Abstract
Primary graft dysfunction (PGD) is a major cause of morbidity and mortality after lung transplantation. Ischemia–reperfusion injury (IRI) is a key event that contributes to PGD, though complex interactions affect donor lungs status, such as preceding brain death (BD), hemorrhagic shock (HS), and pre-engraftment lung management, the latter recognized as important risk factors for PGD. We hypothesized that a multi-hit isogenic mouse model of lung transplantation is more closely linked to PGD than IRI alone. Left lung transplants were performed between inbred C57BL/6 mice. A one-hit model of IRI was established by inducing cold ischemia (CI) of the donor lungs at 0°C for 1, 72, or 96 hours before engraftment. Multi-hit models were established by inducing 24 hours of HS and/or 3 hours of BD before 24 hours of CI. The recipients were killed at 24 hours after transplant and lung graft samples were analyzed. In the one-hit model of IRI, up to 72-hour CI time resulted in minimal cellular infiltration near small arteries after 24-hour reperfusion. Extension of CI time to 96 hours led to increased cellular infiltration and necroptotic pathway activation, without evidence of apoptosis, after 24-hour reperfusion. In a multi-hit model of PGD, “HS + BD + IRI” demonstrated increased lung injury, cellular infiltration, and activation of necroptotic and apoptotic pathways compared with IRI alone. Treatment with an inhibitor of receptor-interacting protein kinase 1 kinase, necrostatin-1, resulted in a significant decrease of downstream necroptotic pathway activation in both single- and multi-hit models of IRI. Thus, activation of necroptosis is a central event in IRI after prolonged CI, though it may not be sufficient to cause PGD alone. Pathological evaluation of donor lungs after CI-induced IRI, in conjunction with pre-engraftment donor lung factors in our multi-hit model, demonstrated early evidence of lung injury consistent with PGD. Our findings support the premise that pre-existing donor lung status is more important than CI time alone for inflammatory pathway activation in PGD, which may have important clinical implications for donor lung retrieval.
Keywords: lung transplantation, primary graft dysfunction, reperfusion injury, necroptosis
Clinical Relevance
This study identifies necroptosis as a form of cell death in orthotopic experimental lung transplantation that may open up molecular targets for therapies.
Primary graft dysfunction (PGD) can have a dramatic impact on outcomes, resulting in increased length of mechanical ventilation, prolonged hospital stays, bronchiolitis obliterans syndrome, and increased early and late mortality after lung transplantation (1, 2). The incidence of grade 3 PGD, which has similar histologic characteristics to adult respiratory distress syndrome, is evident in approximately 30% of patients early after transplant and 15–20% at 48 and 72 hours (1). Ischemia–reperfusion injury (IRI) is generally considered the major contributor to PGD and is often used interchangeably with PGD in animal studies (1–3). This is of clinical importance, as both IRI and PGD are linked to the development and progression of chronic lung allograft dysfunction, the major determinant of morbidity and mortality after lung transplantation (4, 5). Donor lung cell death has been shown to correlate with the extent of IRI, and dynamic changes of apoptosis and necrotic cell death have been reported in rodent models of lung transplantation after 18 hours of cold ischemia (CI) time (6).
In the clinical setting, the median CI time window before lung transplant is approximately 4–6 hours, and is a factor that contributes to less than 20% of donor lungs being ultimately selected for transplant, with many lungs declined due to poor or marginal function (7, 8). Considerably longer CI times are possible in other solid organs, an attribute that increases storage, transport, and recipient transplant options. Extension of total lung preservation time using ex vivo lung perfusion strategies at normothermia can optimize “borderline” donor lungs before transplant and significantly increase the donor lung pool (9). A confounding issue in previous rodent studies of experimental IRI is that the donor lungs are healthy, but are often impaired in brain dead human donors, a factor that may influence donor organ quality (10). The molecular pathways underlying injury in brain dead donor lungs and in IRI, specifically as it relates to other forms of cell death remain largely unknown.
The recognition that necroptosis, a caspase-independent mechanism of programmed cell death, is activated by IRI, through the phosphorylation of receptor-interacting protein kinase (RIPK) 1, RIPK3, and the mixed-lineage kinase domain-like kinase (MLKL), has advanced our understanding of the pathogenic mechanisms of PGD and might potentially lead to future therapeutic interventions in transplant medicine (11). RIPK3-dependent necroptosis is characterized by cellular swelling, rupture, and release of inflammatory damage-associated molecular patterns (DAMPs). Necroptosis has been linked to graft failure in renal and cardiac transplantation (12–14), and, in a recent study, inhibition of necroptosis has been shown to attenuate IRI in a rodent model of lung transplantation (15). In the present study, we hypothesized that RIPK3-mediated necroptosis activation occurs after experimental lung transplantation, and that pre-existent donor lung injury will have a more significant impact than CI time alone in a multi-hit mouse model of brain death (BD), hemorrhagic shock (HS), and CI-induced IRI.
Key Points
-
1.
Necroptotic cell death pathways are activated in IRI after prolonged CI time in lung transplantation.
-
2.
Reperfusion injury induced by 72-hour CI or less leads to minimal pathologic evidence of PGD.
-
3.
Characteristic pathological features of PGD are most evident in a multi-hit model of donor lung injury.
-
4.
Necroptotic cell death may be an important therapeutic target to prevent PGD in lung transplantation in conjunction with strategies that optimize donor lung status before engraftment.
Methods
Animals
All animal procedures were approved and performed according to the protocols established by the Institutional Animal Care and Use Committee of the University of Pittsburgh. Male C57BL/6 (H-2b) mice (weighing 23∼28 g) were purchased from Jackson Laboratory and used as both donors and recipients. Animals received humane care in compliance with standards of care. Each experimental endpoint represents a different set of animals for histologic versus protein immunoassay.
CI Model of IRI
Orthotopic left lung transplants (OLTs) were performed with standard cuff techniques (16). Briefly, the donor lungs were recovered after being flushed with 10 ml of cold Perfadex (XVIVO Perfusion Inc.) and inflated with room air. CI was induced by storing donor lungs at 0°C for 1 to 96 hours. The left donor lung was cuffed and implanted into recipients within 30 minutes. The recipient mice were killed at 2 hours for RNA sequencing analysis or 24 hours after transplantation for other measurements. The grafts were collected, snap frozen in liquid nitrogen, and stored at −80°C until performance of the assays. We used n = 2 mice at 1 hour CI time, and n = 3 mice at 72-hour and 96-hour CI time.
Pulmonary Function Tests
The recipient mouse was anesthetized and mechanically ventilated with 100% oxygen at a rate of 140 breaths/min, a tidal volume of 0.3 ml, and 2 cm H2O of positive end-expiratory pressure. Two minutes after ligation of the right hilum, no less than 300 μl of arterial blood was aspirated directly from the ascending aorta and tested on an i-STAT of Care analyzer (Abbott). The mouse was then killed, and lungs were harvested for other measurements.
Pulmonary Capillary Permeability
Evans-blue dye albumin (30 mg/kg) was injected into the tail vein 2 hours before termination of the experiment to assess vascular leakage (17). The entire left lung was homogenized and suspended in 500 μl formamide for 18 hours at 80°C, specimens were centrifuged at 13,000 × g for 30 minutes, and 100 μl supernatant was placed in 96-well plates for colorimetric analysis by spectrophotometric means (SpectraMax M2) at 620 nm. Relative optical density values were normalized to the weight of the samples.
Lung Wet:Dry Ratio
The lung tissue wet:dry ratio was measured to assess the extent of pulmonary edema. The lower portion of the left lung was cut and weighed before and after drying for 72 hours in an oven at 80°C (15). Wet weight was divided by the dry weight to calculate the wet:dry ratio.
Multi-Hit Model of IRI
HS and/or BD were induced in donor mice to establish two-hit models (BD + IRI, HS + IRI) and a three-hit model (HS + BD + IRI). The CI model of IRI was used as a control. All donor mice were ventilated with a tidal volume of 8 ml/kg room air for 3 hours before donation surgery to simulate donor ventilation before organ procurement (n = 3 mice/group). HS was induced by using continuous tail bleeding techniques. After anesthesia and local disinfection, the tip of the mouse tail was cut and immersed in 50 μl of sterile heparin solution (1:10,000). The tail was ligated after 50% blood volume (58.5 ml/kg) was lost. At 2 hours later, the mice were resuscitated with an intravenous sterile saline injection at three times the volume of shed blood (18, 19). The animals were kept warm with access to food and water for 24 hours before BD and/or mechanical ventilation. To induce BD, the donor mouse was anesthetized, intubated, and put on a heating pad. After excising the scalp, two 2-mm holes were drilled with a #11 General Surgery Blade through the skull 4 mm lateral to the sagittal suture. Saline-soaked gauze strips (1.5 cm × 0.5 cm) were gently inserted intracranially through the burr holes. The mouse was connected to a MiniVent Model 845 ventilator (Harvard Apparatus) once BD was confirmed by cessation of spontaneous respiration and maximally dilated and fixed pupils (20, 21). The animal was kept warm and ventilated for 3 hours before donation.
Treatment with Necrostatin-1
Necrostatin (Nec)-1 (10 mg; Sigma-Aldrich) was dissolved in 2 ml of DMSO (Sigma-Aldrich). In selected experiments, Nec-1 (10 mg/kg) or DMSO (2 ml/kg) was intraperitoneally administered to the recipient 1 hour before lung transplantation (n = 3 mice/group). The grafted lungs were sampled after 24 hours of reperfusion.
Immunoblot Analysis
The upper portion of the left lung was separated from remaining lung (used for RNA) using a forceps, and samples were lysed in lysis buffer containing Triton X-100 (MilliporeSigma), 1× protease inhibitor cocktail tablet, and 1× PhosSTOP Phosphatase Inhibitor Cocktail Tablet (Roche Life Science). Equal amounts of lysates were separated on SDS-PAGE (4∼12% gels) under reducing or nonreducing conditions and transferred onto nitrocellulose membranes (MilliporeSigma). The membranes were blocked and then incubated overnight at 4°C with the after antibodies: anti-RIPK1 (dilution of 1:200; Biorbyt); anti-pRIPK3 (dilution of 1:1,000; Abcam); anti-RIPK3 (dilution of 1:800; Abcam); anti-pMLKL (dilution of 1:1,000; Abcam); anti-MLKL (dilution of 1:1,000; Biorbyt); anti–cleaved-caspase3 (dilution of 1:1,000; Cell Signaling); anti–caspase-3 (dilution of 1:1,000; Cell Signaling); anti–Caspase-8 (dilution of 1:1,000; Novus); anti–caspase-9 (dilution of 1:1,000; Cell Signaling); anti–TNF-α (dilution of 1:1,000; Abcam); anti-IL6 (dilution of 1:200; Santa Cruz); anti–IL1-β (dilution of 1:1,000; Cell Signaling); anti–CXCL-1 (dilution of 1:2,000; Invitrogen); anti–CXCL-2 (dilution of 1:2,000; Invitrogen); anti-HMGB1 (high mobility group box 1; dilution of 1:2,000; Antibody Revolution); anti–pNF-κB (dilution of 1:1,000; Cell Signaling); anti–NF-κB (dilution of 1:1,000; Cell Signaling); anti-pJNK (c-Jun N-terminal kinase; dilution of 1:1,000; Cell Signaling); anti-JNK (dilution of 1:1,000; Cell Signaling); anti-pP38 (dilution of 1:1,000; Cell Signaling); anti-P38 (dilution of 1:1,000; Cell Signaling); anti-pERK (phosphorylated extracellular signal–regulated kinases; dilution of 1:500; Santa Cruz); anti-ERK (dilution of 1:500; Santa Cruz); anti-pAKT (phosphorylated serine/threonine–protein kinases; dilution of 1:1,000; Cell Signaling); anti-AKT (dilution of 1:1,000; Cell Signaling); and anti-actin (loading control, 1:5,000; Sigma). Protein bands were visualized using enhanced chemiluminescence according to the manufacturer’s instructions. The band density was measured with ImageJ (https://imagej.net) and normalized to actin.
Histology and Immunohistochemistry
Entire left lung grafts were harvested, inflated, fixed in 10% phosphate-buffered formalin, and embedded in paraffin. Sections were stained with hematoxylin and eosin. Three observers, who were blinded to the treatment groups, measured epithelial thickness of small airways with similar diameters by using ImageJ imaging software, calculated an acute lung injury score on images (n ≥ 3) obtained in 400× high-power fields (20 per slide). The scoring systems was applied per the American Thoracic Society workshop report (22), which includes evaluation of neutrophils in the alveolar and interstitial space, the presence of hyaline membranes, airspace proteinaceous debris, and alveolar septal thickening. For immunohistochemistry, the tissue sections were incubated with a mouse pMLKL antibody (ab196436; Abcam). Primary and biotinylated secondary antibody (horseradish peroxidase) binding were visualized by DAB Substrate Kit (#8059; Cell Signaling Technology) and counterstained with hematoxylin (#14166; Cell Signaling Technology).
RNA Sequencing
Entire left lung grafts (n = 1 per group) were lysed in RLT buffer plus (Qiagen) and total cellular RNA was extracted using the RNeasy MiniKit (Qiagen) in accordance with the manufacturer’s instructions. RNA integrity was determined with the Agilent 2100 Bioanalyzer, and lung homogenate samples with RNA integrity numbers ranging from 4.9 to 7.4 were processed with an mRNA-Seq sample preparation kit (Illumina, Inc.). Sequencing libraries were prepared from 1 μg of RNA using Nextera XT DNA Library Preparation kit (Illumina), normalized at 2 nM using Tris-HCl (10 mM, pH 8.5) with 0.1% Tween20, diluted and denatured to a final concentration of 1.8 nM using the Illumina Denaturing and Diluting libraries for the NextSeq 500 protocol Revision D (Illumina). Cluster generation and 1 × 75 bp forward end-reads sequencing was performed on an Illumina NextSeq 500 system at the University of Pittsburgh Health Sciences Sequencing Core.
RNA Sequence Analysis
Raw transcript data were imported into CLC Genomics Workbench 11, quality control and trimming of files was performed before alignment of reads to the mouse reference genome. Genes with a mean log2 transcript count value less than 5 were removed from further processing. Differentially expressed genes (DEGs) were determined between the experimental groups using filters to select genes with transcripts per million of 1 or greater, absolute fold change greater than 1.5 using total gene count, and a false discovery rate P value of 0.05 or less. Principal component analysis, Venn diagram creation, hierarchical clustering of DEGs, and heatmap generation were performed using CLC Genomics Workbench RNA sequencing software.
Gene Set Enrichment Analysis
Reactome pathway analysis, to evaluate the functional effects of 24-hour and 96-hour CI time on donor lungs before transplantation and 2 hours after reperfusion, was completed using gene set enrichment of filtered DEGs in Panther (http://pantherdb.org) (23). Statistical overrepresentation testing was performed using Fisher’s exact test with correction for multiple testing using a false discovery rate P value cut-off less than 0.05 for pathway significance.
Statistical Analysis
Data are expressed as mean (±SEM) as indicated in figure legends. Differences were analyzed via two-way ANOVA for group analysis with post hoc Tukey’s or Dunn’s test for multiple comparison testing. A Student’s t test was performed for the comparison of means in normally distributed data. Statistical significance was defined as P less than 0.05. All statistical analyses were performed by using GraphPad Prism V7.03 software.
Results
Lung Isogenic Grafts Tolerate up to 72 Hours of CI
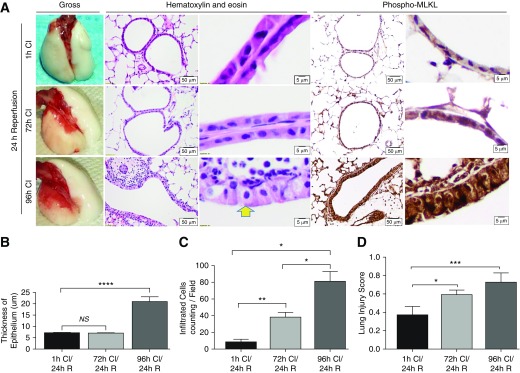
Cold ischemic time was extended up to 96 hours to explore the cellular mechanisms of cold IRI and determine if cold IRI alone is sufficient to cause PGD in mouse lung transplantation. Less than 72 hours of CI only induced mild infiltration near small arteries after 24 hours of reperfusion (Figure 1A). However, the 96-hour CI–time lung grafts after 24-hour reperfusion demonstrated pathologic manifestations of necroptosis, characterized by swelling of airway epithelia and increased cellular translucency, though nuclei and cell membranes remained intact (Figure 1A, high magnification, 5 μM, arrow) (11, 24). Localization of necroptotic cell death was determined using phospho (p) MLKL staining of lung slices. A significant increase in pMLKL signal intensity was most notable at 96 hours of CI in the swollen airway epithelial cells, though was also seen in the alveolar septa to a lesser extent. Longer CI times resulted in increased thickening of the epithelial membrane (Figure 1B) and inflammatory cell infiltration in the interstitial space (Figure 1C). This corresponded to a significant increase in the lung injury score (Figure 1D). The results suggest that, under these experimental conditions, morphologic changes consistent with cellular necroptosis ensue after extended cold ischemic time in the OLT model with predominant effects on the airway epithelium.
Figure 1.
Prolonged cold ischemia (CI) worsens ischemia–reperfusion injury in a mouse model of orthotopic lung transplantation. Using murine syngeneic lung transplantation between C57BL/6 mice (n = 3 mice per group), cold preservation time was prolonged up to 96 hours at 0°C to induce varying severities of CI/repurfusion (R) (A). Shown in A are changes in gross appearance of lungs; low (scale bars = 5 μm) and high magnification (scale bars = 50 μm) of the lung alveolar capillary units. Shown after 96-hour CI, the epithelial cells of the grafted lung manifested swelling with increased translucency, but the nuclei remained intact (A, yellow arrow) using hematoxylin and eosin staining. Phospho-mixed-lineage kinase domain-like kinase (MLKL) staining (right panels) is predominantly localized to the airway epithelium and is markedly increased in the 96-hour CI/24-hour R group compared with the earlier time points. (B) The thickness of the epithelial barrier was measured, demonstrating increased cross-sectional area after 96 hours of CI time. Lung injury scores were determined by evaluating neutrophils in the alveolar and interstitial space, the presence of hyaline membranes, airspace proteinaceous debris, and alveolar septal thickening (22). (C and D) Prolonged CI led to a significant increase of infiltrating inflammatory cells (C) and lung injury (D) of the grafts. Student’s t test. *P < 0.05, **P < 0.01, ***P < 0.001, and ****P < 0.0001. A two-way ANOVA was performed for statistical analysis. Data are expressed as mean (±SEM). NS = not significant.
Necroptosis Is Activated after Prolonged CI
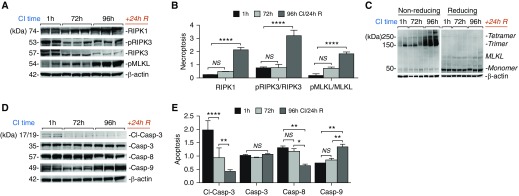
The potential role for necroptosis in experimental IRI after lung transplantation has been recently reported; however, necroptotic cell death pathway activation has not yet been investigated (15). A key step in necroptosis activation is the phosphorylation of MLKL by pRIPK3, which results in pMLKL trimerization, translocation, and channel formation on the plasma membrane through which DAMPs are released (24, 25). Activation of necroptosis was evaluated after 72-hour and 96-hour CI after 24-hour reperfusion, using the transplanted lung with minimal IRI (1-h CI/24 h R [reperfusion]) as a control. In agreement with the pathological findings, the ratio of pRIPK3 to total RIPK3 and pMLKL to total MLKL were substantially increased after 96-hour CI compared with the earlier time points and the control donor lung (Figures 2A and 2B). Moreover, as a marker of necrosome assembly, there was a substantial increase in MLKL trimerization observed in the 96-hour CI group under nonreducing conditions, despite stable total MLKL levels (Figure 2C). Importantly, levels of caspase 8, an inhibitor of necroptosis (24), decreased with prolonged CI (Figures 2D and 2E). Moreover, levels of immune-reactive caspase 9 increased with increasing time of CI, and the ratio of cleaved-to-total caspase 3 levels decreased significantly, indicating that the terminal effector pathway of apoptosis was not activated.
Figure 2.
CI–reperfusion injury activates necroptosis after experimental orthotopic lung transplantation. (A) Immunoblot analysis showing levels of necroptosis effectors receptor-interacting protein kinase (RIPK) 1, pRIPK3, and phospho (p) MLKL are increased after CI–reperfusion injury. (B) Densitometric analysis of immunoblots show significantly increased RIPK1 after normalization to actin (arbitrary units), and ratio of pMLKL/MLKL, pRIPK3/RIPK3 after prolonged CI. (C) As a consequence of MLKL phosphorylation, we observed the formation of MLKL trimer/tetramer complexes consistent with necrosome assembly after 96-hour CI and reperfusion. (D) Levels of death effector caspases are shown by immunoblotting. Cleaved caspase-3 and caspase-8 decreased significantly in the grafted lung after 96-hour CI and reperfusion. (E) Densitometric analysis of immunoblots in D after normalization to actin showing loss of caspase-8, a necroptosis inhibitor, and capase-3, an effector of apoptosis, as the CI time was extended. Each lane in the immunoblots represents protein isolated from a separate mouse whole lung allograft, CI time; n = 2 at 1 hour, n = 3 at 72 hours and 96 hours. A two-way ANOVA was used for statistical analysis. *P < 0.05, **P < 0.01, and ****P < 0.0001. Data are expressed as mean (±SEM).
Cellular Signaling Is Altered after Prolonged CI
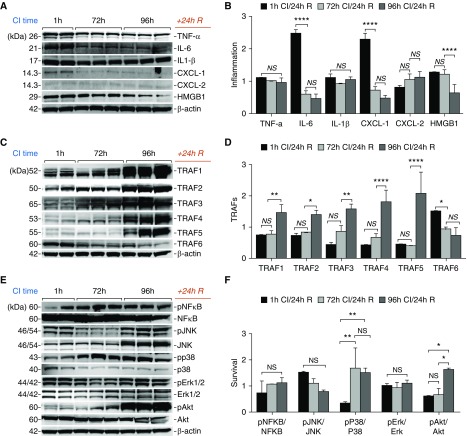
Tissue levels of proinflammatory cytokines, including TNF-α, IL-6, IL1-β, CXCL1, and CXCL2 were found to either decrease or remain at low, undetectable levels with prolonged CI (Figures 3A and 3B). HMGB1, a well-studied DAMP released by necrotic, but not apoptotic, death, decreased significantly in lung tissue after reperfusion after prolonged CI-induced IRI, supporting activation of necroptosis leading to its cellular release (26). TNF-associated factors (TRAFs) are major signal transducers of the TNF superfamily with diverse biological roles that play a crucial role in cell survival signaling, apoptotic signaling, and are recruited by the TNF receptor 1–associated death domain protein for necroptotic pathway activation. Prolonged CI led to a robust increase in levels of TRAF1 to TRAF5, whereas TRAF6 decreased (Figures 3C and 3D). CI triggered an increase in phosphorylation of several cell survival kinase pathways in lung tissue after OLT, including total JNK, phosphorylated AKT, and p38 MAPK (mitogen-activate protein kinase), which was more pronounced after prolonged CI (Figures 3E and 3F). Collectively, these observations indicate that, under these conditions of experimental lung transplantation, multiple signaling pathways are activated to modulate cell survival after sterile inflammation.
Figure 3.
CI–reperfusion injury activates inflammatory signaling after experimental orthotopic lung transplantation. (A) Immunoblot showing unchanged or decreased levels of several proinflammatory cytokines and the damage-associated molecular pattern, HMGB1 (high mobility group box 1). (B) Densitometric analysis after actin normalization of immunoblots in A showing significantly decreased levels of IL-6, CXCL-1, and HMGB1 during CI and R. (C) Levels of TNF-associated factor (TRAF) proteins, which are involved in necroptosis pathway activation, are shown by immunoblotting. (D) Densitometric analysis of immunoblots in C showing that TRAFs (except TRAF6) markedly increased in the grafted lung tissue as CI time was extended to 96 hours. (E and F) Various cell stress and survival kinases were assayed by immunoblot in E and densitometry of bands quantitated as a ratio of phosphorylated to total protein in F. Each lane in the immunoblots represents protein isolated from a separate mouse whole-lung allograft over CI time; n = 2 at 1 hour, n = 3 at 72 hours and 96 hours. A two-way ANOVA was used for statistical analysis. *P < 0.05, **P < 0.01, and ****P < 0.0001. Data are expressed as mean (±SEM).
Comparative Transcriptomic Analysis after Prolonged Donor CI and Lung Transplantation
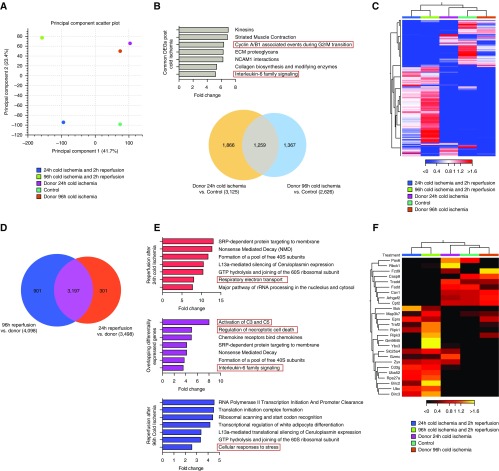
A total of 25,087 genes were identified in a preliminary transcriptomic analysis of prolonged CI time on IRI in lung transplantation by whole-lung RNA-Seq (n = 1 per treatment group). Principal component analysis demonstrated close similarity in the gene profile of 24 hours and 96 hours of CI, with a divergent profile emerging from control lungs along principal component 1 after reperfusion (Figure 4A). There were 3,125 DEGs in donor lungs with 24-hour CI compared with control lungs, and 2,626 DEGs in the 96-hour CI comparator group (Figure 4B). Pathway analysis of 1,259 DEGs shared at the 24-hour and 96-hour CI time points revealed involvement of cell cycle regulation and inflammatory (IL-6) signaling. Hierarchical clustering of the top 500 DEGs revealed clustering of control lungs and those subjected to 24-hour and 96-hour CI alone, compared with a divergent gene expression profile after 2 hours of reperfusion after transplantation (Figure 4C). Reperfusion of donor lungs subjected to prolonged ischemia revealed an overlap of 3,197 DEGs (Figure 4D). Pathway analysis of the shared DEGs after reperfusion indicated that complement activation, regulation of necroptotic cell death, and IL-6 signaling were involved in regulating IRI, and were not present in the donor lung subject to prolonged CI alone (Figure 4E). Activation of pathways involving respiratory electron transport discriminated the earlier 24-hour CI time point from the 96-hour time point, whereas the cellular stress response was enhanced, the latter after 2-hour reperfusion. Consistent with our earlier observations, heatmap visualization of a published necroptotic pathway gene reveals clustering of reperfused donor lungs, suggesting that activation of necroptosis is a potential mechanism for IRI that is not significantly activated before engraftment, despite extended periods of CI (Figure 4F).
Figure 4.
Comparative transcriptomic analysis of the effect of prolonged donor lung CI in experimental lung transplantation. A basic transcriptomic analysis of donor lungs before and after engraftment with reperfusion by RNA sequencing (n = 1 per treatment group). (A) Principal component analysis scatter plot revealing covariance of the 24- and 96-hour donor lung CI, which indicates close similarity in the gene expression profile. After 2 hours of reperfusion, there is a distinct alteration of the projection of the 48- and 96-hour transcriptome compared with control lung reperfusion. (B) Venn diagram representation of differentially expressed genes (DEGs) from 48- and 96-hour donor lung CI compared with control donor lung; 3,544 genes and 2,995 genes, respectively, demonstrated significant overlap of 1,414 genes between the groups; representation factor 3.3, P < 0.0001. Reported genes were filtered with a minimum absolute fold change of 1.5 and a false discovery rate (FDR) P < 0.05. Pathway analysis of genes that were common to both groups revealed processes affecting cell cycle progression, IL-6 signaling, and extracellular matrix (ECM) interactions. (C) Hierarchical cluster heatmap representation of the top 500 DEGs demonstrating clustering of control donor lungs and those subjected to 24- and 96-hour CI compared with a divergent gene expression profile after 2 hours of reperfusion. (D) There is significant overlap of 3,197 genes that are differentially expressed after 2 hours of reperfusion in donor lungs subjected to 24 hours (3,498 DEGs) and 96 hours (4,098 DEGs) of CI time; representation factor 5.6; P < 0.0001. (E) Common and unique DEGs from 24-hour and 96-hour CI after 2 hours of reperfusion, as visualized in the Venn diagram in D, were subjected to pathway analysis using statistical overrepresentation testing in Panther with Benjamini and Hochberg FDR correction. Reperfusion resulted in significant enrichment of genes that involve pathway activation of complement C3 and C5, necroptotic cell death, and inflammatory IL-6 signaling. There was selective enrichment of genes involved in respiratory electron transport in the donor lung after 24-hour CI, and the cellular response to stress in the donor lung after 96-hour CI, after 2 hours of reperfusion, respectively. (F) Heatmap visualization and hierarchical clustering of a necroptotic pathway gene set identified during pathway analysis.
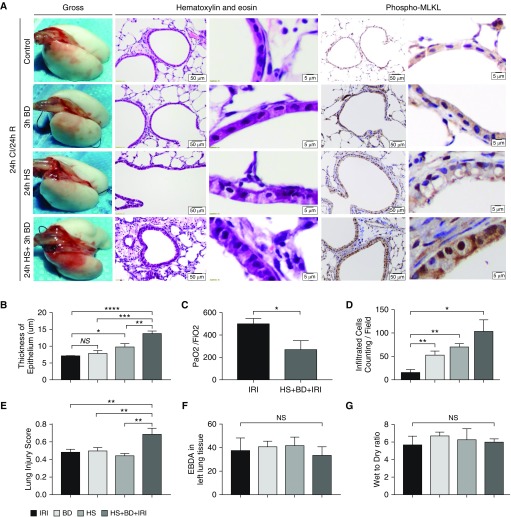
Diffuse Alveolar Damage and Epithelial Edema Are Evident in a Multi-Hit Model of IRI
Findings of PGD were evident in the multi-hit model of IRI after 24 hours of reperfusion as characterized by major pathological features of diffuse alveolar damage and epithelial edema on hematoxylin and eosin stains (Figure 5A). Furthermore, pMLKL staining was observed to be notably increased in the airway epithelium of the “HS + BD + IRI” multi-hit model compared with the “BD + IRI” and the “HS + IRI” models and control lung. The three-hit model, “HS + BD + IRI,” also demonstrated significant changes in epithelial thickness (Figure 5B) and decreased oxygenation (Figure 5C). There was increased cellular infiltration in the “BD + IRI” and the “HS + IRI” groups compared with CI control, and marked increases in the “HS + BD + IRI” group (Figure 5D). Correspondingly, the pathological grading of the severity of donor lung injury showed minimal changes in the “BD + IRI” and the “HS + IRI” groups compared 24-hour CI alone, whereas lung injury was significantly increased in the three-hit model, “HS + BD + IRI” (Figure 5E). We did not detect any significant changes in pulmonary capillary permeability using Evan’s blue dye (Figure 5F) or change in the wet:dry ratio in any of our experimental groups (Figure 5G).
Figure 5.
Pathological characteristics of primary graft dysfunction (PGD) are demonstrated in a clinically relevant multi-hit model of lung injury. To mimic clinical settings, pre-existing donor lung injury was induced by subjecting donor mice to hemorrhagic shock (HS) and/or brain death (BD) in addition to 24-hour CI time (n = 3 mice per group). (A) The three-hit model demonstrates more significant hyaline membrane deposition, bronchial epithelial injury, and inflammatory infiltration around small arteries. Shown are changes in gross appearance of lungs, and low (scale bars = 50 μm) and high magnification (scale bars = 5 μm) of the lung alveolar capillary units. In the three-hit model of HS + BD + ischemia–reperfusion injury (IRI), severity of lung injury was most prominent compared with either one-hit or two-hit models using hematoxylin and eosin staining. pMLKL staining is predominantly localized to the airway epithelium and is increased in the HS + BD + IRI multi-hit model compared with the single-hit models of IRI. (B) Epithelial thickness was increased in the three-hit model compared with control and single-hit models. (C) The ratio of PaO2 to FiO2 showed that graft function remained normal in the one-hit model of IRI but deteriorated significantly in three-hit model of HS + BD + IRI. There was significantly more inflammatory infiltration (D) and higher acute lung injury scores (E) in the three-hit model of HS + BD + IRI. (F and G) The test of Evans-blue dye albumin (EBDA) leakage and wet-to-dry ratio showed no difference in permeability of microvasculature between one-hit and multi-hit models. *P < 0.05, **P < 0.01, ***P < 0.001, and ****P < 0.0001. Two-way ANOVA testing was used for statistical analysis. Data are expressed as mean (±SEM).
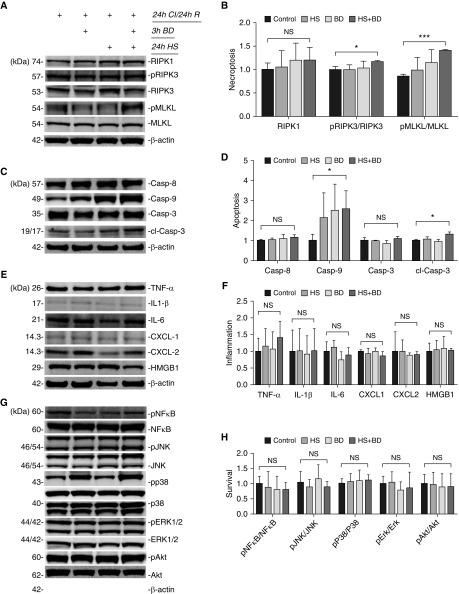
Necroptosis and Apoptosis Cell Death Pathways Are Enhanced in a Multi-Hit Model of IRI
In agreement with the pathological severity of PGD, activation of necroptotic cell death was demonstrated biochemically by a significant increase of pMLKL and modest increases in pRIPK3 in the “HS + BD + IRI” three-hit model, but not in the “BD + IRI” or “HS + IRI” (Figures 6A and 6B). There was a small, but significant, increase in the levels of cleaved caspase 3, caspase 8 levels were unchanged, and caspase 9 levels were increased in the three-hit model (Figures 6C and 6D). Together, these results indicate a small, but contributory, role of apoptosis in cell death pathway activation in our multi-hit model of PGD. In contrast to CI-induced IRI, we did not detect a significant change in either tissue levels of proinflammatory cytokines or phosphorylation of important cell survival signaling pathways, such as p38 MAPK, JNK, and ERK1/2, in any of the multi-hit treatment groups (Figures 6E–6H). Collectively, despite common activation of necroptotic cell death, our findings indicate that intracellular cell death signaling in CI-induced IRI and multi-hit models of donor lung injury are not equivalent, and may belie distinct mechanisms of necroptosis and cell death pathway activation.
Figure 6.
Necroptosis contributes to regulated cell death in a multi-hit model of PGD. Immunoblotting analysis showing effector levels of necroptosis, apoptosis, inflammation, and signaling kinases (n = 3 mice per group). (A) The phosphorylation status of MLKL increased with pre-existing donor lung injury. (B) Densitometric analysis after actin normalization of immunoblots in A showing significantly increased ratios of phosphorylated MLKL (pMLKL) to total MLKL and of pRIPK3 to total RIPK3. (C) In contrast to repressed apoptosis in prolonged CI–reperfusion injury, cleaved caspase-3 increased in the three-hit model of HS + BD + IRI. (D) Densitometric analysis revealed a degree of intrinsic apoptotic pathway activation in addition to necroptosis, which differentiates the mechanisms of cell death activation in the multi-hit model from prolonged cold IRI alone. (E–H) There was no difference detected in levels of proinflammatory cytokines or signaling kinases in lung grafts with or without pre-existing injury. *P < 0.05 and ***P < 0.001. Two-way ANOVA testing was used for statistical analysis. Data are expressed as mean (±SEM).
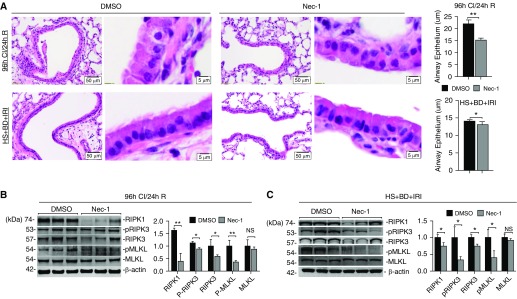
Necrostatin Attenuates Severity of Cellular Death in Experimental Lung Transplantation
Donor lungs subjected to 96-hour CI or to the multi-hit model, “HS + BD + IRI,” of PGD were transplanted into recipient mice that were administered the RIPK1 inhibitor of cell death, Nec-1. There was a significant decrease in airway epithelial thickness of the 96-hour CI/24-hour R group that was evident at low (50 μm) and high (5 μm) magnification after treatment with Nec-1 compared with DMSO control. Epithelial thickening was notably less in the multi-hit model compared with CI IRI model; however, a small, but statistically significant, reduction in airway epithelial thickness was still evident after Nec-1 treatment (see Figure 7A). Immunoblotting showed that levels of RIPK1, pRIPK3, and pMLKL were significantly reduced in mice administered Nec-1 compared with control in the prolonged CI group (see Figure 7B) and in the multi-hit “HS + BD + IRI” group (Figure 7A).
Figure 7.
Necrostatin attenuates necroptotic cell death activation after lung transplantation. Recipient mice were administered DMSO (control) or necrostatin (Nec)-1 (10 mg/kg) 2 hours before lung transplantation (n = 3 mice per group). (A) Pathologic changes of the lung airway epithelium at low (scale bars = 50 μm) and high magnification (scale bars = 5 μm) after treatment with Nec-1 or DMSO. Nec-1 treatment resulted in a significant reduction in airway epithelial thickness in the 96-hour CI/24-hour R group and a small, but statistically significant, reduction in airway epithelial thickness in the HS + BD + IRI group; **P < 0.01 and *P < 0.05, respectively, using a two-tailed Student’s t test. Corresponding immunoblot analysis shows levels of RIPK1 and its downstream effectors are decreased significantly after the treatment of Nec-1 in both one-hit model of prolonged cold IRI (B) and three-hit model of HS + BD + IRI (C); *P < 0.05 and **P < 0.01 versus DMSO using two-way ANOVA. Data are expressed as mean (±SEM).
Discussion
The pathologic findings in our study demonstrate that IRIs in donor lung subjected to prolonged CI time are consistent with activation of necroptotic cell death. Pathway analysis of the donor lung transcriptome revealed that complement activation, the regulation of necroptotic cell death, and chemokine signaling were implicated in IRI. Necroptosis activation was confirmed by immunoassay, demonstrating increased RIPK1 and downstream phosphorylation of RIPK3 and MLKL, which are key steps required for necrosome assembly, and abrogation of this cell death pathway activation and epithelial injury by in vivo administration of necrostatin. Localization of necroptosis pathway activation was determined by a predominant pattern of pMLKL staining in airway epithelia cells and, to a lesser extent, affecting the alveolar septa. Despite notable IRI after prolonged CI alone, characterized by an increased lung injury score, CI was not sufficient to reproduce the classic pathologic features of PGD (27); alveolar structures were not significantly disrupted, there was no significant alveolar edema or hyaline membrane deposition, and minimal cellular influx was observed.
Apoptotic cell death and an excessive inflammatory response have both been implicated in IRI of the lung (28). Here, we found evidence of intrinsic apoptotic signaling via caspase-9 at 96 hours, but not at 72 hours, CI time; however, the key effector of apoptosis, cleaved caspase-3, was reduced at both time points. Though IL1-β was consistently detected in lung tissue undergoing necroptosis, other proinflammatory cytokines, including TNF-α and some chemokines were decreased. Despite this absence of inflammatory signaling after prolonged CI, there was significant activation of the cell stress and survival kinases, p38 MAPK and AKT. Collectively, our findings indicate that intrinsic activation of necroptosis, rather than apoptosis or excessive inflammation, is evident after reperfusion and influences cell death after prolonged CI. Furthermore, PGD is not equivalent to IRI, given the distinct pathologic and biochemical changes induced by CI.
The contribution of donor-related factors, such as activation of the systemic inflammatory response, BD, and HS, are known to be important for the development of PGD in human lung transplantation (29, 30). To explore donor lung factors in the development of PGD, we adopted a multi-hit model of lung injury incorporating antecedent HS and BD in donor mice before lung explant in conjunction with CI-induced IRI. It is reported that 3 hours of BD is sufficient to cause neurogenic pulmonary edema and lung damage in mice (21, 31). We observed increased cellular infiltration and mild epithelial swelling in the two-hit models of “BD + IRI” and “HS + IRI” at 24 hours of reperfusion, which is consistent with that observed in clinical settings, though we did not detect a significant change in lung injury scores (32, 33). However, in the three-hit model of “BD + HS + IRI,” at 24 hours of reperfusion we observed diffuse alveolar damage and pulmonary edema with activation of necroptotic cell death pathways at an earlier time point of 24-hour CI. Indicative of the coinvolvement of apoptotic cell death, levels of caspase-9 and effector cleaved caspase-3 were increased and proinflammatory cytokine responses were absent in the multi-hit model of PGD. Moreover, and contrary to our findings in the prolong CI model, there was no downstream cell stress and survival kinase activation in the multi-hit model.
IRI and the condition of the donor lung before engraftment are major factors contributing to PGD in lung transplantation (8). Experimental models of rodent lung transplantation have demonstrated inconsistencies in the pathological severity of PGD due to IRI at similar time points across studies, which may relate to differences in study design and surgical approach (34–41). It is generally accepted that the limit of CI time in mouse lung transplantation is 18 hours, resulting in severe lung edema, cellular tissue infiltration, and graft failure (34–39). Indeed, in experimental models of orthotopic lung transplantation, donor lung cold ischemic time is varied to produce different degrees of lung injury upon reperfusion (34–41). However, we have demonstrated that the impact of 24-hour CI on isogenic lung grafts in our mouse lung transplant model is minimal, and that isogenic lung grafts can tolerate up 72 hours of CI at 0°C with minimal necroptotic pathway activation. Thus, differences in experimental protocols, as well as potential microsurgical complications, such as anastomotic stenosis or microthrombosis, may have also contributed to the findings of more severe alveolar injury observed in prior studies. However, our work does not exclude other factors, such as animal age, sex, lung ischemia technique, and tissue processing for analysis.
The relative contribution of IRI to the development of PGD has been hitherto poorly defined in lung transplantation. During the preparation of this article, attenuation of necroptotic cell death was shown in rodent lung transplantation using a model of cold IRI (15); this supports the findings in our study and aligns with previous reports regarding the role of necroptosis during IRI of the heart, kidney, liver, retina, and intestine (12; 41–44). Our data demonstrate that necroptotic pathway activation is potentially a critical step leading to necrosome assembly and cell death in IRI after prolonged CI in the lung, and that airway epithelial cells are most susceptible to this form of injury. We have shown necroptosis as a contributory mechanism to the pathology of PGD in a multi-hit model of donor lung injury, a finding that is consistent with the established paradigm of IRI as a risk factor for PGD in human lung transplantation. Furthermore, we have demonstrated that inhibition of necroptosis led to a significant decrease in pathologic epithelial injury in the CI-induced IRI, and, to a lesser extent, in the multi-hit model, which supports the exploration of necroptotic cell death as a therapeutic target in lung transplantation.
In conclusion, our study has elucidated a key role for necroptotic cell death in PGD pathology. Furthermore, we demonstrate that even extreme prolonged CI alone was not sufficient to induce PGD in the mouse OLT model. Our findings show that necroptotic cell death occurs at an earlier time point after CI in donor mice subjected to BD and HS before explant, implicating pre-existing donor lung status as an important factor resulting in the more severe pathobiological changes of PGD. Our findings demonstrate how targeting necroptosis attenuates downstream mediators of necrosome assembly in the mouse OLT model; additional correlative studies are needed to determine whether necroptosis could be a potential therapeutic target to reduce PGD in humans. Our studies also show that longer donor lung CI preservation times are feasible in murine lung transplant, which, if successfully adapted to human transplantation, may complement current clinical strategies, such as ex vivo lung perfusion, to optimize marginal donor lungs before engraftment. Collectively, our findings provide new mechanistic insights into the pathogenesis of PGD, and translation of these concepts may expand the available donor pool and enhance donor selection for lung transplantation.
Supplementary Material
Footnotes
Supported by National Institutes of Health grants R01HL096376, R01HL097376, R01HL098174, R01HL081784, and P01HL114453, and the U.S. Department of Veterans Affairs (VA) Office of Research and Development, Biomedical Laboratory Research and Development and a VA Merit Review award (R.K.M.), the Competitive Medical Research Fund of the University of Pittsburgh and Samuel and Emma Winters Foundation (X.W.), and National Natural Science Foundation of China grant 81470275 (Q.Z.).
Author Contributions: Concept and design: X.W. and R.K.M. Data acquisition: X.W., M.E.O’B., J.Y., C.X., Q.Z., and X.A. Analysis and interpretation: X.W., M.E.O’B., C.X., J.Y., S.L., and L.L. Manuscript preparation and editing: M.E.O’B., X.W., R.K. and J.F.M. All authors read and approved the final version of the manuscript.
Originally Published in Press as DOI: 10.1165/rcmb.2018-0207OC on February 11, 2019
Author disclosures are available with the text of this article at www.atsjournals.org.
References
- 1.Diamond JM, Arcasoy S, Kennedy CC, Eberlein M, Singer JP, Patterson GM, et al. Report of the International Society for Heart and Lung Transplantation Working Group on Primary Lung Graft Dysfunction, part II: epidemiology, risk factors, and outcomes—a 2016 Consensus Group statement of the International Society for Heart and Lung Transplantation. J Heart Lung Transplant. 2017;36:1104–1113. doi: 10.1016/j.healun.2017.07.020. [DOI] [PubMed] [Google Scholar]
- 2.Suzuki Y, Cantu E, Christie JD. Primary graft dysfunction. Semin Respir Crit Care Med. 2013;34:305–319. doi: 10.1055/s-0033-1348474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Gelman AE, Fisher AJ, Huang HJ, Baz MA, Shaver CM, Egan TM, et al. Report of the ISHLT Working Group on Primary Lung Graft Dysfunction part III: mechanisms. A 2016 Consensus Group statement of the International Society for Heart and Lung Transplantation. J Heart Lung Transplant. 2017;36:1114–1120. doi: 10.1016/j.healun.2017.07.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Fiser SM, Tribble CG, Long SM, Kaza AK, Kern JA, Jones DR, et al. Ischemia–reperfusion injury after lung transplantation increases risk of late bronchiolitis obliterans syndrome. Ann Thorac Surg. 2002;73:1041–1047. doi: 10.1016/s0003-4975(01)03606-2. [Discussion, pp. 1047–1048.] [DOI] [PubMed] [Google Scholar]
- 5.Whitson BA, Prekker ME, Herrington CS, Whelan TP, Radosevich DM, Hertz MI, et al. Primary graft dysfunction and long-term pulmonary function after lung transplantation. J Heart Lung Transplant. 2007;26:1004–1011. doi: 10.1016/j.healun.2007.07.018. [DOI] [PubMed] [Google Scholar]
- 6.Fischer S, Maclean AA, Liu M, Cardella JA, Slutsky AS, Suga M, et al. Dynamic changes in apoptotic and necrotic cell death correlate with severity of ischemia–reperfusion injury in lung transplantation. Am J Respir Crit Care Med. 2000;162:1932–1939. doi: 10.1164/ajrccm.162.5.9910064. [DOI] [PubMed] [Google Scholar]
- 7.Israni AK, Zaun D, Rosendale JD, Schaffhausen C, Snyder JJ, Kasiske BL. OPTN/SRTR 2016 annual data report: deceased organ donation. Am J Transplant. 2018;18:434–463. doi: 10.1111/ajt.14563. [DOI] [PubMed] [Google Scholar]
- 8.Chambers DC, Yusen RD, Cherikh WS, Goldfarb SB, Kucheryavaya AY, Khusch K, et al. International Society for Heart and Lung Transplantation. The registry of the International Society for Heart and Lung Transplantation: thirty-fourth adult lung and heart-lung transplantation report–2017; focus theme. Allograft ischemic time. J Heart Lung Transplant. 2017;36:1047–1059. doi: 10.1016/j.healun.2017.07.016. [DOI] [PubMed] [Google Scholar]
- 9.Aigner C, Slama A, Hötzenecker K, Scheed A, Urbanek B, Schmid W, et al. Clinical ex vivo lung perfusion—pushing the limits. Am J Transplant. 2012;12:1839–1847. doi: 10.1111/j.1600-6143.2012.04027.x. [DOI] [PubMed] [Google Scholar]
- 10.Floerchinger B, Oberhuber R, Tullius SG. Effects of brain death on organ quality and transplant outcome. Transplant Rev (Orlando) 2012;26:54–59. doi: 10.1016/j.trre.2011.10.001. [DOI] [PubMed] [Google Scholar]
- 11.Linkermann A, Green DR. Necroptosis. N Engl J Med. 2014;370:455–465. doi: 10.1056/NEJMra1310050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lau A, Wang S, Jiang J, Haig A, Pavlosky A, Linkermann A, et al. RIPK3-mediated necroptosis promotes donor kidney inflammatory injury and reduces allograft survival. Am J Transplant. 2013;13:2805–2818. doi: 10.1111/ajt.12447. [DOI] [PubMed] [Google Scholar]
- 13.Linkermann A, Bräsen JH, Himmerkus N, Liu S, Huber TB, Kunzendorf U, et al. Rip1 (receptor-interacting protein kinase 1) mediates necroptosis and contributes to renal ischemia/reperfusion injury. Kidney Int. 2012;81:751–761. doi: 10.1038/ki.2011.450. [DOI] [PubMed] [Google Scholar]
- 14.Pavlosky A, Lau A, Su Y, Lian D, Huang X, Yin Z, et al. RIPK3-mediated necroptosis regulates cardiac allograft rejection. Am J Transplant. 2014;14:1778–1790. doi: 10.1111/ajt.12779. [DOI] [PubMed] [Google Scholar]
- 15.Kanou T, Ohsumi A, Kim H, Chen M, Bai X, Guan Z, et al. Inhibition of regulated necrosis attenuates receptor-interacting protein kinase 1-mediated ischemia–reperfusion injury after lung transplantation. J Heart Lung Transplant. 2018;37:1261–1270. doi: 10.1016/j.healun.2018.04.005. [DOI] [PubMed] [Google Scholar]
- 16.Okazaki M, Krupnick AS, Kornfeld CG, Lai JM, Ritter JH, Richardson SB, et al. A mouse model of orthotopic vascularized aerated lung transplantation. Am J Transplant. 2007;7:1672–1679. doi: 10.1111/j.1600-6143.2007.01819.x. [DOI] [PubMed] [Google Scholar]
- 17.Gonzales JN, Gorshkov B, Varn MN, Zemskova MA, Zemskov EA, Sridhar S, et al. Protective effect of adenosine receptors against lipopolysaccharide-induced acute lung injury. Am J Physiol Lung Cell Mol Physiol. 2014;306:L497–L507. doi: 10.1152/ajplung.00086.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kohut LK, Darwiche SS, Brumfield JM, Frank AM, Billiar TR. Fixed volume or fixed pressure: a murine model of hemorrhagic shock. J Vis Exp. 2011;(52):pii:2068. doi: 10.3791/2068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wen Z, Fan L, Li Y, Zou Z, Scott MJ, Xiao G, et al. Neutrophils counteract autophagy-mediated anti-inflammatory mechanisms in alveolar macrophage: role in posthemorrhagic shock acute lung inflammation. J Immunol. 2014;193:4623–4633. doi: 10.4049/jimmunol.1400899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Pomper G, Trescher K, Santer D, Hasun M, Baumgartner A, Adelmann K, et al. Introducing a mouse model of brain death. J Neurosci Methods. 2010;192:70–74. doi: 10.1016/j.jneumeth.2010.07.019. [DOI] [PubMed] [Google Scholar]
- 21.Wauters S, Koole M, Vermaelen P, Somers J, Van Laere K, Van Loon J, et al. Fluoro-D-glucose-micro positron emission tomography as a diagnostic tool to confirm brain death in a murine donor lung injury model. J Surg Res. 2013;180:343–348. doi: 10.1016/j.jss.2012.05.011. [DOI] [PubMed] [Google Scholar]
- 22.Matute-Bello G, Downey G, Moore BB, Groshong SD, Matthay MA, Slutsky AS, et al. Acute Lung Injury in Animals Study Group. An official American Thoracic Society workshop report: features and measurements of experimental acute lung injury in animals. Am J Respir Cell Mol Biol. 2011;44:725–738. doi: 10.1165/rcmb.2009-0210ST. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Croft D, O’Kelly G, Wu G, Haw R, Gillespie M, Matthews L, et al. Reactome: a database of reactions, pathways and biological processes. Nucleic Acids Res. 2011;39:D691–D697. doi: 10.1093/nar/gkq1018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Pasparakis M, Vandenabeele P. Necroptosis and its role in inflammation. Nature. 2015;517:311–320. doi: 10.1038/nature14191. [DOI] [PubMed] [Google Scholar]
- 25.Cai Z, Jitkaew S, Zhao J, Chiang HC, Choksi S, Liu J, et al. Plasma membrane translocation of trimerized MLKL protein is required for TNF-induced necroptosis. Nat Cell Biol. 2014;16:55–65. doi: 10.1038/ncb2883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Weber DJ, Allette YM, Wilkes DS, White FA. The HMGB1-RAGE inflammatory pathway: implications for brain injury-induced pulmonary dysfunction. Antioxid Redox Signal. 2015;23:1316–1328. doi: 10.1089/ars.2015.6299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Morrison MI, Pither TL, Fisher AJ. Pathophysiology and classification of primary graft dysfunction after lung transplantation. J Thorac Dis. 2017;9:4084–4097. doi: 10.21037/jtd.2017.09.09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Ng CS, Wan S, Arifi AA, Yim AP. Inflammatory response to pulmonary ischemia–reperfusion injury. Surg Today. 2006;36:205–214. doi: 10.1007/s00595-005-3124-2. [DOI] [PubMed] [Google Scholar]
- 29.Avlonitis VS, Fisher AJ, Kirby JA, Dark JH. Pulmonary transplantation: the role of brain death in donor lung injury. Transplantation. 2003;75:1928–1933. doi: 10.1097/01.TP.0000066351.87480.9E. [DOI] [PubMed] [Google Scholar]
- 30.de Perrot M, Bonser RS, Dark J, Kelly RF, McGiffin D, Menza R, et al. ISHLT Working Group on Primary Lung Graft Dysfunction. Report of the ISHLT Working Group on Primary Lung Graft Dysfunction part III: donor-related risk factors and markers. J Heart Lung Transplant. 2005;24:1460–1467. doi: 10.1016/j.healun.2005.02.017. [DOI] [PubMed] [Google Scholar]
- 31.Wauters S, Somers J, De Vleeschauwer S, Verbeken E, Verleden GM, van Loon J, et al. Evaluating lung injury at increasing time intervals in a murine brain death model. J Surg Res. 2013;183:419–426. doi: 10.1016/j.jss.2013.01.011. [DOI] [PubMed] [Google Scholar]
- 32.Belhaj A, Dewachter L, Rorive S, Remmelink M, Weynand B, Melot C, et al. Mechanical versus humoral determinants of brain death–induced lung injury. PLoS One. 2017;12:e0181899. doi: 10.1371/journal.pone.0181899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Pecoraro Y, Tsushima Y, Opitz I, Benden C, Schüpbach R, Lenherr R, et al. Impact of time interval between donor brain death and cold preservation on long-term outcome in lung transplantation. Eur J Cardiothorac Surg. 2016;50:264–268. doi: 10.1093/ejcts/ezw028. [DOI] [PubMed] [Google Scholar]
- 34.Lama VN, Belperio JA, Christie JD, El-Chemaly S, Fishbein MC, Gelman AE, et al. Models of lung transplant research: a consensus statement from the National Heart, Lung, and Blood Institute workshop. JCI Insight. 2017;2:pii:93121. doi: 10.1172/jci.insight.93121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Watanabe T, Hoshikawa Y, Ishibashi N, Suzuki H, Notsuda H, Watanabe Y, et al. Mesenchymal stem cells attenuate ischemia–reperfusion injury after prolonged cold ischemia in a mouse model of lung transplantation: a preliminary study. Surg Today. 2017;47:425–431. doi: 10.1007/s00595-016-1391-8. [DOI] [PubMed] [Google Scholar]
- 36.Mallavia B, Liu F, Sheppard D, Looney MR. Inhibiting integrin αvβ5 reduces ischemia–reperfusion injury in an orthotopic lung transplant model in mice. Am J Transplant. 2016;16:1306–1311. doi: 10.1111/ajt.13605. [DOI] [PubMed] [Google Scholar]
- 37.Kreisel D, Sugimoto S, Zhu J, Nava R, Li W, Okazaki M, et al. Emergency granulopoiesis promotes neutrophil–dendritic cell encounters that prevent mouse lung allograft acceptance. Blood. 2011;118:6172–6182. doi: 10.1182/blood-2011-04-347823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Yamada Y, Laube I, Jang JH, Bonvini JM, Inci I, Weder W, et al. Sevoflurane preconditioning protects from posttransplant injury in mouse lung transplantation. J Surg Res. 2017;214:270–277. doi: 10.1016/j.jss.2017.03.021. [DOI] [PubMed] [Google Scholar]
- 39.Chang JE, Kim HJ, Yi E, Jheon S, Kim K. Reduction of ischaemia–reperfusion injury in a rat lung transplantation model by low-concentration GV1001. Eur J Cardiothorac Surg. 2016;50:972–979. doi: 10.1093/ejcts/ezw135. [DOI] [PubMed] [Google Scholar]
- 40.Guler L, Tavlasoglu M, Yucel O, Guler A, Sahin MA, Kurkluoglu M, et al. Taurine attenuates lung ischemia–reperfusion injury after lung transplantation in rats. J Anesth. 2014;28:347–353. doi: 10.1007/s00540-013-1741-2. [DOI] [PubMed] [Google Scholar]
- 41.Wen S, Ling Y, Yang W, Shen J, Li C, Deng W, et al. Necroptosis is a key mediator of enterocytes loss in intestinal ischaemia/reperfusion injury. J Cell Mol Med. 2017;21:432–443. doi: 10.1111/jcmm.12987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Hong JM, Kim SJ, Lee SM. Role of necroptosis in autophagy signaling during hepatic ischemia and reperfusion. Toxicol Appl Pharmacol. 2016;308:1–10. doi: 10.1016/j.taap.2016.08.010. [DOI] [PubMed] [Google Scholar]
- 43.Tuuminen R, Holmström E, Raissadati A, Saharinen P, Rouvinen E, Krebs R, et al. Simvastatin pretreatment reduces caspase-9 and RIPK1 protein activity in rat cardiac allograft ischemia–reperfusion. Transpl Immunol. 2016;37:40–45. doi: 10.1016/j.trim.2016.05.001. [DOI] [PubMed] [Google Scholar]
- 44.Kim CR, Kim JH, Park HL, Park CK. Ischemia–reperfusion injury triggers TNFα induced-necroptosis in rat retina. Curr Eye Res. 2017;42:771–779. doi: 10.1080/02713683.2016.1227449. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.