SUMMARY
Acute itch can be generated by either chemical or mechanical stimuli, which activate separate pathways in the periphery and spinal cord. While substantial progress has been made in mapping the transmission pathway for chemical itch, the central pathway for mechanical itch remains obscure. Using complementary genetic and pharmacological manipulations, we show that excitatory neurons marked by the expression of the neuropeptide Y1 receptor (Y1Cre neurons) form an essential pathway in the dorsal spinal cord for the transmission of mechanical but not chemical itch. Ablating or silencing the Y1Cre neurons abrogates mechanical itch, while chemogenetic activation induces scratching. Moreover, using Y1 conditional knockout mice, we demonstrate that endogenous neuropeptide Y (NPY) acts via dorsal horn Y1-expressing neurons to suppress light punctate touch and mechanical itch stimuli. NPY-Y1 signaling thus regulates the transmission of innocuous tactile information by establishing biologically relevant thresholds for touch discrimination and mechanical itch reflexes.
Graphical Abstract
In Brief
Acton et al. identify the excitatory neurons in the dorsal spinal cord that drive mechanical itch. These cells mediate responses to light punctate touch and are inhibited by neuropeptide Y (NPY)::Cre interneurons. Light touch sensitivity and mechanical itch responses are gated by NPY signaling mediated by Y1-expressing neurons in the dorsal horn.
INTRODUCTION
Itch is a protective cutaneous somatosensory modality that drives reflexive scratching to remove harmful parasites and irritants from the skin. Itch is elicited either by chemical pruritogens, including histamine and serotonin, or by light mechanical stimuli, such as an insect crawling across the skin (Dong and Dong, 2018; Hachisuka et al., 2018; Han and Dong, 2014; Ikoma, 2013; Ikoma et al., 2006). Although chemical and mechanical itch function in a complementary manner to protect against cutaneous tissue damage, there is growing evidence that they are mediated by separate neural pathways in the periphery and spinal cord.
The transmission pathway for chemical itch has been characterized in some detail (Bautista et al., 2014; Dong and Dong, 2018; Hachisuka et al., 2018; Ikoma, 2013). Chemical pruritogens activate pruriceptive Ad and C fibers that display little or no mechanical sensitivity (Ringkamp et al., 2011; Schmelz et al., 1997, 2003), and these convey the itch signal from the skin to the superficial dorsal horn. Within the dorsal horn, chemical itch is transmitted by neurons expressing the gastrin releasing peptide receptor (GRPR) (Bautista et al., 2014; Hachisuka et al., 2018; Han and Dong, 2014; Mishra and Hoon, 2013; Sun et al., 2009; Sun and Chen, 2007). Thereafter, the signal is relayed to the thalamus (Davidson et al., 2007, 2012; Johanek et al., 2008; Ma et al., 2012; Papoiu et al., 2012; Simone et al., 2004) and parabrachial nucleus (Campos et al., 2018; Mu et al., 2017) by projection neurons, including those marked by neurokinin-1 receptor (NK1R) expression (Akiyama et al., 2015). Much less is known about mechanical itch, which is initiated by the activation of low-threshold mechanoreceptors (LTMRs) in the skin (Fukuoka et al., 2013; Han and Dong, 2014; Koch et al., 2018). Recently, we showed that mechanical itch is selectively gated by inhibitory interneurons in the dorsal horn that are marked by the expression of Neuropeptide Y::Cre (NPY::Cre interneurons [INs]). Ablation of NPY::Cre INs in mice leads to chronic scratching in the absence of an external stimulus, as well as an increased tendency to scratch in response to low-threshold mechanical stimulation of the nape, consistent with disinhibited transmission of tactile information from the hairy skin (Bourane et al., 2015a). This mechanical itch phenotype is independent of the GRPR+ neuron-dependent chemical itch pathway, providing strong evidence for a separate mechanical itch pathway in the spinal cord. However, the excitatory spinal neurons that transmit the mechanical itch signal and the mechanism by which the inhibitory NPY::Cre INs gate the flow of information through this pathway remain wholly unknown.
In this study, we delineate a central circuit for the processing of mechanical itch that incorporates a peptidergic signaling mechanism to establish normal touch discrimination. We show that neurons in the dorsal spinal cord expressing Y1 and the transcription factor Lbx1, hereafter referred to as Y1Cre neurons, are essential for the central transmission of light punctate touch and mechanical but not chemical itch. Ablating the spinal Y1Cre neurons abolishes the chronic spontaneous scratching that occurs following removal of the NPY::Cre INs, while ablation or silencing spinal Y1Cre neurons renders mice insensitive to mechanical but not chemical stimulation. We go on to demonstrate that endogenous NPY acts on Y1+ dorsal horn neurons to suppress light punctate touch and mechanical itch. Lbx1Cre; Y1f/f conditional knockout mice exhibit spontaneous scratching and hypersensitivity to mechanical itch stimuli, as well as reduced thresholds for hindpaw withdrawal from light punctate stimuli, but, like Y1Cre neuron-ablated mice, they have normal responses to chemical itch and pain stimuli. Conversely, selective activation of Y1 receptors or activation of NPY::Cre INs to stimulate endogenous NPY release reduces mechanical itch responses. These data show that the mechanical itch pathway is gated by NPY-Y1 signaling within the dorsal horn. These data also indicate that the previously reported analgesic effects of NPY and Y1 signaling (Diaz-delCastillo et al., 2018; Duggan et al., 1991; Hua et al., 1991; Intondi et al., 2008; Naveilhan et al., 2001; Solway et al., 2011; Taiwo and Taylor, 2002) are mediated by peripheral sensory neurons.
RESULTS
Y1 Expression Marks a Population of Excitatory Neurons within the LTMR-RZ
Mechanical itch is mediated by light touch information transmitted to the spinal cord by LTMRs innervating the hairy skin (Fukuoka et al., 2013); this information is then subject to inhibitory gating by locally projecting NPY::Cre INs in the dorsal horn (Bourane et al., 2015a). We therefore centered our search for the excitatory neurons that transmit the mechanical itch signal on glutamatergic cells that are located within the LTMR recipient zone (LTMR-RZ) of the dorsal horn (Abraira et al., 2017). This zone (laminae IIi–IV) contains molecularly diverse excitatory populations that are extensively innervated by LTMRs (Abraira et al., 2017; Abraira and Ginty, 2013; Koch et al., 2018). Of particular interest was a subset of dorsal horn excitatory neurons distinguished by expression of the inhibitory Y1 receptor (NPYR1; Häring et al., 2018; Melnick, 2012; Miyakawa et al., 2005; Sathyamurthy et al., 2018). Y1 mRNA-positive neurons are distributed throughout the LMTR-RZ and are also present in laminae I–IIo (Figure 1A). In the spinal cords of adult Y1Cre (NPYR1Cre) knockin mice (Padilla et al., 2016) carrying the Ai14lsl-tdTom reporter (Madisen et al., 2010), Y1Cre-derived cells were enriched in laminae IIi/III, with additional cells present in laminae I, IIo, and IV (Figures 1B and S1C). 88.8% ± 2.0% of Y1+ cells in the dorsal horn co-expressed the Y1Cre-dependent tdTomato reporter, with 62.5% ± 7.9% of tdTomato+ neurons showing Y1 expression (Figures 1C and 1D). Likewise, when Y1Cre; Ai14lsl-tdTom mice were crossed with a Y1::EGFP transgenic reporter line (Gene Expression Nervous System Atlas [GENSAT]), 84.4% ± 4.6% of the EGFP+ neurons co-expressed tdTomato (Figures S1A–S1C) and 53.8% ± 3.4% of the tdTomato+ neurons co-expressed EGFP. These data suggest that Y1Cre captures dorsal horn neurons that exhibit transient or low-level Y1 gene expression in addition to cells that show persistent expression, as has been noted for other Cre drivers (e.g., Bourane et al., 2015a; Duan et al., 2014; Peirs et al., 2015).
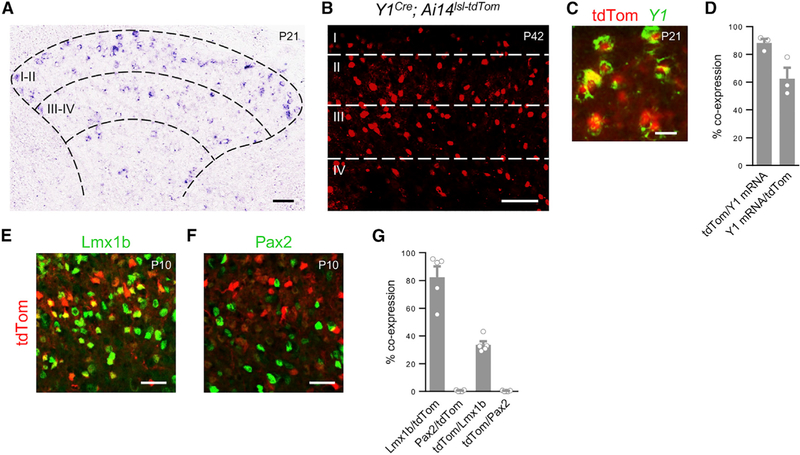
Figure 1. Y1Cre Marks a Population of Excitatory Neurons Concentrated in Laminae II-III.
(A) Transverse section through the lumbar spinal cord of a P21 mouse showing Y1 mRNA expression in the dorsal horn.
(B) Section from a P42 Y1Cre; Ai14lsl-tdTom mouse showing laminar distribution of Y1-tdTomato neurons.
(C) Co-expression of tdTomato and Y1 mRNA in the dorsal horn of a P21 Y1Cre; Ai14lsl-tdTom mouse.
(D) Summary of Y1 mRNA expression (n = 3 mice).
(E and F) Transverse sections through the lumbar spinal cord of a P10 Y1Cre; Ai14lsl-tdTom mouse stained with antibodies against Lmx1b (E) and Pax2 (F).
(G) Quantification of co-expression of Y1-tdTomato with antibody-labeled Lmx1b and Pax2 in P10 Y1Cre; Ai14lsl-tdTom mice (n = 5 mice). Roman numerals denote Rexed’s laminae.
Scale bars: 50 μm (A, B, E, and F) and 10 μm (C). Data: mean ± SEM. See also Figures S1–S3.
The vast majority of Y1Cre-derived neurons are excitatory, with 82.4% ± 6.9% expressing Lmx1b and only 0.4%± 0.2% expressing the inhibitory neuron marker Pax2 (Figures 1E–1G). Further-more, some tdTomato+ cells co-expressed markers of excitatory neuron populations that have been implicated in the transmission of mechanical stimuli, including cMaf+ neurons (Wende et al., 2012), retinoid-related orphan receptor alpha (RORα)+ neurons (Bourane et al., 2015b), and Somatostatin (Sst)+ neurons (Christensen et al., 2016; Duan et al., 2014; Huang et al., 2018). Co-expression was also seen in gastrin releasing peptide (GRP)-expressing neurons (Mishra and Hoon, 2013; Solorzano et al., 2015), as well as in NK1R+ neurons in lamina I and laminae III– IV (Akiyama et al., 2013, 2015; Carstens et al., 2010). By contrast, there was no overlap with neurons that express GRPR, or cells expressing the astrocytic markers S100b or glial fibrillary acidic protein (GFAP) (Figure S2).
In keeping with this analysis, sparse labeling of Y1Cre neurons in laminae I–IV with EnvA-pseudotyped ∆G-dsRed-Express rabies virus revealed cell morphologies that are characteristic of excitatory neurons. These include central-, vertical-, and radial-like cell types in lamina II (Grudt and Perl, 2002), and projection neurons in lamina I that likely correspond to NK1R+ projection neurons (Akiyama et al., 2015; Brumovsky et al., 2006; Szabo et al., 2015). We also observed fusiform neurons in lamina II and multipolar neurons in laminae III–IV, some of which were previously shown to express Y1 (Figure S3; Brumovsky et al., 2006).
To determine whether Y1Cre neurons receive direct LTMR inputs, we performed injections of cholera toxin B (CTb) into either the hairy skin of the thigh or the glabrous skin of the plantar hindpaw of adult Y1Cre; Ai14lsl-tdTom mice. CTb-labeled boutons from fibers innervating either region were found in close apposition to Y1Cre-tdTomato somata, and many of these displayed immunoreactivity for vesicular glutamate transporter 1 (vGluT1), which labels myelinated Aß and Aδ LTMRs in the LTMR-RZ (Figures 2A and 2B; n = 3 mice assessed per condition) (Todd et al., 2003).
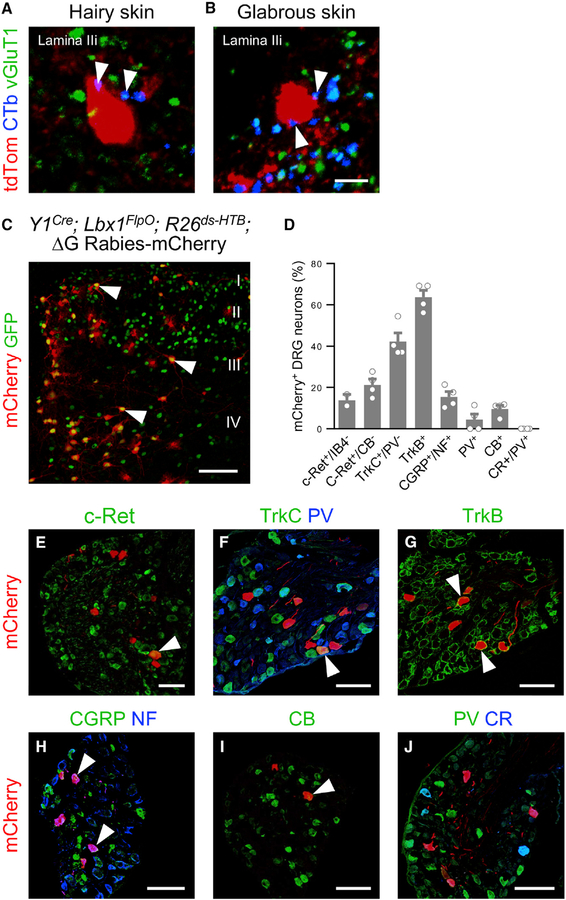
Figure 2. Y1Cre Neurons Receive Extensive LTMR Input.
(A and B) Examples of Y1Cre neurons in lamina IIi from lumbarspinal cord sectionsof P42 Y1Cre; Ai14lsl-tdTom mice injected with CTb into: the hairy skin of the thigh (A) and the glabrous skin of the hindpaw (B). Immunolabeled CTb+ contacts (blue) displayed vGluT1 immunoreactivity (green, arrowheads).
(C) Section through the lumbar dorsal horn of a P10 Y1Cre; Lbx1FlpO; R26ds-HTB mouse injected with EnvA G-deleted rabies-mCherry virus. Arrowheads indicate infected Y1Cre neurons. mCherry+/GFP— cells represent transsynaptically labeled presynaptic neurons.
(D) Summary of antibody-labeled myelinated sensory afferent subtypes that are presynaptic to the Y1Cre neurons, expressed as a percentage of mCherry+ neurons (n = 4 mice).
(E–J) Sections from P10 Y1Cre; Lbx1FlpO; R26ds-HTB lumbar DRGs showing presynaptically labeled sensory neurons(red) thatexpress c-Ret (E), TrkC but not parvalbumin(PV; F), TrkB(G), calcitonin gene-related peptide (CGRP) and neurofilament 200 (NF; H), and calbindin (CB; I), but not PV or calretinin (CR; J). Arrowheads indicate co-labeled sensory afferents. CB, calbindin; CR, calretinin; NF, neurofilament; PV, parvalbumin. Scale bars: 5 μm (B) and 100 μm (C and E–J). Data: mean ± SEM. See also Figure S4.
The LTMR subtypes that innervate Y1Cre neurons were further analyzed by intersectional monosynaptic retrograde tracing with EnvA-pseudotyped ∆G-mCherry rabies virus (Figures 2C–2J) (Albisetti et al., 2017; Bourane et al., 2015a, 2015b; Wickersham et al., 2007). Retrograde mCherry expression was observed in multiple cutaneous LTMR subtypes, including c-Ret+/IB4— Aß-LTMRs, TrkC+/parvalbumin— Aß-LTMRs, TrkB+ Aδ-LTMRs, and putative LTMRs that express calcitonin gene-related peptide (CGRP) and neurofilament 200 (NF200) (Bourane et al., 2015b; Lawson et al., 2002). Dorsal root ganglion (DRG) neurons expressing calbindin, which innervate Meissner corpuscles, were also labeled (Figure 2I). Proprioceptors marked by the expression of parvalbumin were largely spared, and Aß fibers expressing calretinin, which innervate Pacinian corpuscles, were never detected (Figure 2J). In summary, this analysis provides clear evidence that the Y1Cre neurons are extensively innervated by cutaneous LTMR fibers.
Y1Cre Neurons Are Essential Components of the Mechanical Itch Pathway Gated by Inhibitory NPY:: Cre INs
NPY::Cre IN ablation causes a mechanical itch phenotype that is marked by increased spontaneous hindlimb scratching (Figure 3A; Bourane et al., 2015a). We therefore set about examining whether the Y1Cre neurons in the dorsal horn transmit the mechanical itch signal using a genetic epistasis strategy entailing diphtheria toxin (DT)-mediated ablation of both the NPY::Cre/Lbx1 and Y1Cre/Lbx1 populations. Ablation of dorsal horn NPY::Cre INs alone significantly increased spontaneous scratching 1 week after DT treatment, from 12 ± 2 scratch episodes per 30-min observation period in FlpO— controls to 100 ± 17 episodes in ablated animals (Figures 3A, S4A, S4D, and S4E). Strikingly, this increase in spontaneous scratching was completely abolished following ablation of both the Y1Cre and NPY::Cre populations in DT-treated Y1Cre; NPY:: Cre; Lbx1FlpO; Tauds-DTR; Ai65ds-tdTom mice (Figures 3A, S4B, S4D, and S4E). These findings demonstrate that the Y1Cre neurons are required for the expression of the mechanical itch phenotype in mice lacking NPY::Cre-derived inhibitory INs.
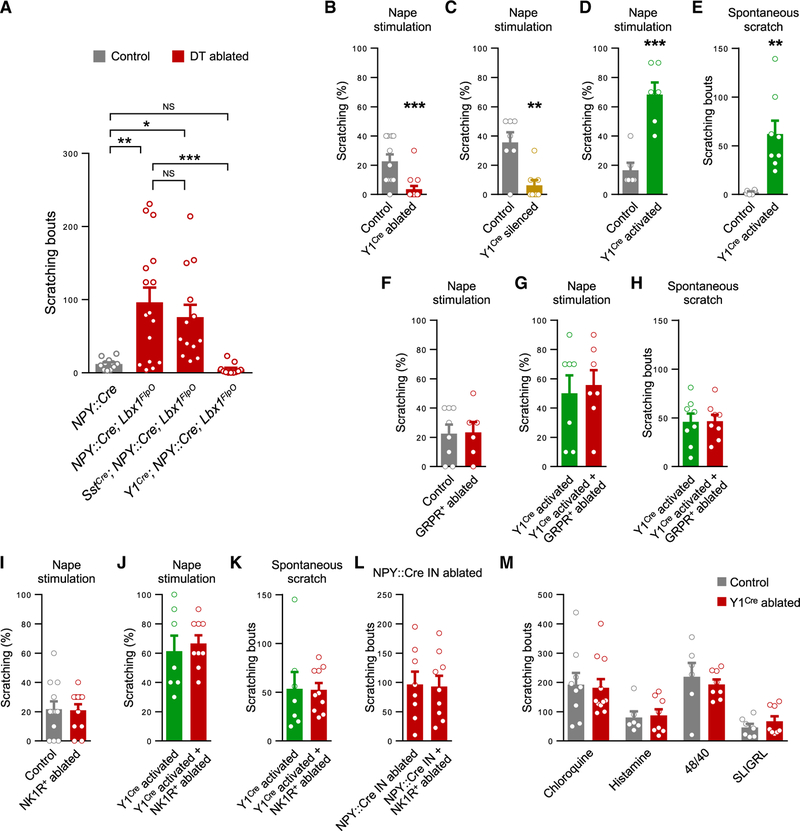
Figure 3. Y1Cre Neurons within the Dorsal Horn Transmit Mechanical but Not Chemical Itch.
(A) Ablation of dorsal horn NPY::Cre INs increases spontaneous scratching in NPY::Cre; Lbx1FlpO; Tauds-DTR; Ai65ds-tdTom mice (n = 16) compared with DT-treated NPY::Cre; Tauds-DTR; Ai65ds-tdTom controls that lack DT-receptor expression (n = 11). Ablation of the Y1Cre and NPY::Cre IN populations in Y1Cre; NPY::Cre; Lbx1FlpO; Tauds-DTR; Ai65ds-tdTom mice abolishes scratching (n = 11). Scratching is unaffected when Sst+ neurons are ablated together with the NPY::Cre INs in SstCre; NPY::Cre; Lbx1FlpO; Tauds-DTR; Ai65ds-tdTom mice (n = 13). One-way ANOVA and Bonferroni post hoc tests were used to assess statistical differences.
(B and C) Reduced scratching in response to stimulation of the nape with a 0.16-g von Frey hair in Y1Cre; Lbx1FlpO; Tauds-DTR; Ai65ds-tdTom mice treated with DT (n = 14) compared with saline-treated controls (n = 11; B), and in Y1Cre; Lbx1FlpO; R26ds-hM4D mice treated with clozapine N-oxide (CNO; n = 8) compared with Y1Cre; R26ds-hM4D controls, which lack FlpO-dependent hM4D expression (n = 8; C).
(D) Enhanced scratching in response to stimulation of the nape in CNO-treated Y1Cre; Lbx1FlpO; R26ds-hM3D mice (n = 6) compared with Y1Cre; R26ds-hM3D controls (n = 6).
(E) Spontaneous scratching in CNO-treated Y1Cre; Lbx1FlpO; R26ds-hM3D mice (n = 8) is enhanced over a 30-min period compared with control mice (n = 6).
(F) Scratching responses are unchanged in wild-type mice treated with bombesin-saporin (BOM-SAP; n = 6) to ablate GRPR+ neurons compared with controls injected with saporin (SAP; n = 8).
(G and H) Ablation of GRPR+ neurons in Y1Cre; Lbx1FlpO; R26ds-hM3D mice does not alter evoked scratching (G; BOM-SAP, n = 8; SAP, n = 7) or spontaneous scratching (H; BOM-SAP, n = 7; SAP, n = 7) following CNO injection.
(I) Scratching responses are unchanged in wild-type mice treated with [Sar9, Met(O2)11]-substance P-SAP (SSP-SAP; n = 10) to ablate NK1R+ neurons compared to SAP-injected controls (n = 11).
(J and K) In Y1Cre; Lbx1FlpO; R26ds-hM3D mice, ablation of NK1R+ neurons does not alter evoked (J; SSP-SAP, n = 9; SAP, n = 7) or spontaneous (K; SSP-SAP, n = 10; SAP, n = 7) scratching following CNO injection.
(L) Unchanged scratching in NPY::Cre; Lbx1FlpO; Tauds-DTR; Ai65ds-tdTom mice 2 weeks after NK1R+ neuron ablation and 1 week following DT administration (SSP-SAP, n = 9; SAP, n = 8).
(M) Unchanged scratching over a 30-min period in Y1Cre neuron-ablated mice following injection of chloroquine (control, n = 9; ablated, n = 11), histamine (control, n = 6; ablated, n = 8), compound 48/80 (control, n = 6; ablated, n = 8), and SLIGRL (control, n = 7; ablated, n = 8).
*p < 0.05, **p < 0.01, ***p < 0.001, NS, not significant. Data: mean ± SEM. See also Figures S4–S6.
Sst expression partially overlaps with Y1 expression in lamina II (Figures S2E–S2G; Zhang et al., 1999), and Sst signaling has been implicated in chemical itch transmission (Christensen et al., 2016; Huang et al., 2018; Kardon et al., 2014). We therefore set out to test whether NPY::Cre IN-gated scratching is mediated by Sst+ neurons in the dorsal horn. Strikingly, in contrast to the negation of scratching that occurs upon Y1Cre neuron depletion in NPY::Cre IN-ablated mice, simultaneous ablation of the NPY::Cre and Sst+ populations did not reduce scratching as compared to NPY::Cre IN-ablated controls (Figures 3A and S4C–S4E; p > 0.05). Furthermore, removing the Sst+ neurons alone did not alter spontaneous scratching or responses to mechanical stimulation of the nape (Figure S5). These data reveal that dorsal horn neurons expressing Y1, either transiently or persistently, are necessary for transmitting the light touch modalities that drive mechanical itch, whereas those that express Sst, including Sst+/Y1+ neurons, are not.
Y1Cre Neurons Selectively Transmit the Mechanosensory Stimuli That Drive Mechanical Itch
We next examined the function of the Y1Cre neurons in transmitting the itch sensation that is generated by low-threshold mechanical irritation of the hairy skin. Mechanical itch induction was assessed using a modified alloknesis assay (Akiyama et al., 2012) in which a low-force (0.16 g) von Frey hair was applied to the hairy skin of the nape, and scratching responses were recorded over ten trials. Whereas nape stimulation elicited scratching in 22.7% ± 4.7% of trials in control mice, it did so in only 3.6% ± 2.3% of trials in which the Y1Cre neurons were ablated (Figures 3B and S6A–S6B). A similar reduction in scratching was observed when the Y1Cre neurons were silenced in Y1Cre; Lbx1FlpO; R26ds-hM4D mice treated with clozapine N-oxide (CNO), as compared with CNO-treated Y1Cre; R26ds-hM4D control mice that lack FlpO-dependent expression of the hM4D receptor (Figure 3C). Conversely, scratching was greatly enhanced when the Y1Cre neurons were activated in Y1Cre; Lbx1FlpO; R26ds-hM3D animals (Figure 3D), which is consistent with heightened sensitivity to low-threshold tactile stimulation. There was also a marked increase in the number of spontaneous hindlimb scratching bouts over a 30-min period following hM3D-mediated activation of the Y1Cre neurons (Figure 3E).
We then considered the possibility that the increases in spontaneous and evoked scratching seen in Y1Cre; Lbx1FlpO; R26ds-hM3D mice might be attributable to the activation of the chemical itch pathway that is dependent on GRPR+ neurons. As previously shown, chloroquine-induced chemical itch was strongly reduced in wild-type mice following ablation of the GRPR+ neurons by intrathecal (i.t.) injection of saporin-conjugated bombesin (BOM-SAP; Figures S6C–S6E) (Bourane et al., 2015a; Huang et al., 2018; Sun et al., 2009). By contrast, scratching responses to nape stimulation were unaffected (Figure 3F). Consistent with these results, neither the evoked nor spontaneous scratching seen following activation of Y1Cre neurons was affected by ablation of the GRPR+ neurons (Figures 3G and 3H). These data agree with our previous report that spontaneous scratching induced by removal of NPY::Cre INs is not affected by ablation of the GRPR+ neurons (Bourane et al., 2015a), despite our finding that the Y1Cre neurons include some GRP-expressing neurons (Figures S2C, S2F, and S2G).
We next considered whether the pathways for chemical and mechanical itch transmission converge downstream of the GRPR+ neurons on lamina I NK1R+ neurons, given that Y1Cre and NK1R expression partially overlap (Figures S2D, S2F, and S2G) and evidence that NK1R+ projection neurons convey chemical itch sensation to key supraspinal regions for the integration of aversive stimuli (Carstens et al., 2010; Akiyama et al., 2013, 2015). Ablation of the NK1R+ neurons by injection of saporin conjugated to the NK1R ligand [Sar9, Met(O2)11]-substance P (SSP-SAP) significantly attenuated chloroquine-induced itch (Figures S6F–S6H) consistent with previous reports (Carstens et al., 2010). However, NK1R+ neuron ablation failed to modulate mechanically evoked itch in wild-type mice (Figure 3I), nor did it alter the rate of evoked or spontaneous scratching that occurs when the Y1Cre neurons are activated (Figures 3J and 3K). Ablation of the NK1R+ neurons also did not alleviate scratching induced by ablation of NPY::Cre INs (Figure 3L).
As a final test to exclude the possibility that Y1Cre neurons are interposed in the chemical itch circuitry, we assessed scratching in Y1Cre neuron-ablated mice following injection of chloroquine, histamine, compound 48/80 or SLIGRL into the skin behind the ear. Scratching responses to all four pruritogens remained intact in Y1Cre neuron-ablated mice (Figure 3M), confirming that these cells are not required for chemical itch, Together, these data indicate that that the minority of Y1Cre neurons that co-express NK1R are dispensable for mechanical itch sensation.
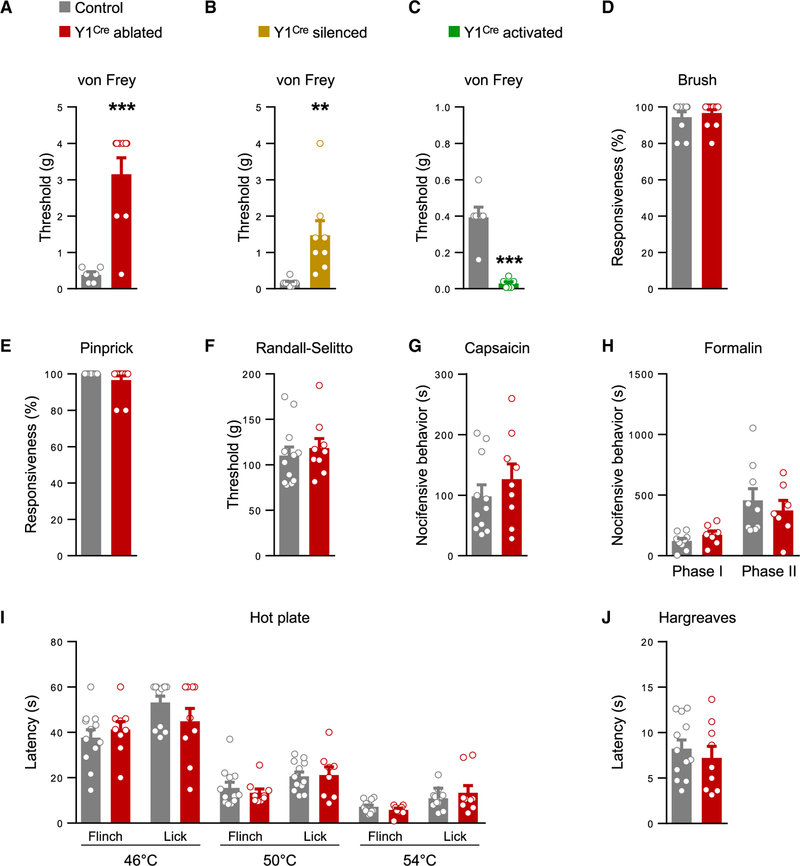
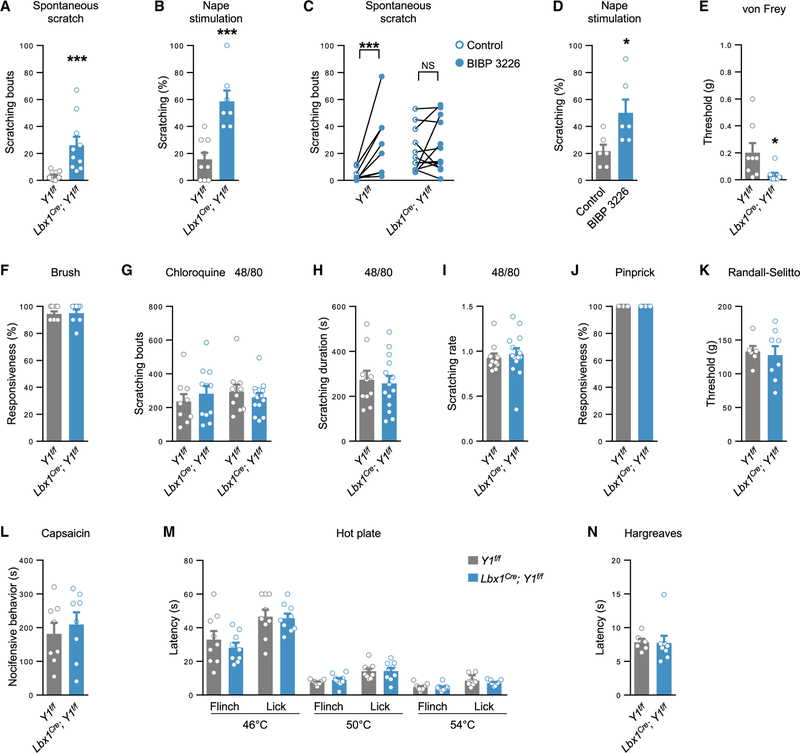
Our demonstration that the Y1Cre neurons are essential for mechanical itch led us to ask whether the Y1Cre neurons have a role in transmitting low-threshold mechanical stimuli. Sensitivity to light punctate touch, as assessed by von Frey hair stimulation of the glabrous skin, was significantly reduced after ablating or silencing the Y1Cre neurons (Figures 4A and 4B). Conversely, sensitivity was elevated by activation of the Y1Cre neurons (Figure 4C). However, responses to light dynamic touch, as assessed by gentle brushing, were unaffected (Figure 4D). This finding is consistent with our observation that there is little, if any, overlap between the Y1Cre and RORα+ neuronal populations (Figures S2B, S2F, and S2G), the latter of which transmits dynamic touch (Bourane et al., 2015b). Sensitivity to acute pain, as assessed by the pinprick and Randall-Sellito tests, was largely unchanged in Y1Cre neuron-ablated mice, as were responses to pain chemically induced by intradermal injection of capsaicin or formalin. Thermal pain, as assessed by the hot-plate and Hargreaves assays, was likewise unaffected (Figures 4E–4J). Together, these findings suggest that the Y1Cre population of neurons is largely specialized for transmitting light touch information, and they play little or no role in relaying chemical itch, noxious mechanical, or thermal information.
Figure 4. Y1Cre Neurons Selectively Transmit Light Touch Information.
(A and B) Sensitivity to von Frey hair stimulation of the hindpaw glabrous skin is reduced following Y1Cre neuron ablation (A; control, n = 6; ablated, n = 9) or silencing (B; control, n = 8; silenced, n = 8).
(C) Glabrous skin sensitivity to von Frey hair stimulation is elevated following Y1Cre neuron activation (control, n = 6; activated, n = 6).
(D–F) Responses to dynamic touch (D; control, n = 11; ablated, n = 13), pinprick (E; control, n = 11; ablated, n = 13), and Randall-Selitto (F; control, n = 12; ablated, n = 9) are unchanged after Y1Cre neuron ablation.
(G and H) Chemical pain responses following hindpaw injection of capsaicin (G; control, n = 11; ablated, n = 9) or formalin (H; control, n = 9; ablated, n = 7) are not altered following Y1Cre neuron ablation.
(I and J) Y1Cre neuron-ablated mice show normal heat responses as assessed by the hot plate (I; control, n = 12; ablated, n = 9) and Hargreaves (J; control, n = 12; ablated, n = 9) assays.
**p < 0.01, ***p < 0.001. Data: mean ± SEM.
NPY Peptide Signaling Regulates Mechanical Itch Transmission
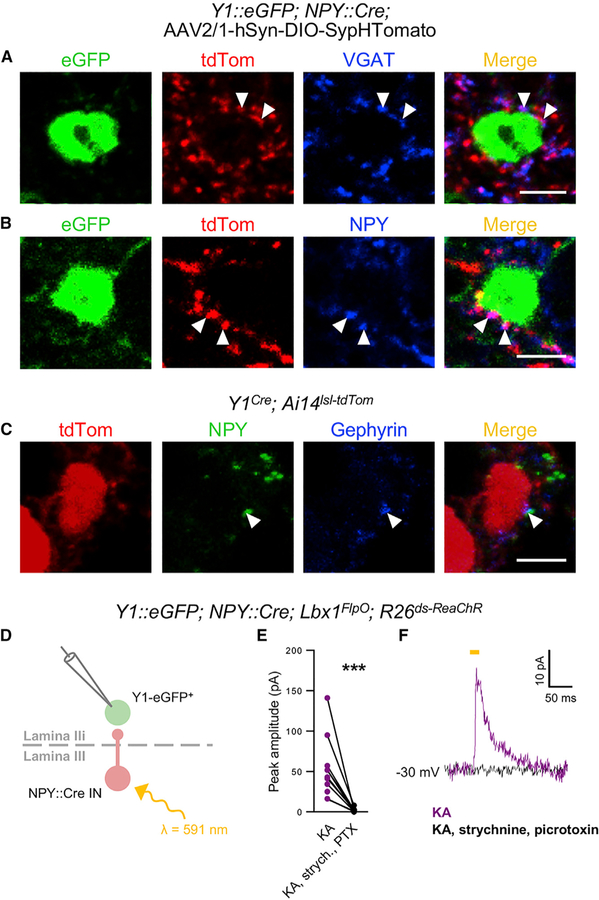
The underlying signaling mechanisms that contribute to the gating of mechanical itch by the NPY::Cre INs have not been established. In Y1::EGFP; NPY::Cre mice injected intraspinally (T13-L1 levels) with an AAV2/1-hSyn-DIO-SypHTomato virus, lamina II EGFP+ cell bodies displayed tdTomato+ synaptic contacts from NPY::Cre INs (Figures 5A and 5B; n = 3 mice). These contacts colocalized with vesicular GABA (gamma-aminobutyric acid) transporter (VGAT), indicating that NPY::Cre INs provide inhibitory synaptic inputs to Y1+ neurons (Figure 5A). Strikingly, many puncta also had NPY immunoreactivity (Figure 5B), with multiple NPY immunoreactive puncta being co-localized with the postsynaptic marker gephyrin on Y1-tdTomato+ somata in Y1Cre; Ai14lsl-tdTom mice (Figure 5C; n = 3).
Figure 5. Y1Cre Neurons Receive VGAT+ and NPY+ Synaptic Contacts from NPY::Cre Ins.
(A and B) Examples of GFP-labeled cells from the lumbar spinal cord of a P60 Y1::EGFP; NPY::Cre transgenic mouse. Presynaptic contacts (red) from NPY::Cre INs onto Y1/EGFP+ cells were visualized with a Cre-dependent AAV2/1-hSyn-DIO-SypHTom virus. Putative synaptic boutons marked by VGAT (blue; A) and NPY (blue; B) immunoreactivity are indicated by arrowheads.
(C) Examples of synaptic puncta labeled with antibodies against NPY (green) and gephyrin (blue) on a Y1-tdTomato+ cell body in the lumbar spinal cord of a P42 Y1Cre; Ai14lsl-tdTom mouse (arrowheads).
(D) Schematic illustrating experimental conditions used to assess synaptic connectivity between NPY::Cre INs and Y1Cre neurons.
(E) ReaChR-mediated activation of NPY::Cre IN generated monosynaptic outward currents in lamina IIi Y1-EGFP+ neurons in P14–21 Y1::EGFP; NPY::Cre; Lbx1FlpO; R26ds-ReaChR spinal cord slices in the presence of kynurenic acid (KA; 1.5 mM) at a holding potential of —30 mV. These currents were abolished following application of 1 μM strychnine and 60 μM picrotoxin (n = 7 cells from 5 mice). ***p < 0.001. The statistical difference was determined by the two-tailed, paired t test.
(F) Traces recorded from a Y1-EGFP+ neuron showing a monosynaptic inhibitory current elicited by NPY::Cre activation (purple trace) and no response following bath application of strychnine and picrotoxin (black trace).
Scale bars: 5 μm.
To confirm that the NPY::Cre INs form functional synapses onto Y1+ neurons, we performed whole-cell patch-clamp recordings from lamina IIi Y1-EGFP cells in spinal cord sections from Y1::EGFP; NPY::Cre; Lbx1FlpO; R26ds-ReaChR mice (Hooks et al., 2015). Following current injection, the Y1-EGFP neurons displayed either single (20%) or phasic firing patterns (80%; n = 32) that are characteristic of dorsal excitatory neurons (Abraira et al., 2017; Grudt and Perl 2002; Koch et al., 2018). We then blocked glutamatergic transmission with kynurenic acid and recorded from Y1-EGFP neurons at a holding potential of —30 mV. Under these conditions, we observed inhibitory currents in 11/30 neurons following red-light excitable channelrhodopsin (ReaChR)-mediated activation of the NPY::Cre INs (mean amplitude: 57.3 ± 15.6 pA). These currents were abolished in 7/7 neurons following application of strychnine and picrotoxin (Figures 5D–5F), demonstrating the NPY::Cre INs form functional inhibitory synapses onto excitatory Y1+ neurons within the dorsal horn.
The intriguing finding that the Y1Cre neurons are contacted by processes containing putative NPY+ dense core vesicles suggested to us that NPY peptidergic signaling via the Y1Cre neurons might also contribute to the gating of the mechanical itch pathway. To assess the role of Gi-mediated inhibitory Y1 signaling within the dorsal horn, we crossed Lbx1Cre (Sieber et al., 2007) with Y1f/f mice (Bertocchi et al., 2011) to remove Y1 from dorsal horn neurons, sparing ventral, peripheral, and supraspinal neurons. Lbx1Cre; Y1f/f mice exhibited spontaneous scratching at P42, as well as elevated sensitivity to nape stimulation (Figures 6A and 6B). Similarly, both spontaneous and evoked scratching were elevated by the Y1 antagonist BIBP 3226 (Wang et al., 2016) acting at dorsal horn Y1 receptors (Figures 6C–6D and S7A–S7B). Likewise, elevated spontaneous scratching was observed following administration of another Y1 antagonist, BMS 193885 (Antal-Zimanyi et al., 2008), and in NPY knockout mice (Figure S7C) (Karl et al., 2008). Conditional knockout of Y1 enhanced hindpaw sensitivity to light punctate touch but did not modulate responses to dynamic touch (Figures 6E and 6F) or the frequency of scratching in response to histaminergic and non-histaminergic chemical itch stimuli (Figure 6G). The duration and rate (duration/frequency) of scratching induced by compound 48/80 were also unchanged (Figures 6H and 6I; Gao et al., 2018). Likewise, responses to mechanical, chemical, and thermal pain stimuli were not affected (Figures 6J–6N). These data indicate that NPY-Y1 signaling in the dorsal horn has a selective role in the modulation of light punctate touch information from both the hairy and glabrous skin and, as a consequence, functions to gate mechanical itch.
Figure 6. NPY-Y1 Receptor Signaling within the Dorsal Horn Gates Mechanical Itch and Light Punctate Touch.
(A and B) Mice lacking the Y1 receptor in dorsal horn neurons exhibit pronounced spontaneous scratching (A; Lbx1Cre; Y1f/f, n = 10; Y1f/f control, n = 12) and hypersensitivity to light punctate mechanical stimulation of the nape (B; Lbx1Cre; Y1f/f,n = 7; Y1f/f control, n = 9).
(C) The Y1 antagonist BIBP 3226 (1 mg kg−1, intraperitoneally [i.p.]) increases spontaneous scratching in control (n = 8) but not conditional knockout mice (n = 12). Two-tailed, paired t tests were used to assess statistical differences.
(D) BIBP 3226 causes hypersensitivity to nape stimulation (n = 12; vehicle, n = 12).
(E and F) Lbx1Cre; Y1f/f mice have reduced hindpaw von Frey thresholds (E; Lbx1Cre; Y1f/f,n= 8; Y1f/f control, n = 8) but responses to dynamic touch are unaltered (F; Lbx1Cre; Y1f/f,n= 8; Y1f/f control, n = 9).
(G–I) Deletion of Y1 from dorsal horn neurons does not alter scratching frequency in response to chloroquine (Lbx1Cre; Y1f/f, n = 11; Y1f/f control, n = 9) or scratching frequency (G), duration (H), or rate (duration/frequency; I) in response to compound 48/80 (Lbx1Cre; Y1f/f, n = 14; Y1f/f control, n = 10).
(J–N) Deletion of Y1 from dorsal horn neurons does not affect sensitivity to acute mechanical pain as assessed by pinprick (J; Lbx1Cre; Y1f/f,n = 9; Y1f/f control, n = 9) or the Randall-Selitto test (K; Lbx1Cre; Y1f/f,n= 8; Y1f/f control, n = 6), chemical nociception (L; Lbx1Cre; Y1f/f,n= 8; Y1f/f control, n = 8), or thermal pain as assessed by the hot plate (M; Lbx1Cre; Y1f/f,n = 9; Y1f/f control, n = 9) or Hargreaves tests (N; Lbx1Cre; Y1f/f,n = 8; Y1f/f control, n = 6).
*p < 0.05 and ***p < 0.001. NS, not significant. Data: mean ± SEM.
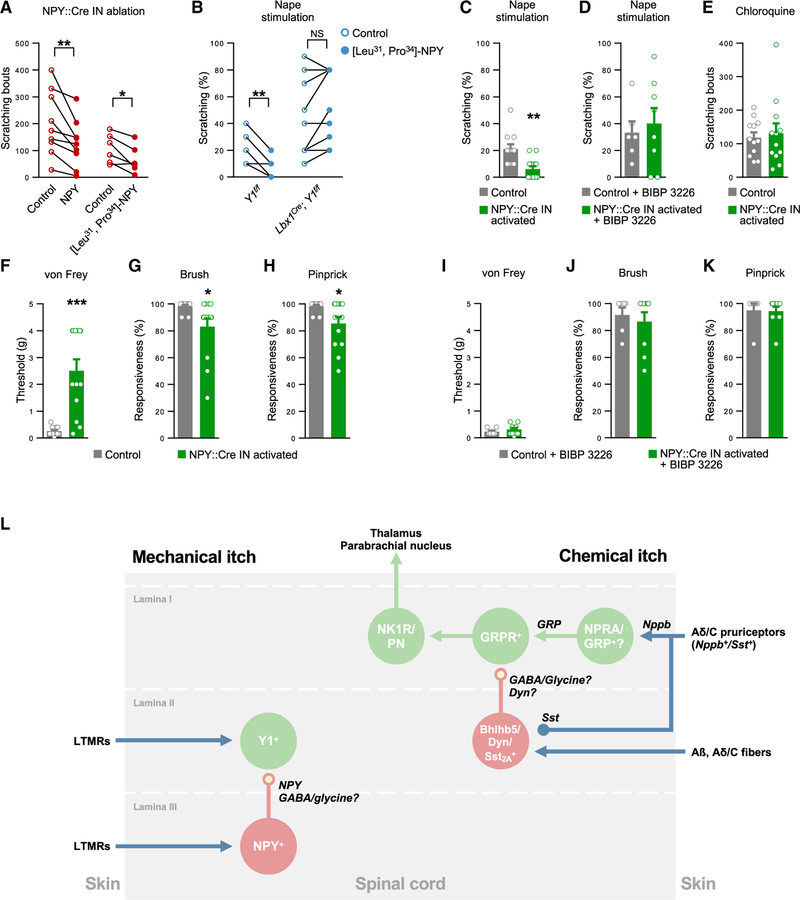
The role of Y1 signaling in the regulation of mechanical itch was further assessed by administering NPY or the selective Y1 agonist [Leu31, Pro34]-NPY (Gelfo et al., 2011) to NPY::Cre; Lbx1FlpO; Tauds-DTR; Ai65ds-tdTom mice 1 week after ablating the NPY::Cre INs. Both agonists relieved spontaneous scratching in the NPY:: Cre IN-ablated mice (Figures 7A and S7D). Furthermore, injection of NPY (Figure S7E) or [Leu31, Pro34]-NPY reduced sensitivity to nape stimulation, with this effect being abolished when [Leu31, Pro34]-NPY was administered to Lbx1Cre; Y1f/f mice, confirming the dorsal horn as the site of NPY action (Figures 7B and S7F).
Figure 7. NPY-Y1 Receptor Signaling Determines Sensitivity to Mechanical Itch Stimuli.
(A) Spontaneous scratching in NPY::Cre; Lbx1FlpO; Tauds-DTR; Ai65ds-tdTom mice 1 week after DT treatment was reduced following injection of NPY (100 μg kg−1, i.p.; n = 9) or the selective Y1 receptor agonist [Leu31, Pro34]-NPY (100 μg kg−1, i.p.; n = 6) when compared with vehicle.
(B) Evoked scratching is reduced when Y1f/f mice (n = 8) but not Lbx1Cre; Y1f/f mice (n = 9) are injected with [Leu31, Pro34]-NPY (100 μg kg−1, i.p., n = 8; vehicle, n = 8).
(C and D) hM3D-mediated activation of the NPY::Cre INs reduces scratching in response to mechanical stimulation of the nape compared with controls (NPY::Cre; Lbx1FlpO; R26ds-hM3D, n = 13; NPY::Cre; R26ds-hM3D controls, n = 11; C) but not when Y1 receptors are inhibited by BIBP 3226 (n = 7; controls, n = 9; D).
(E) Unchanged chloroquine-induced scratching following activation of the NPY::Cre INs (n = 12; controls, n = 12).
(F–H) Activation of NPY::Cre INs reduces sensitivity to von Frey hair (F), brush (G), and pinprick (H) stimulation of the plantar hindpaw (n = 13; controls, n = 11).
(I–K) Activation of NPY::Cre INs does not alter sensitivity to von Frey hair (I), brush (J), and pinprick (K) stimulation following Y1 receptor blockade by BIBP 3226 (n = 9; BIBP 3226-injected controls, n = 7).
(L) Schematic showing proposed dorsal horn circuitry for mechanical and chemical itch. Dyn, dynorphin; Nppb, natriuretic peptide B; NPRA, natriuretic peptide receptor A; PN, projection neuron; Sst2A, Sst 2A receptor. Italics indicate neurotransmitters/neuropeptides.
Two-tailed, paired (A and B) or unpaired (C–K) t tests were used to assess statistical differences. *p < 0.05, **p < 0.01, ***p < 0.001. NS, not significant. Data: mean ± SEM (B–L). See also Figure S7.
As a final test of the functional interaction between the NPY:: Cre and Y1+ neurons, we assessed itch sensitivity following chemogenetic activation of NPY::Cre INs. When NPY::Cre INs were selectively activated, evoked scratching was reduced compared to controls (Figure 7C). This reduction in scratching was negated by co-injecting BIBP 3226 with CNO, indicating that the inhibition of itch by NPY:: Cre INs is in large part dependent on the actions of NPY on the Y1+ neurons (Figure 7D). Importantly, the inhibition of itch by NPY::Cre IN activation was specific to mechanical itch, as chloroquine-induced scratching was unaffected (Figure 7E).
The activation of NPY::Cre INs also caused a pronounced reduction in the sensitivity of the glabrous skin to light punctate touch, as well as more moderate reductions in responses to dynamic touch and acute pain (Figures 7F–7H). These effects were not observed when BIBP 3226 was administered (Figures 7I–7K), implying that they were mediated by enhanced release of NPY. Given that dorsal horn Y1+ neurons do not transmit dynamic touch and acute pain, it is likely that the modulation of these modalities following NPY::Cre IN activation results from ectopic activation of Y1 receptors expressed by sensory neurons. Taken together, these data support a model in which light punctate touch and mechanical itch are strongly regulated by the actions of NPY on inhibitory Y1-bearing neurons in the dorsal horn.
DISCUSSION
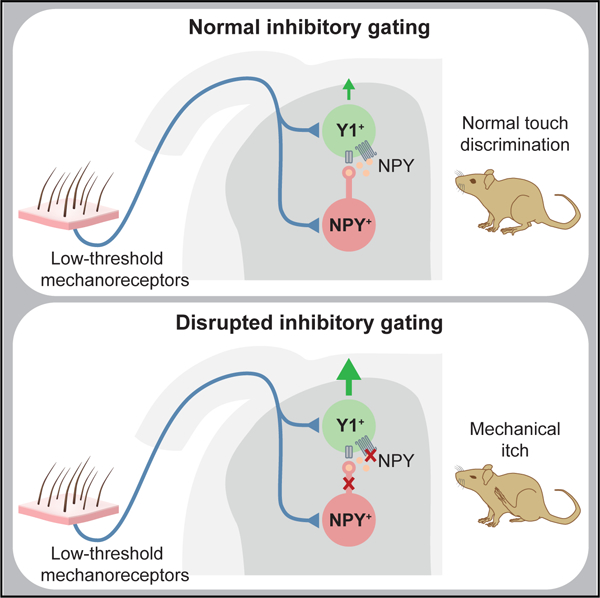
In this study we have identified the population of excitatory neurons in the dorsal spinal cord that are essential for transmitting the mechanosensory stimuli that drive mechanical itch. These neurons are marked by Y1 receptor expression and they define, together with NPY::Cre inhibitory INs in the dorsal horn, a central pathway for mechanical itch transmission that is distinct from the well-described GRP-GRPR chemical itch pathway (Figure 7L). Scratching in response to mechanical stimulation is abolished when the Y1Cre neurons are ablated, while activation of the Y1Cre neurons increases scratching in response to mechanical stimulation. Our results also reveal a prominent role for NPY peptidergic signaling in regulating light punctate touch sensitivity and mechanical itch, which is mediated by Y1+ neurons in the dorsal horn. We propose that NPY inhibitory signaling normally facilitates the discrimination of light touch stimuli, and disrupting it promotes a chronic itch phenotype and mechanical hyperknesis/alloknesis.
Dorsal Horn Y1Cre Neurons Transmit Low-Threshold Mechanical Stimuli
The light punctate touch and mechanical itch phenotypes we observe upon manipulation of the Y1Cre neurons are consistent with their innervation by mechanosensory LTMR afferents from both the glabrous and hairy skin (Figure 2). Whereas the Y1Cre neurons encompass multiple cell types within the LTMR-RZ (Figures S2 and S3), our findings suggest that only a subset of these cells are required for mechanical itch responses, and this subset appears to be distinct from those Y1Cre neurons that express Sst or NK1R, as ablating the Sst+ or NK1R+ neurons fails to disrupt the mechanical itch pathway (Figures 3 and S5).
Touch information appears to be broadly distributed between overlapping molecularly defined populations within the LTMRRZ that include Y1+, RORa+, Sst+, vGlut3+, and calretinin+ neurons (this study; Abraira et al., 2017; Abraira and Ginty, 2013; Bourane et al., 2015b; Duan et al., 2014; Peirs et al., 2015). Interestingly, the role of the Y1+ neurons in processing touch information appears to be limited to punctuate touch, since they are dispensable for transmitting dynamic light touch information (Figure 3), which is instead largely transmitted by excitatory RORα+ INs in laminae IIi/III (Bourane et al., 2015b). The contribution that dynamic touch and the RORa+ INs make to mechanical itch is still unclear.
The possibility that the Y1Cre neurons may contribute to the transmission of other cutaneous modalities is suggested by our observation that some Y1Cre neurons reside in laminae I–IIo, subsets of which are Sst+ neurons, which have been implicated in acute mechanical pain and lamina I NK1R+ neurons, which transmit chemical pain (Brumovsky et al., 2002; Duan et al., 2014; Mantyh et al., 1997; Nichols et al., 1999). Somewhat surprisingly, our functional analyses did not reveal a role for the Y1Cre neurons in acute mechanical pain or responses to noxious heat or chemical stimuli (Figure 4). Moreover, the absence of a pain phenotype following Y1Cre neuron ablation or activation suggests the Y1Cre/SstCre and Y1Cre/NK1R+ neurons are dispensable for pain transmission.
Chemical itch transmission was also unaffected following ablation of the Y1Cre neurons (Figure 3), with this study and our previous functional analysis of the NPY::Cre INs (Bourane et al., 2015a) providing strong evidence that the pathways for mechanical and chemical itch are largely segregated in the periphery and dorsal spinal cord (Figures 3 and 7L). Although the Y1Cre population partially overlaps with NK1R+, Sst+, and GRP+ neurons that have been implicated in chemical itch transmission (Albisetti et al., 2019; Christensen et al., 2016; Fatima et al., 2019; Huang et al., 2018; Kardon et al., 2014; Mishra and Hoon, 2013), those embedded within the Y1Cre population appear to be dispensable for chemical itch.
Mechanical and chemical itch stimuli produce indistinguishable sensations in humans, and both trigger scratching (Fukuoka et al., 2013). This suggests central convergence of the mechanical and chemical itch pathways, a possibility supported by observations that chemical pruritogens can cause alloknesis, a condition in which normally innocuous mechanical stimuli cause itch (Akiyama et al., 2012; Gao et al., 2018; Wahlgren et al., 1991). Moreover, there is evidence that the parabrachial nucleus (Campos et al., 2018; Mu et al., 2017) to which NK1R+ neurons project (Akiyama et al., 2015) encodes both mechanical and chemical itch. Our results show that the NK1R+ neurons are dispensable for mechanical itch, which suggests that the chemical and mechanical itch pathways do not converge in the spinal cord on ascending NK1R+ projection neurons.
NPY-Y1R Signaling Gates Mechanical Itch
Our analysis reveals a key role for NPY in regulating mechanical itch and touch sensitivity. We propose that NPY-Y1 signaling functions in healthy animals as a homeostatic mechanism to adjust the output of the LTMR circuitry, thus ensuring the correct discrimination between itch and other innocuous light touch sensations. Recently, it has been reported that NPY suppresses itch (Gao et al., 2018), although the site of action and the cellular basis for this activity were not described. Our studies show endogenous NPY acts on Y1+ neurons in the dorsal horn to regulate mechanical itch, the same cells that are required for the chronic itch phenotype in NPY::Cre IN ablated mice (Figure 3). We cannot completely exclude the possibility that NPY-Y1 signaling in sensory afferents also contributes to the modulation of mechanosensory transmission; however, this is unlikely, as in these neurons Y1 is co-expressed with CGRP (Brumovsky et al., 2002), a marker of high-threshold mechanoreceptors (Lawson et al., 2002).
While our results show the inhibitory effects of NPY::Cre IN activation are largely abolished by Y1 blockade, the present study does not eliminate a role for GABA or glycine in the regulation of the mechanical itch pathway. Rather, Y1-mediated inhibition may operate in conjunction with fast inhibitory pathways (Akiyama et al., 2011; Ralvenius et al., 2018) to regulate the sensitivity of the dorsal horn mechanosensory circuitry over longer time scales than those controlled by ionotropic feedforward inhibition. It is also possible that peptidergic signaling between NPY::Cre and Y1+ neurons contributes to the inhibition of mechanical itch by counterstimuli, a role that would be consistent with our observation that after-discharge spiking is enhanced following brushing of the hairy skin in NPY::-Cre IN-ablated mice (Bourane et al., 2015a).
The central inhibition of mechanical itch by NPY::Cre INs parallels the inhibitory regulation of chemical itch by the B5-I INs (Figure 7L) (Chiang et al., 2016; Kardon et al., 2014; Ross et al., 2010). B5-I INs are a subset of lamina I-II inhibitory INs that express the transcription factor Bhlhb5. These cells suppress activity in a tonic manner (Huang et al., 2018; Kardon et al., 2014; Ross et al., 2010) and in response to counterstimuli (Hachisuka et al., 2016; Kardon et al., 2014). The B5-I INs include the spinal INs that express the neuropeptide dynorphin, the endogenous agonist of kappa opioid receptors, and as in mechanical itch, a neuropeptidergic mechanism is proposed to regulate the chemical itch pathway in a tonic manner (Huang et al., 2018; Kardon et al., 2014; however, also see Duan et al., 2014).
Several studies have found that NPY exerts analgesic effects via two of the five identified mammalian NPY receptors, the Y1 and Y2 receptors, in neuropathic, inflammatory, and some forms of acute pain (Diaz-delCastillo et al., 2018; Duggan et al., 1991; Hua et al., 1991; Intondi et al., 2008; Naveilhan et al., 2001; Solway et al., 2011; Taiwo and Taylor 2002). Y1 signaling is also required for normal sensitivity to heat (Naveilhan et al., 2001). However, it was previously unclear whether these effects are mediated by central or peripheral mechanisms (Diaz-delCastillo et al., 2018; Gibbs et al., 2004; Moran et al., 2004). We find that ablation of Y1Cre neurons or deletion of the Y1 gene within the dorsal horn has no effect on acute, thermal, or chemical nociception (Figure 4), strongly indicating that they are instead mediated by peptidergic Y1+ nociceptive afferents (Brumovsky et al., 2002), rather than Y1+ neurons in the dorsal horn. This finding further suggests that the mild Y1-dependent inhibition of acute pain and dynamic touch observed following activation of the NPY::Cre INs (Figure 7) entails ectopic inhibition of Y1+ primary afferents, and that other inhibitory neurons play a more prominent role in gating these modalities under physiological conditions.
In summary, this study demonstrates that Y1+ neurons form an excitatory pathway within the dorsal horn for the transmission of mechanical itch, and that the flow of information through this pathway is regulated by inhibitory NPY-Y1 signaling to maintain normal touch discrimination. We propose that dysregulation of this pathway following failure of the gating mechanism may drive the development of neuropathic chronic itch. It has been suggested that increases in mechanical itch sensitivity in mice following the loss of Merkel cells due to aging or dry skin might be due to reduced cutaneous input to inhibitory NPY-expressing INs, although this has not been tested (Feng et al., 2018). There is also evidence that the disruption of NPY signaling in humans may contribute to chronic itch (Reich et al., 2013). Notably, psoriasis patients presenting with pruritus have reduced NPY serum levels, which are negatively correlated with the intensity of itch (Reich et al., 2007). In view of the central contribution NPY signaling makes to the inhibitory regulation of itch sensation, Y1 signaling merits investigation as a therapeutic target for the treatment of chronic itch, a condition that rivals chronic pain in the severity of its impact on quality of life (Kini et al., 2011).
STAR★METHODS
LEAD CONTACT AND MATERIALS AVAILABILITY
Further information and requests for resources and reagents should be directed to and will be fulfilled by the Lead Contact, Martyn Goulding (goulding@salk.edu).
EXPERIMENTAL MODEL AND SUBJECT DETAILS
All protocols for animal experiments were approved by the IACUC of the Salk Institute for Biological Studies according to NIH guidelines for animal experimentation. Male and female mice were used in all studies. Animals were randomized to experimental groups and no sex differences were noted.
The Y1Cre knockin mouse line was generated by Padilla et al. (Padilla et al., 2016). An FRT-flanked neomycin cassette was removed by crossing with the FLPo-10 deleter strain (Wu et al., 2009), as described by Padilla et al. (Padilla et al., 2016). The Y1::eGFP transgenic reporter mouse line (RRID:MMRRC_010554-UCD) was generated by the Gene Expression Nervous System Atlas (GENSAT) project. The following mouse lines were also used in this study: NPY::Cre (Bourane et al., 2015a), SstCre (Taniguchi et al., 2011), SstFlpO (He et al., 2016), Lbx1Cre (Sieber et al., 2007), Lbx1FlpO (Bourane et al., 2015a), Ai14lsl-Tom (Madisen et al., 2010), Ai65ds-Tom (Madisen et al., 2015), R26ds-hM3D (Sciolino et al., 2016), R26ds-hM4D (Bourane et al., 2015a), R26ds-ReaChR (Hooks et al., 2015), Tauds-DTR (Britz et al., 2015), NPY KO (Karl et al., 2008), Y1f/f (Bertocchi et al., 2011), GRP::eGFP (Mishra and Hoon, 2013), R26ds-HTB (Bourane et al., 2015b), R26lsl-TVA (Seidler et al., 2008).
METHOD DETAILS
Immunohistochemistry
Mice were euthanized by a single intraperitoneal (i.p.) injection (10 μl g−1 body weight) of ketamine (10 mg ml−1) and xylazine (1 mg ml−1) immediately prior to perfusion with 20 mL ice-cold 4% paraformaldehyde in PBS. Spinal cords and DRG (lumbar levels L4 and L5) were dissected and post-fixed for 1 h at RT, then washed 3 times in PBS and cryoprotected in 30% sucrose-PBS (w/v) overnight at 4°C. Tissue was embedded in Tissue-Tek OCT Compound (Sakura Finetek) and cryosectioned at 40 μm. Sections were dried at RT and stored at —20°C. Sections were washed once with PBS (5 min), blocked with a solution of 10% donkey serum in PBT (PBS, 0.1% Triton X-100) for 1 h at RT and then incubated overnight at 4°C with primary antibodies in a solution of 1% donkey serum in PBT. Sections were then washed 3 times (15 min each) in PBT before being incubated for 2 h at RT with fluorophore–conjugated secondary antibodies (1:1000; Jackson Laboratories) in a solution of 1% donkey serum in PBT. Sections were again washed 3 times (15 min each) in PBT before being mounted with Aqua-Poly/Mount (Polysciences). A Zeiss LSM 700 confocal microscope was used to capture images. 3–5 spinal cords were analyzed for each condition. ImageJ software was used to assess immunofluorescence, with thresholds set according to signal intensity (Jensen, 2013).
The following primary antibodies were used in this study: rabbit α-Calbindin (1:1,000; Swant), rabbit α-Calretinin (1:1,000; Swant), sheep α-CGRP (1:1,000; Abcam), rabbit α-cMaf (1:5000; C Birchmeier, MDC, Berlin), goat α-cRet (1:250; R&D Systems), goat α-CTB (1:4000; List Laboratories), rabbit α-DsRed (1:1000; Clontech), mouse α-Gephyrin (1:8000; Synaptic systems); rabbit α-GFAP (1:500; Dako), chicken α-GFP (1:000; Aves), goat α-GFP (1:1000; Abcam), rabbit α-GRPR (1:1000; Abcam), rabbit α-GRPR (1:100; MBL), guinea pig α-Lmx1b (1:1000; M Goulding), mouse α-NeuN (1:1000; Millipore), rabbit α-NK1R (1:500; Advanced Targeting Systems), rabbit α-NPY (1:1000; Peninsula Lab), rabbit α-Parvalbumin (1:1000; Swant), rabbit α-Pax2 (1:200; Zymed), rat α-RFP (1:1000; Chromotek), rabbit α-PKCg (1:1000; Santa Cruz), goat α-RORa (1:100; Santa Cruz), rabbit α-S100b (1:500; Dako), goat TrkB (1:1000; R&D Systems), goat α-TrkC (1:1000; R&D Systems), guinea pig α-VGAT (1:1000; Synaptic Systems), guinea pig α-vGluT1 (1:1000; Millipore). In addition, Alexa Fluor 647-conjugated isolectin GS-IB4 from Griffonia simplicifolia (Invitrogen) was used at 1:500.
In Situ Hybridization
For in situ hybridization (ISH), mice were perfused with 4% paraformaldehyde in a solution of 0.1% diethyl pyrocarbonate in PBS (PBS-DEPC), post fixed for 1 h at RT, washed 3 times with PBS-DEPC and cryopreserved overnight in 30% sucrose in PBS-DEPC. Spinal cords were embedded in Tissue-Tek and stored at —80°C. Spinal cords were then cryosectioned at 16 μm, and sections were hybridized with an antisense RNA probe overnight at 64°C. Sections were washed twice in a solution of 1 × saline-sodium citrate buffer, 50% formamide, and 0.1% Tween-20 at 64°C for 30 min and blocked with a solution of 0.1% Tween 20 in maleic acid buffer (MABT) containing 2% blocking reagent and 10% inactivated sheep serum for 2 h at RT. Sections were then incubated overnight with sheep α-digoxigenin-alkaline-phosphatase Fab fragments (1:2000; Roche), washed twice in MABT and revealed with a staining solution of NBT (1:500, Roche) and BCIP (1:600, Roche). An Olympus VS-120 Virtual Slde Scanning Microscope was used for imaging. For double staining analyses, tdTomato fluorescence was imaged before ISH was performed. ISH images were later pseudo-colored and superposed onto the tdTomato signal in Photoshop (Adobe Systems). For quantification, three sections from each of three spinal cords were analyzed per condition, and only cells with clearly visible nuclei were scored.
Rabies Virus Tracing and Morphological Analyses
For the sparse labeling of Y1Cre neurons required for morphological reconstruction, EnvA-pseudotyped, ∆G-dsRed-Express rabies virus (100 μl, 1.28 × 108 units ml−1) was injected unilaterally into the lumbar spinal cord of P10 Y1Cre; R26lsl-TVA mice (Bourane et al., 2015b). For the transsynaptic tracing studies, bilateral injections of EnvA-pseudotyped, DG-mCherry (250 μl, 3.3 × 1010 units ml−1) were made into the lumbar cord of Y1Cre; Lbx1FlpO; R26ds-HTB animals at P5.
Briefly, mice were anesthetized by administering 2.5% isoflurane via a nose cone. The skin over the lumbar region of the dorsal spinal cord was incised and a laminectomy was performed at the T13-L1 level. After removal of the dura mater with a fine needle to expose the spinal cord, virus was injected via a fine glass capillary inserted into the dorsal spinal cord. The needle was left in the cord for 1 min after injection to prevent outflow. The skin was closed using Vetbond (3M) and Reflex Skin Closure System (CellPoint Scientific). Mice were perfused 5 days post-injection and processed for immunohistochemistry.
AAV Virus Tracing of Synaptic Connections
To visualize synaptophysin at presynaptic boutons, injections of AAV2/1-hSyn-DIO-SypHTomato (0.5 μl, 1.6 × 1012 units ml−1) were made into the lumbar spinal cord of P37 Y1::eGFP mice as described above. Spinal cords were then processed for immunohisto-chemistry at P60.
Retrograde Cholera Toxin-b Labeling of Cutaneous Sensory Neurons
Postnatal day (P) 39 Y1Cre; Ai14lsl-Tom mice were anesthetized with 2.5% isoflurane in O2, and Alexa Fluor 647-conjugated cholera toxin subunit B (CTb) (0.5 μl, 2.5 μg μl−1 in 0.9% sterile saline; Molecular Probes) was injected into either the hairy skin of the hindlimb or the glabrous skin of the plantar hindpaw with a fine glass capillary. Mice were perfused at 3 days post injection and processed for immunohistochemistry.
Cell Ablation
For ablation of neurons expressing Cre and FlpO drivers in addition to Tauds-DTR, mice were injected with diphtheria toxin (DT; 50 ng kg−1 in 0.9% sterile saline, i.p.; List Biological Laboratories) at P28 and P31 (Bourane et al., 2015a). Analysis of spontaneous scratching in mice expressing NPY::Cre (and relevant controls) was performed at P35, 7 days following the first DT injection. For the double-Cre ablation experiment, controls were positive for Cre and Tauds-DTR alleles but lacked the Lbx1FlpO allele and did not therefore express the diphtheria toxin receptor; however, all mice received DT. All other behavioral testing was performed 14–21 days following the first injection; controls were littermates of experimental animals and had identical genotypes but received injections of 0.9% sterile saline instead of DT.
To ablate cells cell populations with saporin-conjugated receptor ligands, P28 mice were given a single intrathecal (i.t.) injection of either bombesin-saporin (400 ng in 5 μL 0.9% sterile saline; Advanced Targeting Systems) (Bourane et al., 2015a) to ablate GRPR+ cells or [Sar9, Met(O2)11]-substance P-saporin neurons (100 ng in 5 mL 0.9% sterile saline; Advanced Targeting Systems) to ablate NK1R+ neurons (Wiley and Lappi, 1999). Littermate controls received blank saporin (equal mass in 5 μL 0.9% sterile saline; Advanced Targeting Systems). Behavioral testing and assessment of ablation efficiency by immunohistochemistry were performed 14 days later at P42.
For i.t. injections, mice were anesthetized with 2.5% isoflurane in O2, delivered via a nose cone. The caudal paralumbar region was then secured between the thumb and index finger, and a 30-gauge needle was inserted into the fifth intervertebral space until it elicited a tail flick. To prevent outflow, the needle was held in place for 10 s and turned 90° prior to withdrawal.
Drug Administration
Synthesis of the selective Y1 receptor agonist [Leu31, Pro34]-Neuropeptide Y ([Leu31, Pro34]-NPY; YPSKPDNPGEDAPAEDMARYYS ALRHYINLLTRPRY-NH2) (Fuhlendorff et al., 1990) was performed on a Gyros Protein Technologies, Inc. Tribute peptide synthesizer equipped with real-time UV monitoring, using standard Fmoc chemistry, in the Salk’s Peptide Synthesis Core. The resultant crude peptide was purified by the Salk’s Proteomics Core to > 98% using HPLC. The final, purified product gave a single peak of predicted mass (4240.7 Da) by MS analysis. The peptide was administered in solution at pH 7.
Neuropeptide Y (NPY; Tocris) and [Leu31, Pro34]-NPY were dissolved in 0.9% sterile saline. Clozapine N-oxide (CNO; Sigma) and the Y1 receptor antagonist BIBP 3226 (Tocris) (Jacques et al., 1995) were dissolved in DMSO, which was then diluted with 0.9% sterile saline such that the concentration of DMSO did not exceed 1% in injected solutions. The Y1 receptor antagonist BMS 193885 (Tocris) (Poindexter et al., 2002) was dissolved in sterile water, which was then rendered isotonic with glucose (5% w/v).
CNO was administered by i.p. injection at 2 mg kg-1. Other drugs were administered as indicated.
CNO, NPY, [Leu31, Pro34]-NPY, and BIBP 3226 were injected 15 min prior to behavioral testing. BMS 193885 was observed to cause inactivity for ~30 min following i.p. injection, as previously reported (Antal-Zimanyi et al., 2008); behavioral testing in BMS 193885-injected mice and vehicle-injected controls was therefore conducted from 30 to 60 min post injection. WT mice injected with NPY, [Leu31, Pro34]-NPY, BIBP 3226 or BMS 193885 were compared to vehicle-injected littermate controls.
To take into account differences in phenotype severity between NPY::Cre; Lbx1FlpO; Tauds-DTR mice treated with DT and between Lbx1Cre; Y1f/f mice, paired t tests were conducted to assess the effects of Y1 drugs. Recordings were made at 4 h intervals on the same day.
For chemogenetic silencing and activation experiments, all mice received CNO. Control mice were positive for the Cre and R26ds-hM4D or R26ds-hM3D alleles but lacked the Lbx1FlpO allele and did not therefore express the hM4D or hM3D receptor.
Electrophysiology
For slice preparations, P14–28 mice were anaesthetized by i.p. injection of urethane (10 ml/g) and transcardially perfused with oxygenated ice-cold dissecting/recovery artificial cerebrospinal fluid (ACSF; NaCl, 95 mM; KCl, 2.5 mM; NaHCO3, 26 mM; NaH2PO4H2O, 1.25 mM; MgCl2, 6 mM; CaCl2, 1.5mM; glucose, 20 mM; sucrose, 50 mM; Kynurenic Acid, 1 mM; ethyl pyruvate, 5 mM). The spinal cords were then isolated in ice-cold dissecting/recovery ACSF before being embedded in low-melting agarose at 33°C. A vibratome (Leica VT1000S) was used to cut 300 μm transverse slices from lumbar segments L1-5 in ice cold dissecting/recovery ACSF. Slices were then allowed to recover in dissecting/recovery ACSF at ~34°C for 1 h before being secured in a recording chamber continuously perfused with recording aCSF (NaCl, 125 mM; KCl, 2.5 mM; NaHCO3, 26 mM; NaH2PO4H2O, 1.25 mM; MgCl2, 1 mM; CaCl2, 2 mM; glucose, 20 mM; ethyl pyruvate, 5 mM) at RT. At all stages, ACSF was equilibrated with carbogen (95% O2; 5% CO2). Experiments were performed at RT.
Patch-clamp electrodes (3–5 MU) were pulled on a horizontal puller (Sutter Instrument, Novato, CA) from borosilicate glass (World Precision Instruments, Sarasota, FL). Signals were amplified and filtered (4 kHz low-pass Bessel filter) with a MultiClamp 700B amplifier (Molecular Devices) and acquired at 50 kHz with a Digidata 1440A A/D board and pCLAMP software (Molecular Devices). Neuronal firing was elicited by injecting depolarizing currents ranging from 0 to 200 pA in 20 pA increments for 1 s at intervals of 10 s. For ReaChR-mediated stimulation of NPY::Cre INs, a single LED optic fiber source (~2 mW output at 591 nm) was positioned ~10 mm from the surface of the slice, illuminating its entire surface. Stimulation was delivered at a pulse width of 5–20 ms. All drugs were bath applied. Kynurenic acid (1.5 μM; Sigma) and strychnine (1 mM; Sigma) were dissolved in water. Picrotoxin (60 μM; Sigma) was dissolved in DMSO such that the concentration of DMSO in recording solution did not exceed 0.1% (v/v). A liquid junction potential of 14 mV was corrected offline.
Behavioral Testing
Littermate controls were used for behavioral tests, and the experimenter was blinded to genotype/treatment. Animals were habituated to the behavioral testing apparatus for 1 h on each of the two days preceding data collection. Tests were conducted at P42-P49, or at P35 (7 days following the first injection of DT) in experiments to assess spontaneous scratching induced by ablation of NPY::Cre neurons.
Spontaneous Itch
To quantify scratching induced in the absence of an experimental mechanical stimulus (spontaneous itch), mice were placed in a plastic chamber and video recorded for a period of 30 min; bouts of hindlimb scratching were counted offline (Bourane et al., 2015a).
Nape Stimulation Assay
To quantify itch-related scratching behaviors induced by mechanical stimulation of the hairy skin, mice were placed in a plastic chamber and a 0.16 g von Frey hair was applied to the nape for 3 s (as per Bourane et al., 2015a). Hindlimb scratching responses over 10 trials were counted and reported as a percentage.
von Frey Assay
To assess the sensitivity of the glabrous skin to light punctate mechanical stimulation, mice were placed in a plastic chamber on an elevated wire grid and the lateral plantar surface of the hindpaw was stimulated with calibrated von Frey monofilaments (0.008–4 g). The paw withdrawal threshold for the von Frey assay was determined by Dixon’s up-down method (Chaplan et al., 1994).
Dynamic Touch Test
To assess the sensitivity of the glabrous skin to light dynamic touch, mice were placed in a plastic chamber on an elevated wire grid and the plantar surface of the hindpaw was stimulated by light stroking with a fine paintbrush in a heel-to-toe direction (Bourane et al., 2015b). The test was repeated 10 times at 10 s intervals between trails, and the percentage of positive paw withdrawal trials was calculated.
Pinprick Test
To assess the sensitivity of the glabrous skin to acute painful stimuli, mice were placed in a plastic chamber on an elevated wire grid and the plantar surface of the hindpaw was stimulated with an Austerlitz insect pin (Tip diameter: 0.02 mm; Fine Science Tools). The pin was gently applied to the plantar surface of the hindpaw without moving the paw or penetrating the skin. The pin stimulation was repeated 10 times on different paw areas with a 1–2 min interval between trails, and the percentage of trials in which mice responded with paw withdrawal was calculated.
Randall-Selitto Test
Prior to testing, mice were placed in a plastic restraining tube and allowed 5 min to acclimatize. A Randall-Selitto device (IITC, USA) was used to apply slowly increasing pressure to a point midway along the tail until the animal showed clear signs of discomfort. This pressure was recorded as the pain threshold. Three trials taken at 2 min intervals were performed to calculate the average threshold for each animal.
Chemical Nociception Tests
To assess pain induced by chemical agents, a 30-gauge needle was used to inject 6 mL of either capsaicin (1 mg in 10 mL 9% saline containing 7% Tween-80) or formalin (2% in 9% saline) subcutaneously into the plantar hindpaw. The time spent licking, flinching, and biting the injected hindpaw was recorded for 15 min (capsaicin) or 1 h (formalin) post-injection. Phase I was defined as the first 10 min post injection and phase II was defined as the period 10–60 min post injection.
Hot Plate Test
Mice were placed on a hot plate (IITC, USA) set at 46°C, 50°C or 54°C and the latencies to hindpaw flinching and licking were measured. All animals were tested sequentially with a minimum of 5 min between each test. To prevent tissue damage, a cutoff time was set at 60 s for assays at 46°C and 50°C, and 30 s for 54°C.
Hargreaves Test
To measure radiant heat pain, mice were placed in a plastic chamber and the plantar hindpaw surface was exposed to a beam of radiant heat (IITC, USA). The latency to paw withdrawal was determined in one trial per hindpaw and averaged per animal, with a 10 min interval between trials. A cutoff time of 30 s was set to prevent tissue damage.
Chemical Itch Test
The pruritogens chloroquine (200 mg; Sigma), histamine (100 μg; Sigma), compound 48/80 (100 μg; Sigma) and SLIGRL-NH2 (SLIGRL; 100 nM; Abcam) were dissolved in 0.9% sterile saline and injected intradermally behind the ear in a volume of 50 μl. The behavior of each animal was video-recorded over the following 30 min, and the number of hindpaw scratch bouts was counted.
QUANTIFICATION AND STATISTICAL ANALYSIS
Data were analyzed in GraphPad Prism 6 for Windows, Version 6.01 (Graphpad Software) or Excel 2016 (Microsoft) by two-tailed, unpaired t tests, unless otherwise indicated. See figure legends for details of statistical analyses. p < 0.05 was considered to be statistically significant. All data are presented as the mean ± standard error of the mean (SEM).
All tdTomato signals were enhanced by RFP antibody staining prior to analysis.
Supplementary Material
KEY RESOURCES TABLE
| REAGENT or RESOURCE | SOURCE | IDENTIFIER |
|---|---|---|
| Antibodies | ||
| Rabbit α-Calbindin (1:1000) | Swant | Cat# 300; RRID:AB_10000347 |
| Rabbit α-Calretinin (1:1000) | Swant | Cat# 7699/3H; RRID:AB_10000321 |
| Sheep α-CGRP (1:1000) | Abcam | AB22560; RRID:AB_725809 |
| Rabbit α-cMaf (1:5000) | C Birchmeier, MDC, Berlin | N/A |
| Goat α-cRet (1:250) | R&D Systems | Cat# AF482; RRID:AB_2301030 |
| Goat α-CTb (1:4000), | List Laboratories | Cat# 703; RRID:AB_10013220 |
| Sheep α-Digoxigenin-AP, Fab fragments (1:2000) | Roche | Cat# 11093274910; RRID:AB_514497 |
| Rabbit α-DsRed (1:1000) | Clontech | Cat# 632496; RRID: AB_10013483 |
| Mouse α-Gephyrin (1:8000) | Synaptic Systems | Cat# 147 011; RRID:AB_887717 |
| Rabbit α-GFAP (1:500) | Dako | Cat# Z0334; RRID:AB_10013382 |
| Chicken α-GFP (1:000) | Aves | Cat # GFP-1020; RRID:AB_10000240 |
| Goat α-GFP (1:1000) | Abcam | Cat# ab6673; RRID:AB_305643 |
| Rabbit α-GRPR (1:100) | MBL | Cat# LS-A831; RRID:AB_591750 |
| Rabbit α-GRPR (1:1000) | Abcam | Cat# ab39883; RRID:AB_880315 |
| Guinea pig α-Lmx1b (1:1000) | M Goulding | N/A |
| Rabbit α-MafA (1:5000) | C Birchmeier, MDC, Berlin | N/A |
| Mouse α-NeuN (1:1000) | Millipore | Cat# MAB377; RRID:AB_2298772 |
| Rabbit α-Neuropeptide Y1 Receptor (1:100) | Alomone Labs | Cat# ANR-021: RRID:AB_2040030 |
| Rabbit α-NF200 (1:1000) | Sigma | Cat# N4142; RRID:AB_477272 |
| Rabbit α-NK1R (1:500) | Advanced Targeting Systems | Cat# AB-N33ap; RRID:AB_458739 |
| Rabbit α-NPY (1:1000) | Peninsula Lab | Cat# T-4070.0050; RRID:AB_518504 |
| Rabbit α-Pax2 (1:200) | Zymed | Cat# 71–6000; RRID:AB_2533990 |
| Rabbit α-Parvalbumin (1:2000) | Swant | Cat# PV-25; RRID:AB_10000344 |
| Rabbit α-PKCγ (1:1000) | Santa Cruz | Cat# SC-211; RRID:AB_632234 |
| Rat α-RFP (1:1000) | Chromotek | Cat#5F8; RRID: AB_2336064 |
| Goat α-RORα (1:100) | Santa Cruz | Cat# sc-6062; RRID:AB_655755 |
| Rabbit α-S100β (1:500) | Dako | Cat# Z0311; RRID:AB_10013383 |
| Chicken α-TrkB (1:1000) | R&D Systems | Cat# AF1494; RRID:AB_2155264 |
| Goat α-TrkC (1:1000) | R&D Systems | Cat# AF1404; RRID:AB_2155412 |
| Guinea pig α-VGAT (1:1000) | Synaptic Systems | Cat# 131004; RRID:AB_887873 |
| Guinea pig α-vGluT1 (1:1000) | Millipore | Cat# AB5905; RRID: AB_2301751 |
| Bacterial and Virus Strains | ||
| EnvA-pseudotyped, ∆G-mCherry | Janelia Viral Core/HHMI | Wickersham et al., 2007 |
| EnvA-pseudotyped, ∆G-dsRed-Express | Janelia Viral Core/HHMI | Wickersham et al., 2007 |
| AAV2/1-hSyn-DIO-SypHTom | Koch et al., 2017 | N/A |
| Chemicals, Peptides, and Recombinant Proteins | ||
| Isolectin GS-IB4 From Griffonia simplicifolia, Alexa Fluor 647 Conjugate (1:500) | Invitrogen | Cat# I32450; RRID:SCR_014365 |
| Cholera Toxin Subunit b (Recombinant), Alexa Fluor 647 Conjugate | Invitrogen | Cat# C34778 |
| Diphtheria Toxin, Unnicked, from Corynebacterium diphtheria | List Laboratories | Cat# 150 |
| Clozapine N-oxide | Sigma | Cat# C0832 |
| Blank saporin | Advanced Targeting Systems | Cat# IT-21 |
| Bombesin-saporin | Advanced Targeting Systems | Cat# IT-40 |
| [Sar9, Met(O2)11]-substance P-saporin | Advanced Targeting Systems | Cat# IT-11 |
| Neuropeptide Y (human, rat) | Tocris | Cat# 1153 |
| [Leu31, Pro34]-NPY (100 µg kg−1) | M Goulding | N/A |
| BIBP 3226 trifluoroacetate | Tocris | Cat# 2707/1 |
| BMS 193885 | Tocris | Cat# 3242/10 |
| Chloroquine diphosphate | Sigma | Cat# C6628 |
| Histamine | Sigma | Cat# H7125 |
| Compound 48/40 | Sigma | Cat# C2313 |
| SLIGRL-NH2 | Abcam | Cat# ab120176 |
| Capsaicin | Sigma | Cat# M2028 |
| Formalin | Sigma | Cat# HT501128 |
| Kynurenic acid | Sigma | Cat# K3375 |
| Picrotoxin | Sigma | Cat# P1675 |
| Strychnine | Sigma | Cat# S0532 |
| NBT | Roche | Cat# 11383213001 |
| BCIP | Roche | Cat# 11383221001 |
| Experimental Models: Organisms/Strains | ||
| Mouse: Y1Cre | The Jackson Laboratory | JAX stock #030544; RRID:IMSR_JAX:030544 |
| Mouse: FLPo-10 | The Jackson Laboratory | JAX stock #011065; RRID:IMSR_JAX:011065 |
| Mouse: Y1::eGFP | Gene Expression Nervous System Atlas (GENSAT) | RRID:MMRRC_010554-UCD |
| Mouse: NPY::Cre | Bourane et al., 2015a | N/A |
| Mouse: SstCre | The Jackson Laboratory | JAX stock #013044; RRID:IMSR_JAX:013044 |
| Mouse: SstFlpO | The Jackson Laboratory | JAX stock #028579; RRID:IMSR_JAX:028579 |
| Mouse: Lbx1Cre | Sieber et al., 2007 | MGI: 104867 |
| Mouse: Lbx1FlpO | Bourane et al., 2015a | N/A |
| Mouse: Ai14lsl-Tom | The Jackson Laboratory | JAX stock #007908; RRID:IMSR_JAX:007908 |
| Mouse: Ai65ds-Tom | The Jackson Laboratory | JAX stock #021875; RRID:IMSR_JAX:021875 |
| Mouse: R26ds-hM3D | The Jackson Laboratory | JAX stock #026942; RRID:IMSR_JAX:026942 |
| Mouse: R26ds-hM4D | Bourane et al., 2015a | N/A |
| Mouse: R26ds-HTB | Bourane et al., 2015b | N/A |
| Mouse: R26ds-ReaChR | Hooks et al., 2015 | JAX stock #024846; RRID:IMSR_JAX:024846 |
| Mouse: R26lsl-TVA | Seidler et al., 2008 | MGI:3814188 |
| Mouse: NPY KO | Karl et al., 2008 | N/A |
| Mouse: Y1f/f | Bertocchi et al., 2011 | N/A |
| Mouse: GRP::eGFP | MMRRC | RRID:MMRRC_010444-UCD |
| Software and Algorithms | ||
| Adobe Illustrator and Photoshop CS5 | Adobe | https://www.adobe.com |
| Prism 5 | GraphPad | https://www.graphpad.com |
| Excel 365 | Microsoft | https://www.microsoft.com/en-us/ |
| ImageJ Cell Counter Plugin | Kurt de Vos, Univ. of Sheffield, UK | https://imagej.nih.gov/ij |
| pClamp10.4 and Clampfit | Molecular Devices | https://www.moleculardevices.com |
Highlights.
Excitatory NPYR1Cre (Y1Cre ) neurons are required for mechanical itch transmission
Spinal Y1 neurons receive LTMR input and mediate light punctate touch
NPY::Cre interneurons inhibit Y1-expressing neurons in the dorsal horn
NPY signaling via dorsal horn Y1-expressing neurons gates mechanical itch
ACKNOWLEDGMENTS
This study was supported by grants from the NIH (NS0850586, NS086372, and NS111643 to M.G.) and by the Caterina Foundation (D.A.). Synthesis of [Leu31, Pro34]-NPY was supported by the Peptide Synthesis and Proteomics Core Facilities of the Salk Institute with funding from NIH-NCI CCSG (P30 014195). Rabies viruses were generated and kindly provided by Kim Ritola, HHMI Janelia. M.G. holds the Frederick and Joanne Mitchell Chair at the Salk Institute. We thank Qiufu Ma for insightful comments on the manuscript.
Footnotes
SUPPLEMENTAL INFORMATION
Supplemental Information can be found online at https://doi.org/10.1016/j.celrep.2019.06.033.
DECLARATION OF INTERESTS
The authors declare no competing interests.
REFERENCES
- Abraira VE, and Ginty DD (2013). The sensory neurons of touch. Neuron 79, 618–639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Abraira VE, Kuehn ED, Chirila AM, Springel MW, Toliver AA, Zimmerman AL, Orefice LL, Boyle KA, Bai L, Song BJ, et al. (2017). The Cellular and Synaptic Architecture of the Mechanosensory Dorsal Horn. Cell 168, 295–310.e19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Akiyama T, Iodi Carstens M, and Carstens E (2011). Transmitters and pathways mediating inhibition of spinal itch-signaling neurons by scratching and other counterstimuli. PLoS ONE 6, e22665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Akiyama T, Carstens MI, Ikoma A, Cevikbas F, Steinhoff M, and Carstens E (2012). Mouse model of touch-evoked itch (alloknesis). J. Invest. Dermatol 132, 1886–1891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Akiyama T, Tominaga M, Davoodi A, Nagamine M, Blansit K, Horwitz A, Carstens MI, and Carstens E (2013). Roles for substance P and gastrin-releasing peptide as neurotransmitters released by primary afferent pruriceptors. J. Neurophysiol 109, 742–748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Akiyama T, Nguyen T, Curtis E, Nishida K, Devireddy J, Delahanty J, Carstens MI, and Carstens E (2015). A central role for spinal dorsal horn neurons that express neurokinin-1 receptors in chronic itch. Pain 156, 1240–1246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Albisetti GW, Ghanem A, Foster E, Conzelmann K-K, Zeilhofer HU, and Wildner H (2017). Identification of two classes of somatosensory neurons that display resistance to retrograde infection by rabies virus. J. Neurosci 37, 10358–10371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Albisetti GW, Pagani M, Platonova E, Hösli L, Johannssen HC, Fritschy J-M, Wildner H, and Zeilhofer HU (2019). Dorsal Horn Gastrin-Releasing Peptide Expressing Neurons Transmit Spinal Itch But Not Pain Signals. J. Neurosci 39, 2238–2250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Antal-Zimanyi I, Bruce MA, Leboulluec KL, Iben LG, Mattson GK, McGovern RT, Hogan JB, Leahy CL, Flowers SC, Stanley JA, et al. (2008). Pharmacological characterization and appetite suppressive properties of BMS-193885, a novel and selective neuropeptide Y(1) receptor antagonist. Eur. J. Pharmacol 590, 224–232. [DOI] [PubMed] [Google Scholar]
- Bautista DM, Wilson SR, and Hoon MA (2014). Why we scratch an itch: the molecules, cells and circuits of itch. Nat. Neurosci 17, 175–182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bertocchi I, Oberto A, Longo A, Mele P, Sabetta M, Bartolomucci A, Palanza P, Sprengel R, and Eva C (2011). Regulatory functions of limbic Y1 receptors in body weight and anxiety uncovered by conditional knockout and maternal care. Proc. Natl. Acad. Sci. USA 108, 19395–19400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bourane S, Duan B, Koch SC, Dalet A, Britz O, Garcia-Campmany L, Kim E, Cheng L, Ghosh A, Ma Q, and Goulding M (2015a). Gate control of mechanical itch by a subpopulation of spinal cord interneurons. Science 350, 550–554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bourane S, Grossmann KS, Britz O, Dalet A, Del Barrio MG, Stam FJ, Garcia-Campmany L, Koch S, and Goulding M (2015b). Identification of a spinal circuit for light touch and fine motor control. Cell 160, 503–515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Britz O, Zhang J, Grossmann KS, Dyck J, Kim JC, Dymecki S, Gosgnach S, and Goulding M (2015). A genetically defined asymmetry underlies the inhibitory control of flexor-extensor locomotor movements. eLife 4, e04718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brumovsky PR, Shi TJ, Matsuda H, Kopp J, Villar MJ, and Hökfelt T (2002). NPY Y1 receptors are present in axonal processes of DRG neurons. Exp. Neurol 174, 1–10. [DOI] [PubMed] [Google Scholar]
- Brumovsky P, Hofstetter C, Olson L, Ohning G, Villar M, and Hökfelt T (2006). The neuropeptide tyrosine Y1R is expressed in interneurons and projection neurons in the dorsal horn and area X of the rat spinal cord. Neuroscience 138, 1361–1376. [DOI] [PubMed] [Google Scholar]
- Campos CA, Bowen AJ, Roman CW, and Palmiter RD (2018). Encoding of danger by parabrachial CGRP neurons. Nature 555, 617–622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carstens EE, Carstens MI, Simons CT, and Jinks SL (2010). Dorsal horn neurons expressing NK-1 receptors mediate scratching in rats. Neuroreport 21, 303–308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chaplan SR, Bach FW, Pogrel JW, Chung JM, and Yaksh TL (1994). Quantitative assessment of tactile allodynia in the rat paw. J. Neurosci. Methods 53, 55–63. [DOI] [PubMed] [Google Scholar]
- Chiang MC, Hachisuka J, Todd AJ, and Ross SE (2016). Insight into B5-I spinal interneurons and their role in the inhibition of itch and pain. Pain 157, 544–545. [DOI] [PubMed] [Google Scholar]
- Christensen AJ, Iyer SM, François A, Vyas S, Ramakrishnan C, Vesuna S, Deisseroth K, Scherrer G, and Delp SL (2016). In Vivo Interrogation of Spinal Mechanosensory Circuits. Cell Rep 17, 1699–1710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davidson S, Zhang X, Yoon CH, Khasabov SG, Simone DA, and Giesler GJ Jr. (2007). The itch-producing agents histamine and cowhage activate separate populations of primate spinothalamic tract neurons. J. Neurosci 27, 10007–10014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davidson S, Zhang X, Khasabov SG, Moser HR, Honda CN, Simone DA, and Giesler GJ Jr. (2012). Pruriceptive spinothalamic tract neurons: physiological properties and projection targets in the primate. J. Neurophysiol 108, 1711–1723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Diaz-delCastillo M, Woldbye DPD, and Heegaard AM (2018). Neuropeptide Y and its Involvement in Chronic Pain. Neuroscience 387, 162–169. [DOI] [PubMed] [Google Scholar]
- Dong X, and Dong X (2018). Peripheral and Central Mechanisms of Itch. Neuron 98, 482–494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duan B, Cheng L, Bourane S, Britz O, Padilla C, Garcia-Campmany L, Krashes M, Knowlton W, Velasquez T, Ren X, et al. (2014). Identification of spinal circuits transmitting and gating mechanical pain. Cell 159, 1417–1432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duggan AW, Hope PJ, and Lang CW (1991). Microinjection of neuropeptide Y into the superficial dorsal horn reduces stimulus-evoked release of immunoreactive substance P in the anaesthetized cat. Neuroscience 44, 733–740. [DOI] [PubMed] [Google Scholar]
- Fatima M, Ren X, Pan H, Slade HFE, Asmar AJ, Xiong CM, Shi A, Xiong AE, Wang L, and Duan B (2019). Spinal somatostatin-positive interneurons transmit chemical itch. Pain 160, 1166–1174. [DOI] [PubMed] [Google Scholar]
- Feng J, Luo J, Yang P, Du J, Kim BS, and Hu H (2018). Piezo2 channel-Merkel cell signaling modulates the conversion of touch to itch. Science 360, 530–533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fuhlendorff J, Gether U, Aakerlund L, Langeland-Johansen N, Thøgersen H, Melberg SG, Olsen UB, Thastrup O, and Schwartz TW (1990). [Leu31, Pro34]neuropeptide Y: a specific Y1 receptor agonist. Proc. Natl. Acad. Sci. USA 87, 182–186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fukuoka M, Miyachi Y, and Ikoma A (2013). Mechanically evoked itch in humans. Pain 154, 897–904. [DOI] [PubMed] [Google Scholar]
- Gao T, Ma H, Xu B, Bergman J, Larhammar D, and Lagerström MC (2018). The Neuropeptide Y System Regulates Both Mechanical and Histaminergic Itch. J. Invest. Dermatol 138, 2405–2411. [DOI] [PubMed] [Google Scholar]
- Gelfo F, De Bartolo P, Tirassa P, Croce N, Caltagirone C, Petrosini L, and Angelucci F (2011). Intraperitoneal injection of neuropeptide Y (NPY) alters neurotrophin rat hypothalamic levels: Implications for NPY potential role in stress-related disorders. Peptides 32, 1320–1323. [DOI] [PubMed] [Google Scholar]
- Gibbs J, Flores CM, and Hargreaves KM (2004). Neuropeptide Y inhibits capsaicin-sensitive nociceptors via a Y1-receptor-mediated mechanism. Neuroscience 125, 703–709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grudt TJ, and Perl ER (2002). Correlations between neuronal morphology and electrophysiological features in the rodent superficial dorsal horn. J. Physiol 540, 189–207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hachisuka J, Baumbauer KM, Omori Y, Snyder LM, Koerber HR, and Ross SE (2016). Semi-intact ex vivo approach to investigate spinal somatosensory circuits. eLife 5, e22866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hachisuka J, Chiang MC, and Ross SE (2018). Itch and neuropathic itch. Pain 159, 603–609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Han L, and Dong X (2014). Itch mechanisms and circuits. Annu. Rev. Biophys 43, 331–355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Häring M, Zeisel A, Hochgerner H, Rinwa P, Jakobsson JET, Lönnerberg P, La Manno G, Sharma N, Borgius L, Kiehn O, et al. (2018). Neuronal atlas of the dorsal horn defines its architecture and links sensory input to transcriptional cell types. Nat. Neurosci 21, 869–880. [DOI] [PubMed] [Google Scholar]
- He M, Tucciarone J, Lee S, Nigro MJ, Kim Y, Levine JM, Kelly SM, Krugikov I, Wu P, Chen Y, et al. (2016). Strategies and Tools for Combinatorial Targeting of GABAergic Neurons in Mouse Cerebral Cortex. Neuron 91, 1228–1243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hooks BM, Lin JY, Guo C, and Svoboda K (2015). Dual-channel circuit mapping reveals sensorimotor convergence in the primary motor cortex. J. Neurosci 35, 4418–4426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hua XY, Boublik JH, Spicer MA, Rivier JE, Brown MR, and Yaksh TL (1991). The antinociceptive effects of spinally administered neuropeptide Y in the rat: systematic studies on structure-activity relationship. J. Pharmacol. Exp. Ther 258, 243–248. [PubMed] [Google Scholar]
- Huang J, Polgár E, Solinski HJ, Mishra SK, Tseng P-Y, Iwagaki N, Boyle KA, Dickie AC, Kriegbaum MC, Wildner H, et al. (2018). Circuit dissection of the role of somatostatin in itch and pain. Nat. Neurosci 21, 707–716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ikoma A (2013). Updated neurophysiology of itch. Biol. Pharm. Bull 36, 1235–1240. [DOI] [PubMed] [Google Scholar]
- Ikoma A, Steinhoff M, Ständer S, Yosipovitch G, and Schmelz M (2006). The neurobiology of itch. Nat. Rev. Neurosci 7, 535–547. [DOI] [PubMed] [Google Scholar]
- Intondi AB, Dahlgren MN, Eilers MA, and Taylor BK (2008). Intrathecal neuropeptide Y reduces behavioral and molecular markers of inflammatory or neuropathic pain. Pain 137, 352–365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jacques D, Cadieux A, Dumont Y, and Quirion R (1995). Apparent affinity and potency of BIBP3226, a non-peptide neuropeptide Y receptor antagonist, on purported neuropeptide Y Y1, Y2 and Y3 receptors. Eur. J. Pharmacol 278, R3–R5. [DOI] [PubMed] [Google Scholar]
- Jensen EC (2013). Quantitative analysis of histological staining and fluorescence using Image. J. Anat. Rec. (Hoboken) 296, 378–381. [DOI] [PubMed] [Google Scholar]
- Johanek LM, Meyer RA, Friedman RM, Greenquist KW, Shim B, Borzan J, Hartke T, LaMotte RH, and Ringkamp M (2008). A role for polymodal C-fiber afferents in nonhistaminergic itch. J. Neurosci 28, 7659–7669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kardon AP, Polgár E, Hachisuka J, Snyder LM, Cameron D, Savage S, Cai X, Karnup S, Fan CR, Hemenway GM, et al. (2014). Dynorphin acts as a neuromodulator to inhibit itch in the dorsal horn of the spinal cord. Neuron 82, 573–586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karl T, Duffy L, and Herzog H (2008). Behavioural profile of a new mouse model for NPY deficiency. Eur. J. Neurosci 28, 173–180. [DOI] [PubMed] [Google Scholar]
- Kini SP, DeLong LK, Veledar E, McKenzie-Brown AM, Schaufele M, and Chen SC (2011). The impact of pruritus on quality of life: the skin equivalent of pain. Arch. Dermatol 147, 1153–1156. [DOI] [PubMed] [Google Scholar]
- Koch SC, Del Barrio MG, Dalet A, Gatto G, Günther T, Zhang J, Seidler B, Saur D, Schüle R, and Goulding M (2017). RORb Spinal Interneurons Gate Sensory Transmission during Locomotion to Secure a Fluid Walking Gait. Neuron 96, 1419–1431.e5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koch SC, Acton D, and Goulding M (2018). Spinal Circuits for Touch, Pain, and Itch. Annu. Rev. Physiol 80, 189–217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lawson SN, Crepps B, and Perl ER (2002). Calcitonin gene-related peptide immunoreactivity and afferent receptive properties of dorsal root ganglion neurones in guinea-pigs. J. Physiol 540, 989–1002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma C, Nie H, Gu Q, Sikand P, and Lamotte RH (2012). In vivo responses of cutaneous C-mechanosensitive neurons in mouse to punctate chemical stimuli that elicit itch and nociceptive sensations in humans. J. Neurophysiol 107, 357–363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Madisen L, Zwingman TA, Sunkin SM, Oh SW, Zariwala HA, Gu H, Ng LL, Palmiter RD, Hawrylycz MJ, Jones AR, et al. (2010). A robust and high-throughput Cre reporting and characterization system for the whole mouse brain. Nat. Neurosci 13, 133–140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Madisen L, Garner AR, Shimaoka D, Chuong AS, Klapoetke NC, Li L, van der Bourg A, Niino Y, Egolf L, Monetti C, et al. (2015). Transgenic mice for intersectional targeting of neural sensors and effectors with high specificity and performance. Neuron 85, 942–958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mantyh PW, Rogers SD, Honore P, Allen BJ, Ghilardi JR, Li J, Daughters RS, Lappi DA, Wiley RG, and Simone DA (1997). Inhibition of hyperalgesia by ablation of lamina I spinal neurons expressing the substance P receptor. Science 278, 275–279. [DOI] [PubMed] [Google Scholar]
- Melnick IV (2012). Cell type-specific postsynaptic effects of neuropeptide Y in substantia gelatinosa neurons of the rat spinal cord. Synapse 66, 640–649. [DOI] [PubMed] [Google Scholar]
- Mishra SK, and Hoon MA (2013). The cells and circuitry for itch responses in mice. Science 340, 968–971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miyakawa A, Furue H, Katafuchi T, Jiang N, Yasaka T, Kato G, and Yoshimura M (2005). Action of neuropeptide Y on nociceptive transmission in substantia gelatinosa of the adult rat spinal dorsal horn. Neuroscience 134, 595–604. [DOI] [PubMed] [Google Scholar]
- Moran TD, Colmers WF, and Smith PA (2004). Opioid-Like Actins of Neuropeptide Y in Rat Substantia Gelatinosa: Y1 Suppression of Inhibition and Y2 Suppression of Excitation. J. Neurophysiol 92, 3266–3275. [DOI] [PubMed] [Google Scholar]
- Mu D, Deng J, Liu K-F, Wu Z-Y, Shi Y-F, Guo W-M, Mao Q-Q, Liu X-J, Li H, and Sun Y-G (2017). A central neural circuit for itch sensation. Science 357, 695–699. [DOI] [PubMed] [Google Scholar]
- Naveilhan P, Hassani H, Lucas G, Blakeman KH, Hao J-X, Xu X-J, Wiesenfeld-Hallin Z, Thoén P, and Ernfors P (2001). Reduced antinociception and plasma extravasation in mice lacking a neuropeptide Y receptor. Nature 409, 513–517. [DOI] [PubMed] [Google Scholar]
- Nichols ML, Allen BJ, Rogers SD, Ghilardi JR, Honore P, Luger NM, Finke MP, Li J, Lappi DA, Simone DA, and Mantyh PW (1999). Transmission of chronic nociception by spinal neurons expressing the substance P receptor. Science 286, 1558–1561. [DOI] [PubMed] [Google Scholar]
- Padilla SL, Qiu J, Soden ME, Sanz E, Nestor CC, Barker FD, Quintana A, Zweifel LS, Rønnekleiv OK, Kelly MJ, and Palmiter RD (2016). Agouti-related peptide neural circuits mediate adaptive behaviors in the starved state. Nat. Neurosci 19, 734–741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Papoiu ADP, Coghill RC, Kraft RA, Wang H, and Yosipovitch G (2012). A tale of two itches. Common features and notable differences in brain activation evoked by cowhage and histamine induced itch. Neuroimage 59, 3611–3623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peirs C, Williams S-PG, Zhao X, Walsh CE, Gedeon JY, Cagle NE, Goldring AC, Hioki H, Liu Z, Marell PS, and Seal RP (2015). Dorsal Horn Circuits for Persistent Mechanical Pain. Neuron 87, 797–812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poindexter GS, Bruce MA, LeBoulluec KL, Monkovic I, Martin SW, Parker EM, Iben LG, McGovern RT, Ortiz AA, Stanley JA, et al. (2002). Dihydropyridine neuropeptide Y Y(1) receptor antagonists. Bioorg. Med. Chem. Lett 12, 379–382. [DOI] [PubMed] [Google Scholar]
- Ralvenius WT, Neumann E, Pagani M, Acuna MA, Wildner H, Benke D, Fischer N, Rostaher A, Schwager S, Detmar M, et al. (2018). Itch suppression in mice and dogs by modulation of spinal α2 and α3GABAA receptors. Nat. Commun 9, 3230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reich A, Orda A, Wisnicka B, and Szepietowski JC (2007). Plasma neuropeptides and perception of pruritus in psoriasis. Acta Derm. Venereol 87, 299–304. [DOI] [PubMed] [Google Scholar]
- Reich A, Mędrek K, Adamski Z, and Szepietowski JC (2013). Itchy hair–trichoknesis: a variant of trichodynia or a new entity? Acta Derm. Venereol 93, 591. [DOI] [PubMed] [Google Scholar]
- Ringkamp M, Schepers RJ, Shimada SG, Johanek LM, Hartke TV, Borzan J, Shim B, LaMotte RH, and Meyer RA (2011). A role for nociceptive, myelinated nerve fibers in itch sensation. J. Neurosci 31, 14841–14849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ross SE, Mardinly AR, McCord AE, Zurawski J, Cohen S, Jung C, Hu L, Mok SI, Shah A, Savner EM, et al. (2010). Loss of inhibitory interneurons in the dorsal spinal cord and elevated itch in Bhlhb5 mutant mice. Neuron 65, 886–898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sathyamurthy A, Johnson KR, Matson KJE, Dobrott CI, Li L, Ryba AR, Bergman TB, Kelly MC, Kelley MW, and Levine AJ (2018). Massively Parallel Single Nucleus Transcriptional Profiling Defines Spinal Cord Neurons and Their Activity during Behavior. Cell Rep 22, 2216–2225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmelz M, Schmidt R, Bickel A, Handwerker HO, and Torebjörk HE (1997). Specific C-receptors for itch in human skin. J. Neurosci 17, 8003–8008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmelz M, Schmidt R, Weidner C, Hilliges M, Torebjörk HE, and Handwerker HO (2003). Chemical response pattern of different classes of C-nociceptors to pruritogens and algogens. J. Neurophysiol 89, 2441–2448. [DOI] [PubMed] [Google Scholar]
- Sciolino NR, Plummer NW, Chen Y-W, Alexander GM, Robertson SD, Dudek SM, McElligott ZA, and Jensen P (2016). Recombinase-Dependent Mouse Lines for Chemogenetic Activation of Genetically Defined Cell Types. Cell Rep 15, 2563–2573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seidler B, Schmidt A, Mayr U, Nakhai H, Schmid RM, Schneider G, and Saur D (2008). A Cre-loxP-based mouse model for conditional somatic gene expression and knockdown in vivo by using avian retroviral vectors. Proc. Natl. Acad. Sci. USA 105, 10137–10142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sieber MA, Storm R, Martinez-de-la-Torre M, Müller T, Wende H, Reuter K, Vasyutina E, and Birchmeier C (2007). Lbx1 acts as a selector gene in the fate determination of somatosensory and viscerosensory relay neurons in the hindbrain. J. Neurosci 27, 4902–4909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simone DA, Zhang X, Li J, Zhang J-M, Honda CN, LaMotte RH, and Giesler GJ Jr. (2004). Comparison of responses of primate spinothalamic tract neurons to pruritic and algogenic stimuli. J. Neurophysiol 91, 213–222. [DOI] [PubMed] [Google Scholar]
- Solorzano C, Villafuerte D, Meda K, Cevikbas F, Bráz J, Sharif-Naeini R, Juarez-Salinas D, Llewellyn-Smith IJ, Guan Z, and Basbaum AI (2015). Primary afferent and spinal cord expression of gastrin-releasing peptide: message, protein, and antibody concerns. J. Neurosci 35, 648–657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Solway B, Bose SC, Corder G, Donahue RR, and Taylor BK (2011). Tonic inhibition of chronic pain by neuropeptide Y. Proc. Natl. Acad. Sci. USA 108, 7224–7229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun YG, and Chen ZF (2007). A gastrin-releasing peptide receptor mediates the itch sensation in the spinal cord. Nature 448, 700–703. [DOI] [PubMed] [Google Scholar]
- Sun YG, Zhao ZQ, Meng XL, Yin J, Liu XY, and Chen ZF (2009). Cellular basis of itch sensation. Science 325, 1531–1534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Szabo NE, da Silva RV, Sotocinal SG, Zeilhofer HU, Mogil JS, and Kania A (2015). Hoxb8 intersection defines a role for Lmx1b in excitatory dorsal horn neuron development, spinofugal connectivity, and nociception. J. Neurosci 35, 5233–5246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taiwo OB, and Taylor BK (2002). Antihyperalgesic effects of intrathecal neuropeptide Y during inflammation are mediated by Y1 receptors. Pain 96, 353–363. [DOI] [PubMed] [Google Scholar]
- Taniguchi H, He M, Wu P, Kim S, Paik R, Sugino K, Kvitsiani D, Fu Y, Lu J, Lin Y, et al. (2011). A resource of Cre driver lines for genetic targeting of GABAergic neurons in cerebral cortex. Neuron 71, 995–1013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Todd AJ, Hughes DI, Polgár E, Nagy GG, Mackie M, Ottersen OP, and Maxwell DJ (2003). The expression of vesicular glutamate transporters VGLUT1 and VLUGT2 in neurochemically defined axonal populations in the rat spinal cord with emphasis on the dorsal horn. Eur. J. Neurosci 17, 13–27. [DOI] [PubMed] [Google Scholar]
- Wahlgren CF, Hägermark O, and Bergström R (1991). Patients’ perception of itch induced by histamine, compound 48/80 and wool fibres in atopic dermatitis. Acta Derm. Venereol 71, 488–494. [PubMed] [Google Scholar]
- Wang M, Wang Q, and Whim MD (2016). Fasting induces a form of autonomic synaptic plasticity that prevents hypoglycemia. Proc. Natl. Acad. Sci. USA 113, E3029–E3038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wende H, Lechner SG, Cheret C, Bourane S, Kolanczyk ME, Pattyn A, Reuter K, Munier FL, Carroll P, Lewin GR, and Birchmeier C (2012). The transcription factor c-Maf controls touch receptor development and function. Science 335, 1373–1376. [DOI] [PubMed] [Google Scholar]
- Wickersham IR, Lyon DC, Barnard RJO, Mori T, Finke S, Conzelmann K-K, Young JAT, and Callaway EM (2007). Monosynaptic restriction of transsynaptic tracing from single, genetically targeted neurons. Neuron 53, 639–647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wiley RG, and Lappi DA (1999). Targeting neurokinin-1 receptor-expressing neurons with [Sar9,Met(O2)11 substance P-saporin. Neurosci. Lett 277, 1–4. [DOI] [PubMed] [Google Scholar]
- Wu Y, Wang C, Sun H, LeRoith D, and Yakar S (2009). High-efficient FLPo deleter mice in C57BL/6J background. PLoS ONE 4, e8054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang X, Tong YG, Bao L, and Hökfelt T (1999). The neuropeptide Y Y1 receptor is a somatic receptor on dorsal root ganglion neurons and a postsynaptic receptor on somatostatin dorsal horn neurons. Eur. J. Neurosci 11, 2211–2225. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.