Abstract
P2X7 receptors (Rs) constitute a subclass of ATP-sensitive ionotropic receptors (P2X1-P2X7). P2X7Rs have many distinguishing features, mostly based on their long intracellular C terminus regulating trafficking to the cell membrane, protein–protein interactions, and post-translational modification. Their C-terminal tail is especially important in enabling the transition from the nonselective ion channel mode to a membrane pore allowing the passage of large molecules. There is an ongoing dispute on the existence of neuronal P2X7Rs with consequences for our knowledge on their involvement in neuroinflammation, aggravating stroke, temporal lobe epilepsy, neuropathic pain, and various neurodegenerative diseases. Whereas early results appeared to support the operation of P2X7Rs at neurons, more recently glial P2X7Rs are increasingly considered as indirect causes of neuronal effects. Specific tools for P2X7Rs are of limited value because of the poor selectivity of agonists, and the inherent failure of antibodies to differentiate between the large number of active and inactive splice variants, or gain-of-function and loss-of-function small nucleotide polymorphisms of the receptor. Unfortunately, the available P2RX7 knock-out mice generated by pharmaceutical companies possess certain splice variants, which evade inactivation. In view of the recently discovered bidirectional dialogue between astrocytes and neurons (and even microglia and neurons), we offer an alternative explanation for previous data, which assumedly support the existence of P2X7Rs at neurons. We think that the unbiased reader will follow our argumentation on astrocytic or microglial P2X7Rs being the primary targets of pathologically high extracellular ATP concentrations, although a neuronal localization of these receptors cannot be fully excluded either.
Introduction
P2XR types
ATP-sensitive P2 receptors (Rs) have been classified into two groups: the ligand-gated P2X type and the G-protein-coupled P2Y type (Abbracchio and Burnstock, 1994). Scientific interest in P2Rs considerably increased after their successful cloning in 1983 (Webb et al., 1993; Surprenant et al., 1996; Burnstock, 2013). Within the P2X-type, 7 subtypes have been identified (P2X1–7), which form homotrimeric and heterotrimeric assemblies and mediate rapid transmembrane ionic fluxes. The P2X7R is unique in occurring only as a homotrimer and causing, in addition to immediate effects, also molecular changes on a much longer time-scale, such as proliferation and apoptosis (Surprenant et al., 1996; North, 2016). It has two transmembrane regions, intracellular N- and C-termini, as well as a large extracellular loop. As inferred from crystallization and x-ray diffraction of the zebrafish P2X4R (Hattori and Gouaux, 2012) and, more recently, the human P2X3R (Mansoor et al., 2016), as well as from earlier mutagenesis studies (North, 2002), the binding pouch for ATP is harbored at the intersection of the extracellular loops of two neighboring subunits. The C-terminal region of P2X7Rs is much longer than that of the other subtypes, which regulates trafficking to the cell membrane, protein–protein interactions, and post-translational modification (Feng et al., 2005; Roger et al., 2008, 2010; Costa-Junior et al., 2011). The C terminus is especially important in enabling the transition from the nonselective ion-channel mode to a large membrane pore allowing the diffusion of molecules up to 900 Da (e.g., ATP, glutamate, GABA) (Sperlágh et al., 2006) and fluorescent dyes (Surprenant et al., 1996; Bartlett et al., 2014) according to their concentration gradient. Hence, ATP is an agonist at the receptor pore as well as a molecule that permeates the dilated receptor or its associated pore and subsequently strengthens the agonist-induced receptor activation.
The P2X7R
Kinetic modeling coupled with electrophysiological experiments suggest that, in the absence of extracellular ATP, P2X7Rs remain closed. When one ATP molecule is bound, the receptor becomes desensitized, whereas the binding of two molecules triggers conformational changes leading to the opening of the nonselective cationic channel (Yan et al., 2010). The occupation of three binding sites initiates the generation of a large membrane pore (Yan et al., 2010). There are multiple, mutually exclusive hypotheses explaining the construction of this pore: (1) high concentrations of ATP for extended periods of time or repetitive application of ATP pulses cause channel dilation, which is an inherent property of other P2XRs, such as P2X4 as well (Chaumont and Khakh, 2008); (2) the shift in equilibrium potential characterizing channel conductance leads to time-dependent alterations in the concentration of intracellular ions rather than channel dilation (Li et al., 2015); and (3) pore opening is due to the recruitment of an accessory protein, the pannexin-1 channel (Pelegrin and Surprenant, 2006).
The classic P2X7R in monocytes/macrophages and microglia
Already before the cloning of the P2X7Rs by Surprenant et al. (1996), a native receptor found in lymphocytes and macrophages, originally termed P2Z, has been shown to mediate ATP-induced plasma membrane permeability and cytotoxicity (Gordon, 1986). P2X7Rs are considered to be identical with P2ZRs in both their preferential location at cells of the hematopoietic linage and their necrotic/apoptotic properties (Surprenant et al., 1996; Volonté et al., 2012). Indeed, P2X7Rs have a widespread distribution at lymphocytes, monocytes/macrophages, and dendritic cells, but also in platelets and erythrocytes (Di Virgilio et al., 2001). In the CNS, P2X7R-mRNA and protein were originally thought to be confined to microglia, activated, for instance, by ischemia due to medial cerebral artery occlusion (MCAO) (Collo et al., 1997). Microglia are resident macrophages of the brain and arise from the yolk sac erythromyeloid precursors that invade the brain early in the course of embryonic development (Sperlágh and Illes, 2007; Bhattacharya and Biber, 2016). P2X7Rs confine to microglia a whole range of properties causing indiscriminate damage to neurons as confirmed for neuron/microglia cocultures (Skaper et al., 2006). Immediate consequences of P2X7R stimulation are the release of inflammatory mediators through (1) the costimulation of the post-translational processing of interleukin-1β (IL-1β) upon an LPS stimulus; (2) the shedding of microvesicles and thereby the release of IL-1β into the extracellular environment; (3) the expression of inducible nitric oxide synthase and in consequence increased NO production; (4) the generation of reactive oxygen intermediates, in particular superoxide; and (5) the secretion of diacylglycerol lipase responsible for endocannabinoid production and the simultaneous inhibition of monoacylglycerol lipase responsible for endocannabinoid degradation (Sperlágh et al., 2007; Skaper et al., 2009, 2011; Turola et al., 2012). P2X7Rs induce apoptosis in glial cells via the activation of the caspase pathway, and necrosis via opening of membrane pores and the subsequent loss of intracellular proteins of vital significance. P2X7Rs drive not only microglial activation but also its proliferation, potentially leading to a deleterious cycle of neuroinflammation and neurodegeneration (Monif et al., 2010).
P2X7R-immunoreactivity (IR) in the rodent brain has been shown to be upregulated after MCAO, at first in microglia (Collo et al., 1997) but then, with a temporal lag, also in astrocytes and neurons (Franke et al., 2004; Pedata et al., 2016). Under in vitro conditions, mimicking ischemic damage of HEK293 cells by incubation with an oxygen/glucose-deprived medium facilitated the integration of wild-type P2X7Rs into the plasma membrane, although the integration of their trafficking-deficient mutants did not occur (Milius et al., 2007). Thus, it was concluded that hypoxia/ischemia leads to the increased expression of P2X7Rs in the cellular membrane by promoting trafficking from the endoplasmic reticulum and Golgi apparatus where these receptors are synthesized. Apparently, the direct damage of neurons by MCAO-induced metabolic limitation in the central necrotic area of the parietal cortex is followed in the surrounding penumbra by a more slowly developing disturbance due to the activation of microglia and the secretion of their inflammatory mediators in consequence of P2X7R upregulation (Illes and Verkhratsky, 2016). The participation of microglia in all types of subacute and chronic injuries of CNS neurons, including neurodegenerative diseases, has been discussed in a number of excellent overviews (Sperlágh and Illes, 2007, 2014; Skaper et al., 2010; Burnstock et al., 2011; Volonté et al., 2012; Bartlett et al., 2014; Bhattacharya and Biber, 2016) and will not be reviewed here. We will rather focus on cells of the neural linage, such as neurons and neuroglial cells (astrocytes, Bergmann glia, Müller cells, oligodendrocytes) and their mutual interaction in shaping normal and pathological CNS functions.
The neuronal P2X7R: early arguments for and subsequent doubts in their existence
P2X7R-mRNA and protein in neurons
RT-PCR and Western blotting confirmed the presence of P2X7R-mRNA and protein in extracts of the spinal cord and medulla oblongata (Deuchars et al., 2001), midbrain (Gómez-Villafuertes et al., 2001; Marín-García et al., 2008), hippocampus (Sperlágh et al., 2002), and cerebral cortex (Wirkner et al., 2005) of rodents. In addition, in situ hybridization helped to attribute P2X7R-mRNA to neurons in diverse areas of the brain (Deuchars et al., 2001; Yu et al., 2008). Immunohistochemistry indicated the colocalization of P2X7R-IR with the neuronal marker microtubule-associated protein-2 in cultured cerebrocortical neurons (Wirkner et al., 2005; Fischer et al., 2009a). Antibodies in conjunction with electron microscopy revealed that P2X7R-IR is present in presynaptic nerve terminals of the CNS (Deuchars et al., 2001; Sperlágh et al., 2002). Moreover, P2X7R-IR was detected in synaptosomal preparations (isolated synaptic terminals) of cerebellar granule cells (Sánchez-Nogueiro et al., 2005), hippocampal neurons (Rodrigues et al., 2005), and cerebrocortial neurons (Alloisio et al., 2008).
Evidence for functional P2X7Rs in neurons
Electrophysiological recordings in brain and spinal cord slices (Deuchars et al., 2001; Ireland et al., 2004) or neuronally enriched cerebrocortical cell cultures (Wirkner et al., 2005), as well as measurement of the release of tritiated GABA from brain slices (Sperlágh et al., 2002) were considered to indicate the presynaptic facilitatory effect of ATP and its structure analog dibenzoyl-ATP (Bz-ATP) at presynaptic P2X7Rs. Bz-ATP is a prototypic P2X7R agonist, exhibiting higher potency at this receptor type than ATP itself. The stimulus for transmitter release was the ATP/Bz-ATP-induced rise of intracellular Ca2+ ([Ca2+]i) as demonstrated by Ca2+ imaging in synaptosomes (Miras-Portugal et al., 2003; Sánchez-Nogueiro et al., 2005; Marín-García et al., 2008).
The arguments for the existence of presynaptic P2X7Rs critically depend on the selectivity of the respective antibodies. However, three batches of antibodies, binding to intracellular C-terminal or ectodomain epitopes, yielded different immunoreactivity patterns (Anderson and Nedergaard, 2006). Still more importantly, all antibodies stained not only hippocampal structures of wild-type mice, but also those of two separate P2X7R knock-out strains generated by the pharmaceutical companies Pfizer or Glaxo Smith Kline (Sim et al., 2004). By contrast, the labeling of P2X7Rs in peripheral organs was robust in wild-type and absent in knock-out animals (Sim et al., 2004). The picture became still more confusing when it was reported that midbrain synaptic terminals (synaptosomes) of the Pfizer P2RX7−/− mice possess functional P2X7Rs mediating ATP and Bz-ATP-induced [Ca2+]i transients in a Brilliant Blue G (BBG)-antagonizable manner (Sánchez-Nogueiro et al., 2005; Marín-García et al., 2008). It was concluded that an unidentified P2X7R-like protein is involved in the functional responses. These apparent riddles were resolved only some time afterward. There are several splice variants and small nucleotide polymorphisms of mammalian P2X7Rs, all of which are widely expressed in the nervous system and some of which may escape inactivation by genetic deletion (Sperlágh and Illes, 2014). In humans, 7 naturally occurring splice variants were initially reported and termed P2X7B to P2X7H (Cheewatrakoolpong et al., 2005; Bartlett et al., 2014). A naturally occurring truncated isoform (P2X7B) of the human wild-type P2X7R (P2X7A) has been found in the CNS probably participating in the control of cell growth (Adinolfi et al., 2010). A C-terminally truncated variant of the mouse P2X7R has also been identified, which partly retains its functionality when present in tissues of the P2RX7 gene-deficient Pfizer mice (Masin et al., 2012). Another isoform is the P2X7(k) variant, which, in contrast to the originally identified P2X7(a), is sensitive to ADP ribosylation and is present in the Glaxo mouse (Nicke et al., 2009; Schwarz et al., 2012). ADP ribosylation is a distinctive feature of the mouse P2X7R in that it can be activated by extracellular nicotinamide dinucleotide (NAD+) by means of an ADP-ribosyltransferase 2 coenzyme (Adriouch et al., 2008). Eventually, gain-of-function or loss-of-function small nucleotide polymorphisms may also exist in the two knock-out mouse strains mentioned (Fuller et al., 2009; Bartlett et al., 2014). Hence, functional splice variants and naturally occurring mutants of the P2X7R as well as of their wild-type counterparts are equally labeled by P2X7R antibodies; this may lead to erroneous conclusions with regard to functionality.
Notwithstanding some uncertainty, it was concluded in 2006 that neuronal P2X7Rs exist not only as spurious immunological signals but as true operational units regulating the function of neurons (Anderson and Nedergaard, 2006; Sperlágh et al., 2006). It was suggested that at least two of three criteria have to be fulfilled before a P2XR can be classified as belonging to the P2X7 subtype (Anderson and Nedergaard, 2006). These criteria are the following: (1) the effects of ATP/Bz-ATP should be potentiated in a low divalent cation containing external medium, probably due to the removal of an allosteric blockade of the receptor by Ca2+ (Yan et al., 2011); (2) Bz-ATP should have a higher agonistic potency than ATP; and (3) BBG or other selective P2X7R antagonists should block the receptors already at rather low concentrations. Indeed, these criteria are of considerable help in identifying P2X7Rs in various tissues.
Astroglial P2X7Rs
P2X7R-mRNA and protein in astroglial cells
Even if P2X7Rs were absent at neurons, neuroglial/microglial P2X7Rs could trigger the release of signaling molecules that thereafter initiate seemingly primary neuronal functions. P2X7R-mRNA and protein have been detected in both cultured astrocytes as well as in their counterparts localized in brain/spinal cord slices or in whole animals (Illes et al., 2012). RT-PCR, Western blotting, and immunohistochemistry of astrocytic cultures obtained from the rat cerebral cortex documented the presence of P2X7Rs (Fischer et al., 2009b). In acutely isolated human Müller cells of the retina, single-cell RT-PCR identified the message for P2X7Rs (Pannicke et al., 2000). In cultured astrocyte-like satellite glial cells of rat DRGs, P2X7R-IR was detected (Chen et al., 2008). Hippocampal (Kukley et al., 2001) as well as accumbal and cortical astrocytes of rats stained for P2X7R-IR (Franke et al., 2012), although the latter ones only after mechanical or ischemic injury.
Evidence for functional P2X7Rs in astroglial cells
P2X7R-mediated currents were found to mediate cationic currents in acutely isolated human Müller cells of the retina (Pannicke et al., 2000). Such currents were identified recently also in cultured rat cortical astrocytes (Duan et al., 2003; Nörenberg et al., 2010) and in astrocytes patch-clamped in acute brain slices of rats and mice (Oliveira et al., 2011). Further, in cultured cerebrocortical astrocytes, P2X7Rs were identified as the main pathway for Ca2+ entry, although astroglial P2YRs possibly also contribute to the ATP or Bz-ATP-induced Ca2+ transients (Fumagalli et al., 2003; Nobile et al., 2003). In the cerebellum of mice, Bergmann glial cells expressed P2X7Rs responding to Bz-ATP with inward current and depolarization, potentiated in the absence of extracellular Ca2+/Mg2+ and blocked by P2X7R antagonists (Habbas et al., 2011). Bz-ATP-induced [Ca2+]i transients in mouse cerebellar astrocytes exhibited two components, namely, an initial rapid metabotropic phase followed by a sustained one that depended on extracellular Ca2+ (Salas et al., 2013). This latter phase was absent in astrocytes from P2RX7−/− mice, was modulated by extracellular Mg2+, and was sensitive to the P2X7R-antagonistic BBG and A-438079. Continuous Bz-ATP application caused a gradual increase in membrane permeability to large cations, such as N-methyl-d-glucamine, and led ultimately to the death of the mouse astrocytes (Salas et al., 2013).
The long-term effects of P2X7R stimulation in astrocytic cultures and their second-messenger mechanisms have been extensively investigated (Duan and Neary, 2006; Burnstock et al., 2011; Franke et al., 2012; Illes et al., 2012; Rodrigues et al., 2015). The validity of the conclusions drawn by the reviewed studies is often hampered by the lack of selective P2X7R antagonists used in combination with the prototypic but unselective agonist Bz-ATP. Hence, we will not discuss the effects of P2X7R stimulation continuing over days or weeks and culminating in apoptosis/necrosis.
The neuroinflammatory astroglial P2X7R
As mentioned before in microglia, P2X7Rs tightly control the synthesis and release of the inflammatory cytokine IL-1β (Skaper et al., 2006). P2X7Rs are linked, probably via pannexin channels, to the inflammasome, which is a protein complex consisting of caspase-1, apoptosis-associated speck-like protein, and nod-like receptor protein (NLRP1 or NLRP3) (Pelegrin and Surprenant, 2006, 2009; Di Virgilio, 2007). The inflammasome is a component of the innate immune system and is responsible for the activation of inflammatory processes.
The P2X7R-pannexin complex
Gap junctions in vertebrates are comprised of two docked connexin (Cx) hemichannels linking the cytosol of adjacent cells, whereas in invertebrates they are built up of innexins of related structure. Homologous to innexins are the vertebrate pannexins (Panx), which, however, do not form gap junctions and exist only as channels (Sosinsky et al., 2011; Bennett et al., 2012). In vertebrates, Cx-43 and Panx-1 hemichannels are pathways to release ATP into the extracellular space, which then plays important roles in secondary inflammatory reactions following, for example, traumatic spinal cord or optic nerve damage (C. Huang et al., 2012; Beckel et al., 2014), as well as ischemic brain injury (Bargiotas et al., 2009; Orellana et al., 2014). Fibroblast growth factor-1 (FGF-1) is secreted in response to mechanical traumata inflicted upon the spinal cord (Koshinaga et al., 1993). FGF-1 induces within minutes the release of ATP from astrocytes by a vesicular mechanism; this ATP then activates P2X7Rs and pannexin channels, and consequently triggers a further release of ATP (Garré et al., 2010, 2016). FGF-1, ATP, and P2X7Rs thereby contribute to an early inflammatory phase characterized by the recruitment of phagocytotic and cytokine/chemokine secreting monocytes/macrophages (Garré et al., 2010, 2016). Prenatal exposure to inflammatory conditions by injecting LPS to pregnant mice resulted in the astrocytes of their offspring in an enhanced release of ATP through pannexin unopposed channels (Avendano et al., 2015). This ATP then caused neuronal death by P2X7R activation.
Already before realizing the close association between P2X7Rs and Panx-1 channels, convincing evidence was presented for the release of glutamate (Duan et al., 2003; Fellin et al., 2006) and ATP itself (Suadicani et al., 2006, Suadicani et al., 2009) through P2X7Rs. Of course, other mechanisms of glutamate/ATP release from astrocytes are also possible (Duan et al., 2006). These are, for example, the reversal of neurotransmitter transporters, ABC transporters, Ca2+-dependent exocytosis, lysosomal release, connexin hemichannels, and osmolytic transporters linked to anion channels (Illes and Ribeiro, 2004; Duan and Neary, 2006; Lohman et al., 2012).
Glia-neuron signaling
The tripartite synapse
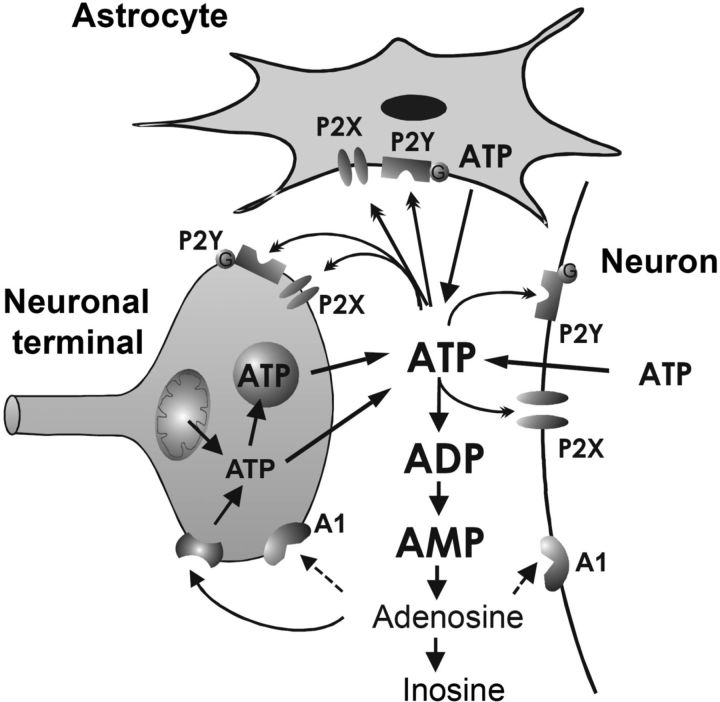
According to the concept of “tripartite synapse” (presynaptic and postsynaptic neuronal elements, as well as fine astrocytic processes being in intimate contact with these synapses), astrocytic processes sense neuronal transmitter release and also signal back to neurons either at the same synapse or at remote ones by the release of so-called gliotransmitters (Fig. 1). Recent studies revealed that small, rapid, and localized Ca2+ responses can be elicited in microdomains of astrocytic processes by minimal synaptic activity (Di Castro et al., 2011; Panatier et al., 2011). These data suggest that astrocytes may integrate the activity of several individual synapses to generate the larger Ca2+ responses upon sustained and intense stimulation (Araque et al., 2014). In turn, transmitter molecules may be secreted from astrocytes by various mechanisms, including Ca2+-dependent exocytosis (Araque et al., 2014; Gundersen et al., 2015; Verkhratsky et al., 2016).
Figure 1.
Schematic of the tripartite synapse, which consists of the presynaptic neuronal terminals, the innervated soma-dendritic structures of the postsynaptic neuron, and the astrocytic processes projecting to the synapse. ATP is synthesized in mitochondria of neurons and astrocytes. It is either stored in the cytosol or is taken up from the cytosol into synaptic vesicles. The release into the extracellular space occurs via transporters or exocytotic mechanisms. In addition, during brain or spinal cord injury, ATP may pour out from the cell interior through the damaged plasma membrane. ATP then binds to its specific receptors (P2X, P2Y) and becomes afterward sequentially degraded to adenosine and finally to the inactive metabolite inosine. Adenosine stimulates its own receptors (e.g., A1). The P2X, P2Y, and adenosine receptors may be located at neuronal terminals, postsynaptic soma/dendritic elements, and astrocytes. We think that an important member of the P2XR family (P2X7) is predominantly astrocytic and regulates neuronal functions by gliotransmitters/signaling molecules. Modified with permission from Illes (2006).
P2X7R involvement in glia-neuron signaling
P2X7Rs can induce the release of glutamate or ATP from astrocytes either via exocytotic (Verkhratsky et al., 2016) or nonexocytotic mechanisms (Illes and Ribeiro, 2004). These receptors may increase [Ca2+]i, which is a trigger of the vesicular/lysosomal gliotransmitter release, but they can be also instrumental in glutamate/ATP outflow through their associated pannexin channels. P2X7R-mediated depolarization generates a massive influx of sodium into the cell, which may cause a reversal of the glutamate transporter and subsequent accumulation of glutamate in the extracellular space. Indeed, Bz-ATP triggered [Ca2+]i elevations in astrocytes in substantia gelatinosa (SG) slices of the rat lumbar spinal cord and thereafter induced in nearby SG neurons slow and irregularly occurring inward currents (SICs) (Bardoni et al., 2010). These SICs were insensitive to TTX but were blocked by AP-5, an antagonist of NMDA-type ionotropic glutamate receptors. Ca2+ imaging revealed that astrocytic glutamate evokes NMDAR-mediated episodes of synchronous activity in groups of SG neurons. A more sustained release of glutamate also occurred from SG astrocytes, which was shown to be inhibited by the P2X7R antagonist A-438079, confirming that a P2X7R is responsible for this type of glutamate release (Fig. 2A,B) (Ficker et al., 2014). Moreover, biosensor measurements indicated that reactive oxygen species are coreleased from astrocytes together with glutamate and potentiate its neuronal effects (Ficker et al., 2014). In perfect agreement with these data, Bz-ATP also induced SICs and tonic currents in CA1 hippocampal pyramidal neurons of rats (Fellin et al., 2006). The SICs were again unaffected by P2X7R antagonism, whereas the tonic currents were sensitive to this pharmacological manipulation, although they did not react to glutamate transporter and gap junction channel inhibitors. It was concluded from these experiments that purinergic receptors other than those belonging to the P2X7 subtype mediate glutamate release that evokes SICs, whereas activation of a P2X7R appears to mediate a sustained glutamate efflux that generates a tonic current in CA1 neurons (Fellin et al., 2006). However, recent experiments in mouse, rather than rat, spinal cord slices showed that, in a subpopulation of SG neurons, the Bz-ATP-induced current is due to a direct P2X7R-mediated effect, which cannot be blocked by a combination of NMDA- and AMPA-type glutamate receptor antagonists (Gao et al., 2017). These results point out the significance of species differences with respect to neuronal P2X7Rs.
Figure 2.
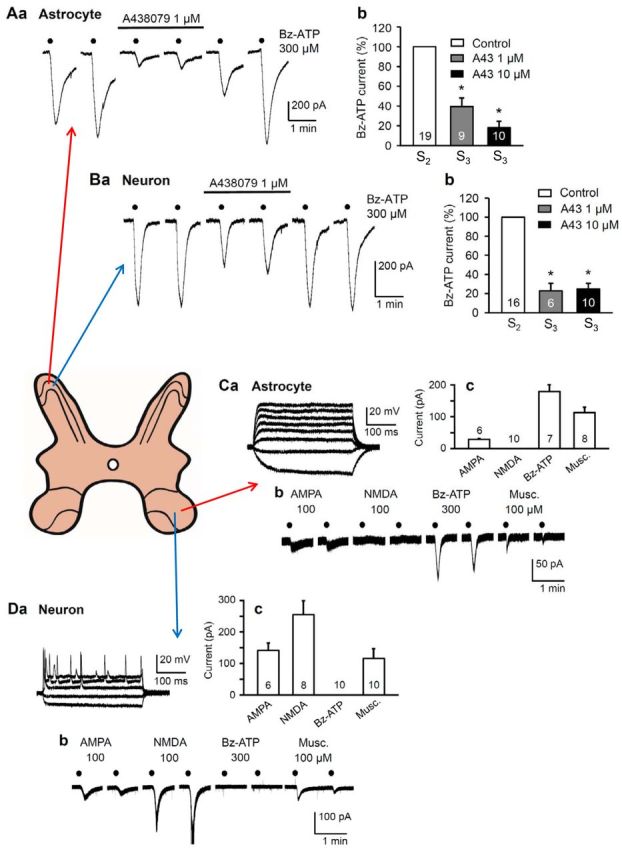
P2X7Rs are located at astrocytes, but not neurons, in the spinal cord dorsal and ventral horns of rats. A–D, Spinal cord slices were made from layers II and VIII of the lumbosacral spinal cord, respectively. Preparation, recording, and drug application protocols were as described by Ficker et al. (2014). In spinal cord slices, astrocytes and neurons were discriminated by a series of current pulses injected into the cell by means of patch-clamp electrodes. Astrocytes responded to depolarizing current pulses with electrotonic membrane potential changes only (Ca), whereas neurons responded above a certain threshold depolarization with series of action potentials (Da). The holding potential was −80 mV for astrocytes and −70 mV for neurons. Membrane currents were induced by 6 consecutive applications of Bz-ATP (300 μm) every 2 min for 10 s in a low Ca2+/no Mg2+-containing external medium (S1-S6). A, Whole-cell recordings of membrane currents in astrocytes from layer II (substantia gelatinosa) of the rat dorsal horn. Aa, The Bz-ATP currents were reversibly depressed by the selective P2X7R antagonist A-438079 (1 μm). Representative tracings. Ab, Percentage mean ± SEM inhibition of the Bz-ATP effect by increasing concentrations of A-438079 (1, 10 μm). The first current response immediately after the application of A-438079 (S3) was compared with the last current response immediately before antagonist application (S2). B, Whole-cell recordings of membrane currents in neurons from layer II of the rat dorsal horn. Ba, Bb, Experiments analogous to those shown in Aa and Ab. *p < 0.05; statistically significant difference from 100% both in Ab and Bb. It is important to note that, in contrast to the astrocytic currents, the neuronal currents were depressed by a combination of the ionotropic glutamate receptor antagonists AP-5 (50 μm) and CNQX (20 μm); a still stronger inhibition was achieved when, in addition, GABAA and glycineA receptors were blocked by gabazine and strychnine (10 μm both). A, B, Modified with permission from Ficker et al. (2014). C, Whole-cell recordings of membrane currents in astrocytes from the layer VIII of the mouse ventral horn. All agonist concentrations were applied every 2 min for 10 s each. Cb, Inward current responses to agonists for ionotropic glutamate receptors (AMPA, 100 μm), P2X7Rs (Bz-ATP; 300 μm), and GABAA-Rs (muscimol; 100 μm), but not to the ionotropic glutamate receptor agonist NMDA (100 μm). Cc, Columns represent the mean of two consecutive responses to each agonist with the corresponding SEM values. D, Whole-cell recordings of membrane currents in neurons from layer VIII of the mouse ventral horn. Db, Inward current responses to AMPA (100 μm), NMDA (100 μm), and muscimol (100 μm), but not to Bz-ATP (300 μm). Dc, Columns represent the mean of two consecutive responses to each agonist with the corresponding SEM values. The number of cells is indicated in each column. C, D, Unpublished experiments of T.M.K. and P.I.
In rat locus ceruleus neurons, contained in a midbrain slice preparation, the frequency, but not amplitude, of glutamatergic spontaneous EPSCs was strongly potentiated by Bz-ATP (Khakpay et al., 2010). BBG as well as fluorocitric acid and aminoadipic acid prevented the activity of Bz-ATP. BBG is a selective antagonist of P2X7Rs; fluorocitric acid and aminoadipic acid are selective astrocytic poisons, reported to interfere with the synthesis, storage, and release of amino acid gliotransmitters (Huck et al., 1984; Clarke, 1991). Hence, it was assumed that the stimulation of astrocytic P2X7Rs leads to the outflow of an unidentified signaling molecule, which presynaptically increases the spontaneous release of glutamate onto LC neurons from their afferent fiber tracts. Interestingly, in the rat hypothalamic paraventricular nucleus, an increase in the amplitude of miniature EPSCs was observed in response to noradrenaline that required the release of ATP from glial cells (Gordon et al., 2005). The increase in quantal efficacy, which likely results from an insertion of AMPA receptors into the postsynaptic membrane, was secondary to the activation of P2X7Rs, increase in postsynaptic Ca2+, and the activation of phosphatidylinositol 3-kinase. The gliotransmitter ATP therefore contributed indirectly to the regulation of the postsynaptic efficacy at glutamatergic synapses in the CNS (Gordon et al., 2005).
Effects attributed to the stimulation of neuronal P2X7Rs may have also other reasons
Conditional humanized P2RX7 mouse
Recently, Metzger et al. (2017) reported the generation of a conditional humanized P2RX7 mouse. This P2RX7 allele is accessible to spatially and temporally controlled Cre recombinase-mediated inactivation. In contrast to previously generated KO mice, none of the described P2X7R splice variants evaded this null allele. By selective disruption and assessment of the humanoid P2X7R expression in different brain regions and cell types, it was demonstrated that the P2X7R-mRNA is expressed in glutamatergic pyramidal neurons of the hippocampal CA3 region. In addition, the receptor message was expressed also in neuroglia (i.e., astrocytes, oligodendrocytes) and microglia in the brain. However, functional experiments, for example, by electrophysiological recordings from CA3 neurons and the target areas of the CA3 axonal projections (e.g., the Schaffer collateral-commissural pathway ending at CA1 neurons), are still indispensably needed to prove the existence of neuronal P2X7Rs.
Cell culture systems
A general problem may arise when data obtained in cell culture systems are uncritically transferred to the in vivo situation. It has been repeatedly reported that astrocytes and neurons may change their functional properties as well as their morphological and immunohistochemical characteristics when kept in cell culture (e.g., Rubini et al., 2006; Nörenberg et al., 2010). For example, the electrophysiological properties of rat striatal neurons dramatically change in dependence on the culture medium used; in neurobasal medium (NBM), they have a low resting membrane potential and fire only a single low amplitude spike in response to depolarizing current injection (Rubini et al., 2006). By contrast, neurons cultured in DMEM have a high resting membrane potential and fire multiple, repetitive spikes in response to the injection of depolarizing current pulses. In addition, NBM, but not DMEM, neurons express immunoreactivity for astrocytic/neural stem cell markers (GFAP, nestin).
P2X7R activation by Bz-ATP has been reported to induce [Ca2+]i transients in the distal domains of their cellular processes rather than in the cell bodies of cultured mouse hippocampal neurons cultured in NBM (Díaz-Hernández et al., 2008; del Puerto et al., 2012). Pharmacological inhibition of P2X7Rs or their silencing by shRNA interference induced longer and more branched axons, coupled with morphological changes to the growth cones. However, Rubini et al. (2014) were unable to reproduce these results in rat hippocampal neurons cultured by identical procedures. Bz-ATP caused large [Ca2+]i transients in the more distal cellular processes only if they grew above a glial carpet, supplying spurious neuronal signals. Moreover, neither the amplitude nor the frequency of sEPSCs measured in rat hippocampal neurons was increased by Bz-ATP; sEPSCs represented the quantal release of glutamate onto neighboring neurons in a network. Thus, even if we assume that cultured mice and rat hippocampal neurons respond in a different manner to Bz-ATP, the general significance of presynaptic P2X7Rs may be questioned in this system. In addition, the control of axonal elongation in cultured neurons by P2X7Rs is an interesting finding, which probably argues for the function of this receptor type in the mouse hippocampus during development, but it fails to prove its neuronal localization in maturity.
Presynaptic P2X7Rs
Another publication had a long-lasting impact on the scientific community by supplying evidence for purported presynaptic P2X7Rs. It was reported that Bz-ATP-inhibits mossy fiber-CA3 pyramidal cell synapses in the rat hippocampus in an oxidized ATP but not PPADS-reversible manner (Armstrong et al., 2002). Several researchers did not notice that, soon after the publication of this article, it was found that, although the findings are correct, their interpretation is misleading. Apparently, the depression of the field EPSPs evoked by mossy fiber stimulation could be fully blocked by the adenosine A1R antagonist 8-cyclopently-1,3-dipropylxanthine (Kukley et al., 2004). Further, the effect of Bz-ATP was mimicked by ATP and adenosine and was completely prevented by adenosine-deaminase, an enzyme specifically degrading adenosine. Last but not least, oxidized ATP is a slowly equilibrating and irreversible, nonselective antagonist of P2X7Rs, which also interferes with inflammatory signal processing and is by itself seriously cytotoxic (Sperlágh et al., 2006). Hence, Kukley et al. (2004) concluded that Bz-ATP is extracellularly catabolized to Bz-adenosine, subsequently hetero-exchanged for intracellular adenosine from neuronal or astrocytic pools, and then depresses mossy fiber excitatory synaptic potentials through presynaptic A1Rs rather than through P2X7Rs.
Spinal cord ventral horn neurons
Weight drop impact inflicted upon the spinal cord of Sprague Dawley rats caused injury and a large increase in the extracellular ATP concentration (Wang et al., 2004; Peng et al., 2009). This ATP induced in neurons an irreversible rise of cytosolic Ca2+ and subsequently cell death. P2X7R antagonism by oxidized ATP, PPADS, and BBG significantly improved functional recovery and diminished cell death in the peritraumatic zone. It was concluded that P2X7Rs are located at the spinal ventral horn neurons themselves because ATP or Bz-ATP induced in these neurons high-frequency spiking with little tendency to inactivate. The increased spiking activity was attenuated by pretreatment with oxidized ATP, but not by combined treatment with AMPA and NMDA receptor antagonists. In similar experiments, Bz-ATP (300 μm) failed to induce inward current in layer VIII neurons of lumbosacral spinal cord slices of Wistar rats (Fig. 2D). By contrast, AMPA, NMDA, and muscimol (100 μm each) caused inward current responses. At the same time, Bz-ATP stimulated the corresponding layer VIII astrocytes, just as AMPA (100 μm) and muscimol (100 μm) did, whereas NMDA (100 μm) was ineffective (Fig. 2C). The disparate results once again point out the extreme species- and strain-dependent variability in the presence or absence of neuronal P2X7Rs and definitely raise doubts about the general functional significance of this receptor type as localized at ventral spinal cord neurons.
Neuroglial P2X7Rs may be involved in secondary neurodegeneration and neurodegenerative illnesses
Trauma, ischemia, and stroke
Immunohistochemical investigations in the nucleus accumbens of rats showed that after stab wound injury previously absent P2X7R-IR was observed on cells labeled for the astrocytic marker GFAP (Franke et al., 2001; Burnstock et al., 2011; Pedata et al., 2016). Very similar findings were reported for the cerebral cortex of spontaneously hypertensive rats, where MCAO led to the upregulation of P2X7R-IR in the penumbra surrounding the necrotic region in the brain (Franke et al., 2004). In support of a secondary damage caused by ATP-induced stimulation of P2X7Rs in the penumbra, the blockade of these receptors by the selective antagonist BBG attenuated postischemic damage after MCAO (Arbeloa et al., 2012; Cisneros-Mejorado et al., 2015). Similarly, inhibition of P2X7Rs ameliorated global cerebral ischemia/reperfusion injury as evidenced by survival rate, neuronal death in the hippocampal CA1 region, and learning memory deficits (Chu et al., 2012; Yu et al., 2013). P2X7Rs appeared to be involved in cerebral edema and neurological damage after traumatic brain injury as well; these receptors were localized at astrocytic end-feet, and the application of BBG decreased the expression of GFAP and aquaporin-4, an astrocytic water channel that promotes cellular edema (Kimbler et al., 2012).
Oxygen/glucose deficiency led to anoxic depolarization in cultured cortical neurons (Arbeloa et al., 2012) and oligodendrocytes (Domercq et al., 2010), which was due to the combined activation of the excitotoxic NMDA and P2X7Rs. Embryonic rat cortical neurons were cultured in NBM to obtain astrocyte- and microglia-free preparations (Arbeloa et al., 2012). However, this procedure has been shown to alter both the immunohistochemical characteristics and functional properties of cultured rat striatal neurons, making a comparison with the in vivo situation questionable (Rubini et al., 2006). In neurons and oligodendrocytes, blocking a subtype of ionotropic glutamate receptors (NMDA, AP-5) and P2X7Rs (BBG) nearly abolished the ischemia-induced cell death (Domercq et al., 2010; Arbeloa et al., 2012). Nonetheless, another study yielded divergent results for hippocampal CA1 neurons and astrocytes studied in their natural environment in brain slice preparations; whereas AP-5 depressed the anoxic depolarization, the selective P2X7R antagonist A-438079 failed to alter it (Leichsenring et al., 2013). Hence, the available evidence fails to support the notion that hypoxia/ischemia induces anoxic depolarization by the activation of neuronal P2X7Rs under in situ conditions.
Ischemic preconditioning using short-lasting MCAO in mice protected from the consequences of a subsequent severe MCAO (Hirayama et al., 2015). The spatiotemporal pattern of astrocytic, but not microglial, activation correlated well with that of ischemic tolerance; inhibiting astrocytes with fluorocitric acid abolished the induction of ischemic tolerance, which was also absent in P2X7R knock-out mice.
Temporal lobe epilepsy
The temporal lobe epilepsy syndrome can be modeled in rodents by chemical induction of status epilepticus using systemic kainic acid or pilocarpine (Kandratavicius et al., 2014). Soon after pilocarpine-induced status epilepticus in rats (12 h), there was no somatic staining for P2X7R-IR in CA1 or CA3 pyramidal neurons, but a pronounced labeling in stratum oriens astrocytes (Doná et al., 2009). At later time points (7 d) after chemically induced epilepsy in rodents, the IR was upregulated in granule neurons and microglia, paralleled by astroglial loss (Kim et al., 2009). Intracerebroventricular application of P2X7R antagonists (BBG, A-438079) attenuated seizures induced by unilateral kainic acid injection into the amygdala of mice (Engel et al., 2012, 2016; Henshall et al., 2013). In this model, Western blotting revealed increased P2X7R protein levels in the CA1 and dentate gyrus samples; EGFP-IR was accordingly upregulated in the dentate gyrus granule cell layer of Tg(P2RX7-EGFP) mice. These mice express EGFP immediately downstream of the P2RX7 promoter; therefore, EGFP antibodies reliably label P2X7Rs. It has been confirmed that P2X7R protein was increased in the hippocampal subfields of mice that underwent status epilepticus and in the resected hippocampus from patients with pharmaco-resistant temporal lobe epilepsy (Jimenez-Pacheco et al., 2016).
Tg(P2RX7-EGFP) mice developed recurrent spontaneous seizures within 5 d after intra-amygdala kainic acid-induced status epilepticus. In these animals, it was shown that, 21 d after status epilepticus, the expression of P2X7Rs was increased in CA1 pyramidal cells and dentate granule neurons, as well as microglia, but not astrocytes (Jimenez-Pacheco et al., 2016). However, another group of authors found that systemic kainic acid or pilocarpine-induced status epilepticus already 24 h after status epilepticus potentiated Bz-ATP currents in astrocyte-like neural progenitor cells (NPCs) of the subgranule zone of Tg(nestin-EGFP) mice (Rozmer et al., 2016; Tang and Illes, 2016). The nestin-positive NPCs of the subgranular zone generate neuroblasts and then granule cells in the dentate gyrus. Rozmer et al. (2016) also reported that neither neuroblasts nor mature granule cells of the dentate gyrus projecting to interneurons and pyramidal neurons of the CA3 area bear functional P2X7Rs. They raised the hypothesis that status epilepticus may cause a proliferation of the subgranular zone NPCs via P2Y1R activation. Such NPCs express necrotic/apoptotic P2X7Rs decreasing the overtly large cell number, which would lead otherwise to a chronic manifestation of recurrent epileptic fits by the generation of ectopically localized granule cells in the hilus hippocampi. Once again, both a neuronal and astrocytic localization of P2X7Rs might explain the secondary neurodegeneration following a status epilepticus. It has to be noted that acute epileptic seizures are inhibited by P2X7R antagonistic drugs (Engel et al., 2012, 2016; Jimenez-Pacheco et al., 2016), whereas recurrent epileptic seizures developing after a one-time status epilepticus are promoted by P2X7R antagonists (Rozmer et al., 2016). In conclusion, evidence for the involvement of neuronal P2X7Rs in the antiepileptic effect of BBG and A-438079 are possible but not unequivocally confirmed.
Diseases of the retina
Genetic association studies have implicated purinergic receptors in the development of age-related macular degeneration (Gu et al., 2013). It was suggested that P2X7Rs identified by their immunoreactivity at retinal ganglion cells may be the reason for the observed apoptotic reactions (Franke et al., 2005). In human organotypic retinal cultures, oxygen-glucose deprivation also caused a BBG-antagonizable cell death of the ganglion cell layer (Niyadurupola et al., 2013). To identify other cell types influenced by P2X7R activation, we searched for P2X7Rs in astroglia-like Müller cells. In the human retina, P2X7Rs are present in Müller cells, where they seem to function as targets of ATP, coupled with Ca2+-mediated signaling pathways (Pannicke et al., 2000; Wurm et al., 2011; Reichenbach and Bringmann, 2016). Hence, Müller cells of healthy humans, but not those of rats and mice, appear to possess functional P2X7Rs. In preparations obtained from patients suffering of proliferative vitreoretinopathy, stimulation by Bz-ATP induced larger membrane currents than in preparations from healthy patients (Bringmann et al., 2001). Currents through Ca2+-activated K+ channels of high conductance that reflect the increase of the intracellular Ca2+ concentration became also larger in consequence of proliferative vitreoretinopathy.
Neuropathic and inflammatory pain
The peripheral equivalents of astrocytes, the so-called satellite glial cells located in sensory ganglia, were reported to bear functional P2X7Rs (X. F. Zhang et al., 2005). Thus, it was suggested that ATP released from DRG neurons may activate P2X7Rs at satellite cells, which in consequence release TNF-α potentiating P2X3R-mediated responses at nearby neurons and thereby regulate pain sensation (X. Zhang et al., 2007). Another group of authors published just opposite results (Chen et al., 2008; Villa et al., 2010). They reported that satellite cells of DRGs tonically inhibit the expression of P2X3Rs in neurons. Reducing P2X7R expression using siRNA or blocking P2X7R activity by antagonists elicited P2X3R upregulation, increased the activity of sensory neurons responding to painful stimuli, and evoked abnormal nociceptive behaviors in rats (hyperalgesia, allodynia). It was hypothesized that activation of P2X7Rs evoked ATP release from satellite cells. ATP in turn stimulated P2Y1Rs in neurons. P2Y1R activation was then necessary and sufficient for the inhibitory control of P2X3R expression. Because P2X7R blockade or its deletion has been shown to ameliorate neuropathic and inflammatory pain (Chessell et al., 2005; McGaraughty et al., 2007), only the increase of P2X3R sensitivity by P2X7R activation would make sense from a pathophysiological view. However, both sets of data support the notion that neurons and surrounding glial cells in DRGs communicate with each other via signaling molecules; this communication has implications for the manifestation of chronic pain (Gosselin et al., 2010). Later it has been pointed out that Cx hemichannels and Panx channels serve as paracrine communication pathways between satellite cells and neurons by modulating the excitability of sensory neurons (Retamal et al., 2014).
The primary mechanism of P2X7R stimulation in neuropathic/inflammatory pain may be the release of IL-1β from activated microglia/macrophages (Clark et al., 2010; Burnstock, 2016). In spinal cord slices taken from wild-type mice, the LPS-induced release of IL-1β was prevented by pharmacological inhibition of the P2X7R with A-438079 and was absent in P2RX7 knock-out animals. Application of ATP did not deliver IL-1β from the dorsal horn unless preceded by a LPS priming stimulus. In behavioral studies, intrathecal injection of LPS in the lumbar spinal cord produced mechanical hyperalgesia in rat hindpaws, which was attenuated by concomitant injections of A-438079 (Clark et al., 2010). The astrocytic release of glutamate initiated by P2X7R stimulation might also be a reason for neuropathic pain (Andó and Sperlágh, 2013). Perfusion of spinal cord slices with ATP elicited larger [3H]glutamate release from spinal cord slices of sciatic nerve-ligated mice (model of neuropathic pain) than from their nonoperated counterparts. Moreover, ATP failed to release [3H]glutamate from spinal cord slices of P2RX7−/− animals or from analogous preparations of P2RX7+/+ animals when electrically stimulated. Hence, glutamate release facilitated by P2X7Rs situated extrasynaptically or on non-neuronal cells (i.e., astrocytes or microglia) may be causally involved in neuropathic pain.
Neurodegenerative diseases
Alzheimer's disease (AD), Parkinson's disease, Huntington's disease, multiple sclerosis, and amyotrophic lateral sclerosis develop on the basis of endogenous/genetic or toxic/infectious factors. However, it is a common characteristic of these diseases that superimposed on neuronal degeneration caused by disease-specific factors secondary neuroinflammation with consecutive neurodegeneration develops because of the deleterious activation of P2X7Rs at microglia (see Introduction) (Sperlágh et al., 2006; Burnstock et al., 2011; Skaper et al., 2011; Turola et al., 2012; Sperlágh and Illes, 2014).
A neuropathological hallmark of AD is the appearance of plaques consisting of extracellular β-amyloid peptide surrounded by reactive microglial cells (Sanz et al., 2009). The amyloid precursor protein is cleaved by β- and γ-secretases to generate β-amyloid peptide, which triggers increases in [Ca2+]i, ATP release, IL-1β secretion, and plasma membrane permeabilization in microglia which consecutively damage neurons. In contrast to microglial P2X7Rs, the same receptors at Neuro2a mouse neuroblastoma cells, human neuroblastoma cells, and mouse primary astrocytes or neural progenitor cells were reported to favorably influence AD-like pathologies (Delarasse et al., 2011). P2X7Rs may stimulate nonamyloidogenic processing of the amyloid precursor protein by α-secretases, leading to proteolytic cleavage within the Aβ peptide sequence and shedding of the soluble amyloid precursor protein ectodomain, which in turn has been reported to exert neuroprotection. Because in vivo applied P2X7R antagonists have been shown to slow down amyloid plaque deposition and disease progression (Díaz-Hernández et al., 2012; Rodrigues et al., 2015), the microglial and astroglial P2X7Rs may display contrasting effects, with the microglial receptor dominating over the astroglial one.
Huntington's disease (HD) is a dominant inherited motor disorder, characterized by progressive striatal neurodegeneration. The mutation responsible for this fatal disease is an abnormally expanded and unstable CAG (cysteine-alanine-glycine) repeat within the coding region of the gene encoding the huntingtin protein, which damages GABAergic output neurons of the basal ganglia. Using mouse and cellular HD models, Díaz-Hernández et al. (2009) demonstrated increased P2X7R levels and altered P2X7R-mediated Ca2+ permeability in somata and terminals of HD neurons. In vivo administration of the P2X7R antagonist BBG to HD mouse models prevented neuronal apoptosis and attenuated body weight loss and motor coordination deficits. However, these data once again do not allow deciding whether the neuronal changes are the immediate causes of the expression of P2X7Rs or whether they are due to astrocytic/microglial signaling mechanisms.
Experimental autoimmune encephalomyelitis (EAE) is an animal model of multiple sclerosis. Matute et al. (2007) identified functional P2X7Rs at oligodendrocytes ensheathing the isolated rat optic nerve and observed that these receptors could kill oligodendrocytes after their activation by ATP. In addition, oligodendrocytic P2X7R-IR was increased in optic nerve samples obtained at autopsy of human patients, and P2X7R antagonism by BBG ameliorated motor deficits in EAE by reducing demyelination and thus improving axonal conduction. In another study, the overexpression of P2X7R-IR was detected at neurons and astrocytes in EAE (Grygorowicz et al., 2010). Although infiltrating cells were detected in brains of both P2RX7−/− and wild-type mice, astroglial activation and axonal damage were reduced in the receptor-deficient animals (Sharp et al., 2008). In contrast, microglial activation was similar in the two strains. These results point to the role of infiltrating T cells as pathological factors and, in addition, point to a role of astroglial P2X7Rs in the development of EAE. In addition, P2RX7−/− mice showed an enhanced susceptibility to EAE, reflecting a loss of apoptotic activity in lymphocytes; this finding supports an important role for this receptor in lymphocyte homeostasis (Chen and Brosnan, 2006). Whereas Sharp et al. (2008) (Glaxo mouse) reported a protective effect in case of P2RX7 deletion, Chen and Brosnan (2006) (Pfizer mouse) found a worsening of the EAE symptoms in the respective knock-out animals. Hence, specific P2X7R isoforms evading inactivation in the Glaxo or Pfizer mice could explain the differences in the functional consequences of P2RX7 deficiency in the respective knock-outs.
Transgenic mice overexpressing human superoxide dismutase 1 (SOD1) are an accepted model of amyotrophic lateral sclerosis (Maier and Chan, 2002). In SOD1 mixed astrocytic/neuronal cultures, stimulation by Bz-ATP caused astrocytes to become neurotoxic, inducing death of motor neurons. Involvement of P2X7Rs was confirmed by BBG inhibition of ATP/Bz-ATP effects (Gandelman et al., 2010). Furthermore, in SOD1-G93A microglial cultures, the stimulation of P2X7Rs by Bz-ATP activated potentially neurotoxic signaling pathways, which were interrupted by knocking out P2X7Rs or were inhibited by P2X7R antagonists (Apolloni et al., 2013a). In contrast to these results, the same group of researchers reported, by using P2X7R-deficient SOD1-G93A mice, that the clinical onset was significantly anticipated and the disease progression worsened in comparison with the normal SOD1-G93A mice (Apolloni et al., 2013b). We have no apparent explanation for this discrepancy.
In conclusion, there is an ongoing and repeatedly escalating discussion about the presence or absence of neuronal P2X7Rs. In the early embryo, the neuroectoderm gives rise to both neurons and neuroglial cells (e.g., astrocytes); microglial cells are of mesenchymal origin. In addition to their direct or indirect participation in neuronal signaling, astrocytes, when activated, also exert immunological (microglia-like) functions in secreting cytokines/chemokines and in phagocytosing cellular debris or microorganisms. By contrast, neurons are specialized to receive, process, and transmit information through electrical and chemical signals. Microglia are resident immunocytes of the peripheral and CNS, which constitute the first defense line in case of an infection. By inspecting the relevant literature, we believe that, although the existence of the necrotic/apoptotic P2X7Rs at neurons cannot be excluded in all cases, primary modulation of neuronal activities by these receptors is with great likelihood usually due to indirect effects exerted by the stimulation of neuroglial/microglial P2X7Rs.
Footnotes
Dual Perspectives Companion Paper: Neuronal P2X7 Receptor: Involvement in Neuronal Physiology and Pathology, by M. Teresa Miras-Portugal, Álvaro Sebastián-Serrano, Laura de Diego García, and Miguel Díaz-Hernández
This study was supported by Deutsche Forschungsgemeinschaft Grant IL 20/21-1 and Sino-German Center for the Promotion of Science GZ 919. T.M.K. was supported by Deutsche Akademische Austauschdienst scholarship.
The authors declare no competing financial interests.
References
- Abbracchio MP, Burnstock G (1994) Purinoceptors: are there families of P2X and P2Y purinoceptors? Pharmacol Ther 64:445–475. 10.1016/0163-7258(94)00048-4 [DOI] [PubMed] [Google Scholar]
- Adinolfi E, Cirillo M, Woltersdorf R, Falzoni S, Chiozzi P, Pellegatti P, Callegari MG, Sandonà D, Markwardt F, Schmalzing G, Di Virgilio F (2010) Trophic activity of a naturally occurring truncated isoform of the P2X7 receptor. FASEB J 24:3393–3404. 10.1096/fj.09-153601 [DOI] [PubMed] [Google Scholar]
- Adriouch S, Bannas P, Schwarz N, Fliegert R, Guse AH, Seman M, Haag F, Koch-Nolte F (2008) ADP-ribosylation at R125 gates the P2X7 ion channel by presenting a covalent ligand to its nucleotide binding site. FASEB J 22:861–869. 10.1096/fj.07-9294com [DOI] [PubMed] [Google Scholar]
- Alberto AV, Faria RX, Couto CG, Ferreira LG, Souza CA, Teixeira PC, Fróes MM, Alves LA (2013) Is pannexin the pore associated with the P2X7 receptor? Naunyn Schmiedebergs Arch Pharmacol 386:775–787. 10.1007/s00210-013-0868-x [DOI] [PubMed] [Google Scholar]
- Alloisio S, Cervetto C, Passalacqua M, Barbieri R, Maura G, Nobile M, Marcoli M (2008) Functional evidence for presynaptic P2X7 receptors in adult rat cerebrocortical nerve terminals. FEBS Lett 582:3948–3953. 10.1016/j.febslet.2008.10.041 [DOI] [PubMed] [Google Scholar]
- Anderson CM, Nedergaard M (2006) Emerging challenges of assigning P2X7 receptor function and immunoreactivity in neurons. Trends Neurosci 29:257–262. 10.1016/j.tins.2006.03.003 [DOI] [PubMed] [Google Scholar]
- Andó RD, Sperlágh B (2013) The role of glutamate release mediated by extrasynaptic P2X7 receptors in animal models of neuropathic pain. Brain Res Bull 93:80–85. 10.1016/j.brainresbull.2012.09.016 [DOI] [PubMed] [Google Scholar]
- Apolloni S, Parisi C, Pesaresi MG, Rossi S, Carrì MT, Cozzolino M, Volonté C, D'Ambrosi N (2013a) The NADPH oxidase pathway is dysregulated by the P2X7 receptor in the SOD1–G93A microglia model of amyotrophic lateral sclerosis. J Immunol 190:5187–5195. 10.4049/jimmunol.1203262 [DOI] [PubMed] [Google Scholar]
- Apolloni S, Amadio S, Montilli C, Volonté C, D'Ambrosi N (2013b) Ablation of P2X7 receptor exacerbates gliosis and motoneuron death in the SOD1–G93A mouse model of amyotrophic lateral sclerosis. Hum Mol Genet 22:4102–4116. 10.1093/hmg/ddt259 [DOI] [PubMed] [Google Scholar]
- Araque A, Carmignoto G, Haydon PG, Oliet SH, Robitaille R, Volterra A (2014) Gliotransmitters travel in time and space. Neuron 81:728–739. 10.1016/j.neuron.2014.02.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arbeloa J, Pérez-Samartin A, Gottlieb M, Matute C (2012) P2X7 receptor blockade prevents ATP excitotoxicity in neurons and reduces brain damage after ischemia. Neurobiol Dis 45:954–961. 10.1016/j.nbd.2011.12.014 [DOI] [PubMed] [Google Scholar]
- Armstrong JN, Brust TB, Lewis RG, MacVicar BA (2002) Activation of presynaptic P2X7-like receptors depresses mossy fiber-CA3 synaptic transmission through p38 mitogen-activated protein kinase. J Neurosci 22:5938–5945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Avendano BC, Montero TD, Chavez CE, von Bernhardi R, Orellana JA (2015) Prenatal exposure to inflammatory conditions increases Cx43 and Panx1 unopposed channel opening and activation of astrocytes in the offspring effect on neuronal survival. Glia 63:2058–2072. 10.1002/glia.22877 [DOI] [PubMed] [Google Scholar]
- Bardoni R, Ghirri A, Zonta M, Betelli C, Vitale G, Ruggieri V, Sandrini M, Carmignoto G (2010) Glutamate-mediated astrocyte-to-neuron signalling in the rat dorsal horn. J Physiol 588:831–846. 10.1113/jphysiol.2009.180570 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bargiotas P, Monyer H, Schwaninger M (2009) Hemichannels in cerebral ischemia. Curr Mol Med 9:186–194. 10.2174/156652409787581646 [DOI] [PubMed] [Google Scholar]
- Bartlett R, Stokes L, Sluyter R (2014) The P2X7 receptor channel: recent developments and the use of P2X7 antagonists in models of disease. Pharmacol Rev 66:638–675. 10.1124/pr.113.008003 [DOI] [PubMed] [Google Scholar]
- Beckel JM, Argall AJ, Lim JC, Xia J, Lu W, Coffey EE, Macarak EJ, Shahidullah M, Delamere NA, Zode GS, Sheffield VC, Shestopalov VI, Laties AM, Mitchell CH (2014) Mechanosensitive release of adenosine 5′-triphosphate through pannexin channels and mechanosensitive upregulation of pannexin channels in optic nerve head astrocytes: a mechanism for purinergic involvement in chronic strain. Glia 62:1486–1501. 10.1002/glia.22695 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bennett MV, Garré JM, Orellana JA, Bukauskas FF, Nedergaard M, Sáez JC (2012) Connexin and pannexin hemichannels in inflammatory responses of glia and neurons. Brain Res 1487:3–15. 10.1016/j.brainres.2012.08.042 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bhattacharya A, Biber K (2016) The microglial ATP-gated ion channel P2X7 as a CNS drug target. Glia 64:1772–1787. 10.1002/glia.23001 [DOI] [PubMed] [Google Scholar]
- Bringmann A, Pannicke T, Moll V, Milenkovic I, Faude F, Enzmann V, Wolf S, Reichenbach A (2001) Upregulation of P2X7 receptor currents in Muller glial cells during proliferative vitreoretinopathy. Invest Ophthalmol Vis Sci 42:860–867. [PubMed] [Google Scholar]
- Burnstock G. (2013) Introduction and perspective, historical note. Front Cell Neurosci 7:227. 10.3389/fncel.2013.00227 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burnstock G. (2016) Purinergic mechanisms and pain. Adv Pharmacol 75:91–137. 10.1016/bs.apha.2015.09.001 [DOI] [PubMed] [Google Scholar]
- Burnstock G, Krügel U, Abbracchio MP, Illes P (2011) Purinergic signalling: from normal behaviour to pathological brain function. Prog Neurobiol 95:229–274. 10.1016/j.pneurobio.2011.08.006 [DOI] [PubMed] [Google Scholar]
- Chaumont S, Khakh BS (2008) Patch-clamp coordinated spectroscopy shows P2X2 receptor permeability dynamics require cytosolic domain rearrangements but not Panx-1 channels. Proc Natl Acad Sci U S A 105:12063–12068. 10.1073/pnas.0803008105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheewatrakoolpong B, Gilchrest H, Anthes JC, Greenfeder S (2005) Identification and characterization of splice variants of the human P2X7 ATP channel. Biochem Biophys Res Commun 332:17–27. 10.1016/j.bbrc.2005.04.087 [DOI] [PubMed] [Google Scholar]
- Chen L, Brosnan CF (2006) Exacerbation of experimental autoimmune encephalomyelitits in P2X7R−/− mice: evidence for loss of apoptotic activity in lymphocytes. J Immunol 176:3115–3126. 10.4049/jimmunol.176.5.3115 [DOI] [PubMed] [Google Scholar]
- Chen Y, Zhang X, Wang C, Li G, Gu Y, Huang LY (2008) Activation of P2X7 receptors in glial satellite cells reduces pain through downregulation of P2X3 receptors in nociceptive neurons. Proc Natl Acad Sci U S A 105:16773–16778. 10.1073/pnas.0801793105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chessell IP, Hatcher JP, Bountra C, Michel AD, Hughes JP, Green P, Egerton J, Murfin M, Richardson J, Peck WL, Grahames CB, Casula MA, Yiangou Y, Birch R, Anand P, Buell GN (2005) Disruption of the P2X7 purinoceptor gene abolishes chronic inflammatory and neuropathic pain. Pain 114:386–396. 10.1016/j.pain.2005.01.002 [DOI] [PubMed] [Google Scholar]
- Chu K, Yin B, Wang J, Peng G, Liang H, Xu Z, Du Y, Fang M, Xia Q, Luo B (2012) Inhibition of P2X7 receptor ameliorates transient global cerebral ischemia/reperfusion injury via modulating inflammatory responses in the rat hippocampus. J Neuroinflammation 9:69. 10.1186/1742-2094-9-69 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cisneros-Mejorado A, Gottlieb M, Cavaliere F, Magnus T, Koch-Nolte F, Scemes E, Pérez-Samartín A, Matute C (2015) Blockade of P2X7 receptors or pannexin-1 channels similarly attenuates postischemic damage. J Cereb Blood Flow Metab 35:843–850. 10.1038/jcbfm.2014.262 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clark AK, Staniland AA, Marchand F, Kaan TK, McMahon SB, Malcangio M (2010) P2X7-dependent release of interleukin-1β and nociception in the spinal cord following lipopolysaccharide. J Neurosci 30:573–582. 10.1523/JNEUROSCI.3295-09.2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clarke DD. (1991) Fluoroacetate and fluorocitrate: mechanism of action. Neurochem Res 16:1055–1058. 10.1007/BF00965850 [DOI] [PubMed] [Google Scholar]
- Collo G, Neidhart S, Kawashima E, Kosco-Vilbois M, North RA, Buell G (1997) Tissue distribution of the P2X7 receptor. Neuropharmacology 36:1277–1283. 10.1016/S0028-3908(97)00140-8 [DOI] [PubMed] [Google Scholar]
- Costa-Junior HM, Sarmento Vieira F, Coutinho-Silva R (2011) C terminus of the P2X7 receptor: treasure hunting. Purinergic Signal 7:7–19. 10.1007/s11302-011-9215-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- del Puerto A, Díaz-Hernández JI, Tapia M, Gómez-Villafuertes R, Benitez MJ, Zhang J, Miras-Portugal MT, Wandosell F, Díaz-Hernández M, Garrido JJ (2012) Adenylate cyclase 5 coordinates the action of ADP, P2Y1, P2Y13 and ATP-gated P2X7 receptors on axonal elongation. J Cell Sci 125:176–188. 10.1242/jcs.091736 [DOI] [PubMed] [Google Scholar]
- Delarasse C, Auger R, Gonnord P, Fontaine B, Kanellopoulos JM (2011) The purinergic receptor P2X7 triggers α-secretase-dependent processing of the amyloid precursor protein. J Biol Chem 286:2596–2606. 10.1074/jbc.M110.200618 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deuchars SA, Atkinson L, Brooke RE, Musa H, Milligan CJ, Batten TF, Buckley NJ, Parson SH, Deuchars J (2001) Neuronal P2X7 receptors are targeted to presynaptic terminals in the central and peripheral nervous systems. J Neurosci 21:7143–7152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Di Virgilio F. (2007) Liaisons dangereuses: P2X7 and the inflammasome. Trends Pharmacol Sci 28:465–472. 10.1016/j.tips.2007.07.002 [DOI] [PubMed] [Google Scholar]
- Díaz-Hernández JI, Gómez-Villafuertes R, León-Otegui M, Hontecillas-Prieto L, Del Puerto A, Trejo JL, Lucas JJ, Garrido JJ, Gualix J, Miras-Portugal MT, Díaz-Hernández M (2012) In vivo P2X7 inhibition reduces amyloid plaques in Alzheimer's disease through GSK3β and secretases. Neurobiol Aging 33:1816–1828. 10.1016/j.neurobiolaging.2011.09.040 [DOI] [PubMed] [Google Scholar]
- Díaz-Hernández M, del Puerto A, Díaz-Hernández JI, Diez-Zaera M, Lucas JJ, Garrido JJ, Miras-Portugal MT (2008) Inhibition of the ATP-gated P2X7 receptor promotes axonal growth and branching in cultured hippocampal neurons. J Cell Sci 121:3717–3728. 10.1242/jcs.034082 [DOI] [PubMed] [Google Scholar]
- Díaz-Hernández M, Diez-Zaera M, Sánchez-Nogueiro J, Gómez-Villafuertes R, Canals JM, Alberch J, Miras-Portugal MT, Lucas JJ (2009) Altered P2X7-receptor level and function in mouse models of Huntington's disease and therapeutic efficacy of antagonist administration. FASEB J 23:1893–1906. 10.1096/fj.08-122275 [DOI] [PubMed] [Google Scholar]
- Di Castro MA, Chuquet J, Liaudet N, Bhaukaurally K, Santello M, Bouvier D, Tiret P, Volterra A (2011) Local Ca2+ detection and modulation of synaptic release by astrocytes. Nat Neurosci 14:1276–1284. 10.1038/nn.2929 [DOI] [PubMed] [Google Scholar]
- Di Virgilio F, Borea PA, Illes P (2001) P2 receptors meet the immune system. Trends Pharmacol Sci 22:5–7. 10.1016/S0165-6147(00)01574-1 [DOI] [PubMed] [Google Scholar]
- Domercq M, Perez-Samartin A, Aparicio D, Alberdi E, Pampliega O, Matute C (2010) P2X7 receptors mediate ischemic damage to oligodendrocytes. Glia 58:730–740. 10.1002/glia.20958 [DOI] [PubMed] [Google Scholar]
- Doná F, Ulrich H, Persike DS, Conceição IM, Blini JP, Cavalheiro EA, Fernandes MJ (2009) Alteration of purinergic P2X4 and P2X7 receptor expression in rats with temporal-lobe epilepsy induced by pilocarpine. Epilepsy Res 83:157–167. 10.1016/j.eplepsyres.2008.10.008 [DOI] [PubMed] [Google Scholar]
- Duan S, Neary JT (2006) P2X7 receptors: properties and relevance to CNS function. Glia 54:738–746. 10.1002/glia.20397 [DOI] [PubMed] [Google Scholar]
- Duan S, Anderson CM, Keung EC, Chen Y, Chen Y, Swanson RA (2003) P2X7 receptor-mediated release of excitatory amino acids from astrocytes. J Neurosci 23:1320–1328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Engel T, Gómez-Villafuertes R, Tanaka K, Mesuret G, Sanz-Rodriguez A, Garcia-Huerta P, Miras-Portugal MT, Henshall DC, Díaz-Hernández M (2012) Seizure suppression and neuroprotection by targeting the purinergic P2X7 receptor during status epilepticus in mice. FASEB J 26:1616–1628. 10.1096/fj.11-196089 [DOI] [PubMed] [Google Scholar]
- Engel T, Alves M, Sheedy C, Henshall DC (2016) ATPergic signalling during seizures and epilepsy. Neuropharmacology 104:140–153. 10.1016/j.neuropharm.2015.11.001 [DOI] [PubMed] [Google Scholar]
- Fellin T, Pozzan T, Carmignoto G (2006) Purinergic receptors mediate two distinct glutamate release pathways in hippocampal astrocytes. J Biol Chem 281:4274–4284. 10.1074/jbc.M510679200 [DOI] [PubMed] [Google Scholar]
- Feng YH, Wang L, Wang Q, Li X, Zeng R, Gorodeski GI (2005) ATP stimulates GRK-3 phosphorylation and β-arrestin-2-dependent internalization of P2X7 receptor. Am J Physiol Cell Physiol 288:C1342–C1356. 10.1152/ajpcell.00315.2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ficker C, Rozmer K, Kató E, Andó RD, Schumann L, Krügel U, Franke H, Sperlágh B, Riedel T, Illes P (2014) Astrocyte-neuron interaction in the substantia gelatinosa of the spinal cord dorsal horn via P2X7 receptor-mediated release of glutamate and reactive oxygen species. Glia 62:1671–1686. 10.1002/glia.22707 [DOI] [PubMed] [Google Scholar]
- Fischer W, Nörenberg W, Franke H, Schaefer M, Illes P (2009a) Increase of intracellular Ca2+ by P2Y but not P2X receptors in cultured cortical multipolar neurons of the rat. J Comp Neurol 516:343–359. 10.1002/cne.22079 [DOI] [PubMed] [Google Scholar]
- Fischer W, Appelt K, Grohmann M, Franke H, Nörenberg W, Illes P (2009b) Increase of intracellular Ca2+ by P2X and P2Y receptor-subtypes in cultured cortical astroglia of the rat. Neuroscience 160:767–783. 10.1016/j.neuroscience.2009.02.026 [DOI] [PubMed] [Google Scholar]
- Franke H, Grosche J, Schädlich H, Krügel U, Allgaier C, Illes P (2001) P2X receptor expression on astrocytes in the nucleus accumbens of rats. Neuroscience 108:421–429. 10.1016/S0306-4522(01)00416-X [DOI] [PubMed] [Google Scholar]
- Franke H, Günther A, Grosche J, Schmidt R, Rossner S, Reinhardt R, Faber-Zuschratter H, Schneider D, Illes P (2004) P2X7 receptor expression after ischemia in the cerebral cortex of rats. J Neuropathol Exp Neurol 63:686–699. 10.1093/jnen/63.7.686 [DOI] [PubMed] [Google Scholar]
- Franke H, Klimke K, Brinckmann U, Grosche J, Francke M, Sperlágh B, Reichenbach A, Liebert UG, Illes P (2005) P2X7 receptor-mRNA and -protein in the mouse retina: changes during retinal degeneration in BALBCrds mice. Neurochem Int 47:235–242. 10.1016/j.neuint.2005.04.022 [DOI] [PubMed] [Google Scholar]
- Franke H, Verkhratsky A, Burnstock G, Illes P (2012) Pathophysiology of astroglial purinergic signalling. Purinergic Signal 8:629–657. 10.1007/s11302-012-9300-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fuller SJ, Stokes L, Skarratt KK, Gu BJ, Wiley JS (2009) Genetics of the P2X7 receptor and human disease. Purinergic Signal 5:257–262. 10.1007/s11302-009-9136-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fumagalli M, Brambilla R, D'Ambrosi N, Volonté C, Matteoli M, Verderio C, Abbracchio MP (2003) Nucleotide-mediated calcium signaling in rat cortical astrocytes: role of P2X and P2Y receptors. Glia 43:218–303. 10.1002/glia.10248 [DOI] [PubMed] [Google Scholar]
- Gandelman M, Peluffo H, Beckman JS, Cassina P, Barbeito L (2010) Extracellular ATP and the P2X7 receptor in astrocyte-mediated motor neuron death: implications for amyotrophic lateral sclerosis. J Neuroinflammation 7:33. 10.1186/1742-2094-7-33 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gao P, Ding X, Khan TM, Rong W, Franke H, Illes P (2017) P2X7 receptor-sensitivity of astrocytes and neurons in the substantia gelatinosa of organotypic spinal cord slices of the mouse depends on the length of the culture period. Neuroscience 349:195–207. 10.1016/j.neuroscience.2017.02.030 [DOI] [PubMed] [Google Scholar]
- Garré JM, Retamal MA, Cassina P, Barbeito L, Bukauskas FF, Sáez JC, Bennett MV, Abudara V (2010) FGF-1 induces ATP release from spinal astrocytes in culture and opens pannexin and connexin hemichannels. Proc Natl Acad Sci U S A 107:22659–22664. 10.1073/pnas.1013793107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garré JM, Yang G, Bukauskas FF, Bennett MV (2016) FGF-1 triggers pannexin-1 hemichannel opening in spinal astrocytes of rodents and promotes inflammatory responses in acute spinal cord slices. J Neurosci 36:4785–4801. 10.1523/JNEUROSCI.4195-15.2016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gómez-Villafuertes R, Gualix J, Miras-Portugal MT (2001) Single GABAergic synaptic terminals from rat midbrain exhibit functional P2X and dinucleotide receptors, able to induce GABA secretion. J Neurochem 77:84–93. 10.1046/j.1471-4159.2001.t01-1-00228.x [DOI] [PubMed] [Google Scholar]
- Gordon GR, Baimoukhametova DV, Hewitt SA, Rajapaksha WR, Fisher TE, Bains JS (2005) Norepinephrine triggers release of glial ATP to increase postsynaptic efficacy. Nat Neurosci 8:1078–1086. 10.1038/nn1498 [DOI] [PubMed] [Google Scholar]
- Gordon JL. (1986) Extracellular ATP: effects, sources and fate. Biochem J 233:309–319. 10.1042/bj2330309 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gosselin RD, Suter MR, Ji RR, Decosterd I (2010) Glial cells and chronic pain. Neuroscientist 16:519–531. 10.1177/1073858409360822 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grygorowicz T, Struzyńska L, Sulkowski G, Chalimoniuk M, Sulejczak D (2010) Temporal expression of P2X7 purinergic receptor during the course of experimental autoimmune encephalomyelitis. Neurochem Int 57:823–829. 10.1016/j.neuint.2010.08.021 [DOI] [PubMed] [Google Scholar]
- Gu BJ, Baird PN, Vessey KA, Skarratt KK, Fletcher EL, Fuller SJ, Richardson AJ, Guymer RH, Wiley JS (2013) A rare functional haplotype of the P2RX4 and P2RX7 genes leads to loss of innate phagocytosis and confers increased risk of age-related macular degeneration. FASEB J 27:1479–1487. 10.1096/fj.12-215368 [DOI] [PubMed] [Google Scholar]
- Gundersen V, Storm-Mathisen J, Bergersen LH (2015) Neuroglial transmission. Physiol Rev 95:695–726. 10.1152/physrev.00024.2014 [DOI] [PubMed] [Google Scholar]
- Habbas S, Ango F, Daniel H, Galante M (2011) Purinergic signaling in the cerebellum: Bergmann glial cells express functional ionotropic P2X7 receptors. Glia 59:1800–1812. 10.1002/glia.21224 [DOI] [PubMed] [Google Scholar]
- Hattori M, Gouaux E (2012) Molecular mechanism of ATP binding and ion channel activation in P2X receptors. Nature 485:207–212. 10.1038/nature11010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Henshall DC, Díaz-Hernández M, Miras-Portugal MT, Engel T (2013) P2X receptors as targets for the treatment of status epilepticus. Front Cell Neurosci 7:237. 10.3389/fncel.2013.00237 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hirayama Y, Ikeda-Matsuo Y, Notomi S, Enaida H, Kinouchi H, Koizumi S (2015) Astrocyte-mediated ischemic tolerance. J Neurosci 35:3794–3805. 10.1523/JNEUROSCI.4218-14.2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang C, Han X, Li X, Lam E, Peng W, Lou N, Torres A, Yang M, Garré JM, Tian GF, Bennett MV, Nedergaard M, Takano T (2012) Critical role of connexin 43 in secondary expansion of traumatic spinal cord injury. J Neurosci 32:3333–3338. 10.1523/JNEUROSCI.1216-11.2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huck S, Grass F, Hörtnagl H (1984) The glutamate analogue α-aminoadipic acid is taken up by astrocytes before exerting its gliotoxic effect in vitro. J Neurosci 4:2650–2657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Illes P, Ribeiro AJ (2004) Molecular physiology of P2 receptors in the central nervous system. Eur J Pharmacol 483:5–17. 10.1016/j.ejphar.2003.10.030 [DOI] [PubMed] [Google Scholar]
- Illes P, Verkhratsky A (2016) Purinergic neurone-glia signalling in cognitive-related pathologies. Neuropharmacology 104:62–75. 10.1016/j.neuropharm.2015.08.005 [DOI] [PubMed] [Google Scholar]
- Illes P, Verkhratsky A, Burnstock G, Franke H (2012) P2X receptors and their roles in astroglia in the central and peripheral nervous system. Neuroscientist 18:422–438. 10.1177/1073858411418524 [DOI] [PubMed] [Google Scholar]
- Ireland MF, Noakes PG, Bellingham MC (2004) P2X7-like receptor subunits enhance excitatory synaptic transmission at central synapses by presynaptic mechanisms. Neuroscience 128:269–280. 10.1016/j.neuroscience.2004.06.014 [DOI] [PubMed] [Google Scholar]
- Jimenez-Pacheco A, Díaz-Hernández M, Arribas-Blázquez M, Sanz-Rodriguez A, Olivos-Oré LA, Artalejo AR, Alves M, Letavic M, Miras-Portugal MT, Conroy RM, Delanty N, Farrell MA, O'Brien DF, Bhattacharya A, Engel T, Henshall DC (2016) Transient P2X7 receptor antagonism produces lasting reductions in spontaneous seizures and gliosis in experimental temporal lobe epilepsy. J Neurosci 36:5920–5932. 10.1523/JNEUROSCI.4009-15.2016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kandratavicius L, Balista PA, Lopes-Aguiar C, Ruggiero RN, Umeoka EH, Garcia-Cairasco N, Bueno-Junior LS, Leite JP (2014) Animal models of epilepsy: use and limitations. Neuropsychiatr Dis Treat 10:1693–1705. 10.2147/NDT.S50371 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khakpay R, Polster D, Köles L, Skorinkin A, Szabo B, Wirkner K, Illes P (2010) Potentiation of the glutamatergic synaptic input to rat locus coeruleus neurons by P2X7 receptors. Purinergic Signal 6:349–359. 10.1007/s11302-010-9198-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim JE, Kwak SE, Jo SM, Kang TC (2009) Blockade of P2X receptor prevents astroglial death in the dentate gyrus following pilocarpine-induced status epilepticus. Neurol Res 31:982–988. 10.1179/174313209X389811 [DOI] [PubMed] [Google Scholar]
- Kimbler DE, Shields J, Yanasak N, Vender JR, Dhandapani KM (2012) Activation of P2X7 promotes cerebral edema and neurological injury after traumatic brain injury in mice. PLoS One 7:e41229. 10.1371/journal.pone.0041229 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koshinaga M, Sanon HR, Whittemore SR (1993) Altered acidic and basic fibroblast growth factor expression following spinal cord injury. Exp Neurol 120:32–48. 10.1006/exnr.1993.1038 [DOI] [PubMed] [Google Scholar]
- Kukley M, Barden JA, Steinhäuser C, Jabs R (2001) Distribution of P2X receptors on astrocytes in juvenile rat hippocampus. Glia 36:11–21. 10.1002/glia.1091 [DOI] [PubMed] [Google Scholar]
- Kukley M, Stausberg P, Adelmann G, Chessell IP, Dietrich D (2004) Ecto-nucleotidases and nucleoside transporters mediate activation of adenosine receptors on hippocampal mossy fibers by P2X7 receptor agonist 2′-3′-O-(4-benzoylbenzoyl)-ATP. J Neurosci 24:7128–7139. 10.1523/JNEUROSCI.2093-04.2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leichsenring A, Riedel T, Qin Y, Rubini P, Illes P (2013) Anoxic depolarization of hippocampal astrocytes: possible modulation by P2X7 receptors. Neurochem Int 62:15–22. 10.1016/j.neuint.2012.11.002 [DOI] [PubMed] [Google Scholar]
- Li M, Toombes GE, Silberberg SD, Swartz KJ (2015) Physical basis of apparent pore dilation of ATP-activated P2X receptor channels. Nat Neurosci 18:1577–1583. 10.1038/nn.4120 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lohman AW, Billaud M, Isakson BE (2012) Mechanisms of ATP release and signalling in the blood vessel wall. Cardiovasc Res 95:269–280. 10.1093/cvr/cvs187 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maier CM, Chan PH (2002) Role of superoxide dismutases in oxidative damage and neurodegenerative disorders. Neuroscientist 8:323–334. 10.1177/107385840200800408 [DOI] [PubMed] [Google Scholar]
- Mansoor SE, Lü W, Oosterheert W, Shekhar M, Tajkhorshid E, Gouaux E (2016) X-ray structures define human P2X3 receptor gating cycle and antagonist action. Nature 538:66–71. 10.1038/nature19367 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marín-García P, Sánchez-Nogueiro J, Gómez-Villafuertes R, León D, Miras-Portugal MT (2008) Synaptic terminals from mice midbrain exhibit functional P2X7 receptor. Neuroscience 151:361–373. 10.1016/j.neuroscience.2007.10.038 [DOI] [PubMed] [Google Scholar]
- Masin M, Young C, Lim K, Barnes SJ, Xu XJ, Marschall V, Brutkowski W, Mooney ER, Gorecki DC, Murrell-Lagnado R (2012) Expression, assembly and function of novel C-terminal truncated variants of the mouse P2X7 receptor: re-evaluation of P2X7 knockouts. Br J Pharmacol 165:978–993. 10.1111/j.1476-5381.2011.01624.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matute C, Torre I, Pérez-Cerdá F, Pérez-Samartín A, Alberdi E, Etxebarria E, Arranz AM, Ravid R, Rodríguez-Antigüedad A, Sánchez-Gómez M, Domercq M (2007) P2X7 receptor blockade prevents ATP excitotoxicity in oligodendrocytes and ameliorates experimental autoimmune encephalomyelitis. J Neurosci 27:9525–9533. 10.1523/JNEUROSCI.0579-07.2007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- McGaraughty S, Chu KL, Namovic MT, Donnelly-Roberts DL, Harris RR, Zhang XF, Shieh CC, Wismer CT, Zhu CZ, Gauvin DM, Fabiyi AC, Honore P, Gregg RJ, Kort ME, Nelson DW, Carroll WA, Marsh K, Faltynek CR, Jarvis MF (2007) P2X7-related modulation of pathological nociception in rats. Neuroscience 146:1817–1828. 10.1016/j.neuroscience.2007.03.035 [DOI] [PubMed] [Google Scholar]
- Metzger MW, Walser SM, Aprile-Garcia F, Dedic N, Chen A, Holsboer F, Arzt E, Wurst W, Deussing JM (2017) Genetically dissecting P2rx7 expression within the central nervous system using conditional humanized mice. Purinergic Signal 13:153–170. 10.1007/s11302-016-9546-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- Milius D, Gröger-Arndt H, Stanchev D, Lange-Dohna C, Rossner S, Sperlágh B, Wirkner K, Illes P (2007) Oxygen/glucose deprivation increases the integration of recombinant P2X7 receptors into the plasma membrane of HEK293 cells. Toxicology 238:60–69. 10.1016/j.tox.2007.05.028 [DOI] [PubMed] [Google Scholar]
- Miras-Portugal MT, Díaz-Hernández M, Giráldez L, Hervás C, Gómez-Villafuertes R, Sen RP, Gualix J, Pintor J (2003) P2X7 receptors in rat brain: presence in synaptic terminals and granule cells. Neurochem Res 28:1597–1605. 10.1023/A:1025690913206 [DOI] [PubMed] [Google Scholar]
- Monif M, Burnstock G, Williams DA (2010) Microglia: proliferation and activation driven by the P2X7 receptor. Int J Biochem Cell Biol 42:1753–1756. 10.1016/j.biocel.2010.06.021 [DOI] [PubMed] [Google Scholar]
- Nicke A, Kuan YH, Masin M, Rettinger J, Marquez-Klaka B, Bender O, Górecki DC, Murrell-Lagnado RD, Soto F (2009) A functional P2X7 splice variant with an alternative transmembrane domain 1 escapes gene inactivation in P2X7 knock-out mice. J Biol Chem 284:25813–25822. 10.1074/jbc.M109.033134 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Niyadurupola N, Sidaway P, Ma N, Rhodes JD, Broadway DC, Sanderson J (2013) P2X7 receptor activation mediates retinal ganglion cell death in a human retina model of ischemic neurodegeneration. Invest Ophthalmol Vis Sci 54:2163–2170. 10.1167/iovs.12-10968 [DOI] [PubMed] [Google Scholar]
- Nobile M, Monaldi I, Alloisio S, Cugnoli C, Ferroni S (2003) ATP-induced, sustained calcium signalling in cultured rat cortical astrocytes: evidence for a non-capacitative, P2X7-like-mediated calcium entry. FEBS Lett 538:71–76. 10.1016/S0014-5793(03)00129-7 [DOI] [PubMed] [Google Scholar]
- Nörenberg W, Schunk J, Fischer W, Sobottka H, Riedel T, Oliveira JF, Franke H, Illes P (2010) Electrophysiological classification of P2X7 receptors in rat cultured neocortical astroglia. Br J Pharmacol 160:1941–1952. 10.1111/j.1476-5381.2010.00736.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- North RA. (2002) Molecular physiology of P2X receptors. Physiol Rev 82:1013–1067. 10.1152/physrev.00015.2002 [DOI] [PubMed] [Google Scholar]
- North RA. (2016) P2X receptors. Philos Trans R Soc Lond B Biol Sci 371:20150427. 10.1098/rstb.2015.0427 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oliveira JF, Riedel T, Leichsenring A, Heine C, Franke H, Krügel U, Nörenberg W, Illes P (2011) Rodent cortical astroglia express in situ functional P2X7 receptors sensing pathologically high ATP concentrations. Cereb Cortex 21:806–820. 10.1093/cercor/bhq154 [DOI] [PubMed] [Google Scholar]
- Orellana JA, Avendaño BC, Montero TD (2014) Role of connexins and pannexins in ischemic stroke. Curr Med Chem 21:2165–2182. 10.2174/0929867321666131228191714 [DOI] [PubMed] [Google Scholar]
- Panatier A, Vallée J, Haber M, Murai KK, Lacaille JC, Robitaille R (2011) Astrocytes are endogenous regulators of basal transmission at central synapses. Cell 146:785–798. 10.1016/j.cell.2011.07.022 [DOI] [PubMed] [Google Scholar]
- Pannicke T, Fischer W, Biedermann B, Schädlich H, Grosche J, Faude F, Wiedemann P, Allgaier C, Illes P, Burnstock G, Reichenbach A (2000) P2X7 receptors in Muller glial cells from the human retina. J Neurosci 20:5965–5972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pedata F, Dettori I, Coppi E, Melani A, Fusco I, Corradetti R, Pugliese AM (2016) Purinergic signalling in brain ischemia. Neuropharmacology 104:105–130. 10.1016/j.neuropharm.2015.11.007 [DOI] [PubMed] [Google Scholar]
- Pelegrin P, Surprenant A (2006) Pannexin-1 mediates large pore formation and interleukin-1β release by the ATP-gated P2X7 receptor. EMBO J 25:5071–5082. 10.1038/sj.emboj.7601378 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pelegrin P, Surprenant A (2009) The P2X7 receptor-pannexin connection to dye uptake and IL-1β release. Purinergic Signal 5:129–137. 10.1007/s11302-009-9141-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peng W, Cotrina ML, Han X, Yu H, Bekar L, Blum L, Takano T, Tian GF, Goldman SA, Nedergaard M (2009) Systemic administration of an antagonist of the ATP-sensitive receptor P2X7 improves recovery after spinal cord injury. Proc Natl Acad Sci U S A 106:12489–12493. 10.1073/pnas.0902531106 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reichenbach A, Bringmann A (2016) Purinergic signaling in retinal degeneration and regeneration. Neuropharmacology 104:194–211. 10.1016/j.neuropharm.2015.05.005 [DOI] [PubMed] [Google Scholar]
- Retamal MA, Alcayaga J, Verdugo CA, Bultynck G, Leybaert L, Sáez PJ, Fernández R, León LE, Sáez JC (2014) Opening of pannexin- and connexin-based channels increases the excitability of nodose ganglion sensory neurons. Front Cell Neurosci 8:158. 10.3389/fncel.2014.00158 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rodrigues RJ, Almeida T, Richardson PJ, Oliveira CR, Cunha RA (2005) Dual presynaptic control by ATP of glutamate release via facilitatory P2X1, P2X2/3, and P2X3 and inhibitory P2Y1, P2Y2, and/or P2Y4 receptors in the rat hippocampus. J Neurosci 25:6286–6295. 10.1523/JNEUROSCI.0628-05.2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rodrigues RJ, Tomé AR, Cunha RA (2015) ATP as a multi-target danger signal in the brain. Front Neurosci 9:148. 10.3389/fnins.2015.00148 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roger S, Pelegrin P, Surprenant A (2008) Facilitation of P2X7 receptor currents and membrane blebbing via constitutive and dynamic calmodulin binding. J Neurosci 28:6393–6401. 10.1523/JNEUROSCI.0696-08.2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roger S, Gillet L, Baroja-Mazo A, Surprenant A, Pelegrin P (2010) C-terminal calmodulin-binding motif differentially controls human and rat P2X7 receptor current facilitation. J Biol Chem 285:17514–17524. 10.1074/jbc.M109.053082 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rozmer K, Gao P, Araujo MG, Khan MT, Liu J, Rong W, Tang Y, Franke H, Krügel U, Fernandes MJ, Illes P (2016) Pilocarpine-induced status epilepticus increases the sensitivity of P2X7 and P2Y1 receptors to nucleotides at neural progenitor cells of the juvenile rodent hippocampus. Cereb Cortex. Advance online publication. Retrieved Jun. 23, 2016. doi: 10.1093/cercor/bhw178. 10.1093/cercor/bhw178 [DOI] [PubMed] [Google Scholar]
- Rubini P, Pinkwart C, Franke H, Gerevich Z, Nörenberg W, Illes P (2006) Regulation of intracellular Ca2+ by P2Y1 receptors may depend on the developmental stage of cultured rat striatal neurons. J Cell Physiol 209:81–93. 10.1002/jcp.20705 [DOI] [PubMed] [Google Scholar]
- Rubini P, Pagel G, Mehri S, Marquardt P, Riedel T, Illes P (2014) Functional P2X7 receptors at cultured hippocampal astrocytes but not neurons. Naunyn Schmiedebergs Arch Pharmacol 387:943–954. 10.1007/s00210-014-1005-1 [DOI] [PubMed] [Google Scholar]
- Salas E, Carrasquero LM, Olivos-Oré LA, Bustillo D, Artalejo AR, Miras-Portugal MT, Delicado EG (2013) Purinergic P2X7 receptors mediate cell death in mouse cerebellar astrocytes in culture. J Pharmacol Exp Ther 347:802–815. 10.1124/jpet.113.209452 [DOI] [PubMed] [Google Scholar]
- Sánchez-Nogueiro J, Marín-García P, Miras-Portugal MT (2005) Characterization of a functional P2X7-like receptor in cerebellar granule neurons from P2X7 knockout mice. FEBS Lett 579:3783–3788. 10.1016/j.febslet.2005.05.073 [DOI] [PubMed] [Google Scholar]
- Sanz JM, Chiozzi P, Ferrari D, Colaianna M, Idzko M, Falzoni S, Fellin R, Trabace L, Di Virgilio F (2009) Activation of microglia by amyloid-β requires P2X7 receptor expression. J Immunol 182:4378–4385. 10.4049/jimmunol.0803612 [DOI] [PubMed] [Google Scholar]
- Schwarz N, Drouot L, Nicke A, Fliegert R, Boyer O, Guse AH, Haag F, Adriouch S, Koch-Nolte F (2012) Alternative splicing of the N-terminal cytosolic and transmembrane domains of P2X7 controls gating of the ion channel by ADP-ribosylation. PLoS One 7:e41269. 10.1371/journal.pone.0041269 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sharp AJ, Polak PE, Simonini V, Lin SX, Richardson JC, Bongarzone ER, Feinstein DL (2008) P2x7 deficiency suppresses development of experimental autoimmune encephalomyelitis. J Neuroinflammation 5:33. 10.1186/1742-2094-5-33 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sim JA, Young MT, Sung HY, North RA, Surprenant A (2004) Reanalysis of P2X7 receptor expression in rodent brain. J Neurosci 24:6307–6314. 10.1523/JNEUROSCI.1469-04.2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Skaper SD. (2011) Ion channels on microglia: therapeutic targets for neuroprotection. CNS Neurol Disord Drug Targets 10:44–56. 10.2174/187152711794488638 [DOI] [PubMed] [Google Scholar]
- Skaper SD, Facci L, Culbert AA, Evans NA, Chessell I, Davis JB, Richardson JC (2006) P2X7 receptors on microglial cells mediate injury to cortical neurons in vitro. Glia 54:234–242. 10.1002/glia.20379 [DOI] [PubMed] [Google Scholar]
- Skaper SD, Debetto P, Giusti P (2009) P2X(7) receptors in neurological and cardiovascular disorders. Cardiovasc Psychiatry Neurol 2009:861324. 10.1155/2009/861324 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Skaper SD, Debetto P, Giusti P (2010) The P2X7 purinergic receptor: from physiology to neurological disorders. FASEB J 24:337–345. 10.1096/fj.09-138883 [DOI] [PubMed] [Google Scholar]
- Sosinsky GE, Boassa D, Dermietzel R, Duffy HS, Laird DW, MacVicar B, Naus CC, Penuela S, Scemes E, Spray DC, Thompson RJ, Zhao HB, Dahl G (2011) Pannexin channels are not gap junction hemichannels. Channels (Austin) 5:193–197. 10.4161/chan.5.3.15765 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sperlágh B, Illes P (2006) Az ATP és az adenozin részvétele az idegsejtek közötti kommunikációban, Természet Világa Suppl II: 30–36.
- Sperlágh B, Illes P (2007) Purinergic modulation of microglial cell activation. Purinergic Signal 3:117–127. 10.1007/s11302-006-9043-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sperlágh B, Illes P (2014) P2X7 receptor: an emerging target in central nervous system diseases. Trends Pharmacol Sci 35:537–547. 10.1016/j.tips.2014.08.002 [DOI] [PubMed] [Google Scholar]
- Sperlágh B, Köfalvi A, Deuchars J, Atkinson L, Milligan CJ, Buckley NJ, Vizi ES (2002) Involvement of P2X7 receptors in the regulation of neurotransmitter release in the rat hippocampus. J Neurochem 81:1196–1211. 10.1046/j.1471-4159.2002.00920.x [DOI] [PubMed] [Google Scholar]
- Sperlágh B, Vizi ES, Wirkner K, Illes P (2006) P2X7 receptors in the nervous system. Prog Neurobiol 78:327–346. 10.1016/j.pneurobio.2006.03.007 [DOI] [PubMed] [Google Scholar]
- Suadicani SO, Brosnan CF, Scemes E (2006) P2X7 receptors mediate ATP release and amplification of astrocytic intercellular Ca2+ signaling. J Neurosci 26:1378–1385. 10.1523/jneurosci.3902-05.2006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suadicani SO, Iglesias R, Spray DC, Scemes E (2009) Point mutation in the mouse P2X7 receptor affects intercellular calcium waves in astrocytes. ASN Neuro 1:e00005. 10.1042/AN20090001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Surprenant A, Rassendren F, Kawashima E, North RA, Buell G (1996) The cytolytic P2Z receptor for extracellular ATP identified as a P2X receptor (P2X7). Science 272:735–738. 10.1126/science.272.5262.735 [DOI] [PubMed] [Google Scholar]
- Tang Y, Illes P (2016) Regulation of adult neural progenitor cell functions by purinergic signaling. Glia 65:213–230. 10.1002/glia.23056 [DOI] [PubMed] [Google Scholar]
- Turola E, Furlan R, Bianco F, Matteoli M, Verderio C (2012) Microglial microvesicle secretion and intercellular signaling. Front Physiol 3:149. 10.3389/fphys.2012.00149 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verkhratsky A, Matteoli M, Parpura V, Mothet JP, Zorec R (2016) Astrocytes as secretory cells of the central nervous system: idiosyncrasies of vesicular secretion. EMBO J 35:239–257. 10.15252/embj.201592705 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Villa G, Fumagalli M, Verderio C, Abbracchio MP, Ceruti S (2010) Expression and contribution of satellite glial cells purinoceptors to pain transmission in sensory ganglia: an update. Neuron Glia Biol 6:31–42. 10.1017/S1740925X10000086 [DOI] [PubMed] [Google Scholar]
- Volonté C, Apolloni S, Skaper SD, Burnstock G (2012) P2X7 receptors: channels, pores and more. CNS Neurol Disord Drug Targets 11:705–721. 10.2174/187152712803581137 [DOI] [PubMed] [Google Scholar]
- Wang X, Arcuino G, Takano T, Lin J, Peng WG, Wan P, Li P, Xu Q, Liu QS, Goldman SA, Nedergaard M (2004) P2X7 receptor inhibition improves recovery after spinal cord injury. Nat Med 10:821–827. 10.1038/nm1082 [DOI] [PubMed] [Google Scholar]
- Webb TE, Simon J, Krishek BJ, Bateson AN, Smart TG, King BF, Burnstock G, Barnard EA (1993) Cloning and functional expression of a brain G-protein-coupled ATP receptor. FEBS Lett 324:219–225. 10.1016/0014-5793(93)81397-I [DOI] [PubMed] [Google Scholar]
- Wirkner K, Köfalvi A, Fischer W, Günther A, Franke H, Gröger-Arndt H, Nörenberg W, Madarász E, Vizi ES, Schneider D, Sperlágh B, Illes P (2005) Supersensitivity of P2X receptors in cerebrocortical cell cultures after in vitro ischemia. J Neurochem 95:1421–1437. 10.1111/j.1471-4159.2005.03465.x [DOI] [PubMed] [Google Scholar]
- Wurm A, Pannicke T, Iandiev I, Francke M, Hollborn M, Wiedemann P, Reichenbach A, Osborne NN, Bringmann A (2011) Purinergic signaling involved in Muller cell function in the mammalian retina. Prog Retin Eye Res 30:324–342. 10.1016/j.preteyeres.2011.06.001 [DOI] [PubMed] [Google Scholar]
- Yan Z, Khadra A, Li S, Tomic M, Sherman A, Stojilkovic SS (2010) Experimental characterization and mathematical modeling of P2X7 receptor channel gating. J Neurosci 30:14213–14224. 10.1523/JNEUROSCI.2390-10.2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yan Z, Khadra A, Sherman A, Stojilkovic SS (2011) Calcium-dependent block of P2X7 receptor channel function is allosteric. J Gen Physiol 138:437–452. 10.1085/jgp.201110647 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu Q, Guo Z, Liu X, Ouyang Q, He C, Burnstock G, Yuan H, Xiang Z (2013) Block of P2X7 receptors could partly reverse the delayed neuronal death in area CA1 of the hippocampus after transient global cerebral ischemia. Purinergic Signal 9:663–675. 10.1007/s11302-013-9379-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu Y, Ugawa S, Ueda T, Ishida Y, Inoue K, Kyaw Nyunt A, Umemura A, Mase M, Yamada K, Shimada S (2008) Cellular localization of P2X7 receptor mRNA in the rat brain. Brain Res 1194:45–55. 10.1016/j.brainres.2007.11.064 [DOI] [PubMed] [Google Scholar]
- Zhang X, Chen Y, Wang C, Huang LY (2007) Neuronal somatic ATP release triggers neuron-satellite glial cell communication in dorsal root ganglia. Proc Natl Acad Sci U S A 104:9864–9869. 10.1073/pnas.0611048104 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang XF, Han P, Faltynek CR, Jarvis MF, Shieh CC (2005) Functional expression of P2X7 receptors in non-neuronal cells of rat dorsal root ganglia. Brain Res 1052:63–70. 10.1016/j.brainres.2005.06.022 [DOI] [PubMed] [Google Scholar]