Abstract
Clostridium difficile infection (CDI) is one of the most common nosocomial infections worldwide and an urgent public health threat. Epidemiological and experimental studies have demonstrated an association between nonsteroidal anti-inflammatory drug (NSAID) exposure and enhanced susceptibility to, and severity of, CDI. NSAIDs target cyclooxygenase enzymes and inhibit the production of prostaglandins (PGs), but the therapeutic potential of exogenous introduction of PGs for the treatment of CDI has not been explored. In this study, we report that treatment with the FDA-approved stable PGE1 analogue, misoprostol, protects mice against C. difficile-associated mortality, intestinal pathology, and CDI-mediated intestinal permeability. Furthermore, we report that the effect of misoprostol on the gastrointestinal tract contributes to increased recovery of the gut microbiota following antibiotic perturbation. Together, these data implicate PGs as an important host-factor associated with recovery to C. difficile-associated disease and demonstrate the potential for misoprostol in the treatment of CDI. Further studies to explore the safety and efficacy of misoprostol treatment of CDI in humans is needed.
Introduction
Clostridium difficile is a nosocomial pathogen that colonizes the colon and causes severe diarrhea and pseudomembranous colitis [1]. C. difficile infection (CDI) is the most common nosocomial infection in the United States [2] Risk for CDI is primarily associated with exposure to antibiotics, which perturbs the resident gut microbiota leading to loss of colonization resistance and susceptibility to C. difficile colonization and disease [3]. Interestingly, recent studies have revealed a growing list of environmental factors, including dietary factors and pharmaceutical drugs, that may work independently or in concert with antimicrobials to shape the epidemiology of CDI [4–11]. Understanding the mechanisms and influence of non-antibiotic factors on CDI promises to provide increased understanding of CDI and the development of novel therapeutics.
Nonsteroidal anti-inflammatory drugs (NSAIDs) are among the most commonly used drugs in the United States [12, 13]. NSAIDs target cyclooxygenase (COX) enzymes and block the production of prostaglandins (PGs), which are lipid mediators associated with many aspects of health and disease. Recent epidemiological studies have established an association between NSAID use and CDI [14, 15]. Early animal studies also suggested this association [16]. Recent work by our group and others has demonstrated that NSAIDs perturb the gut microbiota and dramatically exacerbate CDI severity in mice [15, 17–20]. For example, we determined that NSAID treatment prior to CDI leads to loss of tight junction integrity and increased gut microbiota translocation [17]. These studies provide indirect evidence that PG production and downstream signaling is an essential host response during CDI and PG-mediated tight junction maintenance is necessary for protection against severe CDI and microbiota translocation. Despite this evidence, the role of PGs in CDI remains to be elucidated. A potential protective effect of exogenously-administered (pharmacological) PGs in CDI may provide a novel therapeutic or preventive strategy against C. difficile-colitis. Supporting this rationale are data showing that stable PG analogues prevent colitis caused by other insults in rodent models [21–23].
Exogenous introduction of the synthetic PGE1 analogue misoprostol has been FDA approved to prevent or treat stomach and duodenal ulcers in patients taking NSAIDs because of its protective effects on the gastric mucosa [24, 25]. Misoprostol binds to each of the four unique the PGE2 E prostanoid (EP) receptors and mimics the cytoprotective effects of PGE2 necessary for maintaining epithelial integrity [26]. Based on the detrimental impact of NSAIDs during CDI, we reasoned that misoprostol may have promise as a therapeutic treatment during CDI. In this study, we report that introduction of misoprostol following infection with a highly virulent strain of C. difficile, protects mice against severe CDI and decreases mucosal permeability. Furthermore, we show that misoprostol does not alter the gut microbiota at baseline but promotes the recovery of the community following antibiotic perturbation. Together, this work demonstrates that misoprostol is protective against severe CDI in mice and suggests that this FDA-approved PGE1 analogue may have therapeutic potential for the treatment of severe CDI.
Results
Misoprostol protects against severe Clostridium difficile infection in a mouse model
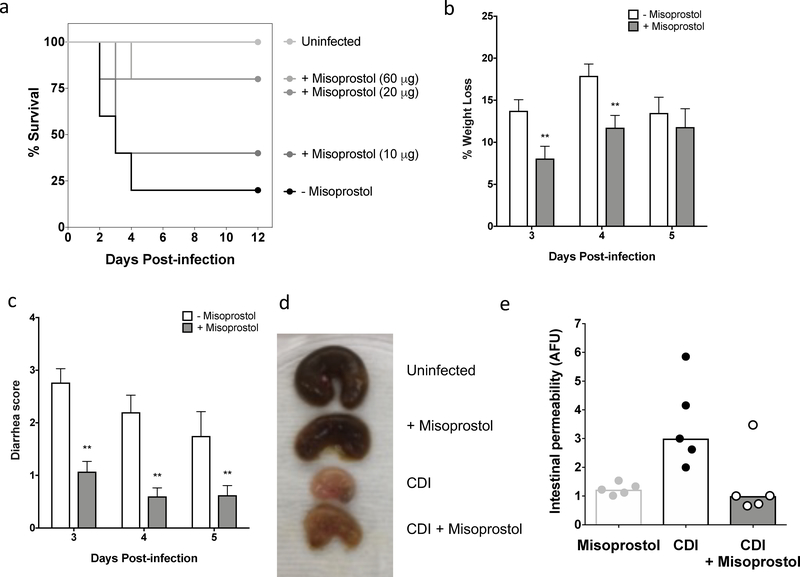
Inhibition of PG production by NSAIDs exacerbates C. difficile-associated disease, suggesting an important role for PGs in the immune response to CDI [17]. To determine the role of PGs in C. difficile-colitis and to explore the therapeutic potential of synthetic PGs in CDI, we treated C. difficile-infected mice with misoprostol. Female C57BL/6 mice were given the broad spectrum antibiotic cefoperazone and subsequently infected with 106 spores of C. difficile strain M7404 (NAP1/BI/027). A high inoculum of this virulent epidemic strain of C. difficile strain was used to induce severe CDI in these animals. Beginning on day 1 post-infection, mice were given daily doses of misoprostol via intraperitoneal administration for 7 days (Fig. 1a). Misoprostol treatment protected mice from CDI-associated mortality in a dose-dependent manner (Figure 2a). Mice treated with misoprostol showed decreased weight loss, less severe diarrhea, and larger and grossly healthier ceca (Figure 2b,c,d).
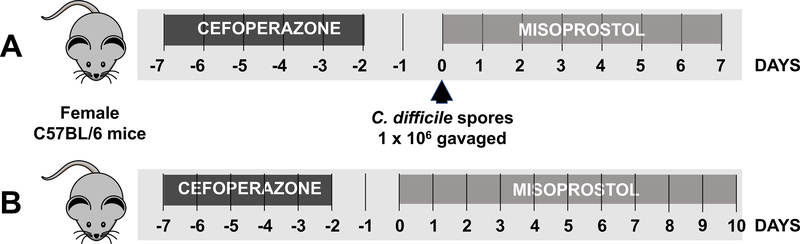
Fig. 1: Experimental design.
(a) Female C57BL/6J mice were exposed to cefoperazone ad libitum in drinking water for days −7 to −2 prior to inoculation with 1 × 106 spores of the C. difficile strain M7404 by gavage on day 0. Following infection mice were administered intraperitoneal injections of misoprostol once daily for 7 days. (b) Mice were exposed to cefoperazone as in (a) then treated with misoprostol (20 μg) daily by intraperitoneal injection through 10 days. Feces were collected for microbiota studies.
Fig. 2: Misoprostol protects animals against severe CDI and decreases intestinal permeability.
Female C57BL/6 mice were treated with cefoperazone for 5 days followed by 2 days of recovery in regular drinking water and then challenged with 1 × 106 spores of strain M7404. (a) Mice received misoprostol by intraperitoneal (i.p.) injection daily starting on the day of inoculation. 60 mg i.p. daily showed the same results as 20 mg (not shown). N = 5 mice per group. * P <0.05 compared to uninfected control by Log-rank (Mantel-Cox) test. (b.) Mice were treated with cefoperazone for 5 days followed by 2 days of recovery in regular drinking water and then challenged with 1 × 104 spores of strain M7404. Mice received misoprostol by i.p. injection (or vehicle) daily and stools were scored for severity of diarrhea on a 4 point scale (1 -- normal, 2 -soft stool/discolored, 3 -- wet stained tail/mucous, 4 -- liquid/no stool). N = 5 mice per group. **P <0.01, ***P < 0.001 by ANOVA followed by Tukey’s multiple comparisons test. (c) To assess intestinal permeability mice were infected with 1×104 spores of M7404 and given misoprostol 20 mg/mouse by IP injection 30 min before C. difficile inoculation, 24 h later and at the time of FITC-dextran treatment. 2 d post infection mice were gavaged with FITC-dextran or vehicle control, then euthanized 4 h later and concentrations of FITC-dextran in plasma were determined. N = 5 mice per group. *P < 0.05 by ANOVA followed by Tukey’s multiple comparisons test.
Previously, we reported that NSAID treatment leads to decreased tight junction integrity and increased intestinal permeability and translocation of microbiota [17]. To determine the extent to which misoprostol promotes maintenance of tight junction integrity following CDI, we quantified escape of luminal FITC-Dextran into the systemic circulation during CDI infection following misoprostol treatment, a measure of barrier integrity. Misoprostol treatment significantly decreased the level of FITC-dextran uptake in infected mice (Figure 2e). These data suggest that misoprostol promotes the maintenance of tight junction integrity and decreased CDI-associated intestinal permeability. Together, these data further reinforce the concept that PGs play an important and protective role in CDI and suggest a pharmacological role for misoprostol in the treatment of severe CDI.
Misoprostol enhances gut microbiota recovery following antibiotic perturbation
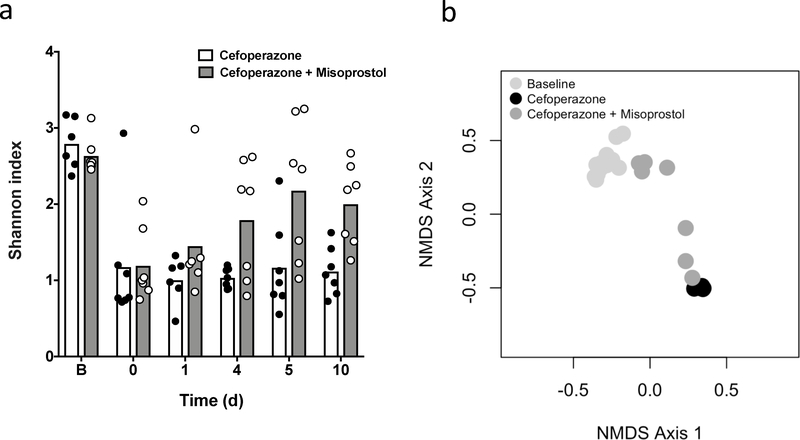
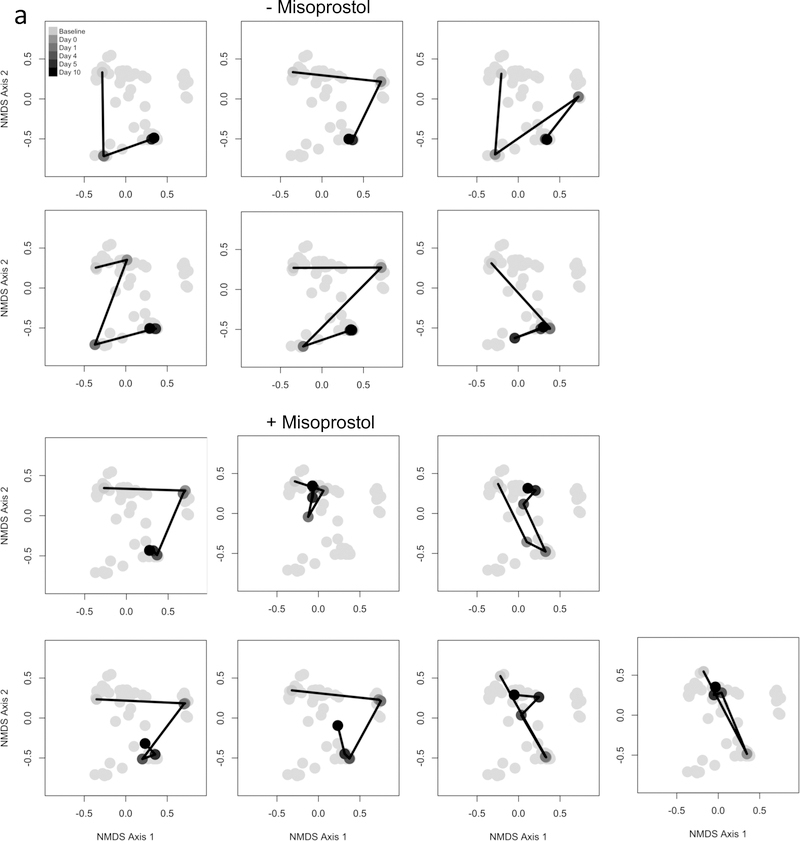
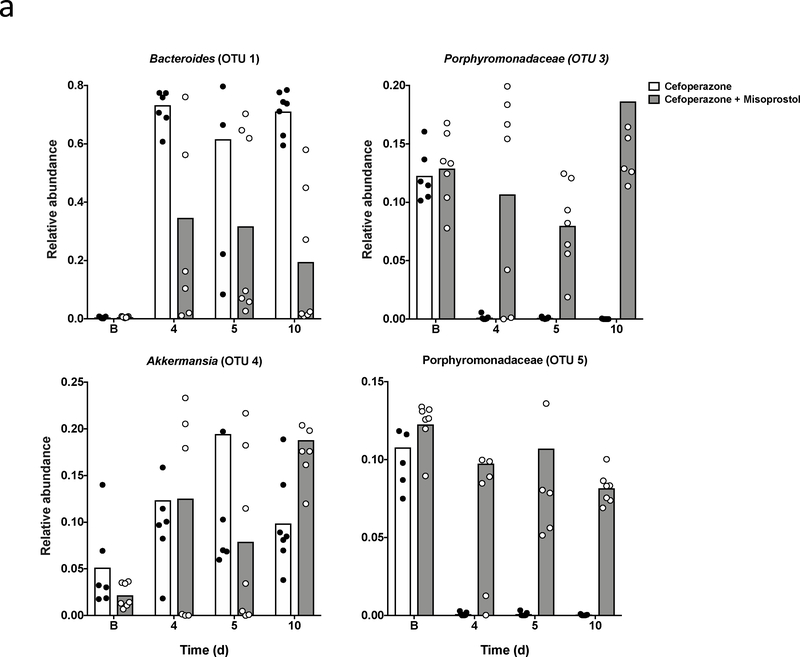
The gut microbiota plays an important role in resistance to CDI, the manifestation of C. difficile-associated disease, and clearance of C. difficile following initial colonization and infection [27, 28]. Moreover, recovery of the healthy microbiota is essential for the prevention of recurrent CDI [29]. Our group and others have reported that NSAIDs have a marked impact on the structure of the gut microbiota, which we reason contributes to increased disease severity during CDI and a lack of microbiota recovery [15, 17, 19]. We reasoned that PGs may have a beneficial impact on the gut microbiota following antibiotic perturbation. To test the effect of PGs on the microbiota, we treated mice with daily IP-injections of misoprostol for 10 days following cefoperazone pre-treatment and surveyed the microbiota (Figure 1b). After the first IP-injection with misoprostol or vehicle, mice treated with cefoperazone showed no significant differences in a-diversity or community structure regardless of treatment group (Figure 3a,b; P = 0.076). However, following 4 days of treatment, we observed that mice given daily misoprostol injections exhibited increased a-diversity and significant recovery of the microbiota towards baseline (Figure 3a; Figure 4). On day 10, five of seven mice that were treated with daily misoprostol injections showed significant recovery of the microbiota (Figure 3b, Figure 4). Community structure in these mice was statistically indistinguishable to the pre-treated community (Figure 3b; P = 0.099). In contrast, mice treated with vehicle and cefoperazone showed continued perturbation of the microbiota on day 10 (Figure 4). Misoprostol-mediated recovery of the microbiota was highlighted by an enrichment in operational taxonomic units (OTUs) affiliated with Porphyromonadaceae (OTUs 3 and 5) and Akkermansia (OTU 4), and decreases in a Bacteriodes (OTU 1) that bloomed following cefoperazone treatment (Figure 5a,b). Taken together these data demonstrate that PGs plays a protective role in acute CDI and suggest that misoprostol promotes recovery of the gut microbiota following antibiotic treatment.
Fig. 3: Misoprostol treatment promotes recovery of the gut microbiota following antibiotic exposure.
(a) Shannon diversity index for cefoperazone treated (black) or cefoperazone + Misoprostol (purple) treated mice. (b) Non-metric multidimensional scaling (NMDS) ordination showing β-diversity as measured by Yue and Clayton’s measure of dissimilarity (θYC) on day 10. Significance between baseline (grey) and cefoperazone- treated (black) or cefoperazone + Misoprostol (grey) samples measured using analysis of molecular variance (AMOVA) (P<0.001).
Fig 4. Gut microbiota dynamics following antibiotic and misoprostol treatment.
(a) NMDS ordination showing the dynamics of recovery (baseline, day 0, day 1, day 4, day 5, and day 10) following antibiotic treatment and subsequent introduction of PBS (grey scale) or Misoprostol (purple scale).
Fig 5. Rebound of taxa following antibiotic treatment following introduction of misoprostol.
(a) Dynamics of recovery of differentially abundant taxa over a 10-day time course. B represents baseline microbiota pre-treatment. Significantly altered taxa were identified using the linear discriminant analysis (LDA) effect size (LEfSe) algorithm.
Discussion
This brief report demonstrates that the PGE1 analogue misoprostol rescues antibioticexposed mice from lethal CDI, an effect correlated with improved colonic barrier function and beneficial effects on microbiota resilience (return to pre-antibiotic homeostasis). The rationale for these studies is supported by studies documenting that pharmacological impairment of PG synthesis increases the risk for, and/or severity of, CDI and by studies showing that misoprostol can prevent gastrointestinal complications of NSAIDs.
Because misoprostol is an FDA-approved PGE1 analogue it might be possible to repurpose the drug for the treatment or prevention of C. difficile-associated disease or other causes of colitis. In fact, other studies in animal models suggest that misoprostol protects the colon against colitis-inducing insults[30–33]. However, it is important to acknowledge several limitations in this study, including a lack of mechanistic data to support exactly how misoprostol demonstrated benefit in mice. In addition, correlative studies of gut barrier function (FITC-dextran permeability) do not provide information related to the exact mechanisms of improved barrier function induced by the drug. Similarly, actions of misoprostol on the microbiome were not shown to be causally protective against CDI in this model. Our studies are best interpreted as pilot studies supporting the rationale for more investigation of this approach.
Methods
Animals and experimental model of Clostridium difficile infection.
All animal experiments were approved by the Animal Care and Use Committees of Vanderbilt University (protocol #M1600272–00). 6–8 weeks old C57BL/6 (male or female fig1 says female mice) were purchased from Jackson Laboratories and given two weeks to equilibrate their microbiota prior to experimentation. Mice were housed with autoclaved water, food and bedding and all experiments manipulations were performed in a biosafety level 2 laminar flow hood. Mice were housed in individual cages under the same conditions during the experiment and all mice were shown to be culture negative for C. difficile prior to infection. For the C. difficile infection model, mice were given cefoperazone at 0.5 mg/ml in drinking water ad libitum for 5 days followed to a 2-day recovery period and subsequent infection. Mice were infected via oral gavage with 1×106 spores of C. difficile (NAP1/BI/027 strain M7404) resuspended in PBS. Uninfected mice were given cefoperazone in parallel and infected with vehicle alone [34]. Mice were monitored for survival or were euthanized after reaching a terminal endpoint of appearing moribund or experiencing weight loss > than 20% from baseline. Diarrhea severity was scored on a 4-point scale (1=normal, 2=soft stool/discolored, 3=wet stained tail/mucous, 4=liquid/no stool).
Misoprostol treatment
Misoprostol was obtained from Cayman Chemicals (Ann Arbor, MI) in a methyl acetate solution that was dried down under nitrogen and resuspended in phosphate buffered saline (PBS) immediately before each administration. Misoprostol, when administered, was delivered via intraperitoneal (i.p.) injection at 20 μg, unless otherwise stated. Mice not treated with misoprostol received an injection of PBS at corresponding timepoints.
DNA extraction and Microbiota Analysis
Stool samples were collected fresh from mice at timepoints described. Microbial genomic DNA was extracted using the PowerSoil DNA isolation kit (MO BIO Laboratories), according to the manufacturer’s instructions. The V4 region of the 16S rRNA gene from each sample was amplified and sequenced using the Illumina Sequencing platform. Sequences were curated using mothur software package [35], as previously described and performed [4]. Sequences were aligned to the SILVA 16S rRNA sequence database [36], chimeric sequences were identified by UCHIME [37] and removed, and the number of sequences in each sample was rarefied to 4,200. FASTQ sequence data used in this study have been deposited to the Sequence Read Archive (SRA) at NCBI under the accession number SRP189862. Sequences were clustered into OTUs based on a 3% distance cutoff calculated using OptiClust. Sequences were classified using the RDP training set (version 16), and OTUs were assigned a taxonomic classification based on the majority consensus of sequences within each OTU using a naive Bayesian classifier [38]. α-diversity was calculated using the Shannon diversity index and β- diversity was calculated using the θYC distance metric [39].
Statistical analyses
Statistical analyses were performed using GraphPad Prism (La Jolla, CA) and R. Data are presented as means ± SEM of values determined from the indicated number of samples. Repeated measures ANOVA were used in body weights over time followed by Tukey multiple comparison post-test or by using Student t test as appropriate. Kruskal Wallis (non-parametric) ANOVA was used for ordinal (ranked categorical) variables in histology and clinical scores. Kaplan-Meier plots were used to analyze survival in the challenged mice by Mantel-Cox Log-rank test. A P value ≤ 0.05 was considered significant a priori. Analysis of molecular variance (AMOVA) was performed to determine significance between the community structures of different groups of samples based on θYC distance matrices [40].The biomarker discovery algorithm LEfSe (linear discriminant analysis (LDA) effect size) was implemented to identify features (OTUs) differentially abundant in each group [41].
Highlights.
Treatment with the FDA-approved stable prostaglandin (PG)E1 analogue, misoprostol, protects mice against C. difficile-associated mortality, intestinal pathology, and CDI- mediated intestinal permeability.
The effect of misoprostol on the gastrointestinal tract contributes to increased recovery of the gut microbiota following antibiotic perturbation.
These data implicate prostaglandins as an important host-factor associated with recovery to C. difficile-associated disease and demonstrate the potential for misoprostol in the treatment of CDI.
Acknowledgments
We thank Dr. Dena Lyras (Monash University) for providing Clostridium difficile strains.
Funding and conflict of interest statements.
This work was supported by the National Institutes of Health (NIH) grants TR001723, AI121796, and the Vanderbilt Digestive Diseases Research Center NIH grant DK058404. J.P.Z was supported by F32AI120553 and K22AI7220. D.M.A. and the Vanderbilt University Medical Center hold patents for the treatment and prevention of Clostridium difficile colitis using misoprostol.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- [1].Kelly CP, LaMont JT Clostridium difficile infection. Annu Rev Med 49 (1998) 375–90. [DOI] [PubMed] [Google Scholar]
- [2].Magill SS, O’Leary E, Janelle SJ, Thompson DL, Dumyati G, Nadle J, et al. Changes in Prevalence of Health Care-Associated Infections in U.S. Hospitals. N Engl J Med 379 (2018) 1732–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [3].Theriot CM, Koenigsknecht MJ, Carlson PE, Hatton GE, Nelson AM, Li B, et al. Antibiotic-induced shifts in the mouse gut microbiome and metabolome increase susceptibility to Clostridium difficile infection. Nat Commun 5 (2014) 3114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [4].Zackular JP, Moore JL, Jordan AT, Juttukonda LJ, Noto MJ, Nicholson MR, et al. Dietary zinc alters the microbiota and decreases resistance to Clostridium difficile infection. Nat Med 22 (2016) 1330–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [5].Hryckowian AJ, Pruss KM, Sonnenburg JL The emerging metabolic view of Clostridium difficile pathogenesis. Curr Opin Microbiol 35 (2017) 42–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [6].Hryckowian AJ, Van Treuren W, Smits SA, Davis NM, Gardner JO, Bouley DM, et al. Microbiota-accessible carbohydrates suppress Clostridium difficile infection in a murine model. Nat Microbiol 3 (2018) 662–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [7].Collins J, Robinson C, Danhof H, Knetsch CW, van Leeuwen HC, Lawley TD, et al. Dietary trehalose enhances virulence of epidemic Clostridium difficile. Nature 553 (2018) 291–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [8].Zackular JP, Skaar EP The role of zinc and nutritional immunity in Clostridium difficile infection. Gut Microbes 9 (2018) 469–76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [9].Rogers MA, Greene MT, Saint S, Chenoweth CE, Malani PN, Trivedi I, et al. Higher rates of Clostridium difficile infection among smokers. PLoS One 7 (2012) e42091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [10].Rogers MA, Greene MT, Young VB, Saint S, Langa KM, Kao JY, et al. Depression, antidepressant medications, and risk of Clostridium difficile infection. BMC Med 11 (2013) 121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [11].Rogers MA, Micic D, Blumberg N, Young VB, Aronoff DM Storage duration of red blood cell transfusion and Clostridium difficile infection: a within person comparison. PLoS One 9 (2014) e89332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [12].Cryer B, Barnett MA, Wagner J, Wilcox CM Overuse and Misperceptions of Nonsteroidal Anti-inflammatory Drugs in the United States. Am J Med Sci 352 (2016) 472–80. [DOI] [PubMed] [Google Scholar]
- [13].Fowler TO, Durham CO, Planton J, Edlund BJ Use of nonsteroidal anti-inflammatory drugs in the older adult. J Am Assoc Nurse Pract 26 (2014) 414–23. [DOI] [PubMed] [Google Scholar]
- [14].Permpalung N, Upala S, Sanguankeo A, Sornprom S Association between NSAIDs and Clostridium difficile-Associated Diarrhea: A Systematic Review and Meta-Analysis. Can J Gastroenterol Hepatol 2016 (2016) 7431838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [15].Rogers MAM, Aronoff DM The influence of non-steroidal anti-inflammatory drugs on the gut microbiome. Clin Microbiol Infect 22 (2016) 178.e1–.e9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [16].Robert A, Bundy GL, Field SO, Nezamis JE, Davis JP, Hanchar AJ, et al. Prevention of cecitis in hamsters by certain prostaglandins. Prostaglandins 29 (1985) 961–80. [DOI] [PubMed] [Google Scholar]
- [17].Maseda D, Zackular JP, Trindade B, Kirk L, Roxas JL, Rogers LM, et al. Nonsteroidal Anti-inflammatory Drugs Alter the Microbiota and Exacerbate Clostridium difficile Colitis while Dysregulating the Inflammatory Response. MBio 10 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- [18].Muñoz-Miralles J, Trindade BC, Castro-Córdova P, Bergin IL, Kirk LA, Gil F, et al. Indomethacin increases severity of Clostridium difficile infection in mouse model. Future Microbiol 13 (2018) 1271–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [19].Liang X, Bittinger K, Li X, Abernethy DR, Bushman FD, FitzGerald GA Bidirectional interactions between indomethacin and the murine intestinal microbiota. Elife 4 (2015) e08973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [20].Xiao X, Nakatsu G, Jin Y, Wong S, Yu J, Lau JY Gut Microbiota Mediates Protection Against Enteropathy Induced by Indomethacin. Sci Rep 7 (2017) 40317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [21].Onizuka Y, Murase K, Furusu H, Isomoto H, Mizuta Y, Takeshima F, et al. Effect of intrarectal prostaglandin E2 analogue (enprostil) on trinitrobenzenesulphonic acid-induced colitis in rats. J Int Med Res 28 (2000) 28–35. [DOI] [PubMed] [Google Scholar]
- [22].Tessner TG, Cohn SM, Schloemann S, Stenson WF Prostaglandins prevent decreased epithelial cell proliferation associated with dextran sodium sulfate injury in mice. Gastroenterology 115 (1998) 874–82. [DOI] [PubMed] [Google Scholar]
- [23].Sasaki S, Hirata I, Maemura K, Hamamoto N, Murano M, Toshina K, et al. Prostaglandin E2 inhibits lesion formation in dextran sodium sulphate-induced colitis in rats and reduces the levels of mucosal inflammatory cytokines. Scand J Immunol 51 (2000) 23–8. [DOI] [PubMed] [Google Scholar]
- [24].Watkinson G, Hopkins A, Akbar FA The therapeutic efficacy of misoprostol in peptic ulcer disease. Postgrad Med J 64 Suppl 1 (1988) 60–77. [PubMed] [Google Scholar]
- [25].Walt RP Misoprostol for the treatment of peptic ulcer and antiinflammatory-drug- induced gastroduodenal ulceration. N Engl J Med 327 (1992) 1575–80. [DOI] [PubMed] [Google Scholar]
- [26].Montrose DC, Nakanishi M, Murphy RC, Zarini S, McAleer JP, Vella AT, et al. The role of PGE2 in intestinal inflammation and tumorigenesis. Prostaglandins Other Lipid Mediat 116–117 (2015) 26–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [27].Abt MC, McKenney PT, Pamer EG Clostridium difficile colitis: pathogenesis and host defence. Nat Rev Microbiol 14 (2016) 609–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [28].Jenior ML, Leslie JL, Young VB, Schloss PD Colonizes Alternative Nutrient Niches during Infection across Distinct Murine Gut Microbiomes. mSystems 2 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- [29].Theriot CM, Young VB Interactions Between the Gastrointestinal Microbiome and Clostridium difficile. Annu Rev Microbiol 69 (2015) 445–61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [30].Fedorak RN, Empey LR, MacArthur C, Jewell LD Misoprostol provides a colonic mucosal protective effect during acetic acid-induced colitis in rats. Gastroenterology 98 (1990) 615–25. [DOI] [PubMed] [Google Scholar]
- [31].Torsher KJ, Empey LR, Fedorak RN Misoprostol therapy following trinitrobenzene sulfonic acid-induced colitis accelerates healing. Prostaglandins Leukot Essent Fatty Acids 45 (1992) 275–81. [DOI] [PubMed] [Google Scholar]
- [32].Yamada T, Specian RD, Granger DN, Gaginella TS, Grisham MB Misoprostol attenuates acetic acid-induced increases in mucosal permeability and inflammation: role of blood flow. Am J Physiol 261 (1991) G332–9. [DOI] [PubMed] [Google Scholar]
- [33].Yamada T, Fujimoto K, Tso P, Fujimoto T, Gaginella TS, Grisham MB Misoprostol accelerates colonic mucosal repair in acetic acid-induced colitis. J Pharmacol Exp Ther 260 (1992) 313–8. [PubMed] [Google Scholar]
- [34].Carter GP, Douce GR, Govind R, Howarth PM, Mackin KE, Spencer J, et al. The anti-sigma factor TcdC modulates hypervirulence in an epidemic BI/NAP1/027 clinical isolate of Clostridium difficile. PLoS Pathog 7 (2011) e1002317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [35].Schloss PD, Westcott SL, Ryabin T, Hall JR, Hartmann M, Hollister EB, et al. Introducing mothur: open-source, platform-independent, community-supported software for describing and comparing microbial communities. Appl Environ Microbiol 75 (2009) 7537–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [36].Pruesse E, Quast C, Knittel K, Fuchs BM, Ludwig W, Peplies J, et al. SILVA: a comprehensive online resource for quality checked and aligned ribosomal RNA sequence data compatible with ARB. Nucleic Acids Res 35 (2007) 7188–96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [37].Edgar RC, Haas BJ, Clemente JC, Quince C, Knight R UCHIME improves sensitivity and speed of chimera detection. Bioinformatics 27 (2011) 2194–200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [38].Wang Q, Garrity GM, Tiedje JM, Cole JR Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl Environ Microbiol 73 (2007) 5261–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [39].Yue JC, Clayton MK A similarity measure based on species proportions. Communications in Statistics - Theory and Methods 34 (2005) 2123–31. [Google Scholar]
- [40].Martin AP Phylogenetic approaches for describing and comparing the diversity of microbial communities. Appl Environ Microbiol 68 (2002) 3673–82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [41].Cutler DR, Edwards TC Jr., Beard KH, Cutler A, Hess KT, Gibson J, et al. Random forests for classification in ecology. Ecology 88 (2007) 2783–92. [DOI] [PubMed] [Google Scholar]