Abstract
CD80 is mainly expressed on Ag-presenting cells (APCs) as a costimulatory molecule but is also detected on T cells. However, the origin and physiological role of CD80 on CD8+ T cells remain unclear. In the present study, we demonstrated that effector and memory CD8+ T cells, but not naïve CD8+ T cells, displayed CD80 molecules on their surfaces after acute lymphocytic choriomeningitis virus infection. Using adoptive transfer of CD80-knockout (KO) CD8+ T cells into a wild type or CD80-KO recipient, we demonstrated that the effector CD8+ T cells displayed CD80 by both intrinsic expression and extrinsic acquisition, while memory CD8+ T cells displayed CD80 only by extrinsic acquisition. Interestingly, the extrinsic acquisition of CD80 by CD8+ T cells was observed only in the lymphoid organs but not in the periphery, indicating the trogocytosis of CD80 molecules via interaction between CD8+ T cells and APCs. We compared the recall immune responses by memory CD8+ T cells that either extrinsically acquired CD80 or were deficient in CD80, and found that CD80, presented by memory CD8+ T cells, played a role in limiting their expansion and IL-2 production upon exposure to secondary challenge. Our study presents the in vivo dynamics of the extrinsic acquisition of CD80 by Ag-specific CD8+ T cells and its role in the regulation of recall immune responses in memory CD8+ T cells.
Keywords: B7-1 antigen, Extrinsic acquisition, Trogocytosis, Recall immune response, Lymphocytic choriomeningitis virus
INTRODUCTION
After acute viral infection, naïve T cells become activated and begin to proliferate following Ag recognition. In the contraction phase, most of the effector T cells undergo apoptosis and only a small population of the T cells survives and differentiates into memory T cells (1). The best-known marker for Ag-experienced T cells, that is, CD44 is expressed not only on activated T cells, but also on some naïve T cells that display a memory-phenotype (2,3). In addition, other markers, including CD62L and CD127, which represent memory T cells, are also upregulated on memory-like naïve T cells, and thus, memory T cells cannot be distinguished from such naïve T cells (1). Therefore, it is important to identify new markers for Ag-experienced T cells, including true-memory T cells.
Previous studies have shown that biotin-labeled surface components of Ag-presenting cells (APCs) were detected in T cells after activation with viral peptide or infection (4,5). Among the APC surface components, CD80 molecules could be acquired by in vitro activated CD4+ T cells, and the amount of acquisition was directly related to the strength of the signal mediated via a peptide-MHC on the APCs (6). In other words, activated T cells can extrinsically acquire CD80 molecules from APCs via trogocytosis, a cell-contact dependent uptake of plasma membranes and associated molecules. However, the in vivo kinetics of trogocytosed CD80 molecules on Ag-specific T cells remain unknown.
The costimulatory molecules, CD80 and CD86, are expressed on APCs and share their ligands, CD28 and cytotoxic T-lymphocyte-associated protein 4 (CTLA-4), on T cells (7). Since CTLA-4 binds to CD80 and CD86 with a higher affinity and avidity than CD28, an inhibitory signal for T cells via CTLA-4 is superior to a costimulatory signal via CD28 (8). A previous study has shown that CD80 interacts with PD-L1 and its affinity is intermediate between the affinity of CD28:CD80 and CTLA-4:CD80. CD80 and PD-L1 can specifically interact to inhibit in vitro T cell proliferation and cytokine production (9). However, the physiological role of extrinsically acquired CD80 on T cells has not yet been studied.
Here, we reported that CD80 molecules are detected on Ag-specific CD8+ T cells for a long time period after virus infection but not on naïve T cells. The persisted CD80 molecules on CD8+ T cells later point after infection was observed only in the lymphoid organs, but not in the periphery, which was mainly caused by extrinsic acquisition rather than intrinsic expression. The memory CD8+ T cells that extrinsically acquired CD80 exhibited less in vivo expansion and IL-2 production in the recall responses than CD80-deficient memory CD8+ T cells, suggesting the potential role of CD80 on memory CD8+ T cells in regulating the intensity of recall immune responses.
MATERIALS AND METHODS
Mice and viral infections
Five- to six-week-old female C57BL/6 and CD80-knockout (KO) mice were purchased from the Jackson Laboratory. Lymphocytic choriomeningitis virus (LCMV) GP33–41 epitope-specific TCR transgenic P14 Thy1.1 mice were provided by Dr. Rafi Ahmed (Emory University School of Medicine, Atlanta, GA, USA). CD80-KO mice and P14 Thy1.1 mice were bred to generate CD80-KO P14 mice. For acute infection, mice were infected with 2×105 plaque-forming units (PFUs) of LCMV armstrong (Arm) via intraperitoneal injection or with 2×106 PFUs of LCMV Arm via intravenous injection. For chronic infection, mice were infected with 2×106 PFUs of LCMV clone 13 (CL13) via intravenous injection. All mice were maintained in a specific pathogen-free facility in accordance with Institutional Animal Care and Use Committee (IACUC) guidelines at Yonsei University. Animal experiments were approved by the IACUC of Yonsei University (IACUC No. 201709-629-02) and conducted in accordance with approved IACUC protocols.
Abs and flow cytometry
PBMCs and lymphocytes from the tissues were maintained as described previously (10). For phenotypic analysis of Ag-specific CD8+ T cells derived from the blood and tissues, cells were stained with the following fluorochrome-conjugated Abs in PBS containing 0.2% FBS: Abs against CD62L (MEL-14), CD44 (IM7), CD4 (GK1.5), CD107a (1D4B), CD90.1 (Thy1.1; OX-7), and CD90.2 (Thy1.2; 53-2.1) (BD Biosciences, San Jose, CA, USA); Abs against CD80 (16-10A1) (Biolegend, San Diego, CA, USA); and Abs against CD8 (53-6.7) and CD127 (A7R34) (eBiosciences, San Diego, CA, USA) in the presence of virus-specific tetramer. H-2Db tetramers bound to GP33–41 peptides were generated and used as previously described (11). For intracellular cytokine staining, splenocytes were re-stimulated ex vivo for 5 h with 0.2 µg/ml of LCMV GP33–41 peptide for CD8+ T cell activation in the presence of GolgiPlug (BD Biosciences) and GolgiStop (BD Biosciences). The stimulated cells were fixed, permeabilized, and stained with the fluorochrome-conjugated Abs against IL-2 (JE6-5H4), IFN-γ (XMG1.2), and TNF-α (MP6-XT22) (BD Biosciences). To remove the dead cell population, the Live/Dead Fixable Dead Cell Stain Kit (Invitrogen, Thermo Fisher Scientific, Waltham, MA, USA) was used in staining procedures. Flow cytometry was performed using the FACSCanto II (BD Biosciences). Data was analyzed using FlowJo software (Treestar, Ashland, OR, USA).
Cell enrichment and adoptive transfer
For P14 cell enrichment, P14 cells were isolated from the spleen (SP) using a CD8+ T cell isolation kit by negative selection (Miltenyi Biotec, Bergisch Gladbach, Germany). To analyze the source of CD80 by T cells, 5×103 wild type (WT) P14 CD8+ (Thy1.1/1.1) and 5×103 CD80-KO P14 CD8+ (Thy1.1/1.2) T cells were adoptively transferred together into either WT or CD80-KO recipient mice. To analyze the location of the extrinsic acquisition of CD80 by T cells, 5×103 WT P14 CD8+ (Thy1.1/1.1) and 5×103 CD80-KO P14 CD8+ (Thy1.1/1.2) T cells were adoptively transferred together into WT recipient mice. For functional analysis of CD80-acquired T cells, 5×103 CD80-KO P14 CD8+ (Thy1.1/1.2) T cells were adoptively transferred into either WT or CD80-KO recipient mice. One day after the adoptive transfer, mice were intraperitoneally infected with 2×105 PFUs LCMV Arm. To analyze the recall responses to LCMV infection, both the 5×104 CD80-acquired memory CD8+ (Thy1.1/1.1) and 5×104 CD80-deficient memory CD8+ (Thy1.1/1.2) T cells were intravenously injected into recipient mice, followed by infection with 2×105 PFUs of LCMV Arm via intraperitoneal injection.
Memory CD8+ T cell generation
For establishment of CD8+ memory T cells in vivo, 5×103 CD80-KO P14 CD8+ (Thy1.1/1.2) T cells were adoptively transferred into either WT or CD80-KO recipient mice, followed by infection with 2×105 PFUs of LCMV Arm. After 60 days, total CD8+ T cells were isolated from the SP by MACS, followed by sorting of memory P14 cells from the bulk CD8+ T cells using the BD LSR II sorting system (BD Biosciences).
Statistical analysis
Data were analyzed using the 2-tailed unpaired Student's t-test with GraphPad Prism software (GraphPad Software, San Diego, CA, USA). Results were presented as the mean±SEM. Differences with a p value less than 0.05 were considered statistically significant.
RESULTS
Upregulated CD80 persists for a long time in Ag-specific CD8+ T cells compared to naïve T cells after virus infection
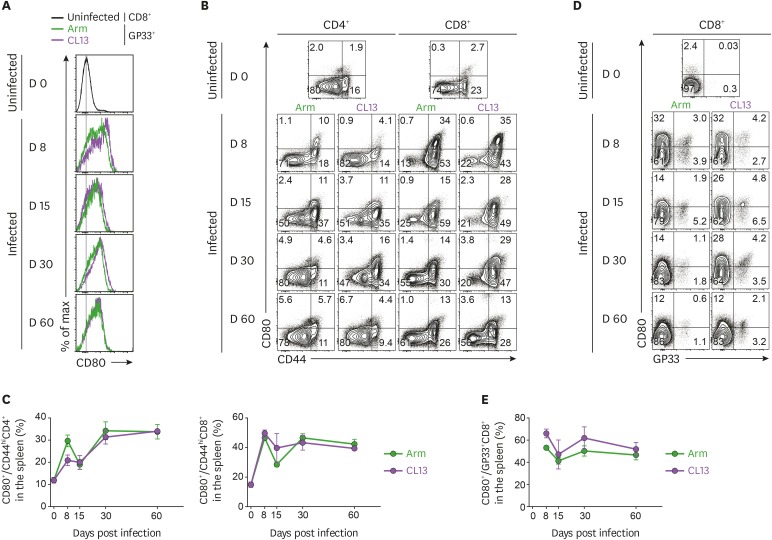
To investigate the kinetics of CD80 molecules within T cells following acute or chronic LCMV infection, mice were infected by the same route with LCMV Arm or CL13 with the same dose. The SP isolated from the infected mice was analyzed by flow cytometry. Compared with the expression of CD80 on naïve CD8+ T cells, the expression of CD80 was upregulated in Ag-specific effector and memory CD8+ T cells after acute virus infection, as well as Ag-specific exhausted CD8+ T cells after chronic virus infection (Fig. 1A). When gated on total CD4+ or CD8+ T cells, CD80 expression was not observed in the naïve population independent of CD44 expression level. However, after infection, the percentage of CD80-expressing cells significantly increased among activated CD44hiCD4+ and CD44hiCD8+ T cells, but not among CD44lo naïve T cells (Fig. 1B and C). The frequency of these CD80-expressing cells was higher in CD44hiCD8+ T cells compared with CD44hiCD4+ T cells. More interestingly, the percentage of CD80-expressing cells in Ag-specific CD8+ T cells was still observed in the SP at day 60 post-infection (Fig. 1D and E). Taken together, CD80 levels were upregulated on Ag-specific CD8+ T cells, and the upregulation of CD80 was maintained on memory or exhausted CD8+ T cells for a long time regardless of LCMV virus strain, suggesting that CD80 could be a subset for Ag-experienced T cells.
Figure 1. Upregulation of CD80 is maintained in Ag-experienced T cells compared to naïve T cells after virus infection. C57BL/6 mice were intravenously infected with 2×106 PFUs of LCMV Arm or CL13. The mice were sacrificed at the days indicated post-infection and the SPs were analyzed. (A) Expression of CD80 on naïve CD8+ and GP33 tetramer-positive CD8+ T cells after Arm or CL13 infection. (B) Representative data showing expression of CD80 on CD44lo and CD44hi population among CD4+ and CD8+ T cells after Arm or CL13 infection. (C) Frequency of CD80+ cells among CD44hiCD4+ and CD44hiCD8+ T cells after Arm or CL13 infection. (D) Representative data showing expression of CD80 on GP33 tetramer-positive cells among CD8+ T cells. (E) Frequency of CD80+ cells among GP33 tetramer-positive cells after Arm or CL13 infection. These data are representative of 3 independent experiments (n=4 mice per group). Results represent mean±SEM.
Ag-specific CD8+ T cells transiently upregulate CD80 expression in a cell-intrinsic manner
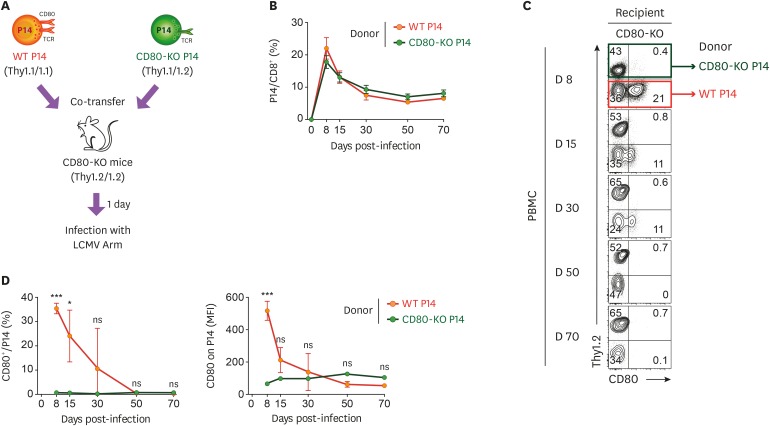
First, we assessed whether or not CD80 molecules found on Ag-specific CD8+ T cells were intrinsically expressed. WT and CD80-KO P14 CD8+ T cells with different congenic markers were adoptively transferred together into CD80-KO mice, followed by infection with LCMV Arm (Fig. 2A). In this setting, the appearance of CD80 molecules on WT P14 CD8+ T cells represents intrinsic expression since CD80-KO recipient mice do not present CD80 molecules. The frequency between donor WT and CD80-KO P14 CD8+ T cells among CD8+ T cells in the blood showed similarity in CD80-KO recipient mice at each day post-infection (Fig. 2B). Interestingly, upregulation of CD80 was observed on donor WT P14 CD8+ T cells during the effector T cell differentiation period, and the CD80 level gradually decreased and disappeared in the blood of CD80-KO mice after day 50 post-infection (Fig. 2C and D). As control, CD80-KO P14 CD8+ T cells did not show any level of CD80 in the blood of CD80-KO mice at all the post-infection time points. In conclusion, these results indicated that the intrinsic expression of CD80 on Ag-specific CD8+ T cells occurs during the early phase of T cell differentiation but not posteriorly.
Figure 2. The intrinsic expression of CD80 on Ag-specific CD8+ T cells gradually decreases in the blood. (A) Experimental scheme for investigation of intrinsic expression and extrinsic acquisition of CD80 on Ag-specific CD8+ T cells in the blood. WT P14 CD8+ and CD80-KO P14 CD8+ T cells were adoptively transferred together into CD80-KO recipient mice. One day after the adoptive transfer, mice were intraperitoneally infected with 2×105 PFUs LCMV Arm. (B) Frequency of donor WT and CD80-KO P14 CD8+ T cells among CD8+ T cells obtained from PBMCs of CD80-KO recipient mice at the days indicated post-infection. (C) Expression of CD80 on WT and CD80-KO P14 CD8+ T cells among Thy1.1+ donor cells obtained from PBMCs of CD80-KO mice at the days indicated post-infection. (D) Frequency of CD80-expressing cells and MFI of CD80 among WT and CD80-KO P14 CD8+ T cells obtained from PBMCs of CD80-KO mice at the days indicated post-infection. Data are representative of 3 independent experiments (n=3−4 mice per group). Results represent the mean±SEM and statistical significance was determined by 2-tailed unpaired Student's t-test.
NS, not significant; MFI, mean fluorescence intensity.
*p<0.05; ***p<0.001.
Extrinsic acquisition of CD80 by Ag-specific CD8+ T cells occurs in the lymphoid organs
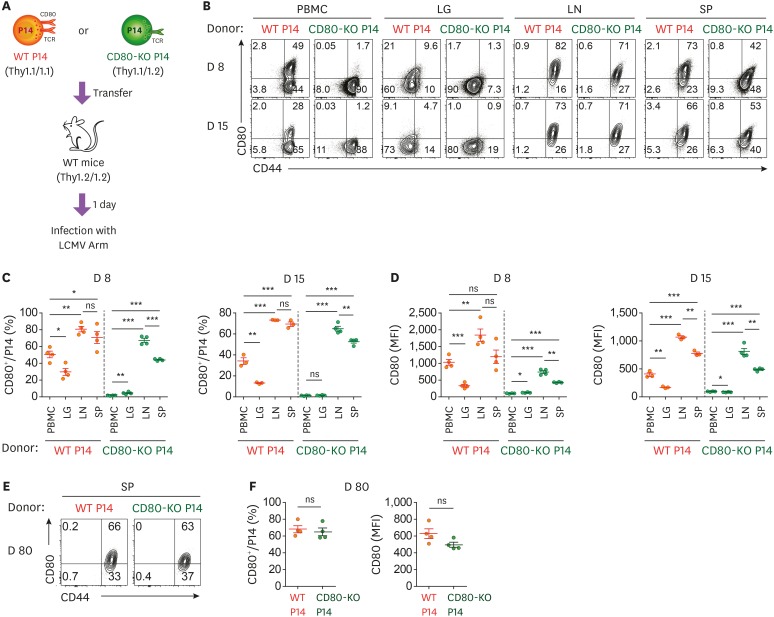
Given that upregulation of CD80 by in vitro activated CD4+ T cells is originated from APCs (6), CD80 expression on Ag-specific T cells may be derived extrinsically after virus infection. In the previous experiment, we found that CD80 molecules on Ag-specific CD8+ T cells were maintained for a long time after virus infection (Fig. 1A and D); however, their intrinsic expression was limited only to the effector phase (Fig. 2C and D). Therefore, we investigated whether the extrinsic acquisition of CD80 actually contributes to the maintenance of CD80 levels on Ag-specific CD8+ T cells and where the phenomenon occurs in vivo. To address this question, WT or CD80-KO P14 CD8+ T cells with different congenic markers were adoptively transferred into WT mice containing CD80, followed by infection with LCMV Arm (Fig. 3A). In this context, the appearance of CD80 molecules on CD80-KO P14 CD8+ T cells represents the extrinsic acquisition of CD80 since WT recipient mice can provide CD80 molecules. In contrast, CD80 molecules on WT P14 CD8+ T cells originated from both extrinsic acquisition and intrinsic expression of CD80.
Figure 3. The extrinsic acquisition of CD80 by CD8+ T cells occurs only in the lymphoid organs but not in the blood and LG. (A) Experimental scheme for analyzing the location of extrinsic acquisition of CD80 by CD8+ T cells. Either WT P14 CD8+ or CD80-KO P14 CD8+ T cells were adoptively transferred into WT recipient mice. One day after the adoptive transfer, mice were intraperitoneally infected with 2×105 PFUs LCMV Arm. (B) Expression of CD80 among donor WT and CD80-KO P14 CD8+ T cells obtained from PBMC and various tissues in WT recipient mice at the days indicated post-infection. (C and D) Frequency of CD80-expressing cells (C) and MFI of CD80 (D) among WT or CD80-KO P14 CD8+ T cells obtained from PBMC, LG, LN, and SP in WT recipient mice at the days indicated post-infection. (E) Expression of CD80 among donor WT and CD80-KO P14 CD8+ T cells obtained from the SP of WT mice at day 80 post-infection. (F) Frequency of CD80-expressing cells and MFI of CD80 among WT or CD80-KO P14 CD8+ T cells obtained from the SP of WT mice at day 80 post-infection. Data are representative of 3 independent experiments (n=3−4 mice per group). Results represent mean±SEM and statistical significance was determined by 2-tailed unpaired Student's t-test.
NS, not significant; MFI, mean fluorescence intensity.
*p<0.05; **p<0.01; ***p<0.001.
The mice were sacrificed at the effector phase (days 8 and 15 post-infection) and each population of donor cells were isolated from lymphoid organs or peripheral tissues (Fig. 3B). We analyzed the extrinsic acquisition of CD80 by Ag-specific CD8+ T cells in the blood, lung (LG), and lymphoid organs, including lymph node (LN) and SP. Interestingly, substantial levels of CD80 were observed on CD80-KO P14 CD8+ T cells in the LN and SP, indicating extrinsic acquisition of CD80 by Ag-specific CD8+ T cells in the lymphoid organs (Fig. 3B and C). In contrast, the extrinsic acquisition of CD80 on CD80-KO P14 CD8+ T cells was rarely observed in the blood and LG. In parallel, the percentage of CD80-expressing cells and the levels of CD80 among WT P14 were much higher in the lymphoid organs compared to the blood or LG (Fig. 3B-D). The highest mean fluorescence intensity of CD80 by Ag-specific CD8+ T cells was found in the LN, suggesting that the LN would be the first site where trogocytosis was induced. Surprisingly, at 80 days post-infection, CD80 molecules were still maintained by CD80-KO P14 CD8+ T cells in the SP similar to WT P14 CD8+ T cells (Fig. 3E and F), indicating that memory CD8+ T cells sustain CD80 molecules through the extrinsic acquisition of CD80 in lymphoid organs as previously mentioned (4). In conclusion, these data suggested that the extrinsic acquisition of CD80 by Ag-specific CD8+ T cells is an evidence of interaction between APCs and T cells at the immunological synapse of the lymphoid organs.
CD80 extrinsically acquired by Ag-specific CD8+ T cells does not alter their capability to produce effector cytokines
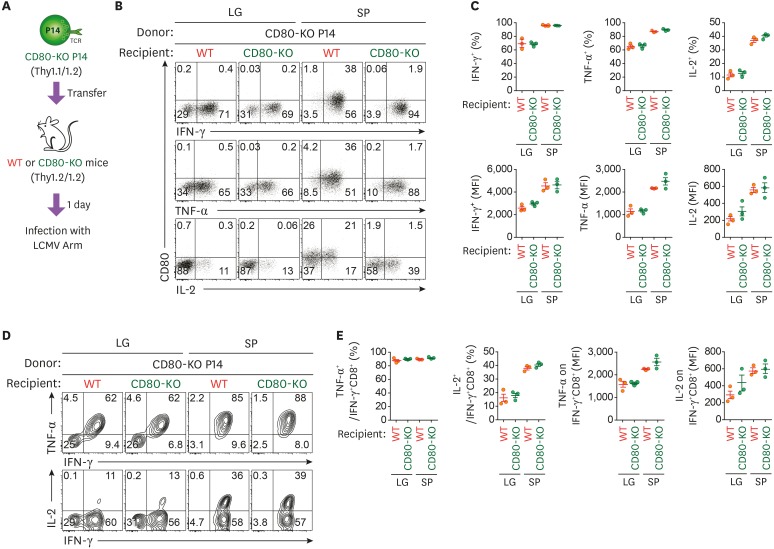
Although there is a possibility that either intrinsic and extrinsic factors may contribute to CD80 upregulation by T cells at the effector stage, CD80 upregulation by CD8+ T cells is mainly derived from the extrinsic acquisition at the memory stage (Fig. 3E and F). To investigate the ability of CD80-acquired T cells to produce cytokines at a later stage of infection during the memory responses, we transferred CD80-KO P14 CD8+ T cells into either WT or CD80-KO recipient mice, followed by infection with LCMV Arm (Fig. 4A). The mice were sacrificed at day 80 post-infection and the production of effector cytokines was analyzed in CD80-acquired and CD80-deficient P14 CD8+ T cells. After restimulation of splenocytes with virus-specific peptide, GP33–41, CD80-acquired CD8+ T cells (CD80-KO P14 cells from WT mice) and CD80-deficient CD8+ T cells (CD80-KO P14 cells from CD80-KO mice) exhibited a similar percentage and number of effector cytokine-producing cells and expression levels of effector cytokines in the SP (Fig. 4B and C). CD80-KO P14 CD8+ T cells in either WT or CD80-KO mice, which did not acquire CD80, also showed similar levels of cytokine production. In addition, the polyfunctionality and cytotoxicity of Ag-specific CD8+ T cells showed no difference between CD80-acquired and CD80-deficient P14 CD8+ T cells (Fig. 4D and E, Supplementary Fig. 1). Thus, these results suggested that the extrinsic acquisition of CD80 by Ag-specific CD8+ T cells does not have an impact on their function regarding cytokine production, at least under in vitro stimulation conditions.
Figure 4. The extrinsic acquisition of CD80 does not affect the ability of Ag-specific memory CD8+ T cells to produce effector cytokines. (A) Experimental scheme for comparison of cytokine productions between CD80-acquired and CD80-deficient memory T cells after in vitro peptide stimulation. CD80-KO P14 CD8+ T cells were adoptively transferred into WT or CD80-KO recipient mice. One day after the adoptive transfer, mice were intraperitoneally infected with 2×105 PFUs LCMV Arm. The LG and SP were obtained from WT and CD80-KO mice at day 60 post-infection and were re-stimulated with GP33 peptide. (B) Representative FACS plots showing production of IFN-γ, TNF-α, and IL-2 versus their CD80 expression in CD80-KO P14 CD8+ T cells obtained from the LG and SP of WT or CD80-KO mice. (C) Frequency and number of IFN-γ+, TNF-α+, and IL-2+ cells and expression levels of IFN-γ, TNF-α, and IL-2 among donor CD80-KO P14 CD8+ T cells obtained from the LG and SP in WT or CD80-KO recipient mice. (D) Representative flow cytometric analysis of production of TNF-α and IL-2 versus IFN-γ in CD80-KO P14 CD8+ T cells obtained from the LG and SP of WT and CD80-KO mice. (E) Frequency and number of TNF-α- and IL-2-producing cells among IFN-γ+CD8+ T cells. MFI of TNF-α and IL-2 among IFN-γ+CD8+ T cells. Data are representative of 3 independent experiments (n=3−4 mice per group). Results represent mean±SEM and statistical significance was determined by 2-tailed unpaired Student's t-test.
MFI, mean fluorescence intensity.
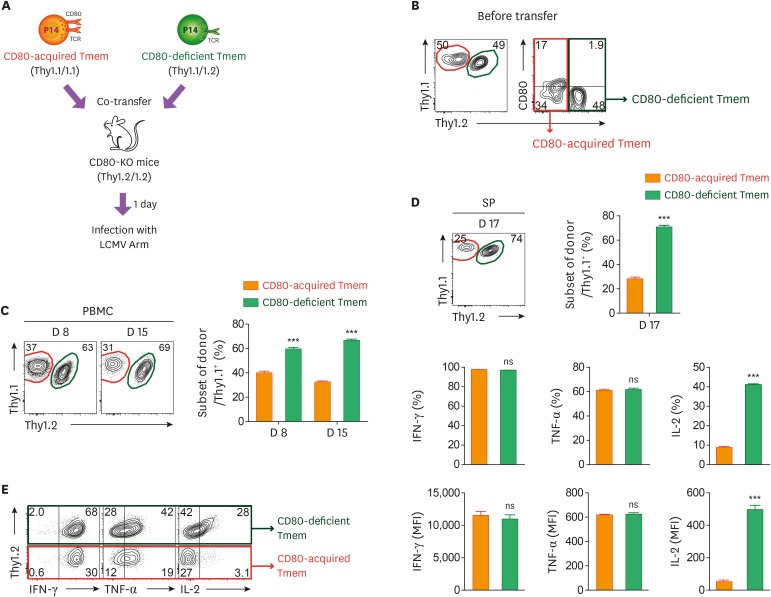
The extrinsic acquisition of CD80 by CD8+ T cells reduces their recall responses upon second LCMV infection
In the previous experiment, we found that the presence or absence of CD80 on memory CD8+ T cells did not affect their role in the production of cytokines under in vitro stimulation. However, since the memory CD8+ T cells can interact again with APCs in lymphoid tissue after secondary exposure to Ags, we tried to compare in vivo expansion and function of CD80-acquired or CD80-deficient memory CD8+ T cells during recall responses. In order to validate these observations, WT and CD80-KO mice were transfected with CD80-KO P14 CD8+ T cells, and subsequently, infected with LCMV Arm. At day 60 post-infection, CD80-acquired and CD80-deficient P14 CD8+ T cells were co-transferred into naïve CD80-KO mice and the recipient mice were challenged with the same virus (Fig. 5A). We confirmed that the frequency of CD80-acquired memory P14 CD8+ T cells was equal to that of CD80-deficient memory P14 CD8+ T cells before transfer (Fig. 5B). During the recall response, CD80-acquired memory P14 CD8+ T cells exhibited a significantly reduced expansion compared to CD80-deficient memory P14 CD8+ T cells in the blood at weeks 1 and 2 post-challenge (Fig. 5C). We also found that the frequency of CD80-acquired donor P14 CD8+ T cells was 3 times lower than that of CD80-deficient donor P14 CD8+ T cells in the SP at day 17 post-challenge (Fig. 5D). When donor P14 CD8+ T cells were stimulated in vitro, there was no difference in the secretion of IFN-γ and TNF-α between 2 different donor P14 CD8+ T cells. However, the ability for IL-2 production significantly decreased in CD80-acquired donor P14 CD8+ T cells compared to CD80-deficient memory T cells (Fig. 5E). In summary, these data indicated that extrinsic acquisition of CD80 on memory CD8+ T cells negatively regulates their in vivo expansion and IL-2 production during recall responses.
Figure 5. CD80-acquired memory CD8+ T cells exhibit a reduced in vivo expansion and a decreased IL-2 production during recall responses. (A) Experimental scheme for comparison of in vivo recall responses between CD80-acquired and CD80-deficient Tmem cells. CD80-acquired and CD80-deficient memory T cells were adoptively transferred together into CD80-KO recipient mice. One day after the adoptive transfer, mice were intraperitoneally infected with 2×105 PFUs LCMV Arm. (B) Representative FACS plots showing the ratio between donor memory T cells and expression of CD80 on each memory T cell before transfer. (C) Frequency of donor memory T cells gated on Thy1.1+ T cells from the PBMCs at day 8 and 15 post-infection. (D) Frequency of donor memory T cells gated on Thy1.1+ T cells from the SP at day 17 post-infection. (E) Frequency of IFN-γ+, TNF-α+, and IL-2+ cells among CD80-acquired and CD80-deficient memory T cells obtained from SP after re-stimulation with GP33 peptide. Data are representative of 2 independent experiments (n=3−5 mice per group). Results represent mean±SEM and statistical significance was determined by 2-tailed unpaired Student's t-test.
NS, not significant; MFI, mean fluorescence intensity; Tmem, memory T.
***p<0.001.
DISCUSSION
In this study, we found that the CD80 levels were maintained by Ag-specific CD8+ T cells during the effector and memory phases. The maintenance of CD80 in the memory phase was caused by the extrinsic acquisition from the lymphoid organs. Previous reports have shown that the CD80 acquisition by human CD4+ T cells was observed during the early stages of activation (12). In agreement with this finding, we confirmed that CD80 was detected during the early stages of activation (Fig. 1B and C). However, to our knowledge, the present study is the first to demonstrate the maintenance of CD80 levels by Ag-specific CD8+ T cells in lymphoid organs after virus infection and for a long period, suggesting that CD80 extrinsically acquired by Ag-specific CD8+ T cells in lymphoid organs could define the characteristics of Ag-experienced CD8+ T cells. More importantly, we newly demonstrated that CD80 present on memory CD8+ T cells was involved in limiting their ability of expansion and IL-2 production, which suggested the in vivo physiological role of CD80 in the firing of recall responses by memory CD8+ T cells.
Importantly, the extrinsic acquisition of CD80 by T cells persisted at the later stages of infection after Ag clearance. One possible explanation is that memory CD8+ T cells interact more efficiently with APCs, such as DCs and macrophages, than naïve CD8+ T cells and thus, might acquire or trogocytose CD80 from APCs via the interaction period, even though these APCs do not load specific Ag peptides. Given that the Ag-independent DCs can interact with T cells at the immunological synapse and induce T cell activation (13), APCs that are physically close to memory T cells might be associated with maintaining high levels of CD80 on memory CD8+ T cells in an Ag-independent manner. Another explanation for CD80 longevity may be the different stabilities of intrinsic expression of CD80 and extrinsic acquisition of CD80 on T cells. While intrinsically expressed CD80 molecules progressively disappeared on Ag-specific CD8+ T cells during the effector T cells differentiation, which began at day 30 post-infection, extrinsically acquired CD80 molecules were maintained even after day 80 post-infection (Fig. 2C and D, Fig. 3E and F). The importance of CD80 trogocytosed by T cells can be suggested based on the previous reports. Firstly, the extrinsic acquisition of CD80 on T cells may be required for the induction and maintenance of Ag-specific memory T cells, since the differentiation and function of those T cells were impaired in primary immune response of CD80/CD86-KO mice (14,15) and in recall responses (15). As the previous study did not identify the levels of extrinsic acquisition of CD80 by T cells during recall response, it is not known exactly whether this effect is derived from the acquisition of CD80 and CD86 or other mechanisms. In the current study, we established the in vivo system to examine the role of extrinsically acquired CD80 by Ag-specific CD8+ T cells. CD80 acquisition during T cell differentiation did not seem to change the frequency and function of Ag-specific CD8+ T cells during their differentiation (Fig. 4). CD80 present on memory CD8+ T cells rather inhibited cell expansion and IL-2 production during recall response (Fig. 5). It should be noted that CD80, extrinsically acquired by CD8+ T cells, plays a role in regulating recall CD8+ T cell response, which is quite different from the immunological role of CD80 intrinsically expressed by APCs. Secondly, based on previous reports that the in vitro CD80 acquisition by T cells from APCs allows T cells to become APCs (6,12), T cells that acquired CD80 may act as APCs by allowing T cell-T cell interactions (16,17). Based on our in vivo effect of CD80, extrinsically acquired by CD8+ T, cells on their recall response (Fig. 5), the possible role of CD80 on CD8+ T cells does not seem to have a substantial impact on delivery of stimulation signals to overcome its regulatory function, at least during recall response. However, the dimension of the impact of CD80 acquired by CD8+ T cells in priming CD8+ T cells during the primary response should be more carefully reconsidered in vivo. Thirdly, CD80 molecules on T cells might inhibit T cell immune responses by interacting with PD-L1, because it has been reported that PD-L1 can deliver negative signals to CD8+ T cells via CD80 engagement (9). This possibility is also supported by some studies indicating that the interaction between PD-L1 and CD80 is required for the induction and maintenance of peripheral T cells tolerance, the regulation of memory T cells homeostasis, and the control of the effector T cells response via inducing apoptosis (18,19,20). Indeed, the current study clearly demonstrated that CD80, extrinsically acquired by Ag-specific CD8+ T cells, significantly inhibited Ag-dependent expansion of memory CD8+ T cells and their IL-2 production under in vivo re-exposure to the same Ag (Fig. 5), which supports the negative regulatory role of CD80 present on CD8+ T cells. According to a recent study showing that CD80 on APCs interact with PD-L1 on the same cells (21,22), CD80 acquired by CD8+ T cells might also interact with PD-L1 expressed on the same CD8+ T cells to activate a recall immune response.
Our data showed that CD80 molecules are extrinsically acquired by Ag-specific CD8+ T cells in the lymphoid organs and further maintained continuously. This continuous existence of CD80 on memory CD8+ T cells appeared to be important in promoting an appropriate recall immune response, dampening excessive CD8+ T cell recall responses. Our study provides information about a novel characteristic of Ag-experienced CD8+ T cells in lymphoid organs. More importantly, the physiological role of CD80 present on memory CD8+ T cells could be applied to generate optimal recall immune responses against repeated infection with pathogens.
ACKNOWLEDGEMENTS
This study was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT) (2015R1A2A1A10056084, 2017R1A5A1014560, 2018R1A2A1A05076997, 2018M3A9H3024850, 2019M3A9B6065221).
Abbreviations
- APC
Ag-presenting cell
- Arm
armstrong
- CL13
clone 13
- CTLA-4
cytotoxic T-lymphocyte-associated protein 4
- IACUC
Institutional Animal Care and Use Committee
- KO
knockout
- LCMV
lymphocytic choriomeningitis virus
- LG
lung
- LN
lymph node
- PFUs
plaque-forming units
- SP
spleen
- WT
wild type
Footnotes
Conflict of Interest: The authors declare no potential conflicts of interest.
- Conceptualization: Son J, Ha SJ.
- Formal analysis: Son J.
- Investigation: Son J.
- Methodology: Son J.
- Supervision: Ha SJ.
- Writing - original draft: Son J.
- Writing - review & editing: Ha SJ.
SUPPLEMENTARY MATERIAL
The extrinsic acquisition of CD80 does not affect the cytotoxicity of Ag-specific memory CD8+ T cells. The LG and SP were obtained from WT and CD80-KO mice at day 60 post-infection and re-stimulated with a GP33 peptide in vitro. Frequency and number of CD107a+ cells and expression levels of CD107a among donor CD80-KO P14 CD8+ T cells obtained from the LG and SP in WT or CD80-KO recipient mice. Data are representative of 3 independent experiments (n=3−4 mice per group). Results represent mean±SEM and statistical significance was determined by 2-tailed unpaired Student's t-test.
References
- 1.Kaech SM, Cui W. Transcriptional control of effector and memory CD8+ T cell differentiation. Nat Rev Immunol. 2012;12:749–761. doi: 10.1038/nri3307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Baaten BJ, Tinoco R, Chen AT, Bradley LM. Regulation of antigen-experienced T cells: lessons from the quintessential memory marker CD44. Front Immunol. 2012;3:23. doi: 10.3389/fimmu.2012.00023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.White JT, Cross EW, Kedl RM. Antigen-inexperienced memory CD8+ T cells: where they come from and why we need them. Nat Rev Immunol. 2017;17:391–400. doi: 10.1038/nri.2017.34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Riond J, Elhmouzi J, Hudrisier D, Gairin JE. Capture of membrane components via trogocytosis occurs in vivo during both dendritic cells and target cells encounter by CD8(+) T cells. Scand J Immunol. 2007;66:441–450. doi: 10.1111/j.1365-3083.2007.01996.x. [DOI] [PubMed] [Google Scholar]
- 5.Rosenits K, Keppler SJ, Vucikuja S, Aichele P. T cells acquire cell surface determinants of APC via in vivo trogocytosis during viral infections. Eur J Immunol. 2010;40:3450–3457. doi: 10.1002/eji.201040743. [DOI] [PubMed] [Google Scholar]
- 6.Sabzevari H, Kantor J, Jaigirdar A, Tagaya Y, Naramura M, Hodge J, Bernon J, Schlom J. Acquisition of CD80 (B7-1) by T cells. J Immunol. 2001;166:2505–2513. doi: 10.4049/jimmunol.166.4.2505. [DOI] [PubMed] [Google Scholar]
- 7.Sansom DM. CD28, CTLA-4 and their ligands: who does what and to whom? Immunology. 2000;101:169–177. doi: 10.1046/j.1365-2567.2000.00121.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Schneider H, Rudd CE. Diverse mechanisms regulate the surface expression of immunotherapeutic target ctla-4. Front Immunol. 2014;5:619. doi: 10.3389/fimmu.2014.00619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Butte MJ, Keir ME, Phamduy TB, Sharpe AH, Freeman GJ. Programmed death-1 ligand 1 interacts specifically with the B7-1 costimulatory molecule to inhibit T cell responses. Immunity. 2007;27:111–122. doi: 10.1016/j.immuni.2007.05.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Barber DL, Wherry EJ, Masopust D, Zhu B, Allison JP, Sharpe AH, Freeman GJ, Ahmed R. Restoring function in exhausted CD8 T cells during chronic viral infection. Nature. 2006;439:682–687. doi: 10.1038/nature04444. [DOI] [PubMed] [Google Scholar]
- 11.Murali-Krishna K, Altman JD, Suresh M, Sourdive DJ, Zajac AJ, Miller JD, Slansky J, Ahmed R. Counting antigen-specific CD8 T cells: a reevaluation of bystander activation during viral infection. Immunity. 1998;8:177–187. doi: 10.1016/s1074-7613(00)80470-7. [DOI] [PubMed] [Google Scholar]
- 12.Tatari-Calderone Z, Semnani RT, Nutman TB, Schlom J, Sabzevari H. Acquisition of CD80 by human T cells at early stages of activation: functional involvement of CD80 acquisition in T cell to T cell interaction. J Immunol. 2002;169:6162–6169. doi: 10.4049/jimmunol.169.11.6162. [DOI] [PubMed] [Google Scholar]
- 13.Revy P, Sospedra M, Barbour B, Trautmann A. Functional antigen-independent synapses formed between T cells and dendritic cells. Nat Immunol. 2001;2:925–931. doi: 10.1038/ni713. [DOI] [PubMed] [Google Scholar]
- 14.Grujic M, Bartholdy C, Remy M, Pinschewer DD, Christensen JP, Thomsen AR. The role of CD80/CD86 in generation and maintenance of functional virus-specific CD8+ T cells in mice infected with lymphocytic choriomeningitis virus. J Immunol. 2010;185:1730–1743. doi: 10.4049/jimmunol.0903894. [DOI] [PubMed] [Google Scholar]
- 15.Eberlein J, Davenport B, Nguyen TT, Victorino F, Sparwasser T, Homann D. Multiple layers of CD80/86-dependent costimulatory activity regulate primary, memory, and secondary lymphocytic choriomeningitis virus-specific T cell immunity. J Virol. 2012;86:1955–1970. doi: 10.1128/JVI.05949-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Helft J, Jacquet A, Joncker NT, Grandjean I, Dorothée G, Kissenpfennig A, Malissen B, Matzinger P, Lantz O. Antigen-specific T-T interactions regulate CD4 T-cell expansion. Blood. 2008;112:1249–1258. doi: 10.1182/blood-2007-09-114389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gérard A, Khan O, Beemiller P, Oswald E, Hu J, Matloubian M, Krummel MF. Secondary T cell-T cell synaptic interactions drive the differentiation of protective CD8+ T cells. Nat Immunol. 2013;14:356–363. doi: 10.1038/ni.2547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Park JJ, Omiya R, Matsumura Y, Sakoda Y, Kuramasu A, Augustine MM, Yao S, Tsushima F, Narazaki H, Anand S, et al. B7-H1/CD80 interaction is required for the induction and maintenance of peripheral T-cell tolerance. Blood. 2010;116:1291–1298. doi: 10.1182/blood-2010-01-265975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Mostböck S, Catalfamo M, Tagaya Y, Schlom J, Sabzevari H. Acquisition of antigen presentasome (APS), an MHC/costimulatory complex, is a checkpoint of memory T-cell homeostasis. Blood. 2007;109:2488–2495. doi: 10.1182/blood-2006-09-047290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Rollins MR, Gibbons Johnson RM. CD80 expressed by CD8+ T cells contributes to PD-L1-induced apoptosis of activated CD8+ T cells. J Immunol Res. 2017;2017:7659462. doi: 10.1155/2017/7659462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sugiura D, Maruhashi T, Okazaki IM, Shimizu K, Maeda TK, Takemoto T, Okazaki T. Restriction of PD-1 function by cis-PD-L1/CD80 interactions is required for optimal T cell responses. Science. 2019;364:558–566. doi: 10.1126/science.aav7062. [DOI] [PubMed] [Google Scholar]
- 22.Park HJ, Park JS, Jeong YH, Son J, Ban YH, Lee BH, Chen L, Chang J, Chung DH, Choi I, et al. PD-1 upregulated on regulatory T cells during chronic virus infection enhances the suppression of CD8+ T cell immune response via the interaction with PD-L1 expressed on CD8+ T cells. J Immunol. 2015;194:5801–5811. doi: 10.4049/jimmunol.1401936. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
The extrinsic acquisition of CD80 does not affect the cytotoxicity of Ag-specific memory CD8+ T cells. The LG and SP were obtained from WT and CD80-KO mice at day 60 post-infection and re-stimulated with a GP33 peptide in vitro. Frequency and number of CD107a+ cells and expression levels of CD107a among donor CD80-KO P14 CD8+ T cells obtained from the LG and SP in WT or CD80-KO recipient mice. Data are representative of 3 independent experiments (n=3−4 mice per group). Results represent mean±SEM and statistical significance was determined by 2-tailed unpaired Student's t-test.