Abstract
Background
Escherichia coli is one of the most common strains of extended-spectrum β-lactam (ESBL)-producing bacteria, and the prevention and treatment of ESBL-producing E. coli infections is an ongoing challenge. The clinical characteristics and outcomes of ESBL-producing E. coli bacteremia in non-transplant patients remain to be elucidated.
Methods
This retrospective study included 491 non-transplant patients with E. coli bloodstream infections (BSIs) from January 2013 to December 2016 and was conducted to investigate the risk factors, clinical features, and outcomes of these infections.
Results
Of the 491 E. coli BSI patients, 57.6% suffered from infections with ESBL-producing strains. A multivariate analysis showed that urinary tract infection, prior use of cephalosporin, and treatment with β-lactam-β-lactamase inhibitor (BLBLI) combination antibiotics were independent risk factors for the development of ESBL-producing E. coli BSIs. The overall mortality rate in E. coli BSI patients was 14.46%, and there was no significant difference in the 28 day mortality rate between ESBL-producing E. coli and non-ESBL-producing E. coli BSI patients (14.8% vs. 14.0%, respectively; P = 0.953). Similarly, there was no difference between the community-acquired infection group and the nosocomial infection group. Hepatobiliary disease, carbapenem exposure, high APACHE II score, and hypoproteinemia were independent risk factors for death in E. coli BSI patients. Multivariate analysis showed that hypoproteinemia and severe disease were independent risk factors for death from ESBL-producing E. coli BSIs. Furthermore, there was no significant difference in the 28 day mortality between patients with ESBL-producing E. coli BSIs treated with carbapenem monotherapy versus those treated with BLBLI combination antibiotics (12.8% vs. 17.9%, respectively; P = 0.384).
Conclusions
Prior use of cephalosporin or BLBLI combination antibiotics increased the risk ratio for ESBL-producing E. coli infection. Hypoproteinemia and severe disease are independent risk factors for death in patients with E. coli BSIs. There was no significant difference in the 28 day prognosis of patients with ESBL-producing E. coli and those with non-ESBL-producing E. coli BSIs. These data do not support the conclusion that carbapenems might be more effective than BLBLI antibiotics for treatment of patients with BSIs caused by ESBL-producing E. coli.
Electronic supplementary material
The online version of this article (10.1186/s13756-019-0599-y) contains supplementary material, which is available to authorized users.
Keywords: Non-transplantation, Extended-spectrum β-lactamase, Escherichia coli, Bloodstream infection, Prognosis
Background
Escherichia coli is one of the most common strains of extended-spectrum β-lactamase (ESBL)-producing bacteria [1]. Since the first ESBL-producing bacterial strains were discovered in the 1980s, new genotypes have frequently emerged [2]. In the past 10 years, the detection rate of ESBL-producing E. coli has continued to increase dramatically worldwide. In Asia, and particularly in China, the prevalence rate is much higher than that in Western developed countries [3, 4]. The production of β-lactamase enzymes is the principal mechanism by which Gram-negative bacteria resist the action of β-lactam antibiotics. According to relevant domestic monitoring data, E. coli accounted for the largest proportion of resistant bacteria detected in China, followed by K. pneumoniae [5].
Previous studies have shown that prior use of broad-spectrum antibiotics, poor general condition, advanced age, and some invasive procedures (such as central venous catheterization) are risk factors for ESBL-producing E. coli infection [6, 7]. For patients with ESBL-producing E. coli bloodstream infections (BSIs), improved prognoses depend on a detailed understanding of the factors that increase mortality. Increased mortality due to inappropriate antibiotic treatment (whether empirical or definite) has been widely reported in previous studies. Furthermore, other risk factors (e.g., poor general health condition, liver disease, septic shock, and antibiotic exposure) prior to infection have also been reported in the literature [8, 9].
ESBL-producing E. coli represents a serious public health issue and infection control challenge, and the selection of appropriate treatments for ESBL-producing E. coli BSIs remains controversial as there are insufficient large-scale randomized controlled trials to support various therapeutic approaches. Carbapenem antibiotics are still considered to be first-line antibiotics for treating ESBL-producing E. coli BSIs [10]. However, in recent years, widespread use of these antibiotics has driven the development of carbapenem-resistant Enterobacteriaceae (CRE). Available monitoring data showed that the resistance rate of Enterobacteriaceae to β-lactam antibiotics such as cefotaxime, ceftazidime, cefepime, and piperacillin-tazobactam decreased from 2005 to 2014, while the meropenem (2005, 2.8%; 2014, 4.5%) and ertapenem (2005, 5.5%; 2014, 8.9%) resistance rates showed an increasing trend [11]. Currently, a growing number of experts believe that there is a wealth of data showing that the effectiveness of treatment with β-lactam-β-lactamase inhibitor (BLBLI) combination antibiotics is similar to that of carbapenems in patients infected with ESBL-producing strains. Antibiotic drugs such as cephalosporins and BLBLI combination antibiotics (e.g., piperacillin-tazobactam) have once again gradually attracted attention.
It is worth noting that ESBL-producing E. coli bacteremia occurs often in solid organ transplant recipients, which may be due to their frequent hospitalization, longer hospital stays, and long-term exposure to immunosuppressive agents. Moreover, some studies have shown that these patients have higher ESBL-producing E. coli infection and mortality rates [12–14]. To the best of our knowledge, no studies have analyzed the risk and prognosis of non-transplanted ESBL-producing E. coli BSIs; therefore, in this study, patients with solid organ and hematopoietic stem cell transplants were excluded.
This retrospective analysis was designed to investigate the clinical characteristics of non-transplant patients with ESBL-producing E. coli infections as well as to analyze the risk and prognostic factors and the therapeutic effects of different antibiotic regimens with the goals of strengthening our understanding of ESBL-producing E. coli infections and providing new guidance for clinical practice.
Methods
Population
This study retrospectively analyzed the clinical and microbiological data of patients with BSIs caused by Escherichia coli in the First Affiliated Hospital of Zhejiang University from January 2013 to December 2016 after receiving approval from the research ethics committee. Data from patients with the following characteristics were included in the analysis: a) a blood culture positive for E. coli, b) clinical manifestations of infection, and c) hospitalization for more than 48 h with a complete clinical data set. Patients aged < 16 years and those without complete medical records were excluded. Patients with histories of organ or hematopoietic stem cell transplantation before the BSI and those with carbapenem-resistant E. coli infections were excluded. If the same patient had more than one BSI within 6 months, only data from the first BSI were included. Ultimately, based on the inclusion and exclusion criteria, 491 patients were included in the study.
Bacterial identification and drug sensitivity testing
The VITEK 2 COMPACT automatic microbial identification system was used for bacterial identification and drug susceptibility testing. According to the guidelines of the Clinical and Laboratory Standards Institute (CLSI) standards (2015) [15], ESBL production was determined using a double-disk potentiation test with amoxicillin-clavulanic acid and cefotaxime, ceftazidime, or cefepime or ESBL-positive results from the VITEK-2 N131 analysis. Carbapenem resistance was defined as a minimum inhibitory concentration (MIC) of ≥2 mg/L for ertapenem or a MIC of ≥4 mg/L for imipenem or meropenem.
Data collection
Demographic and clinical data were collected from the electronic case system, including age, gender, underlying disease, comorbidities, length of hospital stay, admission to the ICU, invasive procedures before and after infection, and antibiotic treatments. The severity of the disease was assessed via the Acute Physiology and Chronic Health Evaluation (APACHE) II scores and Pitt scores [16]. The Charlson index was used to assess the burden of any comorbidities [17]. A three-part analysis was conducted. First, risk factors associated with ESBL-producing E. coli infection were evaluated by comparing the ESBL-producing and non-ESBL-producing patient groups. Second, to investigate the risk factor of mortality, the 491 E. coli BSI patients were divided into survival and death groups according to their survival status after 28 days of infection. Finally, 283 patients with ESBL-producing E. coli BSIs were analyzed to assess the risk factors associated with 28 day mortality and various antibiotic treatments.
Definitions
E. coli BSI was defined as an infection manifested by the presence in at least one blood culture that grew a E. coli strain. BSI refers to the systemic inflammatory response syndrome caused by toxins and metabolites produced by pathogens. Possible sources of E. coli BSIs are based on the Centers for Disease Control (CDC) and Prevention/National Healthcare Safety Network (NHSN) surveillance definitions [18]. Glucocorticoid therapy was defined as prednisolone > 20 mg/day for more than 7 days. Antimicrobial drug exposure referred to the use of antibiotics for > 72 h 30 days prior to the BSI diagnosis. Empirical therapy was defined as a therapeutic drug administered at the time of the blood culture test or before the blood culture report based on clinical experience. Definitive therapy referred to antimicrobial therapy administered based on drug susceptibility results. Treatments were classified as “appropriate” if the regimen contained at least one drug effective against E. coli; otherwise, they were classified as “inappropriate”. All-cause mortality was defined as death from an E. coli BSI within 7, 14 and 28 days of the onset of bacteremia.
Data analysis
In the univariate analysis of ESBL-producing E. coli BSIs, the χ2 or Fisher’s two-tailed test was used for categorical variables, and Student’s t test (for variables with normal distributions) and the Mann-Whitney U test (for variables with non-normal distributions) were used for continuous variables. For continuous variables, the results are expressed as mean ± standard deviation (SD) or median (interquartile range [IQR]). For categorical variables, the percentages for each group are reported. Variables with P values ≤0.05 in the univariate analysis were included in the multivariate analysis, and binary logistic regression (backward: condition) was used to identify independent predictors. The survival distribution functions in different groups were compared via the Kaplan-Meier product limit method. All data were statistically analyzed using SPSS version 23.0, and P values ≤0.05 were considered statistically significant.
Results
Clinical characteristics
From January 2013 to December 2016, 491 patients with E. coli BSIs were included in this study, except for 67 patients who received solid organ or hematopoietic stem cell transplants and 22 patients with CRE BSIs. Among the included patients, 283 (57.6%) had ESBL-producing E. coli BSIs and 208 (42.4%) had non-ESBL-producing E. coli BSIs. There were no significant differences in age and gender between the two groups (60.8 ± 16.7 vs. 60.5 ± 16.3, P = 0.836; male, 112 vs. 150, P = 0.853). Of the primary infection sites of the 491 patients with E. coli BSIs, abdominal infections were the highest (n = 249), followed by respiratory infections (n = 108) and urinary tract infections (n = 84). The annual incidence rates of ESBL-producing E. coli BSIs from 2013 to 2016 were 66.2% (86/130), 59.1% (88/149), 49.1% (53/108), and 53.8% (48/104), respectively, and while the incidence rate fluctuated, it did not increase significantly (P = 0.638).
Risk factors for ESBL-producing E. coli BSIs
A univariate analysis (Table 1) showed that risk factors for ESBL-producing E. coli BSIs included urinary system infection, prior surgery, prior invasive procedures (central venous catheterization, gastric catheterization, and percutaneous catheterization), and antibiotic use within 30 days before infection (mainly cephalosporins and BLBLI combination regimens). In a multivariate analysis, logistic regression analysis showed that urinary tract infections were the primary site of E. coli BSIs (OR = 1.897, P = 0.014) and that cephalosporin exposure (OR = 2.767, P = 0.007) and treatment with BLBLI combination regimens in the 30 days prior to the BSI (OR = 1.950, P = 0.010) were independent risk factors for ESBL-producing E. coli BSIs.
Table 1.
Clinical and Demographic Characteristics of Patients with BSI Caused by E. coli
| Univariate analysis | Multivariable analysis | ||||||
|---|---|---|---|---|---|---|---|
| Non- ESBL- producing E. coli | ESBL- producing E. coli | P-values | P-values | OR | 95% CI for OR | ||
| (n = 208) | (n = 283) | Lower | Upper | ||||
| Demographic | |||||||
| Male, n (%) | 112 (53.8) | 150 (53.0) | 0.853 | ||||
| Age, mean ± SD | 60.8 ± 16.7 | 60.5 ± 16.3 | 0.836 | ||||
| Total hospital stay, days (median, IQR) | 19 (11–28) | 18 (11–37) | 0.188 | ||||
| Hospital stay before BSI, days (median, IQR) | 3 (0–13) | 4 (1–14) | 0.336 | ||||
| ICU stay prior to BSIa | 11 (5.3) | 25 (8.8) | 0.136 | ||||
| Preexisting medical conditions | |||||||
| Hypertension | 58 (27.9) | 80 (28.3) | 0.925 | ||||
| Diabetes | 34 (16.4) | 45 (15.9) | 0.876 | ||||
| Lung disease | 2 (1.0) | 6 (2.1) | 0.477 | ||||
| Cardiovascular diseases | 4 (1.9) | 7 (2.5) | 0.921 | ||||
| Hepatobiliary disease | 59 (28.4) | 78 (27.6) | 0.844 | ||||
| Urinary system disease | 15 (7.2) | 25 (8.8) | 0.516 | ||||
| Nervous system disease | 7 (3.4) | 8 (2.8) | 0.732 | ||||
| Malignant solid tumor | 43 (20.7) | 63 (22.3) | 0.673 | ||||
| Hematological Disease | 39 (18.8) | 35 (12.4) | 0.051 | ||||
| Charlson comorbidity score c (median, IQR) | 2 (0–2) | 2 (0–2) | 0.168 | ||||
| Likely source of BSI | |||||||
| Central vein Catheter-related infections | 7.2 (7.2) | 10 (3.5) | 0.067 | ||||
| Lung infection | 52 (25.0) | 56 (19.8) | 0.168 | ||||
| Abdominal infection | 108 (51.9) | 141 (49.8) | 0.646 | ||||
| Urinary infection | 26 (12.5) | 58 (20.5) | 0.020 | 0.014 | 1.897 | 1.138 | 3.164 |
| Intracranial infection | 1 (0.5) | 4 (1.4) | 0.402 | ||||
| Skin infection | 6 (2.9) | 11(3.9) | 0.548 | ||||
| Primary bloodstream infection | 19 (9.1) | 26 (9.2) | 0.984 | ||||
| Nosocomial-acquired infection | 124 (59.6) | 180 (63.6) | 0.368 | ||||
| Hospitalization prior to BSIb | 75 (56.0) | 110 (59.1) | 0.571 | ||||
| Surgery prior to BSIa | 27 (13.0) | 56 (19.8) | 0.047 | ||||
| Invasive procedure and/or devices prior to BSIa | 46 (22.1) | 83 (29.3) | 0.073 | ||||
| Mechanical ventilation | 7 (3.4) | 11 (3.9) | 0.761 | ||||
| Central venous catheterization | 20 (9.6) | 42 (14.8) | 0.085 | ||||
| Urinary catheterization | 18 (8.7) | 42 (14.8) | 0.039 | ||||
| Gastric catheterization | 10 (4.8) | 31(11.0) | 0.015 | ||||
| Percutaneous catheterization | 6 (2.9) | 21 (7.4) | 0.029 | 0.101 | 2.218 | 0.857 | 5.742 |
| Hemodialysis prior to BSIa | 6 (2.9) | 3 (1.1) | 0.251 | ||||
| Chemotherapy or radiotherapy prior to BSIa | 38 (18.3) | 39 (13.8) | 0.177 | ||||
| Corticosteroid use prior to BSIa | 15 (7.2) | 21 (7.4) | 0.930 | ||||
| Immunosuppressant use prior to BSIa | 1 (0.5) | 2 (0.7) | > 0.050 | ||||
| Antibiotics use prior to BSIa | 68 (32.7) | 134 (47.3) | 0.001 | ||||
| Cepholosporins | 10 (4.8) | 38 (13.4) | 0.001 | 0.007 | 2.767 | 1.325 | 5.779 |
| BLBLI combination antibiotics | 26 (12.5) | 62 (21.9) | 0.007 | 0.010 | 1.950 | 1.173 | 3.241 |
| Tigecycline | 2 (1.0) | 3 (1.1) | > 0.050 | ||||
| Carbapenems | 17 (8.2) | 20 (7.1) | 0.646 | ||||
| Aminoglycosides | 1 (0.5) | 2 (0.7) | > 0.050 | ||||
| Quinolones | 26 (12.5) | 36 (12.7) | 0.942 | ||||
| Laboratory examination | |||||||
| White blood cell | 8.1 (3.8–13.0) | 9.6 (5.5–14.8) | 0.045 | ||||
| Platelet (median, IQR) | 104 (42–183) | 117 (58–192) | 0.161 | ||||
| Serum total protein (median, IQR) | 57.7 (52.0–64.7) | 58.3 (52.3–64.3) | 0.949 | ||||
| Severity of illness | |||||||
| APACHEII score (median, IQR) | 9 (6–12) | 9 (6–13) | 0.540 | ||||
| Appropriate empirical treatment after BSI | 200 (96.2) | 249 (88.3) | 0.001 | ||||
| Appropriate definitive treatments after BSI | 204 (98.1) | 267 (94.3) | 0.039 | ||||
| 7-day mortality | 17 (8.2) | 24 (8.1) | 0.985 | ||||
| 14-day mortality | 23 (11.1) | 28 (9.9) | 0.676 | ||||
| 28-day mortality | 29 (13.9) | 42 (14.8) | 0.780 | ||||
Data are expressed as n (%) unless otherwise stated
Abbreviations: ESBL extended-spectrum beta-lactamase; β-lactam-β-lactamase inhibitor (BLBLI) combination antibiotics, APACHE acute physiology and chronic health evaluation, BSI bloodstream infection, ICU intensive care unit, IQR interquartile range, SD standard deviation
aDuring the 30 days preceding BSI onset
bDuring the 3 months preceding BSI onset
cAt time of BSI onset
Risk factors affecting the mortality of patients with E. coli infection
The non-transplanted patients with E. coli BSIs were classified into survivor and death groups based on the outcome at 28 days. The overall all-cause 28 day mortality rate of the 491 patients was 14.46% (71/491). A survival curve analysis (Fig. 1) showed that the mortality of the patients in the ESBL-producing E. coli group was higher than that of the patients in the non-ESBL-producing E. coli group (14.9% vs. 14.2%, χ2 = 0.003, P = 0.953), and the difference between the mortalities was not statistically significant. Furthermore, the patients were divided into community infection and nosocomial infection groups (Additional file 1: Figure S1). In 187 community-infected BSI patients (8.4%), there was no statistically significant difference in the mortality between those with non-ESBL-producing E. coli infections and those with ESBL-producing E. coli infections (8.3% vs. 11.7%; χ2 = 0.472, P = 0.492). There was also no significant difference in the mortalities between these two groups in the patients with nosocomial infections (17.7% vs. 16.7%; χ2 = 0.240, P = 0.624).
Fig. 1.
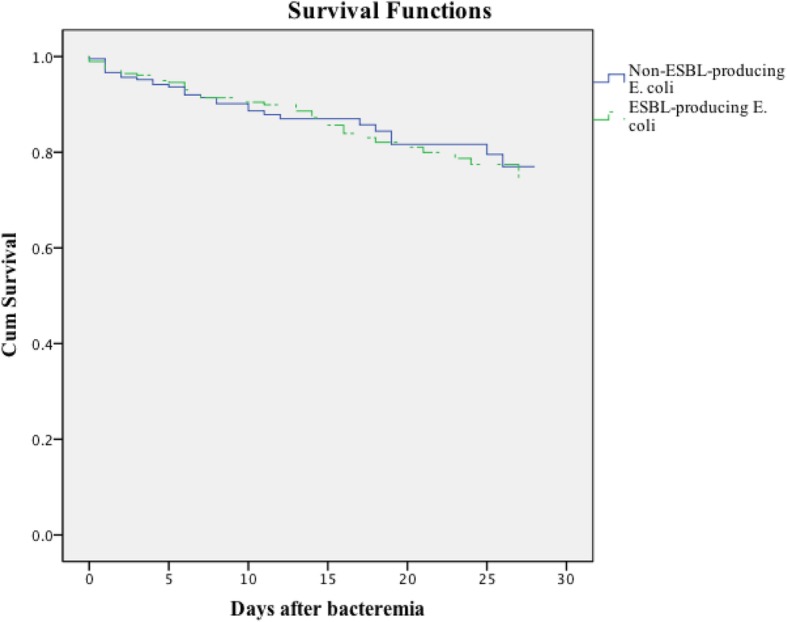
Kaplan-Meier survival estimates among patients with BSIs caused by non-ESBL-producing E. coli and ESBL-producing E. coli
The main characteristics of the E. coli BSI survivor and non-survivor subgroups are shown in Table 2. Based on a multivariate regression analysis, the following factors were independently associated with a higher mortality risk in patients with E. coli BSIs: hepatobiliary disease (OR = 1.890, P = 0.034), prior use of carbapenem antibiotics (OR = 2.839, P = 0.016), and higher APACHE II scores (OR = 1.313, P < 0.001). Normal serum total protein (OR = 0.959, P = 0.006) and appropriate definite antibiotic treatment were independent protective factors (OR = 2.777, P = 0.017) for the outcomes of patients with E. coli BSIs.
Table 2.
Analysis of risk factors for 28-day Mortality in 491 patients with E. coli bloodstream infections
| Univariate analysis | Multivariable analysis | ||||||
|---|---|---|---|---|---|---|---|
| Survivors (n = 420) | Non- survivors (n = 71) | P-values | P-values | OR | 95% CI for OR | ||
| Lower | Upper | ||||||
| Demographic | |||||||
| Male, n (%) | 223 (53.1) | 39 (54.9) | 0.774 | ||||
| Ages, mean ± SD | 60.8 ± 16.4 | 60.0 ± 16.8 | 0.736 | ||||
| Total hospital stay, days (median, IQR) | 20 (11–34) | 15 (7–28) | 0.013 | ||||
| Hospital stay before BSI, days (median, IQR) | 3 (0–13) | 7 (1–17) | 0.028 | ||||
| Preexisting medical conditions | |||||||
| Hypertension | 115 (27.4) | 23 (32.4) | 0.385 | ||||
| Diabetes | 71 (16.9) | 8 (11.4) | 0.249 | ||||
| Lung disease | 5 (1.2) | 3 (4.2) | 0.173 | ||||
| Cardiovascular diseases | 10 (2.4) | 1 (1.4) | 0.937 | ||||
| Hepatobiliary disease | 110 (26.2) | 27 (38.0) | 0.040 | 0.034 | 1.890 | 1.048 | 3.410 |
| Urinary system disease | 34 (8.1) | 6 (8.5) | 0.919 | ||||
| Nervous system disease | 13 (3.1) | 2 (2.8) | > 0.050 | ||||
| Malignant solid tumor | 86 (20.5) | 20 (28.2) | 0.145 | ||||
| Hematological Disease | 63 (15.0) | 11 (15.5) | 0.914 | ||||
| Charlson comorbidity score b (median, IQR) | 2 (0–2) | 2 (1–3) | 0.001 | ||||
| Source of infections | |||||||
| Central venous catheterization | 25 (6.0) | 0 (0) | 0.069 | ||||
| Lung infection | 82 (19.5) | 26 (36.6) | 0.001 | 0.071 | 1.781 | 0.951 | 3.336 |
| Abdominal infection | 212 (50.5) | 37 (52.1) | 0.799 | ||||
| Urinary infection | 74 (17.6) | 10 (14.1) | 0.464 | ||||
| Intracranial infection | 4 (1.0) | 1 (1.4) | 0.544 | ||||
| Skin infection | 15 (3.6) | 2 (2.8) | > 0.050 | ||||
| Primary bloodstream infection | 39 (9.3) | 6 (8.5) | 0.822 | ||||
| Nosocomial- acquired infection | 252 (60.0) | 52 (73.2) | 0.034 | ||||
| ESBL-producing E. coli, n (%) | 241 (57.4) | 42 (59.2) | 0.780 | ||||
| ICU stay prior to BSI a | 30 (7.1) | 6 (8.5) | 0.696 | ||||
| ICU stay after BSI c | 32 (7.6) | 11 (15.5) | 0.030 | ||||
| Prior surgery a | 69 (16.4) | 14 (19.7) | 0.494 | ||||
| Surgery after BSI c | 37 (8.8) | 2 (2.8) | 0.084 | ||||
| Invasive procedure and/or devices prior to BSI a | 106 (25.2) | 23 (32.4) | 0.205 | ||||
| Mechanical ventilation | 14 (3.3) | 4 (5.6) | 0.540 | ||||
| Central venous catheterization | 48 (11.4) | 14 (19.7) | 0.052 | ||||
| Urinary catheterization | 47 (11.2) | 13 (18.3) | 0.090 | ||||
| gastric catheterization | 33 (7.9) | 8 (11.3) | 0.337 | ||||
| Percutaneous catheterization | 24 (5.7) | 3 (4.2) | 0.820 | ||||
| Invasive procedure and/or devices after BSI c | 67 (16.0) | 13 (18.3) | 0.619 | ||||
| Mechanical ventilation | 29 (6.9) | 14 (19.7) | < 0.001 | ||||
| Central venous catheterization | 108 (25.7) | 32 (45.1) | 0.001 | ||||
| Urinary catheterization | 103 (24.5) | 27 (38.0) | 0.017 | ||||
| Gastric catheterization | 73 (17.4) | 19 (26.8) | 0.061 | ||||
| Percutaneous catheterization | 47 (11.2) | 7 (9.9) | 0.740 | ||||
| Hemodialysis prior to BSI a | 8 (1.9) | 1 (1.4) | > 0.050 | ||||
| Chemotherapy or radiotherapy prior to BSI a | 65 (15.5) | 12 (16.9) | 0.760 | ||||
| Corticosteroid use prior to BSI a | 28 (6.7) | 8 (11.3) | 0.169 | ||||
| Hemodialysis after BSI c | 14 (3.3) | 3 (4.2) | 0.977 | ||||
| Corticosteroid use after BSI c | 32 (7.6) | 6 (8.5) | 0.808 | ||||
| Prior Antibiotics use a | 164 (39) | 38 (53.5) | 0.022 | ||||
| Cepholosporins | 43 (10.2) | 5 (7.0) | 0.402 | ||||
| BLBLI combination antibiotics | 72 (17.1) | 16 (22.5) | 0.273 | ||||
| Tigecycline | 3 (0.7) | 2 (2.8) | 0.321 | ||||
| Carbapenems | 25 (6.0) | 12 (16.9) | 0.001 | 0.016 | 2.839 | 1.215 | 6.635 |
| Aminoglycosides | 2 (0.5) | 1 (1.4) | 0.375 | ||||
| Quinolones | 52 (12.4) | 10 (14.0) | 0.689 | ||||
| Laboratory examination b | |||||||
| White blood cell (median, IQR) | 9.1 (5.1–14.2) | 9.6 (3.7–13.3) | 0.849 | ||||
| Platelet (median, IQR) | 117 (52–192) | 80 (37–177) | 0.078 | ||||
| Total protein (median, IQR) | 58.9 (53.0–64.7) | 53.9 (46.0–62.0) | < 0.001 | 0.006 | 0.959 | 0.931 | 0.988 |
| Severity of illness at time of BSI b | |||||||
| APACHEII score (median, IQR) | 9 (6–12) | 13 (9–17) | < 0.001 | < 0.001 | 1.131 | 1.071 | 1.195 |
| Appropriate empirical treatment after BSI c | 389 (92.6) | 60 (84.5) | 0.024 | ||||
| 1. Cepholosporins | 25 (6.0) | 3 (4.2) | 0.761 | ||||
| 2. BLBLI combination antibiotics | 150 (35.7) | 25 (35.2) | 0.935 | ||||
| 3. Carbapenems | 189 (45.0) | 34 (47.9) | 0.651 | ||||
| 4. Quinolones | 49 (11.7) | 6 (8.5) | 0.427 | ||||
| 5. Aminoglycosides | 12 (2.9) | 2 (2.8) | > 0.050 | ||||
| 6. Tigecycline | 5 (1.2) | 2 (2.8) | 0.598 | ||||
| Appropriate definitive treatments after BSI c | 407 (96.9) | 64 (90.1) | 0.008 | 0.017 | 2.777 | 1.198 | 6.437 |
Data are expressed as n (%) unless otherwise stated
Abbreviations: ESBL extended-spectrum beta-lactamase; β-lactam-β-lactamase inhibitor (BLBLI) combination antibiotics, APACHE acute physiology and chronic health evaluation, BSI bloodstream infection, ICU intensive care unit, IQR interquartile range, SD standard deviation
aDuring the 30 days preceding BSI onset
bAt time of BSI onset
cAfter BSI onset
Risk factors for mortality in ESBL-producing E. coli BSIs and treatment regimens
A total of 283 ESBL-producing E. coli BSI patients were included in this analysis, and the 28 day mortality was 14.8%. In the univariate analysis (Additional file 2: Table S1), pulmonary infection, invasive procedures and/or device implementation before and after development of a BSI, antibiotic use within 30 days prior to infection, hypoproteinemia, and higher APACHE II scores were risk factors for increased mortality in patients with ESBL-producing E. coli BSIs. A multivariate analysis revealed that hypoproteinemia (OR = 0.941, P = 0.045) and higher APACHE II scores (OR = 1.103, P = 0.003) were independent risk factors for poor outcomes.
As shown in Additional file 2: Table S1, of the patients infected with ESBL-producing E. coli, 87.99% (249/283) received appropriate empirical antibiotic therapy and 94.35% (267/283) received appropriate definite antibiotic therapy. Appropriate empirical antibiotic treatment was a protective factor in the univariate analysis, and the rate of patients receiving appropriate empirical antibiotic treatment in the death group was significantly lower than that in the survival group (78.2% vs. 89.6%, P = 0.042). Based on our multivariate analysis, appropriate empirical antibiotic treatment also appeared to be a protective factor that can reduce the mortality rate, although this effect was marginally significant (OR = 2.526, P = 0.058).
In terms of empirical treatment, 117 patients with ESBL-producing E. coli BSIs were treated with carbapenem monotherapy, and 95 patients were treated with BLBLI combination monotherapy regimens. Figure 2 shows that a single-antibiotic regimen with carbapenem (n = 117) or a BLBLI regimen (n = 95) resulted in no differences in 28 day mortality between the two groups (12.8% vs. 17.9%; χ2 = 0.759, P = 0.384), and their APACHE II scores were 10 and 9, respectively. Considering the potential confounding effect of severity, the patients were divided into two groups according to their APACHE II scores (< 9 and ≥ 9), and the difference in the prognoses of the two groups was compared (Additional file 1: Figure S2). For patients with APACHE II scores ≥9 at the onset of bacteremia, there was no statistically significant difference in the 28 day mortality rate between the patients who received carbapenem and those who received BLBLI monotherapy (16.4% vs. 22.4%; χ2 = 0.249, P = 0.617); similarly, no significant difference was observed between the two groups in patients with APACHE II scores < 9 (6.8% vs. 10.8%; χ2 = 0.694, P = 0.405).
Fig. 2.
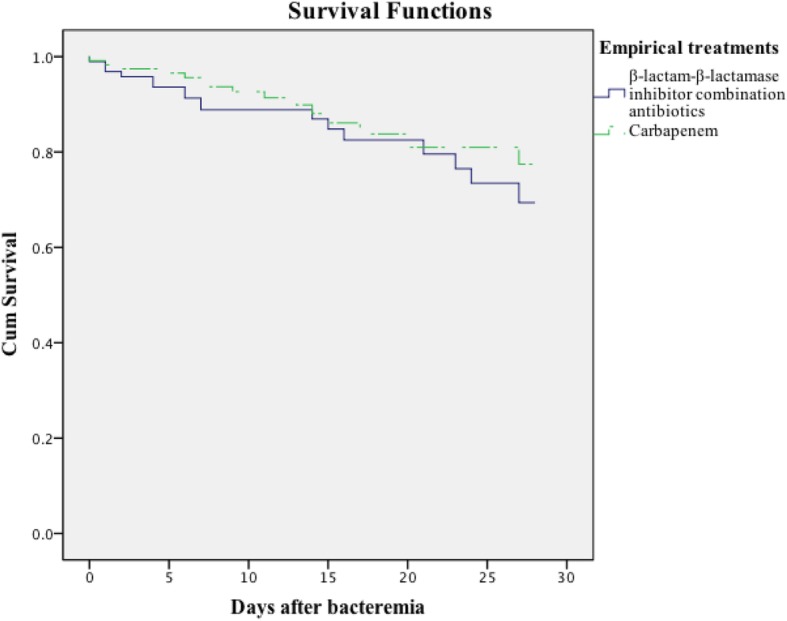
Kaplan-Meier 28 day survival estimates among ESBL-producing E. coli BSI patients depending on treatment with carbapenem or β-lactam-β-lactamase inhibitor (BLBLI) combination antibiotics
Discussion
Escherichia coli is one of the most common pathogens in clinical infections. The primary mechanism of drug resistance in these bacteria is the production of ESBLs, and this mechanism underlies a high proportion of the antibiotic resistance cases in China [5]. This study focused on studying the risk factors associated with ESBL-producing E. coli BSIs in non-transplanted patients.
Antimicrobial use prior to BSI is believed to be an important factor in drug-resistant infections [19, 20], although some studies have shown no association between ESBL-producing E. coli infections and prior antibiotic therapy; for example, Denis et al. reported that there was no significant difference in the prevalence of antibiotic exposure between the ESBL-producing E. coli and non-ESBL-producing E. coli groups, which included 82 patients (P = 0.32) [21]. Our results also demonstrated that cephalosporins and BLBLI combination regimens within 30 days prior to infection were independent risk factors for ESBL-producing E. coli BSIs, which is consistent with the findings of many previous studies. Regarding the likely infectious source of E. coli BSIs, intra-abdominal infections were most common, followed by respiratory tract infections and urinary tract infections. However, most studies showed that ESBL-producing E. coli strains are primarily derived from urinary tract infections [7, 9, 22], followed by respiratory and bloodstream infections. Furthermore, we found that patients with nosocomial infections accounted for 50% of the patients with E. coli urinary tract infections (n = 29) and 66% of the patients with intra-abdominal infections (n = 93); therefore, this significant difference between the frequencies of patients with infections in these two sites is likely due to a higher likelihood of acquiring an abdominal infection in the hospital. Based on this observation, we suspect that the reason for the high proportion of intra-abdominal infections in this study may be related to the high number of patients undergoing hepatology-related procedures and hepatobiliary surgery in our hospital. It is well known that E. coli is an opportunistic bacterial pathogen that can invade the body when invasive procedures disrupt the mucosa. The multivariate analysis of the risk factors associated with ESBL-producing E. coli in this study showed that urinary tract infection was an independent risk factor (P = 0.014), consistent with previous studies [23–25]. Currently, ESBL-producing E. coli BSIs originating from urinary tract infections are more commonly studied, while articles focusing on abdominal infections are rare, suggesting a further research direction.
To explore the high mortality rate associated with E. coli BSIs, we evaluated the patient characteristics and the treatments they received. In this study, there was no significant difference in the 28 day mortality between patients infected with ESBL-producing E. coli and those infected with non-ESBL-producing E. coli (P = 0.953), similar to the findings of some previous studies [21, 26]. Conversely, other studies demonstrated significantly higher mortality in patients infected with ESBL-producing E. coli than in those infected with non-ESBL-producing E. coli [6]. A possible reason for the similar mortality rates in the two groups in the present study is the use of many broad-spectrum antibiotics due to the current high prevalence of ESBL-producing E. coli. For patients with end-stage liver disease, bacterial infection is one of the most common causes of death [27], which explains the observation that hepatobiliary system disease was an independent risk factor for mortality in the present study. Furthermore, an analysis of patients with potential liver disease and E. coli infection by Kang et al. yielded similar results [28]. Our analysis showed that disease severity is also an independent risk factor for mortality in patients with ESBL-producing E. coli BSIs, which is also similar to the results of previous studies [29]. We also found that low serum albumin is an independent risk factor for death in patients with E. coli and ESBL-producing E. coli BSIs. Serum albumin level is a common indicator for assessing a patient’s nutritional status, organ function, and comorbidity. The inflammatory state resulting from bacterial infection, which leads to the production of IL-1, TNF, and other cell mediators, can interfere with liver albumin synthesis, resulting in hypoalbuminemia [30]. There is currently a lack of literature on the relationship between mortality and serum albumin level in patients with E. coli infections. Akirov et al. [31] studied the relationship between serum albumin levels and prognosis in hospitalized patients (without any detailed division) and found that low albumin levels were positively correlated with short- and long-term mortality in the entire hospitalized patient population. Albumin levels are often lower in patients with hepatic insufficiency, which was also associated with higher mortality in the patients with hepatobiliary disease included in this study. Du et al. assessed the outcomes of 85 patients with E. coli infections and found that prior use of BLBLI combination antibiotics increased the mortality rate [32]. In the present study, we also found that use of carbapenems within 30 days prior to infection was an important independent risk factor for the patients who died, which has been rarely reported previously. Previous studies [33] have shown that short-term use of such antibiotics before infection may also lead to the production of and infection by carbapenem-resistant bacteria. These outcomes increase the complexity of treating critical infections, which might explain the increased mortality observed in this study.
In terms of the antibiotic options for treating E. coli BSIs, we believe that appropriate, defined antibiotic treatments are a protective factor for reducing death due to E. coli BSIs, as reported in this study (OR = 2.777, P = 0.017), and this observation is consistent with those of some other studies [7, 34]. The findings of attempts to determine the best treatment options for ESBL-producing E. coli have been inconclusive, although carbapenems have long been considered the best antibiotic for treating infections with ESBL-producing E. coli [35]. However, the wide use of carbapenems has been associated with the emergence of CRE. In recent years, the efficacy advantages of carbapenems over other antibiotics have been increasingly questioned. The latter antibiotics include BLBLI combination antibiotics, cefepime, and quinolones (among others), and the feasibility of using the above-mentioned older antibiotics to treat ESBL-producing E. coli infections is becoming more and more accepted by researchers [36–38]. We analyzed the outcomes of 283 patients with ESBL-producing E. coli BSIs, including 95 patients with BLBLI combination monotherapy regimens and 117 with carbapenem monotherapy (median APACHE II 9 and 10). Their 28 day mortality rates were 12.8 and 17.9%, respectively, and the difference was not statistically significant (χ2 = 0.759, P = 0.384). Because disease severity may affect prognoses, we divided the ESBL-producing E. coli BSI patients into two groups according to their APACHE II scores (< 9, ≥ 9) and assessed their 28 day mortality rates. We found no statistically significant difference in 28 day mortality between the two groups (Additional file 1: Figure S2 P = 0.405; P = 0.617), which confirms that carbapenem antibiotics are no longer more effective than BLBLI combination antibiotics against ESBL-producing E. coli BSIs. These findings are consistent with previous reports that proposed the use of BL/BLIs to treat ESBL-producing E. coli BSIs [22, 39]. For example, Muhammed et al. [39] conducted a meta-analysis of more than 1000 patients in 14 relevant articles and found that there was no significant difference in prognosis between patients treated with carbapenems and those treated with BLBLI combination antibiotics for either empirical or definite antibiotic treatment. However, some reports support the opposite opinion that carbapenem antibiotics can significantly reduce mortality compared with other antibiotics [40]. Currently, as stated above, it remains unclear in all of the relevant studies whether carbapenems were primarily administered to patients with more comorbidities or to those with more severe clinical conditions.
Regarding the limitations of this study, our findings are based mainly on retrospective data and studies, which makes it impossible to eliminate certain types of bias. Furthermore, while this study excluded patients with solid organ and hematopoietic stem cell transplantation, some patients with underlying diseases may have been treated with immunosuppressive agents. Finally, we were unable to perform further stratification of the outcomes based on patient genotyping due to a lack of phenotypic testing. In this context, we should perform more detailed drug resistance gene detection and drug susceptibility testing in ESBL-producing E. coli BSI patients to better understand the drug resistance profiles involved as well as the relevant treatment options.
Conclusion
In summary, prior exposure to cephalosporin and BLBLI combination antibiotics increases the risk of acquiring ESBL-producing E. coli infections. Prior carbapenem use and a poor systemic condition were risk factors for increased mortality in patients with ESBL-producing E. coli and non-ESBL-producing E. coli BSIs; however, no significant difference was found in the prognoses of patients with ESBL-producing E. coli and non-ESBL-producing E. coli BSIs. Hypoproteinemia and severe disease lead to worse outcomes for patients with ESBL-producing E. coli BSIs. Carbapenem offered no significant advantage over BLBLI combination antibiotics for improving the 28 day mortality of patients with ESBL-producing E. coli BSIs.
Additional files
Figure S1. Kaplan-Meier survival estimates among non-transplant patients with BSIs caused by non-ESBL-producing E. coli and ESBL-producing E. coli. (A) Community-acquired infection. (B) Nosocomial-acquired infection. Figure S2. Kaplan-Meier 28 day survival estimates. (A) E. coli BSI patients (APACHE II score < 9) treated with carbapenem and β-lactam-β-lactamase inhibitor (BLBLI) combination antibiotics. (B) E. coli BSI patients (APACHE II score ≥ 9) treated with carbapenem and BLBLIs. (DOCX 122 kb)
Table S1. Analysis of risk factors for 28 day mortality in 283 patients with ESBL- producing E. coli BSI. (DOCX 31 kb)
Acknowledgments
The authors acknowledge the role of all support staff and participating patients in the study.
Abbreviations
- APACHE
Acute Physiology and Chronic Health Evaluation
- BLBLI
β-lactam-β-lactamase inhibitor combination antibiotics
- BSI
Bloodstream infection
- E. coli
Escherichia coli
- ESBL
Extended-spectrum beta-lactamase
- ICU
Intensive care unit
- IQR
Interquartile range
- SD
Standard deviation
Authors’ contributions
YHX conceived the study. TTX and KY were involved in the statistical analysis and drafting the manuscript. YZZ and TSZ participated in the study design and manuscript revision, JRJ, CQY and PS provided assistance with the bacterial identification. All authors agree to be accountable for all aspects of the work.
Funding
This work was partially supported by grants from the Key Research and Development Program of Zhejiang Province (2015C03032).
Availability of data and materials
Full datasets analysed during the current study are available from the corresponding author on reasonable request.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Tingting Xiao and Kai Yang contributed equally to this work.
References
- 1.Pitout JDD, Laupland KB. Extended-spectrum β-lactamase-producing Enterobacteriaceae: an emerging public-health concern. Lancet Infect Dis. 2008;8:159–166. doi: 10.1016/s1473-3099(08)70041-0. [DOI] [PubMed] [Google Scholar]
- 2.Doi Y, Iovleva A, Bonomo RA. The ecology of extended-spectrum beta-lactamases (ESBLs) in the developed world. J Travel Med. 2017;24:S44–S51. doi: 10.1093/jtm/taw102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Chong Y, Shimoda S, Shimono N. Current epidemiology, genetic evolution and clinical impact of extended-spectrum beta-lactamase-producing Escherichia coli and Klebsiella pneumoniae. Infect Genet Evol. 2018;61:185–188. doi: 10.1016/j.meegid.2018.04.005. [DOI] [PubMed] [Google Scholar]
- 4.Padmini N, Ajilda AAK, Sivakumar N, Selvakumar G. Extended spectrum beta-lactamase producing Escherichia coli and Klebsiella pneumoniae: critical tools for antibiotic resistance pattern. J Basic Microbiol. 2017;57:460–470. doi: 10.1002/jobm.201700008. [DOI] [PubMed] [Google Scholar]
- 5.Hu FP, Guo Y, Zhu DM, Wang F, Jiang XF, Xu YC. Antimicrobial resistance profile of clinical isolates in hospitals across China: report from the CHINET surveillance program, 2017. Chin J Infect Chem Other. 2018;18(3):241–25113. doi: 10.16718/j.1009-7708.2018.03.001. [DOI] [Google Scholar]
- 6.Chopra T, Marchaim D, Johnson PC, Chalana IK, Tamam Z, Mohammed M, et al. Risk factors for bloodstream infection caused by extended-spectrum beta-lactamase-producing Escherichia coli and Klebsiella pneumoniae: a focus on antimicrobials including cefepime. Am J Infect Control. 2015;43:719–723. doi: 10.1016/j.ajic.2015.02.030. [DOI] [PubMed] [Google Scholar]
- 7.Ortega M, Marco F, Soriano A, Almela M, Martinez JA, Munoz A, et al. Analysis of 4758 Escherichia coli bacteraemia episodes: predictive factors for isolation of an antibiotic-resistant strain and their impact on the outcome. J Antimicrob Chemoth. 2009;63:568–574. doi: 10.1093/jac/dkn514. [DOI] [PubMed] [Google Scholar]
- 8.Rodriguez-Bano J, Picon E, Gijon P, Hernandez JR, Ruiz M, Pena C, et al. Community-onset bacteremia due to extended-spectrum beta-lactamase-producing Escherichia coli: risk factors and prognosis. Clin Infect Dis. 2010;50:40–48. doi: 10.1086/649537. [DOI] [PubMed] [Google Scholar]
- 9.Hsieh CJ, Shen YH, Hwang KP, Implications C. Risk factors and mortality following community-onset bacteremia caused by extended-spectrum beta-lactamase (ESBL) and non-ESBL producing Escherichia coli. J Microbiol Immunol. 2010;43:240–248. doi: 10.1016/S1684-1182(10)60038-2. [DOI] [PubMed] [Google Scholar]
- 10.Gudiol C, Royo-Cebrecos C, Abdala E, Akova M, Alvarez R, Maestro-de la Calle G, et al. Efficacy of beta-lactam/beta-lactamase inhibitor combinations for the treatment of bloodstream infection due to extended-Spectrum-beta-lactamase-producing Enterobacteriaceae in hematological patients with neutropenia. Antimicrob Agents Chemother. 2017;61. 10.1128/AAC.00164-17. [DOI] [PMC free article] [PubMed]
- 11.Tian AL, Li GX, Elsheikha HM, Gardner DS, Zhang XY, Dong W, et al. Seroepidemiology of toxoplasma gondii infection in patients with liver disease in eastern China. Epidemiol Infect. 2017;145:2296–2302. doi: 10.1017/S0950268817001327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Aguiar EB, Maciel LC, Halpern M, de Lemos AS, Ferreira AL, Basto ST, et al. Outcome of bacteremia caused by extended-spectrum beta-lactamase-producing Enterobacteriaceae after solid organ transplantation. Transplant Proc. 2014;46:1753–1756. doi: 10.1016/j.transproceed.2014.05.003. [DOI] [PubMed] [Google Scholar]
- 13.Cornejo-Juarez P, Suarez-Cuenca JA, Volkow-Fernandez P, Silva-Sanchez J, Barrios-Camacho H, Najera-Leon E, et al. Fecal ESBL Escherichia coli carriage as a risk factor for bacteremia in patients with hematological malignancies. Support Care Cancer. 2016;24:253–259. doi: 10.1007/s00520-015-2772-z. [DOI] [PubMed] [Google Scholar]
- 14.Satlin Michael J., Walsh Thomas J. Multidrug-resistant Enterobacteriaceae, Pseudomonas aeruginosa , and vancomycin-resistant Enterococcus : Three major threats to hematopoietic stem cell transplant recipients. Transplant Infectious Disease. 2017;19(6):e12762. doi: 10.1111/tid.12762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Clinical and Laboratory Standards Institute . CLSI document M100-S25. 2015. Performance standards for antimicrobial susceptibility testing, 25th informational supplement. [Google Scholar]
- 16.Knaus WA, Draper EA, Wagner DP, Zimmerman JE. Apache-ii - a severity of disease classification-system. Crit Care Med. 1985;13:818–829. doi: 10.1097/00003246-198510000-00009. [DOI] [PubMed] [Google Scholar]
- 17.Charlson M, Szatrowski TP, Peterson J, Gold J. Validation of a combined comorbidity index. J Clin Epidemiol. 1994;47:1245–1251. doi: 10.1016/0895-4356(94)90129-5. [DOI] [PubMed] [Google Scholar]
- 18.Centers for Disease Control and Prevention. CDC/NHSN surveillance definitions for specific types of infections. Available at: http://www.cdc.gov/nhsn/PDFs/pscManual/17pscNosInfDef_current.pdf. Accessed 15 January 394 2014.
- 19.Reuland EA, al Naiemi N, Kaiser AM, Heck M, Kluytmans JAJW, Savelkoul PHM, et al. Prevalence and risk factors for carriage of ESBL-producing Enterobacteriaceae in Amsterdam. J Antimicrob Chemoth. 2016;71:1076–1082. doi: 10.1093/jac/dkv441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Park SY, Kang CI, Wi YM, Chung DR, Peck KR, Lee NY, et al. Risk factors and molecular epidemiology of community-onset, multidrug resistance extended-spectrum beta-lactamase-producing Escherichia coli infections. Korean J Intern Med. 2017;32:146–157. doi: 10.3904/kjim.2015.113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Denis B, Lafaurie M, Donay JL, Fontaine JP, Oksenhendler E, Raffoux E, et al. Prevalence, risk factors, and impact on clinical outcome of extended-spectrum beta-lactamase-producing Escherichia coli bacteraemia: a five-year study. Int J Infect Dis. 2015;39:1–6. doi: 10.1016/j.ijid.2015.07.010. [DOI] [PubMed] [Google Scholar]
- 22.Kerneis S, Valade S, Geri G, Compain F, Lavollay M, Rostane H, et al. Cefoxitin as a carbapenem-sparing antibiotic for infections caused by extended-spectrum beta-lactamase-producing Escherichia coli and Klebsiella pneumoniae. Infect Dis (Lond) 2015;47:789–795. doi: 10.3109/23744235.2015.1062133. [DOI] [PubMed] [Google Scholar]
- 23.Peleg AY, Hooper DC. Hospital-acquired infections due to gram-negative bacteria. N Engl J Med. 2010;362:1804–1813. doi: 10.1056/NEJMra0904124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Hertz FB, Schonning K, Rasmussen SC, Littauer P, Knudsen JD, Lobner-Olesen A, et al. Epidemiological factors associated with ESBL- and non ESBL-producing E. coli causing urinary tract infection in general practice. Infect Dis (Lond) 2016;48:241–245. doi: 10.3109/23744235.2015.1103895. [DOI] [PubMed] [Google Scholar]
- 25.Fan NC, Chen HH, Chen CL, Ou LS, Lin TY, Tsai MH, et al. Rise of community-onset urinary tract infection caused by extended-spectrum beta-lactamase-producing Escherichia coli in children. J Microbiol Immunol. 2014;47:399–405. doi: 10.1016/j.jmii.2013.05.006. [DOI] [PubMed] [Google Scholar]
- 26.Harris PN. Clinical management of infections caused by Enterobacteriaceae that express extended-spectrum beta-lactamase and AmpC enzymes. Semin Respir Crit Care Med. 2015;36:56–73. doi: 10.1055/s-0034-1398387. [DOI] [PubMed] [Google Scholar]
- 27.Nanchal RS, Ahmad S. Infections in liver disease. Crit Care Clin. 2016;32:411–41+. doi: 10.1016/j.ccc.2016.03.006. [DOI] [PubMed] [Google Scholar]
- 28.Kang CI, Wi YM, Ko KS, Chung DR, Peck KR, Lee NY, et al. Outcomes and risk factors for mortality in community-onset bacteremia caused by extended-spectrum beta-lactamase-producing Escherichia coli, with a special emphasis on antimicrobial therapy. Scand J Infect Dis. 2013;45:519–525. doi: 10.3109/00365548.2013.775479. [DOI] [PubMed] [Google Scholar]
- 29.Anunnatsiri S, Towiwat P, Chaimanee P. Risk factors and clinical outcomes of extended Spectrum Beta-lactamase (Esbl)-producing Escherichia Coli septicemia at Srinagarind University hospital, Thailand. Se Asian J Trop Med. 2012;43:1169–1177. [PubMed] [Google Scholar]
- 30.Arnau-Barres I, Guerri-Fernandez R, Luque S, Sorli L, Vazquez O, Miralles R. Serum albumin is a strong predictor of sepsis outcome in elderly patients. Eur J Clin Microbiol Infect Dis. 2019;38:743–746. doi: 10.1007/s10096-019-03478-2. [DOI] [PubMed] [Google Scholar]
- 31.Akirov A, Masri-Iraqi H, Atamna A, Shimon I. Low Albumin Levels Are Associated with Mortality Risk in Hospitalized Patients. Am J Med. 2017;130:1465 e11–1465 e19. doi: 10.1016/j.amjmed.2017.07.020. [DOI] [PubMed] [Google Scholar]
- 32.Du B, Long Y, Liu H, Chen D, Liu D, Xu Y, et al. Extended-spectrum beta-lactamase-producing Escherichia coli and Klebsiella pneumoniae bloodstream infection: risk factors and clinical outcome. Intensive Care Med. 2002;28:1718–1723. doi: 10.1007/s00134-002-1521-1. [DOI] [PubMed] [Google Scholar]
- 33.McLaughlin M, Advincula MR, Malczynski M, Qi C, Bolon M, Scheetz MH. Correlations of antibiotic use and carbapenem resistance in enterobacteriaceae. Antimicrob Agents Chemother. 2013;57:5131–5133. doi: 10.1128/AAC.00607-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Curello J, MacDougall C. Beyond susceptible and resistant, part II: treatment of infections due to gram-negative organisms producing extended-Spectrum beta-lactamases. J Pediatr Pharmacol Ther. 2014;19:156–164. doi: 10.5863/1551-6776-19.3.156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Harris PNA, Tambyah PA, Lye DC, Mo Y, Lee TH, Yilmaz M, et al. Effect of piperacillin-Tazobactam vs Meropenem on 30-day mortality for patients with E coli or Klebsiella pneumoniae bloodstream infection and ceftriaxone resistance: a randomized clinical trial. JAMA. 2018;320:984–994. doi: 10.1001/jama.2018.12163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Nguyen HM, Shier KL, Graber CJ. Determining a clinical framework for use of cefepime and beta-lactam/beta-lactamase inhibitors in the treatment of infections caused by extended-spectrum-beta-lactamase-producing Enterobacteriaceae. J Antimicrob Chemother. 2014;69(4):871–880. doi: 10.1093/jac/dkt450. [DOI] [PubMed] [Google Scholar]
- 37.Ng Tat Ming, Khong Wendy X., Harris Patrick N. A., De Partha P., Chow Angela, Tambyah Paul A., Lye David C. Empiric Piperacillin-Tazobactam versus Carbapenems in the Treatment of Bacteraemia Due to Extended-Spectrum Beta-Lactamase-Producing Enterobacteriaceae. PLOS ONE. 2016;11(4):e0153696. doi: 10.1371/journal.pone.0153696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Harris PNA, Tambyah PA, Paterson DL. β-Lactam and β-lactamase inhibitor combinations in the treatment of extended-spectrum β-lactamase producing Enterobacteriaceae: time for a reappraisal in the era of few antibiotic options? Lancet Infect Dis. 2015;15(4):475–485. doi: 10.1016/S1473-3099(14)70950-8. [DOI] [PubMed] [Google Scholar]
- 39.Muhammed M, Flokas ME, Detsis M, Alevizakos M, Mylonakis E. Comparison between Carbapenems and beta-lactam/beta-lactamase inhibitors in the treatment for bloodstream infections caused by extended-Spectrum beta-lactamase-producing Enterobacteriaceae: a systematic review and meta-analysis. Open Forum Infect Dis. 2017;4(2):ofx099. doi: 10.1093/ofid/ofx099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Vardakas KZ, Tansarli GS, Rafailidis PI, Falagas ME. Carbapenems versus alternative antibiotics for the treatment of bacteraemia due toEnterobacteriaceae producing extended-spectrum -lactamases: a systematic review and meta-analysis. J Antimicrob Chemoth. 2012;46(12):2793–2803. doi: 10.1093/jac/dks301. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Figure S1. Kaplan-Meier survival estimates among non-transplant patients with BSIs caused by non-ESBL-producing E. coli and ESBL-producing E. coli. (A) Community-acquired infection. (B) Nosocomial-acquired infection. Figure S2. Kaplan-Meier 28 day survival estimates. (A) E. coli BSI patients (APACHE II score < 9) treated with carbapenem and β-lactam-β-lactamase inhibitor (BLBLI) combination antibiotics. (B) E. coli BSI patients (APACHE II score ≥ 9) treated with carbapenem and BLBLIs. (DOCX 122 kb)
Table S1. Analysis of risk factors for 28 day mortality in 283 patients with ESBL- producing E. coli BSI. (DOCX 31 kb)
Data Availability Statement
Full datasets analysed during the current study are available from the corresponding author on reasonable request.


