Abstract
Objective
Using meta-regression this paper sets out the minimum change in body mass index-SD score (BMI-SDS) required to improve adiposity as percentage body fat for children and adolescents with obesity.
Design
Meta-regression.
Setting
Studies were identified as part of a large-scale systematic review of the following electronic databases: AMED, Embase, MEDLINE via OVID, Web of Science and CENTRAL via Cochrane library.
Participants
Individuals aged 4–19 years with a diagnosis of obesity according to defined BMI thresholds.
Interventions
Studies of lifestyle treatment interventions that included dietary, physical activity and/or behavioural components with the objective of reducing obesity were included. Interventions of <2 weeks duration and those that involved surgical and/or pharmacological components (eg, bariatric surgery, drug therapy) were excluded.
Primary and secondary outcome measures
To be included in the review, studies had to report baseline and post-intervention BMI-SDS or change measurements (primary outcome measures) plus one or more of the following markers of metabolic health (secondary outcome measures): adiposity measures other than BMI; blood pressure; glucose; inflammation; insulin sensitivity/resistance; lipid profile; liver function. This paper focuses on adiposity measures only. Further papers in this series will report on other outcome measures.
Results
This paper explores the potential impact of BMI-SDS reduction in terms of change in percentage body fat. Thirty-nine studies reporting change in mean percentage body fat were analysed. Meta-regression demonstrated that reduction of at least 0.6 in mean BMI-SDS ensured a mean reduction of percentage body fat mass, in the sense that the associated 95% prediction interval for change in mean percentage body fat was wholly negative.
Conclusions
Interventions demonstrating reductions of 0.6 BMI-SDS might be termed successful in reducing adiposity, a key purpose of weight management interventions.
Trial registration number
CRD42016025317.
Keywords: obesity, childhood, adolescence, body mass index, body fat
Strengths and limitations of this study.
We believe that this is the first paper to attempt to bring together all studies that have reported both a change in body mass index-SD score and changes in a marker of adiposity in the paediatric population with obesity.
The systematic methods employed to identify the included studies were stringent, but it is possible that some relevant studies might have been missed.
There was some variation in the reporting of results where there were multiple publications of the same study; in these cases, the results from the most comprehensive paper have been used.
Studies that did not report change in mean percentage body fat could not be included in this meta-regression.
Introduction
Childhood obesity is one of the most serious global public health challenges of the 21st century.1 In England, the latest figures from the National Child Measurement Programme, which measures the height and weight of around 1 million school children every year, showed that 9.5% of children aged 4–5 years and 20.1% of those aged 10–11 years were obese.2 3 Childhood obesity has adverse health consequences in both the short-term and long-term, including an increased risk of developing metabolic disturbances, like hypertension, dyslipidaemia and insulin resistance, and becoming obese adults.4 The presence of adverse changes in cardiac and vascular function and type 2 diabetes, which were previously considered adult morbidities, now being identified in children and adolescents with obesity5–11 illustrates the urgent need for effective weight management treatment interventions to reduce adiposity and improve the metabolic health status of the paediatric population.
Moderate weight loss has been shown to have a positive impact on many metabolic and cardiovascular risk factors.12 13 Weight management interventions for adults with obesity that result in a 5–10% decrease in body weight are associated with significant improvements in blood pressure, serum lipid levels and glucose tolerance14 and reduction in the prevalence of hypertension and diabetes.15 Minimum weight management targets can therefore be set to improve metabolic health in this population.16
During childhood, all measurements over time are complicated by the influence of growth, meaning that cut-offs routinely used in the adult population cannot be used in children and adolescents. However, measured values of body mass index (BMI) can be standardised into SD scores (SDS) with respect to reference populations.17 These standardised scores, referred to as BMI-SDS throughout this paper, provide a normalised measurement for the degree of obesity in children and young people, indicating to what degree an individual BMI lies above or below the median BMI value.
A meta-analysis by Ho et al 18 concluded that lifestyle interventions can lead to improvements in weight and cardiometabolic outcomes in child obesity. However, while numerous lifestyle intervention programmes to tackle childhood obesity are conducted across the UK, and many describe statistically significant reductions in BMI-SDS,19 these results do not necessarily translate into clinical benefit for the individual. How reducing BMI-SDS in a trial translates to a reduction in adiposity is uncertain.
Paediatric weight management guidelines exist in many countries to promote best practice, but at present many of these recommendations are based on low-grade scientific evidence.20 Understanding how much BMI must be reduced to positively affect body composition and metabolic health is important to ensure that treatment interventions are appropriately designed and evaluated.21
Given the scale of the obesity problem and the significant and sustained adverse effects on health, clinically effective paediatric weight management treatment options are vital. A meta-analysis of cardiovascular disease risk in healthy children and its association with BMI has been conducted,22 but there is yet to be a systematic quantification of the reduction in BMI required to improve adiposity in the paediatric population with obesity.
It is important to highlight that when assessing interventions designed to manage overweight and obesity in children and adolescents, it is essential to recognise that measures such as BMI and derived SDS are surrogates of the real purpose: reduction of adiposity, fat being the key organ involved in metabolic complications.23 To rigorously assess the clinical and cost-effectiveness of weight management interventions in young people, it is first necessary to understand what BMI-SDS change means in terms of key outcomes such as effects on adiposity. This paper is designed to put BMI-SDS changes in context when considering improvement in adiposity (fatness). Through meta-regression analysis, we explore the potential impact of BMI-SDS reduction in terms of change in percentage body fat. The outcome of which will both inform clinical guidelines for paediatric weight management interventions and guide outcome measures in future clinical trials.
Objective
This paper aims to establish the minimum change in BMI-SDS needed to effect improvements in adiposity markers of children and adolescents with obesity. This is the first of a series of three papers reporting on the findings from studies identified in a large systematic review (n=90 studies; searched up to May 2017) and focuses on the evidence in relation to adiposity (percentage body fat); the others relating to metabolic and cardiovascular health.
Methods
The studies included in this paper were identified as part of large-scale systematic review (PROSPERO CRD42016025317). The protocol for this systematic review is available: https://doi.org/10.1186/s13643-016-0299-0. The final search was conducted in May 2017, the review was completed in January 2018 and the results are still being evaluated.
Participants
Studies with participants aged 4–19 years with a diagnosis of obesity using defined BMI thresholds were considered for inclusion. BMI-SDS was calculated as a function of the degree of obesity of the subjects when compared with BMI references. BMI standards included, but were not limited to, the 98th percentile on the UK 1990 growth reference chart,24 95th percentile on the US Centre for Disease Control and Prevention growth chart,25 the International Obesity Task Force (IOTF) BMI for age cut-points26 and the WHO growth references,27 28 in addition to country-specific obesity thresholds using BMI reference data from their paediatric populations. Studies that included overweight, as opposed to obese, individuals, pregnant females or those with a critical illness, endocrine disorders or syndromic obesity were excluded from this review.
Interventions
Studies of lifestyle treatment interventions that included dietary, physical activity and/or behavioural components with the objective of reducing obesity were included. Interventions of <2 weeks duration and those that involved surgical and/or pharmacological components (eg, bariatric surgery, drug therapy) were excluded. Studies focused on obesity prevention were also excluded. No restrictions were imposed regarding the setting or delivery of the interventions.
Outcome measures
To meet the inclusion criteria of the full systematic review, interventions had to report baseline (preintervention) and postintervention BMI-SDS or change measurements of BMI-SDS plus one or more markers of metabolic health (please refer to the published protocol paper for a complete list of the metabolic health markers of interest; https://doi.org/10.1186/s13643-016-0299-0).
This paper focuses on change in BMI-SDS and adiposity measures other than BMI, including waist circumference and percentage body fat.
Study design
Completed, published, randomised controlled trials (RCTs) and non-randomised studies (cohort studies) of lifestyle treatment interventions for children and adolescents with obesity, with or without follow-up.
Ethics
Ethical approval was not required as this paper reviewed published studies only.
Patient and public involvement
There was no patient or public involvement in this review of published studies.
Information sources and search methods
Studies were identified by searching five electronic databases from inception to May 2017 (AMED, Embase, MEDLINE via OVID, Web of Science and CENTRAL via Cochrane library), alongside scanning reference lists of included articles and through consultation with experts in the field. The search strategy for MEDLINE database is presented in online supplementary appendix 1.
bmjopen-2018-028231supp001.pdf (11.6KB, pdf)
Study selection and data extraction
Titles and abstracts were assessed for eligibility and the data outcome measures described previously were extracted by two independent reviewers from the review team using a standardised data extraction template, which was piloted by both reviewers before starting the review to ensure consistency.
Quality assessment
The focus of this study is the relationship between change in BMI-SDS and change in metabolic health parameters, rather than the specific treatment interventions that effect those changes. Therefore, risk of bias tools, such as the Cochrane Risk of Bias tool,29 were not considered appropriate. The included studies were assessed for methodological quality by two members of the review team during the data extraction process using the Quality Assessment tool used in the 2004 Health Technology Assessment (HTA) systematic review of the long-term effects and economic consequences of treatments for obesity and implications for health improvement.30 This Quality Assessment tool comprises 20 questions which are added together to give a final score and a percentage rating, from which a level of quality is assigned. Any discrepancies in Quality Assessment scoring were resolved through discussion.
Analysis
We carried out random-effects meta-regression as implemented in Stata31 to try to quantify the relationship between mean change in BMI-SDS (independent, predictor variable) and mean change in percentage body fat (target variable), where these were either reported, or were able to be calculated from reported data. Further details are given below. We were not trying to assess the relative effects of the various interventions, but rather to examine the relationship between these two outcomes. Meta-regression allows for residual heterogeneity in the target variable not explained by the predictor. Subsets from the same study (eg, intervention vs control, boys vs girls, see below) were regarded as independent observations provided there was no data duplication.
Results
Search results
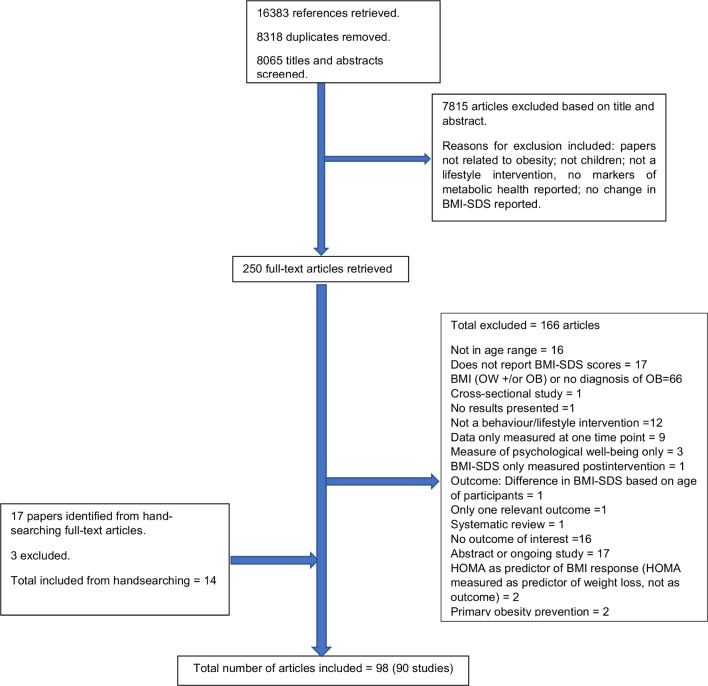
In total, 98 published articles relating to 90 different studies met the inclusion criteria for the entire systematic review. See figure 1 for a flow diagram illustrating the number of papers excluded at each stage of the review. For studies reported in multiple publications, the reference that provided the most comprehensive information has been used (see footnote of table 1 for details).
Figure 1.
Flow diagram from the systematic review that identified the included studies. BMI-SDS: body mass index-SD score; HOMA, homeostatic model assessment (method of assessing insulin resistance); OB: obese; OW: overweight.
Table 1.
Characteristics of studies reporting adiposity outcomes with results of mean change in BMI-SDS and percentage body fat
| Author, Country, (Intervention name) |
Study design: Sample size (n) Analysed (An) |
Obesity definition | Age range (inclusion): Mean ± (SD) Sex (% F) |
Pubertal status measured | Diet D)/ Exercise (E)/ D+E: Setting |
Format & content | Duration (months): Follow up (months) |
Method of % body fat measurement | Δ BMI SDS/ z-score by subgroup when reported |
Δ % body fat score by subgroup when reported |
|
| 1 | Bell et al
52
Australia |
Cohort Total = 14 (14) |
BMI ≥95th %ile | Age range: 9–16 12.70(2.32); F=43% |
Yes—Tanner | E: community |
8 weeks structured circuits exercise training: 3 x 1hr sessions/week. No standard dietary modifications. | 2: 0 |
DXA | All: −0.03 | All: −0.57 |
| 2 | Bock et al
53
Canada HIP KIDS |
Cohort: Total = 42 (41) |
BMI ≥95th %ile (CDC) |
Age range: 8–17 12.8 ±3.14; F=50% |
Yes—Tanner | D+E: Clinic (Hospital) |
Intensive phase (3 months): bi-weekly 90 min counselling. Maintenance phase (9 months): alternating mthly GP or individual sessions (90 mins). Sessions focus on exercise/psychosocial/behavioural aspects. | 12: 0 |
BIA | All: −0.04 | All: −1.39 |
| 3 | Bruyndonckz et al
36
Belgium |
Quasi-RCT: Total = 61 IG = 33 (27) CG = 28 (21) |
BMI ≥97th %ile adolescents <16 years; BMI ≥35 adolescents ≥16 years | Age range: 12–18 IG: 15.4±1.5; F = 79% CG: 15.1±1.2; F=73% |
NR | D+E: Clinic (Hospital) |
Intervention: Dietary restriction 1500-1800 kcal/day + 2 hrs/day supervised play/lifestyle activities + 2hrs/wk PE + 3 x 40min/wk supervised training session. Control: Usual care. |
10: 0 |
Subsample also measured using DXA | IG: −1.21 CG: 0.13 |
IG: −11.30 CG: 0.4 |
| 4 | Bustos et al
54
Chile |
Cohort: Total = 50 (28 completed) |
CDC | Age range: NR 9.5 ±1.9; F=48% |
NR | D+E: Academic Institution |
Nutrition/behavioural modification session 40 min/wk + PA 50 min x2/wk+ Family support every 15 days for first 2 months, then monthly. | 8: 0 |
DXA | All: −0.3 | All: −3.00 |
| 5 | Calcaterra et al 55 Italy | Cohort: Total = 22 (22) | BMI > 95th %ile | Age range: 9−16 13.23 ± 1.76; F=41% |
Yes - Tanner | E: Academic Institution |
2 x 90 mins exercise training sessions/wk | 3: 0 |
BIA | All: −0.15 | All: −3.30 |
| 6 | Dobe et al
46 Germany OBELDICKS – mini |
Cohort: Total = 103 (103) |
>97th to 99.5 percentile | Age range: 4−8 6.1 ±1 F=56% |
NR | D+E: Academic Institution |
Obeldicks mini: focus on training parents (22.5 hrs for parents, 4.5 hrs for children). Group sessions. Parents+children classes every 4th session, Children’s classes: 9 x monthly sessions (30 mins): 1 x introduction; 3 x diet; 5 x eating habits Parenting classes: 13 x monthly sessions (1.5 hrs): 1x introduction 1x medicine 3x nutrition 5x eating habits + education tips 3x discussion circle Individual consultation: every 2 months (30 mins) Exercise: 50 x weekly sessions (1.5 hrs) |
12: 0 |
BIA | Obeldicks-mini: −0.46 | Obeldicks mini: −3.00 |
| 7 | Farpour-Lambert et al
56
Switzerland |
RCT: Total = 44 IG= 22 (22) OC =22 (22) |
BMI >97th %ile | Age range: 6-11 8.9 ± 1.5 IG: F=59% OC: F= 68% |
Yes | E Clinic (Hospital) |
180 min/wk PA + 135 min/wk PE | 3: 0 |
Skinfold measurements | IG: -0.1 CG: 0 |
IG: -1.50 CG: 0.80 |
| 8 | Ford et al
48 57
UK |
RCT: Total = 106 (91) Gp1 SC = 52 (46) Gp 2 Mandometer = 54 (45) |
BMI ≥95th %ile (CDC) | Mandometer: 9.0−16.9 SC: 9.1−17.5 Mandometer: 12.7±2.2 SC: 12.5±2.3 overall F=56% |
Yes | D Clinic (Hospital) |
Mandometer device to regulate rate of eating and total intake vs SC | 12: 0 |
DXA | IG: −0.36 CG: −0.14 |
IG: −4.60 CG: −1.30 |
| 9 | Gajewska et al
37
Poland |
Cohort: Total = 100 (76) With WL =71 (56) Without WL = 29 (20) |
BMI SDS >2 | Age range: 5−10 with WL: 8.1 (6.8−9.2); F= 51% without WL: 8.8(7.3−9.6); F=59% overall F = 53% |
Reported with Tanner stage, any with pubertal develop-ment excluded. | D+E: Community & Academic institution |
3-month intervention, low energy diet (1200−1400kcal), 3−5 meals every day, instructions concerning PA, 10−14 food day diary, 3-day food diary. | 3: 0 |
BIA | WL: −0.98 No WL:−0.2 |
WL: −2.90 No WL:0.30 |
| 10 | Garanty-Bogacka et al
58
Poland |
Cohort: Total = 50 (50) |
BMI >97th %ile (Polish ref pop.) | Age range: 8−18 14.2 ±2.6; F=58% |
Yes | D+E: Clinic (Hospital) |
Exercise therapy (Instructions in PA + reducing sedentary behaviour) + reduction in fat and sugar intake. | 6: 0 |
Skinfold measurements & Lohman’s formula | All:−1 | All: −4.70 |
| 11 | Grønbæk et al
59 & Kazankov et al
60
Denmark Julemaerkehjemmet Hobro (same cohort) |
Cohort: Total = 117 (117) (n=71 attended 12 mth FU) |
NR. Obese. BL BMI-SDS: 2.93±0.52 |
Age range: NR 12.1 ±1.3 F=56% |
NR | D+E: Community |
Individually designed healthy diet + moderately strenuous PA program (at least 1hr/day). | 2.5 months/10 weeks: 12 |
BIA | All: −0.63 | All: −4.30 |
| 12 | Hvidt et al
61
Denmark |
Cohort: Total = 61 (61) |
Children’s Obesity Clinic; BMI >90th %ile (Danish ref pop.) = z-score 1.28. BL BMI-SDS: 2.73±0.60 |
Age range: 10−18 Median: 12.5 F=54% |
NR | D+E: Clinic (Hospital) |
Family-centred approach involving behaviour changing techniques (90 advice and advice strategies on low-calorie diet + activity for example, 10−20 items aimed to reduce obesity). | 12: 0 |
BIA | All: −0.21 | All: −3.40 |
| 13 | Kirk et al
47
USA |
Cohort: Total = 177 (177) Children (5–10yrs) = 85 Adolescents (11–19yrs) = 92 |
BMI >95th %ile | Age range: 5–19 9.0±1.5 Overall F=61% Children: F = 24% Adolescents: F = 59% |
NR | D+E: Clinic (Hospital) |
Behavioural intervention with individualised behavioural goals for nutrition, PA & family support. | 5: 6 |
DXA | GP1: –0.18 GP2: –0.13 All: –0.15 |
GP1: –2.10 GP2: –2.40 All: –2.20 |
| 14 | Klijn et al
62
The Netherlands |
Cohort: Total = 15 (15) |
BMI >30 | Age range: 10–18 14.7 (2.1); F=NR |
NR | E: Community |
Aerobic exercise training programme – 12 weeks; 3 x 30–60 min aerobic group sessions/week (2x gym/outdoors, 1 x swimming pool). P.E teacher led. Diverse indoor, outdoor and swimming activities. | 3: 0 |
% body fat calculated by “dividing fat mass by total body mass” | All: –0.4 | All: –3.80 |
| 15 | Lazzer et al
63
Italy |
Cohort: Total = 19 Boys = 7 (7) Girls = 12 (12) |
BMI >97th %ile | Age range: 8–12 Boys: 9.9±1.6 Girls: 11.2±1.5 Overall F=63% |
Yes – Tanner |
D+E: Community |
2 x 50min/wk endurance training + 2hr/wk PE lessons + 1 x wk child & parent dietetic class + 1 x wk psychological group class. | 8: 12 |
DXA | Boys: –0.4 Girls: –0.2 |
Boys: –4.00 Girls: –2.20 |
| 16 | Meyer et al
64
Germany |
RCT: Total = 67 IG=33 (33) OC=34 (34) |
BMI >97th %ile (German paediatric population) | Age range: 11–16 IG: 13.7±2.1; F=48% OC: 14.1±2.4; F =50% |
Yes - Tanner | E: Clinic (Hospital) |
3 x exercise sessions (Monday: swimming and aqua aerobic training 60 min + Wednesday sports games 90 min + Friday walking 60 min)/ wk; Control: Maintain current level of PA |
6: 0 |
BIA | IG: –0.43 CG: –0.14 |
IG: –1.00 CG: 0.00 |
| 17 | Miraglia et al
65
Brazil |
Cohort: Total = 27 (27) |
BMI z-score >2 | Age range: 6–13 Median 10.3; F=48% |
NR | D+E: Clinic (Hospital) |
AmO: Outpatient Ambulatory. Obesity outpatient clinic - lifestyle change based on goals agreed relative to feeding habits & physical exercise, followed mthly. 12 months: Subjects assessed at inclusion & after 12 months of FU to obtain anthropometric & adipokine measurements. | 12: 0 |
BIA | All: –0.4 | All: –0.10 |
| 18 | Morell-Azanza et al
66 & Rendo-Urteaga et al 67 Spain (same cohort) |
Cohort: Total = 54 (40) high responders =21 low responders = 19 |
OW/OB as per Cole et al 2000 | Age range: 7–15 Mean =11 F=53% (of N analysed) |
Yes – Tanner |
D: Clinic (Hospital) |
Moderate energy-restricted diet + nutritional education sessions with dietitian + family involvement. | 2.5: 0 |
BIA | HR: –0.79 LR: –0.18 HR: –0.64 LR: –0.07 |
HR: –3.10 LR: –0.60 HR: –2.49 LR: –0.37 |
| 19 | Murer et al
68 & Aeberli et al 69 Switzerland (same cohort) |
Cohort: Total = 206 (203) |
BMI >98th %ile | Age range: 10–18 14.1±1.9; F=44% |
NR | D+E: Clinic, hospital |
Moderate caloric restriction.2 x 60–90 min/day endurance exercise + 4–5 hr/wk. exercise session + behaviour modification. | 2: 0 |
BIA | All: –0.42 | All: –5.50 |
| 20 | Murdolo et al 70 Italy | Cohort: Total = 53(53) Responders = 44 Non-responders = 9 |
NR | Age range: 5–13 Responders: 9.0±1.1; F=50% Non-responders: 2.09±0.32; F=33% |
Yes – Tanner |
D+E: Community |
Educational Wt Excess Reduction Program | 24: >6 |
BIA | Responders: –0.44 Non-responders: 0.11 |
Responders:–2.90 Non-responders: –2.00 |
| 21 | Ning et al
71
& BEAN et al 72 USA TEENS (same cohort) |
Cohort: Total = 145**(145) |
BMI ≥95th %ile (CDC) | Age range: 11–18 13.1 F=65% |
NR | D+E: Academic Institution |
12 x 30 min nutritional session with adolescent and parent/s + Education/behavioural support sessions once every 2 wks, or alternating wks + PA 3 x 60 min/wk during initial 12 wks, then minimum of twice/wk. | 6: 0 |
DXA | All: –0.1 | All: –2.40 |
| 22 | Pacifico et al
73
Italy |
Cohort: Total = 120 (120) |
BMI >95th %ile | Age range: (11.5–12.2) 11.9; F=35% |
Yes (method ND) | D+E: Clinic (Hospital) |
Hypocaloric diet (25–30 Kcal/kg/day) + 60 min/day ~ 5 days/wk moderate exercise + Reduce sedentary behaviour. | 12: 0 |
NR | All: –0.32 | All: –2.10 |
| 23 | Racil et al
32
Tunisia |
RCT: Total = 34 HIIT = 11 (11) MIIT = 11 (11) OC = 12 (12) |
BMI >97th %ile (French standards) | Age range: NR HIIT: 15.6±0.7 MIIT: 16.3±0.52 OC:15.9±1.2 Overall F=100% |
Yes -Tanner | D+E: Community |
4-day diet records + HIIT or MIIT. Interval training program 3 x /wk on non-consecutive days. | 3: 0 |
BIA | HIT: –0.4 MIT: –0.3 OC: 0 |
HIT: –2.90 MIT: –2.00 OC: –0.40 |
| 24 | Racil et al
33
Tunisia |
RCT: Total = 47 HIIT = 17 (17) MIIT = 16 (16) OC = 14 |
BMI >97th %ile (French standards) | Age range: NR 14.2±1.2; F=100% |
NR | E: Academic Institution |
HIIT (Warm up + Interval training at 100%/50% MAS + Cooling down); MIIT (Warm up + Interval training 80%/50% MAS + Cooling down) |
3: 0 |
BIA | HIT: –0.3 MIT: –0.3 OC: 0 |
HIT:- –3.90 MIT: –3.40 OC: –0.50 |
| 25 | Reinehr et al
38 Germany OBELDICKS |
Cohort: Total = 42 (42) |
BMI ≥97th %ile | Age range: 6.1–15.1 10.2; F=57% |
Yes - Tanner | D+E: Clinic (Hospital) |
Obeldicks: Intensive phase 3 months (Parents’ course 2x/month + Behaviour therapy 2x/month + Nutritional course 2x/month + Exercise therapy 1x/wk) + Establishing phase 3 months (Talk rounds for parents 1x/month + Psychological therapy + Exercise therapy 1x/wk) + Establishing phase 2 for 3 months (Psychological therapy + Exercise therapy 1x/wk) + Establishing phase 3 for 3 months (Exercise therapy 1x/wk). | 12: 0 |
% body fat skinfold thickness |
Sig. WL –0.9 NS WL: –0.2 |
Sig. WL: –7.50 NS WL: –3.00 |
| 26 | Reinehr et al
74 75
Germany OBELDICKS |
Cohort: Ob + Sub. WL = 25 Ob + no change = 18 Normal control = 19 (BL data only) |
IOTF using pop. -specific data | Ob: 10.8±2.6; F=61% Lean C: 10.3±2.9; F=58% Ob + Sub. WL : F= 68% Ob + no change: F = 50% |
Yes -Tanner | D+E: Clinic (Hospital) |
Obeldicks | 12: 0 |
% body fat skinfold thickness |
WL: –0.6 No WL: –0.1 |
WL: –8.00 No WL: 0.00 |
| 27 | Rohrer et al
76 Germany Fit Kids |
Cohort: Total = 22 (22) Unchanged BMI= 12 Reduced BMI = 10 |
BMI >99.5th %ile (German standard values) or BMI >97th %ile with obesity-associated risk factors or BMI >90th %ile with obesity-associated disease |
Age range: 7–15 Median: 11.9 F=27% Unchanged BM: F = 33% Reduced BMI: F=20% |
NR | D+E: Community |
Physical exercise (2 x wk, 100 hrs in total) + Nutritional/heath education and psychological care for the child (x wk, 43.5 hrs total) and parent/s (2 x wk, 12 hrs total). | 12: 0 |
BIA | Increased BMI: 0.12 Reduced BMI: –0.35 |
Increased BMI: 1.05 Reduced BMI:–0.05 |
| 28 | Rolland-Cachera et al
77
France |
RCT: Total = 99 PROT- = 61 (53) PROT+ =60 (46) |
BMI > 97th %ile (French reference values) | Age range: 11-16 PROT- = 14.1±1.2; F = 74% PROT + =14.4±1.3; F = 72% |
NR | D+E: Academic Institution |
Wt reducing diet; 7hr/wk vigorous sports + 7hr/wk outdoor activities; advice on nutrition & PA during wkends/holidays. | 9: 12+24 |
BIA | PROT- :–2.6 PROT+:–2.5 |
PROT- :–12.40 PROT+:–12.10 |
| 29 | Roth et al
78 Germany OBELDICKS |
Cohort: Total = 69 OB + WL = 32 OB + with WL = 37 |
OB as per IOTF criteria | NR – (see Obeldicks age range) Ob with WL: 11.8±2.0; F=50% Ob without WL: 12.1±2.1; F=51% Normal wt: 12.3±3.0; F=45% |
Yes - Tanner | D+E: Clinic (Hospital) |
Obeldicks | 12: 0 |
% body fat skinfold thickness | WL: –0.69 No WL: 0.03 |
WL: –9.60 No WL: –4.30 |
| 30 | Savoye et al
79
USA Bright Bodies |
Cohort: Total = 33 (25) SMP = 10 (8) BFC = 23 (17) |
BMI ≥95th %ile | Age range: 11–16 13.5±0.3; SMP:13.3±0.6; F=75% BFC: 13.6±0.3; F= 65% |
NR | D+E: Academic Institution |
Bright Bodies Weight Management Program: nutrition education, exercise, behavioural modification. 2 x 30 min exercise sessions + 1 x 45 min nutrition/behaviour medication group session per week. 4 levels: Beginner, Intermediate i, Intermediate ii, Advanced. All levels 12 weeks duration. Monthly maintenance classes after 1 yr (support-group style) |
12: 12 |
BIA | SMP: –0.36 BFC: –0.12 |
SMP:–6.50 BFC: –4.20 |
| 31 | Savoye et al
80 81
USA Bright Bodies (data taken from 2011 paper) |
RCT+ Long term FU results (cohort) RCT Total = 174 BB = 105 CC = 69 1 YR ANALYSIS BB = 75 CC = 44 |
BMI ≥95th %ile (CDC) | Age range: 8–16 BB: 12.0±2.5; F=56% CC: 12.5±2.3; F=68% |
NR | D+E: Academic Institution (local school). |
Bright Bodies Weight Management Program: nutrition education, exercise, behavioural modification. 2 x sessions/wk for 6 months, then biweekly for next 6 months. BB: 2x50 min exercise + 1x40 min nutrition/behaviour modification per wk + 12 months no active intervention. Control group: standard care – paed. obesity clinic (biannual clinic appt; diet + exercise counselling) Structured tx & teaching program (28 x 45 min therapeutic sessions for example, PA, nutrition, healthy cooking) |
12: 12 FU 1.5: 24 |
BIA | IG: –0.21 CG: 0.01 |
IG: –3.90 CG: 2.10 |
| 32 | Savoye et al
82
USA Bright Bodies |
RCT Total = 75 BB = 38 (31) CC = 37 (27) |
BMI ≥95th %ile | Age range: 10–16 BB: 12.7 (1.9); F=68% CC: 13.2 (1.8); F=62% |
Yes-Tanner | D+E: Academic Institution |
Bright Bodies Weight Management Program: nutrition education, exercise, behavioural modification. 2 x 30 min exercise sessions + 1 x 45 min nutrition/behaviour medication group session per week. 4 levels: Beginner, Intermediate I, Intermediate ii, Advanced. All levels 12 weeks duration. Monthly maintenance classes after 1 yr (support-group style) | 6: 0 |
BIA | BB: –0.05 CC: 0.04 |
BB: –3.30 CC: 0.40 |
| 33 | Schiel et al 83Germany | Cohort: Total = 143 (143) |
BMI-SDS ≥97th %ile | Age range: NR 13.9±2.4; F=62% |
NR | D+E: Clinic (Hospital) |
Structured Tx & Teaching Program (STTP): 28 x 45 min therapeutic sessions for example, PA, nutrition, healthy cooking |
1.5: 24 |
NR | All: –0.26 | All: –3.40 |
| 34 | Seabra et al 34 Portugal | Cohort: Total = 88 soccer = 29 (29) Trad. Act. = 29 (29) OC = 30 (30) |
BMI-SDS > 2 | Age range: 8–12 Soccer: 10.5±1.5 Trad. act: 11.0±1.6 OC=10.0±1.3 Overall F=0% |
Yes - Tanner | E: Community |
Soccer & trad. activity programmes (3 x 60-90min/wk) + 2 x 1hr at BL & 3 months later energy balance session. | 6: 0 |
DXA | Soccer: –0.2 Trad.: –0.2 CG: –0.1 |
Soccer:–2.20 Trad:–4.10 CG:3.10 |
| 35 | Truby et al
84
Australia |
RCT: Total = 87 SMC = 37 (33) SLF = 36 (32) WList OC = 14 (14) |
BMI >90th %ile (CDC) | Age range: 10–17 SMC: 13.2±1.9; F=73% SLF: 13.2±2.1; F=72% WList OC: 13.6±1.9; F=71% |
Yes -Tanner | D: Clinic (Hospital) |
Structured modified CHO diet (35% CHO; 30% protein; 35% fat), structured low-fat diet (55% CHO; 20% protein; 25% fat), Control (no dietary advice). |
3: 0 |
BIA | SLF: –0.09 SMC:–0.15 CG: 0.02 |
SLF: –0.13 SMC: –0.40 CG: 2.62 |
| 36 | Van der Baan-Slootweg et al
85
The Netherlands |
RCT: Total = 90 Inpt. = 45 (37) AmO = 45 (36) |
BMI z score ≥ 3.0 or > 2.3 with OB-related health problems | Age range: 8–18 Inpt: 13.8±2.3; F=58% AmO: 13.9±2.5; F=58% |
NR | D+E: Clinic (Hospital) |
Inpt. (Hospitalised 26 wks on working days - 4 days/wk 30-60min exercise + nutrition/BM once/wk + parents/caregivers 3 x 1hr lesson on nutrition/BM); Ambulatory (12 visits at increasing time intervals - 1 hr exercise session + encouraged 3 x exercise/wk + 1 hr educational programme + 30 min nutrition education). |
6: 24 |
BIA | InpT: –0.6 AmO: –0.35 |
InP: –3.34 AmO:–7.87 |
| 37 | Visuthranukul et al
86
Thailand |
RCT: Total = 70 (52) I = 35(25) OC = 35 (27) |
ND. BL BMI z-score: I = 3.7±0.9 C = 3.6±1.6 |
Age range: 9–16 I = 11.9±1.9; F=36% C = 12.0±2.1; F=30% |
Yes -Tanner | D: Clinic (Hospital) |
I (Low GI diet + Energy restriction 1400-1500 kcal/day + Increased exercise); OC (Energy restriction 1200-1300 kcal/day + Low fat/high fibre diet + Increased exercise). |
6: 0 |
BIA | IG:–0.3 CG: –0.3 |
IG:0.10 CG:0.10 |
| 38 | Vitola et al
87
USA |
Cohort: Total = 8(7) |
BMI ≥95th %ile | Age range: NR 15.3±0.6; F=12.8% |
Yes -Tanner | D+E: Clinic (Hospital) |
Individual behavioural therapy sessions with psychologist. Parents involvement encouraged. Self-monitoring of PA & food intake. Gradual reduction of caloric intake to ≈1200-1500 kcal/day. Ongoing therapy - wt loss therapy repeated when 5% body wt lost & wt stable for at least 4 wks | NR | DXA | All: –0.3 | All: –5.30 |
| 39 | Wickham et al
88 & Evans et al 89 USA TEENS (same cohort) |
Cohort: Total = 168 (64)** Completers only = 57 |
BMI ≥95th %ile (CDC) | Age range: 11–18 13.9±1.9; F=62% |
NR | D+E: Academic Institution |
Exercise 1 day/wk at facility + 2 additional exercise days at facility of ppts’ choice + 30 min/wk nutrition education/behavioural support sessions. | 6: 0 |
BIA | Completers: –0.07 |
Completers:–1.30 |
For studies reported in multiple publications, the reference that provided the most comprehensive information has been used (thus Ning et al 71 includes data from Bean et al 72; Evans et al 89 is reported under Wickham et al 88; Aeberli et al 69 is reported under Murer et al 68; Rendo-Urteaga et al 67 is reported under Morell-Azanza et al 66 and Kazankov et al 60 is reported under Grønbæk et al.59
*studies with change in % body fat included in the analysis.
**Minor discrepancies in reporting of data in papers.
KEY: %ile, percentile; AmO, outpatient ambulatory; An., analysed; apt., appointment; BB, Bright Bodies; BIA, bioelectrical impedance analysis; BFC, better food choices; BL, baseline; BM, behaviour modification; BMI, body mass index; C, control; CBT, cognitive behavioural therapy; CDC, Centre for Disease Control; CG, control group; CHO, carbohydrate; D, diet; DXA, Dual-energy X-ray absorption; E, exercise; FBBT, family-based behavioural treatment; F, female; FU, follow up; GI, glycaemic index; GT, group therapy; HGI, high glycaemic index; HIIT, high intensity interval training; hr, hour; HZ, heterozygous; HO, homozygous; ht, height; I, intervention; IG, intervention group; IOTF, International Obesity Task Force; Inpt., inpatient; LGI, low glycaemic index; LMS, least-mean-squares; LS, long stay; MAS, maximal aerobic speed; MIIT, moderate intensity interval training; min, minute; MO, morbidly obese; norm., normal; n, number; NAFLD, Non-alcoholic fatty liver disease; ND, not described; NR, not reported; OB, obese; OC, obese control; OW, overweight; paed., paediatric; PA, physical activity; PE, physical activity; PROT, protein; RCT, randomised controlled trial; SD, standard deviation; SDS, standard deviation score; SMP, structured meal plan; SS, short stay; Sub., substantial; SMC, structured modified carbohydrate diet; trad., traditional; trad. act, traditional activity; tx, treatment; wk, week; WList OC, wait list obese control; WL, weight loss; wt, weight; X-over, crossover; yr, year.
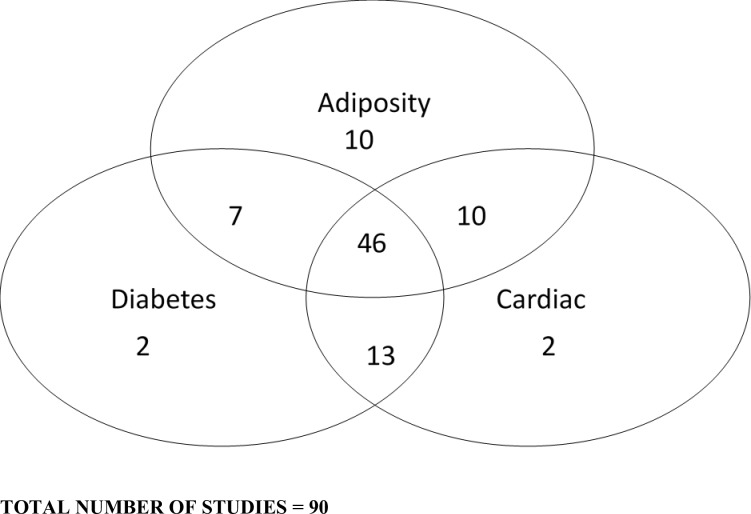
The Venn diagram (figure 2) illustrates how many studies were identified for the various markers of metabolic health. Seventy-three studies assessed and reported adiposity measures. The adiposity measures reported included percentage body fat, body fat-SDS, body mass, fat mass, fat-free mass, waist circumference and waist circumference-SDS. The 68 studies that examined diabetes/inflammation measures (HOMA-IR, insulin, glucose, C reactive protein, interleukin-6, alanine transaminase and the 71 studies examining cardiac measures (eg, lipids, cholesterol, blood pressure) will be reported separately.
Figure 2.
Venn diagram illustrating the markers of metabolic health measured.
Studies for inclusion in meta-regression analysis
Seventy-three studies assessed and reported adiposity measures. Of the different adiposity measures that were reported in these studies (percentage body fat, body fat-SDS, body mass, fat mass, fat-free mass, waist circumference and waist circumference-SDS), we elected to examine percentage body fat as it was far more frequently reported across studies. Therefore, of the 73 adiposity studies, we conducted our meta-regression on 39 studies which reported percentage body fat values. These studies are presented in table 1 with the corresponding changes in BMI-SDS.
The results of five studies were duplicated in multiple papers, thus the reference that reported the most comprehensive information was used in the analysis; see table 1 footnote for details. Thirty-four studies were excluded from the meta-analysis; the characteristics of the excluded studies, along with the reason for exclusion, are summarised in online supplementary appendix 2.
bmjopen-2018-028231supp002.pdf (231.9KB, pdf)
Narrative description of studies that reported BMI-SDS and percentage body fat
Of the 39 studies that reported percentage body fat included in our analysis, 7 were conducted in both Germany and the USA, 4 in Italy, followed by Australia (n=2), Denmark (n=2), the Netherlands (n=2), Poland (n=2), Switzerland (n=2), Tunisia (n=2) and one each in Belgium, Brazil, Canada, Chile, France, Portugal, Spain, Thailand and the UK. There were country-specific variations in the definition of obesity, with most studies defining obesity by participants having a BMI-SDS >2, or a BMI percentile of at least >90th percentile. Most of studies used a cohort design (n=27), 11 were RCTs, of which 1 included results from a cohort of the original RCT. There was also one study which adopted a quasi-randomised design.
Most studies (n=20) conducted their intervention in the hospital clinic setting. Eight studies conducted the intervention in the community setting and 10 in academic institutions. One conducted the intervention in a mixed setting, reporting use of both a community setting and academic institution.
Twenty-eight studies conducted interventions that comprised both diet and exercise components. The remaining studies (n=11) used interventions that focused either on exercise or diet only. The duration of the interventions ranged from 15 days to 24 months. The majority of studies (n=29; 74%) did not report any follow-up after the lifestyle treatment intervention. The duration of follow-up in the studies where it was conducted and reported, ranged from 6 months to 2 years.
The sample sizes of the included studies ranged from 8 to 203 participants. The age of the participants ranged from 4 to 19 years. Studies predominantly had a mix of males and females (95%) with only three studies specifically focused on either only girls32 33 or boys.34 Seventeen studies (44%) measured pubertal development of participants according to Marshall and Tanner staging, with pubertal status categorised into three groups: prepubertal, pubertal and late/postpubertal.35 Four studies (10%) reported that pubertal development was measured but the methodology was not defined. Eighteen studies (46%) did not report any measures of pubertal development.
Quality assessment
The quality of the conduct of each study was assessed using the same criteria as the HTA systematic review of the long-term effects and economic consequences of treatments for obesity and implications for health improvement.30 The results of the quality assessment can be found in table 2. In summary, none of the 39 studies that reported percentage body fat were considered to be of poor quality, 21 studies (54%) were rated as being of moderate quality and 18 studies (46%) achieved a score over 81% indicating high quality.
Table 2.
Quality assessment of included studies
| Sample | Conduct of study | Follow-up | Analysis | Interpretation | Total (x/40) | Overall rating | |||||||||||||||||
| Study | 1. Aims clearly stared | 2. Sample size justified | 3. Age of participant defined | 4. Measurements at start clearly stated? | 5. Measurements likely to be valid and reliable? | 6. Risk factors recorded clearly? | 7. Was the intervention before follow-up defined? | 8. Setting of the study clear? | 9. Is mode of assessment described? | 10. Did untoward events occur during the study? | 11. Was there a follow-up? | 12. Was follow-up necessary? | 13. Are losses to follow-up defined? | 14. Was basic data adequately described? | 15. Do numbers add up? | 16. Did analysis allow for passage of time? | 17. Was statistical significance assessed? | 18. Were the main findings interpreted adequately? | 19. Were null/negative findings interpreted? | 20. Are important effects overlooked? | |||
| 1 | Bell et al 52 | Yes | Yes | Yes | Yes | Yes | No | Yes | ? | Yes | No | No | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | No | 35 | 87.5 |
| 2 | Bock et al 53 | Yes | No | Yes | Yes | Yes | No | Yes | Yes | Yes | No | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | No | 36 | 90 |
| 3 | Bruyndonckx et al 36 | Yes | No | Yes | Yes | Yes | No | Yes | Yes | Yes | No | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | No | 36 | 90 |
| 4 | Bustos et al 54 | Yes | No | Yes | Yes | Yes | No | Yes | Yes | Yes | No | No | Yes | No | Yes | Yes | Yes | Yes | Yes | No | No | 30 | 75 |
| 5 | Calcaterra et al 55 | Yes | No | Yes | Yes | Yes | No | Yes | Yes | Yes | No | No | Yes | No | Yes | Yes | Yes | Yes | Yes | ? | No | 31 | 77.5 |
| 6 | Dobe et al e46 | ? | No | Yes | ? | ? | No | Yes | ? | ? | No | No | Yes | No | Yes | Yes | Yes | Yes | Yes | ? | No | 26 | 65 |
| 7 | Farpour-Lambert, et al 56 | Yes | Yes | Yes | Yes | Yes | No | Yes | ? | Yes | No | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | No | 37 | 92.5 |
| 8 | Ford et al 48 57 | Yes | Yes | Yes | Yes | Yes | No | Yes | Yes | Yes | ? | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | ? | 35 | 87.5 |
| 9 | Gajewska et al 37 | Yes | No | Yes | Yes | Yes | No | Yes | Yes | Yes | No | No | Yes | No | Yes | Yes | Yes | Yes | Yes | ? | No | 31 | 77.5 |
| 10 | Garanty-Bogacka et al 58 | Yes | No | Yes | Yes | Yes | ? | ? | ? | Yes | ? | No | ? | No | Yes | ? | Yes | Yes | Yes | ? | No | 26 | 65 |
| 11 | Grønbæk et al
59
Kazankov et al 60 |
Yes | ? | Yes | Yes | Yes | No | Yes | Yes | Yes | No | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | No | 37 | 92.5 |
| 12 | Hvidt et al 61 | Yes | No | Yes | Yes | Yes | No | Yes | Yes | Yes | No | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | ? | 34 | 85 |
| 13 | Kirk et al 47 | Yes | No | Yes | Yes | Yes | No | Yes | Yes | Yes | ? | No | Yes | ? | Yes | Yes | Yes | Yes | Yes | ? | ? | 29 | 72.5 |
| 14 | Klijn et al 62 | Yes | No | Yes | Yes | Yes | No | Yes | ? | Yes | No | No | Yes | No | No | Yes | Yes | Yes | Yes | No | No | 27 | 67.5 |
| 15 | Lazzer et al 63 | Yes | No | Yes | Yes | Yes | No | Yes | Yes | Yes | No | Yes | Yes | No | Yes | Yes | Yes | Yes | Yes | No | No | 32 | 80 |
| 16 | Meyer et al 64 | Yes | No | Yes | Yes | Yes | No | No | Yes | Yes | No | No | Yes | No | Yes | Yes | Yes | Yes | Yes | Yes | No | 30 | 75 |
| 17 | Miraglia et al 65 | Yes | No | No | Yes | Yes | No | Yes | ? | Yes | No | No | Yes | No | ? | Yes | Yes | Yes | ? | Yes | Yes | 25 | 62.5 |
| 18 | Morell-Azanza et al
66
Rendo-Urteaga et al 67 |
Yes | Yes | Yes | Yes | Yes | No | Yes | Yes | Yes | No | Yes | Yes | No | Yes | Yes | Yes | Yes | Yes | No | No | 32 | 80 |
| 19 | Murer et al
68
Aeberli et al 69 |
Yes | Yes | Yes | Yes | Yes | No | Yes | Yes | Yes | No | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | No | 38 | 92 |
| 20 | Murdolo et al 70 | Yes | No | Yes | Yes | Yes | No | No | No | Yes | No | Yes | Yes | No | Yes | Yes | Yes | Yes | Yes | No | No | 28 | 70 |
| 21 | Ning et al
71
Bean et al 72 |
Yes | No | Yes | Yes | Yes | No | Yes | Yes | Yes | No | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | No | No | 34 | 85 |
| 22 | Pacifico et al 73 | Yes | No | Yes | Yes | Yes | No | Yes | Yes | Yes | No | Yes | Yes | No | Yes | Yes | Yes | Yes | Yes | No | ? | 31 | 77.5 |
| 23 | Racil et al 32 | Yes | No | Yes | Yes | Yes | No | Yes | ? | Yes | No | No | ? | No | Yes | Yes | Yes | Yes | Yes | Yes | ? | 29 | 72.5 |
| 24 | Racil et al 33 | Yes | No | ? | Yes | Yes | No | Yes | ? | Yes | No | No | Yes | No | Yes | Yes | Yes | Yes | Yes | No | No | 28 | 70 |
| 25 | Reinehr et al 38 | Yes | No | Yes | Yes | Yes | No | Yes | Yes | Yes | No | No | Yes | No | Yes | Yes | Yes | Yes | Yes | No | ? | 29 | 72.5 |
| 26 | Reinehr et al 74 75 | Yes | No | Yes | Yes | Yes | No | Yes | Yes | Yes | No | Yes | Yes | No | Yes | Yes | Yes | Yes | Yes | Yes | No | 32 | 80 |
| 27 | Rohrer et al 76 | Yes | No | Yes | Yes | Yes | No | Yes | Yes | Yes | No | Yes | Yes | No | Yes | Yes | Yes | Yes | Yes | ? | No | 33 | 82.5 |
| 28 | Rolland-Cachera et al 77 | Yes | No | Yes | Yes | Yes | No | Yes | Yes | Yes | No | Yes | Yes | Yes | ? | Yes | Yes | Yes | Yes | No | No | 33 | 82.5 |
| 29 | Roth et al 78 | Yes | No | Yes | Yes | Yes | No | Yes | No | Yes | No | No | Yes | No | Yes | Yes | Yes | Yes | Yes | No | No | 28 | 70 |
| 30 | Savoye et al 79 | Yes | No | Yes | Yes | Yes | No | Yes | Yes | Yes | No | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | No | No | 34 | 85 |
| 31 | Savoye et al 80 81 | Yes | Yes | Yes | Yes | Yes | No | Yes | Yes | Yes | No | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | No | No | 36 | 90 |
| 32 | Savoye et al 82 | Yes | Yes | Yes | Yes | Yes | No | Yes | ? | Yes | No | No | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | No | 35 | 87.5 |
| 33 | Schiel et al 83 | Yes | No | ? | Yes | Yes | No | Yes | Yes | Yes | No | Yes | Yes | Yes | Yes | Yes | ? | Yes | No | ? | Yes | 29 | 72.5 |
| 34 | Seabra et al 34 | Yes | Yes | Yes | Yes | Yes | No | Yes | Yes | Yes | No | No | Yes | Yes | Yes | Yes | Yes | Yes | Yes | No | No | 34 | 85 |
| 35 | Truby et al 84 | Yes | Yes | Yes | Yes | Yes | No | Yes | Yes | Yes | No | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | No | 38 | 95 |
| 36 | van der Baan-Slootweg et al 85 | Yes | Yes | Yes | Yes | Yes | No | Yes | Yes | Yes | No | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | No | No | 36 | 90 |
| 37 | Visuthranukul et al 86 | Yes | Yes | Yes | Yes | Yes | No | Yes | Yes | Yes | No | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | No | 38 | 95 |
| 38 | Vitola et al 87 | Yes | No | Yes | Yes | Yes | No | ? | ? | Yes | No | No | Yes | No | Yes | Yes | Yes | Yes | Yes | No | No | 28 | 70 |
| 39 | Wickham et al
88
Evans et al 89 |
Yes | No | Yes | Yes | Yes | No | Yes | Yes | Yes | No | Yes | Yes | ? | Yes | Yes | Yes | Yes | No | Yes | ? | 30 | 75 |
For Q6. Were risk factors clearly recorded? We said ‘no’ rather than ‘unclear’ to all the studies that did not record risk factors.
For Q10. Did untoward events occur during the study? We said ‘no’ rather than unclear if not mentioned.
Rating: not satisfactory 1%–50%; moderate quality=51%–80%; high quality=81%.
?, unclear.
Quantitative analysis
From the 39 studies we identified all data subsets that reported a mean change in BMI-SDS, an associated mean change in percentage body fat (or prestudy and poststudy values from which these could be calculated) and the number of cases analysed. A few studies yielded only aggregated data for the whole study. For the others, typical data subsets included intervention versus control, male versus female or good versus poor responders (table 1), and these were used in preference to aggregated results if both were available. In all, there were 66 subsets, with numbers analysed totalling 2618.
SEs were required for the mean changes in percentage body fat and, if not given explicitly, were calculated, from either the SDs or the 95% CIs of the mean changes. In total, 22 data sets had SEs. For the remainder, the SEs were estimated from the SDs associated with the baseline and the postintervention percentage body fat values, making an assumption about the degree of correlation between them. The median and IQR of the correlation coefficients estimated from the nine data sets where both the SEs of mean change and the SDs for baseline and postintervention percentage body fat values were available was 0.81 (IQR 0.59–0.82) and 0.81 has been used in the following analysis.
A small number of data sets (n=6)36–38 only had medians and IQRs (or range) reported for the baseline and postintervention results; the mean and SDs were estimated from them.39
The meta-regression line was fitted and plotted together with the 95% prediction intervals for the change in percentage body fat across the study data sets. The smallest reduction of mean BMI-SDS associated with a reduction in mean percentage body fat was determined as the smallest reduction in mean BMI-SDS with an associated 95% prediction interval wholly below zero.
A series of sensitivity analyses were conducted. Sensitivity analysis 5A: using the 22 cases where the SEs of the mean change in percentage body fat were actually known, sensitivity analysis 5B: omission of two extreme values and sensitivity analysis 5C: assuming a correlation of 0.50 instead of 0.81. In further exploratory analyses, the percentage of girls and the length of the study (baseline to end of intervention) were added to see if these affected the prediction of mean change in percentage body fat.
Results from the quantitative analysis
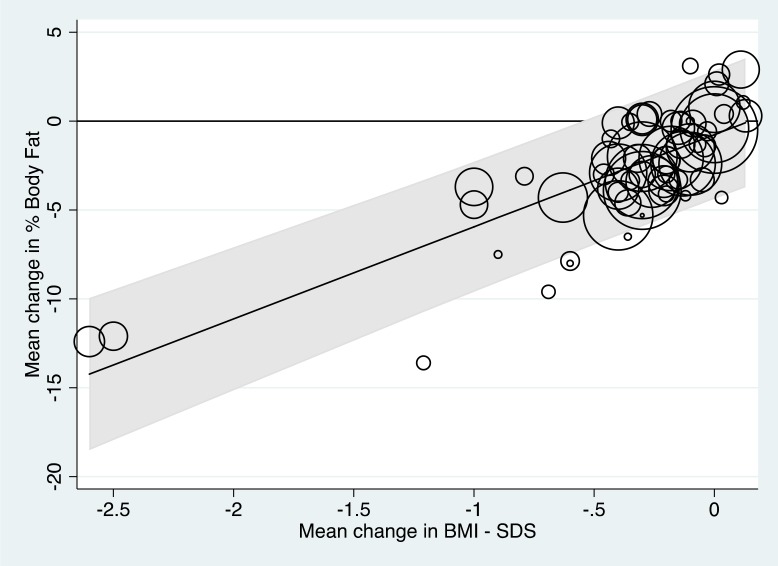
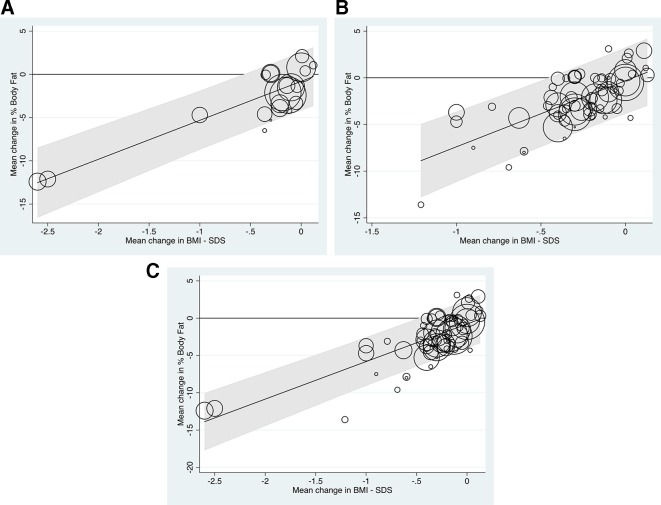
Figure 3 shows the results of the analysis and the fitted regression line. The circles represent the study results (ie, the mean changes in percentage body fat and mean changes in BMI-SDS) analysed for each study, with the size of the circles representing the precision of the mean change in percentage body fat, that is, the reciprocal of the SE squared.
Figure 3.
Meta-regression line showing the relationship between mean change in percentage body fat and body mass index-SD score (BMI-SDS) across the 39 studies (66 subsets) analysed.
The fitted regression line shown in figure 3 is:
Mean change in percentage body fat=5.179×mean change in BMI-SDS−0.767.
The regression slope was statistically significant (p<0.001), confirming a relationship between the mean loss of percentage body fat and the mean change in BMI-SDS across the data subsets; the proportion of the between-subset variance explained by the mean change in BMI-SDS (ie, ‘a type of adjusted R-squared’) was 68%. There was, however, significant between-subset heterogeneity with 89% of the percentage of the total residual variance attributable to this (ie, I2).2 It was further noted that when added to the model, neither the percentage girls in the study sets nor the durations of the interventions significantly improved the prediction of mean change in percentage body fat from the mean change in BMI-SDS (p=0.36, p=0.89, respectively).
Figure 3 also shows the 95% prediction intervals for the mean change in percentage body fat. The upper limit of the prediction interval was below 0 only when the mean reduction in BMI-SDS was >0.6, suggesting that any new study should aim to reduce the BMI-SDS by at least this amount to be confident of achieving a mean reduction of percentage body fat.
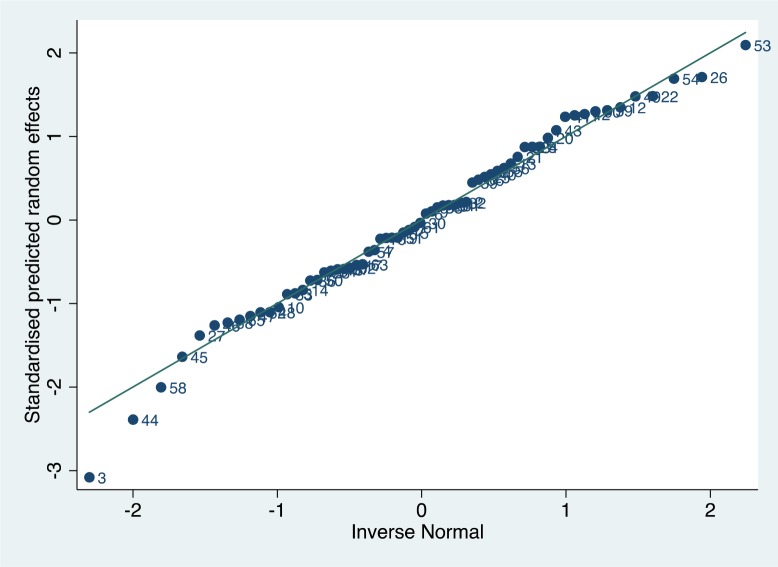
A normal plot for the standardised predicted random effects is shown in figure 4. Most were within ±2, although the data sets themselves were not wholly independent (as some came from the same studies).
Figure 4.
Normal plot for the standardised predicted random effects from the meta-regression.
None of the sensitivity analyses conducted (figure 5) significantly altered the findings, namely that a mean change of 0.6 or more in BMI-SDS was associated with a definitive mean loss in percentage body fat. In figure 5B, with the exclusion of the two extreme data points, the linear trend can be seen more clearly across the range of mean BMI-SDS losses.
Figure 5.
Sensitivity analysis. BMI-SDS, body mass index- SD score. (A) Analyses based on the 22 subsets where the SEs of the mean changes in percentage Body Fat were known (Fitted meta-regression line: Mean change in % body fat = 4.502 x Mean change in BMI-SDS – 0.810). (B) Analysis using all data subsets but excluding two extreme values (reduction of mean BMI-SDS of more than 1.5), leaving 64 subsets (Fitted meta-regression line: Mean change in % body fat = 7.078 x Mean change in BMI-SDS – 0.318). (C) Analysis using all 66 data subsets but using a correlation coefficient of 0.50, rather than 0.81, to estimate the SE of the mean change in % Body Fat for the 66–22=44 subsets where this was not available (Fitted regression line: Mean change in % body fat = 5.039 x Mean change in BMI-SDS - 0.783).
Discussion
Summary of main results
This is the first of a series of papers that report on studies identified in a large systematic review. The objective of this paper was to attempt to establish the minimum change in BMI-SDS needed to achieve improvements in body fat in children and adolescents with obesity; BMI-SDS being by far the most frequently reported outcome in terms of weight management trial interventions in childhood. Seventy-three of the 90 included studies reported adiposity measures, but in our meta-regression only percentage body fat can be used as a reliable, comparable marker of change of adiposity. Thus, the analyses presented in this paper were conducted using data from 39 studies. All of the included studies were considered to be of moderate to high quality according to the HTA quality assessment tool.30 Despite there being a positive relationship between mean change in percentage body fat and mean change in BMI-SDS, our modelling suggested that, in order to be confident of effecting a mean loss in percentage body fat, any future study should aim to reduce the BMI-SDS by at least 0.6.
Strengths and limitations
We believe that this is the first paper to attempt to bring together all studies that have reported both a change in BMI-SDS and changes in a marker of adiposity in the paediatric population with obesity. The systematic methods employed to identify the included studies were stringent, but it is possible that some relevant studies might have been missed. In addition, there was some variation in the reporting of results where there were multiple publications of the same study; in these cases, the results from the most comprehensive paper have been used. An important limitation to address in the broader context going forward is whether BMI-SDS is the best way to represent changes in BMI at extremes of body weight. The US Center for Disease Control cautioned the use of BMI-SDS in weight extremes in 2009.40 Freedman et al have suggested that there are better measures of adiposity in severe obesity, such as percentage of 95th percentile BMI (%BMIp95) or distance in kg/m2 from the 95th percentile (ΔBMIp95).41 Other groups have identified alternate methods when dealing with extremes of obesity such as BMI%42 or percentage above IOTF-25.43 Vanderwell et al have also suggested that BMI-SDS is only a weak to moderate predictor of percentage body fat in children, especially under 9 years of age.44 Notwithstanding these cautions, we based this analysis on the data available to us which was almost entirely reported in terms of BMI-SDS and continues to be the case in most recent publications to date.
It has been suggested that the relationship between change in percentage body fat and change in BMI-SDS may differ between very young and older children.45 Our inclusion criteria stipulated ages from 4 to 19 years. Most of the studies spanned a wide range of ages (table 1) and we did not have access to individual child data to facilitate stratification by age. Data from four subsets of children up to 10 years,37 46 47 however, did not suggest a different relationship from the whole cohort (see online supplementary appendix 3).
bmjopen-2018-028231supp003.docx (16.7KB, docx)
Agreements and disagreements with other research
Previous research has shown that an improvement in body composition and cardiometabolic risk can be achieved with a BMI-SDS reduction of ≥0.25 in adolescents with obesity, with greater benefits achieved when losing at least 0.5 BMI-SDS.48
In clinical practice, the degree of weight loss with lifestyle intervention is moderate and the success rate 2 years after onset of an intervention is low (<20% with a decrease in BMI-SDS <0.25).49 There have been numerous reports of lifestyle-based weight management interventions for children with obesity, many documenting changes in BMI-SDS, but a recent meta-analysis has documented that while such changes may be statistically significant, they are unlikely to lead to clinical improvements in metabolic health.50 51 To our knowledge, this is the first paper to establish the minimum change in BMI-SDS required to be certain of improving adiposity as percentage body fat for children and adolescents with obesity in clinical trials.
Clinical implications
If reducing fat mass is the aim of weight management interventions, our analysis in this review demonstrates that BMI-SDS changes must be of an order seldom achieved in trials worldwide. From our model, to be confident about ensuring an improvement in mean body fat, one should aim to reduce mean BMI-SDS by at least 0.6. Figure 3 and sensitivity analysis 5B (figure 5) suggest that to reduce body fat by 5% requires a much larger BMI-SDS reduction, of the order of 1.3–1.5, although there was a paucity of data in this region.
Recommendations for future research
While we are undertaking further analyses looking at key cardiovascular and metabolic outcomes in childhood obesity that may demonstrate improvements at lesser levels of BMI-SDS reduction, the evidence suggests that very few childhood weight management trials to date are likely to have improved percentage body fat and calls in to question their overall efficacy in terms of health improvement. That said, any trial demonstrating an improvement of the magnitude of 0.6 BMI-SDS might be termed successful with a likely reduction in fat mass. However, given the mounting evidence that BMI-SDS may not accurately reflect adiposity at extremes of obesity, it seems prudent for future trials to report additional indices of derived BMI values which may better reflect changes in actual adiposity. Which of the many measures suggested eventually establishes itself as the ‘optimal’ determinant at extremes of body mass is yet to be determined?
Conclusions
Using our model, to predict any fat mass improvement when reporting a weight management trial outcome requires a BMI-SDS decrease of 0.6. When evaluating key outcomes for future weight management trials and services, this figure needs to be borne in mind by researchers, healthcare professionals and commissioners when assessing apparent success.
Supplementary Material
Footnotes
Contributors: LB and RP provided substantial contributions to the conception and design of the study, designed the data extraction instrument, performed electronic database searches, data screening, extraction and quality assessment, coordinated and supervised data collection and drafted and revised the manuscript. JPHS provided a substantial contribution to the conception and design of the study, conducted data screening and interpretation and assisted with drafting and revision of the manuscript. LPH provided statistical expertise in relation to study design and conducted the data analyses and contributed to the drafting and revision of the manuscript. RM, AC and RB were involved in data acquisition and management. All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Funding: This study was supported by the NIHR Biomedical Research Centre at the University Hospitals Bristol NHS Foundation Trust and the University of Bristol.
Competing interests: JPHS and LPH are authors on two studies included in the systematic review that this paper reports on.
Provenance and peer review: Not commissioned; externally peer reviewed.
Data sharing statement: Dataset will be available from the Dryad repository (not yet set up so DOI currently unavailable).
Patient consent for publication: Not required.
References
- 1. Organization, WHO. Report of the commission on ending childhood obesity: World Health Organization, 2016. [Google Scholar]
- 2. National Child Measurement Programme - England, 2017-18 [National Statistics] National Child Measurement Programme data source: Health and Social Care Information Centre; 2018. Content.digital.nhs.uk. (n.d.). National Child Measurement Programme - NHS Digital. [online}. Available at http://content.digital.nhs.uk/ncmp.
- 3. National Child Measurement Programme - England, 2016-17 [National Statistics] National Child Measurement Programme data source: Health and Social Care Information Centre; 2018. Content.digital.nhs.uk. (n.d.). National Child Measurement Programme - NHS Digital. [online}. Available at http://content.digital.nhs.uk/ncmp.
- 4. Simmonds M, Llewellyn A, Owen CG, et al. . Predicting adult obesity from childhood obesity: a systematic review and meta-analysis. Obesity Reviews 2016;17:95–107. 10.1111/obr.12334 [DOI] [PubMed] [Google Scholar]
- 5. Hannon TS, Rao G, Arslanian SA. Childhood Obesity and Type 2 Diabetes Mellitus. Pediatrics 2005;116:473–80. 10.1542/peds.2004-2536 [DOI] [PubMed] [Google Scholar]
- 6. Haines L, Wan KC, Lynn R, et al. . Rising Incidence of Type 2 Diabetes in Children in the U.K. Diabetes Care 2007;30:1097–101. 10.2337/dc06-1813 [DOI] [PubMed] [Google Scholar]
- 7. Urbina EM, Kimball TR, McCoy CE, et al. . Youth With Obesity and Obesity-Related Type 2 Diabetes Mellitus Demonstrate Abnormalities in Carotid Structure and Function. Circulation 2009;119:2913–9. 10.1161/CIRCULATIONAHA.108.830380 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Pires A, Martins P, Pereira AP, et al. . Insulin Resistance, Dyslipidemia and Cardiovascular Changes in a Group of Obese Children. Arquivos Brasileiros de Cardiologia 2015;104:266–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Cook S, Kavey REW. Dyslipidemia and Pediatric Obesity. Pediatr Clin North Am 2011;58:1363–73. 10.1016/j.pcl.2011.09.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Chiarelli F, Marcovecchio ML. Insulin resistance and obesity in childhood. Eur J Endocrinol 2008;159(suppl_1):S67–74. 10.1530/EJE-08-0245 [DOI] [PubMed] [Google Scholar]
- 11. Tounian P, Aggoun Y, Dubern B, et al. . Presence of increased stiffness of the common carotid artery and endothelial dysfunction in severely obese children: a prospective study. The Lancet 2001;358:1400–4. 10.1016/S0140-6736(01)06525-4 [DOI] [PubMed] [Google Scholar]
- 12. Klein S, Burke LE, Bray GA, et al. . Clinical Implications of Obesity with Specific Focus on Cardiovascular Disease. A Statement for Professionals from the American Heart Association Council on Nutrition, Physical Activity, and Metabolism: Endorsed by the American College of Cardiology Foundation. Circulation 2004;110:2952–67. [DOI] [PubMed] [Google Scholar]
- 13. Krebs JD, Evans S, Cooney L, et al. . Changes in risk factors for cardiovascular disease with body fat loss in obese women. Diabetes, Obesity and Metabolism 2002;4:379–87. 10.1046/j.1463-1326.2002.00231.x [DOI] [PubMed] [Google Scholar]
- 14. Goldstein DJ. Beneficial health effects of modest weight loss. Int J Obes Relat Metab Disord 1992;16:397–415. [PubMed] [Google Scholar]
- 15. Colditz GA, Willett WC, Rotnitzky A, et al. . Weight gain as a risk factor for clinical diabetes mellitus in women. Ann Intern Med 1995;122:481–6. 10.7326/0003-4819-122-7-199504010-00001 [DOI] [PubMed] [Google Scholar]
- 16. National Heart, Lung, and Blood Institute. Clinical guidelines on the identification, evaluation, and treatment of overweight and obesity in adults. Bethesda, MD: National Heart, Lung, and Blood Institute, 1998. [Google Scholar]
- 17. Cole TJ. The LMS method for constructing normalized growth standards. Eur J Clin Nutr 1990;44:45–60. [PubMed] [Google Scholar]
- 18. Ho M, Garnett SP, Baur L, et al. . Effectiveness of Lifestyle Interventions in Child Obesity: Systematic Review With Meta-analysis. Pediatrics 2012;130:e1647–71. 10.1542/peds.2012-1176 [DOI] [PubMed] [Google Scholar]
- 19. Colquitt JL, Loveman E, O’Malley C, et al. . Diet, physical activity, and behavioural interventions for the treatment of overweight or obesity in preschool children up to the age of 6 years. Cochrane Database Syst Rev 2016;3:Cd012105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Oude Luttikhuis H, Baur L, Jansen H, et al. . Cochrane review: Interventions for treating obesity in children. Evidence-Based Child Health: A Cochrane Review Journal 2009;4:1571–729. 10.1002/ebch.462 [DOI] [Google Scholar]
- 21. National Institute for Health and Care Excellence. Quality Standard [QS94]. Obesity in children and young people: prevention and lifestyle weight management programmes. NICE 2015. https://www.nice.org.uk/guidance/qs94. [Google Scholar]
- 22. Friedemann C, Heneghan C, Mahtani K, et al. . Cardiovascular disease risk in healthy children and its association with body mass index: systematic review and meta-analysis. BMJ 2012;345:e4759 10.1136/bmj.e4759 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Prentice AM, Jebb SA. Beyond body mass index. Obesity Reviews 2001;2:141–7. 10.1046/j.1467-789x.2001.00031.x [DOI] [PubMed] [Google Scholar]
- 24. Cole TJ, Freeman JV, Preece MA. Body mass index reference curves for the UK, 1990. Arch Dis Child 1995;73:25–9. 10.1136/adc.73.1.25 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Kuczmarski RJ, Ogden CL, Grummer-Strawn LM, et al. . CDC growth charts: United States. Advance Data 2000;314:1–27. [PubMed] [Google Scholar]
- 26. Cole TJ, Bellizzi MC, Flegal KM, Dietz WH. Establishing a standard definition for child overweight and obesity worldwide: international survey. BMJ 2000;320:1240 10.1136/bmj.320.7244.1240 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. de Onis M, Garza C, Onyango AW, et al. . Comparison of the WHO Child Growth Standards and the CDC 2000 Growth Charts. J Nutr 2007;137:144–8. 10.1093/jn/137.1.144 [DOI] [PubMed] [Google Scholar]
- 28. de Onis M, Onyango AW, Borghi E, et al. . Development of a WHO growth reference for school-aged children and adolescents. Bull World Health Organ 2007;85:660–7. 10.2471/BLT.07.043497 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Higgins J, Green S. Cochrane handbook for systematic reviews of interventions. Chichester: Wiley, 2011. [Google Scholar]
- 30. Avenell A, Broom J, Brown T, et al. . Systematic review of the long-term effects and economic consequences of treatments for obesity and implications for health improvement. Health Technol Assess 2004;8(21). iii-iv:1-182 10.3310/hta8210 [DOI] [PubMed] [Google Scholar]
- 31. Harbord RM, Higgins JPT. Meta-Regression in Stata. Stata J 2008;8:493–519. 10.1177/1536867X0800800403 [DOI] [Google Scholar]
- 32. Racil G, Ben Ounis O, Hammouda O, et al. . Effects of high vs. moderate exercise intensity during interval training on lipids and adiponectin levels in obese young females. Eur J Appl Physiol 2013;113:2531–40. 10.1007/s00421-013-2689-5 [DOI] [PubMed] [Google Scholar]
- 33. Racil G, Coquart J, Elmontassar W, et al. . Greater effects of high- compared with moderate-intensity interval training on cardio-metabolic variables, blood leptin concentration and ratings of perceived exertion in obese adolescent females. Biol Sport 2016;33:145–52. 10.5604/20831862.1198633 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Seabra A, Katzmarzyk P, Carvalho MJ, et al. . Effects of 6-month soccer and traditional physical activity programmes on body composition, cardiometabolic risk factors, inflammatory, oxidative stress markers and cardiorespiratory fitness in obese boys. J Sports Sci 2016;34:1822–9. 10.1080/02640414.2016.1140219 [DOI] [PubMed] [Google Scholar]
- 35. Tanner JM. Growth at adolescence: with a general consideration of the effects of hereditary and environmental factors upon growth and maturation from birth to maturity. 2nd ed Oxford, England: Blackwell Scientific, 1962. [Google Scholar]
- 36. Bruyndonckx L, Hoymans VY, De Guchtenaere A, et al. . Diet, Exercise, and Endothelial Function in Obese Adolescents. Pediatrics 2015;135:e653–e661. 10.1542/peds.2014-1577 [DOI] [PubMed] [Google Scholar]
- 37. Gajewska J, Kuryłowicz A, Mierzejewska E, et al. . Complementary Effects of Genetic Variations in LEPR on Body Composition and Soluble Leptin Receptor Concentration after 3-Month Lifestyle Intervention in Prepubertal Obese Children. Nutrients 2016;8(6):pii: E328 10.3390/nu8060328 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Reinehr T, Roth C, Menke T, et al. . Adiponectin before and after weight loss in obese children. J Clin Endocrinol Metab 2004;89:3790–4. 10.1210/jc.2003-031925 [DOI] [PubMed] [Google Scholar]
- 39. Wan X, Wang W, Liu J, et al. . Estimating the sample mean and standard deviation from the sample size, median, range and/or interquartile range. BMC Med Res Methodol 2014;14:135 10.1186/1471-2288-14-135 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Flegal KM, Wei R, Ogden CL, et al. . Characterizing extreme values of body mass index–for-age by using the 2000 Centers for Disease Control and Prevention growth charts. Am J Clin Nutr 2009;90:1314–20. 10.3945/ajcn.2009.28335 [DOI] [PubMed] [Google Scholar]
- 41. Freedman DS, Butte NF, Taveras EM, et al. . BMI z-Scores are a poor indicator of adiposity among 2- to 19-year-olds with very high BMIs, NHANES 1999-2000 to 2013-2014. Obesity 2017;25:739–46. 10.1002/oby.21782 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Cole TJ, Faith MS, Pietrobelli A, et al. . What is the best measure of adiposity change in growing children: BMI, BMI %, BMI z-score or BMI centile? Eur J Clin Nutr 2005;59:419–25. 10.1038/sj.ejcn.1602090 [DOI] [PubMed] [Google Scholar]
- 43. Júlíusson PB, Roelants M, Benestad B, et al. . Severe obesity is a limitation for the use of body mass index standard deviation scores in children and adolescents. Acta Paediatr 2018;107:307–14. 10.1111/apa.14113 [DOI] [PubMed] [Google Scholar]
- 44. Vanderwall C, Randall Clark R, Eickhoff J, et al. . BMI is a poor predictor of adiposity in young overweight and obese children. BMC Pediatr 2017;17:135 10.1186/s12887-017-0891-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Woo JG, Cole TJ. Assessing adiposity using BMI z -Score in children with severe obesity. Obesity 2017;25:662 10.1002/oby.21800 [DOI] [PubMed] [Google Scholar]
- 46. Dobe M, Geisler A, Hoffmann D, et al. . The Obeldicks concept. An example for a successful outpatient lifestyle intervention for overweight or obese children and adolescents]. Bundesgesundheitsblatt, Gesundheitsforschung, Gesundheitsschutz 2011;54:628–35. [DOI] [PubMed] [Google Scholar]
- 47. Kirk S, Zeller M, Claytor R, et al. . The Relationship of Health Outcomes to Improvement in BMI in Children and Adolescents. Obes Res 2005;13:876–82. 10.1038/oby.2005.101 [DOI] [PubMed] [Google Scholar]
- 48. Ford AL, Hunt LP, Cooper A, et al. . What reduction in BMI SDS is required in obese adolescents to improve body composition and cardiometabolic health? Arch Dis Child 2010;95:256–61. 10.1136/adc.2009.165340 [DOI] [PubMed] [Google Scholar]
- 49. Reinehr T. Lifestyle intervention in childhood obesity: changes and challenges. Nat Rev Endocrinol 2013;9:607–14. 10.1038/nrendo.2013.149 [DOI] [PubMed] [Google Scholar]
- 50. Al-Khudairy L, Loveman E, Colquitt JL, et al. . Diet, physical activity and behavioural interventions for the treatment of overweight or obese adolescents aged 12 to 17 years. Cochrane Database Syst Rev 2017;6:CD012691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Mead E, Brown T, Rees K, et al. . Diet, physical activity and behavioural interventions for the treatment of overweight or obese children from the age of 6 to 11 years. Cochrane Database Syst Rev 2017;6:CD012651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Bell LM, Watts K, Siafarikas A, et al. . Exercise alone reduces insulin resistance in obese children independently of changes in body composition. J Clin Endocrinol Metab 2007;92:4230–5. 10.1210/jc.2007-0779 [DOI] [PubMed] [Google Scholar]
- 53. Bock DE, Robinson T, Seabrook JA, et al. . The Health Initiative Program for Kids (HIP Kids): effects of a 1-year multidisciplinary lifestyle intervention on adiposity and quality of life in obese children and adolescents--a longitudinal pilot intervention study. BMC Pediatr 2014;14:296 10.1186/s12887-014-0296-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Bustos P, Orias J, Sáez K, et al. . Effects of the Bright Bodies Program in Chilean obese children. Revista Medica De Chile 2015;143:1136–43. [DOI] [PubMed] [Google Scholar]
- 55. Calcaterra V, Larizza D, Codrons E, et al. . Improved metabolic and cardiorespiratory fitness during a recreational training program in obese children. J Pediatr Endocrinol Metab 2013;26(3-4):271–6. 10.1515/jpem-2012-0157 [DOI] [PubMed] [Google Scholar]
- 56. Farpour-Lambert NJ, Aggoun Y, Marchand LM, et al. . Physical activity reduces systemic blood pressure and improves early markers of atherosclerosis in pre-pubertal obese children. J Am Coll Cardiol 2009;54:2396–406. 10.1016/j.jacc.2009.08.030 [DOI] [PubMed] [Google Scholar]
- 57. Ford AL, Bergh C, Södersten P, et al. . Treatment of childhood obesity by retraining eating behaviour: randomised controlled trial. BMJ 2009;340:b5388 10.1136/bmj.b5388 [DOI] [PubMed] [Google Scholar]
- 58. Garanty-Bogacka B, Syrenicz M, Goral J, et al. . Changes in inflammatory biomarkers after successful lifestyle intervention in obese children. Endokrynologia Polska 2011;62:499–505. [PubMed] [Google Scholar]
- 59. Grønbæk H, Lange A, Birkebæk NH, et al. . Effect of a 10-week weight loss camp on fatty liver disease and insulin sensitivity in obese Danish children. J Pediatr Gastroenterol Nutr 2012;54:223–8. 10.1097/MPG.0b013e31822cdedf [DOI] [PubMed] [Google Scholar]
- 60. Kazankov K, Møller HJ, Lange A, et al. . The macrophage activation marker sCD163 is associated with changes in NAFLD and metabolic profile during lifestyle intervention in obese children. Pediatr Obes 2015;10:226–33. 10.1111/ijpo.252 [DOI] [PubMed] [Google Scholar]
- 61. Hvidt KN, Olsen MH, Ibsen H, et al. . Effect of changes in BMI and waist circumference on ambulatory blood pressure in obese children and adolescents. J Hypertens 2014;32:1470–7. 10.1097/HJH.0000000000000188 [DOI] [PubMed] [Google Scholar]
- 62. Klijn PHC, van der Baan-Slootweg OH, van Stel HF. Aerobic exercise in adolescents with obesity: preliminary evaluation of a modular training program and the modified shuttle test. BMC Pediatr 2007;7(1). 10.1186/1471-2431-7-19 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Lazzer S, Molin M, Stramare D, et al. . Effects of an eight-month weight-control program on body composition and lipid oxidation rate during exercise in obese children. J Endocrinol Invest 2008;31:509–14. 10.1007/BF03346399 [DOI] [PubMed] [Google Scholar]
- 64. Meyer AA, Kundt G, Lenschow U, et al. . Improvement of early vascular changes and cardiovascular risk factors in obese children after a six-month exercise program. J Am Coll Cardiol 2006;48:1865–70. 10.1016/j.jacc.2006.07.035 [DOI] [PubMed] [Google Scholar]
- 65. Miraglia F, de Moraes Silveira CR, Gomes Beghetto M, et al. . Behavior of adipokines after a year follow-up in the Obesity Outpatient Clinic for Children and Adolescents. Nutricion Hospitalaria 2015;32:1554–9. [DOI] [PubMed] [Google Scholar]
- 66. Morell-Azanza L, García-Calzón S, Rendo-Urteaga T, et al. . Serum oxidized low-density lipoprotein levels are related to cardiometabolic risk and decreased after a weight loss treatment in obese children and adolescents. Pediatr Diabetes 2017;18:392–8. 10.1111/pedi.12405 [DOI] [PubMed] [Google Scholar]
- 67. Rendo-Urteaga T, García-Calzón S, González-Muniesa P, et al. . Peripheral blood mononuclear cell gene expression profile in obese boys who followed a moderate energy-restricted diet: differences between high and low responders at baseline and after the intervention. Br J Nutr 2015;113:331–42. 10.1017/S0007114514003584 [DOI] [PubMed] [Google Scholar]
- 68. Murer SB, Knöpfli BH, Aeberli I, et al. . Baseline leptin and leptin reduction predict improvements in metabolic variables and long-term fat loss in obese children and adolescents: a prospective study of an inpatient weight-loss program. Am J Clin Nutr 2011;93:695–702. 10.3945/ajcn.110.002212 [DOI] [PubMed] [Google Scholar]
- 69. Aeberli I, Jung A, Murer SB, et al. . During Rapid Weight Loss in Obese Children, Reductions in TSH Predict Improvements in Insulin Sensitivity Independent of Changes in Body Weight or Fat. The Journal of Clinical Endocrinology & Metabolism 2010;95:5412–8. 10.1210/jc.2010-1169 [DOI] [PubMed] [Google Scholar]
- 70. Murdolo G, Tortoioli C, Celi F, et al. . Fetuin-A, adiposity-linked insulin resistance and responsiveness to an educational-based weight excess reduction program: a population-based survey in prepubertal schoolchildren. Endocrine 2017;56:357–65. 10.1007/s12020-016-1009-3 [DOI] [PubMed] [Google Scholar]
- 71. Ning Y, Yang S, Evans RK, et al. . Changes in body anthropometry and composition in obese adolescents in a lifestyle intervention program. Eur J Nutr 2014;53:1093–102. 10.1007/s00394-013-0612-9 [DOI] [PubMed] [Google Scholar]
- 72. Bean MK, Mazzeo SE, Stern M, et al. . Six-Month Dietary Changes in Ethnically Diverse, Obese Adolescents Participating in a Multidisciplinary Weight Management Program. Clin Pediatr 2011;50:408–16. 10.1177/0009922810393497 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. Pacifico L, Arca M, Anania C, et al. . Arterial function and structure after a 1-year lifestyle intervention in children with nonalcoholic fatty liver disease. Nutr Metab Cardiovasc Dis 2013;23:1010–6. 10.1016/j.numecd.2012.08.003 [DOI] [PubMed] [Google Scholar]
- 74. Reinehr T, Stoffel-Wagner B, Roth CL. Retinol-binding protein 4 and its relation to insulin resistance in obese children before and after weight loss. J Clin Endocrinol Metab 2008;93:2287–93. 10.1210/jc.2007-2745 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Reinehr T, Roth CL. Fetuin-A and its relation to metabolic syndrome and fatty liver disease in obese children before and after weight loss. J Clin Endocrinol Metab 2008;93:4479–85. 10.1210/jc.2008-1505 [DOI] [PubMed] [Google Scholar]
- 76. Rohrer TR, Rizzo VF, Cäsar JJ, et al. . Changes in hepatic risk factors, metabolic variables, body composition, and physical fitness in obese children after a one-year weight loss program. J Pediatr Endocrinol Metab 2008;21:837–45. 10.1515/JPEM.2008.21.9.837 [DOI] [PubMed] [Google Scholar]
- 77. Rolland-Cachera MF, Thibault H, Souberbielle JC, et al. . Massive obesity in adolescents: dietary interventions and behaviours associated with weight regain at 2 y follow-up. Int J Obes 2004;28:514–9. 10.1038/sj.ijo.0802605 [DOI] [PubMed] [Google Scholar]
- 78. Roth CL, Elfers C, Lass N, et al. . Betatrophin: no relation to glucose metabolism or weight status in obese children before and after lifestyle intervention. Pediatr Diabetes 2017;18:485–91. 10.1111/pedi.12412 [DOI] [PubMed] [Google Scholar]
- 79. Savoye M, Berry D, Dziura J, et al. . Anthropometric and psychosocial changes in obese adolescents enrolled in a Weight Management Program. J Am Diet Assoc 2005;105:364–70. 10.1016/j.jada.2004.12.009 [DOI] [PubMed] [Google Scholar]
- 80. Savoye M, Nowicka P, Shaw M, et al. . Long-term Results of an Obesity Program in an Ethnically Diverse Pediatric Population. Pediatrics 2011;127:402–10. 10.1542/peds.2010-0697 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81. Savoye M, Shaw M, Dziura J, et al. . Effects of a Weight Management Program on Body Composition and Metabolic Parameters in Overweight Children. JAMA 2007;297:2697–704. 10.1001/jama.297.24.2697 [DOI] [PubMed] [Google Scholar]
- 82. Savoye M, Caprio S, Dziura J, et al. . Reversal of early abnormalities in glucose metabolism in obese youth: results of an intensive lifestyle randomized controlled trial. Diabetes Care 2014;37:317–24. 10.2337/dc13-1571 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83. Schiel R, Kaps A, Stein G, et al. . Identification of Predictors for Weight Reduction in Children and Adolescents with Overweight and Obesity (IDA-Insel Survey). Health Care 2016;4:07 10.3390/healthcare4010005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84. Truby H, Baxter K, Ware RS, et al. . A Randomized Controlled Trial of Two Different Macronutrient Profiles on Weight, Body Composition and Metabolic Parameters in Obese Adolescents Seeking Weight Loss. PLoS One 2016;11:e0151787 10.1371/journal.pone.0151787 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85. van der Baan-Slootweg O, Benninga MA, Beelen A, et al. . Inpatient Treatment of Children and Adolescents With Severe Obesity in the Netherlands. JAMA Pediatr 2014;168:807–14. 10.1001/jamapediatrics.2014.521 [DOI] [PubMed] [Google Scholar]
- 86. Visuthranukul C, Sirimongkol P, Prachansuwan A, et al. . Low-glycemic index diet may improve insulin sensitivity in obese children. Pediatr Res 2015;78:567–73. 10.1038/pr.2015.142 [DOI] [PubMed] [Google Scholar]
- 87. Vitola BE, Deivanayagam S, Stein RI, et al. . Weight loss reduces liver fat and improves hepatic and skeletal muscle insulin sensitivity in obese adolescents. Obesity 2009;17:1744–8. 10.1038/oby.2009.171 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88. Wickham EP, Stern M, Evans RK, et al. . Prevalence of the Metabolic Syndrome Among Obese Adolescents Enrolled in a Multidisciplinary Weight Management Program: Clinical Correlates and Response to Treatment. Metab Syndr Relat Disord 2009;7:179–86. 10.1089/met.2008.0038 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89. Evans RK, Franco RL, Stern M, et al. . Evaluation of a 6-month multi-disciplinary healthy weight management program targeting urban, overweight adolescents: effects on physical fitness, physical activity, and blood lipid profiles. Int J Pediatr Obes 2009;4:130–3. 10.1080/17477160802314997 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
bmjopen-2018-028231supp001.pdf (11.6KB, pdf)
bmjopen-2018-028231supp002.pdf (231.9KB, pdf)
bmjopen-2018-028231supp003.docx (16.7KB, docx)