Abstract
Ca2+-activated K+ currents and their Ca2+ sources through high-threshold voltage-activated Ca2+ channels were studied using whole-cell patch-clamp recordings from freshly dissociated mouse neocortical pyramidal neurons. In the presence of 4-aminopyridine, depolarizing pulses evoked transient outward currents and several components of sustained currents in a subgroup of cells. The fast transient current and a component of the sustained currents were Ca2+ dependent and sensitive to charybdotoxin and iberiotoxin but not to apamin, suggesting that they were mediated by large-conductance Ca2+-activated K+ (BK) channels. Thus, mouse neocortical neurons contain both inactivating and noninactivating populations of BK channels. Blockade of either L-type Ca2+ channels by nifedipine or N-type Ca2+ channels by ω-conotoxin GVIA reduced the fast transient BK current. These data suggest that the transient BK current is activated by Ca2+entry through both N- and L-type Ca2+ channels. The physiological role of the fast transient BK current was also examined using current-clamp techniques. Iberiotoxin broadened action potentials (APs), indicating a role of BK current in AP repolarization. Similarly, both the extracellular Ca2+ channel blocker Cd2+ and the intracellular Ca2+chelator BAPTA blocked the transient component of the outward current and broadened APs in a subgroup of cells. Our results indicate that the outward current in pyramidal mouse neurons is composed of multiple components. A fast transient BK current is activated by Ca2+ entry through high-threshold voltage-activated Ca2+ channels (L- and N-type), and together with other voltage-gated K+ currents, this transient BK current plays a role in AP repolarization.
Keywords: Ca2+ channels, Ca2+-activated K+ channels, BK channels, neocortical pyramidal neurons, iberiotoxin, charybdotoxin, nifedipine, ω-conotoxin GVIA, BAPTA
Introduction
Acute hypoxia causes various biological responses in many types of organs or cells. The specific intracellular signaling pathways involved in the cellular responses and adaptation to hypoxia are not yet well defined. Our interest has been focused on the factors that initially trigger these processes. For example, we have shown previously that hypoxia inhibited single large-conductance Ca2+-activated K+ (BK) channel activities in mouse neocortical neurons (Liu et al., 1999). The signaling pathways involved in this inhibition have not been clear. Indeed, the reduction in BK channel activities could have resulted from a hypoxia-evoked decline in Ca2+ influx or a direct effect of cytosolic factors on the channels themselves. Although there is some evidence that hypoxia can reduce the voltage-dependent Ca2+ currents in carotid glomus cells (Lopez-Barneo et al., 1997), other studies have shown either an increase (Summers et al., 2000) or no significant changes in such currents (Keating et al., 2001).
An elevation in intracellular Ca2+ level has been identified, however, as one of the early responses to exposure to hypoxia in dissociated central neurons, including hippocampal neurons (Friedman and Haddad, 1993) and cultured neocortical neurons (Chow and Haddad, 1998). A high cytosolic concentration of Ca2+ may result in the activation of many physiological or pathophysiological processes, including the activation of Ca2+-dependent K+ channels, such as BK channels. In many neurons, calcium influx via voltage-gated calcium channels activates a variety of calcium-dependent K+ currents with different functional roles. In hippocampal neurons, N-type Ca2+ channels activate BK channels selectively, whereas L-type channels provide Ca2+ for small-conductance Ca2+-activated K+ (SK) channel activation (Marrion and Tavalin, 1998). In the present study, we sought to (1) identify the Ca2+-dependent components of the outward currents and (2) characterize BK currents and their Ca2+ sources through Ca2+ channels using whole-cell patch-clamp recordings in neocortical pyramidal neurons. Understanding the relationship of BK channels and their Ca2+sources would be helpful for a better understanding of the pathophysiological roles they play under conditions of hypoxia and ischemia.
Materials and Methods
Preparation of neocortical neurons. In accordance with the guidelines of the Yale Animal Care and Use Committee, mice [postnatal day 9(P9)–P15] were deeply anesthetized with halothane and killed by decapitation. The brain was removed rapidly, chilled in 0–4°C oxygenated isolation buffer containing (in mm): 120 NaCl, 5 KCl, 1 CaCl2, 10 HEPES, and 25 glucose, pH adjusted to 7.0 with NaOH, and sectioned transversely into <800 μm slices. The neocortical slices were dissected from these slices with the aid of a dissecting microscope. Neocortical slices were then incubated for 20 min with oxygenated isolation buffer containing protease (type XIV fromStreptomyces griseus, Sigma P-5147, 0.5 mg/ml, at 32°C). Thick slices and low concentrations of protease were used to minimize enzyme exposure. Slices were then washed in oxygenated buffer and maintained for up to 4 hr. Immediately before recording, a cortical slice was dissociated by gentle trituration with fire-polished Pasteur pipettes. The cells were collected and placed into a plastic Petri dish and were allowed several minutes to settle and adhere to the Petri dish before perfusion was initiated. Recordings were obtained only from pyramidal-shaped neurons that had a single thick proximal dendrite and did not show any visible evidence of injury.
Application of drugs and perfusion. Solutions were applied by a single-pass, gravity-fed perfusion system that delivered medium to the recording chamber (chamber volume, <0.5 ml) at a rate of 2 ml/min. To record Ca2+ channel currents, the chamber was perfused with a bathing solution containing (in mm): 110 NaCl, 3 KCl, 5 CsCl, 15 TEACl, 3 CaCl2, 1 MgCl2, 10 HEPES, 0.0005 TTX, and 10 glucose, with pH adjusted to 7.4 with NaOH. To record calcium-activated potassium channel currents, cells were superfused with a HEPES solution contained (in mm): 140 NaCl, 3 KCl, 1 CaCl2, 1 MgCl2, 10 HEPES, 0.0005 TTX, 1 4-aminopyridine (4-AP), 0.005 glybenclamide, and 10 glucose, pH adjusted to 7.4 with NaOH. TTX, 4-AP, and glybenclamide were included routinely in the K+recording solution unless otherwise indicated. 4-AP (1 mm) was used to reduce voltage-gated K+ currents and unmask Ca2+ dependence of K+ currents. A low concentration of 4-AP was chosen to minimize possible nonspecific blockade on other K+ currents, such as SK currents. Experiments were conducted at room temperature (22–25°C).
Whole-cell patch-clamp recording. Calcium and potassium currents were recorded from mouse neocortical neurons by use of the whole-cell patch-clamp technique. Recording pipettes were made from filament borosilicate capillary glass (1.2 mm outer diameter, 0.69 mm inner diameter) (Warner Instruments, Hamden, CT), using a Flaming/Brown micropipette puller (model P-87; Sutter Instrument, Novato, CA). The pipettes were fire-polished and had resistances of 2–5 MΩ when filled with the solutions listed below. To record calcium current, pipettes were filled with intracellular solution containing (in mm): 120 CsCl, 5 NaCl, 0.5 CaCl2, 2 MgCl2, 10 HEPES, 10 EGTA, 1 ATP, 0.2 GTP, and 0.1 leupeptin, with pH adjusted to 7.4 with CsOH. Patch solutions for potassium current recording contained (in mm): 110 K-gluconate, 10 KCl, 5 NaCl, 2 MgCl2, 10 HEPES, 0.5 EGTA, 1 ATP, 0.2 GTP, and 0.1 leupeptin, with pH adjusted to 7.4 with KOH. BAPTA (10 mm) was included in intracellular solutions in some experiments. Osmolarity was measured and adjusted to between 290 and 310 mOsm (Wescor osmometer; Wescor Inc., Logan, UT). Membrane potential and membrane currents were recorded with an Axopatch 200A amplifier (Axon Instruments, Foster City, CA). Signals were obtained at sampling rates of 5 and 50 kHz for voltage- and current-clamp recordings, respectively, and stored on the hard disk of a personal computer. Stimulus generation and data acquisition were controlled with the ClampEx program in the pClamp6 software package (Axon Instruments). Before seals were made on cells, offset potentials were nulled. Capacitance subtraction was used in all recordings. Recordings were accepted when resting membrane potentials (measured immediately after membrane rupture) were more negative than −40 mV or holding currents at −70 mV were <100 pA. The series resistances were primarily 10–20 MΩ. Leak currents were not subtracted online.
Drugs and solutions. All the channel blockers used in the present study were purchased from Sigma. Charybdotoxin (ChTX) and apamin were first dissolved in water at concentrations of 0.1 and 2 mg/ml, respectively; Bay K 8644 stock solution was prepared in methanol at 10 mm; glybenclamide and nifedipine were dissolved in dimethyl sulfoxide (DMSO) at 20 and 50 mm, respectively; ω-conotoxin GVIA was dissolved in medium at 0.1 mm. TTX stock solution concentration was 1 mm. Aliquots of stock solutions were kept at −80°C and were later diluted with external solution before use. The final concentration of either methanol or DMSO was <1:5000, a concentration that was found to have no effect on Ca2+ current (Sun et al., 2002)
Data analysis. Calcium channel currents were elicited from a holding potential of −70 mV. Single steps to −10 mV or a series of 10 mV steps between −60 and +50 mV for 100 msec were used. Mean values measured at 60 msec in a 100 msec voltage step were used to evaluate calcium channel current amplitude. K+currents were evoked by depolarizing steps (−70 to +50 mV) from a holding potential of −70 mV for 190 msec, with a 5 sec interval. Current-voltage (I–V) relationships were generated by measuring values at the first peak for the transient outward current and at 100 msec for the sustained current. Percentage block of these currents was defined as [1 − (Itest)/(Icontrol)] × 100, and values from depolarizing steps to −10 and +30 mV were used to evaluate Ca2+ and K+ current percentage changes respectively. Raw data were compared for statistical significance using ANOVA for between-group comparison and paired t test for within-group comparison. Data are presented as mean ± SEM.
Results
General properties of outward currents
Using whole-cell patch-clamp techniques, inward Ca2+ currents, outward K+ currents, and membrane potential were studied in a total of 124 neocortical pyramidal neurons. Figure1A shows the morphology of the somata and the apical dendrite of a typical cell studied.
Fig. 1.
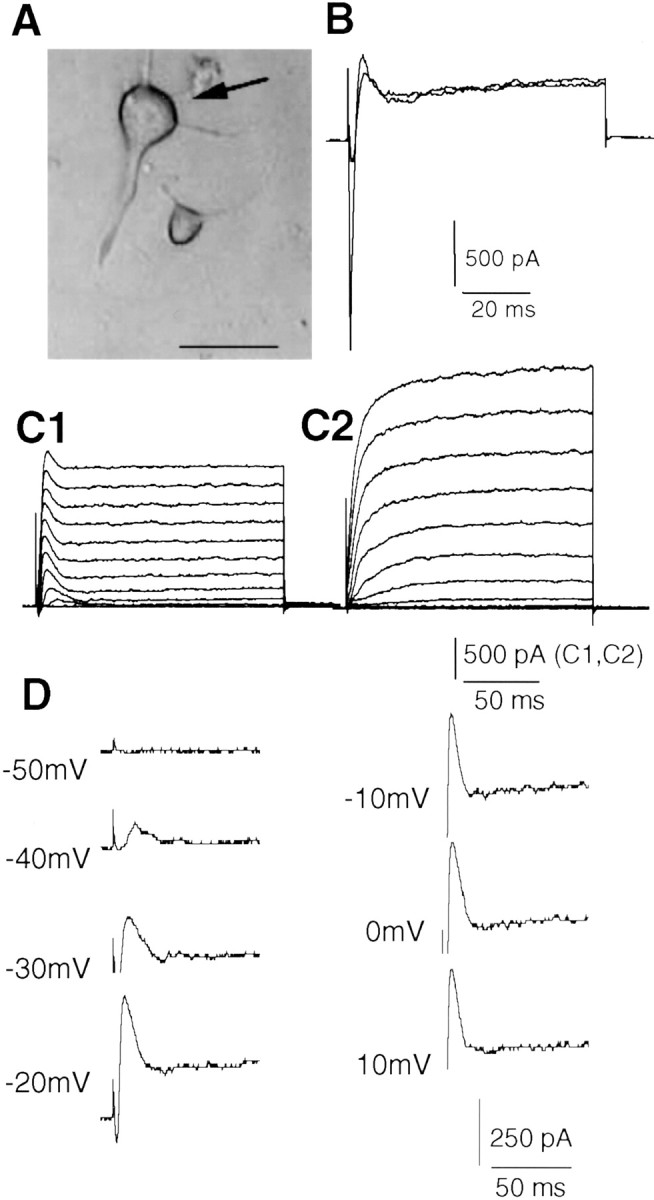
Outward currents in neocortical pyramidal neurons.A, Morphology of freshly dissociated neocortical pyramidal neurons. Bright-field image showing typical shape of the soma and at least one apical dendrite. The arrow indicates a pyramidal cell. Scale bar, 30 μm. B, Superimposed outward current traces in response to a voltage step to −10 mV from a holding potential of −70 mV before and during application of recording solution containing 1 mm 4-AP, 0.5 μm TTX, and 5 μm glybenclamide. Transient sodium currents were blocked by TTX. In the presence of 4-AP, a transient outward current was revealed. C, Currents were elicited by depolarizing voltage steps (−70 to +50 mV) from a holding potential of −70 mV (for 160 msec, in 10 mV steps, every 5 sec) during superfusion. Representative examples of cells expressing an early transient outward current (C1) and cells showing only a sustained current (C2). D, Expanded current traces taken from C1, showing an early transient outward current at depolarizing voltages from −40 to 10 mV.
Whole-cell voltage-clamp experiments were performed using K+-filled pipettes to characterize the K+ currents evoked by depolarization. Extracellular recording solution contained a mixture of channel blockers: 4-AP, 1 mm, for transient voltage-gated K+ current; TTX, 0.5 μm, for Na+ current; and glybenclamide, 5 μm, for ATP-sensitive K+current. These agents were added routinely to the recording perfusate unless otherwise stated. The effects of these blockers were evaluated in five cells. Figure 1 shows an example of inward Na+ and outward K+ currents, evoked by a depolarizing pulse to −10 mV from a holding potential of −70 mV before (control) and after application of channel blockers. The inward Na+ currents were blocked by TTX. The outward K+ currents could be separated into a rapidly activating transient component and a more slowly activating sustained component. The transient component could be partially blocked by 4-AP, revealing the presence of a rapidly activating, partially inactivating, 4-AP-insensitive outward current. The sustained component was not greatly affected.
In a total of 100 control cells using the solutions described above, 64 cells expressed a transient outward current. Figure 1C shows families of currents evoked by holding at −70 mV and stepping to potentials ranging from −70 to +50 mV for 160 msec during superfusion. In the example shown in Figure 1C1, this paradigm resulted in transient outward currents at potentials positive to −40 mV. In another cell shown in Figure 1C2, however, these depolarizing steps evoked only slowly activating, sustained outward currents. The expanded traces shown in Figure 1D are from current traces in Figure 1C1. The transient outward current showed a threshold at −40 mV and became both larger and faster between −40 and −10 mV (Fig. 1D). At membrane potentials >0 mV, however, the peak amplitude of the transient K+ currents did not increase further and even fell slightly at more depolarized potentials, despite an increased driving force on K+. Such a decrease at much depolarized potentials is expected for Ca2+-dependent currents because of the suppression of Ca2+ influx as the membrane potential approaches the equilibrium potential for Ca2+.
Ca2+-dependent component of outward currents
To further test the Ca2+ dependence of the transient outward currents, we examined the effects of blocking Ca2+ influx on this current by applying Cd2+, a nonspecific Ca2+ channel blocker. In the presence of 1 mm 4-AP, eight cells showed a transient outward current with varied amplitude, and the transient component was completely and reversibly blocked by 100 μmCd2+. An example is shown in Figure2A. Outward currents were evoked by stepping from −70 mV to potentials ranging from −70 to +50 mV in control (Fig. 2A1) and with 100 μm Cd2+ (Fig.2A2). Control currents resembled those described in Figure 1 in having an early transient component followed by a sustained current. The transient component was abolished by Cd2+. On average, Cd2+ significantly reduced the transient peak (evoked by single depolarizing steps from −70 to +30 mV) from 1527 ± 179 to 972 ± 124 pA (n = 10;p < 0.05). The voltage dependence of the Cd2+-sensitive current correlates well with that of Ca2+ currents (Fig.2A3,A4) (see also Fig. 8).
Fig. 2.
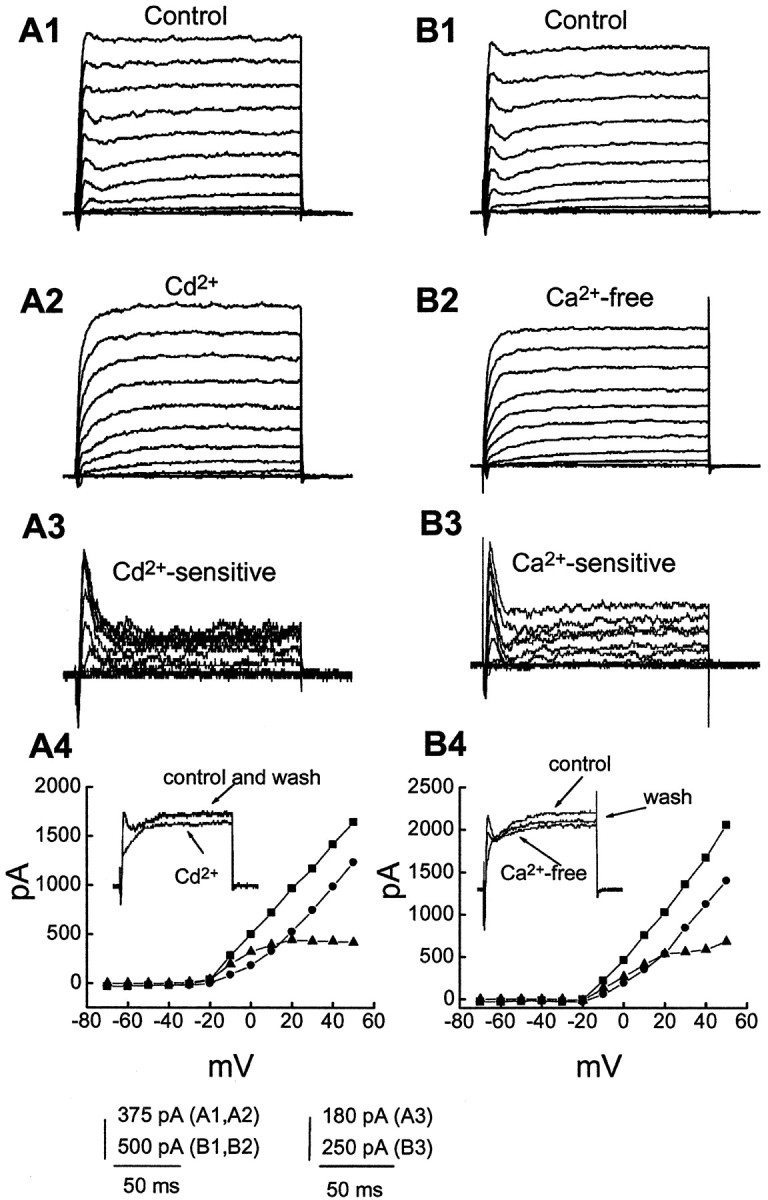
Ca2+ dependence of the transient outward current. Membrane potential was held at −70 mV and stepped from −70 to +50 mV for 160 msec in 10 mV steps every 5 sec.A and B show currents activated by these voltage steps in superfusing medium containing 4-AP (1 mm) before (A1, B1) and during application of 100 μm Cd2+- (A2) or Ca2+-free (B2) solution.A3, B3, Ca2+-sensitive currents (subtraction A1 − A2 orB1 − B2). A4,B4, I–V plots obtained from outward currents in A1–3 and B1–3 measured at the time point of first peak of control transient currents. ▪, Control; ●, in the presence of Cd2+- (A4) or Ca2+-free solution (B4); ▴, Cd2+(A4) or Ca2+(B4)-sensitive currents. Insets, Superimposed current traces in response to a voltage step to −10 from −70 mV showing a reversible effect of 100 μmCd2+- and Ca2+-free medium on outward currents.
Fig. 8.
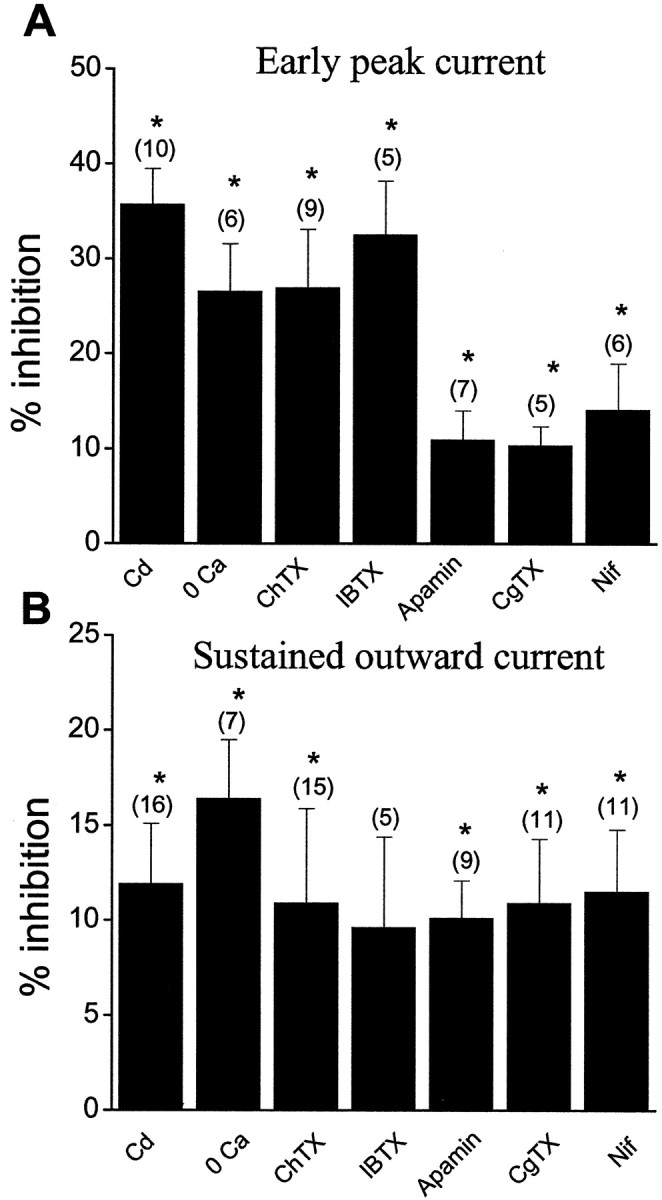
Effects of various blocking agents on components of transient and sustained outward currents. A, Summary of effects of various agents on the early peak outward current. Values are measured at the first peak of outward current evoked by a depolarizing step from −70 to +30 mV before each agent (control). Numbers included only cells expressing transient outward current.B, Group data of effects of various agents on sustained outward current. Values are measured at 100 msec of outward current evoked by a depolarizing step from −70 to +30 mV. Numbers included cells with and without transient outward currents. Data are expressed as mean ± SE. The asterisk indicates a significant difference (p < 0.05) between control and treatments (Student's paired t test).
The effect of free Ca2+ on the outward currents was also examined. Application of Ca2+-free medium, in which Ca2+ was replaced by Mg2+, virtually eliminated the transient outward current. The peak amplitude of this transient outward current (evoked by depolarizing steps from −70 to +30 mV) was reduced from 1339 ± 148 to 989 ± 139 pA (n = 6;p < 0.05). An example is shown in Figure2B. A Ca2+-sensitive current, which was obtained by subtraction of currents evoked in Ca2+-free medium from currents evoked in control medium, consisted of both transient and sustained components (Fig. 2B3). The sensitivity of the transient outward current to treatments that block Ca2+influx indicates that this current is Ca2+dependent.
Effects of block of BK channel blockers on the transient outward current
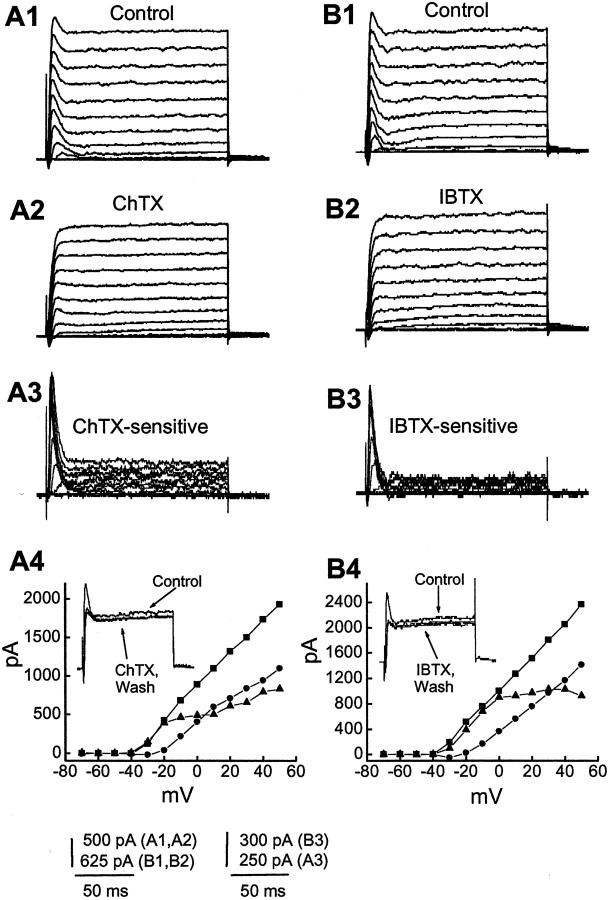
Because the transient outward current is Ca2+-dependent, we also used specific Ca2+-activated potassium channel blockers to determine which channels mediate this current. Evidence that a major part of this current was mediated by BK channels is presented by the experiments shown in Figure 3, in which the effects of two potent BK channel blockers were examined in 14 cells. Figure 3A shows an example of outward currents evoked by stepping from −70 mV to depolarized potentials (−70 to +50 mV) in control (Fig. 3A1) and in the presence of 50 nm ChTX, a BK channel blocker (Fig.3A2). Application of ChTX eliminated most of the transient currents (Fig. 3A). Block of ChTX was irreversible during the time course of these experiments (Fig. 3A4, inset), and the amplitude of the early peak (evoked by depolarizing step to +30 from −70 mV) was reduced from 1370 ± 163 to 1025 ± 165 pA (n = 9; p < 0.05).
Fig. 3.
ChTX and IBTX block the transient outward currents. Membrane potential was held at −70 mV and stepped from −70 to +50 mV for 160 msec in 10 mV steps every 5 sec. A andB show currents activated by these voltage steps in superfusing medium containing 4-AP before (A1,B1) and during application of 50 nm ChTX (A2) and 100 nm IBTX (B2).A3, B3, ChTX-sensitive currents (subtraction A1 − A2) and IBTX-sensitive currents (B1 − B2).A4, B4, I–V plots obtained from outward currents in A1–A3 andB1–B3 measured at the time point of early peak of control transient currents. ▪, Control; ●, in the presence of ChTX (A4) or IBTX (B4); ▴, ChTX (A4) or IBTX (B4)-sensitive currents. Insets, Superimposed current traces evoked by depolarizing steps to −10 from −70 mV, showing an irreversible effect of ChTX and IBTX on outward currents during the time course of experiment.
The effect of another selective blocker for the BK type of Ca2+-activated K+ current, iberiotoxin (IBTX), was also examined. Figure 3B shows that this peptide similarly blocked the transient current in all cells tested (Fig. 3B). On average, the early peak current was 1492 ± 105 pA before and 998 ± 96 pA after 100 nm IBTX (n = 5; p < 0.05). These toxins also reduced a component of the sustained currents. The BK current activated fully within 5 msec and then decayed within 20 msec to a steady level (Fig. 3A3,B3). Inactivation could be fit by a single exponential equation. The inactivation time constant of the transient BK current evoked by a depolarizing step to +30 mV from −70 mV was 3.5 ± 0.2 msec, and time to peak was 4.9 ± 0.4 msec (n = 4), which is not significantly different from the time to peak for calcium current (4.5 ± 0.3 msec;n = 13; one-way ANOVA; p < 0.05) (see also Fig. 5A).
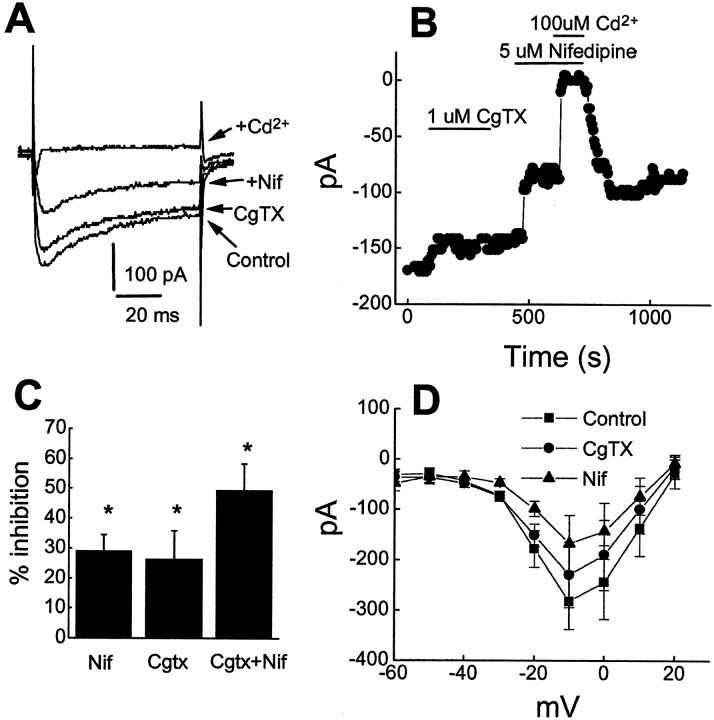
Fig. 5.
Analysis of contribution of L- and N-type Ca2+ channel in total Ca2+currents in mouse neocortical pyramidal neurons. A, A representative experiment illustrating the effect of the L-type Ca2+ channel blocker nifedipine (5 μm), N-type Ca2+ channel blocker ω-CgTx GVIA (1 μm), and Cd2+ (100 μm) on the inward Ca2+ current.B, Time course of the experiment shown inA illustrating the sequential blocking actions of ω-CgTx GVIA, nifedipine, and Cd2+.C, Group data of percentage block of nifedipine (n = 6), and ω-CgTx GVIA (n = 4). D, I–V plots of Ca2+ currents in the absence (control, n = 6) and presence of nifedipine and ω-CgTx GVIA.
Voltage dependence of the transient BK current (Fig. 3) and Ca2+ current (Fig. 5) match well in the voltage range of −40 to +20 mV, where the Ca2+ current is obvious. The observation that the transient K currents show little or no decline at positive membrane potentials is consistent with a series resistance error. TheI–V relationships of ChTX- and IBTX-sensitive currents differed slightly from those of Cd2+- and Ca2+-sensitive currents shown in Figures 2and 3. The ChTX- or IBTX-sensitive currents presumably represented the effects of blocking BK currents only, whereas the Cd2+- or Ca2+-sensitive currents reflected the combined effects of blocking inward Ca2+currents and outward Ca2+-dependent K+ currents (BK and SK).
These results indicate that BK channels mediate a major part of the transient outward current in mouse neocortical pyramidal neurons.
Effect of block of SK channels on the transient component
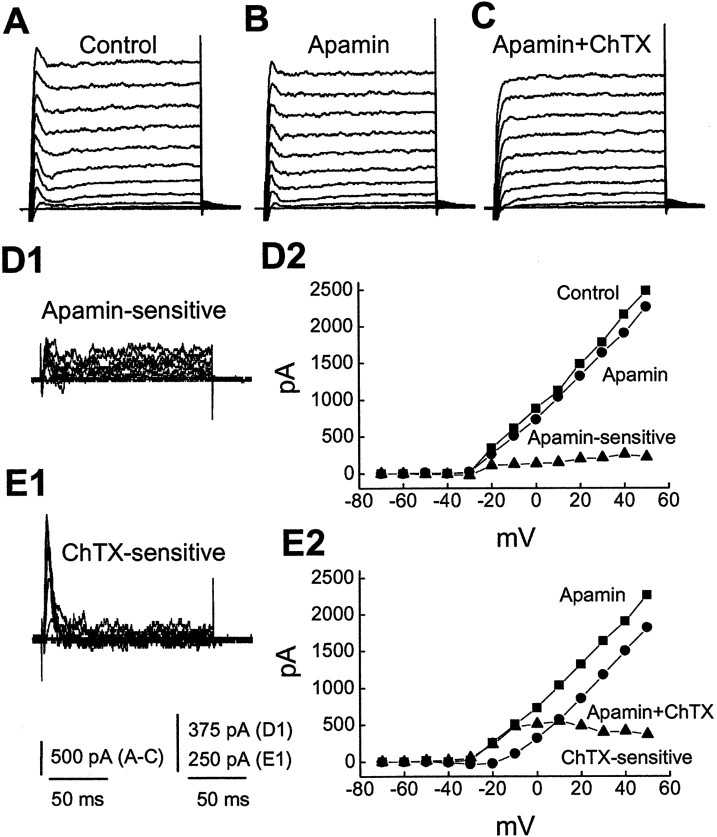
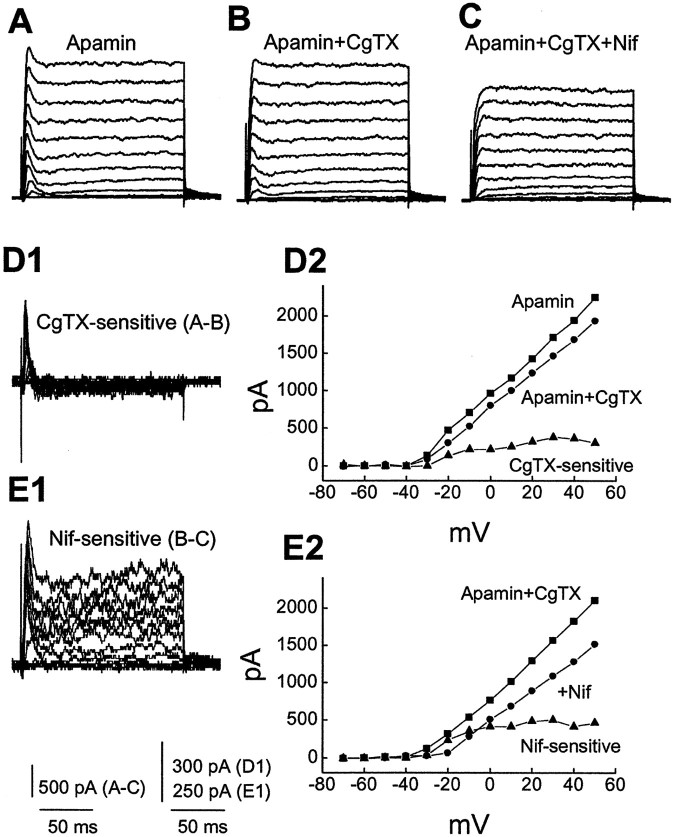
To test whether SK channels were involved, we examined the effect of apamin, which blocks SK channels. In contrast to ChTX and IBTX, application of 500 nm apamin had no obvious effect on the fast transient outward current, suggesting that the transient current is apamin insensitive. In seven of the cells tested, however, apamin affected only the slower current in six cells and did not have an obvious effect in another (data not shown). An example is shown in Figure 4. The fast transient outward current persisted after application of apamin (Fig.4A,B), and addition of ChTX to the solution resulted in complete blockade of the fast transient outward currents (Fig.4C). A sustained component of the outward current is sensitive to apamin (Fig. 4D), whereas ChTX-sensitive current is fast activating and rapidly inactivating (Fig.4E). Thus, the fast transient outward current is sensitive to a toxin specific for BK channels but not to a toxin that blocks SK channels.
Fig. 4.
Charybdotoxin but not apamin blocks the transient outward current. Membrane potential was held at −70 mV and stepped from −70 to +50 mV for 160 msec in 10 mV steps every 5 sec.A–C, Currents activated by these voltage steps in superfusing medium containing 4-AP before (A, Control) and during additive application of 500 nm apamin (B) and 50 nm ChTX (C). ChTX but not apamin resulted in complete blockade of the transient outward current. D1 shows the apamin-sensitive currents obtained by subtracting B fromA. D2, I–V plots obtained from outward currents in A, B, andD1 measured at the time point of peak transient currents in the control. E1, ChTX-sensitive current obtained by subtracting C from B. E2,I–V plots measured from B,C, and E1.
Characterization of the inward calcium currents
Rat neocortical neurons have been shown previously to express pharmacologically distinct types of calcium channels, including N-type and L-type channels (Lorenzon and Foehring, 1995). We used the selective Ca2+ channel blockers ω-conotoxin-GVIA (ω-CgTX, N-type) and nifedipine (L-type) and have confirmed previous observations that neocortical pyramidal neurons express multiple subtypes of high-threshold Ca2+ currents. Figure5 shows Ca2+currents evoked by depolarizing voltage steps from a holding potential of −70 mV. Figure 5A,B illustrates the effects of sequential application of ω-CgTX (1 μm), nifedipine (5 μm), and Cd2+ (100 μm) on the calcium current evoked at −10 mV. Nifedipine produced a rapid and reversible block of Ca2+ current in all cells tested, indicating the presence of L-type Ca2+ channels. Nifedipine alone blocked calcium channel current by approximately one-third (29.8 ± 9.1%;n = 7; p < 0.05) (Fig.5A–C). ω-CgTX produced irreversible inhibition of Ca2+ current by an average of 26 ± 2.9% (n = 4; p < 0.05) (Fig.5A–C). A substantial fraction (50%) of the total Ca2+ current was unaffected by the combined actions of nifedipine and ω-CgTX, indicating the presence of other HVA currents. Cd2+ completely and reversibly blocked all inward calcium current in all cells tested (n = 6; p < 0.05). Figure5D shows the Ca2+ currentI–V relationships obtained in the absence (control) and presence of Ca2+ channel blockers. When elicited by step depolarization from a holding potential of −70 mV, Ca2+ currents were normally activated at approximately −40 mV, peaked at −10 mV, and reversed between +30 and +40 mV. Nifedipine and ω-CgTX exerted an inhibitory effect at all potentials at which currents were activated; this inhibition was not accompanied by a shift in the peak voltage of Ca2+ current.
Contribution of L- and N-type Ca2+ channels in activating transient BK current
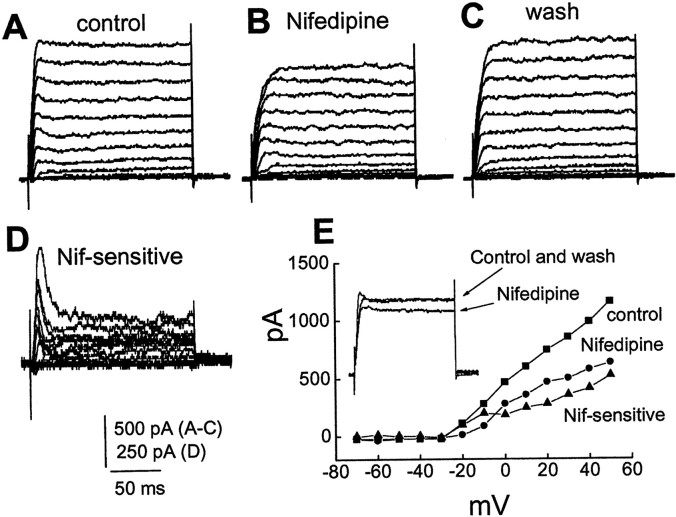
The role of L-type Ca2+ channels in the fast transient BK current activation was examined next with 5 μm nifedipine (antagonist) and 1 μm Bay K8644 (agonist). Figure 6 shows an example of the effects of nifedipine on BK currents. Nifedipine caused a reduction in the magnitude of the fast transient BK current (Fig. 6). Nifedipine-sensitive current, which was obtained by subtractingB (nifedipine) from A (control), had an early transient component followed by a sustained component (Fig.6D). Six cells tested expressed the transient BK current, and nifedipine reduced the early peak current to a mean of 1180 ± 186 pA from a mean of 1343 ± 157 pA (n = 6; p < 0.05). An L-type Ca2+ agonist, Bay K8644, enhanced a transient outward current (n = 3; data not shown).
Fig. 6.
L-type Ca2+ channels contribute to activation of the transient BK currents. Currents were evoked by depolarizing steps (−70 to +50 mV) for 160 msec from a holding potential of −70 mV in control (A), in the presence of nifedipine (5 μm) (B), and after washout (C). Nifedipine inhibits the transient BK current and a sustained outward current. D, Nifedipine-sensitive current was obtained by subtracting B from A. E,I–V plots, which were measured at the early peak time point, showing a slightly N-shaped nifedipine-sensitive transient current. Inset, Superimposed current traces evoked by depolarizing steps to −10 from −70 mV.
We also examined the contribution of N-type channels toward BK current activation. Five cells expressed, to various extents, a transient outward current, and 1 μm ω-CgTX partially blocked the early peak current (average of 1711 ± 254 pA before and 1540 ± 239 pA after ω-CgTX; n = 5; p < 0.05). As shown in Figure 7, in the presence of 500 nm apamin, which removed apamin-sensitive SK current, ω-CgTX resulted in partial reduction in the transient BK current, yielding a small ω-CgTX-sensitive current (Fig. 7B,D). Another addition of nifedipine (5 μm) eliminated most of the transient BK current (Fig. 7C,E). After pretreatment with ChTX, ω-CgTX reduced only the slow component of outward currents, whereas in cells pretreated with apamin, ω-CgTX inhibited the fast transient BK current (data not shown). Thus, these results indicate that N-type Ca2+ channels contributed to the transient BK current activation.
Fig. 7.
Both L- and N-type Ca2+channels contribute to activation of the transient BK currents. Membrane potential was held at −70 mV and stepped from −70 to +50 mV for 160 msec in 10 mV steps every 5 sec. Currents were recorded in the presence of apamin (500 nm) to eliminate SK current contribution. A–C, Currents recorded in superfusing medium containing apamin (A, control) and during additive application of 1 μm CgTX (B) and 5 μm nifedipine (C). Subtracting B fromA and C from B reveals a small transient CgTX-sensitive current (D1) and a nifedipine-sensitive current with both transient and sustained components (E1), respectively. D2,E2, I–V plots obtained fromA–E.
Sustained component of outward currents
As described above, both BK and SK channel blockers and Ca2+ channel blockers (L- and N-type) blocked part of the sustained outward currents. The extent of blockade varied greatly from cell to cell. Figure8 summarizes the effects of all the above-mentioned agents for both the early peak and sustained outward currents. These data suggested that the sustained outward current was composed of multiple components, including both BK and SK currents, and most of all, voltage-gated potassium currents. Ca2+ entry through both L- and N-type Ca2+ channels activated these Ca2+-activated BK and SK currents.
Role of transient BK current in membrane and action potentials
The functional role of the transient BK current has not been determined in neocortical neurons. To examine the role of the BK current, we performed current-clamp experiments. The extracellular solutions used were the same as those for voltage-clamp recordings for K+ currents, except that TTX was omitted. In the presence of 1 mm 4-AP, action potentials were elicited by suprathreshold depolarizing current injection. Because blockade of Ca2+ currents by Ca2+ channel blockers can produce effects unrelated to the reduction of BK current activation, we attempted to selectively remove the contribution of BK currents to action potential repolarization using IBTX. Application of 100 nm IBTX blocked the transient BK current (Fig.9A) and resulted in a slowing of the action potential repolarization recorded in the same cell with current-clamp recording (Fig. 9B). We found no significant differences in action potential amplitude, with a mean of 91.3 ± 6.2 mV before and 90.5 ± 5.9 mV after IBTX (p > 0.05; n = 6). The action potential half-width (width of the spike measured at half-maximal amplitude) was significantly increased by IBTX, from 2.9 ± 0.2 to 3.3 ± 0.3 msec, whereas the threshold duration (width of the spike measured at threshold) was increased to 8.9 ± 1.0 msec from 6.9 ± 0.6 msec (p < 0.05;n = 6). This experiment clearly shows that the transient BK current is normally activated during repolarization.
Fig. 9.
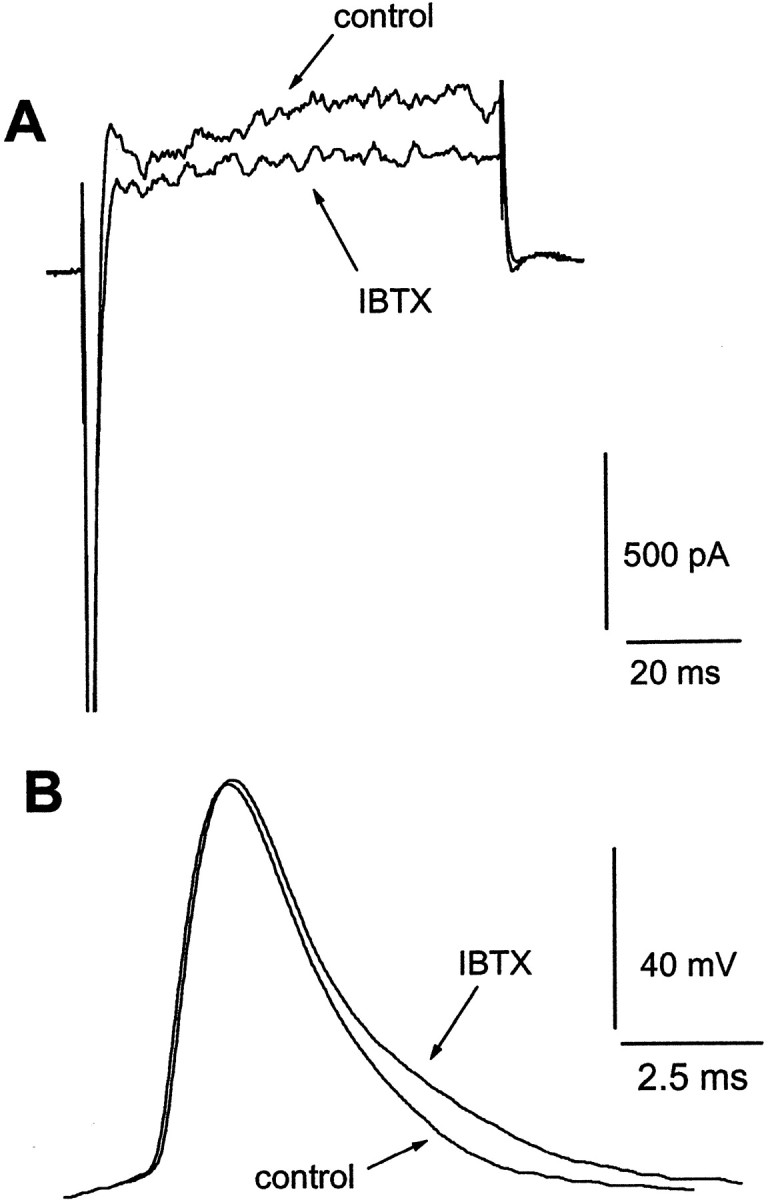
Effects of IBTX on outward currents and action potentials of neocortical pyramidal neurons. A, Voltage-clamp recordings showing superimposed current traces before (control) and after 100 nm IBTX during superfusion with 4-AP. Currents were evoked by a depolarizing step to −10 from −70 mV.B, Current-clamp recordings from the same cell as inA, showing superimposed voltage traces taken in these treatments. Action potentials were elicited by suprathreshold current injections. IBTX blocked the transient BK current and broadened action potentials.
Ca2+ dependence of action potential and membrane currents
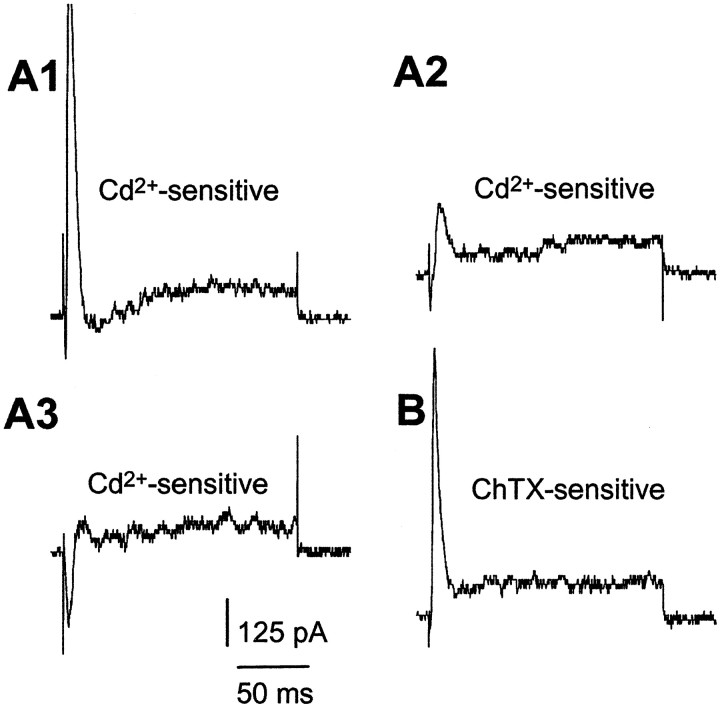
Several previous studies of neocortical pyramidal cells in rats, cats, and guinea pigs have failed to show evidence for Ca2+ dependence of action potential repolarization. We therefore tested this in mice in this work. As shown in Figure 1, cells vary in the expression of total outward currents, and only a subgroup of cells studied showed obvious transient outward currents. We then compared Cd2+-sensitive currents (current obtained by subtracting the current remaining in the presence of 100 μm Cd2+ from control current) during depolarization in different cells. We found that during the first 10 msec depolarization steps from −70 to +10 mV, depolarization evoked varied amounts of Ca2+ (the small inward current that precedes outward currents) and K+ currents (Fig. 10A1–3). Ca2+ dependence of action potential repolarization would then be expected to vary in these different cells.
Fig. 10.
Variations in Cd2+-sensitive currents activated by membrane depolarization in different cells. Currents are activated by 160 msec voltage steps from −70 to +10 mV.A1–A3, Cd2+-sensitive currents from three different cells. Currents are obtained by subtracting remaining currents in the presence of Cd2+ (100 μm) from currents before Cd2+application (control). A small inward Ca2+ current precedes outward currents in A2 and A3. The relative amount of Ca2+ and transient outward currents varies among these cells in the initial 10 msec depolarization period. B, A transient BK current in another cell. ChTX-sensitive current is obtained by subtracting the remaining current in the presence of ChTX (50 nm) from control current.
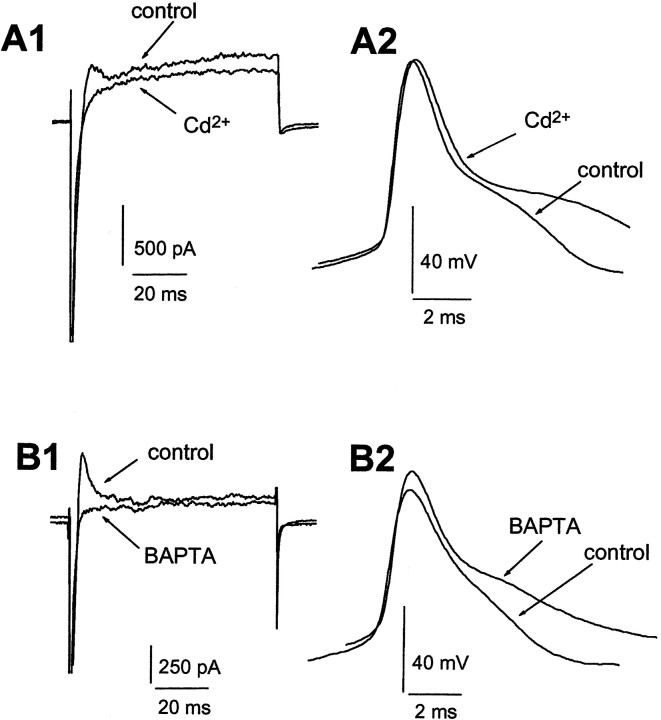
We next examined Ca2+ dependence of action potential repolarization using extracellularly applied Cd2+ (100 μm) and internal BAPTA (10 mm). In the presence of 1 mm 4-AP, membrane currents and action potentials were measured using voltage- and current-clamp recordings, respectively. Again, Cd2+ and BAPTA blocked all the transient outward currents (Fig.11A1,B1), and we found that in a majority of cells (8 of 11), action potential half-width was increased, from 2.6 ± 0.2 msec in control to 3.1 ± 0.5 msec; action potential threshold duration from 6.9 ± 0.6 to 9.7 ± 0.5 msec in the presence of Cd2+ (p < 0.05). In all the cells tested, the afterhyperpolarization (AHP) was consistently attenuated markedly by Cd2+. Examples of the effects of Cd2+ and BAPTA on outward currents and action potential repolarization are illustrated in Fig. 11.
Fig. 11.
Effects of Cd2+ and intracellular BAPTA on outward currents and action potentials of neocortical pyramidal neurons. A1, Voltage-clamp recordings showing superimposed current traces before (control) and after 100 nm IBTX during superfusion with 4-AP. Currents were evoked by a depolarizing step to −10 from −70 mV.A2, Current-clamp recordings from the same cell as inA1, showing superimposed voltage traces taken in these treatments. Action potentials were elicited by suprathreshold current injections. B1, Superimposed current traces from another cell taken with 10 mm intracellular BAPTA in the pipette immediately after rupture (control) and 2 min later (BAPTA).B2, Current-clamp recordings from the same cell as inB1. Both Cd2+ and BAPTA blocked transient outward currents and broadened action potentials.
Discussion
In the present study, we have made several interesting observations. For example, we have identified a fast transient BK current in mouse neocortical pyramidal neurons. In the presence of 4-AP, the fast transient outward current is Ca2+ dependent and sensitive to the specific BK channel blockers ChTX and IBTX but not to the SK channel blocker apamin. Furthermore, the transient BK current is activated by Ca2+ influx from L- and N-type Ca2+ channels during depolarization. Finally, this BK current plays an important role in action potential repolarization.
Variations in the transient BK current
In the present study, the magnitude of the transient and sustained BK currents varied from cell to cell. This may result from varied levels of expression of inactivating and noninactivating BK channels or varied regulation of the current itself. Variations in Ca2+ inward currents would also be a source of variations in the BK currents. Neocortical pyramidal cells are different in size, projections, laminar distribution, and firing pattern. Individual neocortical pyramidal cells vary greatly in the percentage contribution of each Ca2+channel type to the whole-cell Ca2+current (Stewart and Foehring, 2000). Thus, Ca2+ channel subtypes contribute differently to specific cellular events, such as neurotransmitter release, action potential repolarizations (BK channel activation), and AHPs (SK channel activation). Moreover, it is possible that some of the variability may also arise from the technique used by us and others to dissociate cells with different amounts of dendrites still attached to the soma, because there is a subcellular compartmentalization of channel types.
Inactivation of BK channel
In this study, we have shown that a transient outward current is sensitive to the BK channel blockers ChTX and IBTX (Fig. 3) but not to the SK channel blocker apamin (Fig. 4), indicating that the current is mediated by BK channels. In these neocortical neurons, the rapidly activating transient BK current decays rapidly to a steady level within ∼20 msec, with an inactivating time constant of ∼4 msec. In the present study, Ca2+ was used as current carrier. ICa reached its peak rapidly and started to inactivate slowly (Fig. 5). The transient BK current peaked at approximately the same time as Ca2+ current and started to inactivate when Ca2+ current also declined. Thus, it is unlikely that some of the apparent decline in outward current was actually a manifestation of the activation kinetics of the Ca2+ current.
Although BK channels are clearly found in many organisms and cell types, only a few of them exhibit inactivation, including hippocampal neurons (Hicks and Marrion, 1998), Drosophila muscle (Salkoff, 1983), chromaffin cells (Solaro and Lingle, 1992), RINm5f cells (Li et al., 1999), and frog hair cells (Armstrong and Roberts, 2001). The only known mechanism of BK channel inactivation is that conferred by auxiliary (β) subunits, which mediate inactivation through an intracellular “ball and chain” mechanism (Wallner et al., 1999; Xia et al., 1999, 2000). β-Subunit expression was detected in rat brain tissue and human cerebral cortex (Xia et al., 1999). However, to the best of our knowledge, our study is the first to report on inactivating BK currents in cortical pyramidal neurons. From the fast inactivation of this transient BK current in our study, it is possible that inactivation could be the result of interaction between α- and β3-subunits (Xia et al., 2000).
Role of L- and N-type Ca2+ channels in activating BK currents
Although intracellular recording studies have shown that various Ca2+ channel subtypes are involved in the generation of AHPs (SK) in neocortical pyramidal neurons (Pineda et al., 1998, 1999), an association of Ca2+channels with BK channels has not been well delineated. We found that both N- and L-type Ca2+ channel blockers partially eliminated the transient BK current (Figs. 6, 7), indicating that both subtypes provided Ca2+ for the activation of the current. In the present study, we cannot rule out the combination of other Ca2+ sources for the activation of the BK current; however, our data indicate that N- and L-type Ca2+ channels seem to be the major Ca2+ sources for this current.
The relationship between Ca2+ currents and Ca2+-dependent K+ currents depends on the subcellular localization of these channels and on intracellular Ca2+ accumulation during neural activity. Large and localized Ca2+ elevations are thought to occur only in the immediate vicinity of Ca2+ channels because of intracellular buffering. In the present study, intracellular solutions contained low concentrations of EGTA (0.5 mm). The presence of low intracellular concentrations of Ca2+buffers could result in a smaller peak value of [Ca2+]i transient after a fixed influx of Ca2+ and a prolonged decay (Schwindt et al., 1992; Sah and Clements, 1999). Ca2+ buffers have different roles in the control of different Ca2+-activated K+ currents in neurons. It has been shown that in hippocampal neurons, the action potential repolarization and the fast AHP, which is mediated by BK channels, were unaffected by 100 μm to 3 mm of internally applied BAPTA, whereas the slow AHP, which is presumed to be generated by SK-type Ca2+-activated K+ channels, was potentiated (Velumian and Carlen, 1999). Similarly, in cat neocortical neurons, a small reduction of intracellular Ca2+, resulting from low internal Ca2+ chelator concentration, preferentially enhanced a slow, Ca2+-dependent K+ current but did not significantly affect action potential time to half amplitude (Schwindt et al., 1992). Close proximity of BK channels to Ca2+channels has been shown in hippocampal neurons (Marrion and Tavalin, 1998; Velumian and Carlen, 1999). The fast-rising phase of the transient BK current in our study in neocortical neurons (Fig. 3) suggests that a fast increase in [Ca2+]i delivered close to the BK channels activates them rapidly. Thus, it seems likely that the low EGTA in the intracellular solution would not significantly affect the transient BK current (which constitutes our main interest in this study), although the possible enhancing effect on SK current cannot be excluded. In this study, we found that 10 mminternal BAPTA eliminated the transient outward currents (Fig. 11), suggesting that the distance between the transient BK current and its Ca2+ sources is larger than the buffering length constant for BAPTA.
Physiological roles of transient BK current in neocortical pyramidal neurons
In neurons, Ca2+-activated K+ channels have been reported to play an important role in regulating resting and action potentials (Lancaster and Nicoll, 1987; Storm, 1987; Sah and McLachlan, 1992). Because BK channels require simultaneous depolarization and elevated cytosolic calcium to become activated, this necessitates that electrical activity be coupled to changes in intracellular calcium. Furthermore, the large conductance of neuronal BK channels allows them to play an important role in hyperpolarizing the membrane potential when they are activated.
Although a number of K+ currents have been described previously in neocortical neurons, a major part of the action potential repolarization has not been found to be significantly Ca2+ dependent (Spain et al., 1991;Foehring and Surmeier, 1993; Kang et al., 2000). The physiological roles of BK channels in neocortical pyramidal neurons were relatively unclear from previous studies. The effect of Ca2+ entry blockade (Cd2+, BAPTA) on the repolarization phase of action potentials was variable in mouse neocortical neurons in our study, with a majority of cells increasing action potential half-width (Fig. 11). We believe that the difference between cells reflects the relative number of Ca2+ and BK channels activated during the action potential in different cells, with the two types of channels affecting action potential repolarization in opposite directions. The amount of Ca2+ current and BK current activated during depolarization indeed varies among cells (Fig. 10). A transient K+ current was found previously to be sensitive to TEA but not to 4-AP and to be involved in action potential repolarization; however, a BK channel blocker was not tested on these currents (Spain et al., 1991; Kang et al., 2000). Furthermore, it has been shown that a calcium-activated outward current was involved in the late repolarization in a subgroup of pyramidal cells of cat motor cortex (Chen et al., 1996), although the nature of this current was not characterized in that study. In rat neocortical pyramidal neurons, the final phase of repolarization was sensitive to block of N-type Ca2+ current (Pineda et al., 1998). In our present work, the specific BK channel blockers ChTX and IBTX are used, and we demonstrate that a transient BK current is present in these cells in mice. Hence, we have extended previous observations in helix neurons (Crest and Gola, 1993), hippocampal pyramidal neurons (Storm, 1987), and bullfrog sympathetic neurons (MacDermott and Weight, 1982) to demonstrate that a fast inactivating BK current operates during action potential and is involved in action potential repolarization in mouse neocortical pyramidal neurons.
The role of this transient outward current is now made clearer in neocortical pyramidal neurons, because we found that this current is eliminated by Cd2+ and Ca2+ removal from extracellular solutions. It is also sensitive to the BK channel blockers ChTX and IBTX but not to the SK channel blocker apamin. Blocking this transient BK current results in broadening of action potentials, indicating that it is involved in action potential repolarization. Pathophysiological roles of this current, in relation to changes in its Ca2+ sources from voltage-gated Ca2+ channels, under conditions such as hypoxia and ischemia would be of interest for future studies.
Footnotes
This work was supported by National Institutes of Health Grant 1P01NS42202. We appreciate the help of Cate Muenker and Hillary Sunamoto for animal handling. Parts of this paper have been published previously in abstract form (Sun and Haddad, 2002).
Correspondence should be addressed to Dr. Gabriel G. Haddad, Department of Pediatrics, Albert Einstein College of Medicine, Rose Kennedy Center, Room 846, 1410 South Pelham Parkway, Bronx, NY 10461. E-mail:ghaddad@aecom.yu.edu.
References
- 1.Armstrong CE, Roberts WM. Rapidly inactivating and non-inactivating calcium-activated potassium currents in frog saccular hair cells. J Physiol (Lond) 2001;536:49–65. doi: 10.1111/j.1469-7793.2001.00049.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Chen W, Zhang JJ, Hu GY, Wu CP. Different mechanisms underlying the repolarization of narrow and wide action potentials in pyramidal cells and interneurons of cat motor cortex. Neuroscience. 1996;73:57–68. doi: 10.1016/0306-4522(96)00010-3. [DOI] [PubMed] [Google Scholar]
- 3.Chow E, Haddad GG. Differential effects of anoxia and glutamate on cultured neocortical neurons. Exp Neurol. 1998;150:52–59. doi: 10.1006/exnr.1997.6764. [DOI] [PubMed] [Google Scholar]
- 4.Crest M, Gola M. Large conductance Ca2+-activated K+ channels are involved in both spike shaping and firing regulation in helix neurons. J Physiol. 1993;465:265–287. doi: 10.1113/jphysiol.1993.sp019676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Foehring RC, Surmeier DJ. Voltage-gated potassium currents in acutely dissociated rat cortical neurons. J Neurophysiol. 1993;70:51–63. doi: 10.1152/jn.1993.70.1.51. [DOI] [PubMed] [Google Scholar]
- 6.Friedman JE, Haddad GG. Major differences in Ca2+ response to anoxia between neonatal and adult rat CA1 neurons: role of Ca2+ and Na+. J Neurosci. 1993;13:63–72. doi: 10.1523/JNEUROSCI.13-01-00063.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hicks GA, Marrion NV. Ca2+-dependent inactivation of large conductance Ca2+-activated K+ (BK) channels in rat hippocampal neurones produced by pore block from an associated particle. J Physiol (Lond) 1998;508 (Pt 3):721–734. doi: 10.1111/j.1469-7793.1998.721bp.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kang J, Huguenard JR, Prince DA. Voltage-gated potassium channels activated during action potentials in layer V neocortical pyramidal neurons. J Neurophysiol. 2000;83:70–80. doi: 10.1152/jn.2000.83.1.70. [DOI] [PubMed] [Google Scholar]
- 9.Keating DJ, Rychkov GY, Roberts ML. Oxygen sensitivity in the sheep adrenal medulla: role of SK channels. Am J Physiol Cell Physiol. 2001;281:C1434–C1441. doi: 10.1152/ajpcell.2001.281.5.C1434. [DOI] [PubMed] [Google Scholar]
- 10.Lancaster B, Nicoll RA. Properties of two calcium-activated hyperpolarizations in rat hippocampal neurons. J Physiol (Lond) 1987;389:187–203. doi: 10.1113/jphysiol.1987.sp016653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Li Z, Ding JP, Kalyanaraman V, Lingle CJ. RINm5f cells express inactivating BK channels whereas HIT cells express noninactivating BK channels. J Neurophysiol. 1999;81:611–624. doi: 10.1152/jn.1999.81.2.611. [DOI] [PubMed] [Google Scholar]
- 12.Liu H, Moczydlowski E, Haddad GG. O2 deprivation inhibits Ca2+-activated K+ channels via cytosolic factors in mice neocortical neurons. J Clin Invest. 1999;104:577–588. doi: 10.1172/JCI7291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lopez-Barneo J, Ortega-Saenz P, Molina A, Franco-Obregon A, Urena J, Castellano A. Oxygen sensing by ion channels. Kidney Int. 1997;51:454–461. doi: 10.1038/ki.1997.61. [DOI] [PubMed] [Google Scholar]
- 14.Lorenzon NM, Foehring RC. Characterization of pharmacologically identified voltage-gated calcium channel currents in acutely isolated rat neocortical neurons. II. Postnatal development. J Neurophysiol. 1995;73:1443–1451. doi: 10.1152/jn.1995.73.4.1443. [DOI] [PubMed] [Google Scholar]
- 15.MacDermott AB, Weight FF. Action potential repolarization may involve a transient, Ca2+-sensitive outward current in a vertebrate neuron. Nature. 1982;296:746–749. doi: 10.1038/300185a0. [DOI] [PubMed] [Google Scholar]
- 16.Marrion NV, Tavalin SJ. Selective activation of Ca2+-activated K+ channels by co-localized Ca2+ channels in hippocampal neurons. Nature. 1998;395:900–905. doi: 10.1038/27674. [DOI] [PubMed] [Google Scholar]
- 17.Pineda JC, Waters RS, Foehring RC. Specificity in the interaction of HVA Ca2+ channel types with Ca2+-dependent AHPs and firing behavior in neocortical pyramidal neurons. J Neurophysiol. 1998;79:2522–2534. doi: 10.1152/jn.1998.79.5.2522. [DOI] [PubMed] [Google Scholar]
- 18.Pineda JC, Galarraga E, Foehring RC. Different Ca2+ source for slow AHP in completely adapting and repetitive firing pyramidal neurons. NeuroReport. 1999;10:1951–1956. doi: 10.1097/00001756-199906230-00029. [DOI] [PubMed] [Google Scholar]
- 19.Sah P, Clements JD. Photolytic manipulation of [Ca2+]i reveals slow kinetics of potassium channels underlying the afterhyperpolarization in hippocampal pyramidal neurons. J Neurosci. 1999;19:3657–3664. doi: 10.1523/JNEUROSCI.19-10-03657.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Sah P, McLachlan EM. Potassium currents contributing to action potential repolarization and the afterhyperpolarization in rat vagal motoneurons. J Neurophysiol. 1992;68:1834–1841. doi: 10.1152/jn.1992.68.5.1834. [DOI] [PubMed] [Google Scholar]
- 21.Salkoff L. Drosophila mutants reveal two components of fast outward current. Nature. 1983;302:249–251. doi: 10.1038/302249a0. [DOI] [PubMed] [Google Scholar]
- 22.Schwindt PC, Spain WJ, Crill WE. Effects of intracellular calcium chelation on voltage-dependent and calcium-dependent currents in cat neocortical neurons. Neuroscience. 1992;47:571–578. doi: 10.1016/0306-4522(92)90166-y. [DOI] [PubMed] [Google Scholar]
- 23.Solaro CR, Lingle CJ. Trypsin-sensitive, rapid inactivation of a calcium-activated potassium channel. Science. 1992;257:1694–1698. doi: 10.1126/science.1529355. [DOI] [PubMed] [Google Scholar]
- 24.Spain WJ, Schwindt PC, Crill WE. Two transient potassium currents in layer V pyramidal neurons from cat sensorimotor cortex. J Physiol (Lond) 1991;434:591–607. doi: 10.1113/jphysiol.1991.sp018488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Stewart A, Foehring RC. Calcium currents in retrogradely labeled pyramidal cells from rat sensorimotor cortex. J Neurophysiol. 2000;83:2349–2354. doi: 10.1152/jn.2000.83.4.2349. [DOI] [PubMed] [Google Scholar]
- 26.Storm JF. Action potential repolarization and a fast after-hyperpolarization in rat hippocampal pyramidal cells. J Physiol (Lond) 1987;385:733–755. doi: 10.1113/jphysiol.1987.sp016517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Summers BA, Overholt JL, Prabhakar NR. Augmentation of L-type calcium current by hypoxia in rabbit carotid body glomus cells: evidence for a PKC-sensitive pathway. J Neurophysiol. 2000;84:1636–1644. doi: 10.1152/jn.2000.84.3.1636. [DOI] [PubMed] [Google Scholar]
- 28.Sun X, Haddad GG. Calcium influx via L- and N-type calcium channels activates a fast transient BK current in mouse neocortical pyramidal neurons. Soc Neurosci Abstr. 2002;28:340.10. doi: 10.1523/JNEUROSCI.23-09-03639.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Sun X, Barnes S, Baldridge WH. Adenosine inhibits calcium channel currents via A1 receptors on salamander retinal ganglion cells in a mini-slice preparation. J Neurochem. 2002;81:550–556. doi: 10.1046/j.1471-4159.2002.00832.x. [DOI] [PubMed] [Google Scholar]
- 30.Velumian AA, Carlen PL. Differential control of three after-hyperpolarizations in rat hippocampal neurons by intracellular calcium buffering. J Physiol (Lond) 1999;517:201–216. doi: 10.1111/j.1469-7793.1999.0201z.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Wallner M, Meera P, Toro L. Molecular basis of fast inactivation in voltage and Ca2+-activated K+ channels: a transmembrane beta-subunit homolog. Proc Natl Acad Sci USA. 1999;96:4137–4142. doi: 10.1073/pnas.96.7.4137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Xia XM, Ding JP, Lingle CJ. Molecular basis for the inactivation of Ca2+- and voltage-dependent BK channels in adrenal chromaffin cells and rat insulinoma tumor cells. J Neurosci. 1999;19:5255–5264. doi: 10.1523/JNEUROSCI.19-13-05255.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Xia XM, Ding JP, Zeng XH, Duan KL, Lingle CJ. Rectification and rapid activation at low Ca2+ of Ca2+-activated, voltage-dependent BK currents: consequences of rapid inactivation by a novel beta subunit. J Neurosci. 2000;20:4890–4903. doi: 10.1523/JNEUROSCI.20-13-04890.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]