Abstract
Signal transducer and activator of transcription 3 (STAT3) is a cytoplasmic transcription factor that regulates cell proliferation, differentiation, apoptosis, angiogenesis, inflammation and immune responses. Aberrant STAT3 activation triggers tumor progression through oncogenic gene expression in numerous human cancers, leading to promote tumor malignancy. On the contrary, STAT3 activation in immune cells cause elevation of immunosuppressive factors. Accumulating evidence suggests that the tumor microenvironment closely interacts with the STAT3 signaling pathway. So, targeting STAT3 may improve tumor progression, and anti-cancer immune response. In this review, we summarized the role of STAT3 in cancer and the tumor microenvironment, and present inhibitors of STAT3 signaling cascades.
Keywords: Cancer stem cells, Cancer therapy, Immune suppression, STAT3, Tumor microenvironment
INTRODUCTION
Hallmarks of cancer consist of sustaining cellular proliferative signals, attenuating cell death, inappropriate replication with lacking growth suppressors, inducing angiogenesis and promoting invasion and metastasis in tumorigenesis (1). Recently, the impact of the tumor microenvironment and tumor-induced immune suppression on tumor progression, has been subjected to intense investigation, and the STAT3, is a crucial mediator of tumor cell progression and tumor-associated immunosuppression.
STAT3 is integral for transducing signals from receptor and/or non-receptor tyrosine kinases activated in cancer cells, as well as transcriptional factors regulating expression of numerous gene contributing tumor progression (2). STAT3 signaling cascade is triggered by upstream kinase signals, and undergo phosphorylation, homo-dimerization, translocate in to nuclear, and bind to DNA, leading to target gene expression involved in tumor cell proliferation, angiogenesis, metastasis, and immunoediting (3–5).
The tumor microenvironment is composed of tumor cells and their surrounding circumstance, including hypoxic condition, blood vessels and extracellular matrix (ECM), as well as stromal cells, immune cells, and inflammatory cells (6, 7). STAT3 is a key mediator modulating tumor milieu to promote tumor progression, and is a promising target for antitumor immune response (8, 9).
Emerging evidence suggests the key role of STAT3 in cancer cells and their microenvironment. However, there are knowledge gaps remaining regarding interaction between STAT3 signaling, and the tumor microenvironment immune system. Therefore, this review article summarizes recent reports related to the role of STAT3 in cancer cells, and the relationship between cancer cells and tumor microenvironment in tumor progression. Also, this review focuses on the therapeutic agents and inhibitors that specifically target STAT3.
PERSISTENT STAT3 ACTIVATION IN CANCER CELLS
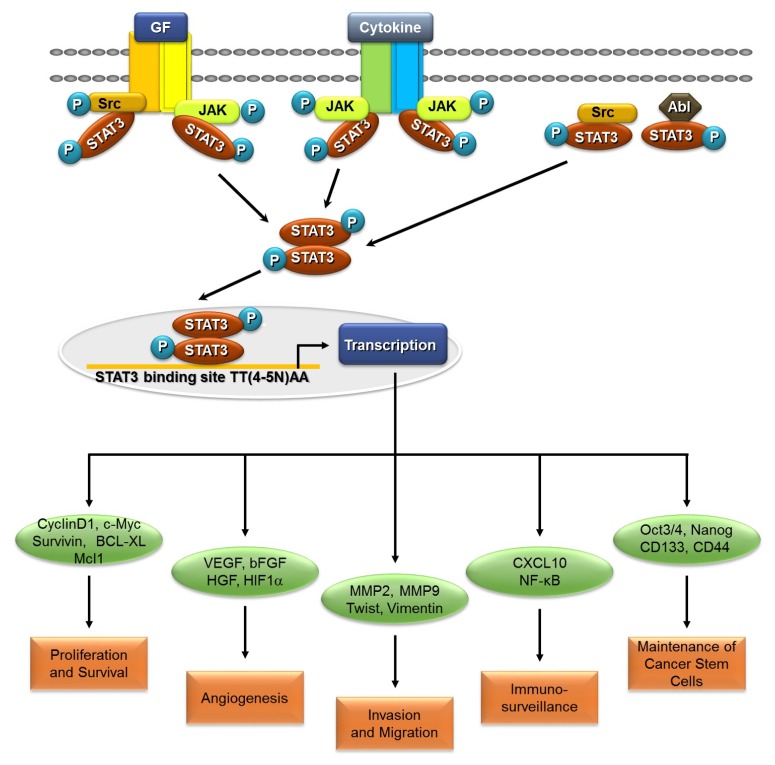
Aberrant activation of STAT3 has been involved in oncogenesis and malignant phenotypes in human cancers (10, 11). Hyperactivation of STAT3 has been reported in several types of tumors, including head-and neck, brain, breast, liver, lung, kidney, pancreas, prostate, ovary cancer, and multiple myeloma, as well as acute myeloid leukemia (AML) (12–21). Expression levels of activated STAT3 are positively correlated with poor prognosis in these cancers. Constitutive STAT3 activation is primarily due to hyperactivation of growth factor receptor tyrosine kinase and overexpression of stimulatory receptor-ligand interactions. Phosphorylation of tyrosine 705 residue, leads to nuclear translocation of STAT3, which allow induction of STAT3 target genes (4). As an oncogene, STAT3 is a major signal transduction pathway involved in multiple cellular processes, including proliferation, survival, angiogenesis, metastasis, invasion, and immune escape (22–24) (Fig. 1).
Fig. 1.
STAT3 signaling in cancer. STAT3 signaling is activated by binding of various ligands to their cell surface receptors, leading to phosphorylation of STAT3. STAT3 also directly phosphorylated by Src and Abl, which are non-receptor tyrosine kinases. Phosphorylated STAT3 further homo-dimerized and translocated, to the nucleus. STAT3 regulate CyclinD1, c-Myc, Survivin, Bcl-XL, and Mcl1, which regulate cellular proliferation and survival. STAT3 up-regulates VEGF, bFGF, HGF, and HIF1α. Additionally, STAT3 also regulates MMP2, MMP9, Twist, and Vimentin, for invasion and migration. STAT3 activation also downregulates immune surveillance, by secretion of pro-inflammatory cytokines. Furthermore, maintaining cancer stem cell properties, STAT3 regulates Oct3/4, Nanog, CD133, and CD44.
STAT3 accelerates the cellular proliferation and survival
Accumulating evidence shows that STAT3 activation, participates in cellular proliferation and survival. Persistent activation of STAT3 induces up-regulated expression of CyclnD1, c-Myc and Survivin, to accelerate cell cycle progression in renal and colon cancers (25–27). Correspond to its role in cellular proliferation, multiple studies have shown that STAT3 signaling pathway suppresses apoptosis in cancer cells. Activated STAT3 also upregulates anti-apoptotic protein such as Bcl-2 (B-cell lymphoma-2), Bcl-XL (B-cell lymphoma-2-like 1), and Mcl1 (myeloid cell leukemia sequence 1) expressions to prevent apoptosis of tumor cells in multiple myeloma (28, 29) (Table 1). Inhibition of STAT3 results in decreased cell proliferation, and promotes apoptosis in various cancers including breast cancer, colorectal cancer, gastric cancer, lung cancer, and so on (30–32). According to these studies, STAT3 is a key regulator of cancer cell proliferation and survival.
Table 1.
The target genes of STAT3
| Function | Upregulated gene | Downregulated gene | Refs. |
|---|---|---|---|
| Proliferation | BCL-XL | (26) | |
| c-MYC | (23) | ||
| Mcl1 | (27) | ||
| Survivin | (25) | ||
| Cyclin-D1 | (24) | ||
| Angiogenesis | VEGF | (32, 33,35) | |
| HIF-1α | (34,35) | ||
| HGF | (36) | ||
| bFGF | (36) | ||
| IL-12 | (4) | ||
| IFNβ | (4) | ||
| IFNγ | (8) | ||
| CXCL10 | (4) | ||
| Metastasis | MMP2 | (42) | |
| MMP9 | (43) | ||
| MMP1 | (44) | ||
| TWIST1 | (37) | ||
| Vimentin | (38) | ||
| HMGB1 | (40) | ||
| ZEB1 | (41) | ||
| Immune escape | IL-6 | (4, 47, 48) | |
| IL-10 | (47) | ||
| IFNβ | (4) | ||
| IFNγ | (8) | ||
| IL-12 | (4) | ||
| CD80 | (4) | ||
| CD86 | (4) | ||
| CCL5 | (4) | ||
| CXCL10 | (4,49) |
STAT3 enhances the angiogenesis
The formation of a new blood vessel called angiogenesis, is a fundamental step in tumor growth and metastasis.
It is well known that STAT3 induces vascular endothelial growth factor (VEGF) directly, which is the most angiogenic molecule (33, 34). Moreover, STAT3 induces hypoxiainducible factor-1α (HIF-1α), another regulator of angiogenesis (35). During hypoxic conditions in core of cancer cells, STAT3 and HIF1α bind to the VEGF promoter, leading to angiogenesis (36). Additionally, pro-angiogenic factors such as bFGF (basic fibroblast growth factor) and HGF (hepatocyte growth factor), also downstream target of STAT3 (37) (Table 1).
STAT3 contributes to promotion of metastasis
Cancer metastasis is a complicated procedure in which cancer cells invade adjacent tissue enabling such cells to accomplish migration and invasion, known as epithelial-mesenchymal transition (EMT). According to previous studies, STAT3 activation is pivotal in regulating expression of Twist, Vimentin, Snail, HMGB1 (high-mobility group box 1), ZEB1 (zinc finger E-box binding homeobox 1), and so on (38–42). Persistent STAT3 activation, leads to upregulated expression of MMP2 (matrix metalloproteinase 2) (43). Moreover, STAT3 activation also regulates other matrix metalloproteinases, such as MMP9 and MMP1 (44, 45) (Table 1). Based on these studies, STAT3 activation promotes cellular invasion. Additionally, earlier studies have shown evidence that aberrant STAT3 activation is required for cell motility, and plays a key role in wound healing and migration (46, 47). Thus, inhibition of neoangiogenic factors and/or migration factors by suppressing STAT3 signaling pathway is an attractive strategy for preventing tumor aggressiveness.
STAT3 induces the immune evasion
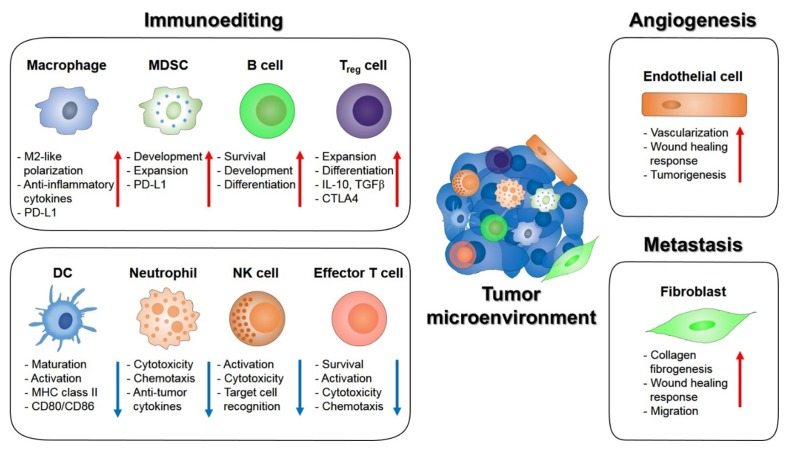
Tumor immune surveillance plays a pivotal role in identifying cancerous and/or precancerous cells, and eliminates them before they abnormally transform. Recent findings show that abnormal cells may evade the immune system, to form malignant cancers. Additionally, hyperactivated STAT3 in tumor cells and tumor-associated immune cells, could enhance tumor immune evasion, or establish immune tolerance (Fig. 2).
Fig. 2.
The role of STAT3 signaling in the tumor microenvironment. STAT3 signaling supports the communication between tumor cells and the tumor microenvironments. STAT3 drives immunosuppressive effects and tumor promoting effects by endothelial cells and fibroblasts. Activation of STAT3 in dendritic (DC) cells suppresses maturation, activation and antigen presentation which promotes immune tolerance. STAT3 activation in neutrophil, NK cells and effector T cells also has immunosuppressive effects. STAT3 signaling in macrophage favors M2-like polarization and increases PD-L1 expression while STAT3 activation proliferates MDSC population. STAT3 exerts immune tolerance in regulatory T (Treg) cells by enhancing CTLA4 expression and tumorigenesis in B cells by promoting survival, proliferation and development. STAT3 effect on endothelial cells to promote tumor vascularization. STAT3 in tumor associated fibroblast also enhance tumor metastasis. Collectively, STAT3 signaling is a key regulator of hallmark of cancers.
Numerous mechanisms which cancer cells escape from detection, include induction of immunosuppressive cytokines such as IL-6, IL-10 and TGF-β and reduction of cancer antigens, and MHC-I and MHC-II (major histocompatibility complex) molecules for T cells (4). Several lines of evidence implicate suppression of STAT3 activation elevates release of proinflammatory cytokines and/or chemokines, suggesting activation of STAT3 negatively regulates the expression of immune stimulating molecules (4). In addition, STAT3 also promotes pro-inflammatory mediators via nuclear factor kappa B (NF-κB) signaling pathways. IL-6/GP130/JAK signaling pathway promotes STAT3 recruitment in colon cancer cells and T cells, which upregulate IL-10 secretion (48, 49). Additionally, STAT3 downregulates C-X-C motif chemokine ligand 10 (CXCL10) expression, which could enhance cytotoxicity of natural killer (NK) cells (50) (Table 1). Emerging evidence indicates that STAT3 inhibitors reduce immune evasion, thus upregulating anti-tumor ability of immune cells.
STAT3 maintains the cancer stem cells
Cancer stem cells (CSCs) have a significant role in cancer initiation and progression. CSCs have characteristics of self-renewal and capacity to generate various tumor cells, thus providing tumor heterogeneity. Additionally, CSCs are responsible for cancer development, metastasis, and drug resistance (51). STAT3 plays significant role in the tumor inflammatory environment with high expression of ROS, leading to DNA damage and oncogene activation (52). This demonstrates that STAT3 activation is also involved in CSCs regulation. Recent studies have shown that STAT3 activation is essential in various cancer types, including prostate, breast cancer, hepatocellular carcinoma (HCC), colorectal cancer, and glioblastoma (53–57).
STAT3 activation by IL-6 or ROS, results in upregulated self-renewal ability of prostate CSCs (58). Additionally, glioma-associated-human mesenchymal stem cells (GA-hMSC) enhance glioma stemness through the IL-6/gp130/STAT3 pathway (59). High levels of aldehyde dehydrogenase (ALDH) activity in endometrial cancer, upregulates CSC activities through IL-6/JAK1/STAT3 signaling pathways. Inhibition of these pathways significantly reduced tumor cell growth (60).
Activated STAT3 in CSCs required co-expression of pluripotent stem cell markers, Oct3/4 and Nanog (61). These signaling pathways upregulate CSC markers such as CD44, thereby increasing CSC properties (62). Moreover, high levels of CSC marker, CD133, positively correlate with poor prognosis and tumor growth in HCC. On the contrary, inhibition of CD133 resulted in cell cycle arrest and tumor suppression, by downregulating cytokine-related genes. Treatment with sorafenib and nifuroxazide lead to inhibition of STAT3 activation, and CD133 expression (55). Recent investigation showed that VEGF promotes self-renewal capacity through VEGFR2/STAT3 signaling pathway, by upregulating Myc and Sox2 expression (63). Highly activated STAT3 correlates with increased self-renewing and radiochemoresistant abilities, in thyroid cancer-derived CD133+ cells (64). Due to the importance of STAT3 maintaining CSC properties such as self-renewing abilities in carcinogenesis, blocking this signaling pathway may eliminate CSCs in preventing cancer.
STAT3 IN THE TUMOR MICROENVIRONMENT
It is well known that tumor cells modify and adapt to their surrounding milieu. Constitutive activation of STAT3 promotes tumor growth through oncogenic signaling pathway, and interacts with tumor cells and their surrounding factors. Aberrant activation of STAT3 recruits immune cells and compromises their functions to benefit tumor cells (65). Additionally, STAT3 is a negative regulator of T helper 1 cells, suggesting inhibition of STAT3 activation, promotes release of proinflammatory cytokines (4).
In the core of tumor tissue, hypoxic stress is generated and therefore induces hypoxia-inducible factors. It is known that STAT3 regulates stability and activity of HIF-1α, inducing expression of cytokines, chemokine, and growth factors to improve cancer development (66, 67). Also, in response to surrounding tumor cells, stromal cells upregulate their C-X-C motif chemokine ligand 12 (CXCL12) receptors, resulting in enhancing metastatic potential in tumor cells (68). Additionally, activation of STAT3 promotes polarization of tumor-associated microphages as M2 phenotype and PD-L1 expression as well, which increase tumor progression. Inhibition of STAT3 activation shows anti-tumor activity by suppressing polarization of macrophages (69). In addition, activation of STAT3 in endothelial cells increases cell adhesion molecule expression and it is important for the tumor metastasis (70).
Tumor cells can evade immune response by regulating their immunological circumstance. Activation of STAT3 is crucial for immune escape of tumor cells, by promoting transforming growth factor-β (TGF-β), VEGF, myeloid-derived suppressor cell (MDSC) expansion and suppressing NK cell function (71–73). Using STAT3 inhibitors has shown reduction of immunosuppressive response, therefore upregulating antitumor activity of immune cells (Fig. 2).
TARGETING STAT3 IN CANCER
Since STAT3 regulates a central role in cell proliferation, differentiation, apoptosis, angiogenesis, immune response and metastasis, STAT3 is rational strategy for development of novel cancer therapeutics (74). STAT3 inhibitors or agents can have two major strategies, in which STAT3 activation is inhibited, directly or indirectly. Direct inhibitors block the SH2 domain, DNA-binding domain, and N-terminal domain, which regulate STAT3 activation by blocking phosphorylation, dimerization, nuclear translocation, and DNA binding (75, 76). Indirect inhibitors target upstream regulators of STAT3 pathway, such as receptor-ligand binding and kinases.
SH2 domain inhibitors
The SH2 domain of STAT3 has a binding pocket to phosphorylated tyrosine (pTyr) residue, and formation of STAT3 dimerization involves pTyr interacting with the SH2 domain. Therefore, inhibiting SH2 domain of STAT3 suppresses activation of STAT3 protein. Numerous kinds of small molecule peptides have been developed as STAT3 inhibitors that directly target the SH2 domain of STAT3 by using high-throughput screening and structure-based virtual screening system. These small molecules and peptides include PY*LKTK (Y* is the phosphorylated tyrosine) (77), S3I-M2001 (78), S3I-1757 (79), curcumin-proline (80), cryptotashinone (81), STA-21 (82), Stattic (83) and S3I-201 (Table 2) (84).
Table 2.
Small molecule inhibitors directly/indirectly targeting STAT3
| Inhibitor Name | Mechanism of Action | Cancer Type | Refs. |
|---|---|---|---|
| PY*LKTK | SH2 domain inhibitor | NIH 3T3/v-Src fibroblasts | (75) |
| S3I-M2001 | SH2 domain inhibitor | Breast cancer | (76) |
| S3I-1757 | SH2 domain inhibitor | Breast and lung cancer | (77) |
| Curcumin-proline | SH2 domain inhibitor | (78) | |
| Cryptotashinone | SH2 domain inhibitor | Prostate cancer | (79) |
| STA-21 | SH2 domain inhibitor | Breast cancer | (80) |
| Stattic | SH2 domain inhibitor | Breast cancer | (81) |
| S3I-201 | SH2 domain inhibitor | Breast cancer, prostate cancer, acute myeloid leukemia and human multiple myeloma | (82) |
| HIC 1 | DNA binding domain inhibitor | Breast cancer | (83) |
| IS3-295 | DNA binding domain inhibitor | Colon cancer | (84) |
| DBD-1 | DNA binding domain inhibitor | Melanoma | (85) |
| KDI1 | RTK inhibitor | Vulval and breast cancer | (86) |
| PD153035 | RTK inhibitor | Oral squamous carcinoma | (87) |
| AG490 | JAK kinase inhibitor | Pancreatic cancer | (88) |
| WP1066 | JAK kinase inhibitor | Acute myelogenous leukemia | (89) |
| TG101209 | JAK2 kinase inhibitor | Acute myeloid leukemia | (90) |
| AZD1480 | JAK kinase inhibitor | Myeloma, Neuroblastoma and Pediatric sarcomas | (91, 92) |
| Dasatinib | Src and PDGF inhibitor | Synovial sarcoma, hepatocellular carcinoma, glioma, prostate cancer | (93) |
| PP2 | Src inhibitor | Intestinal epithelial cell | (97, 100) |
| KX2-391 | Src inhibitor | Prostate cancer | (98) |
| AZD0530 | Src inhibitor | Melanoma | (99) |
| MLS-2384 | Src and JAK inhibitor | Prostate, breast, skin, ovarian, lung, and liver cancer | (108) |
| Sophoraflavanone G | Src and JAK inhibitor | Breast, prostate, lymphoma, human multiple myeloma, large cell lung cancer, colorectal carcinoma | (101) |
DNA binding domain inhibitors
STAT3 has a DNA binding domain, and binds to the gene’s promoter and regulates gene expression. Thus, targeting the DNA binding domain of STAT3 interrupts interaction with the promoter of target gene, thereby inhibiting activity of STAT3, and various inhibitors have been developed. These small molecules include HIC 1 (85), IS3-295 (86) and DBD-1 (Table 2) (87).
STAT3 upstream regulatory inhibitors
Receptor-associated and non-receptor tyrosine kinases are critical upstream regulators of STAT3 activation, so targeting these kinases has attractive potential for STAT3 activation.
KDI1, one of the receptor tyrosine kinases (RTK) inhibitor, complexes with EGFR and inhibits EGF-induced STAT3 phosphorylation (88). Another RTK inhibitor, PD153035, suppresses phosphorylation and activation of EGFR and STAT3 in vivo. This is reported to inhibit the growth of oral squamous cell carcinoma (89).
Additionally, STAT3 is phosphorylated by various protein kinases in the cytoplasmic region. It is well known that JAK and Src kinases are common STAT3 upstream regulators. JAK and Src kinases inhibitors have various anti-cancer effects such as inducing cancer cell apoptosis and reducing metastasis through decrease in the level of STAT3 phosphorylation (90–103). Some of these small molecule inhibitors have recently been in clinical trials for chemotherapy for various cancer treatment, and inflammatory syndromes including rheumatoid arthritis, psoriasis, and inflammatory bowel disease (IBD) (99, 104–110).
CONCLUSION
Although STAT3 expression is properly controlled in normal cells, constitutive activation of STAT3 occurs in various cancers. Aberrant activation of STAT3 provides favorable conditions for tumor metastasis involved in tumor cell proliferation, angiogenesis, migration, and invasion. In addition, induction of STAT3 signaling has a pivotal role in evasion of immune surveillance. Aberrant activation of STAT3 leads to burn out of immune cells, so, STAT3 signaling is an instigator of immune evasion in the tumor microenvironment. STAT3 signaling regulate oncogenic pathway in tumor cells, but also mediate immune evasion. Therefore, targeting STAT3 inhibits tumor progression and improves anti-tumor immune responses as well. Thus, it is a valuable therapeutic target for cancer therapy.
The tumor microenvironment consists of heterogeneous population of cancer cells and various infiltrating cells, secreted factors and extracellular matrix (ECM) proteins, and their surrounding circumstance such as blood vessels and hypoxic region. The interactions of tumor cells with their microenvironments promotes development and progression of tumor cell thought STAT3 signaling pathways, thus interrupting this signaling pathway in the tumor microenvironment is a promising target for cancer therapy.
Despite various small molecule inhibitors effectively inhibiting STAT3 signaling, further studies will be innovatively developed to improve clinical outcomes. Therefore, as this review suggests, future perspectives targeting STAT3 should focus on various combination therapies that regulate tumor cells as well as the tumor microenvironment.
ACKNOWLEDGEMENTS
This work was supported by grants from the National Research Foundation of Korea (NRF) funded by the Korea government (NRF-2014R1A2A1A11053203, NRF-2017R1A2B2006839 and NRF-2018R1A5A2025964), and the Seoul National University Hospital (SNUH) Research Fund (0320190210).
Footnotes
CONFLICTS OF INTEREST
The authors have no conflicting interests.
REFERENCES
- 1.Hanahan D, Weinberg Robert A. Hallmarks of Cancer: The Next Generation. Cell. 2011;144:646–674. doi: 10.1016/j.cell.2011.02.013. [DOI] [PubMed] [Google Scholar]
- 2.Yu H, Jove R. The STATs of cancer — new molecular targets come of age. Nat Rev Cancer. 2004;4:97–105. doi: 10.1038/nrc1275. [DOI] [PubMed] [Google Scholar]
- 3.Chen J, Wang J, Lin L, et al. Inhibition of STAT3 Signaling Pathway by Nitidine Chloride Suppressed the Angiogenesis and Growth of Human Gastric Cancer. Mol Cancer Ther. 2012;11:277–287. doi: 10.1158/1535-7163.MCT-11-0648. [DOI] [PubMed] [Google Scholar]
- 4.Wang T, Niu G, Kortylewski M, et al. Regulation of the innate and adaptive immune responses by Stat-3 signaling in tumor cells. Nat Med. 2004;10:48–54. doi: 10.1038/nm976. [DOI] [PubMed] [Google Scholar]
- 5.Huynh J, Chand A, Gough D, Ernst M. Therapeutically exploiting STAT3 activity in cancer — using tissue repair as a road map. Nat Rev Cancer. 2019;19:82–96. doi: 10.1038/s41568-018-0090-8. [DOI] [PubMed] [Google Scholar]
- 6.Joyce JA, Fearon DT. T cell exclusion, immune privilege, and the tumor microenvironment. Science. 2015;348:74–80. doi: 10.1126/science.aaa6204. [DOI] [PubMed] [Google Scholar]
- 7.Spill F, Reynolds DS, Kamm RD, Zaman MH. Impact of the physical microenvironment on tumor progression and metastasis. Curr Opin Biotechnol. 2016;40:41–48. doi: 10.1016/j.copbio.2016.02.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Herrmann A, Kortylewski M, Kujawski M, et al. Targeting Stat3 in the Myeloid Compartment Drastically Improves the in vivo Antitumor Functions of Adoptively Transferred T Cells. Cancer Res. 2010;70:7455–7464. doi: 10.1158/0008-5472.CAN-10-0736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kortylewski M, Yu H. Role of Stat3 in suppressing anti-tumor immunity. Curr Opin Immunol. 2008;20:228–233. doi: 10.1016/j.coi.2008.03.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Frank DA. STAT3 as a central mediator of neoplastic cellular transformation. Cancer Lett. 2007;251:199–210. doi: 10.1016/j.canlet.2006.10.017. [DOI] [PubMed] [Google Scholar]
- 11.Roeser JC, Leach SD, McAllister F. Emerging strategies for cancer immunoprevention. Oncogene. 2015;34:6029. doi: 10.1038/onc.2015.98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sonnenblick A, Shriki A, Galun E, et al. Tissue microarray-based study of patients with lymph node-positive breast cancer shows tyrosine phosphorylation of signal transducer and activator of transcription 3 (tyrosine705-STAT3) is a marker of good prognosis. Clin Transl Oncol. 2012;14:232–236. doi: 10.1007/s12094-012-0789-z. [DOI] [PubMed] [Google Scholar]
- 13.Schaefer LK, Ren Z, Fuller GN, Schaefer TS. Constitutive activation of Stat3α in brain tumors: localization to tumor endothelial cells and activation by the endothelial tyrosine kinase receptor (VEGFR-2) Oncogene. 2002;21:2058–2065. doi: 10.1038/sj.onc.1205263. [DOI] [PubMed] [Google Scholar]
- 14.Geiger JL, Grandis JR, Bauman JE. The STAT3 pathway as a therapeutic target in head and neck cancer: Barriers and innovations. Oral Oncology. 2016;56:84–92. doi: 10.1016/j.oraloncology.2015.11.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Li S, Priceman SJ, Xin H, et al. Icaritin Inhibits JAK/STAT3 Signaling and Growth of Renal Cell Carcinoma. PLoS One. 2013;8:e81657. doi: 10.1371/journal.pone.0081657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.He G, Karin M. NF-κB and STAT3 – key players in liver inflammation and cancer. Cell Res. 2010;21:159. doi: 10.1038/cr.2010.183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Saini U, Naidu S, ElNaggar AC, et al. Elevated STAT3 expression in ovarian cancer ascites promotes invasion and metastasis: a potential therapeutic target. Oncogene. 2016;36:168. doi: 10.1038/onc.2016.197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Bar-Natan M, Nelson EA, Xiang M, Frank DA. STAT signaling in the pathogenesis and treatment of myeloid malignancies. JAK-STAT. 2012;1:55–64. doi: 10.4161/jkst.20006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Fukuda A, Wang Sam C, Morris John P, et al. Stat3 and MMP7 Contribute to Pancreatic Ductal Adenocarcinoma Initiation and Progression. Cancer Cell. 2011;19:441–455. doi: 10.1016/j.ccr.2011.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Redell MS, Ruiz MJ, Alonzo TA, Gerbing RB, Tweardy DJ. Stat3 signaling in acute myeloid leukemia: ligand-dependent and -independent activation and induction of apoptosis by a novel small-molecule Stat3 inhibitor. Blood. 2011;117:5701–5709. doi: 10.1182/blood-2010-04-280123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Alas S, Bonavida B. Inhibition of Constitutive STAT3 Activity Sensitizes Resistant Non-Hodgkin’s Lymphoma and Multiple Myeloma to Chemotherapeutic Drug-mediated Apoptosis. Clin Cancer Res. 2003;9:316–326. [PubMed] [Google Scholar]
- 22.Yu H, Pardoll D, Jove R. STATs in cancer inflammation and immunity: a leading role for STAT3. Nat Rev Cancer. 2009;9:798. doi: 10.1038/nrc2734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bromberg J, Darnell JE. The role of STATs in transcriptional control and their impact on cellular function. Oncogene. 2000;19:2468–2473. doi: 10.1038/sj.onc.1203476. [DOI] [PubMed] [Google Scholar]
- 24.Kim BH, Yi EH, Ye SK. Signal transducer and activator of transcription 3 as a therapeutic target for cancer and the tumor microenvironment. Arch Pharm Res. 2016;39:1085–1099. doi: 10.1007/s12272-016-0795-8. [DOI] [PubMed] [Google Scholar]
- 25.Horiguchi A, Oya M, Marumo K, Murai M. STAT3, but not ERKs, mediates the IL-6–induced proliferation of renal cancer cells, ACHN and 769P. Kidney Int. 2002;61:926–938. doi: 10.1046/j.1523-1755.2002.00206.x. [DOI] [PubMed] [Google Scholar]
- 26.Lin L, Liu A, Peng Z, et al. STAT3 Is Necessary for Proliferation and Survival in Colon Cancer–Initiating Cells. Cancer Res. 2011;71:7226–7237. doi: 10.1158/0008-5472.CAN-10-4660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Corvinus FM, Orth C, Moriggl R, et al. Persistent STAT3 Activation in Colon Cancer Is Associated with Enhanced Cell Proliferation and Tumor Growth. Neoplasia. 2005;7:545–555. doi: 10.1593/neo.04571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Catlett-Falcone R, Landowski TH, Oshiro MM, et al. Constitutive Activation of Stat3 Signaling Confers Resistance to Apoptosis in Human U266 Myeloma Cells. Immunity. 1999;10:105–115. doi: 10.1016/S1074-7613(00)80011-4. [DOI] [PubMed] [Google Scholar]
- 29.Epling-Burnette PK, Liu JH, Catlett-Falcone R, et al. Inhibition of STAT3 signaling leads to apoptosis of leukemic large granular lymphocytes and decreased Mcl-1 expression. J Clin Invest. 2001;107:351–362. doi: 10.1172/JCI9940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Chen H, Yang Z, Ding C, et al. Discovery of O-Alkylamino-Tethered Niclosamide Derivatives as Potent and Orally Bioavailable Anticancer Agents. ACS Med Chem Lett. 2013;4:180–185. doi: 10.1021/ml3003082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Kanai M, Konda Y, Nakajima T, et al. Differentiation-inducing factor-1 (DIF-1) inhibits STAT3 activity involved in gastric cancer cell proliferation via MEK-ERK-dependent pathway. Oncogene. 2003;22:548–554. doi: 10.1038/sj.onc.1206109. [DOI] [PubMed] [Google Scholar]
- 32.Pancotti F, Roncuzzi L, Maggiolini M, Gasperi-Campani A. Caveolin-1 silencing arrests the proliferation of metastatic lung cancer cells through the inhibition of STAT3 signaling. Cell Signal. 2012;24:1390–1397. doi: 10.1016/j.cellsig.2012.02.015. [DOI] [PubMed] [Google Scholar]
- 33.Wei D, Le X, Zheng L, et al. Stat3 activation regulates the expression of vascular endothelial growth factor and human pancreatic cancer angiogenesis and metastasis. Oncogene. 2003;22:319–329. doi: 10.1038/sj.onc.1206122. [DOI] [PubMed] [Google Scholar]
- 34.Kujawski M, Kortylewski M, Lee H, Herrmann A, Kay H, Yu H. Stat3 mediates myeloid cell–dependent tumor angiogenesis in mice. J Clin Invest. 2008;118:3367–3377. doi: 10.1172/JCI35213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Xu Q, Briggs J, Park S, et al. Targeting Stat3 blocks both HIF-1 and VEGF expression induced by multiple oncogenic growth signaling pathways. Oncogene. 2005;24:5552–5560. doi: 10.1038/sj.onc.1208719. [DOI] [PubMed] [Google Scholar]
- 36.Jung JE, Lee HG, Cho IH, et al. STAT3 is a potential modulator of HIF-1-mediated VEGF expression in human renal carcinoma cells. FASEB J. 2005;19:1296–1298. doi: 10.1096/fj.04-3099fje. [DOI] [PubMed] [Google Scholar]
- 37.Wojcik EJ, Sharifpoor S, Miller NA, et al. A novel activating function of c-Src and Stat3 on HGF transcription in mammary carcinoma cells. Oncogene. 2006;25:2773–2784. doi: 10.1038/sj.onc.1209306. [DOI] [PubMed] [Google Scholar]
- 38.Cheng GZ, Zhang W, Sun M, et al. Twist Is Transcriptionally Induced by Activation of STAT3 and Mediates STAT3 Oncogenic Function. J Biol Chem. 2008;283:14665–14673. doi: 10.1074/jbc.M707429200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Banerjee K, Resat H. Constitutive activation of STAT3 in breast cancer cells: A review. Int J Cancer. 2016;138:2570–2578. doi: 10.1002/ijc.29923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Wendt MK, Balanis N, Carlin CR, Schiemann WP. STAT3 and epithelial-mesenchymal transitions in carcinomas. JAK-STAT. 2014;3:e28975–e28975. doi: 10.4161/jkst.28975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Chen M, Liu Y, Varley P, et al. High-Mobility Group Box 1 Promotes Hepatocellular Carcinoma Progression through miR-21–Mediated Matrix Metalloproteinase Activity. Cancer Res. 2015;75:1645–1656. doi: 10.1158/0008-5472.CAN-14-2147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Xiong H, Hong J, Du W, et al. Roles of STAT3 and ZEB1 proteins in E-cadherin down-regulation and human colorectal cancer epithelial-mesenchymal transition. J Biol Chem. 2012;287:5819–5832. doi: 10.1074/jbc.M111.295964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kalluri R. Basement membranes: structure, assembly and role in tumour angiogenesis. Nat Rev Cancer. 2003;3:422–433. doi: 10.1038/nrc1094. [DOI] [PubMed] [Google Scholar]
- 44.Dechow TN, Pedranzini L, Leitch A, et al. Requirement of matrix metalloproteinase-9 for the transformation of human mammary epithelial cells by Stat3-C. Proc Natl Acad Sci U S A. 2004;101:10602–10607. doi: 10.1073/pnas.0404100101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Itoh M, Murata T, Suzuki T, et al. Requirement of STAT3 activation for maximal collagenase-1 (MMP-1) induction by epidermal growth factor and malignant characteristics in T24 bladder cancer cells. Oncogene. 2006;25:1195–1204. doi: 10.1038/sj.onc.1209149. [DOI] [PubMed] [Google Scholar]
- 46.Sano S, Itami S, Takeda K, et al. Keratinocyte-specific ablation of Stat3 exhibits impaired skin remodeling, but does not affect skin morphogenesis. EMBO J. 1999;18:4657–4668. doi: 10.1093/emboj/18.17.4657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Silver DL, Naora H, Liu J, Cheng W, Montell DJ. Activated signal transducer and activator of transcription (STAT) 3: localization in focal adhesions and function in ovarian cancer cell motility. Cancer Res. 2004;64:3550–3558. doi: 10.1158/0008-5472.CAN-03-3959. [DOI] [PubMed] [Google Scholar]
- 48.Herbeuval J-P, Lelievre E, Lambert C, Dy M, Genin C. Recruitment of STAT3 for Production of IL-10 by Colon Carcinoma Cells Induced by Macrophage-Derived IL-6. J Immunol. 2004;172:4630–4636. doi: 10.4049/jimmunol.172.7.4630. [DOI] [PubMed] [Google Scholar]
- 49.Stumhofer JS, Silver JS, Laurence A, et al. Interleukins 27 and 6 induce STAT3-mediated T cell production of interleukin 10. Nat Immunol. 2007;8:1363. doi: 10.1038/ni1537. [DOI] [PubMed] [Google Scholar]
- 50.Saudemont A, Jouy N, Hetuin D, Quesnel B. NK cells that are activated by CXCL10 can kill dormant tumor cells that resist CTL-mediated lysis and can express B7-H1 that stimulates T cells. Blood. 2005;105:2428–2435. doi: 10.1182/blood-2004-09-3458. [DOI] [PubMed] [Google Scholar]
- 51.Thakur R, Trivedi R, Rastogi N, Singh M, Mishra DP. Inhibition of STAT3, FAK and Src mediated signaling reduces cancer stem cell load, tumorigenic potential and metastasis in breast cancer. Sci Rep. 2015;5:10194. doi: 10.1038/srep10194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Blaylock R. Cancer microenvironment, inflammation and cancer stem cells: A hypothesis for a paradigm change and new targets in cancer control. Surg Neurol Int. 2015;6:92. doi: 10.4103/2152-7806.157890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Liu X, He Z, Li C-H, Huang G, Ding C, Liu H. Correlation analysis of JAK-STAT pathway components on prognosis of patients with prostate cancer. Pathol Oncol Res. 2012;18:17–23. doi: 10.1007/s12253-011-9410-y. [DOI] [PubMed] [Google Scholar]
- 54.Wei W, Tweardy DJ, Zhang M, et al. STAT3 Signaling Is Activated Preferentially in Tumor-Initiating Cells in Claudin-Low Models of Human Breast Cancer. Stem Cells. 2014;32:2571–2582. doi: 10.1002/stem.1752. [DOI] [PubMed] [Google Scholar]
- 55.Won C, Kim B-H, Yi EH, et al. Signal transducer and activator of transcription 3-mediated CD133 up-regulation contributes to promotion of hepatocellular carcinoma. Hepatology. 2015;62:1160–1173. doi: 10.1002/hep.27968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Zhang X, Hu F, Li G, et al. Human colorectal cancer-derived mesenchymal stem cells promote colorectal cancer progression through IL-6/JAK2/STAT3 signaling. Cell Death Dis. 2018;9:25. doi: 10.1038/s41419-017-0176-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Haftchenary S, Luchman HA, Jouk AO, et al. Potent Targeting of the STAT3 Protein in Brain Cancer Stem Cells: A Promising Route for Treating Glioblastoma. ACS Med Chem Lett. 2013;4:1102–1107. doi: 10.1021/ml4003138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Qu Y, Oyan AM, Liu R, et al. Generation of Prostate Tumor–Initiating Cells Is Associated with Elevation of Reactive Oxygen Species and IL-6/STAT3 Signaling. Cancer Res. 2013;73:7090–7100. doi: 10.1158/0008-5472.CAN-13-1560. [DOI] [PubMed] [Google Scholar]
- 59.Hossain A, Gumin J, Gao F, et al. Mesenchymal Stem Cells Isolated From Human Gliomas Increase Proliferation and Maintain Stemness of Glioma Stem Cells Through the IL-6/gp130/STAT3 Pathway. Stem Cells. 2015;33:2400–2415. doi: 10.1002/stem.2053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.van der Zee M, Sacchetti A, Cansoy M, et al. IL6/JAK1/STAT3 Signaling Blockade in Endometrial Cancer Affects the ALDHhi/CD126+ Stem-like Component and Reduces Tumor Burden. Cancer Res. 2015;75:3608–3622. doi: 10.1158/0008-5472.CAN-14-2498. [DOI] [PubMed] [Google Scholar]
- 61.Gibbs CP, Kukekov VG, Reith JD, et al. Stem-like cells in bone sarcomas: implications for tumorigenesis. Neoplasia (New York, N.Y.) 2005;7:967–976. doi: 10.1593/neo.05394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Marotta LLC, Almendro V, Marusyk A, et al. The JAK2/STAT3 signaling pathway is required for growth of CD44+CD24– stem cell–like breast cancer cells in human tumors. J Clin Invest. 2011;121:2723–2735. doi: 10.1172/JCI44745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Zhao D, Pan C, Sun J, et al. VEGF drives cancer-initiating stem cells through VEGFR-2/Stat3 signaling to upregulate Myc and Sox2. Oncogene. 2014;34:3107. doi: 10.1038/onc.2014.257. [DOI] [PubMed] [Google Scholar]
- 64.Tseng L-M, Huang P-I, Chen Y-R, et al. Targeting Signal Transducer and Activator of Transcription 3 Pathway by Cucurbitacin I Diminishes Self-Renewing and Radiochemoresistant Abilities in Thyroid Cancer-Derived CD133+ Cells. J Pharmacol Exp Ther. 2012;341:410–423. doi: 10.1124/jpet.111.188730. [DOI] [PubMed] [Google Scholar]
- 65.Groner B, Lucks P, Borghouts C. The function of Stat3 in tumor cells and their microenvironment. Semin Cell Dev Biol. 2008;19:341–350. doi: 10.1016/j.semcdb.2008.06.005. [DOI] [PubMed] [Google Scholar]
- 66.Jung JE, Kim HS, Lee CS, et al. STAT3 inhibits the degradation of HIF-1α by pVHL-mediated ubiquitination. Exp Mol Med. 2008;40:479. doi: 10.3858/emm.2008.40.5.479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Samavati L, Rastogi R, Du W, Hüttemann M, Fite A, Franchi L. STAT3 tyrosine phosphorylation is critical for interleukin 1 beta and interleukin-6 production in response to lipopolysaccharide and live bacteria. Mol Immunol. 2009;46:1867–1877. doi: 10.1016/j.molimm.2009.02.018. [DOI] [PubMed] [Google Scholar]
- 68.Gao H, Priebe W, Glod J, Banerjee D. Activation of Signal Transducers and Activators of Transcription 3 and Focal Adhesion Kinase by Stromal Cell-Derived Factor 1 Is Required for Migration of Human Mesenchymal Stem Cells in Response to Tumor Cell-Conditioned Medium. Stem Cells. 2009;27:857–865. doi: 10.1002/stem.23. [DOI] [PubMed] [Google Scholar]
- 69.Fujiwara Y, Takeya M, Komohara Y. A novel strategy for inducing the antitumor effects of triterpenoid compounds: blocking the protumoral functions of tumor-associated macrophages via STAT3 inhibition. Biomed Res Int. 20142014 doi: 10.1155/2014/348539. 348539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Kim KJ, Kwon SH, Yun JH, et al. STAT3 activation in endothelial cells is important for tumor metastasis via increased cell adhesion molecule expression. Oncogene. 2017;36:5445. doi: 10.1038/onc.2017.148. [DOI] [PubMed] [Google Scholar]
- 71.Kujawski M, Kortylewski M, Lee H, Herrmann A, Kay H, Yu H. Stat3 mediates myeloid cell-dependent tumor angiogenesis in mice. J Clin Invest. 2008;118:3367–3377. doi: 10.1172/JCI35213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Wu L, Du H, Li Y, Qu P, Yan C. Signal transducer and activator of transcription 3 (Stat3C) promotes myeloid-derived suppressor cell expansion and immune suppression during lung tumorigenesis. Am J Pathol. 2011;179:2131–2141. doi: 10.1016/j.ajpath.2011.06.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Sun X, Sui Q, Zhang C, Tian Z, Zhang J. Targeting Blockage of STAT3 in Hepatocellular Carcinoma Cells Augments NK Cell Functions via Reverse Hepatocellular Carcinoma–Induced Immune Suppression. Mol Cancer Ther. 2013;12:2885–2896. doi: 10.1158/1535-7163.MCT-12-1087. [DOI] [PubMed] [Google Scholar]
- 74.Haura EB, Turkson J, Jove R. Mechanisms of Disease: insights into the emerging role of signal transducers and activators of transcription in cancer. Nat Clin Pract Oncol. 2005;2:315–324. doi: 10.1038/ncponc0195. [DOI] [PubMed] [Google Scholar]
- 75.Debnath B, Xu S, Neamati N. Small Molecule Inhibitors of Signal Transducer and Activator of Transcription 3 (Stat3) Protein. J Med Chem. 2012;55:6645–6668. doi: 10.1021/jm300207s. [DOI] [PubMed] [Google Scholar]
- 76.Jinxia D, Fedora G, Nouri N. Small Molecule Inhibitors of Stat3 Signaling Pathway. Curr Cancer Drug Targets. 2007;7:91–107. doi: 10.2174/156800907780006922. [DOI] [PubMed] [Google Scholar]
- 77.Turkson J, Ryan D, Kim JS, et al. Phosphotyrosyl Peptides Block Stat3-mediated DNA Binding Activity, Gene Regulation, and Cell Transformation. J Biol Chem. 2001;276:45443–45455. doi: 10.1074/jbc.M107527200. [DOI] [PubMed] [Google Scholar]
- 78.Siddiquee KAZ, Gunning PT, Glenn M, et al. An Oxazole-Based Small-Molecule Stat3 Inhibitor Modulates Stat3 Stability and Processing and Induces Antitumor Cell Effects. ACS Chem Biol. 2007;2:787–798. doi: 10.1021/cb7001973. [DOI] [PubMed] [Google Scholar]
- 79.Zhang X, Sun Y, Pireddu R, et al. A Novel Inhibitor of STAT3 Homodimerization Selectively Suppresses STAT3 Activity and Malignant Transformation. Cancer Res. 2013;73:1922–1933. doi: 10.1158/0008-5472.CAN-12-3175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Kumar A, Bora U. Molecular docking studies on inhibition of Stat3 dimerization by curcumin natural derivatives and its conjugates with amino acids. Bioinformation. 2012;8:988–993. doi: 10.6026/97320630008988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Shin D-S, Kim H-N, Shin KD, et al. Cryptotanshinone Inhibits Constitutive Signal Transducer and Activator of Transcription 3 Function through Blocking the Dimerization in DU145 Prostate Cancer Cells. Cancer Res. 2009;69:193–202. doi: 10.1158/0008-5472.CAN-08-2575. [DOI] [PubMed] [Google Scholar]
- 82.Song H, Wang R, Wang S, Lin J. A low-molecular-weight compound discovered through virtual database screening inhibits Stat3 function in breast cancer cells. Proc Natl Acad Sci U S A. 2005;102:4700–4705. doi: 10.1073/pnas.0409894102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Schust J, Sperl B, Hollis A, Mayer TU, Berg T. Stattic: A Small-Molecule Inhibitor of STAT3 Activation and Dimerization. Chem Biol. 2006;13:1235–1242. doi: 10.1016/j.chembiol.2006.09.018. [DOI] [PubMed] [Google Scholar]
- 84.Fletcher S, Page BDG, Zhang X, et al. Antagonism of the Stat3–Stat3 Protein Dimer with Salicylic Acid Based Small Molecules. Chem Med Chem. 2011;6:1459–1470. doi: 10.1002/cmdc.201100194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Lin Y-M, Wang C-M, Jeng J-C, Leprince D, Shih H-M. HIC1 interacts with and modulates the activity of STAT3. Cell Cycle. 2013;12:2266–2276. doi: 10.4161/cc.25365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Turkson J, Zhang S, Palmer J, et al. Inhibition of constitutive signal transducer and activator of transcription 3 activation by novel platinum complexes with potent antitumor activity. Mol Cancer Ther. 2004;3:1533–1542. [PubMed] [Google Scholar]
- 87.Nagel-Wolfrum K, Buerger C, Wittig I, Butz K, Hoppe-Seyler F, Groner B. The Interaction of Specific Peptide Aptamers With the DNA Binding Domain and the Dimerization Domain of the Transcription Factor Stat3 Inhibits Transactivation and Induces Apoptosis in Tumor Cells. Mol Cancer Res. 2004;2:170–182. [PubMed] [Google Scholar]
- 88.Buerger C, Nagel-Wolfrum K, Kunz C, et al. Sequence-specific Peptide Aptamers, Interacting with the Intracellular Domain of the Epidermal Growth Factor Receptor, Interfere with Stat3 Activation and Inhibit the Growth of Tumor Cells. J Biol Chem. 2003;278:37610–37621. doi: 10.1074/jbc.M301629200. [DOI] [PubMed] [Google Scholar]
- 89.Ge H, Liu H, Fu Z, Sun Z. Therapeutic and Preventive Effects of an Epidermal Growth Factor Receptor Inhibitor on Oral Squamous Cell Carcinoma. J Int Med Res. 2012;40:455–466. doi: 10.1177/147323001204000207. [DOI] [PubMed] [Google Scholar]
- 90.Huang C, Cao J, Huang KJ, et al. Inhibition of STAT3 activity with AG490 decreases the invasion of human pancreatic cancer cells in vitro. Cancer Sci. 2006;97:1417–1423. doi: 10.1111/j.1349-7006.2006.00340.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Ferrajoli A, Faderl S, Van Q, et al. WP1066 Disrupts Janus Kinase-2 and Induces Caspase-Dependent Apoptosis in Acute Myelogenous Leukemia Cells. Cancer Res. 2007;67:11291–11299. doi: 10.1158/0008-5472.CAN-07-0593. [DOI] [PubMed] [Google Scholar]
- 92.Pardanani A, Hood J, Lasho T, et al. TG101209, a small molecule JAK2-selective kinase inhibitor potently inhibits myeloproliferative disorder-associated JAK2V617F and MPLW515L/K mutations. Leukemia. 2007;21:1658. doi: 10.1038/sj.leu.2404750. [DOI] [PubMed] [Google Scholar]
- 93.Scuto A, Krejci P, Popplewell L, et al. The novel JAK inhibitor AZD1480 blocks STAT3 and FGFR3 signaling, resulting in suppression of human myeloma cell growth and survival. Leukemia. 2010;25:538. doi: 10.1038/leu.2010.289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Yan S, Li Z, Thiele CJ. Inhibition of STAT3 with orally active JAK inhibitor, AZD1480, decreases tumor growth in Neuroblastoma and Pediatric Sarcomas In vitro and In vivo. Oncotarget. 2013;4:433–445. doi: 10.18632/oncotarget.930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Chen Z, Lee FY, Bhalla KN, Wu J. Potent Inhibition of Platelet-Derived Growth Factor-Induced Responses in Vascular Smooth Muscle Cells by BMS-354825 (Dasatinib) Mol Pharmacol. 2006;69:1527–1533. doi: 10.1124/mol.105.020172. [DOI] [PubMed] [Google Scholar]
- 96.Michels S, Trautmann M, Sievers E, et al. SRC Signaling Is Crucial in the Growth of Synovial Sarcoma Cells. Cancer Res. 2013;73:2518–2528. doi: 10.1158/0008-5472.CAN-12-3023. [DOI] [PubMed] [Google Scholar]
- 97.Chang AY, Wang M. Molecular mechanisms of action and potential biomarkers of growth inhibition of dasatinib (BMS-354825) on hepatocellular carcinoma cells. BMC Cancer. 2013;13:267. doi: 10.1186/1471-2407-13-267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Premkumar D, Jane E, Agostino N, Scialabba J, Pollack I. Dasatinib synergizes with JSI-124 to inhibit growth and migration and induce apoptosis of malignant human glioma cells. J Carcinog. 2010;9:7. doi: 10.4103/1477-3163.65448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Oyaizu T, Fung SY, Shiozaki A, et al. Src tyrosine kinase inhibition prevents pulmonary ischemia-reperfusion-induced acute lung injury. Intensive Care Med. 2012;38:894–905. doi: 10.1007/s00134-012-2498-z. [DOI] [PubMed] [Google Scholar]
- 100.Antonarakis ES, Heath EI, Posadas EM, et al. A phase 2 study of KX2-391 an oral inhibitor of Src kinase and tubulin polymerization, in men with bone-metastatic castration-resistant prostate cancer. Cancer Chemotherapy and Pharmacology. 2013;71:883–892. doi: 10.1007/s00280-013-2079-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Gangadhar TC, Clark JI, Karrison T, Gajewski TF. Phase II study of the Src kinase inhibitor saracatinib (AZD0530) in metastatic melanoma. Invest New Drugs. 2013;31:769–773. doi: 10.1007/s10637-012-9897-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Seltana A, Guezguez A, Lepage M, Basora N, Beaulieu JF. Src family kinase inhibitor PP2 accelerates differentiation in human intestinal epithelial cells. Biochem Biophys Res Commun. 2013;430:1195–1200. doi: 10.1016/j.bbrc.2012.12.085. [DOI] [PubMed] [Google Scholar]
- 103.Kim BH, Won C, Lee YH, et al. Sophoraflavanone G induces apoptosis of human cancer cells by targeting upstream signals of STATs. Biochem Pharmacol. 2013;86:950–959. doi: 10.1016/j.bcp.2013.08.009. [DOI] [PubMed] [Google Scholar]
- 104.Aittomäki S, Pesu M. Therapeutic Targeting of the JAK/STAT Pathway. Basic Clin Pharmacol Toxicol. 2014;114:18–23. doi: 10.1111/bcpt.12164. [DOI] [PubMed] [Google Scholar]
- 105.Buchert M, Burns CJ, Ernst M. Targeting JAK kinase in solid tumors: emerging opportunities and challenges. Oncogene. 2015;35:939. doi: 10.1038/onc.2015.150. [DOI] [PubMed] [Google Scholar]
- 106.Plimack ER, Lorusso PM, McCoon P, et al. AZD1480: a phase I study of a novel JAK2 inhibitor in solid tumors. Oncologist. 2013;18:819–820. doi: 10.1634/theoncologist.2013-0198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Furtek SL, Backos DS, Matheson CJ, Reigan P. Strategies and Approaches of Targeting STAT3 for Cancer Treatment. ACS Chem Biol. 2016;11:308–318. doi: 10.1021/acschembio.5b00945. [DOI] [PubMed] [Google Scholar]
- 108.Puls LN, Eadens M, Messersmith W. Current status of SRC inhibitors in solid tumor malignancies. Oncologist. 2011;16:566–578. doi: 10.1634/theoncologist.2010-0408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Nam S, Wen W, Schroeder A, et al. Dual inhibition of Janus and Src family kinases by novel indirubin derivative blocks constitutively-activated Stat3 signaling associated with apoptosis of human pancreatic cancer cells. Mol Oncol. 2013;7:369–378. doi: 10.1016/j.molonc.2012.10.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Liu L, Gaboriaud N, Vougogianopoulou K, et al. MLS-2384, a new 6-bromoindirubin derivative with dual JAK/Src kinase inhibitory activity, suppresses growth of diverse cancer cells. Cancer Biol Ther. 2014;15:178–184. doi: 10.4161/cbt.26721. [DOI] [PMC free article] [PubMed] [Google Scholar]