Abstract
Preeclampsia is a complication of pregnancy characterized by new-onset hypertension and proteinuria of gestation, with serious consequences for mother and infant. Although a vast amount of research has been performed on the pathogenesis of preeclampsia, the underlying mechanisms of this multisystemic disease have remained to be fully elucidated. Data were retrieved from Gene Expression Omnibus database GSE40182 dataset. After data preprocessing, differentially expressed genes of placental cells cultured in vitro from preeclampsia and normal pregnancy were determined and subjected to Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment analysis to identify the associated pathways. Furthermore, functional principal component analysis (FPCA) was used to calculate the corresponding F-value of each gene. In order to further study the key signaling pathways of preeclampsia, an elastic-net regression model and the Mann-Whitney U (MWU) test were used to estimate the weight of the signaling pathways. Finally, a co-expression network was generated and hub genes were identified based on the topological features. A total of 134 pathways with a role in preeclampsia were identified. The gene expression data of placenta cells cultured in vitro for different durations were determined and F-values of genes were estimated using the FPCA model. The top 1,000 genes were identified as the differentially expressed genes and subjected to further analysis by elastic-net regression and MWU test. Two key signaling pathways were different between the preeclampsia and control groups, namely hsa05142 Chagas disease and hsa05204 Chemical carcinogenesis. Among the genes involved in these two key pathways, 13 hub genes were identified from the co-expression network. Clustering analysis demonstrated that depending on these hub genes, it was possible to divide the sample into four distinct groups based on different incubation time. The top 3 candidates were Toll-like receptor 2 (TLR2), glutathione S-transferase omega 1 (GSTO1) and mitogen-activated protein kinase 13 (MAPK13). TLR2 and associated pathways are known to be closely associated with preeclampsia, indirectly demonstrating the applicability of the analytic process applied. However, the role of GSTO1 and MAPK13 in preeclampsia has remained poorly investigated, and elucidation thereof may be a worthwhile endeavor. The present study may provide a basis for exploring potential novel genes and pathways as therapeutic targets for preeclampsia.
Keywords: pre-eclampsia, pathway enrichment analysis, functional gene set enrichment analyses, elastic-net regression model, Mann-Whitney U-test, co-expression network construction
Introduction
Preeclampsia is a complication of pregnancy characterized by new-onset hypertension and proteinuria of gestation, with serious consequences for mother and infant (1). This gestation-specific syndrome affects 8% of all pregnancies worldwide (2). Preeclampsia may result in eclampsia and hemolysis, elevated liver enzymes and low platelets (HELLP syndrome) in the mother, and preterm birth, intrauterine growth restriction and perinatal death in the fetus (2).
In a normal pregnancy, cytotrophoblasts invade the uterine spiral arteries and cause arterial remodeling, replacing much of the maternal endothelium. Consequently, the high-resistance uterine arteriolar system is transformed into a low-resistance, high-capacity system, allowing for increased fetal blood flow and delivery of oxygen and nutrients to the placenta (3,4). In preeclampsia, a two-stage theory has been proposed as a useful concept to address its pathophysiology. Stage 1 comprises reduced placental perfusion due to inadequate cytotrophoblast invasion of spiral arterioles. This then translates, in certain but not all maternal females, into stage 2, the multisystemic maternal syndrome of preeclampsia (5). In addition, there is a certain association between preeclampsia and maternal rejection, and a correlation between prednisone and trophoblastic insufficiency. Preeclampsia is also closely associated with fetal rejection, gene polymorphisms and trophoblastic insufficiency (6–8). Although a vast amount of studies have been performed on preeclampsia, the pathogenesis of this multisystemic disease has remained to be fully elucidated.
Recent technological advances in high-throughput sequencing and Bioinformatics provide tools to obtain valuable information on the dynamic changes of genes in specific disorders. Gene set enrichment analyses (GSEAs), including Gene Ontology functional term and Kyoto Encyclopedia of Genes and Genomes (KEGG) biological pathway enrichment analysis are widely used to identify the biological mechanisms of gene sets (9,10). The currently available methods have two major inadequacies: i) Designed comparisons or regression analyses are applied to the comparison between control and experimental groups. These traditional methods are not able to effectively exploit time information in time series of transcriptomic measurements; ii) overlapping genes in multiple pathways are thought to have multiple roles in hypothesis testing, while the weight coefficients are overestimated (11).
In the present study, a novel analytical procedure for GSEA was used to overcome these defects. First, KEGG enrichment analysis was used to identify the differential signaling pathways between preeclampsia patients and controls. Furthermore, functional principal component analysis (FPCA), which utilizes the temporal information of gene expression profiles, was performed. Subsequently, the elastic-net regression method was performed to eliminate overlapping gene effects. Combined with the Mann-Whitney U (MWU) test, the key molecular pathways were identified from the gene transcription data of preeclampsia patients vs. controls.
Materials and methods
Data retrieval and pre-processing
Gene expression data for preeclampsia patients that had been deposited in the Gene Expression Omnibus (GEO) database of the National Center for Biotechnology Information (NCBI; www.ncbi.nlm.nih.gov/gds; accession no. GSE40182) were downloaded. A total of 39 samples were contained in the dataset. Villous cytotrophoblasts (CTBs) had been isolated from preeclampsia placentas and placentas of preterm labor patients without signs of infection, which served as gestation-matched controls. In addition, gene expression data were obtained from the in vitro culture of those villous CTBs cultured for different durations. According to the American College of Obstetricians and Gynecologists published criteria (12,13), patients with the most clinically significant forms of this condition that necessitated preterm delivery were included: Maternal females with severe preeclampsia ± intrauterine growth restriction, preeclampsia with superimposed hypertension and HELLP syndrome. The first group named as the preterm labor group included 19 samples and served as gestation-matched controls, and the second group contained 20 samples and was named as the preeclampsia group. After converting the row names of the gene expression profile matrix into GENESYMBOL format, 23,520 genes were obtained. Based on the description of the time-series expression profile, the 39 samples were divided into four time-series groups depending on the culture time: 0, 12, 24 and 48 h, which can provide a common CTB fingerprint for preeclampsia. Meanwhile, CTB cell was under normal culturing conditions. All analyses were performed using the bioinformatics platform from Honghui Biotech Co. Ltd (http://www.genelibs.com).
FPCA analysis of time-series expression profile
Time-series gene signature data were analyzed by functional principal component analysis and an F value was determined for each gene (11). The mean expression of each gene was subtracted and FPCA was adopted across all of the centered expression values. Each gene expression value was calculated according to the following function:
| (i) |
In the above formula, is the FPC score, which quantifies how much (t) may be explained by represents the average expression in the temporal sample and represents the lth eigenfunction.
When this was applied to the time-dependent gene expression, functional F-statistics were used to summarize the gene pattern information for each gene at the different time-points:
| (ii) |
where is the residual sum of squares of null hypotheses, represents the residual sum of squares of alternative hypotheses and δ represents the signal-to-noise ratio. Fi may be considered to indicate the importance a gene, and i stands for a gene (14).
Pathway enrichment analysis
Differentially expressed genes were determined by ranking their F values and ranked list of genes was used for pathway enrichment analysis. KEGG pathway enrichment analysis was performed to obtain significant pathways of the preeclampsia and controls (15). In the present study, Fisher's exact test was performed to select significant pathways. Pathways with P<0.05 and an intersection gene count of >1 were extracted, and were considered as important pathways.
Estimation of the weights of genes using the elastic-net regression model
Regression analysis is a predictive modeling technique to assess the association between dependent and independent variables. This technique has been frequently used in predictive analyses and in identifying causal associations between variables, particularly in time-series models (16). The elastic-net regression model combines the Lasso algorithm and Ridge regression technique (17,18). The major function of the model was as follows:
| (iii) |
| iv |
Where λ is the penalty coefficient (λ1=0.4, λ2=0.01) and is the vector of the set of linear coefficients. When is calculated and estimated, the weights of the overlapping genes may be obtained as follows:
| (v) |
if gene i belongs to the ĸth gene set only. Due to the use of lasso penalty, in some cases both the numerator and denominator may be zero, in which case . The above model would take apart an overlapping gene between gene-sets and eliminate the effects of overlapping genes (11).
Weighted MWU test with correlation using GSEA
The MWU test is a rank-based non-parametric test that is usually used in a competitive GSEA. The MWU test utilizes the gene weight value to test whether the weight of this gene is significantly greater than that of other genes (background genes) (19). In combination with the t-test, key signaling pathways are thereby obtained.
Co-expression network construction and hub gene identification
First, an adjacency matrix of genes in the key pathways was built using Spearman correlation analysis based on the gene expression values. Any two genes were connected and if the adjacency value of a pair of genes was >0.95, the given edge would be retained for inclusion in the co-expression network. Topological features were further studied to identify key nodes in the network. Genes whose degree of connectivity was bigger than the average degree values were considered as hub genes. Heat map and clustering analysis were used to visualize the hub genes in preeclampsia. Receiver operating characteristic (ROC) analysis was performed and the area under the ROC curve (AUC) was calculated by using the ROCR package in R.
Results
FPCA analysis of time-series expression profile
The FPCA model was used to identify temporally differently expressed genes and each gene was assigned with individual F-value F-values were then listed in descending order and the top 1,000 differentially expressed genes were thereby identified. FPCA analysis narrowed the gene search range from 4,419 to 1,000 for the dataset GSE40182. The distribution of the F-values of pathway-associated genes is presented in Fig. 1. FPCA was able to effectively utilize the time series information and overcome the deficiencies of the traditional control design that could not achieve comparison with a control group at one time-point (11). In the FPCA model, the basis functions are estimated from the observed data and the data-adaptive basis has the favorable property to flexibly characterize the major modes of variation in the data (20). F-values were then subjected to the MWU test.
Figure 1.
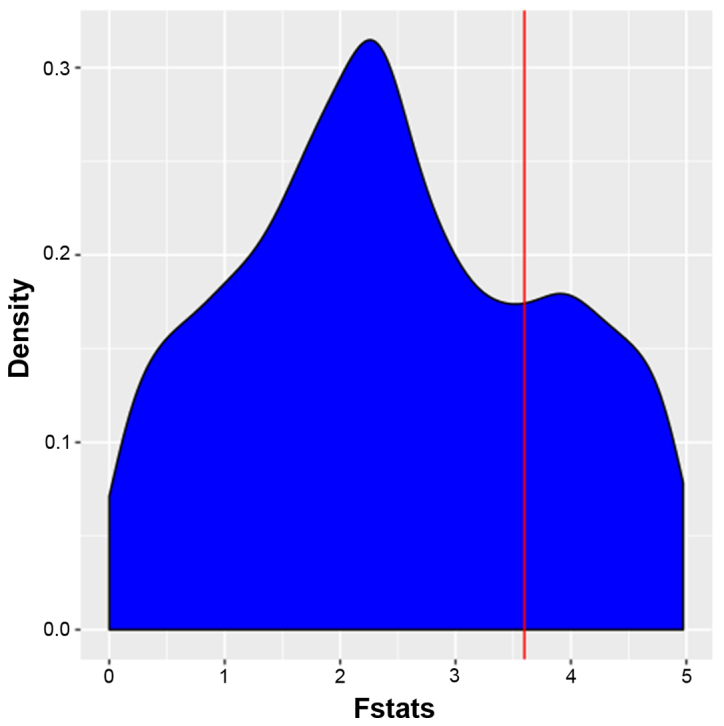
Distribution of F-values of pathway genes from GSE40182. Time-series gene signature data were analyzed by functional principal component analysis and an F value (x-coordinate, F) was determined for each gene. The Y-axis represents the gene density. Genes were ranked in the order of the F-value and the top 1,000 genes were selected. The red line represents the threshold of the top 1,000 genes.
Pathway enrichment analysis based on KEGG
Pathway enrichment analysis of differentially expressed genes between the preeclampsia vs. control group was performed using the KEGG pathway database. A total of 286 pathways comprising 6,893 genes were obtained. After applying Fisher's exact test, 134 pathways comprising 4,419 genes were retained. In Table I, the top 6 signaling pathways associated with preeclampsia are presented in an ascending order based on the P-value. The most significant pathway was ‘olfactory transduction’.
Table I.
The top 6 differentially expressed pathways of GSE40182 according to the KEGG analysis.
| Pathway name | P-value | FDR | Gene count |
|---|---|---|---|
| hsa04740: Olfactory transduction | 5.89×10−191 | 1.69×10-188 | 112 |
| hsa05206: MicroRNAs in cancer | 6.99×10−57 | 1.00×10−54 | 151 |
| hsa05200: Pathways in cancer | 1.87×10−15 | 1.79×10−13 | 391 |
| hsa04151: PI3K/Akt signaling pathway | 9.93×10−10 | 7.13×10−8 | 336 |
| hsa00970: Aminoacyl tRNA biosynthesis | 5.53×10−9 | 3.17×10−7 | 39 |
| hsa04810: Regulation of actin cytoskeleton | 2.23×10−8 | 1.07×10−6 | 211 |
Hsa, Homo sapiens; PI3K, phosphoinositide 3-kinase; tRNA, transfer RNA.
Weights of genes estimated using the elastic-net regression model
Overlapping genes are defined as genes involved in multiple pathways. These genes have multiple roles in hypothesis testing, leading to overestimation of the weight coefficients.
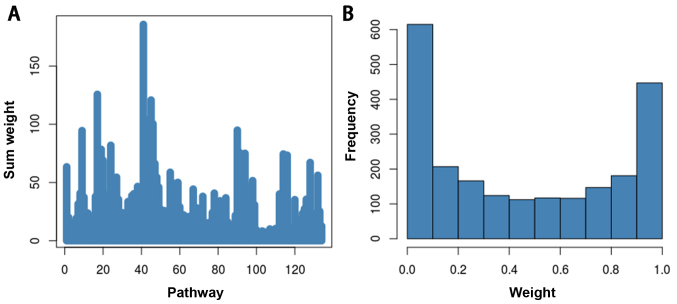
In the present study, an elastic-net regression model was used to take apart an overlapping gene between gene sets and eliminate the overlapping effects. After calculating the weight value of each gene and adding the weight values of the genes in the pathways, the total weight value of the pathways was obtained. The total weight of each pathway in presented in Fig. 2. The weight value of each gene was used for MWU test.
Figure 2.
(A) Sum weight and (B) weight distribution of genes involved in the 134 pathways associated with preeclampsia from GSE40182.
Weighted MWU test with correlation using GSEA
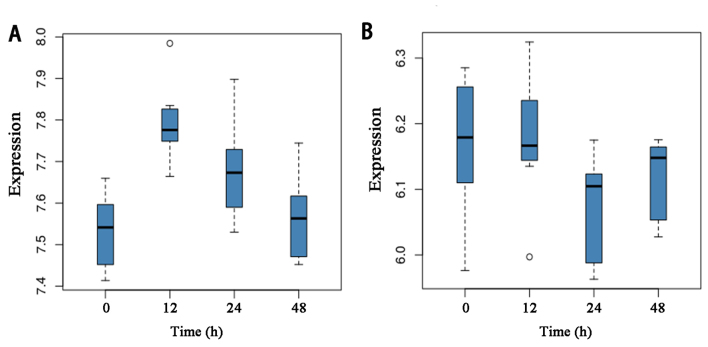
A total of 134 pathways enriched by the differentially expressed were obtained were obtained by KEGG pathway analysis. Using the MWU test, key molecular pathways for differentially expressed genes between preeclampsia and controls were identified. Based on the results of the test, pathways were ranked in a descending order. The top two candidates were pathways in hsa05142: Chagas disease (American trypanosomiasis) and hsa05204: Chemical carcinogenesis. The expression levels of the genes in the two pathways at different time-points are provided in Fig. 3.
Figure 3.
Expression levels of genes in the two key signaling pathways. (A) Expression levels for hsa05142 in Chagas disease (American trypanosomiasis). (B) Expression levels for hsa05204: Chemical carcinogenesis. The data are the expression levels of 39 samples, which were divided into 4 groups based on culturing time (0, 12, 24 and 48 h). Hsa, Homo sapiens.
Hub genes in the co-expression network and clustering analysis
A weighted gene co-expression network was constructed by Spearman correlation analysis. For this, a total of 61 genes involved in the pathways hsa05142 Chagas disease and hsa05204 Chemical carcinogenesis were selected. Edges with an adjacency value for a pair of genes of >0.95 were retained for the network. Subsequently, the co-expression network was built. A total of 30 nodes (genes) were contained in the co-expression network. The co-expression network of the genes of the two most important pathways in preeclampsia is displayed in Fig. 4.
Figure 4.
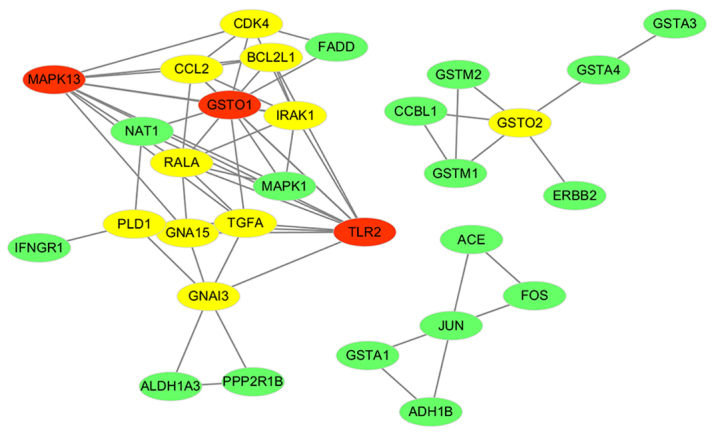
Co-expression network of genes was based on the Chagas disease and Chemical carcinogenesis pathways. The Spearman coefficient of all edges in this network was >0.95. The hub genes are indicated in red and yellow. Red nodes have a higher degree of connectivity than yellow and green nodes.
The topological features of the network were further studied to identify key nodes. Genes with a degree of connectivity above average (average degree, 4.258) were considered as hub genes. In the present study, a total of 13 hub genes (red and yellow nodes represent hub genes, red nodes have a higher degree than yellow nodes and green nodes) were identified, including glutathione S-transferase omega (GSTO)2, GSTO1, BCL2 like 1 (BCL2L1), RAS like proto-oncogene A (RALA), transforming growth factor α (TGFA), cyclin dependent kinase 4 (CDK4), phospholipase D1 (PLD1), Toll-like receptor 2 (TLR2), interleukin 1 receptor associated kinase 1 (IRAK1), mitogen-activated protein kinase (MAPK13), C-C motif chemokine ligand 2 (CCL2), G protein subunit α 15 (GNA15) and G protein subunit alpha I3 (GNAI3). Table II lists the key topological characteristics of the hub genes. The diagnostic value (AUC) of each gene is provided in Fig. S1. The top AUC was 66.84% for the gene TLR2.
Table II.
Key topological characteristics of the hub genes in the co-expression network.
| Gene name | Degree of connectivity | Betweenness centrality | Closeness centrality | No. of directed edges |
|---|---|---|---|---|
| TLR2 | 11 | 0.18228291 | 0.65384615 | 11 |
| GSTO1 | 11 | 0.25733543 | 0.73913043 | 11 |
| MAPK13 | 10 | 0.07893908 | 0.62962963 | 10 |
| RALA | 8 | 0.03345588 | 0.58620690 | 8 |
| IRAK1 | 8 | 0.02447479 | 0.58620690 | 8 |
| TGFA | 7 | 0.08142507 | 0.62962963 | 7 |
| CCL2 | 7 | 0.01197479 | 0.56666667 | 7 |
| PLD1 | 6 | 0.13480392 | 0.54838710 | 6 |
| GNAI3 | 6 | 0.22058824 | 0.54838710 | 6 |
| GNA15 | 6 | 0.03734244 | 0.58620690 | 6 |
| GSTO2 | 5 | 0.76666667 | 0.85714286 | 5 |
| BCL2L1 | 5 | 0.00000000 | 0.53125000 | 5 |
| CDK4 | 5 | 0.01250000 | 0.45945946 | 5 |
TLR2, Toll-like receptor 2; MAPK13, mitogen-activated protein kinase 13; GSTO1, glutathione S-transferase omega 1; GSTO2, glutathione S-transferase o 2; BCL2L1, BCL2 like 1; RALA, RAS like proto-oncogene A; TGFA, transforming growth factor α; CDK4, cyclin dependent kinase 4; PLD1, phospholipase D1; IRAK1, interleukin 1 receptor associated kinase 1; CCL2, C-C motif chemokine ligand 2; GNA15, G protein subunit α 15; GNAI3, G protein subunit α I3.
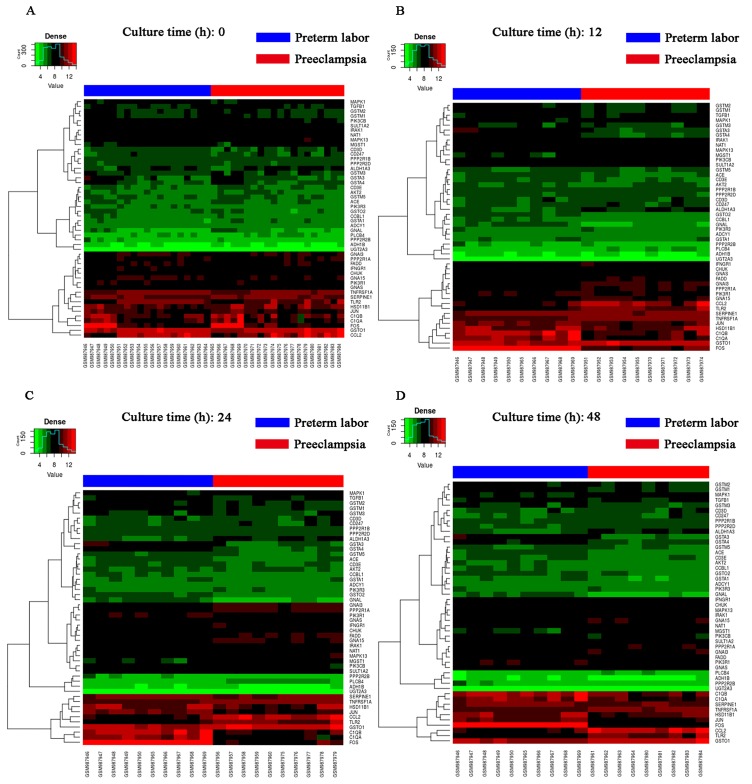
Based on the degree of connectivity, the top 3 candidates were TLR2, GSTO1 and MAPK13. Heat map and clustering analysis were also used to determine the hub genes in preeclampsia. The clustering heat map is presented in Fig. 5. It was observed that there were distinctive expression levels in different groups at 4 different time-points.
Figure 5.
Heat map generated based on 39 samples from GSE40182. The samples were divided into 4 groups. Blue represents preterm labor, red represents preeclampsia. The clustering heat map represents a culture time of (A) 0 h and a culture time of (B) 12 h, (C) 24 h and (D) 48 h of CTB cell from preeclampsia and preterm labor.
Discussion
The causes of preeclampsia remain to be fully elucidated and are a hotspot of current research. The present study focused on the relative expression of the whole genome in primary culture of villous CTB from placentas of subjects with preeclampsia compared with normal pregnancies. The present results enhance the current understanding of the potential factors in preeclampsia. In recent years, novel mathematical models have been widely used in molecular biology and Bioinformatics. Studies have indicated that dysregulation in gene expression at numerous time-points may be calculated by cutting-edge mathematical models (21,22), including high-dimensional differential equations (23,24), dynamic Bayesian networks (21,22) and Granger's model (25).
In the present study, the pathogenesis of preeclampsia was analyzed using Bioinformatics, including KEGG enrichment analysis, FPCA, elastic-net regression and MWU test. According to this novel analytical procedure, two major signaling pathways, hsa05142 Chagas disease and hsa05204 Chemical carcinogenesis, were identified. In the pathway has05142 (Fig. S2; taken from KEGG; www.kegg.jp/dbget-bin/www_bget?map05142), TLR signaling was identified. In the context of pregnancy, it is reported that maternal females with systemic lupus erythematosus have a 3–4-fold higher risk of developing preeclampsia (26). Systemic inflammatory disorders involving extensive tissue damage are characterized by significant changes in the innate immune system. TLRs have an important role in the underlying pathophysiological mechanisms of preeclampsia. TLR2, one of the TLRs, which is expressed on the surface of certain cells and recognizes foreign substances, has a role in the immune system. Previous studies have provided solid evidence that the immune system is closely associated with preeclampsia, and the TLR family has a significant involvement (27–29). Goulopoulou et al (30) proposed a hypothesis that severely necrotic trophoblasts lead to the release of mitocshondrial DNA, stimulating TLR9 to induce an immune response, resulting in hypertension and intra-uterine growth restriction. Circulating mitochondrial DNA may be a potential marker of inchoate preeclampsia, and anti-TLR9 treatments may be promising for this disease (31). Consistent with those studies, the present results also suggested that TLR2 has a diagnostic potential in preeclampsia, which may be used for clinical applications (Fig. S1). Maternal females with preeclampsia had increased TLR2 and TLR4 mRNA and protein expression compared with normal pregnancies (32). MAPK13 is a member of the MAPK family, taking part in a wide variety of cellular processes and acting as an integration point for TLR signaling (33). There are also studies on MAPKs involvement in preeclampsia, which enhance the current understanding of the mechanisms associated with this condition (34).
In order to further elucidate the interrelation of genes involved in those two pathways, a co-expression network was constructed in the present study. Based on the topological features, 13 hub genes were identified. The top 3 candidates were TLR2, GSTO1 and MAPK13.
Apart from TLR2 and MAPK13 mentioned above, GSTO1 in the pathway hsa05204 Chemical carcinogenesis also has an important role in preeclampsia (Fig. S3, taken from KEGG; www.kegg.jp/dbget-bin/www_bget?map05204). GSTO1 has dehydroascorbate reductase activity and has an important role in the glutathione-ascorbate cycle as part of the anti-oxidant metabolism (35).
Numerous studies have verified the influence of TLR pathways on preeclampsia. The result was a demonstration of the feasibility and scientific value of this analytic process. Furthermore, TLR2, GSTO1 and MAPK13 were identified as key molecules according to this analysis, which provides a basis for further studies on the significance of these genes in preeclampsia.
In conclusion, TLR2, GSTO1 and MAPK13 were identified as key genes of preeclampsia and have potential as diagnostic markers or drug targets for preeclampsia; however, further experimental verification should be performed to confirm these in silico results. Since the distribution of clinical factors may be different in other regions or databases, the present results also require verification in clinical samples of a large number of subjects from multiple centers/geographical regions.
Supplementary Material
Acknowledgements
The authors would like to thank Jinan Honghui Biotech Co. Ltd. for providing support for data pre-processing.
Funding
No funding received.
Availability of data and materials
The datasets used and/or analyzed during the present study are available from the corresponding author on reasonable request.
Authors' contributions
XL and YF wrote the manuscript; XL downloaded and analyzed data, and drafted the manuscript; YF interpreted the data and made critical revisions. All authors discussed the results and reviewed the manuscript.
Ethics approval and consent to participate
No applicable.
Patient consent for publication
No applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Chaiworapongsa T, Chaemsaithong P, Yeo L, Romero R. Pre-eclampsia part 1: Current understanding of its pathophysiology. Nat Rev Nephrol. 2014;10:466–480. doi: 10.1038/nrneph.2014.102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hariharan N, Shoemaker A, Wagner S. Pathophysiology of hypertension in preeclampsia. Microvasc Res. 2017;109:34–37. doi: 10.1016/j.mvr.2016.10.002. [DOI] [PubMed] [Google Scholar]
- 3.Yang JI, Kong TW, Kim HS, Kim HY. The proteomic analysis of human placenta with pre-eclampsia and normal pregnancy. J Korean Med Sci. 2015;30:770–778. doi: 10.3346/jkms.2015.30.6.770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Zhou Y, Gormley MJ, Hunkapiller NM, Kapidzic M, Stolyarov Y, Feng V, Nishida M, Drake PM, Bianco K, Wang F, et al. Reversal of gene dysregulation in cultured cytotrophoblasts reveals possible causes of preeclampsia. J Clin Invest. 2013;123:2862–2872. doi: 10.1172/JCI72817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Roberts JM, Gammill HS. Preeclampsia: Recent insights. Hypertension. 2005;46:1243–1249. doi: 10.1161/01.HYP.0000188408.49896.c5. [DOI] [PubMed] [Google Scholar]
- 6.Minassian C, Thomas SL, Williams DJ, Campbell O, Smeeth L. Acute maternal infection and risk of pre-eclampsia: A population-based case-control study. PLoS One. 2013;8:e73047. doi: 10.1371/journal.pone.0073047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Banadakoppa M, Vidaeff AC, Yallampalli U, Ramin SM, Belfort MA, Yallampalli C. Complement split products in amniotic fluid in pregnancies subsequently developing early-onset preeclampsia. Dis Markers. 2015;2015:263109. doi: 10.1155/2015/263109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Song L, Zhong M. Association between Interleukin-10 gene polymorphisms and risk of early-onset preeclampsia. Int J Clin Exp Pathol. 2015;8:11659–11664. [PMC free article] [PubMed] [Google Scholar]
- 9.Alshahrour F, Minguez P, Tárraga J, Montaner D, Alloza E, Vaquerizas JM, Conde L, Blaschke C, Vera J, Dopazo J. BABELOMICS: A systems biology perspective in the functional annotation of genome-scale experiments. Nucleic Acids Res. 2006;34:W472–W476. doi: 10.1093/nar/gkl172. (Web Server Issue) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Tabas-Madrid D, Nogales-Cadenas R, Pascual-Montano A. GeneCodis3: A non-redundant and modular enrichment analysis tool for functional genomics. Nucleic Acids Res. 2012;40:W478–W483. doi: 10.1093/nar/gks402. (Web Server Issue) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Zhang Y, Topham DJ, Thakar J, Qiu X. FUNNEL-GSEA: FUNctioNal ELastic-net regression in time-course gene set enrichment analysis. Bioinformatics. 2017;33:1944–1952. doi: 10.1093/bioinformatics/btx104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.ACOG Committee on Practice Bulletins-Obstetrics, corp-author. ACOG practice bulletin. Diagnosis and management of preeclampsia and eclampsia. Number 33, January 2002. Obstet Gynecol. 2002;99:159–167. doi: 10.1016/s0029-7844(01)01747-1. [DOI] [PubMed] [Google Scholar]
- 13.Haram K, Svendsen E, Abildgaard U. The HELLP syndrome: Clinical issues and management. A review. BMC Pregnancy Childbirth. 2009;9:8. doi: 10.1186/1471-2393-9-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Wu S, Wu H. More powerful significant testing for time course gene expression data using functional principal component analysis approaches. BMC Bioinformatics. 2013;14:6. doi: 10.1186/1471-2105-14-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zhu J, Yao X. Use of DNA methylation for cancer detection: Promises and challenges. Int J Biochem Cell Biol. 2009;41:147–154. doi: 10.1016/j.biocel.2008.09.003. [DOI] [PubMed] [Google Scholar]
- 16.Lu CJ, Wu JY, Lee TS. Application of independent component analysis preprocessing and support vector regression in time series prediction. International Joint Conference on Computational Sciences and Optimization. 2009:468–471. doi: 10.1109/CSO.2009.231. [DOI] [Google Scholar]
- 17.Ogutu JO, Schulzstreeck T, Piepho HP. Genomic selection using regularized linear regression models: Ridge regression, lasso, elastic net and their extensions. BMC Proc. 2012;6(Suppl 2):S10. doi: 10.1186/1753-6561-6-S2-S10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Townsend W. ELASTICREGRESS: Stata module to perform elastic net regression, lasso regression, ridge regression. Statist Software Comp. 2017 [Google Scholar]
- 19.Brandsma CA, van den Berge M, Postma DS, Jonker MR, Brouwer S, Paré PD, Sin DD, Bossé Y, Laviolette M, Karjalainen J, et al. A large lung gene expression study identifying fibulin-5 as a novel player in tissue repair in COPD. Thorax. 2015;70:21–32. doi: 10.1136/thoraxjnl-2014-205091. [DOI] [PubMed] [Google Scholar]
- 20.Zhao Y, Chen MH, Pei B, Rowe D, Shin DG, Xie W, Yu F, Kuo L. A bayesian approach to pathway analysis by integrating gene-gene functional directions and microarray data. Stat Biosci. 2012;4:105–131. doi: 10.1007/s12561-011-9046-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Low ST, Mohamad MS, Omatu S, Chai LE, Deris S, Yoshioka M. Inferring gene regulatory networks from perturbed gene expression data using a dynamic Bayesian network with a Markov Chain Monte Carlo algorithm. IEEE International Conference on Granular Computing. 2014:179–184. [Google Scholar]
- 22.Young WC, Raftery AE, Yeung KY. Fast Bayesian inference for gene regulatory networks using ScanBMA. BMC Syst Biol. 2014;8:47. doi: 10.1186/1752-0509-8-47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Cai TT, Zhang A. Inference for high-dimensional differential correlation matrices. J Multivar Anal. 2016;143:107–126. doi: 10.1016/j.jmva.2015.08.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Chung NC, Storey JD. Statistical significance of variables driving systematic variation in high-dimensional data. Bioinformatics. 2015;31:545–554. doi: 10.1093/bioinformatics/btu674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Basu S, Shojaie A, Michailidis G. Network granger causality with inherent grouping structure. J Mach Learn Res. 2012;16:1–31. [PMC free article] [PubMed] [Google Scholar]
- 26.Abd Rahman R, DeKoninck P, Murthi P, Wallace EM. Treatment of preeclampsia with hydroxychloroquine: A review. J Matern Fetal Neonatal Med. 2018;31:525–529. doi: 10.1080/14767058.2017.1289511. [DOI] [PubMed] [Google Scholar]
- 27.Perez-Sepulveda A, Torres MJ, Khoury M, Illanes SE. Innate immune system and preeclampsia. Front Immunol. 2014;5:244. doi: 10.3389/fimmu.2014.00244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Saito S, Shiozaki A, Nakashima A, Sakai M, Sasaki Y. The role of the immune system in preeclampsia. Mol Aspects Med. 2007;28:192–209. doi: 10.1016/j.mam.2007.02.006. [DOI] [PubMed] [Google Scholar]
- 29.Laresgoiti-Servitje E. A leading role for the immune system in the pathophysiology of preeclampsia. J Leukoc Biol. 2013;94:247–257. doi: 10.1189/jlb.1112603. [DOI] [PubMed] [Google Scholar]
- 30.Goulopoulou S, Matsumoto T, Bomfim GF, Webb RC. Toll-like receptor 9 activation: A novel mechanism linking placenta-derived mitochondrial DNA and vascular dysfunction in pre-eclampsia. Clin Sci (Lond) 2012;123:429–435. doi: 10.1042/CS20120130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Yasuda K, Rutz M, Schlatter B, Metzger J, Luppa PB, Schmitz F, Haas T, Heit A, Bauer S, Wagner H. CpG motif-independent activation of TLR9 upon endosomal translocation of ‘natural’ phosphodiester DNA. Eur J Immunol. 2006;36:431–436. doi: 10.1002/eji.200535210. [DOI] [PubMed] [Google Scholar]
- 32.Xie F, Hu Y, Turvey SE, Magee LA, Brunham RM, Choi KC, Krajden M, Leung PC, Money DM, Patrick DM, et al. Toll-like receptors 2 and 4 and the cryopyrin inflammasome in normal pregnancy and pre-eclampsia. BJOG. 2010;117:99–108. doi: 10.1111/j.1471-0528.2009.02428.x. [DOI] [PubMed] [Google Scholar]
- 33.Cerezo-Guisado MI, Cuenda A. MAPK13 (mitogen-activated protein kinase 13) Atlas Genet Cytogenet Oncol Haematol. 2010;14:911–914. [Google Scholar]
- 34.Luo X, Yao ZW, Qi HB, Liu DD, Chen GQ, Huang S, Li QS. Gadd45α as an upstream signaling molecule of p38 MAPK triggers oxidative stress-induced sFlt-1 and sEng upregulation in preeclampsia. Cell Tissue Res. 2011;344:551–556. doi: 10.1007/s00441-011-1164-z. [DOI] [PubMed] [Google Scholar]
- 35.Whitbread AK, Masoumi A, Tetlow N, Schmuck E, Coggan M, Board PG. Characterization of the omega class of glutathione transferases. Methods Enzymol. 2005;401:78–99. doi: 10.1016/S0076-6879(05)01005-0. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets used and/or analyzed during the present study are available from the corresponding author on reasonable request.