Abstract
Oral ingestion is a common, easy to access, route for therapeutic drugs to be delivered. The conception of the gastrointestinal tract as a passive physiological compartment has evolved towards a dynamic perspective of the same. Thus, microbiota plays an important role in contributing with additional metabolic capacities to its host as well as to its phenotypic heterogeneity. These adaptations in turn influence the efficacy and toxicity of a broad range of drugs. Notwithstanding, xenobiotics and therapeutic drugs affecting the microbiome’s activity also significantly impact metabolism affecting different organs and tissues, and thereby drugs’ toxicity/efficacy effects. Other physiological interfaces (i.e., gut, lungs, skin) also represent complex media with features about microbiota’s composition. Additionally, there have been described key regulatory effects of microbes on immunotherapy, because of its potential harnessing the host immune system, mental disorders by modulating neuroendocrine systems and cancer. These alterations are responsible of physiological variations in the response(s) between individuals and populations. However, the study of population-based differences in intestinal microbial-related drug metabolism has been largely inferential. This review outlines major reciprocal implications between drugs and microbes regulatory capacities in pharmacotherapy.
Keywords: Therapeutics, microbes, efficacy, toxicity, metabolism
1. Introduction
Pharmacological efficacy of therapeutic drugs depends on drug delivery and disposition of the active form responsible for physiological effects. A major challenge of therapeutics is to tailor personalized approaches to minimize population-based differences in bioavailability and biological effects of drugs. The evidence that ‘environmental’ factors greatly impact, to a different extent, drug metabolism and physiological behavior from one individual to another highlight the need of changing the way the efficacy/toxicity of several drugs is being approached.
Increasing evidence demonstrates that microbiome is a critical determinant in human health. Strategies to manipulate and prevent alterations in healthy microbiota to normalize physiological manifestations of metabolic and autoimmune diseases are actively being investigated. Microbiome develops a number of protective, immune and metabolic functions, which altogether have an enormous impact on the nutritional and health status of the host providing biological functions that cannot be performed by human metabolism 1. The application of metagenomics to the gut microbial ecosystem has revealed truly remarkable correlations between imbalances in gut microbiota and metabolic diseases 2. It has also led to the suggestion of the existence of a ‘core microbiome’ that encompasses key functions shared by each individual 3. Research efforts are focused to unravel microbial partners and define the microbiome composition in the gut and other interfaces between host and the external environment, but this task remains incomplete. Gut microbiota and its microbiome exhibit a relevant participation in the development or instauration of major diseases. The latter highlights the need to understand the relationship between intrinsic human genes, its interaction with different environmental factors and the molecular basis of drug efficacy and toxicity. 4,5 In the quest to better understand interactions between microbiota and diseases appearance and progression, new physiological information is needed. This information will contribute to implement personalized therapies tailored to the exact biology or biological state of an individual that constitutes one of the greatest challenges of modern medicine.
Microbiota plays an important role in training and modulating the immune system, preventing growth of harmful bacteria species, producing some useful vitamins and affording the extraction of energy and biotransformation of non-nutritive dietary components as well as therapeutic drugs and other xenobiotics. Besides, therapeutic agents can also end up exerting a significant impact on organs and/or tissues through their effect on gut microbiota. 6 The metabolic capacity of the intestinal microbiota could constitute a crucial factor affecting the disposition, stability and, thereby, efficacy and/or toxicity of several different therapeutic drugs due to extensive transformation of active structures 7–9. This metabolic activity leads to the production of metabolites, which exhibit a different behavior (i.e., solubility, disposition) than that observed for their parental compounds.
Metabolic activity of gut microbiota has led to consider it as a ‘supraorgan’ likely comparable to the liver 8–10. Notably, gut microbiome exhibits high variability between individuals (i.e., genders) and can constitute a source of variation of the effects observed. In fact, experimental studies have evidenced differences in the efficacy or toxicity of therapeutic drugs because of differences in microbial-mediated drug metabolism 11–13. These diverse and complicated interactions between the microbiome, drugs, and host still constitute a novel aspect that should not be ruled out when evaluating the potential, positive or negative, biological effects of drugs 8,14–17. The interaction between microbes and their metabolites with the main physiological interfaces seems to be necessary for health and wellbeing maintenance. 18
The study of human intestinal microbiota is one of the most challenging tasks for the scientist community because of its high complexity. The latter led to join efforts within international collaborative research projects to define microbial composition and characterize their genotype. 19 Additionally, there are intense research programs to develop animal models, including germ-free, antibiotic-treated, and disease-resistant mice as a landmark to unravel the role of the microorganisms that modulates host intestinal physiology. 20,21
This review helps to expand the knowledge about the role of microbiota as important contributor to delivery, disposition and co-metabolism of drugs. To this end, a comprehensive update of the different independently reported aspects about the dynamic interplay between drugs and microbes is reported and completed with current knowledge on novel clinical fields.
2. Impact of microbes on drug disposition.
Limited knowledge exists concerning potential metabolic reactions performed by the bacteria to metabolize therapeutic drugs 7,22,23. The existing studies are mostly related to drugs administered orally, whereas there is a lack of data about the efficacy of drugs administered by several other ways.
Currently, many different therapeutic drugs have been identified as susceptible of biotransformation by microbiota’s metabolism. There have been revealed significant differences in the metabolic activation or inactivation of drugs causing important variations of efficacy 8,24. However, there is scarce evidence that does demonstrate the population-based differences in microbial-related drug metabolism 11–12. Examples of the distinct reactions that have been described (Figure 1) affecting several drugs (Figure 2.) are briefly detailed below.
Figure 1.

Microbial-mediated biotransformation processes influencing drug disposition.
Figure 2.

Examples of drugs susceptible of microbial-mediated biotransformation.
Reductive metabolism
A well-known example of reductive metabolism is the marked decrease in the efficacy of cardiac glycoside drugs such as digoxin 22,23, which is particularly important because of its narrow therapeutic interval. In this sense, comparative studies performed on healthy volunteers from southern India and the United States revealed differences in drug metabolism of digoxin associated to the presence of Eubacterium lentum 11. Similarly, in vitro studies showed that therapeutic drugs such as misonidazole 25, sulfinpyrazone 26, and sulindac are susceptible to undergo reductive-mediated inactivation in the gastrointestinal tract by gut microbiota showing aldehyde oxidase activity. Similarly, studies on rabbits reported the incomplete bioavailability and greater formation of the active metabolite of these drugs when are administered (orally). 27 Similarly, some other in vitro studies have demonstrated the capability of different microbial (Bacillus subtilis MTCC 619, but not Escherichia coli MTCC 118), and fungi (Beauveria bassiana NCIM 1216 used as biological insecticide) cultures to produce the biotransformation of valdecoxib 28. The metabolism of metronidazole 29 and zomisamide 30 has also been evaluated in germ-free and antibiotic-treated animals, respectively. These studies displayed that caecal contents of conventional animals had significant reductase activity, whereas slight or no activity was observed in germ-free and antibiotic-treated animals. In germ-free and antibiotic-treated rats, bacterial nitroreductase activities have also been associated with an increased toxicity and teratogenic potential of drugs such as nitrazepam and clonazepam 31,32. These effects concluded that gut microbiota is essentially involved in the activation of nitroreductase activity. However, these reductive metabolic features of gut microbiota could also be advantageously used when administering pro-drugs to deliver the active structures to the precise location where they are required. For example, the metabolic activation of antimicrobial or anti-inflammatory drugs such as prontosil or sulfasalazine 33, respectively, and their derivatives 34 occurs through the reduction of the “azo” bond in their structures minimizing side effects. The strategy of pro-drugs is also useful for poorly absorbed drugs from the gastrointestinal tract as succinylsulfathiazole, which is activated by elimination of the succinyl group acquiring efficacy as antibacterial. 35
In vitro studies have described a significant decline in ranitidine and nizatidine levels because of the microbiota-mediated in vitro cleavage of the N-oxide bond in these molecules 36. However, if these transformations take place in vivo is not clear as the highest rates of absorption (medians 160–923 μg x 30 cm−1 x min−1) are found in the most proximal region (duodeno/jejunal junction), where bacteria are present at low proportions, and not in the distal part of the intestine or colon 37. Similarly, it has been reported that omeprazole is susceptible of bacterial reduction in vitro 38, but its efficacy should not be affected as it is mostly absorbed before reaching the lower intestine. Animal studies documented that omeprazole administration to rats caused marked shifts in the numbers and genus of enteric bacteria, including a significant reduction (∼80%) of jejunal Actinobacteria, particularly Bifidobacterium spp. 39 considered beneficial to the host’s health.
Hydrolysis / Immunotherapy
The dramatic impact of microbial metabolism on the toxicity of metabolites derived from drugs was clearly manifested in the death of fifteen patients, who were orally administered with sorivudine (SRV, 1-b-d-arabinofuranosyl-(E)-5-(2-bromovinyl) uracil) within forty days 40. This effect was attributed to the enterobacteria-mediated SRV hydrolysis, thus leading to the formation of 5-(2-bromovinyl) uracil. This transformation is mainly carried out by E. coli 40 and Bacteroides spp. (B. vulgatus, B. thetaiotaomicron, B. fragilis, B. uniformis and B. eggerthii) 41 and increases toxicity of the anticancer chemotherapy with 5-fluorouracil pro-drugs. The anticancer activity of 5-fluorouracil relies on the restoration of T-cell immunity following the elimination of myeloid-derived suppressor cells. In this context, coadjutant strategies to restore the population of those microbial groups considered as healthy could provide additional benefits to the pharmacological intervention. For example, recent data provided convincing evidences about the promising potential of defined commensal bacteria (Bifidobacterium longum) as part of immunotherapy because of its potential harnessing the host immune system favoring anti–PD-L1 efficacy 42. The scarce existing data allow us to hypothesize that immune-metabolic activity of microbes could have an important impact favoring antigen presenting processes, thus preventing T cell anergy or exhaustion. In relation to cancer chemotherapy efficacy and toxicity a recent review has focused on defining tools for manipulating microbiota in the clinical practice 43.
Intestinal bacteria can also mediate the conversion of a nitro group to a primary aromatic amine with further hydrolysis of the amide linkage as occurs from orally administered chloramphenicol to conventional, but not antibiotic-treated, animals producing p-aminophenyl-2-amino-1,3-propanediol 44. It is noteworthy to mention that this reaction is not produced by beneficial lactic acid bacteria, which are highly sensitive to chloramphenicol 45.
Denitration, decarboxylation
Denitration of glyceryl trinitrate, commonly used for the treatment of angina pectoris, has also been reported after incubation of this drug with rat colonic contents 46 as well as mixed cultures with wastewater (Pseudomonas putida and Pseudomonas fluorescens) 47,48 in anaerobic and aerobic conditions, respectively. This observation could motivate an important decline on drug delivery, since glyceryl trinitrate is mainly administered through transdermal patches; however, it is difficult to draw out any conclusion due to the lack of studies evaluating the potential impact of skin microbiome on drug metabolism. Another important reaction reported is the reaction of decarboxylation of dyhidroxyphenylalanine residues of L-DOPA, effective in the therapy of Parkinson’s disease, undertaken by bacteria. This biotransformation highlights aspects such as whether variations on the concentrations of L-DOPA reaching the central nervous system affect its desired bioactivity 49.
Functional groups
The bacteria-mediated inactivation of the active component of sulfasalazine (i.e., 5-aminosalicylic acid) has been reported to take place through the incorporation of an acetyl group in the drug’s structure by a plethora of bacteria (Citrobacter amalonaticus, Citrobacter farmeri, Citrobacter freundii, Klebsiella ozaenae, Klebsiella oxytoca, Klebsiella rhinoscleromatis, Morganella morganii, Serratia marcescens, Shigella flexneri, Plesiomonas shigelloides, and Vibrio cholerae) commonly present in the gut 50. The elimination of the acetyl group from another analgesic such as phenacetin leads to the formation of p-phenetidin, and exacerbates the in vitro toxic effects 51, which are connected with methemoglobinemia and nephritis.
N-demethylation seems to be the molecular mechanism by which gut microbiota mediate the alteration of methamphetamine 52. Notably, this reaction has only been reported in vitro using caecal contents of guinea pigs. This alteration can have little or no effect on drug efficacy as methamphetamine is mainly absorbed in the duodenal/jejunal sections of the intestine. In addition, the alteration of amine/amide groups by bacterial metabolism can also take place when they are present as thiazole ring, for example, the biotransformation of levamisole - coadjuvant in vaccines - mainly due to Bacteroides and Clostridium spp. 53. Similarly, the isoxazole ring of risperidone resulted metabolized by gut microbiota showing oxidative dealkylation at the piperidine nitrogen and the scission of the isoxazole in the benzisoxazole ring system 54.
Among all the bacteria-mediated alterations on drugs’ structure, there can be particularly highlighted two major metabolic reactions. These changes are associated with the alteration of bile acid metabolites and derived conjugates affecting drug disposition 55. Additionally, expression and activity of major drug-metabolizing enzymes have also a significant effect on drugs’ efficacy 56. These interactions are especially relevant in the case of parent molecules that render highly toxic metabolites delaying its elimination from blood circulation. Irinotecan is a pro-drug that renders the SN-38 metabolite (7-ethyl-10-hydroxycamptotecin). SN-38, which exerts much more antitumor activity, is eliminated by conjugation with a glucuronide group in the liver. However, this metabolite is majorly deconjugated by the β-glucuronidase activity of gut microbiota, thus contributing to exacerbate intestinal toxicity (structural and functional injuries) 22,57. Other well-known examples of drugs suffering bacteria-mediated deconjugation and enterohepatic recycling are digitoxin 58 or morphine that results in reduced phagocytosis of Gram-positive in comparison to Gram-negative bacteria 59. In addition to classic deglycosylation processes 59,60, human-derived data demonstrate the positive effects of polysaccharides, from gingenosides used in traditional medicine, recovering the microbiota (i.e., Lactobacillus spp. and Bacteroides spp.) responsible for its metabolism 21. These products have been identified as probably responsible for the bioactivity of ginsenosides in humans. 60 Similarly, bacterial-mediated metabolism also occurs on pharmacologically active natural glucuronides such as biacalin (from Scutellariae radix) 61 and glucosinolates like glucorapharin (found in Cruciferous vegetables) with anticarcinogenic properties 23. The bacterial-mediated metabolism favors their rapid appearance in the systemic circulation, as demonstrated in conventional rats in comparison to germ free animals 61. However, there have been identified species- and substrate-specific differences in the capacity of the caecal microbiota to hydrolyze different glucuronide conjugates (phenolphthalein glucuronide, 4-methylumbelliferone glucuronide, morphine-3-glucuronide) 62 pointing to the importance of gut microbiota composition in the disposition and efficacy of these pro-drugs.
3. Dynamic interplay between microbes and drugs.
The mechanisms commonly involved in the host-drug interaction are: i) delayed gastric emptying, ii) alteration of the “first step” of drug metabolism in the intestine and/or liver, iii) increased bile flow, iv) formation of complexes and non-absorbable chelates, v) increased splanchnic blood flow, vi) alteration of intestinal transit time, and vii) modification of gastrointestinal and/or urine pH. In the intestine, detoxifying systems (multidrug resistance, cytochrome P450 or ABC transporters - ATP-binding cassette transporters - in general) work in conjunction metabolizing the administered drugs or xenobiotics by introduction of functional groups. This modification favors their excretion or accumulation into the entero-hepatic circulation of polar metabolites.
Nutrients and metabolites are transported from the intestine to the liver establishing a way of interconnection, known as ‘gut-liver axis’, whereby microbiota-biotransformed metabolites interact with liver metabolism jointly with those compounds transported in the bile (Figure 2.). Liver cells respond to these compounds via different nuclear receptors “xenosensors” that have been demonstrated, in animal studies, be susceptible of regulation by the microbiota 12,17. These comprehensive analyses revealed transcriptional and protein changes in a network connected with the hepatic metabolism of xenobiotics affecting the activity of cytochrome P450, fatty acids and lipid metabolism, amino acid metabolism and bile acid synthesis. A clear example is the recently demonstrated effect of the probiotic microorganism Lactobacillus casei decreasing the expression (mRNA and protein) levels of cytochromes P450 in male rats, an effect that could prevent the development of colorectal cancer and some other interactions with the concurrent administration of pharmacotherapeutic agents 63.
The potential bacteria-mediated modulation in the activity of organs such as the gut and liver, and thereby drugs disposition and metabolism affects the dosage of therapeutic compounds. For example, gut microbiota has been identified as responsible of a less efficient metabolism of pentobarbital 12. The impaired metabolism is reflected in a longer time of anesthesia in conventionally-raised animals compared to germ-free animals. Indeed, there have been reported moderate effects of gut microbiota in the modulation the levels of phase II metabolizing enzymes in germ-free rats 16,64. Intestinal bacteria have shown an important regulatory effect on glutathione S-transferases (p1 and p2), as well as major bile acid transporters (i.e., Na+-taurocholate cotransporting polypeptide and organic anion-transporting polypeptide 1b2), and the cholesterol transporter Abcg5/Abcg8 64.
The bacterial-mediated differences in metabolizing enzymes in conventionally raised animals, as concluded from comparison to germ-free rats, were low protein levels of glutathione transferases, gastrointestinal glutathione peroxidase, epoxy hydrolases and N-acetyltransferases. These results evidence the participation of gut microbiota metabolism to reduce drugs concentration available after oral administration. However, the effects observed displayed a high variability based on the animal model (mouse or rat) and diet 16. The bacteria-mediated modulation of metabolizing systems in complement organs has also been associated with the bioactivation of toxic molecules, for example cycasin – a carcinogenic glycoside from cycads hydrolytically activated - or heterocyclic amines 63,65. In this sense, bacterial metabolizing enzymes can hydrolyze the conjugates produced in the liver and excrete them into bile, thus delaying their elimination and, thereby, exacerbating the potential cytotoxic effects. A growing body of evidence indicates that gut microbiota also impacts on intestinal and hepatic expression and function of transporters (uptake and efflux) 64,66. Notably, the direction of these responses suggests that microbiota-mediated modulation of host’s gene expression and/or function in the gut and/or liver can greatly impact, worsening or improving, drugs’ efficacy/toxicity in mice and patients.
Emerging data have identified the intestinal microbiota as a regulatory component of the intestinal endocannabinoid system (eCB) increasing the expression of endocannabinoid-1 agonist receptor 67. This microbial influence on the eCB system can be hypothesized as responsible of the dyskinetic behavior observed in 6-hydroxyphenylalanine-treated rats 68. In addition, endocannabinoid system participates in the regulation of energy homeostasis and gut permeability. 69 As described, there could be expected inflammatory and hypoxic conditions derived from the endogenous anandamide- and 2-arachidonoyl glicerol-mediated CB1 activation. Thus, the deregulation of cellular energetic metabolism could impair the activation of ABC transporters, which play an important role in the regulation of nutrients and xenobiotics accumulation into cells. The impairment of ABC transporters activity, in conjunction with alterations of gut permeability, condition drugs disposition increasing their toxic effects. There are some examples illustrating how bacteria can influence the regulation (either up or down) of ABC transporters in the duodenum and colon. These sections of the intestine exhibit ability to modulate the absorption, distribution, metabolism, secretion and, thereby the impact of drugs and xenobiotics on human health 70. Besides their role in drug disposition, the hypothesis that ABC transporters can act as potential receptors for pathogenic bacteria to cause infection is gaining strength.
Although it has been demonstrated the important impact of microbiome in host’s phenotype 17, recent comprehensive analyses revealed different transcriptional response(s) to several Lactobacilli (L. acidophilus, L. casei, and L. rhamnosus) 71. These authors evidenced, in a modest number of volunteers, response(s) related to the regulation of major basal mucosal processes with reasonable similarity to response profiles obtained for specific bioactive molecules and drugs 72. These results are in agreement with the previously described microbial modulation of host’s metabolic and detoxifying phenotypes. However, these authors also indicated large person-to-person variations in response to transcriptomes that may lead to measurable effects in some volunteers, but not in others. All these aspects should not be ruled out in research efforts to modeling mammalian biocomplexity 72.
Besides the impact of microbes in drugs disposition and biotransformation, there have also been reported changes in microbiota diversity and numbers as consequence of the drugs-mediated modifications in the internal milieu 39,73, inhibition of intracellular physiological processes 73, deregulation of intestinal homeostasis and/or even modulation of the expression of several different receptors 74,75. Additionally, the microbiota-mediated bioactivation of anti-inflammatory drugs such as mesalazine has also been associated with lower faecal bacteria abundances and rebalancing of the major constituents of the microbiota 73.
A novel area where microbiota composition has emerged as a relevant factor influencing physiological responses is mental disorders. At present, drugs belonging to new classes of antidepressants still have antimicrobial effects. Desipramine is an important antidepressant psychotherapeutic drug that causes important side-effects like dysfunction of salivary gland (i.e., oral dryness, xerostomia). This alteration generates a decrease in amylase protein levels, but an increase of the synthesis of proline-rich proteins and lysozyme as a function of the treatment and the subject’s age 76. Subsequently, oral microbiota changes with the administration of desipramine result in a higher incidence of infections generating gingivitis. However, fundamental questions concerning the microbial taxa that result affected remain unsolved. In this sense, a recent informative review summarized current data concerning the implication of dysbiosis of gut microbiota in the development or exacerbation of mental disorders 77.
The effects of opioids on gut microbiota have been described in vivo and in vitro. Morphine can alter gut microbiota by inducing dissemination of Gram-positive intestinal bacteria (Staphylococcus sciuri, S. cohnii, S. aureus, Enterococcus durans, E. casseliflavus, E. faecium and E. faecalis) in C57BL/6J mice 78. This effect was exerted through the activation of innate immune Toll-like receptor (TLR)-2 and up-regulation of interleukin (IL)-17A 79. There have also been reported morphine-induced inhibitory effects on the in vitro production of interleukins and signaling of the innate immune toll like receptors, thus increasing susceptibility to bacterial infections. Morphine impaired mucosal host defense innate immune signaling processes to Streptococcus pneumoniae-induced IL-23 production through MyD88-IRAK1/4-dependent TLR2 and Nod2 signaling in dendritic cells 74. Similarly, it has been shown the morphine-mediated modulation of gut immunity and homeostasis 80 and the depletion of lymphoid subsets 78. These data point to the impact of opioids on the internal milieu favoring infections and alterations of the health status.
Although this review does not focus on abuse drugs, it is worth to bring here some of the potential contributions to their efficacy and toxicity. Briefly, microbial interaction with abuse drugs seems to contribute in the development of specific phenotypes. This has been identified in populations sharing a social (crack cocaine abuse) network, rather than genetic features, favoring the infection of M. tuberculosis 81. Similarly, cigarette smoke exposure has been linked to high risk of infections evidencing the close relationship between smoking and disruption of the fine-tuned balance of microbiota in the upper human respiratory tract 82. Alcohol consumption has also been identified as one of the causative factors responsible for intestinal dysbiosis as concluded from a reduced mucosal-associated microbial diversity 83. Moreover, alcohol consumption on intestinal microbiota is associated with increased endotoxemia that appears as a mechanism, additional to gut leakiness, of alcoholic steatohepatitis.
Apart from the aforementioned diseases, some metabolic syndromes (i.e., non-alcoholic fatty liver, obesity, and type 2 diabetes) can significantly change bacterial composition and subsequently drug metabolism. 84,85 All these exhibit low-grade inflammation as a common feature, which can be significantly modulated by gut microbiota, thus affecting therapeutics efficacy/toxicity.
4. Conclusions and future perspectives.
Gut microbiota is an essential factor in the metabolic capacities of the host regulating the expression of genes and proteins. Thus, potentially affecting the biotransformation and toxicity/efficacy of therapeutic drugs. The key biological roles that gut microbiota plays in several different metabolic diseases has changed the way it is investigated, since the metabolites and effects depend on particular features of gut microbiota composition and even interindividual variability. Besides, drug-mediated changes in gut microbiota composition can also have important consequences probably modulating its functional effects in mammalian tissues. The existing scientific evidence highlights the potential importance and role of gut microbiota as a new aspect that should be taken into more consideration when establishing pharmacological therapeutic strategies. Advances on the knowledge of the interactions between bioactive food compounds and specific intestinal bacteria will help to better understand population-based differences in microbial-related drug metabolism. The latter will contribute the way for development of therapeutic applications based on personalized medicine, yet important questions such as how far and to what extent can the consequences of bacterial-related metabolic changes in efficacy/toxicity be modulated via targeted intervention.
These are novel perspectives connecting alterations in the microbiota composition of physiological interfaces to risk factors for different metabolic and/or immunological diseases. However, current evidence for an association of these aspects with drugs’ efficacy/toxicity/resistance has largely been inferential. There remain key unanswered questions that require a concerted research effort to arm the scientific and clinical communities and finally society with effective translational strategies. To this end, there is a need to overcome the usually fragmented and compartmentalized approach to evaluate the efficacy/toxicity of therapeutic drugs taking into account the role of microbiota composition in drug delivery/disposition.
Figure 3.
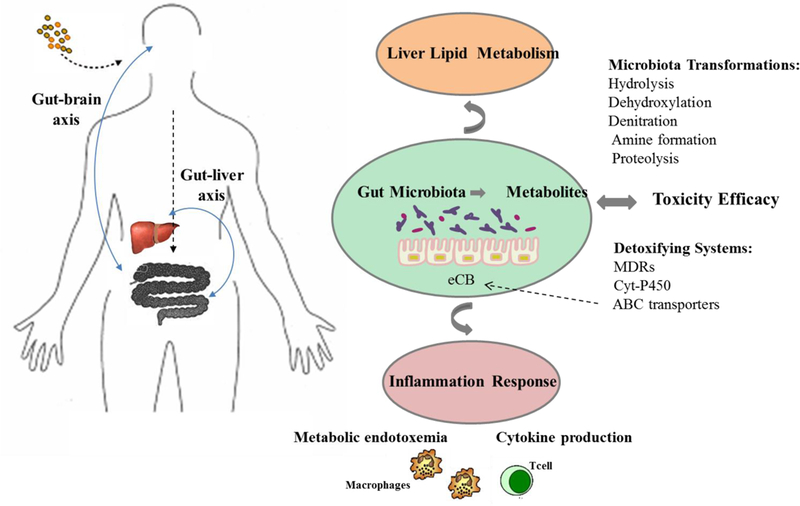
Interactions of gut microbiota with therapeutic drugs.
The black dotted arrow represented the absorption process of drugs. The bold grey arrow represented a schematic interaction of the gut microbiota with the immune-metabolic axis and the different mechanisms proposed to explain its implication in toxicity efficacy and health/disease risk. Mechanisms underlying the connection of drugs-induced host detoxifying metabolism activation influenced by gut microbiota (e.g. multidrug resistance protein, MDRs; the cytochrome P450 superfamily, CYPs; and the ATP-binding cassett transporters, ABC transporters) and microbiota biotransformations (e.g. hydrolysis, dehydroxylation, denitration, amine formation and proteolysis). Drugs metabolism occurs associated to the energetic cell metabolism; and the endocannabinoid system (eCB) participates in the regulation of energy homeostasis and gut permeability (increasing the possibility to appears a metabolic endotoxemia). The deregulation of cellular energetic metabolism could impair the activation of ABC transporters causing together inflammation (cytokines production) and alterations in gut permeability increasing the toxic effects.
Acknowledgements
JML thanks MICINN for his ‘Ramon y Cajal’ contract. J.A.G.-B. is supported by a postdoctoral award from the American Heart Association (16POST30690001).
6. References
- 1.Jia W, Li H, Zhao L, Nicholson JK. Gut microbiota: a potential new territory for drug targeting. Nat Rev Drug Discov. 2008; 7(2): 123–9. [DOI] [PubMed] [Google Scholar]
- 2.Turnbaugh PJ, Ley RE, Mahowald MA, Magrini V, Mardis ER, Gordon JI. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature. 2006; 444: 1027–1031. [DOI] [PubMed] [Google Scholar]
- 3.Blaut M Ecology and Physiology of the Intestinal Tract. Curr Top Microbiol Immunol. 2013; 358: 247–72. [DOI] [PubMed] [Google Scholar]
- 4.Nicholson JK. Global systems biology, personalized medicine and molecular epidemiology. Mol Syst Biol. 2006; 2: 52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Tzika AA, Constantinou C, Bandyopadhaya A, et al. A small volatile bacterial molecule triggers mitocondrial dysfunction in murine skeletal muscle. PLoS One. 2013; 8(9): e74528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Cani PD, Delzenne NM. The gut microbiome as therapeutic target. Pharmacol Ther. 2011; 130(2): 202–12. [DOI] [PubMed] [Google Scholar]
- 7.Sousa T, Paterson R, Moore V, Carlsson A, Abrahamsson B, Basit AW. The gastrointestinal microbiota as a site for the biotransformation of drugs. Int J Pharm. 2008; 363(1–2): 1–25. [DOI] [PubMed] [Google Scholar]
- 8.Wilson ID, Nicholson JK. The role of gut microbiota in drug response. Curr Pharm Des 2009; 15: 1519–23. [DOI] [PubMed] [Google Scholar]
- 9.Carmody RN, Turnbaugh PJ. Host-microbial interactions in the metabolism of therapeutic and diet-derived xenobiotics. J Clin Invest. 2014; 124(10): 4173–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Gill SR, Pop M, Deboy RT, et al. Metagenomic analysis of the human distal gut microbiome. Science. 2006; 312(5778): 1355–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Mathan VI, Wiederman J, Dobkin JF, Lindenbaum J. Geographic differences in digoxin inactivation, a metabolic activity of the human anaerobic gut flora. Gut. 1989; 30(7): 971–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Björkholm B, Bok CM, Lundin A, et al. Intestinal microbiota regulate xenobiotic metabolism in the liver. PLoS One. 2009; 4(9): e6958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Davey KJ, O’Mahony SM, Schellekens H, et al. Gender-dependent consequences of chronic olanzapine in the rat: effects on body weight, inflammatory, metabolic and microbiota parameters. Psychopharmacology (Berl). 2012; 221(1): 155–69. [DOI] [PubMed] [Google Scholar]
- 14.Qin X Gut microbiota: a new aspect that should be taken into more consideration when assessing the toxicity of chemicals or the adverse effects and efficacy of drugs. Regul Toxicol Pharmacol. 2008; 51(2): 251. [DOI] [PubMed] [Google Scholar]
- 15.Li H, He J, Jia W. The influence of gut microbiota on drug metabolism and toxicity. Expert Opin Drug Metab Toxicol. 2016; 12(1): 31–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kim DH. Gut Microbiota-Mediated Drug-Antibiotic Interactions. Drug Metab Dispos. 2015; 43(10): 1581–9. [DOI] [PubMed] [Google Scholar]
- 17.Meini W, Sczesny S, Brigelius-Flohé R, Blaut M, Glatt H. Impact of gut microbiota on intestinal and hepatic levels of phase 2 xenobiotic-metabolizing enzymes in rat. Drug Metab Dispos. 2009; 37(6): 1179–86. [DOI] [PubMed] [Google Scholar]
- 18.Moens E, Veldhoen M. Epithelial barrier biology; Good fences make good neighbours. Immunology. 2012; 135(1): 1–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hattori M, Taylor TD. The human intestinal microbiome: a new frontier of human biology. DNA Res. 2009, 16(1):1–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zhang LS, Davies SS. Genome Medicine, Microbial metabolism of dietary components to bioactive metabolites: opportunities for new therapeutic interventions. Genome Med. 2016, 21;8(1):46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Zhou SS, Xu J, Zhu H, Wu J, Xu JD, Yan R, Li XY, Liu HH, Duan SM, Wang Z, Chen HB, Shen H, Li SL. Gut microbiota-involved mechanisms in enhancing systemic exposure of ginsenosides by coexisting polysaccharides in ginseng decoction. Sci Rep. 2016, 6:22474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Yamamoto M, Kurita A, Asahara T, et al. Metabolism of irinotecan and its active metabolite SN-38 by intestinal microflora in rats. Oncol Rep. 2008; 20(4): 727–30. [PubMed] [Google Scholar]
- 23.Li F, Hullar MA, Beresford SA, Lampe JW. Variation of glucoraphanin metabolism in vivo and ex vivo by human gut bacteria. Br J Nutr. 2011; 106: 408–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Sekirov I, Russell SL, Antunes LCM, Finlay BF. Gut Microbiota in Health and Disease. Physiol Rev. 2010; 90(3): 859–904 [DOI] [PubMed] [Google Scholar]
- 25.Koch RL., Beaulieu BB Jr., Goldman P Role of the intestinal flora in the metabolism of misonidazole. Biochem Pharmacol. 1980; 29(24): 3281–4. [DOI] [PubMed] [Google Scholar]
- 26.Strong HA, Renwick AG, George CF, Liu YF, Hill MJ. The reduction of sulphinpyrazone and sulindac by intestinal bacteria. Xenobiotica. 1987; 17(6): 685–96. [DOI] [PubMed] [Google Scholar]
- 27.Strong HA, Renwick AG, George CF. The site of reduction of sulphinpyrazone in the rabbit. Xenobiotica. 1984; 14(10): 815–26. [DOI] [PubMed] [Google Scholar]
- 28.Keshetti S, Ciddi V. Biotransformation of valdecoxib by microbial cultures. J Microbiol Biotechnol. 2010; 20(4): 809–16. [PubMed] [Google Scholar]
- 29.Chrystal EJ, Koch RL, McLafferty MA, Goldman P. Relationship between metronidazole metabolism and bactericidal activity. Antimicrob Agents Chemother. 1980; 18(4): 566–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kitamura S, Sugihara K, Kuwasako M, Tatsumi K. The role of mammalian intestinal bacteria in the reductive metabolism of zonisamide. J Pharm Pharmacol. 1997; 49(3): 253–6. [DOI] [PubMed] [Google Scholar]
- 31.Elmer GW, Remmel RP. Role of intestinal microflora in clonazepam metabolism in the rat. Xenobiotica. 1984; 14: 829–840. [DOI] [PubMed] [Google Scholar]
- 32.Takeno S, Sakai T. Involvement of the intestinal microflora in nitrazepam-induced teratogenicity in rats and its relationship to nitroreduction. Teratology. 1991; 44(2): 209–14. [DOI] [PubMed] [Google Scholar]
- 33.Lee HJ, Zhang H, Orlovich DA, Fawcett JP. The influence of probiotic treatment on sulfasalazine metabolism in rat. Xenobiotica. 2012; 42(8): 791–7. [DOI] [PubMed] [Google Scholar]
- 34.Hayllar J, Bjarnason I. Sulphasalazine in ulcerative colitis: in memoriam?. Gut. 1991; 32(5): 462–3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Gustafsson BE, Norin KE. Development of germ-free animal characteristics in conventional rats in antibiotics. Acta Pathol Microbiol Scand B. 1977; 85B(1): 1–8. [DOI] [PubMed] [Google Scholar]
- 36.Basit AW, Podczeck F, Newton JM, et al. The use of formulation technology to assess regional gastrointestinal drug absorption in humans. Eur J Pharm Sci. 2004; 21(2–3): 179–89. [DOI] [PubMed] [Google Scholar]
- 37.Gramatté T, el Desoky E, Klotz U. Site-dependent small intestinal absorption of ranitidine. Eur J Clin Pharmacol. 1994; 46(3): 253–9. [DOI] [PubMed] [Google Scholar]
- 38.Watanabe K, Yamashita S, Furuno K, Kawasaki H, Gomita Y. Metabolism of omeprazole by gut flora in rats. J Pharm Sci. 1995; 84(4): 516–7. [DOI] [PubMed] [Google Scholar]
- 39.Wallace JL, Syer S, Denou E, et al. Proton Pump Inhibitors Exacerbate NSAID-Induced Small Intestinal Injury by Inducing Dysbiosis. Gastroenterology, 2011; 141(4): 1314–322. [DOI] [PubMed] [Google Scholar]
- 40.Ogura K, Nishiyama T, Takubo H, et al. Suicidal inactivation of human dihydropyrimidine dehydrogenase by (E)-5-(2-bromovinyl)uracil derived from the antiviral, sorivudine. Cancer Lett. 1998; 122(1–2): 107–13. [DOI] [PubMed] [Google Scholar]
- 41.Nakayama H, Kinouchi T, Kataoka K, et al. Intestinal anaerobic bacteria hydrolyse sorivudine, producing the high blood concentration of 5-(E)-(2-bromovinyl)uracil that increases the level and toxicity of 5-fluorouracil. Pharmacogenetics. 1997; 7(1): 35–43. [DOI] [PubMed] [Google Scholar]
- 42.Sivan A, Corrales L, Hubert N, et al. Commensal Bifidobacterium promotes antitumor immunity and facilitates anti–PD-L1 efficacy. Science. 2015; 350(6264): 1084–1089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Alexander JL, Wilson ID, Teare J, et al. Gut microbiota modulation of chemotherapy efficacy and toxicity. Nat Rev Gastroenterol Hepatol. 2017; 14(6): 356–365. [DOI] [PubMed] [Google Scholar]
- 44.Hwang BY, Oh JM, Kim J, Kim BG. Pro-antibiotic substrates for the identification of enantioselective hydrolases. Biotechnol Lett. 2006; 28(15): 1181–5. [DOI] [PubMed] [Google Scholar]
- 45.Karapetkov N, Georgieva R, Rumyan N, Karaivanova E. Antibiotic susceptibility of different lactic acid bacteria strains. Benef Microbes. 2011; 2(4): 335–9. [DOI] [PubMed] [Google Scholar]
- 46.Abu Shamat M The role of gastrointestinal microflora in the metabolism of drugs. Int J Pharm. 1993; 97: 1–13. [Google Scholar]
- 47.Blehert DS, Knoke KL, Fox BG, Chambliss GH. Regioselectivity of nitroglycerin denitration by flavoprotein nitroester reductases purified from two Pseudomonas species. J Bacteriol. 1997; 179: 6912–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Accashian JV, Vinopal RT, Kim BJ, Smets BF. Aerobic growth on nitroglycerin as the sole carbon, nitrogen, and energy source by a mixed bacterial culture. Appl Environ Microbiol. 1998; 64(9): 3300–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Lee S, Chen Y, Luo H, et al. The first global screening of protein substrates bearing protein-bound 3,4-Dihydroxyphenylalanine in Escherichia coli and human mitochondria. J Proteome Res. 2010; 9(11): 5705–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Deloménie C, Fouix S, Longuemaux S., et al. Identification and functional characterization of arylamine N-acetyltransferases in eubacteria: evidence for highly selective acetylation of 5-aminosalicylic acid. J Bacteriol. 2001; 183(11): 3417–27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Corbett MD, Wei CI, Fernando SY, Doerge DR, Corbett BR. The synthesis and mutagenicity of the N-formyl analog of N-hydroxyphenacetin. Carcinognesis. 1983; 4(12): 1615–8. [DOI] [PubMed] [Google Scholar]
- 52.Caldwell J, Hawksworth GM. The demethylation of methamphetamine by intestinal microflora. J Pharm Pharmacol. 1973; 25(5): 422–4. [DOI] [PubMed] [Google Scholar]
- 53.Shu YZ, Kingston DG, Van Tassell RL, Wilkins TD. Metabolism of levamisole, an anti-colon cancer drug, by human intestinal bacteria. Xenobiotica. 1991; 21(6): 737–50. [DOI] [PubMed] [Google Scholar]
- 54.Meuldermans W, Hendrickx J, Mannens G, et al. The metabolism and excretion of risperidone after oral administration in rats and dogs. Drug Metab Dispos. 1994; 22(1): 129–38. [PubMed] [Google Scholar]
- 55.Schiller LR. Evaluation of small bowel bacterial overgrowth. Curr Gastroenterol Rep. 2007; 9(5): 373–7. [DOI] [PubMed] [Google Scholar]
- 56.Claus SP, Ellero SL, Berger B, et al. Colonization-induced host-gut microbial metabolic interaction. MBio 2011; 2(2): e00271–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Takasuna K, Hagiwara T, Hirohashi M, et al. Inhibition of intestinal microflora beta-glucuronidase modifies the distribution of the active metabolite of the antitumor agent, irinotecan hydrochloride (CPT-11) in rats. Cancer Chemother Pharmacol. 1998; 42(4): 280–6. [DOI] [PubMed] [Google Scholar]
- 58.Songu-Mize E, Nash CB, Elam JT, Caldwell RW. Metabolism and cardiac actions of a polar aminocardenolide and digoxin in the conscious dog. J Pharmacol Exp Ther. 1983; 226(1): 65–70. [PubMed] [Google Scholar]
- 59.Hasegawa H, Sung JH, Matsumiya S, Uchiyama M. Main ginseng saponin metabolites formed by intestinal bacteria. Planta Med. 1996, 62(5):453–7. [DOI] [PubMed] [Google Scholar]
- 60.Tawab MA, Bahr U, Karas M, Wurglics M, Schubert-Zsilavecz M. Degradation of ginsenosides in humans after oral administration. Drug Metab Dispos. 2003, 31(8):1065–71. [DOI] [PubMed] [Google Scholar]
- 61.Akao T, Kawabata K, Yanagisawa E. et al. Baicalin, the predominant flavone glucuronide of scutellariae radix, is absorbed from the rat gastrointestinal tract as the aglycone and restored to its original form. J Pharm Pharmacol. 2000; 52(12): 1563–8. [DOI] [PubMed] [Google Scholar]
- 62.Kenyon EM, Calabrese EJ. Extent and implications of interspecies differences in the intestinal hydrolysis of certain glucuronideconjugates. Xenobiotica. 1993; 23(4): 373–81. [DOI] [PubMed] [Google Scholar]
- 63.Matuskova Z, Siller M, Tunkova A, et al. Effects of Lactobacillus casei on the expression and the activity of cytochromes P450 and on the CYP mRNA level in the intestine and the liver of male rats. Neuro Endocrinol Lett. 2011; 32: S1. [PubMed] [Google Scholar]
- 64.Selwyn FP, Cui JY, Klaassen CD. RNA-Seq Quantification of Hepatic Drug Processing Genes in Germ-Free Mice. Drug Metab Dispos. 2015; 43: 1572–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Overvik E, Lindeskop P, Midtvedt T, Gustafsson JA. Mutagen excretion and cytochrome P-450 dependant activity in germ free and conventional rats fed a diet containing fried meat. Food Chem Toxicol. 1990; 28: 253–61. [DOI] [PubMed] [Google Scholar]
- 66.Rowland IR, Mallett AK, Cole CB, Fuller R. Mutagen activation by hepatic fractions from conventional, germ free and monoassociated rats. Arch Toxicol. 1987; S11: 261–3. [DOI] [PubMed] [Google Scholar]
- 67.Muccioli GG, Naslain D, Bäckhed F, et al. The endocannabinoid system links gut microbiota to adipogenesis. Mol Syst Biol. 2010; 6: 392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Martinez A, Macheda T, Morgese MG, Trabace L, Giuffrida A. The cannabinoid agonist WIN55212–2 decreases l-DOPA-induced PKA activation and dyskinetic behavior in 6-OHDA-treated rats. Neurosci Res. 2012; 72(3): 236–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Karwad MA, Couch DG, Theophilidou E, et al. The role of CB1 in intestinal permeability and inflammation. FASEB J. 2017, 31(8):3267–3277. [DOI] [PubMed] [Google Scholar]
- 70.Mercado-Lubo R, McCormick BA. The interaction of gut microbes with host ABC transporters. Gut Microbes. 2010; 1(5): 301–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Van Baarlen P, Troost F, van der Meer C, Hooiveld G, Boekschoten M, Brummer RJ, Kleerebezem M. Human mucosal in vivo transcriptome responses to three lactobacilli indicate how probiotics may modulate human cellular pathways. Proc Natl Acad Sci U S A. 2011; 108: 4562–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Nicholson JK, Holmes E, Lindon JC, Wilson ID. The challenges of modeling mammalian biocomplexity. Nat Biotechnol. 2004; 22(10): 1268–74. [DOI] [PubMed] [Google Scholar]
- 73.Andrews CN, Griffiths TA, Kaufman J, et al. Mesalazine (5-aminosalicylic acid) alters faecal bacterial profiles, but not mucosal proteolytic activity in diarrhoea-predominant irritable bowel syndrome. Aliment Pharmacol Ther. 2011; 34(3): 374–83. [DOI] [PubMed] [Google Scholar]
- 74.Wang J, Ma J, Charboneau R, Barke R, Roy S. Morphine inhibits murine dendritic cell IL-23 production by modulating Toll-like receptor 2 and Nod2 signaling. J Biol Chem. 2011; 286(12): 10225–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Eisenstein TK. Opioids and the immune system: what is their mechanism of action? Br J Pharmacol. 20122; 164(7): 1826–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Koller MM, Purushotham KR, Maeda N, Scarpace PJ, Humphreys-Beher MG. Desipramine induced changes in salivary proteins, cultivable oral microbiota and gingival health in aging female NIA Fischer 344 rats. Life Sci. 2000; 68(4): 445–55. [DOI] [PubMed] [Google Scholar]
- 77.Macedo D, Filho AJ, Soares de Sousa CN, et al. Antidepressants, antimicrobials or both? Gut microbiota dysbiosis in depression and possible implications of the antimicrobial effects of antidepressant drugs for antidepressant effectiveness. J Affect Disord. 2016; 208: 22–32. [DOI] [PubMed] [Google Scholar]
- 78.Zhang EY, Xiong J, Parker BL, et al. Depletion and recovery of lymphoid subsets following morphine administration. Br J Pharmacol. 2001; 164: 1829–1844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Meng J, Banerjee S, Li D, et al. Opioid Exacerbation of Grampositive sepsis, induced by Gut Microbial Modulation, is Rescued by IL-17A Neutralization. Sci Rep. 2015a; 5: 10918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Meng J, Sindberg GM, Roy S. Disruption of gut homeostasis by opioids accelerates HIV disease progression. Front. Microbiol. 2015b; 6: 643–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Gardy JL, Johnston JC, Ho Sui SJ, et al. Whole-genome sequencing and social-network analysis of a tuberculosis outbreak. N Engl J Med 2011; 364(8): 730–9. [DOI] [PubMed] [Google Scholar]
- 82.Garmendia J, Morey P, Bengoechea JA. Impact of cigarette smoke exposure on host-bacterial pathogen interactions. Eur Respir J. 2012; 39(2): 467–77. [DOI] [PubMed] [Google Scholar]
- 83.Mutlu E, Keshavarzian A, Engen P, Forsyth CB, Sikaroodi M, Gillevet P. Intestinal dysbiosis: A possible mechanism of alcohol-induced endotoxemia and alcoholic steatohepatitis in rats. Alcoholism: Clin Exp Res 2009; 33: 1836–46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Liu C, Hu M, Guo H, Zhang M, Zhang J, Li F, Zhong Z, Chen Y, Li Y, Xu P, Li J, Liu L, Liu X. Combined Contribution of Increased Intestinal Permeability and Inhibited Deglycosylation of Ginsenoside Rb1 in the Intestinal Tract to the Enhancement of Ginsenoside Rb1 Exposure in Diabetic Rats after Oral Administration. Drug Metab Dispos. 2015, 43(11):1702–10. [DOI] [PubMed] [Google Scholar]
- 85.Semenkovich CF, Danska J, Darsow T, Dunne JL, Huttenhower C, Insel RA, McElvaine AT, Ratner RE, Shuldiner AR, Blaser MJ. American Diabetes Association and JDRF Research Symposium: Diabetes and the Microbiome. Diabetes. 2015, 64(12):3967–77. [DOI] [PMC free article] [PubMed] [Google Scholar]


