Abstract
Case series
Patient: Male, 65 • Female, 47
Final Diagnosis: Epithelioid malignant peripheral nerve sheath tumor
Symptoms: Bleeding per rectum
Medication: —
Clinical Procedure: —
Specialty: Pathology
Objective:
Rare disease
Background:
Malignant peripheral nerve sheath tumor (MPNST) with pure epithelioid histology MPNST (epithelioid-MPNST) is a distinctly rare variant of MPNST described by World Health Organization. Occurrence of MPNST in the tubular gut is extremely rare with nearly 20 cases reported in the last 40 years.
Case Report:
In this case report, we described 2 such cases of epithelioid-MPNST occurring in a 65-year male and 47-year-old female, who both presented with bleeding rectal polyps. We also report on our comprehensive systematic literature review.
Conclusions:
To the best of our knowledge, these are possibly the first two cases of pure epithelioid-MPNST at an unusual site. This needs to be differentiated from lesions more commonly encountered at the related anatomical site, such as malignant melanoma, poorly differentiated carcinoma, lymphoma, or even a gastrointestinal stromal tumor, by appropriate panel of antibodies.
MeSH Keywords: Biology, Immunohistochemistry, Recurrence, S100 Proteins
Background
Malignant tumors arising from peripheral nerves or displaying differentiation along the lines of the various elements of the nerve sheath, such as Schwann cell, perineural cell, and fibroblast, are collectively referred to as malignant peripheral nerve sheath tumors (MPNSTs). MPNSTs account for approximately 5–10% of all soft tissue sarcomas; about a fourth to a half occur in the setting of neurofibromatosis 1 (NF1) [1]. Epithelioid-MPNST; defined as ≥50% of polygonal tumor cells in sheets or nodules) is a rare variant of MPNSTs accounting for nearly 5% of all cases; and has a distinct immunophenotypic characteristic. In contrast to conventional MPNST, occurrence from or within a pre-existing benign peripheral nerve sheath tumor is uncommon; and the association with NF1 is exceptional [2].
Although commonly encountered in the soft tissues of the extremities and deep retroperitoneal sites, MPNSTs arising primarily in the tubular gut (gastrointestinal-MPNSTs) are extremely rare; and only nearly 20 such cases have been reported worldwide over the last 4 decades [3–15]. In this case report, we aimed to describe 2 cases of epithelioid-MPNST, both presenting as bleeding rectal polyp posing great diagnostic challenges; we also present a brief comprehensive review of world literature. To the best of our knowledge, these are possibly the first reports of pure epithelioid-MPNSTs occurring at an unusual anatomical site.
Case Report
Case 1
Case 1 was a 65-year-old male who presented with complaints of a mass protruding from his rectum on straining, for last 1 year, which was associated with loss of appetite and weight since the last 6 months; and bleeding per rectum for 1 month. Digital PR (prostate rectum) examination revealed a polypoidal ulcero-proliferative growth extending 4 cm from the anal verge protruding into lumen with stalk at 9 o’clock position. Colonoscopy showed an infiltrative anorectal growth involving half of the circumference with luminal narrowing. Magnetic resonance imaging (MRI) of the pelvis revealed a low mid rectal growth of size 5.5×3.5×3.5 cm extending cranially (5 cm from the anal verge) with luminal narrowing, minimal perirectal fat stranding, enlarged sub-centimeter perirectal lymph nodes; as well as involvement of the left side of the internal sphincter and meso-rectal fascia, anteriorly. Trans anal excision of the rectal mass was performed, and the specimen sent for histopathological examination.
Case 2
Case 2 was a 47-year-old female who presented with bleeding per rectum along with a mass protruding through her rectum, for 3 months. She gave a history of similar complaints 3 years back for which she underwent surgery with a provisional clinical diagnosis of hemorrhoids. Local examination as well as colonoscopy showed a polypoidal growth in her rectum with narrow pedunculated stalk, 1 cm above the anal verge. Contrast enhanced computed tomography scan (CECT) of the abdomen and pelvis showed a polypoidal lesion with mucosal irregularity and thickening (8 mm) involving the anterior and left wall of the anal canal and distal rectum extending for a length of 4.5 cm with an enlarged, necrotic left inguinal node of size 29×20 mm and multiple sub-centimeter perirectal nodes. Besides these, multiple hypodense peripherally enhancing lesions were also seen in both lobes of liver, the largest measuring 19×18 mm in segment VII, suggestive of metastasis. Polypectomy was done and the specimen sent for histopathological examination.
Pathology
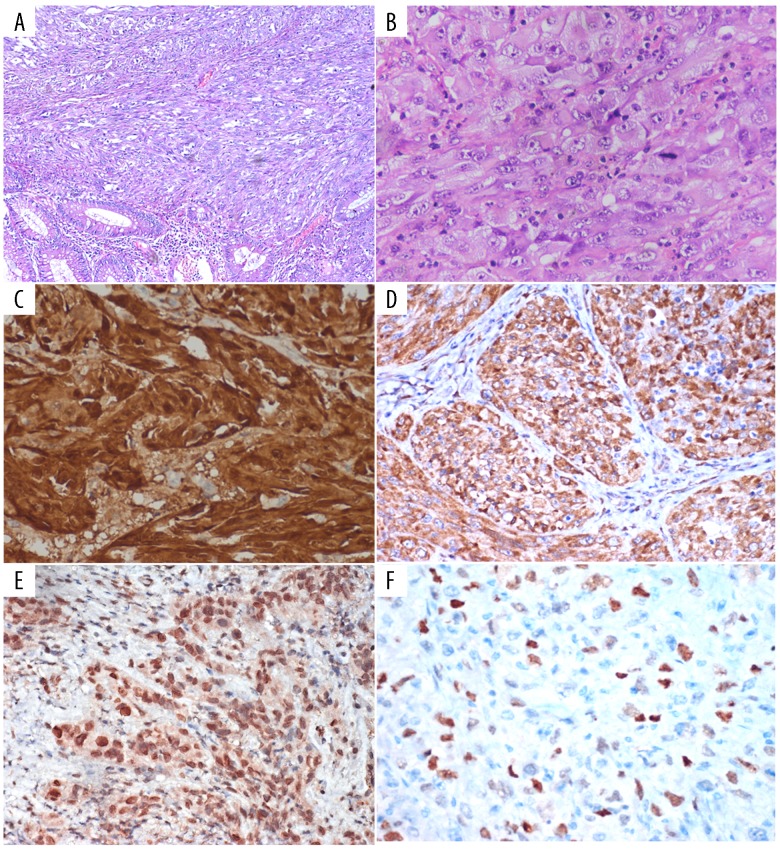
The gross and microscopic pathologies in the 2 cases were similar. Grossly, the lesions were gray brown to gray white solid masses, each measuring 5×4.2×2.4 cm, with exudate on the outer surface of Case 2. Cut surfaces were gray white and solid, firm in consistency with focal gray brown areas (Figure 1A, 1B). On histopathological evaluation, the lesions were comprised predominantly of nodules and sheets of large polygonal tumor cells (comprising >50% of lesioned area) arising underneath the ulcerated rectal mucosa with interspersed fibro collagenous septa. These cells showed moderate pleomorphism, abundant glassy to eosinophilic cytoplasm with prominent eosinophilic macro-nucleoli, increased mitoses (8/10 high power field), along with areas of hemorrhage and necrosis. The histo-morphological findings were reminiscent of poorly differentiated carcinoma, malignant melanoma, epithelioid gastrointestinal stromal tumor, and a non-Hodgkin lymphoma (Figure 2A, 2B).
Figure 1.
Pre-operative photomicrograph demonstrating a polypoidal mass protruding through the anal canal (A) which on cut section had solid, glistening white appearance with areas of hemorrhage (B).
Figure 2.
Hematoxylin and eosin stained tissue sections from the polypoidal mass depicting a cellular tumor underneath ulcerated rectal mucosa. The tumor comprised of sheets and nests of epithelioid type of tumor cells occupying more than 50% of the entire tumor area (A, 100×). These tumor cells were polygonal in shape with abundant glassy cytoplasm (arrow) with pleomorphic nuclei showing macro-nucleoli and brisk mitotic activity; thus, mimicking malignant melanoma, poorly differentiated carcinoma, high risk epithelioid gastrointestinal stromal tumor (B, 400×). On immunohistochemistry evaluation, these tumor cells were diffusely and strongly positive for vimentin and S100 (C); focally positive for neuron specific enolase (D), MDM2 (E), p53 (F), and nuclear integrase interactor 1 (INI-1) (not shown); but negative for other markers such as leukocyte common antigen (LCA), pancytokeratin (PanCK), HMB45, melan A, CD 117, DOG1, glial fibrillary acidic protein, desmin, and CD 34 (peroxidase-anti-peroxidase, all magnifications 400×). The morphological and immunohistochemical characteristics were consistent with the diagnosis of epithelioid malignant peripheral nerve sheath tumor (epithelioid-MPNST).
On immunohistochemistry analysis, tumor cells were strongly and diffusely positive for vimentin and S100; focally positive for neuron specific enolase (NSE), MDM2, nuclear integrase interactor 1 (INI-1), and p53 (in Case 2); but negative for leukocyte common antigen (LCA), desmin, CD 117, DOG1, HMB-45, glial fibrillary acidic protein (GFAP), CD 34, melan A, pancytokeratin (PanCK), smooth muscle actin (SMA), and epithelial membrane antigen (EMA) (Figures 2C–2F). Thus, the histomorphology in correlation with the immunohistochemistry characteristics were consistent with the diagnosis of epithelioid-MPNST in rectum.
Follow-up
The first case did not receive any adjuvant therapy post-surgery; and is presently in close follow-up without any evidence of local recurrence or distal metastasis at 8 months post re-section. The second case received adjuvant localized pelvic radiotherapy. Although there was reduction in size of the inguinal lymph node and the rectal lesion, there was no change in the hepatic lesion; and the patient succumbed to disseminated disease with rapidly accumulating ascites at 6 months post-surgery. The informed consent was obtained from next of kin in both the cases.
Discussion
A PubMed-based comprehensive review of world literature on gastrointestinal-MPNSTs reported during the last 40 years (1980–2019 till report date) is presented in the Table 1 [3–23]. This review excluded all those cases that occurred in the hepatobiliary tree and pancreas. No cases other than 1 reported by Marwah et al. from India were associated with stigmata of NF1; and only 1 case was found to be associated with Schistosoma japonicum infestation [10,23]. The majority of the cases (11 versus 4) occurred in middle-aged to elderly patients (≥40 years of age) with no gender predilection; and the youngest reported case occurred in a 2-day old neonate who presented with features of intestinal obstruction [12]. The signs and symptoms of all cases were site specific.
Table 1.
Malignant peripheral nerve sheath tumor (MPNST) in tubular gastrointestinal tract: a comprehensive review of the literature (1980–2019 till report date) (n=15)#,*.
| Sl. no | Author, [ref] year, place | Age/gender | Site | Presentation | Size | Histopathology | IHC | Management | Outcome | Follow-up duration |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Rawal [4], 2019, India | 25/M | Transverse colon | Abd. mass, peritoneal nodule | 20 cm | Spindle | S-100+ | Excision | NED | 6 months |
| 2 | Zhu [5], 2017, China | 53/F | Distal ileum | Intussusception | 4 cm | Spindle Mitoses; 10/10 HPF | S-100+, CD34+, MIB-1;20% | Right hemicolectomy, anastomosis | Death due to disease | 10 months |
| 3 | Kim [6], 2015, Korea | 48/M | Stomach | Melena | 9 cm | Spindle | S-100+ | Subtotal gastrectomy + LN dissection+ Billroth-II reconstruction | NED | Not available |
| 4 | Pandey [7], 2015, India | 45/F | Ileum | Intussusception | ND | Spindle Mitosis; 8/10 HPF | S-100+, CD34− | Segmental resection and anastomosis | NED | 10 months |
| 5 | Mohtaram [8], 2013, Morocco | 18/M | Proximal jejunum | Abdominal pain, vomiting, diarrhea, | 20 cm | Spindle Mitosis; 9/10 HPF | S-100+, Ki 67; 60% | Resection | Death due to disseminated disease | ND |
| 6 | Marwah [9], 2013, India | 30/F | Splenic flexure of colon | Pain abdomen and mass | 10 cm | Spindle, brisk mitosis, MIB1; 20% | Vimentin+, S-100 + | Segmental resection and anastomosis | Recurrence at anastomotic site with LN metastasis | 18 months |
| 7 | Takemura [10], 2012, Japan | 70/M | Stomach | Epigastric pain and melena | 6 cm | Spindle MIB1; 48.5% Mitosis; 10/50 HPF | S-100+ | Distal gastrectomy | Death due to hepatic metastasis | 5 months |
| 8 | Telem, Pertsemlidis [11], 2008, USA | 71/F | Proximal jejunum | Intussusception, polypoid lesion | 4 cm | Spindle cell Mitosis: 20/50 HPF | CD34+, S-100+ | Resection and anastomosis | NED | Not available |
| 9 | Lee [12], 2006, Korea | 2 days/FCh | Ascending colon | Intestinal obstruction | 5 cm | Spindle cell | Vimentin+, S100+. Genetic study: WNL | Right hemicolectomy | NED | 17 months The youngest case of GI MPNST |
| 10 | Park [13], 2002, Korea | 43/M | Descending colon | Vomiting, diarrhea, liver metastasis | 15 cm | Spindle and epithelioid (EM proven) | S-100+, CD57+, NSE− diffuse+, Pan CK focal+ | Left hemicolectomy | Liver and LN metastasis | ND |
| 11 | Manger [15], 2000, Germany | 60/F | Mid-esophagus | Dysphagia, loss of appetite Pedunculated cauliflower like mass |
10 cm | Spindle cell Mitosis: 2/HPF Tumor giant cells present, EM proven |
Vimentin+, S-100+, NSE+ MIB1; 30% | Resection and anastomosis | NED | 48 months |
| 12 | Rosati [16], 2000, Italy | 73/F | Anorectum | Bleeding PR and change in bowel hobbit | ND | Spindle, focally round cells | Vimentin+, S-100+ | APR + colostomy | Recurrence | 8 months |
| 13 | Nozu [19], 1995, Japan | 65/M | Ileum | Melena, intestinal obstruction | ND | Spindle cell EM-Schwann cell differentiation | GFAP+, S-100+, NSE+, vimentin+ | Resection and anastomosis | Death with peritoneal dissemination | 6 months |
| 14 | Present case (Case 1), India | 65/M | Rectum | Polypoidal mass | 5.5 cm | Epithelioid | Vimentin and, S100; diffuse +, NSE, MDM2, p53; focal+ | Trans anal excision of rectal mass | NED | 8 months |
| 15 | Present case (Case 2), India | 47/F | Rectum | Polypoidal mass Multiple liver metastasis, left inguinal and perirectal LN enlargement | 5 cm | Epithelioid | Vimentin and S-100; diffuse +, NSE and MDM 2; focal + p53; negative | Polypectomy + neoadjuvant radiotherapy | Left inguinal, perirectal lymph nodes-markedly reduced in size, liver metastases, ascites, death | 6 months |
Remainder 5 cases of MPNST are not presented due to inaccessible data;
All cases of hepatobiliary and pancreatic tumors were excluded. MPNST – malignant peripheral nerve sheath tumor; IHC – immunohistochemistry; M – male; F – female; LN – lymph node; ‘±’ – associated with NF1 (neurofibromatosis type 1), positive; ND – not described; ‘+’ – positive (focal); ‘−‘ – negative; WNL – within normal limits; NSE – neuron specific enolase; CK – cytokeratin; EM – electron microscopy; HPF – high power field; GFAP – glial fibrillary acidic protein; APR – abdominoperineal resection; NED – no evidence of disease suggesting no recurrence or metastasis.
The majority of cases occurred in the small intestine (n=9); 5 cases were in the colon; 3 cases were in the stomach; 2 cases were esophageal; and only 1 case (excluding the present 2 cases) involved anorectum. The size varied from small polypoidal masses protruding into the lumen to large lesions occluding the luminal gut producing obstruction/intussusception (range 4 cm to 20 cm). On histopathological evaluation, the lesions were characterized as spindle shaped cells with wavy nuclei arranged in fascicles associated with brisk mitotic activity, moderate to marked pleomorphism along with areas of hemorrhage and/or necrosis; thus, suggestive of a spindle cell sarcoma. The esophageal lesion as reported by Perch et al. demonstrated rhabdomyoblastic differentiation (so called malignant Triton tumor) [21]. On immunohistochemistry evaluation, all lesions demonstrated uniform diffuse positivity for vimentin; focal and weak cytoplasmic positivity for S100, and variable focal positivity for neuron specific enolase, CD 57, and PanCK. Electron microscopy studies, performed in 3 cases, confirmed the true nature of the lesion [13,15, 20].
Pure epithelioid-MPNST has never been reported before in the rectum. The present 2 cases posed immense diagnostic challenge due to the polygonal epithelioid appearance of the tumoral cells that resembled malignant melanoma, poorly differentiated carcinoma, epithelioid gastrointestinal stromal tumor, and lymphomas. Strong and diffuse S100 positivity in these cells was in stark contrast to conventional MPNST, which typically shows weak focal S100 positivity. Furthermore, PanCK, HMB-45, CD 117, DOG-1, and LCA negativity ruled out the possibility of common rectal malignancies. In addition, both of our cases showed retained nuclear integrase interactor 1 (INI-1) expression, and MDM2 positivity; where as p53 positivity was noted in 1 of the case. The immunohistochemistry pattern of our 2 cases were similar to that of a few large case series of epithelioid-MPNST published worldwide (presented in Table 2).
Table 2.
Immunophenotypic characteristics of epithelioid-MPNST: Comparison of present 2 cases with the largest series published in the literature.
| Present cases | Jo and Fletcher [1] | Luzar et al. [2] | Laskin et al. [23] | Lodding et al. [24] | |
|---|---|---|---|---|---|
| Site | Rectum | Extremities | Skin | Extremities | Extremities |
| Number of cases | 2 | 63# | 11 | 25 | 14 |
| Antibody positivity | |||||
| S-100 | 2/2 (100%) | 63/63 (100%) | 11/11(100%) | 20/25 (80%) | 7/14 50%) |
| NSE | 2/2 (focal) | – | 3/11 (focal) | Strong | 3/14 (focal) |
| INI-1 | 2/2 (100%) | 35/52 (67%) | 3/6 (50%) | – | – |
| GFAP | 0/2 | 1/11 | |||
| HMB45 | 0/2 | 24/40 (60%) | 1/11 | – | – |
| MDM2 | 2/2 | ||||
| TP53 | 1/2 | ||||
| CD117 | 0/2 | – | – | – | – |
| DOG1 | 0/2 | – | – | – | – |
| Melan A | 0/2 | 0/58 | 1/11 | – | – |
| Pancytokeratin | 0/2 | 0/33 | 0/11 | 0/15 | – |
| CD34 | 0/2 | ||||
| SMA | 0/2 | 0/3 | 0/6 | – | – |
| EMA | 0/2 | 0/11 | |||
| Desmin | 0/2 | ||||
| LCA | 0/2 |
The biological behavior of MPNST has been reported to be unpredictable in several previous studies. Anatomic sites, larger tumor size (≥10 cm), adequacy of resection margins, American Joint Committee on Cancer (AJCC) stage III, lack of S100 staining, elevated Ki67 expression (>25%), and nuclear TP53 and MDM2 expression were predictors of locoregional recurrence and distant metastasis; thus, contributing to inferior overall survival [3]. In contrast, literature on epithelioid-MPNST of the skin and superficial soft tissues of the extremities has been found to have conflicting results: the largest series to date by Jo and Fletcher reported a favorable outcome [1]; whereas other studies have reported an aggressive biological behavior [2,23,24].
Seven of 13 cases of gastrointestinal-MPNST published to date had evidence of locoregional recurrence with or without lymph nodal and/or distant metastasis following surgery (4 of 7 patients died with their disease) (follow-up duration ranged from 6 months to 48 months) (Table 1). The present 2 cases had close follow-up, with 1 case having liver metastasis and rapidly accumulating ascites, and the patient finally succumbing to the disease (Case 2).
Conclusions
Epithelioid-MPNST is rare at unusual anatomic sites. epithelioid-MPNST presenting as bleeding rectal polyp needs to be differentiated from common mimics, such as bleeding piles, in order to plan appropriate surgical intervention.
Footnotes
Conflict of interest
None.
References:
- 1.Jo VY, Fletcher CD. Epithelioid malignant peripheral nerve sheath tumor: Clinicopathologic analysis of 63 cases. Am J Surg Pathol. 2015;39:673–82. doi: 10.1097/PAS.0000000000000379. [DOI] [PubMed] [Google Scholar]
- 2.Luzar B, Shanesmith R, Ramakrishnan R, et al. Cutaneous epithelioid malignant peripheral nerve sheath tumor: A clinicopathological analysis of 11 cases. Histopathology. 2016;68:286–96. doi: 10.1111/his.12756. [DOI] [PubMed] [Google Scholar]
- 3.McCarthy AJ, Karamchandani DM, Chetty R. Neural and neurogenic tumours of the gastroenteropancreaticobiliary tract. J Clin Pathol. 2018;71:565–78. doi: 10.1136/jclinpath-2017-204895. [DOI] [PubMed] [Google Scholar]
- 4.Rawal G, Zaheer S, Ahluwalia C, Dhawan I. Malignant peripheral nerve sheath tumor of the transverse colon with peritoneal metastasis: A case report. J Med Case Rep. 2019;13:15. doi: 10.1186/s13256-018-1896-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Zhu LB, Li PF, Xiao WH, et al. A distal ileum malignant peripheral nerve sheath tumor causing intussusception in a patient in China: A case report. World J Surg Oncol. 2017;15:29. doi: 10.1186/s12957-017-1101-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kim EY, Lee SH, Yoo HM, et al. Gastric malignant peripheral nerve sheath tumor: A case report. Int J Surg Pathol. 2015;23:505–8. doi: 10.1177/1066896915573570. [DOI] [PubMed] [Google Scholar]
- 7.Pandey D, Verma A, Akhtar A, et al. Malignant peripheral nerve sheath tumour of small intestine presenting as ileo-ileal intussusception – a rare tumour with unusual complication. J Clin Diagn Res. 2015;9(5):XD03–4. doi: 10.7860/JCDR/2015/13318.5933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Mohtaram A, Mesmoudi S, M’rabti H, et al. Malignant peripheral nerve sheath tumor of the small bowel: An unusual presentation with fatal outcome. Case Rep Oncol Med. 2013;2013:423867. doi: 10.1155/2013/423867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Marwah S, Gurawalia JP, Sheoran KD, et al. Malignant peripheral nerve sheath tumor of the colon in a patient with von Recklinghausen’s disease: Report of a case. Clin J Gastroenterol. 2013;6:429–33. doi: 10.1007/s12328-013-0422-x. [DOI] [PubMed] [Google Scholar]
- 10.Takemura M, Yoshida K, Takii M, et al. Gastric malignant schwannoma presenting with upper gastrointestinal bleeding: A case report. J Med Case Rep. 2012;6:37. doi: 10.1186/1752-1947-6-37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Telem DA, Pertsemlidis D. Malignant peripheral nerve sheath tumor: An unusual cause of intussusception. J Gastrointest Surg. 2008;12:1609–11. doi: 10.1007/s11605-007-0395-5. [DOI] [PubMed] [Google Scholar]
- 12.Lee YJ, Moon H, Park ST, et al. Malignant peripheral nerve sheath tumor arising from the colon in a newborn: Report of a case and review of the literatures. J Pediatr Surg. 2006;41:e19–22. doi: 10.1016/j.jpedsurg.2005.11.039. [DOI] [PubMed] [Google Scholar]
- 13.Park YS, Lim SJ, Kim WH, Ham EK. Malignant peripheral nerve sheath tumor in descending colon. Korean J Pathol. 2002;36:179–83. [Google Scholar]
- 14.Catania G1, Puleo C, Cardì F, et al. Malignant schwannoma of the rectum: a clinical and pathological contribution. Chir Ital. 2001;53:873–77. [PubMed] [Google Scholar]
- 15.Manger T, Pross M, Haeckel C, Lippert H. Malignant peripheral nerve sheath tumor of the esophagus. Dig Surg. 2000;17:627–31. doi: 10.1159/000051973. [DOI] [PubMed] [Google Scholar]
- 16.Rosati G, Chlacchlo R, Rossi A, et al. Rectal neurogenic sarcoma: Case report and review of the literature. Tumori. 2000;86:85–87. doi: 10.1177/030089160008600117. [DOI] [PubMed] [Google Scholar]
- 17.Mosca F, Stracqualursi A, Lipari G, et al. Malignant schwannoma of the small intestine: A report of 2 cases. G Chir. 2000;21:149–55. [PubMed] [Google Scholar]
- 18.Bees NR, Ng CS, Dicks-Mireaux C, Kiely EM. Gastric malignant schwannoma in a child. British J Radiol. 1997;70:952–55. doi: 10.1259/bjr.70.837.9486074. [DOI] [PubMed] [Google Scholar]
- 19.Nozu T, Takahashi A, Asakawa H, et al. Malignant intestinal schwannoma: A case report and a review of the literature in Japan. Intern Med. 1995;34:1101–5. doi: 10.2169/internalmedicine.34.1101. [DOI] [PubMed] [Google Scholar]
- 20.Perch SJ, Soffen EM, Whittington R, Brooks JJ. Esophageal sarcomas. J Surg Oncol. 1991;48:194–98. doi: 10.1002/jso.2930480311. [DOI] [PubMed] [Google Scholar]
- 21.Hansen D, Pedersen A, Pedersen KM. Malignant intestinal schwannoma: Case report. Acta Chir Scand. 1990;156:729–32. [PubMed] [Google Scholar]
- 22.Schwartz DA. Malignant schwannoma occurring with Schistosoma japonicum: A case report. Southeast Asian J Trop Med Public Health. 1982;13:601–5. [PubMed] [Google Scholar]
- 23.Laskin WB, Weiss SW, Bratthauer GL. Epithelioid variant of malignant peripheral nerve sheath tumor (malignant epithelioid schwannoma) Am J Surg Pathol. 1991;15:1136–45. doi: 10.1097/00000478-199112000-00004. [DOI] [PubMed] [Google Scholar]
- 24.Lodding P, Kindblom LG, Angervall L. Epitheliod malignant schwannoma: A study of 14 cases. Virchows Arch A Pathol Anat Histopathol. 1986;409:433–51. doi: 10.1007/BF00705415. [DOI] [PubMed] [Google Scholar]