Abstract
Percutaneous transluminal angioplasty (PTA) is a routine procedure for the treatment of peripheral arterial disease. However, its main limitation is late restenosis occurring at a 1-year rate of 6%–60%. Restenosis arises from injury to the arterial wall including overstretching, compression and rupture of the atherosclerotic plaque during balloon inflation. It is hypothesized that better long-term angioplasty results are observed if atherosclerotic plaques are removed rather than compressed and fractured. Laser angioplasty is one method to remove atherosclerotic plaques. We discuss the principles of lasers, physical properties of laser light, history of laser angioplasty and effects of laser radiation on tissues. Large clinical studies using laser angioplasty are critically assessed. In comparison to conventional PTA, there are some advantages of laser angioplasty: easier passage through chronic and calcified occlusions and according to some studies, better short- and medium-term results regarding limb salvage and management of in-stent restenoses. The main drawback of laser angioplasty is that current laser catheters are not able to create a sufficiently wide channel in the occlusion, meaning that adjunctive balloon dilatation is still required. Thus, long-term data may be misleading. Basic and applied research should continue to focus on enlargement of plaque ablation.
Percutaneous transluminal balloon angioplasty (PTA) is a routine and widely accepted method for the treatment of peripheral arterial disease (1). However, its main limitation is an increased risk of late restenosis/reocclusion, which usually develops 3–12 months after PTA. Restenosis rates after PTA vary from 6% to 60% at 1 year depending on the localization, type and length of the original lesion. The restenosis risk is higher for diffuse stenoses, longer occlusions and calcified lesions. It is also more frequent in femoropopliteal than in iliac arteries (1, 2).
The main cause of restenosis is thought to be injury to the arterial wall due to the balloon catheter (barotrauma), specifically overstretching, compression and rupture of the atherosclerotic plaque during balloon inflation. This local trauma results in the production of growth factors that stimulate the proliferation of endothelial and smooth muscle cells. This leads to the development of myointimal hyperplasia and arterial restenosis (3).
As late restenosis represents a permanent issue in the interventional management of occlusive disease, several other methods of angioplasty have been developed since the invention of the balloon catheter in 1973 (4). It is hypothesized that better long-term angioplasty results are achieved if the atherosclerotic plaques are removed rather than compressed and fractured. Nowadays, these methods are called “debulking methods”, including atherectomy, mechanical thrombectomy and laser angioplasty (5).
Laser angioplasty is based on a mechanism of tissue ablation involving the disobliteration of occluded or stenosed arteries using laser radiation.
Principles of lasers
The term “laser” is an acronym of “light amplification by stimulated emission of radiation”. Lasers consist of an excitation source (pump) and an optical resonator (a cavity or a chamber), with mirrors at each end. The optical resonator contains an active medium. The active medium defines the wavelength emitted by the laser, and gives the name to specific types of laser. This medium can be solid (e.g., neodymium yttrium aluminum garnet in Nd:YAG lasers, ruby crystal in ruby lasers), liquid (in dye lasers) or gas (argon ion in argon lasers, carbon dioxide in CO2 lasers, and dimer of a halogen and inert gas in excimer lasers). The excitation source can be a flash lamp, electric current or another laser. When a certain quantum of energy is pumped from the source into the resonator, the atoms of the active medium become excited. After returning to their ground state, they release energy in the form of photons of light. This is called spontaneous emission of radiation, from which only a small amount of light energy is gained. Lasers work on the principle of stimulated emission of radiation, where photons within the optical resonator are reflected between mirrors and collide with atoms of the active medium, which causes them to reach an excited state. Much more light energy in the form of photons is gained from this method than by spontaneous emission. One of the mirrors of the optical resonator is partially transparent, allowing the photons to exit the cavity.
In contrast to nonlaser light, laser light has three unique characteristics: monochromacity, coherence, and collimation. Monochromacity is the emission of light of only one wavelength, or a narrow band of wavelengths. The coherence of laser light is based on stimulated emission, meaning that the light waves are parallel. Collimation can be defined as a narrow beam diameter with no divergence. Hence, laser light does not lose energy, even after covering a large distance.
Lasers can operate in either a continuous mode or in a pulsed mode, which generates short pulses (microsecond, nanosecond, or picosecond duration).
Lasers emit light with very high intensity. In medicine, they can be used for various applications, including cutting, coagulation and ablation of tissues, among others (6–8).
Historical remarks
In 1917, Einstein formulated a theory of stimulated emission of radiation (9). However, the first practical laser was not constructed until 1960, when Maiman demonstrated a laser with a ruby which was stimulated by a flash lamp. This laser emitted a deep red color with a wavelength of 694 nm in a pulsed mode (10).
In 1963, Mc Guff used a pulsed ruby laser for the experimental ablation of atherosclerotic plaques in postmortem human and canine specimens (11). A significant advance in laser angioplasty came in the 1980s, when optical fibers that could transmit laser radiation from the laser source into the artery were developed. In 1983, for the first time clinically, Ginsburg performed laser angioplasty in a patient with severe stenosis of the deep femoral artery and a threatened limb. He used an argon laser with an optical fiber, which was introduced into the inflated balloon catheter enabling coaxial positioning of the optical fiber (12).
In the same year, Fournial and Choy completed perioperative coronary laser angioplasty in 10 patients using an argon laser. This technique was used during bypass surgery in five patients; the other five patients underwent laser therapy alone without bypass grafting. The results were not favorable, as 88% of the treated arteries occluded within three weeks (13). In 1985, a similar procedure was accomplished in the carotid arteries of two patients by Lammer and Choy (14).
At the beginning of laser angioplasty, continuous lasers were more commonly used. These lasers were taken from different fields of medicine where heat production was required, such as for tissue coagulation in surgery, for the management of bleeding ulcers in gastroenterology, and for treating retinal detachment in ophthalmology. For angioplasty, argon lasers and continuous Nd:YAG lasers were important during this period (6, 12–14). However, when ablating atherosclerotic plaques in the arteries, thermal interaction with the tissues is not desirable. This led to an era of intensive research and exploration of pulsed lasers due to their limited thermal effect, which will be discussed later. Initially, a pulsed Nd:YAG laser was used (15–19). The excimer laser (the term excimer is explained later in the text) was launched in clinical practice in the 1990s, when optical fibers for the transmission of radiation from the excimer laser became available (20–23). In USA, the Food and Drug Administration (FDA) has approved the use of excimer laser in patients with peripheral arterial disease only in 2003 (24).
Laser-tissue interactions
Laser radiation absorbed in a tissue has three mechanisms of action: (i) photothermal, (ii) photochemical, and (iii) acoustic mechanical (7).
i) Photothermal effects
Absorbed laser radiation is converted into thermal energy. Temperatures above 60°C lead to the denaturation of proteins and coagulation of the tissue. At temperatures over 100°C, exceeding the boiling point of intra- and extracellular water, vaporization occurs, causing tissue ablation. Photothermal effects are common for continuous lasers. In vivo, thermal injury can cause thrombosis and perforations of arteries. Therefore, continuous lasers are no longer utilized for angioplasty.
ii) Photochemical effects
Photochemical effects are nonthermal. They disintegrate tissues at the molecular level due to the breaking of molecular bonds and atom ionization. This can occur as a result of high energy photons of ultraviolet (UV) radiation from an excimer laser.
iii) Acoustic mechanical
Acoustic mechanical effects are also nonthermal. They arise from pulsed lasers, as irradiation with short, high-energy pulses leads to the local formation of plasma, causing a shock wave. This shock wave induces limited tissue ablation with no thermal damage (6–8, 17, 20, 22).
At present, in peripheral and coronary laser angioplasty, only excimer lasers are used. Its name originates from a combination of “excited” and “dimer”. Its active lasing medium is a molecule consisting of a halogen and inert (rare, noble) gas (such as argon, krypton or xenon). Excimer lasers (“excimers”) are a very powerful source of UV energy, which are very well absorbed by tissue proteins and lipids. The mechanism of action of excimers is nonthermal (“cool lasers”), thus, they cause precise ablation of tissues without thermal damage. Depending on the composition of the lasing medium, emission of various wavelengths of UV light can be achieved, for example, 193 nm for argon fluoride (ArF), 249 nm for krypton fluoride (KrF) and 350 nm for xenon fluoride (XeF) (20–23).
For angioplasty, the most frequently used excimer is the xenon chloride (XeCl) excimer, which operates at a wavelength of 308 nm with relatively long pulses (pulse duration of 135 ns). The long pulse length is required for successful delivery of UV light by silica fiberoptics. Excimer laser energy is strongly absorbed by iodine contrast media and hemoglobin; therefore, a saline infusion must be administered while using the laser to replace blood and contrast (21–23).
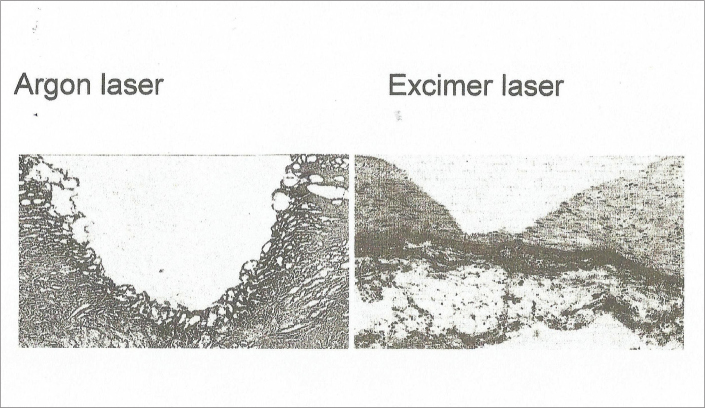
Fig. 1 demonstrates thermal and nonthermal effects of two lasers on vascular tissue.
Figure 1.
Effects of different lasers on human postmortem atherosclerotic coronary arteries: light microscopic findings (own experiments, hematoxylin and eosin staining). Left: a crater after irradiation by argon laser (energy 30 J, beam transmitted through a 250 μm core diameter quartz optical fiber) reveals thermal injury. A superficial zone of coagulation necrosis and a subjacent zone of polymorphous lacunae are present (magnification ×150). Right: After excimer laser irradiation (wavelength 193 nm, energy 100 mJ/pulse, 15 ns pulse duration, 1 Hz repetition rate, energy density 1.5 J cm−2, 250 pulses applied, radiation focused by means of a lens), no thermal damage is evident. The surface of the crater has sharp edges and tissue architecture is preserved (magnification ×120).
Laser system
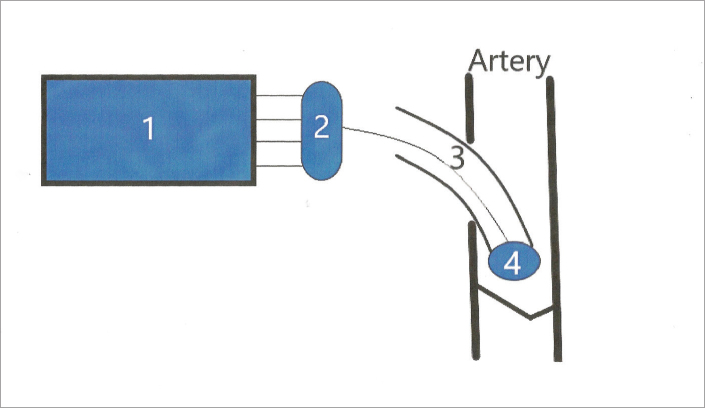
For the application of lasers in the field of laser angioplasty, it is important to consider not only the laser itself, but also the whole laser system. This system consists of a laser, a connector, a laser catheter, and a catheter tip (Fig. 2).
Figure 2.
Laser system consists of a laser (1), a connector (2), a laser catheter with optical fiber(s) transmitting the laser energy into the artery (3) and a catheter tip (4).
The laser was discussed above. The connector acts as a guide to direct the laser light into the laser catheter. The laser catheter is composed of one or more optical fibers, which transmit the laser energy. The catheter tip is a very important component of the laser system, and may be crucial for the success of the laser procedure (25–27). Bare optical fibers were used when laser angioplasty procedures were first developed (12); however, they created only a narrow channel in the occlusion. In addition, the sharp ending of the fiber could perforate the artery, particularly in a tortuous vascular bed. Thus, several different catheter tips have been developed. Aside from their atraumatic shape, they allow the dispersion of radiation and create a wider channel during recanalization of an obstruction. It is worth mentioning the important roles of sapphire contact probes, tips, lens tips and metal contact probes (“hot tips”) in the history of laser angioplasty (15, 16, 19, 25–27).
For excimer laser angioplasty (ELA), a multifiber catheter is used. This catheter contains a bundle of concentrically arranged small optical fibers (50–100 μm in diameter) surrounding the central guidewire lumen. At the end of the fibers is an optical shield. The optical shield is oriented to disperse radiation at the output from the catheter. Should the device work without an optical shield, only limited ablation of the occlusion is obtained, with no ablation between the fibers, a so-called “Swiss cheese effect”. The diameter of the broadest commercially available multifiber catheter is 2.5 mm (21–25).
The major limitation of current multifiber catheters lies in the fact that catheters cannot create a channel in an obstruction that is wider than the diameter of the catheter itself. Hence, laser angioplasty alone can only be performed in coronary arteries. In iliac and femoropopliteal arteries, the recanalized channel is not wide enough, and an additional balloon angioplasty is needed. This may distort long-term outcomes, in particular. To enable the use of multifiber catheters in femoropopliteal arteries, some modifications have been made to the multifiber catheter. One of these modified versions, the Turbo-Power catheter (Spectranetics), involves a guidewire lumen positioned eccentrically, and the catheter rotates around the guidewire by means of an external electric motor. In the Turbo-Tandem catheter (Spectranetics), the multifiber laser catheter is placed within a special guide catheter. The angled ramp at the tip of the guide catheter provides circumferential guidance and positioning of the laser catheter, allowing enlargement of the recanalized channel (21, 23, 28).
Clinical studies
Randomized studies comparing the results of laser angioplasty using continuous wave lasers followed by PTA (“laser-assisted PTA”) to standard PTA alone for femoropopliteal occlusions did not show any significant differences in longer-term patency between the two methods (25–27).
Newer studies only use excimer lasers, referred to as ELA. Sometimes the term CLiRPATH (cool laser revascularization for peripheral artery therapy) is also used.
In a study conducted by Scheinert et al. (29), 411 chronic occlusions of the superficial femoral artery, with an average length of 19.4±6 cm, were managed using ELA with adjunctive PTA or stenting. The technical success rate was reported to be 90.5%. Complications included acute reocclusion (1.0%), perforation (2.2%), and distal embolization (3.9%). However, the primary patency was only 33.6% at 1 year. The 1-year primary assisted and secondary patency rates were 65.1% and 75.9%, respectively.
Steinkamp et al. (30) published a study involving 215 patients with popliteal artery occlusion, with an average length of 10.4 cm. This prospective nonrandomized study compared ELA with subsequent PTA (127 patients) with PTA alone (88 patients). After a mean follow-up of 36 months (range, 6–52 months) the primary and secondary patency rates were 21.7% and 50.8%, respectively, in patients who underwent ELA plus PTA, and 16.3% and 35.2%, respectively, in the PTA group. No statistically significant differences were found between groups.
The multicenter prospective registry CELLO (CliRPATH excimer laser system to enlarge lumen openings) included 65 patients; 52 patients with femoropopliteal stenosis >70% and 13 patients with femoropopliteal occlusion. The primary endpoint was a reduction in the diameter of the stenosis, measured by ultrasound following laser ablation using a Turbo-Tandem catheter prior to any adjunctive therapy. At baseline (before ELA), the stenosis was 77%±15%, which was reduced to 34.7%±17.8% after ELA. Adjunctive PTA was performed in 64.6%, PTA and stenting performed in 23.1%. Of the patients in the trial, 12.3% did not receive any subsequent treatment. Patency rates (restenosis <50%) were 59% and 54% at 6 and 12 months, respectively (31).
In the prospective multicenter LACI (laser angioplasty for critical limb ischemia) study, 155 limbs at high risk for amputation were treated. The patients were poor candidates for surgery due to cardiorespiratory comorbidities. In total, 426 femoropopliteal and infrapopliteal lesions were intervened. After ELA, PTA was performed in all cases, and a stent was implanted in 45% of limbs. In 8%, it was not possible to cross the lesion with the guidewire, so a combination of laser radiation and shifting of the guidewire had to be carried out (“step by step” technique, described below). Procedural success, defined as <50% residual stenosis in all treated lesions, was accomplished in 86% of limbs. At 6 months follow-up, limb salvage was achieved in 92% (32). Favorable outcomes concerning limb salvage using ELA and PTA were confirmed by the Belgian version of the LACI study, in which 51 critically ischemic limbs were intervened with a salvage rate of 90.5% at 6 months (33).
The EXCITE ISR (excimer laser randomized controlled study for treatment of femoropopliteal in-stent restenosis) was the first large, prospective, randomized multicenter study comparing ELA plus PTA and PTA alone. It analyzed 250 patients with femoropopliteal in-stent restenosis. Patients were enrolled and randomly divided into two groups, ELA plus PTA and PTA alone, in a 2:1 ratio. The mean lesion lengths were 19.6±12.0 and 19.3±11.9 cm in the ELA plus PTA and PTA groups, respectively. The ELA plus PTA group demonstrated a superior and significant procedural success rate of 93.5% versus 82.7% obtained for PTA alone (P = 0.01), with significantly fewer procedural complications. The 6-month freedom from target lesion revascularization (TLR) was significantly higher in the ELA plus PTA group at 73.5% compared with 51.8% for the PTA group (P < 0.005), and 30-day major adverse event rates were 5.8% versus 20.5% (P < 0.001), respectively. ELA plus PTA was associated with a 52% reduction in TLR (hazard ratio, 0.48; 95% confidence interval, 0.31–0.74) (34).
Conclusion
In order to critically assess the impact of peripheral laser angioplasty in the percutaneous treatment of peripheral arterial disease, its benefits and drawbacks must be discussed.
The benefits of peripheral laser angioplasty are listed below:
Laser angioplasty is a safe method, as the complication rate does not exceed that of standard PTA. Perforation of the arteries in ELA is relatively rare, only occurring in up to 2.2% of cases (29, 32). Distal embolization ranges from 2.5% to 9% in larger studies (34–36).
An advantage of laser angioplasty is easier passage through chronic and calcified occlusions. According to the literature, crossing such obstructions with a conventional guidewire may fail in 8%–34% of all occlusions (32, 37). In ELA, a “step by step” technique is often used in these cases, whereby laser radiation is applied simultaneously with shifting of the guidewire in the occlusion. Laser catheters with a central lumen for a guidewire advance are also more likely endoluminally than subintimally (22, 38).
In patients with critical limb ischemia, some larger studies have reported positive results for avoiding amputation by means of the ELA plus PTA technique (32, 33). However, these studies were not randomized to standard PTA or stenting, and the follow-up period was relatively short (6 months).
Regarding femoropopliteal in-stent restenoses, a randomized study and meta-analysis found out better short- and medium-term outcomes for ELA plus PTA than for PTA alone (34, 39).
The drawbacks of peripheral laser angioplasty are listed below:
Current multifiber laser catheters (even their novel modifications) cannot create a wide enough channel in the occluded artery, which requires subsequent balloon dilatation. Hence, long-term data for laser angioplasty may be influenced by adjunctive balloon dilatation (21, 23, 28, 29–34).
Peripheral laser angioplasty has a higher cost when compared with plain PTA/stenting (38).
Table highlights the benefits and drawbacks of excimer laser angioplasty.
Table.
Overview of the benefits and drawbacks of ELA
| Benefits | ELA is as safe as standard PTA (29, 32, 34–36) |
| Probably easier passage through chronic and calcified occlusions in ELA than with a conventional guidewire (22, 32, 37, 38) | |
| Positive results for avoiding amputations in patients with critical limb ischemia (Note that randomized studies with standard PTA are missing) (32, 33) | |
| Better short- and medium-term outcomes in femoropopliteal in-stent restenoses than in standard PTA (randomized study and meta-analysis) (34, 39) | |
|
| |
| Drawbacks | Need for adjunctive balloon dilatation in most cases due to a narrow channel created by current laser catheters (21, 23, 28, 29–34) |
| Higher cost than in standard PTA/stenting (38) | |
ELA, excimer laser angioplasty; PTA, percutaneous transluminal balloon angioplasty.
Despite the abovementioned shortcomings, laser angioplasty represents an appealing concept for angioplasty, as it facilitates the removal of atherosclerotic plaques, which may be important for preventing restenosis/reocclusion after angioplasty. In our opinion, basic and applied research should continue, mainly with respect to enlargement of plaque ablation.
We also believe that laser equipment should be present in larger angioplasty centers with the aim to provide complex percutaneous management of peripheral arterial disease.
Main points.
Late restenosis represents the main limitation of PTA of peripheral arteries. It arises from injury to the arterial wall caused by the balloon inflation, specifically overstretching, compression, and rupture of the atherosclerotic plaque. This trauma results in the production of growth factors that stimulate the proliferation of endothelial and smooth muscle cells. This leads to the development of myointimal hyperplasia and arterial restenosis.
It is believed that better long-term angioplasty results are achieved if atherosclerotic plaques are removed rather than compressed and fractured. Laser angioplasty based on a mechanism of tissue ablation using laser radiation is one of such methods and belongs to so called “debulking methods”.
Only excimer lasers with different types of multifiber catheters are used in recent clinical practice, referred to as excimer laser angioplasty (ELA). ELA is a safe method with an easier passage through chronic and calcified occlusions than with conventional guidewire. According to some studies, better short- and medium-term results regarding limb salvage and management of in-stent restenoses are observed in ELA than in standard PTA. The main drawback of ELA is that current laser catheters are not able to create a sufficiently wide channel in the occlusion, thus adjunctive balloon dilatation is still needed.
Footnotes
Conflict of interest disclosure
The author declared no conflicts of interest.
References
- 1.Norgren L, Hiatt WR, Dormandy JA, Nehler MR, Harris KA, Fowkes FG. Inter-society consensus for management of peripheral arterial disease (TASC II) J Vascular Surg. 2007;45(Suppl S):5–67. doi: 10.1016/j.jvs.2006.12.037. [DOI] [PubMed] [Google Scholar]
- 2.Schillinger M, Minar E. Percutaneous treatment of peripheral artery disease. Novel techniques. Circulation. 2012;126:2433–2440. doi: 10.1161/CIRCULATIONAHA.111.036574. [DOI] [PubMed] [Google Scholar]
- 3.Neville RF, Sidawy AN. Myointimal hyperplasia: basic science and clinical considerations. Semin Vasc Surg. 1998;11:142–148. [PubMed] [Google Scholar]
- 4.Grüntzig AR, Hopff H. Rekanalisation chronischer arterieller Verschlüsse mit einem neuen Dilatationskatheter: Modifikation der Dotter-Technik. Dtsch Med Wochenschr. 1974;99:2502–2505. doi: 10.1055/s-0028-1108161. [DOI] [PubMed] [Google Scholar]
- 5.Katsanos K, Spiliopoulos S, Reppas L, Karnabatidis D. Debulking atherectomy in peripheral arteries: Is there a role and what is the evidence? Cardiovasc Intervent Radiol. 2017;40:964–977. doi: 10.1007/s00270-017-1649-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Grundfest WS, Litvack F, Hickey A, et al. The current status of angioscopy and laser angioplasty. J Vasc Surg. 1987;5:667–673. doi: 10.1067/mva.1987.avs0050667. [DOI] [PubMed] [Google Scholar]
- 7.Allemann IB, Kaufman J. Laser principles. In: Alemann IB, Goldberg DJ, editors. Basics in dermatological laser applications. Basel: Karger; 2011. pp. 7–23. [DOI] [Google Scholar]
- 8.Thomas G, Isaacs R. Basic principles of lasers. Anaesth Intens Care Med. 2011;12:574–577. doi: 10.1016/j.mpaic.2011.09.013. [DOI] [Google Scholar]
- 9.Einstein A. Zur Quantentheorie der Strahlung. Physikalische Zeitschrift. 1917;18:121–128. [Google Scholar]
- 10.Maiman TH. Stimulated optical radiation in ruby. Nature. 1960;187:493–494. doi: 10.1038/187493a0. [DOI] [Google Scholar]
- 11.Mc Guff PE, Bushnell D, Soroff HS. Studies of the surgical applications of laser. Surg Forum. 1963;14:143–145. [PubMed] [Google Scholar]
- 12.Ginsburg R, Kim DS, Guthaner D, Toth J, Mitchel RS. Salvage of an ischemic limb by laser angioplasty: Description of a new technique. Clin Cardiol. 1984;7:54–58. doi: 10.1002/clc.4960070112. [DOI] [PubMed] [Google Scholar]
- 13.Fournial G, Choy D, Marco J, et al. Desobstruction des arteres coronaires par faisceau laser. Arch Mal Coeur Vaiss. 1985;78:1061–1065. [PubMed] [Google Scholar]
- 14.Lammer J, Ascher PW, Choy DS. Transfemorale Katheter-Laser-Thrombendartektomie der Arteria carotis. Dtsch Med Wochenschr. 1986;111:607–610. doi: 10.1055/s-2008-1068498. [DOI] [PubMed] [Google Scholar]
- 15.Kvasnicka J, Stanek F, Boudik F, et al. Percutaneous peripheral laser angioplasty with a pulsed Nd-YAG laser and sapphire tips. Lasers Med Sci. 1991;6:43–48. doi: 10.1007/BF02042645. [DOI] [Google Scholar]
- 16.Kvasnicka J, Boudik F, Stanek F, et al. Percutaneous laser angioplasty with a pulsed Nd:YAG laser. Initial clinical experience and early follow-up. Int Angiol. 1991;10:29–33. [PubMed] [Google Scholar]
- 17.Cross FW, Mills TN, Bown SG. Pulsed Nd-YAG laser effects on normal and atheromatous aorta. Lasers Med Sci. 1987;3:193–211. [Google Scholar]
- 18.Stanek F, Kvasnicka J, Boudik F, Kubecek V, Vitkova I, Hamal K. In: Weidelich W, Weidelich R, editors. Pulsed Nd:YAG-laser angioplasty of peripheral arteries - experiments in cadavers; Proceedings of the 9th International Conference Laser 89; 1989; Munich: Springer-Verlag Berlin; 1990. pp. 226–229. [DOI] [Google Scholar]
- 19.Stanek F, Kvasnicka J, Boudik F, et al. Periphere Laserangioplastie mit dem gepulsten Nd:YAG Laser. Radiol Diagn. 1991;32:355–358. [Google Scholar]
- 20.Isner JM, Donaldson RF, Deckelbaum LI, et al. The excimer laser: gross, light microscopic and ultrastructural analysis of potential advantages for use in laser therapy of cardiovascular disease. J Am Coll Cardiol. 1985;6:1102–1109. doi: 10.1016/S0735-1097(85)80316-8. [DOI] [PubMed] [Google Scholar]
- 21.Taylor KD, Reiser C. Next generation catheters for excimer laser coronary angioplasty. Lasers Med Sci. 2001;16:133–140. doi: 10.1007/PL00011343. [DOI] [PubMed] [Google Scholar]
- 22.Biamino G. The excimer laser: science fiction fantasy or practical tool? J Endovasc Ther. 2004;11(Suppl 2):207–222. doi: 10.1583/04-1394.1. [DOI] [PubMed] [Google Scholar]
- 23.Taylor KD, Reiser C. From laser physics to clinical utilization: Design and properties of cardiovascular laser catheters. In: Topaz O, editor. Lasers in cardiovascular interventions. London: Springer; 2015. pp. 1–14. [DOI] [Google Scholar]
- 24.Laird JR, Reiser C, Biamino G, Zeller T. Excimer laser assisted angioplasty for the treatment of critical limb ischemia. J Cardiovasc Surg. 2004;45:239–248. [PubMed] [Google Scholar]
- 25.Lammer J. Laser angioplasty of peripheral arteries: an epilogue? Cardiovasc Intervent Radiol. 1995;18:1–8. doi: 10.1007/BF02807347. [DOI] [PubMed] [Google Scholar]
- 26.Belli AM, Cumberland DC, Procter AE, Welsh CL. Follow-up of conventional angioplasty versus laser thermal angioplasty for total femoropopliteal artery occlusions: results of a randomized trial. J Vasc Interv Radiol. 1991;2:485–488. doi: 10.1016/S1051-0443(91)72229-X. [DOI] [PubMed] [Google Scholar]
- 27.Lammer J, Pilger E, Decrinis M, Quehenberger F, Klein GE, Stark G. Pulsed excimer laser versus continuous-wave Nd: YAG laser versus conventional angioplasty of peripheral arterial occlusions: prospective, controlled, randomised trial. Lancet. 1992;340:1183–1186. doi: 10.1016/0140-6736(92)92891-I. [DOI] [PubMed] [Google Scholar]
- 28.Micari A, Vadala G, Biamino G. Update on the TURBO BOOSTER spectranetics laser for lower extremity occlusive disease. J Cardiovasc Surg. 2010;51:233–243. [PubMed] [Google Scholar]
- 29.Scheinert D, Laird JR, Schröder M, Steinkamp H, Balzer JO, Biamino G. Excimer laser-assisted recanalization of long, chronic superficial femoral artery occlusions. J Endovasc Ther. 2001;8:156–166. doi: 10.1177/152660280100800210. [DOI] [PubMed] [Google Scholar]
- 30.Steinkamp HJ, Rademaker J, Wissgott C, et al. Percutaneous transluminal laser angioplasty versus balloon dilation for treatment of popliteal artery occlusions. J Endovasc Ther. 2002;9:882–888. doi: 10.1177/152660280200900623. [DOI] [PubMed] [Google Scholar]
- 31.Dave RM, Patlola R, Kollmeyer K, et al. Excimer laser recanalization of femoropopliteal lesions and 1-year patency: results of the CELLO registry. J Endovasc Ther. 2009;16:665–675. doi: 10.1583/09-2781.1. [DOI] [PubMed] [Google Scholar]
- 32.Laird JR, Zeller T, Gray BH, et al. Limb salvage following laser-assisted angioplasty for critical limb ischemia: results of the LACI multicenter trial. J Endovasc Ther. 2006;13:1–11. doi: 10.1583/05-1754.1. [DOI] [PubMed] [Google Scholar]
- 33.Bosiers M, Peeters P, Elst FV, et al. Excimer laser assisted angioplasty for critical limb ischemia: results of the LACI Belgium Study. Eur J Vasc Endovasc Surg. 2005;29:613–619. doi: 10.1016/j.ejvs.2005.01.008. [DOI] [PubMed] [Google Scholar]
- 34.Dippel EJ, Makam P, Kovach R, et al. Randomized controlled study of excimer laser atherectomy for treatment of femoropopliteal in-stent restenosis: initial results from the EXCITE ISR trial (EXCImer Laser Randomized Controlled Study for Treatment of FemoropopliTEal In-Stent Restenosis) JACC Cardiovasc Interv. 2015;8:92–101. doi: 10.1016/j.jcin.2014.09.009. [DOI] [PubMed] [Google Scholar]
- 35.Shammas NW, Shammas GA, Arikat L, et al. Five-year freedom from target-lesion revascularization using excimer laser ablation therapy in the treatment of in-stent restenosis of femoropopliteal arteries. J Invasive Cardiol. 2017;29:207–208. [PubMed] [Google Scholar]
- 36.Jayet J, Coscas R, Heim F, et al. Laser uses in non-coronary arterial disease. Ann Vasc Surg. 2019;57:229–237. doi: 10.1016/j.avsg.2018.06.010. [DOI] [PubMed] [Google Scholar]
- 37.Charalambous N, Schäfer PJ, Trentmann J, et al. Percutaneous intraluminal recanalization of long, chronic superficial femoral and popliteal occlusions using the Frontrunner XP CTO device: a single-center experience. Cardiovasc Intervent Radiol. 2010;33:25–33. doi: 10.1007/s00270-009-9700-x. [DOI] [PubMed] [Google Scholar]
- 38.Serino F, Cao Y, Renzi C, et al. Excimer laser ablation in the treatment of total chronic obstructions in critical limb ischaemia in diabetic patients. Sustained efficacy of plaque recanalisation in mid-term results. Eur J Vasc Endovasc Surg. 2010;39:234–238. doi: 10.1016/j.ejvs.2009.10.018. [DOI] [PubMed] [Google Scholar]
- 39.Hajibandeh S, Hajibandeh S, Antoniou SA, Torella F, Antoniou GA. Treatment strategies for in-stent restenosis in peripheral arterial disease: a systematic review. Interact CardioVasc Thorac Surg. 2019;28:253–261. doi: 10.1093/icvts/ivy233. [DOI] [PubMed] [Google Scholar]