Abstract
The risk of gastric cancer (GC) declines after Helicobacter pylori (H. pylori) eradication and long-term aspirin use. We evaluated the effects of H. pylori eradication (Cohort 1) and aspirin use (Cohort 2) on the methylation of microRNAs (miRNAs), such as miR-34c, miR-124a-3, miR-129-2, and miR-137, in the gastric mucosa with and without GC, i.e., in atrophic mucosal glands without intestinal metaplasia (non-IM) and intestinal metaplastic glands (IM). DNA was isolated from non-IM and IM separately using laser caption microdissection. In Cohort 1, H. pylori eradication was associated with a significant reduction of miR-124a-3 methylation only in non-IM, but not in IM. miR-129-2 methylation in non-IM may be a surrogate marker of GC in H. pylori-infected patients. In Cohort 2, aspirin did not reverse miRNA methylation in either non-IM or IM, irrespective of H. pylori infection. miR-129-2 methylation in non-IM was an independent predictive marker of GC in H. pylori-infected but not -eradicated patients. These results indicate that H. pylori eradication and aspirin use were less effective for improving methylation in IM than in non-IM; thus, these interventions are recommended at an early stage prior to the development of IM to prevent GC development. In addition, the effects of the interventions were not uniform for each miRNA gene.
Subject terms: Predictive markers, Gastric cancer, Molecular medicine, Cancer epigenetics, Risk factors
Introduction
Gastric cancer (GC) is a leading cause of cancer death worldwide1, with Helicobacter pylori (H. pylori) considered to be a main risk factor2. In Correa’s hypothesis, chronic infection with H. pylori has been postulated to develop over decades into chronic gastritis, gastric atrophy, usually intestinal metaplasia (IM), dysplasia, and GC2.
To date, some meta-analyses have shown that H. pylori eradication reduced the risk of GC not only in patients with chronic gastritis but also in patients who underwent endoscopic resection for early GC3–8. On the other hand, although several studies have been done regarding the effect of H. pylori eradication in preventing metachronous GC, the results remain controversial: some studies have shown that H. pylori treatment led to a lower incidence of metachronous GC9–11 and others have not12–14. Long-term studies from Japan showed that even after H. pylori eradication, the risk of developing GC remains, and the risk increases under the preneoplastic conditions of the background mucosa, i.e., gastric atrophic mucosa and IM15,16. These results indicate that H. pylori eradication treatment may reduce the risk of GC, though it may not abolish the risk.
Aspirin also has protective effects against certain cancers. Recent reports including meta-analyses have shown that long-term aspirin use (for at least more than 3 years) was associated with a reduced GC risk17–20. However, the majority of these studies included both H. pylori-infected and H. pylori-negative subjects. Cheung et al. showed that the risk significantly decreased with increasing frequency, duration, and dose of aspirin after H. pylori eradication21.
GC develops through the accumulation of genetic and epigenetic alterations. Many studies have reported that several epigenetic alterations, including promoter hypermethylation of multiple tumor-related genes, are associated with GC and precancerous conditions of the stomach that occur in the context of H. pylori infection22–34. These reports demonstrated that H. pylori eradication led to a decrease in methylation levels in some genes22–27, suggesting that the reduction of gene methylation reversed H. pylori-induced gastric carcinogenesis. However, there are no studies except ours33,34 and a study by Huang et al.32 in which molecular events were analyzed in atrophic mucosal glands without IM (non-IM) and with IM separately using laser capture microdissection (LCM); thus, the actual effects of eradication therapy on molecular alteration in patients with the precancerous conditions are not well known.
Currently, microRNAs (miRNAs) are considered to be actively involved in the development, differentiation, and pathogenesis of various malignancies. By comparing the miRNA expression profiles in tumor tissues versus adjacent non-tumor tissues, distinct patterns of up- or down-regulation of miRNAs were found in different types of cancers35. Therefore, these cancer-specific miRNA expressions could be used as molecular biomarkers for GC. Indeed, several studies showed that the dysgeneration of some miRNAs by methylation highlights the useful biomarkers of GC development29–31,35–41. Japanese investigators have recently reported that miR-124a-3 and miR-34b/c methylation are informative markers for predicting the risk of metachronous GC in patients after the endoscopic resection of early GC29,36,37. However, we reported that the incidences of the methylation of miR-124a-3 and miR-34c were mostly observed in IM, with very few in non-IM33,34. Thus, our previous results indicate that the methylation of these miRNA genes might be a specific marker expressed in IM and might not necessarily be a risk marker for GC.
Our aims in this study were: 1) to investigate the methylation changes of several miRNAs related to gastric carcinogenesis in patients before and after H. pylori eradication in patients not taking low-dose aspirin (LDA) or nonsteroidal anti-inflammatory drugs (NSAIDs) (Cohort 1); and 2) to examine the effects of LDA/NSAIDs on the methylation status of those miRNAs before and after H. pylori eradication in patients who had regularly taken LDA/NSAIDs on a long-term basis (≥3 yr) (Cohort 2) while exhibiting a precancerous condition, i.e., non-IM or IM.
Results
Patients’ characteristics
The characteristics of the patients are shown in Table 1. In both Cohorts 1 and 2, there were no significant differences in median age or sex between the atrophic gastritis (AG) and GC groups in H. pylori-infected and -eradicated patients. However, the number of IM samples was significantly higher in the Hp−/GC and Hp−/LDA/GC groups than in the Hp+/AG and Hp+/LDA/AG groups (p = 0.01 and p = 0.0004, respectively).
Table 1.
Patients’ characteristics.
| Cohort 1 | Hp+/AG | Hp+/GC | p–value | Hp−/AG | Hp−/GC | p–value |
Hp+/AG vs Hp−/AG |
Hp+/GC vs Hp−/GC |
|---|---|---|---|---|---|---|---|---|
| (n = 21) | (n = 26) | (n = 30) | (n = 27) | p–value | p–value | |||
|
Past eradication (y) (1st–3rd quartile) |
— | — | — |
5 (3–7) |
5 (4–9) |
0.65 | — | — |
|
Median age (y) (1st–3rd quartile) |
65.0 (59.5–73.0) |
69.0 (65.0–72.3) |
0.22 |
66.5 (54.5–75.3) |
68.0 (64.0–73.0) |
0.24 | 0.72 | 0.68 |
| Male:Female | 13:8 | 18:8 | 0.60 | 14:16 | 19:8 | 0.07 | 0.28 | 0.93 |
| Total no. of samples | 62a | 75b | 90 | 80c | ||||
| Non-IM samples | 41 | 40 | 0.13 | 67 | 45 | 0.01 | 0.27 | 0.72 |
| IM samples | 21 | 35 | 23 | 35 | ||||
| Cohort 2 | Hp +/LDA/AG | Hp +/LDA/GC | Hp −/LDA/AG | Hp −/LDA/GC |
Hp +/LDA/AG vs Hp −/LDA/AG |
Hp +/LDA/GC vs Hp −/LDA/GC |
||
| (n = 3) | (n = 11) | p –value | (n = 21) | (n = 11) | p –value | p –value | p –value | |
|
Median period of prior aspirin use (y) (1st–3rd quartile) |
10 (6–11) |
6 (3–7) |
0.19 |
6 (4–7) |
6 (4–7) |
0.54 | 0.07 | 0.90 |
|
Median age (y) (1st–3rd quartile) |
75.0 (74–76) |
73.0 (67.5–80) |
0.19 |
77.0 (75–78) |
81.0 (71–82) |
0.18 | 0.72 | 0.68 |
| Male:Female | 3:0 | 10:1 | 1 | 12:9 | 10:1 | 0.11 | 0.28 | 0.93 |
| Total no. of samples | 9 | 33 | 63 | 32 d | ||||
| Non-IM samples | 3 | 13 | 1 | 53 | 16 | 0.0004 | 0.27 | 0.72 |
| IM samples | 6 | 20 | 10 | 16 |
aOne sample, bthree samples, cone sample, and done sample could not be analyzed because they were too small.
Hp, H. pylori; AG, atrophic gastritis; GC, gastric cancer; IM, intestinal metaplasia; LDA, low–dose aspirin.
Cohort 1
Molecular events in non-IM: The miR-124a-3 and miR-137 methylation rates were significantly lower in the Hp−/AG group than in the Hp+/AG group (p < 0.0001 and p = 0.06, respectively) (Table 2), thus indicating the effects of H. pylori eradication. Multivariate analysis showed that H. pylori eradication was associated with a significant reduction of miR-124a-3 methylation [odds ratio (OR): 0.03, 95% confidence interval (CI): 0.004–0.27, p = 0.002]. Similarly, the incidences of miR-34c, miR-124a-3, and miR-129-2 methylation were significantly lower in the Hp−/GC group than in the Hp+/GC group (p = 0.005, p = 0.0005, and p = 0.01, respectively), and H. pylori eradication was significantly associated with a reduction of only miR-124a-3 methylation (OR: 0.16, 95% CI: 0.04–0.65, p = 0.01) in a multivariate analysis.
Table 2.
Comparison of molecular alterations in non-IM between patients with and without GC in H. pylori-infected and -eradicated patients (Cohort 1).
| Hp+/AG group | Hp−/AG group | p–value | Multivariate analysis | |||||
|---|---|---|---|---|---|---|---|---|
| (%) | (n) | (%) | (n) | OR | 95% CI | p–value | ||
| miR–34c | 0 | (0/41) | 0 | (0/67) | 1 | — | — | — |
| miR–124a–3 | 31.7 | (13/41) | 1.5 | (1/67) | <0.0001 | 0.03 | 0.004–0.27 | 0.002 |
| miR–129–2 | 10.0 | (4/40) | 7.5 | (5/67) | 0.73 | — | — | — |
| miR–137 | 10.0 | (4/40) | 1.5 | (1/67) | 0.06 | 0.17 | 0.016–2.03 | 0.17 |
| Hp +/GC group | Hp −/GC group | p –value | Multivariate analysis | |||||
| miR–34c | 27.5 | (11/40) | 4.4 | (2/45) | 0.005 | 0.20 | 0.04–1.07 | 0.61 |
| miR–124a–3 | 40.0 | (16/40) | 6.7 | (3/45) | 0.0005 | 0.16 | 0.04–0.65 | 0.01 |
| miR–129–2 | 40.0 | (16/40) | 15.6 | (7/45) | 0.01 | 0.34 | 0.11–1.05 | 0.06 |
| miR–137 | 12.5 | (5/40) | 4.4 | (2/45) | 0.25 | — | — | — |
| Hp +/AG group | Hp +/GC group | p –value | Multivariate analysis | |||||
| miR–34c | 0 | (0/41) | 27.5 | (11/40) | 0.0002 | 1593533.79 | 1.871E–290 | 1.357E302 |
| miR–124a–3 | 31.7 | (13/41) | 40.0 | (16/40) | 0.49 | — | — | — |
| miR–129–2 | 10.0 | (4/40) | 40.0 | (16/40) | 0.004 | 5.21 | 1.46–18.60 | 0.01 |
| miR–137 | 10.0 | (4/40) | 12.5 | (5/40) | 1 | — | — | — |
IM, intestinal metaplasia; Hp, H. pylori; AG, atrophic gastritis; GC, gastric cancer; OR, odds ratio; CI, confidence interval.
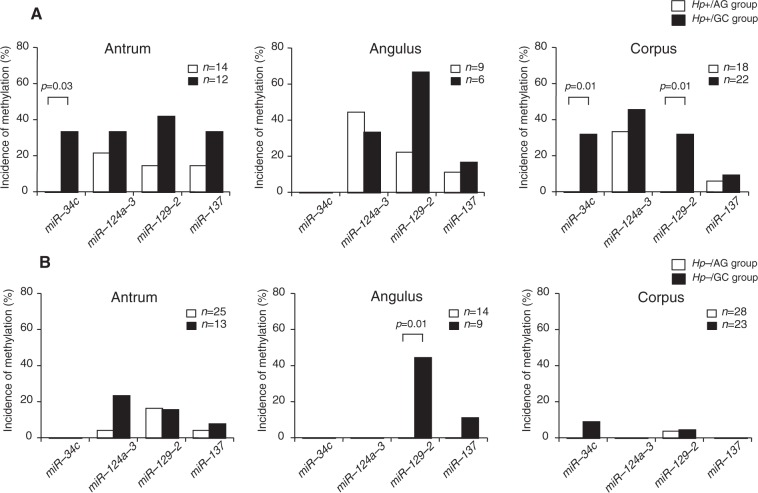
In H. pylori-infected AG patients, the incidence of miR-124a-3 methylation in the Hp+/AG group was 31.7% and that of the methylation of other miRNA genes was very low (Table 2). However, the miR-34c and miR-129-2 methylation rates were significantly higher in the Hp+/GC group than in the Hp+/AG group (p = 0.0002 and p = 0.004, respectively). Multivariate analysis showed that miR-129-2 methylation in non-IM was significantly associated with GC (OR: 5.21, 95% CI: 1.46–18.60, p = 0.01). When comparing the methylation rates of these miRNAs in non-IM among the three different parts of the stomach, the miR-34-c methylation rate in the Hp+/GC group was significantly higher in the antrum (p = 0.03) and corpus (p = 0.01) than in the Hp+/AG group (Fig. 1A). Also, miR-129-2 methylation in non-IM in the corpus was more frequently identified in the Hp+/GC group than in the Hp+/AG group (p = 0.01) (Fig. 1A).
Figure 1.
Incidences of miRNA methylation in non-IM in three different parts of the stomach in patients not taking LDA/NSAIDs. (A) In H. pylori-infected patients, the miR-34-c methylation rate in the antrum (p = 0.03) and corpus (p = 0.01) was significantly higher in the Hp+/GC group than it was in the Hp+/AG group. Also, miR-129-2 methylation in non-IM in the corpus was more frequently identified in the Hp+/GC group than in the Hp+/AG group (p = 0.01). (B) In H. pylori-eradicated patients, the miR-129-2 methylation rate in the angulus in the Hp−/GC group was significantly higher than that in the Hp−/AG group (p = 0.01). LDA, low-dose aspirin; NSAID, nonsteroidal anti-inflammatory drug.
In H. pylori-eradicated cases, there were few methylations in any of the miRNAs in either the Hp−/AG or the Hp−/GC group, and there was no significant difference in the incidence of miRNA methylation between the two groups. However, the miR-129-2 methylation rate in the angulus was significantly higher in the Hp−/GC group than in the Hp−/AG group (p = 0.01) (Fig. 1B).
Molecular events in IM: The incidence of all miRNA genes in IM was demonstrably higher than in non-IM regardless of the presence or absence of H. pylori infection in both the AG and GC groups (Table 3), a finding that was consistent with our previous report33,34. Therefore, there were no significant differences in the methylation rates for each miRNA gene among the four groups, i.e., the Hp+/AG, Hp−/AG, Hp+/GC, and Hp−/GC groups. Furthermore, no significant differences in the incidence of miRNA gene methylation were seen between the AG and GC groups in each part of the stomach in both H. pylori-infected and -eradicated patients, unlike in non-IM (Supplementary Table S1).
Table 3.
Changes of molecular alterations in IM by H. pylori eradication (Cohort 1).
| Hp+/AG group | Hp−/AG group | p–value | Hp+/GC group | Hp−/GC group | p–value |
Hp+/AG group vs Hp+/GC group |
Hp−/AG group vs Hp−/GC group |
|||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| (%) | (n) | (%) | (n) | (%) | (n) | (%) | (n) | p–value | p–value | |||
| miR–34c | 61.9 | (13/21) | 54.5 | (12/22) | 0.63 | 56.0 | (14/25) | 58.8 | (20/34) | 0.83 | 0.77 | 0.79 |
| miR–124a–3 | 100 | (21/21) | 86.4 | (19/22) | 0.23 | 83.3 | (25/30) | 88.6 | (31/35) | 0.72 | 0.07 | 1 |
| miR–129–2 | 100 | (18/18) | 100 | (22/22) | 1 | 100 | (33/33) | 100 | (32/32) | 1 | 1 | 1 |
| miR–137 | 95.0 | (19/20) | 100 | (21/21) | 0.49 | 100 | (34/34) | 100 | (34/34) | 1 | 0.37 | 1 |
IM, intestinal metaplasia; Hp, H. pylori; AG, atrophic gastritis; GC, gastric cancer; OR, odds ratio; CI, confidence interval.
Cohort 2
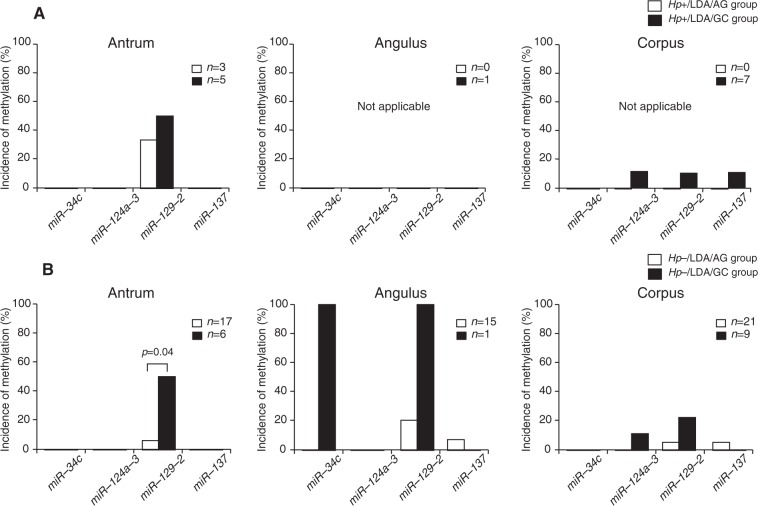
Molecular alterations in non-IM: The incidence of miRNA methylation was not significantly different between the Hp+/LDA/AG and Hp−/LDA/AG groups or between the Hp+/LDA/GC and Hp−/LDA/GC groups (Table 4), a result that was different from the findings in Cohort 1. However, only miR-129-2 methylation was more frequently observed in the Hp−/LDA/GC group than in the Hp−/LDA/AG group (p = 0.02) among the H. pylori-eradicated patients, although there were no significant differences in the incidences of the methylation of other miRNAs between the AG and GC groups regardless of the presence or absence of H. pylori infection. When looking at the methylation rate in each portion of the stomach, the miR-129-2 methylation rate was found to be significantly higher in the Hp−/LDA/GC group than in the Hp−/LDA/AG (p = 0.04) group only in the antrum (Fig. 2B).
Table 4.
Comparison of molecular alterations between patients taking LDA/NSAIDs with and without GC (Cohort 2).
| Non-IM | Hp+/LDA/AG group | Hp−/LDA/AG group | p–value | Hp+/LDA/GC group | Hp−/LDA/GC group | p–value |
Hp+/LDA/AG group vs Hp+/LDA/GC group |
Hp−/LDA/AG group vs Hp−/LDA/GC group |
||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| (%) | (n) | (%) | (n) | (%) | (n) | (%) | (n) | p–value | p–value | |||
| miR–34c | 0 | (0/3) | 0 | (0/53) | 1 | 0 | (0/13) | 6.3 | (1/16) | 1 | 1 | 0.23 |
| miR–124a–3 | 0 | (0/3) | 0 | (0/53) | 1 | 8.3 | (1/12) | 6.3 | (1/16) | 1 | 1 | 0.23 |
| miR–129–2 | 33.3 | (1/3) | 9.4 | (5/53) | 0.29 | 25.0 | (3/12) | 37.5 | (6/16) | 0.69 | 1 | 0.02 |
| miR–137 | 0 | (0/3) | 3.8 | (2/53) | 1 | 7.7 | (1/13) | 0 | (0/16) | 0.45 | 1 | 1 |
| IM | (%) | ( n ) | (%) | ( n ) | p –value | (%) | ( n ) | (%) | ( n ) | p –value | p –value | p –value |
| miR–34c | 40.0 | (2/5) | 60.0 | (6/10) | 0.61 | 75.0 | (9/12) | 58.3 | (7/12) | 0.67 | 0.28 | 1 |
| miR–124a–3 | 80.0 | (4/5) | 100 | (9/9) | 0.36 | 94.4 | (17/18) | 93.3 | (14/15) | 1 | 0.40 | 1 |
| miR–129–2 | 100 | (4/4) | 100 | (7/7) | 1 | 100 | (11/11) | 100 | (10/10) | 1 | 1 | 1 |
| miR–137 | 100 | (4/4) | 100 | (7/7) | 1 | 100 | (12/12) | 100 | (12/12) | 1 | 1 | 1 |
LDA, low–dose aspirin; NSAID, nonsteroidal anti-inflammatory drug; Hp, H. pylori; AG, atrophic gastritis; GC, gastric cancer; IM, intestinal metaplasia.
Figure 2.
Incidences of miRNA methylation in non-IM in the three parts of the stomach in patients regularly taking LDA/NSAIDs. (A) In H. pylori-infected patients, there were no significant differences in the incidences of miRNA methylation in each portion of the stomach. (B) In H. pylori-eradicated patients, the miR-129-2 methylation rate in the antrum was significantly higher in the Hp−/GC group than it was in the Hp−/AG group (p = 0.04). LDA, low-dose aspirin; NSAID, nonsteroidal anti-inflammatory drug.
Molecular alterations in IM: The incidences of methylation of all of the miRNA genes in IM were higher compared to those in non-IM, and they were not significantly different between the Hp+/LDA/AG and Hp−/LDA/AG groups or between the Hp+/LDA/GC and Hp−/LDA/GC groups, findings that were similar to the results in non-IM (Table 4). Similarly, there were no significant differences in the incidences of methylation of all miRNAs between the AG and GC groups regardless of the presence or absence of H. pylori infection. In addition, no positive associations in the incidence of miRNA gene methylation were seen between the AG and GC groups in each part of the stomach in either H. pylori-infected or -eradicated patients, as in IM in Cohort 1 (Supplementary Table S1).
Effects of long-term aspirin use: In H. pylori-infected patients, only miR-34c methylation in non-IM was significantly lower in the Hp+/LDA/GC group compared to the Hp+/GC group (P = 0.047), which may be an effect of long-term aspirin use. In contrast, there were no significant differences in other molecular events between patients who were taking and patients who were not taking LDA/NSAIDs in both non-IM and IM, irrespective of H. pylori infection (Table 5).
Table 5.
Statistical comparison of the incidence of molecular events between patients taking and not taking LDA/NSAIDs.
| Non-IM | H. pylori-infected patients | H. pylori-eradicated patients | ||
|---|---|---|---|---|
| Hp+/AG vs Hp+/LDA/AG | Hp+/GC vs Hp+/LDA/GC | Hp−/AG vs Hp−/LDA/AG | Hp−/GC vs Hp−/LDA/GC | |
| miR–34c | 1 | 0.047 | 1 | 1 |
| miR–124a–3 | 0.54 | 0.08 | 1 | 1 |
| miR–137 | 1 | 1 | 0.58 | 1 |
| miR–129–2 | 0.32 | 0.50 | 0.70 | 0.08 |
| IM | ||||
| miR–34c | 0.62 | 0.31 | 1 | 1 |
| miR–124a–3 | 0.19 | 0.39 | 0.54 | 1 |
| miR–137 | 1 | 1 | 1 | 1 |
| miR–129–2 | 1 | 1 | 1 | 1 |
Values indicate p–values.
LDA, low–dose aspirin; NSAID, nonsteroidal anti-inflammatory drug; IM, intestinal metaplasia; Hp, H. pylori; AG, atrophic gastritis; GC, gastric cancer.
Discussion
To the best of our knowledge, this is the first study to show the effects of H. pylori eradication and LDA/NSAIDs on the methylation of several miRNAs in patients with and without GC.
Cohort 1
In patients not taking LDA/NDAIDs, H. pylori eradication was able to reverse the methylation of most miRNA genes only in non-IM, but not in IM, both in patients with and in those without GC (i.e., the Hp+/AG and Hp+/GC groups). In multivariate analysis, H. pylori eradication was associated with a significant reduction of miR-124a-3 methylation in both groups, a finding that was consistent with our previous study34. In addition, miR-34c and miR-129-2 methylation were associated with GC development in H. pylori-infected cases, and only miR-129-2 methylation in non-IM was an independent risk marker of significant GC.
When evaluating the differences in the methylation of miRNA genes in each of the three parts of the stomach, we found that miR-34c methylation in non-IM in the antrum and corpus portions and miR-129-2 methylation in non-IM in the corpus might be useful biomarkers of GC in H. pylori-infected patients. Meanwhile, miR-129-2 methylation in non-IM in the angulus was associated with GC development in H. pylori-eradicated patients, thus indicating that the biomarkers for GC differ for each part of the stomach according to H. pylori status. Suzuki et al. reported that miR-34b/c methylation in the corpus is an independent predictor of metachronous GC risk37. However, they did not investigate the methylation of miRNA genes in non-IM and IM separately in H. pylori-infected and -eradicated patients.
Cohort 2
In patients taking LDA/NSAIDs, the incidence of miRNA gene methylation was not different between H. pylori-infected and -eradicated patients in either non-IM or IM. Meanwhile, miR-129-2 methylation in non-IM was significantly involved in GC development in H. pylori-eradicated patients. Therefore, this result indicates that patients with miR-129-2 methylation in non-IM, especially in the antrum, may be at risk of GC. In the present study, LDA/NSAIDs decreased miR-34c methylation in non-IM in H. pylori-infected patients with GC (Table 5). This result may suggest that the chemopreventive effect of aspirin is limited to non-IM, and does not occur in IM, in H. pylori-infected patients. Some studies showed in stratified analyses that the chemopreventive effect of aspirin was higher in H. pylori-infected subjects42–44; this finding was in agreement with our results from the viewpoint of miRNA analysis. However, as H. pylori status was judged using only H. pylori IgG antibody in those previous studies42–44, no precise diagnosis of H. pylori infection was performed. In addition, it may be possible that the H. pylori-negative subjects in those studies included two subtypes that were completely different in terms of their GC risk: H. pylori-uninfected patients and patients who were suspected of naturally eradicated H. pylori. In our study, we showed that the long-term use of LDA/NSAIDs did not affect the changes in miRNA methylation in either H. pylori-infected or -eradicated patients, although the number of samples evaluated may have been small. Cheung et al. recently reported a clinical study in which the protective effect of aspirin appeared to be larger in H. pylori-eradicated subjects (hazard ratio = 0.30)45, a finding that was different from our results. Therefore, the mechanism of the chemopreventive effect of aspirin cannot be explained by miRNA methylation alone.
In the present study, the methylation of tumor-suppressor miRNAs was identified more frequently in IM and very infrequently in non-IM. It thus appears that the methylation of these genes is a molecular event that occurs specifically in IM, and also that IM might exhibit a more aggressive state than non-IM with regard to molecular alterations, as shown in our previous reports33,34. Intriguingly, only in H. pylori-eradicated patients, the number of IM samples obtained from the three parts of the stomach was significantly higher in patients with GC (the Hp−/GC and Hp−/LDA/GC groups) than in those without GC (the Hp−/AG and Hp−/LDA/AG groups). This result may support the clinical fact that GC is related to the extent of IM throughout the stomach46, especially after H. pylori treatment. Also, H. pylori eradication and the long-term use of LDA/NSAIDs reversed the methylation of miRNAs in non-IM, but not in IM. Taken together, these results are in agreement with the concept of a “point of no return”47, which holds that the benefits of H. pylori eradication and aspirin diminish after the IM stage is reached through the state in which molecular changes are irreversible. AG is caused by focal inflammation, resulting in a loss of glandular structures in the gastric mucosa, while IM involves the replacement of damaged gastric mucosa by intestinal epithelium, including goblet cells and absorptive cells48. In the inflammatory state, gastric tissue stem cells fail to differentiate normally49, resulting in their progression to IM50. In addition, the appearance of IM is considered to be associated with the aberrant expression of CDX1 and CDX251,52. Based on previous reports48–52 and the present study, the pathogenesis of non-IM and that of IM are likely to be different from the viewpoint not only of histology but also of the differentiation of gastric stem cells and the accumulation of molecular events. It has recently been reported that AG and IM were reversed by H. pylori treatment over the long term48, and the reversibility of IM was associated with a decrease in CDX2 mRNA expression52. Thus, a long-term follow-up study may be necessary to confirm the molecular changes resulting from such interventions in non-IM and IM cases.
The present study had a methodological advantage: as our data from LCM samples delineated many GC-related miRNA genes, this procedure might provide more information from non-IM and IM regarding pathogenesis than would the use of whole-tissue material53. Since whole-biopsy tissues were used for DNA methylation analysis in the previous studies29,30,36–41, the differences in methylation between non-IM and IM were not evaluated. Therefore, those results may be affected by the total volume of IM glands contained in the biopsy samples34.
Our investigation had some limitations. First, this was a study from a single institution with a small number of H. pylori-infected patients who were taking LDA/NSAIDs; the number of patients was especially small considering that molecular alterations in three different parts of the stomach were compared. The second limitation is that this study may have been biased due to our use of the same samples collected in our previous two studies33,34. Third, the methylation-sensitive high-resolution melting (MS-HRM) used in our study may be applicable for semiquantitative but not quantitative assessment of the methylation levels in an unmethylated background. However, we confirmed that the definition of methylation (>10%) by MS-HRM was reasonable on the basis of the correlation coefficient of the calibration curve derived from the fluorescence value of the melting curve using the methylation standard control DNA34. Fourth, a relatively high number of samples could not be analyzed for molecular alterations due to the small amount of DNA that was extracted from the very small biopsy specimen. Therefore, further prospective studies with larger sample sizes are needed to clarify the association between miRNAs and gastric carcinogenesis in the background mucosa with and without GC.
In conclusion, in patients who were not taking LDA/NSAIDs (Cohort 1), (1) H. pylori eradication was associated with a significant reduction of miR-124a-3 methylation only in non-IM in the background mucosa with and without GC, but not in IM, and (2) miR-129-2 methylation in non-IM, especially in the corpus, may be a surrogate marker of GC in H. pylori-infected patients. On the other hand, in patients regularly taking LDA/NSAIDs (Cohort 2), LDA/NSAIDs did not affect the changes in miRNA methylation in either non-IM or IM, irrespective of H. pylori infection. However, miR-129-2 methylation in non-IM, especially in the antrum, was an independent predictive marker of GC in H. pylori-infected patients. These results indicate that H. pylori eradication and LDA/NSAIDs use were less effective in improving the methylation in IM compared with non-IM; thus, these interventions are recommended to prevent GC development in individuals at an early stage prior to the development of IM.
Patients, Materials and Methods
Cohort 1
Patients
We previously conducted a cross-sectional study to define the differences in molecular alterations in non-IM and IM before and after H. pylori eradication (≥3 yr) in patients with and without GC28,33,34. By using the same samples, we herein performed a case-control study on miRNAs in four groups of the same patients based on previous studies:28,33,34 patients with histologically diagnosed AG (GC-free patients) who were positive for H. pylori (Hp+/AG group, n = 21); GC patients with H. pylori infection (Hp+/GC group, n = 26); patients with histologically diagnosed AG (i.e., GC-free patients) who had undergone H. pylori eradication >3 years before and had AG in the background mucosa (Hp−/AG group, n = 30); and patients who developed primary early GC despite the successful eradication of H. pylori at >3 years before (Hp−/GC group, n = 27). Histological diagnosis of GC was made in accordance with the GC criteria of the Japanese Gastric Cancer Association54. Patients with a history of esophagectomy or gastrectomy and who were taking LDA or other NSAIDs were excluded.
H. pylori status and DNA extraction
In our previous studies28,33,34, three biopsy specimens were taken during an endoscopy from three parts of the stomach: the greater curvature at the antrum, the greater curvature at the corpus, and the lesser curvature at the angulus (one from each site). Each biopsy specimen was cut into 4-µm-thick tissue sections and subjected to histological analysis using hematoxylin and eosin staining and Giemsa staining. H. pylori status was analyzed as reported previously28,33,34. A patient was regarded as H. pylori-positive if the result of at least one of the three aforementioned methods, i.e., the urea breath test (UBT), Giemsa staining, and the E-plate anti-H pylori IgG antibody test (Eiken Kagaku, Tokyo, Japan), was positive. H. pylori status following the eradication was determined by the UBT at least 6 weeks or more after the end of the anti-H. pylori treatment. From the paraffin-embedded biopsy specimens, two or three 7-µm-thick tissue sections were cut for DNA extraction. Goblet IM glands were isolated using the PALM MicroBeam LCM system (Microlaser Technologies, Munich, Germany), and DNA was extracted from goblet IM (incomplete type) and non-IM using the QIAamp DNA Micro Kit (Qiagen, Hilden, Germany) as previously reported28,33,34 (Supplementary Fig. S1). Finally, a total of 307 biopsy samples from 104 patients, including Hp+/AG, Hp+/GC, Hp−/AG, and Hp−/GC patients, were analyzed. However, five samples could not be analyzed for molecular alterations due to the small amount of DNA that was extracted from the very small biopsy specimens.
Sodium bisulfite modification of DNA of miRNA genes
As in previous reports28,33,34, purified DNA samples were chemically modified with sodium bisulfite using an EpiTect® Fast Bisulfite Kit (Qiagen). The bisulfite-modified DNA was amplified using primer pairs that specifically amplify the methylated or unmethylated sequences of several miRNAs related to carcinogenesis, including miR-34c, miR-124a-3, miR-129-2, and miR-137.
MS-HRM analyses
MS-HRM analysis was performed as previously described28,33,34. Briefly, polymerase chain reaction (PCR) amplification and MS-HRM analysis were performed using a LightCycler® 480 System II (Roche, Mannheim, Germany). The primer sequences of all genes for the methylated and unmethylated forms and PCR and MS-HRM conditions are summarized in Supplementary Tables S2 and S3. Percentages of methylation (0%, 10%, 50%, and 100%) were used to draw the standard curve (Supplementary Fig. S2). In this study, only samples with >10% methylation were considered to be methylated, as reported previously33,34.
Cohort 2
Patients and samples
In this study, which was based on our previous studies28,33, we analyzed 22 patients who had developed primary GC despite taking LDA (100 mg/day) or NSAIDs for more than 3 years. These patients included 11 H. pylori-infected patients (Hp+/LDA/GC group) and 11 H. pylori-eradicated patients (Hp−/LDA/GC group). We also analyzed 24 patients with histologically diagnosed AG (GC-free patients) who regularly took LDA or NSAIDs for more than 3 years, who included H. pylori-infected cases (n = 3, Hp+/LDA/AG group) and H. pylori-eradicated cases (n = 21, Hp−/LDA/AG group). Biopsy specimens were taken from the same portions of the stomach in the same manner as described for Cohort 1. Patients with a history of esophagectomy or gastrectomy were excluded.
In Cohort 2, the methylation of miRNAs was analyzed. A total of 137 biopsy samples from the 46 patients making up the Hp+/LDA/AG, Hp+/LDA/GC, Hp−/LDA/AG, and Hp−/LDA/GC groups were analyzed. However, one sample could not be analyzed for molecular alterations due to the small amount of DNA that was extracted.
Consent and institutional review board approval
Written informed consent was obtained from all patients prior to these studies. The Ethics Committee of Hyogo College of Medicine approved these trials (Nos. Rin-Hi 136 and 300). These trials were registered with the UMIN Clinical Trials Registry (No. UMIN000021857). The study was performed in accordance with the Declaration of Helsinki.
Statistical analysis
Categorical variables were presented as numbers and percentages and compared between groups using the chi-square test or Fisher’s exact test when appropriate. Continuous variables were expressed by median and interquartile range and compared between groups using the Kruskal-Wallis test or Mann-Whitney U-test. Predictive factors for GC with a p-value of <0.05 in univariate analysis were included in the multiple logistic regression model and analyzed using the backward approach. ORs and 95% CIs were calculated for risk factors. Multivariate logistic regression analysis was performed to identify the molecular alterations associated with GC if a p-value of less than 0.1 in the univariate analysis was identified. A two-tailed p-value less than 0.05 was considered significant. Statistical analyses were performed with SPSS 22.0 (SPSS Inc., Chicago, IL) and StatView version 5.0 (SAS Institute Inc., Cary, NC).
Supplementary information
Acknowledgements
The authors would like to thank Ms. Mayumi Yamada for her excellent technical assistance. This research was supported by funds from Astellas Academic Support (Astellas Pharm. Inc.) and EA Pharma Research Support (EA Pharma Co., Ltd.).
Author Contributions
J.W., T.S., K.M.D. and H.M. designed the study and analyzed the data; J.W. and C.I. recruited the patients, performed the DNA extraction and molecular analysis, and analyzed the data; Y.M., J.W., T.T., T.O. and H.F. performed the endoscopies; J.W. and C.I. performed the statistical analysis; J.W. wrote the manuscript; T.S., K.M.D. and H.M. revised the manuscript. All authors read and approved the final version of the paper.
Competing Interests
The authors declare no competing interests.
Footnotes
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary information accompanies this paper at 10.1038/s41598-019-49069-1.
References
- 1.Ferlay J, et al. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int. J. Cancer. 2015;13:E359–E386. doi: 10.1002/ijc.29210. [DOI] [PubMed] [Google Scholar]
- 2.Correa P. Helicobacter pylori and gastric carcinogenesis. Am. J. Surg. Pathol. 1995;19(Suppl 1):S37–S43. [PubMed] [Google Scholar]
- 3.Fuccio L, et al. Meta-analysis: can Helicobacter pylori eradication treatment reduce the risk for gastric cancer? Ann. Intern. Med. 2009;151:121–128. doi: 10.7326/0003-4819-151-2-200907210-00009. [DOI] [PubMed] [Google Scholar]
- 4.Yoon SB, Park JM, Lim CH, Cho YK, Choi MG. Effect of Helicobacter pylori eradication on metachronous gastric cancer after endoscopic resection of gastric tumors: a meta-analysis. Helicobacter. 2014;19:243–248. doi: 10.1111/hel.12146. [DOI] [PubMed] [Google Scholar]
- 5.Ford AC, Forman D, Hunt RH, Yuan Y, Moayyedi P. Helicobacter pylori eradication therapy to prevent gastric cancer in healthy asymptomatic infected individuals: systematic review and meta-analysis of randomised controlled trials. BMJ. 2014;348:g3174. doi: 10.1136/bmj.g3174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Doorakkers E, Lagergren J, Engstrand L, Brusselaers N. Eradication of Helicobacter pylori and gastric cancer: a systematic review and meta-analysis of cohort studies. J. Natl. Cancer. Inst. 2016;108:jdw132. doi: 10.1093/jnci/djw132. [DOI] [PubMed] [Google Scholar]
- 7.Lee YC, et al. Association between Helicobacter pylori eradication and gastric cancer incidence: a systematic review and meta-analysis. Gastroenterology. 2016;150:1113–1124. doi: 10.1053/j.gastro.2016.01.028. [DOI] [PubMed] [Google Scholar]
- 8.Doorakkers E, Lagergren J, Engstrand L, Brusselaers N. Helicobacter pylori eradication treatment and the risk of gastric adenocarcinoma in a Western population. Gut. 2018;67:2092–2096. doi: 10.1136/gutjnl-2017-315363. [DOI] [PubMed] [Google Scholar]
- 9.Fukase K, et al. Effect of eradication of Helicobacter pylori on incidence of metachronous gastric carcinoma after endoscopic resection of early gastric cancer: an open-label, randomised controlled trial. Lancet. 2008;372:392–397. doi: 10.1016/S0140-6736(08)61159-9. [DOI] [PubMed] [Google Scholar]
- 10.Bae SE, et al. Effect of Helicobacter pylori eradication on metachronous recurrence after endoscopic resection of gastric neoplasm. Am. J. Gastroenterol. 2014;109:60–67. doi: 10.1038/ajg.2013.404. [DOI] [PubMed] [Google Scholar]
- 11.Choi IJ, et al. Helicobacter pylori therapy for the prevention of metachronous gastric cancer. N. Engl. J. Med. 2018;378:1085–1095. doi: 10.1056/NEJMoa1708423. [DOI] [PubMed] [Google Scholar]
- 12.Maehata Y, et al. Long-term effect of Helicobacter pylori eradication on the development of metachronous gastric cancer after endoscopic resection of early gastric cancer. Gastrointest. Endosc. 2012;75:39–46. doi: 10.1016/j.gie.2011.08.030. [DOI] [PubMed] [Google Scholar]
- 13.Kato M, et al. Scheduled endoscopic surveillance controls secondary cancer after curative endoscopic resection for early gastric cancer: a multicentre retrospective cohort study by Osaka University ESD study group. Gut. 2013;62:1425–1432. doi: 10.1136/gutjnl-2011-301647. [DOI] [PubMed] [Google Scholar]
- 14.Choi J, et al. Eradication of Helicobacter pylori after endoscopic resection of gastric tumors does not reduce incidence of metachronous gastric carcinoma. Clin. Gastroenterol. Hepatol. 2014;12:793–800. doi: 10.1016/j.cgh.2013.09.057. [DOI] [PubMed] [Google Scholar]
- 15.Shichijo S, et al. Histologic intestinal metaplasia and endoscopic atrophy are predictors of gastric cancer development after Helicobacter pylori eradication. Gastrointest. Endosc. 2016;84:618–24. doi: 10.1016/j.gie.2016.03.791. [DOI] [PubMed] [Google Scholar]
- 16.Rugge M, et al. Gastritis staging in the endoscopic follow-up for the secondary prevention of gastric cancer: a 5-year prospective study of 1755 patients. Gut. 2019;68:11–17. doi: 10.1136/gutjnl-2017-314600. [DOI] [PubMed] [Google Scholar]
- 17.Kong P, et al. The effects of anti-inflammatory drug treatment in gastric cancer prevention: an update of a meta-analysis. J. Cancer. 2016;7:2247–2257. doi: 10.7150/jca.16524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Huang XZ, et al. Aspirin and non-steroidal anti-inflammatory drugs use reduce gastric cancer risk: A dose-response meta-analysis. Oncotarget. 2017;8:4781–4795. doi: 10.18632/oncotarget.13591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Qiao Y, et al. Associations between aspirin use and the risk of cancers: a meta-analysis of observational studies. BMC Cancer. 2018;18:288. doi: 10.1186/s12885-018-4156-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kim MH, Chang J, Kim WJ, Banerjee S, Park SM. Cumulative dose threshold for the chemopreventive effect of aspirin against gastric cancer. Am. J. Gastroenterol. 2018;113:845–854. doi: 10.14309/00000434-201806000-00015. [DOI] [PubMed] [Google Scholar]
- 21.Cheung KS, Leung WK. Risk of gastric cancer development after eradication of Helicobacter pylori. World J. Gastrointest. Oncol. 2018;10:115–123. doi: 10.4251/wjgo.v10.i5.115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Leung WK, et al. Effects of Helicobacter pylori eradication on methylation status of E-cadherin gene in noncancerous stomach. Clin. Cancer Res. 2006;12:3216–3221. doi: 10.1158/1078-0432.CCR-05-2442. [DOI] [PubMed] [Google Scholar]
- 23.Chan AO, et al. Eradication of Helicobacter pylori infection reverses E-cadherin promoter hypermethylation. Gut. 2006;55:463–468. doi: 10.1136/gut.2005.077776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Perri F, et al. Aberrant DNA methylation in non-neoplastic gastric mucosa of H. Pylori infected patients and effect of eradication. Am. J. Gastroenterol. 2007;102:1361–1371. doi: 10.1111/j.1572-0241.2007.01284.x. [DOI] [PubMed] [Google Scholar]
- 25.Sepulveda AR, et al. CpG methylation and reduced expression of O6-methylguanine DNA methyltransferase is associated with Helicobacter pylori infection. Gastroenterology. 2010;138:1836–1844. doi: 10.1053/j.gastro.2009.12.042. [DOI] [PubMed] [Google Scholar]
- 26.Nakajima T, et al. Persistence of a component of DNA methylation in gastric mucosae after Helicobacter pylori eradication. J. Gastroenterol. 2010;45:37–44. doi: 10.1007/s00535-009-0142-7. [DOI] [PubMed] [Google Scholar]
- 27.Shin CM, et al. Changes in aberrant DNA methylation after Helicobacter pylori eradication: a long-term follow-up study. Int. J. Cancer. 2013;133:2034–2042. doi: 10.1002/ijc.28219. [DOI] [PubMed] [Google Scholar]
- 28.Kawanaka M, et al. Effects of Helicobacter pylori eradication on the development of metachronous gastric cancer after endoscopic treatment: analysis of molecular alterations by a randomised controlled trial. Br. J. Cancer. 2016;114:21–29. doi: 10.1038/bjc.2015.418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Asada K, et al. Demonstration of the usefulness of epigenetic cancer risk prediction by a multicentre prospective cohort study. Gut. 2015;64:388–396. doi: 10.1136/gutjnl-2014-307094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Maeda M, et al. High impact of methylation accumulation on metachronous gastric cancer: 5-year follow-up of a multicentre prospective cohort study. Gut. 2017;66:1721–1723. doi: 10.1136/gutjnl-2016-313387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Liu X, Meltzer SJ. Gastric cancer in the era of precision medicine. Cell. Mol. Gastroenterol. Hepatol. 2017;3:348–358. doi: 10.1016/j.jcmgh.2017.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Huang KK, et al. Genomic and epigenomic profiling of high-risk intestinal metaplasia reveals molecular determinants of progression to gastric cancer. Cancer Cell. 2018;33:137–150. doi: 10.1016/j.ccell.2017.11.018. [DOI] [PubMed] [Google Scholar]
- 33.Michigami Y, et al. Effects of long-term aspirin use on molecular alterations in precancerous gastric mucosa in patients with and without gastric cancer. Sci. Rep. 2017;7:13384. doi: 10.1038/s41598-017-13842-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Michigami Y, et al. Long-term effects of H. pylori eradication on epigenetic alterations related to gastric carcinogenesis. Sci. Rep. 2018;8:14369. doi: 10.1038/s41598-018-32717-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Shin VY, Ng EK, Chan VW, Kwong A, Chu KM. A three-miRNA signature as promising non-invasive diagnostic marker for gastric cancer. Mol. Cancer. 2015;14:202. doi: 10.1186/s12943-015-0473-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Suzuki H, et al. Methylation-associated silencing of microRNA-34b/c in gastric cancer and its involvement in an epigenetic field defect. Carcinogenesis. 2010;31:2066–2073. doi: 10.1093/carcin/bgq203. [DOI] [PubMed] [Google Scholar]
- 37.Suzuki R, et al. Aberrant methylation of microRNA-34b/c is a predictive marker of metachronous gastric cancer risk. J. Gastroenterol. 2014;49:1135–1144. doi: 10.1007/s00535-013-0861-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Chen Q, et al. miR-137 is frequently down-regulated in gastric cancer and is a negative regulator of Cdc42. Dig. Dis. Sci. 2011;56:2009–2016. doi: 10.1007/s10620-010-1536-3. [DOI] [PubMed] [Google Scholar]
- 39.Steponaitiene R, et al. Epigenetic silencing of miR-137 is a frequent event in gastric carcinogenesis. Mol. Carcinog. 2016;55:376–386. doi: 10.1002/mc.22287. [DOI] [PubMed] [Google Scholar]
- 40.Zhang Z, et al. miR-21 plays a pivotal role in gastric cancer pathogenesis and progression. Lab. Invest. 2008;88:1358–1366. doi: 10.1038/labinvest.2008.94. [DOI] [PubMed] [Google Scholar]
- 41.Tsai KW, et al. Epigenetic regulation of miR-34b and miR-129 expression in gastric cancer. Int. J. Cancer. 2011;129:2600–2610. doi: 10.1002/ijc.25919. [DOI] [PubMed] [Google Scholar]
- 42.Zaridze D, Borisova E, Maximovitch D, Chkhikvadze V. Aspirin protects against gastric cancer: results of a case-control study from Moscow, Russia. Int. J. Cancer. 1999;82:473–476. doi: 10.1002/(SICI)1097-0215(19990812)82:4<473::AID-IJC1>3.0.CO;2-K. [DOI] [PubMed] [Google Scholar]
- 43.Akre K, Ekström AM, Signorello LB, Hansson LE, Nyrén O. Aspirin and risk for gastric cancer: a population-based case-control study in Sweden. Br. J. Cancer. 2001;84:965–968. doi: 10.1054/bjoc.2001.1702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Wu CY, et al. Effective reduction of gastric cancer risk with regular use of nonsteroidal anti-inflammatory drugs in Helicobacter pylori-infected patients. J. Clin. Oncol. 2010;28:2952–2957. doi: 10.1200/JCO.2009.26.0695. [DOI] [PubMed] [Google Scholar]
- 45.Cheung KS, et al. Aspirin and risk of gastric cancer after Helicobacter pylori eradication: a territory-wide study. J. Natl. Cancer Inst. 2018;110:743–749. doi: 10.1093/jnci/djx267. [DOI] [PubMed] [Google Scholar]
- 46.Capelle LG, et al. The staging of gastritis with the OLGA system by using intestinal metaplasia as an accurate alternative for atrophic gastritis. Gastrointest. Endosc. 2010;71:1150–1158. doi: 10.1016/j.gie.2009.12.029. [DOI] [PubMed] [Google Scholar]
- 47.Wong BC, et al. Helicobacter pylori eradication to prevent gastric cancer in a high-risk region of China: a randomized controlled trial. JAMA. 2004;291:187–194. doi: 10.1001/jama.291.2.187. [DOI] [PubMed] [Google Scholar]
- 48.Hwang YJ, et al. Reversibility of atrophic gastritis and intestinal metaplasia after Helicobacter pylori eradication - a prospective study for up to 10 years. Aliment. Pharmacol. Ther. 2018;47:380–390. doi: 10.1111/apt.14424. [DOI] [PubMed] [Google Scholar]
- 49.Houghton J, et al. Gastric cancer originating from bone marrow-derived cells. Science. 2004;306:1568–1571. doi: 10.1126/science.1099513. [DOI] [PubMed] [Google Scholar]
- 50.Houghton J, Wang TC. Helicobacter pylori and gastric cancer: a new paradigm for inflammation-associated epithelial cancers. Gastroenterology. 2005;128:1567–1578. doi: 10.1053/j.gastro.2005.03.037. [DOI] [PubMed] [Google Scholar]
- 51.Mutoh H, et al. Cdx1 induced intestinal metaplasia in the transgenic mouse stomach: comparative study with Cdx2 transgenic mice. Gut. 2004;53:1416–1423. doi: 10.1136/gut.2003.032482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Shin CM, et al. Follow-up study on CDX1 and CDX2 mRNA expression in noncancerous gastric mucosae after helicobacter pylori eradication. Dig. Dis. Sci. 2016;61:1051–1059. doi: 10.1007/s10620-016-4048-y. [DOI] [PubMed] [Google Scholar]
- 53.El-Serag HB, et al. Gene expression in Barrett’s esophagus: laser capture versus whole tissue. Scand. J. Gastroenterol. 2009;44:787–795. doi: 10.1080/00365520902898127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Japanese Gastric Cancer Association Japanese Classification of Gastric Carcinoma - 3rd English edition. Gastric Cancer. 2011;14:101–112. doi: 10.1007/s10120-011-0041-5. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.