Abstract
Background
: Recent data suggest that beta blockers are associated with increased perioperative risk in hypertensive patients. We investigated whether beta blockers were associated with an increased risk in elderly patients with raised preoperative arterial blood pressure.
Methods
We conducted a propensity-score-matched cohort study of primary care data from the UK Clinical Practice Research Datalink (2004–13), including 84 633 patients aged 65 yr or over. Conditional logistic regression models, including factors that were significantly associated with the outcome, were constructed for 30-day mortality after elective noncardiac surgery. The effects of beta blockers (primary outcome), renin–angiotensin system (RAS) inhibitors, calcium-channel blockers, thiazides, loop diuretics, and statins were investigated at systolic and diastolic arterial pressure thresholds.
Results
Beta blockers were associated with increased odds of postoperative 30-day mortality in patients with systolic hypertension (defined as systolic BP >140 mm Hg; adjusted odds ratio [aOR]: 1.92; 95% confidence interval [CI]: 1.05–3.51). After excluding patients for whom prior data suggest benefit from perioperative beta blockade (patients with prior myocardial infarction or heart failure), rather than adjusting for them, the point estimate shifted slightly (aOR: 2.06; 95% CI: 1.09–3.89). Compared with no use, statins (aOR: 0.35; 95% CI: 0.17–0.75) and thiazides (aOR: 0.28; 95% CI: 0.10–0.78) were associated with lower mortality in patients with systolic hypertension.
Conclusions
These data suggest that the safety of perioperative beta blockers may be influenced by preoperative blood pressure thresholds. A randomised controlled trial of beta-blocker withdrawal, in select populations, is required to identify a causal relationship.
Keywords: beta blocker, blood pressure, hypertension, surgery, mortality, perioperative outcome, risk factors
Editor's key points.
-
•
The role of preoperative antihypertensive drug therapy as a risk factor for postoperative mortality as a function of preoperative arterial pressure was investigated in a large retrospective data set.
-
•
A propensity-score-matched cohort study of primary care data from the UK Clinical Practice Research Datalink including 84 633 elderly patients 65 yr or over analysed mortality after elective noncardiac surgery.
-
•
Beta blockers were associated with increased risk of 30-day mortality in patients with elevated preoperative BP.
-
•
Renin–angiotensin system inhibitors, calcium-channel blockers, and loop diuretics were not associated with mortality, whilst thiazides and statins were associated with reduced risk.
-
•
Prospective randomised trials are needed to confirm these findings with important implications for perioperative management.
For many years, beta blockers were considered protective medications in the perioperative period; however, recent trials and meta-analyses have challenged this notion.1, 2, 3 Most notably, the Perioperative Ischemic Evaluation Study (POISE) suggested that de novo institution of a high dose of slow-release metoprolol reduces the risk of myocardial infarction (MI), but increases the risk of stroke, sepsis, and mortality.4 Accumulating data from observational studies suggest that beta blockers exert a class effect5 and may be harmful in low-risk patients (defined as low revised cardiac risk index scores6, 7), but beneficial in those with a recent myocardial event or heart failure8 (reviewed by Jorgensen and colleagues3). When excluding high-risk cardiac patients, Jorgensen and colleagues9 found that, amongst hypertensive patients, the use of beta blockers was associated with an increased risk of postoperative major adverse complications. Based on this work, our primary hypothesis was that beta blockers are associated with increased 30-day mortality in patients with numerically high preoperative BP. The working hypothesis is that, in patients with hypertension, the effects on cardiac output, renin–angiotensin system (RAS), and vascular tone by beta blockers in the perioperative period outweigh any direct cardioprotective benefit. As secondary aims, we tested the association of other cardiovascular medications and postoperative 30-day mortality at different BP thresholds defined as hypotension (<80 mm Hg diastolic or <120 mm Hg systolic), normotension (80–89 mm Hg diastolic or 120–139 mm Hg systolic), or hypertension (>90 mm Hg diastolic or >140 mm Hg systolic). There is limited guidance on the impact of other cardiovascular medications in the perioperative period, although RAS inhibitors were suggested to be withheld in some situations.10 It is important to note that, in our recent analysis of these data, hypertensive-level BP values in the elderly were not associated with increased 30-day mortality.11 This a priori planned secondary analysis focuses on whether cardiovascular medications are an important determinant of perioperative outcomes at different BP values in elderly patients undergoing elective noncardiac surgery.
Methods
Data source and study design
The data source for this study was the UK Clinical Practice Research Datalink, which is a primary care database representing about 6% of the country's population. We used medical codes (as listed in the appendix of our original paper11) to identify patients who underwent specific noncardiac surgeries between January 1, 2004 and December 31, 2013. We retained longitudinal data for patients aged 65 yr and over, and who were registered at the general practitioner (GP) practice for >1 yr before their elective noncardiac surgery. This study was approved by the Independent Scientific Advisory Committee (ISAC) for the Medicines and Healthcare Products Regulatory Agency, UK (ISAC protocol number: 11_138A).
Exposure variables
Preoperative cardiovascular medications were the main exposure variables. They included beta blockers (primary outcome) and, as secondary outcomes, statins, calcium-channel blockers, angiotensin-converting-enzyme inhibitors, and angiotensin II receptor blockers referred to as RAS inhibitors, thiazide diuretics, and loop diuretics prescribed within 30 days of surgery (compared with non-users).
Outcome variable
Our outcome variable was perioperative mortality, defined as death occurring within 30 days after noncardiac surgery. In the UK, death certificates issued by the GP are entered directly into the primary care database. Potential biases that may arise from misclassification of death are likely to be non-differential (i.e. similar between both groups: death and survival). We discussed this in more detail in our previous publication.11
Co-variates
We included the following as co-variates in our multivariable models: age, sex, alpha-2 agonists, aspirin, other anti-platelet agents, atrial fibrillation, unstable angina, valvular heart disease, MI, cerebrovascular disease, peripheral vascular disease, chronic pulmonary disease (including asthma), heart failure, diabetes mellitus, renal disease, liver disease, cancer, BMI as a categorical variable (<18.5, 18.5–24.99, 25–29.99, and >30 kg m−2), smoking status, alcohol consumption, and socio-economic status (using the Index of Multiple Deprivation [IMD] 2010 scores in quintiles). We used the surgical risk score to adjust for varying levels of risk posed by the included surgery types. Our surgical procedural risk score was based on a validated surgical risk scale,12, 13 and was included as an ordinal categorical variable ranging from 1 to 5, with 1 as a low-risk procedure and 5 as a high-risk procedure.
Statistical analysis
Based on our prior study,11 we considered the most recent BP measurement before surgery and stratified our study population by BP thresholds as mentioned. We first computed propensity scores14 using multivariable logistic regression models for each of six drugs of interest (statins, beta blockers, calcium-channel blockers, RAS inhibitors, thiazide diuretics, and loop diuretics) using the method of Hirano and Imbens.15 Co-variates that informed the propensity-score derivation models were MI, unstable angina, heart failure, and atrial fibrillation. We generated propensity-score quintiles for each drug of interest, and matched individuals on propensity-score quintile using an interval-matching approach16 with a minimum 1:1 variable matching ratio.
For each BP threshold, we performed conditional logistic regression to investigate the association between preoperative cardiovascular medication and postoperative mortality. We ran unadjusted and adjusted models for each of the six cardiovascular drugs separately comparing the effect of exposure to a given medication to non-exposure. In our adjusted models, we included those co-variates that were statistically significantly (P<0.05) associated with postoperative mortality. Conditional logistic regression models for each of the six drugs had the following co-variates in common and their individual propensity score: age, aspirin, other anti-platelet agents, atrial fibrillation, unstable angina, MI, cerebrovascular disease, peripheral vascular disease, chronic pulmonary disease, diabetes mellitus, renal disease, cancer, BMI, smoking status, and surgical score. Significantly, the associated co-variates included for specific models are presented as follows:
-
(i)
Statins: sex, beta blockers, RAS inhibitors, calcium-channel blockers, loop diuretics, heart failure, alcohol consumption, number of BP measurements, and IMD 2010 scores.
-
(ii)
Beta blockers: sex, statins, RAS inhibitors, calcium-channel blockers, loop diuretics, heart failure, liver disease, alcohol consumption, number of BP measurements, and IMD 2010 scores.
-
(iii)
Calcium-channel blockers: sex, beta blockers, RAS inhibitors, statins, loop diuretics, heart failure, and alcohol consumption.
-
(iv)
RAS inhibitors: sex, statins, beta blockers, calcium-channel blockers, loop diuretics, heart failure, liver disease, alcohol consumption, number of BP measurements, and IMD 2010 scores.
-
(v)
Thiazides: statins, beta blockers, calcium-channel blockers, loop diuretics, RAS inhibitors, liver disease, number of BP measurements, and IMD 2010 scores.
-
(vi)
Loop diuretics: sex, statins, beta blockers, calcium-channel blockers, thiazides, RAS inhibitors, heart failure, liver disease, alcohol consumption, number of BP measurements, and IMD 2010 scores.
Drugs were omitted from the adjusted analysis if the sample size was insufficient. Interaction terms were specified between all drug co-variates included in the multivariable model to account for the impact of drug combinations. All analyses were performed using Stata 15.0 (StataCorp, College Station, TX, USA).
Results
The study population included 84 633 patients aged 65 yr or over who underwent elective noncardiac surgery (Fig. 1). For systolic BP thresholds, there were 7924 hypotensive patients (89.9% were also diastolic hypotensive), 34 531 normotensive patients (36.4% were also diastolic normotensive), and 41 527 hypertensive patients (17.7% were also diastolic hypertensive; Supplementary Fig. S1; Supplementary Table S1). For diastolic BP thresholds, there were 42 821 hypotensive patients, 32 721 normotensive patients, and 8422 hypertensive patients. There were 495 (1.16%) postoperative mortality events recorded (Table 1). Systolic BP was missing for 651 patients, and diastolic BP was missing for 669 patients.
Fig. 1.
Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) diagram. GP, general practitioner.
Table 1.
Demographic and clinical subject characteristics. ∗0 refers to those patients who never received a prescription for any of the cardiovascular drugs of interest examined.
| All (n=84 633) | Survived (n=84 138) | 30-day mortality (n=495) | P-value | |
|---|---|---|---|---|
| Mean age (yr) (standard deviation) | 74.7 (6.9) | 74.7 (6.9) | 81.5 (8.4) | <0.001 |
| Sex | ||||
| Male (%) | 44 349 (52.4) | 44 088 (52.4) | 261 (52.7) | 0.884 |
| Female (%) | 40 284 (47.9) | 40 050 (47.6) | 234 (47.3) | |
| BMI (m kg−2) | ||||
| Underweight (<18.5) | 1528 (1.8) | 1495 (1.8) | 33 (6.7) | <0.001 |
| Normal range (18.5–24.99) | 25 107 (29.7) | 24 914 (29.6) | 193 (39.0) | |
| Overweight (25–29.99) | 31 194 (36.9) | 31 060 (36.9) | 134 (27.1) | |
| Obese (≥30) | 18 992 (22.4) | 18 937 (22.5) | 55 (11.1) | |
| Missing | 7812 (9.2) | 7732 (9.2) | 80 (16.2) | |
| Smoking status | ||||
| Non-smoker | 43 780 (51.7) | 43 557 (51.8) | 223 (45.1) | <0.001 |
| Current smoker | 8371 (9.9) | 8297 (9.9) | 74 (15.0) | |
| Ex-smoker | 31 193 (36.9) | 31 007 (36.9) | 186 (37.6) | |
| Missing | 1289 (1.5) | 1277 (1.5) | 12 (2.4) | |
| Alcohol consumption status | ||||
| Below limit | 38 937 (46.0) | 38 753 (46.1) | 184 (37.2) | <0.001 |
| Above limit | 5858 (6.9) | 5836 (6.9) | 22 (4.4) | |
| Missing | 39 838 (47.1) | 39 549 (47.0) | 289 (58.4) | |
| Comorbidities | ||||
| Atrial fibrillation (%) | 6934 (8.2) | 6845 (8.1) | 89 (18.0) | <0.001 |
| Other cardiac arrhythmia (%) | 48 (0.1) | 48 (0.1) | 0 (0) | 0.595 |
| Unstable angina (%) | 1097 (1.3) | 1086 (1.3) | 11 (2.2) | 0.068 |
| Valvular heart disease (%) | 28 (0.03) | 28 (0.03) | 0 (0) | 0.685 |
| Myocardial infarction (%) | 7671 (9.1) | 7586 (9.0) | 85 (17.2) | <0.001 |
| Congestive heart disease (%) | 3063 (3.6) | 2998 (3.6) | 65 (13.1) | <0.001 |
| Peripheral vascular disease (%) | 5704 (6.7) | 5618 (6.7) | 86 (17.4) | <0.001 |
| Cerebrovascular disease (%) | 5864 (6.9) | 5795 (6.9) | 69 (13.9) | <0.001 |
| Chronic pulmonary disease (%) | 17 023 (20.1) | 16 902 (20.1) | 121 (24.4) | 0.016 |
| Liver disease (%) | 390 (0.5) | 385 (0.5) | 5 (1.0) | 0.070 |
| Diabetes mellitus (%) | 10 690 (12.6) | 10 617 (12.6) | 73 (14.8) | 0.155 |
| Renal disease (%) | 11 421 (13.5) | 11 305 (13.4) | 116 (23.4) | <0.001 |
| Cancer (%) | 20 036 (23.7) | 19 853 (23.6) | 183 (37.0) | <0.001 |
| Statins | 21 617 (25.5) | 21 532 (25.6) | 85 (17.2) | 0.001 |
| Beta blockers | 12 148 (14.4) | 12 078 (14.4) | 70 (14.1) | 0.401 |
| Angiotensin-converting-enzyme inhibitors | 20 888 (24.7) | 20 799 (24.7) | 89 (18) | 0.192 |
| Calcium-channel blockers | 11 786 (13.9) | 11 743 (13.9) | 43 (8.7) | 0.005 |
| Alpha-2 agonists | 1145 (1.4) | 1137 (1.4) | 8 (1.6) | 0.611 |
| Thiazide diuretics | 11 657 (13.8) | 11 627 (13.8) | 30 (6.1) | <0.001 |
| Loop diuretics | 6047 (7.1) | 5977 (7.1) | 70 (14.1) | <0.001 |
| Aspirin | 35 336 (41.8) | 35 052 (41.7) | 284 (57.4) | <0.001 |
| Other anti-platelet drugs | 6941 (8.2) | 6877 (8.2) | 64 (12.9) | <0.001 |
| Number of cardiovascular drugs∗ | ||||
| 0 | 21 870 (25.8) | 21 771 (25.9) | 99 (20) | 0.327 |
| 1 | 15 578 (18.4) | 15 489 (18.4) | 89 (18) | |
| 2 | 13 385 (15.8) | 13 327 (15.8) | 58 (11.7) | |
| 3 | 8773 (10.4) | 8735 (10.4) | 38 (7.7) | |
| 4 | 3200 (3.8) | 3183 (3.8) | 17 (3.4) | |
| 5 | 538 (0.6) | 538 (0.6) | 0 (0) | |
| 6 | 11 (0.01) | 11 (0.01) | 0 (0) | |
| Surgical risk score | ||||
| 1 | 0 (0) | 0 (0) | 0 (0) | <0.001 |
| 2 | 8142 (9.6) | 8114 (9.6) | 28 (5.7) | |
| 3 | 15 033 (17.8) | 14 993 (17.8) | 40 (8.1) | |
| 4 | 8574 (10.1) | 8533 (10.1) | 41 (8.3) | |
| 5 | 52 884 (62.5) | 52 498 (62.4) | 386 (78.0) | |
| Socio-economic status (Index of Multiple Deprivation 2010 quintiles) | ||||
| 1 | 13 153 (15.5) | 13 101 (15.6) | 52 (10.5) | <0.001 |
| 2 | 13 491 (15.9) | 13 409 (15.9) | 82 (16.6) | |
| 3 | 10 104 (11.9) | 10 026 (11.9) | 78 (15.8) | |
| 4 | 7745 (9.15) | 7688 (9.1) | 57 (11.5) | |
| 5 | 5302 (6.3) | 5261 (6.3) | 41 (8.3) | |
| Missing | 34 838 (41.2) | 34 653 (41.2) | 185 (37.4) | |
Of patients who had received cardiovascular drugs, 15 578 (18.4%) had only one prescribed cardiovascular drug, and 21 870 (25.8%) patients did not have any prescriptions for cardiovascular drugs (Table 1). There were 12 148 patients who had received a current prescription for beta blockers. Of these, atenolol 50 mg was the most commonly prescribed beta blocker (29.6% of all beta-blocker prescriptions), followed by atenolol 25 mg (17.3% of all beta-blocker prescriptions) and bisoprolol 2.5 mg (10.5% of all beta-blocker prescriptions).
Primary outcome
The propensity-matched analysis showed that, after adjustment for statistically significant confounders, in patients with systolic hypertension, beta blockers were associated with a statistically significant increase in odds of postoperative mortality (adjusted odds ratio [aOR]: 1.92; 95% confidence interval [CI]: 1.05–3.51; Fig. 2).
Fig. 2.
Forest plots for the impact of preoperative cardiovascular medications on postoperative mortality for different preoperative systolic BP thresholds. Top row shows unadjusted data, and bottom row shows the results after adjusting for confounders. Columns refer to different BP thresholds. ACE, angiotensin-converting enzyme; CI, confidence interval; OR, odds ratio.
Secondary outcomes
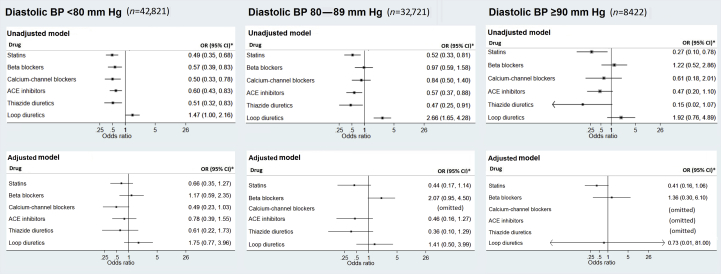
In a secondary analysis, that should be considered at most hypothesis generating, statins were associated with a statistically significant decrease in the adjusted odds of postoperative mortality (aOR: 0.35; 95% CI: 0.17–0.75) in the systolic hypotensive group. For patients with systolic hypertension, statins (aOR: 0.35; 95% CI: 0.17–0.75; Fig. 2) and thiazides (aOR: 0.28; 95% CI: 0.10–0.78; Fig. 2) were associated with a protective effect on postoperative mortality. No significant results were observed based on diastolic BP thresholds (Fig. 3).
Fig. 3.
Forest plots for the impact of preoperative cardiovascular medications on postoperative mortality for different preoperative diastolic BP thresholds. Top row shows unadjusted data, and bottom row shows the results after adjusting for confounders. Columns refer to different BP thresholds. ACE, angiotensin-converting enzyme; CI, confidence interval; OR, odds ratio.
Sensitivity analyses
In 2011, beta blockers were lowered from first-line treatment to third-line treatment for hypertension.17 In order to address any resulting confounding, we performed a post hoc sensitivity analysis in those patients who underwent noncardiac surgery before December 31, 2011. Of 69 686 such patients, 9952 (14.3%) had been prescribed beta blockers. In this population, we found that beta blockers were associated with a two-fold increase in postoperative mortality in the systolic hypertension group (aOR: 2.07; 95% CI: 1.09–3.95).
As confounding by indication could influence the observed associations, and beta blockers are associated with perioperative protection in patients with heart failure and prior MI,8 we conducted a post hoc secondary analysis by excluding patients with heart failure (n=3063) and running our models again. In the remaining 81 570 patients, beta blockers remained statistically significant in systolic hypertension (aOR: 2.13; 95% CI: 1.18–3.85; Table 2). We conducted further analyses excluding patients with prior acute MI (aOR: 1.95; 95% CI: 1.04–3.67), or both heart failure and acute MI (aOR: 2.06; 95% CI: 1.09–3.89). In these groups, significant associations were observed between beta-blocker use and mortality in patients with systolic hypertension (Table 2).
Table 2.
Sensitivity analysis: impact of beta blockers on postoperative mortality at various BP arterial pressure thresholds in patient groups excluding specific disease groups. CI, confidence interval; OR, odds ratio.∗P-value <0.05
| Systolic threshold (mm Hg) | No myocardial infarction |
No heart failure |
No myocardial infarction or heart failure |
|||
|---|---|---|---|---|---|---|
| Unadjusted OR (95% CI) | Adjusted OR (95% CI) | Unadjusted OR (95% CI) | Adjusted OR (95% CI) | Unadjusted OR (95% CI) | Adjusted OR (95% CI) | |
| <120 | 0.79 (0.33–0.89) | 0.28 (0.04–1.84) | 0.60 (0.26–1.43) | 0.28 (0.04–1.84) | 0.61 (0.22–1.73) | 0.28 (0.04–1.84) |
| 120–139 | 1.07 (0.64–1.77) | 1.31 (0.50–3.43) | 1.18 (0.75–1.87) | 1.29 (0.54–3.11) | 1.11 (0.65–1.90) | 1.44 (0.50–4.13) |
| ≥140 | 0.91 (0.57–1.47) | 1.95 (1.04–3.67)∗ | 0.89 (0.57–1.41) | 2.13 (1.18–3.85)∗ | 0.83 (0.50–1.38) | 2.06 (1.09–3.89)∗ |
Discussion
Our main finding confirmed the hypothesis that beta blockers are associated with increased perioperative mortality in patients with raised BP. This hypothesis was based on the recent finding that beta blockers may be harmful in hypertensive patients determined with a different data set.9 The effects of beta blockers on raised systolic BP remained throughout the sensitivity analyses adjusted for year of administration and confounding by indication for heart failure or prior MI.
Before further inference, it is essential to note that, because of the observational nature of this study, the data do not address whether beta-blocker withdrawal (or non-compliance) may be responsible for these findings, as we do not have data on within-hospital administration of beta blockers. This is an important limitation of our data, although beta-blocker continuation has been advocated by guidelines throughout the study period. Furthermore, the exclusion of patients for whom continuation of beta blockers is thought to be critical (high-risk patients with prior MI or heart failure) did not affect the results. Our data support the notion that continuing beta-blocker exposure in the perioperative period may not be advantageous in all patients as we have discussed in a recent review.3 Further epidemiological evidence is required to understand which patients may benefit from beta-blocker withdrawal and which may come to harm. The only way to address these issues definitively will be to conduct an appropriately powered RCT, likely targeting lower-risk patients.
Beta-blocker withdrawal has been associated with adverse outcomes in epidemiological studies,6, 18 although a more recent study provided less clear evidence with both increased mortality and lower morbidity.19 Our data highlight that it is unclear whether the risk/benefit ratio is the same for all levels of patient risk, with patient factors, such as BP, influencing the potential risk, supporting our prior work.8, 9 One hypothesis may be that patients with hypertension are vulnerable to swings in perioperative BP as a result of increased vascular stiffness or suppression of the RAS, combined with higher BP thresholds for organ autoregulation.9 We also hypothesised that they are of low cardiac risk, so to not benefit at a population level from the cardioprotective effects of beta blockers. Hence, the hypothesis is that the risk/benefit ratio of the medications is unclear in this population, although we acknowledge that we are early in evaluating this hypothesis. However, this study validates the prior finding that beta blockers can be associated with harm in patients with hypertension.8 Our approach leveraged the availability of preoperative BP measures that are often used by anaesthesiologists to gauge perioperative risk. We acknowledge that this is slightly discordant with the study by Jorgensen and colleagues,6 in which BP measures were not available. Our results suggest that it is not the diagnosis of hypertension that is most critical, but the actual numerical value of BP, such that an increased risk may not pertain to well-controlled hypertensive patients.
In our secondary analyses, we observed that chronic statins and thiazide diuretics were associated with protective effects against postoperative mortality in elderly patients with systolic hypertension. If these hypothesis-generating findings are confirmed, we suspect that patients on statins may have benefited from improved autoregulation, anti-inflammatory action, and organ protection that statins are thought to afford.20 Whilst observational studies have suggested that statins improve perioperative outcomes,21, 22 the recent Lowering the Risk of Operative Complications Using Atorvastatin Loading Dose RCT did not suggest benefit of de novo institution of statins in the perioperative period (and showed no trend of benefit in elderly patients).23 Hence, the actual benefit of de novo perioperative statin therapy is unproved. The finding that thiazides may be protective is harder to explain, but should not necessarily be dismissed, as we recently observed that thiazides are relatively protective (compared with other antihypertensive medications) in a cohort of Danish patients.9 If real, their protective effect may relate to their small stimulatory effect on the RAS.24
Limitations
Our data suffer from the limitations that affect all observational analyses, particularly that causation cannot be proved. Causality can only be concluded in the setting of experimental studies, such as RCTs. Selection bias and confounding by indication cannot be excluded from influencing these results, hence the importance of specific hypotheses and the conduct of sensitivity analyses (such as excluding patients with heart failure). For example, it is possible that beta blockade in patients without prior MI or heart failure constitutes resistant hypertension. In this context, the risk may be conferred by the underlying pathophysiology, not the drug itself. Given the accumulating data suggesting poor adverse outcomes are associated with beta-blocker exposure in lower-risk populations, an RCT of the safety of beta blockers is warranted. It is important to note that, similar to all observational studies, the analysis is vulnerable to unmeasured confounding, such as from variables on which we lacked data, including comorbidities, such as stable angina or specific subtypes of heart failure. Similar to other perioperative epidemiology studies, we also lack detailed data on perioperative events that influence postoperative mortality, such as non-compliance and withdrawal of medication. Further information is needed from epidemiological data sets about the withdrawal of perioperative medications. Hence, we regard our data as hypothesis generating and requiring confirmation in future epidemiological studies and RCTs.
Conclusions
Beta blockers may be associated with an increased risk of mortality at raised preoperative arterial pressure thresholds in elderly patients undergoing elective noncardiac surgery. Future epidemiological studies and randomised trials should consider analysing outcomes based on preoperative arterial blood pressure thresholds.
Authors' contributions
Research question design: RDS, SV, PRM.
Study analysis plan: RDS, SV, PRM.
Data analysis: SV, PRM, RDS.
Writing paper: RDS, SV, PRM.
Full access to all data in the study, and takes responsibility for the integrity of the data and the accuracy of the data analysis: SV.
All co-authors advised on the analyses. All authors advised on the manuscript content, and contributed to editing and scientific direction. All authors approved the final manuscript.
Declaration of interest
The authors declare that they have no conflicts of interest.
Funding
Association of Anaesthetists of Great Britain and Ireland (WKR0-2012-0076) supported by the National Institute of Academic Anaesthesia, funded a research associate post to conduct this study; National Institutes of Health (K23 AG055700 to RDS).
Handling editor: H.C. Hemmings Jr
Editorial decision: 21 March 2019
Footnotes
This article is accompanied by an editorial: Perioperative beta-adrenergic antagonism: panacea or poison? by Beattie & Yang, Br J Anaesth 2019:123:97–100, doi: 10.1016/j.bja.2019.05.039
Supplementary data to this article can be found online at https://doi.org/10.1016/j.bja.2019.03.042.
Appendix A. Supplementary data
The following are the Supplementary data to this article:
References
- 1.Blessberger H., Kammler J., Domanovits H. Perioperative beta-blockers for preventing surgery-related mortality and morbidity. Cochrane Database Syst Rev. 2014;9:CD004476. doi: 10.1002/14651858.CD004476.pub2. [DOI] [PubMed] [Google Scholar]
- 2.Bouri S., Shun-Shin M.J., Cole G.D., Mayet J., Francis D.P. Meta-analysis of secure randomised controlled trials of beta-blockade to prevent perioperative death in non-cardiac surgery. Heart. 2014;100:456–464. doi: 10.1136/heartjnl-2013-304262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Jorgensen M.E., Andersson C., Venkatesan S., Sanders R.D. Beta-blockers in noncardiac surgery: did observational studies put us back on safe ground? Br J Anaesth. 2018;121:16–25. doi: 10.1016/j.bja.2018.02.004. [DOI] [PubMed] [Google Scholar]
- 4.Devereaux P.J., Yang H., Yusuf S. Effects of extended-release metoprolol succinate in patients undergoing non-cardiac surgery (POISE trial): a randomised controlled trial. Lancet. 2008;371:1839–1847. doi: 10.1016/S0140-6736(08)60601-7. [DOI] [PubMed] [Google Scholar]
- 5.Jorgensen M.E., Sanders R.D., Kober L. Beta-blocker subtype and risks of perioperative adverse events following non-cardiac surgery: a nationwide cohort study. Eur Heart J. 2017;38:2421–2428. doi: 10.1093/eurheartj/ehx214. [DOI] [PubMed] [Google Scholar]
- 6.London M.J., Hur K., Schwartz G.G., Henderson W.G. Association of perioperative beta-blockade with mortality and cardiovascular morbidity following major noncardiac surgery. JAMA. 2013;309:1704–1713. doi: 10.1001/jama.2013.4135. [DOI] [PubMed] [Google Scholar]
- 7.Lindenauer P.K., Pekow P., Wang K., Mamidi D.K., Gutierrez B., Benjamin E.M. Perioperative beta-blocker therapy and mortality after major noncardiac surgery. N Engl J Med. 2005;353:349–361. doi: 10.1056/NEJMoa041895. [DOI] [PubMed] [Google Scholar]
- 8.Andersson C., Shilane D., Go A.S. Beta-blocker therapy and cardiac events among patients with newly diagnosed coronary heart disease. J Am Coll Cardiol. 2014;64:247–252. doi: 10.1016/j.jacc.2014.04.042. [DOI] [PubMed] [Google Scholar]
- 9.Jorgensen M.E., Hlatky M.A., Kober L. Beta-blocker-associated risks in patients with uncomplicated hypertension undergoing noncardiac surgery. JAMA Intern Med. 2015;175:1923–1931. doi: 10.1001/jamainternmed.2015.5346. [DOI] [PubMed] [Google Scholar]
- 10.Kristensen S.D., Knuuti J., Saraste A. 2014 ESC/ESA guidelines on non-cardiac surgery: cardiovascular assessment and management: the Joint Task Force on non-cardiac surgery: cardiovascular assessment and management of the European Society of Cardiology (ESC) and the European Society of Anaesthesiology (ESA) Eur Heart J. 2014;35:2383–2431. doi: 10.1093/eurheartj/ehu282. [DOI] [PubMed] [Google Scholar]
- 11.Venkatesan S., Myles P.R., Manning H.J. Cohort study of preoperative blood pressure and risk of 30-day mortality after elective non-cardiac surgery. Br J Anaesth. 2017;119:65–77. doi: 10.1093/bja/aex056. [DOI] [PubMed] [Google Scholar]
- 12.Brooks M., Sutton R., Sarin S. Comparison of surgical risk score, POSSUM and p-POSSUM in higher-risk surgical patients. Br J Surg. 2005;92:1288–1292. doi: 10.1002/bjs.5058. [DOI] [PubMed] [Google Scholar]
- 13.Sutton R., Bann S., Brooks M., Sarin S. The Surgical Risk Scale as an improved tool for risk-adjusted analysis in comparative surgical audit. Br J Surg. 2009;89:763–768. doi: 10.1046/j.1365-2168.2002.02080.x. [DOI] [PubMed] [Google Scholar]
- 14.Okoli G.N., Sanders R.D., Myles P. Demystifying propensity scores. Br J Anaesth. 2014;112:13–15. doi: 10.1093/bja/aet290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hirano K., Imbens G. The propensity score with continuous treatments. Appl Bayesian Model Causal Inference Incomplete-Data Perspect. 2005:73–84. [Google Scholar]
- 16.Caliendo M., Kopeinig S. Some practical guidance for the implementation of propensity score matching. J Econ Surv. 2008;22:31–72. [Google Scholar]
- 17.Krause T., Lovibond K., Caulfield M., McCormack T., Williams B. Management of hypertension: summary of NICE guidance. BMJ. 2011;343:d4891. doi: 10.1136/bmj.d4891. [DOI] [PubMed] [Google Scholar]
- 18.Wallace A.W., Au S., Cason B.A. Association of the pattern of use of perioperative beta-blockade and postoperative mortality. Anesthesiology. 2010;113:794–805. doi: 10.1097/ALN.0b013e3181f1c061. [DOI] [PubMed] [Google Scholar]
- 19.Kertai M.D., Cooter M., Pollard R.J. Is Compliance with Surgical Care Improvement Project Cardiac (SCIP-Card-2) measures for perioperative beta-blockers associated with reduced incidence of mortality and cardiovascular-related critical quality indicators after noncardiac surgery? Anesth Analg. 2018;126:1829–1838. doi: 10.1213/ANE.0000000000002577. [DOI] [PubMed] [Google Scholar]
- 20.Tseng M.Y., Czosnyka M., Richards H., Pickard J.D., Kirkpatrick P.J. Effects of acute treatment with pravastatin on cerebral vasospasm, autoregulation, and delayed ischemic deficits after aneurysmal subarachnoid hemorrhage: a phase II randomized placebo-controlled trial. Stroke. 2005;36:1627–1632. doi: 10.1161/01.STR.0000176743.67564.5d. [DOI] [PubMed] [Google Scholar]
- 21.Sanders R.D., Nicholson A., Lewis S.R., Smith A.F., Alderson P. Perioperative statin therapy for improving outcomes during and after noncardiac vascular surgery. Cochrane Database Syst Rev. 2013;7:CD009971. doi: 10.1002/14651858.CD009971.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.de Waal B.A., Buise M.P., van Zundert A.A. Perioperative statin therapy in patients at high risk for cardiovascular morbidity undergoing surgery: a review. Br J Anaesth. 2015;114:44–52. doi: 10.1093/bja/aeu295. [DOI] [PubMed] [Google Scholar]
- 23.Berwanger O., de Barros E.S.P.G., Barbosa R.R. Atorvastatin for high-risk statin-naive patients undergoing noncardiac surgery: the lowering the risk of operative Complications using Atorvastatin loading dose (LOAD) randomized trial. Am Heart J. 2017;184:88–96. doi: 10.1016/j.ahj.2016.11.001. [DOI] [PubMed] [Google Scholar]
- 24.Moser M., Feig P.U. Fifty years of thiazide diuretic therapy for hypertension. Arch Intern Med. 2009;169:1851–1856. doi: 10.1001/archinternmed.2009.342. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.