Abstract
Apelin has cardiopulmonary protective properties that promote vasodilation and maintenance of the endothelial barrier. While reductions in apelin have been identified as a contributor to various lung diseases, including pulmonary edema, its role in the effect of air pollutants has not been examined. Thus, in the current study we sought to investigate if apelin is a downstream target of inhaled ozone and if such change in expression is related to altered DNA methylation in the lung. Male, Long-Evans rats were exposed to filtered air or 1.0 ppm ozone for four hours. Ventilation changes were assessed using whole-body plethysmography immediately following exposure, and markers of pulmonary edema and inflammation were assessed in the bronchoaveolar lavage (BAL) fluid. The enzymatic regulators of DNA methylation were measured in the lung, along with methylation and hydroxymethylation of the apelin promoter. Data showed that ozone exposure was associated with increased enhanced pause and protein leakage in the BAL fluid. Ozone exposure reduced DNA cytosine-5-methyltransferase (DNMT) activity and Dnmt3a/b gene expression. Exposure induced upregulation of proliferating cell nuclear antigen, indicative of DNA damage, repair, and maintenance methylation. Increased methylation and reduced hydroxymethylation was measured on the apelin promoter. These epigenetic modifications accompanied ozone-induced reduction of apelin expression and development of pulmonary edema. In conclusion, epigenetic regulation, specifically increased methylation of the apelin promoter downstream of DNA damage, may lead to reductions in protective signaling of the apelinergic system, contributing to the pulmonary edema observed following exposure to oxidant air pollution.
Keywords: apelin, DNA methylation, ozone, pulmonary edema
Introduction
Apelin is a recently identified key hormone involved in the health of various organ systems. Activation of the apelinergic system induces a cascade of effects leading to reduced oxidative stress, inflammation, endoplasmic reticulum stress, and autophagy (Wu et al. 2017). Apelin is widely distributed throughout the body in both humans and rodents, however the highest concentrations are found within the brain, placenta, and lung (O’Carroll et al. 2013). Reduced apelin concentrations have been linked to cardiovascular disease, diabetes, systemic fluid imbalance, activation of the stress response, and importantly, pulmonary disease (O’Carroll et al. 2013).
Reductions in apelin signaling have been noted in patients with pulmonary veno-occlusive disease (Lathen et al. 2014) and high-altitude pulmonary edema (Nickel et al. 2015). Similarly, disruptions in apelin signaling have been found in various rodent models of lung disease (Visser et al. 2010; Chandra et al. 2011; Piairo et al. 2011; Fan et al. 2015). Notably, apelin knockout exposed to hypoxia developed more severe pulmonary hypertension, microvasculature atrophy, and reduced endothelial nitric oxide synthase (eNOS) activation (Chandra et al. 2011). In an acute lung injury model, exogenous apelin administration reduced pulmonary inflammation, pulmonary fluid leakage, and led to improved oxygen saturation following treatment with oleic acid or lipopolysaccharide (Fan et al. 2015). Conversely, blockade of the apelinergic system resulted in more severe lung injury, further suggesting that apelin plays an important role in protection against pulmonary insults. Apelin also appears essential for normal lung development (Piairo et al. 2011); in a 2010 study, treatment with apelin prevented bronchopulmonary dysplasia and inflammation in neonatal rats exposed to hyperoxia (Visser et al. 2010).
Apelin appears to play a key role in maintaining the integrity of the pulmonary vascular endothelium. Apelin is highly expressed in pulmonary vascular endothelial cells (Andersen et al. 2011). Furthermore, the apelin receptor is prominent on both endothelial cells and smooth muscle cells of the lung and the heart (Andersen et al. 2011). Because of the importance of apelin in lung function, it is plausible that apelin may be involved in air pollution-mediated lung injury. Exposure to ozone, a ubiquitous, ambient air pollutant, has been shown to induce robust changes in respiratory function, lung protein leakage and pulmonary gene expression (Ciencewicki et al. 2016; Miller et al. 2016; Henriquez et al. 2017). The relationship between pulmonary edema and ozone exposure is well established in the literature, however the mechanisms behind this phenomenon are not well understood. Reductions in endothelial nitric oxide synthase (eNOS) levels following ozone exposure have been previously identified and were correlated with increased systemic vascular tone in mice (Chuang et al. 2009). Moreover, considerable evidence suggests that ozone exposure may upregulate inducible nitric oxide synthase (iNOS) in the lung (Laskin et al. 1998; Jang et al. 2004; Groves et al. 2013). While iNOS appears important for immune defense, eNOS has been implicated in regulation of vasodilation (Forstermann and Sessa 2012) and thus, may be more important in mediating the permeability changes observed following exposure to ozone. Additionally, ozone may reduce Kruppel-like factor-2 (KLF-2) in the lung (Santana-Rodriguez et al. 2017) and modify tight junctions (Mudway and Kelly 2000). Because apelin signaling may influence the aforementioned pathways (Lin et al. 2010), it is plausible that inhaled exposure to ozone may induce alterations in apelin production.
The purpose of this study was to investigate the effect of inhaled ozone on the regulation of apelin in the lung, where the expression of its receptor is the highest in the rat (Hosoya et al. 2000). The concentration of apelin is primarily regulated at the level of the gene, either through conventional transcription factor activation/repression or by epigenetic mechanisms including DNA methylation and miRNA silencing (Hosoya et al. 2000). Hence, we sought to investigate changes in methylation within the CpG island of apelin following an acute exposure to ozone in the rat. Herein, we report that ozone exposure reduced apelin expression, and increased DNA methylation of the apelin gene; changes that appear to correspond to development of pulmonary edema in male, Long-Evans rats.
Materials and Methods
Experimental animals.
Male, Long-Evans rats (10–12 weeks-of-age) were purchased from Charles River Laboratories (Raleigh, NC). Rats were pair-housed in polycarbonate cages and had ad libitum access to water and food (Purina #5001; Brentwood, MO). Colony rooms were maintained at 21°C, 50% relative humidity under at 12-hour light/dark cycle. All animal procedures were pre-approved by the Institutional Animal Care and Use Committee at the U.S. Environmental Protection Agency.
Generation of ozone and exposure protocol.
Rats were randomized by body weight into groups and were exposed to either filtered air or ozone (1.0 ppm × 4 hours) (n=8/group). A silent arc discharge generator (OREC; Phoenix, AZ) was used to generate ozone from oxygen. Ozone was dispersed into Rochester style “Hinners” chambers, controlled by mass flow controllers (Coastal Instruments, Inc.; Burgaw, NC). Chamber ozone concentrations were monitored throughout the exposure using photometric ozone analyzers (API Model 400, Teledyne Instruments; San Diego, CA). The exposure chambers were maintained at ~23.3°C and 46% relative humidity.
Whole-body plethysmography.
Within 30 minutes of the conclusion of the exposure, rats were placed in whole-body plethysmography (WBP) chambers to acquire ventilatory parameters including inspiratory and expiratory time (Ti and Te), peak inspiratory and expiratory flow (PIF and PEF), breathing frequency (f), tidal volume (TV), minute volume (MV), expiratory flow at 50% of expiratory volume (EF50), and enhanced pause (Penh) using EMKA iox2 software (SCIREQ; Montreal, QC). Rats were allowed a brief adaptation period within the chamber, followed by a 3-minute data collection period.
Necropsy and sample collection.
Approximately one-hour post-exposure, rats were euthanized via barbiturate overdose (pentobarbital sodium, >200mg/kg, i.p.). The left lung was ligated and bronchoalveolar lavage (BAL) was performed on the right lung using warmed Ca2+/Mg2+ free phosphate buffer saline (37°C) equal to 28 mL/kg body weight (total lung capacity) × 0.6 (right lung representing 60% of total lung volume). Three washes using a syringe were performed using the same aliquot of buffer and the lavage was kept on ice until further processing. The number of cells present in the BAL fluid was determined using a Z1 Coulter Counter (Coulter, Inc.; Miami, FL). Cytospin preparations of BAL fluid were stained using Diff-quick (Fischer Scientific; Pittsburgh, PA) and cell differentials were determined. Remaining BAL fluid was centrifuged at 1500 rpm for 10 minutes at 4°C to obtain cell-free supernatant for assessments including: albumin (antibody-based microalbumin assay; Sekisui Diagnostics; Canada), gamma-glutamyltranspeptidase activity (GGT) and lactate dehydrogenase activity (LDH) (Thermo Fisher Diagnostics; Middletown, VA), N-Acetyl-beta-glucosaminidase activity (NAG; Sigma-Aldrich Diagnostics; St. Louis, MO), and total protein (Coomassie Plus protein assay kit, Thermo Fisher Diagnostics; Middletown, VA) using kits modified for use on the Konelab Arena 30 system (Thermo LabSystems; Finland). The ligated left lung lobe was snap frozen in liquid nitrogen and stored at −80°C for subsequent gene and protein analyses.
Pulmonary gene expression.
Total RNA was isolated from left lung lobe using the Direct-Zol RNA MiniPrep kit from Zymo Research (Irvine, CA). The Qubit RNA reagent (Life Technologies; Carlsbad, CA) was used to determine RNA quantity, and cDNA was generated from 1000 ng of RNA isolate using the QScript cDNA Supermix (Quanta Biosciences; Beverly, MA). Primers for selected genes (Table 1) were designed using the National Center for Biotechnology Information database and Integrated DNA Technologies, Inc. (IDT, Inc; Coralville, IA). Quantitative real-time PCR (qRT-PCR) was performed in triplicate using Sybr Green Master Mix (Life Technologies; Carlsbad, CA) on an Applied Biosystems Prism 7900HT Sequence Detection System. Three endogenous control genes were tested across all treatments and the best control was selected based on stability using the BestKeeper excel macro calculation (Pfaffl et al. 2004). The delta delta Ct method was used to determine relative quantification (RQ) of change from air control group for each gene measured.
Table 1.
Primer information for qPCR.
| Gene name | Forward | Reverse |
|---|---|---|
| Rpl13 | GCTGAAGCCTACCAGAAAGT | TCCGTTTCTCCTCCAGAGT |
| Dnmt1 | TTCTGGTACAGCCAGGACTA | CCGGATACAAGATAGGCAGAAC |
| Dnmt3a | ACAGAGAAACCCAAGGTCAAG | GGCTCCCACATGAGATACAAA |
| Dnmt3b | CTGAGATCGCTGCTGACAA | GTGGTACATGGCCTTCCTATAA |
| Tet1 | CCAGAGGATCTTGGTGCTATTA | GGAGGCATAGGAAGGAAGATAG |
| Tet2 | CATAAGCAGGCAGCACAAAC | GTCTGAGGCATCTGCTCTTTAT |
| Tet3 | GGACTTCTGTGCCCACGCCC | TCAGGGTGCAGACCACAGTGC |
| Apln | GGATTCCTGCACATCCATACT | AACACCCAATCCCTCTCAAC |
| Apelin Region 1 | CTGCGCGATCCTCTCTTTC | CCCAGATGCCCTGTCAATAG |
| Apelin Region 2 | GACCGAGTTGCAGCATGAAT | AAAGCCCAAGCGAGCAAG |
| Apelin Region 3 | GCTCACTCTCTCCAGCCA | GTGGCAGCCCTAATGAGC |
Primers were designed using publically available databases including NCBI and Ensembl genome browser and purchased from IDT, Inc. (Coralville, IA). Abbreviations: apelin (Apln) DNA methyltransferase (Dnmt), 60S ribosomal protein L13 (Rpl3), ten-eleven transferase (Tet).
Pulmonary nuclear protein assays.
Nuclear proteins were isolated from the left lung lobe using the EpiQuick Nuclear Extraction Kit II (EpiGentek, Farmingdale, NY). To assess DNA methyltransferase (DNMT) and ten-eleven translocation (TET) enzyme activity, EpiGentek’s EpiQuick DNMT Activity/Inhibition Assay and Epigenase 5mC-Hydroxylase TET Activity/Inhibition Assay kits were used on nuclear protein isolates. DNA damage was measured using the CytoSelect Proliferating Cell Nuclear Antigen (PCNA) ELISA kit (Cell Bioabs, Inc., San Diego, CA). Quantified PCNA levels were normalized to nuclear protein concentrations. The above assays were performed according to the manufacturer’s protocols and were read on a SpectraMax i3 microplate reader (Molecular Devices; Sunnyvale, CA).
Methylated DNA immunoprecipitation (MeDIP)- qPCR.
Isolated DNA from the left lung lobe was first sheared to 200–600 bp fragments using a Bioruptor ultrasonicator device (Diagenode, Inc.; Denville, NJ). Shearing efficacy was tested by gel electrophoresis prior to immunoprecipitation. Fragmented DNA was immunoprecipitated for regions with 5mC or 5hmC using the MagMeDIP kit and the hMeDIP kit for rat, respectively, by Diagenode, Inc. (Denville, NJ) following the manufacturer’s protocols. Targeted primers for the proximal promoter region of apelin were designed from the sequence listed in the Ensembl genome browser and IDT, Inc. (Coralville, IA). Primers were verified to amplify the CpG island of the apelin gene through the UCSC genome In-Silico PCR search and are listed in Table 1. MethylLight qPCR was performed in triplicate using Sybr Green master mix (Life Technologies; Carlsbad, CA) on an Applied Biosystems Prism 7900HT Sequence Detection System. qRT-PCR was run on both input and immunoprecipated samples to determine the % methylation and % hydroxymethylation for each gene region of the lung tissues assessed. An n=6/group was assessed for %5mC due to sample loss.
Statistical analyses.
Group differences in DNMT- and TET-related data and apelin expression were analyzed utilizing two-tailed t-tests. All other data were analyzed by one-tailed t-test. Significance was set at p<0.05. Statistics and graphs were prepared using GraphPad Prism (version 6.07, GraphPad Software, Inc.; San Diego, CA).
Results
Exposure to ozone alters ventilation.
Inhaled ozone is known to alter ventilatory patterns in both humans and various rodent models (e.g., F344, Sprague-Dawley, Wistar, and WKY rat strains) (Stanek et al. 2011; Hatch et al. 2015). However, the effect of an acute ozone exposure on the male Long-Evans rat comparative to other strains is not as well characterized. Acute exposure of male Long-Evans rats to ozone (1.0 ppm × 4 hours) was associated with significant decreases in breathing frequency and significant increases in Ti, PEF, EF50%, and accordingly, Penh (p<0.001 for all; Table 2). Enhanced pause (Penh) is an index of airflow limitation and is a surrogate measure for bronchoconstriction (Hamelmann et al. 1997). Ozone exposure was not associated with significant changes of Te, PIF, or MV.
Table 2.
Effect of ozone on ventilation.
| Air | Ozone | |
|---|---|---|
| Breathing frequency | 252 ± 14.8 | 206 ± 16.2* |
| Inspiratory time (msec) | 106 ± 5.34 | 124 ± 6.56* |
| Expiratory time (msec) | 206 ± 18.3 | 215 ± 24.0 |
| Peak inspiratory flow (mL/s) | 18.9 ± 0.61 | 20.0 ± 1.37 |
| Peak expiratory flow (mL/s) | 12.6 ± 0.75 | 26.4 ± 2.94*** |
| Minute volume (mL/s) | 265 ± 12.1 | 260 ± 25.3 |
| EF50% | 10.2 ± 0.81 | 24.5 ± 3.17*** |
| Enhanced pause | 0.53 ± 0.05 | 4.94 ± 1.02*** |
Data are presented as mean ± SEM (n=8 per group).
p<0.001
p<0.05.
Ozone-induced lung injury and inflammation.
To further characterize ozone’s effects in the Long-Evans rat, we assessed markers of pulmonary injury and inflammatory cell infiltration in the BAL fluid. Ozone exposure resulted in significant increases of albumin and total protein concentrations (2.5 – 3 fold) in the BAL fluid (p<0.001 and p<0.01, respectively; Table 3). These results are consistent with acute lung injury manifesting as significant increases in lung vascular or alveolar epithelial permeability. The degree of lung injury was significantly correlated with the severity of PenH increases (Fig. 1). LDH activity, a marker of airway cell injury, in addition to NAG activity, was also increased in ozone-exposed rats (p<0.05). Exposure did not alter BAL fluid GGT activity.
Table 3.
Effects of ozone on BAL fluid.
| Air | Ozone | |
|---|---|---|
| Albumin (ug/mL) | 17.3 ± 1.70 | 65.8 ± 12.3*** |
| Protein (ug/mL) | 118 ± 7.50 | 329 ± 59.2** |
| LDH (U/L) | 32.4 ± 2.04 | 39.8 ± 1.20* |
| GGT (U/L) | 7.51 ± 0.52 | 7.72 ± 0.37 |
| NAG (U/L) | 3.41 ± 0.37 | 4.61 ± 0.45* |
| Neutrophils / mL | 1,708 ± 782 | 5,545 ± 1,795* |
| Macrophages / mL | 63,281 ± 5,488 | 59,478 ± 6,069 |
| Lymphocytes / mL | 6,109 ± 1,411 | 7,065 ± 1,083 |
| Eosinophils / mL | 1,227 ± 257 | 1,582 ± 431 |
| Epithelial cells / mL | 2,177 ± 472 | 5,304 ± 1,894# |
Data are presented as mean ± SEM (n=8/group).
p<0.001
p<0.01
p<0.05
p<0.07.
Abbreviations: lactate dehydrogenase (LDH), gamma-glutamyltranspeptidase (GGT), N-Acetyl-beta-glucosaminidase (NAG).
Fig. 1.
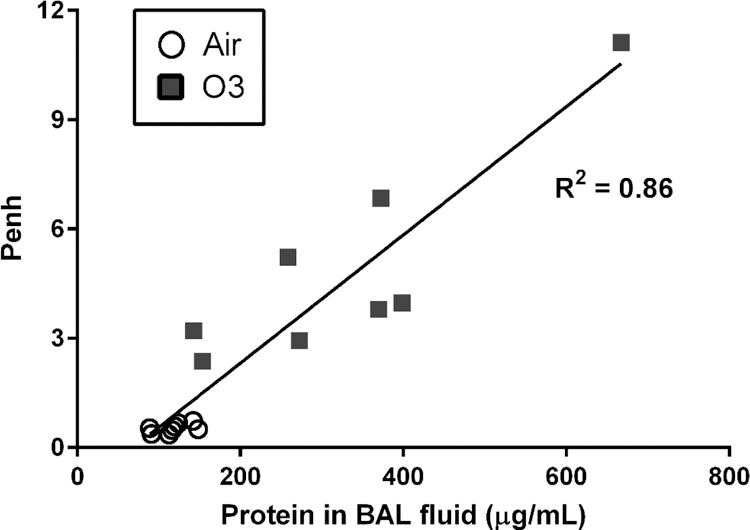
Correlation of BAL fluid protein concentration with Penh in rats exposed to 1.0 ppm ozone. Data are presented as individual samples (n=8/group). Abbreviations: bronchoalveolar lavage (BAL), enhanced pause (Penh).
Cytologic examination of cells recovered in BAL fluid revealed a significant increase in neutrophils following exposure to ozone (p<0.05; Table 3), but did not appear to affect other cell populations except for a trend towards increased epithelial cells (p=0.07), consistent with the minor LDH increase noted above.
Effect of ozone exposure on lung DNA methyltransferase (DNMT) transcripts and activity.
Little information exists on the effects of ozone inhalation on the major regulators of DNA methylation in the lung. DNMTs are commonly recognized as the primary enzymes responsible for methylation of CpG islands on DNA. Hence, in the current study we sought to investigate the effect of ozone exposure on the expression and activity of the DNMTs. Compared to air-exposed controls, transcript levels of both Dnmt3a and Dnmt3b isoforms in the lungs of ozone-exposed rats were reduced (p<0.05; Fig. 2A). The DNMT3 isoforms, both A and B, are generally responsive to environmental changes and mediate de novo methylation (Alvarado et al. 2014). Conversely, expression of Dnmt1, which has a primarily maintenance role in DNA methylation (Kar et al. 2012), was not impacted by ozone exposure (Fig. 2A). Along with downregulated Dnmt3 expression, compared to air-exposed controls, ozone exposure was associated with significant reduction in overall pulmonary DNMT enzyme activity (p<0.05; Fig. 2B).
Fig. 2.
Expression of common regulators of DNA methylation following 1.0 ppm ozone exposure in the left lung lobe. A: Expression of the Dnmts. B: DNMT activity in nuclear protein isolate. C: Expression of the Tets. D: TET activity in the nuclear protein isolate. Data are presented as mean ± SE following normalization to air control (n=7–8/group; n=1 identified outlier removed from DNMT activity from the ozone group). *p<0.05.
Effect of ozone exposure on ten-eleven translocation (TET) enzymes.
The TET enzymes are responsible for demethylation of CpG islands and as such, are important in the process of gene activation. Unlike what was observed in the DNMTs, ozone exposure did not alter Tet1, Tet2, or Tet3 mRNA expression (Fig. 2C). Likewise, the activity of TET enzymes in the lung (Fig. 2D) was not affected by ozone exposure.
Effect of ozone on PCNA concentrations.
It was hypothesized that ozone may induce epigenetic changes, in part, due to oxidative DNA damage. Hence, assessment of DNA repair enzyme concentrations was determined following exposure to ozone. PCNA, a DNA scaffolding protein that interacts with DNMT1 during DNA replication and repair (Mortusewicz et al. 2005), was significantly upregulated in the lung in rats exposed to ozone compared to air-controls (p<0.05; Fig. 3).
Fig. 3.

Quantification of PCNA in the left lung lobe immediately following ozone exposure. Nuclear protein isolate from the lung was assessed for PCNA and was normalized to protein concentration. Data are presented as mean ± SE (n=8/group) following normalization to air control. *p<0.05.
Effect of ozone on Apelin expression in the lung.
Apelin has been identified as a critical hormone that serves to maintain pulmonary vascular permeability (Mishra et al. 2015), a critical function often perturbed by ozone inhalation (Stanek et al. 2011). Therefore, we investigated the regulation of apelin expression in the lung following a single exposure to ozone in the Long-Evans rat. Lung Apln expression was acutely and significantly decreased in ozone-exposed rats compared to air controls (p<0.05; Fig. 4A).
Fig. 4.
Regulation of the apelin gene following ozone exposure. A: Apelin (Apln) gene expression. B: For %5mC (methylation) and %5hmC (hydroxymethylation) quantification on the CpG island in the apelin gene, primer sets were developed that spanned a region within the proximal promoter, transcriptional start site, and exon 1. C: qPCR of the apelin gene following 5mC DNA immunoprecipitation. D: qPCR of the apelin gene following 5hmC DNA immunoprecipitation. Data are presented as mean ± SE (n=7–8/group, n=1 identified outlier removed from %5hmC in region 2; %5mC quantification was assessed in n=6/group). **p<0.01, *p<0.05.
Because apelin has a modifiable CpG island in the promoter region of the gene, we further assessed alterations to the methylation status of apelin. The level of DNA methylation and hydroxymethylation in three areas of the promoter region of apelin were examined in order to better understand regulation of the gene following ozone exposure (Fig. 4B). Whereas exposure to ozone increased the percent methylation on the apelin gene within the proximal promoter (region 1; p<0.01) and in the primer region spanning the transcription start site (region 2; p<0.05) compared to air controls (Fig. 4C), there was no significant increase in CpG methylation in the region solely within exon 1 (region 3). Conversely, ozone exposure reduced the percent hydroxymethylation of the apelin gene within both regions measured in exon 1 (p<0.05; Fig. 4D). However, there was no measurable effect of ozone exposure on hydroxymethylation of the proximal promoter (region 1) of apelin.
Discussion
In the current study, we sought to determine the impact of inhaled ozone on the regulation of the apelin gene, which purportedly plays a role in vascular function including many of the processes affected by ozone exposure. Inhalation of ozone, a ubiquitous oxidant air pollutant, is known to alter ventilation and induce lung injury, edema, and changes in the expression of a large number of genes within the lung tissue (Henriquez et al. 2017). Currently, many people live in areas that exceed the National Ambient Air Quality Standards (NAAQS) for ozone (McCarthy and Lattanzio 2015) and thus, health implications of acute exposures remains highly relevant. Further, gene-specific approaches as used in this study may permit for the identification of novel intervention targets and discovery of possible high-risk populations including those with pertinent genetic polymorphisms, such as apelin.
Having only been discovered in 1998, the role of apelin in the response to air pollutants, to our knowledge, has yet to be explored. In the current study, we are the first to show that the apelinergic system is impacted by an inhaled air pollutant in the lung. Herein, Apln transcription was downregulated acutely following ozone exposure. Therefore, we hypothesized that changes in the regulation of apelin by DNA methylation may be a significant factor in the pulmonary response to ozone exposure. Data showed that in conjunction with reduced expression, methylation of the apelin promoter region was increased, whereas hydroxymethylation of exon 1 of the apelin gene was reduced. Together, the observed patterns in DNA methylation of the apelin gene provides a plausible mechanism to explain the decreased levels of Apln transcript.
Lung apelin expression is negatively associated with both pulmonary hypertension and edema formation (Andersen et al. 2011; Mishra et al. 2015). Apelin is a hormone with metabolic regulatory activity and has a CpG island that spans the promoter region, exon 1, and intron 1 in both humans and rats. Additionally, microRNAs including miRNA-765 (Liao et al. 2015) and miRNA-224 (Wan et al. 2015) regulate apelin processing. The apelinergic systemic is important for the activation of eNOS production, and hence vasodilation (Kim 2014), and the integrity of the endothelial barrier through modulating KLF-2 (Lin et al. 2010). Hence, deficiencies in apelin have been linked to increasing severity of cardiopulmonary diseases (Fan et al. 2015; Gupta et al. 2016). Additionally, it has been recently reported that high-altitude pulmonary edema is positively associated with apelin gene variants as well as increased CpG methylation in humans (Mishra et al. 2015). Collectively, these findings support an epigenetic role in the regulation of apelin expression in the lung that may be impacted by oxidant air pollutants such as ozone.
Based on the aforementioned findings, we propose that the response to inhaled ozone involves dysregulation of the apelin system in the lung and this may be responsible for the decreases in eNOS (Chuang et al. 2009) and KLF-2 (Santana-Rodriguez et al. 2017) observed following exposure to ozone. In addition to the reduction in Apln transcript, ozone-exposed rats had pulmonary edema as determined by the increasing concentration of protein in the BAL fluid that was significantly correlated with Penh. Thus, it could be hypothesized that the downregulation of apelin, observed in rats exposed to ozone, may be important for the loss of endothelial integrity and development of pulmonary edema following exposure to oxidant air pollutants.
Because ozone inhalation appeared to induce epigenetic modification of the apelin promoter, we investigated both the DNMT and TET systems. In healthy Long-Evans rats, ozone exposure resulted in a suppression of DNMT activity and Dnmt3a/b expression, which are the important isoforms that respond to changing cellular and environmental conditions (Alvarado et al. 2014). Conversely, Dnmt1 expression was not impacted by ozone exposure, which is not surprising given the primary role of this isoform in maintenance methylation (Kar et al. 2012). Hence, our data suggests that the DNMT system in the lung is impacted by inhaled ozone. However, it also indicates a lack of coordination between total DNMT activity and apelin methylation, which we hypothesize may be due to DNMT1-related DNA repair activities.
As an oxidant air pollutant, ozone has been reported to have genotoxic properties in a variety of systems including the mouse lung (Bornholdt et al. 2002), A549 alveolar epithelial cells (Cheng et al. 2003), human primary leukocytes (Diaz-Llera et al. 2002), and plants (Tai et al. 2010; Abdelhaliem and Al-Huqai 2016). Furthermore, such DNA strand breaks have been associated with ozone exposure in different populations and cell types in humans (Rojas et al. 2000; Pacini et al. 2003; Tovalin et al. 2006; Palli et al. 2009). Similar to our current work, PCNA has been shown to be upregulated following both acute (Jang et al. 2002; Savov et al. 2004) and chronic ozone exposure (Valacchi et al. 2004). PCNA is a DNA scaffold that binds to proteins involved in DNA replication and repair. Furthermore, PCNA recruits DNMT1 to DNA repair sites (Mortusewicz et al. 2005), where DNMT1 has a higher binding affinity to DNA compared to conditions where PCNA is not present (Iida et al. 2002). In contrast, the DNMT3 isoforms are not recruited to areas with high DNA damage. Hence, we believe that the observed reduction in the DNMT activity is solely reflective of DNMT3 downregulation through gene expression and thus, had little impact on the methylation of apelin. While PCNA has also been shown to be negatively correlated with apelin expression (Kim et al. 2013), further research is necessary to investigate PCNA+DNMT1 recruitment to the apelin promoter following ozone exposure to confirm the relationships observed in this study.
It is important to note that various limitations exist in the current study. We investigated only one time-point acutely following exposure to a relatively high ozone concentration, which was chosen for its known ability to induce a degree of pulmonary damage and epithelial permeability changes. The 1.0 ppm concentration of ozone in this study is several fold higher than the NAAQS. A previous study (Hatch et al. 1994) has demonstrated that ozone exposures of humans during intermittent exercise can result in ozone lung deposition that is 4–5 times higher than that achieved in resting, inactive rodents. As such, we used the 1.0 ppm ozone exposure herein to approximate lung dosimetry of previous 0.2 ppm ozone human clinical studies. Lung oxidative stress, DNA damage and methylation alterations are transient events that can be impacted by susceptibilities including age, sex, underlying disease, and species. We have previously reported differences in the antioxidant response to ozone in between strains of rats (Hatch et al. 2015; Dye et al. 2017). While Long-Evans rats were not included in the aforementioned strain comparisons, the male, Long-Evans rat appears sensitive to the pulmonary damage and epigenetic changes following exposure to ozone. Hence, it is unclear if these results would carry over to less susceptible rodent strains or if lower ozone concentrations were investigated. It may be relevant, however, that epidemiological studies report exposure to warm-season ozone is significantly associated with increased risk of mortality in elderly populations (Di et al. 2017), especially those with congestive heart failure (Zanobetti and Schwartz 2011), and individuals prone to developing cardiogenic pulmonary edema.
Furthermore, the samples used to assess DNA methylation and gene expression in the current study reflect whole tissue alterations. As others have discussed (Mann 2014; Ambati et al. 2016), the use of whole tissue in genomics assessments hinders the interpretation and can be difficult owing to the varying cell populations present that can lead to both under and over-estimation of differences. We observed a significant infiltration of neutrophils in the BAL fluid as a result of ozone exposure which could have impacted our observed changes in gene expression and DNA methylation. However, neutrophils represent a small percentage of the immune cell population in the BAL fluid (~1%) (Patel et al. 2015), making up an even smaller percentage relative to whole tissue. Therefore, the increase in neutrophil recruitment to the lung following ozone exposure is unlikely to be responsible for the changes in the genomic changes observed of this study. However, without the use of single-cell type approaches, it is difficult to state which cell populations in the lung are experiencing the greatest changes in apelin methylation and expression.
Lastly, to date we have not attempted to manipulate apelin levels to further demonstrate that the induction of pulmonary edema caused by ozone inhalation was due to reduced apelin signaling, however, as discussed previously, apelin also has direct cardiac effects. Knockout of apelin in mice is associated with impairment of cardiac contractility and progressive heart failure (Kuba et al. 2007), conditions that both induce secondary pulmonary edema. To our knowledge, apelin in aerosolized form is not yet commercially available, whereas peripheral administration of apelin will likely produce a variety of systemic effects beyond re-establishment of pulmonary apelin signaling. Thus, it is presently difficult to prove with all confidence that ozone-induced pulmonary edema is due to disrupted lung apelin signaling. However, the pre-established importance of apelin in lung health, in addition to our added findings, provides reasoning for continued study.
In summary, results of this study provide initial evidence that ozone exposure results in DNA methylation of the apelin gene and reductions in Apln expression (Fig. 5). The decreased Apln expression in ozone-exposed rats in this study accompanied an increase in pulmonary edema and disrupted ventilatory function. Due to the role of apelin in lung vascular permeability, it may be plausible that re-administration or rescue of apelin signaling may prevent pulmonary edema following exposure to ozone or other lung insults. Lastly, while further study is necessary to understand how ozone inhalation promotes epigenetic modifications, we provide novel evidence that ozone exposure can impact both the DNMT system and promote DNA methylation in a gene-specific manner.
Fig. 5.

Schematic of potential pathways by which ozone inhalation is associated with acute changes in pulmonary epithelial fluid equilibrium.
Acknowledgements
We thank Drs. Brian Chorley, Ian Gilmour, and Mehdi Hazari of the U.S. Environmental Protection Agency for their careful review of this manuscript. We additionally thank Wes Gladwell, National Institute of Environmental Health Sciences, for assistance with the use of the Bioruptor.
Footnotes
Publisher's Disclaimer: Disclaimer
The current study was supported by the U.S. Environmental Protection Agency. The research described in this article has been reviewed by the National Health and Environmental Effects Research Laboratory, U.S. Environmental Protection Agency, and approved for publication. Approval does not signify that the contents necessarily reflect the views and policies of the Agency, nor does the mention of trade names of commercial products constitute endorsement or recommendation for use.
Declaration of Interest
The authors declare no conflict of interest.
References
- Abdelhaliem E, Al-Huqai AA. 2016. Detection of protein and DNA damage induced by elevated carbon dioxide and ozone in Triticum aestivum L. using biomarker and comet assay. Genetics and molecular research : GMR 15(2). [DOI] [PubMed] [Google Scholar]
- Alvarado S, Fernald RD, Storey KB, Szyf M. 2014. The dynamic nature of DNA methylation: a role in response to social and seasonal variation. Integrative and comparative biology 54(1):68–76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ambati S, Yu P, McKinney EC, Kandasamy MK, Hartzell D, Baile CA, Meagher RB. 2016. Adipocyte nuclei captured from VAT and SAT. BMC obesity 3:35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andersen CU, Hilberg O, Mellemkjaer S, Nielsen-Kudsk JE, Simonsen U. 2011. Apelin and pulmonary hypertension. Pulmonary circulation 1(3):334–346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bornholdt J, Dybdahl M, Vogel U, Hansen M, Loft S, Wallin H. 2002. Inhalation of ozone induces DNA strand breaks and inflammation in mice. Mutation research 520(1–2):63–71. [DOI] [PubMed] [Google Scholar]
- Chandra SM, Razavi H, Kim J, Agrawal R, Kundu RK, de Jesus Perez V, Zamanian RT, Quertermous T, Chun HJ. 2011. Disruption of the apelin-APJ system worsens hypoxia-induced pulmonary hypertension. Arteriosclerosis, thrombosis, and vascular biology 31(4):814–820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng TJ, Kao HP, Chan CC, Chang WP. 2003. Effects of ozone on DNA single-strand breaks and 8-oxoguanine formation in A549 cells. Environmental research 93(3):279–284. [DOI] [PubMed] [Google Scholar]
- Chuang GC, Yang Z, Westbrook DG, Pompilius M, Ballinger CA, White CR, Krzywanski DM, Postlethwait EM, Ballinger SW. 2009. Pulmonary ozone exposure induces vascular dysfunction, mitochondrial damage, and atherogenesis. American journal of physiology Lung cellular and molecular physiology 297(2):L209–216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ciencewicki JM, Verhein KC, Gerrish K, McCaw ZR, Li J, Bushel PR, Kleeberger SR. 2016. Effects of mannose-binding lectin on pulmonary gene expression and innate immune inflammatory response to ozone. American journal of physiology Lung cellular and molecular physiology 311(2):L280–291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Di Q, Dai L, Wang Y, Zanobetti A, Choirat C, Schwartz JD, Dominici F. 2017. Association of Short-term Exposure to Air Pollution With Mortality in Older Adults. Jama 318(24):2446–2456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Diaz-Llera S, Gonzalez-Hernandez Y, Prieto-Gonzalez EA, Azoy A. 2002. Genotoxic effect of ozone in human peripheral blood leukocytes. Mutation research 517(1–2):13–20. [DOI] [PubMed] [Google Scholar]
- Dye JA, Gibbs-Flournoy EA, Richards JH, Norwood J, Kraft K, Hatch GE. 2017. Neonatal rat age, sex and strain modify acute antioxidant response to ozone. Inhalation toxicology 29(7):291–303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fan XF, Xue F, Zhang YQ, Xing XP, Liu H, Mao SZ, Kong XX, Gao YQ, Liu SF, Gong YS. 2015. The Apelin-APJ axis is an endogenous counterinjury mechanism in experimental acute lung injury. Chest 147(4):969–978. [DOI] [PubMed] [Google Scholar]
- Forstermann U, Sessa WC. 2012. Nitric oxide synthases: regulation and function. European heart journal 33(7):829–837, 837a-837d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Groves AM, Gow AJ, Massa CB, Hall L, Laskin JD, Laskin DL. 2013. Age-related increases in ozone-induced injury and altered pulmonary mechanics in mice with progressive lung inflammation. American journal of physiology Lung cellular and molecular physiology 305(8):L555–568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gupta MD, Girish MP, Shah D, Rain M, Mehta V, Tyagi S, Trehan V, Pasha Q. 2016. Biochemical and genetic role of apelin in essential hypertension and acute coronary syndrome. International journal of cardiology 223:374–378. [DOI] [PubMed] [Google Scholar]
- Hamelmann E, Schwarze J, Takeda K, Oshiba A, Larsen GL, Irvin CG, Gelfand EW. 1997. Noninvasive measurement of airway responsiveness in allergic mice using barometric plethysmography. American journal of respiratory and critical care medicine 156(3 Pt 1):766–775. [DOI] [PubMed] [Google Scholar]
- Hatch GE, Crissman K, Schmid J, Richards JE, Ward WO, Schladweiler MC, Ledbetter AD, Kodavanti UP. 2015. Strain differences in antioxidants in rat models of cardiovascular disease exposed to ozone. Inhalation toxicology 27 Suppl 1:54–62. [DOI] [PubMed] [Google Scholar]
- Hatch GE, Slade R, Harris LP, McDonnell WF, Devlin RB, Koren HS, Costa DL, McKee J. 1994. Ozone dose and effect in humans and rats. A comparison using oxygen-18 labeling and bronchoalveolar lavage. American journal of respiratory and critical care medicine 150(3):676–683. [DOI] [PubMed] [Google Scholar]
- Henriquez A, House J, Miller DB, Snow SJ, Fisher A, Ren H, Schladweiler MC, Ledbetter AD, Wright F, Kodavanti UP. 2017. Adrenal-derived stress hormones modulate ozone-induced lung injury and inflammation. Toxicology and applied pharmacology 329:249–258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hosoya M, Kawamata Y, Fukusumi S, Fujii R, Habata Y, Hinuma S, Kitada C, Honda S, Kurokawa T, Onda H et al. 2000. Molecular and functional characteristics of APJ. Tissue distribution of mRNA and interaction with the endogenous ligand apelin. The Journal of biological chemistry 275(28):21061–21067. [DOI] [PubMed] [Google Scholar]
- Iida T, Suetake I, Tajima S, Morioka H, Ohta S, Obuse C, Tsurimoto T. 2002. PCNA clamp facilitates action of DNA cytosine methyltransferase 1 on hemimethylated DNA. Genes to cells : devoted to molecular & cellular mechanisms 7(10):997–1007. [DOI] [PubMed] [Google Scholar]
- Jang AS, Choi IS, Koh YI, Park CS, Lee JS. 2002. The relationship between alveolar epithelial proliferation and airway obstruction after ozone exposure. Allergy 57(8):737–740. [DOI] [PubMed] [Google Scholar]
- Jang AS, Choi IS, Lee JU, Park SW, Lee JH, Park CS. 2004. Changes in the expression of NO synthase isoforms after ozone: the effects of allergen exposure. Respiratory research 5:5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kar S, Deb M, Sengupta D, Shilpi A, Parbin S, Torrisani J, Pradhan S, Patra S. 2012. An insight into the various regulatory mechanisms modulating human DNA methyltransferase 1 stability and function. Epigenetics 7(9):994–1007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim J 2014. Apelin-APJ signaling: a potential therapeutic target for pulmonary arterial hypertension. Molecules and cells 37(3):196–201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim J, Kang Y, Kojima Y, Lighthouse JK, Hu X, Aldred MA, McLean DL, Park H, Comhair SA, Greif DM et al. 2013. An endothelial apelin-FGF link mediated by miR-424 and miR-503 is disrupted in pulmonary arterial hypertension. Nature medicine 19(1):74–82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuba K, Zhang L, Imai Y, Arab S, Chen M, Maekawa Y, Leschnik M, Leibbrandt A, Markovic M, Schwaighofer J et al. 2007. Impaired heart contractility in Apelin gene-deficient mice associated with aging and pressure overload. Circulation research 101(4):e32–42. [DOI] [PubMed] [Google Scholar]
- Laskin DL, Sunil V, Guo Y, Heck DE, Laskin JD. 1998. Increased nitric oxide synthase in the lung after ozone inhalation is associated with activation of NF-kappa B. Environmental health perspectives 106 Suppl 5:1175–1178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lathen C, Zhang Y, Chow J, Singh M, Lin G, Nigam V, Ashraf YA, Yuan JX, Robbins IM, Thistlethwaite PA. 2014. ERG-APLNR axis controls pulmonary venule endothelial proliferation in pulmonary veno-occlusive disease. Circulation 130(14):1179–1191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liao YC, Wang YS, Hsi E, Chang MH, You YZ, Juo SH. 2015. MicroRNA-765 influences arterial stiffness through modulating apelin expression. Molecular and cellular endocrinology 411:11–19. [DOI] [PubMed] [Google Scholar]
- Lin Z, Natesan V, Shi H, Dong F, Kawanami D, Mahabeleshwar GH, Atkins GB, Nayak L, Cui Y, Finigan JH et al. 2010. Kruppel-like factor 2 regulates endothelial barrier function. Arteriosclerosis, thrombosis, and vascular biology 30(10):1952–1959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mann DA. 2014. Epigenetics in liver disease. Hepatology 60(4):1418–1425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McCarthy JE, Lattanzio RK. 2015. Ozone Air Quality Standards: EPA’s 2015 Revision
- Miller DB, Snow SJ, Schladweiler MC, Richards JE, Ghio AJ, Ledbetter AD, Kodavanti UP. 2016. Acute Ozone-Induced Pulmonary and Systemic Metabolic Effects Are Diminished in Adrenalectomized Rats. Toxicological sciences : an official journal of the Society of Toxicology 150(2):312–322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mishra A, Kohli S, Dua S, Thinlas T, Mohammad G, Pasha MA. 2015. Genetic differences and aberrant methylation in the apelin system predict the risk of high-altitude pulmonary edema. Proceedings of the National Academy of Sciences of the United States of America 112(19):6134–6139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mortusewicz O, Schermelleh L, Walter J, Cardoso MC, Leonhardt H. 2005. Recruitment of DNA methyltransferase I to DNA repair sites. Proceedings of the National Academy of Sciences of the United States of America 102(25):8905–8909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mudway IS, Kelly FJ. 2000. Ozone and the lung: a sensitive issue. Molecular aspects of medicine 21(1–2):1–48. [DOI] [PubMed] [Google Scholar]
- Nickel NP, Spiekerkoetter E, Gu M, Li CG, Li H, Kaschwich M, Diebold I, Hennigs JK, Kim KY, Miyagawa K et al. 2015. Elafin Reverses Pulmonary Hypertension via Caveolin-1-Dependent Bone Morphogenetic Protein Signaling. American journal of respiratory and critical care medicine 191(11):1273–1286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O’Carroll AM, Lolait SJ, Harris LE, Pope GR. 2013. The apelin receptor APJ: journey from an orphan to a multifaceted regulator of homeostasis. The Journal of endocrinology 219(1):R13–35. [DOI] [PubMed] [Google Scholar]
- Pacini S, Giovannelli L, Gulisano M, Peruzzi B, Polli G, Boddi V, Ruggiero M, Bozzo C, Stomeo F, Fenu G et al. 2003. Association between atmospheric ozone levels and damage to human nasal mucosa in Florence, Italy. Environmental and molecular mutagenesis 42(3):127–135. [DOI] [PubMed] [Google Scholar]
- Palli D, Sera F, Giovannelli L, Masala G, Grechi D, Bendinelli B, Caini S, Dolara P, Saieva C. 2009. Environmental ozone exposure and oxidative DNA damage in adult residents of Florence, Italy. Environmental pollution 157(5):1521–1525. [DOI] [PubMed] [Google Scholar]
- Patel BV, Tatham KC, Wilson MR, O’Dea KP, Takata M. 2015. In vivo compartmental analysis of leukocytes in mouse lungs. American journal of physiology Lung cellular and molecular physiology 309(7):L639–652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pfaffl MW, Tichopad A, Prgomet C, Neuvians TP. 2004. Determination of stable housekeeping genes, differentially regulated target genes and sample integrity: BestKeeper--Excel-based tool using pair-wise correlations. Biotechnology letters 26(6):509–515. [DOI] [PubMed] [Google Scholar]
- Piairo P, Moura RS, Nogueira-Silva C, Correia-Pinto J. 2011. The apelinergic system in the developing lung: expression and signaling. Peptides 32(12):2474–2483. [DOI] [PubMed] [Google Scholar]
- Rojas E, Valverde M, Lopez MC, Naufal I, Sanchez I, Bizarro P, Lopez I, Fortoul TI, Ostrosky-Wegman P. 2000. Evaluation of DNA damage in exfoliated tear duct epithelial cells from individuals exposed to air pollution assessed by single cell gel electrophoresis assay. Mutation research 468(1):11–17. [DOI] [PubMed] [Google Scholar]
- Santana-Rodriguez N, Llontop P, Clavo B, Fiuza-Perez MD, Zerecero K, Ayub A, Alshehri K, Yordi NA, Re L, Raad W et al. 2017. Ozone Therapy Protects Against Rejection in a Lung Transplantation Model: A New Treatment? The Annals of thoracic surgery 104(2):458–464. [DOI] [PubMed] [Google Scholar]
- Savov JD, Whitehead GS, Wang J, Liao G, Usuka J, Peltz G, Foster WM, Schwartz DA. 2004. Ozone-induced acute pulmonary injury in inbred mouse strains. American journal of respiratory cell and molecular biology 31(1):69–77. [DOI] [PubMed] [Google Scholar]
- Stanek LW, Brown JS, Stanek J, Gift J, Costa DL. 2011. Air pollution toxicology--a brief review of the role of the science in shaping the current understanding of air pollution health risks. Toxicological sciences : an official journal of the Society of Toxicology 120 Suppl 1:S8–27. [DOI] [PubMed] [Google Scholar]
- Tai HH, Percy KE, Karnosky DF. 2010. DNA damage in Populus tremuloides clones exposed to elevated O3. Environmental pollution 158(4):969–976. [DOI] [PubMed] [Google Scholar]
- Tovalin H, Valverde M, Morandi MT, Blanco S, Whitehead L, Rojas E. 2006. DNA damage in outdoor workers occupationally exposed to environmental air pollutants. Occupational and environmental medicine 63(4):230–236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Valacchi G, Pagnin E, Corbacho AM, Olano E, Davis PA, Packer L, Cross CE. 2004. In vivo ozone exposure induces antioxidant/stress-related responses in murine lung and skin. Free radical biology & medicine 36(5):673–681. [DOI] [PubMed] [Google Scholar]
- Visser YP, Walther FJ, Laghmani el H, Laarse A, Wagenaar GT. 2010. Apelin attenuates hyperoxic lung and heart injury in neonatal rats. American journal of respiratory and critical care medicine 182(10):1239–1250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wan Y, Zeng ZC, Xi M, Wan S, Hua W, Liu YL, Zhou YL, Luo HW, Jiang FN, Zhong WD. 2015. Dysregulated microRNA-224/apelin axis associated with aggressive progression and poor prognosis in patients with prostate cancer. Human pathology 46(2):295–303. [DOI] [PubMed] [Google Scholar]
- Wu Y, Wang X, Zhou X, Cheng B, Li G, Bai B. 2017. Temporal Expression of Apelin/Apelin Receptor in Ischemic Stroke and its Therapeutic Potential. Frontiers in molecular neuroscience 10:1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zanobetti A, Schwartz J. 2011. Ozone and survival in four cohorts with potentially predisposing diseases. American journal of respiratory and critical care medicine 184(7):836–841. [DOI] [PMC free article] [PubMed] [Google Scholar]




