Abstract
Introduction:
Bioimpedance spectroscopy (BIS) devices utilize biophysical modeling to generate body composition data. The addition of body mass index (BMI) to modified Xitron-Hanai-based mixture equations improved BIS estimates of intracellular water (ICW), particularly at the extremes of BMI. A 3-compartment model for distinguishing excess fluid (ExF) from normally hydrated lean (NHLT) and adipose tissue may further improve BIS estimates.
Objective:
We aimed to validate a BIS approach based on the Chamney model for determining fat mass (FM) in healthy individuals (NHANES) and for measuring FM changes in individuals undergoing massive weight loss.
Methods:
Using adult NHANES 1999–2004 (2821 female, 3063 male) and longitudinal pre-to-post-RYGB (15F) data, we compared dual-energy-X-ray absorptiometry (DXA) and BIS for FM. We applied BIS adiposity-corrected values to Chamney equations for normally hydrated lean and adipose tissue (NHLT, NHAT) and FM. Method agreement was evaluated by correlations, paired t-tests, root mean square error (RMSE), Bland-Altman (B-A) analysis, and concordance correlation coefficients (CCC).
Results:
Method agreement between BIS and DXAFM was good in healthy adults (r=0.96, CCC=0.93, p<.0001), and pre-to-post-RYGB (r=0.93–0.98, CCC=0.81–0.86, p<.001). Although cross-sectional FM measures differed, FM change measures post-RYGB did not (35.6±8.9 vs. 35.2±9.2 kg, BIS vs. DXA) and agreed well (r=0.84, p<.0001). The 15 subjects with follow-up measurements at 1 year lost 11.5±9.8 kg FFM by DXA, but only 1.3±2.5 kg of NHLT by BIS, suggesting that the FFM loss may have been mostly adipose tissue water.
Conclusions:
Incorporation of the Chamney model into BIS algorithms is a major conceptual advancement for assessing and monitoring body composition. Its ability to differentiate ICW and extracellular water (ECW) in NHLT and NHAT, as well as excess ECW is promising, and would facilitate lean tissue monitoring in obesity and acute/chronic disease.
Keywords: Bioelectrical Impedance, Obesity, Body Composition, Bariatric Surgery, Weight Loss, Nutrition Surveys
Abstract
Introdução:
Os dispositivos de espectroscopia de bioimpedância (DEB) utilizam modelagem biofísica para gerar dados de composição corporal. A adição do índice de massa corporal (IMC) às equações de mistura modificadas com Xitron-Hanai modificadas melhorou as estimativas de DEB de água intracelular (AI), particularmente nos casos extremos do IMC. Um modelo de 3 compartimentos para distinguir o excesso de fluido (ExF) de magro normalmente hidratado (NHLT) e tecido adiposo pode ainda melhorar as estimativas do DEB.
Objetivo:
Pretendemos validar uma abordagem do DEB com base no modelo de Chamney para determinar a massa de gordura (MG) em indivíduos saudáveis (NHANES) e para medir mudanças de MG em indivíduos submetidos à perda de peso maciça.
Método:
Usando o NHANES adulto 1999–2004 (2821 mulheres, 3063 homens) e dados longitudinais pré-pós-RYGB (15 F), comparamos a absorção de raios-X de dupla energia (DXA) e DEB para MG. Aplicamos os valores corrigidos de adiposidade do BIS às equações de Chamney para tecidos magros e adiposos normalmente hidratados (NHLT, NHAT) e FM. O acordo de método foi avaliado por correlações, testes t pareados, erro quadrado médio (EQM), análise Bland-Altman (B-A) e coeficientes de correlação de concordância (CCC).
Resultados:
O acordo de método entre DEB e DXA MG foi bom em adultos saudáveis (r=0,96, CCC=0,93, p<.0001) e pré-pós-RYGB (r=0,93–0,98, CCC=0,81–0,86, p<0,001). Embora as medidas de MG transversais diferissem, as medidas de mudança de MG pós-RYGB não (35,6±8,9 vs. 35,2±9,2 kg, DEBvs. DXA) e concordaram bem (r=0,84, p<.0001). Os 15 sujeitos com medidas de seguimento ao 1 ano perderam 11,5±9,8 kg FFM por DXA, mas apenas 1,3±2,5 kg de NHLT pelo DEB, sugerindo que a perda de FFM pode ter sido principalmente água do tecido adiposo.
Conclusões:
A incorporação do modelo de Chamney em algoritmos DEB é um grande avanço conceitual para avaliar e monitorar a composição corporal. A sua capacidade de diferenciar AI e água extracelular (AE) no NHLT e NHAT, bem como o excesso de AE é promissor e facilitará a monitorização do tecido magro na obesidade e doença aguda/crônica.
Keywords: Impedância Bioelétrica, Obesidade, Composição Corporal, Cirurgia Bariátrica, Perda de Peso, Inquéritos Nutricionais
INTRODUCTION
Bioimpedance spectroscopy (BIS) is a non-invasive body composition (BC) assessment technique with high potential for clinical applicability. BIS differs from single- and multi-frequency bioimpedance in that biophysical modeling algorithms are applied to bioimpedance data measured across a spectrum of frequencies in order to generate BC data1. In 1992, Xitron Technologies introduced the first commercial BIS device that also incorporated the Cole mathematical equivalent electrical circuit model of biological tissue, and equations based on Hanai’s mixture theory to estimate whole body extracellular (ECW) and intracellular (ICW) volumes in humans.
The BIS data are fit to the Cole model using nonlinear least squares curve fitting. Cole model terms can then be applied to the Xitron-Hanai-based2 mixture (XHM) equations to determine ECW and ICW. Adipocyte resistivity is greater than skeletal muscle cell resistivity and errors in ICW have been shown to increase with increasing adiposity3,4. To account for this, Moissl et al.5 independently predicted ECW and ICW using BMI as a surrogate of adiposity and substantially improved BIS estimates, particularly for ICW. Using the Moissl equations, ICW resistivity is not assumed to be static as in the XHM equations, but rather changes with increasing fat5. Although an imperfect measure of adiposity, their BMI correction yielded a reduction in ICW error by 24% in all subjects, and 48% in those at BMI extremes (defined as BMI<20 kg/m2 and >30 kg/m2). Increasing adiposity substantially increases the intracellular resistivity term and thus causes errors in the ICW estimates, whereas there is little impact on ECW estimates1.
The 3-compartment model developed by Chamney et al.6 was designed to control for excess fluid in assessment of body composition and is based on data from 104 healthy adults. The model utilizes the improved ECW and ICW estimates to distinguish excess fluid (ExF) from normally hydrated lean (NHLT) and normally hydrated adipose tissue (NHAT). As described, NHAT consists of stored lipids, water, some protein, and minerals, and like lean tissue can be impacted by hydration status6.
Recent applications of this model to dialysis populations to monitor fluid status in normal weight and overweight individuals suggest that it may also have utility for monitoring changes in lean tissue and fat mass (FM) in individuals with abnormal fluid status, including in individuals with severe obesity7–9.
Other clinical populations could benefit from this technology. The clinical assessment of NHLT would allow for more specific evaluation of metabolically active lean tissue; and assessing ExF would allow for the determination of abnormal fluid that can accumulate in both adipose and lean tissue compartments with acute and chronic disease. An accurate measurement of FM at the bedside would also be highly advantageous to the clinician interested in monitoring individuals with obesity undergoing weight loss interventions.
Dual energy X-ray absorptiometry (DXA) is frequently utilized as a reference method for comparison with BIS, particularly for FM, FFM, and non-bone lean soft tissue (LST) measures. As described by Chamney et al.6, measures of LST from DXA and NHLT from BIS are not equivalent and we expect the models to produce differences between these values. Both methods result in measurements of FM that are theoretically equivalent physiological compartments. DXA can therefore be used for comparison to the BIS generated NHLT compartment indirectly through the comparison of FM measures. Both DXA and BIS methods are frequently used in clinical settings and it is unclear how each compare for measurements of various body compartments.
We aimed to compare this BIS approach based on the Chamney model with DXA for determining FM in healthy individuals from the continuous National Health and Nutrition Examination Survey (NHANES). We also aimed to apply the model to measure FM changes in individuals undergoing massive weight loss following Roux-en-Y gastric bypass surgery (RYGB).
METHODS
NHANES 1999–2004 Dataset
NHANES is a cross-sectional, population-based study that collects data from approximately 10,000 non-institutionalized U.S. civilians in two-year blocks. The samples are collected using a complex, multi-stage, clustered survey design that oversamples subgroups and populations of public health interest. Relevant files from cross-sectional datasets containing height, weight, DXA and BIS data from the NHANES years 1999–2000, 2001–2002, and 2003–2004 were combined for analysis.
Whole body DXA scans were acquired with a Hologic QDR-4500A fan-beam densitometer (Hologic, Inc., Bedford, Massachusetts) in mobile examination clinics. DXA data included total FM (DXAFM), total lean body mass with (DXAFFM) and without bone (DXALST). Imputed values for DXA were not included in this analysis. BIS data were collected with a HYDRA ECW/ICW Bio-Impedance Spectrum Analyzer (Hydra Model 4200, Xitron Technologies, Inc., San Diego, California, USA). Individuals were excluded from BIS measurements if they were pregnant, had any non-finger or non-toe amputations, had artificial joints, pins, plates or metal in their bodies, had a pacemaker or automatic defibrillator, had coronary stents or metal suture material in the heart, or weighed over 300 lbs10.
BIS data, including resistance related to ECW (RE) and ICW (RI) estimated from Cole-modeling, were only available for individuals aged between 8 and 49 years, thus we restricted our analysis to adults 18 years and older in this age range. BIS data recorded as having excellent, good, or marginal quality of fit to the Cole model were included in the analysis, while those with questionable, bad, or ‘could not be fit’ were excluded (~3% of initial BIS sample). Poor results for quality of fit to the Cole model typically suggest poor cell membrane integrity, advanced disease conditions, or error in collection methods2.
Longitudinal Dataset
For a longitudinal comparison of methods during weight loss, we separately collected BIS and DXA data from women with obesity before (n=25), 6-months (n=16) and 1-year (n=15) following RYGB as part of a 2005–2009 study at the University of Minnesota-Twin Cities (UM). Partial BC data from this study have been previously described4,11. Ethical approval was obtained from the UM Institutional Review Board and the General Clinical Research Center (GCRC).
Subjects provided written, informed consent before participating. Whole body BIS (Hydra Model 4200, Xitron Technologies, Inc., San Diego, California, USA) measurements were obtained as previously described4,11. We previously reported BIS precision with the Hydra device; inter-day, inter-observer coefficients of variation (CV) for ECW and ICW measures with electrode repositioning were 1.28% and 1.72%, respectively12. Anthropometric measurements were completed following a standard protocol11. Half-body thick-mode DXA scans were performed on the same instrument (GE Lunar Prodigy; GE Medical Systems, Madison, WI, software version 8.8) for each study visit by an experienced technician at the UM GCRC. The precision of the half-body protocol was CV of 5% or less based upon 3 repeat scans with repositioning at baseline, as described previously11.
Calculation of BMI-Corrected ECW and ICW Values and Chamney-Model Values
Table 1 summarizes the equations used throughout this analysis. BMI-corrected XHM equations developed by Moissl et al.5 were used to calculate ECWMoissl and ICWMoissl estimates (Table 1, equations 1–3). The ECWMoissl and ICWMoissl values were converted to mass (0.993 kg/L at 37°C) and applied to the Chamney model equations to generate ExF, NHLT, NHAT, and FMChamney (Table 1, equations 4–7)6.
Table 1.
– Model Equations for Calculating Body Composition by BIS.
| Equation | Equation Number |
|---|---|
| 1 | |
| 2 | |
| 3 | |
| ExF =1.136 × ECWMoissl – 0.430 × ICWMoissl – 0.114 × Weight | 4 |
| NHLT = 2.725 × ICWMoissl + 0.191 × ExF – 0.191 × Weight | 5 |
| NHAT = Weight – ExF − NHLT | 6 |
| FMChamney = 0.753 × NHAT | 7 |
Abbreviations: BIS, Bioimpedance spectroscopy; ECW, extracellular water; ICW, intracellular water. kECW and kICW are functions of BMI based on height, weight and resistance; a = 0.188, b = 0.2883, c = 5.8758 and d = 0.4194. Chamney model compartments: ExF, extracellular fluid; NHLT, normally-hydrated lean tissue; NHAT, normally-hydrated adipose tissue; FMChamney, Chamney calculated fat mass.
Statistical Analysis
Inter-method agreement for FM by BIS and DXA is reported as mean±SD, compared between genders, and evaluated using a variety of comparison metrics including linear regression, Pearson’s correlation (r), the coefficient of determination (r2), and concordance correlation coefficients (CCC, epiR R package)13. CCC indicates the strength of the relationship between two methods and quantifies agreement by combining measures of precision and accuracy.
To further investigate the method agreement we used Bland-Altman (B-A) analysis and evaluated bias and limits of agreement (LOA, calculated as 1.96xSD). B-A percent error (PE) was calculated by dividing the LOA for the inter-method difference by the mean DXAFM. B-A PE <30% was deemed acceptable agreement between methods.
Significant r2 between the difference and average between methods indicated proportional bias. Only r2 values > 0.10 with P<0.01 were considered to indicate proportional bias. Root mean square error (RMSE) and percent RMSE (%RMSE) whereby %RMSE=(RMSE/(mean DXAFM))x100 were computed. %RMSE<15% was deemed acceptable error. Paired t-tests compared means (p<0.05=significant). Analysis was completed using R Studio (version 1.0.143; R version 3.3.2) and SAS (version 9.4 of the SAS System; SAS Institute Inc., Cary, NC, USA).
RESULTS
NHANES Dataset
From the NHANES 1999–2004 datasets, DXA and BIS data were available for 5884 (3063 male, 2821 female) participants. The racial distribution for the sample was 33% Mexican American/Other Hispanic, 40% non-Hispanic white, 22% non-Hispanic black, 4% other/multiracial). Age, weight, height, BMI and BIS Cole model values for RE and RI differed by gender (Table 2). Tissue compartment weights determined by each method also differed by gender, and as expected in a healthy population, calculated ExF was negligible (Figure 1C, Table 2).
Table 2.
– Descriptive Statistics and Composition Data for the NHANES Dataset by Gender.
| All (n=5884) |
Male (n=3063) |
Female (n=2821) |
P-value | |
|---|---|---|---|---|
| Age, years | 31.2 ± 10.1 | 30.8 ± 10.0 | 31.6 ± 10.2 | 0.007 |
| Weight, kg | 76.1 ± 17.0 | 80.8 ± 15.9 | 71.1 ± 16.6 | <0.0001 |
| Height, cm | 168.7 ± 9.7 | 174.9 ± 7.6 | 161.9 ± 6.8 | <0.0001 |
| BMI, kg/m2 | 26.7 ± 5.4 | 26.4 ± 4.7 | 27.1 ± 6.0 | <0.0001 |
| DXA | ||||
| DXALST, kg | 49.9 ± 11.3 | 57.6 ± 8.7 | 41.5 ± 6.9 | <0.0001 |
| DXAFFM, kg | 52.3 ± 11.6 | 60.3 ± 9.0 | 43.7 ± 7.1 | <0.0001 |
| DXAFM, kg | 24.6 ± 10.3 | 21.3 ± 8.7 | 28.1 ± 10.7 | <0.0001 |
| BIS | ||||
| RE, Ω | 645 ± 96 | 597 ± 74 | 697 ± 91 | <0.0001 |
| RI, Ω | 1281 ± 322 | 1087±206 | 1491 ±292 | <0.0001 |
| ECWMoissl, L | 15.8 ± 3.2 | 17.7 ± 2.7 | 13.8 ± 2.3 | <0.0001 |
| ICWMoissl, L | 22.3 ± 5.1 | 26.1 ± 3.7 | 18.2 ± 2.7 | <0.0001 |
| Chamney model | ||||
| ExF, kg | −0.3 ± 1.0 | −0.3 ± 1.1 | −0.3 ± 0.9 | 0.003 |
| NHLT, kg | 46.2 ± 12.3 | 55.6 ± 8.5 | 35.9 ± 5.7 | <0.0001 |
| NHAT, kg | 30.2 ± 16.3 | 25.5 ± 14.6 | 35.4 ± 16.4 | <0.0001 |
| FMChamney, kg | 22.8 ± 12.3 | 19.2 ± 11.0 | 26.7 ± 12.4 | <0.0001 |
Abbreviations: BMI, body mass index; BIS, bioimpedance spectroscopy; RE and RI are Cole model terms: RE, resistance of extracellular water; RI, resistance of intracellular water; ECW, extra cellular water; ICW, intracellular water; Chamney model compartments (derived by applying bioimpedance spectroscopy (BIS)-generated BMI-corrected ECWMoissl and ICWMoissl to Chamney model equations): ExF, extracellular fluid; NHLT, normally-hydrated lean tissue; NHAT, normally-hydrated adipose tissue; FM, fat mass; DXA, dual energy X-ray absorptiometry; LST, lean soft tissue; RYGB, Roux-en-Y gastric bypass. Sample values are mean ± SD (range); male and female values are mean ± SD.
P-values are comparison between males and females by student’s t-test.
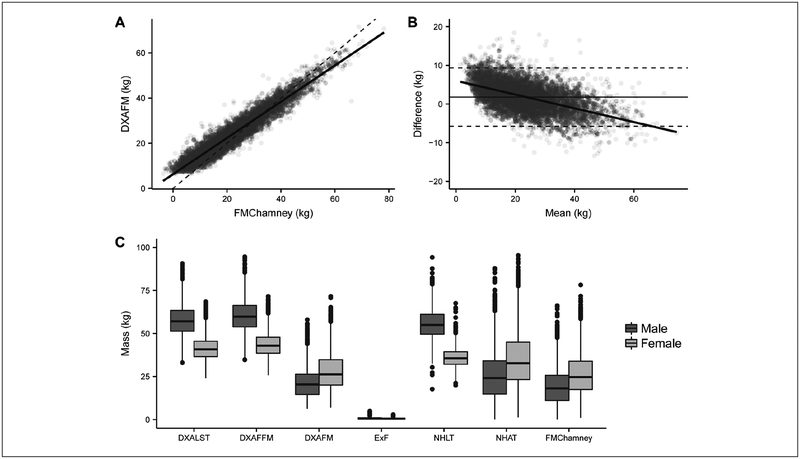
Figure 1 –
Body composition measurement comparisons between DXA and BIS from the NHANES dataset. (A) Correlation and (B) Bland-Altman (B-A) analysis between DXAFM and FMChamney (n=5884). Dashed lines in the B-A plot represent the limits of agreement (1.96xSD). Mean difference, limits of agreement, and other analysis for this comparison is available in Table 3. (C) Box-plotsof DXA and BIS tissue compartments for the NHANES dataset by gender.
Abbreviations: DXA, dual energy X-ray absorptiometry; BIS, bioimpedance spectroscopy; FM, fat mass; FMchamney, FM calculated from the Chamney model using BIS-generated BMI-adjusted ECWmoissl and ICWmoissl LST, lean soft tissue; FFm, fat free mass; EXF, excess fluid (Chamney model); NHAT, normally hydrated lean tissue (chamney model); NHAT, normally hydrated adipose tissue (Chamney model).
Method Agreement for FM
FMChamney was compared to DXAFM (Figure 1, Table 3). For the overall data set, BIS underestimated FM compared to DXA (mean difference=1.8 kg, p<0.0001). FM values for both methods were highly correlated (r=0.96, CCC=0.93; p<0.0001) (Figure 1A), however, B-A analysis revealed a proportional bias (Figure 1B).
Table 3.
– Comparison of FM by DXA and BIS, by BMI Category.
| Mean difference, kg (P-value)a | rb | RMSE, kg | CCC | B-A limits of agreement | FMChamneyMean, kg | DXAFM Mean, kg | B-A PEc | r2 for regression line through B-A plot (P-value)d | |
|---|---|---|---|---|---|---|---|---|---|
| DXAFM-FMChamney | 1.8 ± 3.8* | 0.96 | 4.2 | 0.93 | −5.8 to 9.3 | 22.8 ± 12.3 | 24.6 ± 10.3 | 31% | 0.27 (<0.0001) |
| <18.5 kg/m2 | 3.6 ± 2.8* | 0.74 | 4.5 | 0.43 | −1.9 to 9.0 | 7.2 ± 4.1 | 10.8 ± 2.5 | 51% | 0.34 (<0.0001) |
| 18.5–24.9 kg/m2 | 3.1 ± 3.3* | 0.85 | 4.5 | 0.71 | −3.4 to 9.6 | 13.8 ± 6.2 | 17.0 ± 4.7 | 38% | 0.22 (<0.0001) |
| 25–29.9 kg/m2 | 1.7 ± 3.6* | 0.83 | 4.0 | 0.78 | −5.3 to 8.7 | 23.5 ± 6.5 | 25.2 ± 5.1 | 28% | 0.17 (<0.0001) |
| 30–39.9 kg/m2 | −0.3 ± 4.0 (0.02) | 0.89 | 4.0 | 0.87 | −8.1 to 7.6 | 36.7 ± 8.6 | 36.5 ± 7.0 | 22% | 0.16 (<0.0001) |
| >40 kg/m2 | −2.0 ± 4.2* | 0.84 | 4.6 | 0.80 | −10.2to6.1 | 55.6 ± 7.7 | 53.6 ± 6.4 | 15% | 0.11 (0.001) |
For BMI categories: <18.5 kg/m2, n=173; 18.5–24.9 kg/m2, n=2350; 25–30 kg/m2, n=1914; 30–40 kg/m2, n=1339; >40 kg/m2, n=108.
Mean difference calculated as dual energy X-ray absorptiometry (DXA) – bioimpedance spectroscopy using the Chamney model equations. P-value reported by paired t-test.
indicates P-value <0.0001.
r = Pearson correlation coefficient. All values are significant, P-value <0.0001.
Bland-Altman (B-A) percent error (PE) calculated as limits of agreement for the difference between the two methods (1.96(SD)) divided by the mean for the reference DXA method × 100%.
r2 is reported, P-value indicates significant deviation of regression slope from zero. Unless otherwise noted, all values are mean ± SD. Abbreviations: FM, fat mass; RMSE, root mean square error; CCC, concordance correlation coefficient; FMChamney, FM calculated from the Chamney model using BIS-generated BMI-adjusted ECWMoissl and ICWMoissl.
From the literature3,5 we hypothesized that BMI category may impact the method agreement. Table 3 shows the comparison between DXAFM and FMChamney by BMI category. BMI categories were defined as underweight (<18.5 kg/m2), normal weight (18.5–24.9 kg/m2), overweight (between 25 and 29.9 kg/m2), obesity (30–39.9 kg/m2), and severe obesity (≥40 kg/m2). The best method agreement occurred at BMI 30 kg/m2 or higher, and the B-A PE was lower in women (17%) than men (27%; data not shown). B-A analysis revealed a proportional bias for all BMI categories, except for BMI ≥40 kg/m2.
Method Agreement for Lean Tissue
Because the BIS Chamney approach generates NHLT, a more specific representation of the lean tissue than the more global DXAFFM, we did not directly compare the two. However, we did calculate FFMChamney (i.e. weight – FMChamney) and found that it was well-correlated with DXAFFM for the overall dataset (r=0.94, p<0.001). DXALST and NHLT were also well-correlated (r=0.82, p<0.001). No further analyses on the comparison between lean tissue measures were conducted on the NHANES dataset.
Longitudinal Dataset (RYGB)
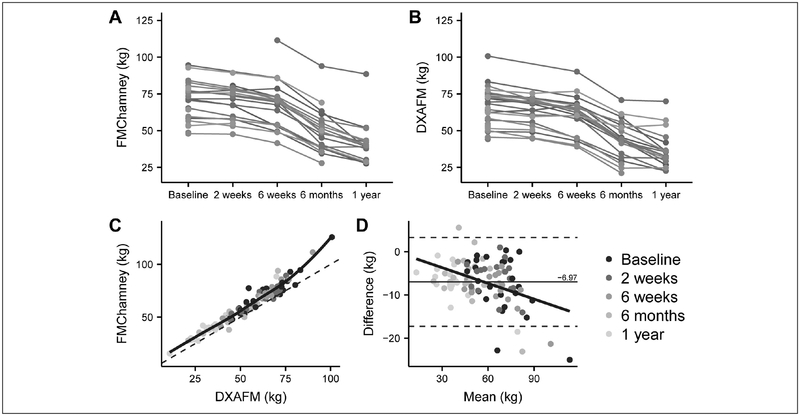
After demonstrating the effectiveness of the Chamney model for FM assessment in healthy adults (NHANES), we then applied the model to non-Hispanic white women with obesity undergoing massive weight loss in the first year after RYGB (Table 4), to evaluate the method’s efficacy for monitoring FM changes. FM measures for each study participant by both methods are shown in Figures 2A and 3B.
Table 4.
– Descriptive Statistics and Body Composition Data for Women Undergoing Weight Loss after RYGB.
| Time of follow-up post-RYGB | |||
|---|---|---|---|
| Baseline (n=25) |
6-months (n=16) |
1-year (n=15) |
|
| Age at time of surgery, years | 48 ± 10 | 47 ± 10 | 47 ± 10 |
| Weight, kg | 127.4 ± 22.4 | 98.99 ± 19.9a | 87.7 ± 19.8b |
| Height, cm | 165 ± 6 | 166 ± 5 | 166 ± 5 |
| BMI, kg/m2 | 46.6 ± 6.8 | 35.7 ± 6.3a | 31.6 ± 6.3b |
| DXA | |||
| DXAFFM | 65.3 ± 12.1 | 55.7 ± 8.4a | 56.0 ± 7.3a |
| DXALST | 62.7 ± 12.2 | 52.7 ± 8.3a | 53.2 ± 7.1a |
| DXAFM | 64.1 ± 13.8 | 44.3 ± 13.6a | 33.2 ± 13.3b |
| BIS | |||
| RE, Ω | 545 ± 61 | 586 ± 56 | 582 ± 56 |
| RI, Ω | 1273±138 | 1602 ±224a | 1618±161a |
| ECWMoissl, L | 19.9 ± 3.1 | 17.8 ± 2.7a | 17.2 ± 2.6b |
| ICWMoissl, L | 21.4 ± 2.6 | 18.3 ± 2.3a | 18.0 ± 1.9b |
| Chamney model: | |||
| ExF, kg | −1.2 ± 1.3 | 1.0 ± 1.0a | 1.8 ± 1.1b |
| NHLT, kg | 33.3 ± 5.5a | 30.7 ± 5.6b | 32.3 ± 4.9ab |
| NHAT, kg | 95.3 ± 22.9 | 67.3 ± 21.7a | 53.6 ± 21.8b |
| FMChamney, kg | 71.8 ± 17.2 | 50.6 ± 16.3a | 40.3 ± 16.4b |
Abbreviations: BMI, body mass index; BIS, bioimpedance spectroscopy; RE and RI are Cole model terms: RE, resistance of extracellular water; RI, resistance of intracellular water; ECW, extra cellular water; ICW, intracellular water; Chamney model compartments (derived by applying bioimpedance spectroscopy (BIS)-generated BMI-corrected ECWMoissl and ICWMoissl to Chamney model equations): ExF, extracellular fluid; NHLT, normally-hydrated lean tissue; NHAT, normally-hydrated adipose tissue; FM, fat mass; DXA, dual energy X-ray absorptiometry; LST, lean soft tissue; RYGB, Roux-en-Y gastric bypass.
Means that do not share a letter within a row are significantly different (P<0.05) by paired t-test; comparison between baseline and 6-months, n=16; between baseline and 1-year, n=15; between 6-months and 1-year, n=14; means that share a letter within a row are not significantly different by paired t-test. All values are mean ± SD
Figure 2 –
FM measurement comparisons between DXA and BIS during the first year after RYGB. (A) FM measured by DXA and (B) BIS (Chamney model) and a comparison between DXAFM and FMchamney post-RYGB by (C)correlation and (D) Bland-Altman analysis.
Abbreviations: DXA, dual energy X-ray absorptiometry; BIS, bioimpedance spectroscopy; RYGB, Roux-en-Y gastric bypass; FM, fat mass; FMchamney FM calculated from the chamney model using BIS-generated BMI-adjusted ECWmoissl and ICWmoissl
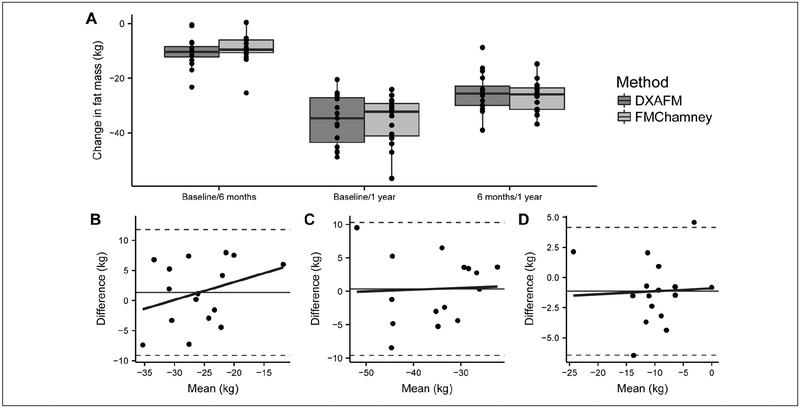
Figure 3 –
Comparison of FM change intervals between DXA and BIS measured in the first year following RYGB. (A) mean changes in FM by each method for each change interval. Bland-Altman analyses for the change in FM from (B) baseline to 6 months, (C) baseline to 1 year, and (D) 6 months to 1 year after surgery. Abbreviations: DXA, dual energy X-ray absorptiometry; BIS, bioimpedance spectroscopy; RYGB, Roux-en-Y gastric bypass; FM, fat mass; FMchamney FM calculated from the Chamney model using BIS-generated BMI-adjusted ECWMoissl and ICWMoissl.
Method Agreement for FM
The Chamney model systematically overestimated FM compared to DXA at each time point (Table 5 and Figure 2C). This agreed with the NHANES dataset, where for women with BMI 30–39.9 kg/m2 there was no inter-method difference (n=709, DXAFM=39.3 kg vs. FMChamney=39.5, p=0.15), and for women with BMI ≥40 kg/m2 the Chamney model overestimated FM (n=89, DXAFM=54.6 vs. FMChamney=56.5, p<0.0001). The methods were highly correlated with low B-A PE and RMSE; however, the B-A LOA were wide and proportional bias was evident (Figure 2D).
Table 5.
– FM by DXA and BIS for the Roux-en-Y Gastric Bypass Longitudinal Data Set.
| Timepoint | n | Mean difference, kg | r | RMSE, kg | CCC | r2 | B-A Limits of agreement | FMChamney, kg | DXAFM, kg | B-A PE | r2 for the regression line through the B-A plot (P-value) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Pre-RYGB | 25 | −7.6 ± 6.7* | 0.93* | 6.47 | 0.81 | 0.86 | −20.8 to 5.5 | 71.8 ± 17.2 | 64.1 ± 13.8 | 20% | 0.27 (0.008) |
| 6-months post-RYGB | 16 | −6.3 ± 5.9* | 0.94* | 5.80 | 0.85 | 0.88 | −17.8 to 5.2 | 50.6 ± 16.3 | 44.3 ± 13.6 | 26% | 0.22 (0.07) |
| 1-year post-RYGB | 15 | −7.2 ± 4.3* | 0.98* | 3.45 | 0.86 | 0.96 | −15.7 to 1.3 | 40.3 ± 16.4 | 33.2 ± 13.3 | 26% | 0.53 (0.002) |
| Δ Baseline to 6-months | 16 | 1.3 ± 5.3 | 0.68* | 4.26 | 0.64 | 0.46 | −9.1 to 11.8 | −26.5 ± 5.6 | −25.2 ± 7.2 | - | 0.11 (0.22) |
| Δ Baseline to 1-year | 15 | 0.36 ± 5.1 | 0.84* | 4.99 | 0.84 | 0.71 | −9.6 to 10.3 | −35.6 ± 8.9 | −35.2 ± 9.2 | - | 0.002 (0.87) |
| Δ 6-months to 1-year | 14 | −1.3 ± 2.8 | 0.88* | 2.83 | 0.86 | 0.78 | −6.9 to 4.2 | −9.2 ± 5.7 | −10.6 ± 5.9 | - | 0.002 (0.87) |
Abbreviations: FM, fat mass; DXA, dual energy X-ray absorptiometry; BIS, bioimpedance spectroscopy; RYGB, Roux-en-Y gastric bypass; RMSE, root mean square error; CCC, concordance correlation coefficient; B-A, Bland-Altman analysis; FMChamney, FM calculated from the Chamney model using BIS-generated BMI-adjusted ECWMoissl and ICWMoissl; B-A percent error (PE) calculated as limits of agreement for the difference between the two methods (1.96(SD)) divided by the mean for the reference DXA method × 100%. PE not calculated for FM change intervals because the magnitude of error in all cases was greater than the mean difference between methods. Values are reported as mean ± SD unless otherwise noted.
indicates significantly different, P-value <0.001
There was no significant inter-method difference for any of the weight change intervals; the best correlations were seen for FM changes between baseline and 1-year and from 6-months to 1-year (Table 5, Figure 3A). The B-A analysis for each change interval revealed slight proportional bias for the change interval between baseline and 6 months (highest weight period), but no proportional bias for the change intervals between baseline and 1 year or 6 months and 1 year, however the LOA were wide (Figures 3B, 3C, and 3D).
Method Agreement for Lean Tissue
DXAFFM and FFMChamney were correlated at each time point [baseline (r=0.71), 6 months (r=0.85), and 1 year post-RYGB (r=0.89); p<0.0001)]. DXAFFM and FFMChamneychanges at 1-year were not different (−11.5±9.8 vs.−9.7±2.5 kg, respectively; p=0.48). Between 6-months and 1-year, the DXAFFM and FFMChamneychanges were different (−0.06±2.8 vs. −1.7±2.2 kg, respectively; p=0.03), but were correlated (r=0.55, p=0.04). Interestingly, by 1-year our subjects lost only 1.3±2.5 kg NHLT.
DISCUSSION
We have demonstrated that a BIS approach that incorporates the Moissl5 BMI-correction to the XHM equations and the Chamney 3-component model6 can estimate FM and FFM in a diverse healthy population sample with comparative accuracy relative to DXA. This BIS approach also shows promise as a monitoring tool for clinical settings. We demonstrated that it could track changes in FM and lean tissue reasonably well (compared to DXA) in women with obesity undergoing massive weight loss after RYGB. In individuals with obesity, BC assessment can be particularly challenging; an accurate method for quantifying lean and fat tissue changes in individuals undergoing weight loss interventions would be of great benefit to clinicians.
Our first priority in this study was to compare DXAFM and FMChamney, in part because the direct comparison of NHLT with DXAFFM or DXALST is complicated by the substantial magnitude of difference between them, particularly at higher BMIs. FMChamney is directly calculated from NHAT, which is derived from NHLT and ExF, incorporating BIS-derived BMI-corrected ECW and ICW. It is not ideal to calculate FFMChamney by simply subtracting FMChamney from body weight in order to compare with DXAFFM because it increases the potential for error to back-convert to a 2-component model. Thus, we narrowed our focus on the FM measures. If the agreement between FMChamney and DXAFM were sufficiently strong, then it should follow that the lean estimates could be considered equally valid.
From our analyses of the NHANES dataset inclusive of mixed ethnicity, gender, and ages 18–49 years, the FMChamney estimates agreed with DXAFM, although proportional bias was evident. Given that BIS algorithms were developed from dilution techniques to measure water compartments, DXA is not the ideal FM reference against which these techniques are compared; i.e. scaling error can occur14 Upon closer inspection by BMI-category, it appeared that FMChamney performed best (minimal mean bias, lowest B-A PE, reasonably good correlations and minimal proportional bias) at BMI ≥30 kg/m2.
One potential concern for future applications of Chamney-like model equations is that they should guard against the generation of negative values for tissue compartments. For example, when the sum of calculated ExF and NHLT is greater than total body weight, which could happen at very low BMIs, the Chamney model equations allow for the generation of non-physiological (i.e.≤0 kg) values of FM. In the NHANES dataset, 0.3% of included observations generated negative FMChamney values.
These were not excluded from analysis (n=21, mean BMI=19.3±1.5, all male). These findings confirm that the Chamney model agrees best with DXA for estimating FM at higher BMIs and that the model needs improvement to prevent bias and enable use in very lean individuals.
Our longitudinal study in women undergoing RYGB provided another way to evaluate the Chamney approach, in a model of rapid weight loss with substantial BC changes. These data are uniquely suited for this evaluation given that mean BMI was Ł30 kg/m2 throughout the study. At all time-points (baseline, 6 months and 1 year post-RYGB) FMChamney was in good agreement with DXAFM by correlation, %RMSE, and CCC, but systematically overestimated FM and the B-A PE was slightly higher than in the NHANES population, indicating wide individual variability. The comparison of changes were more variable across time, with the strongest support (lowest RMSE, strong correlation and CCC, no proportional bias) for the Chamney approach between 6-months and 1-year.
Given that BIS is scaled to dilution, our finding that BIS overestimates FM compared to DXA is not unexpected; we previously reported that DXA underestimated FM compared to multiple dilution in women undergoing RYGB11. Others have reported that DXA underestimates FM and overestimates FFM losses in weight-losing individuals with obesity15,16. DXA may be an imperfect reference technique in this difficult-to-measure population. One concern is excess adiposity and the accompanying expansion of ECW. The overestimation of FFM losses by DXA may be due to its inability to discern fluid changes with weight and FM loss after weight-loss interventions11,15,17. More specifically, DXA cannot differentiate between the ICW and ECW in NHAT or NHLT, nor excess ECW (edema). Changes in DXA-measured FFM could simply be a reflection of changes in ECW accompanied by large FM loss.
The BIS Chamney approach may be well-suited to assessing BC changes in individuals with obesity undergoing weight loss. Of note, the Chamney model was originally developed for the purpose of distinguishing excess fluid from adipose and lean tissue compartments in order to better target the clinical management of individuals on dialysis6,18. Its incorporation in conjunction with an adiposity-correction to the XHM equations into software accompanying the Xitron-based BIS device used by Fresenius Medical Care Europe (BCM Body Composition Monitor) has allowed for ExF assessment during dialysis7,8,18.
In addition to more effective tailoring of dialysis, compelling evidence of the efficacy of this approach for lean tissue assessment came from a recent report that demonstrated that individuals on hemodialysis who had NHLT values below the 10th percentile from healthy reference data had significantly higher mortality, suggesting that it could be indicative of malnutrition and poor clinical status8.
Indeed, the strength of the BIS Chamney approach (and similar models) is the ability to characterize BC in terms beyond the 2-compartment (FM and FFM) model. In addition to estimating pathologic excess fluid, it offers the potential to provide more specific information about the metabolically active lean tissue compartment. The NHLT theoretically represents a compartment closer to what has been termed the body cell mass, reflecting the metabolically active tissue that is deemed most important for long-term weight loss success.
Indeed, Wieskotten et al.19 evaluated a BIS Chamney model-based algorithm and NHLT to identify malnutrition against subjective global assessment in a diverse patient group and found that their algorithm identified malnutrition with 72–83% sensitivity. Further development is needed, however this BIS Chamney approach forms the basis for important advances in bioimpedance as a bedside assessment tool, because of the paramount role that lean tissue has in nutritional status, and given that assessment of muscle loss is one of the defining characteristics of malnutrition as defined in the US by the American Society for Parenteral and Enteral Nutrition and the Academy of Nutrition and Dietetics20.
Although the Moissl BMI-corrected BIS approach has been validated in healthy individuals and those on dialysis for body water compartments5, there are limited published data comparing the Chamney BIS approach for FM and lean tissue compartments against reference methods. Two abstracts describing validation efforts for FM and FFM estimates by this approach in healthy individuals and those with cancer, liver, and renal disease are cited18 but are unavailable in any indexed databases or search engines.
Therefore, our ability to compare our results in terms of measurement error against published literature on this method was limited. Wieskotten et al.19 report some of this data in their evaluation of this BIS Chamney approach for identifying malnutrition. Specifically, the BIS Chamney approach was reported to be in good agreement with DXA for FM in 321 healthy subjects (R2=0.86, p<0.01; minimal bias).
We found that in healthy individuals NHLT was well-correlated with DXAFFM. As expected, the best agreement was found in normal weight individuals who are unlikely to have issues with excess fluid. In support of this, in our overall NHANES dataset, ExF was quite low (Figure 1C). This suggests that with normal weight and hydration status DXAFFM and NHLT are measuring very similar lean tissue compartments. At higher adiposity this relationship may not hold because DXA cannot distinguish between overhydrated lean tissue and fluid compartments. It is this situation that makes the Chamney BIS approach appealing, as it could allow for more accurate BC assessment in individuals with obesity.
Interestingly, when looking at our longitudinal DXA data, our subjects lost a substantial amount of lean tissue, as measured by DXAFFM (~12 kg). However, by the Chamney approach, the loss of NHLT was minimal (~1.5 kg). An abstract presented at The Obesity Society annual meeting in 2012 reported similar findings in 17 ‘Biggest Loser’ contestants21.
They reported that %FM and FM changes by DXA, air-displacement-plethysmography, and the BIS Chamney model were well-correlated and not different. At 7-months, DXAFM and FFM decreased by 46.5kg and 10.8 kg, respectively; BIS NHLT increased 1.4 kg. We cannot be certain that BIS is providing the correct information, but it is possible that the inability of DXA to distinguish fluid from the lean and adipose tissues could be skewing DXAFFM loss estimates upward.
From a slightly different perspective, this could be interpreted to mean that the observed DXAFFM loss was almost entirely adipose tissue water, assuming that the Chamney model was valid in these subjects based on our FM change comparisons. This underscores that DXA may be deficient under these conditions and a method such as the Chamney approach that can distinguish excess fluid from lean tissue mass could offer advantages, particularly for clinical assessment.
CONCLUSIONS
Taken together, the strong agreement between the BIS Chamney approach and DXA for FM and FM changes suggest that it may also be a good option for monitoring lean tissue (i.e. NHLT) in overweight and obesity. Monitoring of lean tissue changes (independent of fluid changes) in response to interventions remains a high priority for nutrition care, in individuals across the weight spectrum. Our data, in addition to the successful application of this BIS approach for assessing excess fluid (ExF) for the tailoring of dialysis regimens7,18 provides compelling reason to consider its evaluation for assessing NHLT in other clinical populations. However, further refinement in the calculation of the 3 compartments is needed to protect against the generation of non-physiologic values and to fine-tune the accuracy of the model components. More advanced methods have been proposed22, and it appears that improved results are being reported9. This is an exciting time, with new advances in bedside assessment that could improve patient care.
Acknowledgments
FUNDING
Research reported in this publication was supported by the Rhoads Research Foundation ofthe American Society for Parenteral and Enteral Nutrition (Earthman); NIH Grant M01-RR00400 which supported the GCRC at the University of Minnesota; USDA National Institute of Food and Agriculture and the Minnesota Agricultural Experiment Station, Project MIN-18–104, Hatch Funding (Earthman); and the Midwest Dairy Association (Earthman).
Footnotes
Local de realização do trabalho: University of Minnesota, Department of Food Science and Nutrition, Saint Paul, MN, USA.
Conflito de interesse: Os autores declaram não haver.
REFERENCES
- 1.Matthie JR. Bioimpedance measurements of human body composition: critical analysis and outlook. Expert Rev Med Devices. 2008;5(2):239–61. [DOI] [PubMed] [Google Scholar]
- 2.De Lorenzo A, Andreoli A, Matthie J, Withers P. Predicting body cell mass with bioimpedance by using theoretical methods: a technological review. J Appl Physiol (1985).1997;82(5):1542–58. [DOI] [PubMed] [Google Scholar]
- 3.Cox-Reijven PL, Soeters PB. Validation of bio-impedance spectroscopy: effects of degree of obesity and ways of calculating volumes from measure dresistance values. Int J Obes Relat Metab Disord. 2000;24(3):271–80. [DOI] [PubMed] [Google Scholar]
- 4.Mager JR, Sibley SD, Beckman TR, Kellogg TA, Earthman CP. Multifrequency bioelectrical impedance analysis and ELRLPSHGDQFH VSHFWURVFRS\ IRU PRQLWRULQJ ÀXLG DQG ERG\ cell mass changes after gastric bypass surgery. Clin Nutr. 2008;27(6):832–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Moissl UM, Wabel P, Chamney PW, Bosaeus I, Levin NW, Bosy-Westphal A et al. body fluid volume determination via body composition spectroscopyin health and disease. Physiol Meas. 2006;27(9):921–33. [DOI] [PubMed] [Google Scholar]
- 6.Chamney PW, Wabel P, Moissl UM, Müller MJ, Bosy-Westphal A, Korth O, et al. A whole-body model to distinguish excess-fluid from the hydration of major body tissues. Am J Clin Nutr. 2007;85(1):80–9. [DOI] [PubMed] [Google Scholar]
- 7.Broers NJ, Martens RJ, Cornelis T, Diederen NM, Wabel P, van der Sande FM, et al. Body composition in dialysis patients: a functional assessment of bioimpedance using different prediction models. J Ren Nutr. 2015;25(2):121–8. [DOI] [PubMed] [Google Scholar]
- 8.Marcelli D, Usvyat LA, Kotanko P, Bayh I, Canaud B, Etter M, et al. ; MONitoring Dialysis Outcomes (MONDO) Consortium. Body composition and survival in dialysis patients: results from an international cohort study. Clin J Am Soc Nephrol. 2015;10(7):1192–200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Keane D, Chamney P, Heinke S, Lindley E. Use of the body composition monitor for fluid status measurements in subjects with high body mass index. Nephron. 2016;133(3):163–8. [DOI] [PubMed] [Google Scholar]
- 10.NHANES 2003 – 2004: Bioelectrical Impedance Analysis Data Documentation, Codebook, and Frequencies. Washington: Centers for Disease Control and Prevention; 2004. [cited 2017 Aug 9]. Available from: https://wwwn.cdc.gov/nchs/nhanes/2003-2004/BIX_C.htm [Google Scholar]
- 11.Levitt DG, Beckman LM, Mager JR, Valentine B, Sibley SD, Beckman TR, et al. Comparison of DXA and water measurements of body fat following gastric bypass surgery and a physiological model of body water, fat, and muscle composition. J Appl Physiol (1985). 2010;109(3):786–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Earthman CP, Matthie JR, Reid PM, Harper IT, Ravussin E, Howell WH. A comparison of bioimpedance methods for detection of body cell mass change in HIV infection. J Appl Physiol (1985). 2000;88(3):944–56. [DOI] [PubMed] [Google Scholar]
- 13.Stevenson M, Nunes T, Heuer C, Marshall J, Sanchez J, Thornton R, et al. epiR: Tools for the analysis of epidemiological data; 2016.
- 14.Earthman CP. Body composition tools for assessment of adult-malnutrition at the bedside: a tutorial on research considerations and clinical applications. JPEN J Parenter Enteral Nutr. 2015;39(7):787–822. [DOI] [PubMed] [Google Scholar]
- 15.Pourhassan M, Schautz B, Braun W, Gluer CC, Bosy-Westphal A, Müller MJ. Impact of body-composition methodology on the composition of weight loss and weight gain. Eur J Clin Nutr. 2013;67(5):446–54. [DOI] [PubMed] [Google Scholar]
- 16.Valentine RJ, Misic MM, Kessinger RB, Mojtahedi MC, Evans EM. Location of body fat and body size impacts DXA soft tissue measures: a simulation study. Eur J Clin Nutr. 2008;62(4):553–9. [DOI] [PubMed] [Google Scholar]
- 17.Das SK, Roberts SB, Kehayias JJ, Wang J, Hsu LK, Shikora SA, et al. Body composition assessment in extreme obesity and after massive weight loss induced by gastric bypass surgery. Am J Physiol Endocrinol Metab. 2003;284(6):E1080–8. [DOI] [PubMed] [Google Scholar]
- 18.Wabel P, Chamney P, Moissl U, Jirka T. Importance of whole-body bioimpedance spectroscopy for the management of fluid balance. Blood Purif. 2009;27(1):75–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wieskotten S, Heinke S, Wabel P, Moissl U, Becker J, Pirlich M, et al. Bioimpedance-based identification of malnutrition using fuzzy logic. Physiol Meas. 2008;29(5):639–54. [DOI] [PubMed] [Google Scholar]
- 20.White JV, Guenter P, Jensen G, Malone A, Schofield M; Academy Malnutrition Work Group; A.S.P.E.N. Malnutrition Task Force; A.S.P.E.N. Board of Directors. Consensus statement: Academy of Nutrition and Dietetics and American Society for Parenteral and Enteral Nutrition: characteristics recommended for the identification and documentation of adult malnutrition (undernutrition). JPEN J Parenter Enteral Nutr. 2012;36(3):275–83. [DOI] [PubMed] [Google Scholar]
- 21.Matthie JR, Huizenga R. Lose fat - not weight! [Internet] In: The Obesity Society - 30th Annual Meeting; 2012. September 20–24; San Antonio, TX, USA: [cited 2016 Jun 16]. p. S152 Available from: https://higherlogicdownload.s3.amazonaws.com/OBESITY/004d4f70-37d5-434e-b24d-08a32dfdfcd9/UploadedImages/Obesity2012-FinalProgram.pdf [Google Scholar]
- 22.Chamney PW, Wabel P, inventors; Fresenius Medical Care Deutschland GmbH, assignee. Method and a device for determining the hydration and/or nutrition status of a patient. US Patent US 7917202 B2. 2011. March 29. [Google Scholar]