Abstract
The objectives of this study were to (1) assess the acceptability, feasibility, and safety of delivering a pulmonary rehabilitation (PR) “taster” session to patients hospitalized with acute exacerbations of chronic obstructive pulmonary disease; (2) evaluate the changes in patient knowledge and readiness to commence PR; and (3) make recommendations for future intervention iterations. Acceptability was measured by the proportion of patients that accepted to participate. Feasibility was measured by the proportion of eligible participants. Knowledge was evaluated using the modified versions of the Understanding COPD (UCOPD) and Bristol COPD Knowledge (BCKQ) questionnaires. Readiness to commence PR was measured by a modified version of the Readiness to Change Exercise Questionnaire. All measures were delivered pre- and post-intervention. Thirty-one of 34 eligible individuals were able to be approached. Prospective acceptability was low, with 24 individuals declining the intervention, 1 being discharged without making a decision, and only 6 participating. Positive median change was recorded in the modified UCOPD questionnaire (+8), but not the BCKQ (0). Three of the patients were already in the action phase pre-intervention, with all but one in that phase post-intervention. The delivery of a PR “taster” session was not prospectively acceptable to a large portion of patients and only feasible with modifications to the original protocol.
Keywords: COPD, pulmonary rehabilitation, acute exacerbation
Introduction
Severe acute exacerbations of chronic obstructive pulmonary disease (AECOPD) are associated with high rates of rehospitalization,1 worsened symptom severity,2 decreased exercise tolerance and physical activity,3–7 and negative impact on skeletal muscle,8,9 quality of life,7 mental health,3,10 and ability to self-care.11,12 It has been argued that there is a clear need for pulmonary rehabilitation (PR) during and/or post-AECOPD,13 with a recent Cochrane review concluding that PR is safe, reduces hospital admissions, and improves quality of life and exercise capacity of patients post-AECOPD.14 Despite the evidence, referral and completion rates of PR are low. One audit in a UK hospital found that of 286 patients with an AECOPD eligible for PR, only 31% were referred, and less than 15% went on to complete the program,13 with another study finding only 9% of post-AECOPD patients completed PR.15
Studies have examined barriers to PR acceptance and uptake post-AECOPD which included a lack of knowledge about PR, the fact that patients exhibit lower self-worth and are less likely to seek help after AECOPD,16 and the perceptions of being too busy or too ill.17 A qualitative study including interviews with health-care professionals and patients was conducted by our team to determine how best to enhance participation of patients post-AECOPD in PR.18 Two common themes arose from these interviews and reinforced the aforementioned barriers: (1) education about PR as early as possible and (2) tailored, flexible interventions (i.e. offering options to patients in terms of mode of delivery and timing of initiation of PR).
A systematic review examining interventions that target uptake and completion of PR only included one study, which focused on an intervention to improve completion in stable patients already enrolled, rather than increasing uptake to PR.19 This reflects the extreme gap in the literature and the need for development of new strategies to improve uptake.19
With the guidance of an intervention development framework, we proposed to deliver a PR “taster” session to patients hospitalized with AECOPD with the aim of increasing knowledge about PR, providing information about services available as early as possible, and answering questions to alleviate concerns regarding participation. The objectives of this study were to:
Assess the acceptability, feasibility, and safety of delivering a PR “taster” session to patients hospitalized with AECOPD;
Evaluate the changes in patient knowledge of, and readiness to commence PR; and
Make recommendations for the refinement of future iterations of the intervention based on the findings of this study.
Methods
Design
We conducted a pre–post, one-site study. Data were collected over a period of 3 months by a trained researcher. Ethics approval was obtained from the Research Ethics Board of the McGill University Health Centre (MUHC).
Setting and participants
Consecutively patients on the respiratory ward of the MUHC were approached as soon as possible after admission. Inclusion criteria included (1) medically confirmed diagnosis of AECOPD (alone or combined with other conditions such as chronic heart failure and pneumonia); (2) being an eligible candidate for PR (all patients with COPD ≥ stage II according to the GOLD guidelines)20; (3) able to provide informed consent; and (4) able to read and speak French or English. Exclusion criteria included (1) major psychological issues relayed by the medical team; (2) being in palliative care; (3) expected survival of less than 6 months; and (4) musculoskeletal or cardiac issues that may have contraindicated exercise.
Intervention development
The ORBIT model for behavioral treatment development guided the intervention development. The ORBIT model comprises a four-phase process to aid the creation of behavior change interventions informed by behavioral and social sciences research.21 The first of these four phases is the “Design” phase, which includes two parts: Define and Refine.21 This project sits within the “Define” phase.
The theory of planned behavior (TPB)22 was selected to guide us throughout the use of the ORBIT model in the development process. The TPB asserts that an individual’s actions are informed by their intentions, and that behavioral intentions can be influenced by (1) their attitude toward performing the behavior (accepting an offer of referral to PR); (2) perceived social pressure (how much emphasis they think others, including nurses or doctors, place on them accepting a referral); and (3) perceived behavioral control (how strongly the individual feels accepting a referral is a choice they have control over).23,24
We set out to design an intervention that would target knowledge of PR, emphasize that health-care professionals believed PR would be beneficial, and address fears of breathlessness/medical conditions worsening. We presented the protocol to a group of researchers and clinicians affiliated with the research institute of the MUHC and to a group of physiotherapists working on the ward in question. Both of these presentations resulted in useful feedback for tweaking the protocol and taster session before beginning its implementation.
Intervention delivered
We delivered a PR “taster” session to patients with AECOPD. We asked patients if they would like to accompany the researcher to one of the hospital gyms to see and/or try equipment used for aerobic exercise (treadmill, stationary cycle) and resistance training in a PR program. The intervention was estimated to take 30–40 minutes and delivered individually. During the intervention, the researcher planned to show and/or involve patients in typical aerobic and resistance exercise with no focus on intensity, explain the general components of a PR program, and provide the participants with a handout that introduced PR and its benefits and covered the general education topics that may be touched on (Appendix 1). At the end of the “taster,” patients were given a “menu” of the PR programs available in the Greater Montreal area, showcasing all options available to them in terms of timing, location, and delivery format (Appendix 2). To ensure the intervention was delivered uniformly, targeting the aforementioned barriers, and informed by the TPB, a script was drafted for the researcher to refer to Table 1.
Table 1.
Script guide for researcher during PR “taster” session.
| Barrier/enabler to overcome/emphasize and/or theoretical concept from the TPB to incorporate | Script guide |
| Recommended by doctors and health-care professionals/perceived social pressure | “It’s great that you could come along and learn a little bit more about pulmonary rehabilitation, because it’s something that your doctors, nurses, and physiotherapists think could be of great benefit to you.” |
| Agency (control over their own behavior) | “It’s up to you whether pulmonary rehabilitation is something that you’d like to take part in, so we want to try and give you some more information than you might usually receive so that you can make an informed decision for yourself and your health.” |
| Information about PR | “Pulmonary rehabilitation is a comprehensive program including exercise, education, and social and psychological support. Today we’re going to show you some of the kinds of exercises you might do, and talk to you about some of the things you might learn about your disease and how to manage it with pulmonary rehabilitation, and hopefully answer some of your questions.” |
| Information about PR | Demonstrations of weights, treadmill → education about exercise component of PR |
| Lack of knowledge of benefits of PR/attitude toward performing the behavior | “Pulmonary rehabilitation improves quality of life, and gives you more stamina, so you can do things that you enjoy more independently and with less breathlessness. It’s also been shown to be helpful no matter what stage of COPD you have.” |
| Lack of knowledge regarding availability and timing of PR programs | “We’re going to give you a couple of handouts to take away, and one of these is what we like to call a ‘menu’ of the pulmonary rehabilitation options available in the Greater Montreal area. You’ll see there are different times of the week, different frequencies, and most of these programs start at several different times during the year, so there is usually something to fit everyone’s schedule.” |
| Fear of breathlessness/medical conditions worsening | “If you enrol in a supervised program, a qualified healthcare professional will be working with you and monitoring you while you exercise, and making sure to adjust the level to suit you. Some breathlessness might occur, but that will keep getting better and they will be there to help you.” |
| Agency (control over their own behavior) | “I know this is a difficult time, no one likes to be in the hospital, but we wanted to offer you this program because it can help you recover from this exacerbation. It is your decision whether you decide to do it or not, but we wanted to give you as much information as possible so you can make a well-informed decision. Do you have any questions that you would like to ask me or you would like me to ask your doctor or nurse while you consider whether this might be something you’d like to do?” |
PR: pulmonary rehabilitation; TPB: theory of planned behavior; COPD: chronic obstructive pulmonary disease.
Descriptive outcome measures
We collected data on age and sex, smoking status, hospital length of stay (LOS), most recent percentage of predicted forced expiratory volume in 1 s (FEV1% pred.) score, symptom severity (measured by the Modified Medical Research Council scale),25,26 disease impact (measured by the COPD assessment test),27 supplemental oxygen use, and depression and anxiety status (measured by the Hospital Anxiety and Depression Scale).28 Sex, age, and hospital LOS were recorded for nonparticipants.
Prospective acceptability, feasibility, and safety
We determined the prospective acceptability by the proportion of patients approached that accepted an offer to participate in the “taster” session.29 Reasons for declining the intervention were collected.
Measures for assessing feasibility included number of patients eligible, proportion of eligible patients approached, how many times a patient was attempted to be approached, proportion of participants who completed the “taster”, and the proportion of times the “taster” could be delivered in the specified time. Changes to the research protocol were also documented.
Safety was measured by the proportion of adverse events occurring as a result of the intervention.
Patient knowledge of PR
Patient knowledge of PR was measured via a modified version of the Bristol COPD Knowledge questionnaire (BCKQ)30 and of the Understanding COPD (UCOPD) questionnaire31 before and after the intervention. The modified version of the UCOPD questionnaire included five questions from the original survey and four additional questions created by the researchers to reflect the level of knowledge of PR and confidence related to taking part in PR. For both questionnaires, the score is presented as a percentage, where 100% represents better understanding for the modified UCOPD questionnaire, and for the modified BCKQ, the percentage represents the percent of correct answers.
Readiness to commence PR
Readiness to commence PR was measured using four tools: (1) A modified version of the Readiness to Change Exercise Questionnaire which places participants into one of four stages: precontemplation, contemplation, preparation, or action32; (2) and (3) two Likert-type scales which assessed motivation and confidence (both adapted from Bourbeau et al.’s behavior-change intervention study in patients with COPD)33; and (4) a modified version of the Treatment Self-Regulation Questionnaire (TSRQ) for exercise which measures the degree to which a patient’s motivation to enroll in PR is self-determined (as provided in the Health-Care, Self-Determination Theory Questionnaire Packet).34 Participants were also asked at the end of the intervention whether or not they had the intention to accept a referral, and which PR option they would prefer (inpatient, outpatient, or home program) if any option were available to them.
Recruitment target, data analysis, and reporting
As the primary intention of this study was to test the feasibility of a PR “taster” session delivered in the peri-hospitalization phase (and to gain information for refinement of the intervention), it was not appropriate to aim for sufficient statistical power to detect changes over time.35 Thus, the aim was to recruit as many patients as were admitted to the MUHC for AECOPD during the data collection period. Due to the small sample size, quantitative statistical analysis was not performed. Descriptive statistics (proportions) were used to report on the acceptability, feasibility, and safety of the intervention.
Results
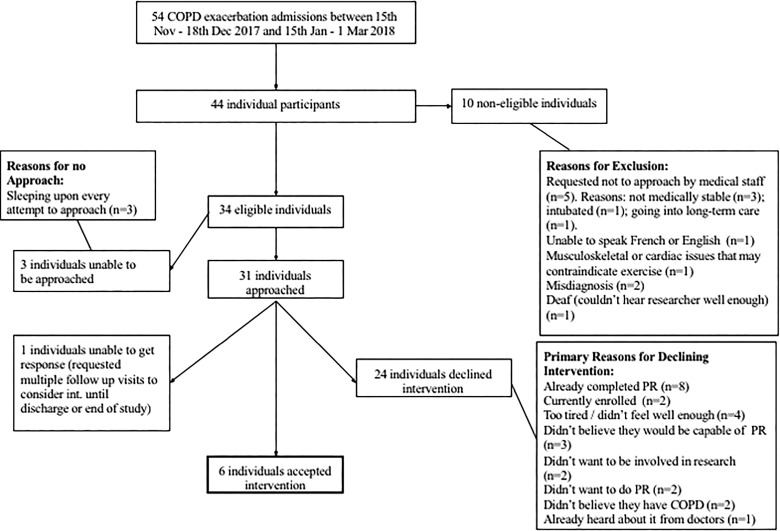
Fifty-four admissions with a primary diagnosis of AECOPD were recorded in the study period. Of these, 44 individual patients were admitted (Figure 1). Participant characteristics are presented in Table 2.
Figure 1.
Flowchart of the study.
Table 2.
Patient and participant characteristics.
| Characteristics | All eligible patients (n = 34)a | Participants (n = 6) |
|---|---|---|
| Age in years, median (range) | 70.5 (52–93) | 71 (66–86) |
| Female, % (n) | 55.9 (19) | 66.7 (4) |
| FEV1% pred., median (range) | — | 37 (22–61) |
| MMRC score, median (range) | — | 3.5 (2–4) |
| CAT score, median (range) | — | 30 (26–35) |
| HADs anxiety score, median (range) | — | 6 (2–11) |
| HADs depression score, median (range) | — | 8.5 (2–10) |
| Using supplemental oxygen, % (n) | — | 66.7 (4) |
| Current/former smoker, % (n) | — | 66.7 (4) |
| In isolation at time of intervention, % (n) | — | 50 (3) |
| Hospital LOS in days, median (range) | 5 (1–56) | 7 (2–41) |
FEV1% pred.: percentage of predicted forced expiratory volume in 1 s; MMRC: modified medical research council; CAT: COPD assessment test; HAD: hospital anxiety and depression; LOS: length of stay.
a As some patients were admitted on multiple occasions, the data presented represent the admission where they were successfully approached and either accepted or declined the intervention (n = 30), or for those with only one admission, their only admission where they were unable to be approached (n = 3) or unable to make a decision about participation (n = 1).
Acceptability
Twenty-four (77%) of the approached patients declined the intervention, one (0.03%) was discharged before they could decide to participate, and six patients (19%) accepted. Reasons for declining are presented in Figure 1.
Feasibility and safety
Of the 44 admitted patients, 10 were ineligible, leaving 34 eligible for participation. Reasons for exclusion are seen in Figure 1. Thirty-one (91%) (n = 31) of the eligible patients were approached as soon as possible after admission (range = 1–10 days). The median number of times to approach a patient was only one (range = 1–11). For all six participants who accepted to take part in the “taster,” the intervention was delivered in less time than the prespecified 30–40 minutes, and all six participants completed the “taster.” However, the originally intended delivery setting (a gym within the hospital) was not feasible. Therefore, the decision had to be made to offer the intervention within patient rooms. The researcher still discussed all of these exercise options, but no demonstration or participation was possible. All six participants received the intervention in their room, due to three being in isolation, and the remaining three not wanting to leave their rooms. There were no adverse events recorded as a result of the intervention.
Patient knowledge of PR
Table 3 presents the individual results of the modified UCOPD and BCKQ questionnaires pre- and post-intervention. The median change post-intervention for the modified UCOPD questionnaire was +8. No median difference was recorded for the percentage of correct answers to the modified BCKQ.
Table 3.
Changes in patient knowledge of PR.
| Patient | Pre-modified UCOPD score (%) | Post-modified UCOPD score (%) | % Change | Pre-modified BCKQ score (%) | Post-modified BCKQ score (%) | % Change |
|---|---|---|---|---|---|---|
| 1 | 58.9 | 70 | +11.1 | 80 | 80 | 0 |
| 2 | 66.7 | 83.3 | +16.6 | 100 | 100 | 0 |
| 3 | 48.9 | 83.3 | +34.4 | 80 | 60 | −20 |
| 4 | 35.6 | 33.3 | −2.3 | 0 | 0 | 0 |
| 5 | 55.6 | 54.3a | −1.3 | 60 | 60 | 0 |
| 6 | 80 | 86.7 | +6.7 | 80 | 80 | 0 |
| Median change | +8.9 | 0 |
PR: pulmonary rehabilitation; COPD: chronic obstructive pulmonary disease; UCOPD: Understanding COPD; BCKQ: Bristol COPD Knowledge
a Incomplete survey data. Missing data procedure followed as laid out in the UCOPD user manual.
Readiness to commence PR
According to the modified Readiness to Change Exercise Questionnaire, three of the patients were already in the action phase pre-intervention. Post-intervention, all but one were in the action phase. One participant was unable to be categorized into a stage both pre- and post-intervention, as they indicated “neither agree nor disagree” for all questions both times.
For both the Confidence and Motivation to Commence PR questionnaires, participant changes post-intervention varied. No median change was observed post-intervention. Results are presented in Table 4. A slight positive change in relative autonomous motivation index (RAMI) (indicating how self-determined the decision to commence PR is) was observed post-intervention. However, this varied between participants (Table 5). Finally, five of the participants reported they intended to enroll in PR. The one participant who did not intend to enroll explained they already had a home program that suited them well. Four of the participants indicated that a home program would be their first choice.
Table 4.
Changes in motivation to commence PR and confidence to commence PR.
| Patient | Pre-motivation score | Post-motivation score | Change | Pre-confidence score | Post-confidence score | Change |
|---|---|---|---|---|---|---|
| 1a | N/A | N/A | N/A | N/A | N/A | N/A |
| 2 | 18 | 32 | +14 | 34 | 50 | +16 |
| 3 | 48 | 45 | −3 | 46 | 48 | +2 |
| 4 | 15 | 15 | 0 | 15 | 15 | 0 |
| 5 | 36 | 28 | −8 | 26 | 22 | −4 |
| 6 | 46 | 48 | +2 | 48 | 39 | −9 |
| Median change | 0 | 0 |
TSRQ: Treatment Self-Regulation Questionnaire; PR: pulmonary rehabilitation.
a After consultation with senior researchers and clinicians during the presentation of the original protocol, the decision was made to add three more questionnaires to the study (motivation to commence PR, confidence to commence PR and the TSRQ), as the ORBIT model allows for iterative processes within the study design. Due to the time it took for the ethics board to approve the protocol changes, the first participant was unable to provide responses to these additional questionnaires.
Table 5.
Pre- and post-intervention RAMI scores for each participant as measured by the TSRQ.
| Patient | Pre-RAMI | Post-RAMI | Change |
|---|---|---|---|
| 1a | N/A | N/A | N/A |
| 2 | 2.5 | 0.5 | −3 |
| 3 | 1 | 5.17 | +4.17 |
| 4 | 0 | 0 | 0 |
| 5 | 1.7 | 2.5 | +0.8 |
| 6 | 4.16 | 4.83 | +0.67 |
| Median change | +0.67 |
TSRQ: Treatment Self-Regulation Questionnaire; RAMI: relative autonomous motivation index; PR: pulmonary rehabilitation.
a After consultation with senior researchers and clinicians during the presentation of the original protocol, the decision was made to add three more questionnaires to the study (motivation to commence PR, confidence to commence PR and the TSRQ), as the ORBIT model allows for iterative processes within the study design. Due to the time it took for the ethics board to approve the protocol changes, the first participant was unable to provide responses to these additional questionnaires.
Discussion
Prospective acceptability of the PR “taster” session to patients was low. Encouragingly, 10 of the 24 patients who declined the intervention did so because they were already enrolled in (n = 2) or had completed (n = 8) PR. This study was carried out at a research hospital affiliated with McGill University. As such, it may be that a positive culture regarding PR exists in this environment, explaining the relatively high proportion of patients who had enrolled in or completed PR. However, almost a third of all patients who declined to take part in the intervention did so because they were not feeling well enough or believed they would not be able to participate. These reasons for declining the intervention are in line with the reasons for declining PR itself in both the general COPD36 and AECOPD17 populations. This raises the question as to whether, despite international recommendations, this may not be the right time to be offering PR to patients.
It is interesting to note that the median anxiety score of our six participants was 6 (absence of anxiety), and the median depression score was 8.5 (borderline depression). Thus, it may be that the patients who opted to participate in our intervention were those who were coping better with their disease and its associated exacerbations. It may be that these “copers” could have been more likely to accept a referral to PR even without undertaking the PR “taster” session.
In terms of feasibility, 34 of the 44 admitted patients were eligible for participation. It was feasible to approach eligible patients, with 31 successfully approached. The intervention was also feasible to deliver in the prespecified time of 30–40 minutes, and all participants completed the intervention. However, the fact that the protocol had to be changed to allow for in-room delivery of the protocol meant that the prespecified design and environment for the “taster” was not feasible. This undoubtedly affected other aspects of feasibility, such as delivery time and safety, and there was no setup time of exercise equipment and no actual exercise carried out or demonstrated. With no participants participating in exercise or even having to leave their rooms, no adverse events were recorded.
A change was observed in the median score for the modified UCOPD questionnaire post-intervention, but not for the modified BCKQ. This may be due to the content of the questionnaires. The original UCOPD questionnaire is designed to measure understanding, self-efficacy use, and satisfaction with the education component of a PR program.31 The questionnaire contains Likert-style questions such as “How confident are you that…” and “How well do you understand….” Thus, patients assess how they perceive their own understanding of PR. Conversely, the section of the BCKQ included in our study asks five specific true or false questions regarding exercise. It could be posited that participants may be more likely to take away the “bigger picture” from the intervention (i.e. PR is beneficial and consists of exercise, education, and support) rather than the details (e.g. exercise is better than breathing exercises at improving fitness). An additional explanation could be that the amount of education delivered was not sufficient to result in a change in knowledge. In a study examining the effects of a brief education program on patients with AECOPD, two one-on-one 30-minute education sessions were delivered to patients covering a variety of topics related to COPD and its management, PR, and maintaining an active lifestyle.37 While this study did see a significant improvement in the BCKQ score in the intervention group, they only saw an 8-point change out of a total possible score of 65.37 As the “taster” session intervention was shorter and only included general information about PR programs, this could also be a potential contributing factor to the lack of change.
The outcome measures which showed positive change for readiness to commence PR were the Readiness to Change Exercise Questionnaire, the RAMI, and the record of intention to enroll in PR. However, as three of the participants were already in the action stage pre-intervention, the idea that the patients who accepted to participate in this intervention may have already been more likely to accept a referral to PR even without exposure to the intervention may have credence. In addition to the possibility of being “copers,” the results of the Readiness to Change Exercise Questionnaire could indicate that those who participated are also more willing and open to taking action to improve their disease and make lifestyle changes. However, two other participants moved to the action stage post-intervention, so a change was recorded in some participants.
We did not observe a change in confidence and motivation to commence PR in the study participants. Considering that four of six participants were in the action or preparation phases of the Readiness to Change Exercise Questionnaire, it may be that participants were already sufficiently motivated and confident to commence PR prior to the “taster,” and thus unlikely to experience changes.
Limitations
This study was conducted in only one site, and this site dealt specifically with patients hospitalized for AECOPD. Thus, results presented may not be generalizable to patients with AECOPD who are not hospitalized for their exacerbation. Due to the small sample size, the study was underpowered to test for significant changes pre- and post-intervention. However, some of the changes recorded help provide an indication of which mediating factors that contribute to PR uptake may be affected by this intervention and could be used to further refine this intervention. Due to the number of admissions per month, we had expected that 3 months would be an acceptable time frame to determine the feasibility and acceptability of the intervention. While we could have extended the data collection, it became clear that the initial protocol and “taster” were not prospectively acceptable to eligible patients. As we were only working in the “Design” phase of the ORBIT model, this initial iteration of the intervention was developed in order to gain feedback on the protocol before moving into the “Preliminary Testing” phase (including Proof-of-Concept and Pilot studies), so the number of participants was not deemed as relevant, as long as we were able to determine whether or not changes needed to be made before proceeding to the next phase.
Additionally, this study took a quantitative approach to assessing the feasibility, acceptability, and safety of the PR “taster.” The addition of a qualitative assessment in future iterations may help to further understand why participants may decline or accept participation in such a “taster” session. Nevertheless, we did record reasons for decline.
Recommendations for future iterations
As this study was guided by the ORBIT model and situated within the “Design” phase, the results of this study can be used to either (a) refine the intervention or (b) contribute to the definition of a new intervention to address the same clinical problem; how to increase patient uptake to PR after AECOPD. Since prospective acceptability of the intervention was low, it may be that the timing of the intervention needs reconsidering. Patients who decline because of feeling too ill could be more likely to partake in the intervention if the initial approach were made at their post-discharge follow-up visit. Or it may be that approaching patients at the day clinic (where the exacerbations are not quite as serious to warrant hospitalization) could result in higher uptake of the intervention. As it was not feasible to deliver the intervention in a gym, future iterations could include a video option which could also be sent home or shown at the follow-up visit. Short testimonials from participants, doctors, nurses, or physiotherapists could also be included to target the social pressure aspect of the TPB. As the majority of participants indicated that a home program would be their first choice, referring health-care practitioners should make patients aware of home program options and other alternatives to traditional in- and outpatient PR. Finally, researchers, physicians, and physiotherapists were consulted in the planning of the initial protocol and “taster,” but it may be helpful to consult with patient representatives of the target group when making future changes to the intervention.
Conclusions
The delivery of a PR “taster” session to patients hospitalized with AECOPD was not prospectively acceptable to a large portion of patients and was only feasible with modifications to the original protocol. It may be that the intervention may be more successful if offered at the time of follow-up after discharge for AECOPD. Additionally, the use of video may improve the acceptance of the intervention by reducing the need for patients to move to a gym to see a demonstration of exercise.
Acknowledgments
The authors would like to acknowledge the suggestions of researchers from the School of Physical and Occupational Therapy at McGill University, and from researchers and clinicians on the eighth floor of the McGill University Health Centre (MUHC), and the physiotherapists from the MUHC for their feedback on initial versions of the protocol, especially Anne Hatzoglu, Catherine Edwards, Chantal C Lefebvre, and Diana Valentini, as well as clinician and physiotherapist assistance during data collection.
Appendix 1
Handout of education topics covered in PR
Appendix 2.
“Menu” of PR options in the Greater Montreal area.
| Area | Establishment | Program(s) | Program duration | Waiting Time | Details |
|---|---|---|---|---|---|
| Montreal | Hôpital Sacré-Coeur de Montréal | Outpatient | 8 weeks, 1 hour of education and 1.5 hour of exercise 2×/week. | 2–3 months | $100 (includes heart rate monitor watch); patient should have a stationary bike at home. |
| Montreal | Hôpital Maisonneuve-Rosemont | Home | Individualized program, no limit on length of time. Home visits once a week at the beginning of the program and then every 2–3 weeks when patient is comfortable with exercises. | 3–4 months | Free. No specific schedule, done at home with patient. Sessions are about 60–90 minutes. |
| Montreal | lnstitut thoracique de Montréal (McGill University Health Centre) | Outpatient, home | 7 weeks, 1.5 hour of exercise 3×/week and 1–2 education sessions a week of 60 minutes at lunchtime. | 4 months | Free. Waiting time: 4 months. Home program is available to all. |
| Montreal | Hôtel Dieu de Montréal | Outpatient, home Maintenance |
Outpatient: 8 weeks, 3×/week (Monday, Wednesday, Friday, two groups: 1:00–2:30 and 2:30–16:00), mix of education and exercise in each session. Maintenance: 2×/week (Tuesday and Thursday). Must have completed the 8-week program first. Personalized to fit the patient. |
Outpatient: 9 months–1 year Home: 3 months Maintenance: No wait |
Free. The home program is operated by another person (a kinesiologist also working at the hospital). If the patient does not want to join ambulatory program, they offer the home program as an option. |
| Montreal | Hôpital Mont Sinaï de Montréal | Outpatient, inpatient | Inpatient: 3–4 weeks. Outpatient: 8 weeks, 1-hour education, 1-hour exercise 3×/week. |
None | Free. |
| Chomedey | Hôpital Juif de réadaptatlon | Outpatient, inpatient | Inpatient: approximately 4 weeks, case by case, not as intense with education. Outpatient: l0 week, 3×/week (2× education, 1× exercise, moving to only exercise in the last 3–4 weeks), 1.5–2 hours. |
Outpatient: 2–3 months Inpatient: case by case |
Free, but patients need to pay for the modules “Living well with COPD” and for a pedometer. Follow-up program—final evaluation once they finish the group, plus final meeting with pneumologist to ensure everything is okay and check progress (1–2 weeks after finishing the group program |
| Legardeur | Centre de santé et de services sociaux du Sud de Lanaudière | Outpatient | Exercise classes are given twice a week (Tuesday and Thursday, 1:30–3:00). Three or four times a year | None | This is not a formal pulmonary rehab program but more an opportunity for the COPO patient to exercise under the supervision of a physiotherapist |
| Longueuil | Hôpital Charles Lemovne | Outpatient | 8 weeks, 3×/week (l× 2-hour session with l-hour education and 1-hour exercise, and 2× 1-hour sessions of exercise). | Less than a year. | Free. Sessions start in autumn, winter, and spring. Patients with severe cardiac problems excluded. |
Footnotes
Data availability: The data used to support the findings of this study are available from the corresponding author upon request.
Declaration of conflicting interests: The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: The author(s) disclosed receipt of the following support for the research, authorship, and/or publication of this article: The senior author (TJF) and SA received a salary award from Fonds de Recherche Sante Quebec (FRQS). JB is a recipient of the research chair GSK-CIHR in COPD.
ORCID iD: Jean Bourbeau  https://orcid.org/0000-0002-7649-038X
https://orcid.org/0000-0002-7649-038X
Sara Ahmed Tania Janaudis-Ferreira  https://orcid.org/0000-0003-0944-3791
https://orcid.org/0000-0003-0944-3791
References
- 1. Harries TH, Thornton H, Crichton S, et al. Hospital readmissions for COPD: a retrospective longitudinal study. NPJ Prim Care Respir Med 2017; 27(1): 31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Jones PW, Chen WH, Wilcox TK, et al. Characterizing and quantifying the symptomatic features of COPD exacerbations. Chest 2011; 139(6): 1388–1394. [DOI] [PubMed] [Google Scholar]
- 3. Kessler R, Stahl E, Vogelmeier C, et al. Patient understanding, detection, and experience of COPD exacerbations: an observational, interview-based study. Chest 2006; 130(1): 133–142. [DOI] [PubMed] [Google Scholar]
- 4. Miravitlles M, Anzueto A, Legnani D, et al. Patient’s perception of exacerbations of COPD—the PERCEIVE study. Respir Med 2007; 101(3): 453–460. [DOI] [PubMed] [Google Scholar]
- 5. Pitta F, Troosters T, Probst VS, et al. Physical activity and hospitalization for exacerbation of COPD. Chest 2006; 129(3): 536–544. [DOI] [PubMed] [Google Scholar]
- 6. Pitta F, Troosters T, Spruit MA, et al. Characteristics of physical activities in daily life in chronic obstructive pulmonary disease. Am J Respir Crit Care Med 2005; 171(9): 972–977. [DOI] [PubMed] [Google Scholar]
- 7. Jones SE, Maddocks M, Kon SS, et al. Sarcopenia in COPD: prevalence, clinical correlates and response to pulmonary rehabilitation. Thorax 2015; 70(3): 213–218. [DOI] [PubMed] [Google Scholar]
- 8. Burtin C, Decramer M, Gosselink R, et al. Rehabilitation and acute exacerbations. Eur Respir J 2011; 38(3): 702–712. [DOI] [PubMed] [Google Scholar]
- 9. Spruit M, Gosselink R, Troosters T, et al. Muscle force during an acute exacerbation in hospitalised patients with COPD and its relationship with CXCL8 and IGF-I. Thorax 2003; 58(9): 752–756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Harrison SL. (2014) Exploring patients’ perceptions following an acute exacerbation of chronic obstructive pulmonary disease to inform tailored strategies to enhance pulmonary rehabilitation. PhD Thesis, University of Leicester, UK. [Google Scholar]
- 11. Chu C, Chan V, Lin A, et al. Readmission rates and life threatening events in COPD survivors treated with non-invasive ventilation for acute hypercapnic respiratory failure. Thorax 2004; 59(12): 1020–1025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Lau AC, Yam LY, Poon E. Hospital re-admission in patients with acute exacerbation of chronic obstructive pulmonary disease. Respir Med 2001; 95(11): 876–884. [DOI] [PubMed] [Google Scholar]
- 13. Man WDC, Puhan MA, Harrison SL, et al. Pulmonary rehabilitation and severe exacerbations of COPD: solution or white elephant? ERJ Open Research 2015; 1(2): 00050–02015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Puhan MA, Gimeno-Santos E, Cates CJ, et al. Pulmonary rehabilitation for people who have been in hospital with an exacerbation of chronic obstructive pulmonary disease. Cochrane Database Syst Rev 2016; 2016: 12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Harrison SL, Robertson N, Graham CD, et al. Can we identify patients with different illness schema following an acute exacerbation of COPD: a cluster analysis. Respir Med 2014; 108(2): 319–328. [DOI] [PubMed] [Google Scholar]
- 16. Harrison SL, Robertson N, Apps L, et al. “We are not worthy”—understanding why patients decline pulmonary rehabilitation following an acute exacerbation of COPD. Disabil Rehabil 2015; 37(9): 750–756. [DOI] [PubMed] [Google Scholar]
- 17. Benzo R, Wetzstein M, Neuenfeldt P, et al. Implementation of physical activity programs after COPD hospitalizations: lessons from a randomized study. Chron Respir Dis 2015; 12(1): 5–10. [DOI] [PubMed] [Google Scholar]
- 18. Janaudis-Ferreira T, Tansey CM, Harrison SL, et al. A qualitative study to inform a more acceptable pulmonary rehabilitation program after acute exacerbation of COPD. Ann Am Thorac Soc. Epub ahead of print 5 May 2019 DOI: 10.1513/AnnalsATS.201812-854OC. [DOI] [PubMed] [Google Scholar]
- 19. Jones AW, Taylor A, Gowler H, et al. Systematic review of interventions to improve patient uptake and completion of pulmonary rehabilitation in COPD. ERJ Open Research 2017; 3(1): 00089–2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. GOLD. GIfCOLD. Global strategy for the diagnosis, management and prevention of COPD, http://goldcopd.org (2017, accessed 3 February 2019). [DOI] [PubMed]
- 21. Czajkowski SM, Powell LH, Adler N, et al. From ideas to efficacy: the ORBIT model for developing behavioral treatments for chronic diseases. Health Psychol 2015; 34(10): 971–982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Wyer SJ, Earll L, Joseph S, et al. Increasing attendance at a cardiac rehabilitation programme: an intervention study using the theory of planned behaviour. Coronary Health Care 2001; 5(3): 154–159. [Google Scholar]
- 23. Ajzen I, Madden TJ. Prediction of goal-directed behavior: attitudes, intentions, and perceived behavioral control. J Exp Soc Psychol 1986; 22(5): 453–474. [Google Scholar]
- 24. Kuhl J, Beckman J. (eds) Action control: From cognition to behavior. New York, NY: Springer-Verlag, 1985. [Google Scholar]
- 25. Brooks SM. Task group on surveillance for respiratory hazards in the occupational setting; surveillance for respiratory hazards. ATS News 1982; 8: 12–16. [PubMed] [Google Scholar]
- 26. Munari AB, Gulart AA, dos Santos K, et al. Modified medical research council dyspnea scale in GOLD classification better reflects physical activities of daily living. Respir Care 2017; 63: 77–85. [DOI] [PubMed] [Google Scholar]
- 27. COPD Assessment Test. Middlesex, UK: GlaxoSmithKline Services Unlimited, http://www.catestonline.org/ (2016, accessed on 3 February 2019). [Google Scholar]
- 28. Zigmond AS, Snaith RP. The hospital anxiety and depression scale. Acta Psychiat Scand 1983; 67(6): 361–370. [DOI] [PubMed] [Google Scholar]
- 29. Sekhon M, Cartwright M, Francis JJ. Acceptability of healthcare interventions: an overview of reviews and development of a theoretical framework. BMC Health Serv Res 2017; 17(1): 88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. White R, Walker P, Roberts S, et al. Bristol COPD knowledge questionnaire (BCKQ): testing what we teach patients about COPD. Chron Respir Dis 2006; 3(3): 123–131. [DOI] [PubMed] [Google Scholar]
- 31. O’Neill B, Cosgrove D, MacMahon J, et al. Assessing education in pulmonary rehabilitation: the understanding COPD (UCOPD) questionnaire. COPD 2012; 9(2): 166–174. [DOI] [PubMed] [Google Scholar]
- 32. Kheawwan P, Chaiyawat W, Aungsuroch Y, et al. Patient readiness to exercise after cardiac surgery: development of the readiness to change exercise questionnaire. J Cardiovasc Nurs 2016; 31(2): 186–193. [DOI] [PubMed] [Google Scholar]
- 33. Bourbeau J, Lavoie KL, Sedeno M, et al. Behaviour-change intervention in a multicentre, randomised, placebo-controlled COPD study: methodological considerations and implementation. BMJ Open 2016; 6(4): e010109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Williams GC, Ryan RM, Deci EL. Health-care, self-determination theory questionnaire packet, http://selfdeterminationtheory.org (accessed 3 February 2019).
- 35. Tickle-Degnen L. Nuts and bolts of conducting feasibility studies. Am J Occup Ther 2013; 67(2): 171–176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Keating A, Lee A, Holland AE. What prevents people with chronic obstructive pulmonary disease from attending pulmonary rehabilitation? A systematic review. Chron Resp Dis 2011; 8(2): 89–99. [DOI] [PubMed] [Google Scholar]
- 37. Janaudis-Ferreira T, Carr SJ, Harrison SL, et al. Can patients with COPD assimilate disease-specific information during an acute exacerbation? Results of a pilot randomized controlled trial. Chest 2018; 154(3): 588–596. [DOI] [PubMed] [Google Scholar]