Abstract
The aim of this study was to investigate the mechanisms responsible for resistance to antimicrobials in a collection of enterobacterial isolates recovered from two hospitals in Saudi Arabia. A total of six strains isolated from different patients showing high resistance to carbapenems was recovered in 2015 from two different hospitals, with four being Klebsiella pneumoniae and two Enterobacter cloacae. All isolates except one K. pneumoniae were resistant to tigecycline, but only one K. pneumoniae was resistant to colistin. All produced a carbapenemase according to the Carba NP test, and all were positive for the EDTA-disk synergy test for detection of MBL. Using PCR followed by sequencing, the four K. pneumoniae isolates produced the carbapenemase NDM-1, while the two E. cloacae isolates produced the carbapenemase VIM-1. Genotyping analysis by Multilocus Sequence Typing (MLST) showed that three out of the four K. pneumoniae isolates were clonally related. They had been recovered from the same hospital and belonged to Sequence Type (ST) ST152. In contrast, the fourth K. pneumoniae isolate belonged to ST572. Noticeably, the NDM-1-producing K. pneumoniae additionally produced an extended-spectrum ß-lactamase (ESBL) of the CTX-M type, together with OXA-1 and TEM-1. Surprisingly, the three clonally related isolates produced different CTX-M variants, namely, CTX-M-3, CTX-M-57, and CTX-M-82, and coproduced QnrB, which confers quinolone resistance, and the 16S rRNA methylase RmtC, which confers high resistance to all aminoglycosides. The AAC(6′)-Ib acetyltransferase was detected in both K. pneumoniae and E. cloacae. Mating-out assays using Escherichia coli as recipient were successful for all isolates. The blaNDM-1 gene was always identified on a 70-kb plasmid, whereas the blaVIM-1 gene was located on either a 60-kb or a 150-kb plasmid the two E. cloacae isolates, respectively. To the best of our knowledge, this is the first report of the coexistence of an MBL (NDM-1), an ESBL (CTX-M), a 16S rRNA methylase (RmtC), an acetyltransferase (AAC[6′]-Ib), and a quinolone resistance enzyme (QnrB) in K. pneumoniae isolates recovered from different patients during an outbreak in a Saudi Arabian hospital.
1. Introduction
Metallo-β-lactamases (MBLs) are enzymes that hydrolyze most β-lactams including carbapenems, the most potent β-lactams. Therefore, resistance mediated by MBLs is considered the most serious mechanism for inactivating β-lactams [1]. Of the clinically relevant MBLs, NDM, VIM, and IMP are the most common worldwide [2, 3]. Klebsiella pneumoniae and Enterobacter cloacae are Enterobacteriaceae that are leading causes of nosocomial infections and can frequently acquire antibiotic resistance [4]. K. pneumoniae and E. cloacae can acquire resistance to third-generation cephalosporins through plasmid-mediated AmpC β-lactamases, such as CMY, FOX, ACC, LAT, MIR, MOX, DHA, and ACT [5], as well as extended-spectrum β-lactamases (ESBL), such as CTX-M, TEM-, and SHV-derivatives. Acquisition of carbapenem resistance in those bacterial species may be related to several carbapenem-hydrolyzing ß-lactamases, such as KPC, NDM, VIM, and OXA-48 [6]. Several mechanisms may confer aminoglycoside resistance to Enterobacteriaceae, including production of aminoglycoside-modifying enzymes, such as aminoglycoside acetyltransferases (AACs), phosphotransferases (APHs), and adenylyltransferases (ANTs), but also target modifications by mutations in ribosomal proteins, or 16S rRNA methylation through the action of 16S rRNA methylases [7]. AACs are the most common aminoglycoside-modifying enzymes and are subdivided into four groups [AAC(1), AAC(2′), AAC(3), and AAC(6′)]. AAC(6′) comprises the most common enzymes in Gram-negative bacteria and can be subdivided into two groups: AAC(6′)-I and AAC(6′)-II [8, 9]. Enterobacteriaceae may also produce 16S rRNA methylases, including ArmA, RmtA to RmtH, and NpmA, which confer resistance to aminoglycosides [10]. The plasmid-mediated quinolone resistance genes qnrA, qnrB, qnrC, qnrD, qnrS, and qnrVC code for Qnr proteins that protect DNA gyrase and topoisomerase IV from the action of quinolones, conferring quinolone resistance [11]. In this study, we aimed to investigate the mechanisms responsible for carbapenem resistance in a collection of enterobacterial isolates recovered from two hospitals in Saudi Arabia. Carbapenemase-producing isolates were also further investigated for additional antibiotic resistance traits.
2. Materials and Methods
2.1. Bacterial Isolates
Four K. pneumoniae and two E. cloacae isolates from two hospitals in Riyadh, the capital of Saudi Arabia and Al-Gouf, the north region of Saudi Arabia were highly resistant to carbapenems. Three K. pneumoniae isolates (KP-Q1, KP-Q2, and KP-Q3) were obtained from hospital A while a single K. pneumoniae (KP-Q4) and two E. cloacae (EN.C.Q5 and EN.C.Q6) isolates were collected from hospital B in 2015 (Table 1). Isolates were identified in the clinical laboratory using the VITEK 2 system (bioMérieux, Marcy l'Etoile, France) and confirmed by conventional morphological and biochemical tests.
Table 1.
Isolate case histories and characteristics.
| Isolate | Hospital/City | Patient age (years) | Patient sex | Specimen source | Diagnosis |
|---|---|---|---|---|---|
| KP-Q1 | A/ Al-Gouf | 65 | Male | Blood | Septic encephalopathy |
| KP-Q2 | A/ Al-Gouf | 95 | Male | Sputum | Pulmonary fibrosis |
| KP-Q3 | A/ Al-Gouf | 70 | Female | Wound | Gangrene of diabetic foot with sepsis |
| KP-Q4 | B/ Riyadh | 50 | Male | Wound | Urogenital infection |
| EN.C.Q5 | B/ Riyadh | 64 | Female | Blood | ND |
| EN.C.Q6 | B/ Riyadh | 78 | Male | Blood | Cystic fibrosis |
KP: Klebsiella pneumoniae
EN.C: Enterobacter cloacae
ND: Not determined
2.2. Antimicrobial Susceptibility Testing and Phenotypic Carbapenemase Detection
Etest strips (bioMérieux,Marcy l'Etoile, France) and disk diffusion methods (Oxoid, UK) were used to determine susceptibility to antimicrobials other than colistin, whose susceptibility was tested using a microbroth dilution method. The guidelines of the Clinical Laboratory Standards Institute (CLSI, 2016) [12] were used to interpret MICs except for tigecycline and colistin, whose data were interpreted according to the 2019 guidelines of the European Committee on Antimicrobial Susceptibility Testing (EUCAST) [13]. The Carba NP test [14] was carried out to detect carbapenemase activity. Production of MBL was screened by an EDTA-disk synergy test [15].
2.3. PCR Amplification of Resistance Genes
Carbapenem-resistant enterobacterial isolates were collected from 200 μL volumes of overnight Luria Bertani broth (Thermo Fischer Scientific, Waltham, MA, USA) cultures and resuspended in equal volumes of sterile distilled water, heated at 99°C for 10 min on a heating block, and then centrifuged at 15,000 rpm for two minutes. The supernatant, which contained released DNA, was used directly in PCR amplification of the resistance genes on a Techne Flexigene Thermocycler (Techne, Duxford, Cambridge, UK). Positive and negative controls were included in all PCR runs. All PCR amplicons were electrophoresed on 0.8% agarose gels containing 0.5 mg/L ethidium bromide and then analyzed under UV light (Pharmacia LKB; Biotechnology AB, Gothenburg, Sweden) and photographed using a documentation system.
The multiplex PCR protocol described by Dallenne et al. [16] was used to amplify four types of class A β-lactamase genes (TEM, SHV, five CTX-M families, and an OXA-1-like). Multiplex PCR was used to seek plasmid-mediated AmpC β-lactamase genes, including ACC, FOX, MOX, CMY, LAT, and MIR, as previously described [17] using six pairs of primers. Ten carbapenemase genes, including IMP, VIM, NDM, SIM, SPM, GIM, AIM, DIM, OXA-48, and KPC, were tested via three multiplex reactions according to the method of Poirel et al. [18]. Moreover, the isolates were screened by multiplex PCR for qnrA, qnrB, and qnrS plasmid-mediated quinolone resistance genes [19], whereas a simplex PCR was performed to detect AAC(6′)-Ib [7]. Additionally, six 16S rRNA methylase genes were tested using two multiplex reactions, with the first detecting armA, rmtB, and npmA and the second rmtA, rmtC, and rmtD according to the previously published method [20]. The PCR products were sequenced and analyzed using the NCBI database with the BLAST program (http://www.ncbi.nlm.nih.gov).
2.4. Multilocus Sequence Typing (MLST) Analysis
Clonal relatedness was determined by MLST according to the Pasteur Institute scheme for the K. pneumoniae isolates (https://bigsdb.pasteur.fr/klebsiella/klebsiella.html) and according to the PubMLST protocol and database for the E. cloacae isolates (https://pubmlst.org/ecloacae/).
2.5. Plasmid Extraction and Conjugation Experiment
A plasmid extraction protocol [21] was used to investigate the presence of plasmids in all isolates and was followed by direct agarose gel electrophoresis of extracted DNA. Bac-tracker (Epicentre, Madison, WI, USA) was used as a plasmid size marker.
Transfer of imipenem resistance genes by conjugation from our isolates as donors was attempted by filter mating [22] with Escherichia coli J53 sodium azide resistant as the recipient strain. The initial donor/recipient ratio was 1:5 or 1:10. Transconjugants were selected on MacConkey agar containing sodium azide (100 mg/L) and ceftazidime (4 mg/L).
3. Results
MICs of different antibiotics for the six carbapenem-resistant K. pneumoniae and E. cloacae isolates are shown in Table 2. All isolates were highly resistant to all tested β-lactams. They all remained susceptible to tigecycline (MIC ≤ 2 mg/L) according to the EUCAST 2016 breakpoint, although the cut-off tigecycline in EUCAST 2019 guidelines is 0.5 mg/L. Consequently, all our isolates, except one K. pneumonia, were resistant to tigecycline using the new EUCAST definition. A single K. pneumoniae isolate (KP-Q2) was resistant to colistin according to the EUCAST definition (MIC > 2 mg/L is considered resistant). Moreover, the four K. pneumoniae isolates were determined to be susceptible to chloramphenicol with inhibition zone diameters > 12 mm according to the 2016 CLSI guidelines, whereas the two E. cloacae isolates were resistant to this compound. Resistance to sulfamethoxazole/trimethoprim was also observed in all isolates. Four isolates (three K. pneumoniae and a single E. cloacae) were resistant to tetracycline. Resistance profiles of aminoglycosides (amikacin and gentamicin) and quinolones/fluoroquinolones (nalidixic acid, ciprofloxacin, and ofloxacin) are presented in Table 2.
Table 2.
Antimicrobial resistance profile of six resistant Klebsiella pneumoniae and Enterobacter cloacae isolates.
| Antibiotics | MIC (mg/L) | |||||
|---|---|---|---|---|---|---|
| KP-Q1 | KP-Q2 | KP-Q3 | KP-Q4 | EN.C.Q5 | EN.C.Q6 | |
| Amoxicillin | >256 | >256 | >256 | >256 | >256 | >256 |
| Piperacillin | >256 | >256 | >256 | >256 | >256 | >256 |
| Ticarcillin | >256 | >256 | >256 | >256 | >256 | >256 |
| Cefoperazone | >256 | >256 | >256 | >256 | >256 | >256 |
| Cefotaxime | >256 | >256 | >256 | >256 | >256 | >256 |
| Ceftazidime | >256 | >256 | >256 | >256 | >256 | >256 |
| Cefepime | >256 | >256 | >256 | >256 | >256 | >256 |
| Aztreonam | >256 | >256 | >256 | >256 | >256 | >256 |
| Cefoxitin | >256 | >256 | >256 | >256 | >256 | >256 |
| Cefotetan | >256 | >256 | >256 | >256 | >256 | >256 |
| Imipenem | >256 | >256 | >256 | >256 | >256 | >256 |
| Meropenem | >32 | >32 | >32 | >32 | >32 | >32 |
| Doripenem | >32 | >32 | >32 | >32 | >32 | >32 |
| Amikacin | >256 | >256 | >256 | >256 | 24 (S) | >256 |
| Gentamicin | >256 | >256 | >256 | >256 | 64 | >256 |
| Nalidixic acid | >256 | >256 | >256 | >256 | >256 | >256 |
| Ciprofloxacin | >32 | >32 | >32 | >32 | 2 (S) | 8 |
| Ofloxacin | >32 | >32 | >32 | >32 | 2 (S) | 8 |
| Tigecycline | 0.75 | 0.75 | 0.75 | 0.19 (S) | 2 | 2 |
| Colistin | 0.75 (S) | 3 | 1.5 (S) | 2 (S) | 0.75 (S) | 0.75 (S) |
| Inhibition zone diameter (mm) by disk diffusion | ||||||
| Sulfamethoxazole/trimethoprim 23.75 / 1.25 µg | 6 | 6 | 6 | 6 | 6 | 6 |
| Tetracycline 30 μg | 6 | 6 | 8 | 15 (S) | 6 | 12 (I) |
| Chloramphenicol 30 μg | 22 (S) | 23 (S) | 21 (S) | 16 (S) | 6 | 6 |
KP: Klebsiella pneumoniae, EN.C: Enterobacter cloacae
S: susceptible, I: intermediate
Disk diameter is 6 mm
MIC: Minimum Inhibitory Concentration
Resistance interpretation for all antimicrobials unless labeled S or I
Interpretation according to CLSI guidelines (2016) except for tigecycline and colistin, which were interpreted according to EUCAST guidelines (2019).
All isolates exhibited a carbapenemase activity as detected by the Carba NP test. MBL production was detected by EDTA-disk synergy tests in all isolates. This was confirmed by PCR, with the blaNDM-1 carbapenemase gene being amplified in the four K. pneumoniae isolates and the blaVIM-1 gene being amplified in the two E. cloacae isolates (Table 3). No additional carbapenemase gene was identified in those six isolates.
Table 3.
Antibiotic resistance enzymes and genotypic screening of six resistant Klebsiella pneumonia and Enterobacter cloacae isolates.
| Strain | Carbapenemase | Size of plasmids harboring carbapenemase-encoding genes | ESBL | BSBL | 16S rRNA methylase | AmpC variant | Aminoglycoside-modifying enzyme (acetyltransferase) | Quinolone resistance enzymes | MLST |
|---|---|---|---|---|---|---|---|---|---|
| KP-Q1 | NDM-1 | 70 kb | CTX-M-3 | TEM-1, OXA-1 | RmtC | - | AAC(6′)-Ib | QnrB | ST152 |
| KP-Q2 | NDM-1 | 70 kb | CTX-M-57 | TEM-1, OXA-1 | RmtC | - | AAC(6′)-Ib | QnrB | ST152 |
| KP-Q3 | NDM-1 | 70 kb | CTX-M-82 | TEM-1, OXA-1 | RmtC | - | AAC(6′)-Ib | QnrB | ST152 |
| KP-Q4 | NDM-1 | 70 kb | CTX-M-15 | TEM-1, OXA-1 | - | - | AAC(6′)-Ib | - | ST572 |
| EN.C.Q5 | VIM-1 | 60 kb | - | TEM-1 | - | CMY-4 | - | - | ST171 |
| EN.C.Q6 | VIM-1 | 150 kb | - | - | - | - | AAC(6′)-Ib | - | ST73 |
KP: Klebsiella pneumoniae
EN.C: Enterobacter cloacae
ESBL: Extended-spectrum β-lactamase
MLST: Multilocus sequence typing
Additionally, three K. pneumoniae isolates harbored a 16S rRNA methylase encoding gene, rmtC, and the quinolone resistance gene, qnrB. Moreover, a single E. cloacae isolate harbored the plasmid-mediated AmpC-encoding genes, blaCMY-4. Additionally, all isolates were found to harbor CTX-M-ESBL-encoding genes. All K. pneumoniae isolates were found to harbor OXA-1 broad spectrum beta-lactamases (BSBL). TEM-1-BSBL was found in all isolates except E. cloacae isolate EN.C.Q6. The aminoglycoside-modifying enzyme AAC(6′)-Ib was found in all isolates except E. cloacae isolate EN.C.Q5 (Table 3).
Transfer of imipenem resistance from all isolates to E. coli J53 was attempted by filter mating. The conjugation experiment demonstrated conjugative transfer of blaNDM-1 from K. pneumoniae and blaVIM-1 from E. cloacae to E. coli J53, confirming carriage of both genes on transferable plasmids. In K. pneumoniae, a single 70-kb plasmid was identified in all four isolates, onto which the blaNDM-1 gene was located. On the other hand, the blaVIM-1 gene was located on a 60-kb plasmid in a single E. cloacae isolate and on a 150-kb plasmid in the other isolate (Figure 1).
Figure 1.
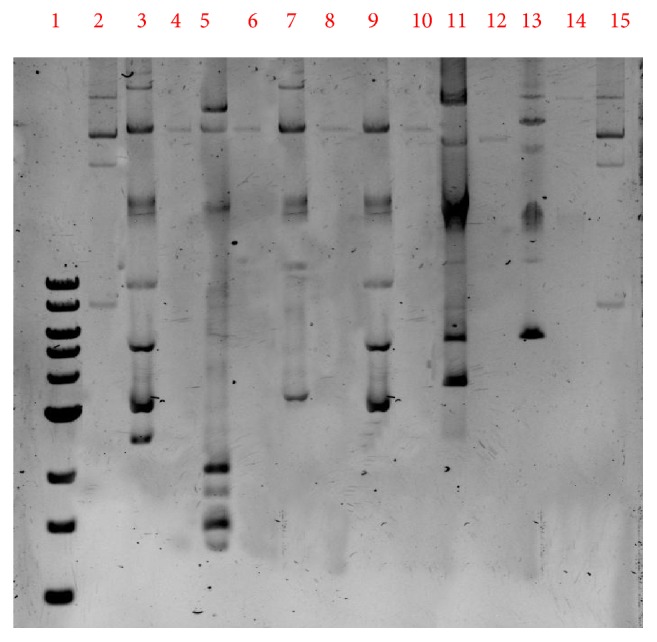
Electrophoretic profile of the plasmid DNA extracts from the strains and their transconjugants to confirm the results. 1. Ladder 1kb (10kb,8kb,6kb,5kb,4kb,3kb,2kb,1.5kb,1kb). 2. Ladder control strain 50192 (154kb, 66kb, 48kb, 7.8kb). 3. KP1 strain (70kb). 4. KP1 Transconjugant strains (70kb). 5. KP 2 (70kb). 6. KP2 Transconjugant strains (70kb). 7. KP 3 (70kb). 8. KP3 Transconjugant strains (70 kb). 9. KP 4 strains (70kb). 10. KP 4 Transconjugant strain 4 (70 kb). 11. EN.C 5 strains (60 kb). 12. EN.C 5 Transconjugant strain (60kb). 13. EN.C 6 strain (150kb). 14. EN.C6 Transconjugant strains (150kb). 15. Ladder control strain 50192 (154kb, 66kb, 48kb, 7.8kb).
MLST genotyping revealed that three K. pneumoniae isolates, which had actually been recovered from same hospital, belonged to ST152. The fourth K. pneumoniae isolate belonged to ST572 and had been recovered from the other hospital.
4. Discussion
The overuse of carbapenems during the last decade has led to increasing levels of bacterial resistance toward these potent β-lactams. Enterobacterial isolates have shown high carbapenem resistance rates worldwide, making alternative antibiotics, such as colistin and tigecycline, urgently needed. In the current study, we investigated by using phenotypic and genotypic methods six carbapenem-resistant enterobacterial isolates for their multiresistance determinants. In a recent study from Saudi Arabia [23], tigecycline resistance was not detected among 31 Enterobacteriaceae clinical isolates (21 K. pneumoniae and 10 E. coli) whereas colistin resistance was seen in 10% (one isolate) and 4.8% (one isolate) of their E. coli and K. pneumonia isolates, respectively. Similarly, colistin and tigecycline susceptibility were 100 and 87.5%, respectively, in 16 extensively drug-resistant K. pneumoniae strains from Saudi Arabia [24]. According to several reports [25–27], colistin and tigecycline remain the most effective antibiotics against carbapenem-resistant Gram-negative pathogens. Interestingly, the 2019 EUCAST guidelines (http://www.eucast.org/fileadmin/src/media/PDFs/EUCAST_files/Breakpoint_tables/v_9.0_Breakpoint_Tables.pdf) [13] changed the tigecycline resistance breakpoint to MIC > 0.5 mg/L. According to this new definition, all our isolates except one K. pneumoniae isolate would be considered resistant, suggesting an increasing challenge to public health. In the current study, resistance to colistin is not determined in our isolates, except one K. pneumonia isolate which has low-level colistin resistance (MIC 3 mg/L).
Two members of MBLs were detected among our isolates, namely, NDM-1-producing K. pneumoniae isolates and VIM-1-producing E. cloacae isolates. Although OXA-48 is commonly identified among K. pneumoniae isolates in some parts of Saudi Arabia [28–30], this resistance determinant was not detected in the present study. Moreover, a multicenter study in Saudi Arabia, Memish et al. [28], reported that OXA-48 and NDM-1 are the dominant carbapenemases among 124 Enterobacteriaceae (E. coli, Klebsiella spp., and Enterobacter spp.) isolated from 12 cities across the Kingdom of Saudi Arabia with low incidence of VIM and complete absence of KPC and IMP.
The coexistence of 16S rRNA methylase genes with β-lactamase genes was previously observed in Saudi Arabia. ESBLs were reported along with armA, rmtB, rmtC, and npmA in Enterobacteriaceae [31]. OXA-48 and NDM were detected along with armA and rmtB in K. pneumoniae [24]. The current study also revealed that the three clonally related K. pneumoniae isolates harbored the rmtC gene along with blaNDM-1. In addition, the plasmid mediated AmpC β-lactamase variant, CMY-4, was determined in our study in one E. cloacae which coproduces VIM-1 as well. This finding is the first report describing CMY carriage by a clinical E. cloacae isolate from Saudi Arabia.
The gene encoding the aminoglycoside-modifying enzyme AAC(6′)-Ib was detected in all but one isolates in our study. This enzyme was previously detected among extensively drug-resistant K. pneumonia isolates in Saudi Arabia [24]. Furthermore, the gene encoding the quinolone resistance protein QnrB was detected in the K. pneumoniae isolates from our study, in accordance with previous reports from Saudi Arabia [23, 24].
Noteworthy, strain EN.C.Q5 of our E. cloacae isolates was resistant to gentamicin although it did not have any of aminoglycoside resistance determinant examined (16S rRNA methylase: armA, rmtA, rmtB, rmtC, rmtD, and npmA, or acetyltransferase AAC(6′)-Ib), suggesting other resistance mechanism involved. Similarly, K. pneumonia strain KP-Q4 and E. cloacae strain EN.C.Q6 were resistant to ciprofloxacin and ofloxacin without qnrA, qnrB, and qnrS plasmid mediated quinolone resistance genes. Quinolone resistance in these isolates may be due to other plasmid mediated genes or mutations in quinolone resistance–determining regions of DNA gyrase (gyrA and gyrB) or DNA topoisomerase IV (parC and parE) [32].
5. Conclusions
Our study reports on a series of threatening resistance determinants responsible for the multidrug resistance pattern observed among clinical isolates. The identification of MBL-encoding genes (blaVIM in E. cloacae and blaNDM in K. pneumoniae) being identified onto conjugative plasmids raises concerns about the real extend of diffusion of those resistance genes in Saudi Arabia. Cooccurrence of multiple resistance determinants in clonally related K. pneumoniae isolates highlights the importance of controlling the dissemination of such isolates by early detection in hospital settings.
Acknowledgments
The authors extend their appreciation to the Deanship of Scientific Research at King Saud University for funding this work through research group project no. RGP-038.
Data Availability
The data used to support the findings of this study are available from the corresponding author upon request.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- 1.Meini M., Llarrull L. I., Vila A. J. Overcoming differences: the catalytic mechanism of metallo-β-lactamases. FEBS Letters. 2015;589(22):3419–3432. doi: 10.1016/j.febslet.2015.08.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Cornaglia G., Giamarellou H., Rossolini G. M. Metallo-β-lactamases: a last frontier for β-lactams? The Lancet Infectious Diseases. 2011;11(5):381–393. doi: 10.1016/S1473-3099(11)70056-1. [DOI] [PubMed] [Google Scholar]
- 3.Rood I. G., Li Q. Review: Molecular detection of extended spectrum-β-lactamase- and carbapenemase-producing Enterobacteriaceae in a clinical setting. Diagnostic Microbiology and Infectious Disease. 2017;89(3):245–250. doi: 10.1016/j.diagmicrobio.2017.07.013. [DOI] [PubMed] [Google Scholar]
- 4.Aubron C., Poirel L., Ash R. J., Nordmann P. Carbapenemase-producing Enterobacteriaceae, U.S. Rivers. Emerging Infectious Diseases. 2005;11(2):260–264. doi: 10.3201/eid1102.030684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Doumith M., Ellington M. J., Livermore D. M., Woodford N. Molecular mechanisms disrupting porin expression in ertapenem-resistant Klebsiella and Enterobacter spp. clinical isolates from the UK. Journal of Antimicrobial Chemotherapy. 2009;63(4):659–667. doi: 10.1093/jac/dkp029. [DOI] [PubMed] [Google Scholar]
- 6.Davin-Regli A., Pagès J.-M. Enterobacter aerogenes and Enterobacter cloacae; Versatile bacterial pathogens confronting antibiotic treatment. Frontiers in Microbiology. 2015;6:p. 392. doi: 10.3389/fmicb.2015.00392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hu X., Xu B., Yang Y., et al. A high throughput multiplex PCR assay for simultaneous detection of seven aminoglycoside-resistance genes in Enterobacteriaceae. BMC Microbiology. 2013;13(1, article no. 58) doi: 10.1186/1471-2180-13-58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Fernandez-Martinez M., Castillo B. R. D., Lecea-Cuello M. J., Rodriguez-Bano J., Pascual A., Martinez-Martinez L. Prevalence of aminoglycoside-modifying enzymes in Escherichia coli and Klebsiella pneumoniae producing extend spectrum ß-lactamases collected in two multicenter studies in Spain. Microbiol Drug Resistance. 2018;24(4):367–376. doi: 10.1089/mdr.2017.0102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Chiem K., Jani S., Fuentes B., Lin D. L., Rasche M. E., Tolmasky M. E. Identification of an inhibitor of the aminoglycoside 6′-N-acetyltransferase type Ib [AAC(6′)-Ib] by glide molecular docking. MedChemComm. 2016;7(1):184–189. doi: 10.1039/C5MD00316D. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Poirel L., Madec J., Lupo A., et al. Antimicrobial resistance in Escherichia coli. Microbiology Spectrum. 2018;6(4) doi: 10.1128/microbiolspec.ARBA-0026-2017. [DOI] [PubMed] [Google Scholar]
- 11.Jacoby G. A., Strahilevitz J., Hooper D. C. Plasmid-mediated quinolone resistance. Microbiology Spectrum. 2014;2(5) doi: 10.1128/microbiolspec.PLAS-0006-2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Clinical And Laboratory Standards Institute. Performance Standards for Antimicrobial Susceptibility Testing. 26th. Wayne, Pa, USA: CLSI; 2016. (Informational Supplement, M100-S). [Google Scholar]
- 13.European Committee on Antimicrobial Susceptibility Testing (EUCAST) http://www.eucast.org/fileadmin/src/media/PDFs/EUCAST_files/Breakpoint_tables/v_9.0_Breakpoint_Tables.pdf.
- 14.Nordmann P., Poirel L., Dortet L. Rapid detection of carbapenemase-producing Enterobacteriaceae. Emerging Infectious Diseases. 2012;18(9):1503–1507. doi: 10.3201/eid1809.120355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lee K., Chong Y., Shin H. B., Kim Y. A., Yong D., Yum J. H. Modified Hodge and EDTA-disk synergy tests to screen metallo-β-lactamase-producing strains of Pseudomonas and Acinetobacter species. Clinical Microbiology and Infection. 2001;7(2):88–91. doi: 10.1046/j.1469-0691.2001.00204.x. [DOI] [PubMed] [Google Scholar]
- 16.Dallenne C., da Costa A., Decré D., Favier C., Arlet G. Development of a set of multiplex PCR assays for the detection of genes encoding important β-lactamases in Enterobacteriaceae. Journal of Antimicrobial Chemotherapy. 2010;65(3):490–495. doi: 10.1093/jac/dkp498.dkp498 [DOI] [PubMed] [Google Scholar]
- 17.Pérez-Pérez F. J., Hanson N. D. Detection of plasmid-mediated AmpC β-lactamase genes in clinical isolates by using multiplex PCR. Journal of Clinical Microbiology. 2002;40(6):2153–2162. doi: 10.1128/jcm.40.6.2153-2162.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Poirel L., Walsh T. R., Cuvillier V., Nordmann P. Multiplex PCR detection of acquired carbapenemase genes. Diagnostic Microbiology and Infectious Disease. 2011;70(1):119–123. doi: 10.1016/j.diagmicrobio.2010.12.002. [DOI] [PubMed] [Google Scholar]
- 19.Cattoir V., Poirel L., Rotimi V., Soussy C.-J., Nordmann P. Multiplex PCR for detection of plasmid-mediated quinolone resistance qnr genes in ESBL-producing enterobacterial isolates. Journal of Antimicrobial Chemotherapy. 2007;60(2):394–397. doi: 10.1093/jac/dkm204. [DOI] [PubMed] [Google Scholar]
- 20.Berçot B., Poirel L., Nordmann P. Updated multiplex polymerase chain reaction for detection of 16S rRNA methylases: high prevalence among NDM-1 producers. Diagnostic Microbiology and Infectious Disease. 2011;71(4):442–445. doi: 10.1016/j.diagmicrobio.2011.08.016. [DOI] [PubMed] [Google Scholar]
- 21.Sinnett D., Montpetit A. Isolation of cosmid and BAC DNA from E. coli. Methods in Molecular Biology (Clifton, N.J.) 2003;235:99–102. doi: 10.1385/1-59259-409-3:99. [DOI] [PubMed] [Google Scholar]
- 22.Pitout J. D., Thomson K. S., Hanson N. D., Ehrhardt A. F., Coudron P., Sanders C. C. Plasmid-mediated resistance to expanded-spectrum cephalosporins among Enterobacter aerogenes strains. Antimicrobial Agents and Chemotherapy. 1998;42(3):596–600. doi: 10.1128/AAC.42.3.596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Al-Agamy M. H., Aljallal A., Radwan H. H., Shibl A. M. Characterization of carbapenemases, ESBLs, and plasmid-mediated quinolone determinants in carbapenem-insensitive Escherichia coli and Klebsiella pneumoniae in Riyadh hospitals. Journal of Infection and Public Health. 2018;11(1):64–68. doi: 10.1016/j.jiph.2017.03.010. [DOI] [PubMed] [Google Scholar]
- 24.Abdalhamid B., Elhadi N., Albunayan S., Alsamman K., Aljindan R. First description of methyltransferases in extensively drug-resistant Klebsiella pneumoniae isolates from Saudi Arabia. Journal of Medical Microbiology. 2017;66(7):859–863. doi: 10.1099/jmm.0.000480. [DOI] [PubMed] [Google Scholar]
- 25.Gamal D., Fernández-Martínez M., Salem D., et al. Carbapenem-resistant Klebsiella pneumoniae isolates from Egypt containing bla NDM-1 on IncR plasmids and its association with rmtF. International Journal of Infectious Diseases. 2016;43:17–20. doi: 10.1016/j.ijid.2015.12.003. [DOI] [PubMed] [Google Scholar]
- 26.Kiaei S., Moradi M., Hosseini-Nave H., Ziasistani M., Kalantar-Neyestanaki D. Endemic dissemination of different sequence types of carbapenem-resistant klebsiella pneumoniae strains harboring blaNDMand 16S rRNA methylase genes in kerman hospitals, iran, from 2015 to 2017. Infection and Drug Resistance. 2019;12:45–54. doi: 10.2147/IDR.S186994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Samonis G., Maraki S., Karageorgopoulos D. E., Vouloumanou E. K., Falagas M. E. Synergy of fosfomycin with carbapenems, colistin, netilmicin, and tigecycline against multidrug-resistant Klebsiella pneumoniae, Escherichia coli, and Pseudomonas aeruginosa clinical isolates. European Journal of Clinical Microbiology & Infectious Diseases. 2012;31(5):695–701. doi: 10.1007/s10096-011-1360-5. [DOI] [PubMed] [Google Scholar]
- 28.Memish Z. A., Assiri A., Almasri M., et al. Molecular characterization of carbapenemase production among gram-negative bacteria in Saudi Arabia. Microbial Drug Resistance. 2015;21(3):307–314. doi: 10.1089/mdr.2014.0121. [DOI] [PubMed] [Google Scholar]
- 29.Shibl A., Al-Agamy M., Memish Z., Senok A., Khader S. A., Assiri A. The emergence of OXA-48- and NDM-1-positive Klebsiella pneumoniae in Riyadh, Saudi Arabia. International Journal of Infectious Diseases. 2013;17(12):e1130–e1133. doi: 10.1016/j.ijid.2013.06.016. [DOI] [PubMed] [Google Scholar]
- 30.uz Zaman T., Aldrees M., Al Johani S. M., Alrodayyan M., Aldughashem F. A., Balkhy H. H. Multi-drug carbapenem-resistant Klebsiella pneumoniae infection carrying the OXA-48 gene and showing variations in outer membrane protein 36 causing an outbreak in a tertiary care hospital in Riyadh, Saudi Arabia. International Journal of Infectious Diseases. 2014;28:186–192. doi: 10.1016/j.ijid.2014.05.021. [DOI] [PubMed] [Google Scholar]
- 31.Al Sheikh Y. A., Marie M. A., John J., Krishnappa L. G., Dabwab K. H. Prevalence of 16S rRNA methylase genes among β-lactamase-producing Enterobacteriaceae clinical isolates in Saudi Arabia. Libyan Journal of Medicine. 2014;9(1):p. 24432. doi: 10.3402/ljm.v9.24432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Acar J. F., Goldstein F. W. Trends in bacterial resistance to fluoroquinolones. Clinical Infectious Diseases. 1997;24, supplement 1:S67–S73. doi: 10.1093/clinids/24.supplement_1.s67. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data used to support the findings of this study are available from the corresponding author upon request.


