Abstract
Spinal activation of the cAMP pathway produces mechanical hyperalgesia, sensitizes nociceptive spinal neurons, and phosphorylates the transcription factor cAMP-responsive element binding protein (CREB), which initiates gene transcription. This study examined the role of the cAMP pathway in a model of chronic muscle pain by assessing associated behavioral changes and phosphorylation of CREB. Bilateral mechanical hyperalgesia of the paw was induced by administering two injections of acidic saline, 5 d apart, into the gastrocnemius muscle of male Sprague Dawley rats. Interestingly, the increases in immunoreactivity for CREB and phosphorylated CREB (p-CREB) in the spinal dorsal horn occur 24 hr, but not 1 week, after the second injection of acidic saline compared with pH 7.2 intramuscular injections. Spinal blockade of adenylate cyclase prevents the expected increase in p-CREB that occurs after intramuscular acid injection. The reversal of mechanical hyperalgesia by adenylate cyclase or protein kinase A inhibitors spinally follows a similar pattern with reversal at 24 hr, but not 1 week, compared with the vehicle controls. The p-CREB immunoreactivity in the superficial dorsal horn correlates with the mechanical withdrawal threshold such that increases in p-CREB are associated with decreases in threshold. Therefore, activation of the cAMP pathway in the spinal cord phosphorylates CREB and produces mechanical hyperalgesia associated with intramuscular acid injections. The mechanical hyperalgesia and phosphorylation of CREB depend on early activation of the cAMP pathway during the first 24 hr but are independent of the cAMP pathway by 1 week after intramuscular injection of acid.
Keywords: protein kinase A, adenylate cyclase, muscle, CREB, pain, spinal cord
Introduction
Chronic pain is an abnormal and nonprotective response. This condition is widespread: an estimated 6 million Americans are affected with fibromyalgia, making it a common pain condition (Bennett, 1995). Chronic pain, including musculoskeletal, is not well understood partially because of a lack of appropriate pain models. Current musculoskeletal pain models typically exhibit short-term hyperalgesia or significant tissue damage, or both (Mense, 1993; Schaible and Grubb, 1993). Our laboratory developed a unique model of chronic muscle hyperalgesia. This model is different from other muscle pain models because there is long-lasting bilateral mechanical hyperalgesia without significant tissue damage and is maintained by changes in the CNS (Sluka et al., 2001; Skyba et al., 2002).
Activation of the cAMP pathway in the spinal cord is implicated in pain processing. Mechanical hyperalgesia is produced by spinal activation of the cAMP pathway (Sluka, 1997, 2002; Dolan and Nolan, 2001). Spinal activation of adenylate cyclase increases responses of spinothalamic tract neurons to pinch but not brushing, which is prevented by pretreatment with a protein kinase A (PKA) inhibitor (Lin et al., 2002). Mice lacking adenylate cyclases 1 and 8 have no changes in acute pain but have decreases in behavioral responses after administration of formalin or complete Freund's adjuvant (Wei et al., 2002). Additionally, mice that carry a null mutation for the type I regulatory subunit (Riβ) of PKA demonstrate a significant reduction in capsaicin-evoked plasma extravasation and nocifensive behaviors in the second phase of the formalin test (Malmberg et al., 1997a). Blocking of adenylate cyclase or PKA prevents the mechanical hyperalgesia and allodynia produced by intradermal, intramuscular, or intraarticular injection of capsaicin (Sluka and Willis, 1997; Sluka, 2002).
Once cAMP activates PKA, the catalytic subunit of PKA translocates to the nucleus and phosphorylates cAMP response element-binding protein (CREB), a transcription factor, at Ser 133 (Gonzalez and Montminy, 1989). An increase in phosphorylated CREB (p-CREB) occurs after carrageenan paw inflammation (Messersmith et al., 1998), subcutaneous formalin (Ji and Rupp, 1997; Anderson and Seybold, 2000; Wei et al., 2002), and neuropathic pain (Ma and Quirion, 2001; Miletic et al., 2002). Phosphorylated CREB corresponds to the time frame of hyperalgesia in neuropathic (Miletic et al., 2002) and inflammatory pain (Ji and Rupp, 1997). Furthermore, the amount of CREB that is phosphorylated appears to be stimulus dependent. Increasing the volume of formalin injected into the rat hindpaw results in an increase in phosphorylated CREB (Ji and Rupp, 1997).
Earlier research has demonstrated the importance of the cAMP pathway in mechanical hyperalgesia and in phosphorylating the transcription factor CREB. Using a chronic muscle pain model that mimics clinically relevant chronic muscle pain such as fibromyalgia, we tested the hypothesis that inhibition of the cAMP pathway by blocking either adenylate cyclase or PKA will result in a decrease in mechanical hyperalgesia. Furthermore, we hypothesized that p-CREB will increase in the early, but not late, maintenance phase of chronic muscle hyperalgesia.
Portions of these data have been published previously (Hoeger and Sluka, 2001, 2002).
Materials and Methods
The following experiments were approved by the University of Iowa's Animal Care and Use Committee and followed the policies issued by the National Institutes of Health and the International Association for the Study of Pain on the use of laboratory animals. Male Sprague Dawley rats were used in this study (250–350 gm; Harlan, St. Louis, MO; n = 147).
Chronic muscle hyperalgesia model. Two injections of pH 4.0 sterile saline (100 μl for each injection) were administered 5 d apart into the left gastrocnemius while the animals were anesthetized with vaporized halothane (2–4%). This model produces long-lasting bilateral mechanical hyperalgesia without significant muscle tissue damage or motor deficits (Sluka et al., 2001). In some animals, two intramuscular injections of pH 7.2 (100 μl) sterile saline were used for controls.
Intrathecal injections. An intrathecal catheter was used to administer drugs to the lumbar spinal cord to inhibit the cAMP pathway (Sakura et al., 1996; Storkson et al., 1996; Pogatski et al., 2000). Rats were anesthetized with vaporized halothane (2–4%). A vertical incision was made in the skin at the L5–L6 vertebral level. A 10 cm 32 ga polyurethane catheter (Recathco, Allison Park, PA) was placed between the L5 and L6 vertebrae and advanced intrathecally to the L5/L6 level. Movement of the tail or hindlimb verified placement. Next, an 8 cm polyethylene 10 catheter was fixed to the remaining portion of the 32 ga catheter and exited through the skin. Rats were allowed to recover 5–7 d before the first injection of pH 4.0 saline, which initiated the chronic muscle hyperalgesia. At the end of the experiments, the catheter placement was verified by injecting lidocaine and methylene blue dye. The catheter was considered appropriately placed if there was (1) loss of pinprick and motor paralysis to lidocaine and (2) dye covering L4–L6 spinal levels.
Behavioral testing. Mechanical withdrawal threshold was used as a measure of hyperalgesia to study the effects of spinal blockade of the cAMP pathway. Von Frey filaments (North Coast Medical, Morgan Hill, CA; 1–350 mN bending force) were used to measure the mechanical withdrawal threshold. We measured hyperalgesia outside the intramuscular acid injection in the skin. This is interpreted as secondary hyperalgesia and thought to reflect changes in the CNS (Willis and Coggeshall, 1991). Rats were placed in clear plastic cubicles on an elevated wire mesh and allowed to acclimate for 20–30 min. The filaments were applied to the paw in ascending order starting with the lowest bending force. Two trials per filament were performed. The paw had to lift for two sequential filaments for the force to be recorded. Behavioral tests were performed in a blinded manner in some animals. This type of behavior testing has been demonstrated to be a reliable measurement of mechanical withdrawal threshold (Gopalkrishnan and Sluka, 2001).
Drugs. Two drugs were used to inhibit the cAMP pathway at separate locations along the pathway. SQ 22536 (Biomol, Plymouth Meeting, PA), used to block adenylate cyclase, was dissolved in 16% DMSO. The following doses were tested: 0.064, 0.2, and 0.715 μmol. DMSO (16%) was used as its control during behavioral testing. Myristoylated protein kinase inhibitor (PKI) (14–22) amide (Biomol) dissolved in saline and deionized water was used to inhibit PKA. This drug was tested at 2, 20, 60, and 100 nmol doses. Saline was used as its control. We chose to use these drugs because of their specificity. For example, PKI peptides are extremely specific and potent inhibitors of the PKA catalytic subunit (Walsh and Glass, 1991). Specifically, myristoylated PKI (14–22) amide is an effective inhibitor of PKA (Harris et al., 1997) and blocks hyperalgesia produced by spinal administration of 8-bromo-cAMP (Sluka, 2002).
Immunohistochemical labeling. Standard immunohistochemical labeling was used to assess the location of cells in which CREB had been phosphorylated after induction of hyperalgesia (Sluka and Westlund, 1993; Messersmith et al., 1998). Rats were anesthetized by sodium pentobarbital (100 mg/kg, i.p.) and perfused through the left ventricle with 100 ml of heparinized saline followed by 1 l of 4% paraformaldehyde in 0.1 m phosphate buffer (PB), pH 7.4, 4°C. Segments L5 and T10 of the spinal cord were removed and placed in 30% sucrose solution overnight.
Tissue was cut on a cryostat at 40 μm thickness and placed in PB. These sections underwent a step-wise procedure that included 0.5% H2O2, 1% Na-borohydride, solution A (avidin), solution B (biotin), and 3% normal goat serum (NGS). Between each step, tissue was rinsed in PBS. Next, sections were incubated overnight in primary antibody in 1% NGS/PBS containing 0.75% Triton X-100 at room temperature as follows: anti-CREB (1:10,000) (Upstate Biotechnology, Lake Placid, NY) and anti-p-CREB (1:5000) (Upstate Biotechnology). The anti-p-CREB recognizes phosphorylation at the PKA site, Ser 133 (Gonzalez and Montminy, 1989). Preliminary dilution series for CREB and p-CREB determined appropriate concentrations for the 1° antibody.
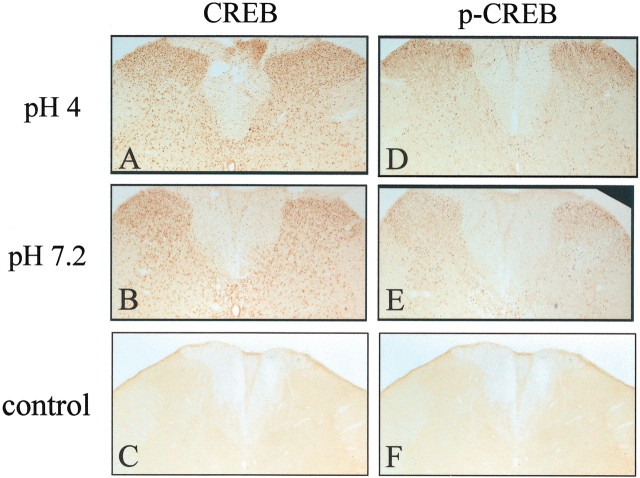
Preabsorption controls for anti-CREB and anti-p-CREB were performed, indicating their specificity to the protein using the exact immunohistochemical protocol except for the addition of the immunizing peptide. Specifically, CREB immunizing peptide (16.7 μg/1 ml) (Upstate Biotechnology) was added to CREB primary antibody, and p-CREB immunizing peptide (16.7 μg/1 ml) (Upstate Biotechnology) was added to p-CREB primary antibody. After 30 min at room temperature, the appropriate solution was added to tissue sections. Simultaneously, another sample of tissue was immunohistochemically stained with antibodies to CREB and p-CREB to ensure that changes were caused by the addition of the immunizing peptide. No staining was observed in tissue sections incubated with the immunizing peptide (see Fig. 3C,F).
Figure 3.
Immunohistochemistry of CREB and p-CREB 24 hr after the second intramuscular injection of pH 4.0, pH 7.2 control, or the preabsorption control. Both CREB and p-CREB densities increase 24 hr after the second intramuscular injection of pH 4.0 (A, D) compared with the animals that received pH 7.2 saline intramuscular injections (B, E). No staining was observed in tissue sections incubated with the immunizing peptide (C, F).
Sections were washed in PBS and 3% NGS followed by incubation for 1 hr in the secondary antibody biotinylated-goat anti-rabbit IgG (Vector Laboratories, Burlingame, CA; 1:1000). After they were washed in PBS and 3% NGS, sections were incubated in avidin horseradish peroxidase (1% NGS with 0.75% Triton X-100; 1:1000) for 1 hr. This was followed by 6 min in 0.05% diaminobenzidine (DAB) and 0.01% H2O2. Sections were washed and mounted on slides, allowed to dry for 24–48 hr before cleaning in ethanol/water and propar, and coverslipped.
To minimize differences in staining between animals, each group was run simultaneously, i.e., pH 4.0 and pH 7.2 or saline and SQ 22536. Furthermore, between groups the exact staining protocol was used, including incubation in primary and secondary antibodies and DAB.
Density readings. Three spinal cord sections (L5) were randomly chosen from each rat. All density readings were performed with the experimenter blinded to group except for initial preliminary data. Images were generated using an Olympus BX-51 microscope. The superficial laminas (I–II) and intermediate and deep dorsal horn (III–VI) were outlined, and the number of pixels occupied by immunoreactive cells was measured using Image J 1.24 software (NIH) (Le Guen et al., 1998; Martin et al., 1999; Wei et al., 2002). Specifically, each tissue section was first converted to eight-bit gray scale, and then each tissue section was calibrated independently using the “uncalibrated OD” function with pixel values ranging from 0 to 255. The density values represent pixels per area. A background reading taken from the white matter of the dorsal column was subtracted from the density reading taken from the gray matter of the same tissue section. This controls for differences in nonspecific staining as a result of the DAB reaction.
Motor testing. The PKA inhibitor produced a significant motor deficit in one rat during the behavioral testing protocol. Therefore, the following experimental drugs were tested for motor effects: 100 nmol PKI (n = 9), 60 nmol PKI (n = 5), and 0.715 μmol SQ22536 (n = 4) against a saline control (n = 4) using the Rota-Rod treadmill test and placing reflex. The rats were trained on the treadmill (Ugo Basile Rota-Rod, Stoelting, Wood Dale, IL) for 2 d before the testing, which included three sessions of three intervals given daily with an intersession interval >2hrand an intertrial interval >5 min. Motor involvement was tested after intrathecal drug administration in 15 min intervals for the first hour followed by 30 min intervals for the second hour.
Experimental design. The following drugs were used to determine the effects of inhibiting the cAMP pathway on chronic muscle hyperalgesia: (1) adenylate cyclase inhibitor, SQ 22536, and (2) PKA inhibitor, myristoylated PKI (14–22) amide. The adenylate cyclase inhibitor was dissolved in 16% DMSO, and behavioral testing was performed at the doses of 0.064, 0.2, and 0.715 μmol. The PKA inhibitor was dissolved in saline and deionized water, and behavioral testing was performed at the doses of 2, 20, 60, and 100 nmol. We demonstrated previously that 60 nmol of the PKA inhibitor (PKI) reverses the mechanical hyperalgesia produced by spinal administration of 8-bromo-cAMP (Sluka, 2002). The drug dose quantities were determined by preliminary data, previously published data (Sluka 1997), and the dissociation constant for binding of inhibitor to enzyme (reported by the manufacturer, Biomol).
In the first series of experiments, intrathecal catheters were placed in male Sprague Dawley rats. After 5–7 d, the chronic muscle hyperalgesia model was induced, which involves administering two intramuscular injections of pH 4.0 saline 5 d apart. Twenty-four hours after the second intramuscular injection of pH 4.0 saline, the following drugs were administered intrathecally: (1) adenylate cyclase inhibitor, SQ 22536 (0.715 μl dose, n = 9; 0.215 μl dose, n = 5; 0.064 μl dose, n = 4), (2) PKA inhibitor, myristoylated PKI (14–22) amide (100 nmol, n = 9; 60 nmol, n = 9; 20 nmol, n = 7; 2 nmol, n = 4), (3) saline control, pH 7.2 (n = 9), or (4) 16% DMSO in saline control (n = 8). Similarly, 1 week after the second injection, the following drugs were tested: (1) adenylate cyclase inhibitor, SQ 22536 (0.715 μl dose; n = 6), (2) PKA inhibitor, myristoylated PKI (14–22) amide (60 nmol; n = 8), (3) saline control, pH 7.2 (n = 6), (4) 16% DMSO in saline control (n = 6). Mechanical withdrawal threshold was measured (1) before each intramuscular injection of pH 4.0 saline, (2) 24 hr or 1 week after the second intramuscular injection of pH 4.0 saline (before drug administration), and (3) after drug administration in 15 min intervals for the first hour followed by 30 min intervals for the second hour.
In the second series of experiments, two injections of pH 4.0 or 7.2 were given into the left gastrocnemius 5 d apart. Twenty-four hours and 1 week after the second injection, immunohistochemistry was performed for CREB and p-CREB. Spinal cord sections from animals injected intramuscularly with pH 4.0 or 7.2 were immunostained simultaneously.
In another group of animals, 24 hr after the second injection into the left gastrocnemius, the adenylate cyclase inhibitor or vehicle control was injected intrathecally. Once the decreased mechanical withdrawal threshold reached its maximum reversal, ∼60 min after intrathecal injection, rats were perfused and immunohistochemistry was performed to identify CREB and p-CREB.
Statistical analysis. The results from the behavior testing using the von Frey filaments were not normally distributed; therefore, a nonparametric Kruskal–Wallis ANOVA was used. If differences were present, a post hoc Wilcoxon signed ranks test was used. Statistical analysis for the density readings was done using a one-way ANOVA. A Pearson product-moment coefficient of correlation was used to determine the relationship between mechanical withdrawal threshold and the density of the p-CREB immunoreactivity 24 hr after the second injection of acidic saline. Statistical significance was determined by p < 0.05.
Results
Behavioral effects of repeated intramuscular acid injections
The animals demonstrated the same pattern of hyperalgesia, as reported previously (Sluka et al., 2001). Specifically, there was a bilateral decrease in mechanical withdrawal threshold 24 hr and 1 week after the second intramuscular injection of acidic saline. The mechanical withdrawal thresholds did not change significantly after spinal administration of saline (PKI control) or 16% DMSO (SQ 22536 control).
Inhibition of adenylate cyclase
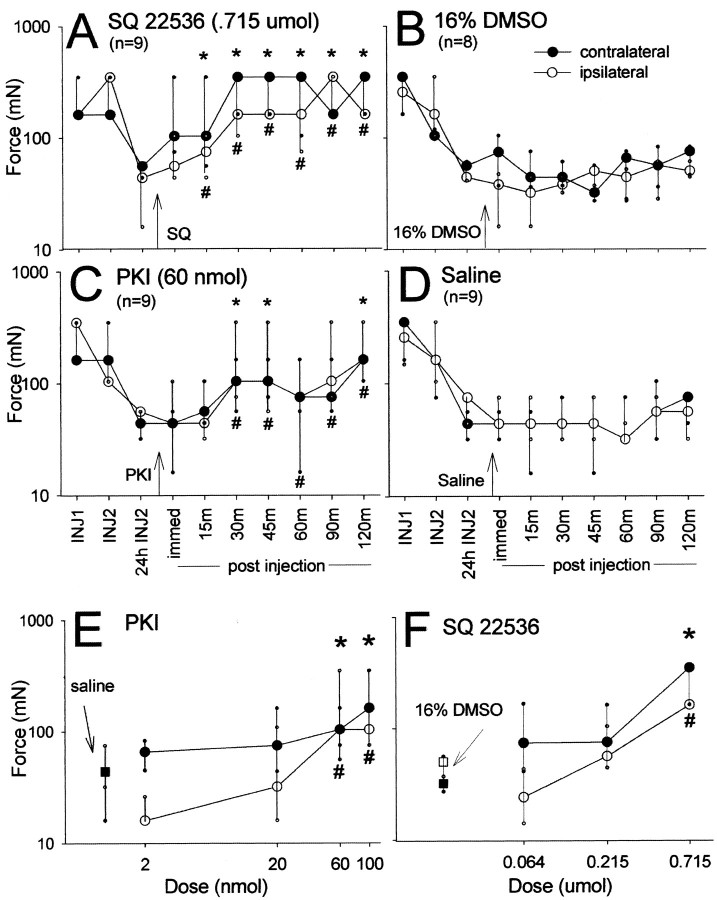
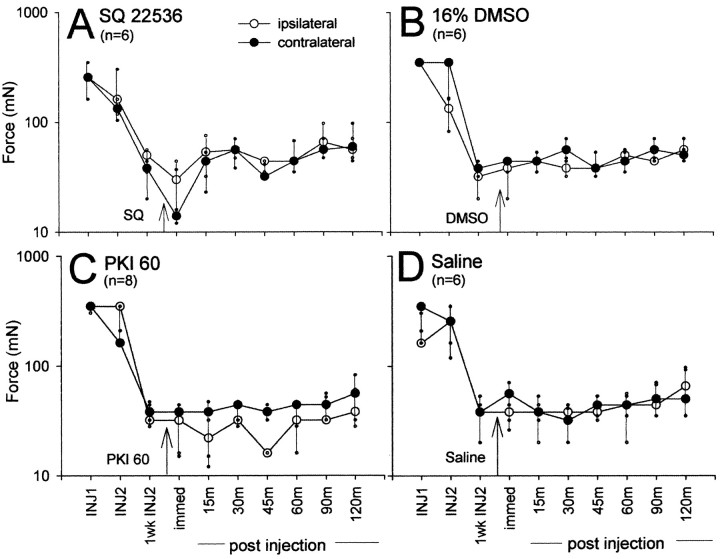
The adenylate cyclase inhibitor, SQ 22536, was injected intrathecally 24 hr or 1 week after the second intramuscular injection of pH 4.0 saline to assess the role of the cAMP pathway in the early and late maintenance phases of chronic muscle hyperalgesia. The highest dose of SQ 22536 administered 24 hr after the second intramuscular injection of pH 4.0 saline increased the mechanical withdrawal threshold bilaterally (Fig. 1A). Significant increases from 16% DMSO control occurred 15 min (p = 0.008), 30 min (p = 0.0001), 45 min (p = 0.0001), 60 min (p = 0.002), 90 min (p = 0.0001), and 120 min (p = 0.001) ipsilaterally, and 15 min (p = 0.027), 30 min (p = 0.006), 45 min (p = 0.008), 60 min (p = 0.006), 90 min (p = 0.001), and 120 min (p = 0.008) contralaterally. The effects of SQ 22536 are dose dependent 45 min after drug administration, with the highest dose showing almost complete reversal of hyperalgesia contralaterally and ipsilaterally (Fig. 1E). The mechanical withdrawal threshold remained unchanged if the SQ 22536 was administered 1 week after the second intramuscular injection of pH 4.0 saline (Fig. 2A).
Figure 1.
Line graphs showing the effects of the inhibitors of the cAMP pathway on the median mechanical withdrawal threshold 24 hr after the second intramuscular acid injection. A, B, Intrathecal administration of SQ22536 (0.7 μmol) increases mechanical withdrawal threshold bilaterally compared with the 16% DMSO control 15 min after drug administration and remains increased for the next 2 hr. C, D, Intrathecal administration of PKI (60 nmol) increases mechanical withdrawal threshold bilaterally compared with the intrathecal saline control. E, F, A dose-dependent increase in mechanical withdrawal threshold is observed 45 min after spinal blockade of adenylate cyclase (SQ22536) or protein kinase A (PKI). INJ1, Before first intramuscular injection of pH 4.0 saline; INJ2, before second intramuscular injection of pH 4.0 saline; 24 hr INJ2, 24 hr after second intramuscular injection of pH 4.0 saline. Data are presented as the median with the 25th and 75th percentiles. *Contralaterally and #ipsilaterally significantly different from vehicle control; p ≤ 0.05.
Figure 2.
Line graphs showing the effects of the inhibitors of the cAMP pathway on the median mechanical withdrawal threshold 1 week after the second intramuscular acid injection. A, B, Intrathecal administration of SQ22536 (0.7 μmol) has no effect on mechanical withdrawal threshold compared with the 16% DMSO control. C, D, Intrathecal administration of PKI (60 nmol) has no effect on mechanical withdrawal threshold compared with the intrathecal saline control. INJ1, Before first intramuscular injection of pH 4.0 saline; INJ2, before second intramuscular injection of pH 4.0 saline; 1wk INJ2, 1 week after second intramuscular injection of pH 4.0 saline. Data are presented as the median with the 25th and 75th percentiles.
Inhibition of protein kinase A
To further demonstrate the role of the cAMP pathway in chronic muscle hyperalgesia, a PKA inhibitor was injected intrathecally 24 hr and 1 week after the second intramuscular injection of pH 4.0 saline. The highest dose of PKI (100 nmol) administered 24 hr after the second intramuscular injection of pH 4.0 saline increased the mechanical withdrawal threshold bilaterally. Significant increases from intrathecally administered saline control occurred 45 min (p = 0.031), 60 min (p = 0.0001), 90 min (p = 0.0001), and 120 min (p = 0.014) ipsilaterally, and 30 min (p = 0.014), 45 min (p = 0.006), 90 min (p = 0.05), and 120 min (p = 0.003) contralaterally. The second highest dose of PKI (60 nmol) administered 24 hr after the second intramuscular injection of pH 4.0 saline also increased the mechanical withdrawal threshold bilaterally (Fig. 1C). Significant increases from saline control occurred 30 min (p = 0.024), 45 min (p = 0.05), 60 min (p = 0.008), 90 min (p = 0.04), and 120 min (p = 0.002) ipsilaterally, and 30 min (p = 0.04), 45 min (p = 0.014), and 120 min (p = 0.006) contralaterally. The effects of PKI are dose dependent, with the two highest doses showing reversal of hyperalgesia ipsilaterally and contralaterally (Fig. 1F). The mechanical withdrawal threshold remained unchanged if the PKI was administered 1 week after the second intramuscular injection of pH 4.0 saline (Fig. 2C).
Motor involvement
The potential effects that the drugs may have on motor impairment were tested for both the SQ 22536 (0.7 mol dose) and PKI (60 and 100 nmol doses) using the Rota-Rod treadmill test. Both drugs were injected intrathecally and monitored for motor function for the same 2 hr duration as the experimental protocol. Rats injected with SQ22536 had no motor involvement compared with the rats injected intrathecally with saline; all of the rats were able to stay on the treadmill for the maximum 150 sec. After intrathecal injection of PKI, three of the nine rats given the 100 nmol dose were excluded from the treadmill testing because of paralysis. Rats injected with PKI that were not paralyzed (n = 6) showed no significant difference when compared with the saline control. In previous behavioral experiments, this exclusive procedure was also used. For instance, if any of the rats appeared to have severe motor involvement after intrathecal administration of the drug, the rats were automatically excluded from behavioral testing.
CREB
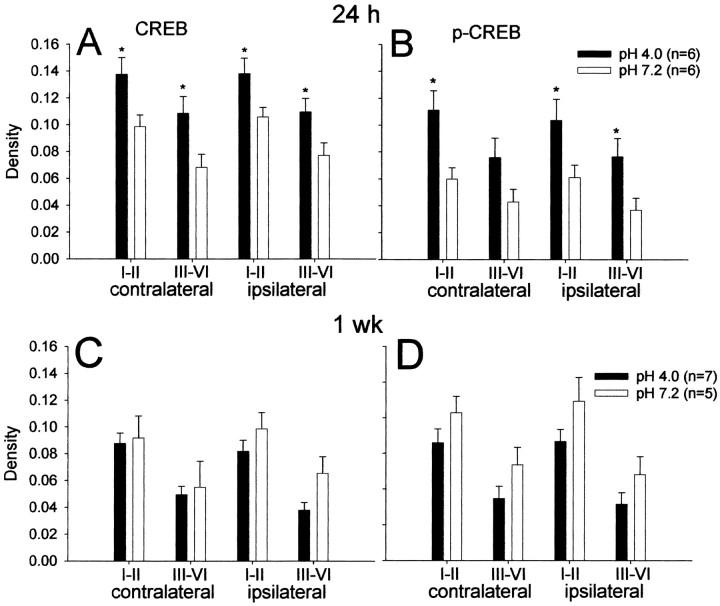
There was a bilateral increase in CREB and p-CREB immunoreactivity in L5 24 hr after intramuscular injection of pH 4.0 saline when compared with pH 7.2 (Fig. 3). CREB immunoreactivity increased ipsilaterally in the superficial (p = 0.038) and deep (p = 0.039) dorsal horn and contralaterally in the superficial (p = 0.03) and deep (p = 0.029) dorsal horn when compared with rats receiving pH 7.2 intramuscular injections (Fig. 4A,B). p-CREB significantly increases ipsilaterally in the superficial (p = 0.04) and deep (p = 0.034) dorsal horn and contralaterally in the superficial dorsal horn (p = 0.012) when compared with rats receiving pH 7.2 intramuscular injections (Fig. 4A,B). CREB and p-CREB immunoreactivity 1 week after the second intramuscular acid injection were not significantly different from the pH 7.2 controls (Fig. 4C,D).
Figure 4.
Density of CREB and p-CREB immunoreactivity 24 hr and 1 week after the second intramuscular injection of pH 4.0 or 7.2 control. A, B, CREB and p-CREB increase in the spinal cord dorsal horn bilaterally 24 hr after the second intramuscular injection of pH 4.0 saline compared with pH 7.2 saline injections. C, D, CREB and p-CREB do not significantly differ 1 week after the second intramuscular injection of pH 4.0 compared with pH 7.2 saline injections. I–II, Laminas I and II; III–VI, laminas III–VI; 24 h, 24 hr; 1 wk, 1 week. Data are presented as the average with the SEM. *p, significantly different from the pH 7.2 control group; p < 0.05.
These changes in immunoreactivity for CREB and p-CREB did not occur in tissue sections from T10. CREB immunoreactivity in rats receiving pH 4.0 intramuscular injections did not significantly differ in the ipsilateral superficial (p = 0.41) and deep (p = 0.29) dorsal horn and contralaterally in the superficial (p = 0.37) and deep (p = 0.38) dorsal horn when compared with rats receiving pH 7.2 intramuscular injections. p-CREB immunoreactivity in rats receiving pH 4.0 intramuscular injections did not differ significantly in the ipsilateral superficial (p = 0.47) and deep (p = 0.43) dorsal horn, and contralaterally in the superficial (p = 0.31) and deep (p = 0.38) dorsal horn when compared with rats receiving pH 7.2 intramuscular injections.
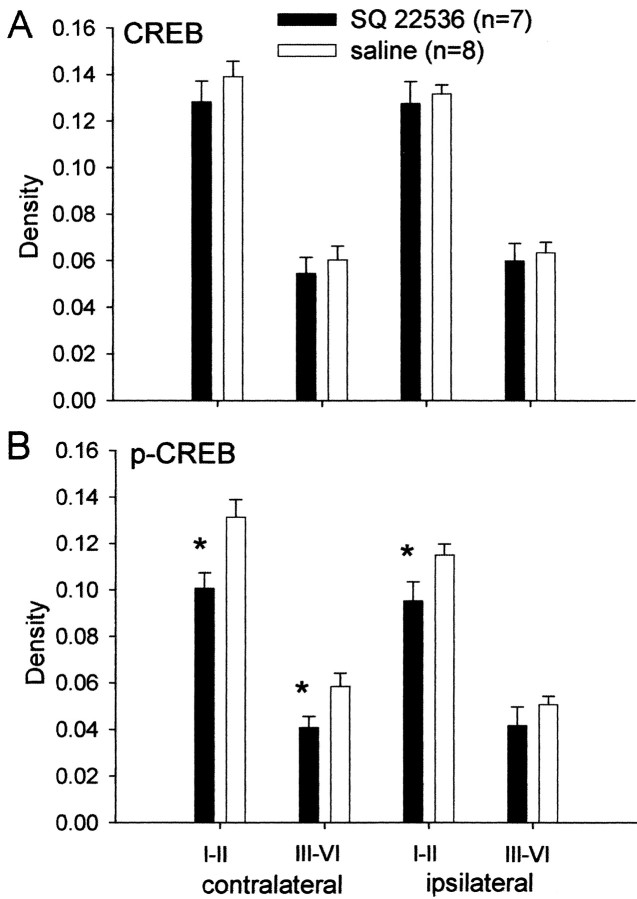
To test whether the increase in p-CREB is mediated by activation of the cAMP pathway, we administered SQ 22536 intrathecally and measured CREB and p-CREB. Spinal administration of SQ 22536 significantly prevented the increase in p-CREB contralaterally in the superficial (p = 0.016) and deep (p = 0.048) dorsal horn and ipsilaterally in the superficial dorsal horn (p = 0.046) compared with the intrathecal saline control (Fig. 5). Spinal application of SQ 22536 had no effect on CREB (Fig. 5). Thus, it appears that the increase in p-CREB is mediated by activation of the cAMP pathway.
Figure 5.
Density of CREB and p-CREB immunoreactivity after intrathecal treatment with the adenylate cyclase inhibitor, SQ 22536, or saline. A, CREB remains unchanged after spinal inhibition of adenylate cyclase with SQ22536. B, The increase in p-CREB is prevented after spinal inhibition of adenylate cyclase with SQ22536 compared with the intrathecal saline control. I–II, Laminas I and II; III–VI, laminas III–VI. Data are presented as the average with the SEM. *Significantly different from saline control; p ≤ 0.05.
To test for the relationship between p-CREB and mechanical hyperalgesia, the density of staining was tested for correlation with the mechanical withdrawal thresholds. The density of p-CREB immunoreactivity in the superficial dorsal horn 24 hr after the second intramuscular injection of pH 4.0 or 7.2 or after the administration of SQ 22536 or saline is significantly correlated to the mechanical withdrawal threshold on the ipsilateral and contralateral paws (Fig. 6). There was no correlation between the mechanical withdrawal threshold with density readings from the deep dorsal horn. Thus, a decrease in mechanical withdrawal threshold, indicating an increase in mechanical hyperalgesia, is associated with an increase in p-CREB immunoreactivity in the superficial dorsal horn.
Figure 6.
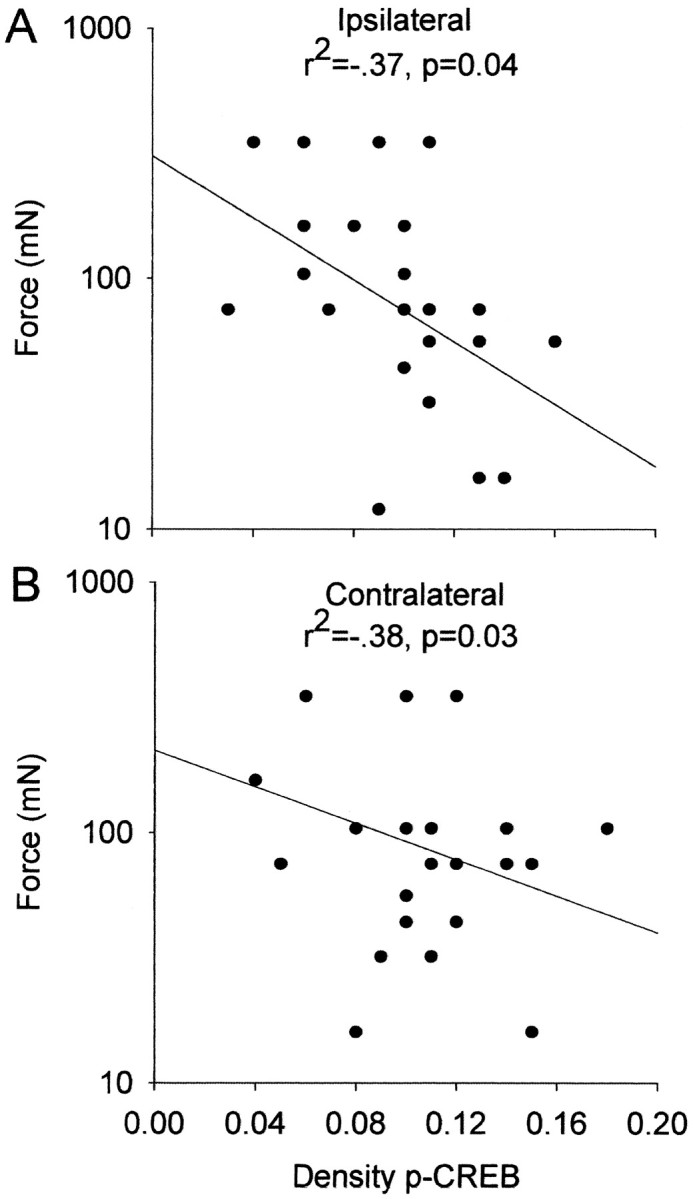
Scatter plots showing the correlation of the mechanical withdrawal threshold ipsilaterally (A) and contralaterally (B) to the density of p-CREB immunoreactivity of the superficial dorsal horn at 24 hr. All animals at 24 hr were included in the analysis, and each point represents an individual animal.
Discussion
This study shows a time-dependent increase in CREB and the phosphorylation of CREB with significant increases 24 hr, but not 1 week, after the second intramuscular acid injection. The increase in p-CREB depends on activation of the cAMP pathway because blockade of adenylate cyclase or PKA prevents this increase at 24 hr. Changes in p-CREB parallel the cAMP-mediated mechanical hyperalgesia. Furthermore, the staining density for p-CREB in the superficial dorsal horn correlates with the mechanical withdrawal threshold 24 hr after the second intramuscular injection, indicating that behavioral changes at 24 hr are associated with increases in p-CREB. A number of intracellular messengers can phosphorylate CREB at Ser 133, i.e., calcium calmodulin kinase IV, mitogen-activated kinase, extracellular regulated kinase, and PKA (Lonze and Ginty, 2002). This study demonstrates that increases in p-CREB after repeated intramuscular acid injections are reversed by blockade of the cAMP pathway. Thus, these data suggest that phosphorylation of CREB in muscle-induced hyperalgesia is mediated by activation of the cAMP pathway and is time dependent.
Activation of the cAMP pathway and CREB
CREB is a nuclear protein that mediates the effects of the activation of the cAMP pathway in the transcriptional regulation of a large number of peptides and proteins. Specifically, CREB is a transcription factor that binds to the cAMP response element (CRE) promoter site (Shaywitz and Greenberg, 1999). Two types of CREB that have opposing actions may bind to the CRE promoter site. CREB-1 represses gene transcription and CREB-2 activates gene transcription, but only when it is phosphorylated (Bear et al., 2001).
The current study shows that activation of the cAMP pathway phosphorylates CREB, which is required for CRE-mediated transcription, suggesting that an increase in gene transcription may occur in chronic muscle pain. These data agree with previous studies showing an increase in p-CREB in various animal models of pain (Ji and Rupp, 1997; Messersmith et al., 1998; Anderson and Seybold, 2000; Ji et al., 2000; Ma and Quirion, 2001; Miletic et al., 2002; Wei et al., 2002). In both neuropathic and inflammatory pain, the phosphorylation of CREB parallels hyperalgesia (Ji and Rupp, 1997; Miletic et al., 2002). In contrast, our study shows that the increases in p-CREB occur in a time-dependent manner, are mediated by activation of the cAMP pathway, and that the increases in the superficial dorsal horn correlate with the mechanical withdrawal threshold. Thus, the increases in p-CREB after muscle-induced hyperalgesia appear to contribute to the cAMP-dependent phase of hyperalgesia associated with stimulation of the muscle.
In the current study there was not an area-dependent increase in CREB or p-CREB immunoreactivity as observed after electrical stimulation of the sciatic nerve (White and Helme, 1985; Klein et al., 1990). The changes in CREB and p-CREB are difficult to explain on the basis of activation solely by muscle nociceptors. Types III and IV muscle nociceptors project predominantly to laminas I and V without projections to lamina II (for review, see Mense, 1993; Mense and Prabhakar, 1986). Similarly, changes in substance P and calcitonin gene-related peptide after knee joint inflammation occur throughout laminas I and II (Sluka et al., 1992; Sluka and Westlund, 1993), although joint afferents terminate primarily in laminas I and V (Craig et al., 1988). One speculation is that increases in CREB and p-CREB are mediated by activation of descending facilitatory pathways secondary to activation of muscle nociceptors (Urban et al., 1999; Porreca et al., 2002). Secondary hyperalgesia involves descending facilitation mediated by supraspinal sites, including the rostral ventral medulla and the anterior cingulate cortex (Urban et al., 1999; Calejesan et al., 2000; Porreca et al., 2002; Wei et al., 2002). Activation of supraspinal sites may also explain the bilateral changes in CREB, p-CREB, and mechanical hyperalgesia.
The phosphorylation of CREB may contribute to the longlasting hyperalgesia observed in this model by allowing CREB to bind the CRE promoter. The CRE promoter is found in a number of “pain genes,” including c-fos (Sassone-Corsi et al., 1988), somatostatin (Gonzales and Montminy, 1989), and neurokinin 1 receptor (Hershey et al., 1991), that are modulated after tissue injury. Furthermore, p-CREB increases in neurokinin-1 receptor immunoreactive neurons in the spinal cord after formalin injection (Anderson and Seybold, 2000). There may be other CRE promoter sites downstream in which consequences of p-CREB are not identified.
The increase in CREB could represent an upregulation attributable to new synthesis or decreased degradation. Increases in CREB protein, which occur in other systems, would result in a greater pool of protein for phosphorylation. For example, CREB mRNA and CREB immunoreactivity increase in the rat hippocampus after chronic administration of antidepressants as well as phosphodiesterase inhibitors (Nibuya et al., 1996). Therefore, this increase in CREB parallels what is seen in other systems that are mediated by the cAMP pathway.
Activation of the cAMP pathway and mechanical hyperalgesia
Activation of adenylate cyclase in neurons can occur through neurotransmitter–receptor interactions or increases in calcium (Xia and Storm, 1997). Adenylate cyclase activation converts ATP to cAMP, which then activates PKA. PKA is involved in neuroplasticity through phosphorylation of various substrates, including ion channels, neurotransmitter receptors, and transcription factors (Gonzalez and Montminy, 1989; Blackstone et al., 1995; Hell et al., 1995). The PKA site on the NR1 subunit of the NMDA receptor is phosphorylated after intradermal capsaicin injection (Zou et al., 2000). The current study provides evidence that PKA can also phosphorylate the transcription factor CREB in an animal model of muscle-induced hyperalgesia.
We propose that activation of the cAMP pathway is involved in a time-dependent manner in the early phase of maintenance but not the later phase or the induction of hyperalgesia. Pretreatment with either an adenylate cyclase or PKA inhibitor has no effect on the hyperalgesia produced by intradermal injection of capsaicin (Sluka, 1997). Treatment with an adenylate cyclase or PKA inhibitor 1 hr after intradermal capsaicin or 24 hr after intra-articular or intramuscular injection of capsaicin reverses secondary mechanical hyperalgesia (Sluka, 1997, 2002). In contrast, treatment 1 week after intra-articular or intramuscular injection of capsaicin has no effect on mechanical hyperalgesia (Sluka, 2002). Our results parallel these previous behavioral studies with a reduction in hyperalgesia at 24 hr, but not 1 week, after repeated intramuscular acid injection. We further these results by showing that phosphorylation of CREB occurs in the same time-dependent manner and that the changes in p-CREB at 24 hr correlate with the mechanical withdrawal threshold, suggesting a role for phosphorylation of CREB in the early maintenance of mechanical hyperalgesia induced by intramuscular acid injection.
The temporal effects of the cAMP pathway activation are seen in other models of neuroplasticity. The early phase of long-term potentiation is dependent on the activation of the cAMP pathway; inhibition of this pathway decreases early long-term potentiation (Blitzer et al., 1995; Otmakhova et al., 2000). In the hippocampus, PKA activity rapidly increases in the initial stages of spatial learning and starts to decrease when protein kinase C (PKC) activity is maximal at later stages (Vazquez et al., 2000). PKC activation appears to be critical in the later maintenance phase of long-term potentiation and memory (Sweatt, 1999; Vazquez et al., 2000). Thus, the mechanical hyperalgesia observed in the current study and a previous report (Sluka, 2002) show a pattern similar to that observed in long-term potentiation and memory. On the basis of these data, we hypothesize that PKC plays a role in the later phase. In support of this hypothesis, mice lacking PKC γ have no differences in their acute pain responses to heat or mechanical stimuli (Malmberg et al., 1997b). In a more chronic neuropathic pain model, however, both mechanical and heat hyperalgesia were markedly reduced (Malmberg et al., 1997b). Therefore, these results indicate that PKA is involved in the early maintenance of chronic muscle hyperalgesia, but some other molecule, such as PKC, is involved in the later maintenance of hyperalgesia.
The cAMP pathway within the spinal cord plays an integral role in nociceptive processing. Specifically, activation of the cAMP pathway potentiates dorsal horn neurons in vitro (Cerne et al., 1992, 1993), sensitizes spinothalamic tract cells to noxious mechanical stimuli in vivo (Lin et al., 2002), and produces mechanical hyperalgesia in vivo (Sluka, 1997, 2002; Dolan and Nolan, 2001). Conversely, spinal blockade of the cAMP pathway reverses mechanical hyperalgesia produced by intradermal, intramuscular, or intra-articular injection of capsaicin (Sluka, 1997, 2002) and reverses capsaicin-induced sensitization of spinothalamic tract cells (Sluka et al., 1997). Thus, activation of the cAMP pathway in the dorsal horn, including spinothalamic tract neurons, could sensitize dorsal horn neurons to noxious mechanical input resulting in mechanical hyperalgesia, as observed after repeated intramuscular acid injection.
Summary
In summary, the phosphorylation of CREB occurs in a time-dependent manner that parallels the cAMP-dependent phase of mechanical hyperalgesia. These increases in p-CREB are reversed by blockade of the cAMP pathway and correlate with the mechanical withdrawal threshold, suggesting that increases in p-CREB may contribute to the mechanical hyperalgesia associated with repeated intramuscular acid injection. Thus, these data provide clinical relevance in that modulation of the cAMP pathway may be beneficial in the early stages of muscle hyperalgesia.
Footnotes
This study was supported by National Institutes of Health Grants RO1 NS 39734, KO2 AR 02201, and F31 NS43962.
Correspondence should be addressed to Dr. K. A. Sluka, Graduate Program in Physical Therapy and Rehabilitation Science, 1-214 Medical Education Building, University of Iowa, Iowa City, IA 52242. E-mail: ksluka@blue.weeg.uiowa.edu.
Copyright © 2003 Society for Neuroscience 0270-6474/03/235437-09$15.00/0
References
- Anderson LE, Seybold VS ( 2000) Phosphorylated cAMP response element binding protein increases in neurokinin-1 receptor immunoreactive neurons in rat spinal cord in response to formalin-induced nociception. Neurosci Lett 283: 29–32. [DOI] [PubMed] [Google Scholar]
- Bear MF, Connors BW, Paradiso MA ( 2001) Neuroscience: exploring the brain. In: Molecular mechanisms of learning and memory (Katz S, ed), pp 804–805. Baltimore: Lippincott Williams & Wilkins.
- Bennett RM ( 1995) Fibromyalgia: the commonest cause of widespread pain. Compr Ther 21: 269–275. [PubMed] [Google Scholar]
- Blackstone C, Murphy TH, Moss SJ, Baraban JM, Huganir RL ( 1995) cAMP and synaptic activity-dependent phosphorylation of AMPA-preferring glutamate receptors. J Neurosci 14: 7585–7593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blitzer RD, Wong T, Nouranifar R, Iyengar R, Landau EM ( 1995) Postsynaptic cAMP pathway gates early LTP in hippocampal CA1 region. Neuron 15: 1403–1414. [DOI] [PubMed] [Google Scholar]
- Calejesan AA, Kim SJ, Zhuo M ( 2000) Descending failitatory modulation of a behavioral nociceptive response by stimulation in the adult rat anterior cingulate cortex. Eur J Pain 4: 83–96. [DOI] [PubMed] [Google Scholar]
- Cerne R, Jaing M, Randic M ( 1992) Cyclic adenosine 3′5′-monophosphate potentiates excitatory amino acid and synaptic responses of rat spinal dorsal horn neurons. Brain Res 596: 111–123. [DOI] [PubMed] [Google Scholar]
- Cerne R, Rusin KI, Randic M ( 1993) Enhancement of the N-methyl-d-aspartate response in spinal dorsal horn neurons by cAMP dependent protein kinase. Neurosci Lett 161: 124–128. [DOI] [PubMed] [Google Scholar]
- Craig AD, Heppelmann B, Schaible HG ( 1988) The projection of the medial and posterior articular nerves of the cat's knee to the spinal cord. J Comp Neurol 276: 279–288. [DOI] [PubMed] [Google Scholar]
- Dolan S, Nolan A ( 2001) Biphasic modulation of nociceptive processing by the cyclic AMP-protein kinase A signaling pathway in sheep spinal cord. Neurosci Lett 309: 157–160. [DOI] [PubMed] [Google Scholar]
- Gonzales GA, Montminy MR ( 1989) Cyclic AMP stimulates somatostatin gene transcription by phosophorylation of CREB at serine 133. Cell 59: 675–680. [DOI] [PubMed] [Google Scholar]
- Gopalkrishnan P, Sluka KA ( 2001) Effect of varying frequency, intensity and pulse duration of TENS on primary hyperalgesia in inflamed rats. Arch Phys Med Rehabil 81: 984–990. [DOI] [PubMed] [Google Scholar]
- Harris TE, Persaud SJ, Jones PM ( 1997) Pseudosubstrate inhibition of cyclic AMP-dependent protein kinase in intact pancreatic islets: effects on cyclic AMP-dependent and glucose-dependent insulin secretion. Biochem Biophys Res Commun 232: 648–651. [DOI] [PubMed] [Google Scholar]
- Hell JW, Yokoyama CT, Breeze LJ, Chavkin C, Catterall WA ( 1995) Phosphorylation of presynaptic and postsynaptic calcium channels by cAMP dependent protein kinase in hippocampal neurons. EMBO J 14: 3036–3044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hershey AD, Dykema PE, Krause JE ( 1991) Organization, structure and expression of the gene encoding the rat substance P receptor. J Biol Chem 266: 4366–4374. [PubMed] [Google Scholar]
- Hoeger MK, Sluka KA ( 2001) The role of the cAMP pathway in a chronic muscle pain model in the rat. Soc Neurosci Abstr 27: 891.3. [Google Scholar]
- Hoeger MK, Sluka KA ( 2002) Increased phosphorylation of CREB and reversal by blockade of adenylate cyclase in a chronic muscle pain model in the rat. Paper presented at 10th World Congress on Pain, San Diego, August. Abstract 23-P19.
- Ji RR, Rupp F ( 1997) Phosphorylation of transcription factor CREB in rat spinal cord after formalin-induced hyperalgesia: relationship to c-fos induction. J Neurosci 17: 1776–1785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ji RR, Brenner GJ, Schmoll R, Baba H, Woolf CJ ( 2000) Phosphorylation of ERK and CREB in nociceptive neurons after noxious stimulation. In: Proceedings of 9th World Congress on Pain (Devor M, Rowbotham MC, Wiesenfeld-Hallin Z, eds), Vol 16, pp 191–198. Seattle: IASP. [Google Scholar]
- Klein CM, Coggeshall RE, Carlton SM, Westlund KN, Sorkin LS ( 1990) Changes in calcitonin gene-related peptide immunoreactivity in the rat dorsal horn following electrical stimulation of the sciatic nerve. Neurosci Lett 115: 149–154. [DOI] [PubMed] [Google Scholar]
- Le Guen S, Gwenaelle C, Besson JM ( 1998) Development of tolerance to the antinociceptive effect of systemic morphine at the lumbar spinal cord level: a c-Fos study in the rat. Brain Res 813: 128–138. [DOI] [PubMed] [Google Scholar]
- Lin Q, Wu J, Willis WD ( 2002) Effects of protein kinase A activation on the responses of primate spinothalamic tract neurons to mechanical stimuli. J Neurophysiol 88: 214–221. [DOI] [PubMed] [Google Scholar]
- Lonze BE, Ginty DD ( 2002) Function and regulation of CREB family transcription factors in the nervous system. Neuron 35: 605–623. [DOI] [PubMed] [Google Scholar]
- Ma W, Quirion R ( 2001) Increased phosphorylation of the cyclic AMP response element-binding protein (CREB) in the superficial dorsal horn neurons following partial sciatic nerve ligation. Pain 93: 295–301. [DOI] [PubMed] [Google Scholar]
- Malmberg AB, Brandon EP, Idzerda RL, Liu H, McKnight GS, Basbaum AI ( 1997a) Diminished inflammation and nociceptive pain with preservation of neuropathic pain in mice with a targeted mutation of the type I regulatory subunit of cAMP dependent protein kinase. J Neurosci 17: 7462–7470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malmberg AB, Chen C, Tonegawa S, Basbaum AI ( 1997b) Preserved acute pain and reduced neuropathic pain in mice lacking PKC gamma. Science 278: 279–283. [DOI] [PubMed] [Google Scholar]
- Martin WJ, Liu H, Wang H, Malmberg AB, Basbaum AI ( 1999) Inflammation-induced up-regulation of protein kinase Cγ immunoreactivity in rat spinal cord correlates with enhanced nociceptive processing. Neuroscience 88: 1267–1274. [DOI] [PubMed] [Google Scholar]
- Mense S ( 1993) Nociception from skeletal muscle in relation to clinical muscle pain. Pain 54: 241–289. [DOI] [PubMed] [Google Scholar]
- Mense S, Prabhakar NR ( 1986) Spinal termination of nociceptive afferent fibres from deep tissues in the cat. Neurosci Lett 66: 169–174. [DOI] [PubMed] [Google Scholar]
- Messersmith DJ, Kim DJ, Iadarola MJ ( 1998) Transcription factor regulation of prodynorphin gene expression following rat hindpaw inflammation. Mol Brain Res 53: 259–269. [DOI] [PubMed] [Google Scholar]
- Miletic G, Pankratz MT, Miletic V ( 2002) Increases in the phosphorylation of cyclic AMP response element binding protein (CREB) and decreases in the content of calcineurin accompany thermal hyperalgesia following chronic constriction injury in rats. Pain 99: 493–500. [DOI] [PubMed] [Google Scholar]
- Nibuya M, Nestler EJ, Duman RS ( 1996) Chronic antidepressant administration increases the expression of cAMP response element-binding protein (CREB) in rat hippocampus. J Neurosci 16: 2365–2372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Otmakhova NA, Otmakhov N, Mortenson LH, Lisman JE ( 2000) Inhibition of the cAMP pathway decrease early long-term potentiation at CA1 hippocampal synapses. J Neurosci 20: 4446–4451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pogatski EM, Zahn PK, Brennan TJ ( 2000) Lumbar catheterization of the subarachanoid space with a 32-gauge polyurethane catheter in the rat. Eur J Pain 4: 111–113. [DOI] [PubMed] [Google Scholar]
- Porreca F, Ossipov MH, Gebhart GF ( 2002) Chronic pain and medullary descending facilitation. Trends Neurosci 25: 319–325. [DOI] [PubMed] [Google Scholar]
- Sakura S, Hashimoto K, Bollen AW, Ciriales R, Drasner K ( 1996) Intrathecal catheterization in the rat. Improved technique for morphologic analysis of drug-induced injury. Anesthesiology 85: 1184–1189. [DOI] [PubMed] [Google Scholar]
- Sassone-Corsi P, Visvader J, Ferland L, Mellon PL, Verma IM ( 1988) Induction of proto-oncogene fos transcription through the adenylate cyclase pathway: characterization of a cAMP responsive element. Genes Dev 2: 1529–1538. [DOI] [PubMed] [Google Scholar]
- Schaible HG, Grubb BD ( 1993) Afferent and spinal mechanisms of joint pain. Pain 55: 5–54. [DOI] [PubMed] [Google Scholar]
- Shaywitz AJ, Greenberg ME ( 1999) CREB: a stimulus-induced transcription factor activated by a diverse array of extracellular signals. Annu Rev Biochem 68: 821–861. [DOI] [PubMed] [Google Scholar]
- Skyba DA, King EW, Sluka KA ( 2002) Effects of NMDA and non-NMDA ionotropic glutamate receptor antagonists on the development and maintenance of hyperalgesia induced by repeated intramuscular injection of acidic saline. Pain 98: 69–78. [DOI] [PubMed] [Google Scholar]
- Sluka KA ( 1997) Activation of the cAMP transduction cascade contributes to the mechanical hyperalgesia and allodynia induced by intradermal injection of capsaicin. Br J Pharmacol 122: 1165–1173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sluka KA ( 2002) Stimulation of deep somatic tissue with capsaicin produces long-lasting mechanical allodynia and heat hypoalgesia that depend on early activation of the cAMP pathway. J Neurosci 22: 5687–5693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sluka KA, Westlund KN ( 1993) Behavioral and immunohistochemical changes in an experimental arthritis model in rats. Pain 55: 367–377. [DOI] [PubMed] [Google Scholar]
- Sluka KA, Willis WD ( 1997) The effects of G-protein and protein kinase inhibitors on the behavioral responses of rats to intradermal injection of capsaicin. Pain 71: 165–178. [DOI] [PubMed] [Google Scholar]
- Sluka KA, Dougherty PM, Sorkin LS, Willis WD, Westlund KN ( 1992) Neural changes in acute arthritis in monkeys. III. Changes in substance P, calcitonin gene-related peptide and glutamate in the dorsal horn in the spinal cord. Brain Res Rev 17: 29–38. [DOI] [PubMed] [Google Scholar]
- Sluka KA, Kalra A, Moore SA ( 2001) Unilateral intramuscular injections of acidic saline produce a bilateral, long-lasting hyperalgesia. Muscle Nerve 24: 37–46. [DOI] [PubMed] [Google Scholar]
- Storkson RV, Kjorsvik A, Tjolsen A, Hole K ( 1996) Lumbar catheterization of the spinal subarachnoid space in the rat. J Neurosci Methods 65: 167–172. [DOI] [PubMed] [Google Scholar]
- Sweatt JD ( 1999) Toward a molecular explanation for long-term potentiation. Learn Mem 6: 399–416. [DOI] [PubMed] [Google Scholar]
- Urban MO, Zahn PK, Gebhart GF ( 1999) Descending facilitatory influences from the rostral medial medulla mediate secondary, but not primary, hyperalgesia in the rat. Neuroscience 90: 349–352. [DOI] [PubMed] [Google Scholar]
- Vazquez SI, Vazquez A, Pena de Ortiz S ( 2000) Different hippocampal activity profiles for PKA and PKC in spatial discrimination learning. Behav Neurosci 114: 1109–1118. [DOI] [PubMed] [Google Scholar]
- Walsh DA, Glass DB ( 1991) Utilization of the inhibitor protein of adenosine cyclic monophosphate-dependent protein kinase, and peptides derived from it, as tools to study adenosine cyclic monophosphate-mediated cellular processes. Methods Enzymol 201: 304–316. [DOI] [PubMed] [Google Scholar]
- Wei F, Qiu CS, Kim SJ, Muglia L, Maas JW, Pineda VV, Xu HM, Chen ZF, Storm DR, Muglia LJ, Zhuo M ( 2002) Genetic elimination of behavioral sensitization in mice lacking calmodulin-stimulated adenylyl cyclases. Neuron 36: 713–726. [DOI] [PubMed] [Google Scholar]
- White DM, Helme RD ( 1985) Release of substance P from peripheral nerve terminals following electrical stimulation of the sciatic nerve. Brain Res 336: 27–31. [DOI] [PubMed] [Google Scholar]
- Willis WD, Coggeshall RE ( 1991) Sensory mechanisms of the spinal cord, Ed 2. New York: Plenum.
- Xia Z, Storm DR ( 1997) Calmodulin-regulated adenylyl cyclases and neuromodulation. Curr Opin Neurobiol 7: 391–396. [DOI] [PubMed] [Google Scholar]
- Zou X, Lin Q, Willis WD ( 2000) Enhanced phosphorylation of NMDA receptor 1 subunits in spinal cord dorsal horn and spinothalamic tract neurons after intradermal injection of capsaicin in rats. J Neurosci 20: 6989–6997. [DOI] [PMC free article] [PubMed] [Google Scholar]