Abstract
Background
Pain is recognised to have both a sensory dimension (intensity) and an affective dimension (unpleasantness). Pain feels like a single unpleasant bodily experience, but investigations of human pain have long considered these two dimensions of pain to be separable and differentially modifiable. The evidence underpinning this separability and differential modifiability is seldom presented. We aimed to fill this gap by evaluating the current evidence base for whether or not the sensory and affective dimensions of pain can be selectively modulated using cognitive manipulations.
Methods
A rigorous systematic search, based on a priori search terms and consultation with field experts, yielded 4270 articles. A detailed screening process was based on the following recommendations: (i) evaluation of effectiveness; (ii) examination of methodological rigour, including each study having an a priori intention to cognitively modulate one of the two dimensions of pain; and (iii) sound theoretical reasoning. These were used to ensure that included studies definitively answered the research question.
Results
After in-depth critique of all 12 articles that met the inclusion criteria, we found that there is no compelling evidence that the sensory and affective dimensions of pain can be selectively and intentionally modulated using cognitive manipulations in humans.
Conclusions
We offer potential explanations for this discrepancy between assumptions and evidence and contend that this finding highlights several important questions for the field, from both the research and clinical perspectives.
Keywords: cognition, hypnosis, imagery, pain measurement, pain perception
Pain commonly drives the sufferer towards safety; it can be considered a motivator of protective behaviour rather than a marker of tissue damage.1, 2 The clear motivational drive of pain makes it more closely resemble feelings such as hunger or thirst than sensory perceptions such as vision or hearing.3 That pain is necessarily perceived—one cannot have pain and not know about it—and its inherent unpleasantness (or indeed its juxtaposition to pleasure) have occupied human thought for millennia: Plato wrote ‘ … All pleasure and pain come to be in the soul, because all pleasure and pain is necessarily perceived … ’.4 This immutable emotional nature of pain was recognised more formally, as one dimension of a wider pain experience, towards the end of the past century, with the proposal that pain is a bidimensional experience consisting of a sensory-discriminative dimension and an affective-motivational dimension.5 This is further highlighted through the International Association for the Study of Pain definition of pain as ‘[a]n unpleasant sensory and emotional experience associated with actual or potential tissue damage, or described in terms of such damage’.6
The sensory-discriminative dimension, often referred to simply as ‘intensity’ or given the label ‘sensory’, includes the spatial, and temporal characteristics and quality of pain.7 The affective-motivational dimension, often referred to simply as ‘unpleasantness’ or given the label ‘affective’, captures how ‘bad’ or how ‘unpleasant’ the pain is.7 That is, it captures the motivational aspect of pain—the aspect that makes us want to take protective action.
That pain is felt at a bodily location and therefore has sensory qualities, while also being definitively unpleasant, led to the development of the most widespread and tested pain assessment tool—the McGill Pain Questionnaire.8 There is a large body of literature concerning the properties of the McGill Pain Questionnaire and recognisable patterns of responses that can differentiate between, for example, neuropathic and non-neuropathic pain.9
This potentially separable bidimensionality of pain is apparent in some clinical studies. For example, people who attribute their pain to a greater threat to their health will rate the pain unpleasantness higher than those who attribute their pain to a lesser threat to their health, despite having similar self-reported pain intensities.10, 11
Cognitive manipulations during brain imaging have been used to tease apart the cortical activation patterns associated with the sensory and affective dimensions of pain.12, 13 Study reports implicate the anterior cingulate cortex (ACC) in subserving pain unpleasantness in particular.14 For example, a cognitive manipulation to increase the affective dimension of pain (i.e. to make it more unpleasant but not more ‘intense’) caused an increase in ACC activation; a cognitive manipulation to decrease the affective dimension (i.e. make it less unpleasant but no less intense) caused a decrease in ACC activation.14 Similar direction-specific modulation of the primary somatosensory cortex (S1) was reported in conjunction with manipulation of the sensory dimension of pain.14
Functional neuroanatomical data have also been used to substantiate the claim that these two dimensions of pain involve distinct neural pathways from the dorsal horn of the spinal cord to cortical centres (see Treede and colleagues15 for further details).16 The lateral spinothalamic pathway has been proposed to subserve the sensory dimension of pain, because of the type of synaptic input and main cerebral projection.16 Strengthening this proposition is the finding that nociceptive neurones within the spinothalamic pathway are predominantly wide dynamic range (WDR) neurones, which by definition are responsive to a wide range of stimuli, from light touch to intense stimulation.17
The spino-parabrachio-amygdaloid and spino-parabrachio-hypothalamic have been proposed to subserve the affective dimension of pain,17, 18 largely because they comprise mainly nociceptive neurones and their central projections terminate in the amygdala and hypothalamus, which are thought to play a role in fear, emotional memory and behaviour, and autonomic and somatomotor responses to threatening stimuli.19
Many neurophysiologists, neuroanatomists, and pain researchers seem accepting of these ideas, but other specialists have argued that pain is in fact an unidimensional construct and that separating it into dimensions is as fallacious as attributing it to single neurones.20 The latter group argue that the apparently successful attempts to independently modulate the two dimensions of pain via cognitive strategies—for example, in the imaging studies mentioned earlier—actually reflect demand effects (or the effects of ‘coaching’ participants), not selective endogenous modulation of pain's two dimensions.21 Indeed, whether people can even differentiate the two dimensions remains contentious. Several studies have shown that participants tend to rate the sensory and affective dimensions of pain similarly, unless they receive clear instructions that explicitly highlight differences between them.22, 23, 24
When such a fundamental matter is soundly endorsed by some parts of the community (predominantly scientists) and soundly rejected by others (predominantly clinicians, philosophers, and laypeople), one might expect the evidence for either position to be clearly presented.13, 14, 25, 26 However, there seems to have been very little attempt to evaluate the actual evidence for either view. This is surprising because experimental studies continue to differentiate them, whereas clinical practice continues to consider pain a single construct—patients are almost always asked ‘How is your pain?’. Within the context of the lived experience of pain and the position it takes within social interactions, we might ask ‘How bad is it?’, or ‘Where is it?’, but such questions consider pain as a unitary unpleasant sensory experience that affects that person.27 By failing to evaluate the actual evidence for whether the separation and independent modulation of the sensory and affective dimensions of pain is possible, studies, clinical interactions, and treatment development based on either viewpoint, remain questionable.
We systematically evaluated the evidence that the two dimensions of pain can be selectively modulated in humans using a cognitive manipulation. Our approach was informed by the seminal review in this field, which also remains the most recent.28 That work concluded that, in order to address the contention that threatened to polarise the field, there needed to be (i) a comprehensive evaluation of the effectiveness of attempts to modulate the two postulated dimensions of pain; (ii) a structured framework and standardised appraisal to examine the methodological rigour of each study, including the potential threats to validity of uncontrolled biases, with each included study having an a priori intention to use a cognitive strategy to selectively modulate one of the two dimensions of pain; and (iii) that all such investigations are planned according to sound theoretical reasoning.
Methods
For the review, the following PICO (Population, Intervention, Comparison, and Outcome) question was devised (see Table 1).
Table 1.
PICO question.
| Population | Intervention | Comparison | Outcome |
|---|---|---|---|
| People with chronic pain and healthy volunteers | Cognitive manipulations | None | The extent to which the affective and sensory dimensions of pain can be selectively and intentionally modulated |
This systematic review was carried out following the guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement and an a priori protocol based on the recommendations of Fernandez and Turk28 (see Supplementary File 1).29
Information sources
The following databases were searched from their inception to February 2017: Cochrane Central Register of Controlled Trials (CENTRAL) in the Cochrane Library; MEDLINE (OvidSP); SciVerse SCOPUS; PsycINFO (OvidSP); and Embase classic and Embase (OvidSP). A sensitive search strategy was devised, using a combination of keywords, relevant subject headings and MeSH terms. The aim was to identify studies that investigated the selective cognitive modulation of the sensory and affective dimensions of pain. The initial search strategy was developed in Medline (OvidSP), and then adapted for other databases (see Supplementary File 2).
Search
Study selection
The titles and abstracts of all records that were identified by the initial search were independently screened and evaluated, using a predetermined list of inclusion criteria, by two reviewers (KAT and VJM). Studies were included if they met all of the following criteria: tested human participants; applied a cognitive technique with the aim of selectively modulating either the sensory or the affective dimension of pain; followed an experimental study design that had at least one of the following: measured a baseline, had a control group, had a pre–post design; assessed both the sensory and affective dimensions. Studies were excluded if they used somatosensory (e.g. transcutaneous electrical nerve stimulation) or exogenous pharmacological (e.g. morphine) interventions to modulate pain. Studies could include participants with acute pain, chronic pain, or no pain, in which case the study would involve experimentally induced pain. Where more than one participant group was included, the groups were evaluated separately. The search was not limited by language, but the title and abstract had to be in English to allow for initial assessment of eligibility. Full texts were retrieved for all eligible records, and independently appraised by two reviewers (KAT and VJM). Non-English full-text articles that were identified as potentially eligible were translated by two independent translators before assessment.
Risk of bias across studies
We developed, tested, and refined a customised risk of bias tool (see Supplementary File 3). This tool was then applied by two independent reviewers (KAT and VJM) to all included studies. For each category of risk, the reviewers highlighted particular factors that could be sources of bias. Minimum criteria were defined for a low risk of bias rating in certain categories. Studies were expected to have confirmed the efficacy of the cognitive manipulation by reporting the results of the manipulation check and reporting the efficacy of participant blinding. Outcome measures were expected to be clearly assessing pain rather than the stimulus. Purposive or convenience sampling were considered to introduce high risk of bias (a deliberately conservative decision although we recognise that, for pragmatic reasons, most research in this field uses convenience sampling).
Data collection process
Data extraction was performed by two independent reviewers (KAT and VJM) using a customised and pre-piloted form (see Supplementary File 4). Extracted data were: participant characteristics such as age and gender, study methods, trial characteristics, and details of the outcome measures.
Outcome measures
The primary outcomes were participant ratings of the sensory and affective dimensions of the experiences that were evoked by painful stimuli. The secondary outcomes were differences in cortical activation, physiological responses, and psychological variables associated with the manipulations. We summarised data by subtracting the change in the ‘non-target’ dimension from the change in the ‘target’ dimension, producing a mean difference (and standard error of the mean difference) as an estimate of the manipulation's effectiveness. Mean and standard error (se) data were then entered in to Review Manager (version 5.2, The Nordic Cochrane Centre, Copenhagen, Denmark) using the generic inverse function and pooled using the random-effects model. For full details, see Supplementary File 5.
Results
Study selection
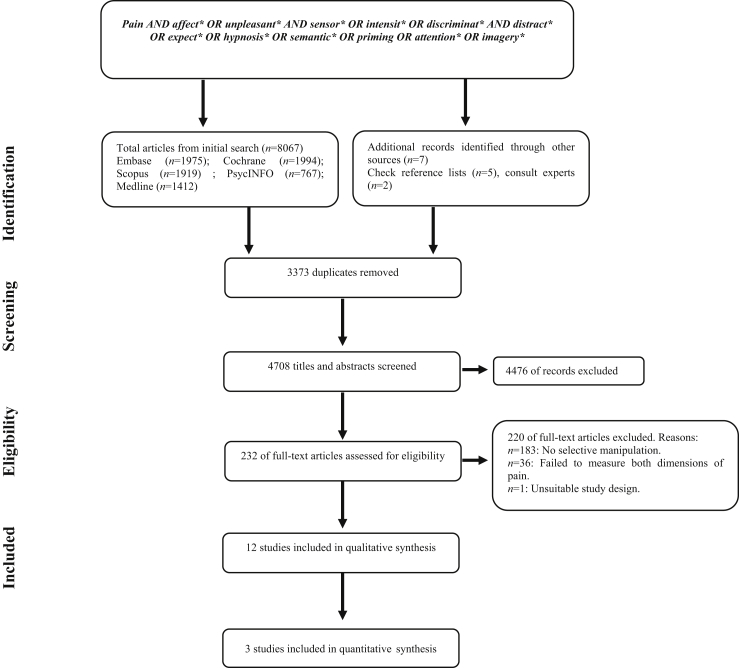
The initial search and removal of duplicates yielded 4708 records. After the screening phase, 232 full-text articles were assessed for eligibility. Two records were reviewed with the help of translators. After assessment of eligibility, 12 articles were included in this systematic review. The details of identification, screening, and appraisal are depicted in Figure 1.
Fig 1.
PRISMA flowchart of systematic search strategy. PRISMA, Preferred Reporting Items for Systematic Reviews and Meta-Analyses.
Study characteristics
Five studies used hypnotic induction with suggestion (n=5),30, 31, 32, 33, 34 one used suggestion alone (without hypnotic induction; n=1),35 four used valence modulation secondary to odour (n=1)13, 14, 25, 26 or visual images (n=3),37, 38, 39 and two used meditation (n=2)40, 41 (Table 2). Although some manipulations were similar, comparability was limited because even studies that used the same class of manipulation used different methods. For example, although Dahlgren and colleagues33 and Rainville and colleagues30 used hypnotic suggestion, Dahlgren and colleagues33 targeted intensity and unpleasantness using different suggestions, whereas Rainville and colleagues30 targeted unpleasantness only, using different suggestions to elicit either an increase or a decrease in unpleasantness.
Table 2.
Key characteristic summary of articles. *Unclear in manuscript/unable to obtain further information. INT, pain intensity; MES, Magnitude Estimation Scale; N/R, not reported; NRS: Numeric Rating Scale; PL, pleasant; UNP, pain unpleasantness; VRS, Verbal Rating Scale.
| First author | Year | Sample size, n (F:M) | Mean age, yr (age range) | Cognitive manipulation | Scale type | Sensory pain anchors (pain intensity) | Affective pain anchors (pain unpleasantness) |
|---|---|---|---|---|---|---|---|
| Hypnotic induction with suggestion | |||||||
| Dahlgren33 | 1995 | 32 (17:15) | N/R* | Hypnotic Suggestion for relaxation or analgesia | VRS | Sensory list of words | Affective list of words |
| Rainville30 | 1997 | 8 (3:5) | 19–53 yr | Hypnotic Suggestion to ↓ and ↑ pain UNP | NRS | 0: No burning, pricking, stinging sensation | 0: Not at all UNP |
| 100: Extremely intense sensation | 100: Extremely UNP | ||||||
| Rainville31 | 1999 | Exp 1:17 (5:12) | Exp 1: 17–28 Controls: 19–21 |
Exp 1: Hypnotic suggestion to ↓ pain UNP | VAS | Burning, pricking, stinging sensation | N/R* |
| Exp 2: 20 (7:13) | Exp 2: 18–37 | Exp 2: Hypnotic suggestion to ↓ ↑ pain UNP | |||||
| Exp 3: 22 (10:12) | Exp 3: 20–29 | Exp 3: Hypnotic suggestion to ↓ and ↑ pain INT | |||||
| Hofbauer32 | 2001 | 10 (6:4) | 20–35 | Hypnotic suggestion to ↓ and ↑ pain INT | MES | N/R* | N/R* |
| Valentini34 | 2013 | 24 (24:0) | 20–25 | Hypnotic suggestion to alter pain INT or UNP | VAS | 0: No pricking/burning/itching | 0: Not at all UNP |
| 101: Intolerable intensity | 101: Intolerable UNP | ||||||
| Meditation | |||||||
| Perlman40 | 2010 | 19 (9:10) | Experimental: 37–59 | Meditation | NRS | N/R* | N/R* |
| Controls: 35–59 | |||||||
| Grant41 | 2009 | 28 (10:18) | Experimental: 23–47 | Meditation | VAS | 0: Not painful | 0: Not UNP |
| Controls: 24–45 | 10: Extremely painful | 10: Extremely UNP | |||||
| Valence induction with exteroceptive stimuli | |||||||
| Kenntner-Mabiala37 | 2007 | 54 (27:27) | 18–40 | Picture valence and attention | VAS | 0: No pressure | –100: Very UNP |
| 100: Just noticeable p | 100: Very PL | ||||||
| 200: Unbearable p | |||||||
| Kenntner-Mabiala38 | 2008 | 30 (15:15) | 21–36 | Picture valence and attention | VAS* | 0: No sensation | 0: Neutral |
| 4: Just noticeable pain | 10: Very UNP | ||||||
| 10: Unbearable pain | |||||||
| Loggia39 | 2008 | 12 (12:0) | 47–55 | Video valence | NRS | 0: No heat | N/R* |
| 200: Most intense pain tolerable | |||||||
| Villemure36 | 2003 | 14 (9:5) | 20–26 | Odour and attention | NRS | 0: No pain | 0: Neutral |
| 10: Most intense pain tolerable | 10: Extremely UNP | ||||||
| Suggestion alone | |||||||
| Kunz35 | 2012 | 22 (10:12) | 18–27 | Suggestion to ↑ pain UNP or INT | VAS | 0: No pain | 0: No pain |
| 100: Extremely strong pain | 100: Extremely UNP pain | ||||||
All the included studies used healthy, pain-free, adult participants. That is, there were no studies that satisfied our a priori criteria and included people with acute or chronic clinical pain. All included studies had biased eligibility criteria. All five hypnosis studies selectively recruited participants with certain susceptibility profiles: four recruited participants who had moderate to high scores of susceptibility30, 31, 32, 33; one recruited two groups—one group of people who had high scores of susceptibility and another group of people who had low scores of susceptibility.34 Both meditation studies recruited a group of expert meditators who had spent a set number of hours in meditative practice, and a group of novices.40, 41 Several studies included only participants who had demonstrated successful attentional or emotional modulation of pain during recruitment.32, 35, 36 Overall, there were more female participants (n=164) than male participants (n=148). Two studies recruited only female participants.34, 39
Risk of bias within studies
All studies were at high risk of selection, performance, or detection bias, or failed to provide sufficient data for us to assess risk of bias (Table 3). The most common reasons for studies to be scored as ‘high risk’ in the selection bias section were selective recruitment or convenience sampling. Full data for risk of bias are presented in Table 3 and in Supplementary File 5.
Table 3.
Risk of bias assessment of included studies. ×, high bias; ✓, low bias; ?, unclear (unclear in manuscript/unable to obtain further information).
| First author (year) | Selection bias | Performance and detection bias | Cognitive technique | Measurement bias | Statistical methods | Reporting |
|---|---|---|---|---|---|---|
| Hypnotic induction with suggestion | ||||||
| Dahlgren (1995)33 | × | ? | × | × | ||
| Rainville (1997)30 | × | × | × | ? | ? | × |
| Rainville (1999)31 | × | ? | × | ✓ | × | ✓ |
| Hofbauer (2001)32 | × | × | × | ✓ | × | ✓ |
| Valentini (2013)34 | × | × | × | ✓ | ✓ | ✓ |
| Meditation | ||||||
| Grant (2009)41 | × | × | × | ✓ | ✓ | ✓ |
| Perlman (2010)40 | × | ? | × | × | × | ✓ |
| Valence induction with exteroceptive stimuli | ||||||
| Kenntner-Mabiala (2007)37 | × | ? | ✓ | ✓ | × | ✓ |
| Kenntner-Mabiala (2008)38 | ? | ? | ✓ | ✓ | × | ✓ |
| Loggia (2008)39 | × | ? | ✓ | ✓ | × | ✓ |
| Villemure (2003)36 | × | ? | ✓ | ✓ | ✓ | ✓ |
| Suggestion alone | ||||||
| Kunz (2012)35 | × | × | ✓ | ✓ | × | ✓ |
Results of individual studies
Hypnosis induction with suggestion
Five studies used hypnotic induction with suggestion. Four of the five studies found that hypnotic suggestion selectively increased or decreased affective dimension ratings in accordance with the type of suggestions given.30, 31, 33, 34 However, they were unable to demonstrate the same effect with the sensory dimension ratings. The three studies that also aimed to selectively modulate the sensory dimension of pain did not achieve selective modulation.31, 32, 34 Interestingly, the reported changes in cortical activation patterns did not match participant pain reports: when hypnotic suggestion targeted the sensory dimension, there was no evidence of selective change in ratings of the sensory or affective dimensions of pain, but there was evidence of selective activation of cortical areas.31, 32, 34 Suggestions that intended to modulate the sensory dimension of pain selectively altered cortical activation in the somatosensory areas (S1 and S2), whereas suggestions that intended to modulate the affective dimension of pain selectively altered ACC activation. The fifth hypnosis study found that hypnosis with suggestion of analgesia selectively decreased ratings of the sensory dimension, whereas hypnosis with suggestion of relaxation selectively decreased the affective dimension of pain.33 Another factor that influenced the effect of the selective modulation was the hypnotic susceptibility of the participants. Three of the studies reported that participants with high susceptibility to hypnosis demonstrated greater selective modulation of the two dimensions than participants with low susceptibility.31, 32, 34
Suggestion alone (without hypnotic induction)
The only study that used non-hypnotic suggestion found that both dimensions of pain could be selectively modulated, depending on the type of suggestion that was given.35 That is, suggestions to decrease the sensory dimension of pain resulted in lower pain intensity but not pain unpleasantness ratings, and vice versa.
Valence modulation using exteroceptive stimuli
Four studies used exteroceptive stimuli to modulate valence, and tested the effect of the modulation of valence on separate dimensions.36, 37, 38, 39 Three of the four studies reported selective modulation of affective dimension ratings,36, 38, 39 and one reported similar shifts in both sensory and affective dimension ratings37; three manipulated participants' attention37, 38, 39; two used a three-way manipulation in which participants were tasked with directing their attention ‘towards the pictures’, ‘towards the sensory experience’, or ‘towards the affective experience’37, 38; one used a two-way manipulation in which participants attended to either heat or an odour.36 Interestingly, in the two studies by Kenntner-Mabiala and colleagues,37, 38 only sensory ratings changed, but the first study involved attention to the sensory experience and the second study involve attention to the affective experience.
Meditation
Two studies compared the modulatory effects of two meditation techniques; one focused on attention,41 and the other on acceptance, sometimes referred to as ‘open monitoring’.40 When performed by experienced meditators, acceptance-based meditation selectively modulated unpleasantness in one study,40 but selectively modulated intensity in the other,41 which is interesting considering that the hypothesised effects focused on unpleasantness. The findings for the focused attention approach were similarly muddled. Focused attention had no effect in one study,40 and increased pain intensity in the other, but only in novice meditators.41 The two studies used focused attention in different ways: Perlman and colleagues40 used attention to a fixation cross, whereas Grant and Rainville41 used attention to the stimulus site.
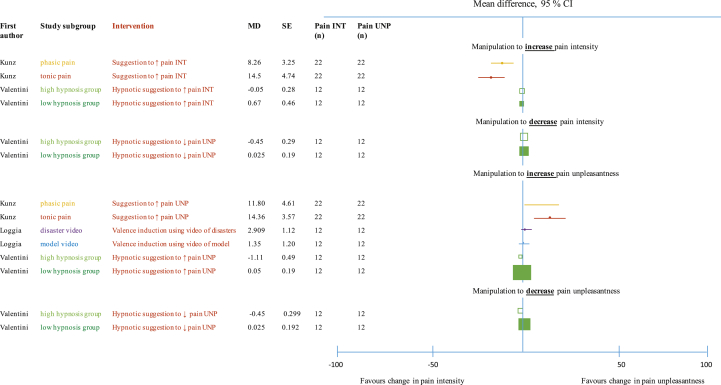
Full data were obtained for three studies.34, 35, 39 They used different manipulations, which prevented pooling. Their data are depicted in forest plots (see Fig. 2).
Fig 2.
The effectiveness of selective modulation of the sensory and affective dimensions of pain.
Discussion
Despite apparent widespread support of the idea that the affective and sensory dimensions of pain can be selectively and intentionally modulated by different cognitive manipulations, our results reveal an evidence base that offers some support to both viewpoints, and is therefore far from clear-cut. Several studies showed selective downregulation of the affective dimension, but studies most compromised by potential bias showed the largest effects. Nine of 12 studies30, 31, 32, 34, 36, 38, 39, 40, 41 could not or did not selectively modulate the sensory dimension and the two studies that reported selective modulation of both dimensions, both using suggestion with or without hypnosis, had a very high risk of bias.33, 35 We conclude that the current evidence suggests that the sensory dimension of pain cannot be selectively modulated using cognitive manipulation, but the affective dimension might be.
Which cognitive manipulation appears most likely to selectively modulate the affective dimension of pain? Of the four cognitive manipulations identified, the five studies that used hypnotic induction with suggestion demonstrated the most consistent effects.30, 31, 32, 33, 34 Most of the studies involved the suggestion to decrease or increase pain unpleasantness. The scripts they used, however, did not necessarily mention unpleasantness. That is, scripts used words such as ‘agreeable’ or ‘comfortable’ or ‘restful’ when aiming to decrease unpleasantness, and words such as ‘uncomfortable’ when aiming to increase unpleasantness, but did not directly refer to pleasantness or unpleasantness specifically.31 Similarly, one study used a suggestion of relaxation intended to decrease pain unpleasantness that was adapted from the Stanford Hypnotic Susceptibility Scale form C (SHSS).30, 42
That three studies demonstrated that hypnosis without suggestion was ineffective, strongly implies that it was the suggestion itself that led to selective modulation of the affective dimension.30, 31, 32 This might appear concerning because it raises the possibility that the effect is simply one of reporting bias. However, two aspects of the literature suggest otherwise. First, the same suggestions were ineffective for the sensory dimension—presumably reporter bias would be equally applicable regardless of dimension. Second, the only study that also investigated neurophysiological markers demonstrated a biologically plausible effect on cortical activation.30 During the two control conditions called ‘wake control’ and ‘hypnotic control’, Rainville and colleagues30 demonstrated that no modulatory effect was observed in the S1, S2, the insular cortex, and the ACC. It was only hypnotic induction with suggestion that altered cortical activation. Therefore, it was hypnotic induction with specific suggestions about the affective dimension that selectively modulated the pain unpleasantness ratings and caused an alteration in the ACC activation. Similarly, it was hypnotic induction with specific suggestions about the sensory dimension that caused an alteration in the S1 activation; however, this did not translate to a selective modulation of pain intensity ratings. Although it clarifies that the effect is not merely a reflection of a participant's being under hypnosis, it is also potentially concerning because it suggests a differential effect of the manipulation on brain activity, but not on brain activity associated with production of pain.
If we are to accept that suggestion can selectively modulate the affective dimension of pain, there are important caveats concerning generalisation and clinical utility. All five hypnosis studies recruited highly hypnotisable participants, but only 10–20% of the population is highly susceptible to hypnosis.30, 31, 32, 33, 34 That the one study that included participants with low susceptibility to hypnosis had clearly lower modulatory effects leaves question marks over the potential utility of using suggestion to change pain unpleasantness.34 Four studies demonstrated that selectively modulating the affective dimension of pain by using exteroceptive stimuli to change the valence of a test stimulus, were small effects—4.4 units or 3 units on a 100-unit scale.36, 37, 38, 39 Clearly, regardless of the mechanism of induction, the magnitude of effect is well short of that which might be considered clinically meaningful and, indeed, that which is measurable at an individual level.43
Why might the affective dimension be more modifiable than the sensory dimension? Relevant here might be data that show that most people conceptualise pain as a marker of tissue damage, which it has now been clearly shown not to be (see Moseley and Butler44 for a comprehensive review of experimental and clinical studies).45, 46 We suspect that the participants in these studies would conceptualise their pain as ‘coming from the tissues’ rather than resulting from a highly complex evaluative process and a ‘best-guess output’ of the brain that reflects the predicted merit of taking protective action.47 If our suspicion is correct, it is not unreasonable to suggest that participants interpret the sensory dimension of pain as pain itself, which would render any cognitive attempt to reduce it seemingly pointless.48, 49 Consistent with this idea is the clinical approach of mindfulness, in which the sufferer learns skills of acknowledging the pain by curbing a behavioural response to it, implying the immutable characteristics of pain's sensory but not affective dimension.50 A review of the meditation literature proposed mechanisms by which mindfulness or meditation may alter pain, focusing on its affective dimension, but compelling empirical data remain lacking.51
It is difficult to exclude demand effects as contributing mechanisms to the successful manipulations.52 Four of the five studies used a hypnotic suggestion that asked participants to either increase or decrease ‘pain intensity’, but none were successful. The fifth hypnosis study used a hypnotic suggestion of ‘analgesia’ to decrease pain intensity, and successfully achieved a selective modulation of sensory intensity ratings.33 It is plausible that the idea of analgesia was critical to this effect. The term ‘analgesia’ is usually used to describe an effect of medication, an agent that participants may understand to be capable of modulating sensory signalling.53 As such, the term ‘analgesia’ might possess conceptual validity that ‘reducing pain intensity’ does not.54 Certainly, expectation of pain relief modulates pain relief. For example, positive expectations about treatment seem to enhance the analgesic effect of opioid derived remifentanil, but negative expectations about treatment completely abolished the analgesic effect of remifentanil.53 Therefore, assessing conceptual understanding, expectation and self-efficacy in future studies of this topic would help with clarity.
Despite our comprehensive and rigorous systematic review, the idea that pain comprises two separable dimensions is likely to remain somewhat controversial.24 Alternative views advocate for an understanding of pain as a unitary experience that involves different sensations, emotions, and cognitive processes that cannot be teased apart.5, 55, 56 This view is supported by the apparent inability of untrained participants to separate pain into different dimensions.21, 22, 23 Those studies that did not train participants to distinguish between the two dimensions found no difference between participants' ratings of the sensory and affective dimensions. Moreover, Fernandez and Turk22 showed that when participants rated each dimension on a separate week they failed to rate them differently, but when they were instructed to rate the two dimensions concurrently, they indeed provided distinct ratings. Such observations underpin the view that prising apart two dimensions from a unitary experience is, at best, pointless and, at worst, fallacious.22
A further and often overlooked point concerns the differences between experimental and clinical pain: experimental participants know that their pain will be short-lived, that they can withdraw at any time, and that there is very little or (most likely) no risk of injury.57 In contrast, chronic pain already has long-lasting pain, which they usually attribute to unresolved tissue damage.58 Given these differences, the unpleasantness of pain in experimental settings may be less compelling, or perhaps more modifiable, because of this reduced biological need to motivate protective behaviour.44 Moreover, participants in experimental pain studies arguably represent a subgroup of the general population—those who willingly volunteer for pain experiments. It is highly plausible that characteristics that render them willing to volunteer for pain experiments modulate their experiences and affect the mechanisms by which, and the extent to which, cognitive manipulations might modulate their pain.59
Future directions and limitations
Several suggestions can be made for future work on the basis of this review. First, experimental studies need to adopt rigorous methodologies so as to avoid demand effects. Second, the risk of bias assessment identified the need to remove ceiling effects and carefully word the verbal anchors in self-report scales so as to avoid ambiguity and to limit response bias. Third, both participants and researchers should be blinded to condition and participants should be naïve to the experimental hypotheses (remarkably, such issues are both commonly emphasised and commonly neglected).60 Fourth, raw data should be retained for longer periods of time to make pooling of data feasible for meta-analyses. This issue led to the main limitation of the current work—we were able to obtain raw data for only three of the 12 studies. Other limitations were as follows: the methodological challenges of the constituent studies and the possibility that our a priori selection criteria may have caused us to miss studies that inadvertently invoked selective modulation of separate dimensions of pain. However, the approach used by this systematic review was based on well-endorsed recommendations. A final consideration is the need to enhance reproducibility, for example full transparency in protocols, data analysis plans, and reporting.61
Conclusion
In contrast to popular opinion, evidence that the dimensions of pain can be selectively modulated using cognitive manipulations is weak. The most parsimonious conclusion seems to be that the sensory component cannot be selectively modulated, but the affective component might be, although significant threats to validity of the primary studies, and the risk of demand effects, warrant only tentative endorsement. If an effect does exist, it is likely to be very small. Although investigations of human pain have long considered these two dimensions of pain to be separable and differentially modifiable, the evidence suggests the alternative view, one that is usually held by people actually in pain—that pain is a unitary unpleasant and sensory experience. Several fundamental questions, with significant implications for the field, remain unanswered.
Authors' contributions
Study design: KT, VJM.
Concept design: GLM.
Data collection and analysis: KT, VJM.
Drafting of the first version of the paper: KT.
Editing of the paper: VJM, SJ, GLM.
All authors approved the final version of the manuscript.
Acknowledgements
The authors acknowledge Karolína Hlávková, Nikolay Kadaryia, Johana Vankava, and Alexandra Sorvina for translating the non-English articles identified for full-text screening.
Handling editor: J.G. Hardman
Editorial decision: 21 March 2019
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.bja.2019.03.033.
Declarations of interest
GLM receives payments for lectures and courses on pain and rehabilitation and he receives royalties for books on pain and rehabilitation. GLM and VM receive speaker fees for lectures on pain and rehabilitation. All the other authors declare no conflicts of interest.
Funding
NHMRC Principal Research Fellowship (#1061279; to GLM). NHMRC Project grant (#1047317; to GLM). Pfizer, Kaiser Permanente, Neuro Orthopaedic Institute, Workers' Compensation Boards in Australia and North America, Arsenal FC, Port Adelaide FC (to GLM). Oppenheimer Memorial Trust, South African National Research Foundation Innovation Postdoctoral Fellowship (to VJM). Australian Government Research Training Program (to KT).
Appendix A. Supplementary data
The following are the Supplementary data to this article:
References
- 1.Moseley G.L., Butler D.S. Fifteen years of explaining pain: the past, present, and future. J Pain. 2015;16:807–813. doi: 10.1016/j.jpain.2015.05.005. [DOI] [PubMed] [Google Scholar]
- 2.Moseley G.L., Vlaeyen J.W. Beyond nociception: the imprecision hypothesis of chronic pain. Pain Forum. 2015;156:35–38. doi: 10.1016/j.pain.0000000000000014. [DOI] [PubMed] [Google Scholar]
- 3.Moseley G.L. Why do people with complex regional pain syndrome take longer to recognize their affected hand? Neurology. 2004;62:2182–2186. doi: 10.1212/01.wnl.0000130156.05828.43. [DOI] [PubMed] [Google Scholar]
- 4.Erginel M.M. Plato on the psychology of pleasure and pain. Phoenix. 2011;65:288–314. [Google Scholar]
- 5.Price D. Psychological and neural mechanisms of the affective dimension of pain. Science. 2000;288:1769–1772. doi: 10.1126/science.288.5472.1769. [DOI] [PubMed] [Google Scholar]
- 6.IASP. International Association for the Study of Pain IASP Terminology 2017 updated December 14, 2017. Available from: https://www.iasp-pain.org/Education/Content.aspx?ItemNumber=1698.
- 7.Auvray M., Myin E., Spence C. The sensory-discriminative and affective-motivational aspects of pain. Neurosci Biobehav Rev. 2010;34:214–223. doi: 10.1016/j.neubiorev.2008.07.008. [DOI] [PubMed] [Google Scholar]
- 8.Melzack R., Katz J. The McGill Pain Questionnaire: appraisal and current status. In: Melzack R., Katz J., editors. Handbook of pain measurement. Guilford Press; New York: 2001. [Google Scholar]
- 9.Treede R.-D. The International Association for the Study of Pain definition of pain: as valid in 2018 as in 1979, but in need of regularly updated footnotes. Pain Rep. 2018;3:e643. doi: 10.1097/PR9.0000000000000643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Taenzer P., Melzack R., Jeans M.E. Influence of psychological factors on postoperative pain, mood and analgesic requirements. Pain. 1986;24:331–342. doi: 10.1016/0304-3959(86)90119-3. [DOI] [PubMed] [Google Scholar]
- 11.Wade J.B., Price D., Hamer R.M., Schwartz S.M., Hart R.P. An emotional component analysis of chronic pain. Pain. 1990;40:303–310. doi: 10.1016/0304-3959(90)91127-5. [DOI] [PubMed] [Google Scholar]
- 12.Maihofner C., Herzner B., Handwerker H. Secondary somatosensory cortex is important for the sensory-discriminative dimension of pain: a functional MRI study. Eur J Neurosci. 2006;23:1377–1383. doi: 10.1111/j.1460-9568.2006.04632.x. [DOI] [PubMed] [Google Scholar]
- 13.Rainville P. Brain mechanisms of pain affect and pain modulation. Curr Opin Neurobiol. 2002;12:195–204. doi: 10.1016/s0959-4388(02)00313-6. [DOI] [PubMed] [Google Scholar]
- 14.Rainville P., Feine J.S., Bushnell M., Duncan G.H. A psychophysical comparison of sensory and affective responses to four modalities of experimental pain. Somatosens Mot Res. 1992;9:265–277. doi: 10.3109/08990229209144776. [DOI] [PubMed] [Google Scholar]
- 15.Treede R.-D., Kenshalo D.R., Gracely R.H., Jones A.K. The cortical representation of pain. Pain. 1999;79:105–111. doi: 10.1016/s0304-3959(98)00184-5. [DOI] [PubMed] [Google Scholar]
- 16.Price D. Central neural mechanisms that interrelate sensory and affective dimensions of pain. Mol Interv. 2002;2:392. doi: 10.1124/mi.2.6.392. [DOI] [PubMed] [Google Scholar]
- 17.Coghill R.C., Mayer D.J., Price D. Wide dynamic range but not nociceptive-specific neurons encode multidimensional features of prolonged repetitive heat pain. J Neurophysiol. 1993;69:703–716. doi: 10.1152/jn.1993.69.3.703. [DOI] [PubMed] [Google Scholar]
- 18.Willis W., Zhang X., Honda C., Giesler G.J. Projections from the marginal zone and deep dorsal horn to the ventrobasal nuclei of the primate thalamus. Pain. 2001;92:267–276. doi: 10.1016/s0304-3959(01)00268-8. [DOI] [PubMed] [Google Scholar]
- 19.Willis W., Westlund K. Neuroanatomy of the pain system and of the pathways that modulate pain. J Clin Neurophysiol. 1997;14:2–31. doi: 10.1097/00004691-199701000-00002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Smart K., Doody C. Mechanisms-based clinical reasoning of pain by experienced musculoskeletal physiotherapists. Physiotherapy. 2006;92:171–178. doi: 10.1016/j.math.2006.02.006. [DOI] [PubMed] [Google Scholar]
- 21.Chapman R., Nakamura Y., Donaldson G.W. Sensory and affective dimensions of phasic pain are indistinguishable in the self-report and psychophysiology of normal laboratory subjects. J Pain. 2001;2:279–294. doi: 10.1054/jpai.2001.25529. [DOI] [PubMed] [Google Scholar]
- 22.Fernandez E., Turk D.C. Demand characteristics underlying differential ratings of sensory versus affective components of pain. J Behav Med. 1994;17:375–390. doi: 10.1007/BF01858009. [DOI] [PubMed] [Google Scholar]
- 23.Chapman R. Limbic processes and the affective dimension of pain. Prog Brain Res. 1996;110:63–81. doi: 10.1016/s0079-6123(08)62565-0. [DOI] [PubMed] [Google Scholar]
- 24.Chapman R., Donaldson G.W., Nakamura Y., Jacobson R.C., Bradshaw D.H., Gavrin J. A psychophysiological causal model of pain report validity. J Pain. 2002;3:143–155. doi: 10.1054/jpai.2002.123771. [DOI] [PubMed] [Google Scholar]
- 25.Fledman J. Expanding hypnotic pain management the affective dimension of pain. Am J Clin Hypn. 2009;51:235–254. doi: 10.1080/00029157.2009.10401674. [DOI] [PubMed] [Google Scholar]
- 26.Younger J., McCue R., Mackey S. Pain outcomes: a brief review of instruments and techniques. Curr Pain Headache Rep. 2009;13:39–43. doi: 10.1007/s11916-009-0009-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Wittgenstein L., von Wright G. Electronic ed. Oxford University Press; Oxford: 1998. Wittgenstein's nachlass the bergen. [Google Scholar]
- 28.Fernandez E., Turk D.C. Sensory and affective components of pain: separation and synthesis. Psychol Bull. 1992;112:205–217. doi: 10.1037/0033-2909.112.2.205. [DOI] [PubMed] [Google Scholar]
- 29.Moher D., Liberati A., Tetzlaff J., Altman D.G., PRISMA Group Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Ann Intern Med. 2009;151:264–269. doi: 10.7326/0003-4819-151-4-200908180-00135. [DOI] [PubMed] [Google Scholar]
- 30.Rainville P., Duncan G., Price D., Carrier B., Bushnell M.C. Pain affect encoded in human anterior cingulate but not somatosensory cortex. Science. 1997;277:968–971. doi: 10.1126/science.277.5328.968. [DOI] [PubMed] [Google Scholar]
- 31.Rainville P., Carrier B., Hofbauer R.K., Bushnell M.C., Duncan G.H. Dissociation of sensory and affective dimensions of pain using hypnotic modulation. Pain. 1999;82:159–171. doi: 10.1016/S0304-3959(99)00048-2. [DOI] [PubMed] [Google Scholar]
- 32.Hofbauer R., Rainville P., Duncan G., Bushnell M.C. Cortical representation of the sensory dimension of pain. J Neurophysiol. 2001;86:402–411. doi: 10.1152/jn.2001.86.1.402. [DOI] [PubMed] [Google Scholar]
- 33.Dahlgren L.A., Kurtz R.M., Strube M.J., Malone M.D. Differential effects of hypnotic suggestion on multiple dimensions of pain. J Pain Symptom Manage. 1995;10:464–470. doi: 10.1016/0885-3924(95)00055-4. [DOI] [PubMed] [Google Scholar]
- 34.Valentini E., Betti V., Hu L., colleagues Hypnotic modulation of pain perception and of brain activity triggered by nociceptive laser stimuli. Cortex. 2013;49:446–462. doi: 10.1016/j.cortex.2012.02.005. [DOI] [PubMed] [Google Scholar]
- 35.Kunz M., Lautenbacher S., LeBlanc N., Rainville P. Are both the sensory and the affective dimensions of pain encoded in the face? Pain. 2012;153:350–358. doi: 10.1016/j.pain.2011.10.027. [DOI] [PubMed] [Google Scholar]
- 36.Villemure C., Slotnick B.M., Bushnell M. Effects of odors on pain perception: deciphering the roles of emotion and attention. Pain. 2003;106:101–108. doi: 10.1016/s0304-3959(03)00297-5. [DOI] [PubMed] [Google Scholar]
- 37.Kenntner-Mabiala R., Weyers P., Pauli P. Independent effects of emotion and attention on sensory and affective pain perception. Cogn Emot. 2007;21:1615–1629. [Google Scholar]
- 38.Kenntner-Mabiala R., Andreatta M., Wieser M.J., Muhlberger A., Pauli P. Distinct effects of attention and affect on pain perception and somatosensory evoked potentials. Biol Psychol. 2008;78:114–122. doi: 10.1016/j.biopsycho.2008.01.007. [DOI] [PubMed] [Google Scholar]
- 39.Loggia M.L., Mogil J.S., Bushnell M. Experimentally induced mood changes preferentially affect pain unpleasantness. J Pain. 2008;9:784–791. doi: 10.1016/j.jpain.2008.03.014. [DOI] [PubMed] [Google Scholar]
- 40.Perlman D.M., Salomons T.V., Davidson R.J., Lutz A. Differential effects on pain intensity and unpleasantness of two meditation practices. Emotion. 2010;10:65. doi: 10.1037/a0018440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Grant J.A., Rainville P. Pain sensitivity and analgesic effects of mindful states in Zen meditators: a cross-sectional study. Psychosom Med. 2009;71:106–114. doi: 10.1097/PSY.0b013e31818f52ee. [DOI] [PubMed] [Google Scholar]
- 42.Weitzenhoffer A.M., Hilgard E.R. Consulting Psychologists Press; Palo Alto, CA: 1962. Stanford hypnotic susceptibility scale, form C. [Google Scholar]
- 43.Hawker G.A., Mian S., Kendzerska T., French M. Measures of adult pain: visual analog scale for pain (VAS pain), numeric rating scale for pain (NRS pain), McGill pain Questionnaire (MPQ), short-form McGill pain Questionnaire (SF-MPQ), chronic pain grade scale (CPGS), short form-36 bodily pain scale (SF-36 BPS), and measure of intermittent and constant osteoarthritis pain (ICOAP) Arthritis Care Res. 2011;63:S240–S252. doi: 10.1002/acr.20543. [DOI] [PubMed] [Google Scholar]
- 44.Moseley G.L., Butler D. Noigroup Publications; Adelaide, Australia: 2017. Explain pain supercharged. [Google Scholar]
- 45.Catley M.J., O'Connell N.E., Moseley G.L. How good is the Neurophysiology of Pain Questionnaire? A Rasch analysis of psychometric properties. J Pain. 2013;14:818–827. doi: 10.1016/j.jpain.2013.02.008. [DOI] [PubMed] [Google Scholar]
- 46.Moseley G.L. A pain neuromatrix approach to patients with chronic pain. Man Ther. 2003;8:130–140. doi: 10.1016/s1356-689x(03)00051-1. [DOI] [PubMed] [Google Scholar]
- 47.Moseley G.L. Evidence for a direct relationship between cognitive and physical change during an education intervention in people with chronic low back pain. Eur J Pain. 2004;8:39–45. doi: 10.1016/S1090-3801(03)00063-6. [DOI] [PubMed] [Google Scholar]
- 48.Tabor A., Thacker M.A., Moseley G.L., colleagues Pain: a statistical account. PLoS Comput Biol. 2017;13 doi: 10.1371/journal.pcbi.1005142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Tabor A., O'Daly O., Gregory R.W. Perceptual inference in chronic pain: an investigation into the economy of action hypothesis. Clin J Pain. 2015;32:588–593. doi: 10.1097/AJP.0000000000000305. [DOI] [PubMed] [Google Scholar]
- 50.Rosenzweig S., Greeson J.M., Reibel D.K., Green J.S., Jasser S.A., Beasley D. Mindfulness-based stress reduction for chronic pain conditions: variation in treatment outcomes and role of home meditation practice. J Psychosom Res. 2010;68:29–36. doi: 10.1016/j.jpsychores.2009.03.010. [DOI] [PubMed] [Google Scholar]
- 51.Grant J.A. Meditative analgesia: the current state of the field. Ann N Y Acad Sci. 2014;1307:55–63. doi: 10.1111/nyas.12282. [DOI] [PubMed] [Google Scholar]
- 52.Turk D.C., Melzack R. The measurement of pain and the assessment of people experiencing pain. In: Turk D.C., Melzack R., editors. Handbook of pain assessment. Guilford Press; New York: 2001. pp. 1–11. [Google Scholar]
- 53.Bingel U., Wanigasekera V., Wiech K. The effect of treatment expectation on drug efficacy: imaging the analgesic benefit of the opioid remifentanil. Sci Transl Med. 2011;3 doi: 10.1126/scitranslmed.3001244. 70ra14. [DOI] [PubMed] [Google Scholar]
- 54.Campbell P., Bishop A., Dunn K.M., Main C.J., Thomas E., Foster N.E. Conceptual overlap of psychological constructs in low back pain. Pain. 2013;154:1783–1791. doi: 10.1016/j.pain.2013.05.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Chapman R., Gavrin J. Suffering: the contributions of persistent pain. Lancet. 1999;353:2233–2237. doi: 10.1016/S0140-6736(99)01308-2. [DOI] [PubMed] [Google Scholar]
- 56.Moseley G.L. Reconceptualising pain according to modern pain science. Phys Ther Rev. 2007;12:169–178. [Google Scholar]
- 57.Price D., Harkins S.W., Baker C. Sensory-affective relationships among different types of clinical and experimental pain. Pain. 1987;28:297–307. doi: 10.1016/0304-3959(87)90065-0. [DOI] [PubMed] [Google Scholar]
- 58.Price D., Harkins S.W., Rafii A., Price C. A simultaneous comparison of fentanyl's analgesic effects on experimental and clinical pain. Pain. 1986;24:197–203. doi: 10.1016/0304-3959(86)90042-4. [DOI] [PubMed] [Google Scholar]
- 59.Knudsen L., Petersen G.L., Nørskov K.N. Review of neuroimaging studies related to pain modulation. Scand J Pain. 2011;2:108–120. doi: 10.1016/j.sjpain.2011.05.005. [DOI] [PubMed] [Google Scholar]
- 60.Park J., Bang H., Cañette I. Blinding in clinical trials, time to do it better. Complement Ther Med. 2008;16:121–123. doi: 10.1016/j.ctim.2008.05.001. [DOI] [PubMed] [Google Scholar]
- 61.Lee H., Lamb S.E., Bagg M.K., Toomey E., Cashin A.G., Mosely G.L. Reproducible and replicable pain research: a critical review. Pain. 2018;159:1683–1689. doi: 10.1097/j.pain.0000000000001254. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.