Abstract
When humans manipulate objects, the sensorimotor system coordinates three-dimensional forces to optimize and maintain grasp stability. To do this, the CNS requires precise information about the magnitude and direction of load force (tangential to skin surface) plus feedback about grip force (normal to skin). Previous studies have shown that there is rapid, precise coordination between grip and load forces that deteriorates with digital nerve block. Obviously, mechanoreceptive afferents innervating fingerpad skin contribute essential information. We quantify human capacity to scale tangential and normal forces using only cutaneous information. Our paradigm simulated natural manipulations (a force tangential to the skin superimposed on an indenting force normal to the skin). Precisely controlled forces were applied by a custom-built stimulator to an immobilized fingerpad. Using magnitude estimation, subjects (n = 8) scaled the magnitude of tangential force (0.25–2.8 N) in two experiments (normal force, 2.5 and 4 N, respectively). Performance was unaffected by normal force magnitude and tangential force direction. Moreover, when both normal (2–4 N) and tangential forces were varied in a randomized-block factorial design, the relationship between applied and perceived tangential force remained near linear, with a minor but statistically significant nonlinearity. Our subjects could also discriminate small differences in tangential force, and this was the case for two different reference stimuli. In both cases, the Weber fraction was 0.16. Finally, scaling functions for magnitude estimates of normal force (1–5 N) were also approximately linear. These data show that the cutaneous afferents provide a wealth of precise information about both normal and tangential force.
Keywords: sensorimotor control, grip force, grasp stability, tactile resolution, mechanoreceptive afferents, psychophysics
Introduction
When humans manipulate objects, the sensorimotor system coordinates the three-dimensional forces applied by the fingers to optimize the grasp (Johansson and Westling, 1984; Reilmann et al., 2001; Gysin et al., 2003). Strategies for successful manipulation necessitate adopting forces that are sufficiently large to prevent slips but are not excessive, so that fatigue is minimized and delicate objects are not damaged; the forces that are typically required by most tasks have magnitudes that are of the order of a few Newtons. It is also necessary to use force magnitudes and directions that suit the shape of the object and the load conditions (Flanagan and Wing, 1995; Cadoret and Smith, 1996; Jenmalm and Johansson, 1997; Goodwin et al., 1998). Such fingertip forces can be resolved into two components: normal forces acting orthogonally to the skin surface and tangential forces acting in the plane of the skin surface. These are commonly referred to as grip forces and load forces, respectively.
There is abundant evidence that the sensorimotor system controlling hand movements must have access to precise information about the grip and load forces (Moberg, 1975; Rothwell et al., 1982; Hager-Ross et al., 1996; Cole et al., 2003). There is also evidence that a significant component of this information arises from the populations of cutaneous afferents, which innervate low-threshold mechanoreceptors in the glabrous skin of the digits, because digital nerve block by application of a local anesthetic compromises precision manipulation (Johansson and Westling, 1984; Jenmalm and Johansson, 1997; Jenmalm et al., 2000; Augurelle et al., 2003; Monzee et al., 2003). Furthermore, it is clear that perception and cognition during haptic exploration of objects require conscious evaluation of fingertip forces (Rinkenauer et al., 1999; Robles-De-La-Torre and Hayward, 2001; Smith et al., 2002).
Despite the critical nature of hand function in human life, there is a paucity of precise information about the human capacity to perceive normal and tangential forces in the range normally used for manipulating objects in everyday life. There is a limited amount of data on the perception of normal forces, but only for forces up to ∼0.7 N (Goodwin and Wheat, 1992). Information about our ability to perceive tangential force is sketchy (Pare et al., 2002).
In the experiments reported here, our aim was to quantify or directly measure the human capacity to scale normal forces and to scale and discriminate tangential forces in the ranges used in typical manipulations. Using a computer-controlled stimulator, the forces were applied passively to the immobilized index fingers of human subjects. This allowed us to simulate natural force conditions but to control them precisely. Moreover, the vast majority of information available to the subjects was confined to that originating from the cutaneous afferents innervating the finger.
Materials and Methods
Stimulator. The stimulus (Fig. 1) consisted of a small probe (24 mm in diameter) with a flat contact surface covered with fine sandpaper (500 grade). A custom-built stimulator, controlled by computer using Lab-view software (National Instruments, Austin, TX), allowed simultaneous application of normal and tangential forces to the fingerpad. Torque motor 1 generated the tangential force, the direction of which could be varied in 15° increments via the indexed hub. This motor was mounted on a balanced beam, which was attached to a second torque motor. Torque motor 2, which generated a normal force on the finger, was coupled to a damper. Before the commencement of an experimental trial, the surface was positioned just above and tangential to the skin, and the damper setting was such that contact was smooth and critically damped. A six axis force–torque transducer (Nano FT; ATI Industrial Automation, Apex, NC) measured the three-dimensional forces and torques applied to the fingerpad with a resolution of 0.0125 N and 0.0625 mNm, respectively. The transducer, located between the contact surface and the arm of the stimulator, is shown enlarged in Figure 1 (left).
Figure 1.
Computer-controlled stimulator. Torque motor 2 (m2), coupled to damper (d), produced a vertical (normal) indenting force on the subject's finger (f), which was immobilized in plasticine (p). Torque motor 1 (m1), attached to m2 via beam (b), produced a tangential force on the finger. The direction of the tangential force in the horizontal plane was set by the hub (h), indexed at 15° increments. The flat surface (s), covered with 500 grade sandpaper, was coupled to m1via a six axis force–torque transducer (t) (an enlargement of this area is shown on the left). The stimulator was mounted on an x–y–z vernier shift to allow accurate positioning in all three dimensions.
General procedure. The same eight subjects, ranging in age from 20 to 25 years, took part in all experiments, each of which complied with the policies on the use of animals and humans in neuroscience research, approved by the Society for Neuroscience, and were approved by the University of Melbourne Human Research Ethics Committee. To ensure consistency of procedures, all subjects were given written instructions, and any subsequent information was read by the experimenter from an instruction sheet with an accompanying checklist. The subject was seated comfortably with the forearm supinated and the index finger of the dominant hand secured in a plasticine finger mold by a small brass post that was temporarily attached to the fingernail with a drop of cyanoacrylate glue (removed with acetone at the end of each session). A curtain prevented the subjects from seeing either the stimulator or their finger. The stimulus in each experiment was applied to the distal portion of the fingerpad and was presented in this same position for all trials.
The stimulus was presented to the skin of the fingerpad with a defined matrix of forces that were combinations of normal and tangential forces. The relationship between tangential and normal forces was selected so that their ratio was less than the previously measured coefficient of friction by a margin sufficient to ensure that there was no slip. Force traces were examined off-line to ensure that there was no slip between the stimulus and the finger and also to determine whether there were any irregularities in force delivery. Normal (indenting) and tangential (load) forces were in the range used by humans during normal manipulation (a few Newtons); thus there was no risk of damage or discomfort. At the commencement of each trial, the stimulus surface was positioned just above and tangential to the surface of the skin, but there was no contact with the skin before the commencement of stimulus presentation.
The experiments were conducted in the order set out below (i.e., normal force scaling first and the discrimination experiment last). Data collection for each experiment was completed before proceeding to the next series. Training took place at the commencement of each series, so that each subject underwent a separate period of training for each of the scaling and discrimination studies described below. For scaling experiments, subjects used magnitude estimation. Data collection for each task commenced once the subject's performance had stabilized. For scaling, one session was usually sufficient to reach stable performance, but more sessions were conducted if necessary. For the discrimination experiments, two to three training sessions were required. No session (training or experimental) lasted >1 hr, and only one session was conducted in a single day.
Normal force scaling. Five levels of normal force (1, 2, 3, 4, and 5 N; tangential force, 0) were presented seven times each in random order in blocks of 35 trials. Six such blocks were presented in a single session so that n = 42 for each stimulus for each subject. Each trial (Fig. 2 A) consisted of the presentation of a single force with a rise time of 0.2 sec, a plateau phase of 1.5 sec, and a fall time of 0.1 sec (1.8 sec total), with 3.5 sec between trials and a 2 min break between blocks. A midrange stimulus (3 N) was used as a reference stimulus and was presented three times at the commencement of each block and twice after every seventh trial within each block; subjects were told when the reference stimulus was being presented. To establish some consistency between subjects' magnitude scales, they were asked to assign the reference stimulus an arbitrary value of 100 and to estimate the magnitude of the test stimuli relative to this [e.g., if they perceived a test force as being twice as “great” as the reference force, they were instructed to assign it a value double that of the reference (i.e., 200)].
Figure 2.
Time sequence of normal and tangential forces. A, For normal force scaling, the plateau force was held for 1.5 sec; the rise time was 0.2 sec, the fall time was 0.1 sec, and successive trials occurred at 5.3 sec intervals. B, For tangential force scaling, normal force (dashed line) was presented first, followed after 1.0 sec by tangential force (solid line). Rise times were 0.2 sec, fall times were 0.1 sec, and successive stimuli occurred at intervals of 6.3 sec. C, For tangential force discrimination, trials comprised a pair of stimuli. The first stimulus in a pair was the standard, and the second stimulus was the comparison; the comparison could be either the same as the standard (Ss) or different (Sd). The interval between standard and comparison was 2.0 sec, and the interval between successive pairs was 3.0 sec.
Tangential force scaling: constant normal force. In this series, a range of tangential forces was scaled while normal force was held constant; two normal forces were used, 2.5 and 4 N. In each trial (Fig. 2 B), the normal force was applied first, and then after a delay of 1 sec, the tangential force was applied for 1.5 sec. Successive trials were separated by intervals of 3.5 sec. Seven tangential forces (0.25, 0.5, 0.75, 1.0, 1.25, 1.5, and 1.75 N) were presented randomly at the normal force of 2.5 N, and seven (0.4, 0.8, 1.2, 1.6, 2.0, 2.4, and 2.8 N) were presented at the normal force of 4 N. For both normal forces, the same ratios of tangential to normal force were used (i.e., 0.1, 0.2, 0.3, 0.4, 0.5, 0.6, and 0.7). The largest ratio of 0.7 was below the measured effective coefficient of friction, which ranged from 0.8 to 1.4, depending on force direction and individual fingers. Thus, the maximum tangential forces of 1.75 and 2.8 N for normal forces of 2.5 and 4 N, respectively, were well below the slip forces. All combinations of normal and tangential force were presented in four cardinal directions on the fingerpad: distal, proximal, ulnar, and radial. Data were first collected for all stimuli presented in all four directions at a normal force of 2.5 N and then at the higher normal force of 4 N. At each normal force, the corresponding seven tangential forces were presented three times each in random order in blocks of 21 trials. Six such blocks were presented in each of the cardinal directions so that n = 18 for each tangential force in each of the cardinal directions for each subject at both normal forces. Opposite directions were run consecutively (i.e., proximal and distal were always paired, as were ulnar and radial), but the order in which directions were presented was varied randomly. For each subject, data were collected over six consecutive sessions. The subjects' instructions were analogous to those given in the normal force scaling experiments. The midrange reference stimuli (assigned values of 100) were tangential forces of 1 and 1.6 N at the constant normal forces of 2.5 and 4 N, respectively.
Tangential force scaling: varying normal force. In this series of experiments, the applied force had both a tangential and a normal component as in the previous series, except that here, both components were varied randomly (simultaneously). The time course of the trials was the same as for the previous series (Fig. 2 B). Fifteen force combinations comprising five tangential forces (0.5, 0.75, 1, 1.25, and 1.5 N) and three normal forces (2, 3, and 4 N) were presented in random order in blocks of 30 trials; a total of 18 such blocks were presented so that n = 36 for each tangential force at each normal force for each subject. For each subject, data were collected over six consecutive sessions. Tangential force was applied in the distal direction. The combination ofa1N tangential force and a 3 N normal force was chosen as the midrange reference (assigned 100), and subjects were instructed to scale only the tangential force component of the test stimuli relative to the tangential component of this reference stimulus.
Tangential force discrimination. A two-alternative forced-choice paradigm was used to measure the subjects' abilities to discriminate small differences in force applied tangentially to the skin of the fingerpad. Stimuli were presented in pairs; the first was always the standard, and the second was a comparison. Two stimulus conditions were used; in one [stimulus same (Ss)], the magnitude of the tangential force component of the comparison was the same as that of the standard, and in the other condition [stimulus different (Sd)], the magnitude of the tangential force component of the comparison was greater than that of the standard. The magnitude of the normal force component was the same for the standard and the comparison. Subjects were required to judge whether the two stimuli in the pair were the same (response same) or different [response different (Rd)]; feedback was given after each response. Two standard stimuli were used. For the standard tangential force of 1 N, the comparison tangential forces were 1.05, 1.1, 1.2, and 1.3 N, and these were all presented with a normal force of 2.5 N. For the standard tangential force of 1.6 N, the comparison tangential forces were 1.68, 1.76, 1.92, and 2.13 N, and these were all presented with a normal force of 4 N. The range of comparison forces was chosen to provide values below and above the likely difference thresholds, as estimated in previous pilot studies.
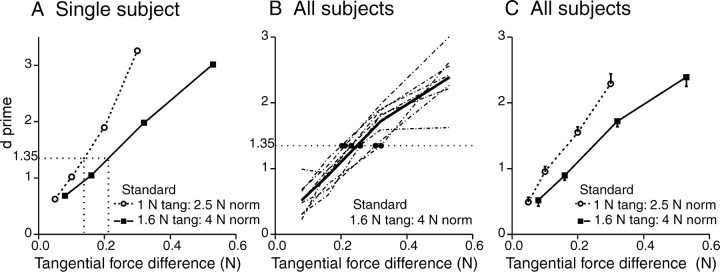
Each trial consisted of the presentation of a pair of stimuli. The sequence and timing were similar to that used for the scaling experiments: the normal force was applied first, and after a delay of 1 sec, the standard stimulus (tangential force) was applied for 1.5 sec. This sequence was repeated after 2 sec for the comparison stimulus (i.e., normal force for 1 sec followed by the comparison stimulus for 1.5 sec). Subjects were given a 3 sec break between subsequent trials. The trials were presented in blocks of 20 (10 Ss trials and 10 Sd trials, presented in random order); for the entire block of 20 trials, all Ss were the same and all Sd were the same. In each experimental session, six blocks of trials were presented with a 2 min break between blocks. Both standards were tested at each session. The order of presentation within and across blocks of trials was varied randomly from session to session for all subjects. At the start of each block, the subject was presented with a number of Ss and Sd pairs and was told which pairs were the same and which were different; no such reminders were given for the remainder of the block. After an initial training period, data were collected over 12 sessions (for each subject and both standards; n = 90 per comparison). From the conditional probabilities p(Rd/Sd) and p(Rd/Ss), the bias-free measure of discrimination (d′) was calculated (Johnson, 1980), and difference limens (d′= 1.35) were determined by linear interpolation of those values for each subject.
Results
Normal force scaling
In the first series of experiments, forces in the range of 1–5 N were applied to the subjects' immobilized index fingers in the normal direction (i.e., orthogonal to the skin surface and equivalent to grip forces in lifting tasks). Subjects were instructed to scale the magnitude of each force relative to the 3 N reference, which was assigned a value of 100. All eight subjects showed similar functions (Fig. 3), with perceived force increasing monotonically as normal force increased. It is clear from the figure that the functions are close to linear. To capture the minor deviation from linearity, a second-order polynomial function was fitted. Simple regression yielded an R2 value of 0.93. However, to separate the effects of the independent parameter from intersubject variability, these and subsequent data were analyzed statistically using a mixed-model approach with functions of the following form: y = (α0 + ϵ0j) + (α1 + ϵ1j)N + (α2 + ϵ2j)N2 + ϵ (where j = 1, 8 subjects). This function described the data well, and a type III test showed that both linear and quadratic effects were significant (p < 0.01 for both) and also showed that if a cubic term was added, it was not significant (p > 0.45).
Figure 3.
Normal force scaling. Dashed lines show perceived magnitude of applied normal force for each of the eight subjects (mean estimates, n = 42 for each level of normal force for each subject). The solid line represents the mean across all eight subjects.
Tangential force scaling
Applying tangential force to the fingerpad is inherently more complicated than applying normal force. To prevent the object from slipping on the skin, any tangential force must be accompanied by a normal force, and the ratio of tangential to normal force must be less than the effective coefficient of friction between the skin and the surface of the object. The time course chosen for the forces (Fig. 2B) ensured that tangential force was always preceded by a stable normal force. Coefficients of friction were estimated in preliminary experiments and were consistent with those ascertained from data reported previously (Westling and Johansson, 1984). After each experiment, the force traces recorded by the six axis transducer (Fig. 1) were examined to verify that there had been no slip between the object and the skin.
First, to determine whether subjects could scale the tangential component of force, they were presented with seven tangential forces in the range 0.25–1.75 N in random order, all with a constant normal force of 2.5 N. As illustrated for a single subject (Fig. 4A, dashed line), the magnitude estimates were consistent and increased monotonically with increasing tangential force. The mean scaling function over the eight subjects (Fig. 4B, dashed line) shows highly consistent scaling behavior across subjects. Second, to determine the effect, if any, of normal force on the ability of subjects to scale tangential force, a range of tangential forces from 0.4 to 2.8 N was presented at a constant normal force of 4 N. As seen in Figure 4, A and B, tangential force scaling performance was comparable for the two normal forces. The ratios of the seven tangential forces to the normal force were the same at both normal forces (2.5 and 4 N). When the two scaling functions are plotted with this ratio as the abscissa (Fig. 4C), the two functions are nearly coincident (see Discussion). The data in Figure 4 were obtained with tangential forces applied in the radial direction.
Figure 4.
Tangential force scaling: normal force invariant. A, Perceived magnitude of tangential force applied with a normal force of 2.5 N (dashed line) and 4 N (solid line) for a single subject (S3) (mean ± SE; n = 18). B, Mean estimates of tangential force across the eight subjects for both normal forces (n = 8; mean ± SE shown at one representative point). C, Mean estimates across all subjects (± one representative SE; n = 8). Tangential force is shown as a fraction of normal force; the seven ratios (0.1, 0.2, 0.3, 0.4, 0.5, 0.6, and 0.7) were the same for both normal forces. Tangential force was applied in the radial direction on the fingerpad for all data shown in this figure.
The distal segment of the finger is anisotropic: the geometry in the direction along the long axis of the finger (proximal–distal) is markedly different from the geometry in the orthogonal direction (radial–ulnar). In addition, the crease at the distal interphalangeal joint results in a difference between proximal and distal directions, and there are differences between the radial and ulnar directions, although these are less obvious. Moreover, differences related to these four cardinal directions have been reported for several aspects of hand function (Hager-Ross et al., 1996). Therefore, we tested for any differences in the subjects' perceptions of tangential force by measuring their scaling ability in all four directions. The four directions were interleaved randomly (see Materials and Methods).
In Figure 5A, scaling functions are shown for a single subject at the 2.5 N normal force. Functions averaged across all eight subjects are shown for all four directions and for both normal forces in Figure 5B. The same data are shown overlaid in Figure 5C by using the ratio of tangential to normal force as the abscissa. These results show clearly that there is no substantial effect of direction. Statistical analysis was performed using a mixed-model approach of the same form as that described for the normal force scaling. The tangential force scaling functions were well described by linear and quadratic components, significant at the p < 0.01 level (type III test). Although the effect of direction was small, it was statistically significant (p < 0.01); Tukey–Kramer post hoc analyses (p < 0.05) revealed that the significant differences were distal versus ulnar and proximal versus ulnar at the normal force of 2.5 N and distal versus ulnar and distal versus radial at the normal force of 4 N.
Figure 5.
Effect of tangential force direction on scaling. A, Perceived magnitude of tangential force applied with a constant normal force of 2.5 N in four directions (radial, ulnar, distal, andproximal) for a single subject (S2) (mean ± one representative SE; n = 18). B, Mean performance across all eight subjects for tangential forces applied in all four directions with normal forces of 2.5 and 4N(± one representative SE for each normal force; n = 8). C, Lines show mean estimates across all eight subjects for both normal forces (dashed lines, 2.5 N; solid lines, 4N) in each of the four directions; tangential force is shown as a proportion of normal force.
The data in Figures 4 and 5 show that humans can scale tangential force magnitude when normal force is held constant and can do so at different normal forces. However, because the data at the two normal forces were collected in separate experimental sequences with different reference or anchor stimuli, there are two crucial questions that cannot be answered. First, can subjects extract the tangential force component when the normal force is varying concurrently? Second, if that is possible, is there any interactive effect between normal and tangential forces on the perception of tangential force magnitude? To answer these questions, we presented subjects with a set of 15 stimuli comprising combinations of five tangential forces and three normal forces presented randomly, with tangential force applied in the distal direction.
Figure 6A shows the capacity of one subject to scale increasing tangential force when normal and tangential forces both varied randomly. Perceived magnitude increased monotonically as tangential force increased, and this was the case for all subjects, as shown in Figure 6B. It is evident from the data that tangential force had a substantial and highly consistent effect; normal force had a much smaller effect, with larger normal forces reducing the perceived magnitude of the tangential component. Statistical analysis with the same mixed-model approach used previously showed that both tangential and normal force had significant effects on performance. The data were described by linear and quadratic components of tangential force (p < 0.01 for both) and a linear component of normal force (p < 0.01).
Figure 6.
Tangential force scaling with concurrently varying normal force. A, Data are mean estimatesforasinglesubject(S8),scalingtangentialforcewithnormalforcevaryingrandomlybetween three magnitudes: 2, 3, and 4 N. Data points are means ± SE; n = 36. B, Data are mean estimates across all eight subjects (±SE; n = 8) at three levels of normal force, which varied randomly throughout blocks of trials. Tangential force was applied in the distal direction for this series.
Tangential force discrimination
To determine the smallest difference in tangential force that could be discriminated, the same eight subjects took part in a series of discrimination experiments using subsets of stimuli from within the range used in the scaling studies. Two series were conducted: in one, the standard tangential force was 1 N and the normal force was 2.5 N; in the other, the standard tangential force was 1.6 N and the normal force was 4 N. These two series were used to determine whether force magnitude would affect acuity. Indices of discrimination (d′), calculated from the conditional probabilities p(Rd/Sd) and p(Rd/Ss), are shown for a single subject for both discrimination series in Figure 7A. The figure also illustrates the calculation of the difference limen (d′= 1.35) by linear interpolation. The discrimination functions of all eight subjects are shown in Figure 7B for the 1.6 N standard tangential force (normal force, 4 N); filled circles show the difference limens, and the solid line shows the mean d′ values across the eight subjects. In Figure 7C, the mean discrimination functions across the eight subjects are compared for the two series. For all subjects in both series, the relationship between tangential force magnitude difference and discrimination performance was approximately linear. Difference limens for each subject are shown in Table 1 for both series. The mean difference limens, 0.163 and 0.252 for standards of 1 and 1.6 N, respectively, were significantly different (p < 0.01; two-tailed paired t test; n = 8). In both cases, the Weber fraction was the same (0.16).
Figure 7.
Tangential (tang) force discrimination. A, Discrimination functions for a single subject (S6) for both series (standard tangential force of1Nata normal force of 2.5 N and standard tangential force of 1.6 N at a normal force of 4 N). Fine dotted lines show difference limens estimated by linear interpolation. B, Discrimination functions for all subjects (dashed lines) for a standard tangential force of 1.6 N (normal force of 4 N). The solid line shows mean d′ values (n = 8). Filled circles show difference limens (d′= 1.35) for each subject. C, Mean d′ values (averaged across all eight subjects; n = 8) for both standards. Error bars show unidirectional SE. Tangential forces in A–C are shown as the differences from the respective standard tangential forces. Tangential force was applied in the distal direction for discrimination studies.
Table 1.
Difference limens (in Newtons) for discriminating tangential force
|
|
Standard tangential force |
||
|---|---|---|---|
|
|
1.0 N |
1.6 N |
|
| Subject | |||
| S1 | 0.216 | 0.258 | |
| S2 | 0.094 | 0.201 | |
| S3 | 0.157 | 0.230 | |
| S4 | 0.188 | 0.231 | |
| S5 | 0.139 | 0.256 | |
| S6 | 0.193 | 0.321 | |
| S7 | 0.177 | 0.304 | |
| S8 | 0.138 | 0.212 | |
| Mean ± SE | 0.163 ± 0.014 | 0.252 ± 0.015 | |
| Weber fraction |
0.16 |
0.16 |
|
For each subject, difference limens were calculated for two series (1 N standard tangential force and 2.5 N normal force; 1.6 N standard tangential force and 4 N normal force). Weber fractions were calculated by dividing the mean difference limens by the respective standard tangential force.
Discussion
Our results show unequivocally that humans are able to scale and discriminate forces acting tangentially to the skin of the fingerpad. Moreover, by applying forces passively to an immobilized finger, we ensured that the vast majority of information, if not all, conveyed to the subjects about these forces was that arising from cutaneous mechanoreceptors in the distal segment of the finger. In principle, it is possible that some subcutaneous mechanoreceptors may have been stimulated, but based on neural recordings with our stimulus, we do not believe this to be the case. Subjects were able to scale tangential forces even in the presence of concurrent independently varying normal forces. Our subjects could discriminate a difference in tangential force of 16%.
Sensory signals in manipulation
During routine manual exploration and manipulation of objects, humans are required to generate forces with precisely controlled normal and tangential components. Our ability to manipulate objects with such precision is testimony itself to the accuracy of the underlying sensorimotor control system for finger movements. There have been many behavioral studies examining the nature of sensorimotor control, and the current view is that of a system with a combination of feedback and feedforward elements, strategies for which rely ultimately on exact sensory information concerning the manipulation (Gordon et al., 1991; Hager-Ross et al., 1996; Ohki et al., 2002; Flanagan et al., 2003). Our experiments demonstrate a rich and accurate source of force feedback (both normal and tangential components) from the mechanoreceptors in the glabrous skin of the digits. How this is combined with other potential sources of sensory input, such as afferents from the joints, muscles, and hairy skin, is not known and points to important future experiments. Nor is it known how these sensory signals are combined with efference copy signals (Gandevia et al., 1992) from the motor cortex and other parts of the brain to control the precise hand movements that are the hallmark of primates.
Experimental constraints
There is an inherent complication in designing experiments to explore tangential forces acting on the skin. To prevent the object from slipping, normal forces must also be present, and the relationship between the normal (grip) and tangential (load) force is limited by the effective coefficient of friction between the object and the skin (Johansson and Westling, 1984; Cadoret and Smith, 1996). How humans coordinate these two force components has been studied behaviorally in a variety of tasks (Cole and Abbs, 1988; Wing and Lederman, 1998; Serrien et al., 1999). Alternatively, normal forces could have been eliminated if the probe were glued to the skin. We ruled out this option because the resultant stimulus would have been very unnatural and would have changed the skin mechanics, leading to results of dubious value.
Although forces were applied passively to an immobilized finger in our experiments, the force patterns were chosen to provide a realistic simulation of events on the digit tips during natural manipulations, with one exception. In our experiments, normal force was applied first and then tangential force was applied after a delay of 1 sec, whereas in grip and lift tasks, the grip and load forces increase more or less simultaneously with only a slight lead for the grip force (Johansson and Westling, 1984). The temporal relationships between normal and tangential forces during more complex tasks, such as fastening a button, or during haptic exploration are not known. Limitations in the dynamics of our stimulator precluded us from achieving precise simultaneity of the two forces; therefore, we decided to separate them clearly. This had the added advantage that all tangential forces were applied on a stable plateau of normal force so that subjects' judgments could be related directly to the tangential force component. It also has the advantage that in matching neural recordings, we have been able to separate the responses of the afferents to normal and tangential forces. Notwithstanding the above, the close analogy between our paradigm and normal manipulation allows valid and important conclusions. However, in the future, similar experiments will need to be done with near-simultaneous normal and tangential forces.
Relationship to previous psychophysical studies
There have been a small number of psychophysical studies of human perception of force, as opposed to displacement, in which the forces were applied normal to the skin of a digit (Goodwin and Wheat, 1992; Cohen and Vierck, 1993b). In these experiments, the forces were applied passively to an immobilized finger, so that essentially only cutaneous mechanoreceptors contributed to the afferent signals that led to perception. These experiments showed that these mechanoreceptor signals are sufficient for humans to scale normal force. Goodwin and Wheat (1992) also showed that normal forces can be perceived accurately even when cutaneous mechanoreceptor signals are altered by other stimulus parameters, such as stimulus curvature.
The major difference in the normal force data reported here is the magnitude of the forces. In previous psychophysical experiments, the forces ranged up to some 0.7 N, whereas in most natural human manipulations, the forces on the digits routinely range up to several Newtons, and on occasion, particularly with heavy objects, are even greater. Therefore, it was important to establish that humans can perceive normal force in a graded manner in the range that is typically used.
Srinivasan et al. (1990) showed that although humans could not detect the slip of a smooth glass plate stroked across the fingerpad, they could detect the direction of skin stretch before slip. However, to our knowledge, the only psychophysical study quantifying perception of forces tangential to the skin without lateral slip is the limited study of Pare et al. (2002). Although their results suggest that humans can perceive differences in tangential force, the forces applied by the experimenter via a hand-held probe could only be controlled approximately, so it is difficult to interpret the data. The forces we used were produced by a computer-controlled stimulator and were in the range commonly used for manipulation. Our data establish unequivocally that humans can scale such tangential forces consistently and accurately and can discriminate small (16%) differences in tangential force.
Neural signals
What is indisputable from our data is that the cutaneous afferents provide a wealth of precise information about both the normal and tangential force components. Viewed another way, they provide information about both the magnitude and direction of the three-dimensional force vector. What is the basis of the neural signals underlying our subjects' abilities to perceive these forces?
It has been known for some time that responses of mechanoreceptive afferents from both monkey and human glabrous skin are influenced by normal forces applied to the skin (Phillips and Johnson, 1981; Cohen and Vierck, 1993a; Goodwin et al., 1995, 1997; Bisley et al., 2000). It has been shown more recently that in humans, the same afferents also respond to tangential forces (Birznieks et al., 2001), and the same is true in monkeys (Srinivasan et al., 1990). These observations themselves are insufficient to explain our psychophysical data. In particular, they do not explain how information about concurrent normal and tangential force components can be extracted independently from the afferent responses. Nor do they explain why other parameters of the stimulus, such as its shape or position on the skin, which also affect afferent responses, do not confound perception of the two force components. It is also not possible from single afferent responses to explain the resolution of human perception.
These issues are common to all multidimensional sensory stimuli and can be resolved, in part, by reconstructing or modeling responses in the entire active population. It is only at this level that the encoding of individual stimulus parameters can be revealed (Goodwin and Wheat, 2004). Population reconstructions have been performed for mechanoreceptive afferents innervating the glabrous skin on the terminal phalanx of the monkey's finger for a variety of stimuli. Taking such an approach enabled the testing of hypotheses about encoding mechanisms for the pertinent stimulus parameters (Johnson, 1974; Blake et al., 1997; Dodson et al., 1998; Khalsa et al., 1998; Goodwin and Wheat, 1999; Wheat and Goodwin, 2000). For example, Goodwin and Wheat (1999) showed how normal force can be perceived independently of the shape of the object and its position on the skin. As of yet, such population analyses in the monkey have not included forces tangential to the skin. A recent study in humans demonstrated clearly that the direction of a force applied to a fingertip is represented in the population of cutaneous afferents; the direction is determined by both normal and tangential components of force (Birznieks et al., 2001). That study needs to be extended to demonstrate how force direction and magnitude can be represented independently or, equivalently, how normal and tangential magnitudes can be represented independently.
Most previous quantitative population models have been based on the responses of afferents terminating in proximity to the stimulus (Khalsa et al., 1998; Wheat and Goodwin, 2001). However, it is now apparent that afferents terminating across the entire extent of the distal segment of digital glabrous skin respond to many or most stimuli applied to the fingerpad, although they may be some distance from the stimulus contact point (Bisley et al., 2000; Birznieks et al., 2001). The geometry of the finger and its skin mechanics are highly complex, and the responses of afferents distributed over the finger are correspondingly complex (Maeno and Kobayashi, 1998; Pawluk and Howe, 1999; Nakazawa et al., 2000). To resolve the issue of independent representation of normal and tangential forces, a quantitative reconstruction of the responses of afferents innervating the entire distal glabrous skin is necessary.
Footnotes
This work was supported by a grant from the National Health and Medical Research Council of Australia.
Correspondence should be addressed to H. E. Wheat, Department of Anatomy and Cell Biology, University of Melbourne, Victoria 3010, Australia. E-mail: hwheat@unimelb.edu.au.
DOI:10.1523/JNEUROSCI.4822-03.2004
Copyright © 2004 Society for Neuroscience 0270-6474/04/243394-08$15.00/0
References
- Augurelle AS, Smith AM, Lejeune T, Thonnard JL (2003) Importance of cutaneous feedback in maintaining a secure grip during manipulation of hand-held objects. J Neurophysiol 89: 665–671. [DOI] [PubMed] [Google Scholar]
- Birznieks I, Jenmalm P, Goodwin AW, Johansson RS (2001) Encoding of direction of fingertip forces by human tactile afferents. J Neurosci 21: 8222–8237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bisley JW, Goodwin AW, Wheat HE (2000) Slowly adapting type I afferents from the sides and end of the finger respond to stimuli on the center of the fingerpad. J Neurophysiol 84: 57–64. [DOI] [PubMed] [Google Scholar]
- Blake DT, Hsiao SS, Johnson KO (1997) Neural coding mechanisms in tactile pattern recognition: the relative contributions of slowly and rapidly adapting mechanoreceptors to perceived roughness. J Neurosci 17: 7480–7489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cadoret G, Smith AM (1996) Friction, not texture, dictates grip forces used during object manipulation. J Neurophysiol 75: 1963–1969. [DOI] [PubMed] [Google Scholar]
- Cohen RH, Vierck Jr CJ (1993a) Population estimates for responses of cutaneous mechanoreceptors to a vertically indenting probe on the glabrous skin of monkeys. Exp Brain Res 94: 105–119. [DOI] [PubMed] [Google Scholar]
- Cohen RH, Vierck Jr CJ (1993b) Relationships between touch sensations and estimated population responses of peripheral afferent mechanoreceptors. Exp Brain Res 94: 120–130. [DOI] [PubMed] [Google Scholar]
- Cole KJ, Abbs JH (1988) Grip force adjustments evoked by load force perturbations of a grasped object. J Neurophysiol 60: 1513–1522. [DOI] [PubMed] [Google Scholar]
- Cole KJ, Steyers CM, Graybill EK (2003) The effects of graded compression of the median nerve in the carpal canal on grip force. Exp Brain Res 148: 150–157. [DOI] [PubMed] [Google Scholar]
- Dodson MJ, Goodwin AW, Browning AS, Gehring HM (1998) Peripheral neural mechanisms determining the orientation of cylinders grasped by the digits. J Neurosci 18: 521–530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flanagan JR, Wing AM (1995) The stability of precision grip forces during cyclic arm movements with a hand-held load. Exp Brain Res 105: 455–464. [DOI] [PubMed] [Google Scholar]
- Flanagan JR, Vetter P, Johansson RS, Wolpert DM (2003) Prediction precedes control in motor learning. Curr Biol 13: 146–150. [DOI] [PubMed] [Google Scholar]
- Gandevia SC, McCloskey DI, Burke D (1992) Kinaesthetic signals and muscle contraction. Trends Neurosci 15: 62–65. [DOI] [PubMed] [Google Scholar]
- Goodwin AW, Wheat HE (1992) Magnitude estimation of contact force when objects with different shapes are applied passively to the fingerpad. Somatosens Mot Res 9: 339–344. [DOI] [PubMed] [Google Scholar]
- Goodwin AW, Wheat HE (1999) Effects of nonuniform fiber sensitivity, innervation geometry, and noise on information relayed by a population of slowly adapting type I primary afferents from the fingerpad. J Neurosci 19: 8057–8070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goodwin AW, Wheat HE (2004) Sensory signals in neural populations underlying tactile perception and manipulation. Annu Rev Neurosci 27: 53–77. [DOI] [PubMed] [Google Scholar]
- Goodwin AW, Browning AS, Wheat HE (1995) Representation of curved surfaces in responses of mechanoreceptive afferent fibers innervating the monkey's fingerpad. J Neurosci 15: 798–810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goodwin AW, Macefield VG, Bisley JW (1997) Encoding of object curvature by tactile afferents from human fingers. J Neurophysiol 78: 2881–2888. [DOI] [PubMed] [Google Scholar]
- Goodwin AW, Jenmalm P, Johansson RS (1998) Control of grip force when tilting objects: effect of curvature of grasped surfaces and applied tangential torque. J Neurosci 18: 10724–10734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gordon AM, Forssberg H, Johansson RS, Westling G (1991) The integration of haptically acquired size information in the programming of precision grip. Exp Brain Res 83: 483–488. [DOI] [PubMed] [Google Scholar]
- Gysin P, Kaminski TR, Gordon AM (2003) Coordination of fingertip forces in object transport during locomotion. Exp Brain Res 149: 371–379. [DOI] [PubMed] [Google Scholar]
- Hager-Ross C, Cole KJ, Johansson RS (1996) Grip-force responses to unanticipated object loading: load direction reveals body- and gravity-referenced intrinsic task variables. Exp Brain Res 110: 142–150. [DOI] [PubMed] [Google Scholar]
- Jenmalm P, Johansson RS (1997) Visual and somatosensory information about object shape control manipulative fingertip forces. J Neurosci 17: 4486–4499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jenmalm P, Dahlstedt S, Johansson RS (2000) Visual and tactile information about object-curvature control fingertip forces and grasp kinematics in human dexterous manipulation. J Neurophysiol 84: 2984–2997. [DOI] [PubMed] [Google Scholar]
- Johansson RS, Westling G (1984) Roles of glabrous skin receptors and sensorimotor memory in automatic control of precision grip when lifting rougher or more slippery objects. Exp Brain Res 56: 550–564. [DOI] [PubMed] [Google Scholar]
- Johnson KO (1974) Reconstruction of population response to vibratory stimulus in quickly adapting mechanoreceptive afferent fiber population innervating glabrous skin of the monkey. J Neurophysiol 37: 48–72. [DOI] [PubMed] [Google Scholar]
- Johnson KO (1980) Sensory discrimination: decision process. J Neurophysiol 43: 1771–1792. [DOI] [PubMed] [Google Scholar]
- Khalsa PS, Friedman RM, Srinivasan MA, LaMotte RH (1998) Encoding of shape and orientation of objects indented into the monkey fingerpad by populations of slowly and rapidly adapting mechanoreceptors. J Neurophysiol 79: 3238–3251. [DOI] [PubMed] [Google Scholar]
- Maeno T, Kobayashi K (1998) FE analysis of the dynamic characteristics of the human finger pad in contact with objects with/without surface roughness. Proceedings of the 1998 American Society of Mechanical Engineers International Mechanical Engineering Congress and Exposition 64: 279–286. [Google Scholar]
- Moberg E (1975) Hand surgery and the development of hand prostheses. Scand J Plast Reconstr Surg 9: 227–230. [DOI] [PubMed] [Google Scholar]
- Monzee J, Lamarre Y, Smith AM (2003) The effects of digital anesthesia on force control using a precision grip. J Neurophysiol 89: 672–683. [DOI] [PubMed] [Google Scholar]
- Nakazawa N, Ikeura R, Inooka H (2000) Characteristics of human fingertips in the shearing direction. Biol Cybern 82: 207–214. [DOI] [PubMed] [Google Scholar]
- Ohki Y, Edin BB, Johansson RS (2002) Predictions specify reactive control of individual digits in manipulation. J Neurosci 22: 600–610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pare M, Carnahan H, Smith AM (2002) Magnitude estimation of tangential force applied to the fingerpad. Exp Brain Res 142: 342–348. [DOI] [PubMed] [Google Scholar]
- Pawluk DT, Howe RD (1999) Dynamic lumped element response of the human fingerpad. J Biomech Eng 121: 178–183. [DOI] [PubMed] [Google Scholar]
- Phillips JR, Johnson KO (1981) Tactile spatial resolution. III. A continuum mechanics model of skin predicting mechanoreceptor responses to bars, edges, and gratings. J Neurophysiol 46: 1204–1225. [DOI] [PubMed] [Google Scholar]
- Reilmann R, Gordon AM, Henningsen H (2001) Initiation and development of fingertip forces during whole-hand grasping. Exp Brain Res 140: 443–452. [DOI] [PubMed] [Google Scholar]
- Rinkenauer G, Mattes S, Ulrich R (1999) The surface-weight illusion: on the contribution of grip force to perceived heaviness. Percept Psychophys 61: 23–30. [DOI] [PubMed] [Google Scholar]
- Robles-De-La-Torre G, Hayward V (2001) Force can overcome object geometry in the perception of shape through active touch. Nature 412: 445–448. [DOI] [PubMed] [Google Scholar]
- Rothwell JC, Traub MM, Day BL, Obeso JA, Thomas PK, Marsden CD (1982) Manual motor performance in a deafferented man. Brain 105: 515–542. [DOI] [PubMed] [Google Scholar]
- Serrien DJ, Kaluzny P, Wicki U, Wiesendanger M (1999) Grip force adjustments induced by predictable load perturbations during a manipulative task. Exp Brain Res 124: 100–106. [DOI] [PubMed] [Google Scholar]
- Smith AM, Gosselin G, Houde B (2002) Deployment of fingertip forces in tactile exploration. Exp Brain Res 147: 209–218. [DOI] [PubMed] [Google Scholar]
- Srinivasan MA, Whitehouse JM, LaMotte RH (1990) Tactile detection of slip: surface microgeometry and peripheral neural codes. J Neurophysiol 63: 1323–1332. [DOI] [PubMed] [Google Scholar]
- Westling G, Johansson RS (1984) Factors influencing the force control during precision grip. Exp Brain Res 53: 277–284. [DOI] [PubMed] [Google Scholar]
- Wheat HE, Goodwin AW (2000) Tactile discrimination of gaps by slowly adapting afferents: effects of population parameters and anisotropy in the fingerpad. J Neurophysiol 84: 1430–1444. [DOI] [PubMed] [Google Scholar]
- Wheat HE, Goodwin AW (2001) Tactile discrimination of edge shape: limits on spatial resolution imposed by parameters of the peripheral neural population. J Neurosci 21: 7751–7763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wing AM, Lederman SJ (1998) Anticipating load torques produced by voluntary movements. J Exp Psychol Hum Percept Perform 24: 1571–1581. [DOI] [PubMed] [Google Scholar]