Abstract
Purpose
Maintenance of a transparent corneal stroma is imperative for proper vision. The corneal stroma is composed of primarily collagen fibrils, small leucine-rich proteoglycans (SLRPs), as well as sparsely distributed cells called keratocytes. The lattice arrangement and spacing of the collagen fibrils that allows for transparency may be disrupted due to genetic mutations and injuries. The purpose of this study is to examine the therapeutic efficacy of human umbilical cord mesenchymal stem/stromal cells (UMSCs) in treating congenital and acquired corneal opacity associated with the loss of collagen V.
Methods
Experimental mice, i.e., wild-type, Col5a1f/f and Kera-Cre/Col5a1f/f (Col5a1∆st/∆st, collagen V null in the corneal stroma) mice in a C57BL/6J genetic background, were subjected to a lamellar keratectomy, and treated with or without UMSC (104 cells/cornea) transplantation via an intrastromal injection or a fibrin plug. In vivo Heidelberg retinal tomograph (HRT II) confocal microscopy, second harmonic generated (SHG) confocal microscopy, histology, and immunofluorescence microscopy were used to assess the corneal transparency of the regenerated corneas.
Results
Col5a1∆st/∆st mice display a cloudy cornea phenotype that is ameliorated following intrastromal transplantation of UMSCs. Loss of collagen V in Col5a1∆st/∆st corneas augments the formation of cornea scarring following the keratectomy. UMSC transplantation with a fibrin plug improves the healing of injured corneas and regeneration of transparent corneas, as determined with in vivo HRT II confocal microscopy. Second harmonic confocal microscopy revealed the improved collagen fibril lamellar architecture in the UMSC-transplanted cornea in comparison to the control keratectomized corneas.
Conclusions
UMSC transplantation was successful in recovering some corneal transparency in injured corneas of wild-type, Col5a1f/f and Col5a1∆st/∆st mice. The production of collagen V by transplanted UMSCs may account for the regeneration of corneal transparency, as exemplified by better collagen fiber organization, as revealed with SHG signals.
Introduction
The cornea plays an important role in proper vision. The cornea does so by having an organization consisting of three tissue layers: the epithelium, stroma, and endothelium. The cornea is transparent, and provides the major refractive power for producing a focused image on the retina. The cornea accounts for two-thirds of the eye’s total optical power [1]. Approximately 90% of the human cornea’s thickness is composed of stroma. The stroma consists of uniformly arranged collagen fibrils with an average diameter of 25 nm arranged in orthogonal lamellae. The stroma also consists of heterogeneously distributed keratocytes that form and maintain a collagenous matrix that aids in corneal curvature and transparency [2,3]. It is thought that the lattice structure and spacing of the collagen fibrils lead to transparency, and that this is dependent on proteoglycan–collagen interactions and collagen–collagen interactions [4-6]. Corneal proteoglycans include the small leucine-rich proteoglycan (SLRP) family [3,7-13].
Collagen V is a member of the fibril-forming subclass of collagens, and is involved in the regulation of fibril assembly [14,15]. Collagen V is a quantitatively minor component of predominantly type I collagen fibrils in most non-cartilaginous tissues. Collagen V makes up only 2–5% of the collagen in tissues, such as bone, tendon, and dermis, and comprises 10–20% of the corneal stroma [16,17]. Collagen V knockout mice (systemic targeted disruption of the Col5a1 gene) die at embryonic day 10 (E10) due to a nearly complete lack of collagen fibril formation [18]. Col5a+/− mice are viable, but display a haplodeficiency phenotype. Because of the 50% reduction in collagen V, there is a corresponding reduction in the fibril number and collagen content [18]. The complete dependence of fibril assembly on collagen V is indicative of its importance in regulating collagen fibrillogenesis.
Disruption of the collagen fibril architecture from conditions such as corneal stromal dystrophies (including congenital dystrophy, cornea plana, corneal injury, or surgical intervention) can lead to loss of visual acuity. Disruption of the collagen fibril architecture can also be due to an insult to the corneal stroma as a result of injury or surgical intervention [19-21]. Laser-assisted in-situ keratomileusis (LASIK), laser epithelial keratomileusis (LASEK), and photorefractive keratectomy (PRK) are some of the most common forms of refractive surgery. It is estimated that millions of individuals have had elective refractive surgery, and the number worldwide continues to grow [22]. These procedures can result in surgical complications, such as corneal stroma opacification. The cellular events leading to complications involve extracellular matrix reorganization, stromal remodeling, as well as wound contraction [23,24]. Other types of corneal insults come from corneal injuries, which can lead to scarring, and in severe cases, to corneal blindness due to persistent inflammation, loss of limbal epithelial stem cells, transformation of keratocytes into myofibrobasts, collagen fibril disorganization, etc. [25-27].
The therapeutic role of umbilical cord mesenchymal stem cells (UMSCs) has been previously demonstrated. Human UMSCs, similar to other MSCs, have been shown to differentiate into chondrocytes, adipocytes, and osteocytes [28,29]. Use of UMSCs has been shown to rescue the thin, cloudy corneas of lumican-null mice with little to no immune rejection or inflammation. Transplanted UMSCs were shown to produce lumican, as well as other keratocyte-derived molecules; thus, compensating for the loss of lumican [30].
Previous research has focused on developing an understanding of the role of collagen V in regulation of the collagen fibril architecture. This study aims to translate this learning into insight into how to efficaciously aid in corneal repair. By disrupting collagen V, we aim to gain a better understanding of the cascade of events involved in collagen fibril assembly, which will help in understanding the etiology of corneal disorders, in improving the safety and efficacy of refractive surgery, and in bringing hope for effective interventions to aid in corneal wound healing. In the scope of this study, the ability of UMSCs to differentiate into keratocytes may compensate for the lack of collagen V in the congenital model, and the irregular arrangement of collagen fibrils in the acquired model to reduce corneal opacity.
Methods
Animals
Animal care and use conformed to the Association for Research in Vision and Ophthalmology (ARVO) Statement for the Use of Animals in Ophthalmic and Vision Research. All animal protocols were approved by the Institutional Animal Care and Use Committee of the University of Cincinnati. Compound transgenic mice were generated by breeding Kera-Cre and Col5a1f/f mice. The bitransgenic mouse (Kera-Cre/Col5a1f/f: Col5a1∆st/∆st, collagen V null in the stroma) expresses Cre in keratocan-positive keratocytes found within the cornea stroma. Col5a1∆st/∆st mice are null for expression of Col5a1 in keratocytes. The alpha 1(V) chain is required for triple helix formation in all collagen V isoforms; therefore, the stroma is collagen V null [15]. Genotypes were identified with PCR using oligonucleotide primers specific for each of the transgenes [15]. PCR was performed as follows: 94 °C for 5 min, followed by 30 cycles of 94 °C for 30 s, 65 °C for 30 s, and 72 °C for 30 s, and 72 °C for 5 min. PCR products were analyzed by electrophoresis in 2% agarose gels. All experimental mice were 10–12 weeks of age for the congenital corneal opacity studies. Male and female mice were used. For the acquired corneal opacity studies, 8-week-old C57BL/6J mice (Jackson Laboratories, Bar Harbor, ME) were used.
Surgical procedures
Keratectomy
Mice were anesthetized with the combined intraperitoneal administration of xylazine (13 mg/kg) and ketamine (95 mg/kg), and the central cornea (2 mm in diameter) was demarcated with a trephine (Tru-Punch; Sklar, West Chester, PA). The corneal epithelium, along with the anterior stroma, was removed using microdissection scissors under a stereomicroscope.
Corneal scarification
A keratectomy wound was performed as described above, and allowed to heal for 14 days. Following this healing period, the mice were anesthetized as described above, and the central corneal epithelium (2 mm in diameter) removed using an Algerbrush Corneal Rust Ring Remover (Sigma Pharmaceuticals, North Liberty, IA).
Isolation and culture of human umbilical cord mesenchymal stem cells
Umbilical cords were obtained from the Christ Hospital according to protocols approved by the Institutional Review Board (IRB) of the University of Cincinnati Medical Center. Upon arrival, umbilical cords were washed with 70% ethanol, and subsequently, excess blood was removed with sequential washes in Earle’s Balanced Salt Solution (Thermo Fisher Scientific, Waltham, MA). Blood vessels were removed, and the tissue minced into fine pieces, and incubated with 0.05% trypsin (Fisher Scientific -Gibco (Waltham, MA)), and 300 U collagenase (Stem Cell Technologies, Vancouver, Canada) in alpha-MEM (Gibco) for 4 h at 37 °C. Thereafter, the cell suspension was filtered, centrifuged at 400 ×g, and the cells were cultured in alpha-MEM supplemented with 10% fetal bovine serum (FBS; Hyclone, Waltham, MA) in 5% CO2 atmosphere at 37 °C. After 16 h, the medium was changed to remove non-adherent cells. Thereafter, the medium was changed every 2 to 3 days, and cells harvested at approximately 70% confluency with Trypsin/EDTA, and subsequently, sub-cultured at a density of 3–6 × 103 cells/cm2. Cells at the fourth passage were stored in liquid nitrogen as previously described [29,30].
Stem cell transplantation
Intrastromal injection
UMSCs were transplanted via intrastromal injection into bitransgenic Col5a1∆st/∆st mice. The mice were anesthetized with 95 μg/g bodyweight of ketamine and 13 μg/g bodyweight of xylazine. A 32-gauge needle was used to make a tunnel in the corneal stroma. A 33-gauge needle was used to deliver 2 × 104 cells in 2 μl into the tunnel. Stem cells were transplanted to the oculus dexter (OD) and oculus sinister (OS), to serve as a contralateral control receiving only alpha-MEM.
Fibrin gel carrier
The fibrin carrier was prepared using fibrinogen and thrombin stock solutions (TISSEEL [Fibrin Sealant], Baxter, Deerfield, IL) dissolved in 1.1% NaCl and 1 mM CaCl2 to a final concentration of 10 mg/ml fibrinogen and 3 U/ml thrombin [31,32]. UMSCs were added to the thrombin solution before transplantation. Immediately following the keratectomy or epithelial debridement (corneal scarification), a 1:1 fibrinogen to thrombin (with or without 2 × 104 UMSCs) was mixed and added as a 7 µl eye drop. Stem cells were added to the fibrin carrier for OD while OS served as a contralateral control with fibrin gel only.
Analysis of corneal stromal thickness and haze
Studies of corneal thickness and haze were performed with in vivo confocal microscopy (Heidelberg Retinal Tomograph, Heidelberg, Germany; HRT II). Mice were given a single drop of GenTeal Gel (Alcon, Forth Worth, TX) on the eye before being placed next to the objective. Three-dimensional images were reconstructed using AxioVision imaging software (Carl Zeiss, Oberkochen, Germany), and assessed for pixel intensity using ImageJ software (National Institutes of Health, Bethesda, MD). The Student t test was used to determine statistical significance (p < 0.05).
Analysis of corneal fibril structure by imaging second harmonic generated signals
Enucleated eyes were fixed overnight in 4% paraformaldehyde, and then washed in PBS (1X: 137 mM NaCL, 2.7 mM KCl, 10mM Na2HPO4, 1.8 mM KH2PO4, pH7.4). The corneal button was excised, and subjected to nuclear staining using Syto59 (Thermo Fisher Scientific; 1:1,000) for 12 h at 4 °C, washed in PBS, and mounted on a slide with Mowiol medium (Calbiochem, La Jolla, CA). Corneas were imaged with a Zeiss LSM510 NLO two-photon microscope (Carl Zeiss, Oberkochen, Germany) with a 40X water-immersion objective. The nuclear Syto59 (Thermo Fisher Scientific) fluorescent signal was obtained using the 633 nm laser line from the He-Ne laser, and detected with a 650 LP filter. Nonlinear optical imaging of second harmonic generated (SHG) signals were obtained using the 800 nm MaiTai laser. Forward-scattered signals were collected using a 0.8 NA condenser lens with a 400/50 band-pass filter, which was placed in front of the transmission light detector. Backscattered SHG signals were collected using the Meta detector on the microscope [30,33].
Immunofluorescence microscopy
Enucleated eyes were fixed overnight in 4% paraformaldehyde, washed in PBS, and treated with 0.2% sodium borohydride for 45 min at room temperature. Following the PBS wash, the eyes were processed for either frozen or paraffin sections. Five micrometer paraffin and 10 µm cryosections were reacted with anti-Col5 collagen antibody [15].
Results
UMSCs ameliorate the cloudy corneas of Col5a1Δst/Δst mice
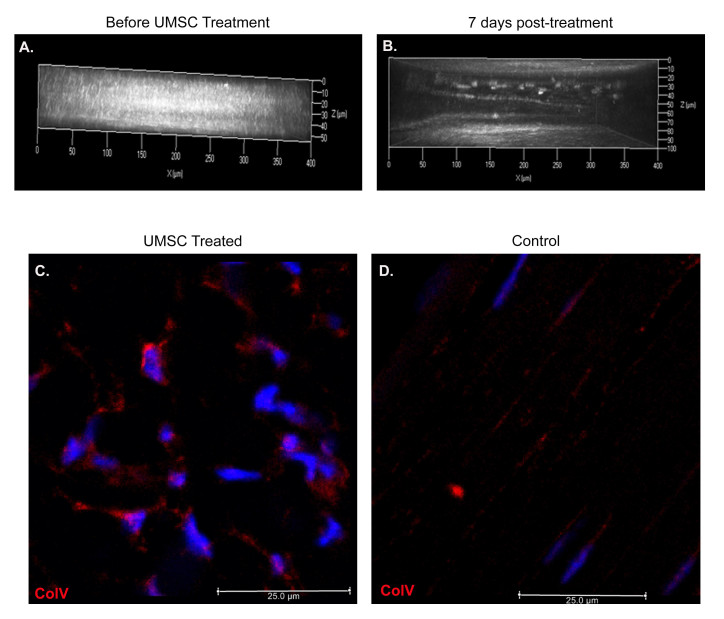
As previously reported, loss of collagen V in the stroma of Col5a1∆st/∆st mice results in a thin, cloudy cornea [15], due to the improper collagen fibril spacing and diameter. We previously showed that mesenchymal stem cells from the umbilical cord are able to correct the thin, cloudy corneas of lumican-null mice. Based on these results, we set out to determine the therapeutic efficacy of UMSCs in treating the opaque cornea of the Col5a1Δst/Δst mouse. UMSCs were transplanted via intrastromal injection into the corneas of 10-week-old Col5a1Δst/Δst mice, and monitored via HRT II to assess corneal opacity. Corneal transparency recovered 7 days following transplantation (Figure 1A,B). Eyes receiving UMSCs showed an increase in Col5a1 expression (Figure 1C); in comparison, Col5a1 expression was not detected in the control eye (Figure 1D).
Figure 1.
Recovery of corneal transparency in Col5a1Δst/Δst mice after UMSC treatment. A,B: Heidelberg retinal tomograph (HRT II) was performed on Col5a1Δst/Δst mice before (A) and 7 days after umbilical cord mesenchymal stem/stromal cell (UMSC) treatment (B). Three-dimensional renderings from volumetric scans were created using AxioVision 4.7 software. Corneal transparency recovers in collagen V-null corneas 7 days post-UMSC transplantation. C,D: Immunofluorescent staining for anti-collagen V antibodies in UMSC-treated (C) and control (D) eyes after 7 days. Note the Col5a expression in the corneal stroma; images from the peripheral cornea.
Recovery of corneal transparency and reorganization of fibril architecture in UMSC-treated eyes following injury
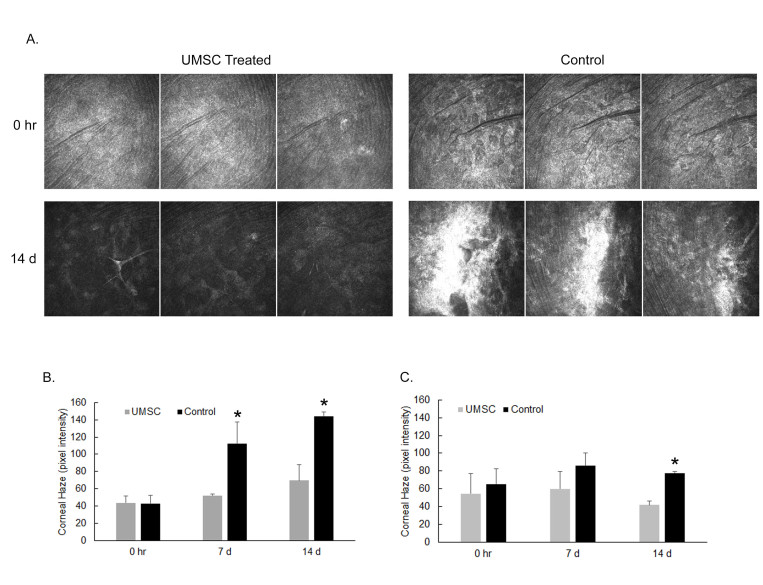
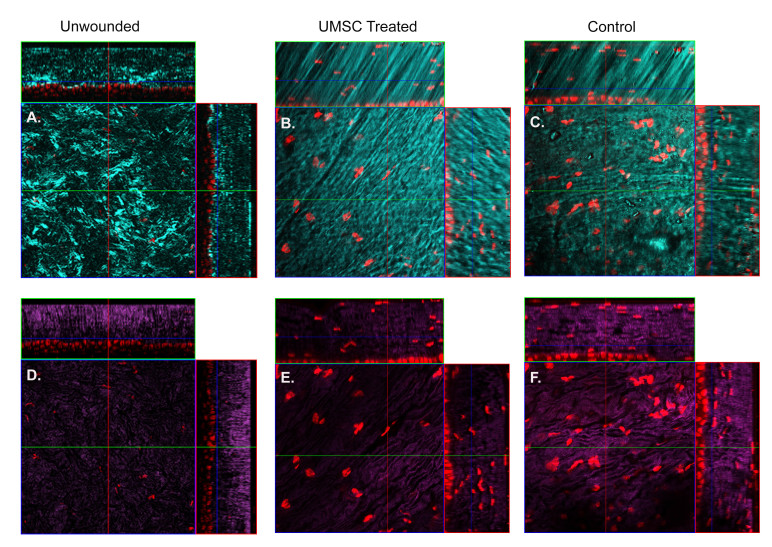
To assess the ability of UMSCs to treat a corneal wound in the Col5a1Δst/Δst mice, we first examined the role of collagen V in the corneal wound healing process. A 2-mm central keratectomy was made in the central cornea. HRT II imaging was performed 8 days and 15 days after injury to assess corneal transparency. Mice lacking Col5a1 failed to resolve the stromal haze; leading to stromal scarring (Figure 2). To determine the therapeutic efficacy of UMSCs in preventing stromal haze and scarring in the Col5a1Δst/Δst mice, the UMSCs were transplanted via fibrin gel immediately following the keratectomy, and analyzed at 7 days and 14 days post-transplant via HRT II. UMSCs were able to reduce the corneal opacity in the control Col5a1f/f and Col5a1Δst/Δst mice following injury, but the efficacy varied. The Col5a1Δst/Δst mice did not show improvement in corneal haze until 14 days compared to 7 days in the Col5a1f/f mice (Figure 3). Given the importance of the collagen arrangement in maintaining the transparent nature of the cornea, we set out to examine the stromal architecture by imaging SHG signals. Forward-scattered signals (cyan) showed that UMSC-treated corneas have a fibril structure more analogous to the control than do control corneas (Figure 4A–C). The backscattered signals (magenta) showed a less transparent and flattened lamellae in the control corneas compared to the corneas treated with UMSCs (Figure 4D–F).
Figure 2.
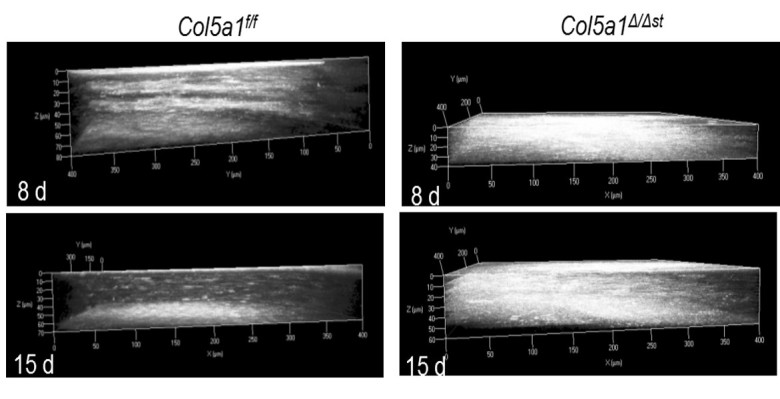
Stromal scarring in the absence of Col5a1 after injury. Heidelberg retinal tomograph (HRT II) was performed on control (Col5a1f/f) and mutant (Col5a1Δst/Δst) mice 8 days before and 15 days after the keratectomy. Three-dimensional renderings from volumetric scans were created using AxioVision 4.7 software. Loss of Col5a1 fails to resolve stromal haze and scarring.
Figure 3.
Recovery of corneal transparency in UMSC-treated eyes following injury in Col5a1-deficient mice. A, B: Representative CutView images acquired from a volumetric scan using in vivo confocal microscopy of Col5a1 control mice (A) and Col5a1-deficient mice (B). C, D: Pixel intensity of the stroma was obtained using Image J software. C: Control mice showed improvement in corneal haze at 7 and 14 days post-umbilical cord mesenchymal stem/stromal cell (UMSC) treatment. D: Col5a1-deficient mice showed significant improvement at 14 days. * p<0.05.
Figure 4.
Reorganization of collagen fibril architecture in UMSC-treated eyes following injury in Col5a1-deficient mice. A–C: Forward-scattered second harmonic generated (SHG) signals (cyan) depicting collagen fibrils. D–F: Backscattered SHG signals representing the overall lamellar structure of the stroma 7 days after treatment. The control eyes show enlarged collagen fibrils (C) and disorganized lamellae (F) compared to the transparent and flattened lamellae in the umbilical cord mesenchymal stem/stromal cells (UMSC)-treated eyes (B–E). Red = Syto59 nuclear stain. Seven days post-UMSC treatment. UMSC treatment more closely resembles an unwounded eye (A, D).
UMSCs prevent corneal opacity after wounding
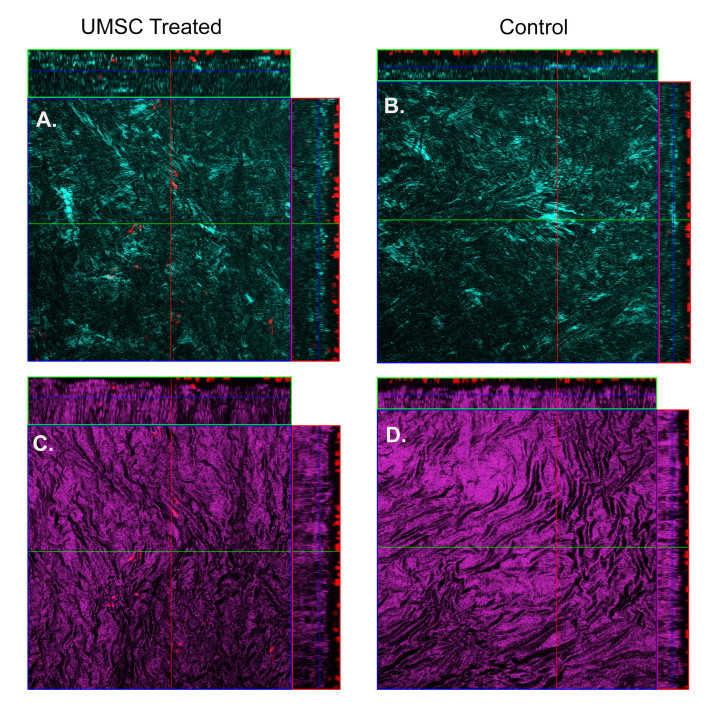
Given the therapeutic efficacy of UMSCs in treating congenital corneal opacity, we set out to determine the ability of these cells to treat acquired corneal opacity. To generate corneal opacity, a keratectomy wound was created, as described above, in C57BL/6J mice. Immediately following injury, UMSCs were transplanted to the surface of the eye via a fibrin carrier, and analyzed with HRT II at 8, 17, and 25 days post-injury. Eyes receiving UMSCs showed a reduced level of corneal opacity compared to the untreated contralateral eyes (Figure 5). UMSC treatment also allowed for the proper organization of the collagen fibril architecture. Forward-scattered images (cyan) showed that the UMSC-treated corneas had a fibril structure more analogous to the control than did the fibrin gel–treated corneas. The backscattered images (magenta) showed less transparent and flattened lamellae in the fibrin gel-treated corneas compared to the corneas treated with UMSCs (Figure 6).
Figure 5.
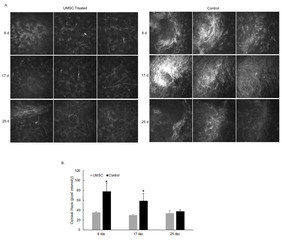
Reduction in acquired corneal haze. Stem cell treatment in C57BL/6J mice immediately following a 2-mm central keratectomy wound. A: A montage acquired from a volumetric scan using in vivo confocal microscope (Heidelberg retinal tomograph, HRT II). Three panels 5 μm apart showing the cornea stroma (anterior to posterior). B: Pixel intensity of the stroma was obtained using Image J software. Umbilical cord mesenchymal stem/stromal cells (UMSC) treatment was able to reduce corneal scarring at 8 days and 17 days post-treatment. * p<0.05.
Figure 6.
Reorganization of collagen fibril architecture in hUMSC-treated eyes. A–D: Forward-scattered second harmonic generated (SHG) images (cyan) show that human umbilical cord mesenchymal stem/stromal cells (hUMSC)-treated corneas (A, C) have a fibril structure more analogous to that of an uninjured eye than do the control corneas (B, D). Backscattered SHG images (magenta) show less transparent and disorganized lamellae control corneas (B, D) compared to the UMSC-treated corneas (A, C).
Treatment of corneal scarification with UMSCs
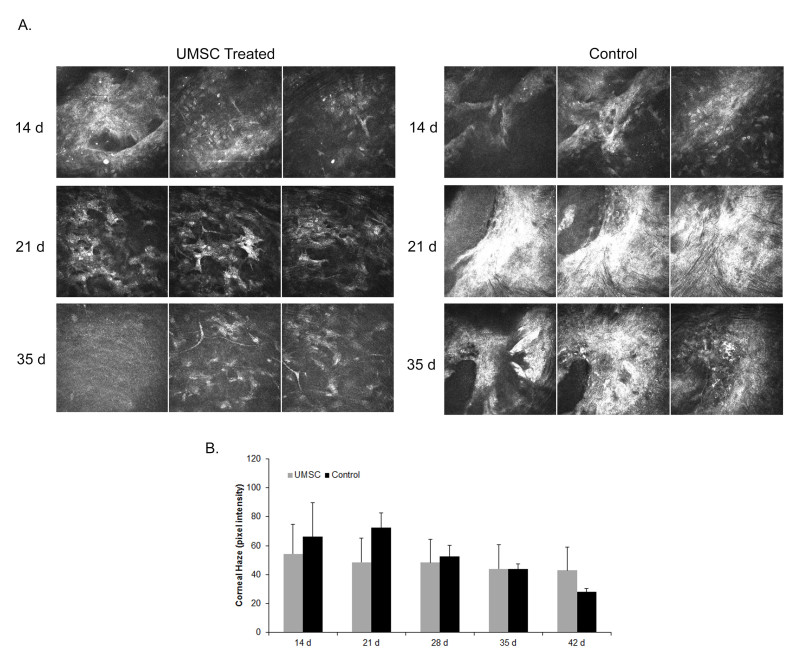
We showed that mice receiving UMSCs immediately following injury showed statistically significant improvement in corneal transparency at 8 days and 17 days post-transplant. Next, we wanted to determine the therapeutic efficacy of the UMSCs if the keratectomy wound was not treated immediately. Could the UMSCs reverse the corneal scar/fibrosis that occurs in mice following a keratectomy wound? To address this question, C57BL/6J mice received a 2-mm central keratectomy wound, as described, and were allowed to heal for 14 days. After 14 days, UMSCs were transplanted to the surface of the eye via the fibrin carrier. The UMSCs were unable to correct the corneal opacity, as the cells were unable to reach the corneal stroma, due to the corneal epithelium that covered the wounded area (data not shown). To address this issue, we debrided the corneal epithelium using a corneal rust ring remover immediately before transplantation. Mice were analyzed via HRT II before UMSC transplantation and weekly thereafter until day 42. Quantification of the corneal haze using Image J software to analyze the pixel intensity showed that the UMSCs trended toward having a therapeutic effect in terms of reducing corneal opacity; however, statistical significance was not reached (Figure 7).
Figure 7.
Treatment of corneal scarification with UMSCs. A 2-mm central keratectomy was created in C57BL/6J mice and left alone for 14 days at which time a scar developed. After 2 weeks, umbilical cord mesenchymal stem/stromal cells (UMSCs) were transplanted via a fibrin carrier onto the surface of the eye. A: A montage acquired from a volumetric scan using in vivo confocal microscopy (Heidelberg retinal tomograph, HRT II). Three panels 5 μm apart showing the cornea stroma (anterior to posterior). B: Pixel intensity of the stroma was obtained using Image J software. Data show no statistically significant difference between the UMSC-treated and control groups. Days represent time after keratectomy with treatment at 14 days.
Discussion
Transparency of the cornea is dependent on the diameter and spacing of collagen fibrils. Damage to the cornea stroma either from injury or refractive surgery can result in disruption of the collagen fibril arrangement, leading to corneal opacity. It has long been thought that collagen fibrils are inert, and not subject to quick turnover. In this study, we disputed this paradigm by showing that UMSC transplantation into Col5a1Δst/Δst mice reduced corneal opacity by reestablishing collagen fibril arrangement through the production of collagen V (Figure 1). Injury to mice lacking collagen V resulted in significant corneal opacity compared to Col5a1Δst/Δst mice that did not receive an injury, as the absence of collagen V does not allow for regulated fibril assembly (Figure 2). Collagen V expression as a result of UMSCs reduced corneal opacity 14 days post-transplantation. Improvement in corneal opacity occurred more quickly (7 days post-transplantation) in the control mice; indicating the importance of collagen fibril organization in the healing process (Figure 3). Forward- and backscattered second harmonic generated signals confirmed the reorganization of the collagen fibrils displaying a flattened lamellae and organized collagen fibrils (Figure 4). It is well understood that the biomechanical properties of the cornea depend, in large part, on the organization of the collagen fibrils [34]. Changes in collagen fibril alignment, diameter, and spacing occur following penetrating injury, which can lead to persistent changes, including corneal scar formation with mechanical strength properties that never achieve uninjured levels [35-39]. Structural changes occur in the cornea during short-term wound healing, including collagen fibril orientation changes and swelling, which reflect the dynamics of healing [38]. In this study, we showed that production of collagen V aided in reestablishing the collagen architecture, leading to a reduction in corneal opacity, including during wound healing. These data suggest that human collagen V can substitute for mouse collagen V; however, it is not clear whether heterotypic fibrils are generated from endogenous collagen I or from human collagen I.
Conventional treatment for corneal injury because of trauma, infection, or congenital defects varies depending on the severity of the injury. Minor corneal injuries are often treated with topical steroid medications that can significantly reduce the amount of scarring, but they can potentially affect vision. Injury resulting in scarring that penetrates deep into the stroma typically involves a phototherapeutic keratectomy, or in severe cases, corneal transplantation surgery. These treatments are restricted by the risk of infection and delayed healing, immune rejection of the transplant, and an overall shortage of donor corneas, due to the increased popularity of laser refractive surgery [40]. Given these limitations of the current treatments and the known therapeutic roles of MSCs, we evaluated the efficacy of UMSCs to treat acquired and congenital corneal opacities. MSCs have long been shown to be efficacious in various tissue injuries and inflammatory diseases [41-43]. MSCs have unique immunosuppressive properties, which enable the cells to evade host rejection [30,44]. UMSCs have been used to treat corneal opacities present in Lum−/− mice, and the congenital lysosomal storage disease MPS VII mouse model [30,44-48]. Similar to Col5a1Δst/Δst mice, Lum−/− mouse corneas are thin and opaque due to changes in the stromal architecture, specifically the irregularly spaced and thickened collagen fibrils that result from the lack of keratan sulfate proteoglycans [30,49]. Intrastromal injection of UMSCs increased corneal thickness, and reduced the corneal opacity, due to the production of keratan sulfate proteoglycans, which reestablished the organization of the collagen fibrils. In another study, it was found that corneal transparency could be restored through secretion of hepatocyte growth factor via MSCs [50]. The fact that UMSC transplantation was able to treat the corneal opacity in the congenital mouse model, but not in the acquired, scarification model, is suggestive of the importance of the mode of transplantation and the complexity of the altered corneal architecture. Intrastromal injection was used in the congenital model, whereas the acquired model used the fibrin application, given that this is more amenable to clinical application. Further research is needed, to optimize the treatment strategy to garner the greatest level of efficacy. Of course, one must also take into account the difference in the two models of corneal opacity and the underlying corneal stromal architecture. Loss of collagen V results in an increased fibril diameter, and reduced fibril density and inter-fibril spacing [15], whereas scarification results in abnormal alignment of the collagen fibrils. The results may suggest that UMSC transplantation may be able to alter fibril diameter and spacing, but may not be able to impact the overall alignment of the fibers. Alternatively, remodeling by removal of the fibrotic scar collagen matrix may be essential for regeneration of a well-organized newly synthesized collagen matrix for corneal transparency, rather than just simply providing the missing collagen V. The data provided in this study further confirm the therapeutic efficacy of MSCs, by demonstrating their ability to treat acquired and congenital opacities. Additionally, the data showed the ability for collagen fibril turnover following injury, when provided an environment amenable to healing. This amenable environment is provided by the ability of UMSCs to suppress inflammation. Future work will delve into the role of UMSCs in collagen fibril turnover and healing following injury, and for optimizing the treatment methodology.
Acknowledgments
This work was supported in part by grants NIH/NEI R01EY013755, Ohio Lions Eye Research Foundation and Ignite grant of Cincinnati Eye Institute.
References
- 1.Cassin B, Solomon S. Dictionary of Eye Terminology. Gainesville, Florida: Triad Publishing Company; 1990. [Google Scholar]
- 2.Hay ED. Development of the verterate cornea. Int Rev Cytol. 1980;63:263–322. doi: 10.1016/s0074-7696(08)61760-x. [DOI] [PubMed] [Google Scholar]
- 3.Linsenmayer TF, Fitch JM, Birk DE. Heterotypic collagen fibrils and stabilizing collagens. Controlling elements in corneal morphogenesis? Ann N Y Acad Sci. 1990;580:143–60. doi: 10.1111/j.1749-6632.1990.tb17926.x. Review. [DOI] [PubMed] [Google Scholar]
- 4.Doane KJ, Yang G, Birk DE. Corneal cell-matrix interactions: type VI collagen promotes adhesion and spreading of corneal fibroblasts. Exp Cell Res. 1992;200:490–9. doi: 10.1016/0014-4827(92)90200-r. [DOI] [PubMed] [Google Scholar]
- 5.Hahn RA, Birk DE. beta-D xyloside alters dermatan sulfate proteoglycan synthesis and the organization of the developing avian corneal stroma. Dev. 1992;115:383–93. doi: 10.1242/dev.115.2.383. [DOI] [PubMed] [Google Scholar]
- 6.Rada JA, Cornuet PK, Hassell JR. Regulation of corneal collagen fibrillogenesis in vitro by corneal proteoglycan (lumican and decorin) core proteins. Exp Eye Res. 1993;56:635–48. doi: 10.1006/exer.1993.1081. [DOI] [PubMed] [Google Scholar]
- 7.Bettelheim FA, Plessy B. The hydration of proteoglycans of bovine cornea. Biochim Biophys Acta. 1975;381:203–14. doi: 10.1016/0304-4165(75)90202-0. [DOI] [PubMed] [Google Scholar]
- 8.Hassell JR, Cintron C, Kublin C, Newsome DA. Proteoglycan changes during restoration of transparency in corneal scars. Arch Biochem Biophys. 1983;222:362–9. doi: 10.1016/0003-9861(83)90532-5. [DOI] [PubMed] [Google Scholar]
- 9.Iozzo RV. The family of the small leucine-rich proteoglycans: key regulators of matrix assembly and cellular growth. Crit Rev Biochem Mol Biol. 1997;32:141–74. doi: 10.3109/10409239709108551. [DOI] [PubMed] [Google Scholar]
- 10.Scott JE. Proteodermatan and proteokeratan sulfate (decorin, lumican/fibromodulin) proteins are horseshoe shaped. Implications for their interactions with collagen. Biochemistry. 1996;35:8795–9. doi: 10.1021/bi960773t. [DOI] [PubMed] [Google Scholar]
- 11.Weber IT, Harrison RW, Iozzo RV. Model structure of decorin and implications for collagen fibrillogenesis. J Biol Chem. 1996;271:31767–70. doi: 10.1074/jbc.271.50.31767. [DOI] [PubMed] [Google Scholar]
- 12.Zhang G, Chen S, Goldoni S, Calder BW, Simpson HC, Owens RT, McQuillan DJ, Young MF, Iozzo RV, Birk DE. Genetic evidence for the coordinated regulation of collagen fribrillogenesis in the cornea by decorin and biglycan. J Biol Chem. 2009;284:8888–97. doi: 10.1074/jbc.M806590200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Chen S, Sun M, Meng X, Iozzo RV, Kao WW, Birk DE. Pathophysiiological mechanisms of autosomal dominant congenital stromal corneal dystrophy: C-terminal-truncated decorin results in abnormal matrix assembly and altered expression of small leucine-rich proteoglycans. Am J Pathol. 2011;179:240–2419. doi: 10.1016/j.ajpath.2011.07.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Birk DE, Fitch JM, Babiarz JP, Linsenmayer TF. Collagen type I and type V are present in the same fibril in the avian corneal stroma. J Cell Biol. 1988;106:999–1008. doi: 10.1083/jcb.106.3.999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sun M, Chen S, Adams SM, Florer JB, Liu H, Kao WWY, Wenstrup RJ, Birk DE. Collagen V is a dominant regulator of collagen fibrillogenesis: dysfunctional regulation of structure and function in a corneal-stroma-specific Col5a1-null mouse model. J Cell Sci. 2011;124:4096–105. doi: 10.1242/jcs.091363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Birk DE. Type V collagen: heterotypic type I/V collagen interactions in the regulation of fibril assembly. Micron. 2001;32:223–37. doi: 10.1016/s0968-4328(00)00043-3. [DOI] [PubMed] [Google Scholar]
- 17.Segev F, Heon E, Cole WG, Wenstrup RJ, Young F, Slomovic AR, Rootman DS, Whitaker-Menezes D, Chervoneva I, Birk DE. Structural abnormalities of the cornea and lid resulting from collagen V mutations. Invest Ophthalmol Vis Sci. 2006;47:565–73. doi: 10.1167/iovs.05-0771. [DOI] [PubMed] [Google Scholar]
- 18.Wenstrup RJ, Florer JB, Brunskill EW, Bell SM, Chervoneva I, Birk DE, Type V. Collagen Controls the Initiation of Collagen Fibril Assembly. J Biol Chem. 2004;279:53331–7. doi: 10.1074/jbc.M409622200. [DOI] [PubMed] [Google Scholar]
- 19.Pellegata NS, Dieguez-Lucena JL, Joensuu T, Lau S, Montgomery KT, Krahe R, Kivela T, Kucherlapati R, Forsius H, de la Chapelle A. Mutations in KERA, encoding keratocan, cause cornea plana. Nat Genet. 2000;25:91–5. doi: 10.1038/75664. [DOI] [PubMed] [Google Scholar]
- 20.Liu CY, Birk DE, Hassell JR, Kane B, Kao WW. Keratocan-deficient Mice Display Alterations in Corneal Structure. J Biol Chem. 2003;278:21672–7. doi: 10.1074/jbc.M301169200. [DOI] [PubMed] [Google Scholar]
- 21.Bredrup C, Knappskog PM, Majewski J, Rodahl E, Boman H. Congenital Stromal Dystrophy of the Cornea Caused by a Mutation in the Decorin Gene. Invest Ophthalmol Vis Sci. 2005;46:420–6. doi: 10.1167/iovs.04-0804. [DOI] [PubMed] [Google Scholar]
- 22.Feder BJ. As economy slows, so do laser eye surgeries. The New York Times. April 24, 2008.
- 23.Netto MV, Mohan RR, Ambrosio RJ, Hutcheon AEK, Zieske JD, Wilson SE. Wound Healing in the Cornea: A Review of Refractive Surgery Complications and New Prospects for Therapy. Cornea. 2005;24:509–22. doi: 10.1097/01.ico.0000151544.23360.17. [DOI] [PubMed] [Google Scholar]
- 24.Marino GK, Santhiago MR, Torricelli AA, Santhanam A, Wilson SE. Corneal molecular and cellular biology for the refractive surgeon: the critical role of the epithelial basement membrane. J Refract Surg. 2016;32:118–25. doi: 10.3928/1081597X-20160105-02. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kao WW. Ocular surface tissue morphogenesis in normal and disease States revealed by genetically modified mice. Cornea. 2006;25(Suppl 1):S7–19. doi: 10.1097/01.ico.0000247207.55520.a4. [DOI] [PubMed] [Google Scholar]
- 26.Saika S, Ohnishi Y, Ooshima A, Liu CY, Kao WW. Epithelial repair: roles of extracellular matrix. Cornea. 2002;21:S23–9. doi: 10.1097/00003226-200203001-00006. [DOI] [PubMed] [Google Scholar]
- 27.Saika S, Okada Y, Miyamoto T, Yamanaka O, Ohnishi Y, Ooshima A, Liu CY, Weng D, Kao WWY. Role of p38 MAP kinase in regulation of cell migration and proliferation in healing corneal epithelium. Invest Ophthalmol Vis Sci. 2004;45:100–9. doi: 10.1167/iovs.03-0700. [DOI] [PubMed] [Google Scholar]
- 28.Weiss ML, Troyer DL. Stem cells in the umbilical cord. Stem Cell Rev. 2006;2:155–62. doi: 10.1007/s12015-006-0022-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Weiss ML, Medicetty S, Bledsoe AR, Rachakatla RS, Choi M, Merchav S, Luo Y, Rao MS, Velagaleti G, Troyer D. Human umbilical cord matrix stem cells: preliminary characterization and effect of transplantation in a rodent model of Parkinson’s disease. Stem Cells. 2006;24:781–92. doi: 10.1634/stemcells.2005-0330. [DOI] [PubMed] [Google Scholar]
- 30.Liu H, Zhang J, Liu CY, Wang IJ, Sieber M, Chang J, Jester JV. Cell Therapy of Congenital Corneal Diseases with Umbilical Mesenchymal Stem Cells: Lumican Null Mice. PLoS One. 2010;5:e10707. doi: 10.1371/journal.pone.0010707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Pellegrini G, Ranno R, Stracuzzi G, Bondanza S, Guerra L, Zambruno G, Micali G, De Luca M. The control of epidermal stem cells (holoclones) in the treatment of massive full-thickness burns with autologous keratinocytes cultured on fibrin. Transplantation. 1999;68:868–79. doi: 10.1097/00007890-199909270-00021. [DOI] [PubMed] [Google Scholar]
- 32.Meyer-Blazejewska EA, Call MK, Yamanaka O, Liu H, Schlotzer-Schrehardt U, Kruse FE, Kao WW. From hair to cornea: towards the therapeutic use of hair follicle-derived stem cells in the treatment of limbal stem cell deficiency. Stem Cells. 2011;29:57–66. doi: 10.1002/stem.550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Morishige N, Petroll WM, Nishida T, Kenney MC, Jester JV. Noninvasive corneal stromal collagen imaging using two-photon-generated second-harmonic signals. J Cataract Refract Surg. 2006;32:1784–91. doi: 10.1016/j.jcrs.2006.08.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Meek KM. Corneal collagen-its role in maintaining corneal shape and transparency. Biophys Rev. 2009;1:83–93. doi: 10.1007/s12551-009-0011-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Cintron C, Schneider H, Kublin C. Corneal scar formation. Exp Eye Res. 1973;7:251–9. doi: 10.1016/0014-4835(73)90176-0. [DOI] [PubMed] [Google Scholar]
- 36.Connon CJ, Meek KM. Organization of corneal collagen fibrils during the healing of trephined wounds in rabbits. Wound Repair Regen. 2003;11:71–8. doi: 10.1046/j.1524-475x.2003.11111.x. [DOI] [PubMed] [Google Scholar]
- 37.Kamma-Lorger CS, Hayes S, Boote C, Burghammer M, Boulton ME, Meek KM. Effects on collagen orientation in the cornea after trephine injury. Mol Vis. 2009;15:378–85. [PMC free article] [PubMed] [Google Scholar]
- 38.Kamma-Lorger CS, Boote C, Hayes S, Albon J, Boulton ME, Meek KM. Collagen ultrastructural changes during stromal wound healing in organ cultured bovine corneas. Exp Eye Res. 2009;88:953–9. doi: 10.1016/j.exer.2008.12.005. [DOI] [PubMed] [Google Scholar]
- 39.Ma J, Wang Y, Wei P, Jhanji V. Biomechanics and structure of the cornea: implications and association with corneal disorders. Surv Ophthalmol. 2018;63:851–61. doi: 10.1016/j.survophthal.2018.05.004. [DOI] [PubMed] [Google Scholar]
- 40.Hamil MB. Mechanical injury. Elsevier. 2011; 1169–1185. [Google Scholar]
- 41.Basu S, Hertsenberg AJ, Funderburgh ML, Burrow MK, Mann MM, Du Y, Lathrop KL, Syed-Picard FN, Adams SM, Birk DE, Funderburgh JL. Human limbal biopsy-derived stromal stem cells prevent corneal scarring. Sci Transl Med. 2014;6:266ra172. doi: 10.1126/scitranslmed.3009644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Uccelli A, Moretta L, Pistoia V. Mesenchymal stem cells in health and disease. Nat Rev Immunol. 2008;8:726–36. doi: 10.1038/nri2395. [DOI] [PubMed] [Google Scholar]
- 43.Wang J, Liao L, Tan J. Mesenchymal-stem-cell-based experimental and clinical trials: current status and open questions. Expert Opin Biol Ther. 2011;11:893–909. doi: 10.1517/14712598.2011.574119. [DOI] [PubMed] [Google Scholar]
- 44.Coulson-Thomas VJ, Gesteira TF, Hascall V, Kao W. Umbilical cord mesenchymal stem cells suppress host rejection: the role of the glycocalyx. J Biol Chem. 2014;289:23465–81. doi: 10.1074/jbc.M114.557447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Liu H, Zhang J, Liu CY, Hayashi Y, Kao WW. Bone marrow mesenchymal stem cells can differentiate and assume corneal keratocyte phenotype. J Cell Mol Med. 2012;16:1114–24. doi: 10.1111/j.1582-4934.2011.01418.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Coulson-Thomas VJ, Caterson B, Kao WW. Transplantation of human umbilical mesenchymal stem cells cures the corneal defects of mucopolysaccharidosis VII mice. Stem Cells. 2013;31:2116–26. doi: 10.1002/stem.1481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Kao WW. Coulson- Thomas VJ. Cell Therapy of Corneal Diseases. Cornea. 2016;35:S9–19. doi: 10.1097/ICO.0000000000001010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Joe AW, Gregory-Evans K. Mesenchymal stem cells and potential applications in treating ocular disease. Curr Eye Res. 2010;35:941–52. doi: 10.3109/02713683.2010.516466. [DOI] [PubMed] [Google Scholar]
- 49.Chakravarti S, Magnuson T, Lass JH, Jepsen KJ, LaMantia C, Carroll H. Lumican regulates collagen fibril assembly: skin fragility and corneal opacity in the absence of lumican. J Cell Biol. 1998;141:1277–86. doi: 10.1083/jcb.141.5.1277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Mittal SK, Omoto M, Amouzegar A, Sahu A, Rezazadeh A, Katikireddy KR, Shah DI, Sahu SK, Chauhan SK. Restoration of corneal transparency by mesenchymal stem cells. Stem Cell Reports. 2016;7:583–90. doi: 10.1016/j.stemcr.2016.09.001. [DOI] [PMC free article] [PubMed] [Google Scholar]