Abstract
Over the last few years, there has been a renewed interest in patients with characteristics of both asthma and COPD. Although the precise definition of asthma-COPD overlap (ACO) is still controversial, patients with overlapping features are frequently encountered in clinical practice, and may indeed have worse clinical outcomes and increased health-care utilization than those with asthma or COPD. Therefore, there is a critical need to set a framework for the therapeutic approach of such patients. There are key distinctions in the therapy between asthma and COPD, particularly regarding the initial choice of therapy. However, there is considerable overlap in the use of existing medications for both diseases. Furthermore, novel therapies approved for asthma, such as monoclonal antibodies, may have a role in patients with COPD and ACO. The use of biomarkers, such as peripheral blood eosinophils, exhaled nitric oxide, and serum IgE, may help in selecting appropriate therapies for ACO. In this review, we provide an overview of available treatments for both asthma and COPD and explore their potential role in the treatment of patients with ACO.
Key Words: asthma, COPD, overlap, therapy
Abbreviations: ACO, asthma-COPD overlap; ICS, inhaled corticosteroid; LABA, long-acting β2 agonist; LAMA, long-acting muscarinic antagonist
Asthma and COPD are the most prevalent chronic respiratory conditions affecting > 500 million people worldwide and resulting in significant morbidity and an increasing health-care expenditure.1, 2, 3 It is therefore not surprising that these diseases are frequently encountered in clinical practice and often coexist. Although in their classic presentations asthma and COPD are usually easy to differentiate, many patients may demonstrate features of both. The exact prevalence of asthma-COPD overlap (ACO) has been difficult to estimate both because of the heterogeneous study populations it represents and the lack of a universal definition. The reported prevalence of ACO increases with age and ranges between 1.6% and 4.5% in general population studies and up to 27% and 33% among asthma and COPD populations, respectively.4, 5
There is considerable debate on how to precisely define ACO.6 The key points of controversy on the utility of defining ACO include (1) the various definitions assume that patients that have features of both asthma and COPD represent a homogenous group, (2) each disease has a different mechanism and probably is composed of multiple endotypes with diverse disease manifestations, (3) the definitions lack assignment of how much weight each disease has on the clinical manifestations and outcomes, and (4) the definitions are sensitive but lack specificity.6, 7 Furthermore, most clinical trials of asthma and of COPD have excluded patients with overlapping features, which limits our understanding of patients who have characteristics of both diseases.7 Definitions that have been previously used to describe ACO include the presence of a postbronchodilator FEV1/FVC < 0.7 in addition to various combinations of clinical features, including significant history of smoking, patient-reported diagnosis of asthma, physician’s diagnosis of asthma, postbronchodilator increase of FEV1 > 200 to 400 mL or 12% to 15% predicted, blood or sputum eosinophilia, and history of wheezing.8, 9, 10, 11 A round table discussion by Sin et al,7 taking into consideration all aspects of both disease and published literature, reached a consensus definition that included major and minor criteria (Table 1). This definition of ACO will likely require further revision as the complexity of this entity is better understood.
Table 1.
Criteria for Diagnosis of Asthma-COPD Overlap7
| Major | Minor |
|---|---|
| 1. Age ≥ 40 y 2. Post bronchodilator FEV1/FVC < 0.70 or LLN 3. Exposure ≥ 10 pack-years of tobacco or equivalent indoor or outdoor air pollution 4. Documentation of asthma before age 40 y or BDR of > 400 mL in FEV1 |
1. Documentation of allergic rhinitis or atopic disease 2. BDR ≥ 200 mL and 12% from baseline values on two or more encounters 3. Peripheral serum eosinophil counts ≥ 300 cells/μL |
The diagnosis of asthma-COPD overlap is reached if all major and at least one minor criteria are present. BDR = bronchodilator response; LLN = lower limit of normal.
Despite the many definitions, patients who meet criteria or are considered within the spectrum of ACO may have worse outcomes than those with either disease alone. Patients with ACO have more respiratory symptoms, greater physical impairment, worse quality of life, and a higher risk for exacerbations and hospitalizations.12, 13, 14 Furthermore, patients with ACO may have more health-care utilization and cost of care than those with a single disease.5, 9, 12, 13, 14 The reasons for these observations are incompletely understood. It is possible that both diseases comprise a continuum of a single disease as described by Orie and Sluiter in the Dutch hypothesis15 because these individuals have genetic and environmental risk factors that may lead to progression of the disease. Alternatively, the British hypothesis posits that asthma and COPD have no common origin. These hypotheses are not necessarily mutually exclusive, and a recent clustering analysis identified the coincidence of features of both conditions in one cohort.16
Why Has ACO Become a Renewed Focus of Interest?
Asthma and COPD have been recognized now for centuries. The potential for overlap has been well described, and the concept that asthma and COPD share common origins was conceived more than half a century ago.15 Why then has there been an increased interest in this subgroup of patients in recent years? This may reflect an increased attention to disease phenotyping because precision medicine approaches require more refined understanding of disease heterogeneity. Along with this is the combination of increased recognition that patients with ACO may have worse outcomes and the development of drugs for specific phenotypes of asthma and COPD may have influenced the increased clinical and research interest in this area. For example, long-acting muscarinic antagonists (LAMAs) (tiotropium, Food and Drug Administration approved for asthma and COPD), a group of bronchodilators that were traditionally used for COPD, have now been found to be effective in asthma, suggesting additional common therapeutic pathways of both diseases.17 Additionally, monoclonal antibodies (ie, omalizumab, mepolizumab), which are typically reserved for patients with asthma, may have applications for patients with COPD who have asthma overlapping characteristics. These therapeutic innovations have economic implications and may influence lines of research and guideline development.18
Management Strategies for Asthma, COPD, and ACO
The Global Strategy for Asthma Management and Prevention and the Global Strategy for the Diagnosis, Management, and Prevention of COPD19 provided approaches for the diagnosis and management of ACO. However, these approaches were based mainly on expert opinion rather than patient outcomes data, reflecting an overall need for well-designed studies that address the management of this population. Nevertheless, these strategies provide a starting point for clinicians who encounter these patients in clinical practice and a framework for approaching management.
In patients with chronic airways disease that cannot be clearly identified as asthma or COPD, case finding strategies should be carried out with an emphasis on clinical history, physical examination, imaging, and screening questionnaires to determine if ACO is present, particularly if there are atypical features or poor response to initial therapy.20, 21 Underlining the importance of gathering more information, the Global Strategy for Asthma Management and Prevention and the Global Strategy for the Diagnosis, Management, and Prevention of COPD particularly emphasized that clinicians should focus on gathering as much phenotypic information as possible from their patients to guide treatment decisions (Table 2).19, 22, 23 Approaching ACO as a single disease is not ideal given the heterogeneity of both asthma and COPD. Even in milder stages, efforts should be carried out to determine phenotypic characteristics that may help the clinician decide if asthma or COPD carry more weight in ACO.
Table 2.
| Obstructive Airways Diseases | Clinical Characteristics | Medical Therapy |
|---|---|---|
| More consistent with asthma |
|
Initial therapy: 1. ICS ± LTRA 2. ICS/LABA 3. ICS/LABA + LAMA Advanced therapy: 1. Anti-IgE or 2. Anti-IL-5 |
| More consistent with COPD |
|
Initial therapy: 1. LABA or LAMA 2. LABA/LAMA 3. LABA/LAMA + ICS Advanced therapy: 1. Chronic macrolides 2. Roflumilast |
ICS = inhaled corticosteroid; LABA = long-acting β2 agonist; LAMA = long-acting muscarinic antagonist; LTRA = leukotriene receptor antagonist.
Contrasting Therapies of Asthma and COPD in Mild/Moderate Disease
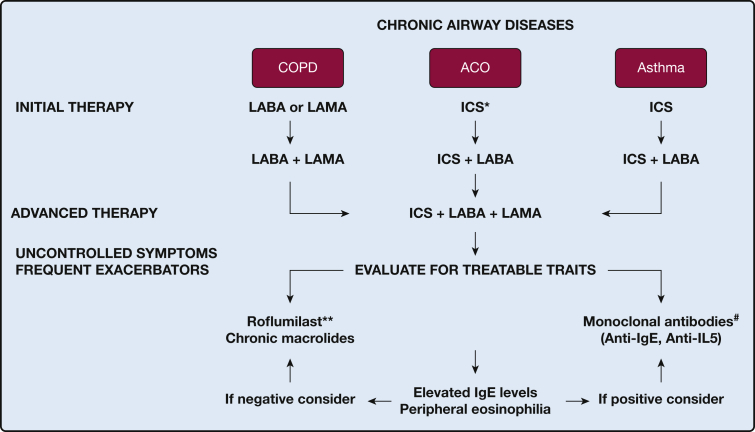
Inhaled medications are the foundation of therapy for both asthma and COPD. The treatment of these diseases uses a stepwise or escalation approach based on symptoms and exacerbations.22, 23 However, there is a key distinction between both treatment algorithms: specifically, the introduction of inhaled corticosteroids (ICSs) (Fig 1). In asthma, ICSs are introduced early in the treatment for patients who are symptomatic. ICSs decrease the risk of severe exacerbations, improve asthma control, and reduce the loss of lung function over time in patients with mild asthma.24, 25 Moreover, patients with asthma who stop ICSs have a higher risk of future exacerbations than those who continue therapy.26 A long-acting β2 agonist (LABA) is recommended as an add-on medication to ICSs only after ICSs and/or other controllers are ineffective in achieving adequate asthma control.23 LABAs are contraindicated for use as a single agent in asthma because of safety concerns because two large clinical trials have findings that link the use of salmeterol to increased rates of asthma-related deaths.27, 28 Although combination therapy (ICS/LABA) was found to be equally safe compared with ICSs alone in patients with persistent asthma and a history of severe exacerbations, there is still controversy related to the potential risk of LABAs in subsets of patients.29
Figure 1.
Treatment algorithm for chronic airway diseases. *Because of risk in patients with asthma with LABA monotherapy, ICS is the preferred therapy in ACO. **For patients with a chronic bronchitis phenotype and an FEV1 < 50% predicted. #For patients with allergic asthma and high IgE levels or with peripheral eosinophilia. ACO = asthma-COPD overlap; ICS = inhaled corticosteroid; LABA = long-acting β2 agonist; LAMA = long-acting muscarinic antagonist.
A LABA and/or LAMA is the preferred initial therapy in COPD, whereas the addition of ICSs is reserved for later stages in treatment (Fig 1). The use of long-acting bronchodilators (LABA or LAMA) alone in COPD has been shown to improve lung function, symptoms, and quality of life and reduce rates of exacerbations and hospitalizations.30, 31, 32 LABA monotherapy is generally considered safer in COPD than in asthma.30 LAMAs can also be used as a single agent and have been shown to be superior to LABAs in the reduction of exacerbations rates.33 However, the overall safety of bronchodilators in COPD is controversial, particularly regarding cardiovascular side effects.34, 35, 36, 37 Close monitoring may be warranted in patients with severe cardiovascular disease or life-threatening cardiovascular events because these were excluded from prior trials.31, 34, 38
The Food and Drug Administration has not approved any of the ICSs for use as monotherapy in patients with COPD. ICS plus LABA combination agents are recommended for those patients with COPD with a history of exacerbations despite adequate bronchodilator therapy. Therapy with ICSs in COPD has been associated with increases in FEV1 and a reduction in lung function decline.39, 40 However, monotherapy with ICS is not recommended in COPD because of an unclear risk-benefit ratio of ICS alone vs ICS/LABA therapy in relation to pneumonia.38, 41, 42, 43
Limited evidence exists regarding the appropriate first-line therapy for individuals with ACO. Several studies have demonstrated increased use of ICS, LABA, and other medications such as leukotriene receptor antagonists among individuals with ACO compared with COPD alone.44 Few trials have evaluated the safety or efficacy of ICS or LABA in ACO populations. One study explored the effects of 3 months of therapy with ICS/LABA in patients with COPD and showed that patients meeting criteria for ACO had a greater increase in FEV1 compared with those with COPD alone.45 These results support ICS use in patients with ACO. However, because ICSs alone are not recommended for patients with COPD, it is unclear if ICS monotherapy is effective in ACO. Likewise, until further data are available, it would be clinically reasonable to avoid LABA monotherapy in patients with ACO because of the previously discussed safety concerns. For those patients who are symptomatic with COPD and concomitant signs of asthma such as significant bronchodilator reversibility, increased blood or sputum eosinophils, or bronchial hyperresponsiveness, most experts advocate that bronchodilators should be continued with the addition of ICSs.19, 46 In addition to inhaled therapy, the clinician should address nonpharmacologic measures for both diseases including appropriate inhaler technique, identification and avoidance of triggers, smoking cessation, vaccination, pulmonary rehabilitation, oxygen therapy, and others.19, 22, 23 There is still much to be learned about the therapy of ACO in the milder stages, and the listed recommendations are based on expert opinion.19, 46
Contrasting Therapies of Asthma and COPD in Severe Disease
Both severe asthma and COPD are characterized by worsening respiratory symptoms, frequent exacerbations and increased health-care utilization.47, 48 In these advanced stages, the treatment may require triple therapy with ICS, LABA, and LAMA (Fig 1). In COPD, triple therapy has been shown to improve lung function and exacerbation rates without increasing adverse events compared with dual therapy and placebo.49, 50, 51 For instance, a recent study by Vestbo et al52 showed that triple therapy decreased the rate of moderate-to-severe COPD exacerbations by 20% compared with tiotropium alone without increasing the risk of pneumonia. Similarly, improvements in lung function and exacerbations rates have been observed with the addition of tiotropium to the combination of ICS and LABA in asthma.16, 53 A study by Magnussen et al54 showed in patients with COPD, concomitant asthma, and at least 1 year of ICS therapy, that tiotropium was superior compared with placebo in improving pulmonary function and reducing the need for rescue albuterol after 12 weeks of therapy. It is reasonable for patients with ACO who remain symptomatic or with frequent exacerbations despite initial inhaler therapy to be treated with triple therapy, but future studies are required to validate this measure. Only tiotropium has been well studied in asthma, and it remains unclear if all LAMAs are equally effective in the therapy of this patient population.
Monoclonal Antibodies
Monoclonal antibodies against IgE and IL-5 have been effective in reducing asthma exacerbations, ED visits, and oral corticosteroids in patients with severe allergic and eosinophilic disease, respectively.55, 56 Responder populations can be identified on the basis of type 2 inflammation phenotypes such as elevated peripheral blood eosinophils or elevated fractional exhaled nitric oxide. It is possible that these therapies could be of benefit for individuals with ACO. Recent data suggest that some individuals with ACO may also share similar disease drivers as type 2 inflammation in patients with asthma.57 This approach was explored in a trial of omalizumab (anti-IgE) in patients with severe allergic asthma and overlapping features of COPD.58 In the ACO subgroup, omalizumab was equally effective in improving asthma control and health-related quality of life compared with patients with severe allergic asthma alone.58 Additionally, an open-label, real-world study designed to evaluate predictors of clinical effectiveness in response to omalizumab in a cohort of asthmatics described the positive effects of this therapy in patients with overlapping features of asthma and COPD.59 The subgroup of patients with ACO experienced significant improvements in exacerbation rates after initiation of therapy compared with 12 months prior (mean, 1.1 vs 3.6). These patients also experienced improvements in asthma control.59 Supporting this concept, a prespecified subgroup analysis demonstrated that patients with COPD with an eosinophilic phenotype had a trend toward improvements in symptoms scores and exacerbations with the use of anti-IL-5 receptor antibody benralizumab, warranting further study in this COPD phenotype.60 More recently, mepolizumab, another anti-IL-5 antibody, showed a decrease in moderate or severe exacerbations in patients with COPD and an eosinophilic phenotype compared with placebo.61 Ongoing studies will further clarify the role of these and other monoclonal antibodies in ACO, but evidence continues to emerge pointing toward the benefits of these therapies in this subgroup of patients.
Phosphodiesterase Inhibitors
Roflumilast, an oral phosphodiesterase-4 inhibitor, has been shown to improve lung function and exacerbation rates in patients with COPD and FEV1 < 50% predicted, chronic bronchitis, and history of frequent or severe exacerbations.62, 63 Roflumilast has also been studied in various asthma populations. An analysis of nine placebo-controlled, double-blind, parallel group phase II or III studies (985 sites across all continents) evaluating the effects of roflumilast in patients with a history of asthma was conducted.64 Of these studies, four phase III monotherapy studies and two phase III combination (with ICS) studies consistently revealed statistically significant improvements in lung function in patients with asthma. The roflumilast dose of 500 μg generally showed more improvement in FEV1 compared with 125 or 250 μg. More recently, a study showed that roflumilast improved lung function and asthma control in patients with moderate-to-severe asthma.65 Importantly, roflumilast has been shown to have an adequate safety profile in asthma.65, 66 Although no trials exist to date evaluating the use of roflumilast in patients with ACO, this therapy could be considered, particularly among those individuals with ACO with frequent exacerbations.
Macrolides
The use of macrolides has been explored in various pulmonary diseases. Chronic azithromycin has been linked to a reduction in exacerbations rates among individuals with COPD and is currently recommended for those with frequent exacerbations who are not actively smoking (Fig 1).22, 67, 68 On the other hand, the use of macrolides for chronic asthma has had mixed results. A double-blind, placebo-controlled trial evaluating the use of 26 weeks of azithromycin did not find any significant benefits in severe asthma exacerbation rates or lower respiratory tract infections.69 In the same study, among those patients with a noneosinophilic inflammatory profile and a fractional exhaled nitric oxide below the upper limit of normal, the use of azithromycin was associated with lower rates of exacerbations. Similar observations, in which macrolides had beneficial effects among patients with severe asthma without eosinophilia, had previously been reported.70 Despite these promising findings, a rigorous systematic review of 23 studies performed to determine the efficacy of macrolides in asthma concluded there was no beneficial effect compared with placebo, but these studies had significant heterogeneity and varying study designs and sample sizes.71 More recently, a study of 420 patients with symptomatic asthma despite ICS/LABA showed that azithromycin 500 mg three times weekly for 48 weeks reduced asthma exacerbations and improved asthma-related quality of life compared with placebo.72 Notably, reductions were seen in both eosinophilic and noneosinophilic phenotypes. Therefore, in patients with ACO, macrolides can be considered for those patients with frequent exacerbations given the evidence in COPD and increased evidenced of benefits in asthma. Future studies, with predefined ACO populations, are still needed to determine the role of macrolides in this setting.
Role of Biomarkers in ACO
The use of biomarkers to identify patients who may benefit from therapy is well established in asthma.55, 56 In COPD, this method has accumulating evidence of benefits.60, 61 Although this approach might appear to be particularly useful to identify the ACO population, to date there is no biomarker that best encompasses the biologic mechanisms of this overlap. In fact, evidence suggests that ACO is a heterogeneous group encompassing several phenotypes of disease, including both eosinophilic and neutrophilic immune activation. For example, sputum analysis has demonstrated both eosinophilic and neutrophilic groups within ACO cohorts.16 Even exacerbations in ACO appear to express different biologic clusters.73 Taken into consideration this heterogeneity, it has been suggested that the use of sputum cellularity can identify three distinct ACO subtypes: eosinophilic, neutrophilic, and paucigranulocytic.74 However, this strategy may be limited because the use of sputum analysis has not been widely used in clinical practice because of the high technical demands of this test. Despite these limitations, the use of biomarkers for type 2 inflammation may better categorize patients with ACO than the traditional clinical taxonomy.75
More recently, airway epithelial genomic signatures were used to identify a subgroup of patients with COPD who may have type 2 inflammation active disease using various COPD cohorts.57 Using this method, it was possible to identify a subgroup of patients with greater airway obstruction and bronchodilator reversibility and increased airway tissue and blood eosinophils.57 Importantly, this group also had a decrease in hyperinflation after treatment with ICSs, suggesting an improvement in small airway inflammation. Although not yet widely available, the use of type 2 inflammation genomic signatures may serve as predictors of response to therapy in patients with ACO.
Along with ACO, there has been renewed attention to eosinophilic COPD.60, 61 Up to 20% to 40% of individuals with COPD may have increased eosinophils in the sputum and blood.76, 77 There is considerable debate regarding what is the exact definition of eosinophilic COPD. Previous studies using monoclonal antibodies have defined the eosinophilic phenotype as sputum eosinophils > 3% or a peripheral eosinophil count > 150/mm3 or > 300/mm3 in the last 12 months.60, 61 These are arbitrary thresholds derived from previous studies in asthma, and controversy remains regarding the best way to define this phenotype. The use of ICSs in patients with eosinophilic COPD has been associated with greater reductions in exacerbation rate and slowing of the rate of lung function decline compared with those without eosinophilia in some studies,78, 79, 80, 81 but other studies have not shown differences in the response to ICS therapy based on eosinophil counts, particularly in those with mild-to-moderate COPD.82, 83 High blood eosinophil counts may help stratify patients with higher risk for pneumonia, particularly those with a FEV1 of 50% predicted or less.84 Moreover, a post hoc analysis of the WISDOM (Withdrawal of Inhaled Steroids during Optimized Bronchodilator Management) trial revealed that patients with a peripheral blood eosinophil count ≥ 4% or ≥ 300 cells/μL had a higher rate of failure after withdrawing ICSs.85 In addition to eosinophils, elevated periostin, a surrogate marker of IL-13 activity and type 2 inflammation, was associated with lung function improvement after therapy with ICSs in patients with stable COPD.86 Although these findings are related to COPD cohorts, it further supports the use of ICSs in patients with ACO, particularly in those with features of type 2 inflammation, such as peripheral eosinophilia.
Even within ACO, phenotypes likely exist. For instance, one might imagine a patient with mild airflow obstruction who is very responsive to ICSs (mild COPD with asthma features) vs a patient with severe, persistent airflow obstruction who is resistant to ICSs (severe asthma with COPD features) might both fall under the ACO umbrella. There is some evidence to suggest that a subset of individuals with ACO may have neutrophilic inflammation driving their disease. However, the benefit of non-type 2 inflammation therapies has yet to be studied in this population. Neutrophilic inflammation can be seen in both COPD and asthma, and neutrophilic inflammation may be more frequently encountered in severe asthma.87 Therefore, targeting this inflammatory pathway is an attractive therapeutic strategy in ACO for future studies.74
Therapeutic Guidance
In patients with ACO who remain symptomatic despite inhaled therapies, an in-depth evaluation is required to determine the presence of treatable traits, which may aid in the diagnosis and provide therapeutic guidance.46 Further work is needed to help clinicians select the most appropriate therapy. Predictors of response in chronic airways disease is an area of intense research. Elevated serum levels of IgE, exhaled fraction of nitric oxide, and peripheral blood eosinophilia not only provide potential insight to the ongoing pathologic processes, but also may predict response to certain therapies, such as omalizumab.88 Future therapies that emphasize biomarker or mechanistic approaches rather than clinical features to segment patient populations may better determine responder populations.
Conclusions
There is still much to learn about the spectrum of ACO. Because asthma and COPD are inherently heterogeneous, future studies that include well-characterized and richly phenotyped cohorts of patients with features of both asthma and COPD, with varying degrees of severity and exacerbation history, are needed to better study disease interactions, risk factors, and prognostic makers.89 A gap clearly exists in the evidence for therapy in ACO, particularly in severe disease. Nevertheless, in clinical practice, patients with ACO, with varying features of asthma and COPD features, present frequently and require optimal therapy. Taking into consideration that two diseases may coexist, with an emphasis in safety and phenotyping, it is necessary to carefully select therapies on a case-by-case basis. Identifying the presence of ACO in a clinic patient is the first step toward developing a treatment plan. However, it remains necessary to develop individualized plans based on concurrent evidence from clinical and biomarker data. Future studies that connect disease phenotypes to targeted therapies may identify improved personalized approaches to all patients with COPD or asthma.
Acknowledgments
Authors contributions: D. J. M. is the guarantor of this article. D. J. M. and F. J. M. contributed to conception, manuscript writing, and manuscript review. M. H. and S. A. C. contributed to manuscript writing and manuscript review. N. A. H., S. G. A, A. A., J. I. P., and M. K. H. contributed to interpretation of data and critical revisions. C. P. H. contributed to interpretation of data and critical revisions.
Financial/nonfinancial disclosures: The authors have reported to CHEST the following: D. J. M. reports consulting fees for GSK, Sunovion, AztraZeneca, and Bayer. M. H. works in the Clinical Discovery Unit at AztraZeneca. S. A. C. reports grants from MedImmune; personal fees from AstraZeneca; and nonfinancial support from Genentech. N. A. H. reports consulting fees for Roche, Teva, Sanofi, Boehringer Ingelheim, and Novartis and has received research support from Chiesi, Boehringer Ingelheim, GlaxoSmithKline, and Roche. C. P. H. reports personal fees from AstraZeneca, grants from Boehringer Ingelheim, personal fees from Mylan, and personal fees from Concert Pharmaceuticals. S. G. A. reports support from NIH, Veterans Affairs, Chest Foundation; grants from AstraZeneca, Boehringer Ingelheim Pharmaceuticals, Daiichi Sankyo, GlaxoSmithKline, Novartis and Sunovion. A. A. reports consultancy for Boehringer-Ingelheim, AstraZeneca, Novartis, and Sunovion. M. K. H. reports support from NIH, FNIH, and the COPD Foundation; consulting fees from GSK, Boehringer Ingelheim, Novartis, AstraZeneca, and Sunovion; and royalties from UptoDate. F. J. M. reports grants from NIH; personal fees from Forest, Janssens, GlaxoSmithKline (GSK), Nycomed/Takeda, Amgen, AstraZeneca, Boehringer Ingelheim, Ikaria/Bellerophon, Genentech, Novartix, Pearl, Pfizer, Roche, Sunovion, Theravance, Axon, CME Incite, California Society for Allergy and Immunology, Annenberg, Informa, Integritas, InThought, Miller Medical, National Association for Continuing Education, Paradigm, Peer Voice, UpToDate, Haymarket Communications, Western Society of Allergy and Immunology, Unity Biotechnology, ConCert, Lucid, Methodist Hospital, Prime, and WebMD. None declared (J. I. P.).
References
- 1.Murphy S.L., Kochanek K.D., Xu J., Arias E. Mortality in the United States, 2014. NCHS Data Brief. 2015;229:1–8. [PubMed] [Google Scholar]
- 2.López-Campos J.L., Ruiz-Ramos M., Soriano J.B. Mortality trends in chronic obstructive pulmonary disease in Europe, 1994-2010: a joinpoint regression analysis. Lancet Respir Med. 2014;2(1):54–62. doi: 10.1016/S2213-2600(13)70232-7. [DOI] [PubMed] [Google Scholar]
- 3.Ehteshami-Afshar S., FitzGerald J.M., Doyle-Waters M.M., Sadatsafavi M. The global economic burden of asthma and chronic obstructive pulmonary disease. Int J Tuberc Lung Dis. 2016;20(1):11–23. doi: 10.5588/ijtld.15.0472. [DOI] [PubMed] [Google Scholar]
- 4.Wurst K.E., Kelly-Reif K., Bushnell G.A., Pascoe S., Barnes N. Understanding asthma-chronic obstructive pulmonary disease overlap syndrome. Respir Med. 2016;110:1–11. doi: 10.1016/j.rmed.2015.10.004. [DOI] [PubMed] [Google Scholar]
- 5.de Marco R., Pesce G., Marcon A. The coexistence of asthma and chronic obstructive pulmonary disease (COPD): prevalence and risk factors in young, middle-aged and elderly people from the general population. PLoS One. 2013;8(5):e62985. doi: 10.1371/journal.pone.0062985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bateman E.D., Reddel H.K., van Zyl-Smit R.N., Agusti A. The asthma-COPD overlap syndrome: towards a revised taxonomy of chronic airways diseases? Lancet Respir Med. 2015;3(9):719–728. doi: 10.1016/S2213-2600(15)00254-4. [DOI] [PubMed] [Google Scholar]
- 7.Sin D.D., Miravitlles M., Mannino D.M. What is asthma-COPD overlap syndrome? Towards a consensus definition from a round table discussion. Eur Respir J. 2016;48(3):664–673. doi: 10.1183/13993003.00436-2016. [DOI] [PubMed] [Google Scholar]
- 8.Cosentino J., Zhao H., Hardin M. Analysis of asthma-chronic obstructive pulmonary disease overlap syndrome defined on the basis of bronchodilator response and degree of emphysema. Ann Am Thorac Soc. 2016;13(9):1483–1489. doi: 10.1513/AnnalsATS.201511-761OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hardin M., Silverman E.K., Barr R.G. The clinical features of the overlap between COPD and asthma. Respir Res. 2011;12:127. doi: 10.1186/1465-9921-12-127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hardin M., Cho M., McDonald M.L. The clinical and genetic features of COPD-asthma overlap syndrome. Eur Respir J. 2014;44(2):341–350. doi: 10.1183/09031936.00216013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Tashkin D.P., Chipps B.E., Trudo F., Zangrilli J.G. Fixed airflow obstruction in asthma: a descriptive study of patient profiles and effect on treatment responses. J Asthma. 2014;51(6):603–609. doi: 10.3109/02770903.2014.895012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Menezes A.M., Montes de Oca M., Pérez-Padilla R. Increased risk of exacerbation and hospitalization in subjects with an overlap phenotype: COPD-asthma. Chest. 2014;145(2):297–304. doi: 10.1378/chest.13-0622. [DOI] [PubMed] [Google Scholar]
- 13.Diaz-Guzman E., Khosravi M., Mannino D.M. Asthma, chronic obstructive pulmonary disease, and mortality in the U.S. population. COPD. 2011;8(6):400–407. doi: 10.3109/15412555.2011.611200. [DOI] [PubMed] [Google Scholar]
- 14.Alshabanat A., Zafari Z., Albanyan O., Dairi M., FitzGerald J.M. Asthma and COPD overlap syndrome (ACOS): a systematic review and meta analysis. PLoS One. 2015;10(9):e0136065. doi: 10.1371/journal.pone.0136065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Orie N.G.M., Sluiter H.J., editors. Bronchitis. Royal van Gorcum; Assen, The Netherlands: 1962. [Google Scholar]
- 16.Ghebre M.A., Bafadhel M., Desai D. Biological clustering supports both “Dutch” and “British” hypotheses of asthma and chronic obstructive pulmonary disease. J Allergy Clin Immunol. 2015;135(1):63–72. doi: 10.1016/j.jaci.2014.06.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kerstjens H.A., Engel M., Dahl R. Tiotropium in asthma poorly controlled with standard combination therapy. N Engl J Med. 2012;367(13):1198–1207. doi: 10.1056/NEJMoa1208606. [DOI] [PubMed] [Google Scholar]
- 18.Hole O.P., Winther F.O., Straume B. Clinical research: the influence of the pharmaceutical industry. Eur J Clin Pharmacol. 2001;56(11):851–853. doi: 10.1007/s002280000238. [DOI] [PubMed] [Google Scholar]
- 19.Global Initiative for Asthma and Global Initiative for Chronic Obstructive Pulmonary Disease. Diagnosis and initial treatment of asthma, COPD, and asthma-COPD overlap. http://ginasthma.org/download/824/. Accessed December 1, 2017.
- 20.Thiadens H.A., de Bock G.H., Dekker F.W. Identifying asthma and chronic obstructive pulmonary disease in patients with persistent cough presenting to general practitioners: descriptive study. BMJ. 1998;316(7140):1286–1290. doi: 10.1136/bmj.316.7140.1286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Tinkelman D.G., Price D.B., Nordyke R.J. Symptom-based questionnaire for differentiating COPD and asthma. Respiration. 2006;73(3):296–305. doi: 10.1159/000090141. [DOI] [PubMed] [Google Scholar]
- 22.Global Strategy for Asthma Management and the Prevention and the Global Strategy for the Diagnosis, Management and Prevention of Chronic Obstructive Pulmonary Disease. 2017 update. https://goldcopd.org/gold-reports/. Accessed May 15, 2018.
- 23.Global Initiative for Asthma. Global strategy for asthma management and prevention. 2017 update. https://ginasthma.org/2018-gina-report-global-strategy-for-asthma-management-and-prevention/. Accessed May 15, 2018.
- 24.Pauwels R.A., Pedersen S., Busse W.W. Early intervention with budesonide in mild persistent asthma: a randomised, double-blind trial. Lancet. 2003;361(9363):1071–1076. doi: 10.1016/S0140-6736(03)12891-7. [DOI] [PubMed] [Google Scholar]
- 25.O'Byrne P.M., Pedersen S., Busse W.W. Effects of early intervention with inhaled budesonide on lung function in newly diagnosed asthma. Chest. 2006;129(6):1478–1485. doi: 10.1378/chest.129.6.1478. [DOI] [PubMed] [Google Scholar]
- 26.Rank M.A., Hagan J.B., Park M.A. The risk of asthma exacerbation after stopping low-dose inhaled corticosteroids: a systematic review and meta-analysis of randomized controlled trials. J Allergy Clin Immunol. 2013;131(3):724–729. doi: 10.1016/j.jaci.2012.11.038. [DOI] [PubMed] [Google Scholar]
- 27.Castle W., Fuller R., Hall J., Palmer J. Serevent nationwide surveillance study: comparison of salmeterol with salbutamol in asthmatic patients who require regular bronchodilator treatment. BMJ. 1993;306(6884):1034–1037. doi: 10.1136/bmj.306.6884.1034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Nelson H.S., Weiss S.T., Bleecker E.R., Yancey S.W., Dorinsky P.M. The Salmeterol Multicenter Asthma Research Trial: a comparison of usual pharmacotherapy for asthma or usual pharmacotherapy plus salmeterol. Chest. 2006;129(1):15–26. doi: 10.1378/chest.129.1.15. [DOI] [PubMed] [Google Scholar]
- 29.Stempel D.A., Raphiou I.H., Kral K.M. Serious asthma events with fluticasone plus salmeterol versus fluticasone alone. N Engl J Med. 2016;374(19):1822–1830. doi: 10.1056/NEJMoa1511049. [DOI] [PubMed] [Google Scholar]
- 30.Kew K.M., Mavergames C., Walters J.A. Long-acting beta2-agonists for chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2013;(10) doi: 10.1002/14651858.CD010177.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Tashkin D.P., Celli B., Senn S. A 4-year trial of tiotropium in chronic obstructive pulmonary disease. N Engl J Med. 2008;359(15):1543–1554. doi: 10.1056/NEJMoa0805800. [DOI] [PubMed] [Google Scholar]
- 32.Wise R.A., Anzueto A., Cotton D. Tiotropium Respimat inhaler and the risk of death in COPD. N Engl J Med. 2013;369(16):1491–1501. doi: 10.1056/NEJMoa1303342. [DOI] [PubMed] [Google Scholar]
- 33.Vogelmeier C., Hederer B., Glaab T. Tiotropium versus salmeterol for the prevention of exacerbations of COPD. N Engl J Med. 2011;364(12):1093–1103. doi: 10.1056/NEJMoa1008378. [DOI] [PubMed] [Google Scholar]
- 34.Celli B., Decramer M., Leimer I., Vogel U., Kesten S., Tashkin D.P. Cardiovascular safety of tiotropium in patients with COPD. Chest. 2010;137(1):20–30. doi: 10.1378/chest.09-0011. [DOI] [PubMed] [Google Scholar]
- 35.Wang M.T., Liou J.T., Lin C.W. Association of cardiovascular risk with inhaled long-acting bronchodilators in patients with chronic obstructive pulmonary disease: a nested case-control study. JAMA Intern Med. 2018;178(2):229–238. doi: 10.1001/jamainternmed.2017.7720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Rogliani P., Matera M.G., Ora J., Cazzola M., Calzetta L. The impact of dual bronchodilation on cardiovascular serious adverse events and mortality in COPD: a quantitative synthesis. Int J Chron Obstruct Pulmon Dis. 2017;12:3469–3485. doi: 10.2147/COPD.S146338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Suissa S., Dell'Aniello S., Ernst P. Concurrent use of long-acting bronchodilators in COPD and the risk of adverse cardiovascular events. Eur Respir J. 2017;49(5) doi: 10.1183/13993003.02245-2016. [DOI] [PubMed] [Google Scholar]
- 38.Calverley P.M., Anderson J.A., Celli B. Salmeterol and fluticasone propionate and survival in chronic obstructive pulmonary disease. N Engl J Med. 2007;356(8):775–789. doi: 10.1056/NEJMoa063070. [DOI] [PubMed] [Google Scholar]
- 39.Celli B.R., Thomas N.E., Anderson J.A. Effect of pharmacotherapy on rate of decline of lung function in chronic obstructive pulmonary disease: results from the TORCH study. Am J Respir Crit Care Med. 2008;178(4):332–338. doi: 10.1164/rccm.200712-1869OC. [DOI] [PubMed] [Google Scholar]
- 40.Calverley P.M., Anderson J.A., Brook R.D. Fluticasone furoate, vilanterol, and lung function decline in patients with moderate chronic obstructive pulmonary disease and heightened cardiovascular risk. Am J Respir Crit Care Med. 2018;197(1):47–55. doi: 10.1164/rccm.201610-2086OC. [DOI] [PubMed] [Google Scholar]
- 41.Yang I.A., Clarke M.S., Sim E.H., Fong K.M. Inhaled corticosteroids for stable chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2012;(7) doi: 10.1002/14651858.CD002991.pub3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Kew K.M., Seniukovich A. Inhaled steroids and risk of pneumonia for chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2014;(3) doi: 10.1002/14651858.CD010115.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Martinez F.J., Vestbo J., Anderson J.A. Effect of fluticasone furoate and vilanterol on exacerbations of chronic obstructive pulmonary disease in patients with moderate airflow obstruction. Am J Respir Crit Care Med. 2017;195(7):881–888. doi: 10.1164/rccm.201607-1421OC. [DOI] [PubMed] [Google Scholar]
- 44.Caillaud D., Chanez P., Escamilla R. Asthma-COPD overlap syndrome (ACOS) vs 'pure' COPD: A distinct phenotype? Allergy. 2017;72(1):137–145. doi: 10.1111/all.13004. [DOI] [PubMed] [Google Scholar]
- 45.Lee S.Y., Park H.Y., Kim E.K. Combination therapy of inhaled steroids and long-acting beta2-agonists in asthma-COPD overlap syndrome. Int J Chron Obstruct Pulmon Dis. 2016;11:2797–2803. doi: 10.2147/COPD.S114964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Postma D.S., Rabe K.F. The Asthma-COPD overlap syndrome. N Engl J Med. 2015;373(13):1241–1249. doi: 10.1056/NEJMra1411863. [DOI] [PubMed] [Google Scholar]
- 47.Wedzicha J.A., Brill S.E., Allinson J.P., Donaldson G.C. Mechanisms and impact of the frequent exacerbator phenotype in chronic obstructive pulmonary disease. BMC Med. 2013;11:181. doi: 10.1186/1741-7015-11-181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Chung K.F., Wenzel S.E., Brozek J.L. International ERS/ATS guidelines on definition, evaluation and treatment of severe asthma. Eur Respir J. 2014;43(2):343–373. doi: 10.1183/09031936.00202013. [DOI] [PubMed] [Google Scholar]
- 49.Welte T., Miravitlles M., Hernandez P. Efficacy and tolerability of budesonide/formoterol added to tiotropium in patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2009;180(8):741–750. doi: 10.1164/rccm.200904-0492OC. [DOI] [PubMed] [Google Scholar]
- 50.Hanania N.A., Crater G.D., Morris A.N., Emmett A.H., O'Dell D.M., Niewoehner D.E. Benefits of adding fluticasone propionate/salmeterol to tiotropium in moderate to severe COPD. Respir Med. 2012;106(1):91–101. doi: 10.1016/j.rmed.2011.09.002. [DOI] [PubMed] [Google Scholar]
- 51.Jung K.S., Park H.Y., Park S.Y. Comparison of tiotropium plus fluticasone propionate/salmeterol with tiotropium in COPD: a randomized controlled study. Respir Med. 2012;106(3):382–389. doi: 10.1016/j.rmed.2011.09.004. [DOI] [PubMed] [Google Scholar]
- 52.Vestbo J., Papi A., Corradi M. Single inhaler extrafine triple therapy versus long-acting muscarinic antagonist therapy for chronic obstructive pulmonary disease (TRINITY): a double-blind, parallel group, randomised controlled trial. Lancet. 2017;389(10082):1919–1929. doi: 10.1016/S0140-6736(17)30188-5. [DOI] [PubMed] [Google Scholar]
- 53.Rodrigo G.J., Castro-Rodríguez J.A. What is the role of tiotropium in asthma?: a systematic review with meta-analysis. Chest. 2015;147(2):388–396. doi: 10.1378/chest.14-1698. [DOI] [PubMed] [Google Scholar]
- 54.Magnussen H., Bugnas B., van Noord J., Schmidt P., Gerken F., Kesten S. Improvements with tiotropium in COPD patients with concomitant asthma. Respir Med. 2008;102(1):50–56. doi: 10.1016/j.rmed.2007.08.003. [DOI] [PubMed] [Google Scholar]
- 55.Rodrigo G.J., Neffen H., Castro-Rodriguez J.A. Efficacy and safety of subcutaneous omalizumab vs placebo as add-on therapy to corticosteroids for children and adults with asthma: a systematic review. Chest. 2011;139(1):28–35. doi: 10.1378/chest.10-1194. [DOI] [PubMed] [Google Scholar]
- 56.Yancey S.W., Ortega H.G., Keene O.N. Meta-analysis of asthma-related hospitalization in mepolizumab studies of severe eosinophilic asthma. J Allergy Clin Immunol. 2017;139(4):1167–1175.e2. doi: 10.1016/j.jaci.2016.08.008. [DOI] [PubMed] [Google Scholar]
- 57.Christenson S.A., Steiling K., van den Berge M. Asthma-COPD overlap. Clinical relevance of genomic signatures of type 2 inflammation in chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2015;191(7):758–766. doi: 10.1164/rccm.201408-1458OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Maltby S., Gibson P.G., Powell H., McDonald V.M. Omalizumab treatment response in a population with severe allergic asthma and overlapping COPD. Chest. 2017;151(1):78–89. doi: 10.1016/j.chest.2016.09.035. [DOI] [PubMed] [Google Scholar]
- 59.Hanania H., Chipps B., Griffin N., Trzaskoma B., Iqbal A., Casale T. Decreased exacerbations and improvement in asthma symptom control in asthma COPD overlap treated with omalizumab: data from the Prospero cohort study. Chest. 2017;152 Supp A776. [Google Scholar]
- 60.Brightling C.E., Bleecker E.R., Panettieri R.A., Jr. Benralizumab for chronic obstructive pulmonary disease and sputum eosinophilia: a randomised, double-blind, placebo-controlled, phase 2A study. Lancet Respir Med. 2014;2(11):891–901. doi: 10.1016/S2213-2600(14)70187-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Pavord I.D., Chanez P., Criner G.J. Mepolizumab for eosinophilic chronic obstructive pulmonary disease. N Engl J Med. 2017;377(17):1613–1629. doi: 10.1056/NEJMoa1708208. [DOI] [PubMed] [Google Scholar]
- 62.Martinez F.J., Rabe K.F., Sethi S. Effect of Roflumilast and Inhaled Corticosteroid/Long-Acting β2-Agonist on Chronic Obstructive Pulmonary Disease Exacerbations (RE(2)SPOND). A randomized clinical trial. Am J Respir Crit Care Med. 2016;194(5):559–567. doi: 10.1164/rccm.201607-1349OC. [DOI] [PubMed] [Google Scholar]
- 63.Martinez F.J., Calverley P.M., Goehring U.M., Brose M., Fabbri L.M., Rabe K.F. Effect of roflumilast on exacerbations in patients with severe chronic obstructive pulmonary disease uncontrolled by combination therapy (REACT): a multicentre randomised controlled trial. Lancet. 2015;385(9971):857–866. doi: 10.1016/S0140-6736(14)62410-7. [DOI] [PubMed] [Google Scholar]
- 64.Meltzer E.O., Chervinsky P., Busse W. Roflumilast for asthma: efficacy findings in placebo-controlled studies. Pulm Pharmacol Ther. 2015;35(suppl):S20–S27. doi: 10.1016/j.pupt.2015.10.006. [DOI] [PubMed] [Google Scholar]
- 65.Bateman E.D., Goehring U.M., Richard F., Watz H. Roflumilast combined with montelukast versus montelukast alone as add-on treatment in patients with moderate-to-severe asthma. J Allergy Clin Immunol. 2016;138(1):142–149. doi: 10.1016/j.jaci.2015.11.035. [DOI] [PubMed] [Google Scholar]
- 66.Chervinsky P., Meltzer E.O., Busse W. Roflumilast for asthma: safety findings from a pooled analysis of ten clinical studies. Pulm Pharmacol Ther. 2015;35(suppl):S28–S34. doi: 10.1016/j.pupt.2015.11.003. [DOI] [PubMed] [Google Scholar]
- 67.Albert R.K., Connett J., Bailey W.C. Azithromycin for prevention of exacerbations of COPD. N Engl J Med. 2011;365(8):689–698. doi: 10.1056/NEJMoa1104623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Han M.K., Tayob N., Murray S. Predictors of chronic obstructive pulmonary disease exacerbation reduction in response to daily azithromycin therapy. Am J Respir Crit Care Med. 2014;189(12):1503–1508. doi: 10.1164/rccm.201402-0207OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Brusselle G.G., Vanderstichele C., Jordens P. Azithromycin for prevention of exacerbations in severe asthma (AZISAST): a multicentre randomised double-blind placebo-controlled trial. Thorax. 2013;68(4):322–329. doi: 10.1136/thoraxjnl-2012-202698. [DOI] [PubMed] [Google Scholar]
- 70.Simpson J.L., Powell H., Boyle M.J., Scott R.J., Gibson P.G. Clarithromycin targets neutrophilic airway inflammation in refractory asthma. Am J Respir Crit Care Med. 2008;177(2):148–155. doi: 10.1164/rccm.200707-1134OC. [DOI] [PubMed] [Google Scholar]
- 71.Kew K.M., Undela K., Kotortsi I., Ferrara G. Macrolides for chronic asthma. Cochrane Database Syst Rev. 2015;(9) doi: 10.1002/14651858.CD002997.pub4. [DOI] [PubMed] [Google Scholar]
- 72.Gibson P.G., Yang I.A., Upham J.W. Effect of azithromycin on asthma exacerbations and quality of life in adults with persistent uncontrolled asthma (AMAZES): a randomised, double-blind, placebo-controlled trial. Lancet. 2017;390(10095):659–668. doi: 10.1016/S0140-6736(17)31281-3. [DOI] [PubMed] [Google Scholar]
- 73.Ghebre M.A., Pang P.H., Diver S. Biological exacerbation clusters demonstrate asthma and COPD overlap with distinct mediator and microbiome profiles. J Allergy Clin Immunol. 2018;141(6):2027–2036. doi: 10.1016/j.jaci.2018.04.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Barnes P.J. Therapeutic approaches to asthma-chronic obstructive pulmonary disease overlap syndromes. J Allergy Clin Immunol. 2015;136(3):531–545. doi: 10.1016/j.jaci.2015.05.052. [DOI] [PubMed] [Google Scholar]
- 75.Cosío B.G., Pérez de Llano L., Lopez Viña A. Th-2 signature in chronic airway diseases: Towards the extinction of asthma-COPD overlap syndrome? Eur Respir J. 2017;49(5) doi: 10.1183/13993003.02397-2016. [DOI] [PubMed] [Google Scholar]
- 76.Singh D., Kolsum U., Brightling C.E., Locantore N., Agusti A., Tal-Singer R. Eosinophilic inflammation in COPD: prevalence and clinical characteristics. Eur Respir J. 2014;44(6):1697–1700. doi: 10.1183/09031936.00162414. [DOI] [PubMed] [Google Scholar]
- 77.Oshagbemi O.A., Burden A.M., Braeken D.C.W. Stability of blood eosinophils in patients with chronic obstructive pulmonary disease and in control subjects, and the impact of sex, age, smoking, and baseline counts. Am J Respir Crit Care Med. 2017;195(10):1402–1404. doi: 10.1164/rccm.201701-0009LE. [DOI] [PubMed] [Google Scholar]
- 78.Siddiqui S.H., Guasconi A., Vestbo J. Blood eosinophils: a biomarker of response to extrafine beclomethasone/formoterol in chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2015;192(4):523–525. doi: 10.1164/rccm.201502-0235LE. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Pascoe S., Locantore N., Dransfield M.T., Barnes N.C., Pavord I.D. Blood eosinophil counts, exacerbations, and response to the addition of inhaled fluticasone furoate to vilanterol in patients with chronic obstructive pulmonary disease: a secondary analysis of data from two parallel randomised controlled trials. Lancet Respir Med. 2015;3(6):435–442. doi: 10.1016/S2213-2600(15)00106-X. [DOI] [PubMed] [Google Scholar]
- 80.Pavord I.D., Lettis S., Locantore N. Blood eosinophils and inhaled corticosteroid/long-acting β-2 agonist efficacy in COPD. Thorax. 2016;71(2):118–125. doi: 10.1136/thoraxjnl-2015-207021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Barnes N.C., Sharma R., Lettis S., Calverley P.M. Blood eosinophils as a marker of response to inhaled corticosteroids in COPD. Eur Respir J. 2016;47(5):1374–1382. doi: 10.1183/13993003.01370-2015. [DOI] [PubMed] [Google Scholar]
- 82.Roche N., Chapman K.R., Vogelmeier C.F. Blood eosinophils and response to maintenance chronic obstructive pulmonary disease treatment. Data from the FLAME trial. Am J Respir Crit Care Med. 2017;195(9):1189–1197. doi: 10.1164/rccm.201701-0193OC. [DOI] [PubMed] [Google Scholar]
- 83.Hastie A.T., Martinez F.J., Curtis J.L. Association of sputum and blood eosinophil concentrations with clinical measures of COPD severity: an analysis of the SPIROMICS cohort. Lancet Respir Med. 2017;5(12):956–967. doi: 10.1016/S2213-2600(17)30432-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Vedel-Krogh S., Nordestgaard B.G., Lange P., Vestbo J., Nielsen S.F. Blood eosinophil count and risk of pneumonia hospitalisations in individuals with COPD. Eur Respir J. 2018;51(5) doi: 10.1183/13993003.00120-2018. [DOI] [PubMed] [Google Scholar]
- 85.Watz H., Tetzlaff K., Wouters E.F. Blood eosinophil count and exacerbations in severe chronic obstructive pulmonary disease after withdrawal of inhaled corticosteroids: a post-hoc analysis of the WISDOM trial. Lancet Respir Med. 2016;4(5):390–398. doi: 10.1016/S2213-2600(16)00100-4. [DOI] [PubMed] [Google Scholar]
- 86.Park H.Y., Lee H., Koh W.J. Association of blood eosinophils and plasma periostin with FEV1 response after 3-month inhaled corticosteroid and long-acting beta2-agonist treatment in stable COPD patients. Int J Chron Obstruct Pulmon Dis. 2015;11:23–30. doi: 10.2147/COPD.S94797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Hastie A.T., Moore W.C., Meyers D.A. Analyses of asthma severity phenotypes and inflammatory proteins in subjects stratified by sputum granulocytes. J Allergy Clin Immunol. 2010;125(5):1028–1036. doi: 10.1016/j.jaci.2010.02.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Hanania N.A., Wenzel S., Rosén K. Exploring the effects of omalizumab in allergic asthma: an analysis of biomarkers in the EXTRA study. Am J Respir Crit Care Med. 2013;187(8):804–811. doi: 10.1164/rccm.201208-1414OC. [DOI] [PubMed] [Google Scholar]
- 89.Celli B.R., Decramer M., Wedzicha J.A. An official American Thoracic Society/European Respiratory Society statement: research questions in COPD. Am J Respir Crit Care Med. 2015;191(7):e4–e27. doi: 10.1164/rccm.201501-0044ST. [DOI] [PubMed] [Google Scholar]