Abstract
Osteoporosis is a disease with a worldwide prevalence that involves a severe loss of bone mineral density and decreased microarchitecture, which increases the risk of bone fracture. The present study evaluated the effects of isopsoralen on osteoblastic OB-6 cells following hydrogen peroxide (H2O2)-induced damage and investigated the molecular mechanisms involved in this process. For in vitro experiments, OB-6 osteoblasts were treated with H2O2 or H2O2 + isopsoralen then the cell viability, apoptosis, reactive oxygen species (ROS) production and calcium accumulation were determined. Results demonstrated that treatment with H2O2 reduced cell viability, runt-related transcription factor 2 (RUNX2) and osteocalcin (OCN) expression levels, and calcium deposition, whilst markedly increasing cell apoptosis and ROS production. However, isopsoralen (1 µM) provided significant protection against H2O2-induced alterations in osteoblasts. In addition, isopsoralen effectively upregulated protein expression of tankyrase and β-catenin which are the main transductors of the Wnt/β-catenin pathway. Of note, the protective effects of isopsoralen against H2O2-induced damage were attenuated in OB-6 cells treated with tankyrase inhibitor XAV-939. In conclusion, the present findings provided evidence that isopsoralen attenuated oxidative stress-induced injury in osteoblasts via the Wnt/β-catenin signaling pathway.
Keywords: isopsoralen, oxidative stress, osteoblast, Wnt/β-catenin pathway
Introduction
Osteoporosis is a skeletal disease where systemic destruction of bone mass and microarchitecture occurs, which often increases the risk of bone fractures and other complications (1,2). Osteoporosis typically affects the elderly, with women more at risk, and the disease negatively impacts the patient's quality of life (3). There is a direct link between osteoporosis and menopause (4), whilst one in five men will suffer from osteoporotic fracture during their lifetime (5). Therefore, the treatment of osteoporosis has attracted intensive attention worldwide.
To date, teriparatide, strontium and bisphosphonates have been widely used for the treatment of osteoporosis by reducing bone resorption and increasing bone formation (6); however, these synthetic medicines have several side-effects (7). Therefore, novel, more effective and safer agents that can be used for the treatment of osteoporosis are highly required. Notably, isopsoralen and its isoformer psoralen are isolated from a number of fruits, vegetables and traditional Chinese medicines (8,9), and they both display antiosteoporotic effects in ovariectomy-induced osteoporotic rats (10) and in vitro (11). In addition, isopsoralen demonstrates osteoprotective effects as well as stimulatory effects on bone formation (12,13) through increasing strength of trabecular bone microstructure (14), inducing apoptosis or necrosis of osteosarcoma (15) and promoting the differentiation of bone marrow stromal stem cells (BMSCs) into osteoblasts (16). However, the underlying osteoprotective mechanisms of isopsoralen have not been widely investigated.
The Wnt/β-catenin signaling pathway is a conserved signaling pathway with its signal transduced by β-catenin and tankyrase proteins (17,18). The alterations of Wnt/β-catenin activity are often associated with developmental disorders and diseases such as cancer, neuronal diseases and skeletal disorders (19). Increasing evidence indicates that the Wnt/β-catenin signaling pathway is involved in the regulation of osteogenesis and bone formation (20,21). In the present study, the protective effects and mechanisms of isopsoralen on oxidative stress-induced osteoblastic OB-6 cells were investigated. Results suggested that isopsoralen could ameliorate oxidative stress-induced damage through the Wnt/β-catenin signaling pathway, indicating that isopsoralen may be a novel treatment approach for osteoporosis.
Materials and methods
Cell culture and treatments
OB-6 osteoblastic cells (Cell Bank of Shanghai Institute of Biological Science) were cultured in α-minimum essential medium (α-MEM; Thermo Fisher Scientific, Inc.) supplemented with 10% FBS (Thermo Fisher Scientific, Inc.) and and 1% penicillin-streptomycin (Thermo Fisher Scientific, Inc.), and incubated at 37°C with 5% CO2. OB-6 cells were divided into four groups including the control group with α-MEM only, an isopsoralen group treated with 1 µM isopsoralen (Shanghai Meilun Biotechnology Co., Ltd.) for 12, 24 and 48 h, a hydrogen peroxide (H2O2) group treated with 100 µM H2O2 for 12, 24 and 48 h or a isopsoralen + H2O2 group pretreated with 1 µM isopsoralen and 100 µM H2O2 for 12, 24 and 48 h. Subsequently, cell viability, apoptosis, runt-related transcription factor (RUNX2) and osteocalcin (OCN) expression, reactive oxygen species (ROS) production, mitochondrial respiratory chain complex I activity, tankyrase protein expression, Wnt3, β-catenin, and calcium accumulation were analyzed, as described below.
Cell viability assay
OB-6 osteoblastic cells at a density of 5×103 cells/ml were seeded in 96-well plates and cultured in α-MEM with 1 µM isopsoralen, 100 µM hydrogen peroxide (H2O2) or 1 µM isopsoralen + 100 µM H2O2. Following 12, 24, 48 h of culture, cell viability was evaluated using 2, 3-bis (2-methoxy-4-nitro-5-sulfophenyl)-2-tetrazolium 5-carboxanilide (XTT, Sigma-Aldrich; Merck KGaA) method in accordance with a previous report (10). Each experiment was performed in triplicate and repeated four times.
Flow cytometry
Cells at a density 5×103 cells/ml were seeded in a 96-well plate, then treated with H2O2 (100 µM) or isopsoralen (1 µM) + H2O2 (100 µM) for 12, 24 and 48 h. Cell apoptotic rate was measured by flow cytometry according to the manufacturer's instructions. In brief, OB-6 osteoblastic cells were collected and fixed in 70% ethanol for 30 min at room temperature. Cells were stained with Annexin V-fluorescein isothiocyanate/propidium iodide (50 µg/ml) using an Annexin V-FITC Apoptosis Detection kit (Roche Molecular Diagnostics) prior to analysis using a FACScanVantage™ SE (BD Biosciences, San Jose, CA, USA). Each experiment was performed in triplicate and repeated four times and data were analyzed using the FloJo software version 10.1.1 (FlowJo, LLC).
Reverse transcription-quantitative polymerase chain reaction (RT-qPCR)
RT-qPCR was used to determine OCN and RUNX2 mRNA expression. Total cellular RNA was isolated using TRIzol reagent (Invitrogen; Thermo Fisher Scientific) and cDNA synthesis was performed using TaKaRa PrimeScript II 1st Strand cDNA Synthesis kit (Invitrogen; Thermo Fisher Scientific, Inc.). qPCR was performed using the SYBR PrimeScript RT-PCR kit (Takara Bio, Inc.) in an Applied Biosystems 7500 Fluorescent Quantitative PCR System (Applied Biosystems; Thermo Fisher Scientific, Inc). The thermocycling conditions were as follows: 40 cycles at 94°C for 5 min, 94°C for 30 sec and 58°C for 1 min. The following primers were provided by Sangon Biotech Co., Ltd.: OCN forward, 5′-CATGAGAGCCCTCACTCT-3′ and reverse, 5′-AGAGCGACACCCTAGAC-3′; RUNX2 forward, 5′-AGCCTCTTCAGCGCAGTGAC-3′ and reverse, 5′-CTGGTGCTCGGATCCCAA-3′ and GAPDH forward, 5′-AACGGATTTGGTCGTATTGGG-3′ and reverse, 5′-CCTGGAAGATGGTGATGGGAT-3′. Each experiment was performed in triplicate and repeated four times. The 2−ΔΔCq method was used to quantify the data (22).
Western blot analysis
Western blotting was used to determine the protein expression levels of tankyrase, Wnt3 and β-catenin in accordance to a previous study (11). Total protein was extracted from cells using RIPA lysis buffer (Beyotime Institute of Biotechnology) and their concentrations were determined using the BCA method. A total of 20 µg protein was separated using 10% SDS-PAGE and transferred to PVDF membranes. The membranes were subsequently blocked with 5% non-fat milk for 2 h at room temperature and incubated at 4°C overnight with primary antibodies against tankyrase (1:2,500; cat. no. sc-365897; Santa Cruz Biotechnology Inc.) Wnt3α (1:2,500; cat. no. 2391; Santa Cruz Biotechnology Inc.), β-catenin (1:1,000; cat. no. 8480; Cell Signaling Technology, Inc.) and β-actin (1:2,500; cat. no. sc-47778; Santa Cruz Biotechnology Inc.). Finally, membranes were incubated with horseradish peroxidase-conjugated anti-rabbit IgG antibody (1:5,000; cat. no. 7074; Cell Signaling Technology, Inc.) at 37°C for 1 h. Image J2X software version 2.1.4.7 (Rawak Software, Inc.) was used to determine protein band density. Each experiment was performed in triplicate and repeated three times.
Calcium accumulation
Calcium Colorimetric Assay kits (cat. no. MAK022; Sigma-Aldrich; Merck KGaA) were used to measure calcium deposition (expressed as µg/mg cellular protein) according to the manufacturer's instructions. Each experiment was performed in triplicate and repeated three times.
ROS production
Intracellular ROS production was measured using a CellROX™ Green Reagent (cat. no. C10444; Invitrogen; Thermo Fisher Scientific, Inc.) according to the manufacturer's guidelines. ROS levels were measured with a FACSCalibur™ flow cytometer (BD Biosciences) with the excitation source at 488 nm and emission at 525 nm. Data were analyzed using the FloJo software version 10.1.1 (FlowJo LLC). Each experiment was performed in triplicate and repeated three times.
Mitochondrial respiratory chain complex I activity
Enzymatic activities of complex I were determined spectrophotometrically as described in a previous study (23). Mitochondrial respiratory chain complex I activity was calculated as the rotenone-sensitive rate of nicotinamide adenine dinucleotide oxidation (ε=6180 M−1 at 340 nm). Each experiment was performed in triplicate and repeated three times.
Blocking the Wnt/β-catenin signaling pathway using inhibitor XAV-939
OB-6 cells were treated with tankyrase inhibitor XAV-939 (10 µM; Abcam) for 30 min followed by addition of H2O2 (100 µM) or H2O2 (100 µM) + isopsoralen (1 µM) for 24 h. The control was treated with just the media. Cell apoptosis, ROS production and calcium accumulation were then evaluated as described above. Each experiment was performed in triplicate and repeated three times.
Statistical analysis
Data are presented as the mean ± standard deviation. SPSS version 17.0 software (SPSS, Inc.) was used to process data. The comparisons between groups were performed using one-way ANOVA and Tukey's honestly significant difference post hoc test. P<0.05 was considered to indicate statistical significance.
Results
Isopsoralen inhibits H2O2-induced apoptosis in osteoblastic OB-6 cells
Osteoblastic OB-6 cell viability was examined by XTT assay. Results indicated that cell viability was significantly inhibited by H2O2 compared with the control, in a time-dependent manner (P<0.05; Fig. 1A). Isopsoralen significantly ameliorated cell viability in H2O2-treated OB osteoblastic cells (P<0.05; Fig. 1A). Subsequently, the apoptotic rate was examined by flow cytometry. Results indicated that there was a significantly increase in the apoptotic rate in H2O2-treated cells compared with the control group (P<0.05; Fig. 1B). By contrast, H2O2-induced apoptosis was significantly inhibited by isopsoralen (P<0.01; Fig. 1B).
Figure 1.
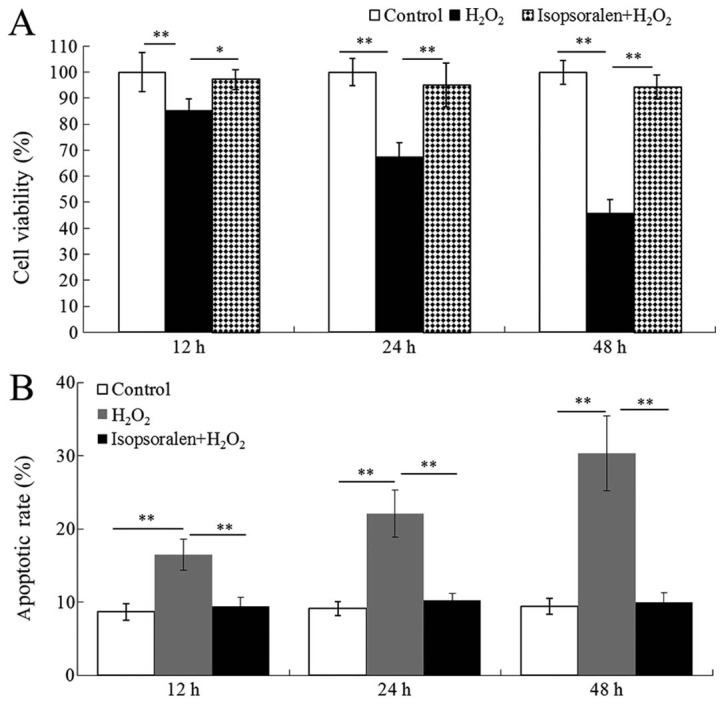
Isopsoralen inhibits H2O2-induced apoptosis. (A) Cell viability and (B) apoptotic rate were analyzed. *P<0.05 or **P<0.01, with comparisons indicated by lines. H2O2, hydrogen peroxide.
Isopsoralen protects cell mitochondrial function in H2O2-treated osteoblastic OB-6 cells
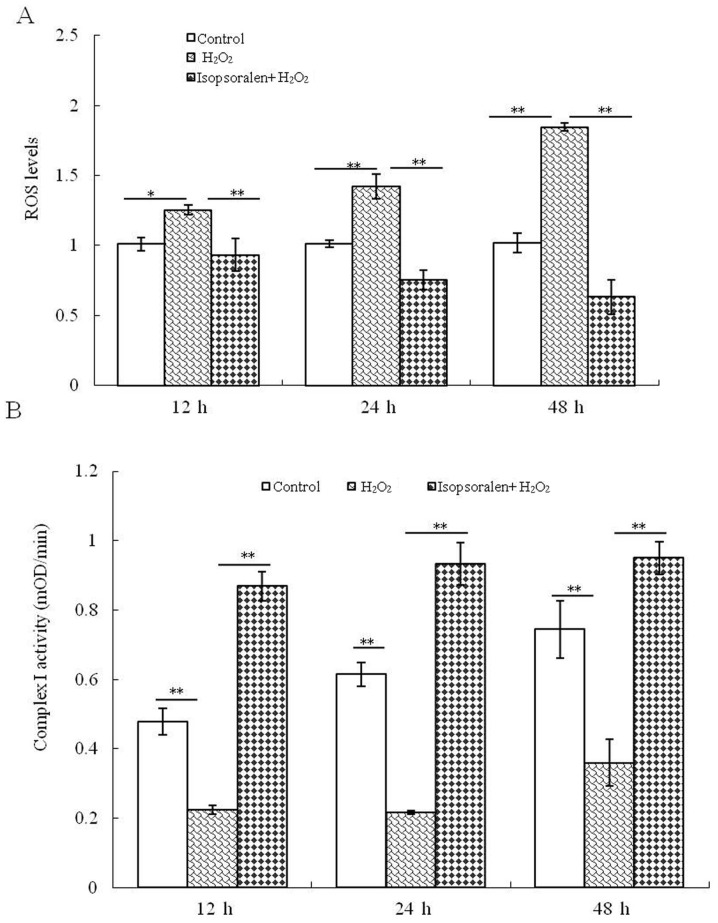
When compared with the control group, a significant downregulation in ROS production was detected in H2O2-treated OB-6 cells at 12, 24 and 48 h. Of note, treatment with isopsoralen significantly reduced H2O2-mediated ROS production (P<0.01; Fig. 2A). In addition, the activity of complex I was examined following treatment with H2O2 or H2O2 + isopsoralen. It was identified that complex I activity decreased significantly in the H2O2-treated cells, but isopsoralen significantly reversed the effects induced by H2O2 (P<0.01; Fig. 2B). These findings suggested that isopsoralen protected the OB-6 osteoblast mitochondrial function following treatment with H2O2.
Figure 2.
Isopsoralen protects against H2O2-induced mitochondrial dysfunction. (A) Levels of ROS and (B) activity of complex I OB-6 osteoblastic cells were measured. *P<0.05 or **P<0.01, with comparisons indicated by lines. H2O2, hydrogen peroxide; ROS, reactive oxygen species.
Isopsoralen restores bone formation in H2O2-treated osteoblastic OB-6 cells
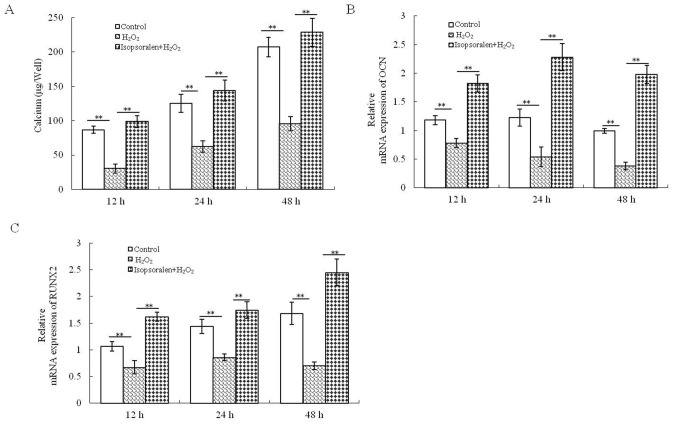
Calcium accumulation was significantly reduced by H2O2, and this effect was reversed by treatment with isopsoralen at all time-points (P<0.01; Fig. 3A). Furthermore, isopsoralen significantly promoted osteogenic gene expression (OCN and RUNX2) in H2O2-treated osteoblasts (P<0.01; Fig. 3B and C). These results further supported the hypothesis that isopsoralen reduced the damage to bone formation following H2O2 treatment.
Figure 3.
Isopsoralen protects against H2O2-induced suppression of bone formation. (A) Calcium accumulation, (B) OCN expression and (C) RUNX2 expression were determined. **P<0.01, with comparisons indicated by lines. H2O2, hydrogen peroxide; OCN, osteocalcin; RUNX2, runt-related transcription factor 2.
Isopsoralen activates the intracellular Wnt/β-catenin signaling pathway in H2O2-treated osteoblastic OB-6 cells
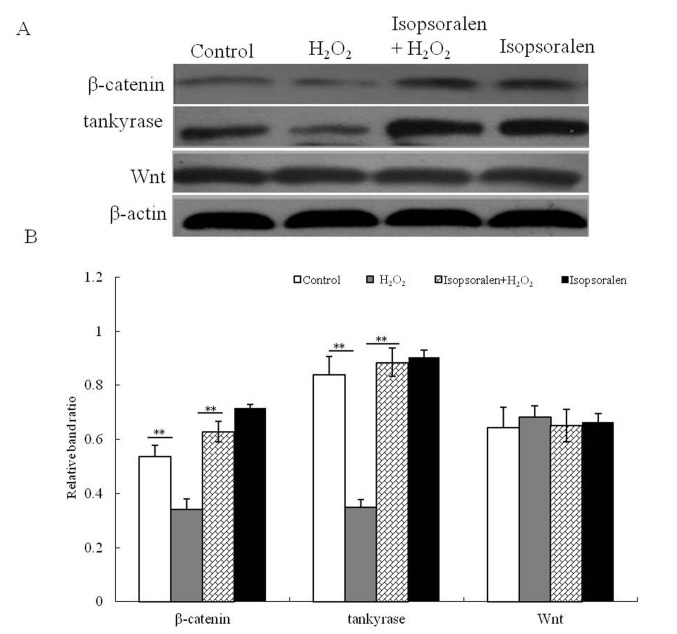
To identify the cytoprotective mechanism of isopsoralen, the activation of the Wnt/β-catenin signaling pathway was investigated. The effect of isosporalen on the protein expression of Wnt, β-catenin and tankyrase in H2O2-treated and non-H2O2-treated osteoblastic OB-6 cells was determined (Fig. 4A). It was demonstrated that β-catenin and tankyrase expression were significantly decreased in osteoblastic OB-6 cells treated with H2O2 compared with the control group (P<0.01; Fig. 4B). When compared with the H2O2-treated cells, the isopsoralen treatment promoted the expression of β-catenin and tankyrase (P<0.01; Fig. 4B), whilst isopsoralen + H2O2 treatment also significantly increased β-catenin and tankyrase protein expression. There was no difference in Wnt expression between the four treatment groups (P>0.05; Fig. 4B). These results suggested that isopsoralen activated the Wnt/β-catenin signaling pathway and was able to attenuate H2O2-induced Wnt/β-catenin signal inhibition in osteoblastic OB-6 cells.
Figure 4.
Isopsoralen activates the Wnt/β-catenin pathway. (A) Protein expression of tankyrase, Wnt and β-catenin was analyzed by Western blot analysis. (B) Western blot results were analyzed quantiatatively. **P<0.01, with comparisons indicated by lines. H2O2, hydrogen peroxide.
The Wnt/β-catenin signaling pathway is the target for isopsoralen in H2O2-treated osteoblastic OB-6 cells
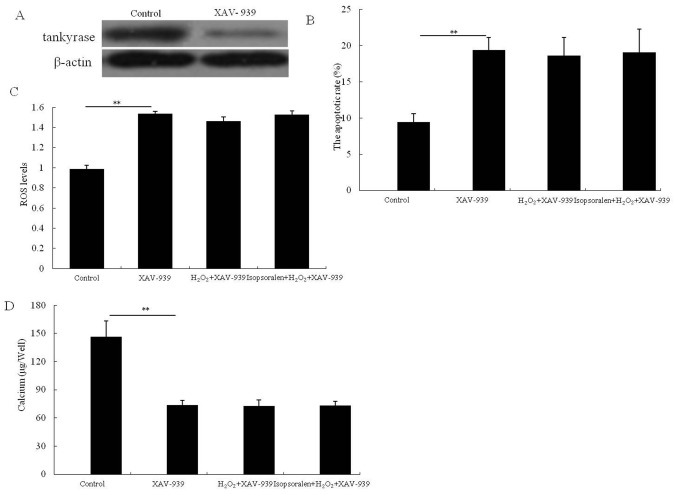
To identify the role of the Wnt/β-catenin signaling pathway in osteoblastic OB-6 cells, cells were pretreated with 10 µM inhibitor XAV-939 for 30 min then treated with H2O2, or isopsoralen + H2O2 for 24 h. OB-6 cells treated with XAV-939 demonstrated an obvious decrease in tankyrase protein expression which confirmed the inhibitory effect of XAV-939. Furthermore, following the XAV-939 pretreatment, there was no significant difference in cell apoptosis (P>0.05; Fig. 5B), ROS production (P>0.05; Fig. 5C) or calcium accumulation (P>0.05; Fig. 5D) between the H2O2-treated group and isopsoralen + H2O2-treated group. Therefore, isopsoralen inhibited the apoptotic rate and protected the mitochondrial function and bone formation in H2O2-treated OB-6 cells via activation of the Wnt/β-catenin signaling pathway.
Figure 5.
Activation of the Wnt/β-catenin pathway is required for isopsoralen-induced protection against H2O2. (A) The expression of tankyrase, (B) cell apoptotic rate, (C) ROS production and (D) calcium accumulation were analyzed. **P<0.01, with comparisons indicated by lines. H2O2, hydrogen peroxide; ROS, reactive oxygen species.
Discussion
Osteoporosis increases the risk of bone fractures and other complications, and affects >200 million people worldwide (24). To date, studies investigating the cellular effects and molecular mechanisms of isopsoralen for osteoporosis treatment are limited. The present study demonstrated that isopsoralen had protective effects against oxidative stress induced by H2O2, indicated by the increased cell proliferation, mitochondrial function and bone formation compared with the H2O2-treated cells. It was also identified that isopsoralen activated the Wnt/β-catenin signaling pathway.
Osteoblasts are crucial in bone formation and remodeling (25). For a number of years, numerous research teams have focused on identifying novel molecular targets to promote osteoblast cell survival (26). H2O2 induces osteoblastic cell apoptosis and also impairs osteoblast differentiation (27,28). Therefore, H2O2 is often used in vitro to establish a cellular model of osteonecrosis (29). A previous study determined that intraperitoneal injection of isopsoralen at 5 and 10 mg/kg per day for two weeks significantly enhanced the hind limb locomotor function of mice with bone damage. The same study also identified significant anti-apoptotic activity of isopsoralen by regulating the PI3K/AKT pathway (30). Isopsoralen at concentrations of 1×10−5 mol/l effectively inhibits H2O2-induced damage to human lens epithelial cells through upregulating two mitochondrial protein spots (with m/z of 6532 and 6809) (31). The present study identified that H2O2 treatment inhibited OB-6 osteoblastic cell proliferation and induced cell apoptosis. By contrast, isopsoralen treatment promoted cell survival and protected against the damage induced by H2O2. However, cell proliferation was only investigated using the XTT method due to the limited financial support for this investigation.
The equilibrium between the formation and resorption of bone exerts a crucial role in modulating bone mass in osteoporosis (32). The present study demonstrated that isopsoralen promoted calcium accumulation, and also OCN and RUNX2 mRNA expression following H2O2 treatment which indicated that isopsoralen enhanced bone formation in OB-6 cells. Previous in vivo and in vitro studies suggested that isopsoralen had osteoprotective effects. For example, following administration of isopsoralen (10 and 20 mg/kg for 8 weeks) to sex hormone-deprived mice, osteoporosis symptoms were ameliorated with mice displaying increased bone strength and improved trabecular bone microstructure (14). In nude rats with osteosarcoma, isopsoralen and psoralen treatment induced growth inhibitory effects on transplanted tumors, and caused apoptosis or necrosis of osteosarcoma without significant toxic effects (15). Isopsoralen suppresses bone BMSCs proliferation in a dose-dependent manner whilst significantly enhancing osteogenesis as determined by the promotion of alkaline phosphatase (ALP) activity, calcium salt sediment yield, and basic fibroblast growth factor, insulin-like growth factor-1, sp7 transcription factor and runx-2 mRNA expression (16). A recent study by Wang et al (33) examined the influence of isopsoralen on the differentiation of MSCs into osteoblasts as well as its effect on bone formation under osteoporotic conditions. Results suggested that isopsoralen promoted BMSC differentiation into osteoblasts due to upregulated ALP, OCN and RUNX2 expression. In addition, bone parameters including trabecular thickness, bone volume and trabecular number in the distal femoral metaphysis were significantly increased in ovariectomized mice following intragastrical administration with isopsoralen. These findings suggest that isopsoralen has potential functions in preventing and treating osteoporosis. The present study demonstrated that treatment with isopsolaren increased OCN and RUNX2 mRNA expression, as well as calcium deposition in OB-6 osteoblastic cells.
ROS are unavoidable by-products of oxidative stress with the concentration determining the influence on cells. ROS at low concentration activates gene expression in cells, but ROS at high concentration damages lipids, proteins and nuclear acids, and therefore is harmful to cells (34). During homeostasis, ROS levels are strictly controlled by antioxidant enzymes, but this equilibrium is disturbed under oxidative stress (35,36). It has been determined that ROS production in mitochondria is promoted under environmental stimuli and this dramatically affects cell growth (37–39). The present study determined that H2O2-induced stress promoted ROS production and inhibited complex I activity in cultured OB-6 cells. Isopsoralen protected mitochondrial damage induced by H2O2 via downregulating ROS production and upregulating complex I activity. ROS is crucial in osteoporosis development with increased ROS production induced by oxidative stress, leading to osteoblast cell damage and apoptosis (40,41). Furthermore, ROS accelerates apoptosis of osteoblasts, inhibits their differentiation and impairs bone formation (42,43). The present findings demonstrated that isopsoralen protected mitochondrial functions in OB cells subjected to oxidative stress through inhibiting ROS production.
To the best of our knowledge, the present study provided the first evidence that isopsoralen attenuated H2O2-induced damage in oxidatively challenged OB-6 osteoblasts via the Wnt/β-catenin signaling pathway. It was demonstrated that isopsoralen activated the Wnt/ß-catenin signaling pathway due to the promotion of β-catenin and tankyrase expression, but no significant effects on Wnt expression. Tankyrase and β-catenin are the main transductors of the Wnt/β-catenin pathway, and therefore alterations in their expression levels demonstrated the effect of isopsoralen on the Wnt/β-catenin signaling pathway. The recent characterization of the Wnt/β-catenin signaling pathway in the regulation of osteoblast proliferation and differentiation provided important insights into understanding of bone metabolism pathophysiology (44). For example, the Wnt/β-catenin signaling pathway serves an essential role in bone formation, regeneration, modeling and remodeling by stimulating the proliferation and differentiation of osteoblasts, and also due to its role in the response of bone to mechanical loading (45–47). Nuclear accumulation of stabilized β-catenin is an important indicator of osteoblast differentiation (48,49). Inactivation of Axin2, a negative regulator of the Wnt/β-catenin pathway, promotes osteoblast proliferation and differentiation, as well as matrix mineralization in vitro and in vivo (50). Activation of the Wnt/β-catenin pathway enhances Runx2 expression, which is a crucial transcription factor for osteoblast differentiation (51). Therefore, the discovery and development of new drugs that activate the Wnt/β-catenin pathway is a promising approach for the development of novel osteoporosis therapies. In conclusion, the present findings demonstrated that isopsoralen protected against damage induced by H2O2 in OB-6 cells, indicated by decreased apoptosis, and increased mitochondrial function and bone formation. Furthermore, it was identified that the Wnt/β-catenin signaling pathway may be the underlying signaling pathway involved in this process. The current study was conducted using only the cultured cell line OB-6, therefore future study into the effects of isopsoralen will involve other cell lines and in vivo work to fully elucidate the underlying mechanism of action. However, the present results did provide evidence for the development of novel therapeutic approaches involving isoporalen for bone-related disease treatment.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Authors' contributions
YPL was guarantor of the current study and designed the study, performed the data analysis and experimental studies, and prepared and edited the manuscript. BW performed the statistical analysis. FL and JL researched the literature and acquired the data. JL also reviewed and revised manuscript. All authors read and approved the final manuscript.
Ethics approval and consent to participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Rachner TD, Khosla S, Hofbauer LC. Osteoporosis: Now and the future. Lancet. 2011;377:1276–1287. doi: 10.1016/S0140-6736(10)62349-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bliuc D, Nguyen ND, Milch VE, Nguyen TV, Eisman JA, Center JR. Mortality risk associated with low-trauma osteoporotic fracture and subsequent fracture in men and women. JAMA. 2009;301:513–521. doi: 10.1001/jama.2009.50. [DOI] [PubMed] [Google Scholar]
- 3.Dueregger A, Heidegger I, Ofer P, Perktold B, Ramoner R, Klocker H, Eder IE. The use of dietary supplements to alleviate androgen deprivation therapy side effects during prostate cancer treatment. Nutrients. 2014;6:4491–4519. doi: 10.3390/nu6104491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Tuck SP, Datta HK. Osteoporosis in the aging male: Treatment options. Clin Interv Aging. 2007;2:521–536. doi: 10.2147/CIA.S820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Khosla S. Update in male osteoporosis. J Clin Endocrinol Metab. 2010;95:3–10. doi: 10.1210/jc.2009-1740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Wang ZQ, Li JL, Sun YL, Yao M, Gao J, Yang Z, Shi Q, Cui XJ, Wang YJ. Chinese herbal medicine for osteoporosis: A systematic review of randomized controlled trails. Evid. Based. Complement. Alternat. Med. 2013;2013:356260. doi: 10.1155/2013/356260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Uebelhart D, Frey D, Frey-Rindova P, Goerres G, Michel BA. Therapy of osteoporosis: Bisphosphonates, SERM's, teriparatide and strontium. Z Rheumatol. 2003;62:512–517. doi: 10.1007/s00393-003-0560-5. (In German) [DOI] [PubMed] [Google Scholar]
- 8.Wang Y, Hong C, Zhou C, Xu D, Qu HB. Screening antitumor compounds Psoralen and Isopsoralen from Psoralea coryl folia L. Seeds. Evid Based Complement Alternat Med. 2011;2011:363052. doi: 10.1093/ecam/nen087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wu CR, Chang CL, Hsieh PY, Lin LW, Ching H. Psoralen in isopsoralen, two coumarins of Psoraleae Fructus, can alleviate scopolamine-induced amnesia in rats. Planta Med. 2007;73:275–278. doi: 10.1055/s-2007-967127. [DOI] [PubMed] [Google Scholar]
- 10.Yang Z, Huang JH, Liu SF, Zhao YJ, Shen ZY, Wang YJ, Bian Q. The osteoprotective effect of psoralen in ovariectomy-induced osteoporotic rats via stimulating the osteoblastic differentiation from bone mesenchymal stem cells. Menopause. 2012;19:1156–1164. doi: 10.1097/gme.0b013e3182507e18. [DOI] [PubMed] [Google Scholar]
- 11.Tang DZ, Yang F, Yang Z, Huang J, Shi Q, Chen D, Wang YJ. Psoralen stimulates osteoblast differentiation through activation of BMP signaling. Biochem Biophys Res Commun. 2011;405:256–261. doi: 10.1016/j.bbrc.2011.01.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Tsai MH, Huang GS, Hung YC, Bin L, Liao LT, Lin LW. Psoralea corylifolia extract ameliorates experimental osteoporosis in ovariectomized rats. Am J Chin Med. 2007;35:669–680. doi: 10.1142/S0192415X07005168. [DOI] [PubMed] [Google Scholar]
- 13.Wong RW, Rabie AB. Effect of psoralen on bone formation. J Orthop Res. 2011;29:158–164. doi: 10.1002/jor.21124. [DOI] [PubMed] [Google Scholar]
- 14.Yuan X, Bi Y, Yan Z, Pu W, Li Y, Zhou K. Psoralen and Isopsoralen ameliorate sex hormone deficiency-induced osteoporosis in female and male mice. Biomed Res Int. 2016;2016:6869452. doi: 10.1155/2016/6869452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lu H, Zhang L, Liu D, Tang P, Song F. Isolation and purification of psoralen and isopsoralen and their efficacy and safety in the treatment of osteosarcoma in nude rats. Afr Health Sci. 2014;14:641–647. doi: 10.4314/ahs.v14i3.20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ming L, Ge B, Chen K, Ma H, Zhai Y. Effects of isopsoralen on bone marrow stromal stem cells differentiate and proliferate in vitro. Zhongguo Zhong Yao Za Zhi. 2011;36:2124–2128. (In Chinese) [PubMed] [Google Scholar]
- 17.Neben CL, Merrill AE. Signaling pathways in craniofacial development: Insights from rare skeletal disorders. Curr Top Dev Biol. 2015;115:493–542. doi: 10.1016/bs.ctdb.2015.09.005. [DOI] [PubMed] [Google Scholar]
- 18.Alfaro MP, Pagni M, Vincent A, Atkinson J, Hill MF, Cates J, Davidson JM, Rottman J, Lee E, Young PP. The Wnt modulator sFRP2 enhances mesenchymal stem cell engraftment, granulation tissue formation and myocardial repair. Proc Natl Acad Sci USA. 2008;105:18366–18371. doi: 10.1073/pnas.0803437105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lorenzon A, Calore M, Poloni G, De Windt LJ, Braghetta P, Rampazzo A. Wnt/β-catenin pathway in arrhythmogenic cardiomyopathy. Oncotarget. 2017;8:60640–60655. doi: 10.18632/oncotarget.17457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Burgers TA, Williams BO. Regulation of Wnt/β-catenin signaling within and from osteocytes. Bone. 2013;54:244–249. doi: 10.1016/j.bone.2013.02.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Duan P, Bonewald LF. The role of the wnt/β-catenin signaling pathway in formation and maintenance of bone and teeth. Int J Biochem Cell Biol. 2016;77:23–29. doi: 10.1016/j.biocel.2016.05.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 23.Skemiene KJ, Liobikas V. Borutaite. Anthocyanins as substrates for mitochondrial complex I- protective effect against heart ischemic injury. FEBS J. 2015;282:963–971. doi: 10.1111/febs.13195. [DOI] [PubMed] [Google Scholar]
- 24.Schurman L, Bagur A, Claus-Hermberg H, Messina OD, Negri AL, Sánchez A, González C, Diehl M, Rey P, Gamba J, et al. Guidelines for the diagnosis, prevention and treatment of osteoporosis, 2012. Medicina (B Aires) 2013;73:55–74. (In Spanish) [PubMed] [Google Scholar]
- 25.Himburg HA, Muramoto GG, Daher P, Meadows SK, Russell JL, Doan P, Chi JT, Salter AB, Lento WE, Reya T, et al. Pleiotrophin regulates the expansion and regeneration of hematopoietic stem cells. Nat Med. 2010;16:475–482. doi: 10.1038/nm.2119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Guo S, Xie Y, Fan JB, Ji F, Wang S, Fei H. A-Melanocyte stimulating hormone attenuates dexamethasone-induced osteoblast damages through activating melanocortin receptor 4-SphK1 signaling. Biochem Biophys Res Commun. 2016;469:281–287. doi: 10.1016/j.bbrc.2015.11.104. [DOI] [PubMed] [Google Scholar]
- 27.Liu H, Bian W, Liu S, Huang K. Selenium protects bone marrow stromal cells against hydrogen peroxide-induced inhibition of osteoblastic differentiation by suppressing oxidative stress and ERK signaling pathway. Biol Trace Elem Res. 2012;150:441–450. doi: 10.1007/s12011-012-9488-4. [DOI] [PubMed] [Google Scholar]
- 28.Yang Y, Su Y, Wang D, Chen Y, Wu T, Li G, Sun X, Cui L. Tanshinol attenuates the deleterious effects of oxidative stress on osteoblastic differentiation via Wnt/FoxO3a signaling. Oxid Med Cell Longev. 2013;2013:351895. doi: 10.1155/2013/351895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Liang D, Xiang L, Yang M, Zhang X, Guo B, Chen Y, Yang L, Cao J. ZnT7 can protect MC3T3-E1 cells from oxidative stress-induced apoptosis via PI3K/Akt and MAPK/ERK signaling pathways. Cell Signal. 2013;25:1126–1135. doi: 10.1016/j.cellsig.2013.02.003. [DOI] [PubMed] [Google Scholar]
- 30.Li XM, Yang Q, Li XB, Cheng Q, Zhang K, Han J, Zhao JN, Liu G, Zhao MG. Estrogen-like neuroprotection of isopsoralen against spinal cord injury through estrogen receptor ERα. Metab Brain Dis. 2017;32:259–265. doi: 10.1007/s11011-016-9913-z. [DOI] [PubMed] [Google Scholar]
- 31.Feng CY, Huang XR, Qi MX, Tang SW, Hu YH, Chen S, Ke FJ. Mitochondrial proteomic analysis of isopsoralen protection against oxidative damage in human lens epithelial cells. Chin J Integr Med. 2012;18:529–533. doi: 10.1007/s11655-012-1144-5. [DOI] [PubMed] [Google Scholar]
- 32.Manolagas SC. Birth and death of bone cells: Basic regulatory mechanisms and implications for the pathogenesis and treatment of osteoporosis. Endocr Rev. 2000;21:115–137. doi: 10.1210/er.21.2.115. [DOI] [PubMed] [Google Scholar]
- 33.Wang J, Li SF, Wang T, Sun CH, Wang L, Huang MJ, Chen J, Zheng SW, Wang N, Zhang YJ, Chen TY. Isopsoralen- mediated suppression of bone marrow adiposity and attenuation of the adipogenic commitment of bone marrow-derived mesenchymal stem cells. Int J Mol Med. 2017;39:527–538. doi: 10.3892/ijmm.2017.2880. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 34.Son Y, Kim S, Chung HT, Pae HO. Reactive oxygen species in the activation of MAP kinases. Methods Enzymol. 2013;528:27–48. doi: 10.1016/B978-0-12-405881-1.00002-1. [DOI] [PubMed] [Google Scholar]
- 35.Andreyev AY, Kushnareva YE, Murphy AN, Starkov AA. Mitochondrial ROS metabolism: 10 years later. Biochemistry (Mosc) 2015;80:517–531. doi: 10.1134/S0006297915050028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Mailloux RJ. Teaching the fundamentals of electron transfer reactions in mitochondria and the production and detection of reactive oxygen species. Redox Biol. 2015;4:381–398. doi: 10.1016/j.redox.2015.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Brand MD. Mitochondrial generation of superoxide and hydrogen peroxide as the source of mitochondrial redox signaling. Free Radic Biol Med. 2016;100:14–31. doi: 10.1016/j.freeradbiomed.2016.04.001. [DOI] [PubMed] [Google Scholar]
- 38.Reczek CR, Chandel NS. ROS-dependent signal transduction. Curr Opin Cell Biol. 2015;33:8–13. doi: 10.1016/j.ceb.2014.09.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Kowaltowski AJ, de Souza-Pinto NC, Castilho RF, Vercesi AE. Mitochondria and reactive oxygen species. Free Radic Biol Med. 2009;47:333–343. doi: 10.1016/j.freeradbiomed.2009.05.004. [DOI] [PubMed] [Google Scholar]
- 40.Herbst RS. Review of epidermal growth factor receptor biology. Int J Radiat Oncol Biol Phys. 2004;59(Suppl 2):S21–S26. doi: 10.1016/j.ijrobp.2003.11.041. [DOI] [PubMed] [Google Scholar]
- 41.Talasila KM, Soentgerath A, Euskirchen P, Rosland GV, Wang J, Huszthy PC, Prestegarden L, Skaftnesmo KO, Sakariassen PØ, Eskilsson E, et al. EGFR wild-type amplification and activation promote invasion and development of glioblastoma independent of angiogenesis. Acta Neuropathol. 2013;125:683–698. doi: 10.1007/s00401-013-1101-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Li J, He W, Liao B, Yang J. FFA-ROS-P53-mediated mitochondrial apoptosis contributes to reduction of osteoblastogenesis and bone mass in type 2 diabetes mellitus. Sci Rep. 2015;5:12724. doi: 10.1038/srep12724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.She C, Zhu LQ, Zhen YF, Wang XD, Dong QR. Activation of AMPK protects against hydrogen peroxide-induced osteoblast apoptosis through autophagy induction and NADPH maintenance: New implications for osteonecrosis treatment? Cell Signal. 2014;26:1–8. doi: 10.1016/j.cellsig.2013.08.046. [DOI] [PubMed] [Google Scholar]
- 44.Krishnan V, Bryant HU, Macdougald OA. Regulation of bone mass by wnt signaling. J Clin Invest. 2006;116:1202–1209. doi: 10.1172/JCI28551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Valenta T, Hausmann G, Basler K. The many faces and functions of β-catenin. EMBO J. 2012;31:2714–2736. doi: 10.1038/emboj.2012.150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Rossini M, Gatti D, Adami S. Involvement of WNT/β-catenin signaling in the treatment of osteoporosis. Calcif Tissue Int. 2013;93:121–132. doi: 10.1007/s00223-013-9749-z. [DOI] [PubMed] [Google Scholar]
- 47.Baron R, Kneissel M. WNT signaling in bone homeostasis and disease: From human mutations to treatments. Nat Med. 2013;19:179–192. doi: 10.1038/nm.3074. [DOI] [PubMed] [Google Scholar]
- 48.Baron R, Rawadi G, Roman-Roman S. Wnt signaling: A key regulator of bone mass. Curr Top Dev Biol. 2006;76:103–127. doi: 10.1016/S0070-2153(06)76004-5. [DOI] [PubMed] [Google Scholar]
- 49.Zhang J, Zhang X, Zhang L, Zhou F, van Dinther M, Ten Dijke P. LRP8 mediates Wnt/beta-catenin signaling and controls osteoblast differentiation. J Bone Miner Res. 2012;27:2065–2074. doi: 10.1002/jbmr.1661. [DOI] [PubMed] [Google Scholar]
- 50.Yu HM, Jerchow B, Sheu TJ, Liu B, Costantini F, Puzas JE, Birchmeier W, Hsu W. The role of Axin2 in calvarial morphogenesis and craniosynostosis. Development. 2005;132:1995–2005. doi: 10.1242/dev.01786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Gaur T, Lengner CJ, Hovhannisyan H, Bhat RA, Bodine PV, Komm BS, Javed A, van Wijnen AJ, Stein JL, Stein GS, Lian JB. Canonical WNT signaling promotes osteogenesis by directly stimulating Runx2 gene expression. J Biol Chem. 2005;280:33132–33140. doi: 10.1074/jbc.M500608200. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.