Abstract
Context
It is a challenge to determine the origin of Cushing syndrome (CS), especially in patients with low-normal adrenocorticotropic hormone (ACTH) concentrations.
Objective
To evaluate the reliability of the corticotropin-releasing hormone (CRH) stimulation test in patients with CS whose origin of disease was not clearly identified using ACTH values, the high-dose dexamethasone suppression test (HDDST), and imaging in a single tertiary referral center.
Design and Methods
Twenty-one patients with CS who were admitted to the endocrinology-metabolism clinic between 2004 and 2016 whose ACTH concentrations were 5-20 pg/mL and needed CRH stimulation test were retrospectively assessed.
Results
Nine out of 21 patients were diagnosed as having Cushing’s disease (CD) and 12/21 had adrenal CS. The CRH stimulation test had a sensitivity and specificity of 100% and 8%, and positive and negative predictive values of 100% and 45% according to the current diagnostic criteria, respectively. An increase in ACTH ≥115% at 15 minutes and cortisol ≥86% at 60 minutes after CRH were associated with the highest likelihood ratio. The sensitivity and specificity of ACTH was 67% and 83% (AUC=0.75±0.12, 95% CI: [0.5-0.9]; p=0.03), and for cortisol it was 75% and 78% (AUC=0.71±0.15, 95% CI: [0.5-0.9]; p=0.03). Cortisol suppression of more than 64% from basal level in the HDDST suggested CD with the highest likelihood ratio. When these cut-off values were used together, both tests were negative in the patients with CD.
Conclusion
The CRH stimulation test has low specificity to localize CS in patients with ACTH concentrations of 5-20 pg/mL according to the current diagnostic criteria. Different diagnostic criteria may be used in the CRH stimulation test and also in the HDDST in this group of patients.
Keywords: ACTH, CRH stimulation test, Cushing’s syndrome
INTRODUCTION
Cushing’s syndrome (CS) is a rare disorder characterized by excessive cortisol secretion from the adrenal glands (1). Once hypercortisolism is definite, it can be problematic to localize the origin of the hypersecretion. Imaging modalities may not always provide a precise localization; moreover, an adrenal or pituitary incidentaloma discovered using diagnostic imaging modalities may further complicate efforts for focus-identification (2, 3).
Adrenocorticotropic hormone (ACTH) concentrations can be a good predictor of the localization of disease; concentrations higher than 15-20 pg/mL suggest ACTH-dependent CS, whereas concentrations less than 5 pg/mL suggest adrenal glands as the origin of the disease (4-6). However, it is difficult to localize patients with ACTH concentrations between these values. Both ACTH-dependent and independent causes of CS can be seen in this gray zone of 5-20 pg/mL.
The corticotropin-releasing hormone (CRH) stimulation test is a second-line non-invasive dynamic test for precise localization of disease in which at least a 35-50% increase of ACTH and 20% increase of cortisol concentrations from baseline indicate the pituitary gland as the origin of hypercortisolemia (7-9). In the literature, publications concerning the CRH stimulation test have been mostly conducted in groups of patients with definite ACTH-dependent CS (8-14). However, it is also suggested that the CRH stimulation test can be used in patients whose origin of disease is uncertain (4-6, 15). There are few studies about the CRH test in patients with undetermined origin of hypercortisolism and the interpretation of the test in these studies was made according to the data of ACTH-dependent CS (7, 16). In the literature, there are scarce data on the diagnostic accuracy of the CRH test in this patient group and there is still no consensus on this topic.
In this study, we aimed to retrospectively evaluate the reliability of the CRH stimulation test in patients with CS whose origin of disease was not clearly identified using ACTH concentrations, the high-dose dexamethasone suppression test (HDDST), and imaging techniques in a single tertiary referral center.
MATERIALS AND METHODS
Subjects and study design
The study was approved by the local ethics committee of Cerrahpasa Medical Faculty, Istanbul University – Cerrahpasa, in accordance with the ethical standards of the 1964 Helsinki Declaration.
The medical records of 430 patients with CS who were followed at Cerrahpasa Medical Faculty, Istanbul University - Cerrahpasa, Department of Endocrinology and Metabolism between 2004 and 2016 were retrospectively reviewed. Forty-five of the 430 patients needed to have the CRH stimulation test. Twenty-one of these 45 patients who had ACTH concentrations between 5 and 20 pg/mL and had definitive diagnosis after surgery according to pathology results were enrolled in the study. The remaining 24 patients were excluded from the study; five patients were lost to follow-up, seven patients did not undergo surgery, and 12 patients did not have biochemical remission after surgery, thus the localization could not be determined.
The diagnosis of CS was established according to the Endocrine Society clinical practice guideline on the diagnosis of CS (17). Patients with 24-hour urinary free cortisol (UFC) concentrations within normal levels and serum cortisol concentrations below 1.8 μg/dL during low-dose 1 mg the LDDST were identified as having biochemical remission after surgery (17).
The demographic characteristics of the patients, the imaging techniques used to determine the localization of disease, basal ACTH, basal cortisol, UFC concentrations, 1 mg dexamethasone suppression test (DST), HDDST results, and the cortisol-ACTH concentrations found in the CRH stimulation test were recorded from the medical files of the patients. The pituitary was accepted to be the origin of hypercortisolism if there was a 50% increase in ACTH concentrations or a 20% increase in cortisol concentrations from baseline in any measurements performed during the CRH stimulation test, and if there was a ≥50% suppression of serum cortisol after HDDST (8, 9, 17).
High-dose (8 mg) DSTs were performed at 23:00 PM, and serum cortisol concentrations were measured at 08:00 AM the next morning.
The CRH test was performed in the morning after an overnight fast. A cannula was inserted into an antecubital vein, 30 min before the beginning of the test. Blood samples for serum cortisol and plasma ACTH were drawn at -15, 0, 15, 30, 45, 60, 90, 120 minutes after the injection of 100 mg of ovine CRH. The origin of hypercortisolism was interpreted as the pituitary gland if there was a 50% increase in ACTH concentrations and a 20% increase in cortisol concentrations at any measurements performed during the test from baseline.
Plasma cortisol concentrations were analyzed using a solid phase competitive chemiluminescent enzyme immunoassay (normal range = 5-25 μg/dL). ACTH concentrations were analyzed using a solid phase two-site sequential chemiluminescent immunometric assay (ICMA) (normal range = 5-46 pg/mL). Basal ACTH and cortisol values were used to calculate the percentage increases (%B) from baseline in all measurements. Urinary free cortisol (UFC) (normal range = 36-137 μg/24 h) concentrations were measured using an RIA.
Statistical analyses
SPSS Version 21.0 (Armonk, NY: IBM Corp) was used for statistical analyses. For descriptive statistics, continuous data are presented as mean, standard error, median, interquartile range (IQR), and categorical data using numbers and percentages. In statistical comparisons, the Kolmogorov-Smirnov and Shapiro-Wilk tests were used to assess the normality of the distribution of data. The Chi-square test, t-test, and Mann-Whitney U test were used for comparisons between independent groups, and the Wilcoxon test was used for comparisons between dependent groups.
The optimal cut-off level and associated sensitivity and specificity were evaluated using receiver operator characteristics (ROC) curve analyses based on the highest positive likelihood ratio (+LR), positive predictive value (PPV) and negative predictive value (NPV) using MedCalc for Windows, version 17.4.4 (MedCalc Software, Ostend, Belgium). P values less than 0.05 with corresponding confidence intervals of 95% were considered as significant.
RESULTS
Twenty-one patients with CS were included in the study. The diagnostic dates of the study patients were between 2004 and 2016. Nine patients were diagnosed as having CD and all of these patients underwent trans-sphenoidal adenomectomy and their histologic findings were consistent with ACTH-producing adenoma. Twelve patients were diagnosed as having adrenal adenoma-related CS and each achieved biochemical remission following surgery. In our study, 4 out of 12 patients with CS of adrenal origin and 5 out of 9 patients with CD had pre-existing adrenal incidentaloma.
The demographic characteristics and biochemical parameters are shown in Table 1. The median basal ACTH concentration was 8.7 (range: 5-21.4) pg/mL. Baseline ACTH concentrations were higher in patients with CD than in those with CS of adrenal origin; however, this difference did not reach statistical significance (p = 0.508). A pituitary adenoma was detected with pituitary MRI in 4 (44.4%) patients in the CD group, adrenal adenoma was detected in 11 patients, and bilateral macronodular hyperplasia was detected in one patient in the adrenal CS group. Tests for aberrant adrenal expression of various hormone receptors were performed and a patient with ACTH-independent macronodular adrenal hyperplasia was excluded; the patient underwent bilateral adrenalectomy. Two (16.6%) patients in the adrenal Cushing group had a pituitary adenoma and 5 (55.5%) patients in the CD group had adrenal adenoma concurrently. CRH stimulation test results were consistent with CD in all patients with CD (9/9), whereas the test indicated the pituitary as the origin of hypercortisolemia in 11 of 12 patients with adrenal CS (11/12).
Table 1.
Demographic characteristics and laboratory data of patients with adrenal Cushing’s syndrome and Cushing’s disease
| Adrenal CS (n=12) | CD (n=9) | p value | |
| Age (years) | 50 (38-70) | 51 (33-64) | 0.697 |
| Sex (F/M) | 9 / 3 | 7 / 2 | 0.606 |
| 24-hour UFC (μg/24 h) | 147.5 (99.6-225.0) | 142.5 (103.0-391.5) | 0.702 |
| ACTH (pg/mL) | 7.4 (5.0-21.4) | 11.4 (5.2-17.9) | 0.508 |
| Cortisol after 1 mg DST (μg/dL) | 3.4 (2.1-7.2) | 3.6 (2.12-8.1) | 0.776 |
| Cortisol suppression after HDDST (%) | 55.5 (23.5-81.4) | 83.9 (67.5-91.7) | 0.006 |
| Patients with pituitary adenoma n (%) | 2 (16.6) | 4 (44.4) | - |
| Patients with adrenal adenoma n (%) | 11 (91.7) | 5 (55.6) | - |
| CRH test (+)* | 11 (91.7) | 9 (100) | 0.571 |
Values are expressed as median (IQR: interquartile range) or number (%). ACTH: Adrenocorticotropic hormone, DST: dexamethasone suppression test, UFC: urinary free. Cortisol, CRH: Corticotropin-releasing hormone, HDDST: High-dose dexamethasone suppression test. *CRH test (+), 50 % increase at the level of ACTH or 20 % increase at the level of cortisol after CRH injection.
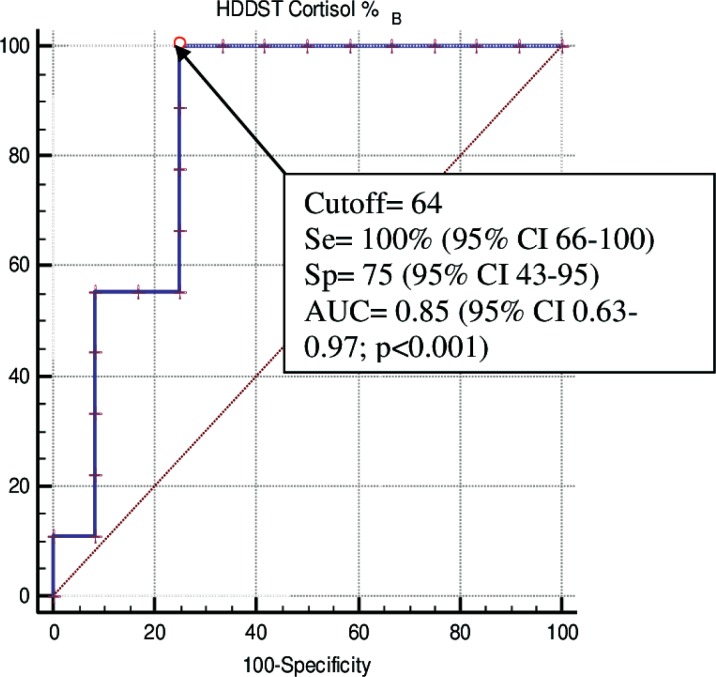
The percentage suppression of serum cortisol after the HDDST is shown in Table 1. Inter- group comparisons revealed statistically significant differences between the suppression level in the CD group (median=83.9%, range 67.5-91.7) and adrenal CS group (median=55.5%, range, 23.5-81.4) (p=0.006) (Table 1). The ROC curve analysis of the HDDST with recently determined criteria is shown in Figure 1. Suppression of basal cortisol concentrations more than ≥64% from baseline cortisol after the administration of 8 mg of dexamethasone was associated with the highest likelihood ratio and the sensitivity and specificity of the test were found as 100% and 75%, respectively (AUC=0.85±0.11, 95% CI: [0.63-0.97]; p<0.001) (Fig. 1).
Figure 1.
ROC curve analysis of high-dose dexamethasone test (HDDST) with recently determined criteria.
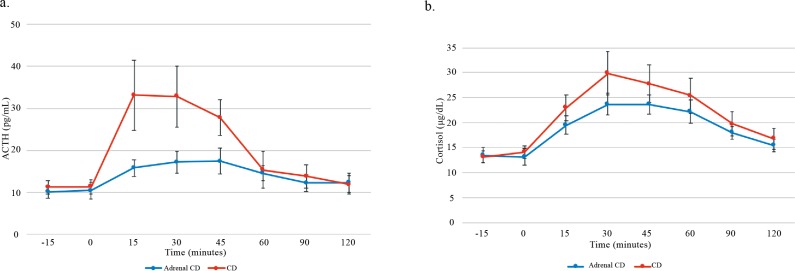
ACTH and cortisol concentrations during the CRH test are shown in Figure 2. In patients with CD, the percentage increment in ACTH and cortisol concentrations after IV CRH administration was found to be higher than in patients in the adrenal group, but this difference did not reach statistical significance (p=0.055 and p=0.15, respectively).
Figure 2.
ACTH (a) and Cortisol (b) concentrations following CRH administration in patients with adrenal CS and CD.
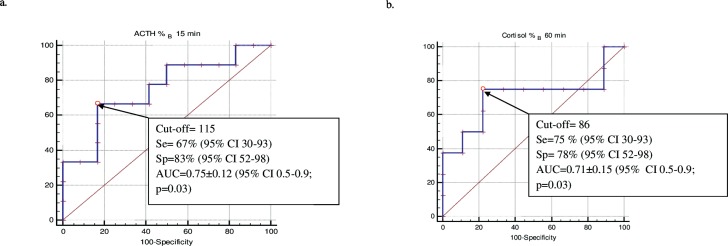
In the assessment of the CRH test results on the basis of the current assessment criteria, the test had a specificity of 8%, a sensitivity of 100%, and a positive and negative predictive value 100% and 45%, respectively. The accuracy of the test was 48% in patients with ACTH concentrations of 5-20 pg/mL (Table 2). ROC curve analysis of ACTH (a) and cortisol (b) concentrations in the CRH test with recently determined criteria are shown in Figure 3. More than a 115% increase in ACTH concentrations 15 minutes after the injection of CRH was associated with the highest likelihood ratio and the sensitivity and specificity of the test were found as 67% and 83%, respectively (AUC=0.75±0.12, 95% CI: [0.5-0.9]; p=0.03) (Fig. 3a). More than an 86% increase in cortisol concentrations at minute 60 after the injection of CRH was associated with the highest likelihood ratio and the sensitivity and specificity of the test were found as 75% and 78%, respectively (AUC=0.71±0.15, 95% CI: [0.5-0.9]; p=0.03) (Fig. 3b).
Figure 3.
ROC curve analysis of ACTH (a) and cortisol (b) concentrations of corticotrophin- releasing hormone (CRH) test with recently determined criteria.
Table 2.
Receiver operating characteristics curves of different test variables
| Test variable | Cut-off value (%) | ROC AUC (SEM) | Positive LR | Sensitivity (%) | Specificity (%) | PPV (%) | NPV (%) |
| ACTH 15 min | >115 | 0.75 (0.12) | 4.00 | 67 | 83 | 75 | 77 |
| ACTH 30 min | >47 | 0.75 (0.11) | 1.71 | 100 | 42 | 56 | 100 |
| ACTH 45 min | >161 | 0.68 (0.13) | 3.44 | 63 | 82 | 71 | 75 |
| ACTH 60 min | >22 | 0.54 (0.17) | 1.71 | 57 | 67 | 57 | 67 |
| Cortisol 15 min | >26 | 0.66 (0.13) | 1.78 | 89 | 50 | 57 | 86 |
| Cortisol 30 min | >60 | 0.69 (0.12) | 1.87 | 78 | 58 | 58 | 78 |
| Cortisol 45 min | >82 | 0.69 (0.14) | 2.25 | 75 | 67 | 60 | 80 |
| Cortisol 60 min | >86 | 0.71 (0.15) | 3.37 | 75 | 78 | 75 | 78 |
| CRH original* | - | - | 1.09 | 100 | 8 | 100 | 45 |
ACTH: Adrenocorticotropic hormone, CRH: Corticotropin-releasing hormone. *CRH test (+), 50% increase at the level of ACTH or 20% increase at the level of cortisol after CRH injection.
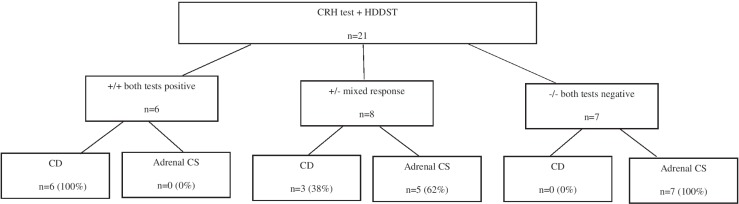
When the strongest recently determined criteria of the CRH test (ACTH rise of ≥115% increase at 15 min, and cortisol rise ≥86% increase at 60 min after the injection of ovine CRH) and the HDDST (cortisol suppression of ≥64% after 8 mg dexamethasone) were concurrently applied in the study population, 66% of the patients were correctly diagnosed as having CD, and both tests were negative in the patients with CD, whereas 58% of the patients with adrenal CS had negative results when both tests were applied together (Fig. 4).
Figure 4.
The comparison of the diagnosis of the study population and the results of CRH and HDSST combinations performed according to newly determined criteria (ACTH increase of ≥115% at 15 min, and cortisol increase ≥86% at 60 min after the injection of ovine CRH and cortisol suppression of ≥64% after 8 mg dexamethasone).
A cut-off value concerning ACTH concentrations at baseline was added to the diagnostic criteria in order to increase the reliability of the CRH test, and the most adequate cut-off value was found as 5.6 pg/mL (sensitivity=89%, specificity=42%) based on the ROC analysis for the group of patients with CD and patients with adrenal CS. However, the addition of a cut-off value did not lead to a statistically significant difference (AUC=0.59±0.13, 95% CI: [0.4-0.8]; p=0.482).
DISCUSSION
In this study, we found that the CRH stimulation test has low specificity in determining the origin of hypercortisolism in patients with ACTH concentrations between 5 and 20 pg/mL according to the current diagnostic criteria. We found that a 115% increase of ACTH concentrations at 15 minutes and a 86% increase of cortisol concentrations at 60 minutes were the highest likelihood ratio of the CRH stimulation test. Additionally, if cortisol suppression in the HDDST with a cut-off value of more than 64% from baseline was also added to CRH stimulation test, the specificity increased.
Sometimes, it can be a challenge to find the origin of hypercortisolism in CS, especially in patients with moderately increased ACTH concentrations. There are a few non-invasive tests for this purpose; however, each has limited diagnostic value. This situation may lead to unnecessary imaging, invasive procedures, and unnecessary surgeries, which delays the treatment and is also not cost-effective.
ACTH is a good marker for localization in CS; however, the diagnostic accuracy decreases at concentrations between 5 and 20 pg/mL (4,5). In this respect, the transportation and storage of blood has great importance. Plasma should be separated from whole blood rapidly and stored as frozen to prevent ACTH from degrading by plasma proteases (18). Moreover, the assay used for ACTH measurement is important in the evaluation of disease. Pecori et al. showed that ACTH assays had high variability and may give incorrect results, especially in patients with suppressed ACTH secretion (19). In various studies, it was suggested to measure ACTH concentrations twice on separate days to improve sensitivity (4,6,8). We calculated ACTH concentrations at least twice and the mean of these measurements was considered as the basal ACTH concentration in our study.
The method used to measure ACTH concentrations is also important for the accuracy of the measurements. The development of two-site immunometric assays [immunoradiometric (IRMA) or chemiluminescent immunometric (ICMA) assays] has improved the specificity and ability to measure low-normal ACTH concentrations (6,18). Gibson et al. found that two-site immunometric assays were more sensitive and had a wider working range than RIAs (20). In a study by Invitti et al. IRMA detected ACTH concentrations below the lower limit of normal better than RIA (7). In our study, the diagnostic dates of the study patients were between 2004 and 2016 and we used the ICMA method for analyzing ACTH concentrations for all study patients.
Some dynamic tests were used when ACTH concentrations fail to definitely localize the origin of hypercortisolism, one of which is the CRH stimulation test. In the literature, the CRH stimulation test is generally used to differentiate ACTH-dependent CS etiologies; however, various articles have demonstrated the usefulness of this test in differentiating ACTH-dependent and independent causes (4-7,15,16). One of the most comprehensive studies on this subject was by Invitti et al. In that study, 158 patients with CD, 13 patients with ectopic CS, and 41 patients with adrenal CS with pathologically-proven disease were screened using the CRH stimulation test. All patients with CD were correctly identified with this test in this group of patients, which had a wide range of ACTH concentrations (7). In another study by al-Saadi et al., 26 patients with CD and 13 patients with adrenal CS were assessed using the CRH stimulation test and none of 13 patients with CS of adrenal origin responded to the test (16). However, in this study, patients with adrenal CS had basal ACTH concentrations <5 pg/mL and the ACTH concentrations of patients with CD were >20 pg/mL. Despite these studies, there are no clear data about patients with ACTH concentrations of 5-20 pg/mL, thus we decided to design our study in order to answer this question.
The definitive diagnostic criteria for the CRH stimulation test are still unclear in the literature. Reimondo et al. reported that an ACTH increase of at least 50% gave the best diagnostic accuracy with an 86% sensitivity and 90% specificity for CD (9). Niaman et al. demonstrated that a 35% increase in ACTH and a 20% increase in cortisol were associated with 100% sensitivity and 93% specificity for ACTH, and 91% sensitivity and 88% specificity for cortisol (8). However, these studies were conducted in patients with ACTH-dependent CS.
When a 50% increase in plasma ACTH concentrations or a 20% increase in plasma cortisol concentrations from baseline was accepted as CD, we found the sensitivity of this test as excellent (100%), whereas the specificity was very low (8%) in our patients with ACTH concentrations between 5-20 pg/mL. Moreover, the sensitivity and specificity of the test for ACTH were found as 67% and 83%, and the sensitivity and specificity for cortisol were found as 75% and 78%, respectively.
In our study, the time to reach the peak concentrations of ACTH and cortisol following intravenous administration of CRH was consistent with literature (7, 8, 10, 14). However, we demonstrated that a much higher increase (115%) of ACTH concentrations was needed for the best predictor of CD. Furthermore, we showed that an 86% increase of cortisol at 60 minutes was also a good predictor of CD.
One of the remarkable results of our study was the false-positive response to the CRH stimulation test in patients with adrenal CS. It has been shown in the literature that the ACTH response to CRH stimulation is blunted in adrenal CS due to negative feedback, and in CD, the ACTH response is exaggerated if tumor cells contain CRH receptors (10, 21, 22). However, various studies in healthy subjects have shown that CRH stimulation may increase basal ACTH concentrations by up to 35 to 900% (23-26). The exaggerated response found in our study in the adrenal group may also be related to this issue.
Another non-invasive localization test used in the diagnosis of CD, the HDDST, has sensitivity of 65-100% and specificity of 60-100% (15, 16, 27). The test has little diagnostic use in clinical practice, regardless of the test protocol and the cut-off value used for cortisol suppression. Moreover, previous studies have reported the poor diagnostic accuracy of the HDDST in discriminating adrenal CS from CD; the routine use of the HDDST alone in the differential diagnosis of CS is not recommended because suppression of cortisol with the HDDST can also be seen in adrenal CS (20, 27, 28). In our study, in accordance with literature, some patients from the adrenal CS group had suppression higher than 50% from their basal cortisol concentrations. When the strongest criteria of the CRH test and the HDDST were concurrently applied to the study population, 66% of patients were correctly diagnosed as having CD, both tests were negative in the patients with CD, whereas 58% of the patients with adrenal CS had negative results when both tests were applied together. According to the results from our study, although the HDDST had contradictory results, it can be helpful in excluding the diagnosis of adrenal CS when both tests are positive.
Another consideration is the differentiation of subclinical CS (SCS) from CS because the diagnosis may be challenging in patients who present with mild CS symptoms and have concurrent adrenal incidentaloma. In a large series of adrenal incidentalomas, hormonal evaluation in patients with SCS whose ACTH concentrations were mostly low revealed that a sufficient plasma ACTH response to CRH was observed in 45% of patients (2). In a study conducted by Giordano et al., ACTH concentrations did not differ significantly between patients with adrenal incidentaloma and those with SC (29). Thus, in patients with SCS, clinical presentation and blood chemistry may be similar to those with patients with mild CS. Therefore, a cortisol cut-off level of >5 mcg/dL (>138 nmol/L) for 1-mg DST may be reasonable in patients with adrenal incidentaloma to detect glucocorticoid secretory autonomy (30). Although we did not have any patients with SCS, 4/12 patients with adrenal CS and 5/9 patients with CD had pre-existing adrenal incidentaloma. In patients with adrenal incidentalomas who have mild CS symptoms, the wait-and-see strategy may be appropriate instead of referring to a quick surgery.
The study has some limitations. First, the study is retrospectively designed and the number of patients was small. Cushing’s Syndrome is a relatively rare disease so it was not easy to increase the number of the patients. Moreover, if confirmed in larger series of patients, the data presented here could potentially improve the differential diagnosis of CS, and thus the management of these patients. Secondly, we conducted this study in patients who were followed during a wide period of time, between 2004 and 2016. During this long period, the literature data changed and recent studies suggest raising the cut-off value of ACTH from 5 to 10 pg/mL in the differentiation of dependent from independent CS, and the CRH test may be performed in patients with undetermined localization of CS with ACTH concentrations above 10 pg/mL (31). In accordance with the literature, we also suggest the cut-off point of 10 pg/mL to be used for the CRH test in order to differentiate ACTH-dependent and independent CS.
In conclusion, it can still be challenging to find the origin of CS in patients with ACTH concentrations of 5-20 pg/mL. The CRH stimulation test may inadequately localize the origin of CS according to the current diagnostic criteria. It may be useful to assess both the HDDST and CRH tests with renovated cut-offs in these patients. Larger and prospectively designed studies are needed to develop definitive diagnostic criteria for the CRH test and also for the HDDST in patients with CS whose disease origin has not been clearly identified.
Conflict of interest
The authors declare that they have no conflict of interest.
Acknowledgement
This research did not receive any specific grant from any funding agencies in the public, commercial, or non-profit sector. We thank Mr. David F. Chapman for his help regarding English language editing.
References
- 1.Bertagna X, Guignat L, Groussin L, Bertherat J. Cushing’s disease. Best Pract Res Clin Endocrinol Metab. 2009;23(5):607–623. doi: 10.1016/j.beem.2009.06.001. [DOI] [PubMed] [Google Scholar]
- 2.Mantero F, Terzolo M, Arnaldi G, Osella G, Masini AM, Alì A, Giovagnetti M, Opocher G, Angeli A, Study Group on Adrenal Tumors of the Italian Society of Endocrinology A survey on adrenal incidentaloma in Italy. J Clin Endocrinol Metab. 2000;85(2):637–644. doi: 10.1210/jcem.85.2.6372. [DOI] [PubMed] [Google Scholar]
- 3.Lindsay JR, Nieman LK. Differential diagnosis and imaging in Cushing’s syndrome. Endocrinol Metab Clin North Am. 2005;34(2):403–421. doi: 10.1016/j.ecl.2005.01.009. [DOI] [PubMed] [Google Scholar]
- 4.Juszczak A, Grossman A. The management of Cushing’s disease - from investigation to treatment. Endokrynol Pol. 2013;64(2):166–174. [PubMed] [Google Scholar]
- 5.Newell-Price J, Bertagna X, Grossman AB, Nieman LK. Cushing’s syndrome. Lancet. 2006;367(9522):1605–1617. doi: 10.1016/S0140-6736(06)68699-6. [DOI] [PubMed] [Google Scholar]
- 6.Boscaro M, Arnaldi G. Approach to the patient with possible Cushing’s syndrome. J Clin Endocrinol Metab. 2009;94(9):3121–3131. doi: 10.1210/jc.2009-0612. [DOI] [PubMed] [Google Scholar]
- 7.Invitti C, Pecori Giraldi F, de Martin M, Cavagnini F, Study Group of the Italian Society of Endocrinology on the Pathophysiology of the Hypothalamic-Pituitary-Adrenal Axis Diagnosis and management of Cushing’s syndrome: results of an Italian multicentre study. J Clin Endocrinol Metab. 1999;84(2):440–448. doi: 10.1210/jcem.84.2.5465. [DOI] [PubMed] [Google Scholar]
- 8.Nieman LK, Oldfield EH, Wesley R, Chrousos GP, Loriaux DL, Cutler GB., Jr A simplified morning ovine corticotropin-releasing hormone stimulation test for the differential diagnosis of adrenocorticotropin-dependent Cushing’s syndrome. J Clin Endocrinol Metab. 1993;77(5):1308–1312. doi: 10.1210/jcem.77.5.8077325. [DOI] [PubMed] [Google Scholar]
- 9.Reimondo G, Paccotti P, Minetto M, Termine A, Stura G, Bergui M, Angeli A, Terzolo M. The corticotrophin-releasing hormone test is the most reliable noninvasive method to differentiate pituitary from ectopic ACTH secretion in Cushing’s syndrome. Clin Endocrinol (Oxf) 2003;58(6):718–724. doi: 10.1046/j.1365-2265.2003.01776.x. [DOI] [PubMed] [Google Scholar]
- 10.Newell-Price J, Morris DG, Drake WM, Korbonits M, Monson JP, Besser GM, Grossman AB. Optimal response criteria for the human CRH test in the differential diagnosis of ACTH-dependent Cushing’s syndrome. J Clin Endocrinol Metab. 2002;87(4):1640–1645. doi: 10.1210/jcem.87.4.8357. [DOI] [PubMed] [Google Scholar]
- 11.Orth DN, De Bold CR, De Cherney GS, Jackson RV, Alexander AN, Rivier J, Rivier C, Spiess J, Vale W. Pituitary microadenomas causing Cushing’s disease respond to corticotropin-releasing factor. J Clın Endocrinol Metab. 1982;55(5):1017–1019. doi: 10.1210/jcem-55-5-1017. [DOI] [PubMed] [Google Scholar]
- 12.Pecori Giraldi F, Invitti C, Cavagnini F, Study Group of the Italian Society of Endocrinology on the Pathophysiology of the Hypothalamic-pituitary-adrenal axis The corticotropin-releasing hormone test in the diagnosis of ACTH-dependent Cushing’s syndrome: a reappraisal. Clin Endocrinol (Oxf) 2001;54(5):601–607. doi: 10.1046/j.1365-2265.2001.01258.x. [DOI] [PubMed] [Google Scholar]
- 13.Barbot M, Trementino L, Zilio M, Ceccato F, Albiger N, Daniele A, Frigo AC, Mardari R, Rolma G, Boscaro M, Arnaldi G, Scaroni C. Second-line tests in the differential diagnosis of ACTH-dependent Cushing’s syndrome. Pituitary. 2016;19(5):488–495. doi: 10.1007/s11102-016-0729-y. [DOI] [PubMed] [Google Scholar]
- 14.Ritzel K, Beuschlein F, Berr C, Osswald A, Reisch N, Bidlingmaier M, Schneider H, Honegger J, Geyer LL, Schopohl J, Reincke M. ACTH after 15 min distinguishes between Cushing’s disease and ectopic Cushing’s syndrome: a proposal for a short and simple CRH test. Eur J Endocrinol. 2015;173(2):197–204. doi: 10.1530/EJE-14-0912. [DOI] [PubMed] [Google Scholar]
- 15.Arnaldi G, Angeli A, Atkinson AB, Bertagna X, Cavagnini F, Chrousos GP, Fava GA, Findling JW, Gaillard RC, Grossman AB, Kola B, Lacroix A, Mancini T, Mantero F, Newell Price J, Nieman LK, Sonino N, Vance ML, Giustina A, Boscaro M. Diagnosis and complications of Cushing’s syndrome: a consensus statement. J Clin Endocrinol Metab. 2003;88(12):5593–5602. doi: 10.1210/jc.2003-030871. [DOI] [PubMed] [Google Scholar]
- 16.Al-Saadi N, Diederich S, Oelkers W. A very high dose dexamethasone suppression test for differential diagnosis of Cushing’s syndrome. Clin Endocrinol (Oxf) 1998;48(1):45–51. doi: 10.1046/j.1365-2265.1998.00345.x. [DOI] [PubMed] [Google Scholar]
- 17.Nieman LK, Biller BM, Findling JW, Newell-Price J, Savage MO, Stewart PM, Montori VM. The diagnosis of Cushing’s syndrome: an Endocrine Society Clinical Practice Guideline. J Clın Endocrinol Metab. 2008;93(5):1526–1540. doi: 10.1210/jc.2008-0125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lindsay JR, Shanmugam VK, Oldfield EH, Remaley AT, Nieman LK. A comparison of immunometric and radioimmunoassay measurement of ACTH for the differential diagnosis of Cushing’s syndrome. J Endocrinol Invest. 2006;29(11):983–988. doi: 10.1007/BF03349211. [DOI] [PubMed] [Google Scholar]
- 19.Pecori Giraldi F, Saccani A, Cavagnini F. Study Group on the Hypothalamo-Pituitary-Adrenal Axis of the Italian Society of Endocrinology Assessment of ACTH assay variability: a multicenter study. Eur J Endocrinol. 2011;164(4):505–512. doi: 10.1530/EJE-10-0962. [DOI] [PubMed] [Google Scholar]
- 20.Gibson S, Pollock A, Littley M, Shalet S, White A. Advantages of IRMA over RIA in the measurement of ACTH. Ann Clin Biochem. 1989;6(26):500–507. doi: 10.1177/000456328902600608. [DOI] [PubMed] [Google Scholar]
- 21.Findling JW, Raff H. Cushing’s Syndrome: important issues in diagnosis and management. J Clın Endocrinol Metab. 2006;91(10):3746–3753. doi: 10.1210/jc.2006-0997. [DOI] [PubMed] [Google Scholar]
- 22.Vetter H, Strass R, Bayer JM, Beckerhoff R, Armbruster H, Vetter W. Short-term fluctuations in plasma cortisol in Cushing’s syndrome. Clin Endocrinol (Oxf) 1977;6(1):1–4. doi: 10.1111/j.1365-2265.1977.tb01989.x. [DOI] [PubMed] [Google Scholar]
- 23.Sheldon WR, Jr, De Bold CR, Evans WS, De Cherney GS, Jackson RV, Island DP, Thorner MO, Orth DN. Rapid sequential intravenous administration of four hypothalamic releasing hormones as a combined anterior pituitary function test in normal subjects. J Clın Endocrinol Metab. 1985;60(4):623–630. doi: 10.1210/jcem-60-4-623. [DOI] [PubMed] [Google Scholar]
- 24.DeBold CR, De Cherney GS, Jackson RV, Sheldon WR, Alexander AN, Island DP, Rivier J, Vale W, Orth DN. Effect of synthetic ovine corticotropin-releasing factor: prolonged duration of action and biphasic response of plasma adrenocorticotropin and cortisol. J Clin Endocrinol Metab. 1983;57(2):294–298. doi: 10.1210/jcem-57-2-294. [DOI] [PubMed] [Google Scholar]
- 25.Kaye TB, Crapo L. The Cushing’s Syndrome: an update on diagnostic tests. Ann Intern Med. 1990;112(6):434–444. doi: 10.7326/0003-4819-76-3-112-6-434. [DOI] [PubMed] [Google Scholar]
- 26.DeCherney GS, De Bold CR, Jackson RV, Sheldon WR, Jr, Island DP, Orth DN. Diurnal variation in the response of plasma adrenocorticotropin and cortisol to intravenous ovine corticotropin-releasing hormone. J Clin Endocrinol Metab. 1985;61(2):273–279. doi: 10.1210/jcem-61-2-273. [DOI] [PubMed] [Google Scholar]
- 27.Aron DC, Raff H, Findling JW. Effectiveness versus efficacy: the limited value in clinical practice of high dose dexamethasone suppression testing in the differential diagnosis of adrenocorticotropin-dependent Cushing’s syndrome. J Clin Endocrinol Metab. 1997;82(6):1780–1785. doi: 10.1210/jcem.82.6.3991. [DOI] [PubMed] [Google Scholar]
- 28.Antonio ID, Sandoval MA, Lantion-Ang FL. Crossing the otherside of the algorithm: a challengingcase of adrenal Cushing’s syndrome. BMJ Case Rep. 2011 doi: 10.1136/bcr.08.2011.4694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Giordano R, Marinazzo E, Berardelli R, Picu A, Maccario M, Ghigo E, Arvat E. Long-term morphological, hormonal, and clinical follow-up in a single unit on 118 patients with adrenal incidentalomas. Eur J Endocrinol. 2010;162(4):779–785. doi: 10.1530/EJE-09-0957. [DOI] [PubMed] [Google Scholar]
- 30.Terzolo M, Stigliano A, Chiodini I, Loli P, Furlani L, Arnaldi G, Reimondo G, Pia A, Toscano V, Zini M, Borretta G, Papini E, Garofalo P, Allolio B, Dupas B, Mantero F, Tabarin A, Italian Association of Clinical E AME position statement on adrenal incidentaloma. Eur J Endocrinol. 2011;164(6):851–870. doi: 10.1530/EJE-10-1147. [DOI] [PubMed] [Google Scholar]
- 31.Lacroix A, Feelders RA, Stratakis CA, Nieman LK. Cushing’s syndrome. Lancet. 2015;386(9996):913–927. doi: 10.1016/S0140-6736(14)61375-1. [DOI] [PubMed] [Google Scholar]