexin, revealing important cross-talk between the LHA Nts and orexin populations in maintenance of behavior and body weight. In contrast, chemogenetic inhibition of intact LHA Nts neurons did not disrupt orexin expression, but it suppressed locomotor activity and the adaptive response to leptin. Taken together, these data reveal the necessity of LHA Nts neurons and their activation for controlling energy balance, and that LHA Nts neurons influence behavior and body weight via orexin-dependent and orexin-independent mechanisms.
Lesion or inhibition of a specific group of neurotensin neurons disrupts ingestive behavior and physical activity and causes adiposity, revealing neurotensin neurons as vital for energy balance.
Overweight and obesity affects millions of individuals worldwide, predisposing them to chronic conditions and increased mortality (1, 2). However, there are few efficacious interventions for the disease, and diet and exercise remain the most prescribed treatment (3). Although such lifestyle changes can induce weight loss, they are difficult to maintain and are thwarted by bodily adaptations that oppose sustained weight loss (4–7). Understanding how the brain regulates feeding and locomotor behaviors that impact body weight may suggest strategies to support weight loss and maintenance of healthy weight.
Experimental brain lesions have yielded invaluable information about how specific brain regions regulate physiology, and this method exposed the essential role of the lateral hypothalamic area (LHA) in control of body weight. Lesion of the entire LHA causes adipsia, aphagia, and impaired motivation to move that leads to profound weight loss and death (8, 9). Global LHA lesion, however, disrupts all of the neurochemically and projection-specified populations of LHA neurons, hampering determination of which specific neurons mediate facets of energy balance. For example, LHA neurons defined by their expression of the neuropeptide melanin-concentrating hormone (MCH) are activated by glucose and facilitate feeding and drinking, but they suppress locomotor activity and arousal (10–13). Separate neurons expressing the neuropeptide orexin (OX)/hypocretin are activated by signals of energy deficit and promote arousal along with feeding, drinking, and locomotor activity (14–17). LHA neurons are also defined by their expression of either γ-aminobutyric acid (GABA) or glutamate; whereas GABA neurons promote voracious ingestive behavior in part via projections to the mesolimbic dopamine (DA) system, glutamate neurons suppress motivated intake via projections to the lateral habenula (18–21). These LHA populations, however, do not explain the entirety of LHA actions, particularly how the LHA intercepts anorectic cues such as leptin or dehydration and coordinates appropriate ingestive behavior (22, 23).
The LHA also contains a large population of neurotensin (Nts)-expressing neurons that are distinct from, and more numerous than, OX and MCH neurons (24), yet their requirement for energy balance was unknown. Unlike MCH and OX neurons, LHA Nts neurons specifically respond to feeding-suppressing cues such as dehydration and the adipose derived hormone, leptin (24–26). Indeed, a subset of LHA Nts neurons coexpress the long form of the leptin receptor (LepRb), and developmental deletion of LepRb from these cells causes weight gain (24, 25). Activation of the entire population of LHA Nts neurons suppresses feeding but promotes voracious drinking and locomotor activity, indicating that LHA Nts neurons are sufficient to modify ingestive behaviors, but do so differently from the generalized ingestion induced by activating neighboring MCH and OX neurons (27). Projection mapping from LHA Nts neurons suggests two circuit mechanisms by which they might modulate behavior and energy balance. At least some LHA Nts neurons project to the ventral tegmental area (VTA) and thus modify DA release to the nucleus accumbens, where DA modulates motivated ingestive and locomotor behavior (28). Alternately, LHA Nts neurons project to and inhibit OX neurons, which impedes OX-mediated regulation of feeding, including in response to the orexigenic hormone ghrelin (24, 25). Taken together, these data suggest that LHA Nts neurons are sufficient to coordinate unique aspects of energy balance and behavior from neighboring LHA neurons, but it remained unclear whether they are necessary for maintaining normal body weight. Necessity is not a foregone conclusion, as sufficiency and necessity do not always go hand in hand in the neural regulation of behavior and energy balance. For example, chemogenetic activation of glucagon-like peptide-1 neurons in the nucleus tractus solitarius is sufficient to reduce feeding, but inhibiting these neurons causes no alteration of feeding, confirming that they are not necessary for the behavior (29). We therefore used genetic cell lesion and chemogenetic inhibition techniques to define the necessity of LHA Nts neurons in control of behavior and body weight.
Materials and Methods
Reagents
Unique experimental resources with RRIDs are cited at their first use and included in the “References.” Clozapine-N-oxide (CNO) was obtained from the National Institutes of Health as part of the Rapid Access to Investigative Drug Program funded by the National Institute of Neurological Disorders and Stroke. Aliquots of 40× CNO stock solution were made by diluting with PBS/10% β-cyclodextrin (Sigma-Aldrich, St. Louis, MO) and stored at −20°C until use. CNO stock solution was diluted to 1× with PBS just prior to use. PBS was used for vehicle (VEH) control treatments. Recombinant leptin was purchased from the National Hormone and Peptide Program (Torrance, CA). For genetic ablation experiments we used Cre-dependent adeno-associated virus (AAV) expressing Cre-dependent diphtheria toxin A (DTA; or AAV-DTA) (lox-mCherry-loxDTA-WPRE-AAV, serotype 10) (30) that was developed and provided by Dr. P. M. Fuller. AAV-GFP was used as a control (rAAV2/hSyn-DIO-eGFP) (31) and was purchased from the University of North Carolina Vector Core (Chapel Hill, NC). For chemogenetic inhibition experiments we used the Cre-dependent AAV-hM4D-mCherry [AAV-hSyn-DIO-hM4D(Gi)-mCherry] (32) developed by Bryan Roth that was purchased from the University of North Carolina Vector Core.
Animals
All procedures were approved by the Michigan State University and the University of Michigan Institutional Animal Care and Use Committees, in accordance with the Association for Assessment and Accreditation of Laboratory Animal Care and National Institutes of Health guidelines. Mice were bred in-house, maintained on a 12-hour light: 12-hour dark cycle with ad libitum access to food and water unless otherwise noted in experimental methods. Male mice were used in all metabolic and chronic studies. Male and female mice were studied in the acute leptin refeeding studies, but no significant differences were observed between sexes.
Mice
Ntscre mice (33) (The Jackson Laboratory, stock no. 017525) were crossed with homozygous Rosa26EGFP-L10a mice (34) that were provided by Dr. David P. Olson (University of Michigan), and progeny heterozygous for both alleles were used for ablation studies (Ntscre;GFP mice). Heterozygous Ntscre mice were used for other experiments, including chemogenetic inhibition studies. Genotyping was performed using standard PCR using the following primer sequences: NtsCre: common forward, 5′-ATA GGC TGC TGA ACC AGG AA-3′; Cre reverse, 5′-CCA AAA GAC GGC AAT ATG GT-3′; and WT reverse, 5′-CAA TCA CAA TCA CAG GTC AAG AA-3′; Rosa26EGFP-L10: mutant forward, 5′-TCT ACA AAT GTG GTA GAT CCA GGC-3′; wild-type forward, 5′-GAG GGG AGT GTT GCA ATA CC-3′; and common reverse, 5′-CAG ATG ACT ACC TAT CCT CCC-3′.
Generation and phenotyping of LHA Nts-ablated mice
NtsCre;GFP males (8 to 10 weeks old) received bilateral LHA stereotaxic injections of AAVs as described previously (24) using coordinates to target the LHA (A/P, 1.34; M/L, 1.13; and D/V, 5.20; in accordance with the mouse brain atlas of Paxinos and Franklin (35). One cohort of mice received 500 nL of AAV-DTA, which induces Cre-dependent expression of the cytotoxic subunit A of diphtheria toxin that leads to cell death; we refer to these as LHA Nts-ablated mice. A separate group of mice received 500 nL of AAV-GFP, which induces Cre-dependent GFP expression and leaves LHA Nts neurons intact; these are controls. Mice were single-housed and assessed weekly for food intake and body weight. At 16 weeks postsurgery, mice were analyzed for body composition using an nuclear magnetic resonance–based instrument (Minispec mq7.5, Bruker Optics). Mice were then acclimated for 24 hours in TSE cages (PhenoMaster, TSE Systems, Chesterfield, MO), followed by 4 days of continuous measurement of food and water intake, locomotor activity, and energy expenditure. Ambient temperature was maintained at 20 to 23°C and the airflow rate through the chambers was adjusted to maintain an oxygen differential around 0.3% at resting conditions. Metabolic parameters including oxygen consumption (VO2), respiratory exchange ratio (RER), and energy expenditure were assessed via indirect calorimetry by comparing O2 and CO2 concentrations relative to a reference cage. Data are presented as the mean intake, locomotor counts, and metabolic measures during the last 2 full days (48 hours) of the 4-day analysis period ± SEM. Next, mice were analyzed in TSE cages with a running wheel using a similar experimental time course (e.g., 2 days acclimation, 4 days measurement, of which the last 2 full days of data are plotted). Average running wheel time, rotations, and maximum running bout length were graphed as well as the intake, locomotor, and metabolic measures ± SEM. After metabolic analysis the mice were assessed for 1% sucrose preference as described previously (24). Preference was assessed by determining the percentage of total volume drunk that was sucrose solution. Mice were included in the final data set when post hoc analysis confirmed that injections were localized to and contained within the LHA. AAV-DTA–injected mice were only included when they exhibited ≥50% reduction in Nts-GFP neurons relative to uninjected animals and when loss of Nts-GFP neurons was confined to the LHA and not in adjacent regions. After post hoc examination and quantification, 3 of 8 control mice and 5 of 15 LHA Nts-ablated mice and were excluded from analysis due to unilateral targeting, spread of the injection site outside of the LHA, or inadequate reduction in Nts-GFP; thus, final bilateral groups consisted of n = 5 control mice and n = 10 LHA Nts-ablated mice. An equipment malfunction led to loss of TSE data for one LHA Nts-ablated mouse from the standard run, so n = 5 control and n = 9 LHA Nts-ablated mice for that data set. Owing to bottle leakage in two cages during the sucrose preference assay, n = 5 for control and n = 8 LHA Nts-ablated mice in this experiment.
For qualitative assessment of LHA cell types, a separate cohort of NtsCre;GFP mice received unilateral LHA injections of AAV-DTA and were perfused at 2 weeks, 6 weeks, and 10 weeks later (six mice in each treatment group). After post hoc analysis and exclusion of mistargeted animals, there were n = 4, 4, and 5, respectively, well-targeted mice in each group that were included in the final analysis.
Gene expression of LHA Nts-ablated mice
NtsCre mice (8 to 10 weeks old) received unilateral LHA injections of AAV-DTA. Mice were euthanized 2 or 10 weeks postsurgery to recover the uninjected (control) and AAV-DTA–injected (LHA Nts-ablated) sides of the LHA (2 weeks, n = 6; 10 weeks, n = 8). Briefly, brains were removed to an ice-cold stainless-steel coronal mouse brain matrix with 0.5-mm channels (Electron Microscopy Services). Ice-cooled razor blades were placed into the matrix to obtain a 1.0-mm-thick slice containing the OX, MCH, and Nts-enriched portion of the LHA (approximately bregma −1.34 to −0.34 mm). The LHA was microdissected from each hemisphere of the slice (with bregma −1.34 facing up) by collecting the tissue lateral and ventral to the mammillothalamic tract, above the fornix and medial to the optic tract. This tissue from each uninjected and AAV-DTA–injected side was collected in separate microcentrifuge tubes, then snap frozen on dry ice and stored at −80°C for later processing. RNA was extracted using TRIzol (Invitrogen, Carlsbad, CA) and 200-ng samples were converted to cDNA using the SuperScript first-strand synthesis system for RT-PCR (Invitrogen). Sample cDNAs were analyzed in triplicate via quantitative RT-PCR for gene expression using TaqMan reagents and an ABI 7500 (Applied Biosystems, Foster City, CA). Using GAPDH expression as an internal control, the relative mRNA expression values were calculated by the 2−∆∆Ct method and the AAV-DTA–injected values were normalized to the uninjected values. To verify AAV-DTA–mediated ablation, the fold change in Nts was compared between the ablated and intact sides of the LHA. Mice were only included in final analysis when the ablated side fold change in Nts expression was greater than 1 SD from the mean fold change of the control side. By this method, three mice were excluded from analysis (final 2 weeks, n = 5; 10 weeks, n = 6).
Chemogenetic inhibition of LHA Nts neurons
Ntscre mice (8 to 10 weeks old) were injected bilaterally with 500 μL of AAV-hM4D-mCherry into the LHA and analysis began 3 weeks later. Note that this method of virally induced expression of hM4D-mCherry provides ample receptor reserve to sustain an inhibitory response, thereby mitigating concerns regarding desensitization (36). Mice received twice-daily IP injections (8:00 to 9:00 am and 6:00 to 7:00 pm) of VEH for 2 days to acclimatize them to treatment. Mice were then separated into two groups that received twice-daily treatments for 28 days: one group received VEH (controls) and the other received CNO (0.3 mg/kg, to inhibit LHA Nts neurons). We chose this low dose of CNO and twice-daily treatment schedule to match prior studies using chronic designer receptor exclusively activated by designer drugs (DREADD)i inhibition, which were shown to provide sustained suppression and to avoid chronic desensitization (37). Additionally, we have shown that this low dose of twice-daily CNO is sufficient to sustain DREADDq-mediated activation for days (27). Mice were analyzed within TSE cages during the first 5 days and then again for the final 6 days of treatment. In the interim, mice were housed in home cages, and daily food intake and body weight were assessed via hand measuring. Mice were only included in the final study when post hoc analysis confirmed that mCherry expression was localized to and confined within the LHA on both sides of the brain. Of 14 mice injected, 2 VEH-treated and 2 CNO-treated mice were exclude as misses (chronic VEH treated, n = 7; chronic CNO-treated, n = 7).
Fasting-induced refeeding
Mice were divided into four groups and then underwent four rounds of overnight fasting followed by refeeding and treatment. During a round, each group received a different treatment (VEH/PBS, VEH/leptin, CNO/PBS, or CNO/leptin) to control for possible treatment order bias. During the four repeated rounds each mouse ultimately received every treatment and so could serve as its own control. Each experimental round was separated by a 2-day “washout” period in which mice had ad libitum food. Briefly, each experimental round began by removing chow from home cages at 6:00 pm and providing a clean cage bottom to prevent intake of food that may have fallen into the bedding. The following morning between 8:00 and 9:00 am, fasted mice were treated with IP VEH or CNO to inhibit LHA Nts neurons. Then, 30 minutes later mice were treated with PBS or leptin (5 mg/kg IP). Chow was returned 30 minutes after PBS/leptin treatment, and food intake and body weight were measured 3, 9, and 24 hours after return of food. Mice had ad libitum access to water throughout. Mice were then given at least 2 full days of recovery from fasting to ensure complete weight regain before another fasting round was initiated.
Imaging and quantification of neuronal populations
Immunohistochemistry and immunofluorescence (IF) were performed as in Brown et al. (24). Brain sections were exposed to primary antibodies for MCH (38) (1:1000) or IBA1 (39) (1:500) (see data in Supplemental Fig. 1, respectively). Sections were then exposed to species-specific biotinylated antibodies (40, 41) at a dilution of 1:100, avidin–biotinylation reaction (Vectastain, Vector Laboratories, Youngstown, OH), and diaminobenzidine detection (Sigma-Aldrich). Other antibodies were subsequently added and visualized via IF, using species-specific Alexa Fluor 488–conjugated (42, 43) or Alexa Fluor 568–conjugated antibodies (44) at a dilution of 1:200. Primary antibodies used included chicken anti-GFP (45) (1:1000), goat anti-OX (46) (1:1000), rabbit anti-DLK1 (47) (1:500), and rabbit anti-GFAP (48) (1:1000). Brain sections were analyzed using an Olympus BX53 fluorescence microscope outfitted with fluorescein isothiocyanate and Texas Red filters. Microscope images were collected using Cell Sens software and a Qi-Click 12-bit cooled camera, and images were analyzed using Photoshop software (Adobe, San Jose, CA). To verify LHA Nts ablation, the hypothalami from the 10 LHA Nts-ablated mice that underwent metabolic phenotyping and from four uninjected male NtsCre;GFP mice were coronally sectioned into four series of 30-µm sections, and one series was used to count the number of LHA Nts-GFP neurons. Briefly, three representative sections spanning the LHA were selected from each brain and the numbers of Nts-GFP neurons were counted from these sections. Because the GFP expression in AAV-GFP–injected control mice so intensely labeled fibers and soma that it obscured counting of cell bodies, the uninjected NtsCre;GFP mice were used to determine the baseline number of LHA Nts neurons. The percentage of ablation in each LHA Nts-ablated brain was determined as (no. of LHA Nts-ablated neurons − average uninjected LHA Nts-GFP neurons)/average uninjected LHA Nts-GFP neurons.
We characterized the two most OX-rich sections of the LHA (approximately bregma −1.34 and −1.46 from each mouse) to focus on the distribution of OX and coexpressed proteins. Because Nts neurons are highly codistributed with OX neurons, these sections should also contain numerous LHA Nts neurons in control mice. The neurons were counted from the two sections in the area defined as ventral and lateral to the mammillothalamic tract, as well as dorsal to the fornix. Because each brain is sectioned into four equally representative series of sections, the total number of each neuron is multiplied by four for final analysis. This counting method is not intended to represent the total number of neurons of each type within the LHA, but to provide a relative comparison between control and LHA Nts-ablated mice in the regions that should contain significant numbers of Nts-, OX-, and DLK1-containing neurons.
Statistical analysis
A Student t test (to compare two groups) or one-way ANOVA with Bonferroni posttesting (for comparisons between multiple groups) was used to determine significant differences between groups and the calculations were made using GraphPad Prism (GraphPad Software, San Diego, CA). Error bars depict ±SEM. Differences were considered significant for P < 0.05 (*P < 0.05; **P < 0.01; ***P < 0.001).
Results
Loss of LHA Nts neurons causes obesity
To determine whether LHA Nts neurons are required for the control of energy balance, we genetically lesioned them (49, 50) (Fig. 1A). Adult NtsCre;GFP mice were injected unilaterally in the LHA with AAV-DTA, thus inducing the cytoxic DTA in the Cre-expressing Nts cells of the LHA. Adjacent LHAs lacking Cre do not express DTA and hence remain intact. We confirmed that AAV-DTA resulted in ablation of GFP (Nts) neurons compared with the control side by 2 weeks postsurgery, whereas adjacent MCH neurons remained intact (Fig. 1B). Next, we generated cohorts of mice bilaterally injected in the LHA with AAV-DTA (LHA Nts-ablated mice) or with AAV-GFP, which drives Cre-dependent expression of GFP so that Nts neurons remain intact (control). We confirmed that this method resulted in a significant depletion of LHA Nts-GFP neurons in LHA Nts-ablated mice by 16 weeks postinjection (Fig. 1C). Because DTA-mediated cellular degeneration and clearance can drive inflammation that might itself impact feeding and body weight, we also assessed inflammation in AAV-DTA–injected mice (Supplemental Fig. 1). At the peak of DTA-mediated neuronal ablation and debris clearance (2 weeks postinjection) the LHA Nts-ablated mice exhibited increases of inflammatory markers GFAP and IBA1 within the LHA. Similar to other models of DTA-mediated ablation (51), this inflammatory signature subsided over the subsequent weeks. Hence, whereas there is an inevitable increase in inflammation with all DTA-mediated ablation models at early time points, changes in metabolism or behavioral phenotypes at later time points are not attributable to residual or ongoing inflammation.
Figure 1.
Genetic ablation of LHA Nts neurons increases adiposity. (A) To study the requirement for LHA Nts neurons, we compared NtsCre;GFP mice that received bilateral LHA injections of AAV-GFP (control, leaves LHA Nts neurons intact) or AAV-DTA to specifically ablate LHA Nts neurons (control, n = 5; LHA Nts-ablated, n = 9). (B) Proof of the genetic ablation method showing that AAV-DTA depletes Nts-GFP neurons but leaves MCH neurons intact compared with the control LHA. MCH neurons were detected via IF. Magnification ×40. (C) Number of Nts-GFP neurons in uninjected NtsCre;GFP mice (n = 4) and LHA Nts-ablated mice (n = 10) 16 wk after injection. (D) Percentage weight change in control mice (green line) and AAV-DTA–injected LHA Nts-ablated mice (orange line) during 16 wk. (E and F) At 16 wk postinjection, LHA Nts-ablated mice are visibly obese compared with control mice. A quarter is included for size reference. (G) Total body weight and (H) body weight gain were similar between control and LHA Nts-ablated mice. (I) LHA Nts-ablated mice have a significantly higher percentage of body fat compared with control mice. (J) Percentage lean mass and (K) percentage body fluid are similar between control and LHA Nts-ablated mice. (L) Correlation of the percentages of LHA Nts ablation and body fat. Quantitative data were analyzed by an unpaired test, and graphed data represent mean ± SEM. For the graphs in (G)–(L) each point represents a mouse. For (D)–(L): control, n = 5; LHA Nts ablated, n = 10. *P < 0.05; **P < 0.01; ****P < 0.0001. 3V, third ventricle; f, fornix; mt, mammillothalamic tract.
Interestingly, the control and LHA Nts-ablated mice exhibited comparable weight changes at 2 weeks postinjection, when AAV-DTA–mediated ablation is complete and inflammation is at its peak. Body weights began to diverge 5 weeks postinjection (Fig. 1D). By 16 weeks postinjection the LHA Nts-ablated mice appeared visibly larger than the controls (Fig. 1E and 1F). Although the total body weight and weight gain of LHA Nts-ablated mice tended higher than those of controls, the differences were not statistically significant (Fig. 1G and 1H). However, the LHA Nts-ablated mice accumulated a significantly higher percentage of body fat, essentially double that of the control mice (Fig. 1I). Control and LHA Nts-ablated mice had comparable percentages of lean mass and body fluid (Fig. 1J and 1K). Because body fat takes up more space than lean or fluid mass, the expansion of body fat in LHA Nts-ablated mice may account for why they appear visibly larger compared with controls (Fig. 1E and 1F). The weak correlation between the percentages of LHA Nts ablation and body fat indicates that the small variance in LHA Nts neuronal ablation did not account for the degree of body fat composition. Overall, these data suggest that an intact population of LHA Nts neurons is required to prevent excess adiposity and for maintaining appropriate body weight.
Loss of LHA Nts neurons blunts water intake and locomotor activity
Next, we sought to determine the cause of the obesity in LHA Nts-ablated mice and whether it resulted from disrupted ingestive behavior and/or energy expenditure. LHA Nts-ablated mice ate similar amounts of chow as did control mice during 48 hours, and thus their obesity is not due to excess caloric intake (Fig. 2A). Interestingly, LHA Nts-ablated mice drank less water than did control mice, suggesting some disruption of ingestive behavior (Fig. 2B). Additionally, during 48 hours of continuous measurements the LHA Nts-ablated mice displayed reduced ambulatory locomotor activity relative to control mice (Fig. 2C) as well as decreased VO2, energy expenditure, and a trend for lower carbon dioxide production (VCO2) (Fig. 2D–2F). Notably, the reduced locomotor activity of LHA Nts-ablated mice is confined to the dark cycle, when mice are generally awake and active (Supplemental Fig. 2). LHA Nts-ablated and control mice exhibited comparable locomotor activity during the light phase and ramped up locomotor activity at the light–dark transition (Supplemental Fig. 2), suggesting that they retain some measure of circadian-regulated locomotor activity. The RER was not different between groups (data not shown), suggesting that substrate usage is not changed with loss of LHA Nts neuronal function. Taken together, these data suggest that loss of LHA Nts neurons diminishes locomotor activity and energy expenditure and thus impair energy homeostasis to promote the development of obesity.
Figure 2.
Loss of LHA Nts neurons blunts water intake and locomotion. Control and LHA Nts-ablated mice were analyzed in TSE metabolic cages for (A) chow intake, (B) water intake, (C) ambulatory locomotor activity measured as beam breaks, (D) VO2, (E) VCO2, and (F) RER. Data were analyzed by an unpaired test, and graphed data represent mean ± SEM during 48 h. Control, n = 5; LHA Nts-ablated, n = 9. *P < 0.05. (Although 10 LHA Nts-ablated mice were analyzed, an error in one of the TSE cages prevented data acquisition from 1 mouse.)
Loss of LHA Nts neurons decreases motivated activity and adaptive energy balance
One of the main projection targets of LHA Nts neurons is the VTA DA cell group, a population known to influence the motivation to ingest and move (52). We therefore reasoned that loss of LHA Nts neurons, in addition to disrupting homeostatic energy balance, might specifically impair motivated behaviors that impact body weight. To examine this, we gave control and LHA Nts-ablated mice access to running wheels within metabolic cages, allowing us to examine their nonobligatory, motivated locomotor activity (wheel running) and its effects on other metabolic parameters. As expected, control mice with intact LHA Nts neurons engaged the running wheels, but the LHA Nts-ablated mice exhibited vastly reduced running time, wheel rotations, and shorter run bouts during 48 hours (Fig. 3A–3C). Some LHA Nts-ablated mice displayed nearly complete lack of wheel activity (see Fig. 3A–3C, data points near 0); we verified that the wheels in their cages worked, and thus the low values reflect that these mice simply did not use the wheels. The wheel-running control mice consumed significantly more chow and water than did LHA Nts-ablated mice (Fig. 3D and 3E). The wheel-running control mice also displayed increased feeding and drinking compared with their baseline intake (Fig. 2A and 2B), which reflects the additional energy and fluid intake necessary to support elevated physical activity. Similarly, heat energy expenditure, VO2, and VCO2 were higher in control mice compared with LHA Nts-ablated mice, consistent with their increased energy output via wheel running (Fig. 3F–3H). RER remained similar between groups, indicating that substrate usage is not altered even with the higher energy demands of the wheel-running control mice (Fig. 3I). These data reveal that loss of LHA Nts neurons reduces volitional motivated locomotor activity and associated energy expenditure, whereas mice with intact LHA Nts neurons choose to engage in exercise-like behavior. Thus, LHA Nts neurons may be necessary for the motivation to exercise, which in tandem with a healthy diet can protect against obesity (53).
Figure 3.
Motivational locomotor activity is blunted by loss of LHA Nts neurons. Control and LHA Nts-ablated mice were analyzed in TSE metabolic cages containing a running wheel to measure volitional locomotor activity (wheel running). (A–C) Compared with control mice, LHA Nts-ablated mice spent significantly less time running on wheels, with fewer wheel rotations and shorter running bouts. (D and E) LHA Nts-ablated mice consumed less food and water relative to the more highly active control mice. (F–I) Consistent with their decreased wheel running, LHA Nts-ablated mice exhibit (F) lower heat/energy expenditure, (G) VO2, and (H) VCO2 relative to controls. (I) RER was similar between groups, indicating no divergence in energy substrate use. Taken together, these data show that loss of LHA Nts neurons blunts volitional locomotor activity, and therefore the adaptive ingestive and metabolic increases necessary to support increased movement. Data were compared by an unpaired test. Graphed data represent mean ± SEM during 48 h. Control, n = 5; LHA Nts-ablated, n = 10. *P < 0.05; **P < 0.01.
Loss of LHA Nts neurons disrupts drinking but not sucrose preference
LHA Nts neurons project to and release Nts into the VTA, a brain region implicated in modifying the motivation to work for food rewards (e.g., DA-dependent “wanting”) and their hedonic value (e.g., opioid-dependent “liking”) (54, 55). Our data suggest that LHA Nts neurons are required for motivated wheel-running behavior (Fig. 3) that is known to be DA-dependent (56), but we also examined whether these neurons are required for hedonic intake. We therefore tested control and LHA Nts-ablated mice via a two-bottle sucrose preference assay, which is well established to identify hedonic intake (57). Prior to receiving sucrose, LHA Nts-ablated mice consumed less water than did control mice (Fig. 4A), consistent with our findings from metabolic cages (Fig. 2B). Whereas LHA Nts-ablated and control mice both increased intake of sucrose over water and exhibited similar sucrose preference, the LHA Nts-ablated mice consumed less total volume of each liquid compared with control mice (Fig. 4B–4D). Taken together, these data implicate a role for LHA Nts neurons in promoting drinking behavior, but they are not required for discrimination of palatable liquids or hedonic consumption.
Figure 4.
Loss of LHA Nts neurons decreases drinking but not sucrose preference. Control and LHA Nts-ablated mice were given a two-bottle choice sucrose preference test to assess hedonic drive. (A) Prior to sucrose access, LHA Nts-ablated animals drank less water than did controls. (B) When given access to sucrose, both groups exhibited similar sucrose preference, but (C) LHA Nts-ablated mice drank less total volume compared with control mice (D) with reductions in both water and sucrose intake. These data show that loss of LHA Nts neurons does not decrease hedonic preference for sucrose, but it does lead to a decrease in overall drinking. Data were compared by an (A–C) unpaired t test or (D) ordinary two-way ANOVA with a Sidak posttest. Graphed data represent mean ± SEM during 96 h. Control, n = 5; LHA Nts-ablated, n = 8. **P < 0.01; ****P < 0.0001. (Although 10 LHA Nts-ablated mice were analyzed for sucrose preference, bottle leakage in two of the cages prevented accurate measurement.)
Lesion of LHA Nts neurons precedes reduction in OX expression
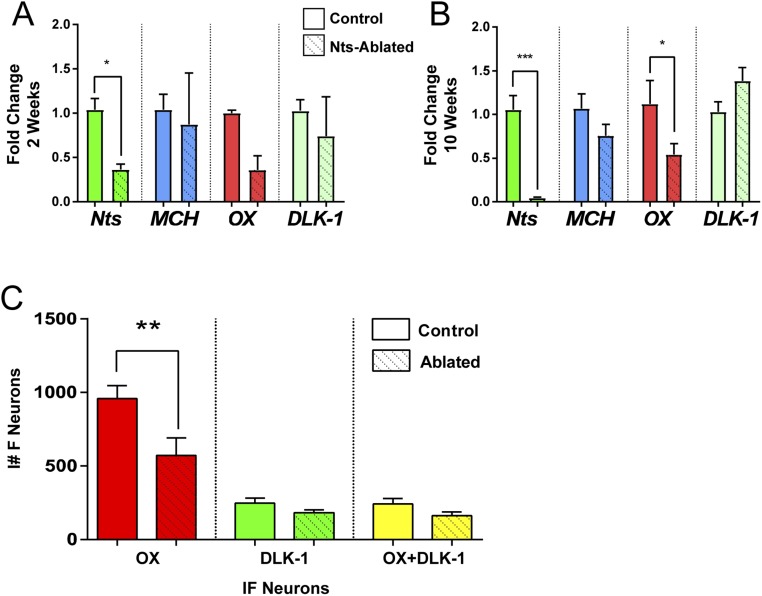
Some LHA Nts neurons synaptically modulate OX neurons (24, 58, 59), and thus loss of LHA Nts neurons might impair OX function over time and indirectly contribute to the behavioral derangements and obesity of LHA Nts-ablated mice. We therefore investigated the impact of LHA Nts ablation at 2 weeks postinjection, when AAV-DTA–mediated ablation should be complete, yet control and LHA Nts-ablated mice exhibit similar weight gain at this time point (Fig. 1D). We also examined mice 10 weeks after injection, when LHA Nts-ablated mice show a trend toward increased weight gain compared with controls (Fig. 1D). We evaluated NtsCre;GFP mice that were unilaterally injected with AAV-DTA for their distribution of Nts-GFP neurons and MCH and OX peptide expression via IF; in these experiments, the contralateral uninjected LHA serves as an intact control (Fig. 5A). As observed previously (Fig. 1B), AAV-DTA injection depletes LHA Nts-GFP neurons at 2 weeks relative to the control side but the MCH population remained intact (Fig. 5B and 5C). OX-IF appeared somewhat reduced, but bright, highly expressing OX neurons were still apparent at 2 weeks postinjection (Fig. 5B and 5C). Depletion of LHA Nts-GFP neurons was also evident at 10 weeks after AAV-DTA injection compared with the control side. MCH-IF was somewhat reduced at 10 weeks postinjection, but OX-IF was more obviously diminished, with fewer brightly labeled OX-expressing cell bodies compared with the control LHA or 2 week–ablated mice (Fig. 5D and 5E). These data hinted that long-term loss of LHA Nts neurons could lead to a reduction in MCH and OX neuropeptide expression and/or loss of these neurons. To investigate these possibilities, we counted all Nts-GFP cells, and also all IF-detected MCH or OX neurons regardless of their brightness, to ensure inclusion of cells that may have downregulated MCH or OX peptide expression. Cell counts confirmed a significant reduction in Nts-GFP neurons in the ablated LHA compared with the control side at 2 and 10 weeks (Fig. 4F). The number of IF-detected MCH neurons was similar in the ablated and control LHA at 2 weeks, with a nonsignificant decrease in MCH-IF–detected neurons by 10 weeks. In contrast, the number of OX-IF neurons was similar in the ablated and control LHA at 2 weeks but was significantly decreased at 10 weeks (Fig. 5F). Taken together, these data confirm that the genetic lesion method causes rapid, sustained, and cell-specific loss of LHA Nts neurons, because the LHA Nts neurons were depleted by 2 weeks postinjection, at which point adjacent MCH and OX neurons remained intact. However, the reduction in OX-IF–labeled cells at 10 weeks suggests that prolonged loss of LHA Nts neurons may lead to impairments in OX expression and cell function.
Figure 5.
Distribution of LHA neurons after LHA Nts lesion. (A) Brains from NtsCre;GFP mice with unilateral LHA injections of AAV-DTA were examined via IF to detect Nts-GFP, MCH, and OX neurons at 2 and 10 wk postinjection. The uninjected LHA served as an intact control. (B and C) At 2 wk postinjection there was a dramatic decrease in Nts-GFP in the Nts-ablated LHA compared with the control hemisphere, but MCH and OX-IF was comparable between sides. (D and E) Decreased Nts-GFP was still apparent in the Nts-ablated LHA at 10 wk postinjection. MCH-IF was also present, but OX IF was decreased compared with the control LHA. (F) Counts of LHA Nts-GFP and MCH and OX-IF neurons in the control and ablated LHA were compared by at each time point by an unpaired test. These data indicate that permanent LHA Nts ablation occurred by 2 wk postsurgery, and sustained loss of LHA Nts neurons led to loss of OX-IF. Graphed data represent mean ± SEM. 2 wk, n = 4; 10 wk, n = 5. *P < 0.05. (B–E) The first images were collected at ×10 magnification. Inset images were digitally magnified from ×20 images of the same brain section. mt, mammillothalamic tract.
Because LHA Nts neurons act, in part, via modulation of OX neurons, sustained loss of LHA Nts input could disrupt the gene expression and/or the viability of OX-expressing neurons. To examine the repercussions of LHA Nts lesions on transcription, we assessed gene expression in NtsCre mice that received AAV-DTA in one side of the LHA compared with the other side, which was uninjected. MCH and OX expression is identical in the LHA of uninjected and AAV-GFP–injected mice (Supplemental Fig. 3), and hence the uninjected LHA is a reasonable internal control for this experiment. As expected, Nts expression was significantly decreased in the Nts-ablated LHA at 2 and 10 weeks after AAV-DTA injection. MCH expression remained unchanged between the control and Nts-ablated LHA at both time points, consistent with our previous observation of intact MCH-IF in LHA Nts lesioned mice (Fig. 5F). Conversely, we observed a trend for decreased OX expression in the Nts-ablated LHA at 2 weeks postinjection and a significant OX reduction by 10 weeks (Fig. 6A and 6B). This downregulation of OX could represent a specific consequence of loss of LHA Nts signaling input but could also result from general suppression of transcription in OX neurons and/or decreased viability of the OX-expressing cells. To assess these possibilities, we measured DLK1 expression in the LHA because the DLK1 protein is coexpressed by many OX neurons and can be detected specifically within OX-containing soma via IF, but is not detected in non-OX LHA neurons (60, 61). DLK1 expression was similar in the intact and Nts-ablated LHA at 2 and 10 weeks postinjection, indicating that loss of LHA Nts neurons does not generally impair gene expression within OX neurons. We also examined DLK1 protein expression via IF, which weakly labels cell bodies in the LHA. This analysis revealed similar numbers of DLK1 and OX plus DLK1–expressing neurons in the Nts-ablated and control LHA, suggesting that at least some OX neurons remain intact (Fig. 6C; Supplemental Fig. 4). In contrast, we observed significantly fewer neurons with OX-IF in the Nts-ablated LHA compared with the control side. Taken together, these data indicate that lesion of LHA Nts neurons specifically downregulates OX transcription, which reduces OX peptide and hence impairs detection of the “OX neurons” via OX-IF. This is not necessarily indicative of mass OX neuronal cell death, however; the retention of DLK1 protein known to be coexpressed within OX neurons confirms that at least some of them remain intact. Thus, loss of LHA Nts neurons leads to decreased OX expression without disrupting all function and viability of the OX population. Hence, it is possible that diminished or absent OX peptide could have contributed to the behavioral and energy balance impairments observed in LHA Nts-ablated mice.
Figure 6.
Effect of LHA Nts lesion on LHA gene and protein expression. NtsCre mice received unilateral LHA injections of AAV-DTA, and brains were assessed at 2 and 10 wk postinjection. The uninjected LHA served as an intact control. (A and B) Fold changes in gene expression for Nts, MCH, OX, and DLK1 (which is coexpressed within some OX neurons) relative to the control LHA. Nts is decreased in the Nts-ablated vs control LHA at both 2 and 10 wk, but OX is only decreased at 10 wk. 2 wk, n = 5; 10 wk, n = 6. (C) Counts of OX-IF– and DKL-1–IF-labeled neurons at 10 wk postinjection (control and ablated, n = 4 each). Consistent with the gene expression data, there is a significant decrease in OX-IF in the Nts-ablated LHA compared with the control. The similar numbers of DLK1 and OX plus DLK1 neurons in the control and Nts-ablated LHA suggest that at least the OX plus DLK1 neurons remain intact. Collectively, these data suggest that loss of LHA Nts neurons disrupts LHA OX neurons, leading to specific downregulation of the OX transcript and peptide. *P < 0.05; **P < 0.01; ***P < 0.001.
Chronic inhibition of LHA Nts neurons decreases locomotion
Our LHA Nts-ablation data confirm that LHA Nts neurons are necessary for proper regulation of OX neurons, and that prolonged loss of LHA Nts neurons disrupts Nts, OX, and energy balance. Although these findings reinforce the interdependence of Nts and OX neurons (24), they impaired determination of which facets of the LHA Nts-ablated phenotype are specifically due to loss of LHA Nts neurons (rather than indirect effects due to loss of OX). To circumvent this issue, we used chemogenetics (DREADD technology) to selectively inhibit LHA Nts neurons for 4 weeks, thereby avoiding long-term structural changes that might impair OX neurons and obfuscate LHA Nts effects. Adult NtsCre mice were bilaterally injected with Cre-inducible AAV-hM4Di-mCherry in the LHA to drive expression of inhibitory DREADD-mCherry selectively on LHA Nts-GFP neurons (Fig. 7A and 7B). In a pilot slice electrophysiology experiment (data not shown) we verified that CNO indeed hyperpolarized hM4Di-mCherry–expressing LHA Nts neurons, consistent with other reports using this inhibitory strategy (37, 62). We then treated mice with either VEH or the DREADD agonist CNO to inhibit LHA Nts neurons and examined how acute (24 hour) or chronic (4 week) inhibition impacted energy balance (Fig. 7C and 7D). Neither acute nor chronic CNO treatment altered body weight, adiposity, feeding, or drinking relative to VEH-treated controls (Fig. 7E–7H). However, locomotor activity was significantly diminished by acute and chronic CNO treatment. The finding that acute and chronic CNO treatment elicited identical reductions in locomotor activity indicates that this treatment paradigm yielded similar, sustained inhibition at both time points. Although not significant, CNO-treated mice also showed a trend toward decreased ambulatory activity speed. Despite the reduction in locomotor activity, CNO-mediated inhibition of LHA Nts neurons did not alter respiration rates (VO2, VCO2) or substrate usage (RER) (Fig. 7K–7M). These data indicate that inhibition of LHA Nts neurons specifically blunts locomotor activity.
Figure 7.
Inhibition of LHA Nts neurons blunts locomotion. (A) NtsCre;GFP mice were injected with AAV-HM4D-mCherry into the LHA, which (B) induced Cre-dependent expression of DREADDi-mCherry (shown in blue) within LHA Nts-GFP neurons (shown in green). The large image was collected at ×10 magnification. Insets were digitally magnified from ×20 images of the same sections. (C) Treatment with the DREADD agonist CNO allowed for chemogenetic inhibition of LHA Nts neurons, (D) including acute (24 h) and chronic inhibition (4 wk). CNO-mediated LHA Nts inhibition did not alter (E) body weight, (F) body fat, (G) chow intake, or (H) water intake compared with VEH-treated mice and were not different between CNO and VEH groups acutely or chronically. (I) Acute and chronic LHA Nts inhibition decreased locomotor activity as assessed by beam breaks but did not significantly alter (J) speed of activity, (K) VO2, (L) VCO2, or (M) RER. Data were compared by two-way ANOVA with Sidak posttests. Graphed data represent mean ± SEM, and (G)–(M) reflect data gathered during 24 h. Data are from VEH-treated (n = 7) or CNO-treated (n = 7) mice. Data were not included for one CNO-treated mouse due to an error during TSE cage analysis. *P < 0.05. 3V, third ventricle; mt, mammillothalamic tract.
Inhibition of LHA Nts neurons does not reduce OX
We next examined the distribution of Nts-DREADDi-mCherry neurons and OX-IF in chronically CNO-treated mice to determine whether prolonged inhibition of LHA Nts neurons impaired OX expression similar to the effect of LHA Nts ablation. Nts-DREADDi-mCherry was robustly expressed in the LHA from mice treated for 4 weeks with VEH or CNO (Fig. 8A). We also observed a similar distribution of OX-IF–labeled cells in both groups (Fig. 8B). These data suggest that, unlike prolonged LHA Nts lesion, chronic inhibition of LHA Nts neurons does not derange OX peptide expression. Thus, the inhibition model presumably reflects the behaviors governed by LHA Nts neurons alone, without effects mediated by disruption of OX neurons.
Figure 8.
Intact OX-IF in LHA Nts-inhibited mice. NtsCre mice injected in the LHA with AAV-HM4D-mCherry were treated with VEH or CNO to inhibit LHA Nts neurons. Brains were examined for AAV-induced Nts-DREADDi-mCherry (cyan) and OX-IF (red). (A) Nts-DREADDi-mCherry and OX-IF were robustly expressed within the LHA but did not overlap, consistent with the separate populations of LHA Nts and OX neurons (merged panel). (B) The distribution of Nts-DREADDi-mCherry and OX-IF was similar after CNO-mediated inhibition of LHA Nts neurons, suggesting that OX expression was not interrupted by this manipulation. CNO, n = 8; VEH, n = 7. (A and B) The first panels were collected at ×10 magnification. Insets were digitally magnified from ×20 images of the same sections. f, fornix; mt, mammillothalamic tract.
Inhibition of LHA Nts neurons disrupts leptin sensitivity
As loss of LHA Nts neurons caused subsequent disruption of OX neurons in LHA Nts-ablated mice, we could not determine how LHA Nts neurons selectively contribute to adaptive energy balance. However, because OX neurons are preserved in mice with chronically inhibited LHA Nts neurons, they can be used to study the specific requirement for LHA Nts neuronal activity in mediating adaptive energy balance without the influence of OX neurons. Because ∼15% of LHA Nts neurons express LepRb, we reasoned that suppression of LHA Nts neural activity would blunt the anorectic response to leptin (24). To test this, we fasted mice expressing DREADDi-mCherry in LHA Nts neurons overnight to increase their motivation to eat. Mice were then treated with either VEH or CNO to inhibit LHA Nts neurons just prior to injection with PBS or leptin, and then chow was returned to their cages. The 2 days between each fasting/treatment session were sufficient to “wash out” the prior treatment and for mice to regain any lost weight, verified by their comparable starting weights prior to each test (Fig. 9G and 9H). As expected, leptin decreased refeeding and prevented weight gain even in the hungry VEH-treated mice at 3 and 9 hours posttreatment (Fig. 9A–9F). In comparison, CNO-mediated inhibition of LHA Nts neurons blunted the anorectic effect of leptin at 3 and 9 hours posttreatment (Fig. 9A and 9B). Direct comparison of the leptin-treated mice suggested a trend toward increased feeding with CNO-induced inhibition of LHA Nts neurons, although this did not reach the level of significance (Fig. 9D and 9E). At 3 and 9 hours the CNO-treated mice gained less body weight in response to leptin treatment compared with PBS treatment, suggesting that leptin-mediated effects via non-LHA Nts neurons can adequately restrain weight regain (Fig. 9G, 9H, 9J, and 9K). Both VEH and CNO-treated mice resumed normal feeding and body weight 24 hours after treatment, indicating that LHA Nts inhibition does not cause permanent impairments in homeostatic energy balance (Fig. 9C, 9F, 9I, and 9L). These data confirm that inhibition of LHA Nts neurons impairs the adaptive anorectic response to leptin. Although they are not the sole mediators of leptin-mediated feeding restraint and weight loss, LHA Nts neurons are required for the appropriate coordination of leptin and reduced feeding behavior, as might occur in response to elevated adiposity. Loss of function via LHA Nts neurons may therefore impair both locomotor activity and appropriate adaptive feeding in response to peripheral energy cues that could, over time, potentiate weight gain.
Figure 9.
Chemogenetic inhibition of LHA Nts neurons blunts the adaptive response to leptin. NtsCre mice expressing AAV-HM4D-mCherry in the LHA were fasted overnight. The next morning, mice were treated with either VEH or CNO to inhibit LHA Nts neurons, followed by treatment with either PBS or leptin (5 mg/kg IP). Food was then returned to the cage and chow intake and body weight were assessed at 3, 9, and 24 h. Mice were given 2 d to recover any lost body weight and to wash out treatments prior to retesting, such that each mouse was exposed to all four treatment combinations. A crossover study design was used so that each mouse received all four treatments (n = 10). (A–C) Comparison of feeding at 3, 9, and 24 h in mice that were VEH treated (no inhibition of LHA Nts neurons) and CNO treated to inhibit LHA Nts neurons. (D and E) Comparison of the change in feeding at 3, 9, and 24 h of mice treated with VEH and then CNO to inhibit LHA Nts neurons in response to PBS treatment (control, black lines) or leptin (purple lines). (G–I) Comparison of body weight gain at 3, 9, and 24 h in mice that were VEH treated (no inhibition of LHA Nts neurons) and CNO treated to inhibit LHA Nts neurons. (J and K) Comparison of the change in body weight at 3, 9, and 24 h of mice treated with VEH and then CNO to inhibit LHA Nts neurons in response to PBS treatment (control, black lines) or leptin (purple lines). (M) Body weights of mice were similar prior to each treatment and (N) prior to each of the four fasting bouts in the crossover experimental design, indicating that the 2-d recovery period between tests was sufficient for mice to regain any lost weight. Data were compared by repeated-measures ANOVA with Holm–Sidak multiple-comparison posttests, and graphed data represent mean ± SEM. *P < 0.05; **P < 0.01; ***P < 0.001.
Discussion
We previously demonstrated that experimental activation of LHA Nts neurons is sufficient to modify ingestive and locomotor behavior and to reduce body weight, but our current study is, to our knowledge, the first to demonstrate that LHA Nts neurons are necessary for normal energy balance and adiposity. In particular, loss of LHA Nts neurons decreases OX expression, drinking behavior, locomotor activity, and energy expenditure and leads to obesity. In comparison, inhibition of LHA Nts neurons blunts locomotor activity and the anorectic response to leptin without disrupting OX expression. Taken together, these data implicate a specific role for LHA Nts neurons in regulating locomotor activity and the adaptive response to leptin, as these effects were not contingent upon loss of action via downstream OX neurons. Because deficits in drinking and energy expenditure were only observed after sustained LHA Nts ablation and with OX reduction, these behaviors may depend on sustained communication between LHA Nts and OX neurons. Collectively, our findings support different LHA Nts circuit mechanisms in controlling behaviors relevant to energy balance, and that chronic disruption of LHA Nts neurons may contribute to maladaptive behaviors that potentiate obesity. Indeed, endogenous Nts expression is decreased in the brains of obese rodents (63–65), although it was previously unclear whether this was a cause or effect of weight gain. Our findings show that loss of LHA Nts causes homeostatic disruption and adiposity. Going forward, it will be important to understand the precise mechanisms by which LHA Nts neurons coordinate energy balance to determine whether they may be novel targets to treat or prevent obesity.
Experimental lesions of the entire LHA revealed its important role in energy balance, but they masked the individual contributions of discrete, neurochemically defined LHA populations. Owing to the ability to easily detect MCH and OX via immunoreactivity, the roles of these neurons have been well studied within the LHA, and both are considered to be orexigenic, glucose responsive, and to modulate arousal in opposing directions (MCH promotes sleep and limits locomotor activity, OX facilitates waking and locomotor activity) (62, 66). Genetic ablation has previously been used to discern the necessity of MCH or OX neurons for energy balance, as well as their distinct contributions to energy balance. Ablation of MCH neurons in adult mice caused significantly increased locomotor activity and improved glucose tolerance and leanness, but it did not significantly alter food intake (67). Conversely, complete ablation of adult OX neurons decreased arousal/wake time, locomotor activity, and drinking, whereas it increased cataplexy and body weight, with no observed change in feeding (51). In comparison, we found that adult lesion of LHA Nts neurons decreased locomotor activity and drinking and increased adiposity, with no effect on homeostatic or hedonic feeding. The phenotypes resulting from LHA Nts or OX ablation are somewhat similar, and they may be due to the fact that LHA Nts neurons synaptically regulate OX neurons and coordinate at least some physiology via OX neurons (24, 68). Hence, loss of LHA Nts neurons and the subsequent downregulation of OX presumably impairs OX-mediated functions and accounts for the overlapping LHA Nts and OX-ablation phenotypes. Prolonged loss of action via the LHA Nts → OX circuit could also impair MCH neurons, which are synaptically regulated by OX neurons (69); indeed, this could account for the delayed trend for reduced MCH expression that we observed in LHA Nts-ablated mice. Collectively, these data suggest that LHA Nts neurons are crucial master controllers of OX neurons and perhaps the LHA as a whole.
LHA neurons have also recently been parsed by their classical neurotransmitter content, and at least some LHA Nts neurons coexpress GABA (70). Accordingly, we note some similarities between mice with genetic lesion of GABA neurons or disruption of LHA Nts neurons. For example, loss of LHA GABA neurons blunts fasting-induced refeeding (20), and in our study fasted LHA Nts-inhibited mice had impaired anorectic responses to leptin (Fig. 9). However, the phenotypes of LHA GABA vs Nts-ablated neurons do not perfectly overlap. Interestingly, ablation of LHA GABA neurons did not affect locomotor behavior (20), whereas loss or inhibition of LHA Nts neurons resulted in substantial reductions in physical activity. Although modulation of LHA GABA neurons must include the subset of LHA Nts-GABA neurons (28), the differences in observed ablation phenotypes suggest that the LHA GABA and LHA Nts neurons are not fully overlapping populations. Thus, there are likely to be some separate behaviors mediated via GABA-only and Nts-GABA neurons, which have yet to be disentangled. Nonetheless, because the GABAergic LHA Nts neurons are synaptically connected with OX neurons they presumably release GABA and Nts onto them (42). GABA and Nts would thus seem to be the obvious targets to directly modulate OX activity and expression, yet this may not be the case. Activating LHA Nts neurons leads to inhibition of OX neurons but via GABA-independent mechanisms (58). Furthermore, OX neurons lack NtsR1 and are unlikely to express NtsR2 because it is predominantly located on glia (71, 72), suggesting that they may not be directly regulated by Nts either. Given the conflicting reports concerning how Nts alters OX neuronal activity (58, 73, 74), as well as the lack of data on how it impacts OX transcription, we cannot yet speculate on how LHA Nts neurons modify OX expression. One possibility is that Nts acts via non-OX intermediary cells to indirectly modulate regulation of OX neurons, and this will be important to investigate in the future.
Intriguingly, ablation of MCH, OX, or LHA Nts neurons does not impair homeostatic feeding, despite the well-established roles of these peptides to modify food intake (51, 67) (Fig. 2). This may suggest that the LHA is more important for regulating adaptive feeding in response to changing peripheral signals of energy status, and it may suggest why we did not observe any reduction in homeostatic feeding of LHA Nts-ablated mice. One important signal for adaptive energy balance is leptin, which communicates high peripheral energy stores to the brain and suppresses resultant feeding to prevent weight gain. At least some LHA Nts neurons coexpress LepRb and mediate the adaptive leptin response. We therefore reasoned that loss of LHA Nts neurons would impair the adaptive feeding and body weight response to leptin. Indeed, inhibition of LHA Nts neurons blunted leptin-mediated suppression of feeding and resulted in increased body weight. Thus, loss of LHA Nts neurons disrupts leptin-mediated adaptive energy balance, particularly under conditions of increased appetitive drive (as in the fasted mice we tested). Individuals who lose weight via a long-term diet and exercise regimen also have an increased appetitive drive, which may prompt feeding and weight regain (75). We postulate that this could be due, in part, to diminished actions of LHA Nts neurons and the inability to suppress feeding drive necessary for sustained weight loss. Furthermore, this would impair individuals from responding to elevated leptin, as might occur due to increased adiposity, and from being able to curb the motivation to eat. Loss or inhibition of LHA Nts neurons also blunts locomotor activity without a commensurate or leptin-regulated decrease in energy intake, such that both behaviors could contribute to energy surfeit. Both of these behaviors are relevant to thwarting the obesity epidemic, which has been linked to increasingly sedentary lifestyles as well as an obesogenic food environment (76, 77).
At least some LHA Nts neurons that project to the VTA coexpress LepRb, and presumably they coordinate anorectic leptin response with DA regulation (24, 25). Indeed, LHA Nts neurons regulate mesolimbic DA neurons in the VTA, and they promote DA release to the nucleus accumbens that modulates motivated ingestion and physical activity (24, 50, 52). Loss of LHA Nts neurons presumably impairs DA-dependent actions, which may be more apparent in conditions that elevate motivation. For example, LHA Nts-ablated mice exhibit reduced ambulatory locomotor activity, but they have more profound deficits in motivated locomotor activity (e.g., wheel running). Importantly, note that LHA Nts-ablated mice remained capable of moving and did engage their running wheels, although to a lesser extent than control mice. Likewise, prior studies confirm that obese rodents can and do run on wheels (56, 78, 79), so the increased adiposity of LHA Nts-ablated mice is not likely to account for their diminished running behavior. Given that loss of mesolimbic DA blunts locomotor activity (80), it is possible that loss of the LHA Nts → VTA DA circuit contributes to the diminished physical activity, which over time may potentiate fat accumulation despite normal caloric intake (Fig. 2A). LHA Nts-inhibited mice also demonstrated a decrease in activity, without any apparent downregulation of OX, providing rationale that this effect might be mediated via an OX-independent pathway. Thus, the blunting of locomotor activity in both the LHA Nts-ablated and inhibited models suggests that at least some portion of LHA Nts-mediated control of physical activity occurs independently of OX and might be mediated via projections to the VTA or other LHA Nts projection sites. Although our data do not rule out a contribution of LHA Nts → OX neurons in regulating physical activity, this circuit is not the sole mediator of the behavior. In contrast, the blunted drinking and energy balance was only noted in the LHA Nts-ablated mice with downregulation of OX neurons, suggesting that these may be predominantly mediated via an LHA Nts → OX circuit. Going forward it will be crucial to dissect the circuit-specific roles of LHA Nts neurons, which may reveal specific targets to modify drinking, feeding, and locomotor activity.
OX is a vital mediator of arousal; hence, it is possible that the diminished OX expression in LHA Nts-ablated mice might blunt arousal-dependent behaviors to contribute to their altered physiology. Indeed, loss of OX induces narcolepsy and cataplexy, obesity, and decreased water intake and locomotor activity (51, 81, 82). Although we lack the expertise or equipment to directly assess arousal in mice, we did not observe any overt increases in sleeping or cataplexy-like behaviors in LHA Nts-ablated mice despite their ∼50% reduction in IF-detected OX neurons (Fig. 6). Nearly complete (∼90%) genetic ablation of OX neurons is required before there are detectable changes in arousal and cataplexy (51); hence, the comparably modest OX decrease in LHA Nts-ablated mice may not be sufficient to produce obvious changes in these behaviors. Moreover, there are differences in the degree of locomotor deficit between mice with nearly complete ablation of OX neurons vs the LHA Nts-ablated mice suggestive of different underlying mechanisms. Mice with nearly complete OX ablation exhibit reduced locomotor activity throughout the light and dark cycles (51), but LHA Nts-ablated mice have decreased locomotor activity selectively during the dark cycle, which argues against a general blunting of arousal and activity (Supplemental Fig. 2). It is possible that compensatory wake-promoting mechanisms may have masked any overt alertness/sleep phenotype in LHA Nts-ablated mice, as has been observed in other models with only partial loss of OX neurons (83). For example, other transmitters in OX neurons may be sufficient to maintain arousal via the intact circuitry despite downregulation of OX. Collectively, we cannot rule out a role for alertness and cataplexy in LHA Nts-ablated mice, but our data diverge sufficiently from fully OX-ablated mice to suggest that their blunted locomotor and drinking behaviors are not solely attributable to reduced arousal.
In contrast to global LHA manipulations, we selectively modulated LHA Nts neurons to reveal their contributions to energy balance and their necessity to prevent obesity. A caveat of our studies is that LHA Nts neurons are not homogeneous, and hence by modulating all LHA Nts neurons, we have obscured the specific roles of any subpopulations of LHA Nts neurons. For example, LHA Nts neurons can be divided into at least two subpopulations: ∼15% of LHA Nts neurons express LepRb and are regulated by leptin (24, 25), whereas other LHA Nts neurons are regulated by dehydration (26). As such, simultaneous loss of all LHA Nts neuronal subtypes may yield a mixed phenotype by disrupting function of LepRb-expressing neurons that modify feeding, as well as dehydration-modulated neurons that presumably coordinate drinking to restore fluid balance. Identification of genetic markers to discern molecularly specified subpopulations of LHA Nts neurons will be necessary to selectively ablate them and reveal their specific functions. Alternately, ablating LHA Nts neurons that project to local OX neurons vs those that project to the VTA may be useful to assess whether these circuits separately modify drinking vs feeding and locomotor behavior. Although there is still much to be learned about how LHA Nts neurons modify physiology, our data reveal that they are necessary and important mediators for locomotor activity, adaptive feeding responses, drinking, and control of normal adiposity. Thus, loss of action via LHA Nts neurons may contribute to maladaptive behaviors that increase the risk to develop obesity. In the future, defining the signaling and circuit mechanisms by which LHA Nts neurons coordinate ingestive and locomotor behavior may suggest strategies to restore LHA Nts function and healthy body weight.
Supplementary Material
Acknowledgments
We thank Sandra O’Reilly for assistance with mouse metabolic phenotyping and Michelle Mazei-Robison and A.J. Robison for guidance on behavioral experiments. We are also grateful to the Michigan State University College of Natural Science and the Honors College for providing research awards that supported undergraduate students who contributed to this project (A.S., T.M., and A.W.).
Financial Support: This research was supported by grants from the National Institutes of Health to J.B. (National Institute of Environmental Health Sciences Grant T32-ES00725527, National Institute of Diabetes and Digestive and Kidney Diseases Grant F31-DK107081) and G.L. (pilot and feasibility grants from the National Institute of Diabetes and Digestive and Kidney Diseases–funded Michigan Diabetes Research Center, Grants P30-DK020572 and R01-DK103808), as well as by National Institute of Neurologic Disorders and Stroke Grants NS073613 and NS092652 to P.M.F.
Disclosure Summary: The authors have nothing to disclose.
Glossary
Abbreviations:
- AAV
adeno-associated virus
- AAV-DTA
adeno-associated virus expressing Cre-dependent diphtheria toxin A
- CNO
clozapine-N-oxide
- DA
dopamine
- DREADD
designer receptor exclusively activated by designer drugs
- DTA
diphtheria toxin A
- GABA
γ-aminobutyric acid
- IF
immunofluorescence
- LepRb
long form of the leptin receptor
- LHA
lateral hypothalamic area
- MCH
melanin-concentrating hormone
- Nts
neurotensin
- OX
orexin
- RER
respiratory exchange ratio
- VCO2
carbon dioxide production
- VEH
vehicle
- VO2
oxygen consumption
- VTA
ventral tegmental area
References
- 1. Finucane MM, Stevens GA, Cowan MJ, Danaei G, Lin JK, Paciorek CJ, Singh GM, Gutierrez HR, Lu Y, Bahalim AN, Farzadfar F, Riley LM, Ezzati M; Global Burden of Metabolic Risk Factors of Chronic Diseases Collaborating Group (Body Mass Index) . National, regional, and global trends in body-mass index since 1980: systematic analysis of health examination surveys and epidemiological studies with 960 country-years and 9.1 million participants. Lancet. 2011;377(9765):557–567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Kelly T, Yang W, Chen CS, Reynolds K, He J. Global burden of obesity in 2005 and projections to 2030. Int J Obes. 2008;32(9):1431–1437. [DOI] [PubMed] [Google Scholar]
- 3. Apovian CM, Aronne LJ, Bessesen DH, McDonnell ME, Murad MH, Pagotto U, Ryan DH, Still CD; Endocrine Society . Pharmacological management of obesity: an endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2015;100(2):342–362. [DOI] [PubMed] [Google Scholar]
- 4. Sumithran P, Proietto J. The defence of body weight: a physiological basis for weight regain after weight loss. Clin Sci (Lond). 2013;124(4):231–241. [DOI] [PubMed] [Google Scholar]
- 5. Sumithran P, Prendergast LA, Delbridge E, Purcell K, Shulkes A, Kriketos A, Proietto J. Long-term persistence of hormonal adaptations to weight loss. N Engl J Med. 2011;365(17):1597–1604. [DOI] [PubMed] [Google Scholar]
- 6. Wing RR, Hill JO. Successful weight loss maintenance. Annu Rev Nutr. 2001;21(1):323–341. [DOI] [PubMed] [Google Scholar]
- 7. Fothergill E, Guo J, Howard L, Kerns JC, Knuth ND, Brychta R, Chen KY, Skarulis MC, Walter M, Walter PJ, Hall KD. Persistent metabolic adaptation 6 years after “The Biggest Loser” competition. Obesity (Silver Spring). 2016;24(8):1612–1619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Anand BK, Brobeck JR. Hypothalamic control of food intake in rats and cats. Yale J Biol Med. 1951;24(2):123–140. [PMC free article] [PubMed] [Google Scholar]
- 9. Anand BK, Brobeck JR. Localization of a “feeding center” in the hypothalamus of the rat. Proc Soc Exp Biol Med. 1951;77(2):323–324. [DOI] [PubMed] [Google Scholar]
- 10. Burdakov D, Gerasimenko O, Verkhratsky A. Physiological changes in glucose differentially modulate the excitability of hypothalamic melanin-concentrating hormone and orexin neurons in situ. J Neurosci. 2005;25(9):2429–2433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Konadhode RR, Pelluru D, Blanco-Centurion C, Zayachkivsky A, Liu M, Uhde T, Glen WB Jr, van den Pol AN, Mulholland PJ, Shiromani PJ. Optogenetic stimulation of MCH neurons increases sleep. J Neurosci. 2013;33(25):10257–10263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Tsunematsu T, Ueno T, Tabuchi S, Inutsuka A, Tanaka KF, Hasuwa H, Kilduff TS, Terao A, Yamanaka A. Optogenetic manipulation of activity and temporally controlled cell-specific ablation reveal a role for MCH neurons in sleep/wake regulation. J Neurosci. 2014;34(20):6896–6909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Vetrivelan R, Kong D, Ferrari LL, Arrigoni E, Madara JC, Bandaru SS, Lowell BB, Lu J, Saper CB. Melanin-concentrating hormone neurons specifically promote rapid eye movement sleep in mice. Neuroscience. 2016;336:102–113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Venner A, Karnani MM, Gonzalez JA, Jensen LT, Fugger L, Burdakov D. Orexin neurons as conditional glucosensors: paradoxical regulation of sugar sensing by intracellular fuels. J Physiol. 2011;589(Pt 23):5701–5708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Yamanaka A, Beuckmann CT, Willie JT, Hara J, Tsujino N, Mieda M, Tominaga M, Yagami K, Sugiyama F, Goto K, Yanagisawa M, Sakurai T. Hypothalamic orexin neurons regulate arousal according to energy balance in mice. Neuron. 2003;38(5):701–713. [DOI] [PubMed] [Google Scholar]
- 16. Adamantidis A, de Lecea L. The hypocretins as sensors for metabolism and arousal. J Physiol. 2009;587(1):33–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Inutsuka A, Inui A, Tabuchi S, Tsunematsu T, Lazarus M, Yamanaka A. Concurrent and robust regulation of feeding behaviors and metabolism by orexin neurons. Neuropharmacology. 2014;85:451–460. [DOI] [PubMed] [Google Scholar]
- 18. Nieh EH, Matthews GA, Allsop SA, Presbrey KN, Leppla CA, Wichmann R, Neve R, Wildes CP, Tye KM. Decoding neural circuits that control compulsive sucrose seeking. Cell. 2015;160(3):528–541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Stamatakis AM, Van Swieten M, Basiri ML, Blair GA, Kantak P, Stuber GD. Lateral hypothalamic area glutamatergic neurons and their projections to the lateral habenula regulate feeding and reward. J Neurosci. 2016;36(2):302–311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Jennings JH, Ung RL, Resendez SL, Stamatakis AM, Taylor JG, Huang J, Veleta K, Kantak PA, Aita M, Shilling-Scrivo K, Ramakrishnan C, Deisseroth K, Otte S, Stuber GD. Visualizing hypothalamic network dynamics for appetitive and consummatory behaviors. Cell. 2015;160(3):516–527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Navarro M, Olney JJ, Burnham NW, Mazzone CM, Lowery-Gionta EG, Pleil KE, Kash TL, Thiele TE. Lateral hypothalamus GABAergic neurons modulate consummatory behaviors regardless of the caloric content or biological relevance of the consumed stimuli. Neuropsychopharmacology. 2016;41(6):1505–1512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Brown JA, Woodworth HL, Leinninger GM. To ingest or rest? Specialized roles of lateral hypothalamic area neurons in coordinating energy balance. Front Syst Neurosci. 2015;9:9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Kurt G, Woodworth HL, Leinninger GM. Lateral Hypothalamic Control of Energy Balance. San Rafael, CA: Morgan & Claypool; 2017.
- 24. Brown JA, Bugescu R, Mayer TA, Gata-Garcia A, Kurt G, Woodworth HL, Leinninger GM. Loss of action via neurotensin-leptin receptor neurons disrupts leptin and ghrelin-mediated control of energy balance. Endocrinology. 2017;158(5):1271–1288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Leinninger GM, Opland DM, Jo YH, Faouzi M, Christensen L, Cappellucci LA, Rhodes CJ, Gnegy ME, Becker JB, Pothos EN, Seasholtz AF, Thompson RC, Myers MG Jr. Leptin action via neurotensin neurons controls orexin, the mesolimbic dopamine system and energy balance. Cell Metab. 2011;14(3):313–323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Watts AG, Sanchez-Watts G, Kelly AB. Distinct patterns of neuropeptide gene expression in the lateral hypothalamic area and arcuate nucleus are associated with dehydration-induced anorexia. J Neurosci. 1999;19(14):6111–6121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Woodworth HL, Beekly BG, Batchelor HM, Bugescu R, Perez-Bonilla P, Schroeder LE, Leinninger GM. Lateral hypothalamic neurotensin neurons orchestrate dual weight loss behaviors via distinct mechanisms. Cell Reports. 2017;21(11):3116–3128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Patterson CM, Wong JM, Leinninger GM, Allison MB, Mabrouk OS, Kasper CL, Gonzalez IE, Mackenzie A, Jones JC, Kennedy RT, Myers MG Jr. Ventral tegmental area neurotensin signaling links the lateral hypothalamus to locomotor activity and striatal dopamine efflux in male mice. Endocrinology. 2015;156(5):1692–1700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Wang XF, Liu JJ, Xia J, Liu J, Mirabella V, Pang ZP. Endogenous glucagon-like peptide-1 suppresses high-fat food intake by reducing synaptic drive onto mesolimbic dopamine neurons. Cell Reports. 2015;12(5):726–733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. RRID:SCR_012527.
- 31. RRID:SCR_002448.
- 32. RRID:SCR_002448.
- 33. RRID:IMSR_JAX:017525.
- 34. RRID:SCR_015373.
- 35. Paxinos G, Franklin B. The Mouse Brain in Stereotaxic Coordinates. 2nd ed. San Diego, CA: Academic Press; 2001.
- 36. Roth BL. DREADDs for neuroscientists. Neuron. 2016;89(4):683–694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Soumier A, Sibille E. Opposing effects of acute versus chronic blockade of frontal cortex somatostatin-positive inhibitory neurons on behavioral emotionality in mice. Neuropsychopharmacology. 2014;39(9):2252–2262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. RRID: AB_2237276.
- 39. RRID: AB_2636859.
- 40. RRID: AB_2340397.
- 41. RRID: AB_2340593.
- 42. RRID: AB_2340375.
- 43. RRID: AB_2556546.
- 44. RRID: AB_2534104.
- 45. RRID: AB_300798.
- 46. RRID: AB_653611.
- 47. RRID: AB_2292943.
- 48. RRID: AB_305808.
- 49. Pei H, Sutton AK, Burnett KH, Fuller PM, Olson DP. AVP neurons in the paraventricular nucleus of the hypothalamus regulate feeding. Mol Metab. 2014;3(2):209–215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Woodworth HL, Batchelor HM, Beekly BG, Bugescu R, Brown JA, Kurt G, Fuller PM, Leinninger GM. Neurotensin receptor-1 identifies a subset of ventral tegmental dopamine neurons that coordinates energy balance. Cell Reports. 2017;20(8):1881–1892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Tabuchi S, Tsunematsu T, Black SW, Tominaga M, Maruyama M, Takagi K, Minokoshi Y, Sakurai T, Kilduff TS, Yamanaka A. Conditional ablation of orexin/hypocretin neurons: a new mouse model for the study of narcolepsy and orexin system function. J Neurosci. 2014;34(19):6495–6509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Opland D, Sutton A, Woodworth H, Brown J, Bugescu R, Garcia A, Christensen L, Rhodes C, Myers M Jr, Leinninger G. Loss of neurotensin receptor-1 disrupts the control of the mesolimbic dopamine system by leptin and promotes hedonic feeding and obesity. Mol Metab. 2013;2(4):423–434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Mann T, Tomiyama AJ, Westling E, Lew AM, Samuels B, Chatman J. Medicare’s search for effective obesity treatments: diets are not the answer. Am Psychol. 2007;62(3):220–233. [DOI] [PubMed] [Google Scholar]
- 54. Berridge KC, Ho CY, Richard JM, DiFeliceantonio AG. The tempted brain eats: pleasure and desire circuits in obesity and eating disorders. Brain Res. 2010;1350:43–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Salamone JD, Correa M. The mysterious motivational functions of mesolimbic dopamine. Neuron. 2012;76(3):470–485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Novak CM, Burghardt PR, Levine JA. The use of a running wheel to measure activity in rodents: relationship to energy balance, general activity, and reward. Neurosci Biobehav Rev. 2012;36(3):1001–1014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Sclafani A. Sucrose motivation in sweet “sensitive” (C57BL/6J) and “subsensitive” (129P3/J) mice measured by progressive ratio licking. Physiol Behav. 2006;87(4):734–744. [DOI] [PubMed] [Google Scholar]
- 58. Goforth PB, Leinninger GM, Patterson CM, Satin LS, Myers MG Jr. Leptin acts via lateral hypothalamic area neurotensin neurons to inhibit orexin neurons by multiple GABA-independent mechanisms. J Neurosci. 2014;34(34):11405–11415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Louis GW, Leinninger GM, Rhodes CJ, Myers MG Jr. Direct innervation and modulation of orexin neurons by lateral hypothalamic LepRb neurons. J Neurosci. 2010;30(34):11278–11287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Meister B, Perez-Manso M, Daraio T. Delta-like 1 homologue is a hypothalamus-enriched protein that is present in orexin-containing neurones of the lateral hypothalamic area. J Neuroendocrinol. 2013;25(7):617–625. [DOI] [PubMed] [Google Scholar]
- 61. Dalal J, Roh JH, Maloney SE, Akuffo A, Shah S, Yuan H, Wamsley B, Jones WB, de Guzman Strong C, Gray PA, Holtzman DM, Heintz N, Dougherty JD. Translational profiling of hypocretin neurons identifies candidate molecules for sleep regulation. Genes Dev. 2013;27(5):565–578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Burdakov D, Luckman SM, Verkhratsky A. Glucose-sensing neurons of the hypothalamus. Philos Trans R Soc Lond B Biol Sci. 2005;360(1464):2227–2235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Beck B, Stricker-Krongrad A, Burlet A, Nicolas JP, Burlet C. Changes in hypothalamic neurotensin concentrations and food intake in rats fed a high fat diet. Int J Obes Relat Metab Disord. 1992;16(5):361–366. [PubMed] [Google Scholar]
- 64. Wilding JP, Gilbey SG, Bailey CJ, Batt RA, Williams G, Ghatei MA, Bloom SR. Increased neuropeptide-Y messenger ribonucleic acid (mRNA) and decreased neurotensin mRNA in the hypothalamus of the obese (ob/ob) mouse. Endocrinology. 1993;132(5):1939–1944. [DOI] [PubMed] [Google Scholar]
- 65. Williams G, Cardoso H, Lee YC, Ghatei MA, Flatt PR, Bailey CJ, Bloom SR. Reduced hypothalamic neurotensin concentrations in the genetically obese diabetic (ob/ob) mouse: possible relationship to obesity. Metabolism. 1991;40(10):1112–1116. [DOI] [PubMed] [Google Scholar]
- 66. Saper CB, Fuller PM, Pedersen NP, Lu J, Scammell TE. Sleep state switching. Neuron. 2010;68(6):1023–1042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Whiddon BB, Palmiter RD. Ablation of neurons expressing melanin-concentrating hormone (MCH) in adult mice improves glucose tolerance independent of MCH signaling. J Neurosci. 2013;33(5):2009–2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Funato H, Tsai AL, Willie JT, Kisanuki Y, Williams SC, Sakurai T, Yanagisawa M. Enhanced orexin receptor-2 signaling prevents diet-induced obesity and improves leptin sensitivity. Cell Metab. 2009;9(1):64–76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69. Apergis-Schoute J, Iordanidou P, Faure C, Jego S, Schöne C, Aitta-Aho T, Adamantidis A, Burdakov D. Optogenetic evidence for inhibitory signaling from orexin to MCH neurons via local microcircuits. J Neurosci. 2015;35(14):5435–5441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. Leinninger GM, Jo YH, Leshan RL, Louis GW, Yang H, Barrera JG, Wilson H, Opland DM, Faouzi MA, Gong Y, Jones JC, Rhodes CJ, Chua S Jr, Diano S, Horvath TL, Seeley RJ, Becker JB, Münzberg H, Myers MG Jr. Leptin acts via leptin receptor-expressing lateral hypothalamic neurons to modulate the mesolimbic dopamine system and suppress feeding. Cell Metab. 2009;10(2):89–98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71. Woodworth HL, Perez-Bonilla PA, Beekly BG, Lewis TJ, Leinninger GM. Identification of neurotensin receptor expressing cells in the ventral tegmental area across the lifespan. eNeuro. 2018;5(1):ENEURO.0191-17.2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Yamauchi R, Wada E, Kamichi S, Yamada D, Maeno H, Delawary M, Nakazawa T, Yamamoto T, Wada K. Neurotensin type 2 receptor is involved in fear memory in mice. J Neurochem. 2007;102(5):1669–1676. [DOI] [PubMed] [Google Scholar]
- 73. Furutani N, Hondo M, Kageyama H, Tsujino N, Mieda M, Yanagisawa M, Shioda S, Sakurai T. Neurotensin co-expressed in orexin-producing neurons in the lateral hypothalamus plays an important role in regulation of sleep/wakefulness states. PLoS One. 2013;8(4):e62391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. Sheng Z, Santiago AM, Thomas MP, Routh VH. Metabolic regulation of lateral hypothalamic glucose-inhibited orexin neurons may influence midbrain reward neurocircuitry. Mol Cell Neurosci. 2014;62:30–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Soleymani T, Daniel S, Garvey WT. Weight maintenance: challenges, tools and strategies for primary care physicians. Obes Rev. 2016;17(1):81–93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Church TS, Thomas DM, Tudor-Locke C, Katzmarzyk PT, Earnest CP, Rodarte RQ, Martin CK, Blair SN, Bouchard C. Trends over 5 decades in U.S. occupation-related physical activity and their associations with obesity. PLoS One. 2011;6(5):e19657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77. Berthoud HR. The neurobiology of food intake in an obesogenic environment. Proc Nutr Soc. 2012;71(4):478–487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78. Carpenter KC, Strohacker K, Breslin WL, Lowder TW, Agha NH, McFarlin BK. Effects of exercise on weight loss and monocytes in obese mice. Comp Med. 2012;62(1):21–26. [PMC free article] [PubMed] [Google Scholar]
- 79. Laing BT, Do K, Matsubara T, Wert DW, Avery MJ, Langdon EM, Zheng D, Huang H. Voluntary exercise improves hypothalamic and metabolic function in obese mice. J Endocrinol. 2016;229(2):109–122. [DOI] [PubMed] [Google Scholar]
- 80. Zhou QY, Palmiter RD. Dopamine-deficient mice are severely hypoactive, adipsic, and aphagic. Cell. 1995;83(7):1197–1209. [DOI] [PubMed] [Google Scholar]
- 81. Chemelli RM, Willie JT, Sinton CM, Elmquist JK, Scammell T, Lee C, Richardson JA, Williams SC, Xiong Y, Kisanuki Y, Fitch TE, Nakazato M, Hammer RE, Saper CB, Yanagisawa M. Narcolepsy in orexin knockout mice: molecular genetics of sleep regulation. Cell. 1999;98(4):437–451. [DOI] [PubMed] [Google Scholar]
- 82. Hara J, Beuckmann CT, Nambu T, Willie JT, Chemelli RM, Sinton CM, Sugiyama F, Yagami K, Goto K, Yanagisawa M, Sakurai T. Genetic ablation of orexin neurons in mice results in narcolepsy, hypophagia, and obesity. Neuron. 2001;30(2):345–354. [DOI] [PubMed] [Google Scholar]
- 83. Black SW, Sun JD, Santiago P, Laihsu A, Kimura N, Yamanaka A, Bersot R, Humphries PS. Partial ablation of the orexin field induces a sub narcoleptic phenotype in a conditional mouse model of orexin neurodegeneration [published online ahead of print 31 May 2018]. Sleep. [DOI] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.