Abstract
Substance P plays a pivotal role in human cancer development and progression by binding to its receptor, neurokinin-1. Neurokinin-1 has 2 isoforms: full-length neurokinin-1 and truncated neurokinin-1, the latter lacking the cytoplasmic terminal 96-amino acid residues of the full-length protein. We have identified 3 candidate miR-206 target sites within the 3′-untranslated region of the full-length neurokinin-1 gene from bioinformatics database searches. In the present study, real-time quantitative polymerase chain reaction was performed to quantify the expression of miR-206, and the expression of neurokinin-1 and full-length neurokinin-1 was detected by immunohistochemistry in 82 clinical cases of breast cancer and paired adjacent normal tissues. The miR-206 target gene was demonstrated by using a dual-luciferase reporter assay, quantitative real-time polymerase chain reaction, and Western blotting. Transwell migration and invasion, colony formation, and proliferation assays were performed to evaluate the effects of miR-206 expression on various aspects of breast cancer cell behavior in vitro. We showed that miR-206 expression is upregulated in breast cancer cell lines and breast cancer tissues when compared to that in adjacent normal tissues, and full-length neurokinin-1 expression inversely correlates with Tumor Lymph Node Metastasis (TNM) stage and lymph node metastasis. Western blotting, quantitative real-time polymerase chain reaction, and dual-luciferase reporter assays demonstrated that miR-206 binds the 3′-untranslated region of full-length neurokinin-1 messenger RNA, regulating protein expression. We showed that the overexpression of miR-206 promotes breast cancer cell invasion, migration, proliferation, and colony formation in vitro. The present study furthers the current understanding of the mechanisms underlying breast cancer pathogenesis and may be useful for the development of novel targeted therapies.
Keywords: microrna-206, neurokinin-1 receptor, breast cancer, invasion, proliferation
Introduction
Yearly, almost 350 000 women die from breast cancer, which is the most common cancer in women.1 One factor is the high mortality owing to the spread of difficult-to-identify tumor cells to distant organs during the early stages of the disease. A number of studies have shown diverse genetic alterations related to breast cancer,2,3 but the extremely complex molecular mechanisms in the progression of this disease remain obscure. Therefore, it is necessary to identify novel markers for breast cancer that can more accurately define the biological characteristics of tumors to further improve therapeutic measures and the prediction of clinical outcomes.
Substance P (SP), a member of the tachykinin family of peptides, is widely distributed throughout the mammalian central nervous system and peripheral tissues and is associated with inflammation and viral infections, such as HIV infection.4,5 Substance P plays a biological function by binding the transmembrane G-protein-coupled receptor neurokinin-1 (NK1R). Neurokinin-1 regulates a variety of cellular processes including pain transmission,6 exocrine and endocrine secretion,7 vasodilatation, cell proliferation, and immune and inflammatory responses.8 Neurokinin-1 has 2 isoforms: full-length NK1R (NK1R-FL) and truncated NK1R (NK1R-Tr), the latter lacking the cytoplasmic terminal 96-amino acid residues of the full-length protein. Differences in the binding and signaling properties of NK1R-Tr and NK1R-FL have been observed following their expression in various cell systems. These studies have demonstrated that SP exerted different biological functions through NK1R-Tr and NK1R-FL, resulting in distinct tumor-promoting properties.9 We have previously shown that both NK1R isoforms are present in breast tumor and benign and normal tissues but that NK1R-FL expression is significantly decreased in tumor specimens, especially in metastatic carcinomas.10 Furthermore, we have shown that NK1R-FL is highly expressed in the nontumorigenic breast epithelial cell line MCF-10A, whereas the MDA-MB-231, T47D, and MCF-7 breast cancer cell lines express only NK1R-Tr. Decreased NK1R-FL expression was associated with tumor-grade and lymph node metastases of breast cancer, while NK1R-Tr expression levels were increased gradually with the advancement of malignancy in breast cancer cells. And these 2 forms of NK1R were inversely expressed in cancer cells: one was upregulated, and the other was downregulated.
The C-terminal domain of NK1R is important for SP-induced cellular Ca2+ influx because the ligand-dependent response differs between NK1R-FL and NK1R-Tr. The phosphatidylinositol pathway is a major signal transduction pathway downstream of G-protein-coupled receptor. The binding of extracellular signaling molecules to cell surface G-protein-coupled receptors results in the recruitment and activation of phospholipase C-β (PLC-β) at the plasma membrane, where it catalyzes the hydrolysis of phosphatidylinositol-4,5-diphosphate (PIP2) into the second messengers inositol 1,4,5-triphosphate (IP3) and diacylglycerol. Once liberated, IP3, which is a soluble second messenger, binds the ligand-gated calcium channels of the endoplasmic reticulum and induces intracellular Ca2+ release. Therefore, to examine the role of miR-206 in the regulation of NK1R-FL-dependent intracellular signaling, we monitored changes in the concentration of cellular IP3.
MicroRNAs (miR-206s) are a series of endogenous short noncoding RNAs and have been demonstrated to suppress the expression level of protein-coding genes at the transcriptional and posttranscriptional level by binding to the 3′-untranslated region (3′-UTR) of specific target mRNA molecules, resulting in its degradation or translational inhibition.11 miR-206 plays an important role in several malignancies, such as lung cancer, gastric cancer, colorectal cancer, renal cell carcinoma, endometrioid adenocarcinoma, glioma and neuroblastoma, rhabdomyosarcoma, and osteosarcoma.12-14 Sun et al demonstrated that miR-206 could impact lung cancer cell proliferation, migration, and colony formation by directly binding the 3′-UTR of c-Met and Bcl2 mRNA.15 The downregulation of miR-206 suppressed clear renal cell carcinoma proliferation and invasion by targeting vascular endothelial growth factor A.16 However, the role and direct functional targets of miR-206 in human breast cancer remain to be clarified. By using a bioinformatics approach, we identified 3 candidate miR-206 target sequences within the 3′-UTR region of NK1R-FL, followed by searches of the microRNA.org, miRanda, Target-Scan, and Pictar databases. This finding suggests that miR-206 can target NK1R-FL to influence breast cancer malignancy.
In the present study, we explored the role of miR-206 in breast cancer development. miR-206, as a potent regulator of NK1R, demonstrated that miR-206 is quickly upregulated in tumor specimens, particularly in metastatic carcinomas. Expression level of miR206 in breast cancer tissues was 0.76 ± 0.24 and in paracancerous tissues was 0.12 ± 0.08. Expression level of miR206 in breast cancer tissues was significantly higher than that in paracancerous tissues (t = 48.79, P < .001).17 In addition, functional studies have shown that miR-206 plays an important role in NK1R-mediated tumor cell growth and migration.
Materials and Methods
Clinical Specimens
A total of 82 freshly frozen breast tumor tissues and 82 adjacent normal tissues were obtained from the Tissue Bank Facility of Tianjin Cancer Hospital. Hematoxylin and eosin (H&E) staining was used for histologically confirming both tumor and normal tissues. Before surgery, none of the patients received radiotherapy or chemotherapy. According to the 2009 International Union Against Cancer (UICC) TNM staging system, the following cases were included: breast cancer stage I, 40 cases; stage II, 34 cases; stage III, 8 cases; stage IV, 0 cases; and invasive ductal carcinoma and intraductal carcinoma, 68 cases. The collection of specimens was achieved after obtaining the patient’s consent and received approval from the Institutional Research Ethics Committee.
Immunohistochemical Analyses
Immunohistochemistry and H&E staining on human breast tumor tissues and paired adjacent normal tissues were performed with the following primary antibodies: anti-N-terminus-polyclonal (1:1000, Novus, Cat. NB 300-119) specific for NK1R-FL and anti-C-terminus-polyclonal NK1R (1:2000, Sigma, Cat. S8305) for NK1R-total. A normal breast tissue that intensively expresses NK1R-FL and weakly expresses NK1R-Tr was employed as an NK1R-FL-positive control. The proportion of positive cells and the staining intensity of tumor cells were determined by 2 experienced pathologists using a light microscope (Olympus Optical Co., Japan). According to the percentage of positive cells, the samples were divided into 4 grades: 0, positive cells <5%; 1, 5% to 25%; 2, 26% to 50%; 3, 51% to 75%; and 4, 76% to 100%. The staining intensity was categorized as follows: 0, negative result; 1, weakly positive result; 2, moderately positive result; and 3, strongly positive result. The final score was determined from these 2 scores as follows: 0, negative result; 1 to 4, weakly positive result; 5 to 8, moderately positive result; and 9 to 12, strongly positive result.
Cell Culture
The human breast cancer cell lines MDA-MB-231, SK-BR-3, and the nontumorigenic breast MCF-10A cells were obtained from the American Type Culture Collection (Manassas, Virginia). MDA-MB-231 and SK-BR-3 cells were cultured in RPMI-1640 (HyClone) medium. All media were supplemented with 10% fetal bovine serum (FBS; Gibco, Tianjin yibo hengtai biotechnology co., LTD, China). MCF-10A cells were cultured in Dulbecco modified Eagle medium/F12 (HyClone). The media were supplemented with 5% horse serum, 100 ng/mL Cholera toxin, 10 ug/mL insulin, and 20 ng/mL epidermal growth factor (Gibco). The cells were incubated at 37°C in a 5% CO2 humidified atmosphere.
Cell Transfection
The miR-206 mimic control, negative control (NC), antisense miR-206 (As-miR-206), and inhibitor control (IC) sequences were synthesized by the RuiBo Company (Guangzhou, China). SK-BR-3, MCF-10A, and MDA-MB-231 cells were seeded onto 6-cm dishes and incubated overnight, and then SK-BR-3 and MCF-10A cells were transiently transfected with miR-206 mimic control or NC, while MDA-MB-231 cells were transfected with As-miR-206 or IC using Lipofectamine™ 2000 (Invitrogen, Tianjin boldi biotechnology co., LTD, China) according to the manufacturer’s instructions.
Messenger RNA/miR-206 Isolation and Quantitative Real-Time Polymerase Chain Reaction
Total RNA from tissue samples and cell lines was extracted using TRIzol Reagent (Invitrogen) Real-time quantitative polymerase chain reaction (PCR) was performed according to the manufacturer’s instructions. The expression of miR-206 was quantified by Taqman microRNA assays from Applied Biosystems (RuiBo Company). Briefly, 1 µg of total RNA was reverse transcribed to complementary DNA (cDNA) with specific stem-loop RT primers, and real-time quantitative PCR with miR-206-specific stem-loop primers was executed on an Applied Biosystems 7900 Real-time PCR System. The miR-206 expression in each sample was uniformly normalized to that of U6 as an internal control. The following primer sequences were used: NK1R-FL (NM001058) forward 5′-AAAGTCAGCCGCCTGGAGAC-3′; NK1R-FL reverse 5′-TCATGGTCTTGGAGTCACTTCG-3′; miR-206 forward 5′-GATTCGCCAAAGGAAATAGC-3′; and miR-206 reverse 5′- GTTACAAGGTCATCCAAGAC-3′. The quantitative real-time PCR (qRT-RCR) results were expressed relative to the expression levels of miR-206 at the threshold cycle (Ct) and converted to fold-changes (2−ΔΔCt) in which ΔΔCt = (CtmiR-206 − CtU6)target − (CtmiR-206 − CtU6)control.
Western Blot Analysis
Briefly, the cells were lysed in lytic buffer (1× sodium dodecyl sulfate sample buffer) with a phosphatase inhibitor and protease inhibitor cocktail. Approximately 120 µg of total protein was separated by 10% sodium dodecyl sulfate polyacrylamide gel electrophoresis for 60 minutes and then transferred to a polyvinylidene fluoride membrane. The membrane was blocked with 5% milk in Tris-buffered saline with Tween 20 for 1 hour and then incubated with the following primary antibodies at 4°C overnight: anti-NK1R primary antibody, which recognizes the N-terminal domain (1:100; Novus, Cat. NB300-119), anti-Erk1/2 (1:800, Cell Signaling Technology, Beverly, Massachusetts), and anti-phosphorylated Erk1/2 (1:800, Cell Signaling Technology, followed by a horseradish peroxidase-conjugated secondary antibody. Mouse monoclonal antibody to β-actin (1:8000; Sigma, Tianjin kangsi pute biotechnology co., LTD, China) was used as an endogenous reference.
Luciferase Reporter Assay
The cDNA of NK1R-FL in MCF-10A cells was amplified by PCR, and the general method of recombinant DNA was used to clone the wild-type (WT) 3′-UTR and mutation type (MUT) 3′-UTR of NK1R-FL. The WT and MUT sequences of NK1R-FL were cloned into the pmirGLO-REPORT luciferase reporter vector (Ambion, Tianjin laibo technology co., LTD, China) to generate constructs of Luc-NK1R-FL-3′-UTR-WT and Luc-NK1R-FL-3′-UTR-MUT, followed by DNA sequencing verification. SK-BR-3 and MCF-10A cells cultured in 6-well plates were cotransfected with NK1R-FL-3′-UTR-WT or NK1R-FL-3′-UTR-MUT, miR-206 mimic control or NC, while MDA-MB-231 cells were cotransfected with NK1R-FL-3′-UTR-WT or NK1R-FL-3′-UTR-MUT, As-miR-206, or inhibitor control with a 50 nM final concentration by using Lipofectamine™ 2000 reagent (Invitrogen) according to the manufacturer’s instructions. After transfection for 48 hours, the cell lysates were analyzed by using a luciferase reporter assay system (Promega, Beijing errui medical technology development co., LTD, China).
Measurement of Ins(1, 4, 5)P3 Content
To detect the character of miR-206 in the regulation of NK1R-FL-dependent intracellular signaling, we monitored changes in the concentration of cellular Ins(1, 4, 5)P3. Thus, we selected SK-BR-3 and MCF-10A cell lines that express NK1R-FL as target groups and the MDA-MB-231 cell line, which does not express NK1R-FL, as a control group. These cells were treated with 10−7 M SP at different times. In some cases, cells transfected with miR-206 or As-miR-206 were incubated with the NK1R antagonist Aprepitant (C23H21F7N4O3, Merck) and then stimulated with 10−7 M SP. In other cases, the cells that had been transfected with miR-206 or As-miR-206 and subsequently stimulated with 10−7 M SP at the same time points. Then, the 3 groups of cells that with different treatments were all harvested and used to detect the Ins(1, 4, 5)P3 content with the d-myo-inositol-1,4,5-trisphosphate[3H] assay kit (GE healthcare, General Electric Co., USA). A standard curve was drawn according to the levels of Ins(1, 4, 5)P3.
Transwell Migration and Invasion Assays
The migration and invasion assays were performed using 24-well Transwell chambers (8 µm; with MilliCell chambers; Millibo company, China). For the migration assay, the breast cancer cells, at 24 hours after transfection, were resuspended in RPMI-1640 medium and adjusted to 2 × 105 cells/mL and subsequently plated in the upper chambers. Then, in the bottom chambers, we added in 0.5 mL RPMI-1640 medium comprising 10% FBS. After incubating for 24 hours, the cells were scrubbed off the upper surface of the membrane, and the migrated cells were fixed with ice-cold ethanol and then stained using a 3-step stain set kit and counted with a light microscope (100×, 3 random fields per well). Except that the upper chambers covered with a mixture of serum-free RPMI-1640 medium and Matrigel (1:3; BD Biosciences, Tianjin taierui technology co., LTD, China), the invasion assay was similar to the migration assay protocol.
Cell Counting Kit 8 Assay
With cell counting kit 8 (Dojindo, Gaithersburg, Maryland), we performed the cell proliferation assay according to manufacturer’s protocol. Logarithmic growth phase SK-BR-3 cells transfected with miR-206 mimic control or NC were plated in 96-well plates at a concentration of 2 × 103 cells per well. Additionally, MDA-MB-231 cells were transfected with As-miR-206 or IC, and the cell viability at different points (24, 48, 72, 96, and 120 hours) was calculated by a micro-enzyme linked immunosorbent assay reader using a filter for 3 hours at 37°C. Each experiment was repeated 3 times using 6 replicates for each time.
Colony Formation Assay
Breast cancer cells were seeded on to 6-cm dishes. After 24 hours, the cells were transfected with miR-206 mimic or As-miR-206 and then incubated at 37°C in a 5%CO2 humidified atmosphere. For colony formation, the cells were counted, and 900 cells were seeded onto 6-cm dishes and cultured for 7 to 10 days in a sterile incubator maintained 37°C with 5% CO2. We fixed the colonies with ice-cold methanol at 4°C and then stained the cells with a 3-Step Stain Set kit (Thermo Scientific, Kalamazoo, Michigan).
Nude Mice Assay
Female BALB/c-nu mice (4-5 weeks of age, 18-20 g) were purchased from Speyfor Biotechnology Company of Beijing and housed in barrier facilities on a 12-hour light–dark cycle. All experimental procedures were approved by the Animal Ethical Committee of Tianjin Medical University Cancer Institute and Hospital. For tumor formation assay, the BALB/c nude mice were randomly divided into 2 groups (n = 5/group). MDA-MB-231 cells were infected with miR-206 agomir and its control. The cells were resuspended in phosphate-buffered saline at a concentration of 2.5 × 106 cells in 0.25 mL and injected into the mammary fat pads of female athymic mice. Tumors were examined twice weekly. The length, width, and thickness measurements were obtained with calipers, and the tumor volumes were calculated. On day 27, the animals were euthanized, and the tumors were excised and weighed.
For metastasis assay in vivo, other 10 mice were randomly divided into 2 groups (n = 5/group). MDA-MB-231 and SK-BR-3 miR-206 overexpressing and mimic control cells were injected into the tail vein of SCID mice. All mice were observed on a weekly basis after 5 weeks of intravenous injection; the lung tissues of each mouse were dissected and fixed in 100-mL buffered formalin for 24 hours. The number of metastases per lung was analyzed both macroscopically and microscopically and was quantified for each treatment group. Surface metastases were counted first, and then paraffin-embedded sections of each tissue were stained routinely with H&E. The histopathological examination was carried out using microscopy, and the metastatic nodules were recorded. The incidence of metastasis was calculated as the percentage of mice that had ≥1 lung metastases.
Statistical Analysis
All statistical analyses were performed with the SPSS statistical software package (version 20.0). Each in vitro quantitative test was independently replicated, and all data are presented as the means ± standard deviations. One-way analysis of variance (ANOVA) was used to compare the expression levels of luciferase activities, transfer cell numbers, and grayscale values of immunohistochemistry staining among the different groups. Two-way repeated measures ANOVAs and Bonferroni tests were used to measure the proliferation curves in vitro. Spearman rank correlation coefficient was used as a statistical measure of association. All the statistical tests were 2-sided, and P < .05 was considered to indicate statistical significance.
Results
mir-206 and NK1R-FL Expression in Breast Tumor Tissues and Cell Lines
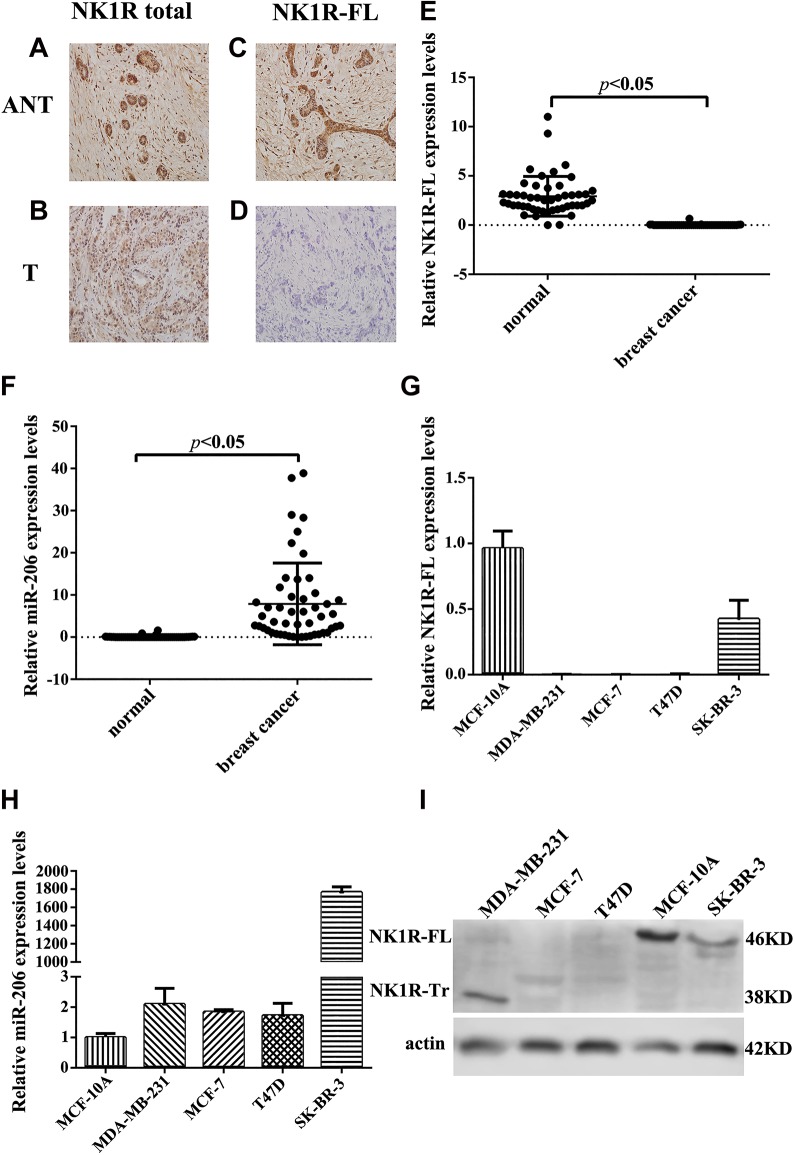
To investigate the role of total NK1R and NK1R-FL in the progression of human breast cancer, we examined the expression of total receptor and the NK1R-FL isoform in clinical breast cancer tissue and paired adjacent normal tissue samples from 82 patients by immunohistochemistry. The present results showed that total NK1R and NK1R-FL were mainly located at the membrane of breast cancer cells, especially within the cytoplasm (Figure 1A-D). Immunohistochemical analysis also showed that NK1R-FL expression was significantly deceased in breast cancer tissues when compared to paired adjacent normal tissues (Figure1E). Full-length neurokinin-1 (NK1R-FL) expression was lower in invasive ductal carcinoma (33.8%) than in intraductal carcinoma (78.6%). We also confirmed that lower NK1R-FL expression was associated with tumor metastasis and advanced TNM stage, especially where tumors were of more advanced histological grade. Moreover, NK1R-FL expression was significantly lower in ERα-negative, PR-negative, and HER-2 positive breast cancer tissues (P < .05, Table 1). To further examine correlations between the level of miR-206 expression in human breast cancer tissues and clinicopathological features, the 82 patients were assigned to either a positive or a negative expression group according to their median miR-206 expression score. Expression of miR-206 was significantly higher in breast cancer tissues than in paired adjacent normal tissues, especially in ERα-negative, PR-negative, and HER-2 positive breast cancer tissues (P < .05, Table 1). In contrast to NK1R-FL, the expression of miR-206 was higher in invasive ductal carcinoma (38.3%) than in intraductal carcinoma (14.3%). We also confirmed that miR-206 positivity was significantly associated with tumor metastasis and advanced TNM stage, especially when the tumors were of advanced histological grade. Expression of miR-206 and NK1R-FL was also examined in human breast cell lines (metastatic breast cancer cell lines MDA-MB-231, T47D, MCF-7, and SK-BR-3) and the nontumorigenic breast epithelial cell line MCF-10A by qRT–PCR and normalized to U6 small nuclear RNA or β-actin. In MDA-MB-231, T47D, and MCF-7 breast cancer cell lines, miR-206 expression was significantly upregulated compared to that in control MCF-10A cells (Figure 1H and I). As shown in Figure 1G and I, MDA-MB-231, T47D, and MCF-7 cells expressed only low levels of NK1R-FL. Given the inverse relationship between NK1R-FL and miR-206 expression, we hypothesized that miR-206 could regulate NK1R-FL expression in breast cancer cell lines. Levels of NK1R-FL were much higher in MCF-10A cells than in SK-BR-3 cells as determined by qRT–PCR and Western blot analysis. SK-BR-3, MCF-10A, and MDA-MB-231 cells were therefore selected for analysis in further studies to confirm the role of miR-206 in the regulation of NK1R-FL expression.
Figure 1.
Immunohistochemical analysis of total NK1R and NK1R-FL protein expression in breast tumor tissues (T) and paired adjacent normal tissues (ANT). (A and B) Positive staining of total NK1R in ANT and breast tumor tissues (at ×100). (C) Positive staining of NK1R-FL in ANT (at ×100). (D) Weak staining of NK1R-FL in breast tumor tissues (at ×100). The expression levels of NK1R-FL and miR-206 in human breast tissues and cell lines. (E and F) Average expression level of NK1R-FL and miR-206 in human breast tumor tissues (n = 82) and adjacent normal tissues (n = 82). mRNA and miR-206 abundance was assessed by real-time quantitative PCR and normalized to β-actin or U6 RNA. (G and H) Real-time quantitative PCR analysis of NK1R-FL and miR-206 in human breast cancer cell lines (metastatic breast cancer cell lines MDA-MB-231, T47D, MCF-7, and SK-BR-3) and nontumorigenic epithelial cell line MCF-10A. *P < .05 versus normal control. (I) The expression levels of NK1R-FL in human breast cancer cells (metastatic breast cancer cells MDA-MB-231, T47D, MCF-7, and SK-BR-3) and MCF-10A in nontumorigenic epithelial cells were detected by Western blotting.
Table 1.
Relationship Between NK1R, NK1R-FL,miRNA-206, and Clinic Pathological Features in Breast Cancer Patients
| Clinical Pathology | n | NK1R Positive (%) | P | NK1R-FL Positive (%) | P | miRNA-206 positive (%) | P | |
|---|---|---|---|---|---|---|---|---|
| Age, years | <50 | 44 | 93.2 | .382 | 47.77 | .16 | 36.4 | .263 |
| ≥50 | 38 | 97.4 | 34.2 | 31.6 | ||||
| Histological type | Invasive ductal carcinoma | 68 | 97.1 | .681 | 33.8 | <.01a | 38.3 | <.01a |
| Intraductal carcinoma | 14 | 100.0 | 78.6 | 14.3 | ||||
| TNM stages | Ⅰ | 40 | 100.0 | .234 | 67.5 | <.01a | 27.5 | <.01a |
| Ⅱ | 34 | 94.1 | 20.6 | 35.3 | ||||
| Ⅲ | 8 | 100.0 | 0.00 | 58.5 | ||||
| Histological grade | Ⅰ | 17 | 100.0 | .533 | 50.0 | <.01a | 29.4 | <.01a |
| Ⅱ | 50 | 96.0 | 44.0 | 24.0 | ||||
| Ⅲ | 15 | 100.0 | 14.3 | 73.3 | ||||
| Lymphnode metastasis | no | 52 | 98.1 | .688 | 61.5 | <.01a | 13.4 | <.01a |
| yes | 30 | 96.7 | 6.7 | 69.4 | ||||
| ER | negative | 20 | 100.0 | .415 | 15.0 | <.01a | 76.1 | <.01a |
| positive | 62 | 96.8 | 50.0 | 20.9 | ||||
| PR | negative | 32 | 100.0 | .252 | 12.5 | <.01a | 58.9 | <.01a |
| positive | 50 | 96.0 | 60.0 | 17.4 | ||||
| HER-2 | negative | 54 | 96.3 | .304 | 47.9 | <.05b | 11.2 | <.01a |
| positive | 28 | 100.0 | 17.9 | 78.6 | ||||
a P < .01.
b P < .05.
mir-206 Targets the 3′UTR of the NK1R-FL mRNA and Downregulates its Expression
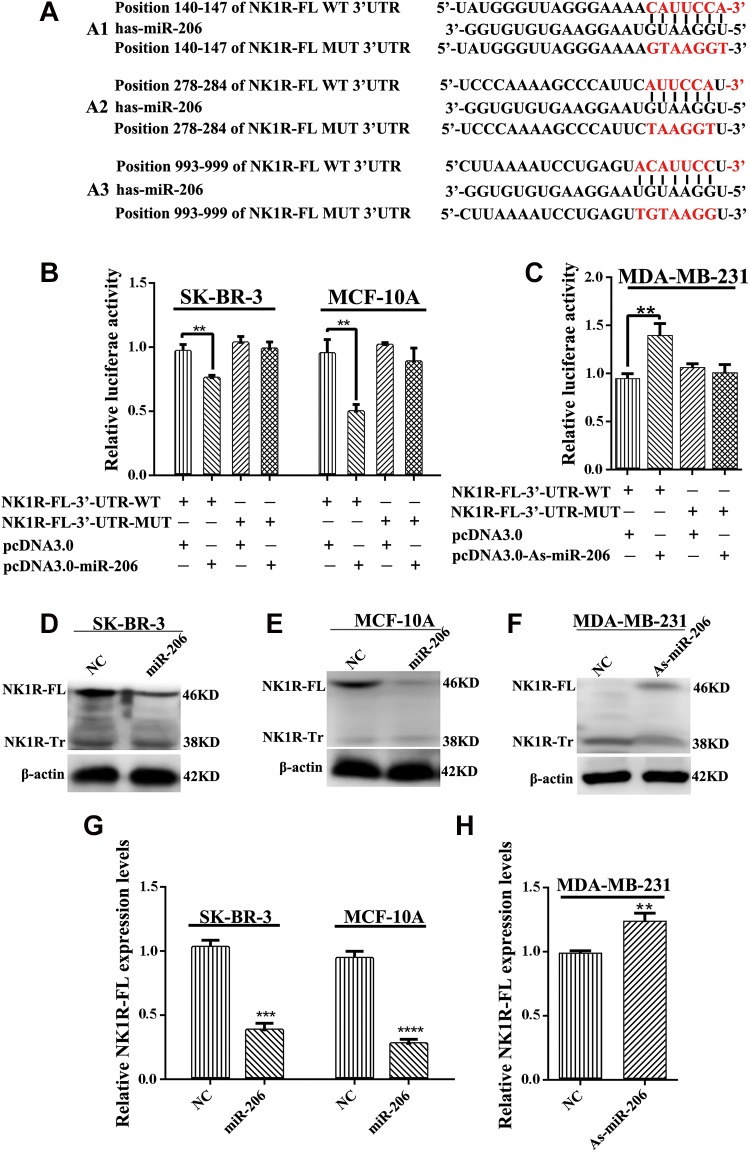
Using microRNA.org, miRanda, Target-Scan, and Pictar databases, we identified 3 candidate miR-206 target sequences within the 3′-UTR of NK1R-FL, and there is no miR-206 binding site within the 3′-UTR of NK1R-Tr. To determine whether miR-206 can regulate NK1R-FL by binding to its 3′-UTR, we constructed a luciferase reporter encoding the 3′-UTR of NK1R-FL and a variant that contained 3 mutated bases within the predicted miR-206 target sites of the 3′-UTR (Figure 2 A1-3). SK-BR-3 and MCF-10A cells were transiently transfected with pmirGLO-NK1R-FL 3′-UTR and miR-206 mimics or NC. In addition, MDA-MB-231 cells were transiently transfected with As-miR-206 or IC.
Figure 2.
miR-206 downregulation NK1R-FL expression. (A) A1, A2, and A3 Discovery of 3 potential miR-206 target sites within the 3′-UTR of NK1R-FL according to the bioinformatics analysis. A putative miR-206 binding target sites in the 3′-UTR of NK1R-FL mRNA among mammalian species (upper panel); site-directed mutations in the sequence complimentary to the seed region for miR-206 (lower panel). (B and C) Relative luciferase activity of NK1R-FL-3′-UTR-WT and NK1R-FL-3′-UTR-MUT in SK-BR-3 and MCF-10A cells cotransfected with miR-206 mimic or negative control, and in MDA-MB-231 cells with As-miR-206 or inhibitor control. (D, E, and F) Protein levels of NK1R-FL were determined by western blotting in (D, E) SK-BR-3 and MCF-10A cells transfected with miR-206 mimic or negative control; MDA-MB-231 cells transfected with As-miR-206 or inhibitor control (F). (G and H) After transfection, real-time quantitative PCR was used to examine the mRNA expression of NK1R-FL in SK-BR-3, MCF-10A and MDA-MB-231 cells. *P < .05, **P < .01, ***P < .0005, ****P < .0001 versus normal control.
As shown in Figure 2B and C, luciferase activity in SK-BR-3 and MCF-10A cells substantially decreased after miR-206 transfection, and this inhibition could be prevented when the 3 putative miR-206 binding sites were mutated. In contrast, luciferase activity increased in MDA-MB-231 cells following transfection with As-miR-206.
To further show that NK1R-FL is a target gene of miR-206, SK-BR-3 and MCF-10A cells were transiently transfected with miR-206 and the expression of NK1R-FL subsequently examined. Western blot analysis revealed that NK1R-FL expression was decreased in SK-BR-3 and MCF-10A cells following transfection with miR-206 when compared to that in the control group (Figure 2D and E). Furthermore, the transfection of MDA-MB-231 cells with As-miR-206 resulted in significant upregulation of NK1R-FL expression. The abovementioned findings were also confirmed by qRT–PCR analysis (Figure 2G and H). There are 2 naturally occurring forms of NK1R: NK1R-FL and NK1R-Tr. Previous studies have shown that these 2 forms of NK1R were inversely expressed in cancer cells: one was upregulated and the other was downregulated. In the present study, MDA-MB-231 cell expression of NK1R-FL after transient transfection with As-miR-206, but the NK1R-Tr expression was downregulated (Figure 2F). Therefore, luciferase activity increased in MDA-MB-231 cells following transfection with As-miR-206. Collectively, these results demonstrate that NK1R-FL is a direct target of miR-206 and that miR-206 regulates NK1R-FL expression by directly binding the 3′-UTR of NK1R-FL mRNA.
mir-206 Regulate SP-Induced Ca2+ Influx and Erk1/2 Activation by Targeting NK1R-FL
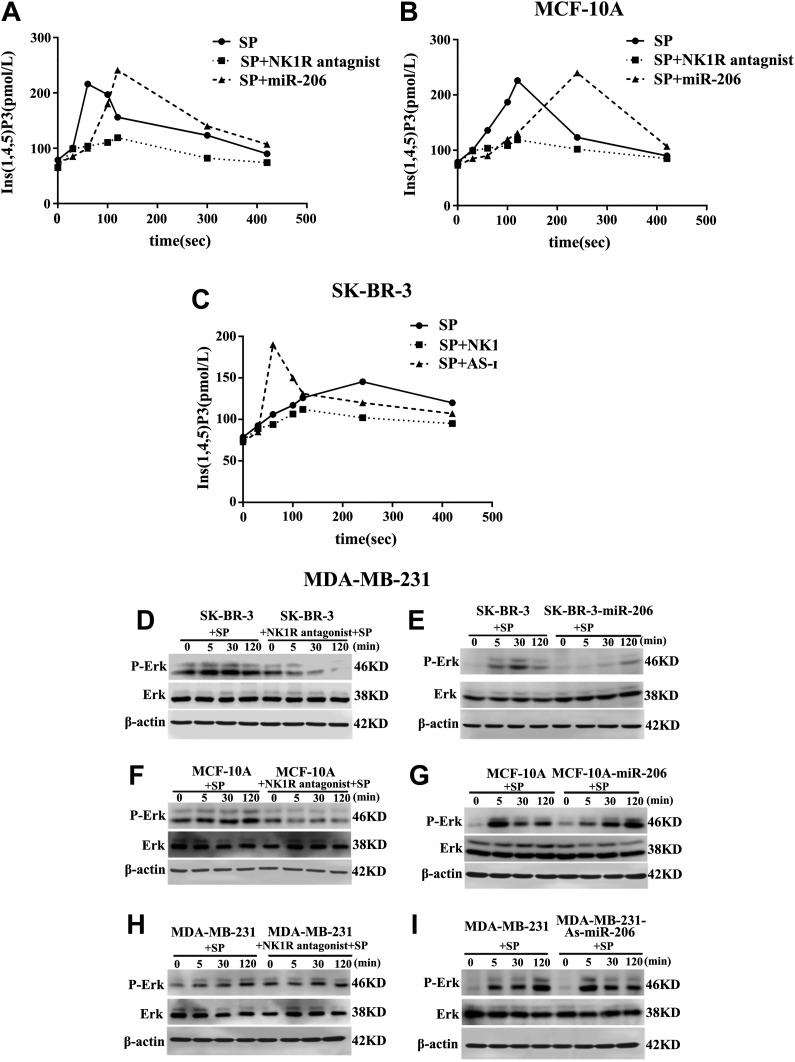
In a previous study, we examined NK1R-FL and NK1R-Tr function in breast cancer cells, and the results showed that in cells with high NK1R-FL expression, such as MCF-10A and HMSC-bm, SP triggered rapid and high level of Ins(1,4,5)P3 response, while in SK-BR-3 cells, Ins(1,4,5)P3 levels were lower and slowly increased after SP stimulation. In MDA-MB-231 cells, there was low Ins(1,4,5)P3 response by SP stimulation. In the extracellular signal-regulated kinase (ERK) phosphorylation assay, SP potently and rapidly activated ERK1/2 as early as 5 minutes in MCF-10A cells, but in MDA-MB-231 cells, the ERK1/2 phosphorylation with activation of truncated NK1R increased slowly from 5 to 120 minutes with SP stimulation. In the present study, Ins(1,4,5)P3 and ERK1/2 phosphorylation was also evaluated NK1R function.
Compared to the control group, after cells were transfected with miR-206 and then stimulated 100 s by 10−7 mol/L SP, IP3 levels were significantly decreased, and the increased speed and magnitude were also lower in nontumorigenic MCF-10A breast cells and breast cancer cells SK-BR-3 (Figure 3A and B). After treatment with the NK1R antagonist Aprepitant for 30 minutes, the IP3 levels were almost unchanged after 10−7 mol/L SP stimulation when compared to the control group in MCF-10A and SK-BR-3 cells that transfected with miR-206 (Figure 3A and B). However, in MDA-MB-231 cells transfected with As-miR-206, the IP3 levels increased, and the speed and magnitude were higher (Figure 3C). Compared to the control group, the time of elevating IP3 was prolonged in transfected with miR-206 of MCF-10A and SK-BR-3 cell after stimulation with SP, while the time of elevating IP3 was increased in MDA-MB-231 cells transfected with As-miR-206.
Figure 3.
Effect of activated NK1R-FL on intracellular Ins(1,4,5)P3 response and ERK1/2 phosphorylation. (A, B, and C) The rise of intracellular IP3 in cells that express NK1R-FL or NK1R-Tr. Cells transfected with miR-206 or As-miR-206 were stimulated with 10−7 mol/L SP only or treated for 30 minutes with the NK1R antagonist Aprepitant after that. (D, F, and H) SK-BR-3, MCF-10A, and MDA-MB-231 cells that express NK1R-FL or NK1R-Tr transfected with miR-206 or As-miR-206, were treated for 30 minutes with the NK1R antagonist Aprepitant and then stimulated by 10−7 mol/L SP or untreated as control. (E, G, and I) SK-BR-3 and MCF-10A cells that express NK1R-FL or NK1R-Tr were transfected with miR-206 mimic or negative control, and in MDA-MB-231 cells, with As-miR-206 or inhibitor control or untransfected as a control. Phospho-ERK1/2 was detected by antiphospho-ERK1/2 antibody.
To study differences in the signaling downstream of NK1R-FL and NK1R-Tr, we next examined Erk1/2 activity in SK-BR-3, MCF-10A, and MDA-MB-231 cells. In MDA-MB-231 cells expressing the NK1R-Tr receptor, Erk1/2 phosphorylation was detected at approximately 30 minutes after treatment with SP, whereas in MCF-10A cells and SK-BR-3 cells stably expressing the NK1R-FL, Erk1/2 phosphorylation was detected within 5 minutes of SP treatment (Figure 3 D, F, and H). Next, we treated SK-BR-3 and MCF-10A cells with the NK1R antagonist Aprepitant 30 minutes to investigate whether the SP-dependent induction of Erk1/2 phosphorylation was specifically mediated by this receptor. As shown in Figure 3D, F, and H, SP-dependent phosphorylation of Erk1/2 was prevented in cells treated with Aprepitant, a known antagonist of SP. Treatment with this inhibitor had no effect on total cellular Erk1/2 levels.
Interestingly, in MCF-10A cells transfected with miR-206, a delay in Erk1/2 phosphorylation was observed following SP treatment when compared to that in controls. This result was also confirmed in SK-BR-3 (Figure 3E and G). Conversely, the transfection of MDA-MB-231 cells with As-miR-206 resulted in a more rapid phosphorylation of Erk1/2 (Figure 3I).
miR-206 Regulates the Invasive, Migration, and Proliferative Capacities of Breast Cancer Cells In Vitro
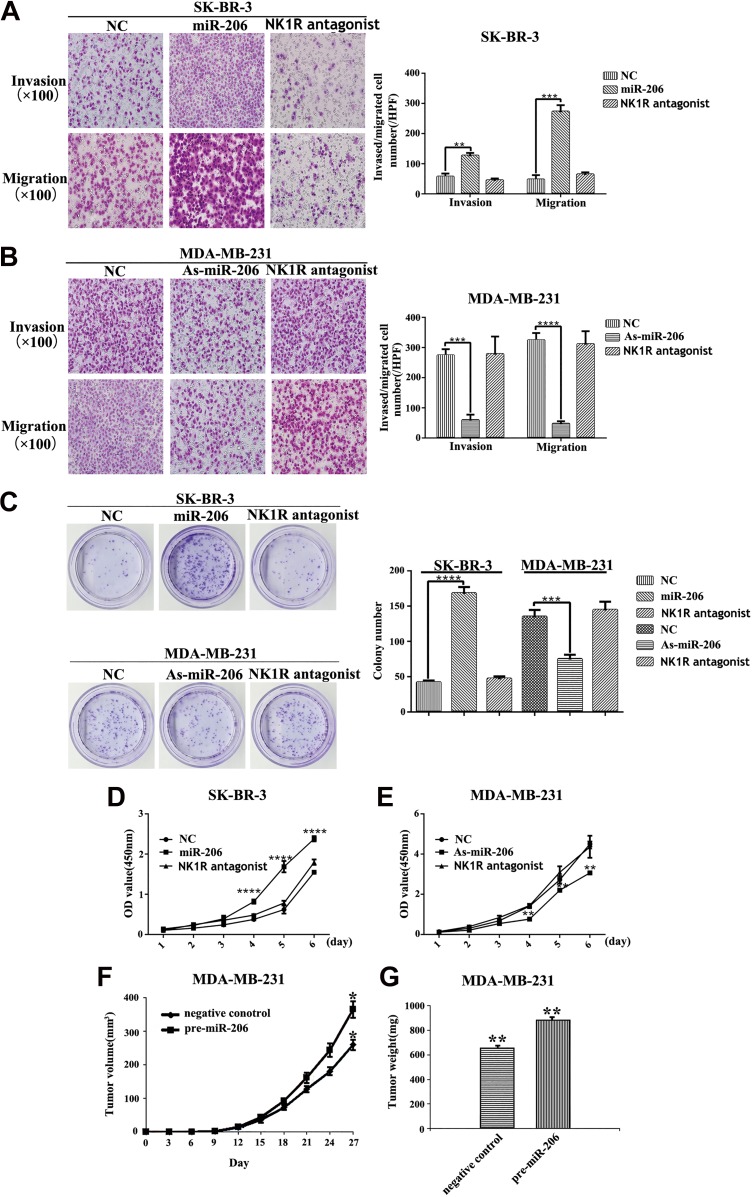
To investigate whether miR-206 regulates human breast cancer cell migration, invasion, proliferation, and colony formation, we selected MDA-MB-231 cells, which show strong migration and invasion potential, and SK-BR-3 cells, which show weak migration and invasion potential, for further study. We first performed an in vitro gain-of-function analysis by overexpression of miR-206 in SK-BR-3 cells. We found that transfection of miR-206 caused enhanced SK-BR-3 cell invasion and migration when compared with that of controls (Figure 4A). Ectopic expression of miR-206 also enhanced the proliferation, and meanwhile, the number of colony-forming potential of SK-BR-3 cells in vitro (Figure 4C and D). Furthermore, the inhibition of NK1R-FL with NK1R antagonist Aprepitant eliminated the effects of miR-206 transfection on human breast cancer cell invasion, migration, proliferation, and colony formation. These data clearly demonstrate that depletion of NK1R-FL drives SK-BR-3 cells proliferation and invasion when compared to the control groups.
Figure 4.
miR-206 promoted human breast cancer cell migration, invasion, proliferation, and colony formation. Growth of the SK-BR-3 cells transfected with miR-206 mimic or negative control (NC), and in MDA-MB-231 cells, with As-miR-206 or inhibitor control at an indicated time (at ×100). (A and B) Transwell assays on SK-BR-3 and MDA-MB-231 cells were employed to analyze the effect of miR-206 on migration. For the invasion assay, 1:3 Matrigel was added. **P < .01, ***P < .0005, ****P < .0001. (C) Representative photomicrographs of colony formation assay of SK-BR-3 and MDA-MB-231 cells with the transfected miR-206 ***P < .0005, ****P < .0001. (D and E) The cell counting kit-8 assay was performed to analyze the effect of miR-206 on proliferation in SK-BR-3 and MDA-MB-231 cells. Data are presented as the mean ± SD from 3 independent experiments. **P < .01 versus normal, control ****P < .0001 versus normal. The agomirs of miR-206 and their controls were inoculated into nude mice carrying MDA-MB-231 xenografts (n = 5/group). As shown in Figure 4F, tumors transfected with miR-206 agomirs grew significantly faster than the control tumors at different time points (*P < .05), which was consistent with the present in vitro cell proliferation results. Four weeks later, the weights of tumors in mice transfected by miR-206 silencing cells were significantly increased compared to control cells (**P < .01, Figure 4G).
Furthermore, to determine whether miR-206 can promote the invasive, migration, colony formation, and proliferative capacities of breast cancer cell lines in vitro, we next performed transwell invasion and migration assays by using MDA-MB-231 cells transfected with As-miR-206. As shown in Figure 4B, downregulation of miR-206 expression impaired cell invasion and migration compared to controls. The CCK-8 assay revealed that downregulation of miR-206 also significantly inhibited cell proliferation (Figure 4C). In MDA-MB-231 cells transfected with As-miR-206, colony formation was suppressed (Figure 4E). Furthermore, the pretreatment of MDA-MB-231 with NK1R antagonist Aprepitant inhibited the SP-dependent invasion, migration, proliferation, and colony formation of these cells when compared to that of the controls (Figure 4A-E). These data clearly showed that miR-206 promotes breast cancer cell invasion, migration, proliferation, and colony formation in vitro by binding to the 3′-UTR region of NK1R-FL.
Transfection of mir-206 Promotes Tumor Formation of Breast Cancer Cells In Vivo
Because overexpression of miR-206 facilitated breast cancer cell growth in vitro, we evaluated the effect on tumor formation in vivo. The agomirs of miR-206 and their controls were inoculated into nude mice carrying MDA-MB-231 xenografts (n = 5/group). As shown in Figure 4F, tumors transfected with miR-206 agomir grew significantly faster than the control tumors at different time points (P < .05), which was consistent with the present in vitro cell proliferation results. At 4 weeks later, the weights of tumors in mice transfected by miR-206 silencing cells were significantly increased compared to those in mice transected with control cells (P < .01, Figure 4G). These results suggest that miR-206 plays an important role in increasing the tumorigenicity of breast cancer cells in vivo. As for distant metastases, mice injected with miRNA-206-MDA-MB-231 and miR-206-SK-BR-3 cells exhibited much more distant lung metastases compared to mimic control groups. Together, these data suggest that miR-206 promotes in vivo tumorigenciy of MDA-MB-231 and SK-BR-3 cells and increase the distal pulmonary metastases, and NK1R-FL may be an important target involved in this process.
Discussion
The protumorigenic and antitumorigenic activities of the miR-206 are not mutually exclusive, as their functions will be dictated by the presence or absence of particular targets within in a given setting. In the present study, we demonstrated that miR-206 functions as a tumor promoter by targeting NK1R-FL in human breast cancer.
To date, 2 naturally occurring variants of NK1R have been reported in humans, the full-length receptor, NK1R-FL, and the truncated form of the receptor, NK1R-Tr.4. SP, a member of the tachykinin family of peptides encoded by TACR1 gene, plays an important signaling role in both the nervous and the immune systems.18 The 3 tachykinin receptors, NK1, NK2 and NK3, are all G protein-coupled receptors.19 SP is the natural ligand, shows the highest affinity for the NK1 receptor, and regulates the biological functions encoded by the C-terminal sequence of tachykinins receptors20. SP controls important biological functions related to cancer, such as tumor cell proliferation, tumor cell migration, invasion, and metastasis, neoangiogenesis, and tumor cell apoptotic.20-22 Numerous studies have reported the involvement of the SP/NK1R system in various types of cancers,23 including glioblastoma,24 pancreatic carcinoma,25 retinoblastoma,26 and breast cancer.27 SP induces mitogenesis in both normal cells and tumor cells and also protects tumor cells from apoptosis and to control their migration.28-30 Chronic inflammation may facilitate the development of pancreatic cancer through the SP/NK1R system because this system is upregulated in inflammatory processes.31 To further probe the function of NK1R forming in human breast cancer, we examined the expression of this receptor in human breast cancer tissues and a panel of human breast cancer cell lines. Initially, we analyzed correlation between the level of NK1R-FL and the clinical characteristics of 82 patients with breast cancer. We found that NK1R-FL expression was downregulated in breast cancer tissues when compared to adjacent normal tissues. Proliferation and colony formation assays revealed that NK1R-FL inhibited the growth of human breast cancer cells. In a previous study, we showed that the NK1R-FL expression is higher in the nontumorigenic epithelial cell lines MCF-10A and HMSC-bm when compared to that in the SK-BR-3 human breast cancer cell lines, and these findings were further confirmed in the present study. These results suggest that NK1R-FL performs the role of proto-oncogene in human breast cancer.
The dysregulation of miR-206s has been reported in various types of human cancers,32-34 but few studies have examined the role of miR-206 in breast cancer. miR-206 is highly expressed in human skeletal muscle and plays specific roles in modulating skeletal muscle proliferation and differentiation in vitro and in vivo. Previous data indicate that miR-206 may function as a tumor suppressor gene in colorectal cancer,35 lung cancer,36 and laryngeal squamous cell carcinoma,37 but its importance in breast cancer has, until now, largely been unexplored. NK1R-FL can be regulated by miR-206s at the post-transcriptional level. For example, miR-519b can bind to the 3′-UTR of NK1R-FL and suppress its translation, resulting in higher level of NK1R-Tr protein expression and the promotion of cancer progression,38 However, until now, it has not been clear whether miR-206 regulates the expression of NK1R-FL in human breast cancer. The present study indicates that miR-206 directly targets and downregulates the expression of NK1R-FL in human breast cancer cells. Given that cell invasion, migration, proliferation, and colony formation induced by miR-206 expression were abrogated by the inhibition of NK1R-FL. Moreover, the observed inverse correlation between NK1R-FL and miR-206 expression in breast cancer tissues and cell lines indicates that the up-regulation of miR-206 may contribute to the malignant phenotype of breast cancer by maintaining a low level of NK1R-FL. Through binding to NK1R, SP promotes the activation of members of the mitogen-activated protein kinase (MAPK) family, which includes ERK1/2 and p38 MAPK.20,39,40 These pathways play a critical role in cell cycle progression, proliferation, and apoptosis.41 The ERK1/2 response differed following the activation of NK1R-FL and NK1R-Tr, which are coupled to different second messenger systems. The present studies demonstrated that the overexpression of miR-206 in MCF-10A and SK-BR-3 cells prolonged the duration of elevated intracellular Ca2+ concentrations and ERK1/2 activation in response to SP stimulation and that this effect could be prevented following the inhibition of NK1R-FL.
In this study, miR-206 and NK1R antagonist showed different effects on IP3 release, ERK activation, cell migration, and colony formation. miR-206s recognize their target sites by incomplete base pairing, and individual miR-206s may regulate a cohort of mRNAs. Consequently, miR-206s affect a multitude of signaling pathways. In addition to NK1R-FL gene, other oncogenes were also identified as targets of miR-206. miR-206 targets the estrogen receptor and is downregulated in estrogen receptor-positive breast cancer. miR-206, as a proapoptotic activator of cell death, was associated with its inhibition of notch3 signaling and tumor formation. miR-206 is a negative regulator of NF-κB signaling and, thereby, miR-206 as a tumor suppressor by inhibiting tumor growth, cancer cell invasiveness, and release of an NF-κB-dependent circuit of proangiogenic cytokines and growth factors. Therefore, estrogen receptor and notch3 could also be regulated by miR-206, except the NK1R-FL gene in the present study. Compared to miR-206, NK1R antagonist has high affinity for NK1R, could significantly abolish the effect of substance P on NK1R, and exerted antitumor action in breast cancer cells. miR-206 targets many important genes, such as estrogen receptor and Notch. Therefore, regulated by miR-206, changes in these genes and other unclear mechanisms result in the duration of elevated intracellular Ca2+ concentration and ERK1/2 activation in response to SP stimulation and promotes the invasive, migration, and proliferative capacities of breast cancer cells. Finally, the inhibition of tumor formation by miR-206 agomir and NK1R silencing in the present in vivo mouse experiments confirmed the suppression of proliferation in vitro. We also demonstrated that miR-206 overexpression promoted breast cancer cell metastasis by targeting MDA-MB-231 and SK-BR-3 in vitro and in vivo, and its upregulation in breast cancer tissues was positively correlated with distant metastasis in patients.
Altogether, these results indicated that miR-206 can directly regulate NK1R-FL expression. We have further showed that NK1R-FL is potentially involved in the miR-206-mediated repression of cellular growth in NK1R-FL-positive breast cancer cells. Numerous studies have reported that NK1R antagonist may prove useful as therapeutic agents in the treatment of cancer, for example, in head and neck cancer.42 Thus, it would be interesting is to further explore whether miR-206 could serve as a potential therapeutic agent in the treatment of cancer.
Supplemental Material
Supplemental Material, Abbreviations for miR-206 Promotes Cancer Progression by Targeting Full-Length Neurokinin-1 Receptor in Breast Cancer by Yu Zhou, Meng Wang, Yingna Tong, Xiaobin Liu, Lufang Zhang, Dong Dong, Jie Shao and Yunli Zhou in Technology in Cancer Research & Treatment
Abbreviations
- ANOVA
analysis of variance
- As-miR-206
antisense miR-206
- cDNA
complementary DNA
- Ct
threshold cycle
- ERK
extracellular signal-regulated kinase
- FBS
fetal bovine serum
- H&E
hematoxylin and eosin
- IC
inhibitor control
- IP3
inositol 1,4,5-triphosphate
- MAPK
mitogen-activated protein kinase
- MUT
mutation type
- NC
negative control
- NK1R
neurokinin-1
- NK1R-FL
full-length neurokinin-1
- NK1R-Tr
truncated neurokinin-1
- PCR
polymerase chain reaction
- qRT-PCR
quantitative real time–polymerase chain reaction
- SP
substance P
- 3′-UTR
3′-untranslated region
- WT
wild-type
Footnotes
Author Notes: Zhou, Zhou, and Wang equally contribute to this work.
Ethical Approval: All procedures performed in studies involving human participants were conducted in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. Our study was approved by The Tianjin Medical University Cancer Hospital Ethics Committee (approval no. 201310011). All patients provided written informed consent prior to enrollment in the study.
Declaration of Conflicting Interests: The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: The author(s) received the financial support for the research, authorship, and/or publication of this article: The present study was financially supported by grants from the National Natural Science foundation of China [grant number 81201653] to Zhou Y., the Tianjin natural science foundation [grant number 16JCYBJC26000] to Zhou Y., the National Natural Science foundation of China [grant number 81502519] to DongDong, and the National Natural Science foundation of China [grant number 3150109] to ShaoJie, and the Science foundation of Tianjin Medical University [grant number 2014KYM04] to Pengyu Zhang.
ORCID iD: Yu Zhou  https://orcid.org/0000-0003-0059-6265
https://orcid.org/0000-0003-0059-6265
Yunli Zhou  https://orcid.org/0000-0002-9547-3366
https://orcid.org/0000-0002-9547-3366
Supplemental Material: Supplemental material for this article is available online.
References
- 1. DeSantis C, Ma J, Bryan L, Jemal A. Breast cancer statistics, 2013. CA Cancer J Clin. 2014;64(1):52–62. [DOI] [PubMed] [Google Scholar]
- 2. Liu Y, Cai Q, Bao PP, et al. Tumor tissue microRNA expression in association with triple-negative breast cancer outcomes. Breast Cancer Res Treat. 2015;152(1):183–191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Matamala N, Vargas MT, Gonzalez-Campora R, et al. Tumor microRNA expression profiling identifies circulating microRNAs for early breast cancer detection. Clin Chem. 2015;61(8):1098–1106. [DOI] [PubMed] [Google Scholar]
- 4. Sloniecka M, Le Roux S, Boman P, Bystrom B, Zhou Q, Danielson P. Expression profiles of neuropeptides, neurotransmitters, and their receptors in human keratocytes in vitro and in situ. PloS One. 2015;10(7):e0134157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Tebas P, Spitsin S, Barrett JS, et al. Reduction of soluble CD163, substance P, programmed death 1 and inflammatory markers: phase 1B trial of aprepitant in HIV-1-infected adults. Aids. 2015;29(8):931–939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Zhang J, Li H, Teng H, et al. Regulation of peripheral clock to oscillation of substance P contributes to circadian inflammatory pain. Anesthesiology. 2012;117(1):149–160. [DOI] [PubMed] [Google Scholar]
- 7. Mukda S, Moller M, Ebadi M, Govitrapong P. The modulatory effect of substance P on rat pineal norepinephrine release and melatonin secretion. Neurosci Lett. 2009;461(3):258–261. [DOI] [PubMed] [Google Scholar]
- 8. Sideri A, Bakirtzi K, Shih DQ, et al. Substance P mediates pro-inflammatory cytokine release form mesenteric adipocytes in inflammatory bowel disease patients. Cell Mol Gastroenterol Hepatol. 2015;1(4):420–432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Spitsin S, Stevens KE, Douglas SD. Expression of substance P, neurokinin-1 receptor and immune markers in the brains of individuals with HIV-associated neuropathology. J Neurol Sci. 2013;334(1-2):18–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Zhou Y, Zhao L, Xiong T, et al. Roles of full-length and truncated neurokinin-1 receptors on tumor progression and distant metastasis in human breast cancer. Breast Cancer Res Treat. 2013;140(1):49–61. [DOI] [PubMed] [Google Scholar]
- 11. Osman A. MicroRNAs in health and disease--basic science and clinical applications. Clin Lab. 2012;58(5-6):393–402. [PubMed] [Google Scholar]
- 12. Zhang L, Liu X, Jin H, et al. MiR-206 inhibits gastric cancer proliferation in part by repressing cyclinD2. Cancer Lett. 2013;332(1):94–101. [DOI] [PubMed] [Google Scholar]
- 13. Parasramka MA, Dashwood WM, Wang R, et al. A role for low-abundance miRNAs in colon cancer: the miR-206/kruppel-like factor 4 (KLF4) axis. Clin Epigenetics. 2012;4(1):16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Wang R, Hu Y, Song G, et al. MiR-206 regulates neural cells proliferation and apoptosis via Otx2. Cell Physiol Biochem.2012;29(3-4):381–390. [DOI] [PubMed] [Google Scholar]
- 15. Sun C, Liu Z, Li S, et al. Down-regulation of c-Met and Bcl2 by microRNA-206, activates apoptosis, and inhibits tumor cell proliferation, migration and colony formation. Oncotarget. 2015;6(28):25533–25574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Cai Y, Li H, Zhang Y. Downregulation of microRNA-206 suppresses clear cell renal carcinoma proliferation and invasion by targeting vascular endothelial growth factor A. Oncol Rep. 2016;35(3):1778–1786. [DOI] [PubMed] [Google Scholar]
- 17. Quan Y, Huang X, Quan X. Expression of miRNA-206 and miRNA-145 in breast cancer and correlation with prognosis. Oncol Lett. 2018;16(5):6638–6642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Park JH, Kim S, Hong HS, Son Y. Substance P promotes diabetic wound healing by modulating inflammation and restoring cellular activity of mesenchymal stem cells. Wound Repair Regen. 2016;24(2):337–348. [DOI] [PubMed] [Google Scholar]
- 19. Pennefather JN, Lecci A, Candenas ML, Patak E, Pinto FM, Maggi CA. Tachykinins and tachykinin receptors: a growing family. Life Sci. 2004;74(12):1445–1463. [DOI] [PubMed] [Google Scholar]
- 20. Munoz M, Covenas R. Involvement of substance P and the NK-1 receptor in cancer progression. Peptides. 2013;48:1–9. [DOI] [PubMed] [Google Scholar]
- 21. Munoz M, Covenas R. Involvement of substance P and the NK-1 receptor in human pathology. Amino Acids. 2014;46(7):1727–1750. [DOI] [PubMed] [Google Scholar]
- 22. Munoz M, Covenas R. Involvement of substance P and the NK-1 receptor in pancreatic cancer. World J Gastroenterol. 2014;20(9):2321–2334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Munoz M, Rosso M, Covenas R. The NK-1 receptor: a new target in cancer therapy. Curr Drug Targets. 2011;12(6):909–921. [DOI] [PubMed] [Google Scholar]
- 24. Akazawa T, Kwatra SG, Goldsmith LE, et al. A constitutively active form of neurokinin 1 receptor and neurokinin 1 receptor-mediated apoptosis in glioblastomas. J Neurochem. 2009;109(4):1079–1086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Friess H, Zhu Z, Liard V, et al. Neurokinin-1 receptor expression and its potential effects on tumor growth in human pancreatic cancer. Lab Invest. 2003;83(5):731–742. [DOI] [PubMed] [Google Scholar]
- 26. Munoz M, Rosso M, Covenas R, Montero I, Gonzalez-Moles MA, Robles MJ. Neurokinin-1 receptors located in human retinoblastoma cell lines: antitumor action of its antagonist, L-732,138. Invest Ophthalmol Vis Sci. 2007;48(6):2775–2781. [DOI] [PubMed] [Google Scholar]
- 27. Ramkissoon SH, Patel PS, Taborga M, Rameshwar P. Nuclear factor-kappaB is central to the expression of truncated neurokinin-1 receptor in breast cancer: implication for breast cancer cell quiescence within bone marrow stroma. Cancer Res. 2007;67(4):1653–1659. [DOI] [PubMed] [Google Scholar]
- 28. DeFea KA, Vaughn ZD, O’Bryan EM, Nishijima D, Dery O, Bunnett NW. The proliferative and antiapoptotic effects of substance P are facilitated by formation of a beta -arrestin-dependent scaffolding complex. Proc Natl Acad Sci U S A. 2000;97(20):11086–11091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Feng F, Yang J, Tong L, et al. Substance P immunoreactive nerve fibres are related to gastric cancer differentiation status and could promote proliferation and migration of gastric cancer cells. Cell Biol Int. 2011;35(6):623–629. [DOI] [PubMed] [Google Scholar]
- 30. Munoz M, Rosso M, Covenas R. A new frontier in the treatment of cancer: NK-1 receptor antagonists. Curr Med Chem. 2010;17(6):504–516. [DOI] [PubMed] [Google Scholar]
- 31. Munoz M, Covenas R. Targeting NK-1 receptors to prevent and treat pancreatic cancer: a new therapeutic approach. Cancers(Basel). 2015;7(3):1215–1232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Chen Y, Gao Y, Zhang K, et al. MicroRNAs as regulators of cisplatin resistance in lung cancer. Cell Physiol Biochem. 2015;37(5):1869–1880. [DOI] [PubMed] [Google Scholar]
- 33. Deng K, Wang H, Guo X, Xia J. The cross talk between long, non-coding RNAs and microRNAs in gastric cancer. Acta Biochim Biophys Sin (Shanghai).2016;48(2):111–116. [DOI] [PubMed] [Google Scholar]
- 34. Huang YK, Yu JC. Circulating microRNAs and long non-coding RNAs in gastric cancer diagnosis: an update and review. World J Gastroenterol. 2015;21(34):9863–9886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Ren XL, He GY, Li XM, et al. MicroRNA-206 functions as a tumor suppressor in colorectal cancer by targeting FMNL2. J Cancer Res Clin Oncol. 2016;142(3):581–592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Zhang YJ, Xu F, Zhang YJ, Li HB, Han JC, Li L. . MiR-206 inhibits non small cell lung cancer cell proliferation and invasion by targeting SOX9. Int J Clin Exp Med. 2015;8(6):9107–9113. [PMC free article] [PubMed] [Google Scholar]
- 37. Yu WF, Wang HM, Lu BC, Zhang GZ, Ma HM, Wu ZY. miR-206 inhibits human laryngeal squamous cell carcinoma cell growth by regulation of cyclinD2. Eur Rev Med Pharmacol Sci.2015;19(14):2697–2702. [PubMed] [Google Scholar]
- 38. Navarro P, Ramkissoon SH, Shah S, et al. An indirect role for oncomir-519b in the expression of truncated neurokinin-1 in breast cancer cells. Exp Cell Res. 2012;318(20):2604–2615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Sun J, Ramnath RD, Zhi L, Tamizhselvi R, Bhatia M. Substance P enhances NF-kappaB transactivation and chemokine response in murine macrophages via ERK1/2 and p38 MAPK signaling pathways. Am J Physiol Cell Physiol. 2008;294(6):C1586–C596. [DOI] [PubMed] [Google Scholar]
- 40. Miyano K, Morioka N, Sugimoto T, Shiraishi S, Uezono Y, Nakata Y. Activation of the neurokinin-1 receptor in rat spinal astrocytes induces Ca2+ release from IP3-sensitive Ca2+ stores and extracellular Ca2+ influx through TRPC3. Neurochem Int. 2010;57(8):923–934. [DOI] [PubMed] [Google Scholar]
- 41. Refaat A, Abdelhamed S, Saiki I, Sakurai H. Inhibition of p38 mitogen-activated protein kinase potentiates the apoptotic effect of berberine/tumor necrosis factor-related apoptosis-inducing ligand combination therapy. Oncol Lett. 2015;10(3):1907–1911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Kanazawa T, Misawa K, Misawa Y, et al. G-Protein-coupled receptors: next generation therapeutic targets in head and neck cancer? Toxins(Basel). 2015;7(8):2959–2984. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplemental Material, Abbreviations for miR-206 Promotes Cancer Progression by Targeting Full-Length Neurokinin-1 Receptor in Breast Cancer by Yu Zhou, Meng Wang, Yingna Tong, Xiaobin Liu, Lufang Zhang, Dong Dong, Jie Shao and Yunli Zhou in Technology in Cancer Research & Treatment