Abstract
Study Objectives:
Supplemental oxygen has been shown to decrease the frequency of obstructive respiratory events during sleep, but may result in alveolar hypoventilation. Limited information exists on the effect of supplemental oxygen on sleep and respiratory events in infants with obstructive sleep apnea (OSA).
Methods:
We conducted a retrospective study of infants with OSA who had sleep studies performed from 2007–2012. All infants underwent a room air diagnostic sleep study (RA-PSG), followed by a sleep study while breathing supplemental oxygen via nasal cannula (O2-PSG) on a separate night. Infants with split-night studies or with inadequate sleep time were excluded.
Results:
Fifty-nine infants met criteria for entry into analysis. The mean age of infants at the time of RA-PSG was 13.0 ± 11.7 weeks and at O2-PSG was 15.4 ± 13.0 weeks. The obstructive AHI decreased from 19.7 ± 13.0 during RA-PSG to 10.6 ± 11.7 during O2-PSG (P < .001). The duration of longest obstructive apnea increased from 11.0 ± 4.2 seconds to 13.4 ± 7.4 seconds (P = .01). The lowest saturation associated with obstructive apneas increased from 80.7 ± 6.8% to 90.0 ± 6.7% (P < .001). Carbon dioxide data showed no difference in ventilation after supplemental oxygen administration. There was no significant change in the spontaneous arousal index, however, the percentage of respiratory events associated with arousal increased from 20.7 ± 11.1% to 35.7 ± 19.7% (P < .001).
Conclusions:
Infants with OSA who received supplemental oxygen had a significant decrease in the frequency of obstructive respiratory events and improved oxygenation without adverse effect on alveolar ventilation. These data suggest that supplemental oxygen may be an effective treatment for infants with OSA who are not good candidates for continuous positive airway pressure or surgery.
Citation:
Brockbank J, Astudillo CL, Che D, Tanphaichitr A, Huang G, Tomko J, Simakajornboon N. Supplemental oxygen for treatment of infants with obstructive sleep apnea. J Clin Sleep Med. 2019;15(8):1115–1123.
Keywords: infant sleep, oxygen therapy, pediatric OSA, respiratory control
BRIEF SUMMARY
Current Knowledge/Study Rationale: Treatment of obstructive sleep apnea (OSA) in infants is challenging given the technical difficulties of administering continuous positive airway pressure to that population and given the invasive surgical treatments available. This study evaluates the effect of supplemental oxygen on sleep and respiratory parameters in infants with OSA.
Study Impact: Our study demonstrates that supplemental oxygen administration to infants with OSA reduces the frequency of obstructive respiratory events without having an adverse effect on ventilation. These findings suggest that low flow supplemental oxygen for infant OSA may be an effective treatment option.
INTRODUCTION
Obstructive sleep apnea (OSA) is characterized by periods of complete or partial upper airway obstruction during sleep resulting in intermittent hypoxemia and sleep fragmentation. If left untreated, it can lead to cardiovascular and neurocognitive morbidity.1 The prevalence of OSA has been estimated to be 1% to 4% in older children,2–6 but there is limited information on the prevalence in infants. Snoring is present in 11.8% to 26% of infants, but fewer than 10% of snoring infants have OSA.7,8 OSA is more common in preterm infants, those with genetic syndromes, and those with neuromuscular or craniofacial abnormalities.9–11 It is reported that children younger than 3 years have more severe OSA compared to older children.12 Because of this, and although long-term outcomes data are not available, infants with sleep disordered breathing may be at risk for cognitive sequelae of OSA, including learning or memory impairment and hyperactivity.1,13
Treatment of infants with OSA is challenging because of difficulties administering continuous positive airway pressure (CPAP), which is the standard therapy in adults and in older children in whom adenotonsillectomy has failed. Surgical treatments including supraglottoplasty, mandibular distraction, and tracheostomy may not be ideal in some circumstances. For these reasons low flow supplemental oxygen delivered via nasal cannula has been used to treat infants with OSA.11 Studies have shown the benefit of supplemental oxygen on central apnea, periodic breathing, and sleep architecture in infants with apnea of prematurity.14–16 The safety and efficacy of supplemental oxygen administration to infants with OSA, however, is not well studied.
There are several studies evaluating the effect of supplemental oxygen on OSA in adults. A recent systematic review and meta-analysis showed supplemental oxygen led to a significant improvement in the apnea-hypopnea index (AHI) and overall oxygenation status.17 There have been two published studies that examined the effects of supplemental oxygen in children with OSA18,19; however, there are few data on oxygen use for OSA in the infant population. In this study, we examined the effect of supplemental oxygen on sleep and respiratory parameters in infants with OSA. We hypothesized that supplemental oxygen administration would improve oxygenation and reduce the frequency of respiratory events without causing alveolar hypoventilation.
METHODS
This retrospective observational study was performed at Cincinnati Children’s Hospital Medical Center, a quaternary care children’s hospital. The study protocol was approved by the institutional review board. At our institution, we often use supplemental oxygen for treatment of infants with OSA who are poor candidates for surgical intervention or CPAP. Before starting treatment at home, patients typically undergo an inpatient oxygen titration sleep study to evaluate the effectiveness and safety of oxygen use, and to titrate to the appropriate flow. We obtained retrospective data on infants 12 months of age or younger with significant OSA who underwent polysomnography (PSG) from 2007 to 2012. All patients had a room air diagnostic PSG (RA-PSG), followed by PSG on a separate night during which the patient received supplemental oxygen via nasal cannula (O2-PSG). Significant OSA was defined as an obstructive apnea-hypopnea index (OAHI) ≥ 4 to 5 events/h. Those with inadequate sleep time (< 3 hours) or with split-night studies were excluded from analysis. The split-night studies were excluded due to limited time, often less than 3 hours, during the diagnostic portion. The short diagnostic time would affect not only the estimation of respiratory parameters but also the accuracy of sleep parameters. In addition, when oxygen use was split in these patients there may have been insufficient time to completely titrate to the most effective flow. Demographic data, including sex, gestational age, medical history, age at the time of study, pre- and post-O2-PSG blood gases, and sleep and respiratory parameters from both RA-PSG and O2-PSG were collected.
Sleep studies were performed in accordance with 2007 American Academy of Sleep Medicine (AASM) guidelines.20 A standard infant montage was used, and the following variables were recorded simultaneously: body position, left and right electrooculogram (ROC/A1, LOC/A2), four-channel electroencephalogram (O1A2, O2A1, C4A1, C3A2), chin electromyogram, electrocardiogram, pulse oximetry, pulse waveform, thoracic and abdominal inductance plethysmography, airflow with thermistor and nasal pressure transducer, end-tidal partial pressure of carbon dioxide (pCO2) (BCI Capnocheck, Smiths Medical, St. Paul, Minnesota, USA), and transcutaneous pCO2 (Tina TCM4/40, Radiometer, Copenhagen, Denmark). In our sleep laboratory, both end-tidal and transcutaneous CO2 are simultaneously monitored during routine PSG in all infants. Sleep scoring was performed by registered sleep technicians and verified by board-certified pediatric sleep specialists using standard infant criteria. Studies were analyzed in 30-second epochs and staged as wake, rapid eye movement (REM) sleep, or non-rapid eye movement (NREM) sleep. Sleep efficiency was calculated by dividing total sleep time by time in bed and expressing as a percentage. An arousal was defined as a shift in the electroencephalogram pattern to frequencies of 8 to 13 Hz, or above 16 Hz, for a minimum of 3 seconds. Arousals in REM sleep required a concurrent increase in submental EMG amplitude. The arousal index was defined as the number of arousals per hour of sleep. A respiratory arousal was defined as an arousal occurring at the termination of, or within 5 seconds following, a respiratory event. The respiratory arousal percentage was the percent of respiratory events that were associated with an arousal. Apnea was defined as at least a 90% reduction in airflow from baseline over two or more respiratory cycles. An obstructive apnea was defined as apnea in the presence of persistent or increased respiratory effort. A central apnea was defined as apnea with absence of both chest wall and abdominal movement. Hypopnea was defined as a 50% or greater reduction in airflow accompanied by either an oxygen desaturation of at least 3% or arousal. The reduction in airflow was determined primarily by the nasal pressure transducer, and if this signal was inadequate, the thermistor was used as backup. The apnea-hypopnea index (AHI) was defined as the total number of apneas and hypopneas per hour of sleep. The OAHI was defined as the number of obstructive apneas, mixed apneas, and obstructive hypopneas per hour. The obstructive apnea index (OAI) was the number of obstructive and mixed apneas per hour. The central index (CI) was defined as the number of central respiratory events per hour. For oxygen titration studies, analysis of capillary blood gas (CBG) was performed in some infants; blood was collected just before starting, and at the conclusion of the sleep study. The oxygen titration protocol was to initiate supplemental oxygen at 1/8 L/min flow using a divided end-tidal CO2 cannula with simultaneous oxygen delivery (Infant 4701F, Salter Labs, Arvin, California, USA). Flow was increased by 1/8–1/4 L/min increments to maintain oxygen saturation greater than 90% to 93% and to minimize oxygen desaturations associated with respiratory events.
Data were stored in an Excel spreadsheet (Microsoft, Redmond, Washington, USA). The SAS program (Cary, North Carolina, USA) was used for statistical analysis. Our primary outcomes were OAHI, average end-tidal carbon dioxide (EtCO2), and average transcutaneous pCO2 (TrCO2). Secondary outcomes were effects of oxygen on sleep architecture and additional respiratory parameters including AHI, CI, OAI, average and longest duration of obstructive apnea, obstructive hypopnea, and central apnea, and percentage of respiratory arousals. RA-PSG and O2-PSG data were presented as means and standard deviations. CBG pCO2 obtained at the initiation and completion of O2-PSGs were compared. Because the analyzed variables were not normally distributed, the Wilcoxon signed-rank test was applied. A value of P < .05 was considered statistically significant.
RESULTS
Three hundred sixty-two individuals were identified for possible inclusion as being infants age 1 year or younger for whom PSG was performed at least once. Exclusions included undergoing only one PSG, no O2-PSG, insufficient sleep time, or interval surgery between RA-PSG and O2-PSG. Fifty-nine were included for analysis (Figure 1). Demographic data and underlying diagnoses are shown in Table 1. Of the 59 infants, 49 (83%) had an airway obstruction that was identified by flexible and/or rigid bronchoscopy. Some had more than one level of upper airway obstruction and some had more than one diagnosis. In nine infants (15%), an apparent life-threatening event was diagnosed. Of those, a definite upper airway obstruction was identified in seven. Upper airway abnormalities were defined in all but six of the infants with genetic syndromes, and in all but two of the infants with significant neurological disorders. Two were classified as having both a genetic syndrome and neurological disorder. One infant had OSA of uncertain etiology without any underlying disorder or identified upper airway pathology; however, there was a family history of sudden infant death syndrome.
Figure 1. Study population identification and exclusion rationale.
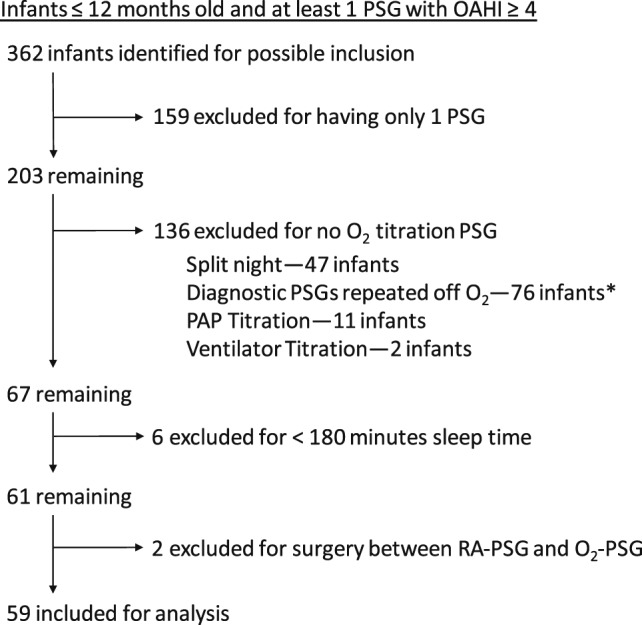
* Most had surgical interventions such as supraglottoplasty or were placed on oxygen empirically.
Table 1.
Baseline characteristics and diagnoses of infants included for analysis (n = 59).

The optimal flow of supplemental oxygen during the oxygen titration PSG was determined to be 1/8 L/min in 29%, 1/4 L/min in 34%, 1/2 L/min in 17%, 3/4 L/min in 12% , and 1 L/min in 8% of the study population. Table 2 shows the effect of supplemental oxygen on sleep architecture and respiratory parameters. There was a significant increase in the percentage of NREM sleep in the O2-PSG group compared to the RA-PSG group. Total sleep time was shorter and sleep efficiency was lower in the oxygen-treated group. Although both the total and spontaneous arousal indices remained unchanged, the percentage of respiratory events terminated by arousal (respiratory arousal percentage) was significantly higher in the O2-PSG group. As oxygen decreased the frequency of respiratory events, comparing the respiratory arousal index would be misleading. Therefore, we compared respiratory arousals using a percentage of respiratory events that were terminated by arousal.
Table 2.
Comparison of sleep and respiratory parameters between RA-PSG and O2-PSG expressed as mean (standard deviation).
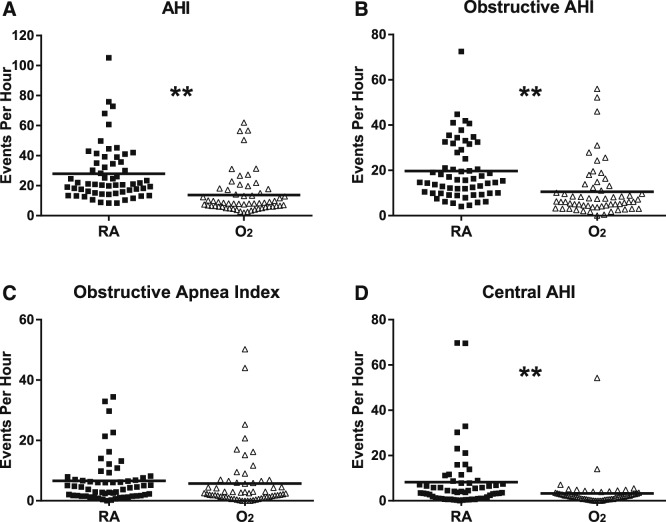
Comparison of respiratory parameters between RA-PSG and O2-PSG groups demonstrated a significant decrease in the AHI, OAHI, and central apnea-hypopnea index when infants were placed on supplemental oxygen. There was also a trend toward reduction in the OAI (Table 2 and Figure 2). Using a 50% or greater reduction in AHI as an indicator of response to supplemental oxygen,21 57.6% of infants classify as oxygen responders. Obstructive apnea duration was longer in the O2-PSG group, whereas there was no significant change in duration of obstructive hypopneas or central apneas. Average and lowest oxygen desaturations associated with obstructive apneas and hypopneas were significantly improved in the O2-PSG group. Similarly, the average and lowest oxygen saturation during central apneas were improved with administration of supplemental oxygen (Table 2).
Figure 2. Effect of supplemental oxygen on respiratory events during sleep.
Scatter plot with mean shown. (A) Apnea-hypopnea index expressed as number of events per hour of sleep. (B) Obstructive apnea hypopnea index expressed as number of events per hour of sleep. (C) Obstructive apnea index expressed as number of events per hour of sleep. (D) Central apnea hypopnea index expressed as number of events per hour of sleep. ** P < .001. AHI = apnea-hypopnea index, O2 = oxygen titration sleep study, RA = diagnostic sleep study on room air.
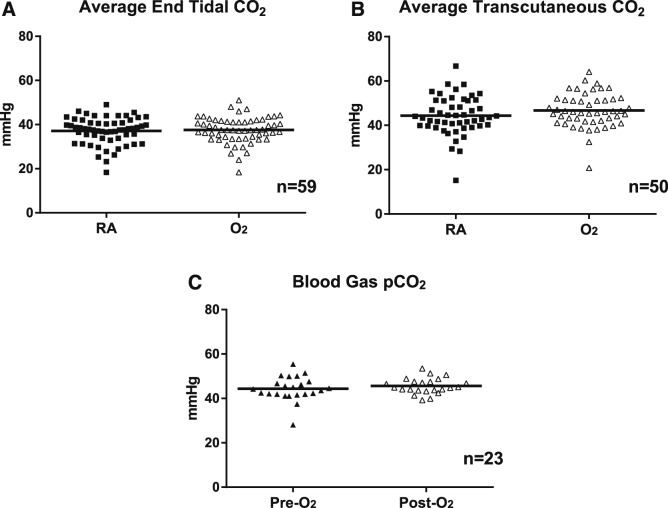
Figure 3 shows the effect of supplemental oxygen on CO2 parameters. There were no significant differences in average EtCO2, average TrCO2, or in percentage of time EtCO2 was greater than 45 or 50 mmHg. Although the percentage of time TrCO2 was greater than 45 mmHg showed no significant change with oxygen administration, the percentage of time TrCO2 was greater than 50 mmHg was higher in the O2-PSG group. Twenty-three infants had blood gases performed just prior to, and immediately following, their O2-PSG studies. There was no significant difference in pCO2 when comparing pre-O2-PSG to post-O2-PSG blood gas data (44.4 ± 5.5 mmHg [pre-O2-PSG] versus 45.6 ± 3.5 [post-O2-PSG], P = .26).
Figure 3. Effect of supplemental oxygen on alveolar ventilation during sleep.
Scatter plot with mean shown. (A) Average end tidal carbon dioxide in mmHg. (B) Average transcutaneous carbon dioxide in mmHg. (C) Partial pressure of carbon dioxide (pCO2) from capillary blood gas obtained at the beginning of oxygen titration sleep study (Pre-O2) compared to end of oxygen titration sleep study (Post-O2). O2 = oxygen titration sleep study, RA = diagnostic sleep study on room air.
DISCUSSION
Our main finding is that infants with OSA treated with supplemental oxygen demonstrated a significant reduction in the frequency of obstructive respiratory events, although they failed to completely normalize their OAHI. Furthermore, supplemental oxygen administration for infants with OSA was associated with a reduction in respiratory event-related oxygen desaturations and improved overall oxygenation. Supplemental oxygen was associated with a slight prolongation of obstructive apneas but did not have a significant adverse effect on alveolar ventilation in this population. An improvement in overall sleep architecture also occurred in infants with OSA treated with supplemental oxygen as evidenced by an increased proportion of NREM sleep. In this study, a greater proportion of respiratory events were terminated by arousal in infants receiving low flow oxygen, suggesting that supplemental oxygen administration may have a protective effect.
This study investigates the effect of supplemental oxygen on OSA in infants. Strengths of our study include the study population consisting of a large cohort of infants with OSA who underwent an RA-PSG, followed by an O2-PSG on 2 separate nights, providing a better assessment of sleep and respiratory parameters in both conditions. In addition, this was a heterogeneous population, allowing for generalization of conclusions across diagnostic categories. Several limitations exist, however. First, this is a retrospective study, and individuals were selected clinically as appropriate candidates for supplemental oxygen therapy. Second, there is a possibility of night-to-night variation in sleep and respiratory events due to the first- night effect; however, this likely has little effect on infants. In fact, it has been shown that the reliability of sleep and respiratory parameters is adequate when the sleep duration exceeds 3 hours.22 Third, the oxygen flow rate was changed throughout the oxygen titration study. Analysis of data obtained during optimal flow could provide more accurate information. Fourth, it was technically difficult to score hypopneas when desaturations were mitigated by supplemental oxygen. There may have been an artificial reduction in the number of hypopneas in patients receiving supplemental oxygen due to utilization of AASM scoring rules in which a 3% desaturation or arousal is required to score a reduction in airflow as a hypopnea. However, we also observed a trend toward reduction in the frequency of obstructive apneas, as well as a reduction in the overall OAHI while infants breathed supplemental oxygen. One interesting observation in our cohort is that not all infants responded to oxygen in a similar manner; some responded well and others failed to respond. In fact, 57.6% of infants can be classified as oxygen responders based on a 50% or greater reduction in AHI. If the reduction in frequency of apneas and hypopneas was exclusively due to the inability to score hypopneas or central apneas because of the lack of desaturation, the response should have been consistent across all infants. Finally, our study excluded many infants with OSA who did not have an oxygen titration study or for whom oxygen was not recommended. This exclusion may have led to selection bias. A significant proportion of these excluded infants were those who underwent surgical intervention and were likely to have had significant anatomical obstruction. Therefore, the findings of this study may not be applicable to that group.
Our results demonstrating a reduction in the frequency of obstructive respiratory events in infants treated with supplemental oxygen are consistent with those found in several prior adult and pediatric studies. In a meta-analysis conducted to evaluate the effects of supplemental oxygen on adults with OSA, six pooled studies showed a significant reduction in sleep- disordered breathing events with oxygen administration compared to room air.17 Although several adult studies showed a reduction in the frequency of obstructive events with oxygen use,23–26 one showed no change in AHI.27 Some showed a prolongation of event duration in adults treated with supplemental oxygen,25,26 and others demonstrated no change in respiratory event duration.23,28 Two published studies exist on supplemental oxygen in children with OSA. Marcus et al showed no significant change in the OAI with oxygen administration, however, hypopneas were not included. There was no significant change in average duration of longest obstructive apneas when children breathed supplemental oxygen.18 Aljadeff et al found a reduction in the hypopnea index and OAI on supplemental oxygen, without prolongation of apnea duration.19
The mechanisms underlying the effect of oxygen on frequency of respiratory events in infants are unknown, although several potential explanations exist. First, supplemental oxygen delivered via nasal cannula could produce a positive pressure effect on the upper airway of infants. Although positive end-distending pressure may be generated with use of a 0.3 cm outer diameter nasal cannula, in one study there was no detection of a positive pressure effect with use of a 0.2 cm cannula even with flows up to 2 L/min.29 The outer diameter of the cannula used in the current study was measured at 0.19 cm, and most patients were studied on flows of 1/8 to 1/4 L/min, which would be very unlikely to produce positive pressure in our infant cohort.
Second, supplemental oxygen administration may reverse upper airway dilator muscle fatigue that has been demonstrated to occur even after short periods of hypoxia.30 Hypoxia-induced activation of upper airway dilator muscles likely contributes to termination of apneic events; however, muscle fatigue may develop quickly, leading to worsening apnea. Supplemental oxygen may prevent upper airway muscle fatigue. Interestingly, hyperoxia has been shown to inhibit stimulation of upper airway dilator muscles, which may be the mechanism by which supplemental oxygen prolongs apnea duration.31,32 However, this mechanism is unlikely because contraction of dilator muscles may still occur under hyperoxic conditions through mechanoreceptor activation by negative pressure in the pharynx generated during apnea.32
Third, supplemental oxygen acts to stabilize ventilatory control. In addition to chemoreceptor and mechanoreceptor effects on upper airway dilator muscles, an increase in ventilatory drive also stimulates contraction of those muscles to promote upper airway patency.33,34 Patients with unstable ventilatory control or high loop gain tend to experience airway collapse as drive oscillates. Adults with OSA and high loop gain were found to have a reduction in both loop gain and AHI when breathing supplemental oxygen.34 Recent studies have shown changes in loop gain associated with infant development. In fact, infants were found to have a substantial rise in loop gain over the first 4 weeks of life, only to fall to normal adult levels by 6 months of age.35 The population in the current study had a mean age of 13 weeks at the time of RA-PSG and 15 weeks at O2-PSG, corresponding to a period of high loop gain, which may have contributed to a favorable response to oxygen. Another interesting finding in our cohort is that more than half were oxygen responders. Adult data have shown that good oxygen response corresponds with a high loop gain phenotype.21 From our recent preliminary data based on a small number of infants, loop gain may play a role in the variation of responses to oxygen, similar to data in the adult population36; however, further study is needed to evaluate the effect of oxygen on loop gain in infants with OSA.
In the current study, administration of supplemental oxygen to infants with OSA resulted in a slight prolongation of both the average and longest duration of obstructive apneas. The mechanism underlying prolongation of apnea duration is unknown but could be related to a delayed arousal response. Arousals following obstructive respiratory events can be triggered by several respiratory-related stimuli including hypoxia, hypercapnia, and respiratory effort (changes in upper airway resistance).37 One potential mechanism to explain a prolongation of obstructive apneas with administration of supplemental oxygen is removal of the hypoxemic respiratory drive and subsequent reliance on the other stimuli for arousal. Hypercapnia is unlikely to play a role because it was not observed in our study. Increased respiratory effort may underlie the increased proportion of arousals following respiratory events (respiratory arousals) in our cohort of infants treated with supplemental oxygen. Because hypoxia is a relatively weak stimulus for arousal compared to hypercapnia or changes in upper airway resistance,37–39 it is unclear whether attenuation of hypoxia with use of supplemental oxygen plays a role in apnea prolongation. Regardless, it is unlikely that a slight prolongation of apnea in infants treated with supplemental oxygen would lead to any adverse effects, because overall improvement in both oxygenation status and respiratory arousals have been demonstrated.
Arousal response during sleep-associated airway obstruction is an important protective mechanism that allows for recovery from oxygen desaturations and hypoventilation. Studies have shown the important role of arousal in termination of respiratory events in infants.40,41 In our study, a significantly higher proportion of respiratory events was terminated by arousal in infants given supplemental oxygen. Despite the prolongation of apneas observed, the increased arousal frequency following respiratory events in infants on supplemental oxygen may offer protection from sequelae associated with failure to arouse during airway occlusion.
Since the initial study published by Motta and Guilleminault in 1978 regarding the effects of oxygen administration on sleep apnea in adults with OSA, a concern has been raised about the potential development of hypoventilation.42 That study examined three adult males with OSA and found increased PaCO2 from blood gases during oxygen administration. Two additional studies examining the effects of supplemental oxygen on adults with chronic obstructive pulmonary disease also demonstrated development of hypoventilation.43,44 However, two other adult studies showed no significant change in EtCO2 with oxygen administration.25,26 As described previously, two studies examined the effect of supplemental oxygen on children with OSA.18,19 Although Marcus et al found no significant change in mean EtCO2 in those breathing supplemental oxygen, there were two children who experienced an increase in peak EtCO2 of more than 10 mmHg.18 Aljadeff et al found no significant change in average or highest EtCO2, or in hypoventilation time as a percent of total sleep time.19 Supplemental oxygen has been studied in preterm infants with central sleep apnea, and has been shown to decrease the frequency of central apneas and periodic breathing without producing an adverse effect on alveolar ventilation.14,15 The current study found no significant adverse effect on alveolar ventilation when supplemental oxygen was delivered to infants with OSA, consistent with our previous study in preterm infants. Although the average percentage of time spent with TrCO2 greater than 50 mmHg increased in the O2-PSG group, both EtCO2 and CO2 from blood gas did not show baseline hypoventilation or significant increase in CO2 with oxygen. The standard deviation was quite high for TrCO2 data, indicating that some infants did have significantly elevated TrCO2; however, our study is not designed to evaluate whether there is a subgroup of infants in whom hypoventilation may develop while being treated with supplemental oxygen. For that reason we do not recommend use of supplemental oxygen for treatment of infants with OSA without first performing PSG for the purpose of titrating to the appropriate flow, and for evaluation of pCO2 to rule out development of hypoventilation while on supplemental oxygen.
In the current study, oxygen administration is associated with an increased percentage of NREM sleep and a concomitant decreased percentage of REM sleep. This observed increase in percentage of NREM sleep with oxygen administration is consistent with previous studies in preterm infants with CSA and infants with chronic lung disease.14,45 Although the decreased REM sleep could potentially have an adverse effect, there is no existing literature to support this notion in infants. On the contrary, various studies have shown benefits of quiet (NREM) sleep compared to active (REM) sleep, including decreased energy expenditure in quiet sleep that may favor growth,46 increased cerebral hemoglobin concentration,47 decreased oxygen consumption and energy conservation in quiet sleep,48 and fewer neurodevelopmental disabilities in infants who have a greater proportion of quiet sleep.49 Increased quiet sleep may contribute to improved growth in infants with chronic lung disease who maintain higher oxygenation, coinciding with improved growth hormone urinary excretion.50 By producing an increase in quiet sleep time, supplemental oxygen administration may result in a reduction in sleep fragmentation and promote growth and development. Of note, the use of supplemental oxygen use was associated with a decrease in total sleep time and sleep efficiency, which could potentially lead to adverse outcome.
CONCLUSIONS
Our study evaluated the effect of administering low-flow supplemental oxygen to infants with significant OSA. Infants experienced a significant improvement in overall oxygenation and a reduction in frequency of both obstructive and central respiratory events. Although there was slight prolongation of obstructive apneas, the proportion of arousals following respiratory events was increased. In addition, there was no significant adverse effect of oxygen on alveolar ventilation. Sleep architecture was improved with oxygen administration as evidenced by an increase in NREM sleep density. Supplemental oxygen may be considered as an alternative treatment for infants who are not good candidates for CPAP or surgery. Further study is needed to evaluate maturational changes of respiratory stability and neurocognitive developmental outcomes in infants treated with supplemental oxygen.
DISCLOSURE STATEMENT
Work for this study was performed at Cincinnati Children’s Hospital Medical Center. All authors have seen and approved the manuscript. Part of this work was presented at the International Conference of the American Thoracic Society 2013, Philadelphia, Pennsylvania. Dr. Brockbank received an ATS Abstract Scholarship Award from the Pediatric Assembly for this research project. Financial support was provided by the Cincinnati Children’s Hospital Research Fund. None of the authors received any additional financial support. The authors report no conflicts of interest.
ABBREVIATIONS
- AHI
apnea-hypopnea index
- CBG
capillary blood gas
- CI
central index
- CPAP
continuous positive airway pressure
- EtCO2
end-tidal carbon dioxide
- NREM
non-rapid eye movement
- O2-PSG
oxygen titration sleep study
- OAHI
obstructive apnea-hypopnea index
- OAI
obstructive apnea index
- OSA
obstructive sleep apnea
- PSG
polysomnography
- RA-PSG
room air diagnostic sleep study
- REM
rapid eye movement
- TrCO2
transcutaneous carbon dioxide
REFERENCES
- 1.Gozal D. Sleep, sleep disorders and inflammation in children. Sleep Med. 2009;10(Suppl 1):S12–S16. doi: 10.1016/j.sleep.2009.07.003. [DOI] [PubMed] [Google Scholar]
- 2.Lumeng JC, Chervin RD. Epidemiology of pediatric obstructive sleep apnea. Proc Am Thorac Soc. 2008;5(2):242–252. doi: 10.1513/pats.200708-135MG. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Rosen CL, Larkin EK, Kirchner HL, et al. Prevalence and risk factors for sleep-disordered breathing in 8- to 11-year-old children: association with race and prematurity. J Pediatr. 2003;142(4):383–389. doi: 10.1067/mpd.2003.28. [DOI] [PubMed] [Google Scholar]
- 4.Gislason T, Benediktsdottir B. Snoring, apneic episodes, and nocturnal hypoxemia among children 6 months to 6 years old. An epidemiologic study of lower limit of prevalence. Chest. 1995;107(4):963–966. doi: 10.1378/chest.107.4.963. [DOI] [PubMed] [Google Scholar]
- 5.Anuntaseree W, Kuasirikul S, Suntornlohanakul S. Natural history of snoring and obstructive sleep apnea in Thai school-age children. Pediatr Pulmonol. 2005;39(5):415–420. doi: 10.1002/ppul.20207. [DOI] [PubMed] [Google Scholar]
- 6.Brunetti L, Rana S, Lospalluti ML, et al. Prevalence of obstructive sleep apnea syndrome in a cohort of 1,207 children of southern Italy. Chest. 2001;120(6):1930–1935. doi: 10.1378/chest.120.6.1930. [DOI] [PubMed] [Google Scholar]
- 7.Kahn A, Groswasser J, Sottiaux M, et al. Clinical symptoms associated with brief obstructive sleep apnea in normal infants. Sleep. 1993;16(5):409–413. doi: 10.1093/sleep/16.5.409. [DOI] [PubMed] [Google Scholar]
- 8.Montgomery-Downs HE, Gozal D. Sleep habits and risk factors for sleep-disordered breathing in infants and young toddlers in Louisville, Kentucky. Sleep Med. 2006;7(3):211–219. doi: 10.1016/j.sleep.2005.11.003. [DOI] [PubMed] [Google Scholar]
- 9.Sharma PB, Baroody F, Gozal D, Lester LA. Obstructive sleep apnea in the formerly preterm infant: an overlooked diagnosis. Front Neurol. 2011;2:73. doi: 10.3389/fneur.2011.00073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Arens R, Marcus CL. Pathophysiology of upper airway obstruction: a developmental perspective. Sleep. 2004;27(5):997–1019. doi: 10.1093/sleep/27.5.997. [DOI] [PubMed] [Google Scholar]
- 11.Ramgopal S, Kothare SV, Rana M, Singh K, Khatwa U. Obstructive sleep apnea in infancy: a 7-year experience at a pediatric sleep center. Pediatr Pulmonol. 2014;49(6):554–560. doi: 10.1002/ppul.22867. [DOI] [PubMed] [Google Scholar]
- 12.Don DM, Geller KA, Koempel JA, Ward SD. Age specific differences in pediatric obstructive sleep apnea. Int J Pediatr Otorhinolaryngol. 2009;73(7):1025–1028. doi: 10.1016/j.ijporl.2009.04.003. [DOI] [PubMed] [Google Scholar]
- 13.Landau YE, Bar-Yishay O, Greenberg-Dotan S, Goldbart AD, Tarasiuk A, Tal A. Impaired behavioral and neurocognitive function in preschool children with obstructive sleep apnea. Pediatr Pulmonol. 2012;47(2):180–188. doi: 10.1002/ppul.21534. [DOI] [PubMed] [Google Scholar]
- 14.Simakajornboon N, Beckerman RC, Mack C, Sharon D, Gozal D. Effect of supplemental oxygen on sleep architecture and cardiorespiratory events in preterm infants. Pediatrics. 2002;110(5):884–888. doi: 10.1542/peds.110.5.884. [DOI] [PubMed] [Google Scholar]
- 15.Weintraub Z, Alvaro R, Kwiatkowski K, Cates D, Rigatto H. Effects of inhaled oxygen (up to 40%) on periodic breathing and apnea in preterm infants. J Appl Physiol. 1992;72(1):116–120. doi: 10.1152/jappl.1992.72.1.116. [DOI] [PubMed] [Google Scholar]
- 16.Rigatto H, Brady JP. Periodic breathing and apnea in preterm infants. I. Evidence for hypoventilation possibly due to central respiratory depression. Pediatrics. 1972;50(2):202–218. [PubMed] [Google Scholar]
- 17.Mehta V, Vasu TS, Phillips B, Chung F. Obstructive sleep apnea and oxygen therapy: a systematic review of the literature and meta-analysis. J Clin Sleep Med. 2013;9(3):271–279. doi: 10.5664/jcsm.2500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Marcus CL, Carroll JL, Bamford O, Pyzik P, Loughlin GM. Supplemental oxygen during sleep in children with sleep-disordered breathing. Am J Respir Crit Care Med. 1995;152(4 Pt 1):1297–1301. doi: 10.1164/ajrccm.152.4.7551385. [DOI] [PubMed] [Google Scholar]
- 19.Aljadeff G, Gozal D, Bailey-Wahl SL, Burrell B, Keens TG, Ward SL. Effects of overnight supplemental oxygen in obstructive sleep apnea in children. Am J Respir Crit Care Med. 1996;153(1):51–55. doi: 10.1164/ajrccm.153.1.8542162. [DOI] [PubMed] [Google Scholar]
- 20.Iber C, Ancoli-Israel S, Chesson AL, Jr, Quan SF. for the American Academy of Sleep Medicine . The AASM Manual for the Scoring of Sleep and Associated Events: Rules, Terminology and Technical Specifications. 1st ed. Westchester, IL: American Academy of Sleep Medicine; 2007. [Google Scholar]
- 21.Sands SA, Edwards BA, Terrill PI, et al. Identifying obstructive sleep apnoea patients responsive to supplemental oxygen therapy. Eur Respir J. 2018;52(3):1800674. doi: 10.1183/13993003.00674-2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Duke JC, Stahl ML, Parrish WE, Rundell OH, Orr WC. Predicting sleep apnea rates in infants. Sleep Res. 1984;13:201. [Google Scholar]
- 23.Smith PL, Haponik EF, Bleecker ER. The effects of oxygen in patients with sleep apnea. Am Rev Respir Dis. 1984;130(6):958–963. doi: 10.1164/arrd.1984.130.6.958. [DOI] [PubMed] [Google Scholar]
- 24.Gold AR, Schwartz AR, Bleecker ER, Smith PL. The effect of chronic nocturnal oxygen administration upon sleep apnea. Am Rev Respir Dis. 1986;134(5):925–929. doi: 10.1164/arrd.1986.134.5.925. [DOI] [PubMed] [Google Scholar]
- 25.Martin RJ, Sanders MH, Gray BA, Pennock BE. Acute and long-term ventilatory effects of hyperoxia in the adult sleep apnea syndrome. Am Rev Respir Dis. 1982;125(2):175–180. doi: 10.1164/arrd.1982.125.2.175. [DOI] [PubMed] [Google Scholar]
- 26.Hudgel DW, Hendricks C, Dadley A. Alteration in obstructive apnea pattern induced by changes in oxygen- and carbon-dioxide-inspired concentrations. Am Rev Respir Dis. 1988;138(1):16–19. doi: 10.1164/ajrccm/138.1.16. [DOI] [PubMed] [Google Scholar]
- 27.Landsberg R, Friedman M, Ascher-Landsberg J. Treatment of hypoxemia in obstructive sleep apnea. Am J Rhinol. 2001;15(5):311–313. [PubMed] [Google Scholar]
- 28.Gold AR, Bleecker ER, Smith PL. A shift from central and mixed sleep apnea to obstructive sleep apnea resulting from low-flow oxygen. Am Rev Respir Dis. 1985;132(2):220–223. doi: 10.1164/arrd.1985.132.2.220. [DOI] [PubMed] [Google Scholar]
- 29.Locke RG, Wolfson MR, Shaffer TH, Rubenstein SD, Greenspan JS. Inadvertent administration of positive end-distending pressure during nasal cannula flow. Pediatrics. 1993;91(1):135–138. [PubMed] [Google Scholar]
- 30.Salmone RJ, Van Lunteren E. Effects of hypoxia and hypercapnia on geniohyoid contractility and endurance. J Appl Physiol. 1991;71(2):709–715. doi: 10.1152/jappl.1991.71.2.709. [DOI] [PubMed] [Google Scholar]
- 31.Gauda EB, Carroll JL, McColley S, Smith PL. Effect of oxygenation on breath-by-breath response of the genioglossus muscle during occlusion. J Appl Physiol. 1991;71(4):1231–1236. doi: 10.1152/jappl.1991.71.4.1231. [DOI] [PubMed] [Google Scholar]
- 32.Gauda EB. Upper-Airway Muscle Control During Development. In: Mathew OP, ed. Respiratory Control and Disorders in the Newborn. Vol. 173. New York, NY: Taylor & Francis; 2003:115–148. [Google Scholar]
- 33.Badr MS, Skatrud JB, Simon PM, Dempsey JA. Effect of hypercapnia on total pulmonary resistance during wakefulness and during NREM sleep. Am Rev Respir Dis. 1991;144(2):406–414. doi: 10.1164/ajrccm/144.2.406. [DOI] [PubMed] [Google Scholar]
- 34.Wellman A, Malhotra A, Jordan AS, Stevenson KE, Gautam S, White DP. Effect of oxygen in obstructive sleep apnea: role of loop gain. Respir Physiol Neurobiol. 2008;162(2):144–151. doi: 10.1016/j.resp.2008.05.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Edwards BA, Sands SA, Berger PJ. Postnatal maturation of breathing stability and loop gain: the role of carotid chemoreceptor development. Respir Physiol Neurobiol. 2013;185(1):144–155. doi: 10.1016/j.resp.2012.06.003. [DOI] [PubMed] [Google Scholar]
- 36.Brockbank J, Edwards BA, Ishman S, Amin R, McConnell K, Simakajornboon N. The effect of loop gain on efficacy of supplemental oxygen for treatment of infants with obstructive sleep apnea (OSA) Sleep. 2017;40(Suppl 1):A332–A333. [Google Scholar]
- 37.Berry RB, Gleeson K. Respiratory arousal from sleep: mechanisms and significance. Sleep. 1997;20(8):654–675. doi: 10.1093/sleep/20.8.654. [DOI] [PubMed] [Google Scholar]
- 38.Ward SL, Bautista DB, Keens TG. Hypoxic arousal responses in normal infants. Pediatrics. 1992;89(5 Pt 1):860–864. [PubMed] [Google Scholar]
- 39.Marcus CL, Lutz J, Carroll JL, Bamford O. Arousal and ventilatory responses during sleep in children with obstructive sleep apnea. J Appl Physiol. 1998;84(6):1926–1936. doi: 10.1152/jappl.1998.84.6.1926. [DOI] [PubMed] [Google Scholar]
- 40.Stark AR, Thach BT. Recovery of airway patency after obstruction in normal infants. Am Rev Respir Dis. 1981;123(6):691–693. doi: 10.1164/arrd.1981.123.6.691. [DOI] [PubMed] [Google Scholar]
- 41.Mograss MA, Ducharme FM, Brouillette RT. Movement/arousals. Description, classification, and relationship to sleep apnea in children. Am J Respir Crit Care Med. 1994;150(6 Pt 1):1690–1696. doi: 10.1164/ajrccm.150.6.7952634. [DOI] [PubMed] [Google Scholar]
- 42.Motta J, Guilleminault C. Effects of oxygen administration in sleep-induced apneas. In: Guilleminault C, Dement WC, eds. Sleep apnea syndromes. New York, NY: A. R. Liss; 1978:137–144. [Google Scholar]
- 43.Guilleminault C, Cummiskey J, Motta J. Chronic obstructive airflow disease and sleep studies. Am Rev Respir Dis. 1980;122(3):397–406. doi: 10.1164/arrd.1980.122.3.397. [DOI] [PubMed] [Google Scholar]
- 44.Alford NJ, Fletcher EC, Nickeson D. Acute oxygen in patients with sleep apnea and COPD. Chest. 1986;89(1):30–38. doi: 10.1378/chest.89.1.30. [DOI] [PubMed] [Google Scholar]
- 45.Fitzgerald D, Van Asperen P, Leslie G, Arnold J, Sullivan C. Higher SaO2 in chronic neonatal lung disease: does it improve sleep? Pediatr Pulmonol. 1998;26(4):235–240. doi: 10.1002/(sici)1099-0496(199810)26:4<235::aid-ppul1>3.0.co;2-3. [DOI] [PubMed] [Google Scholar]
- 46.Thureen PJ, Phillips RE, Baron KA, DeMarie MP, Hay WW., Jr Direct measurement of the energy expenditure of physical activity in preterm infants. J Appl Physiol. 1998;85(1):223–230. doi: 10.1152/jappl.1998.85.1.223. [DOI] [PubMed] [Google Scholar]
- 47.Münger DM, Bucher HU, Duc G. Sleep state changes associated with cerebral blood volume changes in healthy term newborn infants. Early Hum Dev. 1998;52(1):27–42. doi: 10.1016/s0378-3782(98)00002-4. [DOI] [PubMed] [Google Scholar]
- 48.Telliez F, Bach V, Dewasmes G, Leke A, Libert JP. Effects of medium- and long-chain triglycerides on sleep and thermoregulatory processes in neonates. J Sleep Res. 1998;7(1):31–39. doi: 10.1046/j.1365-2869.1998.00083.x. [DOI] [PubMed] [Google Scholar]
- 49.Whitney MP, Thoman EB. Early sleep patterns of premature infants are differentially related to later developmental disabilities. J Dev Behav Pediatr. 1993;14(2):71–80. [PubMed] [Google Scholar]
- 50.Fitzgerald D, Van Asperen P, O’Leary P, et al. Sleep, respiratory rate, and growth hormone in chronic neonatal lung disease. Pediatr Pulmonol. 1998;26(4):241–249. doi: 10.1002/(sici)1099-0496(199810)26:4<241::aid-ppul2>3.0.co;2-1. [DOI] [PubMed] [Google Scholar]