Abstract
The induction of apoptosis is a useful strategy in anti-cancer research. Various Moon Hyung Yang (MHY) compounds have been developed as novel anti-cancer drug candidates; in the present study, the pro-apoptotic effects of (Z)-5-(3-ethoxy-4- hydroxybenzylidene)-2-thioxothiazolidin-4-one (MHY695) on HCT116 human colon cancer cells were assessed. MTT assays were performed to investigate the dose-dependent cytotoxic effects of MHY695 on HCT116 cells. Immunofluorescence staining and flow cytometry analyses were performed to identify apoptotic cell death, and western blot analysis was used to investigate the apoptotic-signaling pathways. A mouse xenograft model was also used to determine the effects of MHY695 in vivo. MHY695 decreased the viability of HCT116 cells and induced apoptotic cytotoxicity. The apoptotic mechanisms induced by MHY695 involved the dephosphorylation of Bcl-2-associated agonist of cell death protein following protein kinase B inactivation, induced myeloid leukaemia cell differentiation protein and BH3-interacting domain death agonist truncation, caspase-3 and −9 activation and poly (ADP-ribose) polymerase cleavage. In addition, MHY695 significantly suppressed tumor growth in the mouse xenograft model, compared with the vehicle control. Notably, MHY695 exhibited potent anti-cancer effects in four different types of human colon cancer cell line, including Caco-2, DLD-1, HT-29 and HCT116. Additionally, MHY695 showed reduced cytotoxicity in NCM460, normal colonic epithelial cells. Furthermore, MHY-induced cytotoxicity in colon cancer cells was independent of the tumor suppressor protein p53. Collectively, these observations suggested that MHY695 may be a novel drug for the treatment of colon cancer.
Keywords: colon cancer, apoptosis, caspase, poly(ADP-ribose) polymerase, p53
Introduction
Colon cancer is the third most frequently occurring cancer in the United States, following prostate cancer in males (or breast cancer in females) and lung cancer (1). In the Republic of Korea, colon cancer is the third most commonly diagnosed type of cancer, following thyroid and stomach cancer, and in 2014, the mortality rate of patients with colon cancer was the fourth highest of all cancer-associated deaths (2). Therefore, the development of novel anti-colorectal cancer drugs is urgently required. Numerous types of anti-cancer drug have been developed, and a select few, including 5-fluorourical (antimetabolite), oxaliplatin (a platinum-based antineoplastic), irinotecan (a topoisomerase inhibitor) and bevacizumab (a monoclonal antibody) are commercially available for the treatment of colon cancer. These drugs are used in different combinations and dosages depending on the cancer stage and condition of the patient (3).
Anti-cancer drugs exert their effects by inducing apoptosis in cancer cells. Apoptosis is a distinct form of programmed cell death and apoptotic cells possess certain characteristic features. Morphologically, apoptotic cells exhibit shrinkage, pyknosis and the formation of apoptotic bodies (4–6). Apoptosis incorporates a number of crucial biochemical steps. In the cytosol, BH3-interacting domain death agonist (Bid) becomes truncated and translocates to the mitochondria (7). In addition, the phosphorylation of Bcl-2-associated agonist of cell death (Bad) induced by phospho-protein kinase B (p-Akt) is interrupted (8). Upon apoptotic signal transduction, cytochrome C is released from mitochondria, causing the activation of pro-caspase-9 by forming an apoptosome with apoptotic protease-activating factor 1. In turn, activated caspase-9 is able to activate pro-caspase-3 by proteolytic cleavage (9). During apoptosis, activated caspases promote the degradation or cleavage of diverse cytoskeletal and nuclear proteins, including poly(ADP-ribose) polymerase (PARP) (10).
Various MHY compounds that exert growth inhibitory effects against different types of cancer have previously been synthesized; these include the hydroxamic acid derivatives MHY218 and MHY219. MHY218 enhanced apoptotic cell death and exhibited anti-cancer activity in gastric, colon, breast and ovarian cancer. In the AGS human gastric cancer cell line, MHY218 inhibited autophagy and enhanced apoptotic cell death (11–14). MHY219, a novel histone deacetylase inhibitor, promoted the upregulation of androgen receptor expression levels, and induced apoptosis in human prostate cancer cells (15,16). The oxirane derivatives MHY336, MHY407, MHY412 and MHY449 have also been investigated. MHY336, a novel epoxypropoxy flavonoid, induced apoptosis and cell cycle arrest in colon and prostate cancer cells by inhibiting topoisomerase II (17,18). Additionally, the novel carbazole derivative MHY407 produced anti-cancerous effects in breast cancer cells by sensitizing the cells to doxorubicin-, etoposide- and radiation-induced DNA damage (19). The anthracene derivative MHY412 induced apoptosis in doxorubicin-resistant breast cancer cells by modulating cell cycle arrest and the downregulation of P-glycoprotein expression (20). Furthermore, MHY449, a novel dihydrobenzofuro[4,5-b][1,8]naphthyridine-6-one derivative, exerted potent anti-cancerous effects on prostate, gastric and lung cancer cells. Furthermore, in human lung cancer cells, MHY449 induced cell cycle arrest and apoptosis by inhibiting the activation of Akt (21–24). Further studies have also reported that benzylidene compounds exert anti-cancerous effects in various types of cancer cells by inducing apoptosis (25–27). Therefore in the present study, a novel benzylidene derivative, (Z)-5-(3-ethoxy-4-hydroxybenzylidene)- 2-thioxothiazolidin-4-one (MHY695) was synthesized and investigated for cytotoxic effects in human colon cancer cells.
Materials and methods
Synthesis of MHY695
A suspension of 3-ethoxy-4-hydroxybenzaldehyde (purity, 99%; 300 mg; 1.81 mmol; Sigma-Aldrich; Merck KGaA) and rhodanine (purity, 97%; 2-thioxothiazolidin-4-one; 264.5 mg; 1.99 mmol; Sigma-Aldrich; Merck KGaA) in 4 ml ethanol was refluxed for 5 h in the presence of piperidine (purity, 99%; 0.05 ml; 0.54 mmol; Sigma-Aldrich; Merck KGaA) (28). After cooling to room temperature in the air, water (20 ml) was added to the reaction mixture to induce precipitation. The precipitate was filtered and washed using water (50 ml), ethyl acetate (20 ml) and methylene chloride (20 ml) and air-dried to yield MHY695 (purity, >98%; 124.9 mg; 24.6%) as an amorphous orange solid. Melting point, 207.8–210.1°C; 1H-NMR (500 MHz, DMSO-d6, δ in ppm): 13.70 (brs, 1H, NH), 10.02 (s, 1H, OH), 7.55 (s, 1H, vinylic H), 7.12 (d, 1H, J =2.0 Hz, 2′-H), 7.06 (dd; 1H; J=2.0, 8.5 Hz; 6′-H), 6.93 (d, 1H, J=8.5 Hz, 5′-H), 4.07 (q, 2H, J=7.0 Hz, 3′-OCH2), 1.35 (t, 3 H, J=7.5 Hz, CH2CH3); 13C-NMR (100 MHz, DMSO-d6, δ in ppm): 196.1 (C2), 170.1 (C4), 150.9 (C3′), 147.9 (C4′), 133.5 (benzylic C), 125.8 (C6′), 125.0 (C1), 121.7 (C5), 117.1 (C5′), 116.1 (C2′), 64.6 (3′-OCH2), 15.3 (CH2CH3); LRMS (ESI-) m/z 280 (M-H)−; HRMS (ESI) m/z C12H11NO3S2 (M-H)− calculated mass: 280.0108; observed mass: 280.0122.
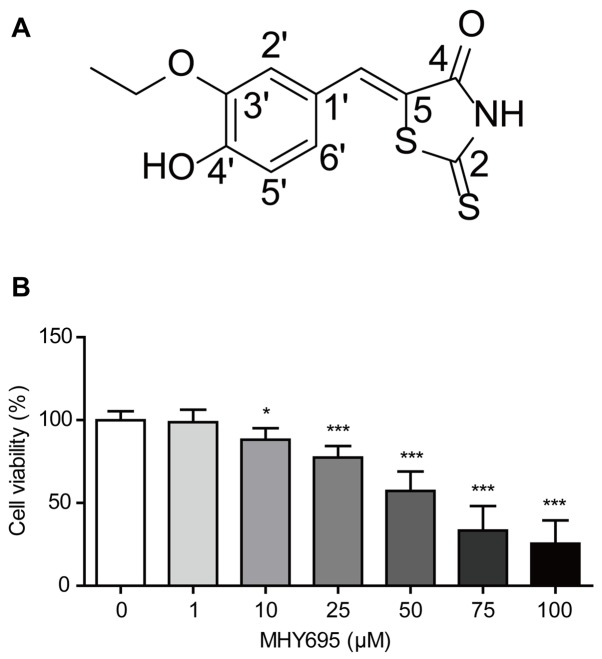
To produce a stock solution, MHY695 was dissolved in DMSO to a concentration of 100 mM (Fig. 1A). The solution was serially diluted in RPMI-1640 or DMEM/High glucose medium (HyClone; GE Healthcare Life Sciences) as required; the final DMSO concentration was <0.1%.
Figure 1.
Viability of HCT116 cells investigated by MTT assay. (A) Structure of MHY695. (B) HCT116 cells were treated with vehicle or MHY695 for 24 h at the indicated concentrations. *P<0.05 and ***P<0.001 vs. the vehicle-treated group. MHY695, (Z)-5-(3-ethoxy-4-hydroxybenzylidene)-2- thioxothiazolidin-4-one.
Cell culture
NCM460, a normal human colonic epithelial cell line, was obtained from INCELL Corporation LLC. The human colon cancer cell line HCT116 [p53 wild-type (wt)] was obtained from the Korea Cell Line Bank, and p53-null HCT116 cells were kindly provided by Dr Young-Chae Chang of the Department of Cell Biology, Catholic University of Daegu School of Medicine (Daegu, Republic of Korea). Human colon cancer DLD-1, Caco-2 and HT-29 cells were purchased from the American Type Tissue Culture Collection. The HCT116 and DLD-1 cells were cultured in RPMI-1640 medium supplemented with 10% fetal bovine serum (HyClone; GE Healthcare Life Sciences) and 1% penicillin/streptomycin (HyClone; GE Healthcare Life Sciences) at 37°C, 5% CO2. NCM460, Caco-2 and HT-29 cells were cultured in DMEM/High glucose medium (HyClone; GE Healthcare Life Sciences) supplemented in the same manner as the RPMI-1640.
Cell viability assay
An MTT assay was performed to investigate the effects of MHY695 on the viabilities of NCM460, Caco-2, DLD-1, HT-29 and HCT116 cells. Thiazolyl blue tetrazolium bromide (Sigma-Aldrich; Merck KGaA) was dissolved in PBS to a stock concentration of 5 mg/ml, and further diluted with RPMI-1640 or DMEM/High glucose medium to a working concentration of 0.5 mg/ml. Cells were cultured in a 96-well cell culture plate at a density of 5×103 cells/well for 24 h prior to treatment. The vehicle control (0.05% DMSO in culture medium) or MHY695 samples were treated with 25 or 50 µM in sextuplicate for 24 h. Thiazolyl blue tetrazolium bromide (0.5 mg/ml) was subsequently added, and the cells were incubated in the dark for 2 h. The formazan crystals were dissolved in DMSO and absorbance was measured at 540 nm using a microplate spectrophotometer (Thermo Fisher Scientific, Inc.). Each experiment was performed three times independently. The half maximal inhibitory concentration (IC50) values were obtained using GraphPad Prism 5 (GraphPad Software).
Cytochrome C and DAPI staining
HCT116 cells were cultured in 4-well chamber slides at a density of 5.0×104 cells/well for 24 h, and treated with the vehicle or MHY695 (25 or 50 µM) for 24 h at 37°C. The cells were fixed with 10% neutral-buffered formalin solution (Sigma-Aldrich; Merck KGaA) for 1 h at room temperature. The fixed cells were incubated in blocking/permeabilization solution, containing 0.1% triton-X-100 in protein block serum-free reagent (Dako; Agilent Technologies, Inc.) for 1 h at room temperature. The cells were subsequently incubated overnight with cytochrome C primary antibody (1:100; cat. no. sc-7159; Santa Cruz Biotechnology) at 4°C. A goat anti-rabbit IgG, fluorescein isothiocyanate (FITC)-conjugated secondary antibody (1:200; cat. no. A120-101F; Bethyl Laboratories, Inc.) was added and incubated for a further 3 h in the dark at room temperature. Vectashield mounting medium with DAPI (Vector Laboratories, Inc.) was used for mounting, and the cells were washed with PBS between each step. Fluorescence was visualized using a confocal microscope at ×480 magnification (Olympus).
FITC Annexin V staining
HCT116 cells were cultured in a 6-well culture plate at a density of 1×106 cells/well for 24 h, and treated with the vehicle or MHY695 (25 or 50 µM) for 24 h at 37°C. The treated cells were collected by centrifugation at 380 × g for 3 min at room temperature and stained using a FITC Annexin V Apoptosis Detection Kit I (BD Biosciences) per the manufacturer's protocol. The stained cells were counted using a flow cytometer (BD Biosciences) within 1 h, and the data were analyzed using C Flow Plus software (version 1.0.227.4; BD Biosciences).
Western blot analysis
HCT116 cells were cultured in a 6-well culture plate at a density of 1×106 cells/well for 24 h, and treated with the vehicle or MHY695 (25 or 50 µM) for a further 24 h at 37°C. The cells were harvested with cell scrapers and lysed in RIPA assay buffer (Elpis BioMed). The total protein was quantified using the Pierce® BCA Protein Assay Kit (Thermo Fisher Scientific, Inc.) and separated using SDS-PAGE on a 10 or 15% gel. The proteins were transferred to PVDF membranes (Merck KGaA) and incubated in 5% bovine serum albumin, fraction V (cat. no. 160069; MP Biomedicals, LLC) or 5% non-fat dry milk (BD Difco™) for 1 h. The membranes were subsequently incubated overnight at 4°C with the appropriate primary antibodies, diluted in blocking buffer. All of the primary antibodies were purchased from Cell Signaling Technology, with the exception of β-actin (Sigma-Aldrich; Merck KGaA). The catalog numbers and the dilution factors of the primary antibodies were as follows: p-Akt (Ser 473; 1:1,000; cat. no. #9275), Akt (1:2,000; cat. no. #9272), p-Bad (Ser136; 1:500; cat. no. #4366), Bad (1:1,000; cat. no. #9239), induced myeloid leukaemia cell differentiation protein (Mcl-1; 1:1,000; cat. no. #5453), Bid (1:1,000; cat. no. #2002), caspase-9 (1:1,000; cat. no. #9508), cleaved caspase-9 (Asp330; 1:500; cat. no. #9501), caspase-3 (1:1,000; cat. no. #9662), cleaved caspase-3 (1:500; cat. no. #9661), cleaved PARP (Asp214; 1:1,000; cat. no. #9541), Bcl-2-binding component 3 (PUMA;1:1,000; cat. no. #12450), Bcl-xL (1:1,000; cat. no. #2764), Apoptosis regulator BAX (Bax; 1:1,000; cat. no. #5023), Bcl-2 homologous antagonist/killer (Bak; 1:1,000; cat. no. #12105) and β-actin (1:10,000; cat. no. #A5316). The membranes were washed using TBS-T buffer (10 mM Tris, 150 mM NaCl, pH 8.0, 0.2% Tween-20) after each step. The secondary antibodies were also diluted in blocking buffer and added for 2 h at room temperature. HRP conjugated goat anti-rabbit IgG polyclonal antibody (1:5,000; cat. no. ADI-SAB-300-J) and HRP conjugated goat anti-mouse IgG polyclonal antibody (1:20,000; cat. no. ADI-SAB-100-J) were purchased from Enzo Life Sciences Inc.. The blots were visualized using an enhanced chemiluminescence solution (Advansta Inc.) and the ChemiDoc™ Touch Gel Imaging System (Bio-Rad Laboratories, Inc.).
Mouse xenograft model
A total of 20 female BALB/c nude mice (four weeks of age) were obtained from OrientBio, Inc. The mice were housed under a 12 h light/dark cycle and fed rodent chow (Samtako Bio Korea) and tap water ad libitum. After a 1-week acclimation period, the mice were subcutaneously inoculated with HCT116 cells (5×106 resuspended in 100 µl PBS) in each flank. After 1 week, vehicle solution or MHY695 (2 mg/kg diluted in PBS) was subcutaneously injected near the inoculated tumor site once a day. No animal exhibited signs of toxicity following the administration of MHY695. All inoculations were performed under anesthesia with isoflurane (Hana Pharm Co., Ltd) using the Small Animal O2 Single Flow Anesthesia System for Rat, Mouse (LMS Co., Ltd). The concentration of isoflurane was 3% for induction and 2% for maintenance, with 1 l/min oxygen. Inoculations were performed when the mice didn't respond to physical stimuli when under anesthesia. The diameter of each tumor was measured daily and tumor volume (mm3) was calculated as (long diameter)2 × (short diameter) × 0.5. The mice were euthanized using carbon dioxide (CO2; with a flow rate 20% per min) and the tumor was dissected following the 12th injection of vehicle solution or MHY695. All animal research and protocols were reviewed and approved by the Pusan National University Institutional Animal Care and Use Committee (PNU-IACUC).
In vivo fluorescence imaging
GFP vectors, kindly provided by Dr Bum-Joon Park (Department of Molecular Biology, College of Natural Science, Pusan National University, Busan, Republic of Korea), were transfected into HCT116 colon cancer cells using the Lipofectamine® 3000 transfection kit (Invitrogen; Thermo Fisher Scientific, Inc.) and Opti-MEM medium (Thermo Fisher Scientific). A total of 4 female BALB/c nude mice (four weeks of age) were obtained from OrientBio, Inc., and acclimated for one week. The GFP-tagged HCT116 cells (1.5×107 cells resuspended in 100 µl PBS) were inoculated subcutaneously into the flanks of each mouse. The vehicle or MHY695 (5 mg/kg diluted in PBS) was administered subcutaneously once a day near each tumor. Tumor fluorescence was observed daily using a fluorescence-labeled Organism Bioimaging Instrument (NeoScience Co., Ltd.) After the 4th injection of vehicle or MHY695, the mice were sacrificed using CO2 inhalation and the tumors were dissected for analysis.
Statistical analysis
Statistical analysis was conducted using GraphPad Prism 5 (GraphPad Software, Inc.). The data were analyzed using one-way analysis of variance followed by Tukey's test for multiple comparisons. The Student's t-test was performed for the comparison of two groups. The results are expressed as the mean ± standard deviation, and P<0.05 was considered to indicate a statistically significant difference.
Results
MHY695 decreases cell viability of HCT116 human colon cancer cells
To investigate the effects of MHY695 on the viability of colon cancer cells, MTT assays were performed using the HCT116 human colon cancer cell line. HCT116 cells were treated with various concentrations of MHY695 for 24 h, and cell viability was determined using an MTT assay. As shown in Fig. 1B, MHY695 significantly reduced the viability of HCT116 cells in a concentration-dependent manner. The percentages of viable cells were 98.8, 88.2, 77.4, 57.3, 33.4 and 25.5% following treatment with 1, 10, 25, 50, 75 and 100 µM MHY695, respectively.
MHY695 induces apoptotic cell death in HCT116 human colon cancer cells
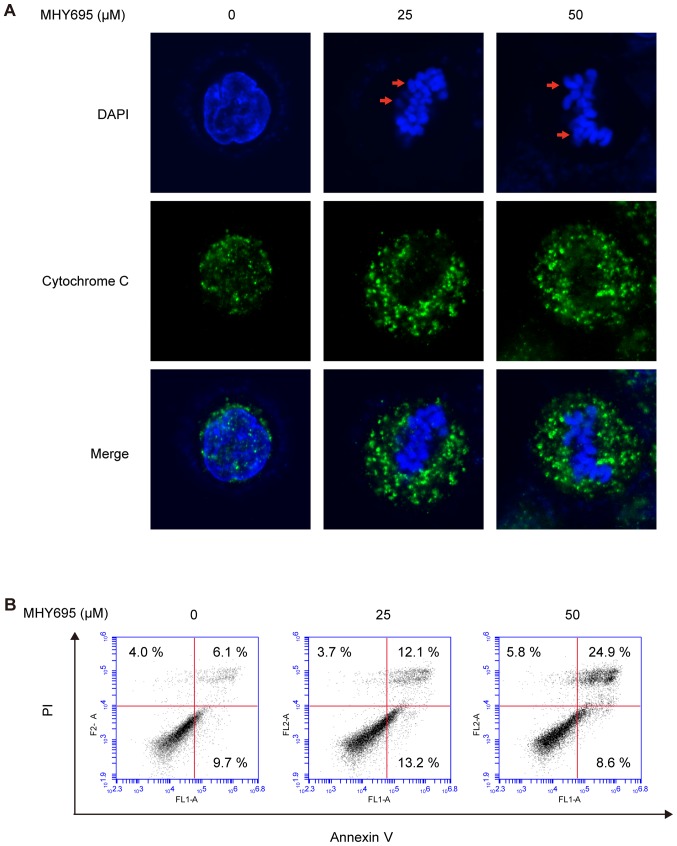
To determine whether MHY695-induced cytotoxicity was the result of apoptotic cell death, nuclear morphological alterations were observed using DAPI staining. Confocal microscopic analysis of HCT116 cells treated with MHY695 (25 or 50 µM) for 24 h showed cellular shrinkage, nuclear condensation and the formation of apoptotic bodies. Moreover, cytochrome C release from the mitochondria into the cytosol was also observed in MHY695-treated HCT116 cells (Fig. 2A). To further investigate MHY695-induced apoptotic cell death, flow cytometric analysis of HCT116 cells treated with MHY695 was conducted after Annexin V/PI staining. As shown in Fig. 2B, the number of apoptotic cells increased in a concentration-dependent manner following MHY695 treatment. The percentages of early and late apoptotic cells (quadrants IV and I, respectively) were 15.8, 25.3 and 33.5% following treatment with MHY695 at 0, 25 and 50 µM, respectively. In addition, the percentages of MHY695-induced cytotoxicity appeared to be higher in the MTT assays (Fig. 1B) compared with the Annexin V/PI assays (Fig. 2B), suggesting that MHY695 may also induce cell cycle arrest.
Figure 2.
MHY695 induces cytotoxicity by promoting apoptosis. (A) Morphological changes in HCT116 colon cancer cells were observed by confocal microscopy. Cells were treated with vehicle or MHY695 (25 or 50 µM) for 24 h. Apoptotic body formation (red arrows) was observed using DAPI staining (blue) in the MHY695-treated groups. The release of cytochrome C (green) was also observed in the MHY695-treated groups. (B) Apoptotic or necrotic cells were identified using flow cytometry. HCT116 cells were treated with MHY695 or vehicle for 24 h, prior to staining with Annexin V and PI. Annexin V-bound or PI-positive cells were subsequently quantified. MHY695, (Z)-5-(3-ethoxy-4-hydroxybenzylidene)-2-thioxothiazolidin-4-one; PI, propidium iodide.
MHY695 modulates the expression of apoptosis-related proteins
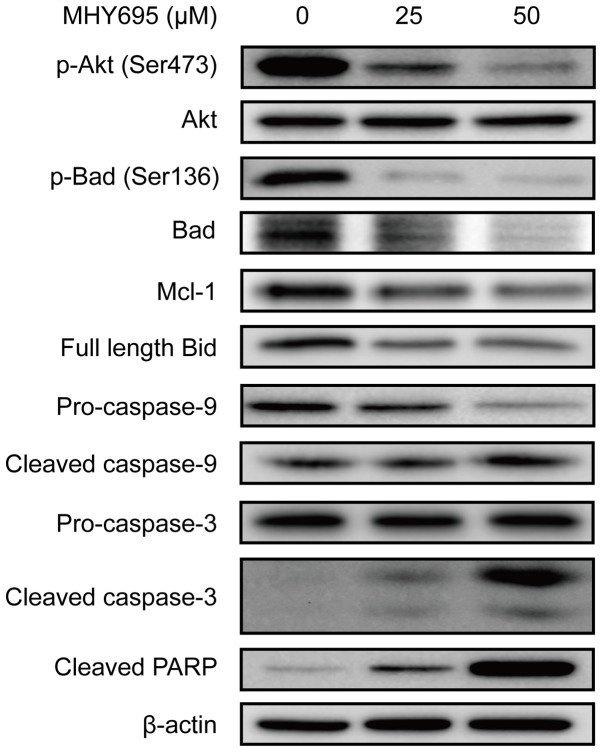
To elucidate the apoptotic signal transduction induced by MHY695 in HCT116 cells, the expression levels of apoptosis-associated proteins were analyzed by western blot analysis. As shown in Fig. 3, Akt and Bad phosphorylation was inhibited by MHY695 treatment at 25 and 50 µM for 24 h. Mcl-1 expression was also inhibited by MHY695. The expression of full-length Bid was also decreased, suggesting that Bid truncation was increased by MHY695. As a result, caspase-9, caspase-3 and PARP cleavage was increased in a concentration-dependent manner.
Figure 3.
MHY695 modulates the expression of apoptosis-associated proteins. HCT116 cells were treated with vehicle or MHY695 (25 or 50 µM) for 24 h. Cellular proteins were separated using SDS-PAGE and transferred to PVDF membranes. The bands were detected using ECL solution and a chemiluminescence system. The bands are representative of three independent experiments. MHY695, (Z)-5-(3-ethoxy- 4-hydroxybenzylidene)-2-thioxothiazolidin-4-one; p-, phospho-; Akt, protein kinase B; Bad, Bcl-2-associated agonist of cell death; Mcl-1, induced myeloid leukaemia cell differentiation protein; PARP, poly(ADP-ribose) polymerase.
MHY695 suppresses tumor growth in a mouse xenograft model
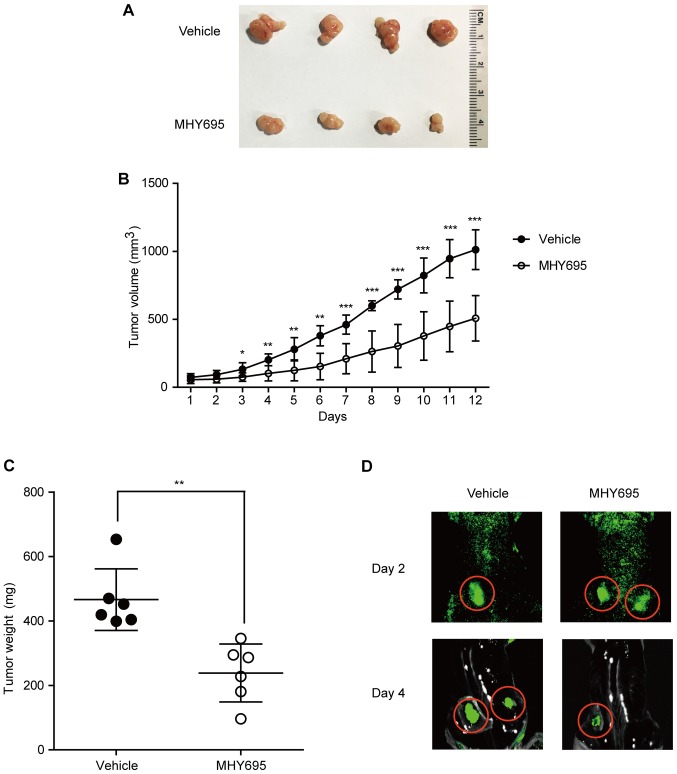
To investigate the anti-cancerous effects of MHY695 in vivo, a mouse xenograft model was employed. HCT116 cells were implanted into the left and right flanks of BALB/c nude mice; 1 week after cancer cell inoculation, the vehicle or MHY695 (2 mg/kg) were injected subcutaneously next to each tumor on a daily basis. As shown in Fig. 4A and B, MHY695 significantly suppressed tumor growth compared with vehicle. Although tumor volume was increased in both the vehicle and MHY695 groups, the growth rate of the MHY695-treated group was significantly lower than that of the vehicle group. As illustrated in Fig. 4C, tumor weight was also lower in the MHY695-treated group compared with the vehicle-treated group. Furthermore, xenograft tumors were visualized using an in vivo fluorescence imaging system, which revealed that four days after treatment, 5 mg/kg MHY695 resulted in reduced tumor growth compared with the vehicle (Fig. 4D).
Figure 4.
In vivo anti-cancer effects of MHY695 in a mouse xenograft model. (A) Tumors were resected from the mice following euthanasia. (B) Tumor size was measured daily with a caliper. Tumor volume (mm3) was calculated as L2 × S × 0.5. L, long diameter; S, short diameter. (C) Tumor weight was measured following resection. MHY695 (2 mg/kg) was subcutaneously injected into each tumor daily. *P<0.05, **P<0.01 and ***P<0.001 vs. the vehicle-treated group. (D) In vivo anti-cancer effects of MHY695 (5 mg/kg) were visualized using GFP-tagged HCT116 stem cells. MHY695, (Z)-5-(3-ethoxy-4-hydroxybenzylidene)-2-thioxothiazolidin-4-one.
Cytotoxic effect of MHY695 is independent of p53
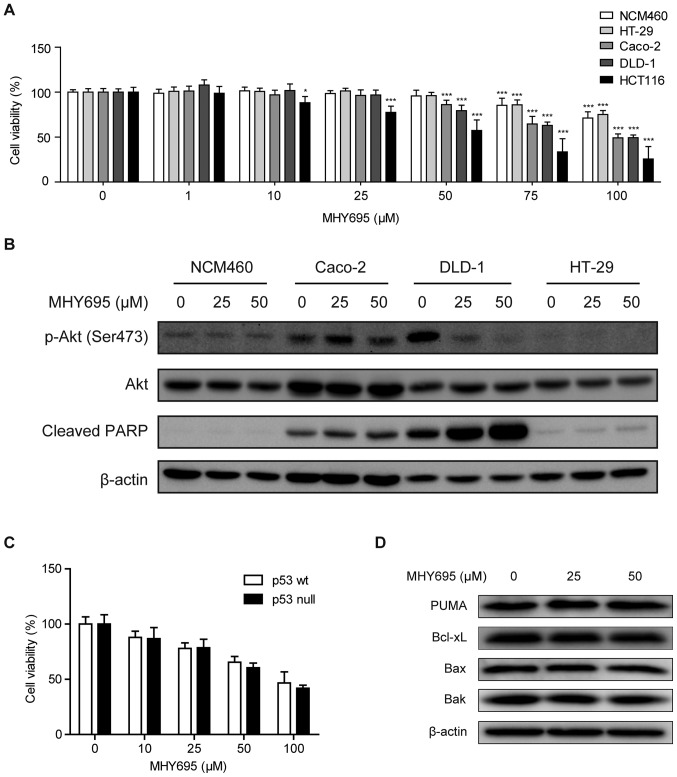
To further investigate the cytotoxic effects of MHY695, a normal colon epithelial cell line and additional human colon cancer cell lines with different genetic features were employed. MTT assays were performed in NCM460, Caco-2, DLD-1, HT-29 and HCT116 cells. Fig. 5A illustrates that concentrations of MHY695 ≤50 µM had no cytotoxic effect on NCM460 or HT-29 cells, while 50 µM was able to reduce the viability of the other human colon cancer cell lines. Specifically, MHY695 showed significant inhibitory effects towards HCT116 cells at a concentration of 10 µM. In addition, the lowest IC50 value among the five cell lines was observed in HCT116 cells (Table I). Moreover, the expression levels of apoptosis-associated proteins were analyzed using western blotting. Fig. 5B illustrates that in DLD-1 cells, Akt phosphorylation (Ser473) was inhibited, but PARP cleavage was increased by 25 or 50 µM MHY695 after 24 h. However, Akt phosphorylation and PARP cleavage were not altered by MHY695 treatment in Caco-2, HT-29 and NCM460 cells, suggesting that these effects are cell type-specific.
Figure 5.
Cytotoxic effects of MHY695 are independent of p53. (A) NCM460, HT-29, Caco-2, DLD-1 and HCT116 cells were treated with various concentrations of MHY695. *P<0.05, **P<0.01 and ***P<0.001 vs. the vehicle-treated group. (B) Expression levels of phospho-Akt and cleaved PARP were investigated in NCM460, Caco-2, DLD-1 and HT-29 cells by western blot analysis. Cells were treated with vehicle or MHY695 (25 or 50 µM) for 24 h. (C) Cell viability of p53 wt and p53 null HCT116 cells were investigated by MTT assay. p53 wt or p53 null HCT116 cells were treated with vehicle or MHY695 at the specified concentrations for 24 h. (D) Downstream proteins of the p53-associated apoptosis pathway were investigated by western blot analysis. p53 wt HCT116 cells were treated with vehicle or MHY695 at the specified concentrations for 24 h. MHY695, (Z)-5-(3-ethoxy-4-hydroxybenzylidene)-2-thioxothiazolidin-4-one; wt, wild-type; p-Akt, phospho-protein kinase B; PARP, poly(ADP-ribose) polymerase; PUMA, Bcl-2-binding component 3; Bax, Apoptosis regulator BAX; Bak, Bcl-2 homologous antagonist/killer.
Table I.
IC50 of MHY695 in human colon cancer cells.
| Cell line | IC50 (µM) |
|---|---|
| HT-29 | 268.7 |
| NCM460 | 236.8 |
| Caco-2 | 110.2 |
| DLD-1 | 104.8 |
| HCT116 | 47.9 |
MHY695, (Z)-5-(3-ethoxy-4-hydroxybenzylidene)-2-thioxothiazolidin- 4-one; IC50, half maximal inhibitory concentration.
It is well known that the p53 gene is mutated or deleted in the majority of cancer cell types. Therefore, the p53 status of the cells was also determined to elucidate the different cytotoxic effects. As shown in Table II, the p53 gene is mutated in Caco-2, DLD-1 and HT-29 cells, but HCT116 cells express wild-type p53 (29). Based on these findings, the cytotoxic effects of MHY695 on p53 wt and p53 null HCT116 cells were investigated using an MTT assay. As shown in Fig. 5C, the cytotoxic effect of MHY695 did not differ between p53 wt and p53 null HCT116 cells. Moreover, the expression levels of PUMA, Bcl-xL, Bax and Bak, downstream proteins of the p53-related apoptosis pathway, were not altered by MHY695 treatment, suggesting that MHY695-mediated cytotoxicity was independent of p53 (Fig. 5D).
Table II.
Genetic features of human colon cancer cells.
| Cell line | KRAS | BRAF | PIK3CA | PTEN | TP53 |
|---|---|---|---|---|---|
| HT-29 | wt | V600E | P449T | wt | R273H |
| Caco-2 | wt | wt | wt | wt | E204X |
| DLD-1 | G13D | wt | E545K, D549N | wt | S241H |
| HCT116 | G13D | wt | H1047R | wt | wt |
KRAS, GTPase KRas; BRAF, serine/threonine-protein kinase B-raf; PIK3CA, phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit α isoform; PTEN, phosphatase and tensin homolog; TP53, cellular tumor antigen p53; wt, wild-type.
Discussion
In the present study, MHY695 was demonstrated to induce cytotoxicity and apoptosis in HCT116 colon cancer cells. When used to treat various colon cancer cell lines, including Caco-2, DLD-1, HT-29 and HCT116 cells, MHY695 showed the greatest potency towards HCT116 cells. In an attempt to establish the reason for this finding, the mutation status of the p53 gene was investigated. The tumor suppressor protein p53 is a transcription factor that serves a pivotal role in apoptosis and cell cycle arrest. When p53 is mutated, cancer cells exhibit resistance to chemotherapy. According to a previous study, p53 is mutated in Caco-2 (E204X), DLD-1 (S241F) and HT-29 (R273H) cells, but not in HCT116 cells (29). Thus, the effects of MHY695 were compared between p53 wt and null HCT116 cells. MHY695 showed equivalent cytotoxicity in both cell types. Also, PUMA, Bcl-xL, Bax and Bak, which are downstream proteins of p53, were not affected by MHY695 (30,31). Collectively, these findings suggested that p53 was not associated with the cytotoxic effect of MHY695. The reasoning for the selectivity of MHY695 towards different types of cancer cell remains a future challenge.
When the results of MTT assays (Fig. 1B) were compared with those of Annexin V/PI staining (Fig. 2B), MHY695 appeared to exhibit lesser cytotoxicity during Annexin V/PI analysis. This result suggests that MHY695 may also promote cell cycle inhibition in HCT116 cells. Therefore, future studies using FACS analysis and/or immunoblot analysis of cell cycle proteins are warranted to determine whether MHY695 induces cell cycle arrest in other colon cancer cell lines.
The expression levels of numerous apoptosis-associated proteins were investigated using western blot analysis. Among them, Bax and Bak, which oligomerize in the mitochondrial membrane and form pores to facilitate the release of cytochrome C, were not affected by MHY695 treatment. Generally speaking, pro-apoptotic proteins such as Bax and Bak are upregulated in apoptotic cells (32). However, MHY695 significantly inhibited the phosphorylation of Akt and Bad. Also, caspase-3 and PARP cleavage, reliable markers of apoptosis, were markedly apparent. According to Datta et al (8) reduced phosphorylation of Akt precedes Bad-induced cell death, hence it was concluded that MHY695 may induce apoptosis by influencing the phosphoinositide 3-kinase (PI3K)/Akt signaling pathway. Furthermore, as the PI3K/Akt pathway is primarily activated by growth factors, such as epidermal growth factor and fibroblast growth factor, it was suggested that MHY695 may target those growth factor receptors, the specifics of which present a future research objective (33,34).
In addition to in vitro studies, the in vivo anti-cancer effects of MHY695 we investigated using a mouse xenograft model. Subcutaneous injection of MHY695 around the tumor significantly inhibited tumor growth compared with the vehicle. However, intraperitoneal injection of MHY695 did not inhibit the growth of tumors located in the flanks (data not shown). Therefore, the route of administration and optimal in vivo dose may be another focus of future research. Taken together, these findings suggested that MHY695 may constitute a candidate for the development of anti-cancer drugs against colon cancer.
Acknowledgements
Not applicable.
Funding
This work was supported by a 2-year research grant from Pusan National University (EI).
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Authors' contributions
GH conducted and analyzed the in vitro cell based and animal experiments, and wrote the manuscript. DK, CP and HRM synthesized the chemical reagents. SJK, JC and YL conducted in vitro experiments. JWY, YJ, JL, NDK and HYC substantially contributed to the design of the study, interpreted the data, and wrote the manuscript. HRM and EI analyzed the data, conceived, supervised and directed the study, and wrote the manuscript. All the authors approved the final version of this manuscript and agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and revised.
Ethics approval and consent to participate
All animal research and protocols were reviewed and approved by the Pusan National University Institutional Animal Care and Use Committee (PNU-IACUC).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Siegel RL, Miller KD, Jemal A. Cancer Statistics, 2017. CA Cancer J Clin. 2017;67:7–30. doi: 10.3322/caac.21387. [DOI] [PubMed] [Google Scholar]
- 2.Jung KW, Won YJ, Oh CM, Kong HJ, Lee DH, Lee KH, Community of Population-Based Regional Cancer Registries. Cancer Statistics in Korea: Incidence, mortality, survival, and prevalence in 2014. Cancer Res Treat. 2017;49:292–305. doi: 10.4143/crt.2017.118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Benson AB, III, Venook AP, Cederquist L, Chan E, Chen YJ, Cooper HS, Deming D, Engstrom PF, Enzinger PC, Fichera A, Grem JL, et al. Colon cancer, version 1.2017, NCCN clinical practice guidelines in oncology. J Natl Compr Canc Netw. 2017;15:370–398. doi: 10.6004/jnccn.2017.0059. [DOI] [PubMed] [Google Scholar]
- 4.Kerr JF, Wyllie AH, Currie AR. Apoptosis: A basic biological phenomenon with wide-ranging implications in tissue kinetics. Br J Cancer. 1972;26:239–257. doi: 10.1038/bjc.1972.33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Levin S, Bucci TJ, Cohen SM, Fix AS, Hardisty JF, LeGrand EK, Maronpot RR, Trump BF. The nomenclature of cell death: Recommendations of an ad hoc Committee of the society of toxicologic pathologists. Toxicol Pathol. 1999;27:484–490. doi: 10.1177/019262339902700419. [DOI] [PubMed] [Google Scholar]
- 6.Elmore S. Apoptosis: A review of programmed cell death. Toxicol Pathol. 2007;35:495–516. doi: 10.1080/01926230701320337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Li H, Zhu H, Xu CJ, Yuan J. Cleavage of BID by caspase-8 mediates the mitochondrial damage in the Fas pathway of apoptosis. Cell. 1998;94:491–501. doi: 10.1016/S0092-8674(00)81590-1. [DOI] [PubMed] [Google Scholar]
- 8.Datta SR, Dudek H, Tao X, Masters S, Fu H, Gotoh Y, Greenberg ME. Akt phosphorylation of BAD couples survival signals to the cell-intrinsic death machinery. Cell. 1997;91:231–241. doi: 10.1016/S0092-8674(00)80405-5. [DOI] [PubMed] [Google Scholar]
- 9.Cai J, Yang J, Jones DP. Mitochondrial control of apoptosis: the role of cytochrome c. Biochim Biophys Acta. 1998;1366:139–149. doi: 10.1016/S0005-2728(98)00109-1. [DOI] [PubMed] [Google Scholar]
- 10.Slee EA, Adrain C, Martin SJ. Executioner caspase-3, −6, and −7 perform distinct, non-redundant roles during the demolition phase of apoptosis. J Biol Chem. 2001;276:7320–7326. doi: 10.1074/jbc.M008363200. [DOI] [PubMed] [Google Scholar]
- 11.Choi PR, Kang YJ, Sung B, Kim JH, Moon HR, Chung HY, Kim SE, Park MI, Park SJ, Kim ND. MHY218-induced apoptotic cell death is enhanced by the inhibition of autophagy in AGS human gastric cancer cells. Int J Oncol. 2015;47:563–572. doi: 10.3892/ijo.2015.3031. [DOI] [PubMed] [Google Scholar]
- 12.Jeon HS, Ahn MY, Park JH, Kim TH, Chun P, Kim WH, Kim J, Moon HR, Jung JH, Kim HS. Anticancer effects of the MHY218 novel hydroxamic acid-derived histone deacetylase inhibitor in human ovarian cancer cells. Int J Oncol. 2010;37:419–428. doi: 10.3892/ijo_00000690. [DOI] [PubMed] [Google Scholar]
- 13.Kim MK, Kang YJ, Kim DH, Hossain MA, Jang JY, Lee SH, Yoon JH, Chun P, Moon HR, Kim HS, et al. A novel hydroxamic acid derivative, MHY218, induces apoptosis and cell cycle arrest through downregulation of NF-κB in HCT116 human colon cancer cells. Int J Oncol. 2014;44:256–264. doi: 10.3892/ijo.2013.2163. [DOI] [PubMed] [Google Scholar]
- 14.Park JH, Ahn MY, Kim TH, Yoon S, Kang KW, Lee J, Moon HR, Jung JH, Chung HY, Kim HS. A new synthetic HDAC inhibitor, MHY218, induces apoptosis or autophagy-related cell death in tamoxifen-resistant MCF-7 breast cancer cells. Invest New Drugs. 2012;30:1887–1898. doi: 10.1007/s10637-011-9752-z. [DOI] [PubMed] [Google Scholar]
- 15.De U, Kundu S, Patra N, Ahn MY, Ahn JH, Son JY, Yoon JH, Moon HR, Lee BM, Kim HS. A new histone deacetylase inhibitor, MHY219, inhibits the migration of human prostate cancer cells via HDAC1. Biomol Ther (Seoul) 2015;23:434–441. doi: 10.4062/biomolther.2015.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Patra N, De U, Kim TH, Lee YJ, Ahn MY, Kim ND, Yoon JH, Choi WS, Moon HR, Lee BM, Kim HS. A novel histone deacetylase (HDAC) inhibitor MHY219 induces apoptosis via up-regulation of androgen receptor expression in human prostate cancer cells. Biomed Pharmacother. 2013;67:407–415. doi: 10.1016/j.biopha.2013.01.006. [DOI] [PubMed] [Google Scholar]
- 17.Lee SH, Kang YJ, Kim DH, Sung B, Kang JA, Chun P, Yoon JH, Moon HR, Kim HS, Chung HY, Kim ND. A novel oxiranylchromenone derivative, MHY336, induces apoptosis and cell cycle arrest via a p53- and p21-dependent pathway in HCT116 human colon cancer cells. Int J Oncol. 2014;44:943–949. doi: 10.3892/ijo.2013.2226. [DOI] [PubMed] [Google Scholar]
- 18.Patra N, De U, Kang JA, Kim JM, Ahn MY, Lee J, Jung JH, Chung HY, Moon HR, Kim HS. A novel epoxypropoxy flavonoid derivative and topoisomerase II inhibitor, MHY336, induces apoptosis in prostate cancer cells. Eur J Pharmacol. 2011;658:98–107. doi: 10.1016/j.ejphar.2011.02.015. [DOI] [PubMed] [Google Scholar]
- 19.Yoon S, Kim JH, Lee YJ, Ahn MY, Choi G, Kim WK, Yang Z, Lee HJ, Moon HR, Kim HS. A novel carbazole derivative, MHY407, sensitizes cancer cells to doxorubicin-, etoposide-, and radiation treatment via DNA damage. Eur J Pharmacol. 2012;697:24–31. doi: 10.1016/j.ejphar.2012.10.001. [DOI] [PubMed] [Google Scholar]
- 20.De U, Chun P, Choi WS, Lee BM, Kim ND, Moon HR, Jung JH, Kim HS. A novel anthracene derivative, MHY412, induces apoptosis in doxorubicin-resistant MCF-7/Adr human breast cancer cells through cell cycle arrest and downregulation of P-glycoprotein expression. Int J Oncol. 2014;44:167–176. doi: 10.3892/ijo.2013.2160. [DOI] [PubMed] [Google Scholar]
- 21.Hwang HJ, Kang YJ, Hossain MA, Kim DH, Jang JY, Lee SH, Yoon JH, Moon HR, Kim HS, Chung HY, Kim ND. Novel dihydrobenzofuro[4,5-b][1,8]naphthyridin-6-one derivative, MHY-449, induces apoptosis and cell cycle arrest in HCT116 human colon cancer cells. Int J Oncol. 2012;41:2057–2064. doi: 10.3892/ijo.2012.1659. [DOI] [PubMed] [Google Scholar]
- 22.Kim SH, Kang YJ, Sung B, Kim DH, Lim HS, Kim HR, Kim SJ, Yoon JH, Moon HR, Chung HY, Kim ND. MHY-449, a novel dihydrobenzofuro[4,5-b][1,8]naphthyridin-6-one derivative, mediates oxidative stress-induced apoptosis in AGS human gastric cancer cells. Oncol Rep. 2015;34:288–294. doi: 10.3892/or.2015.3984. [DOI] [PubMed] [Google Scholar]
- 23.Lee SH, Kang YJ, Sung B, Kim DH, Lim HS, Kim HR, Kim SJ, Yoon JH, Moon HR, Chung HY, Kim ND. MHY-449, a novel dihydrobenzofuro[4,5-b][1,8] naphthyridin-6-one derivative, induces apoptotic cell death through modulation of Akt/FoxO1 and ERK signaling in PC3 human prostate cancer cells. Int J Oncol. 2014;44:905–911. doi: 10.3892/ijo.2014.2257. [DOI] [PubMed] [Google Scholar]
- 24.Lim HS, Kang YJ, Sung B, Kim SH, Kim MJ, Kim HR, Kim SJ, Choi YH, Moon HR, Chung HY, Kim ND. Novel dihydrobenzofuro[4,5-b][1,8]naphthyridin-6-one derivative, MHY-449, induces cell cycle arrest and apoptosis via the downregulation of Akt in human lung cancer cells. Oncol Rep. 2015;34:2431–2438. doi: 10.3892/or.2015.4208. [DOI] [PubMed] [Google Scholar]
- 25.Jada SR, Matthews C, Saad MS, Hamzah AS, Lajis NH, Stevens MF, Stanslas J. Benzylidene derivatives of andrographolide inhibit growth of breast and colon cancer cells in vitro by inducing G(1) arrest and apoptosis. Br J Pharmacol. 2008;155:641–654. doi: 10.1038/bjp.2008.368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Zuliani V, Carmi C, Rivara M, Fantini M, Lodola A, Vacondio F, Bordi F, Plazzi PV, Cavazzoni A, Galetti M, et al. 5-Benzylidene-hydantoins: Synthesis and antiproliferative activity on A549 lung cancer cell line. Eur J Med Chem. 2009;44:3471–3479. doi: 10.1016/j.ejmech.2009.01.035. [DOI] [PubMed] [Google Scholar]
- 27.Yang X, Wang W, Qin JJ, Wang MH, Sharma H, Buolamwini JK, Wang H, Zhang R. JKA97, a novel benzylidene analog of harmine, exerts anti-cancer effects by inducing G1 arrest, apoptosis, and p53-independent up-regulation of p21. PLoS One. 2012;7:e34303. doi: 10.1371/journal.pone.0034303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Pinson JA, Schmidt-Kittler O, Zhu J, Jennings IG, Kinzler KW, Vogelstein B, Chalmers DK, Thompson PE. Thiazolidinedione-based PI3Kα inhibitors: An analysis of biochemical and virtual screening methods. Chem Med Chem. 2011;6:514–522. doi: 10.1002/cmdc.201000467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ahmed D, Eide PW, Eilertsen IA, Danielsen SA, Eknæs M, Hektoen M, Lind GE, Lothe RA. Epigenetic and genetic features of 24 colon cancer cell lines. Oncogenesis. 2013;2:e71. doi: 10.1038/oncsis.2013.35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Nakano K, Vousden KH. PUMA, a novel proapoptotic gene, is induced by p53. Mol Cell. 2001;7:683–694. doi: 10.1016/S1097-2765(01)00214-3. [DOI] [PubMed] [Google Scholar]
- 31.Ashkenazi A. Directing cancer cells to self-destruct with pro-apoptotic receptor agonists. Nat Rev Drug Discov. 2008;7:1001–1012. doi: 10.1038/nrd2637. [DOI] [PubMed] [Google Scholar]
- 32.Wei MC, Zong WX, Cheng EH, Lindsten T, Panoutsakopoulou V, Ross AJ, Roth KA, MacGregor GR, Thompson CB, Korsmeyer SJ. Proapoptotic BAX and BAK: A requisite gateway to mitochondrial dysfunction and death. Science. 2001;292:727–730. doi: 10.1126/science.1059108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Wang X, McCullough KD, Franke TF, Holbrook NJ. Epidermal growth factor receptor-dependent Akt activation by oxidative stress enhances cell survival. J Biol Chem. 2000;275:14624–14631. doi: 10.1074/jbc.275.19.14624. [DOI] [PubMed] [Google Scholar]
- 34.Hu Y, Lu H, Zhang J, Chen J, Chai Z, Zhang J. Essential role of AKT in tumor cells addicted to FGFR. Anticancer Drugs. 2014;25:183–188. doi: 10.1097/CAD.0000000000000034. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.