Abstract
Esophageal adenocarcinoma (EAC) has one of the fastest rising incidence rates in the U.S. and many other Western countries. One of the unique risk factors for EAC is gastroesophageal reflux disease (GERD), a chronic digestive condition in which acidic contents from the stomach, frequently mixed with duodenal bile, enter the esophagus resulting in esophageal tissue injury. At the cellular level, progression to EAC is underlined by continuous DNA damage caused by reflux and chronic inflammatory factors that increase the mutation rate and promote genomic instability. Despite recent successes in cancer diagnostics and treatment, EAC remains a poorly treatable disease. Recent research has shed new light on molecular alterations underlying progression to EAC and revealed novel treatment options. This review focuses on the genetic and molecular studies of EAC. The molecular changes that occur during the transformation of normal Barrett’s esophagus to esophageal adenocarcinoma are also discussed.
Keywords: Esophageal adenocarcinoma, GERD, Barrett’s esophagus, Esophageal cancer, p53
1. Introduction
1.1. Epidemiology, Etiology, and Pathology
Esophageal cancer is a group of diseases characterized by uncontrolled proliferation of cells, which may originate from epithelial, neuroendocrine, lymphoid or mesenchymal tissues. The vast majority of esophageal tumors are carcinomas (i.e. derived from epithelial cells), with two main histological types: squamous cell carcinoma (ESCC) and adenocarcinoma (EAC). Both tumor types primarily affect older individuals and are three to four times more common in men than in women [1]. It was estimated that 572,034 new esophageal cancer cases and 508,585 deaths occurred in 2018 worldwide [2]. Geographical variation in the incidence rate and tumor type is striking. The highest incidence rate is found in Asia and Southern and Eastern Africa. In Asian region, often referred to as the “esophageal cancer belt”, which stretches from Northern Iran through the Central Asia to Mongolia and North-Central China, 90% of cases are squamous cell carcinomas [3]. In contrast, in Western countries, where the incidence rate for esophageal cancer is typically low, EAC has been rapidly increasing and in some countries has overtaken ESCC. In the US, the incidence of EAC among men surpassed that for ESCC around 1990 and continues to increase [4, 5].
Despite recent successes in cancer diagnostics and treatment, esophageal cancer remains a poorly treatable disease, and the surgery that is the mainstay of current therapy carries notable morbidity and mortality. In the US, the overall 5-year survival rate for individuals diagnosed with esophageal cancer was estimated 19% [6]. It is significantly lower for patients diagnosed with metastatic disease, at this point the 5-year survival rate declines to 5%. Unfortunately, most esophageal tumors are found when metastases already have occurred [6].
Cigarette smoking and excessive alcohol consumption account for the majority of ESCC cases in the US and Western countries [7]. Chronic gastroesophageal reflux disease (GERD), Barrett’s esophagus, obesity, and cigarette smoking are the risk factors for EAC. Among these risk factors, GERD is considered being most prominent [8]. Weekly symptoms of GERD increase the odds of EAC fivefold and daily symptoms sevenfold compared with individuals without symptoms or less frequent symptoms [9]. In the center of tumorigenic alterations induced by GERD is a persistent cycle of damage and regeneration of esophageal tissues. At the cellular level, esophageal epithelial cells are periodically exposed to a refluxate that contains acidic gastric juice frequently mixed with duodenal bile, causing cellular and DNA damages. It also induces inflammatory esophagitis, which in turn, may exacerbate mucosal injury [10-12]. If the damage persists, it can cause hyperplasia and Barrett’s esophagus (BE), a condition in which the normal squamous epithelial lining is replaced by a metaplastic intestinal type of epithelium. About 5 to 15% of patients with GERD are found to have BE [13]. GERD can lead to further accumulation of genetic alterations in BE cells and progression to EAC at a rate approximately 0.12%-0.6% per year [8].
In this review, we will discuss genetic and epigenetic changes that play an integral role in the progression of Barrett’s esophagus to esophageal adenocarcinoma. The effect of cell cycle dysregulation and alterations of key oncogene and tumor suppressor signaling networks will also be discussed.
2. Genetic and Epigenetic alterations
Based on a comprehensive comparison across more than 3000 cancers and 27 tumor types, EAC was included in a group of tumors with the most frequent copy-number alterations (CNA) [5, 14, 15]. In EAC, the median frequency of chromosomal rearrangements was reported at 172 per tumor (range of 77-402). Approximately 20% of these rearrangements were classified as interchromosomal translocations. The mutation frequency was estimated to be 9.9 mutations/Mb (range of 7.1-25.2) relative to a haploid genome. This frequency translates into a median of 26,161(range of 18,881-66,225) mutations across the genome per tumor [5].
During the past three decades, our understanding of genetic changes in EAC has been evolving following the development of new and more advanced techniques. Early cytogenetic studies using chromosome-banding and flow cytometry have identified aneuploidy, tetraploidy, and specific losses of chromosomes 4, 18, 21, and Y, as well as recurrent gains of chromosomes 14 and 20 in dysplastic BE and adenocarcinoma. The loss of the Y chromosome has been found as a one of the most common numeric chromosomal abnormalities. Recurrent structural rearrangements at 1p, 3q, 11p, and 22p and frequent mutations in the TP53 gene, which encodes p53, a well-known tumor suppressor, have also been found [16, 17].
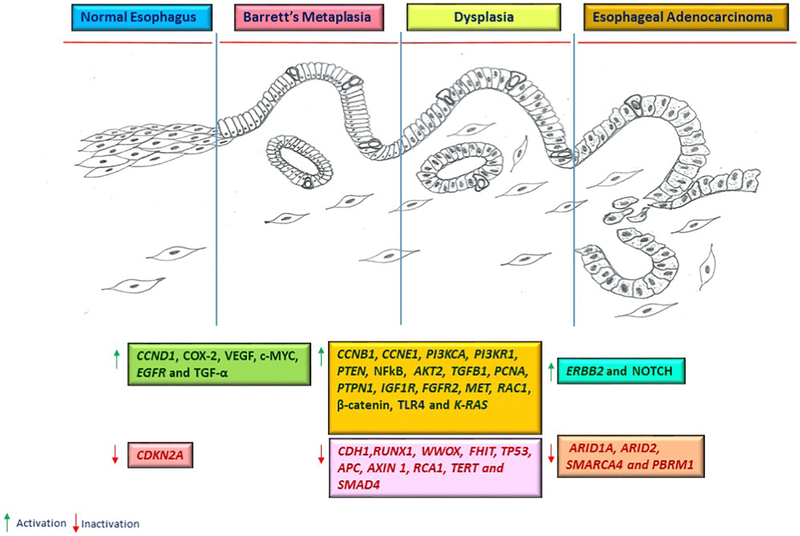
Fluorescent in situ Hybridization (FISH) has helped to identify additional numeric changes in chromosomes 4, 6, 7, 8, 9, 10, 11, 12, 17, 18, Y, and X that were found to be an early change in dysplastic regions [18, 19]. Further studies using comparative genomic hybridization (CGH) revealed common regions of gain at 20pq, 17q, 8q, 7p, 13q, 10q, 6p, 15q, 2pq and loss at 4pq, 14q, 18q, 5q, 16q, 17q, 9p, 7q and Y [20-23]. The frequencies of losses and gains were found to correlate with aneuploidy and significantly increased during neoplastic progression from low-grade to high-grade dysplasias and invasive carcinomas [19, 24]. Chromosomal alterations were also found in BE adjacent to cancer sites [23, 25]. Several studies have suggested that recurrent gains (8q, 6p, 10q) and losses (13q, Y, 9p, 17p) occur in Barrett’s metaplastic cells even in the absence of dysplasia and adenocarcinoma, although to a lesser extend [23, 26]. These studies outlined complex structural and whole chromosome abnormalities in EAC. The CGH analyses also yielded a wealth of data for identification of specific EAC related genes. Multiple candidate tumor-suppressor genes (APC, MCC, MTS1, CDKN2, TSHR, DCC, PI5, FHIT, RCA1) and oncogenes (MLVI2, NRASL3 EGFR, MYC, IGF1R, ERBB2/HER2-neu, TGFB1, BCL3, AKT2, PCNA, MYBL2, PTPN1) have been identified (Figure 1) [20, 21].
Figure 1. Key molecular alterations associated with progression of Barrett’s metaplasia to esophageal adenocarcinoma.
Gastroesophageal reflux and other carcinogenic stimuli cause genetic and epigenetic alterations. Multiple pathways regulating cell growth, proliferation, apoptosis, differentiation, inflammation and angiogenesis are commonly affected. The molecular changes occur at early stages of tumorigenic process and increase with tumor progression. COX-2 – Cyclooxygenase-2; VEGF – Vascular endothelial growth factor; TGF-α – Transforming growth factor α; NFκB – Nuclear factor kappa-light-chain-enhancer of activated B cells.
Further development of array-based profiling and next generation sequencing (NGS) improved resolution of genomic analyses and helped to identify new recurrent genetic alterations and specific signaling pathways associated with EAC. The initial systematic NGS study by Dulak et al, which included 149 EAC tumor – normal pairs, found mutations in 8,331 genes, of which 199 were mutated in 5% or more of the EACs. A search for genes with significantly recurring mutations identified 26 genes. The TP53 gene was found to be the most frequently mutated gene in EACs. Seventy two percent of EACs carry p53 mutations. Similar frequencies of p53 mutations (71-72%) were reported in studies conducted by the Cancer Genome Atlas (TCGA) Research Network [27] and the Oesophageal Cancer Clinical and Molecular Stratification (OCCAMS) Study Group [28].
High frequency of mutations was also found in the CDKN2 gene, which is known to regulate the cell cycle. The TCGA studies revealed that the CDKN2A gene is inactivated by deletions, epigenetic silencing, or mutations in 76% of EACs. Cell cycle regulation is affected not only by inactivation of CDKN2A but also by amplification of CDK6, CCNE1 and CCND1 genes, which encode cell division protein kinase 6 (CDK6) and cyclins E1 and D1, respectively (Figure 1). The MYC gene, which regulates proliferation, is also amplified in approximately 30% of EACs. Among frequently altered genes are receptor tyrosine kinases (RTKs) of the EGFR family and their downstream mediators. Amplification of the ERBB2 gene is the most prominent receptor alteration in EACs that was found in 32% of tumors. EGFR gene is amplified in 15% of EACs [27]. The phosphatidylinositol-3-kinase (PI3K) pathway was the most frequently altered oncogenic pathway by mutations and CNAs found in 24% of tumors [5, 27]. In addition to ERBB2 and EGFR gene amplifications, which can potentially activate the PI3K pathway, mutations were reported in PI3KCA, PI3KR1, PTEN and other related genes. K-RAS gene amplifications were found in 14% of tumors. The Rho family GTPase, RAC1, is also frequently activated primarily by mutations in DOCK2 and ELM01 genes that are important regulators of RAC1. Given that dysregulation of DOCK2 and ELMO1 is associated in cancers with enhanced cell migration and invasion, it may help to explain the highly invasive nature of EACs. In addition, EACs shows amplifications of VEGFA, FGFR2, IGF1R, and MET genes (Figure 1). Given that many receptor and non-receptor kinases can be inhibited with specific drugs, these findings open new opportunities for targeted therapy in EAC.
NGS analyses also revealed dysregulation of the TGFβ pathway. Its components were mutated in 18% of tumors; the most recurrently altered gene in this pathway was SMAD4. The product of this gene forms transcription complexes with other members of the SMAD protein family and regulates TGFβ-mediated transcription. Interestingly, SMAD4 is primarily mutated in EAC, but not in high grade dysplasia (HGD) providing a genetic distinction between EAC and HGD [29].
In addition, some EACs showed activation of the WNT/β-catenin pathway by mutations or loss of AXIN1, APC or CDH1 genes, although dysregulation of this pathway was less frequent than in other tumor types. Mutations of the CTNNB1 gene, which encodes β-catenin, were found to be relatively uncommon.
EAC also shows loss-of-function mutations and CNAs of ARID1A, ARID2, SMARCA4, and PBRM1 genes that encode components of the SWI/SNF (SWItch/Sucrose Non-Fermentable) chromatin-remodelling complex (Figure 1). The SWI/SNF complex is an evolutionarily conserved multi-subunit complex involved in chromatin restructuring that contribute to transcriptional activation and repression. Alterations of the SWI/SNF complex are not unique to EAC and are found in over 20% of human malignancies.
Among other prominent alterations were amplifications of GATA4/6 genes, deletions of RUNX1, WWOX, FHIT genes that have potential tumor suppressor roles, and mutations in genes that regulate the adherens junctions, CDH1, HEWCW 1, AJAP1, and inflammatory response, TLR4. It is important to mention, however, that many significantly altered genes are poorly characterized and their functions remain unclear.
The mutational signatures revealed three distinct molecular subtypes for EAC: (i) enrichment for BRCA signature with prevalent defects in the homologous recombination pathway; (ii) dominant T>G mutational pattern associated with a high mutational load and neoantigen burden; and (iii) C>A/T mutational pattern with evidence of an aging imprint [30].
WGS studies assessed the molecular relationship between BE and EAC. BE was found to be polyclonal and highly mutated even in the absence of dysplasia [28]. It was reported that early stages of disease and BE often have a higher rate of mutations than many common dysplastic tumors [31]. At the same time, comparison of EAC and adjacent BE often showed surprisingly little overlap (< 20%) in the spectrum of mutations [28]. This is in contrast to early studies showing that many mutations in EAC are already present in BE [32]. The underlying reason for these differences is unclear but it may possibly be attributable to clonal variations and the presence of dysplastic cells in analyzed specimens.
The mutational landscape of BE and EAC differs more dramatically at the chromosomal level. Genomes of BE tissues were found to be relatively more stable than those of invasive tumors [31]. It was shown that approximately a third of EAC cases (32%) are characterized by massive localized chromosome translocations (chromothripsis) that may cause rapid activation of oncogenes and inactivation of tumor suppressors. These catastrophic genome rearrangements may potentially explain fast progression of EAC in some BE patients [33].
Similar to other tumor types, genetic alterations in EAC are accompanied by significant changes of the epigenome. DNA methylation is the most studied epigenetic mark in the esophagus. Methylation of DNA was assessed by many researchers using a broad spectrum of methylation assays including methylation arrays and whole genome bisulfite sequencing. The latter methods permit to obtain genome-wide epigenetic information on the entire regulatory regions and compare DNA methylation of normal tissues with precancerous and cancerous lesions. These studies not only revealed a vast amount of new groundbreaking data on cancer-related alterations but also demonstrated that regulation of DNA methylation is complex and significantly affected by age, obesity, tobacco smoking and other risk factors [34].
Increased levels of the CpG island methylation were found in Barrett’s metaplasia compared to normal squamous epithelium [35-37]. Comparison with other types of normal tissues suggests that epigenetic alterations in BE may reflect the actual tumorigenic process, rather than simply due to acquisition of metaplastic phenotype [38]. Methylation of the CpG islands is further increased following progression to HGD and EAC, which between them have significant similarities in the methylation profiles [39]. Tumorigenic process in the esophagus is characterized not only by hypermethylation of the CpG islands, but also by decreasing DNA methylation outside of the CpG islands [40]. These two coexisting epigenetic phenomena force global transcriptome alterations that play significant roles in the development and progression of EAC [35].
Several studies attempted to find methylation markers that discriminate between high- and low-risk BE. It was shown that the promoter hypermethylation of MGMT [41], p16/ RUNX3/HPP1 [42], HPP1/p16/RUNX3 [43], TIPM3/APC/TERT [44] genes or gene combinations such as SLC22A18+PIGR+GJA2+RIN2 [39], p16+APC [45], RUNX3+p16+HPP1+NELL1+TAC1+SST+AKAP12+CDH13 and hypomethylation of ORF3A4 gene [46] may help to stratify the risk of cancer development in patients with non-dysplastic BE. Clinical application of these epigenetic biomarkers, although promising, requires additional investigation in large-scale clinical trials.
Among other critical epigenetic alterations that contribute to the development and progression of EAC are posttranslational modifications of histones and alterations of multiple non-coding RNAs, including microRNA and lncRNA. We refer to recent comprehensive reviews on the subject [47, 48].
3. DNA damage in conditions of esophageal reflux injury
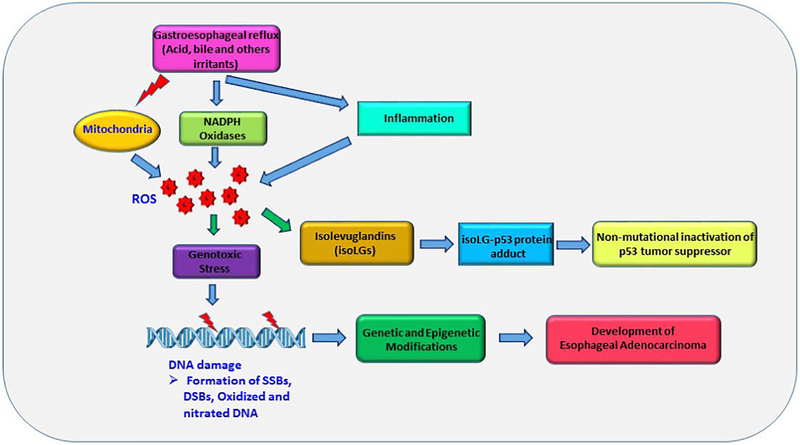
As discussed above, GERD is a prominent risk factor for EAC. Due to its complex local and systemic effects, many questions remain on how reflux promotes esophageal adenocarcinomas. Among known tumorigenic factors associated with reflux is DNA damage. It has been demonstrated that reflux have genotoxic effect on esophageal cells. Hydrochloric acid (HCl) and bile salts are the most characterized components of the refluxate that induce DNA damage, although other ingredients of gastric juice, pancreatic and duodenal secretions, and consumed food may also have additive effects. In the experimental setting in vitro a short exposure to acidic pH and bile salts, which mimics an episode of reflux, induces reactive oxygen species (ROS), oxidative stress and DNA damage [11, 49-52]. Both mitochondria and NADPH oxidases (NOX) have been found to be responsible for excessive production of ROS (Figure 2) [52, 53]. Reflux activates NOX1 and NOX2 enzymes in the esophagus of GERD and BE patients [53]. NOX5-S, a truncated variant of NOX5, is also activated in acidic conditions [44, 54, 55]. These enzymes produce superoxide anion O2−and hydrogen peroxide H2O2 that damage genomic DNA. ROS is also thought to induce mutations in mitochondrial DNA in Barrett’s metaplasia [56].
Figure 2. Gastroesophageal reflux induces genotoxic stress and promotes esophageal tumorigenesis.
Gastroesophageal reflux causes aberrant production of reactive oxygen species (ROS), which cause DNA damage and multiple genetic and epigenetic alterations. Mitochondria and NADPH oxidases are strong inducers of ROS in the esophagus. Reflux-induced ROS mediate the isoLG adduction of p53 protein and its inhibition. SSBs – Single strand breaks; DDBs – Double strand breaks.
Multiple studies reported DNA damage in esophageal tissues of GERD and BE patients [57-59]. Experiments with esophageal perfusion were especially demonstrative. Perfusion of the esophagus of BE patients with HCl acid or deoxycholic acid (DCA) increased DNA damage in the esophagus even after a short exposure to these reflux ingredients [50, 60]. Another strong evidence on the reflux-induced DNA damage was produced by animal studies in which reflux was purposely induced by surgical procedures [61-63]. It was also shown that induction of bile reflux increases the mutational rate (primarily transitions C to T and G to A) in the rat esophagus [64]. These data are consistent with the preponderance of C to T transitions in human esophageal adenocarcinomas, suggesting that reflux may be responsible for their generation [5].
Although the entire spectrum of reflux-induced DNA lesions is currently unknown, it has been shown that exposure of esophageal cells to acid and bile salts promote formation of single- and double-strand DNA breaks, oxidized and nitrated DNA lesions. Nucleotide derivatives such as 8-oxo-deoxyguanineosine (8-oxo-dG) and 8-nitroguanine (8-nitro-dG), which are formed as a result of reflux, increase mutagenesis. Double strand breaks of DNA are even more detrimental as these lesions are extremely difficult to repair resulting in highly cytotoxic and mutagenic effects.
In addition to direct genotoxic effects, chronic inflammatory reactions caused by reflux significantly contribute to tissue and DNA damage in the esophagus (Figure 2) [65]. Under inflammatory conditions, inflammatory and epithelial cells release ROS. The produced superoxide radical O2− can react with nitric oxide (NO) resulting in generation of peroxynitrite (ONOO–), highly reactive species that cause oxidation, nitration, and deamination reactions of different biomolecules including DNA. During gastroesophageal reflux, large quantities of nitric oxide are produced from dietary nitrate at the GE junction and gastric cardia [66]. Another source of NO is inducible nitric oxide synthase (iNOS), an enzyme that is activated by multiple inflammatory stimuli. Formed reactive nitrogen species (RNS) can nitrate, deaminate DNA, and produce DNA strand breaks and mutations [67].
Normally, the integrity of DNA is restored by the DNA repair machinery, which detects and promptly repairs damaged DNA. A number of studies suggested inhibitory role of reflux on DNA damage repair (DDR). For example, levels of DDR enzymes MUTYH and OGG1, which are involved in repair of oxidative DNA damage, were significantly decreased after treatment of esophageal cells with bile acid [68]. Inhibition of MUTYH was also shown in rats, in which reflux was induced by duodenoesophageal anastomosis [69].
DDR is inhibited by various mechanisms. MGMT protein, which is involved in repair of alkylated DNA lesions, is downregulated by promoter hypermethylation in BE [41]. Decreased efficiency of DNA damage repair may also occur due to polymorphisms in DNA repair genes [70-72]. Not all DDR enzymes are inhibited by reflux. Induction of APE1, an enzyme involved in base excision repair, is activated by acidic bile salts and provide a survival advantage to esophageal tumor cells [73].
Reflux also negatively affects the redox homeostasis resulting in an increased production of ROS. Activities of redox regulating enzymes, such as superoxide dismutase (SOD) [62, 74] and glutathione peroxidases (GPxs), were found to be inhibited by reflux [75-77].
Inhibition of DNA damage induced by reflux can be a promising strategy for chemoprevention of esophageal cancer [11, 49, 75]. Suppression of excessive production of reactive oxygen radicals and other reactive compounds is one obvious possibility. Prevention of chronic inflammation, which, in turn, can help to control production of ROS and RNS, also holds great promises. Studies of antioxidants showed their ability to suppress DNA damage induced by acid and bile [11, 78-80]. Among antioxidants, natural products are of particular interest because of their low toxicity, health safety, and general acceptance as dietary supplements. One interesting example is a natural antioxidant apocynin, which not only scavenges ROS but also suppresses NOX activity and activates DNA damage repair [11].
4. Molecular signaling networks in Esophageal Adenocarcinoma
4.1. p53 protein family
Over the last two decades, significant progress has been achieved in defining the key signaling molecules and pathways involved in development and progression of EAC. p53 tumor suppressor is among the most affected proteins in EAC. As discussed above, p53 is frequently inactivated by mutations, which typically occur during transition from non-dysplastic BE to high-grade dysplasia [29]. TP53gene mutations in BE tissues increased the adjusted risk of progression 13.8-fold (95% confidence interval, 3.2-61.0; p < 0.001). The comparison of BE tissues from patients with or without later progression to HGD or EAC found significantly higher numbers of TP53 mutations in BE from patients with subsequent progression [81].
These mutations are primarily missense variants that inhibit the binding of p53 protein to DNA causing inhibition of p53-dependent transcription. The p53 gene is also characterized by high frequency of loss of heterozygosity (LOH) [82]. LOH for chromosome 17p, which harbors the p53 gene, has shown promise as a biomarker for neoplastic progression in Barrett’s esophagus [83-88]. Activity of p53 protein is also inhibited by non-mutational mechanisms during the early stages of tumorigenesis. Several studies including ours have also demonstrated significant inhibition of wild type p53 protein in conditions of acidic reflux [63, 89-91]. One recently discovered mechanism includes the formation of p53 protein adducts [90]. It was shown that gastroesophageal reflux produces reactive isolevuglandins (isoLG), a family of γ-ketoaldehydes generated by the free radical-induced peroxidation of lipids and COX2 enzyme, that form adducts on the p53 molecule [92]. This results in inhibition of p53 activity and protein precipitation (Figure 2) [90].
Given the important role played by p53 in tumor suppression and chemotherapeutic drug response, a number of compounds, such as STIMA-1, PRIMA-1, MIRA-1, RITA and others, have been identified to restore activity of mutant p53 (reviewed in [93]). PRIMA-1 and its analog APR-246 are the most investigated compounds in this category of the p53-targeting compounds. APR-246 was tested in EAC cells harboring mutant p53 and found to upregulate p53 target genes and induce apoptosis [94, 95]. It can also enhance the inhibitory effects of chemotherapeutic drugs cisplatin and 5-fluorouracil through p53 accumulation in tumor cell [94]. Notably, APR-246 showed limited cytotoxic effect on normal cells. An initial phase I clinical trial has shown APR-246 to be safe in humans. Phase Ib/II study () evaluating the efficacy of APR-246 in the treatment of advanced and metastatic esophageal or gastro-esophageal junction cancers is currently ongoing. In contrast to the TP53, TP63 and TP73 genes, which encode other members of the p53 protein family, are rarely mutated in EAC [96, 97]. In esophageal tissues, p63 and p73 proteins are expressed as an intertwined mix of protein isoforms that are generally divided into two groups, termed TA and ΔN. The former isoforms have “p53-like” properties. Similar to p53, TA isoforms can transactivate the set of target genes overlapping with p53, induce cell cycle arrest, and apoptosis. In contrast, ΔN isoforms lack the N-terminal transactivation domain and exert a dominant negative effect toward TA isoforms. However, some ΔN isoforms, such as ΔNp63α retain transcriptional activity through additional transactivation domains (reviewed in [97]). Normal esophageal squamous epithelium shows strong nuclear staining for ΔNp63α in cells of the basal and in the suprabasal cell layers. p63 has also been detected in the ducts of esophageal mucosal and submucosal glands. This is in contrast to Barrett’s metaplastic and EAC epithelia, where levels of p63 isoforms are typically low [98, 99]. Treatment of esophageal cells with acidic bile salts results in decreased levels of ΔNp63 and upregulation of TA73 [11,63, 89]. The latter isoform is important for DNA damage repair as p73 regulates transcription of multiple DNA damage repair proteins [63]. Esophageal cells deficient in p73 activity are characterized by high levels of DNA damage [63]. Expression of p73 isoform, ΔNp73α, which is a dominant-negative inhibitor of TAp73 and p53, is upregulated in GERD and EAC and associated with poor prognosis in patients with EAC [100]. Pro-inflammatory cytokines IL-1β and TNFα were found to induce expression of ΔNp73 in conditions of esophageal reflux injury [100].
4.2. Cell cycle regulation
The loss of proper control of the cell cycle is one of the main mechanisms that promotes tumorigenic transformation. A number of cell cycle regulators are affected in EAC. Among them are tumor suppressors p16INK4aand p14ARF that are encoded by the CDKN2A gene. p16INK4a is a specific inhibitor of the cyclin D/CDK4/6 complexes. Its inhibition leads to disruption of normal cell cycle and uncontrolled cell growth [101]. Immunohistochemical staining showed loss of p16INK4a expression in 20-68% of BE and 60-100% of EAC cases [102, 103]. p14ARF is downregulated in 20% of BE and 75% of EAC cases [104]. Notably, downregulation of p14ARF interferes with the proper p53 response, because p14ARF is a critical upstream regulator of p53 that activates p53 protein by blocking its Mdm2-mediated degradation. Another CDK inhibitor, p27KIP1, which regulates the cell cycle by inhibiting the cyclin E/CDK2 and cyclin D/CDK4 complexes, is also affected in BE and EAC. Low levels of p27 KIP1 protein was found in 30-70% of BE and 83-100% of EAC and correlated with higher histological grade, depth of invasion, presence of lymph node metastasis, and survival [105-107]. p27KIP1 knockout mice develop BE and EAC following the esophagostomy and treatment with carcinogen [108].
Frequent amplification at 7q21, which harbors the CDK6 gene, was found in 35% of EACs [109, 110]. The CDK4 gene was also found amplified in EAC, but to a lesser extent (10%). Amplifications of both genes are associated with poor survival of EAC patients [110]. In addition, EAC are characterized by upregulation of several cyclins. Cyclin D1 is upregulated in 25-38% of BE and 36-44% of EAC patients [111-114]. Increased expression of cyclin D1 has prognostic significance and is associated with poor survival of EAC patients [115]. Protein expression of another cyclin, cyclin E, was found to be significantly increased during progression from non-dysplastic esophageal lesions to high grade dysplasia. High expression of cyclin E was observed in 5.8% of BE, 19.0% of LGD, 35.7% of HGD, and 16.7% of EACs [116]. The same study found amplification of the CCNE1 gene, which encodes cyclin E1, in 19.0% of EAC cases.
4.3. Activation of Oncogenic Signaling Pathways
Receptor Tyrosine Kinases
A large group of RTKs, such as epidermal growth factor receptor (EGFR), ERBB2/HER2, insulin-like growth factor receptor 1 (IGF1R), hepatocyte growth factor receptor (HGFR/c-MET) and vascular endothelial growth factor receptor (VEGFR) play a significant role in the development and progression of EAC (Figure 3) [117, 118]. Aberrant activation of EGFR signaling is caused by overproduction of EGFR protein and its ligands TGF-α and EGF [119, 120]. An increased expression of EGFR protein was found in 22.2-35% of BE and 46.5-80% of EAC patients [118, 121]. Several studies reported correlation between expression of EGFR protein and poor survival of EAC patients [122-124]. In vitro studies found that treatment of esophageal cells with acidic bile salts activates the EGFR signaling [125]. In addition to EGFR, protein expression of another member of the EGFR family, ERRB2/HER2, is increased in 18 – 28% of esophageal dysplasias and 22 – 24% of tumors [55, 126, 127].
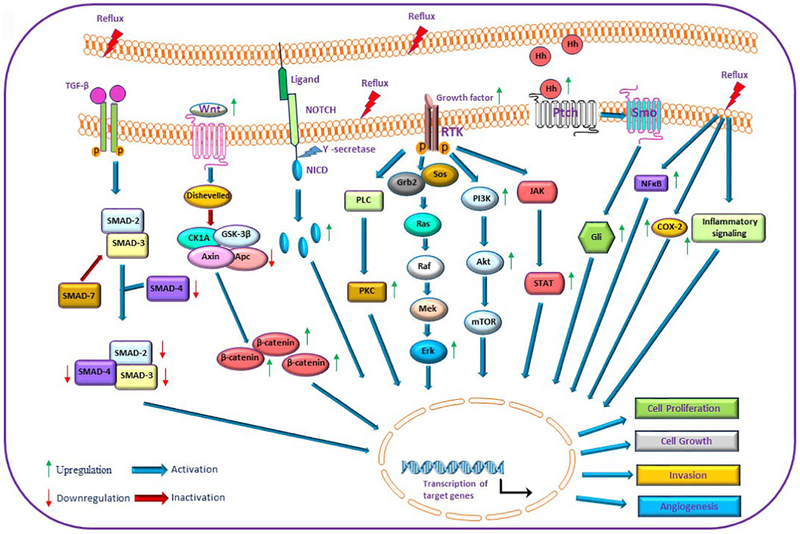
Figure 3. Schematic representation of major signalling pathways involved in the development and progression of esophageal adenocarcinoma.
Exposure of esophageal cells to gastroesophageal reflux causes the dysregulation of multiple signalling pathways that promote Barrett’s esophagus and esophageal adenocarcinoma. Activation of RTKs, Notch, Wnt, Hedgehog and other signalling cascades were found in esophageal adenocarcinoma. RTK – receptor tyrosine kinase; NICD- Notch intracellular domain; Hh – Hedgehog; TGF-β – transforming growth factor β; COX-2 – Cyclooxygenase-2; PKC – protein kinase C.
EAC is also characterized by strong activation of the IGF1R pathway. An increased staining for phosphorylated insulin receptor substrate 1 (pIRS1) that transmits signals from the IGFR receptors was found in 43.2% of BE and 70% of EAC patients [128]. Levels of IGF-1 ligand are increased in the sera of BE patients [129]. Another tyrosine kinase receptor, c-MET, which is regulated by hepatocyte growth factor (HGF), is highly induced in BE and EAC. c-Met immunoreactivity was found in 100% of dysplastic BE and EAC patients and correlated with poor prognosis [130, 131]. Activation of MET results in induction of β-catenin in EAC [130, 131].
The VEGF (vascular endothelial growth factor) signaling is also increased in EAC and found to regulate angiogenesis in BE and EAC. Both BE and tumor cells produce VEGF protein and its expression correlates with esophageal vascularization [132]. Among members of the VEGF family, expression of VEGF-A, -C are increased during progression from Barrett’s to EAC and suggested to correlate with metastasis and advanced disease [132, 133].
Downstream RTK signaling includes multiple effectors that regulate cell proliferation, survival, apoptosis, and angiogenesis (Figure 3). Among them, RAS and PI3K are frequently altered in EAC. Several studies have reported K-RAS activating mutations and amplifications of the K-RAS gene [134-137]. The central effector pathway downstream of RAS (ERK/MAPK) was found to be activated in 60% of EACs [138]. Similarly, serine/threonine kinase Akt, an effector of the PI3K pathway, was phosphorylated and activated in approximately 80% of HGD and EACs. This is in striking contrast to BE, where 62% of specimens showed low activity of Akt and the remaining cases were negative for p-Akt [139]. Reflux is thought to be responsible for activation of the PI3K-AKT and ERK/MAPK pathways [140-142].
TGF-β signaling
The transforming growth factor beta (TGF-β) pathway is implicated in regulation of cell growth, apoptosis, differentiation, and development. It is well known for its ability to inhibit proliferation and inflammation in normal tissues. However, during EAC development the TGF-β pathway can facilitate epithelial to mesenchymal transition (EMT), invasion, and metastasis [143-146]. Several studies reported unchanged or decreased levels of TGF-β mRNA in BE compared to normal squamous epithelium [144, 147, 148]. TGF-β expression is significantly increased in advanced stages of EAC [144]. EAC is also characterized by elevated expression of TGF-β-related proteins BMP4 and Activin A that are thought to promote invasive phenotype [148-150]. Notably, exposure to bile salts induces BMP4 and TGF-β1 [151, 152]. In contrast, TGF-β signal transducers (SMADs) are commonly lost in EACs (Figure 3). Among them, SMAD2 and SMAD4 are most affected [5]. Loss of expression of SMAD 2/4 was found in 30% to 70% of EAC cases [5, 153-155]. Expression of TGF- β receptor 2 is also downregulated in BE and EAC resulting in dysregulation of TGF-β signaling [154].
Notch signaling
The Notch signaling pathway is involved in different aspects of normal development and disease, from stem cell regulation and tissue morphogenesis to cancers and other diseases. Mechanistically, Notch signaling is mediated by a group of Notch receptors that are regulated by various ligands, such as Delta-like and Jagged. The binding of ligands leads to a series of proteolytic cleavages in the receptors, which release the Notch intracellular domain (NICD), which translocates into the nucleus and activates transcription of multiple target genes (Figure 3) [156]. In the esophagus, Notch signaling is active in the basal epithelial cell layer. Its inhibition contributes to the development of BE via the KLF4-dependent mechanism [154,157,158]. Reflux is likely to play role in this process [158, 159]. In contrast to BE, EAC shows induction of the NICD in 72% of EAC cases. Elevated Notch activity is associated with the state of differentiation and clinical stage of EAC [160]. Levels of JAG1/2, DDL1/3/4 ligands and Notch targets Hes-1, HEY1/2, NEYL are also increased in EAC patients [154, 160]. Elevated Notch signaling is thought to promote cancer stem cell phenotype, increases cancer cell survival and resistance to chemotherapy [160].
Hedgehog signaling
The Hedgehog (Hh) signaling pathway is critical for normal gut development. It also contributes to progression of intestinal metaplasia in the esophagus. In canonical signaling, it is activated by the binding of Hh ligands (Sonic, Indian, and Desert) to transmembrane receptors Patched (PTCH). This relieves PTCH repression of Smoothened (SMO) protein and subsequently activates Gli transcription factors that regulates transcription of Hh target genes (Figure 3). It has been demonstrated that Sonic Hedgehog signaling is suppressed in normal esophageal epithelium. However, it is strongly activated in BE that happens likely due to reflux [161-164]. Strong staining for proteins regulating the Hh pathway was found in 96% of EAC cases [164]. Approximately 90% of EAC patients also showed aberrant expression of Gli1 and Gli2 proteins [165]. This in contrast to ESCC, where levels of these proteins were found to be lower [164]. FOXA2 protein was recently identified among Shh targets that are upregulated in BE and EAC. This transcription factor was suggested to contribute to the development of Barrett’s metaplasia [163]. The Shh signaling may also promote BE through induction of BMP4 and SOX9 [162].
Wnt signaling
Aberrant activation of the Wnt/β-catenin signaling is a common event during the late stages of BE neoplastic transformation (Figure 3). This process underlies tumor progression [166-168]. Strong nuclear expression of β-catenin, which is indicative of its activation, was found in 44-53% of LGD, 42-93% of HGD and 61-63% of EAC [166-169]. Nuclear expression of β-catenin is uncommon in normal esophageal tissues and Barret’s metaplasia, although activation of β-catenin without its nuclear accumulation was reported in BE [166, 170]. In contrast to colonic and other tumors, dysregulation of the Wnt/β-catenin pathway is rarely caused by mutations in the APC, AXIN1, CDH1 or the β-catenin genes [5]. Instead, upregulation of the WNT2 loss of the WNT inhibitory factor 1 (WIF1), and promoter hypermethylation of sFRP1 (secreted Frizzled Related Protein 1) and APC genes have been reported in EAC [167]. Nuclear accumulation of β-catenin can be also induced by HGF and TNFβ in esophageal cells [130, 171].
Other significant signaling factors
Cyclooxygenase-2 (COX-2) protein, which catalyzes the formation of prostanoids, contributes to inflammation and tumorigenesis in various tissues (Figure 3). It is significantly upregulated in more than half of patients with BE and EAC [172-174]. Acid and bile strongly stimulate COX2 in vitro and in vivo [175,176]. Since inhibition of COX-2 activity suppresses inflammation and induce apoptosis, COX2 is considered as a target for prevention and treatment of esophageal cancer [177, 178]. Several studies have reported promising results for testing of COX inhibitors (aspirin and other non-steroidal anti-inflammatory drugs) in EAC (reviewed in [179, 180])
CDX2 is a homeobox transcription factor that is known for its role in processes of normal intestinal development. It shows low expression in the normal esophagus. CDX2 upregulation (by acid and bile) contributes to the development of BE [181-187]. Another group of transcription factors that is involved in Barrett’s pathogenesis belongs to the GATA family [45, 188, 189]. As discussed above, GATA 4/6 genes are amplified in EAC. Their protein expression is also progressively increased during EAC development from 30% in BE to 82% in high grade dysplasia [190].
5. Targeted therapy for esophageal cancer
RTKs are promising targets for EAC treatment. The FDA has approved trastuzumab, a monoclonal antibody against the HER2 ectodomain, for treatment of metastatic gastroesophageal tumors. Trastuzumab increased survival of advanced carcinoma patients in the phase-III ToGA trial, where HER2-positive patients were enrolled [191]. The second FDA approved biological agent is ramucirumab, a monoclonal antibody against human VEGFR2. Ramucirumab showed survival advantage in two randomized phase-III trials, REGARD and RAINBOW [192, 193]. The addition of bevacizumab, another VEGF antibody, to mFOLFOX6 also provided clinical benefits to patients with metastatic gastroesophageal adenocarcinoma [194]. Among other RTKs, suppression of MET with small molecule inhibitor, crizotinib, showed a promising response in MET-positive gastroesophageal adenocarcinoma patients [195]. The selective inhibition of MET using AMG-337 also showed anti-tumor activity in MET-amplified patients [196, 197].
However, most of the studies did not find significant survival benefits for anti-EGFR antibodies and small molecule inhibitors in patients with esophageal and gastroesophageal junction carcinomas [198-205]. Multiple factors may contribute to negative outcomes of these trials: EGFR mutations that prevents binding of inhibitors, RAS mutations, deletion of PTEN, amplifications of HER2 and MET, activation of downstream mediators such as PI3KCA and MAPK-ERK and/or activation of alternative oncogenic pathways in response to EGFR inhibition [202, 206].
A large leap forward in esophageal cancer treatment is immunotherapy. Suppression of immune checkpoints using antibodies against T-cell surface receptor (programmed cell death 1; PD-1) and its ligand programmed cell death ligand 1 (PD-L1) showed promising outcomes for patients with advanced gastric and gastroesophageal cancers. Among tested drugs is pembrolizumab, a monoclonal antibody against PD-L1 approved by the FDA. The KEYNOTE-028 trial (), which include a cohort of 23 patients with squamous cell carcinoma (SCC) and EAC showed manageable toxicity and durable antitumor activity of pembrolizumab in 29% of SCC and 40% of EAC [207]. In the phase-II KEYNOTE-180 study, objective response for pembrolizumab was observed in 13.8% patients with PD-L1 positive and 6.3% patients with PD-L1 negative tumors [208]. In the Checkmate-032 trial, patients with chemorefractory gastroesophageal junction cancer showed objective response to another PD-1 inhibitor, nivolumab. The response was seen in 12% of PD-L1 negative and 18% of PD-L1 positive patients [209]. The anti-PD-1 therapy also showed some response in two other clinical trials in gastroesophageal cancer patients [210, 211]. [210, 211]. Currently, the efficacy of immune checkpoint inhibitors is being investigated in several phase II/III trails such as KEYNOTE-061, KEYNOTE-062, KEYNOTE-181, KEYNOTE-182, Checkmate-577, JAVELIN-100 and JAVELIN-300. Further investigations of immune checkpoint inhibitors hold great promises and is expected to improve treatment of esophageal adenocarcinoma patients.
Concluding remarks
EAC has a complex etiology with involvement of multiple genetic, dietary, behavioral and environmental factors. GERD has been identified as one of the strongest risk factors for EAC. The currently accepted paradigm is that GERD leads to tissue damage and subsequent development of Barrett’s metaplasia (BE), which then progresses to esophageal dysplasia and invasive cancer. Development of EAC is underlined by continuous damage of DNA caused by reflux that promotes genomic instability and alterations of multiple tumor-suppressor and oncogenic pathways. Development of novel and more advanced techniques helped to better understand the molecular and cellular underpinning of this cancer. However, many questions about mechanisms of reflux-induced cellular damage and interactions between various signaling networks remain unanswered, thus limiting the development of effective preventive and treatment modalities. Further research will ultimately overcome these difficulties and help to identify novel molecular targets for EAC treatment.
Acknowledgements:
This work was supported by grants from the National Cancer Institute RO1 206564, R01 138833, and the Department of Veterans Affairs BX002115. The contents of this work are solely the responsibility of the authors and do not necessarily represent the official views of the Department of Veterans Affairs, the National Institutes of Health or University of Miami.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Conflict of interest: The authors have no conflict of interest to disclose.
References
- [1].Torre LA, Siegel RL, Ward EM.Jemal A, Global Cancer Incidence and Mortality Rates and Trends--An Update, Cancer Epidemiol Biomarkers Prev, 25 (2016) 16–27. [DOI] [PubMed] [Google Scholar]
- [2].Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA.Jemal A, Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries, CA Cancer J Clin, 68 (2018) 394–424. [DOI] [PubMed] [Google Scholar]
- [3].A.A.C. Society, Global Cancer Facts & Figures 3rd Edition. , (2015). [Google Scholar]
- [4].Brown LM, Devesa SS.Chow WH, Incidence of adenocarcinoma of the esophagus among white Americans by sex, stage, and age, J Natl Cancer Inst, 100 (2008) 1184–1187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [5].Dulak AM, Stojanov P, Peng S, Lawrence MS, Fox C, Stewart C, et al. , Exome and whole-genome sequencing of esophageal adenocarcinoma identifies recurrent driver events and mutational complexity, Nat Genet, 45 (2013) 478–486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [6].Siegel RL, Miller KD.Jemal A, Cancer statistics, 2019, CA Cancer J Clin, 69 (2019) 7–34. [DOI] [PubMed] [Google Scholar]
- [7].Engel LS, Chow WH, Vaughan TL, Gammon MD, Risch HA, Stanford JL, et al. , Population attributable risks of esophageal and gastric cancers, J Natl Cancer Inst, 95 (2003) 1404–1413. [DOI] [PubMed] [Google Scholar]
- [8].Rubenstein JH.Shaheen NJ, Epidemiology, Diagnosis, and Management of Esophageal Adenocarcinoma, Gastroenterology, 149 (2015) 302–317 e301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [9].Rubenstein JH.Taylor JB, Meta-analysis: the association of oesophageal adenocarcinoma with symptoms of gastro-oesophageal reflux, Aliment Pharmacol Ther, 32 (2010) 1222–1227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [10].Abdel-Latif MM, Duggan S, Reynolds JV.Kelleher D, Inflammation and esophageal carcinogenesis, Curr Opin Pharmacol, 9 (2009) 396–404. [DOI] [PubMed] [Google Scholar]
- [11].Bhardwaj V, Horvat A, Korolkova O, Washington MK, El-Rifai W, Dikalov SI, et al. , Prevention of DNA damage in Barrett's esophageal cells exposed to acidic bile salts, Carcinogenesis, 37 (2016) 1161–1169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [12].McQuaid KR, Laine L, Fennerty MB, Souza R.Spechler SJ, Systematic review: the role of bile acids in the pathogenesis of gastro-oesophageal reflux disease and related neoplasia, Aliment Pharmacol Ther, 34 (2011) 146–165. [DOI] [PubMed] [Google Scholar]
- [13].Runge TM, Abrams JA.Shaheen NJ, Epidemiology of Barrett's Esophagus and Esophageal Adenocarcinoma, Gastroenterol Clin North Am, 44 (2015) 203–231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [14].Lawrence MS, Stojanov P, Polak P, Kryukov GV, Cibulskis K, Sivachenko A, et al. , Mutational heterogeneity in cancer and the search for new cancer-associated genes, Nature, 499 (2013) 214–218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [15].Beroukhim R, Mermel CH, Porter D, Wei G, Raychaudhuri S, Donovan J, et al. , The landscape of somatic copy-number alteration across human cancers, Nature, 463 (2010) 899–905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [16].Menke-Pluymers MB, van Drunen E, Vissers KJ, Mulder AH, Tilanus HW.Hagemeijer A, Cytogenetic analysis of Barrett’s mucosa and adenocarcinoma of the distal esophagus and cardia, Cancer Genet Cytogenet, 90 (1996) 109–117. [DOI] [PubMed] [Google Scholar]
- [17].Rodriguez E, Rao PH, Ladanyi M, Altorki N, Albino AP, Kelsen DP, et al. , 11p13–15 is a specific region of chromosomal rearrangement in gastric and esophageal adenocarcinomas, Cancer Res, 50 (1990) 6410–6416. [PubMed] [Google Scholar]
- [18].Persons DL, Croughan WS, Borelli KA.Cherian R, Interphase cytogenetics of esophageal adenocarcinoma and precursor lesions, Cancer Genet Cytogenet, 106 (1998) 11–17. [DOI] [PubMed] [Google Scholar]
- [19].Paulson TG, Maley CC, Li X, Li H, Sanchez CA, Chao DL, et al. , Chromosomal instability and copy number alterations in Barrett’s esophagus and esophageal adenocarcinoma, Clin Cancer Res, 15 (2009) 3305–3314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [20].Moskaluk CA, Hu J.Perlman EJ, Comparative genomic hybridization of esophageal and gastroesophageal adenocarcinomas shows consensus areas of DNA gain and loss, Genes Chromosomes Cancer, 22 (1998) 305–311. [PubMed] [Google Scholar]
- [21].van Dekken H, Geelen E, Dinjens WN, Wijnhoven BP, Tilanus HW, Tanke HJ, et al. , Comparative genomic hybridization of cancer of the gastroesophageal junction: deletion of 14Q31–32.1 discriminates between esophageal (Barrett’s) and gastric cardia adenocarcinomas, Cancer Res, 59 (1999) 748–752. [PubMed] [Google Scholar]
- [22].Varis A, Puolakkainen P, Savolainen H, Kokkola A, Salo J, Nieminen O, et al. , DNA copy number profiling in esophageal Barrett adenocarcinoma: comparison with gastric adenocarcinoma and esophageal squamous cell carcinoma, Cancer Genet Cytogenet, 127 (2001) 53–58. [DOI] [PubMed] [Google Scholar]
- [23].Walch AK, Zitzelsberger HF, Bruch J, Keller G, Angermeier D, Aubele MM, et al. , Chromosomal imbalances in Barrett's adenocarcinoma and the metaplasia-dysplasia-carcinoma sequence, Am J Pathol, 156 (2000) 555–566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [24].Riegman PH, Vissers KJ, Alers JC, Geelen E, Hop WC, Tilanus HW, et al. , Genomic alterations in malignant transformation of Barrett's esophagus, Cancer Res, 61 (2001) 3164–3170. [PubMed] [Google Scholar]
- [25].Chaves P, Crespo M, Ribeiro C, Laranjeira C, Pereira AD, Suspiro A, et al. , Chromosomal analysis of Barrett's cells: demonstration of instability and detection of the metaplastic lineage involved, Mod Pathol, 20 (2007) 788–796. [DOI] [PubMed] [Google Scholar]
- [26].Suspiro A, Pereira AD, Afonso A, Albuquerque C, Chaves P, Soares J, et al. , Losses of heterozygosity on chromosomes 9p and 17p are frequent events in Barrett's metaplasia not associated with dysplasia or adenocarcinoma, Am J Gastroenterol, 98 (2003) 728–734. [DOI] [PubMed] [Google Scholar]
- [27].N. Cancer Genome Atlas Research, M. Albert Einstein College of, S. Analytical Biological, H. Barretos Cancer, M. Baylor College of, H. Beckman Research Institute of City of, et al. , Integrated genomic and molecular characterization of cervical cancer, Nature, 543 (2017) 378–384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [28].Ross-Innes CS, Becq J, Warren A, Cheetham RK, Northen H, O'Donovan M, et al. , Whole-genome sequencing provides new insights into the clonal architecture of Barrett's esophagus and esophageal adenocarcinoma, Nat Genet, 47 (2015) 1038–1046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [29].Weaver JMJ, Ross-Innes CS, Shannon N, Lynch AG, Forshew T, Barbera M, et al. , Ordering of mutations in preinvasive disease stages of esophageal carcinogenesis, Nat Genet, 46 (2014) 837–843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [30].Secrier M, Li X, de Silva N, Eldridge MD, Contino G, Bornschein J, et al. , Mutational signatures in esophageal adenocarcinoma define etiologically distinct subgroups with therapeutic relevance, Nat Genet, 48 (2016) 1131–1141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [31].Contino G, Vaughan TL, Whiteman D.Fitzgerald RC, The Evolving Genomic Landscape of Barrett's Esophagus and Esophageal Adenocarcinoma, Gastroenterology, 153 (2017) 657–673 e651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [32].Agrawal N, Jiao Y, Bettegowda C, Hutfless SM, Wang Y, David S, et al. , Comparative genomic analysis of esophageal adenocarcinoma and squamous cell carcinoma, Cancer Discov, 2 (2012) 899–905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [33].Nones K, Waddell N, Wayte N, Patch AM, Bailey P, Newell F, et al. , Genomic catastrophes frequently arise in esophageal adenocarcinoma and drive tumorigenesis, Nat Commun, 5 (2014) 5224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [34].Kaz AM, Wong CJ, Varadan V, Willis JE, Chak A.Grady WM, Global DNA methylation patterns in Barrett's esophagus, dysplastic Barrett's, and esophageal adenocarcinoma are associated with BMI, gender, and tobacco use, Clin Epigenetics, 8 (2016) 111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [35].Xu E, Gu J, Hawk ET, Wang KK, Lai M, Huang M, et al. , Genome-wide methylation analysis shows similar patterns in Barrett's esophagus and esophageal adenocarcinoma, Carcinogenesis, 34 (2013) 2750–2756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [36].Eads CA, Lord RV, Kurumboor SK, Wickramasinghe K, Skinner ML, Long TI, et al. , Fields of aberrant CpG island hypermethylation in Barrett’s esophagus and associated adenocarcinoma, Cancer Res, 60 (2000) 5021–5026. [PubMed] [Google Scholar]
- [37].Kaz AM, Wong CJ, Luo Y, Virgin JB, Washington MK, Willis JE, et al. , DNA methylation profiling in Barrett's esophagus and esophageal adenocarcinoma reveals unique methylation signatures and molecular subclasses, Epigenetics, 6 (2011) 1403–1412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [38].Agarwal A, Polineni R, Hussein Z, Vigoda I, Bhagat TD, Bhattacharyya S, et al. , Role of epigenetic alterations in the pathogenesis of Barrett’s esophagus and esophageal adenocarcinoma, Int J Clin Exp Pathol, 5 (2012) 382–396. [PMC free article] [PubMed] [Google Scholar]
- [39].Alvi MA, Liu X, O'Donovan M, Newton R, Wernisch L, Shannon NB, et al. , DNA methylation as an adjunct to histopathology to detect prevalent, inconspicuous dysplasia and early-stage neoplasia in Barrett's esophagus, Clin Cancer Res, 19 (2013) 878–888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [40].Alvarez H, Opalinska J, Zhou L, Sohal D, Fazzari MJ, Yu Y, et al. , Widespread hypomethylation occurs early and synergizes with gene amplification during esophageal carcinogenesis, PLoS Genet, 7 (2011) e1001356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [41].Kuester D, El-Rifai W, Peng D, Ruemmele P, Kroeckel I, Peters B, et al. , Silencing of MGMT expression by promoter hypermethylation in the metaplasia-dysplasia-carcinoma sequence of Barrett's esophagus, Cancer Lett, 275 (2009) 117–126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [42].Schulmann K, Sterian A, Berki A, Yin J, Sato F, Xu Y, et al. , Inactivation of p16, RUNX3, and HPP1 occurs early in Barrett's-associated neoplastic progression and predicts progression risk, Oncogene, 24 (2005) 4138–4148. [DOI] [PubMed] [Google Scholar]
- [43].Jin Z, Cheng Y, Gu W, Zheng Y, Sato F, Mori Y, et al. , A multicenter, double-blinded validation study of methylation biomarkers for progression prediction in Barrett’s esophagus, Cancer Res, 69 (2009) 4112–4115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [44].Clement G, Braunschweig R, Pasquier N, Bosman FT.Benhattar J, Methylation of APC, TIMP3, and TERT: a new predictive marker to distinguish Barrett’s oesophagus patients at risk for malignant transformation, J Pathol, 208 (2006) 100–107. [DOI] [PubMed] [Google Scholar]
- [45].Wang J, Qin R, Ma Y, Wu H, Peters H, Tyska M, et al. , Differential gene expression in normal esophagus and Barrett's esophagus, J Gastroenterol, 44 (2009) 897–911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [46].Dilworth MP, Nieto T, Stockton JD, Whalley CM, Tee L, James JD, et al. , Whole Genome Methylation Analysis of Nondysplastic Barrett Esophagus that Progresses to Invasive Cancer, Ann Surg, (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- [47].Clark RJ, Craig MP, Agrawal S.Kadakia M, microRNA involvement in the onset and progression of Barrett's esophagus: a systematic review, Oncotarget, 9 (2018) 8179–8196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [48].Kailasam A, Mittal SK.Agrawal DK, Epigenetics in the Pathogenesis of Esophageal Adenocarcinoma, Clin Transl Sci, 8 (2015) 394–402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [49].Olyaee M, Sontag S, Salman W, Schnell T, Mobarhan S, Eiznhamer D, et al. , Mucosal reactive oxygen species production in oesophagitis and Barrett's oesophagus, Gut, 37 (1995) 168–173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [50].Huo X, Juergens S, Zhang X, Rezaei D, Yu C, Strauch ED, et al. , Deoxycholic acid causes DNA damage while inducing apoptotic resistance through NF-kappaB activation in benign Barrett's epithelial cells, Am J Physiol Gastrointest Liver Physiol, 301 (2011) G278–286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [51].Dvorak K, Chavarria M, Payne CM, Ramsey L, Crowley-Weber C, Dvorakova B, et al. , Activation of the interleukin-6/STAT3 antiapoptotic pathway in esophageal cells by bile acids and low pH: relevance to barrett's esophagus, Clin Cancer Res, 13 (2007) 5305–5313. [DOI] [PubMed] [Google Scholar]
- [52].Jenkins GJ, D'Souza FR, Suzen SH, Eltahir ZS, James SA, Parry JM, et al. , Deoxycholic acid at neutral and acid pH, is genotoxic to oesophageal cells through the induction of ROS: The potential role of anti-oxidants in Barrett’s oesophagus, Carcinogenesis, 28 (2007) 136–142. [DOI] [PubMed] [Google Scholar]
- [53].Bhardwaj V, Gokulan RC, Horvat A, Yermalitskaya L, Korolkova O, Washington KM, et al. , Activation of NADPH oxidases leads to DNA damage in esophageal cells, Sci Rep, 7 (2017) 9956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [54].Dikalov SI.Harrison DG, Methods for detection of mitochondrial and cellular reactive oxygen species, Antioxid Redox Signal, 20 (2014) 372–382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [55].Almhanna K, Rosa M, Henderson-Jackson E, Jiang K, Shamekh R, Sayegh Z, et al. , Her-2 Expression in Gastroesophageal Intestinal Metaplasia, Dysplasia, and Adenocarcinoma, Appl Immunohistochem Mol Morphol, 24 (2016) 633–638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [56].Lee S, Han MJ, Lee KS, Back SC, Hwang D, Kim HY, et al. , Frequent occurrence of mitochondrial DNA mutations in Barrett's metaplasia without the presence of dysplasia, PLoS One, 7 (2012) e37571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [57].Olliver JR, Hardie LJ, Dexter S, Chalmers D.Wild CP, DNA damage levels are raised in Barrett's oesophageal mucosa relative to the squamous epithelium of the oesophagus, Biomarkers, 8 (2003) 509–521. [DOI] [PubMed] [Google Scholar]
- [58].von Holzen U, Chen T, Boquoi A, Richter JE, Falk GW, Klein-Szanto AJ, et al. , Evidence for DNA damage checkpoint activation in barrett esophagus, Transl Oncol, 3 (2010) 33–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [59].Olliver JR, Hardie LJ, Gong Y, Dexter S, Chalmers D, Harris KM, et al. , Risk factors, DNA damage, and disease progression in Barrett's esophagus, Cancer Epidemiol Biomarkers Prev, 14 (2005) 620–625. [DOI] [PubMed] [Google Scholar]
- [60].Zhang HY, Hormi-Carver K, Zhang X, Spechler SJ.Souza RF, In benign Barrett's epithelial cells, acid exposure generates reactive oxygen species that cause DNA double-strand breaks, Cancer Res, 69 (2009) 9083–9089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [61].Chen X, Ding YW, Yang G, Bondoc F, Lee MJ.Yang CS, Oxidative damage in an esophageal adenocarcinoma model with rats, Carcinogenesis, 21 (2000) 257–263. [DOI] [PubMed] [Google Scholar]
- [62].Piazuelo E, Cebrian C, Escartin A, Jimenez P, Soteras F, Ortego J, et al. , Superoxide dismutase prevents development of adenocarcinoma in a rat model of Barrett's esophagus, World J Gastroenterol, 11 (2005) 7436–7443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [63].Zaika E, Wei J, Yin D, Andl C, Moll U, El-Rifai W, et al. , p73 protein regulates DNA damage repair, FASEB J, 25 (2011) 4406–4414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [64].Theisen J, Peters JH, Fein M, Hughes M, Hagen JA, Demeester SR, et al. , The mutagenic potential of duodenoesophageal reflux, Ann Surg, 241 (2005) 63–68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [65].Thanan R, Ma N, Hiraku Y, lijima K, Koike T, Shimosegawa T, et al. , DNA Damage in CD133-Positive Cells in Barrett's Esophagus and Esophageal Adenocarcinoma, Mediators Inflamm, 2016 (2016) 7937814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [66].Iijima K.Shimosegawa T, Involvement of luminal nitric oxide in the pathogenesis of the gastroesophageal reflux disease spectrum, J Gastroenterol Hepatol, 29 (2014) 898–905. [DOI] [PubMed] [Google Scholar]
- [67].Halliwell B, Oxygen and nitrogen are pro-carcinogens. Damage to DNA by reactive oxygen, chlorine and nitrogen species: measurement, mechanism and the effects of nutrition, Mutat Res, 443 (1999) 37–52. [DOI] [PubMed] [Google Scholar]
- [68].Burnat G, Majka J.Konturek PC, Bile acids are multifunctional modulators of the Barrett's carcinogenesis, J Physiol Pharmacol, 61 (2010) 185–192. [PubMed] [Google Scholar]
- [69].Bonde P, Gao D, Chen L, Miyashita T, Montgomery E, Harmon JW, et al. , Duodenal reflux leads to down regulation of DNA mismatch repair pathway in an animal model of esophageal cancer, Ann Thorac Surg, 83 (2007) 433–440; discussion 440. [DOI] [PubMed] [Google Scholar]
- [70].Casson AG, Zheng Z, Evans SC, Veugelers PJ, Porter GA.Guernsey DL, Polymorphisms in DNA repair genes in the molecular pathogenesis of esophageal (Barrett) adenocarcinoma, Carcinogenesis, 26 (2005) 1536–1541. [DOI] [PubMed] [Google Scholar]
- [71].Tse D, Zhai R, Zhou W, Heist RS, Asomaning K, Su L, et al. , Polymorphisms of the NER pathway genes, ERCC1 and XPD are associated with esophageal adenocarcinoma risk, Cancer Causes Control, 19 (2008) 1077–1083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [72].Kadioglu E, Sardas S, Ergun M, Unal S.Karakaya AE, The role of oxidative DNA damage, DNA repair, GSTM1, SOD2 and OGG1 polymorphisms in individual susceptibility to Barrett's esophagus, Toxicol Ind Health, 26 (2010) 67–79. [DOI] [PubMed] [Google Scholar]
- [73].Hong J, Chen Z, Peng D, Zaika A, Revetta F, Washington MK, et al. , APE1-mediated DNA damage repair provides survival advantage for esophageal adenocarcinoma cells in response to acidic bile salts, Oncotarget, 7 (2016) 16688–16702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [74].Jimenez P, Piazuelo E, Sanchez MT, Ortego J, Soteras F.Lanas A, Free radicals and antioxidant systems in reflux esophagitis and Barrett’s esophagus, World J Gastroenterol, 11 (2005) 2697–2703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [75].Peng D, Hu T, Soutto M, Belkhiri A, Zaika A.El-Rifai W, Glutathione peroxidase 7 has potential tumour suppressor functions that are silenced by location-specific methylation in oesophageal adenocarcinoma, Gut, 63 (2014) 540–551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [76].van Lieshout EM, Tiemessen DM, Witteman BJ, Jansen JB.Peters WH, Low glutathione and glutathione S-transferase levels in Barrett's esophagus as compared to normal esophageal epithelium, Jpn J Cancer Res, 90 (1999) 81–85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [77].Rasanen JV, Sihvo EI, Rantanen TK, Ahotupa MO, Farkkila MA, Harjula A, et al. , Gastroesophageal reflux patients' defective antioxidative capacity in the proximal esophageal mucosa before antireflux surgery and also after 4-year follow-up, Ann Med, 40 (2008) 74–80. [DOI] [PubMed] [Google Scholar]
- [78].Kubo A, Levin TR, Block G, Rumore GJ, Quesenberry CP Jr., Buffler P, et al. , Dietary antioxidants, fruits, and vegetables and the risk of Barrett's esophagus, Am J Gastroenterol, 103 (2008) 1614–1623; quiz 1624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [79].Oh TY, Lee JS, Ahn BO, Cho H, Kim WB, Kim YB, et al. , Oxidative damages are critical in pathogenesis of reflux esophagitis: implication of antioxidants in its treatment, Free Radic Biol Med, 30 (2001) 905–915. [DOI] [PubMed] [Google Scholar]
- [80].Terry P, Lagergren J, Ye W, Nyren O.Wolk A, Antioxidants and cancers of the esophagus and gastric cardia, Int J Cancer, 87 (2000) 750–754. [PubMed] [Google Scholar]
- [81].Stachler MD, Camarda ND, Deitrick C, Kim A, Agoston AT, Odze RD, et al. , Detection of Mutations in Barrett's Esophagus Before Progression to High-Grade Dysplasia or Adenocarcinoma, Gastroenterology, 155 (2018) 156–167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [82].Flejou JF.Svrcek M, Barrett's oesophagus--a pathologist's view, Histopathology, 50 (2007) 3–14. [DOI] [PubMed] [Google Scholar]
- [83].Spechler SJ, Sharma P, Souza RF, Inadomi JM, Shaheen.A NJ. American Gastroenterological, American Gastroenterological Association technical review on the management of Barrett's esophagus, Gastroenterology, 140 (2011) e18–52; quiz e13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [84].Kaye PV, Haider SA, Ilyas M, James PD, Soomro I, Faisal W, et al. , Barrett's dysplasia and the Vienna classification: reproducibility, prediction of progression and impact of consensus reporting and p53 immunohistochemistry, Histopathology, 54 (2009) 699–712. [DOI] [PubMed] [Google Scholar]
- [85].Kastelein F, Biermann K, Steyerberg EW, Verheij J, Kalisvaart M, Looijenga LH, et al. , Aberrant p53 protein expression is associated with an increased risk of neoplastic progression in patients with Barrett's oesophagus, Gut, 62 (2013) 1676–1683. [DOI] [PubMed] [Google Scholar]
- [86].Skacel M, Petras RE, Rybicki LA, Gramlich TL, Richter JE, Falk GW, et al. , p53 expression in low grade dysplasia in Barrett's esophagus: correlation with interobserver agreement and disease progression, Am J Gastroenterol, 97 (2002) 2508–2513. [DOI] [PubMed] [Google Scholar]
- [87].Reid BJ, Prevo LJ, Galipeau PC, Sanchez CA, Longton G, Levine DS, et al. , Predictors of progression in Barrett's esophagus II: baseline 17p (p53) loss of heterozygosity identifies a patient subset at increased risk for neoplastic progression, Am J Gastroenterol, 96 (2001) 2839–2848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [88].Kalatskaya I, Overview of major molecular alterations during progression from Barrett's esophagus to esophageal adenocarcinoma, Ann N Y Acad Sci, 1381 (2016) 74–91. [DOI] [PubMed] [Google Scholar]
- [89].Roman S, Petre A, Thepot A, Hautefeuille A, Scoazec JY, Mion F, et al. , Downregulation of p63 upon exposure to bile salts and acid in normal and cancer esophageal cells in culture, Am J Physiol Gastrointest Liver Physiol, 293 (2007) G45–53. [DOI] [PubMed] [Google Scholar]
- [90].Caspa Gokulan R, Adcock JM, Zagol-Ikapitte I, Mernaugh R, Williams P, Washington KM, et al. , Gastroesophageal Reflux Induces Protein Adducts in the Esophagus, Cell Mol Gastroenterol Hepatol, 7 (2019) 480–482 e487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [91].Qiao D, Gaitonde SV, Qi W.Martinez JD, Deoxycholic acid suppresses p53 by stimulating proteasome-mediated p53 protein degradation, Carcinogenesis, 22 (2001) 957–964. [DOI] [PubMed] [Google Scholar]
- [92].Yan HP, Roberts LJ, Davies SS, Pohlmann P, Parl FF, Estes S, et al. , Isolevuglandins as a gauge of lipid peroxidation in human tumors, Free Radic Biol Med, 106 (2017) 62–68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [93].Parrales A.Iwakuma T, Targeting Oncogenic Mutant p53 for Cancer Therapy, Front Oncol, 5 (2015) 288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [94].Liu DS, Read M, Cullinane C, Azar WJ, Fennell CM, Montgomery KG, et al. , APR-246 potently inhibits tumour growth and overcomes chemoresistance in preclinical models of oesophageal adenocarcinoma, Gut, 64 (2015) 1506–1516. [DOI] [PubMed] [Google Scholar]
- [95].Liu DS, Duong CP, Haupt S, Montgomery KG, House CM, Azar WJ, et al. , Inhibiting the system xC(−)/glutathione axis selectively targets cancers with mutant-p53 accumulation, Nat Commun, 8 (2017) 14844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [96].Nimura Y, Mihara M, Ichimiya S, Sakiyama S, Seki N, Ohira M, et al. , p73, a gene related to p53, is not mutated in esophageal carcinomas, Int J Cancer, 78 (1998) 437–440. [DOI] [PubMed] [Google Scholar]
- [97].Zaika AI.El-Rifai W, The role of p53 protein family in gastrointestinal malignancies, Cell Death Differ, 13 (2006) 935–940. [DOI] [PubMed] [Google Scholar]
- [98].Glickman JN, Chen YY, Wang HH, Antonioli DA.Odze RD, Phenotypic characteristics of a distinctive multilayered epithelium suggests that it is a precursor in the development of Barrett's esophagus, Am J Surg Pathol, 25 (2001) 569–578. [DOI] [PubMed] [Google Scholar]
- [99].Daniely Y, Liao G, Dixon D, Linnoila RI, Lori A, Randell SH, et al. , Critical role of p63 in the development of a normal esophageal and tracheobronchial epithelium, Am J Physiol Cell Physiol, 287 (2004) C171–181. [DOI] [PubMed] [Google Scholar]
- [100].Zaika E, Bhardwaj V, Wei J, Washington MK, Souza R, El-Rifai W, et al. , Proinflammatory cytokines and bile acids upregulate DeltaNp73 protein, an inhibitor of p53 and p73 tumor suppressors, PLoS One, 8 (2013) e64306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [101].Bian YS, Osterheld MC, Fontolliet C, Bosman FT.Benhattar J, p16 inactivation by methylation of the CDKN2A promoter occurs early during neoplastic progression in Barrett's esophagus, Gastroenterology, 122 (2002) 1113–1121. [DOI] [PubMed] [Google Scholar]
- [102].Shi XY, Bhagwandeen B.Leong AS, p16, cyclin D1, Ki-67, and AMACR as markers for dysplasia in Barrett esophagus, Appl Immunohistochem Mol Morphol, 16 (2008) 447–452. [DOI] [PubMed] [Google Scholar]
- [103].M.A.K. Kotzev Andrey Iskrenov, Petrov Alexander Tcherveniakov Immunohistochemical Expression of Ki-67, PCNA, pRb, p16, p53, Bcl-2 and Bax in Esophageal Adenocarcinoma and Barrett's Associated Dysplasia Journal of Cancer Therapy, 3 (2012) 1092–1100. [Google Scholar]
- [104].Huang Y, Peters CJ, Fitzgerald RC.Gjerset RA, Progressive silencing of p14ARF in oesophageal adenocarcinoma, J Cell Mol Med, 13 (2009) 398–409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [105].Singh SP, Lipman J, Goldman H, Ellis FH Jr., Aizenman L, Cangi MG, et al. , Loss or altered subcellular localization of p27 in Barrett's associated adenocarcinoma, Cancer Res, 58 (1998) 1730–1735. [PubMed] [Google Scholar]
- [106].Merola E, Mattioli E, Minimo C, Zuo W, Rabitti C, Cicala M, et al. , Immunohistochemical evaluation of pRb2/p130, VEGF, EZH2, p53, p16, p21waf-1, p27, and PCNA in Barrett's esophagus, J Cell Physiol, 207 (2006) 512–519. [DOI] [PubMed] [Google Scholar]
- [107].Xiong DD, He RQ, Lan AH, Chen WJ, Luo YH, Ye ZH, et al. , Clinical significances of p27 in digestive tract cancers: a comprehensive analysis on immunohistochemistry staining, published literatures, microarray and RNA-seq data, Oncotarget, 9 (2018) 12284–12303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [108].Ellis FH Jr., Xu X, Kulke MH, LoCicero J 3rd.Loda M, Malignant transformation of the esophageal mucosa is enhanced in p27 knockout mice, J Thorac Cardiovasc Surg, 122 (2001) 809–814. [DOI] [PubMed] [Google Scholar]
- [109].N. The Cancer Genome Atlas Research, Kim J, Bowlby R, Mungall AJ, Robertson AG, Odze RD, et al. , Integrated genomic characterization of oesophageal carcinoma, Nature, 541 (2017) 169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [110].Ismail A, Bandla S, Reveiller M, Toia L, Zhou Z, Gooding WE, et al. , Early G(1) cyclin-dependent kinases as prognostic markers and potential therapeutic targets in esophageal adenocarcinoma, Clin Cancer Res, 17 (2011) 4513–4522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [111].Arber N, Gammon MD, Hibshoosh H, Britton JA, Zhang Y, Schonberg JB, et al. , Overexpression of cyclin D1 occurs in both squamous carcinomas and adenocarcinomas of the esophagus and in adenocarcinomas of the stomach, Hum Pathol, 30 (1999) 1087–1092. [DOI] [PubMed] [Google Scholar]
- [112].Han EK, Ng SC, Arber N, Begemann M.Weinstein IB, Roles of cyclin D1 and related genes in growth inhibition, senescence and apoptosis, Apoptosis, 4 (1999) 213–219. [DOI] [PubMed] [Google Scholar]
- [113].Bani-Hani K, Martin IG, Hardie LJ, Mapstone N, Briggs JA, Forman D, et al. , Prospective study of cyclin D1 overexpression in Barrett's esophagus: association with increased risk of adenocarcinoma, J Natl Cancer Inst, 92 (2000) 1316–1321. [DOI] [PubMed] [Google Scholar]
- [114].Kuwahara M, Hirai T, Yoshida K, Yamashita Y, Hihara J, Inoue H, et al. , p53, p21(Waf1/Cip1) and cyclin D1 protein expression and prognosis in esophageal cancer, Dis Esophagus, 12 (1999) 116–119. [DOI] [PubMed] [Google Scholar]
- [115].Izzo JG, Malhotra U, Wu TT, Ensor J, Babenko IM, Swisher SG, et al. , Impact of cyclin D1 A870G polymorphism in esophageal adenocarcinoma tumorigenesis, Semin Oncol, 32 (2005) S11–15. [DOI] [PubMed] [Google Scholar]
- [116].Zhou Z, Bandla S, Ye J, Xia Y, Que J, Luketich JD, et al. , Cyclin E involved in early stage carcinogenesis of esophageal adenocarcinoma by SNP DNA microarray and immunohistochemical studies, BMC Gastroenterol, 14 (2014) 78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [117].Jankowski J, Murphy S, Coghill G, Grant A, Wormsley KG, Sanders DS, et al. , Epidermal growth factor receptors in the oesophagus, Gut, 33 (1992) 439–443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [118].Pretto G, Gurski RR, Binato M, Navarini D, Aguiar WW.Meurer L, Increase of epidermal growth factor receptor expression in progression of GERD, Barrett, and adenocarcinoma of esophagus, Dig Dis Sci, 58 (2013) 115–122. [DOI] [PubMed] [Google Scholar]
- [119].Jankowski J, Coghill G, Tregaskis B, Hopwood D.Wormsley KG, Epidermal growth factor in the oesophagus, Gut, 33 (1992) 1448–1453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [120].Jankowski J, McMenemin R, Hopwood D, Penston J.Wormsley KG, Abnormal expression of growth regulatory factors in Barrett's oesophagus, Clin Sci (Lond), 81 (1991) 663–668. [DOI] [PubMed] [Google Scholar]
- [121].Cronin J, McAdam E, Danikas A, Tselepis C, Griffiths P, Baxter J, et al. , Epidermal growth factor receptor (EGFR) is overexpressed in high-grade dysplasia and adenocarcinoma of the esophagus and may represent a biomarker of histological progression in Barrett's esophagus (BE), Am J Gastroenterol, 106 (2011) 46–56. [DOI] [PubMed] [Google Scholar]
- [122].Wang KL, Wu TT, Choi IS, Wang H, Resetkova E, Correa AM, et al. , Expression of epidermal growth factor receptor in esophageal and esophagogastric junction adenocarcinomas: association with poor outcome, Cancer, 109 (2007) 658–667. [DOI] [PubMed] [Google Scholar]
- [123].Langer R, Von Rahden BH, Nahrig J, Von Weyhern C, Reiter R, Feith M, et al. , Prognostic significance of expression patterns of c-erbB-2, p53, p16INK4A, p27KIP1, cyclin D1 and epidermal growth factor receptor in oesophageal adenocarcinoma: a tissue microarray study, J Clin Pathol, 59 (2006) 631–634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [124].Navarini D, Gurski RR, Madalosso CA, Aita L, Meurer L.Fornari F, Epidermal growth factor receptor expression in esophageal adenocarcinoma: relationship with tumor stage and survival after esophagectomy, Gastroenterol Res Pract, 2012 (2012) 941954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [125].Ghatak S, Reveiller M, Toia L, Ivanov A, Godfrey TE.Peters JH, Bile acid at low pH reduces squamous differentiation and activates EGFR signaling in esophageal squamous cells in 3-D culture, J Gastrointest Surg, 17 (2013) 1723–1731. [DOI] [PubMed] [Google Scholar]
- [126].Reichelt U, Duesedau P, Tsourlakis M, Quaas A, Link BC, Schurr PG, et al. , Frequent homogeneous HER-2 amplification in primary and metastatic adenocarcinoma of the esophagus, Mod Pathol, 20 (2007) 120–129. [DOI] [PubMed] [Google Scholar]
- [127].Geddert H, Zeriouh M, Wolter M, Heise JW, Gabbert HE.Sarbia M, Gene amplification and protein overexpression of c-erb-b2 in Barrett carcinoma and its precursor lesions, Am J Clin Pathol, 118 (2002) 60–66. [DOI] [PubMed] [Google Scholar]
- [128].Greer KB, Kresak A, Bednarchik B, Dawson D, Li L, Chak A, et al. , Insulin/Insulin-Like Growth Factor-1 Pathway in Barrett's Carcinogenesis, Clin Transl Gastroenterol, 4 (2013) e31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [129].Sohda M, Kato H, Miyazaki T, Nakajima M, Fukuchi M, Manda R, et al. , The role of insulin-like growth factor 1 and insulin-like growth factor binding protein 3 in human esophageal cancer, Anticancer Res, 24 (2004) 3029–3034. [PubMed] [Google Scholar]
- [130].Anderson MR, Harrison R, Atherfold PA, Campbell MJ, Darnton SJ, Obszynska J, et al. , Met receptor signaling: a key effector in esophageal adenocarcinoma, Clin Cancer Res, 12 (2006) 5936–5943. [DOI] [PubMed] [Google Scholar]
- [131].Herrera LJ, El-Hefnawy T, Queiroz de Oliveira PE, Raja S, Finkelstein S, Gooding W, et al. , The HGF receptor c-Met is overexpressed in esophageal adenocarcinoma, Neoplasia, 7 (2005) 75–84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [132].Couvelard A, Paraf F, Gratio V, Scoazec JY, Henin D, Degott C, et al. , Angiogenesis in the neoplastic sequence of Barrett's oesophagus. Correlation with VEGF expression, J Pathol, 192 (2000) 14–18. [DOI] [PubMed] [Google Scholar]
- [133].Lord RV, Park JM, Wickramasinghe K, DeMeester SR, Oberg S, Salonga D, et al. , Vascular endothelial growth factor and basic fibroblast growth factor expression in esophageal adenocarcinoma and Barrett esophagus, J Thorac Cardiovasc Surg, 125 (2003) 246–253. [DOI] [PubMed] [Google Scholar]
- [134].Lord RV, O'Grady R, Sheehan C, Field AF.Ward RL, K-ras codon 12 mutations in Barrett's oesophagus and adenocarcinomas of the oesophagus and oesophagogastric junction, J Gastroenterol Hepatol, 15 (2000) 730–736. [DOI] [PubMed] [Google Scholar]
- [135].Trautmann B, Wittekind C, Strobel D, Meixner H, Keymling J, Gossner L, et al. , K-ras point mutations are rare events in premalignant forms of Barrett's oesophagus, Eur J Gastroenterol Hepatol, 8 (1996) 799–804. [PubMed] [Google Scholar]
- [136].Sommerer F, Vieth M, Markwarth A, Rohrich K, Vomschloss S, May A, et al. , Mutations of BRAF and KRAS2 in the development of Barrett's adenocarcinoma, Oncogene, 23 (2004) 554–558. [DOI] [PubMed] [Google Scholar]
- [137].Abdelatif OM, Chandler FW, Mills LR, McGuire BS, Pantazis CG.Barrett JM, Differential expression of c-myc and H-ras oncogenes in Barrett's epithelium. A study using colorimetric in situ hybridization, Arch Pathol Lab Med, 115 (1991) 880–885. [PubMed] [Google Scholar]
- [138].Keld R, Guo B, Downey P, Gulmann C, Ang YS.Sharrocks AD, The ERK MAP kinase-PEA3/ETV4-MMP-1 axis is operative in oesophageal adenocarcinoma, Mol Cancer, 9 (2010) 313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [139].Sagatys E, Garrett CR, Boulware D, Kelley S, Malafa M, Cheng JQ, et al. , Activation of the serine/threonine protein kinase Akt during the progression of Barrett neoplasia, Hum Pathol, 38 (2007) 1526–1531. [DOI] [PubMed] [Google Scholar]
- [140].Mariette C, Perrais M, Leteurtre E, Jonckheere N, Hemon B, Pigny P, et al. , Transcriptional regulation of human mucin MUC4 by bile acids in oesophageal cancer cells is promoter-dependent and involves activation of the phosphatidylinositol 3-kinase signalling pathway, Biochem J, 377 (2004) 701–708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [141].Song S, Byrd JC, Guha S, Liu KF, Koul D.Bresalier RS, Induction of MUC5AC mucin by conjugated bile acids in the esophagus involves the phosphatidylinositol 3-kinase/protein kinase C/activator protein-1 pathway, Cancer, 117 (2011) 2386–2397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [142].Souza RF, Shewmake K, Terada LS.Spechler SJ, Acid exposure activates the mitogen-activated protein kinase pathways in Barrett's esophagus, Gastroenterology, 122 (2002) 299–307. [DOI] [PubMed] [Google Scholar]
- [143].Hong S, Lee HJ, Kim SJ.Hahm KB, Connection between inflammation and carcinogenesis in gastrointestinal tract: focus on TGF-beta signaling, World J Gastroenterol, 16 (2010) 2080–2093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [144].von Rahden BH, Stein HJ, Feith M, Puhringer F, Theisen J, Siewert JR, et al. , Overexpression of TGF-beta1 in esophageal (Barrett's) adenocarcinoma is associated with advanced stage of disease and poor prognosis, Mol Carcinog, 45 (2006) 786–794. [DOI] [PubMed] [Google Scholar]
- [145].Rees JR, Onwuegbusi BA, Save VE, Alderson D.Fitzgerald RC, In vivo and in vitro evidence for transforming growth factor-beta1-mediated epithelial to mesenchymal transition in esophageal adenocarcinoma, Cancer Res, 66 (2006) 9583–9590. [DOI] [PubMed] [Google Scholar]
- [146].Kim AH, Lebman DA, Dietz CM, Snyder SR, Eley KW.Chung TD, Transforming growth factor-beta is an endogenous radioresistance factor in the esophageal adenocarcinoma cell line OE-33, Int J Oncol, 23 (2003) 1593–1599. [PubMed] [Google Scholar]
- [147].Lee SW, Lien HC, Lin CC, Wen MC.Chang CS, Low Expression of Transforming Growth Factor beta in the Epithelium of Barrett's Esophagus, Gastroenterology Res, 11 (2018) 189–194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [148].Taylor C, Loomans HA, Le Bras GF, Koumangoye RB, Romero-Morales AI, Quast LL, et al. , Activin a signaling regulates cell invasion and proliferation in esophageal adenocarcinoma, Oncotarget, 6 (2015) 34228–34244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [149].Kestens C, Siersema PD, Offerhaus GJ.van Baal JW, BMP4 Signaling Is Able to Induce an Epithelial-Mesenchymal Transition-Like Phenotype in Barrett's Esophagus and Esophageal Adenocarcinoma through Induction of SNAIL2, PLoS One, 11 (2016) e0155754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [150].Seder CW, Hartojo W, Lin L, Silvers AL, Wang Z, Thomas DG, et al. , INHBA overexpression promotes cell proliferation and may be epigenetically regulated in esophageal adenocarcinoma, J Thorac Oncol, 4 (2009) 455–462. [DOI] [PubMed] [Google Scholar]
- [151].Perng DW, Chang KT, Su KC, Wu YC, Wu MT, Hsu WH, et al. , Exposure of airway epithelium to bile acids associated with gastroesophageal reflux symptoms: a relation to transforming growth factor-beta1 production and fibroblast proliferation, Chest, 132 (2007) 1548–1556. [DOI] [PubMed] [Google Scholar]
- [152].Zhou G, Sun YG, Wang HB, Wang WQ, Wang XW.Fang DC, Acid and bile salt upregulate BMP4 expression in human esophageal epithelium cells, Scand J Gastroenterol, 44 (2009) 926–932. [DOI] [PubMed] [Google Scholar]
- [153].Onwuegbusi BA, Aitchison A, Chin SF, Kranjac T, Mills I, Huang Y, et al. , Impaired transforming growth factor beta signalling in Barrett's carcinogenesis due to frequent SMAD4 inactivation, Gut, 55 (2006) 764–774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [154].Mendelson J, Song S, Li Y, Maru DM, Mishra B, Davila M, et al. , Dysfunctional transforming growth factor-beta signaling with constitutively active Notch signaling in Barrett's esophageal adenocarcinoma, Cancer, 117 (2011) 3691–3702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [155].Song S, Maru DM, Ajani JA, Chan CH, Honjo S, Lin HK, et al. , Loss of TGF-beta adaptor beta2SP activates notch signaling and SOX9 expression in esophageal adenocarcinoma, Cancer Res, 73 (2013) 2159–2169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [156].Bray SJ, Notch signalling in context, Nat Rev Mol Cell Biol, 17 (2016) 722–735. [DOI] [PubMed] [Google Scholar]
- [157].Vega ME, Giroux V, Natsuizaka M, Liu M, Klein-Szanto AJ, Stairs DB, et al. , Inhibition of Notch signaling enhances transdifferentiation of the esophageal squamous epithelium towards a Barrett's-like metaplasia via KLF4, Cell Cycle, 13 (2014) 3857–3866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [158].Wang YC, Wang ZQ, Yuan Y, Ren T, Ni PZ.Chen LQ, Notch Signaling Pathway Is Inhibited in the Development of Barrett's Esophagus: An In Vivo and In Vitro Study, Can J Gastroenterol Hepatol, 2018 (2018) 4149317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [159].Tamagawa Y, Ishimura N, Uno G, Yuki T, Kazumori H, Ishihara S, et al. , Notch signaling pathway and Cdx2 expression in the development of Barrett's esophagus, Lab Invest, 92 (2012) 896–909. [DOI] [PubMed] [Google Scholar]
- [160].Wang Z, Da Silva TG, Jin K, Han X, Ranganathan P, Zhu X, et al. , Notch signaling drives stemness and tumorigenicity of esophageal adenocarcinoma, Cancer Res, 74 (2014) 6364–6374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [161].Milano F, van Baal JW, Buttar NS, Rygiel AM, de Kort F, DeMars CJ, et al. , Bone morphogenetic protein 4 expressed in esophagitis induces a columnar phenotype in esophageal squamous cells, Gastroenterology, 132 (2007) 2412–2421. [DOI] [PubMed] [Google Scholar]
- [162].Wang DH, Clemons NJ, Miyashita T, Dupuy AJ, Zhang W, Szczepny A, et al. , Aberrant epithelial-mesenchymal Hedgehog signaling characterizes Barrett's metaplasia, Gastroenterology, 138 (2010) 1810–1822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [163].Wang DH, Tiwari A, Kim ME, Clemons NJ, Regmi NL, Hodges WA, et al. , Hedgehog signaling regulates FOXA2 in esophageal embryogenesis and Barrett's metaplasia, J Clin Invest, 124 (2014) 3767–3780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [164].Yang L, Wang LS, Chen XL, Gatalica Z, Qiu S, Liu Z, et al. , Hedgehog signaling activation in the development of squamous cell carcinoma and adenocarcinoma of esophagus, Int J Biochem Mol Biol, 3 (2012) 46–57. [PMC free article] [PubMed] [Google Scholar]
- [165].Wang L, Jin JQ, Zhou Y, Tian Z, Jablons DM.He B, Gli is activated and promotes epithelial-mesenchymal transition in human esophageal adenocarcinoma, Oncotarget, 9 (2018) 853–865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [166].Bian YS, Osterheld MC, Bosman FT, Fontolliet C.Benhattar J, Nuclear accumulation of beta-catenin is a common and early event during neoplastic progression of Barrett esophagus, Am J Clin Pathol, 114 (2000) 583–590. [DOI] [PubMed] [Google Scholar]
- [167].Clement G, Braunschweig R, Pasquier N, Bosman FT.Benhattar J, Alterations of the Wnt signaling pathway during the neoplastic progression of Barrett's esophagus, Oncogene, 25 (2006) 3084–3092. [DOI] [PubMed] [Google Scholar]
- [168].Moyes LH, McEwan H, Radulescu S, Pawlikowski J, Lamm CG, Nixon C, et al. , Activation of Wnt signalling promotes development of dysplasia in Barrett's oesophagus, J Pathol, 228 (2012) 99–112. [DOI] [PubMed] [Google Scholar]
- [169].Osterheld MC, Bian YS, Bosman FT, Benhattar J.Fontolliet C, Beta-catenin expression and its association with prognostic factors in adenocarcinoma developed in Barrett esophagus, Am J Clin Pathol, 117 (2002) 451–456. [DOI] [PubMed] [Google Scholar]
- [170].Lyros O, Rafiee P, Nie L, Medda R, Jovanovic N, Otterson MF, et al. , Wnt/beta-Catenin Signaling Activation beyond Robust Nuclear beta-Catenin Accumulation in Nondysplastic Barrett's Esophagus: Regulation via Dickkopf-1, Neoplasia, 17 (2015) 598–611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [171].Tselepis C, Perry I, Dawson C, Hardy R, Darnton SJ, McConkey C, et al. , Tumour necrosis factor-alpha in Barrett's oesophagus: a potential novel mechanism of action, Oncogene, 21 (2002) 6071–6081. [DOI] [PubMed] [Google Scholar]
- [172].Morris CD, Armstrong GR, Bigley G, Green H.Attwood SE, Cyclooxygenase-2 expression in the Barrett's metaplasia-dysplasia-adenocarcinoma sequence, Am J Gastroenterol, 96 (2001) 990–996. [DOI] [PubMed] [Google Scholar]
- [173].Lagorce C, Paraf F, Vidaud D, Couvelard A, Wendum D, Martin A, et al. , Cyclooxygenase-2 is expressed frequently and early in Barrett's oesophagus and associated adenocarcinoma, Histopathology, 42 (2003) 457–465. [DOI] [PubMed] [Google Scholar]
- [174].Nguyen T, Tang Z, Younes M, Alsarraj A, Ramsey D, Fitzgerald S, et al. , Esophageal COX-2 expression is increased in Barrett's esophagus, obesity, and smoking, Dig Dis Sci, 60 (2015) 65–73. [DOI] [PubMed] [Google Scholar]
- [175].Shirvani VN, Ouatu-Lascar R, Kaur BS, Omary MB.Triadafilopoulos G, Cyclooxygenase 2 expression in Barrett's esophagus and adenocarcinoma: Ex vivo induction by bile salts and acid exposure, Gastroenterology, 118 (2000) 487–496. [DOI] [PubMed] [Google Scholar]
- [176].Zhang F, Altorki NK, Wu YC, Soslow RA, Subbaramaiah K.Dannenberg AJ, Duodenal reflux induces cyclooxygenase-2 in the esophageal mucosa of rats: evidence for involvement of bile acids, Gastroenterology, 121 (2001) 1391–1399. [DOI] [PubMed] [Google Scholar]
- [177].Souza RF, Shewmake K, Beer DG, Cryer B.Spechler SJ, Selective inhibition of cyclooxygenase-2 suppresses growth and induces apoptosis in human esophageal adenocarcinoma cells, Cancer Res, 60 (2000) 5767–5772. [PubMed] [Google Scholar]
- [178].Buttar NS, Wang KK, Leontovich O, Westcott JY, Pacifico RJ, Anderson MA, et al. , Chemoprevention of esophageal adenocarcinoma by COX-2 inhibitors in an animal model of Barrett's esophagus, Gastroenterology, 122 (2002) 1101–1112. [DOI] [PubMed] [Google Scholar]
- [179].Hao W, Shen Y, Feng M, Wang H, Lin M, Fang Y, et al. , Aspirin acts in esophageal cancer: a brief review, J Thorac Dis, 10 (2018) 2490–2497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [180].Zhang S, Zhang XQ, Ding XW, Yang RK, Huang SL, Kastelein F, et al. , Cyclooxygenase inhibitors use is associated with reduced risk of esophageal adenocarcinoma in patients with Barrett's esophagus: a meta-analysis, Br J Cancer, 110 (2014) 2378–2388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [181].Groisman GM, Amar M.Meir A, Expression of the intestinal marker Cdx2 in the columnar-lined esophagus with and without intestinal (Barrett's) metaplasia, Mod Pathol, 17 (2004) 1282–1288. [DOI] [PubMed] [Google Scholar]
- [182].Pera M, Pera M, de Bolos C, Brito MJ, Palacin A, Grande L, et al. , Duodenal-content reflux into the esophagus leads to expression of Cdx2 and Muc2 in areas of squamous epithelium in rats, J Gastrointest Surg, 11 (2007) 869–874. [DOI] [PubMed] [Google Scholar]
- [183].Tatsuta T, Mukaisho K, Sugihara H, Miwa K, Tani T.Hattori T, Expression of Cdx2 in early GRCL of Barrett's esophagus induced in rats by duodenal reflux, Dig Dis Sci, 50 (2005) 425–431. [DOI] [PubMed] [Google Scholar]
- [184].Kazumori H, Ishihara S, Rumi MA, Kadowaki Y.Kinoshita Y, Bile acids directly augment caudal related homeobox gene Cdx2 expression in oesophageal keratinocytes in Barrett's epithelium, Gut, 55 (2006) 16–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [185].Marchetti M, Caliot E.Pringault E, Chronic acid exposure leads to activation of the cdx2 intestinal homeobox gene in a long-term culture of mouse esophageal keratinocytes, J Cell Sci, 116 (2003) 1429–1436. [DOI] [PubMed] [Google Scholar]
- [186].Liu T, Zhang XY, So CK, Wang S, Wang P, Yan LY, et al. , Regulation of Cdx2 expression by promoter methylation, and effects of Cdx2 transfection on morphology and gene expression of human esophageal epithelial cells, Carcinogenesis, 28 (2007) 488–496. [DOI] [PubMed] [Google Scholar]
- [187].Huo X, Zhang HY, Zhang XI, Lynch JP, Strauch ED, Wang JY, et al. , Acid and bile salt-induced CDX2 expression differs in esophageal squamous cells from patients with and without Barrett's esophagus, Gastroenterology, 139 (2010) 194–203 e191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [188].Chen X, Qin R, Liu B, Ma Y, Su Y, Yang CS, et al. , Multilayered epithelium in a rat model and human Barrett's esophagus: similar expression patterns of transcription factors and differentiation markers, BMC Gastroenterol, 8 (2008) 1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [189].Miller CT, Moy JR, Lin L, Schipper M, Normolle D, Brenner DE, et al. , Gene amplification in esophageal adenocarcinomas and Barrett's with high-grade dysplasia, Clin Cancer Res, 9 (2003) 4819–4825. [PubMed] [Google Scholar]
- [190].Pavlov K, Honing J, Meijer C, Boersma-van Ek W, Peters FT, van den Berg A, et al. , GATA6 expression in Barrett's oesophagus and oesophageal adenocarcinoma, Dig Liver Dis, 47 (2015) 73–80. [DOI] [PubMed] [Google Scholar]
- [191].Bang YJ, Van Cutsem E, Feyereislova A, Chung HC, Shen L, Sawaki A, et al. , Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer (ToGA): a phase 3, open-label, randomised controlled trial, Lancet, 376 (2010) 687–697. [DOI] [PubMed] [Google Scholar]
- [192].Fuchs CS, Tomasek J, Yong CJ, Dumitru F, Passalacqua R, Goswami C, et al. , Ramucirumab monotherapy for previously treated advanced gastric or gastro-oesophageal junction adenocarcinoma (REGARD): an international, randomised, multicentre, placebo-controlled, phase 3 trial, Lancet, 383 (2014) 31–39. [DOI] [PubMed] [Google Scholar]
- [193].Wilke H, Muro K, Van Cutsem E, Oh SC, Bodoky G, Shimada Y, et al. , Ramucirumab plus paclitaxel versus placebo plus paclitaxel in patients with previously treated advanced gastric or gastro-oesophageal junction adenocarcinoma (RAINBOW): a double-blind, randomised phase 3 trial, Lancet Oncol, 15 (2014) 1224–1235. [DOI] [PubMed] [Google Scholar]
- [194].Li J, Yao X, Kortmansky JS, Fischbach NA, Stein S, Deng Y, et al. , Phase II Study of Modified FOLFOX6 With Bevacizumab in Metastatic Gastroesophageal Adenocarcinoma, Am J Clin Oncol, 40 (2017) 146–151. [DOI] [PubMed] [Google Scholar]
- [195].Lennerz JK, Kwak EL, Ackerman A, Michael M, Fox SB, Bergethon K, et al. , MET amplification identifies a small and aggressive subgroup of esophagogastric adenocarcinoma with evidence of responsiveness to crizotinib, J Clin Oncol, 29 (2011) 4803–4810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [196].Van Cutsem E, Karaszewska B, Kang YK, Chung HC, Shankaran V, Siena S, et al. , A Multicenter Phase II Study of AMG 337 in Patients with MET-Amplified Gastric/Gastroesophageal Junction/Esophageal Adenocarcinoma and Other MET-Amplified Solid Tumors, Clin Cancer Res, 25 (2019) 2414–2423. [DOI] [PubMed] [Google Scholar]
- [197].Kwak PLEL , Hamid O , Janku F , Kittaneh M , Virgil D et al. , , Clinical activity of AMG 337, an oral MET kinase inhibitor, in adult patients (pts) with MET-amplified gastroesophageal junction (GEJ), gastric (G), or esophageal (E) cancer., Journal of Clinical Oncology, 33 (2015) 1–1.25332246 [Google Scholar]
- [198].Enzinger PC, Burtness BA, Niedzwiecki D, Ye X, Douglas K, Ilson DH, et al. , CALGB 80403 (Alliance)/E1206: A Randomized Phase II Study of Three Chemotherapy Regimens Plus Cetuximab in Metastatic Esophageal and Gastroesophageal Junction Cancers, J Clin Oncol, 34 (2016) 2736–2742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [199].Tebbutt NC, Price TJ, Ferraro DA, Wong N, Veillard AS, Hall M, et al. , Panitumumab added to docetaxel, cisplatin and fluoropyrimidine in oesophagogastric cancer: ATTAX3 phase II trial, Br J Cancer, 114 (2016) 505–509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [200].Waddell T, Chau I, Cunningham D, Gonzalez D, Okines AF, Okines C, et al. , Epirubicin, oxaliplatin, and capecitabine with or without panitumumab for patients with previously untreated advanced oesophagogastric cancer (REAL3): a randomised, open-label phase 3 trial, Lancet Oncol, 14 (2013) 481–489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [201].Dutton SJ, Ferry DR, Blazeby JM, Abbas H, Dahle-Smith A, Mansoor W, et al. , Gefitinib for oesophageal cancer progressing after chemotherapy (COG): a phase 3, multicentre, double-blind, placebo-controlled randomised trial, Lancet Oncol, 15 (2014) 894–904. [DOI] [PubMed] [Google Scholar]
- [202].Yoon H, Karapetyan L, Choudhary A, Kosozi R, Bali GS, Zaidi AH, et al. , Phase II Study of Irinotecan Plus Panitumumab as Second-Line Therapy for Patients with Advanced Esophageal Adenocarcinoma, Oncologist, 23 (2018) 1004–e1102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [203].Suntharalingam M, Winter K, Ilson D, Dicker AP, Kachnic L, Konski A, et al. , Effect of the Addition of Cetuximab to Paclitaxel, Cisplatin, and Radiation Therapy for Patients With Esophageal Cancer: The NRG Oncology RTOG 0436 Phase 3 Randomized Clinical Trial, JAMA Oncol, 3 (2017) 1520–1528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [204].Stahl M, Maderer A, Lordick F, Mihaljevic AL, Kanzler S, Hoehler T, et al. , Perioperative chemotherapy with or without epidermal growth factor receptor blockade in unselected patients with locally advanced oesophagogastric adenocarcinoma: Randomized phase II study with advanced biomarker program of the German Cancer Society (AIO/CAO STO-0801), Eur J Cancer, 93 (2018) 119–126. [DOI] [PubMed] [Google Scholar]
- [205].Ruhstaller T, Thuss-Patience P, Hayoz S, Schacher S, Knorrenschild JR, Schnider A, et al. , Neoadjuvant chemotherapy followed by chemoradiation and surgery with and without cetuximab in patients with resectable esophageal cancer: a randomized, open-label, phase III trial (SAKK 75/08), Ann Oncol, 29 (2018) 1386–1393. [DOI] [PubMed] [Google Scholar]
- [206].Maron SB, Alpert L, Kwak HA, Lomnicki S, Chase L, Xu D, et al. , Targeted Therapies for Targeted Populations: Anti-EGFR Treatment for EGFR-Amplified Gastroesophageal Adenocarcinoma, Cancer Discov, 8 (2018) 696–713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [207].Doi SAP-PT , Jalal SI , Mai-Dang H , Saraf S , Csiki MK et al. , , Updated results for the advanced esophageal carcinoma cohort of the phase Ib KEYNOTE-028 study of pembrolizumab (MK-3475). Journal of Clinical Oncology 34 (2016) 7–7. [Google Scholar]
- [208].Shah MA, Kojima T, Hochhauser D, Enzinger P, Raimbourg J, Hollebecque A, et al. , Efficacy and Safety of Pembrolizumab for Heavily Pretreated Patients With Advanced, Metastatic Adenocarcinoma or Squamous Cell Carcinoma of the Esophagus: The Phase 2 KEYNOTE-180 Study, JAMA Oncol, (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- [209].Le JCBDT , Calvo E , Kim JW , Ascierto PA , Ott PSPA et al. ,, Safety and activity of nivolumab monotherapy in advanced and metastatic (A/M) gastric or gastroesophageal junction cancer (GC/GEC): Results from the CheckMate-032 study., Journal of Clinical Oncology 34 (2016) 6–6.26578608 [Google Scholar]
- [210].Kang YK, Boku N, Satoh T, Ryu MH, Chao Y, Kato K, et al. , Nivolumab in patients with advanced gastric or gastro-oesophageal junction cancer refractory to, or intolerant of, at least two previous chemotherapy regimens (ONO-4538-12, ATTRACTION-2): a randomised, double-blind, placebo-controlled, phase 3 trial, Lancet, 390 (2017) 2461–2471. [DOI] [PubMed] [Google Scholar]
- [211].Fuchs CS, Doi T, Jang RW, Muro K, Satoh T, Machado M, et al. , Safety and Efficacy of Pembrolizumab Monotherapy in Patients With Previously Treated Advanced Gastric and Gastroesophageal Junction Cancer: Phase 2 Clinical KEYNOTE-059 Trial, JAMA Oncol, 4 (2018) e180013. [DOI] [PMC free article] [PubMed] [Google Scholar]