Abstract
Purpose
Ambient light is both a stimulus for visual function and a regulator of photoreceptor physiology. However, it is not known if light can regulate any aspect of photoreceptor development. The purpose of this study was to investigate whether ambient light is required for the development of mouse rod photoreceptors.
Methods
Newborn mouse pups (C57BL/6) were reared in either cyclic light (LD) or constant dark (DD). Pups were collected at postnatal day (P)5, P10, P17, or P24. We performed retinal morphometric and cell death analysis at P5, P10, and P17. Rhodopsin expression was assessed using immunofluorescence, Western blot, and quantitative RT-PCR analysis. Electroretinograms were performed at P17 and P24. Radioimmunoassay and ELISA were used to follow changes in thyroid hormone levels in the serum and vitreous.
Results
In the DD pups, the outer nuclear layer was significantly thinner at P10 and there were higher numbers of apoptotic cells at P5 compared to the LD pups. Rhodopsin expression was lower at P10 and P17 in DD pups. Electroretinogram a-waves were reduced in amplitude at P17 in the DD pups. The DD animals had lower levels of circulating thyroid hormones at P10. Light-mediated changes in thyroid hormones occur as early as P5, as we detected lower levels of total triiodothyronine in the vitreous from the DD animals. Drug-induced developmental hypothyroidism resulted in lower rhodopsin expression at P10.
Conclusions
Our data demonstrate that light exposure during postnatal development is required for rod photoreceptor development and that this effect could be mediated by thyroid hormone signaling.
Keywords: light, retinal development, retina, rhodopsin, photoreceptors, thyroid
Photoreceptors are specialized sensory neurons with elaborate outer segments within which reside the light-sensitive opsins necessary to initiate phototransduction.1–4 In mice, almost 97% of the photoreceptors are made up of rhodopsin-expressing rod photoreceptors, and the rest are cones.5,6 Though the actual numbers, ratios, and the spatial arrangements of the photoreceptors vary across species, a conserved feature is the protracted time of photoreceptor development after they exit the cell cycle to when they become fully functional.7–9 A combination of transcription factor activities and external signals including hormones and growth factors is known to coordinate the precise timing of photoreceptor development.10
Several studies have suggested that environmental light regulates both photoreceptor function and physiology.11–14 Retinal pigment epithelium–mediated shedding and phagocytosis of the rod outer segment is a light-dependent process. It has been demonstrated that the transport of rhodopsin to the rod outer segment is under circadian control as well.15–18 Whether light is one of the external factors that can influence photoreceptor development is a question that remains to be answered.
Thyroid hormones play an important role in tissue development, metabolic homeostasis, reproduction, and a wide range of other biological processes.19–21 Deficiency in thyroid hormones during fetal and early postnatal development impairs photoreceptor development.22 Thyroid hormone levels are under circadian control, and hence environmental light would have an important role in their developmental regulatory roles.23,24 In this study, we demonstrate that early light exposure affects rod photoreceptor development. As thyroid hormones have been implicated in photoreceptor development, we also evaluated the levels of these hormones in mice that were reared in cyclic light and constant dark.
We compared rhodopsin expression, rod outer segment morphology and function, and thyroid hormone levels in mouse pups reared under cyclic light (LD) or in constant darkness (DD). In DD as compared to LD animals, rods had shorter outer segments, reduced photoreceptor electrophysiology, and lower levels of circulating thyroid hormones. These results demonstrate that light exposure is critical during the first postnatal weeks and that light deprivation can have a profound effect on photoreceptor morphology and function, via regulation of thyroid hormones.
Methods
Animals
All animal studies were approved by the Institutional Animal Care and Use Committee of the Cleveland Clinic and conformed to current National Institutes of Health guidelines (Guide for the Care and Use of Laboratory Animals in Research, 8th ed., 2011). The animals were cared for in accordance with the ARVO Statement for the Use of Animals in Ophthalmic and Vision Research. C57BL/6 mice (6–8 weeks old) were purchased from the Jackson Laboratory (Bar Harbor, ME, USA). Breeding cages containing one male and one female were set under regular LD housing (14 hours/10 hours, LD), and vaginal plugs were checked the following day to ensure embryonic day of pregnancy and marked as E1 (embryonic day 1). At birth (postnatal day [P]1), cages were randomly assigned to either LD or DD housing, and the DD cages were transferred to an adjacent animal housing room without lights. The DD animals were maintained in constant darkness throughout the experimental period, and husbandry was accomplished using infrared night vision goggles. Pups were euthanized at P5, P10, P17, or P24 using isoflurane (Piramal Critical Care, Inc., Bethlehem, PA, USA) in an appropriate inhalation chamber. Eyes were enucleated for histology, immunofluorescence, Western blot, and RT-PCR as described in the following sections.
Light Microscopy and Retinal Morphometric Analysis
Eyes from P17 and P24 pups were fixed overnight in a 2% paraformaldehyde–2% glutaraldehyde mixture, dehydrated using ethanol, and embedded in Epon as described in detail previously.25 Images were acquired using a light microscope (Zeiss, Thornwood, NY, USA). Outer nuclear layer (ONL) thickness was measured at constant intervals, starting from the optic nerve head (ONH) and extending toward dorsal and ventral ora serrata.
Immunofluorescence
Eyes from P10, P17, and P24 pups were fixed in 4% paraformaldehyde for 1 hour at room temperature (RT), cryoprotected sequentially in 15%, 20%, and 30% sucrose. Sections 10 μm thick were blocked with 4% bovine serum albumin (BSA) and 0.1% Triton X-100 in PBS for 1 hour at RT. Sections were incubated overnight at 4°C with anti-rhodopsin antibody (ab98887; Abcam, Cambridge, MA, USA) in blocking solution and Alexa Fluor 594 conjugated secondary antibody (Life Technologies, Carlsbad, CA, USA). Sections were analyzed and imaged using a Leica laser scanning confocal microscope (TCSSP2; Leica, Exton, PA, USA).
Western Blot
Eyes were removed and retinas were isolated in cold PBS. Pooled retinas (three or four) were lysed in lysis buffer composed of 20 mM Tris-HCl, pH 8, 150 mM NaCl, 2.5 mM EDTA, 10% glycerol, 0.5% Triton X-100, 0.01% Nonidet P-40 substitute, and protease inhibitor cocktail tablets (Roche Diagnostics, Indianapolis, IN, USA). Blots were probed with antibodies against rhodopsin (ab98887; Abcam, Cambridge, MA, USA) and b-tubulin (Cell Signaling Technology, Danvers, MA, USA). Blots were digitized using a densitometer (GS800; Bio-Rad, Hercules, CA, USA), and densitometry quantification of the bands was done using Quantity One 4.6.8 software (Bio-Rad).
Reverse Transcription Polymerase Chain Reaction
Total RNA was extracted from neural retina using RNeasy mini kit (Qiagen, Germantown, MD, USA) according to the manufacturer's instructions. The Verso cDNA kit (Thermo Scientific, Rockford, IL, USA) was used to generate cDNA for RT-PCR from 250 ng total RNA. Gene-specific primers were used. Gene specific primers were used as described for Rho,26,27 Crx,28 rod arrestin,29 Rp1l1 F(5′-CACCGGCCAAGAAGATAACCT-3′), R(5′-TGAGACAGCTCATCCATTAGGG-3′), Aldh1a1 F(5′-CACTGGCCGACTTGAAGATT-3′), R(5′-ACCTCCTCAGTTGCAGGGTT-3′), Aldh1a3 F(5′-CTTGGAGGTCAAGTTCACCAA-3′), R(5′-TCGCTCGGTCCCTTTCTACAA-3′), Cyp26a1,30 Cyp26b1,30 Cyp26c1 F(5′-ATACCTTTGAACAGCTGGTGGA-3′), R(5′-CCCTAGCGCTGTTAATAATCA-3′), Cntf F(5′-TGGCTAGCAAGGAAGATTCG-3′), R(5′-TCAGTCATCTCACTCCAGCG-3′), Dio2,31 Egf F(5′-GAACTGTCAGCCAGGTCCTC-3′), R(5′-GGTGATTTGTTCCATCTGGG-3′), and for β-Actin F(5′-TTCTTTGCAGCTCCTTCGTT-3′), R(5′-ATGGAGGGGAATACAGCC-3′). Real-time PCR was performed on the 7900 HT fast real-time PCR system (Life Technologies, Carlsbad, CA, USA) and using a Bullseye EvaGreen qPCR master mix (Midwest Scientific, Valley Park, MO, USA). Relative fold changes in mRNA expression were determined using the comparative Ct method (2−ΔΔCt method).
Electroretinography
For ERG studies, P17 and P24 pups were dark-adapted overnight and anesthetized with ketamine (80 mg/kg) and xylazine (16 mg/kg). Electroretinograms were obtained using a published stimulation and recording procedure.32
Radioimmunoassay and ELISA
Serum thyroxine (T4) and triiodothyronine (T3) concentrations were measured by a solid-phase radioimmunoassay (Applied Biosciences, College Station, TX, USA) adapted for mice, using 25 and 50 μL serum, respectively. Total T3 concentrations in vitreous at P5 were measured using mouse/rat T3 ELISA kit (Calbiotech, Spring Valley, CA, USA). Assays were performed by following manufacturer's guidelines, and absorbance was recorded at 450 nm. For P5 mice, vitreous was extracted immediately after enucleating the eyes, and vitreous from all the pups in same litter was pooled to obtain a sufficient amount of sample to perform ELISA assays.
Drug Treatment
For inducing developmental hypothyroidism in newborn pups, nursing mothers were treated with methimazole (MMI; 0.05–0.08% wt/vol; Sigma-Aldrich Corp., St. Louis, MO, USA), sodium perchlorate monohydrate (PM) (1.0% wt/vol; Sigma-Aldrich Corp.), and sucrose (5.0% wt/vol; Fisher Scientific, Waltham, MA, USA) in the drinking water from P1 (day of birth) until P10.33 A placebo group received sucrose (5.0% wt/vol) in drinking water. Pups were killed on P10, and retinal samples were collected for rhodopsin analysis using Western blot and immunofluorescence. Total T3 concentrations in serum at P10 were measured using mouse/rat T3 ELISA kit (Calbiotech).
Statistical Analysis
Statistical analysis was performed using SigmaPlot 10.0 software (Systat Software, San Jose, CA, USA). A Student's t-test was performed to evaluate the difference between control group (LD) and constant darkness group (DD). P values less than 0.05 were considered statistically significant. Data were plotted as the mean ± standard error of the mean (SEM). Electroretinogram data were analyzed using 2-way repeated measures ANOVA.
Results
Effect of Dark Rearing on Outer Nuclear Layer Thickness and Photoreceptor Apoptosis
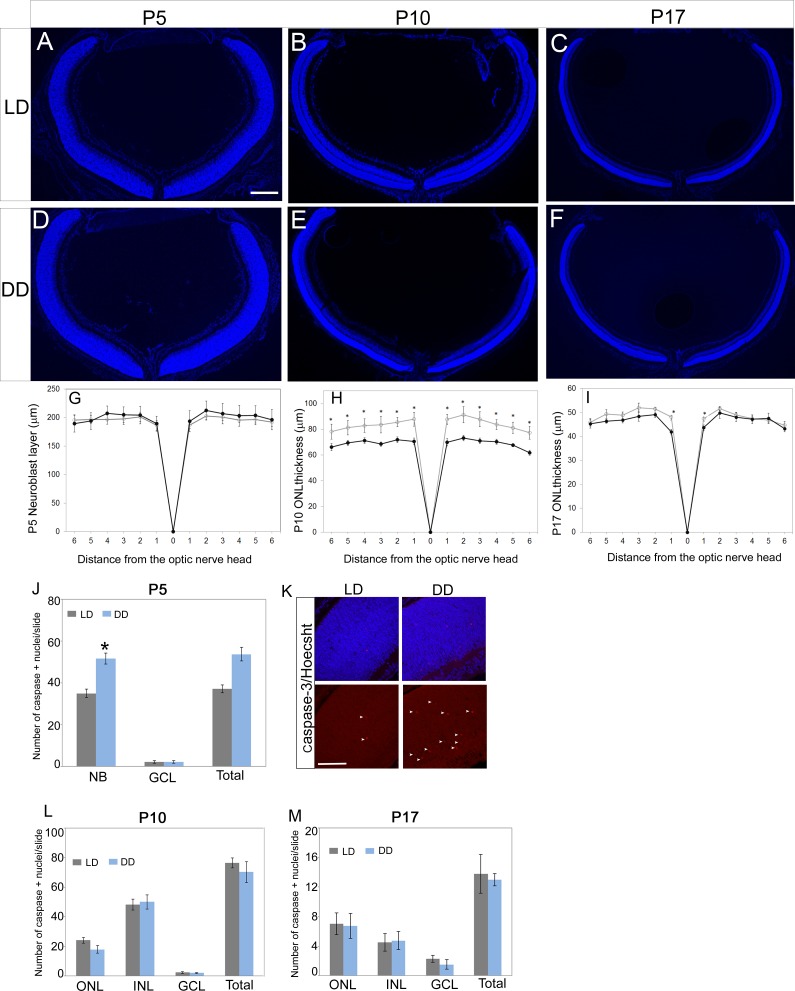
To determine whether postnatal light deprivation affects rod development, we compared retinal sections obtained from LD and DD animals. At P5, LD and DD animals have a neuroblast layer that is comparable in thickness (Figs. 1A, 1D, 1G). However, at P10 we noted a significant reduction in ONL thickness of DD animals (Figs. 1B, 1E, 1H). This decrease in ONL thickness was more prominent near the ONH and was less severe toward the periphery (Fig. 1H). At P17, the difference was less pronounced and was not significant (except for the region closest to ONH) (Figs. 1C, 1F, 1I). These data suggest that light has no effect on the initial generation of the retinal progenitors but is required after P5 for the maintenance of the rod photoreceptor numbers. The inner nuclear layer (INL) thickness was comparable between LD and DD retina at P10 (Supplementary Fig. S1).
Figure 1.
Retinal thickness measurements and apoptotic cell counts. Images (×5) of retinal sections around optic nerve region from P5 (A, D), P10 (B, E), and P17 (C, F) pups from LD and DD groups. (G–I) Spider plots represent the thickness measurements for neuroblastic layer at P5 (G) and ONL at P10 (H) and P17 (I). Outer nuclear layer thickness at P10 was significantly decreased in DD pups compared to the LD pups. Pups from the LD group were raised in normal cyclic light-dark (LD) conditions (14 hours light, 8 hours dark), and pups from the DD group were raised in constant darkness (24 hours dark) from their day of birth. Outer nuclear layer thickness was measured at constant intervals (230 or 320 μm), starting from the optic nerve head (ONH) and extending toward the periphery. (J–M) Bar graph represents the total number of active caspase-3–positive cells per retinal section across different retinal layers at P5 (J), P10 (L), and P17 (M). (K) Active caspase-3 (red)-positive nuclei in P5 LD and DD retinas are shown by arrowheads. In this study, four to six eyes from at least two different litters were used in each group, and two to five sections around the optic nerve region from each eye were imaged and used for quantitative analysis. Data points indicate average ± SEM. Student's t-test was performed between the groups at each data point, and the asterisk indicates statistical significance with P < 0.05. NB, neuroblastic layer; ONL, outer nuclear layer; GCL, ganglion cell layer. Scale bars: 300 μm (A); 25 μm (K).
At P5, the comparable thickness of the neuroblast layer between LD and DD animals suggested that differences in the rate of photoreceptor progenitor proliferation could not account for the differences observed in ONL thickness at P10. To examine the possibility that dark rearing altered the rate of developmental apoptosis, we quantitated the total number of active caspase-3–positive apoptotic cell nuclei at P5, P10, and P17. In comparison to the P5 LD pups, the DD pups had significantly higher numbers of active caspase-3–positive nuclei in the neuroblast layer (Figs. 1J, 1K). At P10, though there was not a significant difference in the total numbers of apoptotic cells between the LD and DD retinas (Fig. 1L), overall there were fewer apoptotic cells in the DD retina at P10. We detected very few apoptotic cells in the LD retina at P17, and others have reported similar findings.34,35 There was no detectable difference between LD and DD retina at P17 and P24 (Fig. 1M). These results indicate that increased rate of early developmental apoptosis of the photoreceptor cells is an important contributor to the decreased ONL thickness of DD pups at P10.
Expression of Rhodopsin Is Altered by Light
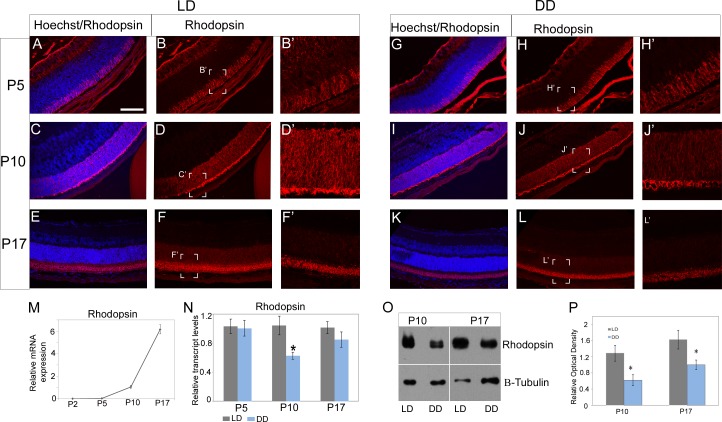
As the ONL was most affected in the DD pups, we next wanted to determine if light had any effect on rhodopsin levels. We analyzed for the transcript using quantitative RT-PCR and for the protein using Western blot and immunofluorescence. Developmentally, rhodopsin expression is very minimal at P2, increases gradually from P5 to P10, and then increases rapidly between P10 and P17 (Fig. 2M). We did not see any change in transcript levels at P5; however, we did observe a significant (∼40%) reduction in rhodopsin mRNA expression at P10 in DD pups compared to the LD pups (Fig. 2N). Although rhodopsin mRNA was slightly decreased at P17 in the DD retina, the difference was not significant (Fig. 2N). However, using an antibody to visualize rhodopsin in the retinal sections, we did detect lower immunofluorescence signal in the DD retinas at P10 and at P17 (Figs. 2C, 2E, 2I, 2K). We validated these observations by using Western blots to quantitate the total amount of rhodopsin between the LD and DD retinas. There was a ∼50% reduction in protein levels of DD as compared to LD retinas at P10, and this difference persisted at P17 (Figs. 2O, 2P). These results indicate that postnatal light is required for the continued expression of rhodopsin in the developing retina.
Figure 2.
Rhodopsin immunofluorescence and protein quantification. Rhodopsin immunofluorescence (red) at P5 (A, B, B'; G, H, H'), P10 (C, D, D'; I, J, J') and P17 (E, F, F'; K, L, L') in the LD and DD pups. (M) Expression of rhodopsin mRNA during development. Rhodopsin mRNA expression is very minimal at P2, increases gradually from P5 to P10, and then increases rapidly between P10 and P17. (N) Real-time qPCR analysis of rhodopsin mRNA between LD and DD pups at P5, P10, and P17. (O) Western blot image of rhodopsin and β-tubulin (housekeeping protein) at P10 and P17. (P) Quantification of rhodopsin protein optical density normalized to β-tubulin levels. Data points indicate average ± SEM. n = 6 in each group, and each sample (n) consisted of two to four pooled retinas. Samples were collected from at least three different litters in each group. Student's t-test was performed between the groups at each data point, and asterisk indicates statistical significance with P < 0.05. Scale bar: 50 μm.
Dark Rearing Results in Delayed Growth of Rod Outer Segment
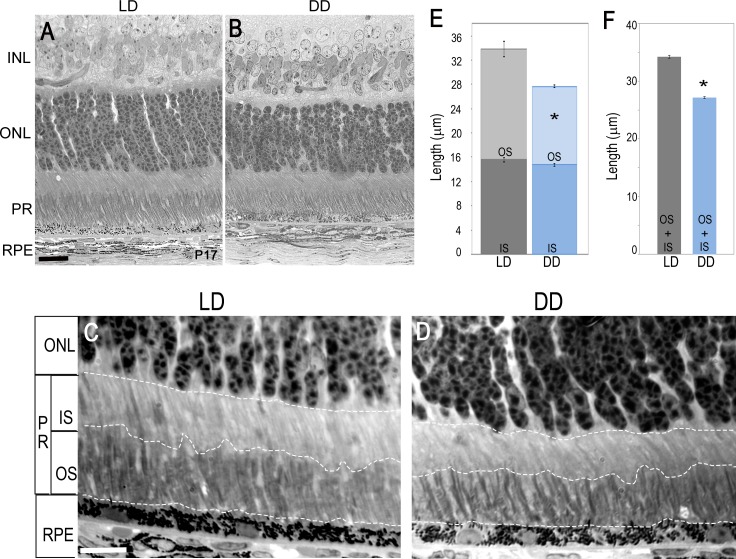
Rod photoreceptors mature postnatally, and during this process of maturation there is an increase in the length of the outer segment (OS) that occurs at an exponential rate between P10 and P17.36 Although the rod outer segments continue to elongate after P17, the change in OS length is not significantly different at P17 versus P24. Since DD pups had a lower amount of rhodopsin protein compared to LD pups, we wanted to assess if reduction in rhodopsin protein was associated with disruption in rod OS development. We measured rod outer segment (ROS) and rod inner segment (RIS) length from LD and DD pups at P17 and P24. As expected, overall the rod photoreceptors were shorter in DD pups and, interestingly, most of the difference was in the length of the OS (Figs. 3A–E). At P17, the combined length of ROS and RIS was ∼20% shorter in DD pups as compared to the LD pups (Figs. 3E, 3F), whereas reduction in only ROS was ∼30% in DD pups as compared to the LD pups. We did not detect any obvious changes between LD and DD groups at P24 (data not shown). This suggests that during retinal development, light exposure is required for the growth of the ROS. This effect seems to be specific to the eyes, as we did not observe any significant differences in the weights of the animals at P17 (Supplementary Fig. S2).
Figure 3.
Light micrographs of retina from LD and DD animals. Light microscope images of retinal sections of LD (A) and DD (B) pups at P17. Outer nuclear layer appears normal; however. the PR layer is shorter in length in DD animals compared to LD animals at P17. (C, D) Representative high-magnification images of retinal cross sections at P17 from LD (C) and DD (D) animals used for measuring outer segment and inner segment length. Dotted white lines indicate the demarcation for measurement of IS and OS separately. (E, F) Quantification of OS and IS length analysis at P17. Overall the PR length is shorter (F), and most of the defect is seen in the outer segment (E). Each group consisted of five or six eyes. Data points indicate average ± SEM. Student's t-test was performed between the groups, and asterisk indicates statistical significance with P < 0.05. INL, inner nuclear layer; ONL, outer nuclear layer; PR, photoreceptors; OS, outer segment; IS, inner segment; RPE, retinal pigment epithelium. Scale bar: 20 μm.
Dark Rearing Decreases Retinal ERG Amplitude
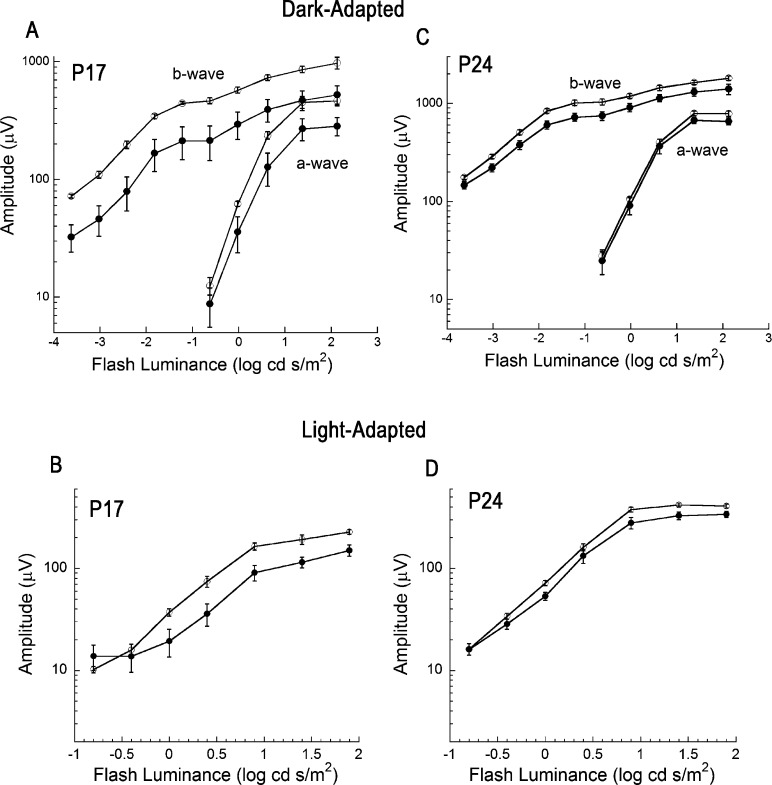
To evaluate if dark rearing had any functional consequences we performed ERGs at P17 and P24; ERGs are not present at P10 or earlier.37 At P17, the dark-adapted ERG a-waves were significantly lower in DD as compared to LD mice (Fig. 4A). This correlates well with our data indicating a difference in ROS length as well as reduced rhodopsin levels at P17. By P24, a-waves were comparable between LD and DD animals (Fig. 4C), consistent with our observation that LD and DD retinas had comparable morphology at this age. We noted a different impact of light exposure history on the ERG b-wave, which is generated by depolarizing bipolar cells in response to photoreceptor activity.38 At P17, b-waves were significantly reduced in DD as compared to LD animals; this was true for dark-adapted conditions (Fig. 4A) where responses are dominated by rod pathway activity and for light-adapted conditions (Fig. 4B) reflecting activity of the cone pathway. At P24, the magnitude of the difference was decreased partially for the dark-adapted (Fig. 4B) and light-adapted (Fig. 4D) ERG b-wave, although DD b-waves were statistically reduced as compared to those of LD mice. These results suggest that dark rearing might delay the developmental maturation of synaptic connectivity, although there were no obvious defects in the outer plexiform layer where photoreceptors and bipolar cells synapse.
Figure 4.
Electroretinogram recordings from LD and DD mice. Summary luminance-response functions obtained from LD (open circles) and DD (filled circles) mice obtained under dark-adapted (A, C) and light-adapted (B, D) stimulus conditions at P17 (A, B) and at P24 (C, D). At P17, the amplitudes of the a-wave (F1,7 = 10.6; P < 0.02), b-wave (F1,7 = 23.3; P < 0.01), and cone ERG (F1,7 = 16.1; P < 0.01) were significantly reduced in DD (n = 4) as compared to LD (n = 4) mice (A, B). At P24, there was no difference in a-wave amplitude (F1,12 = 2.4; NS) between DD (n = 5) and LD (n = 9) mice (C), but the b-wave was reduced, under both dark-adapted (F1,12 = 6.9; P < 0.05) and light-adapted (F1,12 = 6.8; P < 0.05) conditions (C, D). Data points indicate average ± SEM.
Dark Rearing Results in Lower Levels of Circulating Thyroid Hormones
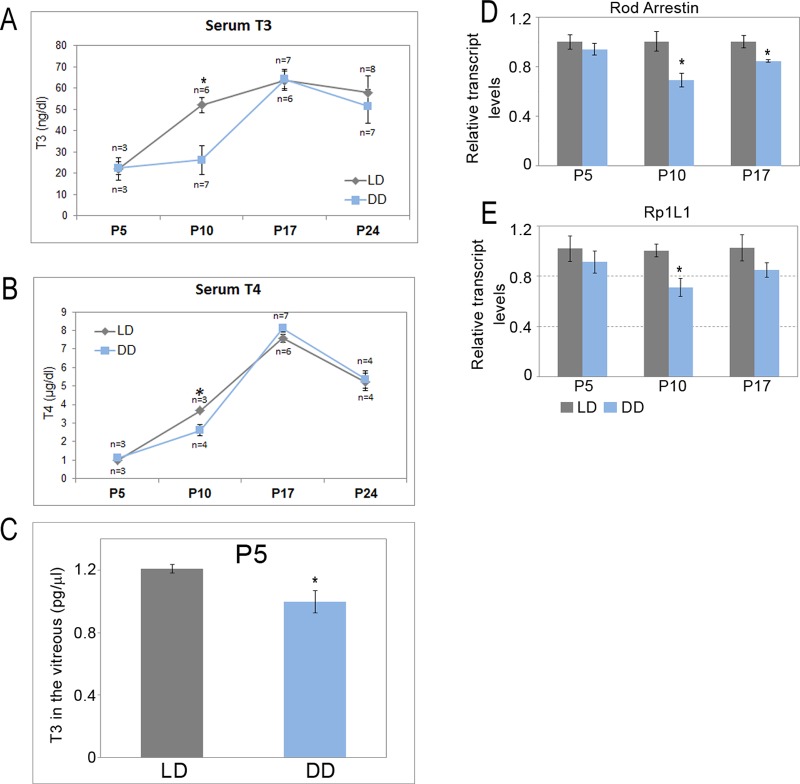
Thyroid hormones have been implicated in regulating retinal neurogenesis and development. In rats with induced hypothyroidism from early gestation, there was a marked decrease in growth of the eye as well as proliferation of all retinal cell types.22 Circadian control of thyroid hormone levels is well known,23,24 and we explored whether light could be a mediator of this process. Hence we wondered if animals maintained in constant darkness had changes in their thyroid hormones. We measured the serum levels of both T3 (triiodothyronine), the active form of the hormone, as well as the prohormone T4 (thyroxine) in LD and DD pups at P5, P10, P17, and P24. The levels of total T3 and total T4 were significantly lower in the DD animals at P10 (P = 0.038 and 0.032, respectively) (Figs. 5A, 5B). At P17 and P24, the levels were indistinguishable between the LD and DD animals. It is well known that the circulating levels of thyroid hormones could be very different than the local levels, and it is the local levels that will ultimately dictate the fate of the cells. Therefore, we also measured the levels of T3 in the vitreous from the P5 animals; this is a more accurate representation of the local differences in the thyroid hormone levels. Just as we would have predicted, the total T3 levels were significantly lower in the vitreous of DD animals as compared to LD (P = 0.039) (Fig. 5C). These data support our observations that thyroid hormones are likely required for the growth and development of the rod photoreceptors and also provide an explanation for the increased photoreceptor apoptosis observed in DD pups at P5.
Figure 5.
Quantitation of thyroid hormone levels in the vitreous and serum of LD and DD animals. Serum T3 (A) and serum T4 (B) levels in LD and DD pups at P5, P10, P17, and P24 quantified using radioimmunoassay. (C) Total T3 levels in vitreous at P5. n = 3–7, and each serum and vitreous sample was pooled from two to eight pups. Real-time qPCR analysis of rod arrestin (D) and Rp1l1 (E) mRNA expression between LD and DD pups at P5, P10, and P17. For real-time qPCR analysis, n = 3, and each sample was pooled from four retinas from at least two different litters. Data points indicate average ± SEM. Student's t-test was performed between the groups at each data point, and asterisk indicates statistical significance with P < 0.05.
Light Alters the Expression of Retinal Target Genes of Thyroid Hormones
Thyroid hormones can govern proliferation and cell fate determination by regulating transcription of various target genes.39 In the dark-reared animals, the levels of T3 and T4 in the serum were lower compared to those in LD animals. We wanted to investigate if lower levels of thyroid hormones resulted in transcriptional changes of rod-specific genes and could provide an explanation for the defects seen in rod photoreceptors development. Using quantitative RT-PCR, we measured the relative transcript levels for Crx, rod arrestin, and Rp1l1, known targets of thyroid hormones that are involved in rod development.39 The Crx transcript was unaltered in the DD retina (Supplementary Fig. S3). In comparison, rod arrestin was significantly reduced at P10 and P17 in DD retinas compared to the LD retinas (P = 0.031 and 0.042, respectively) (Fig. 5D). Rp1l1 was significantly decreased at P10 but not at P17 (P = 0.011) (Fig. 5E). These results suggest that both light and thyroid hormones have a transient role during development and are required during specific stages of retinal development. This effect seems to be specific, as we did not see any changes in Nr2e3 and Nrl transcripts at any of the ages tested, though these genes have been shown to be critical for rod development (data not shown). Our data further confirm that light regulates photoreceptor development by altering thyroid hormone signaling in the retina.
Drug-Induced Hypothyroidism Results in Lower Rhodopsin Levels
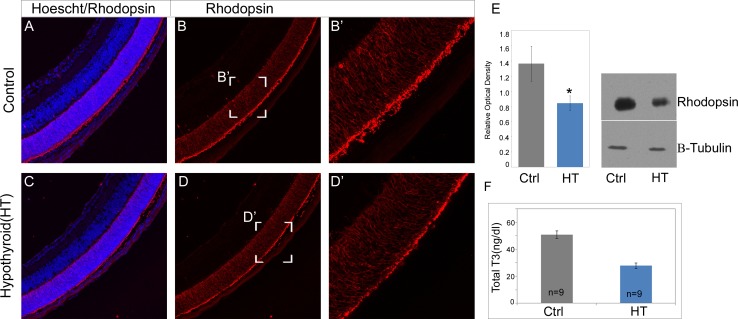
To further validate our findings that lower levels of thyroid hormones could result in decreased rhodopsin expression, we measured rhodopsin levels in P10 neonatal pups with induced hypothyroidism. We observed lower immunofluorescence signal in the hypothyroid retinas at P10 (Figs. 6A–D). We validated these observations by using Western blots to quantitate the total amount of rhodopsin between placebo (control) and hypothyroid groups. There was a ∼38% reduction in rhodopsin protein levels in the hypothyroid retinas as compared to placebo retinas at P10 (Fig. 6E). These data suggest that some of the effects we observe on rod photoreceptor development could be mediated by light-regulated thyroid hormones.
Figure 6.
Rhodopsin immunofluorescence and protein quantification in hypothyroid pups. Rhodopsin immunofluorescence (red) in placebo (control) and hypothyroid pups at P10 (A–D). Western blot image of rhodopsin and β-tubulin (housekeeping protein) and quantification of relative optical density at P10 (E). Serum T3 levels in placebo (Ctrl) and hypothyroid pups at P10 (F). Data points indicate average ± SEM. n = 6–9 in each group from two to four different litters in each group. Student's t-test was performed between the groups at each data point, and asterisk indicates statistical significance with P < 0.05.
Discussion
In the present study, we demonstrate that cyclic light–dark exposure during postnatal development is critical for the maturation and function of rod photoreceptors. Animals that are maintained in constant darkness for the first 2 weeks after birth have shorter outer segments, lower amounts of rhodopsin, and reduced photoreceptor function. We found lower levels of circulating T3 and T4 in dark-reared pups. Similarly, in animals with induced hypothyroidism we detected lower amounts of rhodopsin compared to the control animals, suggesting that some of the phenotypes observed in dark-reared animals could be due to impaired thyroid hormone signaling. Although mice do not open their eyelids until P10, many of the changes we observed occur as early as P5.40 This is similar to results of other studies demonstrating that neonatal pups have the ability to sense and respond to light even before eyelid opening.41 We and others have previously demonstrated an embryonic role for environmental light in regulating survival of the retinal neurons.41–44 Thus, light could regulate developmental processes well before the animals have opened their eyelids.
In the DD pups we detected higher numbers of apoptotic cells at P5 and a decrease in ONL thickness at P10. These data suggest a critical requirement for light in photoreceptor development during the postnatal weeks. Though we have not confirmed the identity of the apoptotic cells at P5, we assume those to be the newly born photoreceptor cells based on the observed phenotypes in the DD retina. It is worth noting that despite the increased number of apoptotic cells in the DD retina, we saw a minimal difference in ONL thickness between the LD and DD groups at P17 and P24. Based on these data, we think that in the DD pups the timing of the normal developmental apoptosis is disrupted and occurs precociously compared to what is seen in the control animals. A possible explanation could be the involvement of the circadian clock in this process. Circadian clock genes are known to play important regulatory roles for timing of cell cycle progression and developmental neurogenesis.45–47 Rhythmic light–dark cycles can regulate the phagocytic activity of the RPE and the transport of rhodopsin to the ROS.15–18 Our data now demonstrate a requirement for cyclic light for the development and maturation of the rod photoreceptors.
We also detected lower levels of thyroid hormones in the DD animals compared to LD animals, suggesting that light could be regulating the thyroid hormone levels in the neonatal animals. Similarly we observed lower rhodopsin levels in animals with induced hypothyroidism. Thus our data suggest a role for light and thyroid hormones in the development of photoreceptors. Several studies have suggested a role for thyroid hormone in photoreceptor development. In rats with induced hypothyroidism there was a marked decrease in growth of the eye as well as proliferation of all retinal cell types.22 In rodents, alteration of thyroid hormone levels can affect expression of rhodopsin as well as other opsins.48–52 Our analysis suggests that light exposure is crucial in the neonatal animals to regulate the levels of thyroid hormones. We speculate that the link between light exposure and thyroid hormone regulation may involve the maturation of the hypothalamic–pituitary–thyroid axis, and this will be worth investigating in the future. Thyroid hormones can affect growth and development, thus raising the possibility that the observed changes in DD animals are due to growth retardation.53 We weighed the LD and DD pups and did not detect any significant weight difference between the groups, making it less likely that growth retardation could result in the observed changes (Supplementary Fig. S2). So far our data suggest that light-mediated changes in thyroid hormones have a specific role for the development and maturation of rod photoreceptors that may be completely different from the effect in adults.
Light could have a pleiotropic effect, and conceivably some of the phenotypes that we observed in the dark-reared animals are the consequence of multiple signaling pathways being affected by light. Soluble factors like epidermal growth factor (EGF), ciliary neurotrophic factor (CNTF), and retinoic acid have been implicated in rod photoreceptor survival and differentiation.54 Activation of these signaling pathways can have both a stimulatory and an inhibitory effect on rod photoreceptor differentiation. In cell culture systems, removal of EGF from the culture media can lead to accelerated differentiation of the rod progenitors to rod photoreceptors.55 Similarly, in rodents, CNTF acts as a negative regulator of rod differentiation.56 We did not observe any significant differences between LD and DD groups for Egf and Cntf expression at P10 (Supplementary Fig. S4).56,57 Conversely, retinoic acid promotes rod development, and in the presence of retinoic acid, rhodopsin-negative cells accelerate their differentiation into rhodopsin-positive cells.58 To understand if light deprivation is altering synthesis or degradation of retinoic acid, we analyzed mRNA expression of retinoic acid-synthesizing enzymes aldehyde dehydrogenase (Aldh1a1 and Aldh1a3) and retinoic acid-degrading enzymes Cyp26a1, Cyp26b1, and Cyp26c1 at P10.30 We did not observe any significant differences between LD and DD groups for any of the retinoic acid-associated enzymes (Supplementary Fig. S4). However, we did observe a significant decrease in retinal Dio2 expression in the DD group compared to the LD group at P10 (Supplementary Fig. S4). Type II deiodinase (DIO2) is required for converting prohormone T4 into biologically active form T3.31 This finding strengthens our claim that thyroid hormone signaling is required for light-dependent rod photoreceptor development.
The DD animals had a lower ERG response at both P17 and P24. Lower ERG responses could be due to depletion in renewal of 11-cis retinal. Photoreceptor function depends on a continuous supply of 11-cis retinal and isomerization to the all-trans configuration in the presence of light.59 It has been well documented that inability to replenish 11-cis retinal results in reduced photosensitivity.60 To our knowledge, there is a paucity of literature addressing how dark rearing would affect developmental expression of 11-cis retinal and enzymes associated with its renewal.
In summary, our findings show that light deprivation during the postnatal period alters rod photoreceptor development and that some of these changes could be mediated by thyroid hormones. Disruption in retinal function has been observed in clinical cases of depression, seasonal affective disorder, hypothyroid patients, in children born to hypothyroid mothers, and in children born to mothers whose first or second trimester was during winter months when photoperiods are shorter.61–66 These results highlight the importance of early environmental light exposure in driving long-term adaptive changes in photoreceptor development and function.
Supplementary Material
Acknowledgments
Supported by Cleveland Clinic Foundation startup funds, a Research to Prevent Blindness Career development award, an E. Matilda Ziegler award (SR), a Challenge Grant from Research to Prevent Blindness to Cleveland Clinic Lerner College of Medicine of Case Western Reserve University, and the Foundation Fighting Blindness.
Disclosure: O. Sawant, None; A.M. Horton, None; M. Shukla, None; M.E. Rayborn, None; N.S. Peachey, None; J.G. Hollyfield, None; S. Rao, None
References
- 1.Ng L, Lu A, Swaroop A, Sharlin DS, Swaroop A, Forrest D. Two transcription factors can direct three photoreceptor outcomes from rod precursor cells in mouse retinal development. J Neurosci. 2011;31:11118–11125. doi: 10.1523/JNEUROSCI.1709-11.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Morris AC, Fadool JM. Studying rod photoreceptor development in zebrafish. Physiol Behav. 2005;86:306–313. doi: 10.1016/j.physbeh.2005.08.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Altshuler D, Lillien L. Control of photoreceptor development. Curr Opin Neurobiol. 1992;2:16–22. doi: 10.1016/0959-4388(92)90155-e. [DOI] [PubMed] [Google Scholar]
- 4.Morrow EM, Furukawa T, Cepko CL. Vertebrate photoreceptor cell development and disease. Trends Cell Biol. 1998;8:353–358. doi: 10.1016/s0962-8924(98)01341-5. [DOI] [PubMed] [Google Scholar]
- 5.Jeon CJ, Strettoi E, Masland RH. The major cell populations of the mouse retina. J Neurosci. 1998;18:8936–8946. doi: 10.1523/JNEUROSCI.18-21-08936.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Carter-Dawson LD, LaVail MM. Rods and cones in the mouse retina. I. Structural analysis using light and electron microscopy. J Comp Neurol. 1979;188:245–262. doi: 10.1002/cne.901880204. [DOI] [PubMed] [Google Scholar]
- 7.Morrow EM, Belliveau MJ, Cepko CL. Two phases of rod photoreceptor differentiation during rat retinal development. J Neurosci. 1998;18:3738–3748. doi: 10.1523/JNEUROSCI.18-10-03738.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Watanabe T, Raff MC. Rod photoreceptor development in vitro: intrinsic properties of proliferating neuroepithelial cells change as development proceeds in the rat retina. Neuron. 1990;4:461–467. doi: 10.1016/0896-6273(90)90058-n. [DOI] [PubMed] [Google Scholar]
- 9.Treisman JE, Morabito MA, Barnstable CJ. Opsin expression in the rat retina is developmentally regulated by transcriptional activation. Mol Cell Biol. 1988;8:1570–1579. doi: 10.1128/mcb.8.4.1570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Cepko CL, Austin CP, Yang X, Alexiades M, Ezzeddine D. Cell fate determination in the vertebrate retina. Proc Natl Acad Sci U S A. 1996;93:589–595. doi: 10.1073/pnas.93.2.589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Liu C, Peng M, Laties AM, Wen R. Preconditioning with bright light evokes a protective response against light damage in the rat retina. J Neurosci. 1998;18:1337–1344. doi: 10.1523/JNEUROSCI.18-04-01337.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Penn JS, Anderson RE. Effect of light history on rod outer-segment membrane composition in the rat. Exp Eye Res. 1987;44:767–778. doi: 10.1016/s0014-4835(87)80040-4. [DOI] [PubMed] [Google Scholar]
- 13.Penn JS, Williams TP. Photostasis: regulation of daily photon-catch by rat retinas in response to various cyclic illuminances. Exp Eye Res. 1986;43:915–928. doi: 10.1016/0014-4835(86)90070-9. [DOI] [PubMed] [Google Scholar]
- 14.Organisciak DT, Xie A, Wang HM, Jiang YL, Darrow RM, Donoso LA. Adaptive changes in visual cell transduction protein levels: effect of light. Exp Eye Res. 1991;53:773–779. doi: 10.1016/0014-4835(91)90113-s. [DOI] [PubMed] [Google Scholar]
- 15.LaVail MM. Rod outer segment disc shedding in relation to cyclic lighting. Exp Eye Res. 1976;23:277–280. doi: 10.1016/0014-4835(76)90209-8. [DOI] [PubMed] [Google Scholar]
- 16.LaVail MM. Circadian nature of rod outer segment disc shedding in the rat. Invest Ophthalmol Vis Sci. 1980;19:407–411. [PubMed] [Google Scholar]
- 17.Besharse JC, Hollyfield JG, Rayborn ME. Photoreceptor outer segments: accelerated membrane renewal in rods after exposure to light. Science. 1977;196:536–538. doi: 10.1126/science.300504. [DOI] [PubMed] [Google Scholar]
- 18.Besharse JC, Hollyfield JG. Turnover of mouse photoreceptor outer segments in constant light and darkness. Invest Ophthalmol Vis Sci. 1979;18:1019–1024. [PubMed] [Google Scholar]
- 19.Kim B. Thyroid hormone as a determinant of energy expenditure and the basal metabolic rate. Thyroid. 2008;18:141–144. doi: 10.1089/thy.2007.0266. [DOI] [PubMed] [Google Scholar]
- 20.Morreale de Escobar G, Obregon MJ. Escobar del Rey F. Role of thyroid hormone during early brain development. Eur J Endocrinol. 2004;151(suppl 3):U25–U37. doi: 10.1530/eje.0.151u025. [DOI] [PubMed] [Google Scholar]
- 21.Wagner MS, Wajner SM, Maia AL. The role of thyroid hormone in testicular development and function. J Endocrinol. 2008;199:351–365. doi: 10.1677/JOE-08-0218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Sevilla-Romero E, Munoz A, Pinazo-Duran MD. Low thyroid hormone levels impair the perinatal development of the rat retina. Ophthalmic Res. 2002;34:181–191. doi: 10.1159/000063885. [DOI] [PubMed] [Google Scholar]
- 23.Russell W, Harrison RF, Smith N, et al. Free triiodothyronine has a distinct circadian rhythm that is delayed but parallels thyrotropin levels. J Clin Endocrinol Metab. 2008;93:2300–2306. doi: 10.1210/jc.2007-2674. [DOI] [PubMed] [Google Scholar]
- 24.Lucke C, Hehrmann R, von Mayersbach K. von zur Muhlen A. Studies on circadian variations of plasma TSH, thyroxine and triiodothyronine in man. Acta Endocrinol. 1977;86:81–88. doi: 10.1530/acta.0.0860081. [DOI] [PubMed] [Google Scholar]
- 25.Xi Q, Pauer GJ, Ball SL, et al. Interaction between the photoreceptor-specific tubby-like protein 1 and the neuronal-specific GTPase dynamin-1. Invest Ophthalmol Vis Sci. 2007;48:2837–2844. doi: 10.1167/iovs.06-0059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Chen YC, Gatchel JR, Lewis RW, et al. Gcn5 loss-of-function accelerates cerebellar and retinal degeneration in a SCA7 mouse model. Hum Mol Genet. 2012;21:394–405. doi: 10.1093/hmg/ddr474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Aguila M, Bevilacqua D, McCulley C, et al. Hsp90 inhibition protects against inherited retinal degeneration. Hum Mol Genet. 2014;23:2164–2175. doi: 10.1093/hmg/ddt613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Jia L, Oh EC, Ng L, et al. Retinoid-related orphan nuclear receptor RORbeta is an early-acting factor in rod photoreceptor development. Proc Natl Acad Sci U S A. 2009;106:17534–17539. doi: 10.1073/pnas.0902425106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Bibb LC, Holt JK, Tarttelin EE, et al. Temporal and spatial expression patterns of the CRX transcription factor and its downstream targets. Critical differences during human and mouse eye development. Hum Mol Genet. 2001;10:1571–1579. doi: 10.1093/hmg/10.15.1571. [DOI] [PubMed] [Google Scholar]
- 30.Alfano G, Conte I, Caramico T, et al. Vax2 regulates retinoic acid distribution and cone opsin expression in the vertebrate eye. Development. 2011;138:261–271. doi: 10.1242/dev.051037. [DOI] [PubMed] [Google Scholar]
- 31.Medina MC, Molina J, Gadea Y, et al. The thyroid hormone-inactivating type III deiodinase is expressed in mouse and human beta-cells and its targeted inactivation impairs insulin secretion. Endocrinology. 2011;152:3717–3727. doi: 10.1210/en.2011-1210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Samuels IS, Bell BA, Sturgill-Short G, et al. Myosin 6 is required for iris development and normal function of the outer retina. Invest Ophthalmol Vis Sci. 2013;54:7223–7233. doi: 10.1167/iovs.13-12887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Ma H, Thapa A, Morris L, Redmond TM, Baehr W, Ding XQ. Suppressing thyroid hormone signaling preserves cone photoreceptors in mouse models of retinal degeneration. Proc Natl Acad Sci U S A. 2014;111:3602–3607. doi: 10.1073/pnas.1317041111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Mervin K, Stone J. Developmental death of photoreceptors in the C57BL/6J mouse: association with retinal function and self-protection. Exp Eye Res. 2002;75:703–713. doi: 10.1006/exer.2002.2063. [DOI] [PubMed] [Google Scholar]
- 35.Young RW. Cell death during differentiation of the retina in the mouse. J Comp Neurol. 1984;229:362–373. doi: 10.1002/cne.902290307. [DOI] [PubMed] [Google Scholar]
- 36.LaVail MM. Kinetics of rod outer segment renewal in the developing mouse retina. J Cell Biol. 1973;58:650–661. doi: 10.1083/jcb.58.3.650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Bakall B, Marmorstein LY, Hoppe G, Peachey NS, Wadelius C, Marmorstein AD. Expression and localization of bestrophin during normal mouse development. Invest Ophthalmol Vis Sci. 2003;44:3622–3628. doi: 10.1167/iovs.03-0030. [DOI] [PubMed] [Google Scholar]
- 38.Pardue MT, Peachey NS. Mouse b-wave mutants. Doc Ophthalmol. 2014;128:77–89. doi: 10.1007/s10633-013-9424-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Liu Y, Fu L, Chen DG, Deeb SS. Identification of novel retinal target genes of thyroid hormone in the human WERI cells by expression microarray analysis. Vis Res. 2007;47:2314–2326. doi: 10.1016/j.visres.2007.04.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Findlater GS, McDougall RD, Kaufman MH. Eyelid development fusion and subsequent reopening in the mouse. J Anat. 1993;183(pt 1):121–129. [PMC free article] [PubMed] [Google Scholar]
- 41.Rao S, Chun C, Fan J, et al. A direct and melanopsin-dependent fetal light response regulates mouse eye development. Nature. 2013;494:243–246. doi: 10.1038/nature11823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Tian N, Copenhagen DR. Visual deprivation alters development of synaptic function in inner retina after eye opening. Neuron. 2001;32:439–449. doi: 10.1016/s0896-6273(01)00470-6. [DOI] [PubMed] [Google Scholar]
- 43.Tian N, Copenhagen DR. Visual stimulation is required for refinement of ON and OFF pathways in postnatal retina. Neuron. 2003;39:85–96. doi: 10.1016/s0896-6273(03)00389-1. [DOI] [PubMed] [Google Scholar]
- 44.Melamed E, Frucht Y, Vidauri J, Uzzan A, Rosenthal J. Effect of postnatal light deprivation on the ontogenesis of dopamine neurons in rat retina. Brain Res. 1986;391:280–284. doi: 10.1016/0165-3806(86)90293-2. [DOI] [PubMed] [Google Scholar]
- 45.Bouchard-Cannon P, Mendoza-Viveros L, Yuen A, Kaern M, Cheng HY. The circadian molecular clock regulates adult hippocampal neurogenesis by controlling the timing of cell-cycle entry and exit. Cell Rep. 2013;5:961–973. doi: 10.1016/j.celrep.2013.10.037. [DOI] [PubMed] [Google Scholar]
- 46.Schnell A, Chappuis S, Schmutz I, et al. The nuclear receptor REV-ERBalpha regulates Fabp7 and modulates adult hippocampal neurogenesis. PLoS One. 2014;9:e99883. doi: 10.1371/journal.pone.0099883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Borgs L, Beukelaers P, Vandenbosch R, et al. Period 2 regulates neural stem/progenitor cell proliferation in the adult hippocampus. BMC Neurosci. 2009;10:30. doi: 10.1186/1471-2202-10-30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Roberts MR, Srinivas M, Forrest D, Morreale de Escobar G, Reh TA. Making the gradient: thyroid hormone regulates cone opsin expression in the developing mouse retina. Proc Natl Acad Sci U S A. 2006;103:6218–6223. doi: 10.1073/pnas.0509981103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Pessoa CN, Santiago LA, Santiago DA, et al. Thyroid hormone action is required for normal cone opsin expression during mouse retinal development. Invest Ophthalmol Vis Sci. 2008;49:2039–2045. doi: 10.1167/iovs.07-0908. [DOI] [PubMed] [Google Scholar]
- 50.Glaschke A, Weiland J, Del Turco D, Steiner M, Peichl L, Glosmann M. Thyroid hormone controls cone opsin expression in the retina of adult rodents. J Neurosci. 2011;31:4844–4851. doi: 10.1523/JNEUROSCI.6181-10.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Lu A, Ng L, Ma M, et al. Retarded developmental expression and patterning of retinal cone opsins in hypothyroid mice. Endocrinology. 2009;150:1536–1544. doi: 10.1210/en.2008-1092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Kelley MW, Turner JK, Reh TA. Ligands of steroid/thyroid receptors induce cone photoreceptors in vertebrate retina. Development. 1995;121:3777–3785. doi: 10.1242/dev.121.11.3777. [DOI] [PubMed] [Google Scholar]
- 53.Burstein PJ, Draznin B, Johnson CJ, Schalch DS. The effect of hypothyroidism on growth, serum growth hormone, the growth hormone-dependent somatomedin, insulin-like growth factor, and its carrier protein in rats. Endocrinology. 1979;104:1107–1111. doi: 10.1210/endo-104-4-1107. [DOI] [PubMed] [Google Scholar]
- 54.Levine EM, Fuhrmann S, Reh TA. Soluble factors and the development of rod photoreceptors. Cell Mol Life Sci. 2000;57:224–234. doi: 10.1007/PL00000686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Reh TA. Cellular interactions determine neuronal phenotypes in rodent retinal cultures. J Neurobiol. 1992;23:1067–1083. doi: 10.1002/neu.480230811. [DOI] [PubMed] [Google Scholar]
- 56.Elliott J, Cayouette M, Gravel C. The CNTF/LIF signaling pathway regulates developmental programmed cell death and differentiation of rod precursor cells in the mouse retina in vivo. Dev Biol. 2006;300:583–598. doi: 10.1016/j.ydbio.2006.09.002. [DOI] [PubMed] [Google Scholar]
- 57.Kelley MW, Turner JK, Reh TA. Regulation of proliferation and photoreceptor differentiation in fetal human retinal cell cultures. Invest Ophthalmol Vis Sci. 1995;36:1280–1289. [PubMed] [Google Scholar]
- 58.Wallace VA, Jensen AM. IBMX. taurine and 9-cis retinoic acid all act to accelerate rhodopsin expression in postmitotic cells. Exp Eye Res. 1999;69:617–627. doi: 10.1006/exer.1999.0741. [DOI] [PubMed] [Google Scholar]
- 59.Kiser PD, Golczak M, Maeda A, Palczewski K. Key enzymes of the retinoid (visual) cycle in vertebrate retina. Biochim Biophys Acta. 2012;1821:137–151. doi: 10.1016/j.bbalip.2011.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Rohrer B, Goletz P, Znoiko S, et al. Correlation of regenerable opsin with rod ERG signal in Rpe65-/- mice during development and aging. Invest Ophthalmol Vis Sci. 2003;44:310–315. doi: 10.1167/iovs.02-0567. [DOI] [PubMed] [Google Scholar]
- 61.Yang MB, Rao S, Copenhagen DR, Lang RA. Length of day during early gestation as a predictor of risk for severe retinopathy of prematurity. Ophthalmology. 2013;120:2706–2713. doi: 10.1016/j.ophtha.2013.07.051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Szabo Z, Antal A, Tokaji Z, et al. Light therapy increases visual contrast sensitivity in seasonal affective disorder. Psychiatry Res. 2004;126:15–21. doi: 10.1016/j.psychres.2003.12.013. [DOI] [PubMed] [Google Scholar]
- 63.Hebert M, Beattie CW, Tam EM, Yatham LN, Lam RW. Electroretinography in patients with winter seasonal affective disorder. Psychiatry Res. 2004;127:27–34. doi: 10.1016/j.psychres.2004.03.006. [DOI] [PubMed] [Google Scholar]
- 64.Lam RW, Beattie CW, Buchanan A, Mador JA. Electroretinography in seasonal affective disorder. Psychiatry Res. 1992;43:55–63. doi: 10.1016/0165-1781(92)90141-o. [DOI] [PubMed] [Google Scholar]
- 65.Mastaglia FL, Black JL, Collins DW, Gutteridge DH, Yuen RW. Slowing of conduction in visual pathway in hypothyroidism. Lancet. 1978;1:387–388. doi: 10.1016/s0140-6736(78)91110-8. [DOI] [PubMed] [Google Scholar]
- 66.Mirabella G, Westall CA, Asztalos E, Perlman K, Koren G, Rovet J. Development of contrast sensitivity in infants with prenatal and neonatal thyroid hormone insufficiencies. Pediatr Res. 2005;57:902–907. doi: 10.1203/01.PDR.0000157681.38042.B4. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.