Abstract
We evaluated the spectral properties of four stilbene derivatives containing the boronic acid group [–B(OH)2]: stilbene-4-boronic acid (STBA), 4′-cyanostilbene-4-boronic acid (CSTBA), 4′-methoxystilbene-4-boronic acid (MSTBA), and 4′-(dimethylamino)stilbene-4-boronic acid (DSTBA). The emission spectrum of DSTBA displays a large solvent-polarity dependence showing the formation of a photoinduced charge transfer state (CT). This state is weakly present in MSTBA and not present for CSTBA and STBA for the neutral form of the boronic acid group. These results show the donor withdrawing property of the neutral form of the boronic acid group. At higher pH, the boronic acid group is present in the anionic form [–B(OH)3−], resulting in a change of the configuration around the boron atom from the triangular planar (sp2 hybridization) to the tetrahedral conformation (sp3 hybridization). This change induced a blue shift of about 50 nm and an increase of intensity in the emission spectrum of DSTBA because of the loss of the electron-withdrawing properties for the anionic form of the boronic acid group, leading to the loss of the CT effect. The same effect is also observed for MSTBA. In contrast, a red shift of about 35 nm and a decrease of intensity are observed for CSTBA from the neutral to the anionic forms of the boronic acid group. These observations lead to the conclusion that the anionic form of the boronic acid group acts as an electron donor group and a photoinduced CT state can be formed when an electron withdrawing group is present on the fluorophore. The usefulness of this effect for the development of saccharide probes is also demonstrated. After addition of sugar, the emission spectra of DSTBA and MSTBA showed a blue shift and an increase of the intensity. On the other hand, a red shift and a decrease of the intensity are observed in the emission spectra of CSTBA after addition of sugar. A change from the neutral to the anionic form of the boronic acid group is used to explain these changes. These results show that the use of the combination of electron donor or withdrawing groups with the boronic acid group is a new and promising way to develop ratiometric fluorescent probes for glucose and other saccharides.
1. Introduction
Recent interest in the boron–aromatic systems stems from the concept of π-electron aromaticity and conjugation across sp2-hybridized boron.1 Recent reports highlight the potential use of boron-containing conjugated polymers in the emerging optoelectronic applications.2–5 Lee et al. investigated the effect of the B−-for-C substitution on the photophysics and photochemistry of borastilbenes and borastyrylstilbenes.6–7
Phenyl boronic acid group [phe–B(OH)2] had attracted a lot of interest during the past decade for its ability to covalently bind diols and sugars.8–11 Lorand and Edwards investigated the structure of the neutral and anionic forms [phe–B(OH)3−] of the phenylboronic acid group.12 Their results showed that the neutral form of the boronic acid group linked to the phenyl moiety has a planar triangular conformation with a sp2-hybridized boron atom. On the other hand, the anionic form has a tetrahedral conformation with a sp3-hybridized boron atom. Two research groups have investigated the effect of this change on the emission of fluorophores in order to evaluate their use for the development of fluorescent probes for saccharides. Yoon and Czarnik used anthrylboronic acid13 and Suenaga et al. used naphthyl, biphenyl, pyrenyl, and stilbeneboronic acid.14 In the case of the anthrylboronic acid, a decrease of 40% of the emission intensity was observed following the formation of the anionic form of the boronic acid group. Complexation of the boronic acid moiety with saccharides decreases the pKa of boronic acid group, 8.8 to 5.9 in saturated fructose solution.13 As a result, complexation with the saccharide induces the formation of the anionic form of the boronic acid and then a decrease of the emission intensity. This decrease is relatively small, 30% for fructose and about 10% for glucose. Suenaga et al. obtained similar results. For this reason, the direct insertion of the boronic acid group on a fluorophore has not been deeply investigated.
Several laboratories have investigated the ability of the boronic acid group to interact with amino groups. Fluorescence probes based on a decrease of the photoinduced electron transfer (PET) of amino-substituted fluorophores, mainly anthracene, have been synthesized.15–18 This mechanism resulted in a significant intensity increase, up to 7-fold, and a fluorescence lifetime change after binding saccharides.18 Molecular rigidification induced by saccharides interaction using the boronic acid group as a chelator group has also been used with a cyanine dye for the development of fluorescence probes.19 Excimer formation between two pyrene moieties has also been used.20 Despite these interesting approaches to use the combination of the boronic acid group and fluorophores, they are mostly restricted to a few fluorophores. The PET mechanism is expected to become ineffective for long wavelength fluorophore.21 Rigidification and excimer formation can be applied only to few fluorophores.
In an attempt to extend the usefulness of fluorescent probes for saccharides based on the boronic acid group, we investigated the possibility of using excited state charge transfer (CT) between the boronic acid moiety and a donor and/or acceptor groups. The excited charge transfer phenomenon has been widely used and described in the development of many luminescent probes for ions.22,23 As discussed above, the sp2-hybridized boron atom inserted directly on the fluorophore shows resonance with the aromatic system of a chromophore (compound 1 in Scheme 1). Because of the empty p orbital present on the boron atom, the boronic acid group should act as an electron acceptor group. The incorporation of a donor group (R in Scheme 1) on the same chromophore should result in excited charge transfer. As the boron with an sp2 hybridization changes to a sp3 hybridization for the anionic form (compound 2 in Scheme 1), the boronic acid group is no longer an electron acceptor group. This should lead to a change in the spectroscopic and photophysical properties of the probes. At pH 7–8, the form 1 (Scheme 1) should be dominant in solution; after, the addition of sugar compound 4 should become predominant as the pKa of the boronic acid group decreases following the complexation with sugar. The difference in the hybridization of compounds 1 and 4 should result in a change of the optical properties of the probes induced by the presence of sugar. This would lead to new fluorescent probes for sugar sensing. Also, the CT mechanism is applicable to a wide range of different luminescent probes and not restricted to only some probes as for the PET, rigidification, or excimer mechanism.
SCHEME 1.
Equilibrium and Conformation of the Different Forms of the Boronic Acid Group with and without Sugar
To check this hypothesis, we synthesized five different stilbene derivatives combining the boronic acid group in position 4 and donor or acceptor groups in position 4′. The molecular structures of the substituted stilbenes investigated are shown in Figure 1. Dimethylamino and cyano groups are well-known and used as donor and acceptor groups, respectively. Both groups have been widely used in the investigation of excited charge transfer in stilbene derivatives.24–26 We also investigated the insertion of the methoxy group, which is a weaker donor group. Stilbeneboronic acid (STBA) is used for a control molecule. A nitro group derivative has also been synthesized, but no fluorescence was observed in methanol and water for this compound. The results show that the insertion of a donor group and the boronic acid group directly on the stilbene in positions 4 and 4′, respectively, lead to an excited charge transfer state. At higher pH, a new blue shifted emission band appears because of the loss of the acceptor properties of the anionic form of the boronic acid group. On the other hand, the incorporation of the cyano group does not lead to any excited charge transfer state, but for the anionic form, an excited charge transfer can be observed. This is observed by the appearance of a new red shifted band in the emission spectrum. The application of both effects for glucose sensing are shown and discussed.
Figure 1.
Molecular structure of the substituted stilbenes investigated.
2. Experimental Section
d-Glucose, d-galactose, and d-fructose were purchased from Sigma and used as received. All solvents used were HPLC grade and purchased from Aldrich. Stilbene-4-boronic acid (STBA), 4′-cyanostilbene-4-boronic acid (CSTBA), 4′-methoxystilbene-4-boronic acid (MSTBA), and 4′-(dimethylamino)stilbene-4-boronic acid (DSTBA) were synthesized by the Wittig reaction between the para-substituted benzaldehydes and the para-boronic acid derivative of the benzyltriphenylphosphonium bromide. This latter was synthesized by the reaction of the p-bromomethylphenylboronic acid16 with triphenylphosphine in toluene. All compounds were purified by recrystalization in methanol, and all NMR spectra were consistent with the structure and showed only the presence of the trans conformation.
Absorption spectra were recorded with a Cary 50 UV–vis spectrophotometer from Varian. Emission spectra were recorded with a Varian Eclipse spectrofluorometer from Varian. In both case, the measurements were taken at room temperature in a 1 cm quartz cuvette. For all measurements, the absorbances of the solutions were about 0.1 corresponding to a concentration range of (2–3) × 10−6 M of the fluorophore. Fluorescence quantum yields were measured against p-quaterphenyl in cyclohexane (ϕF = 0.89)27 for trans-stilbene (ST) and STBA, anthracene in cyclohexane (ϕF = 0.36)27 for CSTBA and MSTBA, and Quinine sulfate in 0.1N sulfuric acid (ϕF = 0.577)28 for DSTBA.
Titration curves against pH were measured in buffer solutions: acetate buffer for pH 4.0–5.5 and phosphate buffer for pH 6.0–9.0 and carbonate buffer for pH 10.0–11.0. Titration curves were fitted and pKa (pKa = −log Ka) values were obtained using the equation:
| (1) |
where Iacid and Ibase are the intensity limits in the acid and basic region, respectively. Titration curves against sugar were fitted, and dissociation constant (KD) values were obtained using the equation
| (2) |
where I0 and If are the initial (no sugar) and final (plateau) intensities of the titration curves. All solutions in water and buffer contained also 33.3% (2:1 v/v) methanol to avoid any problem because of the aggregation.
Frequency-domain (FD) measurements were performed using the instrumentation described previously.29 Excitation was provided by a rhodamine 6G dye laser at ~305 nm for STBA, CSTBA, and MSTBA and by a pyridine 2 dye laser at ~350 nm for DSTBA. Emission was observed through a combination of a cutoff and glass filters to remove scattered and Raman scattered light. The measurements were taken in a 1 cm cuvette With continuously stirring. The frequency intensity profiles were analyzed by nonlinear least squares in terms of the multiexponential model
| (3) |
where αi are the preexponential factors associated with the decay time τi, with ∑iαi = 1.0. The mean lifetime is given by
| (4) |
where fi are the fractional steady-state intensities of each lifetime component
| (5) |
Errors of 0.5 and 0.05 on the phase angle and modulation have been used, respectively.
3. Results and Discussion
3.1. Spectroscopic and Photophysical Properties
Figure 2 shows the absorption and fluorescence spectra of the four stilbene derivatives investigated and the unsubstituted trans-stilbene. Spectral parameters are shown in Table 1. Insertion of the boronic acid in the 4 position induces a small red shift in the absorption spectrum in comparison with ST (Figure 1A) This shift is due to the hyperconjugation of the aromatic system with the empty p orbital of the boron atom. The addition of cyano, methoxy, and dimethylamino groups in the 4′ position also induced a red shift in the absorption spectra (Figure 1A) The maximum of the absorption spectrum of DSTBA (346 nm, Table 1) is relatively similar to that of the 4-(dimethylamino)-stilbene (DS) (351 nm in acetonitrile).25 This shows that no particular effects are involved in the ground state following the insertion of a donor or acceptor group in position 4′ of the stilbeneboronic acid.
Figure 2.
Absorption (A) and emission (B) spectra of the stilbenes investigated in water/methanol 1:1 (v/v) at room temperature. ST is for trans-stilbene.
TABLE 1.
Spectral Properties and Fluorescence Quantum Yield (ϕF) of the Stilbene Derivatives in Water/Methanol 1:1 (v/v) at Room Temperature
| λabs (nm) |
ε (M−1 cm−1) |
λF (nm) |
Δa (cm−1) |
ϕF | |
|---|---|---|---|---|---|
| STb | 296 (295)c |
33 800 (31 500) |
348 (349) |
5100 (5250) |
0.021 (0.044) |
| STBA | 314 (317) |
39 700 (31 300) |
358 (359) |
3950 (3690) |
0.009 (0.054) |
| CSTBA | 327 (330) |
47 300 (34 900) |
385 (373) |
4600 (3500) |
0.006 (0.013) |
| MSTBA | 324 (325) |
38 000 (25 700) |
391 (370) |
5320 (3750) |
0.025 (0.31) |
| DSTBA | 346 (347) |
32 800 (36 500) |
485 (400) |
8300 (3820) |
0.088 (0.64) |
Δ: Stokes shift.
ST: trans-stilbene.
In cyclohexane.
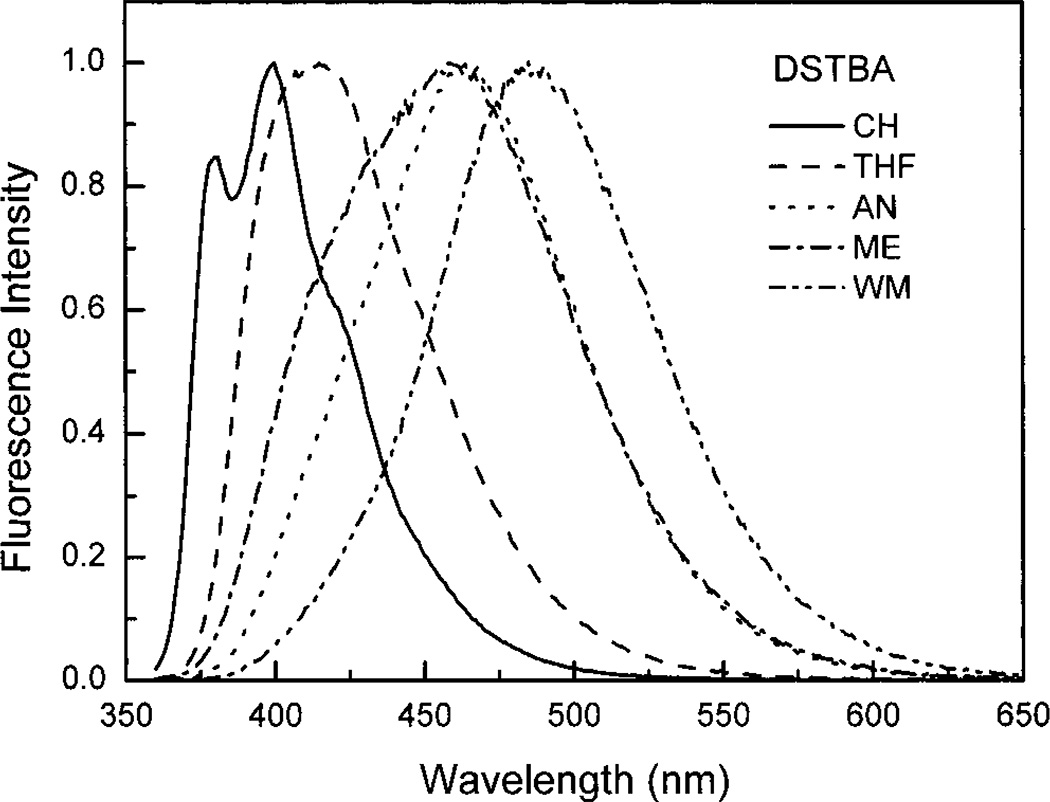
Fluorescence spectra follow the same trend as the absorption spectra. STBA, CSTBA, and MSTBA are red shifted in comparison with ST (Figure 1B). These shifts are similar to those observed in the absorption spectra. The Stokes shifts for these three compounds are similar to the one observed for ST, showing that no major effects are involved in the excited state for these compounds. On the other hand, the emission spectrum of DSTBA shows a large bathochromic shift in comparison with STBA. Because this shift, 127 nm, is much larger than the shift observed in the absorption spectrum, 32 nm, the extent of the hyperconjugation due to the insertion of the amino group is not enough to explain this shift, and this shows that an additional excited-state relaxation process is involved. The emission spectrum of DSTBA shows also an important bathochromic shift in comparison to that with DS (440 nm in acetonitrile). These results suggest the formation of an excited induced CT state for DSTBA. To verify this hypothesis, we recorded the emission spectrum of DSTBA in various solvents of different polarity (Figure 3). The emission spectrum of DSTBA in cyclohexane shows vibronic structure and is centered at 400 nm. Although the polarity of the solvent is increased, the emission spectrum shows a large bathochromic shift (85 nm from CH to WM) and the vibronic structure is lost. On the other hand, the absorption spectra of DSTBA in the same series of solvents do not show any significant shift (not shown). Also, the Stokes shift increases from 3820 cm−1 in cyclohexane (CH) to 8300 cm−1 in the water/methanol (50:50 v/v) mixture (WM). These observations are consistent with the formation of an excited induced CT state due to the presence of the donor amino group and the acceptor boronic acid group on the fluorophore. These results on DSTBA are comparable with the results reported for 4-(dimethylamino)-4-cyanostilbene (DCS) where a bathochromic shift of 115 nm is observed from methylcyclohexane to acetonitrile.24 ST and STBA do not show any solvent effect, but CSTBA and MSTBA show a little bathochromic shift with the increase of the polarity of the solvent (results not shown). These shifts are much smaller, 12 nm for CSTBA and 21 nm for MSTBA from CH to WM, than that observed for DSTBA, 85 nm from CH to WM.
Figure 3.
Normalized emission spectra of DSTBA in different solvents. CH, cyclohexane; THF, tetrahydrofuran; AN, acetonitrile; ME, methanol; and WM, water/methanol 1:1 (v/v) at room temperature, λex = 350 nm.
Table 1 also reports the fluorescence quantum yields of the derivatives investigated. For all stilbenes reported in this study, the fluorescence quantum yields decreased with the increase of the polarity of the solvent. This decrease is about 2–5 time smaller, from CH to WM, for ST, STBA, and CSTBA, whereas this decrease is more important for MSTBA and DSTBA, 10–15 time smaller from CH to WM. Despite this more important solvent effect, ϕF remains larger for MSTBA and DSTBA than for the other three compounds in all of the solvents.
Fluorescence decay parameters of the stilbenes investigated are listed in Table 2. Mean fluorescence lifetimes of STBA and CSTBA are similar and comparable to the mean lifetime of ST, 26 ps in WM. For these three compounds, fluorescence decay profiles were satisfactorily fitted with a single exponential and lifetimes do not show any effect of solvent. MSTBA and DSTBA show much longer lifetimes. For these two compound, a single-exponential model was used to fit the fluorescence decay curves in CH and THF, whereas a two exponential model was needed for the decay profiles in acetonitrile (AN), methanol (ME), and WM. The observed mean lifetime decreased with the polarity of the solvent for MSTBA and DSTBA. Examples of the fluorescence decay profiles of DSTBA in CH and WM are displayed in Figure 4. The decrease of the mean lifetime was larger for MSTBA, 670–80 ps from CH to WM, than for DSTBA, 840–350 ps from CH to WM. The similar solvent effects observed for DSTBA and MSTBA for the steady state and intensity decays could suggest that a CT state is also involved in the excited state of MSTBA.
TABLE 2.
Fluorescence Decay Parameters of the Stilbenes Investigated in Water/Methanol 1:1 (v/v) at Room Temperature
| τ1 (ps) |
τ2 (ps) |
α1 | α2 | τ̄ (ps) |
χR2 | |
|---|---|---|---|---|---|---|
| STBA | 23 | 1.0 | 23 | 1.8 | ||
| CSTBA | 21 | 1.0 | 21 | 5.4 | ||
| MSTBA | 30 | 130 | 0.82 | 0.18 | 80 | 2.2 |
| DSTBA | 40 | 360 | 0.20 | 0.80 | 350 | 0.9 |
Figure 4.
Frequency decay profiles of DSTBA in cyclohexane (CH) and water/methanol 1:1 (v/v) (WM) at room temperature.
3.2. pH and Sugar Effects on the Optical Spectra
3.2.1. STBA
Figure 5 shows the intensity changes after addition of sugars for STBA. A decrease of the fluorescence emission is observed after the addition of sugar. The overall decrease is relatively weak, about 15%. Because the same intensity change is observed by increasing the pH, this change is attributed to the formation of the anionic form of the boronic acid group. The pKa of STBA is 8.86 (Table 3) and decreases to 6.4 for the complex STBA:fructose. A pKa of 8.86 is similar to the pKa of a multitude of phenylboronic acid derivatives reported in the literature.13,30–32 The decrease of this pKa for the complex with sugar is also a general observation for this chelator group.13,30–32 For example, Yoon and Czarnik reported a pKa change of 8.8–5.9 for the anthrylboronic acid and its complex with fructose.13 At pH 8.0, STBA is present mostly in its neutral form. After the addition of sugar, the complex exists under the anionic form because of its lower pKa (see Scheme 1). This change from the neutral to the anionic form is at the origin of the intensity changes observed for STBA and the other complexes presented in this study. Dissociation constants (KD) for the three different sugars are presented in Table 3. All titration curves against sugars have been taken at pH 8.0. This pH does not correspond necessarily to the maximum effect of the optical change but is an average pH that allows measurements of all derivatives at the same pH. The pH effect on the KD values will be discussed later (section 3.3). Monophenylboronic acid groups are well-known to be more sensitive to d-fructose and the sensitivity decreases for d-galactose and decreases again for d-glucose.10,12,13,33,34 All stilbene derivatives presented in this study follow this rule and show KD similar to the other monophenylboronic group. For example, phenylboronic acid shows KD of 0.2, 3.6, and 9.1 mM for d-fructose, d-galactose, and d-glucose, respectively.34
Figure 5.
Titration curves of STBA in phosphate buffer pH 8.0/ methanol 2:1 (v/v) at room temperature, λex = 310 nm. Insert: emission spectra changes upon the addition of d-fructose. Only the representative spectra are shown.
TABLE 3.
pKa and Dissociation Constant (KD) of the Different Stilbenes Investigated in the Absence and Presence of Sugarsa
| pKa | KD (mM) | |||||
|---|---|---|---|---|---|---|
| alone | + d-fructose (50 mM) | + d-glucose (50 mM) | d-fructose | d-galactose | d-glucose | |
| STBA | 8.86 (± 0.08) |
6.4 (± 0.1) |
7.63 (± 0.06) |
3.4 (± 0.7) |
6 (± 1) |
11 (± 1) |
| CSTBA | 8.17 (± 0.03) |
5.84 (± 0.04) |
7.30 (± 0.04) |
0.65 (± 0.04) |
12.5 (± 0.8) |
18.3 (± 0.8) |
| MSTBA | 8.58 (± 0.03) |
6.36 (± 0.03) |
7.88 (± 0.01) |
1.0 (± 0.1) |
26 (± 2) |
43 (± 4) |
| DSTBA | 9.14 (± 0.03) |
6.61 (± 0.02) |
8.34 (± 0.01) |
2.5 (± 0.2) |
49 (± 1) |
98 (± 3) |
All solutions contained 33.3% methanol.
KD measured at pH 8.0.
Intensity changes observed for STBA are similar to the intensity changes reported for anthryl-2-boronic acid.13 For this anthracene derivative, the boronic acid group is also linked directly on the anthracene fluorophore. Intensity decreases of 30 and 10% were reported after the addition of fructose and glucose at pH 7.4, respectively. To explain these decreases, the authors suggested a photoinduced electron transfer (PET) mechanism where the negative charge on the boron atom acts as the quencher. This statement was based on the oxidizability of the borate. In the insert of Figure 5, we show the emission spectra of STBA with different concentrations of fructose. We can observe that the emission is slightly blue shifted and an isosbestic point appears at about 338 nm after the addition of fructose. This could suggest that the effects of sugars on the emission spectra of STBA could be induced by the change of conformation of the boron atom from the neutral to the anionic forms, sp2 to sp3, as presented in the Introduction and schematically shown in Scheme 1. We would like to suggest that the loss of the empty p orbital of the boron atom could result in a partial loss of the resonance between the aromatic system and the boronic acid group. Further studies would be necessary to clarify the nature of these fluorescence changes.
3.2.2. DSTBA
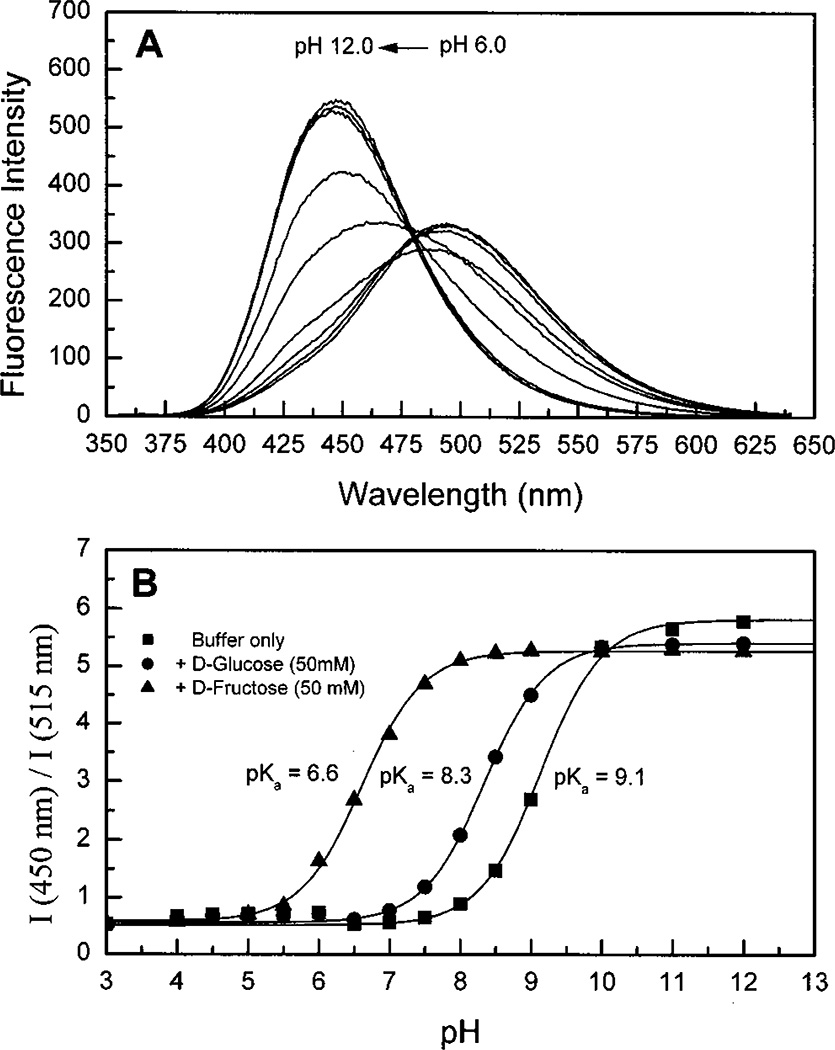
Figure 6A displays the pH dependence of the fluorescence spectra of DSTBA. The emission spectrum shows a hypsochromic shift of about 45 nm and an increase of the intensity as the pH is increased from 6 to 12. These dramatic changes in the emission band of DSTBA are explained by the loss of the electron withdrawing property of the boronic acid group following the formation of the anionic form at high pH (Scheme 1). This results in the loss of the CT excited state, resulting in a blue shift and the increase of the intensity. The absorption spectrum of DSTBA also shows a hypsochromic shift of about 15 nm (results not shown), showing that the formation of the anionic forms also perturbs the ground state of the compound. We also observed significant blue shifts in both absorption and emission spectra at pH below 4.0 (results not shown), which was explained by the protonation of the dimethylamino group. Titration curves against pH with and without sugar are presented in Figure 6B. The pKa of DSTBA is slightly higher than that observed for STBA, and the presence of sugar induces a decrease of the pKa.
Figure 6.
(A) pH dependence on the emission spectra of DSTBA without sugar at room temperature, λex = 330 nm. (B) Titration curves of DSTBA with and without sugar.
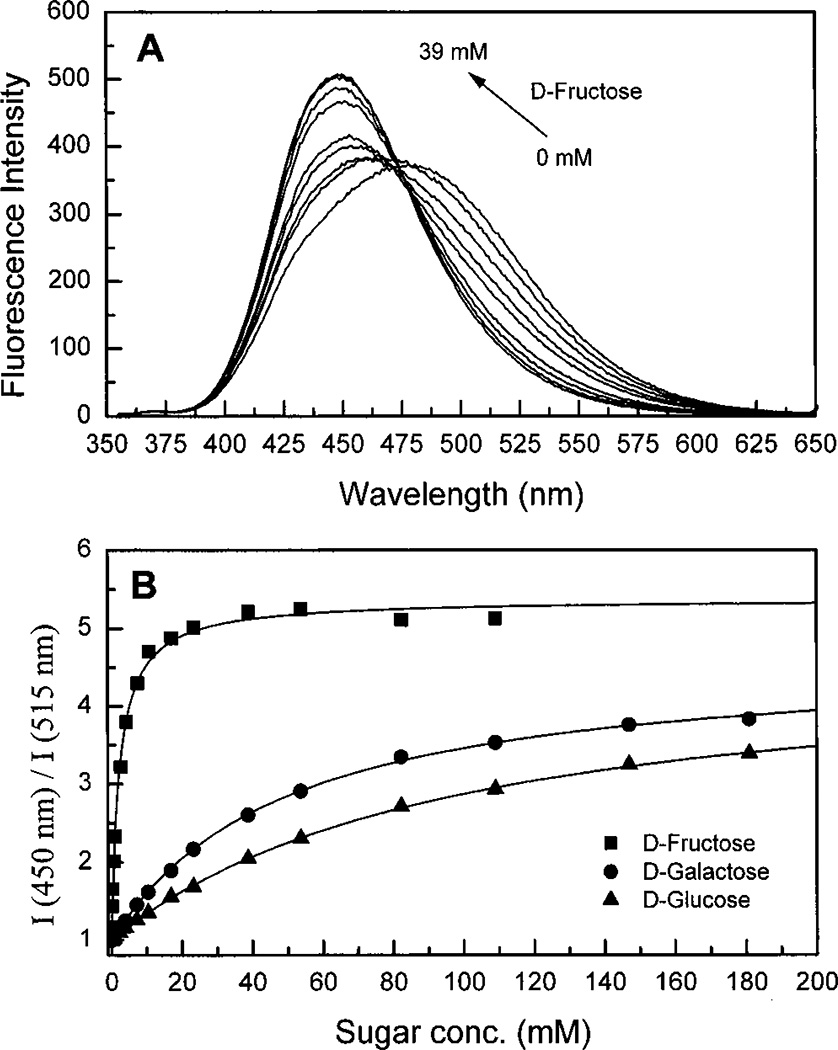
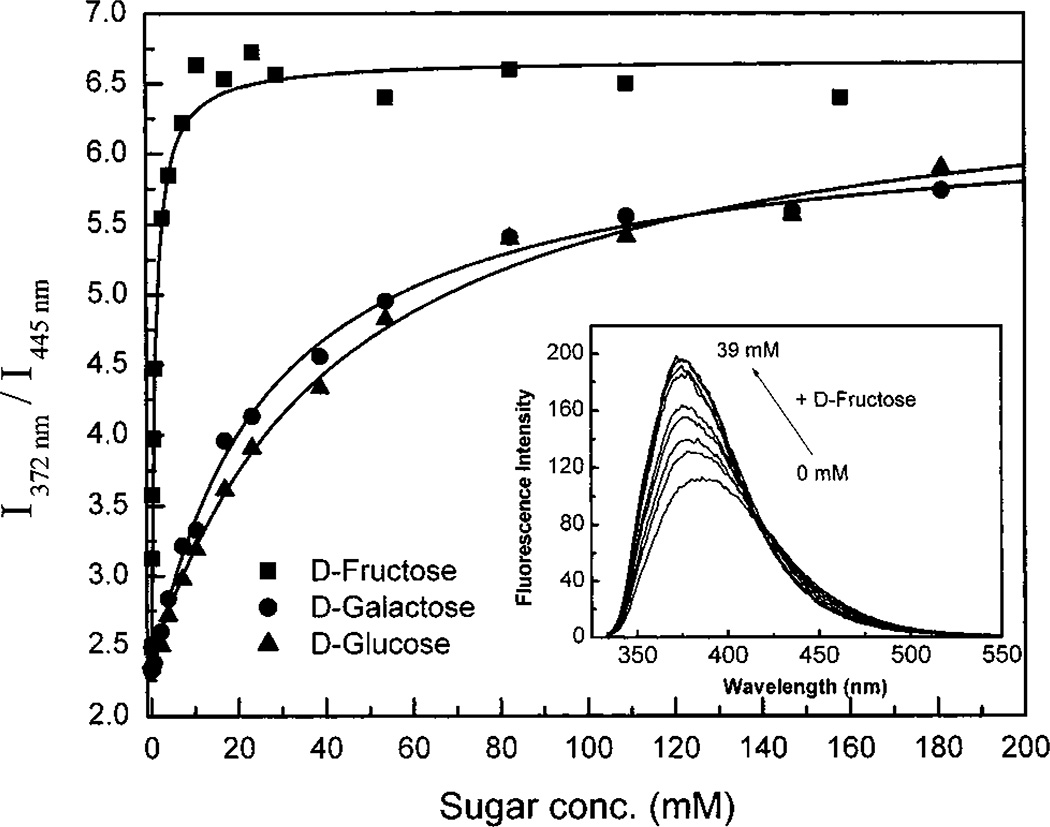
Figure 7A displays the effect of fructose on the emission spectrum of DSTBA. As observed for the pH, the addition of fructose induces a hypsochromic shift and an increase of the intensity of emission. The same spectral changes are observed for the other sugars. As explained above, the results are interpreted with the formation of the anionic form of the sugar complex because of the decrease of the pKa of this complex. In comparison with STBA, the formation of the anionic form in DSTBA induces not only a change in the emission intensity but also a blue shift in the emission band. This important effect, resulting from the insertion of a electron donating group on the fluorophore, provides a wavelength-ratiometric probe for the analysis of sugars using changes in the donor–acceptor properties of this kind of compound. The ratiometric method is well-known to be a superior technique for quantitative measurements of analytes in comparison with simple intensity changes.35–37 Titration curves against sugars for DSTBA are shown in Figure 7A, and KD values are listed in Table 3. This compound shows similar affinity for fructose than the other phenylboronic acid groups10,33,34 but much less sensitivity for galactose and glucose.
Figure 7.
(A) Change in the emission spectra of DSTBA after addition of d-fructose, in phosphate buffer pH 8.0/methanol 2:1 (v/v) at room temperature, λex = 330 nm. (B) Titration curves of DSTBA with the different sugars.
3.2.3. MSTBA
The pH dependence of the fluorescence of MSTBA (results not shown) is quite similar to what we observed for DSTBA. By increasing the pH, we can observe a modest hypsochromic shift and an increase of the intensity of the emission. The blue shift is smaller, 29 nm, in comparison with that of DSTBA. This lead to the conclusion that a CT state is also involved in the excited state of MSTBA. Because of a smaller effect of solvent (section 3.1) and pH on the emission spectrum of MSTBA in comparison with that of DSTBA, we can say that the extent of charge transfer is less than that for DSTBA. pKa values with and without sugars are listed in Table 3 and are comparable with those observed for the other stilebene derivatives.
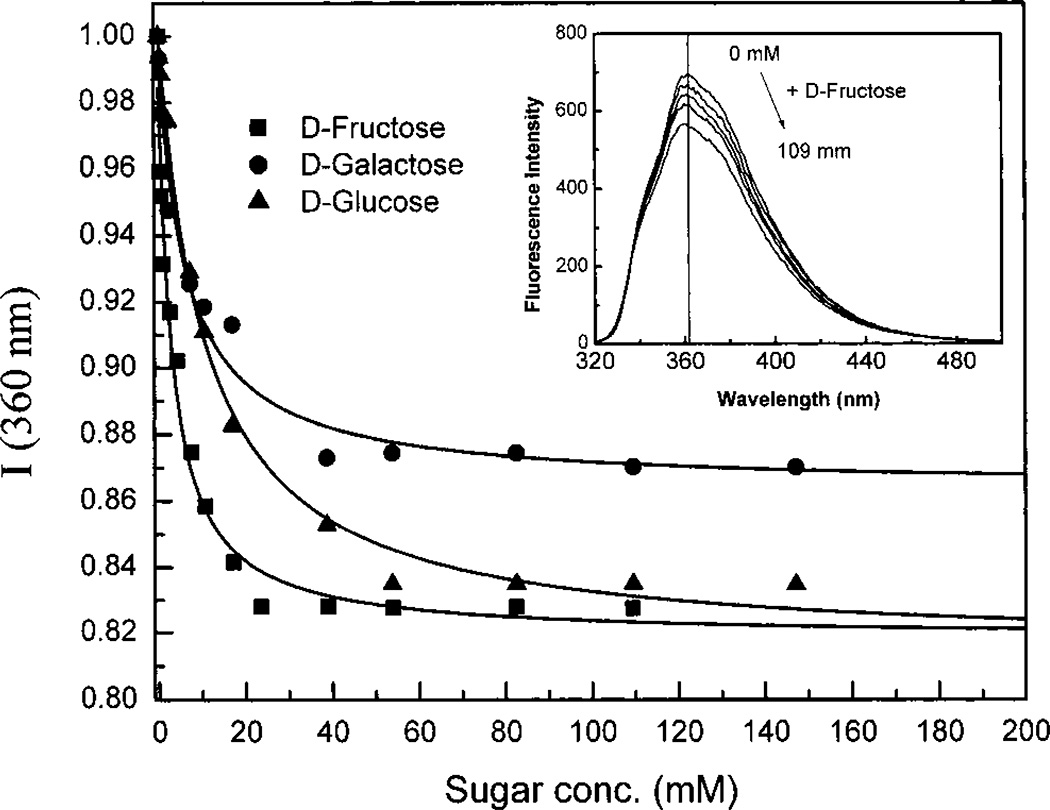
The effect of fructose on the emission spectrum of MSTBA is shown in the inset of Figure 8. As described for the pH, the effect of sugar is smaller for the shift of the band but the intensity change is comparable to what we observed for DSTBA. Titration curves and KD are shown in Figure 8 and Table 3, respectively.
Figure 8.
Titration curves of MSTBA against sugars in phosphate buffer pH 8.0/methanol 2:1 (v/v) at room temperature, λex = 325 nm. Insert: emission spectra changes with the addition of d-fructose.
3.2.4. CSTBA
CSTBA is a different compound than the previous two compounds because it possesses two electron withdrawing groups, the cyano and boronic acid groups. As expected for this kind of compounds, no CT states are observed for the neutral form of the boronic acid group (section 3.1). On the other hand, we can observe a large bathochromic shift, 40 nm, and a decrease of the intensity in the emission spectrum of CSTBA with an increase of pH (Figure 9A). A smaller red shift, 8 nm, is also observed in the absorption spectrum following the increase of pH (results not shown). The shift and the intensity change are very similar but in opposite direction with respect to those observed for DSTBA and MSTBA. We attributed this new red shifted band to an excited CT state present for the anionic form of CSTBA. This suggests that the anionic form of the boronic acid group can act as an electron donor group. Titration curves against pH and pKa values of CSTBA are shown in Figure 9A and Table 3. pKa values are comparable to those observed for the other stilbene derivatives previously presented. Figure 10A shows the effect of addition of fructose on the emission spectrum of CSTBA. The same effects as those observed for the pH are observed upon the addition of sugars. Titration curves against sugars and KD values are presented in Figure 10B and Table 3, respectively.
Figure 9.
(A) pH dependence on the emission spectra of CSTBA without sugar at room temperature, λex = 325 nm. (B) Titration curves of CSTBA with and without sugar.
Figure 10.
(A) Change in the emission spectra of CSTBA after addition of d-fructose, in phosphate buffer pH 8.0/methanol 2:1 (v/v) at room temperature, λex = 335 nm. (B) Titration curves of CSTBA with the different sugars.
3.3. Additional Observations
By comparing the pKa and KD of all stilbene derivatives investigated in this study (Table 3), we can observe a decrease of these values from DSTBA to MSTBA and CSTBA. Only STBA does not follow the trend. As the strength of the donor group increased, we could expect a larger negative partial charge on the boron atom in the ground state. A partial negative charge on the boron would reduce the affinity of the boronic acid group to link with a hydroxyl group. This effect could explain the trend observed for the pKa and the KD values because the methoxy group is a weaker donor and the cyano group is not a donor at all. However, further studies would be necessary to verify this hypothesis.
We also evaluated the effect of pH on the dissociation constant of CSTBA for d-fructose (results not shown). We observed a decrease of the dissociation constant with the increase of pH (3.22, 1.19, and 0.65 mM for pH of 7.0, 7.5, and 8.0, respectively), suggesting that the affinity of the boronic acid group for fructose increases when the pH increases. This could be useful to setting the concentration range of the applicability of a boronic acid sensor.
We also measured the effect of adding fructose on the fluorescence lifetime of the derivatives investigated. For all derivatives, except for MSTBA, a decrease of the mean lifetime was observed (results not shown). For example, the lifetime of DSTBA decreased to 320 ps in the presence of 110 mM of fructose. However, because this change of the lifetime is relatively small, deeper studies would be necessary to link this decrease of lifetime with electronic properties involved in the molecule following the binding with sugars and the formation of the anionic form.
4. Conclusion
The combination of an electron withdrawing and/or donating group and the boronic acid group both directly linked to a fluorophore could lead to the formation of an excited CT state. The neutral form of the boronic acid group acts as an electron withdrawing group, whereas the anionic form could act as an electron donor group. After complexation with a sugar molecule, boronic acid changes from the neutral form to the anionic one and a change in the CT properties of the fluorophore occurs. A shifting and a change in the intensity of the emission bands are then observed. This leads to a new optical and ratiometric approach for the analysis of sugar using fluorescence probes having the boronic acid group. This donor–acceptor combination gives much more optical changes than the presence of only the boronic acid group on a fluorophore opening the perspective to a new class of fluorescence probes for sugars.
Acknowledgments
N.D. is grateful to the National Science and Engineering Research Council of Canada (NSERC) for a postdoctoral fellowship. This work was supported by the Juvenile Diabetes Foundation International, 1-2000-546, with partial support from the NIH National Center for Research Resources, RR-08119.
References and Notes
- 1.Elschenbroich C, Salzer A. Organometallics. New York: VCH; 1989. [Google Scholar]
- 2.Matsumi N, Naka K, Chujo Y. J. Am. Chem. Soc. 1998;120:5112. [Google Scholar]
- 3.Matsumi N, Naka K, Chujo Y. J. Am. Chem. Soc. 1998;120:10776. [Google Scholar]
- 4.Corriu RJ-P, Daforth T, Douglas WE, Guerrero G, Siebert WS. Chem. Commun. 1998:963. [Google Scholar]
- 5.Matsumi N, Naka K, Chujo Y. Macromolecules. 1999;32:4467. [Google Scholar]
- 6.Lee BY, Wang S, Putzer M, Bartholomew GP, Bu X, Bazan GC. J. Am. Chem. Soc. 2000;122:3969. [Google Scholar]
- 7.Lee BY, Bazan GC. J. Am. Chem. Soc. 2000;122:8577. [Google Scholar]
- 8.Sienkiewicz PA, Roberts DC. J. Inorg. Nucl. Chem. 1980;42:1559. [Google Scholar]
- 9.James TD, Samankumara Sandanayake KRA, Shinkai S. Nature. 1995;374:345. [Google Scholar]
- 10.James TD, Samankumara Sandanayake KRA, Iguchi R, Shinkai S. J. Am. Chem. Soc. 1995;117:8982. [Google Scholar]
- 11.Hartley JH, James TD, Ward CJ. J. Chem. Soc. Perkin Trans. 1. 2000:3155. [Google Scholar]
- 12.Lorand JP, Edwards JO. J. Org. Chem. 1959;24:769. [Google Scholar]
- 13.Yoon J, Czarnik AW. J. Am. Chem. Soc. 1992;114:5874. [Google Scholar]
- 14.Suenaga H, Yamamoto H, Shinkai S. Pure Appl. Chem. 1996;68:2179. [Google Scholar]
- 15.James TD, Sandanayake KRAS, Shinkai S. Nature. 1995;374:345. [Google Scholar]
- 16.James TD, Sandanayake KRAS, Iguchi R, Shinkai S. J. Am. Chem. Soc. 1995;117:8982. [Google Scholar]
- 17.James TD, Shinmori H, Takeuchi M, Shinkai S. Chem. Commun. 1996:705. [Google Scholar]
- 18.DiCesare N, Lakowicz JR. Anal. Biochem. 2001 doi: 10.1006/abio.2001.5170. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Takeuchi M, Mizuno T, Shinmori H, Nakashima M, Shinkai S. Tetrahedron. 1996;52:1195. [Google Scholar]
- 20.Sandanayake KRAS, James TD, Shinkai S. Chem. Lett. 1995:503. [Google Scholar]
- 21.Rehm D, Weller A. Israel J. Chem. 1970;8:259. [Google Scholar]
- 22.Valeur B. In: Topics in Fluorescence Spectroscopy. Lakowicz JR, editor. New York: Plenum Press; 1994. pp. 21–50. [Google Scholar]
- 23.Rettig W, Lapouyade R. In: Topics in Fluorescence Spectroscopy. Lakowicz JR, editor. New York: Plenum Press; 1994. pp. 109–150. [Google Scholar]
- 24.Lewis FD, Weigel W. J. Phys. Chem. A. 2000;104:8146. [Google Scholar]
- 25.Létard J-F, Lapouyade R, Rettig W. J. Am. Chem. Soc. 1993;115:2441. [Google Scholar]
- 26.Lapouyade R, Czeschka K, Majenz W, Rettig W, Gilabert E, Rullière C. J. Phys. Chem. 1992;96:9643. [Google Scholar]
- 27.Berlan IB. Handbook of Fluorescence Spectra of Aromatic Molecules. 2nd. New York: Academic Press; pp. 238–356. [Google Scholar]
- 28.Eastman JW. Photochem. Photobiol. 1967;6:55. [Google Scholar]
- 29.Lakowicz JR, Gryczynski I. In: Topics in Fluorescence Spectroscopy. Lakowicz JR, editor. New York: Plenum Press; 1991. pp. 293–335. [Google Scholar]
- 30.Shinmori H, Takeuchi M, Shinkai S. J. Chem. Soc. Perkin Trans. 1996;2:1. [Google Scholar]
- 31.Yam VW-W, Kai AS-F. Chem. Commun. 1998:109. [Google Scholar]
- 32.Mizuno T, Fukumatsu T, Takeuchi M, Shinkai S. J. Chem. Soc. Perkin Trans. 1. 2000:407. [Google Scholar]
- 33.Sananayake KRAS, James TD, Shinkai S. Chem. Lett. 1995:503. [Google Scholar]
- 34.Sananayake KRAS, James TD, Shinkai S. Pure Appl. Chem. 1996;68:1207. [Google Scholar]
- 35.Valeur B. In: Topics in Fluorescence Spectroscopy. Lakowicz JR, editor. New York: Plenum Press; 1994. pp. 21–48. [Google Scholar]
- 36.Poenie M, Chen C-S. In: New Fluorescence Probes for Cell Biology. Herman B, Lemasters JJ, editors. New York: Academic Press; 1993. pp. 1–25. [Google Scholar]
- 37.Smacinski H, Lakowicz JR. In: Topics in Fluorescence Spectroscopy. Lakowicz JR, editor. New York: Plenum Press; 1994. pp. 295–334. [Google Scholar]