Abstract
Damage accumulated in the genome and macromolecules is largely attributed to increased oxidative damage and a lack of damage repair in a cell, and this can eventually trigger the process of aging. Alleviating the extent of oxidative damage is therefore considered as a potential way to promote longevity. SFPS, a heteropolysaccharide extracted from the brown alga Sargassum fusiforme, has previously been shown to alleviate oxidative damage during the aging process in mice, but whether SFPS could extend the lifespan of an organism was not demonstrated. Furthermore, the precise component within SFPS that is responsible for the antioxidant activity and the underlying mechanism of such activity was also not resolved. In this study, SP2, a fucoidan derived from SFPS, was shown to exhibit strong antioxidant activity as measured by in vitro radical-scavenging assays. SP2 also improved the survival rate of D. melanogaster subjected to oxidative stress. The flies that were fed with a diet containing SP2 from the time of eclosion displayed significant enhancement in lifespan and reduced accumulation of triglyceride at the old-age stage. In addition, SP2 markedly improved the activities of the antioxidant enzymes, superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GSH-Px) and reduced the contents of the malondialdehyde (MDA) and oxidized glutathione (GSSG) in old flies. Furthermore, SP2 also upregulated the expression levels of the nuclear factor-erythroid-2-like 2 (nfe2l2 or nrf2) and its downstream target genes, accompanied by a dramatic reduction in the expression of kelch-like ECH-associated protein 1 (keap1, a canonical inhibitor of the Nrf2) in old flies. Additional support linking the crucial role of the Nrf2/ARE pathway to the antioxidant effect of SP2 was the relatively high survival rate under heat stress for D. melanogaster individuals receiving SP2 supplement, an effect that was abolished by the inclusion of inhibitors specific for the Nrf2/ARE pathway. Collectively, the results indicated that SP2, a S. fusiforme fucoidan, could promote longevity in D. melanogaster by enhancing the Nrf2-mediated antioxidant signaling pathway during the aging process.
1. Introduction
Aging is a time-dependent and gradual decline in the physiological function of an organism, which ultimately results in diseases and death. It is now well accepted that the cause of aging and the factors that promote aging are related to genes, oxidative stress, signaling pathways, and energy metabolism [1–3]. Although the exact cause of aging is still a controversy, it is well acknowledged that these factors are interconnected and they also interact with each other. The concept that oxidative damage can promote aging is well recognized [4, 5]. Cumulative genomic and cellular damages might induce the time-dependent loss of function in living organisms, and this is predominantly a result of oxidative damage [4, 6]. However, the antiaging effect conferred by antioxidants remains inconclusive, since the extension of lifespan achieved by long-term consumption of antioxidants such as vitamin E, vitamin A, beta-carotene, and glutathione (GSH) is not consistent among different organisms [7], and in human, an increase in the mortality has even been observed [8, 9]. Therefore, the strategy of using antioxidant to promote longevity should be optimized.
Free radicals such as reactive oxygen species (ROS) and reactive nitrogen species (RNS) are considered as the major drivers of oxidative damage. These free radicals and the onset of oxidative damage can be neutralized or alleviated by detoxifying and antioxidant enzymes such as SOD, CAT, and GSH-Px, which are transcriptionally regulated by the Nrf2/ARE signaling pathway [10]. Nrf2 is a transcriptional factor that plays an indispensable role in the detoxification and antioxidant systems, and nef2l2, the gene encoding Nrf2, has even been proposed to act as a longevity gene [11, 12]. Unsurprisingly, knocking out the nfe2l2 gene in mice has been shown to dramatically reduce the capacity of stress resistance and cellular protection, thus accelerating the aging process [13, 14]. Additionally, Nrf2 is highly expressed in young mice and naked mole rats [13, 15, 16]. There have been numerous studies showing that activation of the Nrf2/ARE signaling pathway can slow down the aging process or ameliorate aging-related diseases [17, 18]. However, more substantial evidence is still needed to confirm the antiaging effect conferred by the activation of the Nrf2/ARE signaling.
Sargassum fusiforme is a seaweed that has earned its reputation as a longevity-promoting vegetable in Northeastern Asia since it can modulate metabolism, strengthen immune response, and maintain redox homeostasis [19]. Many studies have shown that polysaccharides extracted from S. fusiforme possess antioxidant, antitumor, and immunomodulatory activities [19–22]. Our previous studies have revealed that SFPS, a heteropolysaccharide extracted from S. fusiforme, can significantly improve the antioxidant defense in aging mice by promoting Nrf2-dependent stress resistance and cellular protection [23, 24]. However, whether SFPS might also extend the lifespans of organisms, in general, remains undetermined.
In this study, we screened several fractions of polysaccharides derived from SFPS. The screening procedure combined in vitro cell-free antioxidant assay with in vivo oxidative-resistant test. The results suggested that long-term SP2 supplement could significantly prolong the lifespan of D. melanogaster and enhance its antioxidant capacity. Subsequent experiment further showed that the SP2 could significantly activate the CncC/Nrf2-mediated antioxidant signaling pathway, thus enhancing the stress resistance and antiaging capacity of D. melanogaster. Collectively, the data suggested that SP2 might promote longevity in D. melanogaster through activating the Nrf2/ARE signaling pathway.
2. Materials and Methods
2.1. Materials and Reagents
Sargassum fusiforme was harvested from the coast of Wenzhou, Zhejiang Province, China. L-Rhamnose (L-Rha), D-galacturonic acid (D-GalA), D-mannose (D-Man), and D-xylose (D-Xyl) were purchased from China National Institute for the Control of Pharmaceutical and Biological Products. Luteolin, all-trans-retinoic-acid (ATRA), D-glucuronic acid (D-GlcA), D-glucose (D-Glc), D-galactose (D-Gal), and L-arabinose (L-Ara) were purchased from Sigma-Aldrich. 1-Phenyl-3-methyl-5-pyrazolone (PMP) was purchased from Major Chemicals Co. Ltd. (Hangzhou, China). Acetonitrile (HPLC-grade) was obtained from Merck (E. Merck, Darmstadt, Germany). Other utilized chemicals used were of analytical reagent grade.
2.2. Extraction of Polysaccharides from S. fusiforme
Fresh S. fusiforme was dried to a constant weight and then pulverized to powder, which was then used for polysaccharide extraction as described previously [23, 24]. In brief, the powder was initially defatted in 95% ethanol, and then the heteropolysaccharide SFPS was isolated from the residual fraction by hot-water extraction and ethanol precipitation. Subsequently, fractionation of SFPS, which involved CaCl2 precipitation to separate the fucoidan from the alginate, yielded three fractions, designated as SP1, SP2, and SP3 (Supplementary Figure 1).
2.3. Stress-Induced Survival Rate Assays and Lifespan Assay
Drosophila melanogaster individuals (male and female) were maintained at 25°C and 60% relative humidity and under a 12 h light : 12 h dark cycle. According to the gender, the virgin flies were separated within 8 h after eclosion. They were reared on basal medium (10.5% cornmeal (w/v), 10.5% sucrose (w/v), 2.1% yeast (w/v), 1.3% agar (w/v), and 0.4% propionic acid (v/v)) without or with polysaccharide. Male flies were randomly divided into four groups, with at least 100 flies per group. One group was reared on basal medium only (Con group) while the remaining groups were each reared on basal medium plus 1.6 g/L SP1, SP2, or SP3 for 10 days. After that, the flies were transferred to a medium containing 5 mmol/L paraquat, and the number of dead flies from each group was recorded every 10 h. Lifespan assay: the lifespan assay was carried out from the time of eclosion to death. The flies were reared on basal medium without or with different concentrations of SP2 (0.4 g/L: low concentration of SP2, LSP; 0.8 g/L: median concentration of SP2, MSP; and 1.6 g/L: high concentration of SP2, HSP). The number of dead flies from each group was recorded every day until all the flies were dead. Heat stress-induced survival rate assay with/without inhibition of the Nrf2/ARE signaling pathway: to block the Nrf2/ARE signaling, flies were reared on basal medium containing, in addition to SP2, the chemical inhibitor of the Nrf2/ARE pathway, luteolin or all-trans-retinoic-acid (ATRA). The flies were divided into five groups: C or control group (no inhibitor), L1 group (15 μmol/L luteolin), L2 group (30 μmol/L luteolin), A1 group (0.125 g/L ATRA), and A2 group (0.25 g/L ATRA). The concentration of SP2 used was the concentration that yielded the best antiaging effect for each sex, as determined from the result of the lifespan experiment. The flies were reared in a 30°C incubator to provide heat stress, and the number of dead flies from each group was recorded every day until all the flies were dead. In addition, another lot of flies were prepared in the same way; but after 10 days, 15 flies per sample and 3 samples per group were taken for RNA extraction, whereas 25 flies per sample and 3 samples per group were used taken for protein extraction at a specific time. The RNA and protein samples were used for subsequent qRT-PCR and antioxidant activity assays, respectively.
2.4. HPLC Analysis
The molecular weight of SP2 was determined by high-performance gel-permeation chromatography (HPGPC) using a Waters HPLC apparatus equipped with a TSK-gel G-5000PWXL column (Φ7.8 mm × 300 mm ID, TOSOH, Japan) and a Waters 2414 refractive index detector. The SP2 sample (20 μL of 5 mg/mL preparation) was injected into the column and eluted with 0.2 M NaCl at a flow rate of 0.6 mL/min and a column temperature of 45°C. The molecular weights were estimated by reference to the calibration curve obtained from the elution of Dextran T-series standards of known molecular weights. HPLC was performed with a Hypersil ODS-3 column (4.6 mm × 250 mm) using a mobile phase consisting of 83.0% (v/v) phosphate-buffered saline (PBS, 0.1 mol/L, pH 6.9) and 17% acetonitrile (v/v). The flow rate was set at 0.8 mL/min, and the eluent was monitored by absorbance at 254 nm. D-Glc, D-Gal, L-Ara, L-Rha, D-Xyl, D-Man, L-Fuc, D-GalA, and D-GlcA were used as reference monosaccharides.
2.5. Fourier Transform-Infrared (FT-IR) Spectroscopy
The FT-IR spectrum of the SP2 was obtained with a Tensor 27 spectrophotometer (Bruker Daltonics, Ettlingen, Germany). The SP2 sample was mixed with KBr powder and then pressed into a 1 mm thick disk, which was then used to obtain the spectrum, recorded in the frequency range of 4000–400 cm−1.
2.6. Detection of Triglyceride (TG) Content
Twenty-five flies from each experimental group were randomly taken at a specific time point to determine the content of triglyceride. Triglyceride content was determined by measuring the concentration of soluble triglyceride. The flies were first combined and homogenized in normal saline. The homogenate was centrifuged at 3000 × g, and the concentration of TG in the supernatant was measured using a Genzyme Triglyceride Kit purchased from Nanjing Jiancheng Incorporation (Nanjing, China).
2.7. Scavenging Activity of DPPH (1,1-Diphenyl-2-Picrylhydrazyl Radical 2,2-Diphenyl-1-(2,4,6-Trinitrophenyl) Hydrazyl) Radical
The effect of SP2 on free radical-scavenging activity was evaluated by measuring the level of DPPH-scavenging activity using a DPPH-scavenging assay kit purchased from Nanjing Jiancheng Incorporation (Nanjing, China). Polysaccharide solution was prepared by dissolving the polysaccharide powder in distilled water as previously described [23]. Next, the polysaccharide solution was added to DPPH reagent (in 95% ethanol) to yield a final concentration of 0.4, 0.8, or 1.6 g/L, and the reaction mixture was thoroughly mixed by shaking and then incubated in the dark for 30 min at room temperature. After that, the absorbance of the resulting solution was read at 517 nm against a blank. DPPH radical-scavenging activity was calculated using the equation: DPPH − scavenging rate (%) = [1‐(A 1‐A 2)/A 0] × 100%, where A 0 is the absorbance of the DPPH alone, A 1 is the absorbance of DPPH+polysaccharide, and A 2 is the absorbance of the polysaccharide only.
2.8. FRAP (Ferric Ion Reducing Antioxidant Power) and ABTS (2,2′-Azino-Bis(3-Ethylbenzothiazoline-6-Sulfonic Acid)) Assays
FRAP and ABTS assays were performed using assay kits purchased from Nanjing Jiancheng Incorporation (Nanjing, China) but with an additional control consisting of the sample only to account for the absorbance due entirely to the polysaccharide [25]. FRAP reagent was prepared by mixing 300 mmol/L acetate buffer (pH 3.6) with 10 mmol/L 2,4,6-tripyridyl-s-triazine (TPTZ) and 20 mmol/L FeCl3 in a volume ratio of 10 : 1 : 1, while ABTS radical was prepared by mixing the ABTS solution (7 mmol/L) with potassium persulfate (2.45 mmol/L).
2.9. Measurements of the MDA Content and the Enzymatic Activities of CAT and SOD
Twenty-five flies from each group were randomly collected at a specific time and then homogenized as one sample. After centrifugation at 3000 × g, the supernatant was used for MDA, CAT, and SOD assays performed with assay kits purchased from Nanjing Jiancheng Incorporation (Nanjing, China). The principles of the assay kits were based on previously described methods [26, 27].
2.10. qRT-PCR Analysis
Total RNA was extracted from whole flies using the TRIzol reagent. Fifteen flies per sample and three samples per group were taken for total RNA extraction. The total RNA extracted from each sample was then subjected to qRT-PCR analysis using the AffinityScript qPCR cDNA synthesis kit. The primers for the target genes were designed based on the sequences retrieved from FlyBase (http://flybase.org). The sequences of the primers are listed in Table 1. Quantitative RT-PCR assay was performed in a Roche LightCycler 480 platform using the SYBR Green II method. The transcript level of each gene was calculated by the ΔΔCt method with reference to that of the rp49 gene and then normalized to the corresponding control group.
Table 1.
The sequences of the primers.
| Gene | FlyBase no. | Forward primers (5′-3′) | Reverse primers (5′-3′) |
|---|---|---|---|
| cncC | FBgn0262975 | GTCGCCACTAAAACCGCATC | TTGTTCTTTCCACGCCGACG |
| keap1 | FBgn0038475 | TACGAAGATAGTGACGCCCC | GTGAAAGACGCTGGTGGAGT |
| ho | FBgn0037933 | ATGACGAGGAGCAGCAGAAG | ACAAAGATTAGTGCGAGGGC |
| gclc | FBgn0040319 | GAGCCATTAGTGCCGTTAGT | GTCTTTCGTCTTCGTCTTGG |
| rp49 | FBgn0002626 | CCCTCTTCCAGCCATCGTTC | CCACCGATCCAGACGGAGTA |
2.11. Statistical Analysis
The data were expressed as mean ± SD of, at least, three separate experiments. Statistical analyses were carried out by two-way ANOVA and Fisher's LSD multiple comparison tests by SPSS 17.0. Differences among groups were considered statistically significant at the P < 0.05 level.
3. Results
3.1. The Fucoidan SP2 Exhibits Excellent Antioxidant Activity
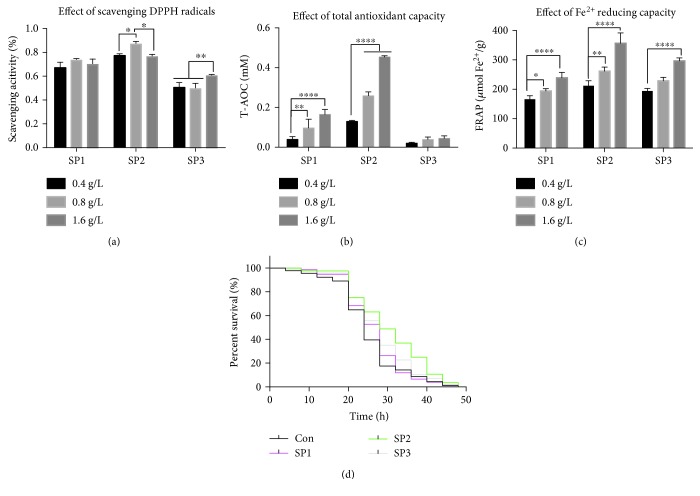
It is well acknowledged that oxidative damage contributes to aging, and therefore, factors that enhance resistance to oxidative stress are considered to have antiaging effects [28–31]. We have previously reported that the SFPS, an S. fusiforme heteropolysaccharide, possesses antioxidant activity and shown that it can significantly alleviate aging-related stresses [23, 24]. However, the active component of SFPS responsible for the antiaging effect was not further evaluated. To address this issue, SFPS was fractionated into three fractions, which were designated as SP1, SP2, and SP3 (Supplementary Figure 1). SP1 and SP2 were both fucoidan whereas SP2 was alginate. In order to select the most promising candidate, the antioxidant capacities of SP1, SP2, and SP3 were determined by both in vitro and in vivo methods. SP2 displayed the highest antioxidant activity as shown by radical- and ion-scavenging activities (Figures 1(a)–1(c)), in particular, the ABTS clearance rate of SP2 was twice the rate of SP1 (Figure 1(b)). All three polysaccharides significantly enhanced the average survival time of D. melanogaster individuals that were treated with 5 mmol/L paraquat to induce oxidative stress. The flies that did not receive polysaccharide supplement died within 48 h of paraquat treatment. Twenty-four hours after paraquat treatment, the survival rate of the flies that had been given SP2 was 70%, whereas those of flies that had been given SP2 and SP3 were 55.4% and 57.2%, respectively, while flies that did not receive polysaccharide had a survival rate of just 39.5% (Figure 1(d)). The results suggested that SP2 might be an effective antioxidant, and therefore, it was chosen for further antiaging assessment.
Figure 1.
Antioxidant capacity of the different polysaccharides fractionated from SFPS. Antioxidant activity of the polysaccharides was assayed in three different cell-free systems. (a) DPPH-scavenging activity, (b) ABTS-scavenging activity, and (c) radical-scavenging activities and Fe2+ reducing. Data are shown as mean ± SD from three determinations. “∗,” “∗∗,” and “∗∗∗∗” indicate a significant difference at the P < 0.05, P < 0.01, and P < 0.0001 levels, respectively. (d) Fruit flies were randomly collected (100 flies/group) and reared on basal medium without (control group) or with 1.6 g/L of SP1, SP2, or SP3 for 10 days. Subsequently, the in vivo oxidative resistant capacity was evaluated by determining the survival rate of the flies following exposure to oxidative stress induced by 5 mmol/L paraquat, given as a diet.
3.2. Chemical Composition of SP2
The bioactivity of a compound is closely related to its chemical composition and structure. Therefore, it is vital to explore the structure-activity relationship of SP2. SP2 was determined to be a fucoidan with fucose being the major monosaccharide (52.89%), while sulfate radical, which is a unique modification of fucoidan, accounted for about 15.3% (Table 2). SP2 also contained a relatively high content of Xyl (12.1%), Gal (11.51%), and Glc (11.33%) (Table 2).
Table 2.
Chemical composition of SP2.
| Molecular weight (kDa) | Sulfate content (%) |
Monosaccharide composition (%) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 52.7 | 15.3 | Man | Rha | GlcA | GalA | Glc | Gal | Xyl | Ara | Fuc |
| 3.50 | 1.77 | 1.11 | 2.61 | 11.33 | 11.51 | 12.10 | — | 52.89 | ||
Notes: Man: D-mannose; Rha: L-rhamnose; GlcA: D-glucuronic acid; GalA: D-galacturonic acid; Glc: D-glucose; Gal: D-galactose; Xyl: D-xylose; Ara: L-arabinose; Fuc: L-fucose.
3.3. SP2 Promotes Longevity in D. melanogaster
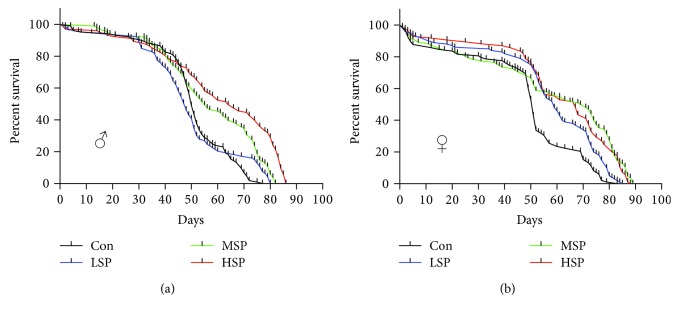
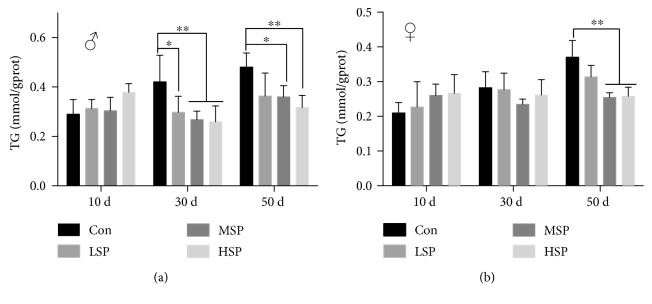
The in vitro antioxidant activity assay and the in vivo oxidative stress resistance assay both demonstrated the remarkable antioxidant capacity of SP2. However, the question as to whether SP2 could promote longevity under normal condition needed to be addressed. First, the flies were reared, from the time of eclosion, on media supplemented with different concentrations of SP2. The result revealed a significant increase in lifespan for the flies receiving SP2 supplement (Figure 2, Table 3). Interestingly, the effect of SP2 on the lifespan of the flies varied with gender. At a concentration of 1.6 g/L in the medium, SP2 extended the mean lifespan by about 22%, but it extended the male lifespan by a maximum of 10%. At 0.8 g/L, SP2 extended the mean lifespan by about 24% and the female lifespan by a maximum of 10% (Table 3). Furthermore, SP2 also slowed down the accumulation of triglyceride (TG) in the aging flies (Figure 3). Old flies are known to accumulate high levels of TG because of a significant decline in their metabolism, and thus, the accumulation of TG has been considered as an aging index [32, 33]. Our data was therefore consistent with SP2 having an antiaging effect, which was clearly demonstrated by its ability to increase the lifespan of D. melanogaster.
Figure 2.
SP2 extends the lifespan of D. melanogaster. Male (a) and female (b) fruit flies were reared on medium containing no SP2 (Con) or different concentrations of SP2 (0.4 g/L (LSP), 0.8 g/L (MSP), and 1.6 g/L (HSP)); the number of dead flies from each group was counted daily; n > 250 flies.
Table 3.
Effect of the SP2 to the lifespan of D. melanogaster.
| Male | Female | |||||||
|---|---|---|---|---|---|---|---|---|
| Con | LSP | MSP | HSP | Con | LSP | MSP | HSP | |
| N | 291 | 283 | 272 | 275 | 270 | 288 | 281 | 288 |
| Max. lifespan | 80.2 ± 1.2 | 84.3 ± 3.6 | 84.5 ± 2.2 | 88.4 ± 1.9∗ | 83.0 ± 1.7 | 85.1 ± 1.0 | 91.5 ± 1.1∗ | 87.1 ± 2.3 |
| Mean lifespan | 58.2 ± 2.5 | 56.0 ± 3.6 | 65.3 ± 1.9∗ | 71.1 ± 3.0∗ | 59.7 ± 1.3 | 66.9 ± 1.9∗ | 74.1 ± 0.9∗ | 70.4 ± 0.8∗ |
Notes: mean ± SD, ∗ P < 0.05.
Figure 3.
SP2 decreases the accumulation of triglyceride (TG) in D. melanogaster during the aging process. The flies were supplemented with varied concentrations of SP2 (0 (Con), 0.4 g/L (LSP), 0.8 g/L (MSP), and 1.6 g/L (HSP), and the contents of soluble TG in the male (a) and female (b) flies of different ages were then measured. Data are shown as the mean ± SD from three determinations, each used the extract obtained from 25 flies. “∗” and “∗∗” indicate a significant difference at the P < 0.05 and P < 0.01 levels, respectively.
3.4. SP2 Enhances the Endogenous Antioxidant Capacity of D. melanogaster
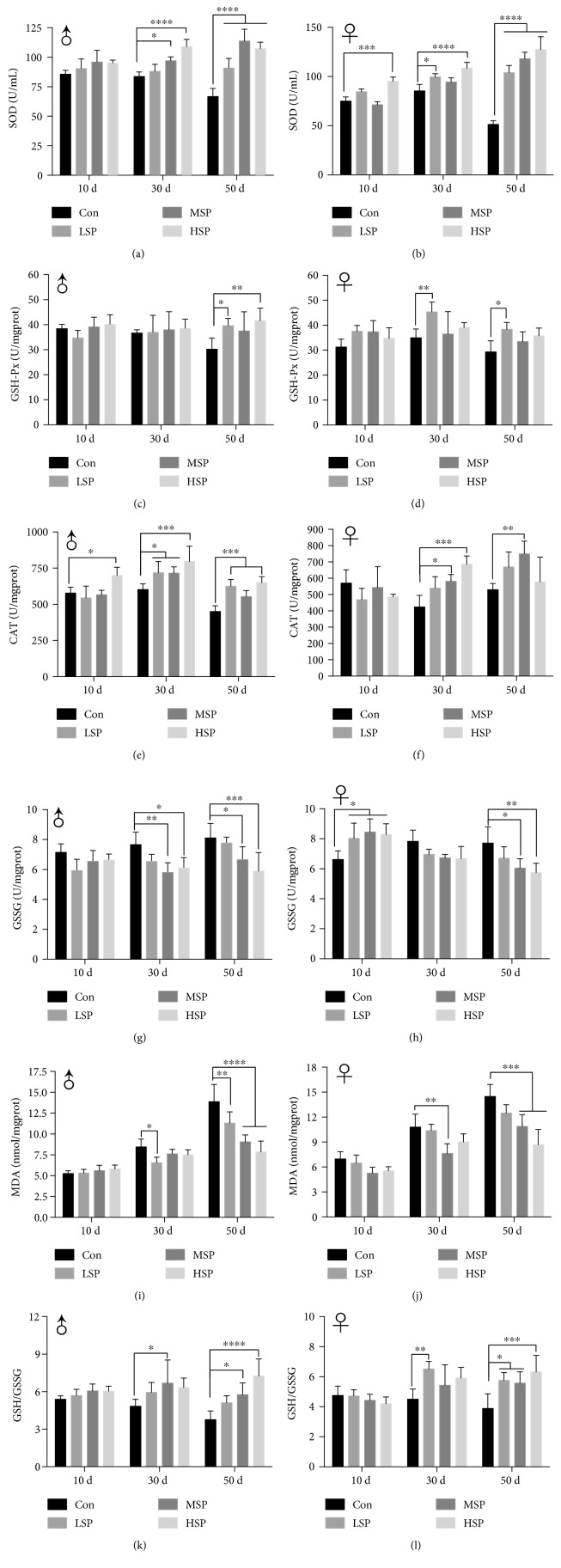
Endogenous antioxidant activity is an important indicator for determining the aging status of an organism. For example, significant declines in peroxidase and superoxide dismutase activities have been observed in aging organisms, from yeast to human [34]. Analysis of the antioxidant capacity of D. melanogaster over time revealed an overall decrease in 50-day-old individuals (Figures 4 and 5). Furthermore, the content of MDA in these individuals also increased during the aging process, and the ratio of GSH/GSSG, commonly used as an oxidant index, was reduced. In contrast, 50-day-old flies that had been given SP2 supplement exhibited significant increases in the levels of SOD, CAT, and GSH-Px activities (Figures 4(a)–4(f)). SP2 supplement also reduced the content of MDA in these individuals by as much as 50% and increased the GSH/GSSG ratio by about two folds (Figures 4(i)–4(l)). Taken together, these results suggested that SP2 supplement could slow the decline in antioxidant defense of D. melanogaster during the aging process.
Figure 4.
SP2-supplemented diet slows down the decline in the antioxidant capacity of D. melanogaster during the aging process. The fruit flies were reared on a medium containing no SP2 (Con) or different concentrations of SP2 (0.4 g/L (LSP), 0.8 g/L (MSP), and 1.6 g/L (HSP)), and their antioxidant capacity was analyzed at different ages. SOD activity (a, b); GSH-Px level (c, d); CAT activity (e, f); CSSG content (g, h); MDA content (i, j), and GSH/GSSG (k, l). The antioxidant capacity was determined for both male flies (a, d, e, g, i, and k) and female flies (b, d, f, h, j, and l). Data are shown as mean ± SD from three determinations, each used the extract obtained from 25 flies. “∗,” “∗∗,” and “∗∗∗” indicate a significant difference at the P < 0.05, P < 0.01, and P < 0.001 levels, respectively.
Figure 5.
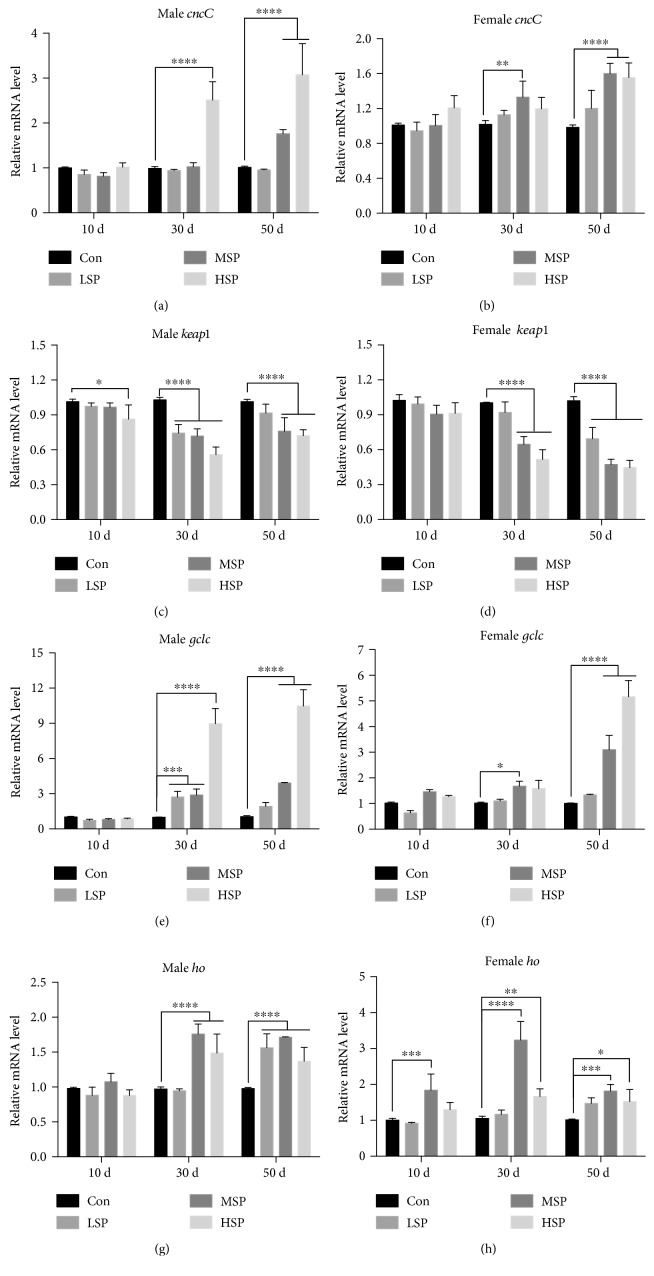
SP2 supplement upregulates the Keap1/CncC/ARE signaling pathway in aging flies. Fruit flies were reared on basal medium containing no SP2 (Con) or different concentrations of SP2 (0.4 g/L (LSP), 0.8 g/L (MSP), and 1.6 g/L (HSP)) over 50 days. The flies were taken at different time intervals, and the mRNA levels of the representative genes (cncC, keap1, gclc, and ho) of the Keap1/CncC/ARE signaling pathway were measured by qRT-PCR using the rp49 gene as the reference gene. (a, b) cncC, (c, d) keap1, (e, f) gclc, and (g, h) ho. The expression levels of the genes were evaluated by the ΔΔCt method and normalized to those of the corresponding control. Data are shown as mean ± SD from three determinations, each used the RNA extracted from 15 flies. “∗,” “∗∗,” and “∗∗∗” indicate a significant difference at the P < 0.05, P < 0.01, and P < 0.001 levels, respectively.
3.5. Expansion of D. melanogaster Lifespan by SP2 Involves the CncC/Nrf2/ARE Signaling Pathway
Nuclear factor-erythroid 2-related factor 2/antioxidant responsive element (Nrf2/ARE) is one of the most important antioxidant pathways that counteract oxidative stress and damage. Nrf2 is conserved in metazoan, and in D. melanogaster, it is called CncC. Previous studies have shown that SFPS can upregulate the Nrf2/ARE signaling pathway to alleviate aging stress in mice [23, 24]. Whether SP2 might stimulate the Nrf2/ARE signaling pathway to enhance the lifespan of D. melanogaster was the focus of this study. First, the mRNA levels of cncC (nrf2) and its classical inhibitor keap1 in the flies of different ages were measured. The results revealed a remarkable upregulation of the cncC, and dramatic downregulation of keap1 mRNA levels in the old flies that had been given moderate (MSP) and high (HSP) concentrations of SP2 supplement (Figures 5(a)–5(d)). The transcriptional activity of Nrf2 was further determined by measuring the mRNA levels of its representative downstream target genes: ho and gclc. As expected, the mRNA levels of these downstream antioxidant genes were markedly upregulated at the 30-day-old and 50-day-old old stages in both sexes (Figures 5(e)–5(h)). Collectively, the results suggested that the Keap1/Nrf2/ARE signaling pathway might be significantly activated by SP2 during the aging process, thereby enhancing the antioxidant capacity of D. melanogaster individuals that had been given SP2 supplement, with the consequence of extending their lifespans.
3.6. SP2-Alleviated Heat Stress Depends on the CncC/Nrf2 Signaling Pathway
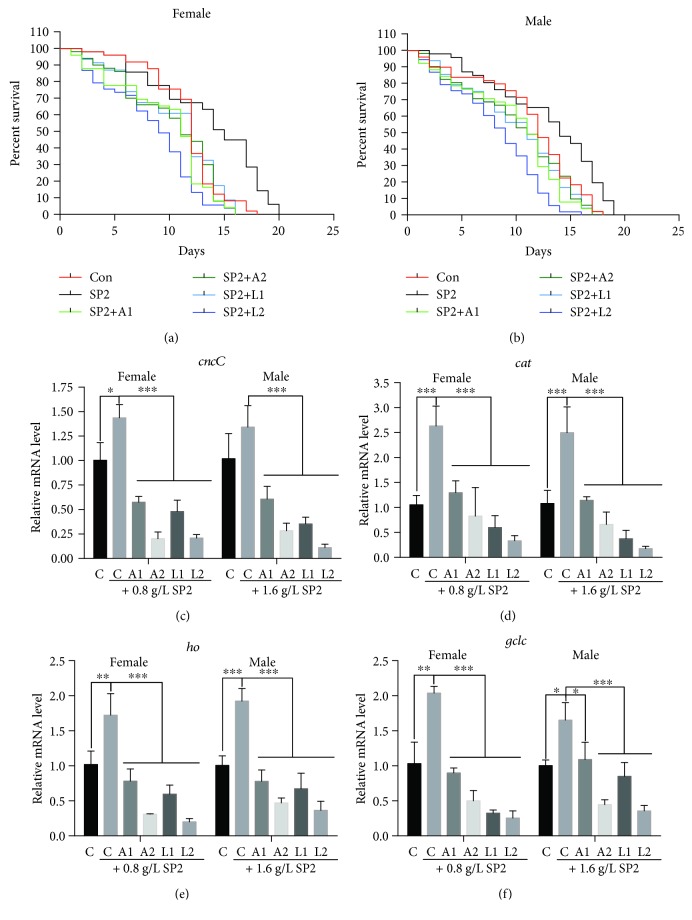
To validate that SP2 might exhibit antistress effect via the Nrf2-mediated signaling pathway, the Nrf2/ARE signaling was blocked by the chemical inhibitor, luteolin or ATRA [35, 36]. In order to accelerate the aging process, the flies were subjected to heat stress at 30°C. SP2 supplement significantly extended the lifespan of the flies under heat stress, but this effect was neutralized when the flies also received luteolin or ATRA in the medium (Figures 6(a) and 6(b)). Furthermore, the mRNA levels of the cncC gene and its representative downstream target genes, ho, cat, and gclc, in flies that were given just SP2 supplement were significantly upregulated compared with the control (no SP2 supplement) but declined relative to the control when the flies received both SP2 plus luteolin or ATRA (Figures 6(c)–6(f)). The effect of SP2 on stress resistance, therefore, appeared to be largely dependent on the CncC/Nrf2/ARE signaling pathway.
Figure 6.
SP2-mediated stress-resistant effect in fruit flies is dependent on the CncC/Nrf2/ARE signaling. Fruit flies were reared on medium containing no SP2 (Con) or different concentrations of SP2 (0.4 g/L (LSP), 0.8 g/L (MSP), and 1.6 g/L (HSP)) without and with all-trans-retinoic-acid (A1, 0.125 g/L; A2, 0.25 g/L) or luteolin (L1, 15 μmol/L; L2, 30 μmol/L), and the survival rates of the flies were determined. (a) Survival rate of male flies; (b) survival rate of female flies. In addition, samples of the flies were also taken after 10 days of treatment, and the transcript levels of cncC (c), cat (d), ho (e), and gclc (f) were then measured by qRT-PCR using the rp49 gene as a reference gene. The expression levels of the genes were evaluated by the ΔΔCt method, and then normalized to those of the corresponding control. Data are shown as mean ± SD from three determinations, each used the RNA extracted from 15 flies. “∗,” “∗∗,” and “∗∗∗” indicate a significant difference at the P < 0.05, P < 0.01, and P < 0.001 levels, respectively.
3.7. SP2 Has No Significant Effect on Body Weight
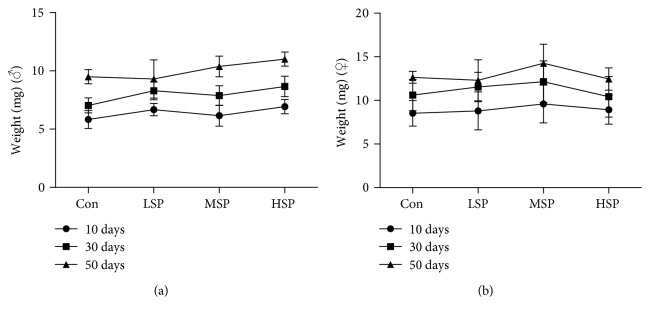
To investigate whether SP2 might affect food intake or trigger calorie restriction in fruit flies, the body weights of the flies given SP2 supplement were compared with those not receiving SP2. SP2 appeared to increase the body weights of the flies as measured at 10, 30, and 50 days, but the increases were not significant for both male or female groups (Figure 7). This suggested that SP2 did not restrict the food intake in D. melanogaster.
Figure 7.
Effect of SP2 on D. melanogaster body weight. Body weights of male (a) and female (b) flies at different ages and reared on medium without (Con) or with different concentrations of SP2: LSP (0.4 g/L), MSP (0.8 g/L), and HSP (1.6 g/L). Data are shown as mean ± SD from three determinations, each based on 10 flies.
4. Discussion
The use of Sargassum fusiforme by Traditional Chinese Medicine to treat thyroid diseases and as a health maintenance agent was first recorded in the ancient pharmaceutical book Shennong Bencaojing, but information concerning the effective components and the related mechanisms is still lacking. S. fusiforme has even been regarded as a longevity-promoting vegetable because it helps to modulate metabolism, strengthen immune response, and maintain redox homeostasis [19]. However, the claim that it promotes longevity has not been supported by any direct evidence, despite numerous studies demonstrating that extracts prepared from S. fusiforme, which contain predominantly polysaccharides, have different biological activities, such as antitumor and antioxidant activities [20–22]. We have previously reported that the heteropolysaccharide SFPS extracted from S. fusiforme can enhance resistance to oxidative stress and even ameliorate the aging process in mice [23, 24]. However, there was no evidence to prove that S. fusiforme can extend the lifespan of an organism. In this study, we showed that the fucoidan SP2, which was extracted from SFPS, could markedly prolong the lifespan of D. melanogaster, and to the best of our knowledge, this could be the first direct evidence to support the claim that S. fusiforme is a longevity-promoting vegetable.
We have integrated in vitro and in vivo antioxidant assays to screen for a promising antiaging candidate from among the different fractions extracted from SFPS. Initial tests revealed SP2 to be a promising candidate, as it exhibited the best antioxidant activity and gave the highest survival rate when fed to the flies (Figure 1). SP2 was therefore chosen for further study to examine its longevity-promoting effect. Indeed, SP2 could increase the lifespan of the flies, further supporting its role as a longevity-promoting polysaccharide (Figures 2 and 3). Although the integrated screening method described in this study was robust, the extent of its validity and practicality may require further study, as the in vitro antioxidant assay may simply depend on a redox reaction, while the in vivo antioxidant assay depends on both nonenzymatic antioxidant and enzymatic systems. Previous studies that attempted to evaluate the antiaging effect of some antioxidants such as vitamin C, vitamin E, and β-carotene did not produce conclusive. Good consistency in antioxidant capacity between in vivo and in vitro assays was obtained for SP2 (Figure 1). Therefore, regardless of the extent of application for this antiaging screening, the data gathered for SP2 alone indicated that at least, our integrated method might provide a feasible and effective screening approach for other polysaccharides derived from brown alga, which might have the ability to slow down aging caused by oxidative damages.
It is worth noting that we merely provided one evidence of a promising antiaging compound based on its strong antioxidant activity. However, aging is a complex process that also correlates with the downregulation of metabolism and protection of cellular components against internal or external inflicted damage [37]. A decrease in antioxidant capacity and an accumulation of TG have been suggested as the hallmarks of aging [38]. The content of TG decreased significantly in 50-day-old flies but increased slightly in 10-day-old flies when the flies were given SP2 supplement (Figure 3). This suggested that SP2 significantly promoted metabolism during the aging process and yet exerted no negative impact on development in young flies. The activities of SOD, GSH-Px, and CAT and the contents of MDA and GSH are usually used to provide a comprehensive assessment of the antioxidant capacity of an organism. SP2 supplement significantly upregulated the levels of SOD, CAT, and GSH-Px activities in the aging flies but downregulated the content of MDA and the ratio of GSSG/GSH in these flies (Figure 4). Intriguingly, SP2 did not interfere with the redox balance in 10-day-old flies but comprehensively slowed down both the decline in antioxidant capacity and the increase in oxidative stress in 30-day-old and 50-day-old flies (Figure 4). In addition, the lack of statistical significance in body weight increase for the flies given SP2 supplement compared with their control counterparts suggested that long-term SP2 supplement could significantly restore the loss of antioxidant capacity in D. melanogaster during the aging process without having any significant effect on growth (Table 3), and this applied to both male and female flies.
Aging is also a process of time-dependent decline in function, with cumulative damage in biomacromolecules and downregulation of cellular defense and damage repair. This is reminiscent of the expression pattern of Nrf2, a crucial stress regulator in the aging process, which is downregulated in aging organisms [18, 39], although the expression starts to decrease from the middle-age stage [23]. Defect in Nrf2 can result in a decline of stress resistance and hence a shortened lifespan [13, 14]. Thus, it is of great importance to show whether reversing the decline of the Nrf2 expression would slow down the aging process. It has been validated that upregulation of the Nrf2 expression, either by genetic manipulation or by pharmacological interference, can significantly ameliorate the extent of aging-related diseases and/or retard the aging process [25, 28, 29, 40–42]. In flies given SP2 supplement, significant upregulation in the expression of cncC and its downstream target genes (gclc, cat, and ho) occurred, which further promoted stress resistance and longevity (Figures 2 –5). Conversely, the stress-resistant effect of SP2 was dramatically repressed by inhibitors of the CncC/Nrf2/ARE signaling, such as trans-retinoic-acid and luteolin (Figure 6). Although our data have clearly demonstrated the involvement of CncC/Nrf2/ARE signaling in enhancing lifespan, other anti-stress-related signaling pathways should not be excluded, since these inhibitors might also act on other genes or their products. Nevertheless, the data did indicate that the Nrf2/ARE signaling pathway plays pivotal roles in antiaging, and targeting this pathway would be a promising approach for the screening of antiaging compounds [43, 44].
However, it must be stressed that the ectopic expression of Nrf2 in an organism can adversely affect its development [45]. For example, constitutive activation of the Nrf2 gene induces hyperkeratosis in the esophagus and forestomach, leading to postnatal lethality [46]. It is also fatal to overexpress Nrf2 during embryonic development, as Nrf2 also modulates decisions concerned with the fate of the cell [47, 48]. Therefore, spatiotemporal manipulation of Nrf2 should also be considered. Intriguingly, under normal condition, SP2 did not seem to influence the overall antioxidant capacity and CncC/Nrf2/ARE signaling pathway at the young-age stage of the flies (Figures 4 and 5), suggesting that it might have no adverse effects on postnatal development and growth. Though the underlying mechanism requires further study, this could be an important property of SP2 when carrying out future antiaging development.
An increasing number of studies suggest that polysaccharides from other sources such as mushroom, hemp seed, and okra can also upregulate the Nrf2/ARE signaling thus ameliorating oxidative damages, aging, diabetes, and other aging-related diseases [49–51]. In addition to S. fusiform fucoidan, the fucoidans from other algae have also been shown to enhance the Nrf2/ARE signaling, which can ameliorate liver injury and neurodegenerative diseases [52, 53]. However, the underlying mechanism by which polysaccharides upregulate the Nrf2/ARE signaling pathway remains unclear. It is possible that the bioactivities of polysaccharides may be intensively related to their molecular structures, which are determined by the molecular weight, chemical modification, monosaccharide composition, linkage types, and chain conformation of the polysaccharides. Fucoidans consist of a group of certain fucose-containing sulfated polysaccharides, which have been reported to possess many biological activities, including immunomodulatory, anti-inflammatory, antitumor, antioxidation, antivirus, and anticoagulant activities [54, 55]. However, the precise structure-activity relationships (SAR) for these polysaccharides remain largely undetermined. A fucoidan usually has a backbone of (1 → 3)-linked or (1 → 3)- and (1 → 4)-linked α-L-fucopyranosyl but also contains sulfated galactofucans, glucuronic acid, glucose, or xylose at different locations and to different extents [56]. Relatively high contents of xylose (12.10%), galactofucans (11.51%), and glucose substitutions (11.33%) were found in SP2 (Table 2), and this might be crucial for its antioxidant and antiaging functions. Thus, future study should focus on the relationship between monosaccharides and their bioactivity in oxidative aging.
Polysaccharides administered via diets would not be directly absorbed by the small intestine [24, 57, 58], and thus, their biological effects in the organisms are thought to be associated with their roles in modulating gut environment [59]. For this reason, we speculated that SP2 might modulate the gut environment of D. melanogaster, but confirmation of this aspect, including the elucidation of the relationship between microbiota composition and the structure of the SP2, will be a subject of further investigation.
5. Conclusion
We have optimized an effective method for screening the antiaging property of polysaccharides by integrating in vitro antioxidant screening and in vivo antioxidant resistance assay. Based on this method, SP2, a fucoidan extracted from the S. fusiforme heteropolysaccharide SFPS, was shown to have longevity-promoting activity. SP2 significantly activated the Nrf2/ARE signaling pathway, hence slowing down the decline in antioxidant defense capacity of D. melanogaster and increased its lifespan. This study has provided direct evidence of a longevity-promoting polysaccharide and revealed the worthiness of further research into SP2 as a health supplement.
Acknowledgments
This work was supported by the National Natural Science Foundation of China (31301148, 41876197, and 81872952), the Public Welfare Technology Application Research Plan Project of Zhejiang Science and Technology Department (2017C32103, LGN18C020004), and the talent training foundation of the Natural Science Foundation of Shandong (ZR2015PH014). The authors thank Dr. Alan K. Chang (Wenzhou University) for revising the language of the manuscript.
Contributor Information
Mingjiang Wu, Email: wmj@wzu.edu.cn.
Peichao Chen, Email: chenpeichao@wzu.edu.cn.
Data Availability
The data used to support the findings of this study are available from the corresponding authors upon request.
Conflicts of Interest
The authors declare that there are no conflicts of interest regarding the publication of this paper.
Supplementary Materials
Figure 1: the extract technological process of the polysaccharides from S. fusiforme. The fresh alga was dried at 50°C and ground to powder by an electric grinder. The alga powder was defatted by 95% ethanol, and the extracts were combined and concentrated. Thereafter, isolation and purification of the heteropolysaccharides were done by centrifugation, followed by washing with 95% ethanol. The nondialyzable phase was dried by lyophilization, and the harvested polysaccharides were named the SFPS. The SFPS was resolved and fractionated into SP1, SP2, and SP3.
References
- 1.Sohal R. S., Orr W. C. The redox stress hypothesis of aging. Free Radical Biology & Medicine. 2012;52(3):539–555. doi: 10.1016/j.freeradbiomed.2011.10.445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hernandez-Segura A., Nehme J., Demaria M. Hallmarks of cellular senescence. Trends in Cell Biology. 2018;28(6):436–453. doi: 10.1016/j.tcb.2018.02.001. [DOI] [PubMed] [Google Scholar]
- 3.López-Otín C., Blasco M. A., Partridge L., Serrano M., Kroemer G. The hallmarks of aging. Cell. 2013;153(6):1194–1217. doi: 10.1016/j.cell.2013.05.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Finkel T., Holbrook N. J. Oxidants, oxidative stress and the biology of ageing. Nature. 2000;408(6809):239–247. doi: 10.1038/35041687. [DOI] [PubMed] [Google Scholar]
- 5.Harman D. Free radical theory of aging: an update: increasing the functional life span. Annals of the New York Academy of Sciences. 2006;1067(1):10–21. doi: 10.1196/annals.1354.003. [DOI] [PubMed] [Google Scholar]
- 6.Back P., de Vos W. H., Depuydt G. G., Matthijssens F., Vanfleteren J. R., Braeckman B. P. Exploring real-time in vivo redox biology of developing and aging Caenorhabditis elegans. Free Radical Biology & Medicine. 2012;52(5):850–859. doi: 10.1016/j.freeradbiomed.2011.11.037. [DOI] [PubMed] [Google Scholar]
- 7.Lustgarten M., Muller F. L., Van Remmen H. An Objective Appraisal of the Free Radical Theory of Aging. In: Masoro E. J., Austad S. N., editors. Handbook of the Biology of Aging. Seventh Edition. San Diego: Academic Press; 2011. pp. 177–202. [DOI] [Google Scholar]
- 8.Yang C. S., Suh N., Kong A. N. T. Does vitamin E prevent or promote cancer? Cancer Prevention Research. 2012;5(5):701–705. doi: 10.1158/1940-6207.CAPR-12-0045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Marantz P. R., Kritchevsky D., Goldstein M. R., et al. Beta carotene, vitamin E, and lung cancer. The New England Journal of Medicine. 1994;331(9):611–614. doi: 10.1056/nejm199409013310912. author reply 613. [DOI] [PubMed] [Google Scholar]
- 10.Malhotra D., Portales-Casamar E., Singh A., et al. Global mapping of binding sites for Nrf2 identifies novel targets in cell survival response through ChIP-Seq profiling and network analysis. Nucleic Acids Research. 2010;38(17):5718–5734. doi: 10.1093/nar/gkq212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lewis K. N., Mele J., Hayes J. D., Buffenstein R. Nrf2, a guardian of healthspan and gatekeeper of species longevity. Integrative and Comparative Biology. 2010;50(5):829–843. doi: 10.1093/icb/icq034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sykiotis G. P., Bohmann D. Keap1/Nrf2 signaling regulates oxidative stress tolerance and lifespan in Drosophila. Developmental Cell. 2008;14(1):76–85. doi: 10.1016/j.devcel.2007.12.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bruns D. R., Drake J. C., Biela L. M., Peelor F. F., Miller B. F., Hamilton K. L. Nrf2 signaling and the slowed aging phenotype: evidence from long-lived models. Oxidative Medicine and Cellular Longevity. 2015;2015:15. doi: 10.1155/2015/732596.732596 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Miller C. J., Gounder S. S., Kannan S., et al. Disruption of Nrf2/ARE signaling impairs antioxidant mechanisms and promotes cell degradation pathways in aged skeletal muscle. Biochimica et Biophysica Acta. 2012;1822(6):1038–1050. doi: 10.1016/j.bbadis.2012.02.007. [DOI] [PubMed] [Google Scholar]
- 15.Lewis K. N., Wason E., Edrey Y. H., Kristan D. M., Nevo E., Buffenstein R. Regulation of Nrf2 signaling and longevity in naturally long-lived rodents. Proceedings of the National Academy of Sciences of the United States of America. 2015;112(12):3722–3727. doi: 10.1073/pnas.1417566112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Leiser S. F., Miller R. A. Nrf2 signaling, a mechanism for cellular stress resistance in long-lived mice. Molecular and Cellular Biology. 2010;30(3):871–884. doi: 10.1128/MCB.01145-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Zamponi E., Zamponi N., Coskun P., et al. Nrf2 stabilization prevents critical oxidative damage in Down syndrome cells. Aging Cell. 2018;17(5, article e12812) doi: 10.1111/acel.12812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Zhang H., Davies K. J. A., Forman H. J. Oxidative stress response and Nrf2 signaling in aging. Free Radical Biology and Medicine. 2015;88:314–336. doi: 10.1016/j.freeradbiomed.2015.05.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Liu L., Heinrich M., Myers S., Dworjanyn S. A. Towards a better understanding of medicinal uses of the brown seaweed Sargassum in traditional Chinese medicine: a phytochemical and pharmacological review. Journal of Ethnopharmacology. 2012;142(3):591–619. doi: 10.1016/j.jep.2012.05.046. [DOI] [PubMed] [Google Scholar]
- 20.Fan S., Zhang J., Nie W., et al. Antitumor effects of polysaccharide from Sargassum fusiforme against human hepatocellular carcinoma HepG2 cells. Food and Chemical Toxicology. 2017;102:53–62. doi: 10.1016/j.fct.2017.01.020. [DOI] [PubMed] [Google Scholar]
- 21.Chen H., Zhang L., Long X., et al. Sargassum fusiforme polysaccharides inhibit VEGF-A-related angiogenesis and proliferation of lung cancer in vitro and in vivo. Biomedicine & Pharmacotherapy. 2017;85:22–27. doi: 10.1016/j.biopha.2016.11.131. [DOI] [PubMed] [Google Scholar]
- 22.Chen L., Chen P., Liu J., et al. Sargassum Fusiforme Polysaccharide SFP-F2 Activates the NF-κB Signaling Pathway via CD14/IKK and P38 Axes in RAW264.7 Cells. Mar Drugs. 2018;16(8):p. 264. doi: 10.3390/md16080264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Chen P., He D., Zhang Y., et al. Sargassum fusiforme polysaccharides activate antioxidant defense by promoting Nrf2-dependent cytoprotection and ameliorate stress insult during aging. Food & Function. 2016;7(11):4576–4588. doi: 10.1039/C6FO00628K. [DOI] [PubMed] [Google Scholar]
- 24.Chen P., Yang S., Hu C., et al. Sargassum fusiforme polysaccharide rejuvenates the small intestine in mice through altering its physiology and gut microbiota composition. Current Molecular Medicine. 2017;17(5):350–358. doi: 10.2174/1566524018666171205115516. [DOI] [PubMed] [Google Scholar]
- 25.Pallauf K., Duckstein N., Hasler M., Klotz L. O., Rimbach G. Flavonoids as Putative Inducers of the Transcription Factors Nrf2, FoxO, and PPARγ . Oxidative Medicine and Cellular Longevity. 2017;2017:11. doi: 10.1155/2017/4397340.4397340 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Peng C., Zuo Y., Kwan K. M., et al. Blueberry extract prolongs lifespan of Drosophila melanogaster. Experimental Gerontology. 2012;47(2):170–178. doi: 10.1016/j.exger.2011.12.001. [DOI] [PubMed] [Google Scholar]
- 27.Zhang Z., Han S., Wang H., Wang T. Lutein extends the lifespan of Drosophila melanogaster. Archives of Gerontology and Geriatrics. 2014;58(1):153–159. doi: 10.1016/j.archger.2013.07.007. [DOI] [PubMed] [Google Scholar]
- 28.Wedel S., Manola M., Cavinato M., Trougakos I., Jansen-Dürr P. Targeting protein quality control mechanisms by natural products to promote healthy ageing. Molecules. 2018;23(5):p. 1219. doi: 10.3390/molecules23051219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lu M.-C., Ji J. A., Jiang Z. Y., You Q. D. The Keap1-Nrf2-ARE pathway as a potential preventive and therapeutic target: an update. Medicinal Research Reviews. 2016;36(5):924–963. doi: 10.1002/med.21396. [DOI] [PubMed] [Google Scholar]
- 30.Zhong W., Liu N., Xie Y., Zhao Y., Song X., Zhong W. Antioxidant and anti-aging activities of mycelial polysaccharides from Lepista sordida. International Journal of Biological Macromolecules. 2013;60:355–359. doi: 10.1016/j.ijbiomac.2013.06.018. [DOI] [PubMed] [Google Scholar]
- 31.Manayi A., Saeidnia S., Gohari A. R., Abdollahi M. Methods for the discovery of new anti-aging products--targeted approaches. Expert Opinion on Drug Discovery. 2014;9(4):383–405. doi: 10.1517/17460441.2014.885014. [DOI] [PubMed] [Google Scholar]
- 32.Michalakis K., Goulis D. G., Vazaiou A., Mintziori G., Polymeris A., Abrahamian-Michalakis A. Obesity in the ageing man. Metabolism. 2013;62(10):1341–1349. doi: 10.1016/j.metabol.2013.05.019. [DOI] [PubMed] [Google Scholar]
- 33.Guebre-Egziabher F., Alix P. M., Koppe L., et al. Ectopic lipid accumulation: a potential cause for metabolic disturbances and a contributor to the alteration of kidney function. Biochimie. 2013;95(11):1971–1979. doi: 10.1016/j.biochi.2013.07.017. [DOI] [PubMed] [Google Scholar]
- 34.del Valle L. G. Oxidative stress in aging: theoretical outcomes and clinical evidences in humans. Biomedicine & Aging Pathology. 2011;1(1):1–7. doi: 10.1016/j.biomag.2011.03.001. [DOI] [PubMed] [Google Scholar]
- 35.Wang X. J., Hayes J. D., Henderson C. J., Wolf C. R. Identification of retinoic acid as an inhibitor of transcription factor Nrf2 through activation of retinoic acid receptor alpha. Proceedings of the National Academy of Sciences of the United States of America. 2007;104(49):19589–19594. doi: 10.1073/pnas.0709483104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Huppke P., Weissbach S., Church J. A., et al. Activating de novo mutations in NFE2L2 encoding NRF2 cause a multisystem disorder. Nature Communications. 2017;8(1) doi: 10.1038/s41467-017-00932-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Navarro-Cruz A. R., Ramírez y Ayala R., Ochoa-Velasco C., et al. Effect of chronic administration of resveratrol on cognitive performance during aging process in rats. Oxidative Medicine and Cellular Longevity. 2017;2017:8. doi: 10.1155/2017/8510761.8510761 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Cartwright M., Tchkonia T., Kirkland J. Aging in adipocytes: potential impact of inherent, depot-specific mechanisms. Experimental Gerontology. 2007;42(6):463–471. doi: 10.1016/j.exger.2007.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Silva-Palacios A., Ostolga-Chavarría M., Zazueta C., Königsberg M. Nrf2: molecular and epigenetic regulation during aging. Ageing Research Reviews. 2018;47:31–40. doi: 10.1016/j.arr.2018.06.003. [DOI] [PubMed] [Google Scholar]
- 40.de la Vega M. R., Dodson M., Chapman E., Zhang D. D. NRF2-targeted therapeutics: new targets and modes of NRF2 regulation. Current Opinion in Toxicology. 2016;1:62–70. doi: 10.1016/j.cotox.2016.10.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Ren D., Villeneuve N. F., Jiang T., et al. Brusatol enhances the efficacy of chemotherapy by inhibiting the Nrf2-mediated defense mechanism. Proceedings of the National Academy of Sciences of the United States of America. 2011;108(4):1433–1438. doi: 10.1073/pnas.1014275108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Guo D., Yu K., Sun X. Y., Ouyang J. M. Structural characterization and repair mechanism of Gracilaria lemaneiformis sulfated polysaccharides of different molecular weights on damaged renal epithelial cells. Oxidative Medicine and Cellular Longevity. 2018;2018:15. doi: 10.1155/2018/7410389.7410389 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Pomatto L. C. D., Davies K. J. A. Adaptive homeostasis and the free radical theory of ageing. Free Radical Biology & Medicine. 2018;124:420–430. doi: 10.1016/j.freeradbiomed.2018.06.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Liu X. F., Hao J. L., Xie T., et al. Nrf2 as a target for prevention of age-related and diabetic cataracts by against oxidative stress. Aging Cell. 2017;16(5):934–942. doi: 10.1111/acel.12645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Loboda A., Damulewicz M., Pyza E., Jozkowicz A., Dulak J. Role of Nrf2/HO-1 system in development, oxidative stress response and diseases: an evolutionarily conserved mechanism. Cellular and Molecular Life Sciences. 2016;73(17):3221–3247. doi: 10.1007/s00018-016-2223-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Wakabayashi N., Itoh K., Wakabayashi J., et al. Keap1-null mutation leads to postnatal lethality due to constitutive Nrf2 activation. Nature Genetics. 2003;35(3):238–245. doi: 10.1038/ng1248. [DOI] [PubMed] [Google Scholar]
- 47.Jang J., Wang Y., Kim H. S., Lalli M. A., Kosik K. S. Nrf2, a regulator of the proteasome, controls self-renewal and pluripotency in human embryonic stem cells. Stem Cells. 2014;32(10):2616–2625. doi: 10.1002/stem.1764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Yoon D. S., Choi Y., Lee J. W. Cellular localization of NRF2 determines the self-renewal and osteogenic differentiation potential of human MSCs via the P53-SIRT1 axis. Cell Death & Disease. 2016;7(2):p. e2093. doi: 10.1038/cddis.2016.3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Wen Z. S., Xue R., du M., et al. Hemp seed polysaccharides protect intestinal epithelial cells from hydrogen peroxide-induced oxidative stress. International Journal of Biological Macromolecules. 2019;135:203–211. doi: 10.1016/j.ijbiomac.2019.05.082. [DOI] [PubMed] [Google Scholar]
- 50.Wen Z. S., Tang Z., Ma L., et al. Protective effect of low molecular weight seleno-aminopolysaccharide on the intestinal mucosal oxidative damage. Marine Drugs. 2019;17(1):p. 64. doi: 10.3390/md17010064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Liao Z., Zhang J., Liu B., et al. Polysaccharide from okra (Abelmoschus esculentus (L.) Moench) improves antioxidant capacity via PI3K/AKT pathways and Nrf2 translocation in a type 2 diabetes model. Molecules. 2019;24(10):p. 1906. doi: 10.3390/molecules24101906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Zhang L., Hao J., Zheng Y., et al. Fucoidan protects dopaminergic neurons by enhancing the mitochondrial function in a rotenone-induced rat model of Parkinson’s disease. Aging and Disease. 2018;9(4):590–604. doi: 10.14336/AD.2017.0831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Wang Y. Q., Wei J. G., Tu M. J., Gu J. G., Zhang W. Fucoidan alleviates acetaminophen-induced hepatotoxicity via oxidative stress inhibition and Nrf2 translocation. International Journal of Molecular Sciences. 2018;19(12):p. 4050. doi: 10.3390/ijms19124050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Fitton J., Stringer D., Karpiniec S. Therapies from fucoidan: an update. Marine Drugs. 2015;13(9):5920–5946. doi: 10.3390/md13095920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Ma X. T., Sun X. Y., Yu K., Gui B. S., Gui Q., Ouyang J. M. Effect of content of sulfate groups in seaweed polysaccharides on antioxidant activity and repair effect of subcellular organelles in injured HK-2 cells. Oxidative Medicine and Cellular Longevity. 2017;2017:13. doi: 10.1155/2017/2542950.2542950 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Ale M. T., Mikkelsen J. D., Meyer A. S. Important determinants for fucoidan bioactivity: a critical review of structure-function relations and extraction methods for fucose-containing sulfated polysaccharides from brown seaweeds. Marine Drugs. 2011;9(10):2106–2130. doi: 10.3390/md9102106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Shang Q., Shan X., Cai C., Hao J., Li G., Yu G. Dietary fucoidan modulates the gut microbiota in mice by increasing the abundance of Lactobacillus and Ruminococcaceae. Food & Function. 2016;7(7):3224–3232. doi: 10.1039/C6FO00309E. [DOI] [PubMed] [Google Scholar]
- 58.Huang X., Nie S., Xie M. Interaction between gut immunity and polysaccharides. Critical Reviews in Food Science and Nutrition. 2017;57(14):2943–2955. doi: 10.1080/10408398.2015.1079165. [DOI] [PubMed] [Google Scholar]
- 59.Shang Q., Song G., Zhang M., et al. Dietary fucoidan improves metabolic syndrome in association with increased Akkermansia population in the gut microbiota of high-fat diet-fed mice. Journal of Functional Foods. 2017;28:138–146. doi: 10.1016/j.jff.2016.11.002. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Figure 1: the extract technological process of the polysaccharides from S. fusiforme. The fresh alga was dried at 50°C and ground to powder by an electric grinder. The alga powder was defatted by 95% ethanol, and the extracts were combined and concentrated. Thereafter, isolation and purification of the heteropolysaccharides were done by centrifugation, followed by washing with 95% ethanol. The nondialyzable phase was dried by lyophilization, and the harvested polysaccharides were named the SFPS. The SFPS was resolved and fractionated into SP1, SP2, and SP3.
Data Availability Statement
The data used to support the findings of this study are available from the corresponding authors upon request.