Abstract
The role of magnetic resonance (MR) imaging inpatients withpancreatic endocrine tumors such as Zollinger-Ellison syndrome (ZES) is controversial. In the present study we have examined the ability of current MR imaging compared with other imaging modalities, to localize gastrinomas in 43 patients with ZES. All results were subsequently assessed at exploratory laparotomy (N = 34) or by liver biopsy (N = 9). For the 18 patients with metastatic gastrinoma in the liver, MR imaging had a sensitivity of 83%, ultrasound 50%, CT 56%, and angiography 61%. The combination of MR imaging, ultrasound, and CT were the same as MR imaging alone. For MR imaging, both T1 and STIR sequences had equal sensitivity, although tumors were more easily seen with STIR sequences. Specificity of MR imaging was slightly lower (88%) than the other modalities (96–100%) because MR imaging incorrectly identified small hemangiomas as possible tumors in four patients. MR imaging was better than CT in identifying metastatic lesions in the liver. For the localization of primary gastrinoma, assessed in 32 patients, MR imaging had a sensitivity of 25%, ultrasound 19%, CT 28%, all three together 38%, and angiography 59%. Localization of metastatic gastrinoma in the liver or primary gastrinomas in 16 patients was assessed before and after gadolinium-DTPA (0.1 mmol/kg). The sensitivity and specificity of MR imaging was unchanged but bolus injection and rapid MR acquisition techniques were not used. These results indicate that recent advances in MR imaging have greatly improved its sensitivity for the detection and assessment of the extent of metastatic gastrinoma. MR imaging is now the imaging study of choice to assess metastatic pancreatic endocrine tumors in the liver. In contrast, the detection of primary tumors by MR imaging has not improved; therefore, angiography remains the study of choice.
Keywords: gastrinoma, islet cell tumor, tumor localization
Patients with functional pancreatic endocrine tumors have two problems: the control of the symptoms due to hormone overproduction and the malignant nature of the tumor (1, 2). With the increased ability to control the effects of hormone overproduction medically, the malignant potential of the tumor is becoming the most important determinant of long-term survival (2, 3). Therefore, tumor localization and assessment of the extent of the tumor is becoming increasingly important, not only to localize tumors for possible surgical resection, but also to establish metastatic disease to prevent unnecessary surgery (3, 4). In addition, because the five-year survival rate of patients with advanced metastatic disease is only 20%, it is becoming increasingly important to have sensitive imaging methods to assess the results of various treatments on metastatic disease (3, 5–7). Lastly, with many pancreatic endocrine tumors such as Zollinger-Ellison syndrome, approximately 50% of the patients cured will develop recurrent disease within five years. Because these tumors are slow growing, these patients have an excellent long-term survival; however, repeated imaging studies over a number of years are usually performed to assess tumor growth (3, 5–7); thus sensitive, noninvasive imaging studies involving minimal or no radiation, such as ultrasound or magnetic resonance (MR) imaging would be preferable.
Ultrasound in most studies (3, 4, 8, 9) has not been sufficiently sensitive to be generally useful. MR imaging has been reported by a number of groups to be useful in localizing hypovascular or nonneuroendocrine abdominal tumors, especially those metastatic to the liver (10, 11). However, we previously reported (12) from the years 1985 to 1987 that MR imaging had a sensitivity similar to ultrasound of (43%) for detecting tumor metastatic to the liver and only 20% for detecting primary tumors. In contrast, computerized tomography (CT) had a sensitivity of 71% for hepatic metastases and 45% for primary tumors (12). In that study (12) angiography had the highest sensitivity for detecting both primary tumors (80%) and hepatic metastases (71%). Because angiography (ANGIO) is an invasive procedure, its usefulness is limited mainly to the initial, preoperative evaluation. In our previous study (12) it was concluded that MR imaging, because of its low sensitivity, should not be routinely used in patients with islet cell tumors such as gastrinoma.
Recent advances in MR imaging technology, such as improved hardware and coil design, motion suppression techniques, short inversion recovery (STIR) sequences, and paramagnetic contrast agents (13, 14), may have increased the potential usefulness of MR imaging in patients with pancreatic endocrine tumors. In the present study we have studied prospectively the ability of current MR imaging to localize either primary or metastatic tumor in patients with gastrinomas, the most common malignant pancreatic endocrine tumor (13). We have also compared these results to those of ultrasound, CT, and ANGIO for the detection of both metastatic tumor in the liver and the primary gastrinomas. We also studied whether gadolinium-DTPA, a paramagnetic contrast agent, further increases the sensitivity for detecting either the primary or metastatic pancreatic endocrine tumors as suggested by a study reported in abstract form (15).
MATERIALS AND METHODS
Forty-three patients with Zollinger-Ellison syndrome (25 males, 18 females) all underwent gastrinoma tumor localization studies prior to either exploratory laparotomy (N = 34) or percutaneous liver biopsy (N = 9) (Table 1). None of the results in this study were from the previous study comparing MR imaging from the years 1985 to 1987 (12).
Table 1.
Patient Characteristics
| Patients (N) | 43 |
| Males | 25 |
| Females | 18 |
| Age (years) | |
| Mean ± SEM | 51 ± 2 |
| Range | 29–86 |
| Multiple endocrine neoplasia type 1 (N) | 0 |
| Disease duration (years) | |
| Mean ± SEM | 6.9 ± 1.1 |
| Range | 0.5–10.2 |
| Fasting serum gastrin (pg/ml) | |
| Mean ± SEM | 969 ± 162 |
| Range | 87–3862 |
| Histologically confirmed tumor location | |
| Primary tumor (N patients)* | 32 |
| Pancreas | 19† |
| Duodenum | 14 |
| Other | 3‡ |
| Hepatic metastases (N patients)§ | 18¶ |
Primary tumor location was determined by laparotomy. In two patients no primary tumor was found.
Four patients had two primary tumors with a tumor in the pancreas and duodenum and these are both included in tumor location.
One patient had a primary tumor in the left kidney; one patient had a primary tumor in a common bile duct lymph node; one patient had a primary tumor in the porta hepatitis.
Metastatic tumor in the liver was found at surgery in nine patients and by percutaneous liver biopsy in nine patients.
Seven patients are included in both the primary tumor and liver tumor groups.
Each patient included in the study met the criteria for Zollinger-Ellison syndrome (11, 12) as defined by having an elevated fasting serum gastrin (> 100 pg/ml), positive secretin provocative test (gastrin increase >200 pg/ml), and elevated gastric acid hypersecretion (3, 18, 19). Prior to surgery or biopsy, each patient underwent abdominal imaging studies (MR imaging, CT, ultrasound, and angiography). MR imaging, CT, and ANGIO studies were interpreted separately and in a blinded fashion by the same radiologist (J.L.D.) without knowledge of the findings at surgery or liver biopsy. Ultrasound studies were performed and reported by radiologists, most experienced with ultrasound abdominal imaging without knowledge of the results at surgery or biopsy.
MR imaging was performed on a 0.5 Tesla scanner (Picker International, Highland Heights, Ohio). All images were obtained in the transaxial plane with contiguous 10-mm sections. In each patient, two pulse sequences were obtained: a Tl-weighted spin-echo (SE) sequence with a repetition time (TR) of 400–600 msec and an echo time (TE) of 10 msec; and a short T1 inversion-recovery (STIR) pulse sequence, TR = 1500 msec, T1 = 100 msec, and TE = 30 msec. Gradient moment scaling was used to correct for respiratory movement in each patient. Gadolinium-DTPA (0.1 mmol/kg) intravenously 5 min prior to MR imaging was given in 30 patients.
Computerized tomography (CT) of the abdomen was performed as described previously (20) (CT-T 9800 Quick, GE Medical Systems, Milwaukee, Wisconsin). Contiguous scans of 10-mm thickness were obtained through the abdominal cavity from the dome of the liver to the pelvis. An oral contrast agent (diatrizoate sodium, Winthrop-Breon, Rensselaer, New York) was given 12 hr, 2 hr, and 15 min prior to CT. All patients were scanned initially without intravenous contrast. Liver scanning was then performed using a bolus enhanced dynamic mode with the injection of 150 ml of intravenous contrast agent (iopamidol 30%, Winthrop-Breon) at a rate of 1m1/sec. On follow-up CT scans either the unenhanced or the contrast enhanced study was performed, depending upon which one had optimally demonstrated liver pathology.
Selective abdominal angiography (ANGIO) was performed with selective injection of the splenic, superior mesenteric, gastroduodenal, and hepatic arteries in all patients as previously described (3, 21, 22). Abdominal ultrasound with a real-time mechanical sector unit (Diasonics, Milpitas, California) with a 5-mHz transducer was performed as described previously (8). An extensive surgical exploration was performed as described previously (5, 23). Endoscopic transillumination of the duodenum (24) and a routine duodenotomy (5) were performed as described previously. Intraoperative ultrasonography was performed to detect primary tumors and of the liver to detect metastatic tumors as described previously (25).
The presence of gastrinoma at surgery or by biopsy constituted the basis for the determination of sensitivity, specificity, and predictive value for each imaging modality. Each imaging modality was analyzed separately for its ability to detect hepatic gastrinoma and/or extrahepatic or primary gastrinoma because, as discussed previously (12), the clinical implications of tumor metastatic to the liver are different from those of primary gastrinoma or gastrinoma not metastatic to the liver (3). Sensitivity, specificity, and predictive values for the results of hepatic and extrahepatic imaging results were calculated as described previously (26). The findings at angiography and MR imaging were compared by means of the McNemar test with the Yates’ correction (27) to assess the significance of differences between the various imaging modalities.
RESULTS
The clinical characteristics of the patients studied are included in Table 1. Of the 34 patients undergoing laparotomy, primary gastrinoma was histologically proved in 32 patients, and two patients had no tumor found at surgery. Four patients had two different primary sites (duodenum and pancreas) (Table 1). Primary gastrinomas were found primarily in the pancreas and in the duodenum (Table 1).
In Table 2, the ability of MR imaging to detect hepatic metastases is compared to corresponding results for other imaging studies. Hepatic metastases were histologically confirmed in 18 patients. With MR imaging, T1 and STIR sequences were equally sensitive in detecting metastatic gastrinoma to the liver. Specifically, for both sequences, the metastatic tumor was correctly identified in 15/18 patients with a sensitivity of 83% (Tables 2 and 3). By comparison, ultrasound and CT detected metastases in 9/18 and 10/18 patients for a sensitivity of 50% and 56%, respectively. ANGIO was not significantly more sensitive than CT or ultrasound, being positive in 11/18 patients for a sensitivity of 61% for metastatic gastrinoma to the liver (Table 3). The combination of the noninvasive studies (MR imaging, CT, and ultrasound) detected metastatic disease to the liver in 15/18 patients for a sensitivity of 83% and thus was not significantly better than MR imaging alone (Tables 2 and 3). Surgery was not performed in the nine patients who underwent liver biopsy to establish the diagnosis of metastatic gastrinoma because the preoperative imaging studies were positive for metastatic disease. In this group of patients, the MRI was positive for metastatic disease in 8/9 (92%), CT in 6/9 (66%), US in 5/9 (48%), and ANGIO in 7/9 (74%). In 25 patients no hepatic metastases were found at surgery. In these patients, the Tl-weighted MR imaging revealed false positive results in 4/25 patients (16%) and STIR sequences revealed false positive results in 3/25 patients (12%) with the T1 or STIR being positive in 4/25 (16%) (Table 2). Both ultrasound and CT gave only one false positive (4%) result, while ANGIO had no false positive results (0%) (Table 2). MR imaging had a specificity of 88% for the detection of hepatic metastases (Table 3), which was not significantly different from the value of 95% with CT or ultrasound or 100% with ANGIO (Table 3). Tl-weighted and STIR sequences were equally specific (79% vs 88%) and sensitive (Table 3) for detecting hepatic metastases; however, the lesions were generally more easily seen on the STIR sequences. A typical result from a patient is shown in Figure 1. In this patient both the CT scan (top) and the Tl-weighted images (center) did not show the metastatic gastrinoma whereas the STIR sequence (bottom) clearly showed metastatic deposits in the right lobe of the liver. At surgery this patient had diffuse metastatic disease with deposits in the sites indicated in Figure 1.The increased sensitivity of MR imaging for detecting tumor in the liver not only allowed metastatic disease to be identified in a larger number of patients, but it also identified additional metastases (Figure 2). In Figure 2 is shown the MR imaging and CT in a patient thought to have an isolated hepatic metastasis by CT and thus considered for possible surgical resection. On MR imaging small metastatic deposits were seen in both the right and left hepatic lobes, excluding the possibility of surgical resection. Figure 3 shows a comparison of a CT and MR imaging in another patient. CT failed to unequivocally identify a lesion in the inferior aspect of the right lobe of the liver, but it is clearly identified by both T1 and STIR MR imaging and confirmed by liver biopsy. Retrospectively, however, it is apparent that a hypodense area on CT corresponds to the location of the metastases.
Table 2.
Results of Localization Studies for Gastrinoma Metastatic to Liver*
| Imaging results (N patients positive) |
|||||||
|---|---|---|---|---|---|---|---|
| Metastatic gastrinoma in liver (N patients) | MR imaging |
US | CT | MR imaging, CT, and US | ANGIO | ||
| T1 | STIR | Tl/STIR | |||||
| Present (18) | 15 | 15 | 15 | 9 | 10 | 15 | 11 |
| Absent (25) | 4 | 3 | 4 | 1 | 1 | 3 | 0 |
All patients with gastrinoma metastatic to the liver had this determined either at surgery (N = 9) or by percutaneous biopsy or laparoscopy (N = 9) and all patients (N = 25) without gastrinoma in the liver had this confirmed at surgery. Abbreviations: US = ultrasound; CT = computed tomography scan; ANGIO = selective abdominal angiography.
Table 3.
Sensitivity, Specificity and Predictive Value of Imaging Studies for Assessing Presence or Absence of Gastrinoma Metastatic to Liver*
| Index | MR imaging |
US | CT | MR imaging, CT, and US | ANGIO | ||
|---|---|---|---|---|---|---|---|
| T1 | STIR | T1/STIR | |||||
| Sensitivity (%) | 83 | 83 | 83 | 50 | 56 | 83 | 61 |
| Specificity (%) | 84 | 88 | 88 | 96 | 96 | 100 | 100 |
| Positive predictive value (%) | 78 | 83 | 83 | 90 | 91 | 100 | 84 |
| Negative predictive value (%) | 88 | 88 | 88 | 75 | 75 | 92 | 81 |
Sensitivity, specificity, positive and negative predictive values were calculated as described in Materials and Methods. Abbreviations: see Table 2.
Fig 1.
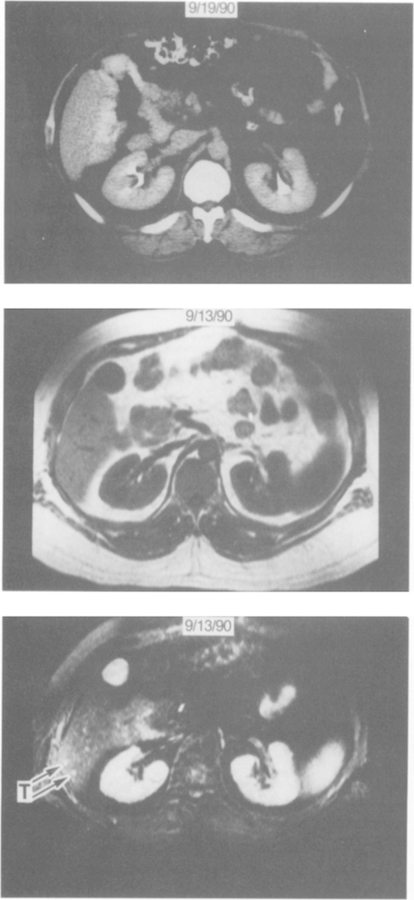
Comparison of the ability of CT and MR imaging to demonstrate hepatic metastases. CT scan (top panel) of the inferior right hepatic lobe on a bolus-enhanced dynamic scan shows no hepatic lesion. By comparison, the T1 sequence MR imaging (middle panel) shows multiple low-signal-intensity foci within the liver that cannot be distinguished from vessels. The STIR sequence MR imaging (bottom panel) shows multiple foci of high signal intensity (the largest two lesions identified by arrows). Exploratory laparotomy demonstrated multiple small (<1 cm) metastatic gastrinomas throughout both the right and left hepatic lobes.
Fig 2.
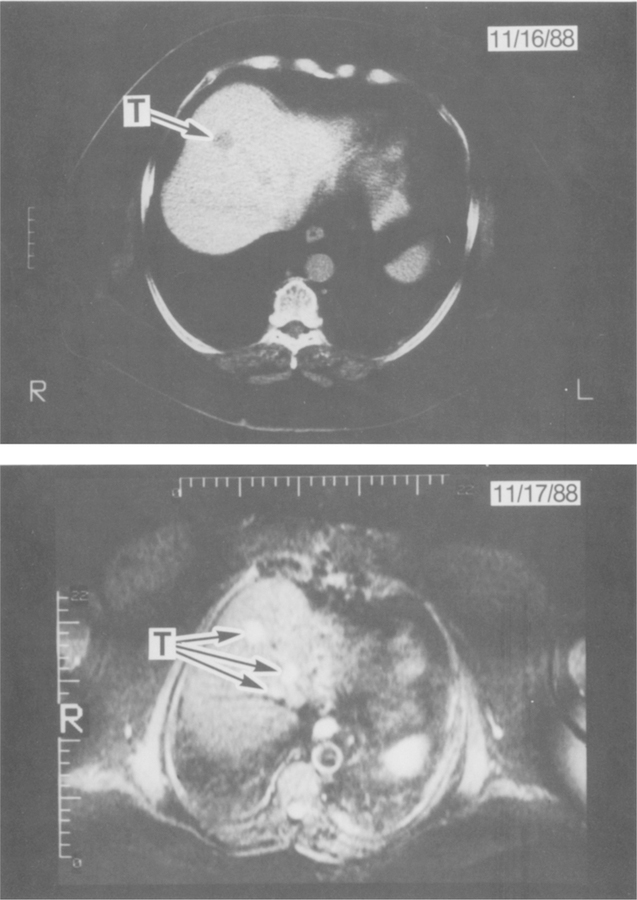
Comparison of CT and MR imaging in a patient with multiple hepatic metastases. The top panel shows a CT with one clearly distinguishable hypodense area (arrow) and one other questionable focus that cannot be distinguished from the hepatic vein. The lower panel is a STIR sequence MR image at the same level of the liver unequivocally showing three hyperintense lesions (T = tumor) confirmed by biopsy to be metastatic gastrinomas.
Fig 3.
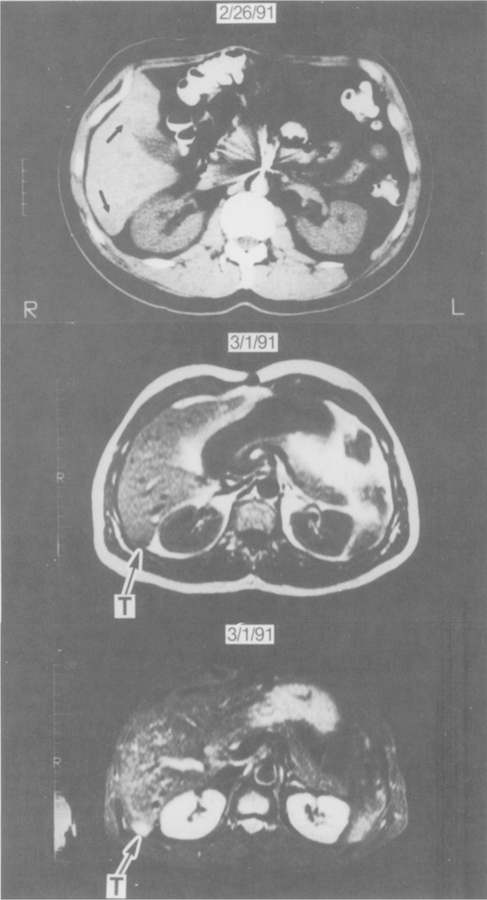
Comparison of CT and MR imaging. Bolus-enhanced dynamic CT (top panel) demonstrates suspicious low-density areas (arrows) in the posterior segment of the right lobe and the medial segment of the left lobe. STIR sequences (bottom panel) unequivocally demonstrate a small focus of high signal intensity compatible with metastatic gastrinoma (T). Biopsy of this lesion confirmed the lesion to be gastrinoma. On the T1-weighted image (middle panel) it was difficult to distinguish the tumor (T) from normal vasculature.
For identifying primary and extrahepatic gastrinomas, MR imaging was less sensitive than for identifying metastatic tumor to liver (Tables 4 and 5). Figure 4 shows the T1 and STIR sequences of a typical patient in whom MR imaging localized the primary gastrinoma. For extrahepatic gastrinomas, the combination of T1 and STIR sequences were equally sensitive with T1 weighted sequences detecting primary gastrinomas in 8/32 patients for a sensitivity of 25% (Tables 4 and 5) compared to STIR sequences, which were positive in 7/32 patients for a sensitivity of 22% (Tables 4 and 5). This was significantly less than the 83% sensitivity for either sequence to detect liver metastases (Table 3). Ultrasound detected primary tumors in 6/32 patients (19%) and CT was positive in 8/32 patients (25%). In contrast, ANGIO provided the greatest sensitivity for primary tumor detection, being positive in 19/32 patients for a sensitivity of 59% (P < 0.05). For localizing extrahepatic tumor, the combination of all the noninvasive tests (ultrasound, CT, and MR imaging) was not significantly better than any individual study alone and was less sensitive than ANGIO (P < 0.05) (Tables 4 and 5). False positive localizations of extrahepatic gastrinomas were detected in 1/9 patients (11%) by ultrasound and in 1/9 (11%) patients both by ANGIO and MR imaging. There were no false positive tumors detected by CT (Table 5). The specificities for each of the imaging modalities varied from 80 to 100% and were not significantly different because of the small number of patients in which extrahepatic tumor was not found.
Table 4.
Results of Localization Studies for Primary Gastrinoma*
| Imaging modality (N patients with positive results) |
|||||||
|---|---|---|---|---|---|---|---|
| MR imaging |
MR imaging CT, and US | ||||||
| Extrahepatic gastrinoma | T1 | STIR | Tl/STIR | US | CT | ANGLO | |
| Tumor found at surgery (N = 32) | 8 | 7 | 8 | 6 | 9 | 12 | 19 |
Two patients had no gastrinomas found. Abbreviations: see Table 2.
Table 5.
Sensitivity and Positive Predictive Value for Imaging Studies to Detect Primary Gastrinoma*
| MR imaging |
MR imaging, CT, and US | ||||||
|---|---|---|---|---|---|---|---|
| Index | T1 | STIR | Tl/STIR | US | CT | ANGIO | |
| Sensitivity (%) | 25 | 22 | 25 | 19 | 28 | 38 | 59 |
| Positive predictive value (%) | 89 | 88 | 89 | 75 | 100 | 85 | 92 |
Sensitivity and positive predictive values were calculated as described in Materials and Methods. Only two patients undergoing surgery had no tumor found at surgery; therefore, there were insufficient numbers of patients without tumors found to calculate reliably specificity or negative predictive value. Abbreviations: see Table 2.
Fig 4.
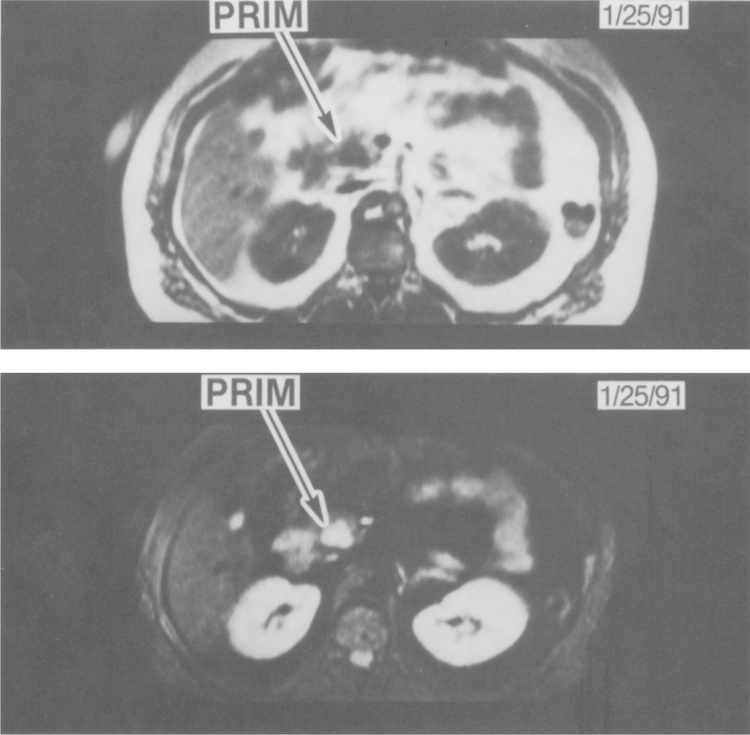
MR imaging of a primary gastrinoma in the head of the pancreas. The two panels show MR imaging at the same level, demonstrating a primary gastrinoma (PRIM) that was later confirmed at surgery. The top panel is a T1-weighted MR image showing a hypointense lesion in the head of the pancreas (PRIM). The bottom panel shows the corresponding STIR MR image, demonstrating the hyperintense tumor in the pancreatic head (PRIM).
Gadolinium-DTPA has been reported to enhance MR imaging of pancreatic endocrine tumors such as gastrinomas (15, 17). To evaluate the usefulness of this paramagnetic agent, MR imaging studies were performed with and without gadolinium-DTPA in 16 patients with histologically confirmed hepatic metastases and in 14 patients with surgically proven extrahepatic gastrinoma. Gadolinium-DTPA did not increase the sensitivity for the detection of either primary tumors or metastatic tumors (Table 6). Furthermore, gadolinium-DTPA did not increase the specificity or predictive values (Table 6). No change in the appearance of hepatic metastases or primary tumors was detected after gadolinium-DTPA.
Table 6.
Sensitivity, Specificity, and Predictive Value for Detection of Gastrinoma by MR Imaging with and Without Gadolinium-DPTA*
| T1 with gadoliniutn-DPTA (%) |
||
|---|---|---|
| Present | Absent | |
| Hepatic metastases (N = 16) | ||
| Sensitivity | 71 | 71 |
| Specificity | 78 | 78 |
| Positive predictive value | 71 | 71 |
| Negative predictive value | 78 | 78 |
| Primary tumors (N = 14)† | ||
| Sensitivity | 14 | 29 |
| Positive predictive value | 100 | 100 |
Sensitivity, specificity, and positive and negative predictive values were calculated as described in Materials and Methods.
Specificity and negative predictive values were not calculated because all patients had primary tumors found at surgery.
DISCUSSION
The role of MR imaging in patients with pancreatic endocrine tumors such as gastrinomas based on earlier reported studies was unclear (3). In our initial prospective study of the ability of MR imaging performed in the years 1985–1987 to localize gastrinoma, we concluded that MR imaging had lower sensitivity than CT or angiography and should not be used routinely (12). However, a study in 1989 (31) of patients with advanced Zollinger-Ellison syndrome showed that MR imaging was equally sensitive to the CT scan. The difference between these two studies can probably be accounted for by the fact that only half the patients studied at the NIH with Zollinger-Ellison syndrome had advanced disease, whereas most patients had advanced disease with large tumor bulk in the other study (3, 18). Because of recent technical improvements in MR imaging (13, 14), MR imaging is likely now to be more useful for examining for metastatic pancreatic endocrine tumors than either of these older studies suggest. In order to evaluate the usefulness of current MR imaging in patients with pancreatic endocrine tumors, in the present study 43 patients with Zollinger-Ellison syndrome with different tumor staging were examined with MR imaging and the results compared to those with ultrasound, CT, and ANGIO. The patients studied represented all consecutive patients evaluated at our center during the period from 1988 to 1991 and thus are representative of the spectrum of disease seen in large series of patients with Zollinger-Ellison syndrome (3, 18, 28–30).
Our interest in assessing MR imaging in this disease is because of the increasing clinical importance of localizing the primary tumor, determining the presence of metastatic disease, and assessing the extent of metastatic disease (2, 3). In this study, we analyzed data separately for gastrinoma metastatic to the liver and primary tumors because in terms of their clinical implications they are assessed for different reasons (1–4). Primary gastrinomas are imaged preoperatively in order to localize tumors for possible resection (2, 3, 5, 23). Hepatic metastases are assessed initially to determine whether surgical resection should be attempted (32). In patients with diffuse hepatic metastases, repeated imaging studies determine whether progression is occurring and to monitor response in these patients during various treatment protocols (chemotherapy, interferon, somatostatin, etc) (3, 33–35). Previously, hepatic metastases were reported to be localized in 40–60% of patients by ultrasound (3, 8), in 50–80% of patients with CT (3, 4, 20), and greater than 90% of the time with ANGIO (3, 4, 21). Because the sensitivity of ultrasound is too low to be generally useful and ANGIO is an invasive test, making repeated studies difficult, CT has been recommended as the best initial study (20); however, it frequently misses small metastatic lesions and thus in some patients lacks the sensitivity needed to determine the true extent of the tumor.
In the present study, MR imaging is shown to be superior to ultrasound, CT, and ANGIO for the detection of gastrinoma in the liver. The enhanced sensitivity observed with the newer generation of MR scanners compared to those reported in previous studies (12, 36) is likely a result of improvements in motion suppression as well as the increased use of STIR sequences. Also in contrast to our previous report (12), T2-weighted sequences (SE 2000/80–100) were not used in this study because STIR sequences have been shown in our previous experience (10, 13) to be more sensitive in detecting hepatic metastases. By using a combination of STIR and T1-weighted sequences, gastrinomas were detected in the liver in 15/18 (83%) of patients with biopsy-proven metastatic disease. In contrast, ultrasound, CT, and ANGIO were positive in only 9/18 (50%), 10/18 (56%), and 11/18 of patients (61%), respectively. The combination of all noninvasive studies (MR imaging, CT, and ultrasound) was not more sensitive than MRI alone, demonstrating that only MR imaging need be done. These results have similarities and differences from other reported studies (12, 31, 36, 37). In our earlier study of seven patients with metastatic gastrinoma (12), we reported that MR imaging had a sensitivity of only 43% for detecting metastatic gastrinoma in the liver, which was equivalent to the sensitivity of ultrasound (43%) and was less sensitive than CT (71%) or ANGIO (71%). The lower sensitivity in CT and ANGIO observed in the current study compared to our previous study may be accounted for by the fact that we are currently evaluating patients earlier in the course of their metastatic disease because of active recruitment of patients with minimal metastatic disease who may benefit from surgery. Another study (31) reported that MR imaging detected metastatic hepatic gastrinoma in 8/8 patients (100%); however, these patients all had large metastatic tumors and CT was also positive in 7/8 patients (88%). Stark et al (37), in two patients with advanced metastases, showed that MR imaging was equally sensitive in detecting hepatic gastrinoma compared to CT imaging. Anderson et al (36) reported in 17 patients with endocrine tumors metastatic to the liver that MR imaging was less sensitive than ultrasound (36).
MR imaging in the present study was found to be more useful clinically than CT in management of metastatic disease. Compared to CT scanning, MR imaging changed clinical management in 9/18 patients (50%) with metastatic disease. The increased sensitivity of MR imaging allowed liver metastases to be detected in five patients thought to be surgical candidates for primary resection after CT revealed no hepatic metastases and altered the surgical approach. MR imaging identified bilobar disease in three additional patients with CT scan results that showed potential resectability. In two recent studies (32, 33), 15% of patients with metastatic disease to the liver with Zollinger-Ellison syndrome were found to have metastases confined primarily to one lobe and thus might benefit from partial hepatectomy. Because MR imaging identified additional lesions not seen on CT scan in the other lobe, it proved to be more useful than CT scanning in determining which patients could benefit from hepatic resection and those who might require chemotherapy. Furthermore, MR imaging identified additional hepatic metastases compared to CT scan in five of these patients and an additional patient, thereby altering the approach to antitumor therapy. Even though the sensitivity and specificity of MR imaging to detect hepatic gastrinoma using STIR sequences was equal to that observed with T1-weighted images, pancreatic endocrine tumors were more clearly seen on STIR sequences because they were hyperintense. False positive results were obtained in 4/25 (16%) patients using Tl-weighted images compared with 3/25 (12%) observed with STIR sequences. All four of the patients with false positive MR imaging results were found at surgery to have hepatic hemangiomas. Hepatic hemangiomas may appear similar to pancreatic endocrine tumors and are difficult to differentiate using conventional MR imaging sequences. Very heavily T2-weighted pulse sequences were not used in this study to distinguish hemangiomas from metastatic gastrinomas. Only by showing rapid growth of a lesion, or increased numbers of hyperintense lesions, can the diagnosis of pancreatic endocrine tumor be made with more certainty from that of hepatic hemangioma. In contrast to MR imaging, neither CT nor ultrasound identified these small hepatic hemangiomas, whereas in two patients the hemangiomas were correctly identified preoperatively by ANGIO. It has been suggested that the use of gadolinium-DTPA might be helpful in differentiating hepatic hemangiomas from metastatic disease in the liver (17, 31, 39, 40, 45) because of prolonged enhancement of hemangiomas with gadolinium-DTPA scans (40, 45).
For the detection of primary gastrinoma tumors, MR imaging is only half as sensitive as ANGIO and equally sensitive as ultrasound and CT. Furthermore, the combination of all noninvasive imaging was better than any one alone, but still significantly less sensitive than ANGIO, demonstrating that angiographic studies should continue to be done to localize the primary tumor. The results from our study are in marked contrast to the study of Tjon et al (31), in patients with advanced disease (60% had metastatic disease) in which CT or MR imaging localized 61% of the primary tumors. The most likely explanations are differences in the extent of the disease at the time of the study and the size of the primary tumors.
Gadolinium-DTPA, a paramagnetic contrast agent, was shown previously (15, 17) to improve the detection of both primary and metastatic pancreatic endocrine tumors. In the present study gadolinium did not improve the sensitivity of detecting either metastatic gastrinomas in the liver or the primary tumor, but the addition of dynamic scanning with bolus injection of gadolinium-DTPA and rapid acquisition may improve these results, because recent reports suggest that dynamic, bolus-enhanced MR using recently introduced rapid acquisition may improve the sensitivity of contrast enhanced MR for detection of primary or metastatic gastrinoma (40–44).
In conclusion, the present study demonstrates that recent developments in MR imaging technology have now made this the diagnostic modality of choice for the detection of metastatic pancreatic endocrine tumors to the liver. Current MR imaging using a 0.5 Tesla scanner is not only more sensitive than other imaging modalities, but it also is superior to CT in assessing involvement of both hepatic lobes. Furthermore, because STIR sequences require an imaging time of only 20 min, it is comparable to ultrasound and CT and requires less time than ANGIO. It is important to remember that the MR imager used in this study is a 0.5 Tesla unit, which may allow greater detection of hepatic metastases because of improved body imaging due to greater motion artifact suppression compared to 1.5 Tesla units, which are now commonly employed in clinical practice. This study also demonstrates that for the detection of primary gastrinomas, selective angiography remains the localizing study of choice. The recent advances in MR imaging have not improved its sensitivity for detecting the primary lesion. Therefore, MR imaging should be the primary imaging study for detecting the presence of metastatic gastrinoma to the liver and for following these metastatic tumors during chemotherapy to assess response. ANGIO should be the principal imaging study for detecting, preoperatively, the location and resectibility of primary gastrinoma. For preoperative staging, MR imaging to exclude metastatic disease and ANGIO to detect the location and resectibility of primary gastrinoma should be performed in every patient with Zollinger-Ellison syndrome.
REFERENCES
- 1.Jensen RT, Norton JA: Endocrine neoplasms. In Textbook of Gastroenterology Yamada T (ed). Philadelphia, JB: Lippincott, 1991, pp 1912–1937 [Google Scholar]
- 2.Norton JA, Doppman JL, Jensen RT: Cancer of the endocrine system. In Principles and Practice of Oncology, 3rd ed. DeVita VT, Helman S, Rosenberg SA, (eds). Philadelphia, JB: Lippincott, 1989, pp 1269–1344 [Google Scholar]
- 3.Jensen RT, Maton PN: Zollinger-Ellison syndrome. In The Stomach Gustavsson S, Kumar D, Graham DY (eds). London, Churchill-Livingstone, 1991, pp 341–374 [Google Scholar]
- 4.Saeed ZA, Doppman JL, Norton J, Maton PN, Gardner JD, Jensen RT: Gastrinoma localization in Zollinger-Ellison syndrome. Intern Med Spec 9:79–99, 1988 [Google Scholar]
- 5.Norton JA, Doppman JL, Jensen RT: Results of curative resection in patients with Zollinger-Ellison syndrome: A ten-year prospective study. Ann Surg 215:8–18, 1992 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Stabile BE, Passaro E: Benign and malignant gastrinoma. Am J Surg 149:144–148, 1985 [DOI] [PubMed] [Google Scholar]
- 7.Malagelada JR, Edis AJ, Adson MA, von Heerden JA, Go VLW: Medical and surgical options in the management of patients with gastrinoma. Gastroenterology 84:1524–1532, 1983 [PubMed] [Google Scholar]
- 8.London JF, Shawker TH, Doppman JL, Fruct H, Vinayek R, Stark HA, Miller LS, Miller DL, Norton JA, Jensen RT, Gardner JD, Maton PN: Prospective assessment of abdominal ultrasound in patients with Zollinger-Ellison syndrome. Radiology 178:763–767, 1991 [DOI] [PubMed] [Google Scholar]
- 9.Shawker TH, Doppman JL, Dunnick NR, McCarthy DM: Ultrasound investigation of pancreatic islet cell tumors. J Ultrasound Med 1:193–200, 1982 [DOI] [PubMed] [Google Scholar]
- 10.Ferrucci JT, Freeny PC, Stark DD, Foley WD, Mueller PR, May G, Burhenne HJ: Advances in hepatobiliary radiology. Radiology 168:319–338, 1988 [Google Scholar]
- 11.Kressel HY: Strategies for magnetic resonance imaging of focal liver disease. Radio Clin North Am 26:607–615, 1988 [PubMed] [Google Scholar]
- 12.Frucht H, Doppman JL, Norton JA, Miller DL, Dwyer AJ, Frank JA, Vinayek R, Maton PN, Jensen RT: Gastrinomas: Comparison of MR imaging with CT, angiography and US. Radiology 171:713–717, 1989 [DOI] [PubMed] [Google Scholar]
- 13.Dwyer AJ; Frank JA, Sank VJ, Reinig JW, Hickey AM, Doppman JL: Short-T1 inversion-recovery pulse sequence: Analysis and initial experience in cancer imaging. Radiology 168:827–836, 1988 [DOI] [PubMed] [Google Scholar]
- 14.Stark DD, Hendrick RE, Hahn PF, Ferrucci JT: Motion artifact reduction with fast spin-echo imaging. Radiology 164:183–191, 1987 [DOI] [PubMed] [Google Scholar]
- 15.Legman P, Ruszniewski P, Sibert A, Somveille E, Hercot O, Benacertaf R, Mignon M: Computerized tomography (CT) and magnetic imaging (MRI) in liver metastases (LM) of digestive endocrinology tumors (DET): A prospective study in 24 patients. Gastroenterology 96:A294, 1989 [Google Scholar]
- 16.Carr DH, Gadian DG: Contrast agents in magnetic resonance imaging. Clin Radiol 36:561–568, 1985 [DOI] [PubMed] [Google Scholar]
- 17.Tjon A, Tham RT, Jansen JBMJ, Falke THM, Jansen MJ, Lamers CBHW: MRI and CT of gastrinoma in ZollingerEllison syndrome. Radiology 169:412(A), 1988 [Google Scholar]
- 18.Jensen RT; Doppman JL, Gardner JD: Gastrinoma. In The Exocrine Pancreas: Biology, Pathobiology and Disease, 2nd ed. Go VLW, DiMagno EP, Gardner JD, Lebenthal E, Reber H, Scheele GA (eds). New York, Raven Press; (in press) [Google Scholar]
- 19.Frucht H, Howard JM, Slaff JE, McCarthy DM, Maton PN, Wank SA, Vinayek R, Gardner JD, Jensen RT: Secretin and calcium provocative tests in patients with Zollinger-Ellison syndrome: A prospective study. Ann Intern Med 111:713–722, 1989 [DOI] [PubMed] [Google Scholar]
- 20.Wank SA, Doppman JL, Miller DL, Collen MJ, Maton PN, Vinayek R, Slaff JI, Norton JA, Gardner JD, Jensen RT: Prospective study of the ability of computerized axial tomography to localize gastrinomas in patients with ZollingerEllison syndrome. Gastroenterology 92:905–912, 1987 [DOI] [PubMed] [Google Scholar]
- 21.Maton PN, Miller DL, Doppman JD, Collen MJ, Norton JA, Vinayek R, Slaff JI, Wank SA, Gardner JD, Jensen RT: Role of selective angiography in the management of ZollingerEllison syndrome.·Gastroenterology 92:913–919, 1987 [DOI] [PubMed] [Google Scholar]
- 22.Cherner JA, Doppman JL, Norton JA, Collen MJ, Maton PN, Gardner JD, Jensen RT: Prospective assessment of selective venous sampling for gastrin to localize gastrinomas. Ann Intern Med 105:841–847, 1986 [DOI] [PubMed] [Google Scholar]
- 23.Doppmart JL, Miller DL, Chang R, Maton PN, London JF, Gardner JD, Jensen RT, Norton JA: Prospective study of gastrinoma localization and resection in patients with Zollinger-Ellison syndrome. Radiology 174:25–29, 1990. 2294556 [Google Scholar]
- 24.Frucht H, Norton JA, London JF, Vinayek R, Doppman JL, Gardner JD, Jensen RT, Maton PN: Detection of duodenal gastrinomas by operative transillumination: A prospective study. Gastroenterology 99:1622–1627, 1990 [DOI] [PubMed] [Google Scholar]
- 25.Norton JA, Cromack DT, Shawker TH, Doppman JL, Cumi R, Gorden P, Maton PN, Gardner JD, Jensen RT: Intraoperative ultrasonographic localization of islet cell tumors: A prospective comparison to palpation. Ann Surg 207:160–168, 1988 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Weinstein MC, Feinberg HV: Clinical Decision Analysis Philadelphia, WB: Saunders, 1980, pp 84–88 [Google Scholar]
- 27.McNemar Q: Psychological Statistics, 4th ed. New York, John Wiley & Sons, 1969 [Google Scholar]
- 28.Mignon M, Ruszniewski R, Haffar S, Rigaud D, Rene E, Bonfils S: Current approach to the management of tumoral process in patients with gastrinoma. World J Surg 10:702–709, 1986 [DOI] [PubMed] [Google Scholar]
- 29.Stadil F, Stage JG: The Zollinger-Ellison syndrome. Clin Endocrinol Metab 9:433–446, 1979 [DOI] [PubMed] [Google Scholar]
- 30.Bonfils S, Landor JH, Mignon M, Hervoir P: Results of surgical management in 92 consecutive patients with Zollinger-Ellison syndrome. Ann Surg 194:692–697, 1981 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Tjon A, Tham RTO, Falke THM, Jansen JBMJ, Lamers CBHW. CT artd MR imaging of advanced Zollinger-Ellison syndrome. J Comput Assist Tomogr 13:821–828, 1989 [DOI] [PubMed] [Google Scholar]
- 32.Norton JA, Sugarbaker PH, Doppman JL, Wesley RA, Maton PN, Gardner JD, Jensen RT: Aggressive resection of metastatic disease in selected patients with malignant gastrinoma. Ann Surg 203:352–359, 1986 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.von Schrenck T, Howard JM, Doppman JL, Norton JA, Smith F, Maton PN, Vinayek R, Frucht H, Wank SA, Gardner JD, Jensen RT: Prospective study of chemotherapy in patients with metastatic gastrinoma. Gastroenterology 94:1326–1334, 1988 [DOI] [PubMed] [Google Scholar]
- 34.Jensen RT, Gardner JD: Zollinger-Ellison syndrome. Clinical presentation, pathology, diagnosis and treatment. In Peptic Ulcer and Other Acid-Related Diseases Zakim D (ed). New York, Spectrum Publishing, 1991, pp 117–211 [Google Scholar]
- 35.Ericksson B, Oberg K, Alm G, Karlsson A, Lundquist G, Andersson T, Wilander E, Wide L: Treatment of malignant endocrine pancreatic tumors with human leukocyte interferon. Lancet 2:1307–1309, 1986 [DOI] [PubMed] [Google Scholar]
- 36.Andersson T, Eriksson B, Hemmingsson A, Lindgren PG, Oberg K: Angiography, computerized tomography, magnetic resonance imaging and ultrasonography in detection of liver metastases from endocrine gastrointestinal tumors. Acta Radiol 28:535–539, 1987 [PubMed] [Google Scholar]
- 37.Stark DD, Moss AA, Goldberg HI, Deveney CW, Way L: Computed tomography and nuclear magnetic resonance imaging of pancreatic islet cell tumors. Surgery 94:1024–1027, 1983 [PubMed] [Google Scholar]
- 38.Ohtomo K, Itai Y, Yoshikawa K, Kokubo T, Vashiro N, Iio N: Hepatic hemangioma: Dynamic MRI using gadolinium DPTA. Eur J Radiol 7:257–259, 1987 [PubMed] [Google Scholar]
- 39.Freeny GC, Marks WM: Hepatic hemangioma: Dynamic bolus CT. AJR 147:711–719, 1986 [DOI] [PubMed] [Google Scholar]
- 40.Chezmar JL, Rumancik WM, Megibow AJ, Hulnick DH, Nelson RC, Bernardino ME: Liver and abdominal screening in patients with cancer: CT versus MR imaging. Radiology 168:43–47, 1988 [DOI] [PubMed] [Google Scholar]
- 41.Mirowitz SA, Gutierrez E, Lee JKT, Brown JJ: Normal abdominal enhancement patterns with dynamic gadolinium enhanced MR imaging. Radiology 180:637–640, 1991 [DOI] [PubMed] [Google Scholar]
- 42.Mirowitz SA, Lee JKT, Guiterrez E, Brown JJ, Heiken JP, Eilenberg SS: Dynamic gadolinium-enhanced rapid acquisition spin-echo MR imaging of the liver. Radiology 179:371–376, 1991 [DOI] [PubMed] [Google Scholar]
- 43.Hamm B, Wolf KJ, Felix R: Conventional and rapid MR imaging of the liver with gadonlinium-DTPA. Radiology 164:313–320, 1987 [DOI] [PubMed] [Google Scholar]
- 44.Edelman RR, Wallmer B, Singer A, Atkinson DJ, Saini S: Segmental turbo FLASH; method for breathhold MR imaging of the liver with Flexible contrast. Radiology 177:515–521, 1990 [DOI] [PubMed] [Google Scholar]
- 45.Hamm B, Fisher E, Taupitz M: Differentiation of hepatic hemangiomas from metastases by dynamic contrast enhanced MR imaging. J Comput Assist Tomogr 14:205–216, 1990 [DOI] [PubMed] [Google Scholar]