Abstract
Cerambycidae is one of the most diversified groups within Coleoptera and includes nearly 35,000 known species. The relationships at the subfamily level within Cerambycidae have not been convincingly demonstrated and the gene rearrangement of mitochondrial genomes in Cerambycidae remains unclear due to the low numbers of sequenced mitogenomes. In the present study, we determined five complete mitogenomes of Cerambycidae and investigated the phylogenetic relationship among the subfamilies of Cerambycidae based on mitogenomes. The mitogenomic arrangement of all five species was identical to the ancestral Cerambycidae type without gene rearrangement. Remarkably, however, two large intergenic spacers were detected in the mitogenome of Pterolophia sp. ZJY-2019. The origins of these intergenic spacers could be explained by the slipped-strand mispairing and duplication/random loss models. A conserved motif was found between trnS2 and nad1 gene, which was proposed to be a binding site of a transcription termination peptide. Also, tandem repeat units were identified in the A + T-rich region of all five mitogenomes. The monophyly of Lamiinae and Prioninae was strongly supported by both MrBayes and RAxML analyses based on nucleotide datasets, whereas the Cerambycinae and Lepturinae were recovered as non-monophyletic.
Keywords: Mitochondrial genome, Cerambycidae, Intergenic spacer, Phylogenetic relationship
Introduction
Coleoptera (Hexapoda: Insecta) are a highly diverse group of insects consisting of about 360,000 known species of beetles that account for almost 40% of all described insect species (Lawrence & Newton, 1982; Hunt et al., 2007). Cerambycidae (longicorn beetles) is one of the species-rich families of Coleoptera and is a group of phytophagous insects with over 4,000 genera and 35,000 species in the world (Monné, Monné & Mermudes, 2009; Sama et al., 2010). Longicorn beetles are morphologically and ecologically diverse, and have significant effects on almost all terrestrial ecosystems (Ponomarenko & Prokin, 2015). Nevertheless, owing to their remarkable species richness, variable morphological features and sparse gene data, the resolution of the phylogeny of longicorn beetles has turned out to be a difficult challenge (Bologna et al., 2008; Zhang et al., 2018a; Zhang et al., 2018b; Zhang et al., 2018c). Cerambycidae s. s. (sensu stricto) has usually been divided into eight subfamilies: Lamiinae, Cerambycinae, Lepturinae, Prioninae, Dorcasominae, Parandrinae, Spondylidinae and Necydalinae (Svacha, Wang & Chen, 1997) whereas Cerambycidae s. l. (sensu lato) was considered to consist of Cerambycidae s. s., Disteniidae, Oxypeltidae and Vesperidae (Napp, 1994; Reid, 1995; Svacha, Wang & Chen, 1997). Even if the number and definition of Cerambycidae gradually stabilizes, the relationships at the subfamily level remained unclear.
The mitochondrial genome is widely considered to be an informative molecular marker for species identification, molecular evolution, and comparative genomic research (Moritz, Dowling & Brown, 1987; Boore, 1999) due to its maternal inheritance and high evolutionary rate properties (Avise et al., 1987). In the last few years, studies of animal mitogenomes have grown rapidly in number and approximately 40,000 mitogenome sequences have now been published in the NCBI database (Tan et al., 2017). By contrast, a mere 18 sequenced mitogenomes of Cerambycidae have been reported, among them being eight mitogenomes belonging to the subfamily Lamiinae, four mitogenomes of the subfamily Cerambycinae, three mitogenomes of the subfamily Prioninae, and three mitogenomes of the subfamily Lepturinae (Kim et al., 2009; Chiu et al., 2016; Fang et al., 2016; Guo et al., 2016; Li et al., 2016a; Li et al., 2016b; Wang et al., 2016; Lim et al., 2017; Liu et al., 2017; Song et al., 2017; Liu et al., 2018; Que et al., 2019; Wang et al., 2019). These few mitogenomes seriously restrict the capacity for phylogenetic analyses and phylogeography of the Cerambycidae.
The gene organization of the known mitogenomes of Coleoptera, especially the arrangements of protein-coding genes, are mostly in accordance with those of ancestral insects (Timmermans & Vogler, 2012). Nevertheless, recent evidence suggested that gene rearrangements had occurred in the tRNA of Mordella atrata (Coleoptera: Mordellidae) and Naupactus xanthographus (Coleoptera: Curculionidae) (Song et al., 2010). In addition to these, recombination in the control region was observed in Phrixtothrix hirtus (Coleoptera: Phengodidae) and Teslasena femoralis (Coleoptera: Elateridae) (Amaral et al., 2016). The mitogenome structure was originally found with no introns, sparse intergenic spacers and no overlapping genes (Ojala, Montoya & Attardi, 1981). Nevertheless, large non-coding regions (except the A + T-rich region) in mitogenomes have been observed within beetles, including a 1724-bp long intergenic spacer region in Pyrocoelia rufa (Coleoptera: Lampyridae), a 494-bp region in Hycleus chodschenticus (Coleoptera: Meloidae) and two large intergenic spacers of more than 30 bp in Hycleus species (Bae et al., 2004; Yuan et al., 2016; Haddad et al., 2018). Previously reported tandem repeat units or an additional origin of replication were identified among large intergenic regions (Dotson & Beard, 2001; Rodovalho et al., 2014).
The phylogenetic relationships within Cerambycidae have yet to be fully resolved due to a lack of adequately convincing taxon sampling, and the monophyly of subfamilies within Cerambycidae need further discussion (Haddad et al., 2018; Kim et al., 2018). With the aim to discuss the monophyly of subfamilies of Cerambycidae and gene arrangements of the mitogenome, complete mitogenomes of the five longicorn beetle species were determined. We also described the structural and compositional features of the newly sequenced mitogenomes and analyzed the intergenic spacers to explain the possible evolutionary mechanisms.
Materials and Methods
Sampling collection and DNA extraction
Five longicorn beetle specimens (Oberea yaoshana, Thermistis croccocincta, Blepephaeus succinctor, Nortia carinicollis, Pterolophia sp. ZJY-2019) were captured from Jinxiu, Guangxi Zhuang Autonomous Region, China and were stored at −40 °C in the lab of JY Zhang (College of Chemistry and Life Science, Zhejiang Normal University). The specimens were identified by Dr. JY Zhang based on morphology. Total genomic DNA was extracted from the thorax muscle using Ezup Column Animal Genomic DNA Purification Kit (Sangon Biotech Company, Shanghai, China).
PCR amplification and sequencing
In order to obtain the entire mitogenome of samples, we used eleven universal primer pairs to amplify eleven adjacent and overlapping fragments (Simon et al., 2006; Zhang et al., 2008; Zhang et al., 2018a; Zhang et al., 2018b). Then specific primers were designed from the initial overlapping fragments using Primer Premier 5.0 (Premier Biosoft International, Palo Alto, CA). A total of 45 pairs of primers were used in the present study to amplify and sequence the remaining gaps (Table S1). The cycling conditions and reaction volume of PCR amplifications were as in Cheng et al. (2016) and Gao et al. (2018). All PCR products were sequenced by Sangon Biotech Company (Shanghai, China).
Mitogenome annotation and sequence analyses
Manual proofreading and assembling of contiguous and overlapping sequences used DNASTAR Package v.6.0 (Burland, 2000). We annotated the tRNA genes by MITOS (freely available at http://mitos.bioinf.uni-leipzig.de/index.py) (Bernt et al., 2013). Two rRNA genes and the A + T-rich region were identified using the Clustal W in Mega 7.0 (Kumar, Stecher & Tamura, 2016) based on alignments of homologous sequences from other species of Cerambycidae available in GenBank (Kim et al., 2009; Fang et al., 2016; Lim et al., 2017). The nucleotide sequences of the 13 protein-coding genes (PCGs) were translated into amino acids based on the invertebrate mitogenome genetic code (Cameron, 2014). We used Mega 7.0 (Kumar, Stecher & Tamura, 2016) to find the open reading frames of the 13 PCGs and calculate AT content along with codon usage for the five newly sequenced mitogenomes. Circular mitogenome maps were generated by CG View server V 1.0 (Grant & Stothard, 2008). Composition skew analysis was calculated on the basis of the formula AT-skew = (A − T)/(A + T) and GC-skew = (G − C)/(G + C) (Perna & Kocher, 1995). Tandem Repeat Finder V 4.07 (http://tandem.bu.edu/trf/trf.html) (Benson, 1999) was used to find tandem repetitive sequences.
Phylogenetic analyses
For the purpose of reconstructing the phylogenetic relationships of Cerambycidae, a nucleotide dataset (13P26) of the 13 protein-coding genes of 26 complete mitogenomes was used (Table 1) according to the methods of Zhang et al. (2019), this included the 5 newly determined sequences and 18 published complete mitogenomes of Cerambycidae (Kim et al., 2009; Chiu et al., 2016; Fang et al., 2016; Guo et al., 2016; Li et al., 2016a; Li et al., 2016b; Wang et al., 2016; Lim et al., 2017; Liu et al., 2017; Song et al., 2017; Liu et al., 2018; Que et al., 2019; Wang et al., 2019). Three species of Galerucinae, Paleosepharia posticata, Diabrotica barberi and Diabrotica virgifera served as the outgroups (Coates, 2014; Wang & Tang, 2017). To verify whether the lack of samples affects the relationships among the Cerambycidae, we reconstructed Cerambycidae phylogeny based on the nucleotide data (12P38) of 12 PCGs (omitting the nad2 gene) from 38 complete or nearly complete mitogenomes (Table 1). These include all species of the 13P26 dataset, 8 directly submitted partial mitogenomes of Cerambycidae, one mitogenome of Necydalinae, two mitogenomes of Vesperidae and one mitogenome of Disteniidae (Nie et al., 2017). Each of the 13 protein-coding genes in 13P26 dataset or 12 protein-coding genes in 12P38 dataset was aligned using Clustal W in the program Mega 7.0 (Kumar, Stecher & Tamura, 2016). Conserved regions were identified by the program Gblock 0.91b (Castresana, 2000). Protein-coding genes were partitioned a priori by codon position. Accodrding to the analyses methods of Zhang et al. (2008), Ma et al. (2015a), Ma et al. (2015b) and Cheng et al. (2016), we excluded the third codon positions because of the saturated third codon positions and obtained a 12P38 dataset with 5584 nucleotide sites and 13P26 dataset with 6960 nucleotide sites. So 12P38 dataset with 24 partitions and 13P26 dataset with 26 partitions were used. The optimal partitioning scheme and best-fitting models were selected by the program PartitionFinder 1.1.1 (Lanfear et al., 2012) based on the Bayesian information criterion (BIC) (Tables 2 and 3). Bayesian Inference (BI) and Maximum likelihood (ML) methods were used for phylogenetic analyses. BI analyses were carried out in MrBayes 3.2 (Ronquist et al., 2012) with the model of GTR + I + G. The runs were set for 10 million generations with sampling every 1,000 generations. The first 25% of generations were removed as burn-in and the average standard deviation of split frequencies in Bayesian was below 0.01. ML analyses were performed by RAxML 8.2.0 with the best-fitting model of GTRGAMMAI. Branch support values were inferred from a rapid bootstrap method applied with 1,000 replications (Stamatakis, 2014).
Table 1. Species used to construct the phylogenetic relationships along with GenBank accession numbers.
| Order | Family | Species | GenBank No. | References |
|---|---|---|---|---|
| Cerambycidae | Lamiinae | Anoplophora glabripennis | DQ768215 | Fang et al. (2016) |
| Psacothea hilaris | FJ424074 | Kim et al. (2009) | ||
| Thyestilla gebleri | KY292221 | Yang et al. (2017) | ||
| Monochamus alternatus | KJ809086 | Li et al. (2016a) | ||
| Anoplophora chinensis | KT726932 | Li et al. (2016b) | ||
| Apriona swainsoni | NC_033872 | Que et al. (2019) | ||
| Batocera lineolata | MF521888 | Liu et al. (2017) | ||
| Oberea yaoshana | MK863509 | This study | ||
| Thermistis croccocincta | MK863511 | This study | ||
| Blepephaeus succinctor | MK863507 | This study | ||
| Pterolophia sp.ZJY-2019 | MK863510 | This study | ||
| Olenecamptus subobliteratus* | KY796054 | Directly submitted | ||
| Eutetrapha metallescens* | KY796053 | Directly submitted | ||
| Cerambycinae | Xylotrechus grayii | NC_030782 | Guo et al. (2016) | |
| Xystrocera globosa | MK570750 | Wang et al. (2019) | ||
| Nortia carinicollis | MK863508 | This study | ||
| Massicus raddei | KC751569 | Wang et al. (2016) | ||
| Aeolesthes oenochrous | AB703463 | Chiu et al. (2016) | ||
| Obrium sp. NS-2015 | KT945156 | Song et al. (2017) | ||
| Pyrrhidium sanguineum* | KX087339 | Directly submitted | ||
| Chlorophorus simillimus* | KY796055 | Directly submitted | ||
| Prioninae | Callipogon relictus | MF521835 | Lim et al. (2017) | |
| Dorysthenes paradoxus | MG460483 | Liu et al. (2018) | ||
| Aegosoma sinicum | NC_038089 | Directly submitted | ||
| Lepturinae | Leptura arcuata* | KY796051 | Directly submitted | |
| Stictoleptura succedanea* | KY796052 | Directly submitted | ||
| Rhagium mordax* | JX412743 | Directly submitted | ||
| Stenurella nigra* | KX087348 | Directly submitted | ||
| Cortodera humeralis | KX087264 | Directly submitted | ||
| Anastrangalia sequensi | KY773687 | Directly submitted | ||
| Brachyta interrogationis | KX087246 | Directly submitted | ||
| Necydalinae | Necydalis ulmi* | JX220989 | Directly submitted | |
| Disteniidae | Disteniinae | Disteniinae sp. BMNH 899837 | KX035158 | Directly submitted |
| Vesperidae | Philinae | Spiniphilus spinicornis | KT781589 | Nie et al. (2017) |
| Vesperinae | Vesperus conicicollis* | JX220996 | Directly submitted | |
| Chrysomelidae | Galerucinae | Paleosepharia posticata | KY195975 | Wang & Tang (2017) |
| Diabrotica barberi | KF669870 | Coates (2014) | ||
| Diabrotica virgifera | KF658070 | Coates (2014) |
Notes.
Partial genome.
Table 2. The partition schemes and best-fitting models selected of 13 protein-coding genes in 13P26 data.
| Nucleotide sequence alignments | ||
|---|---|---|
| Subset | Subset partitions | Best model |
| Partition 1 | atp6_pos1, cox1 pos 1, cox2_pos1, cox3_pos1, cytb_pos1 | GTR + I + G |
| Partition 2 | atp6_pos2, cox1_pos2, cox2_pos2, cox3_pos2, cytb_pos2, nd3_pos2 | TVM + I + G |
| Partition 3 | atp8_pos1, atp8_pos2, nd2_pos2, nd3_pos3, nd6_pos2 | GTR + I + G |
| Partition 4 | nd1_pos1, nd4l_pos1, nd4_pos1, nd5 pos1 | GTR + I + G |
| Partition 5 | nd1_pos2, nd4_pos2, nd4l_pos2, nd5_pos2 | GTR + I + G |
| Partition 6 | nd2_pos2, nd3_pos2, nd6_pos2 | TVM + I + G |
Table 3. The partition schemes and best-fitting models selected of 12 protein-coding genes in 12P38 data.
| Nucleotide sequence alignments | ||
|---|---|---|
| Subset | Subset partitions | Best model |
| Partition 1 | atp6_pos1, cox2_pos1, cox3_pos1, cytb_pos1 | GTR + I + G |
| Partition 2 | atp6_pos2, cox2_pos2, cox3_pos2, cytb_pos2, nd3_pos2 | TVM + I + G |
| Partition 3 | atp8_pos1, atp8_pos2, nd6_pos2 | HKY + G |
| Partition 4 | cox1 pos 1 | SYM + G |
| Partition 5 | cox1_pos2 | F81 + G |
| Partition 6 | nd1_pos1, nd4l_pos1, nd4_pos1, nd5 pos1 | GTR + I + G |
| Partition 7 | nd1_pos2, nd4_pos2, nd4l_pos2, nd5_pos2, | GTR + I + G |
| Partition 8 | nd3_pos1, nd6_pos1 | GTR + I + G |
Results and Discussion
Mitogenome organization and composition
In this study, the complete mitogenomes of five species of the subfamilies Cerambycinae and Lamiinae (O. yaoshana, T. croccocincta, B. succinctor, N. carinicollis, Pterolophia sp. ZJY-2019) were determined. Structures of the five newly sequenced entire mitogenomes are shown in Figs. S1–S5. The lengths of the five mitogenomes were basically within the range of the published Cerambycidae species in the GenBank database, covering sizes between 15,503 bp in T. croccocincta to 16,063 bp in Pterolophia sp. ZJY-2019. Every mitogenome of the five species possessed similar compositional profiles and featured the typical gene arrangement and orientation that have been hypothesized for most coleopteran insects (Wolstenholme, 1992; Boore, Lavrov & Brown, 1998), with the trnW-trnC-trnY triplet (Tables S2–S6). Twenty-three genes were coded on the majority strand (J-strand), with the remaining fourteen genes coded on the minority strand (N-strand) (Figs. S1–S5). The nucleotide composition of the five longicorn beetle mitogenomes was strongly biased towards A and T, which made up 73.2% (N. carinicollis) to 79.1% (O. yaoshana) of the base pairs. A comparison of AT-skew and GC-skew showed that the AT skew of all mitogenomes was positive and the GC-skew was negative (Table 4).
Table 4. Base composition of Cerambycidae mitochondrial genomes.
| Species | A + T(%) | AT-skew | GC-skew | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mito | PCGs | rRNAs | AT-richregion | Mito | PCGs | rRNAs | AT-richregion | Mito | PCGs | rRNAs | AT-richregion | |
| O. yaoshana | 79.1 | 77.8 | 81.1 | 87.1 | 0.03 | 0.14 | 0.04 | 0.04 | 0.20 | 0.01 | 0.38 | 0.24 |
| T. croccocincta | 76.4 | 76.4 | 78.6 | 87.4 | 0.15 | 0.15 | 0.04 | 0.04 | 0.13 | 0.01 | 0.49 | 0.45 |
| B. succinctor | 75.3 | 73.2 | 78.6 | 86.2 | 0.023 | 0.17 | 0.06 | 0.02 | 0.26 | 0.02 | 0.39 | 0.32 |
| N. carinicollis | 73.2 | 71.1 | 75.7 | 80.3 | 0.10 | 0.17 | 0.16 | 0.07 | 0.18 | 0.03 | 0.36 | 0.21 |
|
Pterolophia sp.ZJY-2019 |
76.7 | 75.1 | 81.7 | 82.8 | 0.02 | 0.18 | 0.02 | 0.04 | 0.22 | 0.04 | 0.36 | 0.18 |
Protein-coding genes and codon usages
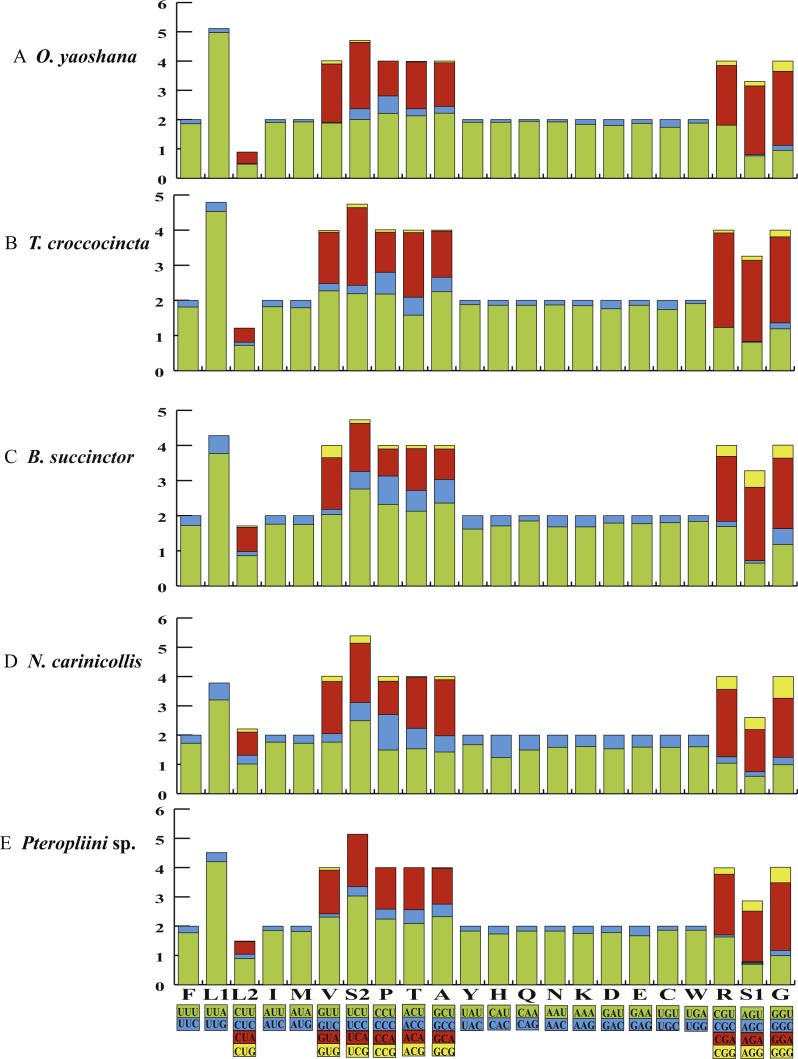
The orientations of the 13 the PCGs of the five longicorn beetles were identical to most coleopteran species (Tables S2–S6). Conventional initiation codons were assigned to the majority of the PCGs, except for nad1, which started with TTG in all five beetles. Most putative protein sequences showed typical stop codons (TAA/TAG), but the nad4 and nad5 genes of O. yaoshana, T. croccocincta, B. succinctor used a single T residue as the terminal codon. The cox1 and cox2 genes of O. yaoshana, T. croccocincta and Pterolophia sp. ZJY-2019 also used a single T residue as the terminal codon. Functional terminal codons can be produced by partial terminal codons in polycistronic transcription cleavage and polyadenylation processes (Anderson et al., 1981; Ojala, Montoya & Attardi, 1981; Du et al., 2016). The relative synonymous codon usage (RSCU) of the five Cerambycidae mitochondrial genomes was calculated (Fig. 1, Table S7). The results showed an over-utilization of A or T nucleotides in the third codon position as compared to other synonymous codons, this is normally considered to be caused by genome bias, optimum choice of tRNA usage or the benefit of DNA repair (Chai & Du, 2012; Ma et al., 2015a; Ma et al., 2015b).
Figure 1. The RSCU of five longicorn beetle mitochondrial genomes.
Codon families are provided on the x-axis along with the different combinations of synonymous codons that code for that amino acid. RSCU (relative synonymous codon usage) is defined on the Y axis.
Comparative analyses also indicated that the major customarily utilized codons and the codon usage patterns of the five samples were conservative. For instance, each of the five mitogenomes possessed UUA (Leu), AUU (Ile), UUU (Phe), and AUA (Met) as the most frequently used codons. All codons contained A or T nucleotides, indicating that the strong AT mutation bias obviously influenced the codon usage (Powell & Moriyama, 1997; Rao et al., 2011). Furthermore, the codons rich in AT encoded the most abundant amino acids, e.g., Leu (15.6–16.4%), indicating that the AT bias also influences the amino acid constituents of the proteins encoded by the mitochondrial genes (Foster, Jermiin & Hickey, 1997; Min & Hickey, 2007).
Ribosomal RNAs and transfer RNAs
The two expected rRNAs (16S rRNA and 12S rRNA) were found in the mitochondrial genomes of all five longicorn beetles. The 16S rRNA gene was situated between trnL and trnV whereas the 12S rRNA gene was between trnV and the A + T-rich region. Due to the impossibility of faultless determination by DNA sequence alone, the terminus of the rRNA genes in coleopteran mitogenomes has been presumed to stretch to the border of the flanking genes (Boore, 2001). Therefore, the 16S rRNA was presumed to fill the blank between trnL and trnV whereas the border between 12S rRNA and the putative A + T-rich region was defined based on alignments of homologous sequences of known longicorn beetles (Boore & Brown, 2000). The sizes of 16S rRNA in the five beetle mitogenomes varied from 1261 bp for N. carinicollis to 1283 bp for O. yaoshana, and the sizes of 12S rRNA ranged between 759 bp for Pterolophia sp. ZJY-2019 to 787 bp for T. croccocincta. These fit within the lengths detected in other coleopteran mitogenomes. The A + T content of the rRNA genes was the highest (81.7%) in the Pterolophia sp. ZJY-2019 mitogenome and the lowest in the N. carinicollis mitogenome (75.7%). The AT-skew of 16S rRNA and 12S rRNA showed great positivity, whereas the GC-skew was somewhat negative (Table 4), which indicated the occurrence of less As and Cs than Ts and Gs (Eyrewaker, 1997).
The 22 typical tRNAs were detected in all five species like other published longicorn beetles. All the anticodons were also highly conserved compared to other beetle species. Twenty-two tRNAs excluding trnS1 displayed the classic clover-leaf secondary structure, whereas trnS1 lacked the dihydrouridine (DHU) arm and formed a simple loop (Fig. S6). Nevertheless, this abnormal tRNA has proven to be functional, although somewhat less effective than conventional tRNAs (Steinberg & Cedergren, 1994; Hanada et al., 2001; Stewart & Beckenbach, 2003). Another unusual feature was the use of TCT as the trnS1 anticodon in Cerambycidae, whereas most arthropods use a GCT anticodon in trnS1. In many other coleopteran mitogenomes the trnS1 anticodon (TCT) can also be observed (Friedrich & Muqim, 2003; Bae et al., 2004). Mismatched pairs also exist in stems of tRNAs. For example, the mismatched pairs U-G existed in the DHU stem of trnY and trnQ; U-U existed in the T ΨC stem of trnC and in the anticodon stem of trnL1; G-U existed in acceptor stem of trnC. It has been verified that mismatched pairs can be revised via editing processes or may symbolize abnormal pairings (Negrisolo, Babbucci & Patarnello, 2011).
A + T-rich region
A large non-coding region between 12S rRNA and trnI, ranging between 861 bp for O. yaoshana to 1137 bp for Pterolophia sp. ZJY-2019, was found in the mitogenomes of the five beetles. Owing to the high AT content levels of the overall mitogenome, this non-coding element was defined as the A + T-rich region. It has been verified that the A + T-rich region harbors the origin sites and essential regulatory elements for transcription and replication (Wolstenholme, 1992; Taanman, 1999; Yukuhiro et al., 2002; Saito, Tamura & Aotsuka, 2005). The sequence of this region is relatively conserved owing to its high A + T content, and thus it is impossible to use as a molecular marker (Zhang & Hewitt, 1997). The existence of tandem repeats in the mitochondrial A + T-rich region has been observed in many coleopteran species. Some studies such as that conducted by Sheffield et al. (2008) have shown that the A + T-rich region of Trachypachus holmbergi (Coleoptera: Trachypachidae) possessed 21 similar copies of tandem repeats consisting of a 58-bp fragment. The A + T-rich region of Priasilpha obscura (Coleoptera: Phloeostichidae) is known to possess 6 tandem repeats of a 132-bp fragment and Psacothea hilaris (Coleoptera: Cerambycidae) possesses 7 identical copies of a 57 bp tandem repeat (Kim et al., 2009). In the present study, we found tandem repetitive sequences in all five newly sequenced mitogenomes. The mitogenomes of T. croccocincta and B. succinctor contained three copies of tandem repetitive sequences with lengths of 19 and 43 bp, respectively. Four tandem repeats of a 19-bp fragment were found in the mitogenome of Pterolophia sp. ZJY-2019, whereas two tandem repeats of a 25-bp fragment existed in N. carinicollis. The tandem repeats generally exhibited high A + T contents. Moreover, two poly-T stretches were detected in the mitogenome of N. carinicollis: one stretch was 16 bp in length (position: 14,880–14,895) near the 12S rRNA gene and the other stretch was 17-bp in length (position: 15,283–15,299). Previous studies have confirmed that the two poly-T stretches were structural signals for the recognition of proteins that performed a role in replication initiation (Andrews, Kubacka & Chinnery, 1999).
Intergenic regions
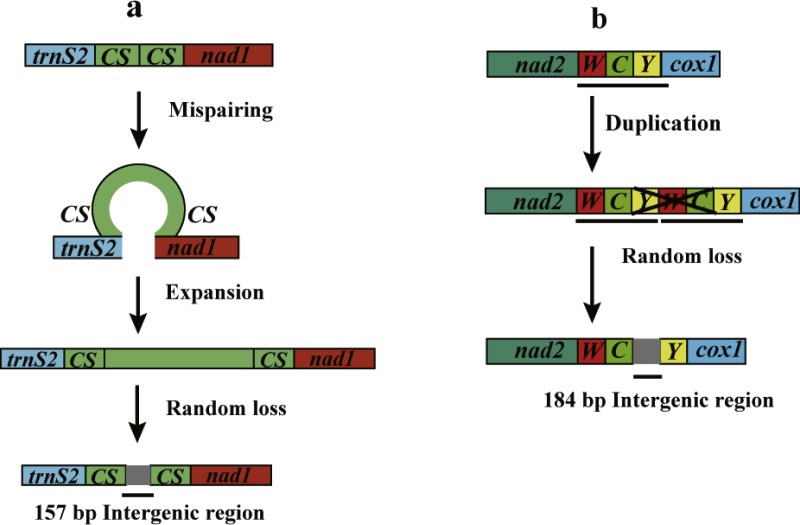
The mitogenomes of O. yaoshana, T. croccocincta, and N. carinicollis contain 6, 7, 9 non-coding intergenic spacer sequences, with total lengths of 28 bp, 28 bp, and 31 bp, respectively, whereas B. succinctor has 8 non-coding intergenic spacer sequences of 52 bp in total length. Unexpectedly, a total of 354-bp of intergenic spacer, whose elements ranged from 1 to 184 bp in length was found in the mitogenome of Pterolophia sp. ZJY-2019. The sequences are divided into 9 regions, containing two large intergenic spacers. The largest one is 184 bp long situating between trnC and trnY, and the other is 157 bp long situated between trnS2 and nad1 (Table S6). Consequently, the total length of the mitogenome of Pterolophia sp. ZJY-2019 is longer than that of other longicorn beetle species. The longer mitogenome length is due to the existence of its extended large intergenic spacers not the A + T-rich region. Previously reported tandem repeat units or additional origins of replication have been identified within this region (Dotson & Beard, 2001; Rodovalho et al., 2014). Proven by the lack of introns, rare intergenic spacers, defective terminal codons and overlapping fragments, mitogenomes characteristically show exceptional compactness of organization (Ojala, Montoya & Attardi, 1981). Nevertheless, according to Yuan et al. (2016) and Haddad et al. (2018), large non-coding regions (except the A + T-rich region) in mitochondrial genomes were observed in Pyrocoelia rufa (Coleoptera: Lampyridae) and some Hycleus species (Coleoptera: Meloidae). Coincidentally, a 5 bp consensus motif (TACTA) exists in the intergenic regions situated between trnS2 and nad1 of all five species studied here. This pentanucleotide motif is conserved across coleopteran lineages (Kim et al., 2009; Liu et al., 2018), similar to the findings that Evania appendigaster (Hymenoptera: Evaniidae) possessed a 6 bp motif ‘THACWW’ and Chilo suppressalis (Lepidoptera: Pyralidae) possessed a 7 bp motif ‘ATACTAA’, respectively (Wei et al., 2010; Gong et al., 2018).
In the mitogenome of Pterolophia sp. ZJY-2019, the large intergenic region was situated between trnS2 and nad1, which included two copies of a 22 bp long consensus sequence (TTACTAAATTTAATTAACTAAA) in both ends of the intergenic region. The formation of an intergenic region may be explained by slipped-strand mispairing (Levinson & Gutman, 1987; Du et al., 2017). Based on this theory, mispairing occurred during replication of DNA strands, and what followed next was misaligned reassociation and then replication or repair was caused by insertions of several repeat units. The resulting tandem repeat underwent random loss and/or point mutation, with only the repeat units in both extremities remaining (Fig. 2A). However, a tandem repeat was not found in the intergenic region located between trnC and trnY of Pterolophia sp. ZJY-2019. We conjectured that some errors in DNA replication can lead to tandem duplication in tRNA clusters of trnW-trnC-trnY, followed by the random loss of partial duplicated genes, and leading to the large intergenic region formed by the residues (Fig. 2B). In addition, Hua et al. (2008) suggested that the duplication-random loss model caused the rearrangements in Hemiptera. Du et al. (2017) also suggested that the duplication-random loss model was an evolutionary ancient mechanism in Coleoptera, which led to the random loss of nucleotides.
Figure 2. Putative mechanisms for formation of the two large intergenic regions (IGRs) that exist in Pterolophia sp. ZJY-2019.
(A) The slipped-strand mispairing and random loss model to explain the 157 bp-IGR between trnS2 and nad1. The CS indicates the 18 bp conservative sequence TTACTAAATTTAATTAACTAAA. (B) The duplication/random loss model to explain the 184 bp-IGR between trnC and trnY.
Consequently, compared to the original tRNAs, the residual intergenic region was not conserved. According to Du et al. (2017), four species of Hycleus genera harbored similar location and sequence of non-coding regions, which indicated that the region may serve as a latent symbol to distinguish Hycleus from the other genera. Thus, we speculated the large intergenic region of Pterolophia sp. ZJY-2019 may be a molecular feature in Pterolophia, though we were unable to adequately confirm it owing to the lack of enough samples.
Phylogenetic analyses
The phylogenetic relationships were reconstructed based on the nucleotide data (13P26) with BI and ML methods (Fig. 3). BI and ML phylogenetic analyses yielded a similar topology except for the position of Lepturinae, which was in the sister group of (Cerambycinae + Prioninae) with high values in BI, but supported as the basal group of Cerambycidae in ML analyses. The BI tree indicated that Cerambycidae split into 2 major groups (0.73): a clade of (Lepturinae + (Cerambycinae + Prioninae)) and a clade of Lamiinae. The monophyly of Lamiinae, Lepturinae and Prioninae was supported by both BI and ML analyses, whereas the monophyly of Cerambycinae was not recovered. Within the subfamily Lamiinae, the clade of (Lamiinae + (Batocera lineolata + Thyestilla gebleri)) was supported. However, Liu et al. (2018) favoured T. gebleri as the basal position of Lamiinae with a high value, and B. lineolata and Apriona swainsoni were reliably recovered as a sister group. Our results concurred with the suggestion that B. lineolata was closely related to A. swainsoni, rather than T. gebleri. The results also placed Pterolophia sp. ZJY-2019 as a sister group of all remaining Lamiinae. Moreover, our results suggested that O. yaoshana clustered with Trachypachus holmbergi, as a sister group of T. gebleri. For the relationship within Cerambycinae, M. raddei, A. oenochrous and Obrium sp. NS-2015 were gathered into one clade and most closely related to the subfamily Prioninae rather than the remaining Cerambycinae, consistent with the morphological and molecular analyses in previous reports (Liu et al., 2018).
Figure 3. Phylogenetic relationships of Cerambycidae in BI and ML analyses.
The data includes 23 species of Cerambycidae as the ingroup and three species of Chrysomelidae as the outgroup. The GenBank accession numbers of all species are also shown.
The results from the BI trees of the nucleotide dataset showed that Lepturinae cluster with the clade (Cerambycinae + Prioninae) with a high support value (Fig. 3). However, in the ML tree, a close relationship between Lamiinae and (Cerambycinae + Prioninae) was supported with 100% posterior probabilities (Fig. 3). The relationship between Cerambycinae and Prioninae is not currently understood in great detail. Prioninae were traditionally considered basal in Cerambycidae by morphology (Hatch, 1958; Svacha, Wang & Chen, 1997; Farrell, 1998). In addition, Hunt et al. (2007) and Haddad et al. (2018) pointed out that Prioninae could be placed at the basal position of Cerambycidae based on molecular phylogenetic studies. However, in BI and ML analyses of the 13P26 dataset Prioninae clustered into Cerambycinae, which was consistent with the phylogenetic position of Prioninae recovered by Raje, Ferris & Holland (2016).
The most controversial point in our results was in Cerambycinae (Fig. 3), which was represented by five different genera and rendered non-monphyletic in Prioninae. However, Cerambycinae was not supported as monophyletic based on molecular by Liu et al. (2018) and Haddad et al. (2018), but was recovered in other molecular studies (Lim et al., 2017; Liu et al., 2017).
To further discuss the monophyly of subfamilies within Cerambycidae, more samples were needed to confirm and rebuild the phylogenetic relationship of Cerambycidae using 12 protein-coding genes. The phylogenetic relationships were reconstructed based on the nucleotide data (12P38) with BI and ML methods (Fig. 4). Prioninae still clustered into Cerambycinae in BI and ML analyses of the 12P38 dataset, which agreed with the phylogenetic position of Prioninae recovered using the 13P26 dataset. In BI and ML analyses, all trees recovered the monophyly of Lamiinae (although the relationships within Lamiinae were different). The Lamiinae formed a sister group to a clade comprising Disteniidae, Prioninae, Cerambycinae and Vesperidae. The clade of Lepturinae and Necydalinae was a sister to the remaining species of Cerambycidae s. l. In addition, BI and ML analyses recovered the monophyly of Prioninae including Callipogon relictus, Dorysthenes paradoxus and Aegosoma sinicum, as proposed by Wang et al. (2019). However, BI and ML results did not support the monophyly of Cerambycinae with respect to Prioninae and Spiniphilus spinicornis (Vesperidae). It has been well accepted that Necydalinae and Lepturinae have a close relationship. The monophyly of Lepturinae was recovered in both BI and ML analyses of the 13P26 dataset. However, BI and ML trees from the 12P38 dataset returned a paraphyletic Lepturinae, due to a sister relationship between Necydalis ulmi (Necydalinae) and Brachyta interrogationis (Fig. 4).
Figure 4. Phylogenetic relationships of Cerambycidae in BI and ML analyses.
The data includes 35 species of Cerambycidae as the ingroup and three species of Chrysomelidae as the outgroup. The GenBank accession numbers of all species are also shown.
Previous studies recognized S. spinicornis as a species of Vesperinae in Cerambycidae (Napp, 1994). Nevertheless, subsequent studies considered it to belong to the subfamily Philinae of Vesperidae (Svacha, Wang & Chen, 1997; Lin & Bi, 2011; Nie et al., 2017). Further phylogenetic studies put S. spinicornis in the fairly controversial placements (Bi & Lin, 2015; Liu et al., 2018). In addition to our results, a recent molecular study also indicated a similar relationship (Liu et al., 2018).
Conclusion
In this study, we present five completely sequenced mitogenomes of Cerambycidae. The five longicorn beetle species shared similar gene organization with the insects previously reported. The gene sequences and composition of the mitogenomes were relatively conservative with no rearrangements, duplications or deletions. Two large intergenic spacers existed in Pterolophia sp. ZJY-2019. The duplication/random loss model and slipped-strand mispairing may explain the existence of these regions. The phylogenetic results inferred from mitogenomes supported the monophyly of Lamiinae and Prioninae in BI and ML analyses, whereas the Cerambycinae and Lepturinae were recovered as non-monophyletic. Although data collected thus far could not resolve the phylogenetic relationships within Cerambycidae, this study will increase the richness of the Cerambycidae genome information and assist in phylogenetic, molecular systematics and evolutionary studies of Cerambycidae.
Supplemental Information
The first circle shows the gene map (PCGs, rRNAs, tRNAs and the AT-rich region) and the genes outside the map are coded on the majority strand (J-strand) whereas the genes inside the map are coded on the minority strand (N-strand). The second circle shows the GC content and the third shows the GC skew. Both GC content and GC skew are plotted as the deviation from the average value of the total sequence.
The first circle shows the gene map (PCGs, rRNAs, tRNAs and the AT-rich region) and the genes outside the map are coded on the majority strand (J-strand) whereas the genes inside the map are coded on the minority strand (N-strand). The second circle shows the GC content and the third shows the GC skew. Both GC content and GC skew are plotted as the deviation from the average value of the total sequence.
The first circle shows the gene map (PCGs, rRNAs, tRNAs and the AT-rich region) and the genes outside the map are coded on the majority strand (J-strand) whereas the genes inside the map are coded on the minority strand (N-strand). The second circle shows the GC content and the third shows the GC skew. Both GC content and GC skew are plotted as the deviation from the average value of the total sequence.
The first circle shows the gene map (PCGs, rRNAs, tRNAs and the AT-rich region) and the genes outside the map are coded on the majority strand (J-strand) whereas the genes inside the map are coded on the minority strand (N-strand). The second circle shows the GC content and the third shows the GC skew. Both GC content and GC skew are plotted as the deviation from the average value of the total sequence.
The first circle shows the gene map (PCGs, rRNAs, tRNAs and the AT-rich region) and the genes outside the map are coded on the majority strand (J-strand) whereas the genes inside the map are coded on the minority strand (N-strand). The second circle shows the GC content and the third shows the GC skew. Both GC content and GC skew are plotted as the deviation from the average value of the total sequence.
(1): O. yaoshana (2): T. croccocincta (3): B. succinctor (4): N. carinicollis (5): Pterolophia sp. ZJY-2019 A: trnI ; B: trnQ; C: trnM; D: trnW; E: trnC; F: trnY; G: trnL (UUA); H: trnK; I: trnD; J: trnG; K: trnA; L: trnR; M: trnN; N: trnS (AGN); O: trnE; P: trnF; Q: trnH; R: trnT; S: trnP; T: trnS (UCN); U: trn L (CUA); V: trnV.
Acknowledgments
We are grateful to Wen-Yong Feng for his help in sample collection.
Funding Statement
This research was supported by the Zhejiang provincial Natural Science Foundation (Y18C040006), the National Natural Science Foundation of China (31370042), the College students’ Innovation and Entrepreneurship Project in China (No. 201810345043), the College students in Zhejiang Normal University Innovation and Entrepreneurship Plan (2018-317) for the study design, data collection and analyses. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Additional Information and Declarations
Competing Interests
Kenneth B. Storey and Jia-Yong Zhang are Academic Editors for PeerJ.
Author Contributions
Jun Wang and Xin-Yi Dai conceived and designed the experiments, performed the experiments, analyzed the data, contributed reagents/materials/analysis tools, prepared figures and/or tables, authored or reviewed drafts of the paper.
Xiao-Dong Xu and Zi-Yi Zhang analyzed the data, prepared figures and/or tables, authored or reviewed drafts of the paper.
Dan-Na Yu conceived and designed the experiments, analyzed the data, contributed reagents/materials/analysis tools, authored or reviewed drafts of the paper.
Kenneth B. Storey authored or reviewed drafts of the paper.
Jia-Yong Zhang conceived and designed the experiments, analyzed the data, contributed reagents/materials/analysis tools, authored or reviewed drafts of the paper, approved the final draft.
Data Availability
References
- Amaral et al. (2016).Amaral DT, Mitani Y, Ohmiya Y, Viviani VR. Organization and comparative analysis of the mitochondrial genomes of Bioluminescent Elateroidea (Coleoptera: Polyphaga) Gene. 2016;586(2):254–262. doi: 10.1016/j.gene.2016.04.009. [DOI] [PubMed] [Google Scholar]
- Anderson et al. (1981).Anderson S, Bankier AT, Barrell BG, Bruijin MHL, Droujn ARJ, Eperon IC, Nierlich DP, Roe BA, Sanger F, Schreier PH. Sequence and organization of the human mitochondrial genome. Nature. 1981;290(5806):457–465. doi: 10.1038/290457a0. [DOI] [PubMed] [Google Scholar]
- Andrews, Kubacka & Chinnery (1999).Andrews RM, Kubacka I, Chinnery PF. Reanalysis and revision of the Cambridge reference sequence for human mitochondrial DNA. Nature Genetics. 1999;23(2):147. doi: 10.1038/13779. [DOI] [PubMed] [Google Scholar]
- Avise et al. (1987).Avise JC, Arnold J, Ball RM, Bermingham E, Lamb T, Neigel JE, Reeb CA, Saunders NC. Intraspecific phylogeography: the mitochondrial DNA bridge between population genetics and systematics. Annual Review of Ecology and Systematics. 1987;18(1):489–522. doi: 10.1146/annurev.es.18.110187.002421. [DOI] [Google Scholar]
- Bae et al. (2004).Bae JS, Kim I, Sohn HD, Jin BR. The mitochondrial genome of the firefly, Pyrocoelia rufa: complete DNA sequence, genome organization, and phylogenetic analysis with other insects. Molecular Phylogenetics and Evolution. 2004;32(3):978–985. doi: 10.1016/j.ympev.2004.03.009. [DOI] [PubMed] [Google Scholar]
- Benson (1999).Benson G. Tandem repeats finder: a program to analyze DNA sequences. Nucleic Acids Research. 1999;27(2):573–580. doi: 10.1093/nar/27.2.573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bernt et al. (2013).Bernt M, Donath A, Jühling F, Externbrink F, Florentz C, Fritzsch G, Pütz J, Middendorf M, Stadler PF. MITOS: improved de novo metazoan mitochondrial genome annotation. Molecular Phylogenetics and Evolution. 2013;69(2):313–319. doi: 10.1016/j.ympev.2012.08.023. [DOI] [PubMed] [Google Scholar]
- Bi & Lin (2015).Bi W, Lin M. Discovery of second new species of the genus Spiniphilus Lin & Bi, and female of Heterophilus scabricollis Pu with its biological notes (Coleoptera: Vesperidae: Philinae: Philini) Zootaxa. 2015;3949(4):575–583. doi: 10.11646/zootaxa.3949.4.7. [DOI] [PubMed] [Google Scholar]
- Bologna et al. (2008).Bologna MA, Oliverio M, Pitzalis M, Mariottini P. Phylogeny and evolutionary history of the blister beetles (Coleoptera, Meloidae) Molecular Phylogenetics and Evolution. 2008;48(2):679–693. doi: 10.1016/j.ympev.2008.04.019. [DOI] [PubMed] [Google Scholar]
- Boore (1999).Boore JL. Animal mitochondrial genomes. Nucleic Acids Research. 1999;27(8):1767–1780. doi: 10.1093/nar/27.8.1767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boore (2001).Boore JL. Complete mitochondrial genome sequence of the polychaete annelid Platynereis dumerilii. Molecular Biology and Evolution. 2001;18(7):1413–1416. doi: 10.1186/1471-2164-5-67. [DOI] [PubMed] [Google Scholar]
- Boore & Brown (2000).Boore JL, Brown WM. Mitochondrial genomes of Galathealinum, Helobdella, and Platynereis: sequence and gene arrangement comparisons indicate that Pogonophora is not a Phylum and Annelida and Arthropoda are not sister taxa. Molecular Biology and Evolution. 2000;17(1):87–106. doi: 10.1093/oxfordjournals.molbev.a026241. [DOI] [PubMed] [Google Scholar]
- Boore, Lavrov & Brown (1998).Boore JL, Lavrov DV, Brown WM. Gene translocation links insects and crustaceans. Nature. 1998;392(6677):667–668. doi: 10.1038/33577. [DOI] [PubMed] [Google Scholar]
- Burland (2000).Burland TG. DNASTAR’s Lasergene sequence analysis software. In: Misener S, Krawetz SA, editors. Bioinformatics methods and protocols. Methods in molecular biology. Humana Press; Totowa: 2000. pp. 71–91. [DOI] [PubMed] [Google Scholar]
- Cameron (2014).Cameron SL. How to sequence and annotate insect mitochondrial genomes for systematic and comparative genomics research. Systematic Entomology. 2014;39(3):400–411. doi: 10.1111/syen.12071. [DOI] [Google Scholar]
- Castresana (2000).Castresana J. Selection of conserved blocks from multiple alignments for their use in phylogenetic analysis. Molecular Biology and Evolution. 2000;17(4):540–552. doi: 10.1093/oxfordjournals.molbev.a026334. [DOI] [PubMed] [Google Scholar]
- Chai & Du (2012).Chai HN, Du YZ. The complete mitochondrial genome of the pink stem borer, Sesamia inferens, in comparison with four other noctuid moths. International Journal of Molecular Sciences. 2012;13(8):10236–10256. doi: 10.3390/ijms130810236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng et al. (2016).Cheng XF, Zhang LP, Yu DN, Storey KB, Zhang JY. The complete mitochondrial genomes of four cockroaches (Insecta: Blattodea) and phylogenetic analyses within cockroaches. Gene. 2016;586:115–122. doi: 10.1016/j.gene.2016.03.057. [DOI] [PubMed] [Google Scholar]
- Chiu et al. (2016).Chiu WC, Yeh WB, Chen ME, Yang MM. Complete mitochondrial genome of Aeolesthes oenochrous (Fairmaire) (Coleoptera: Cerambycidae): an endangered and colorful longhorn beetle. Mitochondrial DNA Part A. 2016;27(1):686–687. doi: 10.3109/19401736.2014.913143. [DOI] [PubMed] [Google Scholar]
- Coates (2014).Coates BS. Assembly and annotation of full mitochondrial genomes for the corn rootworm species, Diabrotica virgifera virgifera and Diabrotica barberi (Insecta: Coleoptera: Chrysomelidae), using next generation sequence data. Gene. 2014;542(2):190–197. doi: 10.1016/j.gene.2014.03.035. [DOI] [PubMed] [Google Scholar]
- Dotson & Beard (2001).Dotson EM, Beard CB. Sequence and organization of the mitochondrial genome of the Chagas disease vector, Triatoma dimidiata. Insect Molecular Biology. 2001;10(3):205–215. doi: 10.1046/j.1365-2583.2001.00258.x. [DOI] [PubMed] [Google Scholar]
- Du et al. (2016).Du C, He SL, Song XH, Liao Q, Zhang XY, Yue BS. The complete mitochondrial genome of Epicauta chinensis (Coleoptera: Meloidae) and phylogenetic analysis among coleopteran insects. Gene. 2016;578(1):274–280. doi: 10.1016/j.gene.2015.12.036. [DOI] [PubMed] [Google Scholar]
- Du et al. (2017).Du C, Zhang LF, Lu T, Ma JN, Zeng CJ, Yue BS, Zhang XY. Mitochondrial genomes of blister beetles (Coleoptera, Meloidae) and two large intergenic spacers in Hycleus genera. BMC Genomics. 2017;18(1):698. doi: 10.1186/s12864-017-4102-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eyrewaker (1997).Eyrewaker A. Differentiating between selection and mutation bias. Genetics. 1997;147(4):1983–1987. doi: 10.1093/genetics/147.4.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fang et al. (2016).Fang J, Qian L, Xu M, Yang X, Wang B, An Y. The complete nucleotide sequence of the mitochondrial genome of the Asian longhorn beetle, Anoplophora glabripennis (Coleoptera: Cerambycidae) Mitochondrial DNA Part A. 2016;27(5):3299–3300. doi: 10.3109/19401736.2015.1015012. [DOI] [PubMed] [Google Scholar]
- Farrell (1998).Farrell BD. Inordinate fondness explained: why are there so many beetles? Science. 1998;281(5376):555–559. doi: 10.1126/science.281.5376.555. [DOI] [PubMed] [Google Scholar]
- Foster, Jermiin & Hickey (1997).Foster PG, Jermiin LS, Hickey DA. Nucleotide composition bias affects amino acid content in proteins coded by animal mitochondria. Journal of Molecular Evolution. 1997;44(3):282–288. doi: 10.1007/pl00006145. [DOI] [PubMed] [Google Scholar]
- Friedrich & Muqim (2003).Friedrich M, Muqim N. Sequence and phylogenetic analysis of the complete mitochondrial genome of the flour beetle Tribolium castanaeum. Molecular Phylogenetics and Evolution. 2003;26(3):502–512. doi: 10.1016/s1055-7903(02)00335-4. [DOI] [PubMed] [Google Scholar]
- Gao et al. (2018).Gao XY, Cai YY, Yu DN, Storey KB, Zhang JY. Characteristics of the complete mitochondrial genome of Suhpalacsa longialata (Neuroptera, Ascalaphidae) and its phylogenetic implications. PeerJ. 2018;6:e5914. doi: 10.7717/peerj.5914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gong et al. (2018).Gong R, Guo X, Ma J, Song X, Shen Y, Geng F, Yue B. Complete mitochondrial genome of Periplaneta brunnea (Blattodea: Blattidae) and phylogenetic analyses within Blattodea. Journal of Asia-Pacific Entomology. 2018;21(3):885–895. doi: 10.1016/j.aspen.2018.05.006. [DOI] [Google Scholar]
- Grant & Stothard (2008).Grant JR, Stothard P. The CG view server: a comparative genomics tool for circular genomes. Nucleic Acids Research. 2008;36(2):181–184. doi: 10.1093/nar/gkn179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guo et al. (2016).Guo K, Chen J, Xu CQ, Qiao HL, Xu R, Zhao XJ. The complete mitochondrial genome of the longhorn beetle Xylotrechus grayii (Coleoptera: Cerambycidae) Mitochondrial DNA Part A. 2016;27(3):2133–2134. doi: 10.3109/19401736.2014.982592. [DOI] [PubMed] [Google Scholar]
- Haddad et al. (2018).Haddad S, Shin S, Lemmon AR, Lemmon EM, Svacha P, Farrell B, McKenna DD. Anchored hybrid enrichment provides new insights into the phylogeny and evolution of longhorned beetles (Cerambycidae) Systematic Entomology. 2018;43(1):68–89. doi: 10.1111/syen.12257. [DOI] [Google Scholar]
- Hanada et al. (2001).Hanada T, Suzuki T, Yokogawa T, Takemoto-Hori C, Sprinzl M, Watanabe K. Translation ability of mitochondrial tRNAsSer with unusual secondary structures in an in vitro translation system of bovine mitochondria. Genes to Cells. 2001;6(12):1019–1030. doi: 10.1046/j.1365-2443.2001.00491.x. [DOI] [PubMed] [Google Scholar]
- Hatch (1958).Hatch MH. Blind beetles in the fauna of the Pacific Northwest. Proceedings of the Tenth International Congress of Entomology, 1956, 1; 1958. pp. 207–211. [DOI] [Google Scholar]
- Hua et al. (2008).Hua J, Li M, Dong P, Cui Y, Xie Q, Bu W. Comparative and phylogenomic studies on the mitochondrial genomes of Pentatomomorpha (Insecta: Hemiptera: Heteroptera) BMC Genomics. 2008;9(1):610. doi: 10.1186/1471-2164-9-610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hunt et al. (2007).Hunt T, Bergsten J, Levkanicova Z, Papadopoulou A, John OS, Wild R, Gómez-Zurita J. A comprehensive phylogeny of beetles reveals the evolutionary origins of a superradiation. Science. 2007;318(5858):1913–1916. doi: 10.1126/science.1146954. [DOI] [PubMed] [Google Scholar]
- Kim et al. (2009).Kim KG, Hong MY, Kim MJ, Im HH, Kim MI, Bae CH. Complete mitochondrial genome sequence of the yellow-spotted long-horned beetle Psacothea hilaris (Coleoptera: Cerambycidae) and phylogenetic analysis among coleopteran insects. Molecules and Cells. 2009;27(4):429–441. doi: 10.1007/s10059-009-0064-5. [DOI] [PubMed] [Google Scholar]
- Kim et al. (2018).Kim S, De Medeiros BA, Byun BK, Lee S, Kang JH, Lee B, Farrell BD. West meets East: how do rainforest beetles become circum-Pacific? Evolutionary origin of Callipogon relictus and allied species (Cerambycidae: Prioninae) in the new and old worlds. Molecular Phylogenetics and Evolution. 2018;125:163–176. doi: 10.1016/j.ympev.2018.02.019. [DOI] [PubMed] [Google Scholar]
- Kumar, Stecher & Tamura (2016).Kumar S, Stecher G, Tamura K. Mega 7: molecular evolutionary genetics analysis version 7.0 for bigger datasets. Molecular Biology and Evolution. 2016;33(7):1870–1874. doi: 10.1093/molbev/msw054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lanfear et al. (2012).Lanfear R, Calcott B, Ho SYW, Guindon S. PartitionFinder: combined selection of partitioning schemes and substitution models for phylogenetic analyses. Molecular Biology and Evolution. 2012;29(6):1695–1701. doi: 10.1093/molbev/mss020. [DOI] [PubMed] [Google Scholar]
- Lawrence & Newton (1982).Lawrence JF, Newton AF. Evolution and classification of beetles. Annual Review of Ecology and Systematics. 1982;13(1):261–290. doi: 10.1146/annurev.es.13.110182.001401. [DOI] [Google Scholar]
- Levinson & Gutman (1987).Levinson G, Gutman GA. Slipped-strand mispairing: a major mechanism for DNA sequence evolution. Molecular Biology and Evolution. 1987;4(3):203–221. doi: 10.1093/oxfordjournals.molbev.a040442. [DOI] [PubMed] [Google Scholar]
- Li et al. (2016a).Li F, Zhang H, Wang W, Weng H, Meng Z. Complete mitochondrial genome of the Japanese pine sawyer, Monochamus alternatus (Coleoptera: Cerambycidae) Mitochondrial DNA Part A. 2016a;27(2):1144–1145. doi: 10.3109/19401736.2014.936321. [DOI] [PubMed] [Google Scholar]
- Li et al. (2016b).Li W, Yang X, Qian L, An Y, Fang J. The complete mitochondrial genome of the citrus long-horned beetle, Anoplophora chinensis (Coleoptera: Cerambycidae) Mitochondrial DNA Part A. 2016b;27(6):4665–4667. doi: 10.3109/19401736.2015.1106493. [DOI] [PubMed] [Google Scholar]
- Lim et al. (2017).Lim J, Yi DK, Kim YH, Lee W, Kim S, Kang JH. Complete mitochondrial genome of Callipogon relictus Semenov (Coleoptera: Cerambycidae): a natural monument and endangered species in Korea. Mitochondrial DNA Part B. 2017;2(2):629–631. doi: 10.1080/23802359.2017.1372718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin & Bi (2011).Lin M, Bi W. A new genus and species of the subfamily Philinae (Coleoptera: Vesperidae) Zootaxa. 2011;2777(1):54–60. doi: 10.11646/zootaxa.2777.1.4. [DOI] [Google Scholar]
- Liu et al. (2017).Liu JH, Jia PF, Luo T, Wang QM. Complete mitochondrial genome of white-striped long-horned beetle, Batocera lineolata (Coleoptera: Cerambycidae) by next-generation sequencing and its phylogenetic relationship within superfamily Chrysomeloidea. Mitochondrial DNA Part B. 2017;2(2):520–521. doi: 10.1080/23802359.2017.1361797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu et al. (2018).Liu YQ, Chen DB, Liu HH, Hu HL, Bian HX, Zhang RS. The complete mitochondrial genome of the longhorn beetle Dorysthenes Paradoxus (Coleoptera: Cerambycidae: Prionini) and the implication for the phylogenetic relationships of the Cerambycidae species. Journal of Insect Science. 2018;18(2) doi: 10.1093/jisesa/iey012. Article 21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma et al. (2015a).Ma Y, He K, Yu PP, Cheng XF, Zhang JY. The complete mitochondrial genomes of three bristletails (Insecta: Archaeognatha): the paraphyly of Machilidae and insights into Archaeognathan phylogeny. PLOS ONE. 2015a;10:e0117669. doi: 10.1371/journal.pone.0117669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma et al. (2015b).Ma ZH, Yang XF, Bercsenyi M, Wu JJ, Yu Y, Wei K, Qi XF, Yang RB. Comparative mitogenomics of the genus Odontobutis (Perciformes: Gobioidei: Odontobutidae) revealed conserved gene rearrangement and high sequence variations. International Journal of Molecular Sciences. 2015b;16(10):25031–25049. doi: 10.3390/ijms161025031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Min & Hickey (2007).Min XJ, Hickey DA. DNA asymmetric strand bias affects the amino acid composition of mitochondrial proteins. DNA Research. 2007;14(5):201–206. doi: 10.1093/dnares/dsm019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Monné, Monné & Mermudes (2009).Monné ML, Monné MA, Mermudes JRM. Inventário das espécies de Cerambycinae (Insecta, Coleoptera, Cerambycidae) do Parque Nacional do Itatiaia, R.J. Brasil. Biota Neotropica. 2009;12(12):40–76. doi: 10.1590/S1676-06032012000100004. [DOI] [Google Scholar]
- Moritz, Dowling & Brown (1987).Moritz C, Dowling TE, Brown WM. Evolution of animal mitochondrial DNA: relevance for population biology and systematics. Annual Review of Ecology and Systematics. 1987;18(1):269–292. doi: 10.2307/2097133. [DOI] [Google Scholar]
- Napp (1994).Napp DS. Phylogenetic relationships among the subfamilies of Cerambycidae (Coleoptera, Chrysomeloidea) Revista Brasileira de Entomologia. 1994;38(2):265–419. [Google Scholar]
- Negrisolo, Babbucci & Patarnello (2011).Negrisolo E, Babbucci M, Patarnello T. The mitochondrial genome of the ascalaphid owlfly Libelloides macaronius, and comparative evolutionary mitochondriomics of neuropterid insects. BMC Genomics. 2011;12(1):221. doi: 10.1186/1471-2164-12-221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nie et al. (2017).Nie R, Lin M, Xue H, Bai M, Yang X. Complete mitochondrial genome of Spiniphilus spinicornis (Coleoptera: Vesperidae: Philinae) and phylogenetic analysis among Cerambycoidea. Mitochondrial DNA Part A. 2017;28(1):145–146. doi: 10.3109/19401736.2015.1111363. [DOI] [PubMed] [Google Scholar]
- Ojala, Montoya & Attardi (1981).Ojala D, Montoya J, Attardi G. tRNA punctuation model of RNA processing in human mitochondria. Nature. 1981;290(5806):470–474. doi: 10.1038/290470a0. [DOI] [PubMed] [Google Scholar]
- Perna & Kocher (1995).Perna NT, Kocher TD. Patterns of nucleotide composition at fourfold degenerate sites of animal mitochondrial genomes. Journal of Molecular Evolution. 1995;41(3):353–358. doi: 10.1007/bf01215182. [DOI] [PubMed] [Google Scholar]
- Ponomarenko & Prokin (2015).Ponomarenko AG, Prokin AA. Review of paleontological data on the evolution of aquatic beetles (Coleoptera) Paleontological Journal. 2015;49(13):1383–1412. doi: 10.1134/S0031030115130080. [DOI] [Google Scholar]
- Powell & Moriyama (1997).Powell JR, Moriyama EN. Evolution of codon usage bias in drosophila. Proceedings of the National Academy of Sciences of the United States of America. 1997;94(15):7784–7790. doi: 10.2307/2097133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Que et al. (2019).Que S, Yu A, Liu P, Jin M, Xie GA. The complete mitochondrial genome of Apriona swainsoni. Mitochondrial DNA Part B. 2019;4(1):931–932. doi: 10.1080/23802359.2019.1567284. [DOI] [Google Scholar]
- Raje, Ferris & Holland (2016).Raje KR, Ferris VR, Holland JD. Phylogenetic signal and potential for invasiveness. Agricultural and Forest Entomology. 2016;18(3):260–269. doi: 10.1111/afe.12158. [DOI] [Google Scholar]
- Rao et al. (2011).Rao Y, Wu G, Wang Z, Chai X, Nie Q, Zhang X. Mutation bias is the driving force of codon usage in the Gallus gallus genome. DNA Research. 2011;18(6):499–512. doi: 10.1093/dnares/dsr035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reid (1995).Reid CAM. A cladistic analysis of subfamilial relationships in the Chrysomelidae s. l. (Chrysomeloidea) In: Pakaluk J, Slipinski SA, editors. Biology, phylogeny and classification of Coleoptera: papers celebrating the 80th birthday of Roy A. Crowson. Muzeum i Instytut Zoologii PAN; Warzawa: 1995. pp. 559–631. [Google Scholar]
- Rodovalho et al. (2014).Rodovalho CM, Lyra ML, Ferro M, Jr MB. The mitochondrial genome of the leaf-cutter ant Atta laevigata: a mitogenome with a large number of intergenic spacers. PLOS ONE. 2014;9(5):e97117. doi: 10.1371/journal.pone.0097117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ronquist et al. (2012).Ronquist F, Teslenko M, Mark PVD, Ayres DL, Darling A, Höhna S, Larget B, Liu L, Suchard MA, Huelsenbeck JP. MrBayes 3.2: efficient Bayesian phylogenetic inference and model choice across a large model space. Systematic Biology. 2012;61(3):539–542. doi: 10.1093/sysbio/sys029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saito, Tamura & Aotsuka (2005).Saito S, Tamura K, Aotsuka T. Replication origin of mitochondrial DNA in insects. Genetics. 2005;171(4):1695–1705. doi: 10.1534/genetics.105.046243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sama et al. (2010).Sama G, Buse J, Orbach E, Friedman A, Rittner O, Chikatunov V. A new catalogue of the Cerambycidae (Coleoptera) of Israel with notes on their distribution and host plants. Munis Entomology and Zoology. 2010;5(1):1–55. doi: 10.1007/s12032-010-9513-4. [DOI] [Google Scholar]
- Sheffield et al. (2008).Sheffield NC, Song H, Cameron SL, Whiting MF. A comparative analysis of mitochondrial genomes in Coleoptera (Arthropoda: Insecta) and genome descriptions of five new beetles. Molecular Biology and Evolution. 2008;25(11):2499–2509. doi: 10.1093/molbev/msn198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simon et al. (2006).Simon C, Buckley TR, Frati F, Stewart JB, Beckenbach AT. Incorporating molecular evolution into phylogenetic analysis, and a new compilation of conserved polymerase chain reaction primers for animal mitochondrial DNA. Annual Review of Ecology Evolution and Systematics. 2006;37(1):545–579. doi: 10.1146/annurev.ecolsys.37.091305.110018. [DOI] [Google Scholar]
- Song et al. (2010).Song H, Sheffield NC, Cameron SL, Miller KB, Whiting MF. When phylogenetic assumptions are violated: base compositional heterogeneity and among-site rate variation in beetle mitochondrial phylogenomics. Systematic Entomology. 2010;35(3):429–448. doi: 10.1111/j.1365-3113.2009.00517.x. [DOI] [Google Scholar]
- Song et al. (2017).Song N, Zhang H, Yin X, Lin A, Zhai Q. The complete mitochondrial genome sequence from the longicorn beetle Obrium sp. (Coleoptera: Cerambycidae) Mitochondrial DNA Part A. 2017;28(3):326–327. doi: 10.3109/19401736.2015.1122766. [DOI] [PubMed] [Google Scholar]
- Stamatakis (2014).Stamatakis A. RAxML version 8: a tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics. 2014;30(9):1312–1313. doi: 10.1093/bioinformatics/btu033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steinberg & Cedergren (1994).Steinberg S, Cedergren R. Structural compensation in atypical mitochondrial tRNAs. Nature Structural Biology. 1994;1(8):507–510. doi: 10.1038/nsb0894-507. [DOI] [PubMed] [Google Scholar]
- Stewart & Beckenbach (2003).Stewart JB, Beckenbach AT. Phylogenetic and genomic analysis of the complete mitochondrial DNA sequence of the spotted asparagus beetle Crioceris duodecimpunctata. Molecular Phylogenetics and Evolution. 2003;26(3):513–526. doi: 10.1016/S1055-7903(02)00421-9. [DOI] [PubMed] [Google Scholar]
- Svacha, Wang & Chen (1997).Svacha P, Wang J, Chen S. Larval morphology and biology of Philus antennatus and Heterophilus punctulatus, and systematic position of the Philinae (Coleoptera: Cerambycidae: Vesperidae) Annales de la Société Entomologique de France. 1997;33:323–369. [Google Scholar]
- Taanman (1999).Taanman JW. The mitochondrial genome: structure, transcription, translation and replication. Biophysica Acta (BBA)-Bioenergetics. 1999;1410(2):103–123. doi: 10.1016/S0005-2728(98)00161-3. [DOI] [PubMed] [Google Scholar]
- Tan et al. (2017).Tan MH, Gan HM, Lee YP, Poore GC, Austin CM. Digging deeper: new gene order rearrangements and distinct patterns of codons usage in mitochondrial genomes among shrimps from the Axiidea, Gebiidea and Caridea (Crustacea: Decapoda) PeerJ. 2017;5:e2982. doi: 10.7717/peerj.2982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Timmermans & Vogler (2012).Timmermans MJ, Vogler AP. Phylogenetically informative rearrangements in mitochondrial genomes of Coleoptera, and monophyly of aquatic elateriform beetles (Dryopoidea) Molecular Phylogenetics and Evolution. 2012;63(2):299–304. doi: 10.1016/j.ympev.2011.12.021. [DOI] [PubMed] [Google Scholar]
- Wang et al. (2019).Wang J, Lan DY, Dai XY, Yu DN, Storey KB, Zhang JY. The complete mitochondrial genome of Xystrocera globosa (Coleoptera: Cerambycidae) and its phylogeny. Mitochondrial DNA Part B. 2019;4(1):1647–1649. doi: 10.1080/23802359.2019.1605852. [DOI] [Google Scholar]
- Wang & Tang (2017).Wang Q, Tang G. Genomic and phylogenetic analysis of the complete mitochondrial DNA sequence of walnut leaf pest Paleosepharia posticata (Coleoptera: Chrysomeloidea) Journal of Asia-Pacific Entomology. 2017;20(3):840–853. doi: 10.1016/j.aspen.2017.05.010. [DOI] [Google Scholar]
- Wang et al. (2016).Wang YT, Liu YX, Tong XL, Ren QP, Jiang GF. The complete mitochondrial genome of the longhorn beetle, Massicus raddei. Mitochondrial DNA Part A. 2016;27(1):209–211. doi: 10.3109/19401736.2014.880892. [DOI] [PubMed] [Google Scholar]
- Wei et al. (2010).Wei SJ, Tang P, Zheng LH, Shi M, Chen XX. The complete mitochondrial genome of Evania appendigaster (Hymenoptera: Evaniidae) has low A+T content and a long intergenic spacer between atp8 and atp6. Molecular Biology Reports. 2010;37(4):1931–1942. doi: 10.1007/s11033-009-9640-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wolstenholme (1992).Wolstenholme DR. Animal mitochondrial DNA: structure and evolution. International Review of Cytology. 1992;141:173–216. doi: 10.1016/S0074-7696(08)62066-5. [DOI] [PubMed] [Google Scholar]
- Yang et al. (2017).Yang J, Cao LJ, Geng YX, Wei DF, Chen M. Determination of the complete mitochondrial genome of Thyestilla gebleri and comparative analysis of the mitochondrial genome in Cerambycidae. Chinese Journal of Applied Entomology. 2017;54(5):755–766. doi: 10.7679/j.issn.2095-1353.2017.092. (In Chinese) [DOI] [Google Scholar]
- Yuan et al. (2016).Yuan M, Zhang Q, Zhang L, Guo Z, Liu Y, Shen Y. High-level phylogeny of the Coleoptera inferred with mitochondrial genome sequences. Molecular Phylogenetics and Evolution. 2016;104:99–111. doi: 10.1016/j.ympev.2016.08.002. [DOI] [PubMed] [Google Scholar]
- Yukuhiro et al. (2002).Yukuhiro K, Sezutsu H, Itoh M, Shimizu K, Banno Y. Significant levels of sequence divergence and gene rearrangements have occurred between the mitochondrial genomes of the wild mulberry silkmoth, Bombyx mandarina, and its close relative, the domesticated silkmoth, Bombyx mori. Molecular Biology and Evolution. 2002;19(8):1385–1389. doi: 10.1093/oxfordjournals.molbev.a004200. [DOI] [PubMed] [Google Scholar]
- Zhang & Hewitt (1997).Zhang DX, Hewitt GM. Insect mitochondrial control region: a review of its structure, evolution and usefulness in evolutionary studies. Biochemical Systematics and Ecology. 1997;25(2):99–120. doi: 10.1016/s0305-1978(96)00042-7. [DOI] [Google Scholar]
- Zhang et al. (2008).Zhang JY, Zhou CF, Gai YH, Song DX, Zhou KY. The complete mitochondrial genome of Parafronurus youi (Insecta: Ephemeroptera) and phylogenetic position of the Ephemeroptera. Gene. 2008;424(1–2):18–24. doi: 10.1016/j.gene.2008.07.037. [DOI] [PubMed] [Google Scholar]
- Zhang et al. (2018a).Zhang LP, Cai YY, Yu D.N. Storey KB, Zhang JY. Gene characteristics of the complete mitochondrial genomes of Paratoxodera polyacantha and Toxodera hauseri (Mantodea: Toxoderidae) PeerJ. 2018a;6:e4595. doi: 10.7717/peerj.4595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang et al. (2019).Zhang LP, Ma Y, Yu DN, Storey KB, Zhang JY. The mitochondrial genomes of Statilia maculata and S. nemoralis (Mantidae: Mantinae) with different duplications of trnR genes. International Journal of Biological Macromolecules. 2019;121:839–845. doi: 10.1016/j.ijbiomac.2018.10.038. [DOI] [PubMed] [Google Scholar]
- Zhang et al. (2018b).Zhang LP, Yu DN, Storey KB, Cheng HY, Zhang JY. Higher tRNA gene duplication in mitogenomes of praying mantises (Dictyoptera, Mantodea) and the phylogeny within Mantodea. International Journal of Biological Macromolecules. 2018b;111:787–795. doi: 10.1016/j.ijbiomac.2018.01.016. [DOI] [PubMed] [Google Scholar]
- Zhang et al. (2018c).Zhang SQ, Che LH, Li Y, Liang D, Pang H, Ślipiński A, Zhang P. Evolutionary history of Coleoptera revealed by extensive sampling of genes and species. Nature Communications. 2018c;9(1) doi: 10.1038/s41467-017-02644-4. Article 205. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
The first circle shows the gene map (PCGs, rRNAs, tRNAs and the AT-rich region) and the genes outside the map are coded on the majority strand (J-strand) whereas the genes inside the map are coded on the minority strand (N-strand). The second circle shows the GC content and the third shows the GC skew. Both GC content and GC skew are plotted as the deviation from the average value of the total sequence.
The first circle shows the gene map (PCGs, rRNAs, tRNAs and the AT-rich region) and the genes outside the map are coded on the majority strand (J-strand) whereas the genes inside the map are coded on the minority strand (N-strand). The second circle shows the GC content and the third shows the GC skew. Both GC content and GC skew are plotted as the deviation from the average value of the total sequence.
The first circle shows the gene map (PCGs, rRNAs, tRNAs and the AT-rich region) and the genes outside the map are coded on the majority strand (J-strand) whereas the genes inside the map are coded on the minority strand (N-strand). The second circle shows the GC content and the third shows the GC skew. Both GC content and GC skew are plotted as the deviation from the average value of the total sequence.
The first circle shows the gene map (PCGs, rRNAs, tRNAs and the AT-rich region) and the genes outside the map are coded on the majority strand (J-strand) whereas the genes inside the map are coded on the minority strand (N-strand). The second circle shows the GC content and the third shows the GC skew. Both GC content and GC skew are plotted as the deviation from the average value of the total sequence.
The first circle shows the gene map (PCGs, rRNAs, tRNAs and the AT-rich region) and the genes outside the map are coded on the majority strand (J-strand) whereas the genes inside the map are coded on the minority strand (N-strand). The second circle shows the GC content and the third shows the GC skew. Both GC content and GC skew are plotted as the deviation from the average value of the total sequence.
(1): O. yaoshana (2): T. croccocincta (3): B. succinctor (4): N. carinicollis (5): Pterolophia sp. ZJY-2019 A: trnI ; B: trnQ; C: trnM; D: trnW; E: trnC; F: trnY; G: trnL (UUA); H: trnK; I: trnD; J: trnG; K: trnA; L: trnR; M: trnN; N: trnS (AGN); O: trnE; P: trnF; Q: trnH; R: trnT; S: trnP; T: trnS (UCN); U: trn L (CUA); V: trnV.
Data Availability Statement
The following information was supplied regarding data availability:
Five new sequenced mitochondrial genomes are available at GenBank: MK863507–MK863511.