Abstract
Purpose
Low 25-hydroxyvitamin D (25[OH]D) concentrations have been associated with adverse outcomes in selected populations with established chronic heart failure (CHF). However, it remains unclear whether 25[OH]D deficiency is associated with mortality and hospitalisation in unselected patients receiving contemporary medical and device therapy for CHF.
Methods
We prospectively examined the prevalence and correlates of 25[OH]D deficiency in 1802 ambulatory patients with CHF due to left ventricular systolic dysfunction (left ventricular ejection fraction ≤ 45%) attending heart failure clinics in the north of England.
Results
73% of patients were deficient in 25[OH]D (< 50 nmol/L). 25[OH]D deficiency was associated with male sex, diabetes, lower serum sodium, higher heart rate, and greater diuretic requirement. During a mean follow-up period of 4 years, each 2.72-fold increment in 25[OH]D concentration (for example from 32 to 87 nmol/L) is associated with 14% lower all-cause mortality (95% confidence interval (CI) 1, 26%; p = 0.04), after accounting for potential confounding factors.
Conclusions
Low 25-hydroxyvitamin D deficiency is associated with increased mortality in patients with chronic heart failure due to left ventricular systolic dysfunction. Whether vitamin D supplementation will improve outcomes is, as yet, unproven.
Electronic supplementary material
The online version of this article (10.1007/s00394-018-1806-y) contains supplementary material, which is available to authorized users.
Keywords: Vitamin D, Chronic heart failure, Mortality
Introduction
Vitamin D is receiving increasing attention as epidemiological evidence links it to many chronic illnesses. Low serum 25-hydroxyvitamin D (25[OH]D) concentrations have been associated with an increased risk of adverse outcomes generally, [1, 2] but especially in patients with suspected coronary artery disease, hypertension and chronic heart failure (CHF) [3–10]. However, published data pertaining to CHF are derived from registry data, or somewhat selected cohorts such as patients referred for coronary angiography, on a waiting list for cardiac transplantation or in-patients. Vitamin D has multiple effects throughout the body that may be of particular relevance in people with CHF [11]. We have recently described in a randomised, placebo-controlled trial in unselected ambulatory patients with CHF and 25[OH]D deficiency that vitamin D3 supplementation (100 mcg (4000 IU) daily) improves left ventricular structure and function [12]. Whether 25[OH]D deficiency independently predicts outcomes in unselected patients with CHF remains unclear.
In a prospective cohort study, we examined the prevalence and correlates of 25[OH]D deficiency and whether 25[OH]D levels were related to hospitalisation and mortality outcomes in unselected patients referred to heart failure clinics in the north of England.
Methods
As described in our previous publications [13, 14], adult patients (> 18 years) were eligible to take part in the cohort study if they had stable symptoms and signs of CHF for 3 months, in addition to echocardiographic evidence of left ventricular systolic dysfunction (left ventricular (LV) ejection fraction (LVEF) ≤ 45%). Between June 2006 and July 2014, we recruited patients from the outpatient heart failure clinics of four hospitals in the north of England. Ethical approval was provided by Leeds West Research Ethics Committee (07/Q1205/17), and all patients provided written informed consent to participate.
Baseline assessment
All patients underwent resting 12-lead electrocardiographs, and blood testing for measurement of full blood count, electrolytes, serum creatinine and serum 25(OH)D2 and 25(OH)D3 concentration. Estimated glomerular filtration rate (eGFR) was calculated using the Modification of Diet in Renal Disease method [15]. Functional status was assessed using the New York Heart Association (NYHA) classification. Two-dimensional transthoracic echocardiography was performed in all participants by sonographers blinded to patient characteristics; left ventricular (LV) ejection fraction (LVEF) was calculated according to the Simpson’s biplane method. Doses of diuretic therapy, angiotensin-converting enzyme inhibitors (ACEi), angiotensin receptor blockers (ARB), and beta-blockers were normalised to maximum licensed CHF dose as previously described [10].
25[OH]D concentration
Serum 25(OH)D2 and 25(OH)D3 concentrations were analysed by tandem mass spectrometry. Samples were prepared using a protein precipitation reagent containing deuterated 25(OH)D3. The supernatant was analysed on an API5000 LC-MS/MS (AB SCIEX, Warrington, UK) in APCI mode. The inter-assay CV was < 10% at all concentrations ranging from 12 to 159 nmol/L. 25(OH)D2 and D3 concentrations were summed and reported as 25(OH)D. We defined deficiency of 25[OH]D concentrations based upon the threshold outlined by the Endocrine Society calling 25[OH]D < 50 nmol/L deficient [16].
Hospitalisation and mortality
The nature and duration of non-elective hospitalisation within the first year after enrolment was determined using hospital databases. Each hospitalisation was sub-classified independently by two cardiologists as cardiovascular if the principal presenting complaint was related to cardiac, cerebrovascular or peripheral vascular disease; consensus was sought in all initial cases of disagreement. Cardiovascular admissions were further sub-classified as heart failure related if the patient presented with symptoms and signs of heart failure and evidence of fluid overload requiring intravenous diuretic therapy for at least 24 h. All patients were registered with the United Kingdom Office of National Statistics (ONS) to provide details of death until the censoring date of 8th May 2016.
Statistical methods
Analyses were conducted in Stata (StataCorp. 2015. Stata Statistical Software: Release 14. College Station, TX). All significance tests were two sided and called significant at the 5% level. Continuous data are expressed as mean (SEM) or median (IQR) depending on normality of distribution, and categorical variables as n (%).
Odds ratios (binary logistic regression) and hazard ratios (Cox proportional hazards regression) were derived for the association between 25[OH]D and hospitalisation or mortality, respectively. 25[OH]D concentrations were natural log transformed during these analyses to account for their non-normal distribution. After defining crude (unadjusted) associations, models were used to adjust for patient and clinical demographics (age, sex, month and year of recruitment, sodium, eGFR, albumin, log transformed QRS interval, NYHA class, LVEF, LV end diastolic dimension), comorbidities and aetiology (diabetes mellitus, chronic obstructive pulmonary disease (COPD), ischaemic aetiology) and treatment (ramipril dose, bisoprolol dose, furosemide dose and device therapy).
Multiple imputation by chained equations, under the missing at random assumption, was used to impute 30 sets of data to minimise any potential bias caused by missing data (see Table 1) [17, 18]. The imputation model specification was such that it included all variables included in the analysis model, including outcome variables (hospitalisation, Nelson–Aalen survival estimator and censoring indicator [19]), as well as auxiliary variables as indicated in supplementary table 1, following previously defined methods [20]. Results presented in the manuscript contain estimates averaged over 30 imputed data sets, according to Rubin’s rules, and were compared to complete case analyses (i.e. without imputation), presented in supplementary tables 2 and 3, to check for consistency.
Table 1.
Variables according to 25[OH]D concentration (≥/<50 nmol/l)
| Whole cohort | 25[OH]D < 50 | 25[OH]D ≥ 50 | p value | Missing | |
|---|---|---|---|---|---|
| n = 1802 | n = 914 | n = 338 | 25[OH]D groups | n (%) | |
| Age (years) | 69.6 (12.5) | 69.9 (12.7) | 71.2 (11.2) | 0.07 | 0 (0) |
| Heart rate (bpm) | 75.3 (17.9) | 76.4 (18.3) | 73.6 (17.2) | 0.017 | 158 (8.8) |
| QRS interval (ms) | 123.2 (31) | 123.8 (31.4) | 123.7 (31.4) | 0.94 | 157 (8.7) |
| Haemoglobin (g/dl) | 13.5 (1.8) | 13.5 (1.8) | 13.4 (1.8) | 0.35 | 20 (1.1) |
| Sodium (mmol/L) | 139.4 (3.4) | 139.2 (3.5) | 139.9 (3) | 0.001 | 4 (0.2) |
| eGFR (ml/kg/1.73 m2) | 57.8 (19.7) | 58.5 (19.5) | 57.3 (20.7) | 0.36 | 8 (0.4) |
| Albumin (g/l) | 43.1 (11.1) | 42.8 (3.7) | 43.1 (3.8) | 0.29 | 56 (3.1) |
| LV end diastolic dimension (mm) | 57.2 (8.9) | 57 (8.6) | 56.7 (8.9) | 0.53 | 71 (3.9) |
| LV ejection fraction (%) | 32 (9.5) | 31.6 (9.3) | 32.2 (9.4) | 0.26 | 45 (2.5) |
| Ramipril dose (mg/day) | 4.9 (3.5) | 4.8 (3.6) | 5 (3.5) | 0.37 | 5 (0.3) |
| Bisoprolol dose (mg/day) | 3.9 (3.4) | 4 (3.4) | 4 (3.3) | 0.89 | 5 (0.3) |
| Prescribed ACEi/ARB (n, %) | 1626 (90.4) | 818 (89.7) | 309 (91.4) | 0.36 | 5 (0.3) |
| Prescribed beta-blocker (n, %) | 1523 (84.7) | 782 (85.7) | 289 (85.5) | 0.91 | 5 (0.3) |
| Prescribed mineralocorticoid receptor antagonist (n, %) | 689 (38.2) | 367 (40.2) | 111 (32.8) | 0.017 | 5 (0.3) |
| Furosemide (mg/day) | 51.2 (1.2) | 54.4 (1.6) | 44.1 (2.6) | 0.001 | 5 (0.3) |
| Male sex (n, %) | 1319 (73.2) | 678 (74.2) | 230 (68) | 0.031 | 0 (0) |
| Ischaemic aetiology (n, %) | 1067 (59.2) | 544 (59.5) | 187 (55.3) | 0.18 | 0 (0) |
| Diabetes (n, %) | 504 (28) | 287 (31.4) | 77 (22.8) | 0.003 | 0 (0) |
| COPD (n, %) | 284 (15.8) | 144 (15.8) | 52 (15.4) | 0.87 | 0 (0) |
| Device therapy (n, %) | 504 (28) | 247 (27) | 109 (32.2) | 0.07 | 0 (0) |
| NYHA class 1 (n, %) | 333 (18.5) | 132 (14.4) | 66 (19.5) | 0.05 | 2 (0.1) |
| 2 | 912 (50.7) | 478 (52.3) | 179 (53) | ||
| 3 | 534 (29.7) | 294 (32.2) | 92 (27.2) | ||
| 4 | 21 (1.2) | 10 (1.1) | 1 (0.3) |
Continuous data all as mean (SD), categorical data are n, %
eGFR estimated glomerular filtration rate, LV left ventricular, COPD chronic obstructive pulmonary disease, NYHA New York Heart Association Class
Results
A total of 1802 patients were recruited to the study [mean (SEM) age 69.6 (0.3) and 1319 (73.2%) male]. Median (IQR) 25[OH]D concentration within the 1252 patients with available data was 32.1 (20–52) nmol/L. Few patients (9.1%) were sufficient in 25[OH]D (≥ 75 nmol/L) at baseline (Fig. 1), and 914 (73%) were deficient (< 50 nmol/L). Although 25[OH]D concentrations varied throughout the year (Fig. 2), even during months with greater daylight, median concentrations remained in the deficiency range (April–September − 35.4 (21.9–56) nmol/L, and October–March − 30 (19.9–48) nmol/L; p = 0.001 by Mann–Whitney test).
Fig. 1.
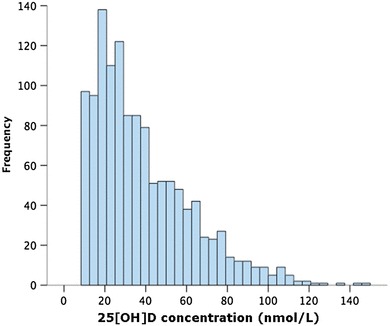
25[OH]D concentrations in patients with chronic heart failure. Distribution of 25(OH)D concentrations in 1252 patients, indicating deficiency (< 50 nmol/L) in 73%
Fig. 2.
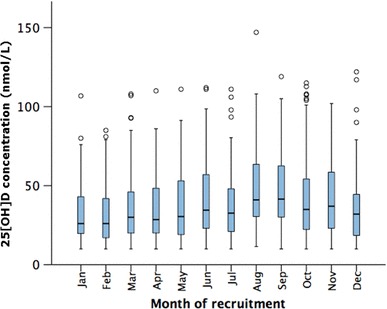
25[OH]D concentrations in patients with chronic heart failure. Monthly variation of 25[OH]D concentration (median and interquartile range) in 1252 patients. Boxes represent median and interquartile range (IQR), with whiskers denoting 1.5 × IQR and circles outliers beyond this range
Patients deficient in 25[OH]D were more likely to be male, and have diabetes, whilst the distribution of heart failure aetiology was not different between the groups (Table 1). Deficient patients also had higher heart rates, lower serum sodium levels, and a greater furosemide requirement. Median 25[OH]D concentrations were significantly related to symptoms: 36.4 (22–57), 33.0 (21–52), 28.5 (19.8–48) and 18.4 (10–42)nmol/L for NYHA classes I–IV, respectively, (p = 0.0005 by Kruskal–Wallis test).
Hospitalisation and mortality outcomes
Using routine data collection, we had 100% follow-up. Table 2 describes the association of natural log-transformed 25[OH]D concentration with hospitalisation during the first year of follow-up, both before and after adjustment for potential confounding factors. Although 25[OH]D concentrations were associated with heart failure-specific, cardiovascular and all-cause hospitalisation in unadjusted analyses, there was no association in all cases after accounting for potential confounding factors. The same conclusions were reached when analyses were repeated without multiple imputation (supplementary table 2).
Table 2.
Association of 25[OH]D with hospitalisation at 1 year (logistic regression analysis) after multiple imputation
| Model | OR** | Lower 95% CI | Upper 95% CI | p value |
|---|---|---|---|---|
| Heart failure hospitalisation (n = 112, 6.2%) | ||||
| Unadjusted | 0.67 | 0.46 | 0.97 | 0.034 |
| Adjusted for patient and clinical demographicsa | 0.76 | 0.52 | 1.12 | 0.165 |
| Adjusted for patient and clinical demographics, and comorbidities and aetiologyb | 0.79 | 0.54 | 1.16 | 0.223 |
| Adjusted for patient and clinical demographics, comorbidities and aetiology, and treatmentc | 0.80 | 0.54 | 1.19 | 0.270 |
| Cardiovascular hospitalisation (n = 227, 12.6%) | ||||
| Unadjusted | 0.78 | 0.59 | 1.01 | 0.061 |
| Adjusted for patient and clinical demographicsa | 0.84 | 0.64 | 1.10 | 0.202 |
| Adjusted for patient and clinical demographics, and comorbidities and aetiologyb | 0.86 | 0.65 | 1.13 | 0.280 |
| Adjusted for patient and clinical demographics, comorbidities and aetiology, and treatmentc | 0.87 | 0.66 | 1.15 | 0.330 |
| All non-elective hospitalisations (457, 25.4%) | ||||
| Unadjusted | 0.78 | 0.64 | 0.95 | 0.011 |
| Adjusted for patient and clinical demographicsa | 0.84 | 0.68 | 1.03 | 0.086 |
| Adjusted for patient and clinical demographics, and comorbidities and aetiologyb | 0.85 | 0.69 | 1.04 | 0.111 |
| Adjusted for patient and clinical demographics, comorbidities and aetiology, and treatmentc | 0.86 | 0.70 | 1.05 | 0.139 |
Multiple imputation by chained equations was performed with 30 imputations and 20 iterations, and all model estimates are averaged over all imputed datasets
**OR per 2.72-fold increase in 25[OH]D (due to natural log transformation to achieve normality)
aIncluding age, sex, month and year of recruitment, sodium, eGFR, albumin, log-transformed QRS interval, NYHA class, LV ejection fraction, LV end diastolic dimension
bDiabetes, COPD, ischaemic aetiology
cRamipril dose, bisoprolol dose, furosemide dose, and device therapy
The association between natural log-transformed 25[OH]D and mortality was significant in all of the unadjusted and adjusted analyses (Table 3) with a 19% lower mortality in non-deficient patients. These analyses suggest that even after accounting for all other variables described in Table 1, a 2.72-fold increase in 25[OH]D (for example, an increase of 25[OH]D from 32 to 87 nmol/L) is associated with a 14% lower risk of death. Broadly similar effect size and statistical significance were noted when repeating analyses without multiple imputation (supplementary table 3). Adjusted survival curves with 95% confidence intervals for 25[OH]D deficient and non-deficient patients are shown in Fig. 3, again revealing significantly higher mortality in deficient patients (HR 1.24, 95% CI 1.05, 1.46).
Table 3.
Association of 25[OH]D with all-cause mortality using Cox proportional hazards modelling after multiple imputation
| Model | HRa | Lower 95% CI | Upper 95% CI | p value |
|---|---|---|---|---|
| All-cause mortality (n = 737, 40.9%) | ||||
| Unadjusted | 0.79 | 0.69 | 0.91 | 0.001 |
| Adjusted for patient and clinical demographicsb | 0.83 | 0.72 | 0.96 | 0.011 |
| Adjusted for patient and clinical demographics, and comorbidities and aetiologyc | 0.84 | 0.72 | 0.97 | 0.016 |
| Adjusted for patient and clinical demographics, comorbidities and aetiology, and treatmentd | 0.86 | 0.74 | 0.99 | 0.042 |
Multiple imputation by chained equations was performed with 30 imputations and 20 iterations, and all model estimates are averaged over all imputed datasets
aHR per 2.72-fold increase in 25[OH]D (due to natural log transformation to achieve normality)
bIncluding age, sex, month and year of recruitment, sodium, eGFR, albumin, log-transformed QRS interval, NYHA class, LV ejection fraction, LV end diastolic dimension
cDiabetes, COPD, Ischaemic aetiology
dRamipril dose, bisoprolol dose, furosemide dose, and device therapy
Fig. 3.
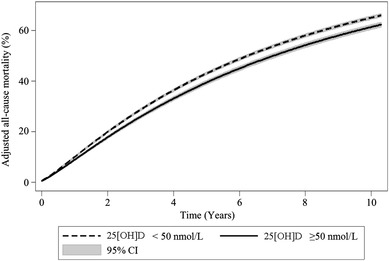
Adjusted survival according to 25[OH]D status. Adjusted survival curves according to 25[OH]D status (deficient and adequate) after multiple imputation, showing that after accounting patient and clinical demographics, comorbidities and aetiology, and treatment factors, 25[OH]D deficiency is associated with decreased survival (HR 1.24, 95% CI 1.05–1.46)
Discussion
Our data from a large prospectively collected cohort of unselected ambulatory patients with CHF suggest that 25[OH]D deficiency (25[OH]D < 50 nmol/L) is highly prevalent, persists during summer and autumn, and is an independent predictor of increased mortality. After adjusting for multiple confounders, the outcome data from the cohort study are consistent with previous smaller reports in ambulatory patients [9, 21, 22], hospitalised patients [10] and registry data [3, 6, 23], and reveal increased mortality in patients with low 25[OH]D concentrations compared with those without. In all of these previous studies, 25[OH]D deficiency was less frequent than in our cohort ranging from 28 to 75% [10, 24], whilst overall mortality of the cohorts was higher. Our cohort study was prospectively designed to assess predictors of outcome in an unselected, consecutive group of patients with heart failure due to left ventricular systolic dysfunction on optimal contemporary medical and device therapy, and provides the strongest evidence to date that a low 25[OH]D concentration is an independent predictor of mortality.
Why might vitamin D be important in chronic heart failure?
Clinical evidence linking micronutrients and cardiovascular disease overall remains neutral [24]. Vitamin D, on the other hand, has multiple effects on the cardiovascular system [25] that might be especially pertinent to the heart failure syndrome [9, 26, 27].
Abnormalities of the vitamin D–parathyroid (PTH) axis have a direct effect upon a wide range of mammalian cells including cardiomyocytes. Through increased urinary excretion of calcium and magnesium, enhanced by loop diuretic use [28, 29], elevated aldosterone levels drive PTH release. This response is exacerbated in people with 25[OH]D deficiency [30, 31]. The consequences of 25[OH]D deficiency [32] and elevated PTH levels [33–36] are calcium loading, with cardiomyocyte and skeletal muscle contractile dysfunction, cellular hypertrophy, oxidative stress, immune activation, endothelial dysfunction (including enhanced endothelin-1 release) [30, 34, 37–41]. These influences are reflected clinically with an increased risk of hospitalisation [7, 42], and worsening renal function [43], whilst vitamin D supplementation may be associated with a reduction of plasma renin and aldosterone levels [44, 45]. Consistent with these observations, we found lower mean sodium levels and higher mean heart rates in 25[OH]D-deficient heart failure patients. Hence, the beneficial remodelling seen in VINDICATE [9], and the adverse effects of 25[OH]D deficiency on outcomes demonstrated in the present report might reflect the interaction of vitamin D with several of the contributory pathophysiological pathways specific to CHF due to left ventricular systolic dysfunction.
Vitamin D as an intervention
A recent Cochrane review of 56 randomised trials with ~ 95 K participants (> 99% healthy volunteers) showed that vitamin D3 (given for mean 4.4 years) decreased all-cause mortality (RR 0.94, 95% CI 0.91–0.98). Vitamin D2 had no effect. The authors called for more research in non-healthy populations [46]. A further systematic review and meta-analysis of observational and interventional studies suggested that vitamin D3 supplementation might reduce mortality [47], with particular benefit in CHF patients. A subsequent trial analysis, systematic review and meta-analysis suggest that vitamin D supplementation might protect against fatal and non-fatal CHFs in older, healthy people (HR 0.75, 95% CI 0.58–0.97), but not against MI or stroke [48]. The most recent meta-analysis on non-skeletal effects of vitamin D supplementation demonstrates that most trials have been done in subjects without low 25[OH]D levels limiting the credibility of the argument that vitamin D supplementation has no potential benefit on outcomes in people with 25[OH]D insufficiency [1].
The Vitamin D Assessment study (ViDA) in 5100 healthy subjects, aged > 50 years, recently reported that 100,000 IU oral vitamin D3 monthly was neutral for the prevention of CV disease [49, 50], with a low rate of 25[OH]D deficiency in participants (25%), a lower than expected endpoint rate and monthly doses cited as possible reasons [51]. Meta-analysis and data from ViDA and other studies describe that benefits on clinical outcomes are greatest in the most deficient, and in studies using daily dosing regimens (as in VINDICATE) rather than monthly [52–55].
Early trials of vitamin D supplementation in CHF were inconclusive possibly due to inclusion criteria, calcium-based placebo, dosing regimen, use of vitamin D2 (rather than D3) and heterogeneous CHF population [56–58], limiting their ability to provide clarity of benefit [25], whilst no trials have shown any adverse safety signals including up to 10,000 IU daily [59]. Recent meta-analyses have suggested that vitamin D might reduce inflammation (assessed by tumour necrosis factor-alpha levels (TNF-α) [60]), but that it is neutral for heart function [61]. However, the latter analysis did not include data from the VINDICATE paper which presented data from two independent randomised, placebo-controlled, double-blind, parallel group studies using two imaging modalities demonstrating consistent improvements in left ventricular structure and function [9].
The single-centre ‘Effect of Vitamin D on all-cause mortality in heart failure’ (EVITA) study is the only study of patient-orientated outcomes in CHF that has been reported. EVITA was neutral for mortality and heart failure hospitalisation [62]. EVITA recruited only 400 of an intended 950 subjects (which would have given 80% power to detect a 36% reduction in total mortality) and had a drop-out rate of 42% at 3 years. Moreover, the median age of participants was 54yrs, with an unusually high (80%) device therapy rate, the study included people who were sufficient in 25[OH]D (concentrations 50–75 mmol/L) and the group assigned vitamin D supplementation had significantly worse renal function and was 2 years older. In contrast, participants in our two studies and the present cohort study were ambulant outpatients, mean age 70 years with a more typical device therapy rate (30%) and all had 25[OH]D concentrations < 50 nmol/L [12].
Limitations
Although the results from our cohort study are significant despite the inclusion of numerous potential confounders in the models presented, our cohort outcome data remain observational. We accept, therefore, that it is possible that we have not accounted for all relevant confounding variables including, for example, social background and lifestyle that are difficult to measure. For example, the main source of vitamin D is not nutritional [63], rather the result of skin sunlight exposure. Hence, patients with chronic disease, immobility and the elderly (who require more sun exposure to make the same amount of vitamin D as younger individuals) are at higher risk of 25[OH]D deficiency since they spend less time outdoors [64]. 25[OH]D deficiency could, therefore, merely be a bystander, a marker of chronic disease or frailty, the result of limited sun exposure. This is, however, a limitation of all of the studies carried out so far and is countered by VINDICATE in which we observed important beneficial left ventricular remodelling with 12 months of vitamin D supplementation [9].
A potential limitation of our study is that although the blood of 25[OH]D vitamin D across centres was assessed using an accepted and validated process, we did not standardise samples across centres. However, decisions on intervention in the form of vitamin D supplementation for CHF patients would be based upon results from local or regional services, abrogating the relevance of this limitation somewhat in real-world practice.
The headline results from the cohort study include data achieved through a robust and recognised multiple imputation process, which aims to reduce the bias introduced by excluding patients with missing 25[OH]D data. The results of the complete case analysis (i.e. without using multiple imputation) are presented in the supplementary materials and describe similar hazard ratios for mortality to those derived from analyses using multiple imputation.
Conclusion
Even after accounting for potential confounding factors, CHF patients with 25[OH]D sufficiency have a lower risk of all-cause mortality. In conjunction with our recent finding that vitamin D supplementation leads to beneficial cardiac remodelling, these data support the need for a longer term, fully recruited, randomised placebo-controlled study, with ‘hard’ clinical endpoints, of high-dose vitamin D3 supplementation in patients with CHF due to left ventricular systolic dysfunction.
Clinical perspectives
25[OH]D deficiency (< 50 nmol/L) is common in patients with chronic heart failure and persists throughout the year. 25[OH]D deficiency is an independent predictor of higher mortality in patients with CHF on optimal medical and device therapy.
Implications
Based upon these data and our previous work demonstrating improvements in cardiac function, it is possible that vitamin D3 supplementation could improve outcomes in patients with heart failure due to left ventricular systolic dysfunction.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Acknowledgements
The cohort study work was funded by the British Heart Foundation (PG/08/020/24617). KKW holds an NIHR Clinician Scientist Award. JG holds an NIHR-HCS Fellowship Award. JEL is funded by a Leeds Charitable Foundation Fellowship. MH is funded by a Wellcome Trust Sir Henry Wellcome Fellowship. MP holds an NIHR-ICA-DRF Fellowship Award. RMC holds a British Heart Foundation Intermediate Fellowship. MTK is a British Heart Foundation Professor of Cardiology. The authors acknowledge the consistent administrative support provided by Mrs Andrea Marchant and additional statistical advice from Dr David Cairns of the Leeds Clinical Trials Research Unit. This research took place in the National Institute for Health Research Leeds Cardiovascular Clinical Research Facility at Leeds Teaching Hospitals NHS Trust.
Abbreviations
- CHF
Chronic heart failure
- LV
Left ventricular
- LVEF
Left ventricular ejection fraction
- eGFR
Estimated glomerular filtration rate
- NYHA
New York Heart Association
- ACEi
Angiotensin-converting enzyme inhibitor
- ARB
Angiotensin receptor blocker
- ONS
Office of National Statistics
- CI
Confidence interval
- HR
Hazard ratio
- VINDICATE study
Vitamin D treating patients with chronic heart failure
- EVITA study
Effect of Vitamin D on all-cause mortality in heart failure study
- VIDA study
Vitamin D assessment study
- IQR
Interquartile range
- PTH
Parathyroid hormone
Author contributions
KW researched the topic and devised the study. He and RC provided the first draft of the manuscript. RC collated the database and RC and MH undertook primary statistical analysis. All other co-authors contributed equally to data collection and manuscript preparation.
Compliance with ethical standards
Conflict of interest
There are no conflicts of interest for any authors.
References
- 1.Rejnmark L, Bislev LS, Cashman KD, Eiríksdottir G, Gaksch M, Grübler M, Grimnes G, Gudnason V, Lips P, Pilz S, van Schoor NM, Kiely M, Jorde R. Non-skeletal health effects of vitamin D supplementation: a systematic review on findings from meta-analyses summarizing trial data. PLoS One. 2017;12:e0180512. doi: 10.1371/journal.pone.0180512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Gaksch M, Jorde R, Grimnes G, Joakimsen R, Schirmer H, Wilsgaard T, Mathiesen EB, Njølstad I, Løchen ML, März W, Kleber ME, Tomaschitz A, Grübler M, Eiriksdottir G, Gudmundsson EF, Harris TB, Cotch MF, Aspelund T, Gudnason V, Rutters F, Beulens JW, van ‘t Riet E, Nijpels G, Dekker JM, Grove-Laugesen D, Rejnmark L, Busch MA, Mensink GB, Scheidt-Nave C, Thamm M, Swart KM, Brouwer IA, Lips P, van Schoor NM, Sempos CT, Durazo-Arvizu RA, Škrabáková Z, Dowling KG, Cashman KD, Kiely M, Pilz S. Vitamin D and mortality: Individual participant data meta-analysis of standardized 25-hydroxyvitamin D in 26916 individuals from a European consortium. PLoS One. 2017;12:e0170791. doi: 10.1371/journal.pone.0170791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Anderson JL, May HT, Horne BD, Bair TL, Hall NL, Carlquist JF, Lappé DL, Muhlestein JB, Intermountain Heart Collaborative (IHC) Study Group Relation of vitamin D deficiency to cardiovascular risk factors, disease status, and incident events in a general healthcare population. Am J Cardiol. 2010;106:963–968. doi: 10.1016/j.amjcard.2010.05.027. [DOI] [PubMed] [Google Scholar]
- 4.Dobnig H, Pilz S, Scharnagl H, Renner W, Seelhorst U, Wellnitz B, Kinkeldei J, Boehm BO, Weihrauch G, Maerz W. Independent association of low serum 25-hydroxyvitamin D and 1,25-dihydroxyvitamin D levels with all-cause and cardiovascular mortality. Arch Intern Med. 2008;168:1340–1349. doi: 10.1001/archinte.168.12.1340. [DOI] [PubMed] [Google Scholar]
- 5.Pilz S, März W, Wellnitz B, Seelhorst U, Fahrleitner-Pammer A, Dimai HP, Boehm BO, Dobnig H. Association of vitamin D deficiency with heart failure and sudden cardiac death. J Clin Endocrinol Metab. 2008;93:3927–3935. doi: 10.1210/jc.2008-0784. [DOI] [PubMed] [Google Scholar]
- 6.Zhao G, Ford ES, Li C, Croft JB. Serum 25-hydroxyvitamin D levels and all-cause and cardiovascular disease mortality among US adults with hypertension: the NHANES linked mortality study. J Hypertens. 2012;30:284–289. doi: 10.1097/HJH.0b013e32834e1f0a. [DOI] [PubMed] [Google Scholar]
- 7.Zittermann A, Schleithoff SS, Frisch S, Götting C, Kuhn J, Koertke H, Kleesiek K, Tenderich G, Koerfer R. Circulating calcitriol concentrations and total mortality. Clin Chem. 2009;55:1163–1170. doi: 10.1373/clinchem.2008.120006. [DOI] [PubMed] [Google Scholar]
- 8.Zittermann A, Schleithoff SS, Götting C, Dronow O, Fuchs U, Kuhn J, Kleesiek K, Tenderich G, Koerfer R. Poor outcome in end-stage heart failure patients with low circulating calcitriol levels. Eur J Heart Fail. 2008;10:321–327. doi: 10.1016/j.ejheart.2008.01.013. [DOI] [PubMed] [Google Scholar]
- 9.Schierbeck LL, Jensen TS, Bang U, Jensen G, Køber L, Jensen JE. Parathyroid hormone and vitamin D-markers for cardiovascular and all cause mortality in heart failure. Eur J Heart Fail. 2011;13:626–632. doi: 10.1093/eurjhf/hfr016. [DOI] [PubMed] [Google Scholar]
- 10.Liu LC, Voors AA, van Veldhuisen DJ, van der Veer E, Belonje AM, Szymanski MK, Silljé HH, van Gilst WH, Jaarsma T, de Boer RA. Vitamin D status and outcomes in heart failure patients. Eur J Heart Fail. 2011;13:619–625. doi: 10.1093/eurjhf/hfr032. [DOI] [PubMed] [Google Scholar]
- 11.Witte KK, Byrom R. Micronutrients for chronic heart failure: end of the road or path to enlightenment? JACC Heart Fail. 2014;2:318–320. doi: 10.1016/j.jchf.2014.04.001. [DOI] [PubMed] [Google Scholar]
- 12.Witte KK, Byrom R, Gierula J, Paton MF, Jamil HA, Lowry JE, Gillott RG, Barnes SA, Chumun H, Kearney LC, Greenwood JP, Plein S, Law GR, Pavitt S, Barth JH, Cubbon RM, Kearney MT. Effects of vitamin D on cardiac function in patients with chronic HF: the VINDICATE study. J Am Coll Cardiol. 2016;67:2593–2603. doi: 10.1016/j.jacc.2016.03.508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Cubbon RM, Gale CP, Kearney LC, Schechter CB, Brooksby WP, Nolan J, Fox KA, Rajwani A, Baig W, Groves D, Barlow P, Fisher AC, Batin PD, Kahn MB, Zaman AG, Shah AM, Byrne JA, Lindsay SJ, Sapsford RJ, Wheatcroft SB, Witte KK, Kearney MT. Changing characteristics and mode of death associated with CHF due to left ventricular systolic dysfunction: a study across therapeutic eras. Circ Heart Fail. 2011;4:396–403. doi: 10.1161/CIRCHEARTFAILURE.110.959882. [DOI] [PubMed] [Google Scholar]
- 14.Cubbon RM, Woolston A, Adams B, Gale CP, Gilthorpe MS, Baxter PD, Kearney LC, Mercer B, Rajwani A, Batin PD, Kahn M, Sapsford RJ, Witte KK, Kearney MT. Prospective development and validation of a model to predict heart failure hospitalisation. Heart. 2014;100:923–929. doi: 10.1136/heartjnl-2013-305294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Coresh J, Astor BC, McQuillan G, Kusek J, Greene T, Van Lente F, Levey AS. Calibration and random variation of the serum creatinine assay as critical elements of using equations to estimate glomerular filtration rate. Am J Kidney Dis. 2012;39:920–929. doi: 10.1053/ajkd.2002.32765. [DOI] [PubMed] [Google Scholar]
- 16.Holick MF, Binkley NC, Bischoff-Ferrari HA, Gordon CM, Hanley DA, Heaney RP, Murad MH, Weaver CM, Endocrine Society Evaluation, treatment, and prevention of vitamin D deficiency: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96:1911–1930. doi: 10.1210/jc.2011-0385. [DOI] [PubMed] [Google Scholar]
- 17.White IR, Royston P, Wood AM. Multiple imputation using chained equations: issues and guidance for practice. Stat Med. 2011;30:377–399. doi: 10.1002/sim.4067. [DOI] [PubMed] [Google Scholar]
- 18.Sterne JA, White IR, Carlin JB, Spratt M, Royston P, Kenward MG, Wood AM, Carpenter JR (2009) Multiple imputation for missing data in epidemiological and clinical research: potential and pitfalls. BMJ338:b2393 [DOI] [PMC free article] [PubMed]
- 19.White IR, Royston P. Imputing missing covariate values for the Cox model. Stat Med. 2009;28:1982–1998. doi: 10.1002/sim.3618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Cattle BA, Baxter PD, Greenwood DC, Gale CP, West RM. Multiple imputation for completion of a national clinical audit dataset. Stat Med. 2011;30:2736–2753. doi: 10.1002/sim.4314. [DOI] [PubMed] [Google Scholar]
- 21.Gruson D, Ferracin B, Ahn SA, Zierold C, Blocki F, Hawkins DM, Bonelli F, Rousseau MF. 1,25-Dihydroxyvitamin D to PTH(1–84) ratios strongly predict cardiovascular death in heart failure. PLoS One. 2015;10:e0135427. doi: 10.1371/journal.pone.0135427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Belen E, Sungur A, Sungur MA. Vitamin D levels predict hospitalization and mortality in patients with heart failure. Scand Cardiovasc J. 2016;50:17–22. doi: 10.3109/14017431.2015.1098725. [DOI] [PubMed] [Google Scholar]
- 23.Gotsman I, Shauer A, Zwas DR, Hellman Y, Keren A, Lotan C, Admon D. Vitamin D deficiency is a predictor of reduced survival in patients with heart failure; vitamin D supplementation improves outcome. Eur J Heart Fail. 2012;14:357–366. doi: 10.1093/eurjhf/hfr175. [DOI] [PubMed] [Google Scholar]
- 24.Schwingshackl L, Boeing H, Stelmach-Mardas M, Gottschald M, Dietrich S, Hoffmann G, Chaimani A. Dietary supplements and risk of cause-specific death, cardiovascular disease, and cancer: a systematic review and meta-analysis of primary prevention trials. Adv Nutr. 2017;8:27–39. doi: 10.3945/an.116.013516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Pilz S, Verheyen N, Grübler MR, Tomaschitz A, März W. Vitamin D and cardiovascular disease prevention. Nat Rev Cardiol. 2016;13:404–417. doi: 10.1038/nrcardio.2016.73. [DOI] [PubMed] [Google Scholar]
- 26.Witte KK, Clark AL, Cleland JG. Chronic heart failure and micronutrients. J Am Coll Cardiol. 2001;37:1765–1774. doi: 10.1016/S0735-1097(01)01227-X. [DOI] [PubMed] [Google Scholar]
- 27.Hossein-nezhad A, Spira A, Holick MF. Influence of vitamin D status and vitamin D3 supplementation on genome wide expression of white blood cells: a randomized double-blind clinical trial. PLoS One. 2013;8:e58725. doi: 10.1371/journal.pone.0058725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Weber KT. Furosemide in the long-term management of heart failure: the good, the bad, and the uncertain. J Am Coll Cardiol. 2004;44:1308–1310. doi: 10.1016/j.jacc.2004.06.046. [DOI] [PubMed] [Google Scholar]
- 29.Rejnmark L, Vestergaard P, Pedersen AR, Heickendorff L, Andreasen F, Mosekilde L. Dose-effect relations of loop- and thiazide-diuretics on calcium homeostasis: a randomized, double-blinded Latin-square multiple cross-over study in postmenopausal osteopenic women. Eur J Clin Invest. 2003;33:41–50. doi: 10.1046/j.1365-2362.2003.01103.x. [DOI] [PubMed] [Google Scholar]
- 30.Chokar VS, Sun Y, Bhattacharya SK, Ahokas RA, Myers LK, Xing Z, Smith RA, Gerling IC, Weber KT. Hyperparathyroidism and the calcium paradox of aldosteronism. Circulation. 2005;111:871–878. doi: 10.1161/01.CIR.0000155621.10213.06. [DOI] [PubMed] [Google Scholar]
- 31.Laguardia SP, Dockery BK, Bhattacharya SK, Nelson MD, Carbone LD, Weber KT. Secondary hyperparathyroidism and hypovitaminosis D in African-Americans with decompensated heart failure. Am J Med Sci. 2006;332:112–118. doi: 10.1097/00000441-200609000-00003. [DOI] [PubMed] [Google Scholar]
- 32.Weisshaar RE, Simpson RU. Involvement of vitamin D3 with cardiovascular function. Am J Physiol. 1987;253:E675–E683. doi: 10.1152/ajpendo.1987.253.6.E675. [DOI] [PubMed] [Google Scholar]
- 33.Khouzam RN, Dishmon DA, Farah V, Flax SD, Carbone LD, Weber KT. Secondary hyperparathyroidism in patients with untreated and treated congestive heart failure. Am J Med Sci. 2006;331:30–34. doi: 10.1097/00000441-200601000-00009. [DOI] [PubMed] [Google Scholar]
- 34.Zia AA, Kamalov G, Newman KP, McGee JE, Bhattacharya SK, Ahokas RA, Sun Y, Gerling IC, Weber KT. From aldosteronism to oxidative stress: the role of excessive intracellular calcium accumulation. Hypertens Res. 2010;33:1091–1101. doi: 10.1038/hr.2010.159. [DOI] [PubMed] [Google Scholar]
- 35.Ahokas RA, Sun Y, Bhattacharya SK, Gerling IC, Weber KT. Aldosteronism and a proinflammatory vascular phenotype. Role of Mg2+, Ca2+ and H2O2 in peripheral blood mononuclear cells. Circulation. 2005;111:51–57. doi: 10.1161/01.CIR.0000151516.84238.37. [DOI] [PubMed] [Google Scholar]
- 36.Cozzolino M, Ketteler M, Zehnder D. The vitamin D system: a crosstalk between the heart and kidney. Eur J Heart Fail. 2010;12:1031–1041. doi: 10.1093/eurjhf/hfq112. [DOI] [PubMed] [Google Scholar]
- 37.Boxer RS, Dauser DA, Walsh SJ, Hager WD, Kenny AM. The association between vitamin D and inflammation with the 6-minute walk and frailty in patients with heart failure. J Am Geriatr Soc. 2008;56:454–461. doi: 10.1111/j.1532-5415.2007.01601.x. [DOI] [PubMed] [Google Scholar]
- 38.Vidal A, Sun Y, Bhattacharya SK, Ahokas RA, Gerling IC, Weber KT. Calcium paradox of aldosteronism and the role of the parathyroid glands. Am J Physiol Heart Circ Physiol. 2006;290:H286–H294. doi: 10.1152/ajpheart.00535.2005. [DOI] [PubMed] [Google Scholar]
- 39.Rutledge MR, Farah V, Adeboye AA, Seawell MR, Bhattacharya SK, Weber KT. Parathyroid hormone, a crucial mediator of pathologic cardiac remodelling in aldosteronism. Cardiovasc Drugs Ther. 2013;27:161–170. doi: 10.1007/s10557-012-6378-0. [DOI] [PubMed] [Google Scholar]
- 40.Xiang W, Kong J, Chen S, Cao LP, Qiao G, Zheng W, Liu W, Li X, Gardner DG, Li YC. Cardiac hypertrophy in vitamin D receptor knockout mice: role of the systemic and cardiac renin angiotensin systems. Am J Physiol Endocrinol Metab. 2005;288:E125–E132. doi: 10.1152/ajpendo.00224.2004. [DOI] [PubMed] [Google Scholar]
- 41.Chang JM, Lai YH, Tsai JH. Interaction between extracellular calcium and endothelin-1 influences parathyroid hormone secretion from bovine parathyroid cells through the increase in intracellular calcium. Miner Electrolyte Metab. 1997;23:113–120. [PubMed] [Google Scholar]
- 42.Hagström E, Ingelsson E, Sundström J, Hellman P, Larsson TE, Berglund L, Melhus H, Held C, Michaëlsson K, Lind L, Arnlöv J. Plasma parathyroid hormone and risk of congestive heart failure in the community. Eur J Heart Fail. 2010;12:1186–1192. doi: 10.1093/eurjhf/hfq134. [DOI] [PubMed] [Google Scholar]
- 43.Masson S, Barlera S, Colotta F, Magnoli M, Bonelli F, Moro M, Marchioli R, Tavazzi L, Tognoni G, Latini R. A low plasma 1,25(OH)2 vitamin D/PTH (1–84) ratio predicts worsening of renal function in patients with chronic heart failure. Int J Cardiol. 2016;224:220–225. doi: 10.1016/j.ijcard.2016.09.014. [DOI] [PubMed] [Google Scholar]
- 44.Li YC, Kong J, Wei M, Chen ZF, Liu SQ, Cao LP. 1,25-Dihydroxyvitamin D(3) is a negative endocrine regulator of the renin–angiotensin system. J Clin Invest. 2002;110:229–238. doi: 10.1172/JCI0215219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Grübler MR, Gaksch M, Kienreich K, Verheyen N, Schmid J, Ó Hartaigh BW, Richtig G, Scharnagl H, Meinitzer A, Pieske B, Fahrleitner-Pammer A, März W, Tomaschitz A, Pilz S. Effects of vitamin D supplementation on plasma aldosterone and renin—a randomized placebo-controlled trial. J Clin Hypertens (Greenwich) 2016;18:608–613. doi: 10.1111/jch.12825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Bjelakovic G, Gluud LL, Nikolova D, Whitfield K, Wetterslev J, Simonetti RG, Bjelakovic M, Gluud C. Vitamin D supplementation for prevention of mortality in adults. Cochrane Database Syst Rev. 2014;1:CD007470. doi: 10.1002/14651858.CD007470.pub3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Chowdhury R, Kunutsor S, Vitezova A, Oliver-Williams C, Chowdhury S, Kiefte-de-Jong JC, Khan H, Baena CP, Prabhakaran D, Hoshen MB, Feldman BS, Pan A, Johnson L, Crowe F, Hu FB, Franco OH. Vitamin D and risk of cause specific death: systematic review and meta-analysis of observational cohort and randomised intervention studies. BMJ. 2014;348:g1903. doi: 10.1136/bmj.g1903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Ford JA, Maclennan GS, Avenell A, Bolland M, Grey A, Witham M, RECORD Trial Group Cardiovascular disease and vitamin D supplementation: trial analysis, systematic review, and meta-analysis. Am J Clin Nutr. 2014;100:746–755. doi: 10.3945/ajcn.113.082602. [DOI] [PubMed] [Google Scholar]
- 49.Scragg R, Stewart AW, Waayer D, Lawes CM, Toop L, Sluyter J, Murphy J, Khaw KT, Camargo CA., Jr Effect of monthly high-dose vitamin D supplementation on cardiovascular disease in the vitamin D assessment study: a randomized clinical trial. JAMA Cardiol Apr. 2017 doi: 10.1001/jamacardio.2017.0175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Scragg R, Waayer D, Stewart AW, Lawes CM, Toop L, Murphy J, Khaw KT, Camargo CA., Jr The vitamin D assessment (ViDA) study: design of a randomized controlled trial of vitamin D supplementation for the prevention of cardiovascular disease, acute respiratory infection, falls and non-vertebral fractures. J Steroid Biochem Mol Biol. 2016;164:318–325. doi: 10.1016/j.jsbmb.2015.09.010. [DOI] [PubMed] [Google Scholar]
- 51.Hollis BW, Wagner CL. The role of the parent compound vitamin D with respect to metabolism and function: why clinical dose intervals can affect clinical outcomes. J Clin Endocrin Metab. 2013;98:4619–4628. doi: 10.1210/jc.2013-2653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Martineau AR, Jolliffe DA, Hooper RL, Greenberg L, Aloia JF, Bergman P, Dubnov-Raz G, Esposito S, Ganmaa D, Ginde AA, Goodall EC, Grant CC, Griffiths CJ, Janssens W, Laaksi I, Manaseki-Holland S, Mauger D, Murdoch DR, Neale R, Rees JR, Simpson S, Jr, Stelmach I, Kumar GT, Urashima M, Camargo CA., Jr Vitamin D supplementation to prevent acute respiratory tract infections: systematic review and meta-analysis of individual participant data. BMJ. 2017;356:i6583. doi: 10.1136/bmj.i6583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Reid IR, Horne AM, Mihov B, Gamble GD, Al-Abuwsi F, Singh M, Taylor L, Fenwick S, Camargo CA, Stewart AW, Scragg R. Effect of monthly high-dose vitamin D on bone density in community-dwelling older adults substudy of a randomized controlled trial. J Intern Med. 2017;282:452–460. doi: 10.1111/joim.12651. [DOI] [PubMed] [Google Scholar]
- 54.Sluyter JD, Camargo CA, Waayer D, Lawes CMM, Toop L, Khaw KT, Scragg R. Effect of monthly, high-dose, long-term vitamin D on lung function: a randomized controlled trial. Nutrients. 2017;9:E1353. doi: 10.3390/nu9121353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Sluyter JD, Camargo CA, Jr, Stewart AW, Waayer D, Lawes CMM, Toop L, Khaw KT, Thom SAM, Hametner B, Wassertheurer S, Parker KH, Hughes AD, Scragg R. Effect of monthly, high-dose, long-term vitamin D supplementation on central blood pressure parameters: a randomized controlled trial substudy. J Am Heart Assoc. 2017;6:e006802. doi: 10.1161/JAHA.117.006802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Boxer RS, Hoit BD, Schmotzer BJ, Stefano GT, Gomes A, Negrea L. The effect of vitamin D on aldosterone and health status in patients with heart failure. J Card Fail. 2014;20:334–342. doi: 10.1016/j.cardfail.2014.01.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Schleithoff SS, Zittermann A, Tenderich G, Berthold HK, Stehle P, Koerfer R. Vitamin D supplementation improves cytokine profiles in patients with congestive heart failure: a double-blind, randomized, placebo-controlled trial. Am J Clin Nutr. 2006;83:754–759. doi: 10.1093/ajcn/83.4.754. [DOI] [PubMed] [Google Scholar]
- 58.Witham MD, Crighton LJ, Gillespie ND, Struthers AD, McMurdo ME. The effects of vitamin D supplementation on physical function and quality of life in older patients with heart failure: a randomized controlled trial. Circ Heart Fail. 2010;3:195–201. doi: 10.1161/CIRCHEARTFAILURE.109.907899. [DOI] [PubMed] [Google Scholar]
- 59.Moretti HD, Colucci VJ, Berry BD. Vitamin D3 repletion versus placebo as adjunctive treatment of heart failure patient quality of life and hormonal indices: a randomized, double-blind, placebo-controlled trial. BMC Cardiovasc Disord. 2017;17:274. doi: 10.1186/s12872-017-0707-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Rodriguez AJ, Mousa A, Ebeling PR, Scott D, de Courten B. Effects of vitamin D supplementation on inflammatory markers in heart failure: a systematic review and meta-analysis of randomized controlled trials. Sci Rep. 2018;18:1169. doi: 10.1038/s41598-018-19708-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Jiang WL, Gu HB, Zhang YF, Xia QQ, Qi J, Chen JC. Vitamin D supplementation in the treatment of chronic heart failure: a meta-analysis of randomized controlled trials. Clin Cardiol. 2016;39:56–61. doi: 10.1002/clc.22473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Zittermann A, Ernst JB, Prokop S, Fuchs U, Dreier J, Kuhn J, Knabbe C, Birschmann I, Schulz U, Berthold HK, Pilz S, Gouni-Berthold I, Gummert JF, Dittrich M, Börgermann J. Effect of vitamin D on all-cause mortality in heart failure (EVITA): a 3-year randomized clinical trial with 4000 IU vitamin D daily. Eur Heart J. 2017;38:2279–2286. doi: 10.1093/eurheartj/ehx235. [DOI] [PubMed] [Google Scholar]
- 63.Vieth R. Why “Vitamin D” is not a hormone, and not a synonym for 1,25-dihydroxy-vitamin D, its analogs or deltanoids. J Steroid Biochem Mol Biol. 2004;89–90:571–573. doi: 10.1016/j.jsbmb.2004.03.037. [DOI] [PubMed] [Google Scholar]
- 64.Holick MF. Sunlight and vitamin D for bone health and prevention of autoimmune diseases, cancers, and cardiovascular disease. Am J Clin Nutr. 2004;80:1678S–88S. doi: 10.1093/ajcn/80.6.1678S. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


