Abstract
Drug resistance to anti-cancer agents is a major concern regarding the successful treatment of malignant tumors. Recent studies have suggested that acquired resistance to anti-epidermal growth factor receptor (EGFR) therapies such as cetuximab are in part caused by genetic alterations in patients with oral squamous cell carcinoma (OSCC). However, the molecular mechanisms employed by other complementary pathways that govern resistance remain unclear. In the current study, we performed gene expression profiling combined with extensive molecular validation to explore alternative mechanisms driving cetuximab-resistance in OSCC cells. Among the genes identified, we discovered that a urokinase-type plasminogen activator receptor (uPAR)/integrin β1/Src/FAK signal circuit converges to regulate ERK1/2 phosphorylation and this pathway drives cetuximab-resistance in the absence of EGFR overexpression or acquired EGFR activating mutations. Notably, the polyphenolic phytoalexin resveratrol, inhibited uPAR expression and consequently the signaling molecules ERK1/2 downstream of EGFR thus revealing additive effects on promoting OSCC cetuximab-sensitivity in vitro and in vivo. The current findings indicate that uPAR expression plays a critical role in acquired cetuximab resistance of OSCC and that combination therapy with resveratrol may provide an attractive means for treating these patients.
Subject terms: Oral cancer, Oral cancer
Introduction
Targeted molecular therapies inhibit specific regulators of oncogenic signaling cascades essential for development of malignant tumors1. Considerable evidence indicates that numerous genetic alterations contribute to tumorigenesis of human oral squamous cell carcinomas (OSCCs)2,3. This has prompted researchers to accelerate investigations aimed at creating targeted molecular therapies for this malignant tumor by exploiting therapeutic vurnerabilites in pathways that are dysregulated as a result of these genetic alterations. For example, expectations have changed regarding therapeutic strategies such as monoclonal antibodies and small-molecule kinase inhibitors that target the epidermal growth factor receptor (EGFR). Given that most OSCCs overexpress EGFR4, anti-EGFR antibodies, like cetuximab, are promising candidate therapeutic agents for human OSCCs. However, unlike conventional cisplatin-based chemotherapy, cetuximab does not have sustained effects due to emergence of acquired resistance caused by genetic alterations such as EGFR and KRAS gain-of-function mutations5,6. In addition, as reported by Stein-O’Brien et al.7, other dynamic molecular changes may be associated with drug-resistant phenotypes. Therefore, it is of clinical importance to identify potential novel pathway(s) that mediate this acquired resistance phenotype to achieve better responses to cetuximab. In fact, recent studies have shown that the expression status of a glutamine transporter, solute carrier 1 family member 5 (SLC1A5), in human colorectal cancer patients dictates their response to cetuximab regardless of EGFR/KRAS mutational status, thus highlighting the importance of currently unknown/unrecognized novel molecular pathways driving cetuximab resistance8,9. Moreover, inhibiting SLC1A5 significantly enhanced the inhibitory efficacy of cetuximab, suggesting that unknown molecular mechanism(s) may be involved in other human malignancies such as cetuximab-resistant SCC of the oral cavity.
In the current study, we generated three independent cetuximab-resistant OSCC cell lines that lack known hotspot EGFR/KRAS mutations. We then analyzed these cetuximab-resistant lines relative to their parental cetuximab-sensitive counterparts to search for novel molecular pathway(s) that govern drug resistance and whose targeting confers renewed sensitivity. Herein, we identified a urokinase-type plasminogen activator receptor (uPAR)/integrin β1/Src/FAK signaling circuit as a novel mechanism governing cetuximab-resistance in OSCC cells that can be effectively inhibited by resveratrol (a polyphenolic phytoalexin). These findings reveal an alternative strategy for re-sensitizing the cetuximab-resistance phenotype via blockade of this signaling pathway in OSCC.
Materials and Methods
Reagents
Cetuximab and resveratrol were purchased from Merck KGaA (Darmstadt, Germany). Cell Signaling Technology (Dancers, MA) provided the following antibodies: anti-EGFR (#4267), anti-Phospho-EGFR (#2234), anti-Integrinβ1 (#4706), anti-Src (#2108), anti-Phospho-Src Family (#2101), anti-FAK (#3285), anti-Phospho-FAK (#3283), anti-ERK1/2 (#4695), and anti-Phospho-ERK1/2 (#4370). Santa Cruz Biotechnology, Inc. (Dallas, TX) provided the antibodies against uPAR (sc-10815) and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) (sc-3223).
Cell lines
All human oral SCC-derived cell lines (SAS, Sa3, and HSC-3) were purchased from and authenticated by the Human Science Research Resources Bank or the RIKEN Bio Resource Center and cultured as described previously10,11. All cells used in this study were within 10 passages from thawing, were routinely tested for Mycoplasma contaminations using EZ-PCR Mycoplasma Test Kit (Biological Industries, Cromwell, CT).
To establish cetuximab-resistant cells, the parental cell lines (SAS-P, Sa3-P, and HSC-3-P) were exposed to varying concentrations of cetuximab (10, 20, 50, and 100 μg/mL for 24 hr) as described previously12,13. After the medium was replaced with normal medium for 7 days until the next mitosis, the cells were exposed to double-dosed of cetuximab for 24 hr. This process was performed successively until the final concentration of cetuximab reached 1,800 μg/mL. Genomic DNA was extracted from all cell lines studied, and they were assessed the known mutation status of the EGFR (exon 18 [G719X], exon 19 [E746_A750 deletion], exon 20 [V769_V774 insersions], exon 20 [T790M], and exon 21 [L858R]) and KRAS (codon 12/ 13) genes as described previously14,15.
Gene expression profiling
RNA samples (50 ng) from each cell line, which passed quality control using the NanoDrop ND-1000 spectrophotometer (NanoDrop Technologies, Inc., Wilmington, DE), were analyzed by an Agilent SurePrint G3 Human GE microarray (Agilent Technologies, Santa Clara, CA). These microarray data have been reported in Gene Expression Omnibus (GEO) (https://www.ncbi.nlm.nih.gov/geo/) in under the reference number 114928. The expression intensity values of significantly differentially expressed genes were obtained by a fold-change cutoff greater than 2.0 or less than 0.5 (P < 0.01) and visualized by volcano plots16. Hierarchical clustering then was conducted using the genes that were shown to divide the cell lines into two groups (cetuximab-sensitive and cetuximab-resistant). The genes were considered to be associated significantly with the z-score at a false discovery rate exceeding a two-fold change. The biologic networks and pathway analyses for identified genes were performed using IPA, and the canonical pathways and Ingenuity Tox List tools were overlaid on the networks.
To refine the genes associated with cetuximab-resistance in OSCC, we compared our gene expression data to 3 independent gene expression datasets downloaded from GEO (accession numbers: GSE98812, GSE63916, and GSE32975).
RT-qPCR
Total RNA was prepared using TRIZOL reagent (Invitrogen, Carlsbad, CA). RT-qPCR was performed as described previously17–19, with the following primers designed at the Universal Probe Library Assay Design Center (https://www.roche-applied-science.com/sis/rtpcr/upl/index.jsp?id=uplct_030000): uPAR (forward, 5′-ACACCACCAAATGCAACGA-3′; reverse, 5′-CCCCTTGCAGCTGTAACAC-3′); ARRDC4: (forward, 5′-GGAGGTGGAGTACCTGAACG-3′; reverse, 5′- AAATTCATGTTTTCCAGGCTGT-3′); LEPROT: (forward, 5′-TGTTGTTTCTGCCTTTGGATT-3′; reverse, 5′-GCAGGCTCCCCATTTGAT-3′); PHF15: (forward, 5′-GTTGGTCAGTCGTGTTTTAAAGAG-3′; reverse, 5′-TTCGCCTCTTCTCTTCCATC3′); DDRGK1: (forward, 5′-GTGGGCCTACGCACTCAG-3′; reverse, 5′-CCCGGTCGTCAATCACAC-3′); HLA-A: (forward, 5′-TTGAGAGCCTACCTGGATGG-3′; reverse, 5′-TGGTGGGTCATATGTGTCTTG-3′), and LAMC2: (forward, 5′-CTCAGCCCAACGACTAGACC-3′; reverse, 5′-TCACCTGTTGATTCCCAAGA-3′). The quantified values were normalized to the GAPDH gene, and the results are presented as relative values compared with the controls.
Cellular viability assay
After treatment with cetuximab or resveratrol, the cellular viability was measured by the 3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium (MTS) assay using CellTiter 96®AQueous Assay kit (Promega, Fitchburg, WI). Twenty microliters of the MTS reagent was added directly to the adherent cells, which were incubated at 37 °C for 24 hr. Absorbance then was recorded at 490 nm using a Benchmark Plus Microplate Reader (Bio-Rad, Philadelphia, PA). Each assay was repeated three times independently.
Cellular proliferation assay
To evaluate the difference between cetuximab-resistant cells and the parental cell lines, we analyzed cellular growth. These cells were seeded in 6-cm dishes at a density of 1 × 104 viable cells and cultured for 168 hours and counted every 24 hours. At the indicated time points, the cells were trypsinized and counted in triplicate using a hemocytometer.
Immunoblotting
After the respective treatments, the cells were harvested and washed twice with phosphate buffered saline and centrifuged. Immunoblotting was performed as reported previously20 with each appropriate antibody already mentioned and visualized by exposing the membranes to ChemiDoc XRS Plus system and the signal intensities were quantified using the Image Lab system (Bio-Rad Laboratories)11,21. Densitometric uPAR protein data were normalized to GAPDH protein levels. (Bio-Rad Laboratories).
Transfection with shRNA plasmid
To knock down uPAR expression in the resistance strains (SAS-R, Sa3-R, and HSC-3-R), they were transfected with shRNA targeting (shuPAR) and control shRNA (shMock) (Santa Cruz Biotechnology) using Lipofectamine 3000 (Invitrogen), according to the manufacturers’ instructions. The stable transfectants were isolated in a culture medium containing puromycin (2 µg/ml) (Santa Cruz Biotechnology). The efficiency of uPAR knockdown was assessed by RT-qPCR and immunoblotting.
Murine experiments
Animal handling and all animal experiments followed the international guidelines. Specifically, to investigate whether uPAR expression contributed to cetuximab resistance and the antitumor activity of cetuximab combined with resveratrol, we used the SAS-R and Sa3-R cells for in vivo validation. The cells (1 × 106 cells) were injected subcutaneously into the backs of the female athymic nude mice, the BALB/cAnNCrj-nu/nu strain (Oriental Yeast Co., Ltd., Andover, MA) as described previously22. The mice were subjected to different treatments, with three mice in each group: shMock + DMSO, shMock + cetuximab, shuPAR + DMSO, shuPAR + cetuximab, controls (DMSO), cetuximab, resveratrol, and cetuximab combined with resveratrol. The mice were treated with cetuximab (10 mg/kg intraperitoneally three times weekly) and/or resveratrol (100 mg/kg intraperitoneally daily). The body weight was measured at least every 2 weeks. The excised tissues were fixed in 10% formalin and embedded in paraffin followed by deparaffinization and hydration. The specimens were stained with appropriate antibodies such as anti-uPAR (1:50 dilution), anti-Integrinβ1 (1:250 dilution), anti-ERK1/2 (1:250 dilution), and anti-Phospho-ERK1/2 (1:250 dilution) at 4 °C. As a negative control, triplicate sections were immunostained without exposure to primary antibodies (Supplementary Fig. S4).
Clinical subjects for immunohistochemical analysis (IHC)
Tissue samples from primay tumors and matched normal oral tissues were obtained from patients undergoing cetuximab therapy at the Chiba University Hospital. All patients had given written informed consent and were enrolled in institutional protocols approved by the Chiba University bioethics review committee (reference No. 562 (300)), which was in accordance with the Declaration of Helsinki and Good Clinical Practice. IHC for detecting uPAR was carried out with the same condition as mentioned above. The immunostaining images were quantified by the IHC Profiler (https://sourceforge.net/projects/ihcprofiler/, Source Forge) for IHC score as described previously23 (Supplementary Fig. S5).
Statistical analysis
All data are expressed as the mean ± standard error of the mean. Statistical differences were analyzed by Welch’s t test (two group comparisons) or ANOVA (multiple group comparisons). P < 0.05 was considered significant.
Ethical approval
All protocols involving mice were approved by the ethical committee of the Graduate School of Medicine, Chiba University in accordance with relevant guidelines and regulations.
Results
mRNA profiling to identify genes related to cetuximab resistance in OSCC
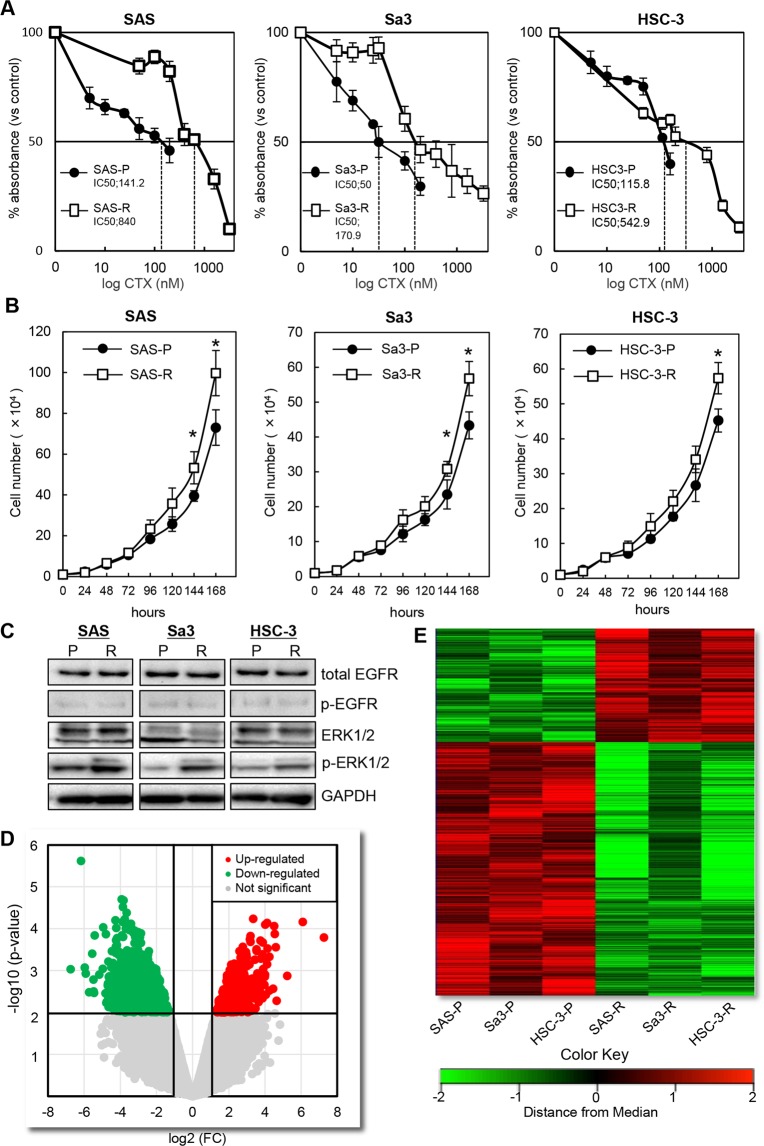
To determine the molecular mechanisms governing acquired resistance to cetuximab, we established three drug resistance cellular models by culturing OSCC-derived cell lines, including SAS, Sa3, and HSC-3, with increasing cetuximab concentrations. While the inhibitory concentration (IC) 50 values for cetuximab in parental cell lines were 141.2 ± 65.4 nM, 50.0 ± 24.1 nM, and 115.8 ± 17.5 nM for the SAS-P, Sa3-P, and HSC-3-P lines, respectively, the IC50 increased to 840.0 ± 91.7 nM, 170.9 ± 33.6 nM, and 542.9 ± 42.9 nM in the resistant cell lines (Fig. 1A). Ninety days later, these cetuximab-resistant cell lines, i.e., SAS-R, Sa3-R, and HSC-3-R, were cultured stably in cetuximab (12 nM)-containing medium, which confirmed a three-fold increase in the IC50 values in all cetuximab-resistant cells generated compared to the parental cells. Cells that were resistant to the anti-proliferative effects of cetuximab displayed an increased proliferative rate compared to untreated controls (Fig. 1B). Acquired resistance to cetuximab targeted therapy has been shown to occur via several mechanisms including EGFR overexpression, altered EGFR degradation, gain-of-function EGFR mutations, and/or gain-of-function mutations in downstream pathway effectors such as KRAS5,6,24. However, Sanger sequencing confirmed that all cell lines lacked any nucleotide variations in EGFR and KRAS regardless of cetuximab sensitivity (Supplementary Fig. S1). Intriguingly, while the cetuximab-resistant SAS-R, Sa3-R, and HSC-3-R cell lines showed no significant change in total EGFR, phosphorylated EGFR (p-EGFR), or total ERK1/2 protein expression, immunoblotting revealed elevated p-ERK1/2 levels in all cetuximab-resistant cell lines (treated with 100 nM of cetuximab) compared to each parental cell line (Fig. 1C). These results suggest that alternative pathway(s) for cetuximab-resistance exist that converge on downstream ERK1/2 signaling.
Figure 1.
Characteristics and mRNA expression profilings for the cetuximab-resistant cells. (A) An MTS assay of the cetuximab-resistant cells and the parental cells in the presence of cetuximab at various concentrations (0–3200 nM) for 24 hr. More than a three-fold increase is seen in the IC50 value in cetuximab-resistant cells compared to parental cells. (B) Proliferation assay of the cetuximab-resistant cells and the parental cells without cetuximab. Cetuximab-resistant cells display significant increases in proliferative rates (*P < 0.05) compared to the parental cells at least after 7 days (168 hours) in culture. All graphs represent mean of three independent experiments ± SEM. (C) Increased phosphorylation of ERK1/2 in the cetuximab-resistant cells with cetuximab (100 nM) by immunoblot analysis. The expression levels of EGFR, phosphorylated EGFR (p-EGFR), and ERK1/2 do not change in any of the cell lines examined. P: parental cells, R: cetuximab-resistant cells. (D) Volcano plots of significant genes with P < 0.01 and more than a two-fold change or less than a 0.5-fold change expressed in 450 up-regulated genes and 992 down-regulated genes. The x-coordinate represents the (log2) fold-change (FC) and y represents the t-statistic or − log10 of the P value. (E) Heat maps of the 450 up-regulated genes or the 992 down-regulated genes in cetuximab-resistant cells. The red transcripts are up-regulated at least two-fold in cetuximab-resistant cells compared with the parental cells, and those in green are similarly down-regulated.
To elucidate the mechanisms responsible for emergence of acquired drug resistance, we performed gene expression analyses on microarray data collected for the parental cell lines relative to their cetuximab-resistant counterparts. Volcano plots show that 450 up-regulated genes and 992 down-regulated genes were significantly changed (P < 0.01) more than two-fold (Fig. 1D). Unsupervised hierarchical clustering of all differentially expressed genes showed clear cetuximab resistance-related gene expression patterns (Fig. 1E). Moreover, based on the common differentially expressed genes with more than a two-fold z-score (Supplementary Table S1), we selected the top 20 up-regulated genes for further validation by reverse transcriptase quantitative polymerase chain reaction (RT-qPCR) and confirmed eight commonly up-regulated genes, including uPAR, ARRDC4, LEPORT, PHF15, DDRGK1, HLA-A, LAMC2, and CD44 (Supplementary Fig. S3). Notably, 12 genes including uPAR formed part of a common overlapping cetuximab-resistance gene signature when we cross-examined several independent gene expression datasets by Hatakeyama et al.14 and performed this comparison with our up-regulated gene list (900 genes) relative to parental cetuximab-sensitive cells (Supplementary Fig. S2 and Table S1). Interestingly, uPAR is the only gene within this 12-gene signature previously shown to regulate an EGFR/p-ERK1/2-related signal pathway25, therefore, we ultimately selected uPAR for further in vitro and in vivo validation based on these data and Ingenuity Pathway Analysis (IPA) analysis.
Effects of uPAR inhibition in vitro
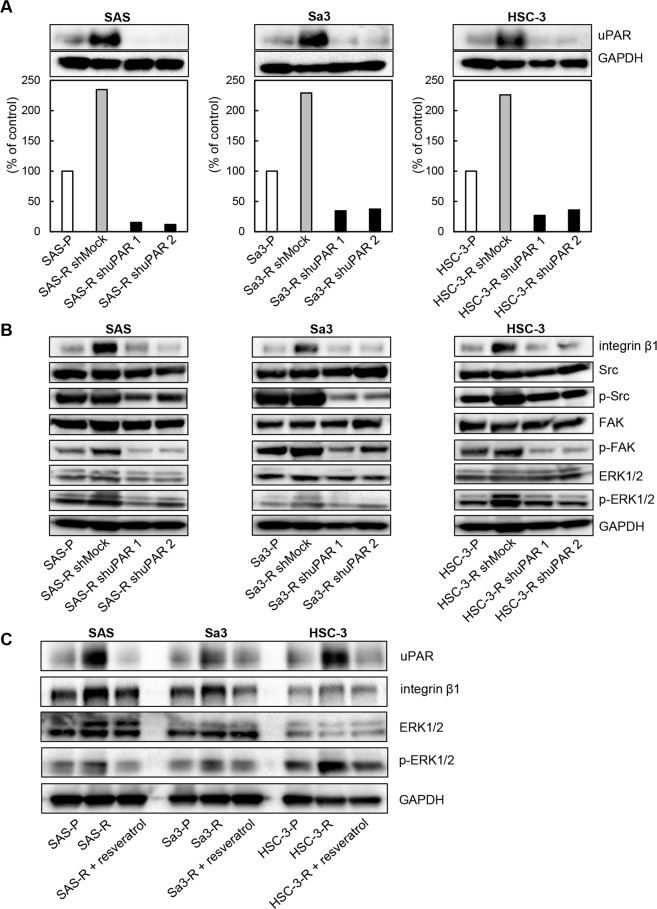
The observed significant increase in uPAR expression suggests a direct role in mediating cetuximab resistance. To test whether acquired resistance to cetuximab requires uPAR, we performed genetic knockdown using validated shRNA molecules specific to uPAR (Fig. 2A). Immunoblot analysis showed that targeted silencing by shuPAR knockdown significantly down-regulated uPAR levels (Fig. 2A). Moreover, downstream signaling pathways, such as integrin β1, p-Src, p-FAK, and p-ERK1/2 were all reduced in shuPAR treated but not in the shMock-treated cetuximab-resistant cell lines (Fig. 2B).
Figure 2.
Effects of uPAR in cetuximab-resistance cells in vitro. (A) Establishment of uPAR knockdown cells. Expression levels of uPAR in the cetuximab-resistant shuPAR cells are down-regulated compared to shMock cells. (B) The expression of uPAR-related genes in the cells treated with cetuximab (100 nM) for 24 hr. While integrin β1, p-Src, p-FAK, and p-ERK1/2 expression levels are up-regulation in the cetuximab-resistant shMock cells compared with the parental cells, and they are down-regulated in the shuPAR cells compared to the shMock cells. The total Src, FAK, and ERK1/2 protein levels remain unchanged in all cell lines examined. (C) The effects of resveratrol on the cetuximab-resistant cells. While uPAR, integrin β1, and p-ERK1/2 are down-regulated significantly in the presence of cetuximab (100 nM)/resveratrol (20 µM) in cetuximab-resistant cells, the steady-state level of total ERK remains constant in all cells under all conditions tested.
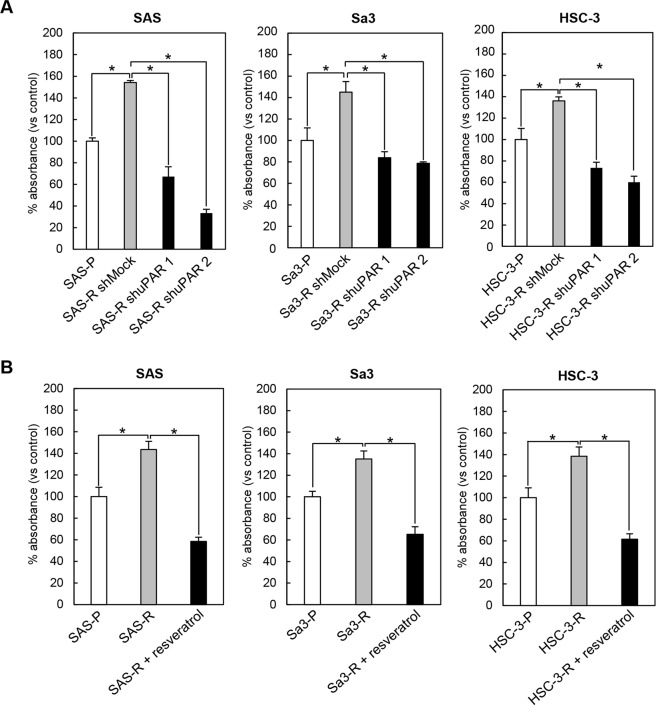
To investigate the effect of uPAR knockdown via a pharmacologic agent on cetuximab-resistant OSCC cells, we chose to focus on resveratrol (3,4′,5-trihydroxystilbene), a naturally occurring compound with anti-cancer effects26–31 found in a variety of fruits and plants32 that has previously been shown to potently downregulate uPAR expression, as a therapeutic strategy for targeting cetuximab-resistant OSCC cells. Similar to the shuPAR condition, resveratrol-treated cetuximab-resistant cell lines expressed similar levels of total ERK1/2 compared to the parental cell lines, however, resveratrol treatment lead to a significant decrease in pERK1/2 levels and a robust downregulation of integrin β1 and uPAR was observed (Fig. 2C). We also found that both shRNA knockdown of uPAR or resveratrol treatment lead to a significant reduction of cellular viability in all cetuximab-resistant cell lines examined (Fig. 3A,B). There was no additional effect of combining shRNA knockdown of uPAR and resveratrol on cellular viability and ERK1/2 phosphorylation of SAS-R against cetuximab (Supplementary Fig. S6). This data suggest that the uPAR-dependent pathway is a major pathway that regulates resensitizing the cetuximab-resistant phenotype by resveratrol in SAS-R cells.
Figure 3.
Analysis of oral SCC cells viability under the conditions of (A) shuPAR and (B) resveratrol treatment. The cellular viability measured by the MTS assay shows a significant reduction in cellular viability in the cetuximab-resistant shuPAR cells. Absorbance was measured at 490 nm. Data are expressed as the mean and standard deviation. All graphs represent mean of three independent experiments ± SEM. Asterisks indicate a significance difference (*P < 0.05).
Effects of uPAR inhibition in vivo
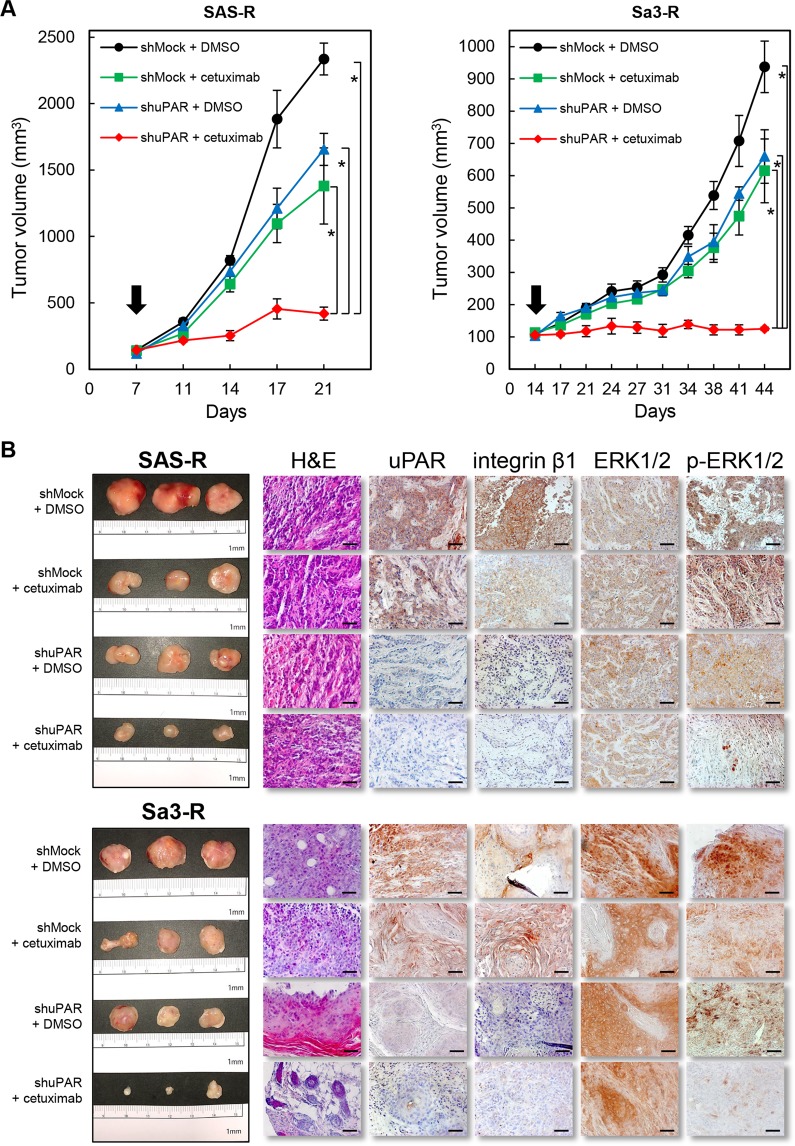
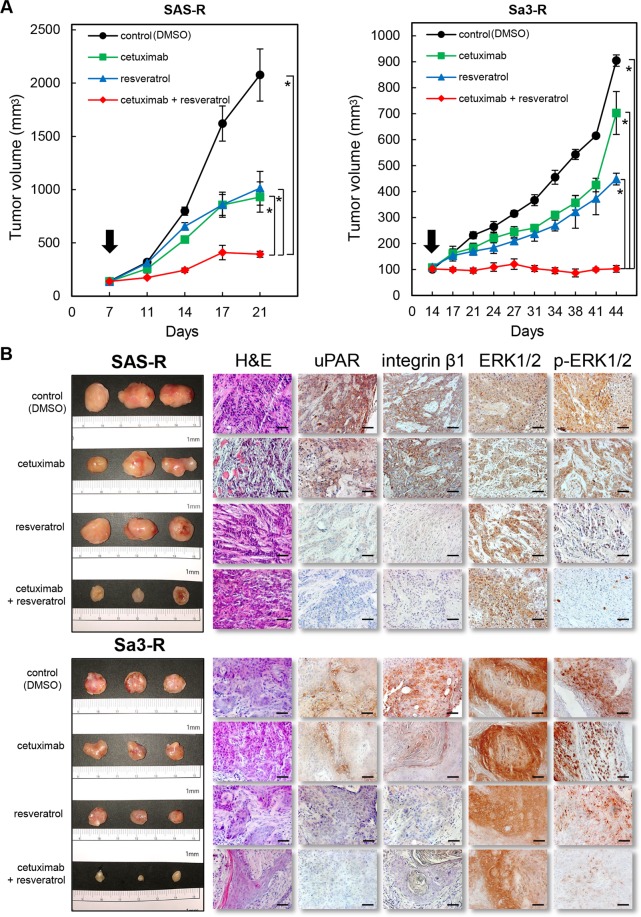
We next examined the effects of uPAR knockdown in combination with EGFR inhibition on tumor growth in vivo. As expected, conditions that combined cetuximab treatment with shuPAR displayed dramatic and synergistic antitumor activity, with growth inhibition levels of about 82.0% of the SAS-R cells at day 21 and 86.6% of the Sa3-R cells at day 44 (Fig. 4A). However, we did not see adequate antitumor growth inhibition in the SAS-R and Sa3-R cells treated with cetuximab or shuPAR alone (Fig. 4A). Immunohistochemical analyses showed dramatic differences in the expression status of uPAR, integrin β1, and p-ERK1/2 of the resected xenograft tumors. More importantly, the combination of cetuximab and shuPAR was more efficient than cetuximab alone or shuPAR alone in the down-regulation of protein expression levels of integrin β1 and uPAR and displayed a robust downregulation of pERK1/2 levels without affecting total ERK1/2 protien levels. (Fig. 4B, Supplementaly Fig. S5A). Consistently, and similar to the effect of uPAR depletion with shRNA in vivo, resveratrol significantly suppressed tumor growth and down-regulated proteins downstream of uPAR (Fig. 5A,B, and Supplementaly Fig. S5B). We also found that the average body weight of the resveratrol-treated mice never dropped lower than that of the control group at any time point (Supplementary Fig. S7).
Figure 4.
The effects of uPAR in cetuximab-resistant cells in vivo. (A) The tumor volume was calculated twice weekly after the injection. when the volume of the transplantation tumor reached 100 mm3 (arrows), the mice were treated with different drugs as indicated below (n = 3 per group). Significant antitumor growth activity by uPAR knockdown (shuPAR) combined with cetuximab is evident in SAS-R and Sa3-R cell xenografts. *P < 0.05. (B) The tumoral tissues from the shMock + DMSO, shMock + cetuximab, shuPAR + DMSO, and shuPAR + cetuximab were fixed in 10% formalin, and paraffin sections were prepared for hematoxylin and eosin (H&E) staining and immunohistochemistry (uPAR, integrin β1, ERK1/2, and p-ERK1/2). The scale bar indicates 50 μm.
Figure 5.
Effects of resveratrol in cetuximab-resistant cells in vivo. (A) The tumor volume was calculated twice weekly after the injection. when the volume of the transplantation tumor reached 100 mm3 (arrows), the mice were treated with different drugs as indicated below (n = 3 per group). Significant antitumor growth activity by resveratrol combined with cetuximab is evident in SAS-R and Sa3-R cell xenografts. *P < 0.05. (B) The tumoral tissues from the controls (DMSO), cetuximab, resveratrol, and cetuximab combined with resveratrol were fixed in 10% formalin, and paraffin sections were prepared for hematoxylin and eosin (H&E) staining and immunohistochemistry (uPAR, integrin β1, ERK1/2, and p-ERK1/2). Scale bars indicate 50 μm.
The uPAR expression level in OSCC tissues with or without cetuximab resistance
We next performed IHC to assess uPAR protein levels in primary OSCCs obtained from cetuximab treated patients. We observed significant and intense uPAR immunoreactivity in all human oral SCC tissues examined compared to the corresponding normal tissues. More importantly, uPAR protein expression levels in cetuximab-resistant OSCCs was much higher than that for cetuximab-sensitive cases (Supplementary Fig. S8). Collectively, these data indicate that up-regulating uPAR gene expression confers acquired cetuximab resistance in OSCC and that the uPAR inhibitor resveratrol is capable of restoring drug sensitivity to cetuximab.
Discussion
Pharmacologic targeting of EGFR with the specific monoclonal antibody cetuximab is now a protagonist of molecular targeted therapy for recurrent/metastatic SCCs of the oral cavity33. However, its clinical use is limited due to acquired drug resistance. A few previous studies have shown that activation of the EGFR-dependent pathways is regarded as the main cause of drug resistance9,34,35.
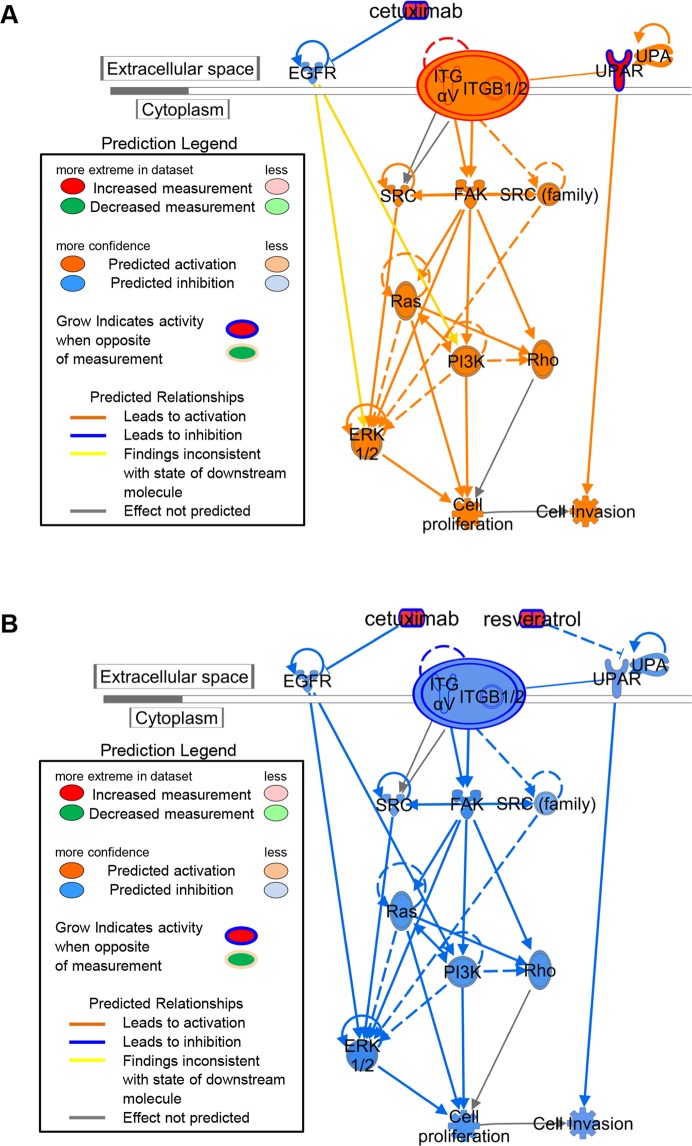
In the current study, we identified gene expression changes common to cetuximab-resistant cell lines and validated select up-regulated targets. Among 1,448 differentially expressed genes identified, we found that the expression levels of a small subset of candidate genes increased significantly in the cetuximab-resistant cells (Supplementary Table S1). Moreover, coupled with available gene expression data published by Hatakeyama et al.14, we have characterized uPAR as the only molecule reported to be a potential regulator of the EGFR-dependent signals25. Given that uPAR up-regulation has been detected in the primary OSCC tumors from patients with cetuximab resistance (Supplementary Fig. S8), we sought to test the hypothesis that cetuximab-resistance may be associated with uPAR and its downstream signal pathways. The IPA analysis (Fig. 6A) indicated that uPAR regulates an integrin β1 signaling axis that converges with activated EGFR pathways at the ERK1/2 siganling node. Our results reveal that both signals are involved in the acquired cetuximab-resistance mechanism, and at the same time, the effect of cetuximab is expected to be enhanced if these signals are controlled (Fig. 6B). Indeed, sphere-forming head and neck SCC cells possibly resistant to molecular targeting were insensitive to both anti-integrin β1 and anti-EGFR inhibitory antibodies36. In human pancreatic ductal adenocarcinoma cells, active integrin β1 is associated closely with primary resistance to cetuximab37. In addition, owing to the frequent aberrant expression levels of integrin β1 and EGFR in human cancer, previous studies have indicated that the integrin β1-EGFR signal pathway is the targeted option that can efficiently reduce tumor radioresistance38–40. Although it is apparent that multiple strategies for cetuximab-resistance have been assumed in human cancers, the above-mentioned observations and our results showed that active uPAR/integrin β1 signaling in cetuximab resistant OSCC cell lines (Figs 2B and 6A) supports our hypothesis that this pathway may play an alternative role in OSCC refractory to cetuximab treatment.
Figure 6.
Ingenuity Pathway Analysis (IPA) of the canonical pathways for the cetuximab-resistant cells based on (A) increased expression of uPAR and (B) the predicted signaling pathway for uPAR inhibition by resveratrol leading to suppression of cetuximab-resistance in cancer cells. The pathways analyses were generated through the use of IPA (QIAGEN Inc., https://www.qiagenbioinformatics.com/products/ingenuity-pathway-analysis).
The next step in translational research is identification of highly selective chemical inhibitors for uPAR in cetuximab-resistant cell lines. Similar to the shuPAR effect, the combination of resveratrol and cetuximab led to almost complete suppression of tumor growth in mice resistant to cetuximab (SAS-R, Sa3-R) with significant down-regulation of the uPAR/integrin β1 signaling pathway (Figs 5A,B and 6B). Resveratrol, a multifunctional natural compound, has a variety of beneficial biologic activities in metabolic and neurodegenerative diseases and cardioprotection41–43.
Intriguingly, recent accumulated evidence shows that resveratrol inhibits specific human carcinogenesis44. Considering the impact of this anti-oncogenic activation, more concerted efforts are required toward finding a chemosensitization effect for resveratrol. Previous studies have reported a chemosensitizing effect of resveratrol in combined treatments with several anti-cancer drugs, such as docetaxel, cisplatin, 5-fluorouracyl, and oxaliplatin45–49. It is noteworthy that resveratrol induces down-regulation of uPAR and exerts an anti-proliferative effect on malignant tumor cells27,28. The current study found that uPAR was highly expressed in the resistant cell lines compared to the parental strains (Fig. 2A, and Supplementaly Table S1), suggesting that resveratrol may be more effective for treating patients refractory to cetuximab. Importantly, the combination of resveratrol and cetuximab significantly inhibited the growth of cetuximab-resistant tumor xenografts in nude mice without inducing systemic toxicity compared to resveratrol or cetuximab alone, and lead to a significant down-regulation of uPAR/integrinβ1/p-ERK1/2 (Fig. 5A,B), indicating that this combination may inhibit tumor growth in mice by suppressing two independent but convergent pathways (EGFR-dependent and uPAR-dependent pathways) simultaneously (Fig. 6B). Moreover, these findings suggest that the combination of resveratrol and cetuximab will be generalizable to all OSCC that acquire cetuximab resistance, either by EGFR overexpression or through activating mutations, since the uPAR forms part of a common downstream pathway that regulates ERK1/2 signaling. While no data exist for cetuximab-resistant OSCC cell lines that harbor mutant EGFR/KRAS, Hatakeyama et al.14 reported up-regulation of uPAR in cetuximab-resistant SCC1 cells that corresponds with increased phosphorylated EGFR in the presence of cetuximab.
Based on our findings and the canonical pathways outlined in Fig. 6, we speculate that cetuximab-resistance is mediated by uPAR upregulation regardless of EGFR mutational status. Thus, resveratrol may be effective for both EGFR mutant and wild-type OSCC cells. In fact, considerable evidence has shown widespread overexpression of uPAR in many cancers from disparate anatomic sites including the colon, lung, liver, breast, prostate, pancreas, kidney, bladder, and thyroid50. Therefore, this combination therapy might be expanded for use in additional other human malignancies that are refractory to anti-EGFR therapies.
Consequently, our preclinical in vitro and in vivo data may provide insights into additional and alternative treatments for cetuximab-resistant OSCC. Although the sample size for the clinical tumor tissues is relatively small, it will be important to design clinical studies to assess the prognostic value of increasing uPAR expression in patients with OSCC based on the status of uPAR expression in primary tumors (Supplementary Fig. S8), since cetuximab-treated OSCC patients have traditionally been divided into two groups (i.e., those with and without cetuximab resistance). Finally, future work focusing on combined treatment using an inhibitor of uPAR expression combined with cetuximab is needed to advance our understanding of drug resistance for other EGFR-targeted drugs, such as panitumumab, erlotinib, and gefitinib.
Supplementary information
Acknowledgements
This study was supported by a Grant-in-Aid for Scientific Research (B) from The Ministry of Education, Culture, Sports, Science and Technology (MEXT), Japan (15H05039 to K. Ogawara), and in part by the Dental Foundation of North Carolina and by a UNC Lineberger Tier 3 Developmental Award (to A.L. Amelio).
Author Contributions
K. Uzawa, A.L. Amelio, Y. Takiguchi, M. Yamauchi and H. Tanzawa designed the experiments and wrote the manuscript, K. Uzawa, A. Kasamatsu, M. Sunohara, M. Fukamachi, Y. Sawai, Y. Toeda, K. Eizuka, F. Hayashi and I. Kase-Kato performed experiments. K. Koike, D. Nakashima, K. Ogawara Y. Sakamoto-Endo and M. Shiiba acquired the data. K. Uzawa, A.L. Amelio, Y. Takiguchi, M. Yamauchi and H. Tanzawa supervised the study. All authors reviewed, edited and approved the final version of manuscript.
Data Availability
All data generated or analyzed during this study are included in this published article. The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Competing Interests
The authors declare no competing interests.
Footnotes
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Katsuhiro Uzawa, Email: uzawak@faculty.chiba-u.jp.
Antonio L. Amelio, alamelio@email.unc.edu
Supplementary information
Supplementary information accompanies this paper at 10.1038/s41598-019-48717-w.
References
- 1.Sawyers Charles. Targeted cancer therapy. Nature. 2004;432(7015):294–297. doi: 10.1038/nature03095. [DOI] [PubMed] [Google Scholar]
- 2.Pezzuto Francesca, Buonaguro Luigi, Caponigro Francesco, Ionna Franco, Starita Noemy, Annunziata Clorinda, Buonaguro Franco M., Tornesello Maria Lina. Update on Head and Neck Cancer: Current Knowledge on Epidemiology, Risk Factors, Molecular Features and Novel Therapies. Oncology. 2015;89(3):125–136. doi: 10.1159/000381717. [DOI] [PubMed] [Google Scholar]
- 3.Palka Kevin T., Slebos Robbert J., Chung Christine H. Update on Molecular Diagnostic Tests in Head and Neck Cancer. Seminars in Oncology. 2008;35(3):198–210. doi: 10.1053/j.seminoncol.2008.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Grandis, J. R., Melhem, M. F., Barnes, E. L. & Tweardy, D. J. Quantitative immunohistochemical analysis of transforming growth factor- α and epidermal growth factor receptor in patients with squamous cell carcinoma of the head and neck. Cancer, https://doi.org/10.1002/(SICI)1097-0142(19960915)78:6<1284::AID-CNCR17>3.0.CO;2-X (1996). [DOI] [PubMed]
- 5.Boeckx, C. et al. Mutation analysis of genes in the EGFR pathway in Head and Neck cancer patients: Implications for anti-EGFR treatment response. BMC Research Notes, 10.1186/1756-0500-7-337 (2014). [DOI] [PMC free article] [PubMed]
- 6.Janjigian Y. Y., Smit E. F., Groen H. J. M., Horn L., Gettinger S., Camidge D. R., Riely G. J., Wang B., Fu Y., Chand V. K., Miller V. A., Pao W. Dual Inhibition of EGFR with Afatinib and Cetuximab in Kinase Inhibitor-Resistant EGFR-Mutant Lung Cancer with and without T790M Mutations. Cancer Discovery. 2014;4(9):1036–1045. doi: 10.1158/2159-8290.CD-14-0326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Stein-O’Brien, G. et al. Integrated time course omics analysis distinguishes immediate therapeutic response from acquired resistance. Genome Medicine, 10.1186/s13073-018-0545-2 (2018). [DOI] [PMC free article] [PubMed]
- 8.Passiglia, F. et al. New findings on primary and acquired resistance to anti-EGFR therapy in metastatic colorectal cancer: do all roads lead to RAS? Oncotarget, 10.18632/oncotarget.4959 (2015). [DOI] [PMC free article] [PubMed]
- 9.Ma Huanrong, Wu Zhenzhen, Peng Jianjun, Li Yang, Huang Hongxiang, Liao Yi, Zhou Minyu, Sun Li, Huang Na, Shi Min, Bin Jianping, Liao Yulin, Rao Jinjun, Wang Lin, Liao Wangjun. Inhibition of SLC1A5 sensitizes colorectal cancer to cetuximab. International Journal of Cancer. 2018;142(12):2578–2588. doi: 10.1002/ijc.31274. [DOI] [PubMed] [Google Scholar]
- 10.Ishigami T, Uzawa K, Fushimi K, Saito K, Kato Y, Nakashima D, Higo M, Kouzu Y, Bukawa H, Kawata T, Ito H, Tanzawa H. Inhibition of ICAM2 induces radiosensitisation in oral squamous cell carcinoma cells. British Journal of Cancer. 2008;98(8):1357–1365. doi: 10.1038/sj.bjc.6604290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Saito T, et al. Semaphorin7A promotion of tumoral growth and metastasis in human oral cancer by regulation of g1 cell cycle and matrix metalloproteases: Possible contribution to tumoral angiogenesis. PLoS ONE. 2015;10:1–20. doi: 10.1371/journal.pone.0137923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Zuo Qiang, Shi Min, Li Libo, Chen Jinzhang, Luo Rongcheng. Development of cetuximab-resistant human nasopharyngeal carcinoma cell lines and mechanisms of drug resistance. Biomedicine & Pharmacotherapy. 2010;64(8):550–558. doi: 10.1016/j.biopha.2010.03.003. [DOI] [PubMed] [Google Scholar]
- 13.Boeckx, C. et al. Establishment and characterization of cetuximab resistant head and neck squamous cell carcinoma cell lines: Focus on the contribution of the AP-1 transcription factor. American Journal of Cancer Research (2015). [PMC free article] [PubMed]
- 14.Hatakeyama Hiromitsu, Cheng Haixia, Wirth Pamela, Counsell Ashley, Marcrom Samuel R., Wood Carey Burton, Pohlmann Paula R., Gilbert Jill, Murphy Barbara, Yarbrough Wendell G., Wheeler Deric L., Harari Paul M., Guo Yan, Shyr Yu, Slebos Robbert J., Chung Christine H. Regulation of Heparin-Binding EGF-Like Growth Factor by miR-212 and Acquired Cetuximab-Resistance in Head and Neck Squamous Cell Carcinoma. PLoS ONE. 2010;5(9):e12702. doi: 10.1371/journal.pone.0012702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Poehlmann Angela, Kuester Doerthe, Meyer Frank, Lippert Hans, Roessner Albert, Schneider-Stock Regine. K-ras mutation detection in colorectal cancer using the Pyrosequencing technique. Pathology - Research and Practice. 2007;203(7):489–497. doi: 10.1016/j.prp.2007.06.001. [DOI] [PubMed] [Google Scholar]
- 16.Kulkarni Kirti K., Bankar Kiran Gopinath, Shukla Rohit Nandan, Das Chandrima, Banerjee Amrita, Dasgupta Dipak, Vasudevan Madavan. Global gene expression profiling data analysis reveals key gene families and biological processes inhibited by Mithramycin in sarcoma cell lines. Genomics Data. 2015;3:8–14. doi: 10.1016/j.gdata.2014.11.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Minakawa Yasuyuki, Kasamatsu Atsushi, Koike Hirofumi, Higo Morihiro, Nakashima Dai, Kouzu Yukinao, Sakamoto Yosuke, Ogawara Katsunori, Shiiba Masashi, Tanzawa Hideki, Uzawa Katsuhiro. Kinesin Family member 4A: A Potential Predictor for Progression of Human Oral Cancer. PLoS ONE. 2013;8(12):e85951. doi: 10.1371/journal.pone.0085951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Baba Takao, Sakamoto Yosuke, Kasamatsu Atsushi, Minakawa Yasuyuki, Yokota Satoshi, Higo Morihiro, Yokoe Hidetaka, Ogawara Katsunori, Shiiba Masashi, Tanzawa Hideki, Uzawa Katsuhiro. Persephin: A potential key component in human oral cancer progression through the RET receptor tyrosine kinase-mitogen-activated protein kinase signaling pathway. Molecular Carcinogenesis. 2013;54(8):608–617. doi: 10.1002/mc.22127. [DOI] [PubMed] [Google Scholar]
- 19.Koyama Tomoyoshi, Ogawara Katsunori, Kasamatsu Atsushi, Okamoto Atsushi, Kasama Hiroki, Minakawa Yasuyuki, Shimada Ken, Yokoe Hidetaka, Shiiba Masashi, Tanzawa Hideki, Uzawa Katsuhiro. ANGPTL3 is a novel biomarker as it activates ERK/MAPK pathway in oral cancer. Cancer Medicine. 2015;4(5):759–769. doi: 10.1002/cam4.418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Uzawa K, Ishigami T, Fushimi K, Kawata T, Shinozuka K, Kasamatsu A, Sakamoto Y, Ogawara K, Shiiba M, Bukawa H, Ito H, Tanzawa H. Targeting fibroblast growth factor receptor 3 enhances radiosensitivity in human squamous cancer cells. Oncogene. 2011;30(43):4447–4452. doi: 10.1038/onc.2011.159. [DOI] [PubMed] [Google Scholar]
- 21.Kimura Y, et al. ARNT2 regulates tumoral growth in oral squamous cell carcinoma. Journal of Cancer. 2016;7:702–710. doi: 10.7150/jca.14208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Sawai Yuki, Kasamatsu Atsushi, Nakashima Dai, Fushimi Kazuaki, Kasama Hiroki, Iyoda Manabu, Kouzu Yukinao, Shiiba Masashi, Tanzawa Hideki, Uzawa Katsuhiro. Critical role of deoxynucleotidyl transferase terminal interacting protein 1 in oral cancer. Laboratory Investigation. 2018;98(8):980–988. doi: 10.1038/s41374-018-0070-3. [DOI] [PubMed] [Google Scholar]
- 23.Yamamoto Jun-ichiro, Kasamatsu Atsushi, Okubo Yasuhiko, Nakashima Dai, Fushimi Kazuaki, Minakawa Yasuyuki, Kasama Hiroki, Shiiba Masashi, Tanzawa Hideki, Uzawa Katsuhiro. Evaluation of tryptophan-aspartic acid repeat-containing protein 34 as a novel tumor-suppressor molecule in human oral cancer. Biochemical and Biophysical Research Communications. 2018;495(4):2469–2474. doi: 10.1016/j.bbrc.2017.12.138. [DOI] [PubMed] [Google Scholar]
- 24.Sforza Vincenzo, Martinelli Erika, Ciardiello Fortunato, Gambardella Valentina, Napolitano Stefania, Martini Giulia, della Corte Carminia, Cardone Claudia, Ferrara Marianna L, Reginelli Alfonso, Liguori Giuseppina, Belli Giulio, Troiani Teresa. Mechanisms of resistance to anti-epidermal growth factor receptor inhibitors in metastatic colorectal cancer. World Journal of Gastroenterology. 2016;22(28):6345. doi: 10.3748/wjg.v22.i28.6345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Guerrero J, Santibañez JF, González A, Martínez J. EGF receptor transactivation by urokinase receptor stimulus through a mechanism involving Src and matrix metalloproteinases. Experimental Cell Research. 2004 doi: 10.1016/j.yexcr.2003.08.011. [DOI] [PubMed] [Google Scholar]
- 26.Menicacci Beatrice, Margheri Francesca, Laurenzana Anna, Chillà Anastasia, Del Rosso Mario, Giovannelli Lisa, Fibbi Gabriella, Mocali Alessandra. Chronic Resveratrol Treatment Reduces the Pro-angiogenic Effect of Human Fibroblast “Senescent-Associated Secretory Phenotype” on Endothelial Colony-Forming Cells: The Role of IL8. The Journals of Gerontology: Series A. 2018;74(5):625–633. doi: 10.1093/gerona/gly175. [DOI] [PubMed] [Google Scholar]
- 27.Ryu J, et al. Resveratrol reduces TNF-α-induced U373MG human glioma cell invasion through regulating NF-κB activation and uPA/uPAR expression. Anticancer Research. 2011;31:4223–4230. [PubMed] [Google Scholar]
- 28.Menicacci Beatrice, Laurenzana Anna, Chillà Anastasia, Margheri Francesca, Peppicelli Silvia, Tanganelli Elisabetta, Fibbi Gabriella, Giovannelli Lisa, Del Rosso Mario, Mocali Alessandra. Chronic Resveratrol Treatment Inhibits MRC5 Fibroblast SASP-Related Protumoral Effects on Melanoma Cells. The Journals of Gerontology: Series A. 2017;72(9):1187–1195. doi: 10.1093/gerona/glw336. [DOI] [PubMed] [Google Scholar]
- 29.Baur Joseph A., Sinclair David A. Therapeutic potential of resveratrol: the in vivo evidence. Nature Reviews Drug Discovery. 2006;5(6):493–506. doi: 10.1038/nrd2060. [DOI] [PubMed] [Google Scholar]
- 30.Delmas Dominique, Lancon Allan, Colin Didier, Jannin Brigitte, Latruffe Norbert. Resveratrol as a Chemopreventive Agent: A Promising Molecule for Fighting Cancer. Current Drug Targets. 2006;7(4):423–442. doi: 10.2174/138945006776359331. [DOI] [PubMed] [Google Scholar]
- 31.Hudson Tamaro S., Hartle Diane K., Hursting Stephen D., Nunez Nomeli P., Wang Thomas T.Y., Young Heather A., Arany Praveen, Green Jeffrey E. Inhibition of Prostate Cancer Growth by Muscadine Grape Skin Extract and Resveratrol through Distinct Mechanisms. Cancer Research. 2007;67(17):8396–8405. doi: 10.1158/0008-5472.CAN-06-4069. [DOI] [PubMed] [Google Scholar]
- 32.Shakibaei Mehdi, Harikumar Kuzhuvelil B., Aggarwal Bharat B. Resveratrol addiction: To die or not to die. Molecular Nutrition & Food Research. 2009;53(1):115–128. doi: 10.1002/mnfr.200800148. [DOI] [PubMed] [Google Scholar]
- 33.Licitra L, et al. A randomized, phase 2 study of cetuximab plus cisplatin with or without paclitaxel for the first-line treatment of patients with recurrent and/or metastatic squamous cell carcinoma of the head and neck. Annals of Oncology. 2017;28:2820–2826. doi: 10.1093/annonc/mdx439. [DOI] [PubMed] [Google Scholar]
- 34.Wheeler Deric L., Dunn Emily F., Harari Paul M. Understanding resistance to EGFR inhibitors—impact on future treatment strategies. Nature Reviews Clinical Oncology. 2010;7(9):493–507. doi: 10.1038/nrclinonc.2010.97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Wang D, et al. HER3 targeting sensitizes HNSCC to cetuximab by reducing HER3 activity and HER2/HER3 dimerization: Evidence from cell line and patient-derived xenograft models. Clinical Cancer Research. 2017;23:677–686. doi: 10.1158/1078-0432.CCR-16-0558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Zscheppang Katja, Kurth Ina, Wachtel Nicole, Dubrovska Anna, Kunz-Schughart Leoni A., Cordes Nils. Efficacy of Beta1 Integrin and EGFR Targeting in Sphere-Forming Human Head and Neck Cancer Cells. Journal of Cancer. 2016;7(6):736–745. doi: 10.7150/jca.14232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Kim Y-J, Jung K, Baek D-S, Hong S-S, Kim Y-S. Co-targeting of EGF receptor and neuropilin-1 overcomes cetuximab resistance in pancreatic ductal adenocarcinoma with integrin β1-driven Src-Akt bypass signaling. Oncogene. 2016;36(18):2543–2552. doi: 10.1038/onc.2016.407. [DOI] [PubMed] [Google Scholar]
- 38.Eke, I. et al. Simultaneous β1 integrin-EGFR targeting and radiosensitization of human head and neck cancer. Journal of the National Cancer Institute, 10.1093/jnci/dju419 (2015). [DOI] [PubMed]
- 39.Klapproth, E. et al. Whole exome sequencing identifies mTOR and KEAP1 as potential targets for radiosensitization of HNSCC cells refractory to EGFR and Beta1 integrin inhibition. Oncotarget, 10.18632/oncotarget.24266 (2018). [DOI] [PMC free article] [PubMed]
- 40.Poschau Mandy, Dickreuter Ellen, Singh-Müller Jenny, Zscheppang Katja, Eke Iris, Liersch Torsten, Cordes Nils. EGFR and β1-integrin targeting differentially affect colorectal carcinoma cell radiosensitivity and invasion. Radiotherapy and Oncology. 2015;116(3):510–516. doi: 10.1016/j.radonc.2015.06.005. [DOI] [PubMed] [Google Scholar]
- 41.Jara Paola, Spies Johana, Cárcamo Constanza, Arancibia Yennyfer, Vargas Gabriela, Martin Carolina, Salas Mónica, Otth Carola, Zambrano Angara. The Effect of Resveratrol on Cell Viability in the Burkitt’s Lymphoma Cell Line Ramos. Molecules. 2017;23(1):14. doi: 10.3390/molecules23010014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Kulkarni SS, Cantó C. The molecular targets of resveratrol. Biochimica et Biophysica Acta - Molecular Basis of Disease. 2015 doi: 10.1016/j.bbadis.2014.10.005. [DOI] [PubMed] [Google Scholar]
- 43.Sun Albert Y., Wang Qun, Simonyi Agnes, Sun Grace Y. Resveratrol as a Therapeutic Agent for Neurodegenerative Diseases. Molecular Neurobiology. 2010;41(2-3):375–383. doi: 10.1007/s12035-010-8111-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Elshaer Mohamed, Chen Yeru, Wang Xiu Jun, Tang Xiuwen. Resveratrol: An overview of its anti-cancer mechanisms. Life Sciences. 2018;207:340–349. doi: 10.1016/j.lfs.2018.06.028. [DOI] [PubMed] [Google Scholar]
- 45.Vinod, B. S. et al. Resveratrol chemosensitizes HER-2-overexpressing breast cancer cells to docetaxel chemoresistance by inhibiting docetaxel-mediated activation of HER-2–Akt axis. Cell Death Discovery, 10.1038/cddiscovery.2015.61 (2015). [DOI] [PMC free article] [PubMed]
- 46.Engelke Laura H., Hamacher Alexandra, Proksch Peter, Kassack Matthias U. Ellagic Acid and Resveratrol Prevent the Development of Cisplatin Resistance in the Epithelial Ovarian Cancer Cell Line A2780. Journal of Cancer. 2016;7(4):353–363. doi: 10.7150/jca.13754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Hotnog, D. et al. Resveratrol modulates apoptosis in 5-fluorouracyl treated colon cancer cell lines. Romanian Archives of Microbiology and Immunology (2013). [PubMed]
- 48.Frampton GA, Lazcano EA, Li H, Mohamad A, Demorrow S. Resveratrol enhances the sensitivity of cholangiocarcinoma to chemotherapeutic agents. Laboratory Investigation. 2010 doi: 10.1038/labinvest.2010.99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Park D-G. Antichemosensitizing effect of resveratrol in cotreatment with oxaliplatin in HCT116 colon cancer cell. Annals of Surgical Treatment and Research. 2014 doi: 10.4174/astr.2014.86.2.68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Mazar A. P. Urokinase Plasminogen Activator Receptor Choreographs Multiple Ligand Interactions: Implications for Tumor Progression and Therapy. Clinical Cancer Research. 2008;14(18):5649–5655. doi: 10.1158/1078-0432.CCR-07-4863. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All data generated or analyzed during this study are included in this published article. The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.