Abstract
The receptor subtypes that mediate the effects of neuropeptide Y (NPY) on food intake have not been clearly defined. The NPY Y4 receptor has been identified recently as a potential mediator of the regulation of food intake. The purpose of the present study was to characterize the central site of action of the Y4 receptor using a combination of neuroanatomical and physiological approaches. Using immunocytochemistry, Y4-like immunoreactivity was found to be colocalized with orexin cell bodies in the lateral hypothalamic area (LHA) and orexin fibers throughout the brain.In situ hybridization confirmed the expression of Y4 mRNA in orexin neurons. To determine the functional interaction between Y4 receptors and orexin neurons, we examined the effects of rat pancreatic polypeptide (rPP), a Y4-selective ligand, or NPY, a nonselective ligand, administered directly into the LHA on the stimulation of food and water intake and c-Fos expression. Both rPP and NPY significantly increased food and water intake when they were administered into the LHA, although NPY was a more potent stimulator of food intake. Furthermore, both NPY and rPP significantly stimulated c-Fos expression in the LHA. However, whereas rPP stimulated c-Fos expression in orexin neurons, NPY did not. Neither rPP nor NPY stimulated c-Fos in melanin-concentrating hormone neurons, but both activated neurons of an unknown phenotype in the LHA. These results suggest that a functional Y4 receptor is expressed on orexin neurons and that these neurons are activated in response to a ligand with high affinity for the Y4 receptor (rPP). Although these data suggest a role for central Y4 receptors, the endogenous ligand for this receptor has yet to be clearly established.
Keywords: neuropeptide Y, pancreatic polypeptide, rat, c-Fos, orexin, feeding
Introduction
Neuropeptide Y (NPY) is a member of the pancreatic polypeptide hormone family that also includes pancreatic polypeptide (PP) and peptide YY (PYY). NPY is most notably characterized as a potent orexigenic agent, and exogenous NPY administration causes a robust increase in food intake and body weight (Morton and Schwartz, 2001; Williams et al., 2001). In addition, NPY is also an important modulator of the neuroendocrine reproductive axis (Kalra and Horvath, 1998). It has been suggested that NPY may act as an integrating factor between signals for energy balance and neuroendocrine reproductive function (Smith and Grove, 2002); however, the neuronal circuitry responsible for these effects remains to be elucidated.
Five receptor subtypes have been characterized that bind NPY, PYY, and PP with different affinities; these subtypes have been distinguished pharmacologically as Y1, Y2, Y4, Y5, and Y6. They all belong to the superfamily of G-protein-coupled receptors and cause the inhibition of adenylate cyclase and, in some instances, the activation of the phospholipase C/protein kinase C pathway (Selbie et al., 1995; Parker et al., 1998). Currently, the Y1 and Y5 receptor subtypes are implicated in mediating the effects of NPY on food intake based on a variety of studies using partially selective pharmacological tools and antisense approaches. Both Y1 and Y5 antagonists can inhibit nocturnal feeding as well as fasting and NPY-induced food intake (Crisioine et al., 1998; Kanatani et al., 1999), and administration of a selective Y5 agonist significantly stimulates food intake (Cabrele et al., 2000). Likewise, antisense oligonucleotides directed against both the Y1 and the Y5 receptors reduce NPY-induced food intake (Lopez-Valpuesta et al., 1996; Schaffhauser et al., 1997; Campbell et al., 2001). However, knock-out studies have provided some unexpected results. Mice lacking the Y1 or Y5 receptor gene exhibit late-onset obesity and even hyperphagia rather than the expected lean/hypophagic phenotype (Kushi et al., 1998; Marsh et al., 1998; Pedrazzini et al., 1998). In contrast, Y4 receptor knock-out mice do display the expected phenotype, because they are leaner and hypophagic (Sainsbury et al., 2002).
The Y4 receptor has been thought of primarily as a peripheral receptor, because it binds with a much higher affinity to peripherally produced PP than to NPY (Bard et al., 1995). However, both Y4 receptor mRNA and binding within the brain have been reported, including hypothalamic areas involved in the regulation of food intake (Parker and Herzog, 1999; Dumont and Quirion, 2000). Also, administration of 1229U91, a Y4 agonist, or rat PP (rPP) into the third ventricle stimulates luteinizing hormone secretion, providing additional evidence for centrally active Y4 receptors (Jain et al., 1999; Raposinho et al., 2000). Therefore, although it is unclear whether the lean phenotype of the Y4 knock-out mouse is attributable to peripheral or central mechanisms, it is reasonable to hypothesize that hypothalamic Y4 receptors may play a role in the central regulation of food intake. The present study was designed using both anatomical and physiological approaches to investigate Y4 receptor expression in the hypothalamus and the potential role of Y4 receptors in the central regulation of food intake.
Materials and Methods
Animals and tissue
Adult rats (Simonsen, Gilroy, CA) were maintained under a 12 hr light/dark cycle (lights on at 7:00 A.M.) and constant temperature (23 ± 2°C). Food and water were available ad libitum. All animal procedures were approved by the Oregon National Primate Research Center Institutional Animal Care and Use Committee.
For immunocytochemical studies, male and female rats were killed under pentobarbital anesthesia by cardiac infusion with ice-cold PBS followed by ice-cold paraformaldehyde in NaPO4 buffer, pH 7.4. The brains and peripheral tissues then were removed, saturated in 25% sucrose, frozen in cooled isopentane, and stored at −80°C until sectioned on a microtome (25 μm). For Western blot analysis, in situ hybridization, and reverse transcriptase (RT)-PCR studies, animals were killed by rapid decapitation. Brains and various peripheral tissues were removed, immediately frozen on crushed dry ice, and stored at −80°C until use. Tissue collected for RT-PCR was stored in RNAlater (Ambion, Austin, TX).
Characterization of Y4 receptor antibody
Production of the Y4 receptor antibody. Rabbit polyclonal antisera were raised against a peptide derived from a sequence unique to the rat NPY Y4 protein (FVTTRQKEKSNVTN). The Y4 receptor antibody was produced by J. M. H. ffrench-Mullen. The Y4 synthetic peptide was cross-linked with the keyhole limpet carrier and injected into rabbits, and sera were collected from rabbits after five and six boosters.
Single-label immunocytochemistry. These methods have been described previously (Campbell et al., 2001). Briefly, tissue sections (n = 3–4, both male and female rats were used in each experiment; one series of sections from a 1:3 series) were removed from cryoprotectant and washed in 0.05 m potassium PBS (KPBS) followed by preincubation in blocking buffer (KPBS plus 0.4% Triton X-100 plus 2% normal donkey serum) for 30 min at room temperature. Sections were then incubated in the Y4 primary antibody (1:10,000) in blocking buffer for 48 hr at 4°C. After washes in KPBS, tissue was incubated for 1 hr in biotinylated donkey anti-rabbit antibody (1:600; Jackson ImmunoResearch, West Grove, PA), and then washed and incubated in avidin–biotin solution (Vectastain; Vector Laboratories, Burlingame, CA) for 1 hr. Y4-immunoreactive (-IR) signal was further amplified using a commercial kit (tyramide signal amplification-indirect kit; NEN Life Sciences Products, Boston, MA) and was visualized with a chromagen label, 3,3′-diaminobenzidine enhanced with nickel chloride, or a fluorescent label FITC conjugated to streptavidin (1:1000; Jackson ImmunoResearch).
To confirm antibody specificity, serial sections were stained either with the primary antibody alone, as described above, or with the primary antibody preadsorbed with control peptide (Phoenix Pharmaceuticals, Belmont, CA) for 24 hr at 4°C. Preadsorption with control peptide eliminated specific antibody staining (data not shown).
Western blot analysis. Western blot analysis was used to investigate the central and peripheral distribution of the Y4 protein. Tissue was homogenized in ice-cold hypotonic buffer and then centrifuged at 1500 rpm (200 × g) for 5 min at 4°C. The resulting pellet containing the membrane-bound protein was resuspended in sample buffer (50 mm Tris, 150 mm NaCl, 1 mm EDTA, 0.1% SDS, pH 7.4), frozen, and stored at −80°C. For Western blot analysis, samples were thawed on ice and 25 μg protein samples were further solubilized, separated by SDS-PAGE using PAGEr Gold precast gels (BioWhittaker Molecular Applications, Rockland, ME), and then electroblotted onto polyvinylidene difluoride membranes (Boehringer Mannheim, Indianapolis, IN). NPY Y4 protein was detected with the NPY Y4 antibody (1:5000) and visualized using enhanced chemiluminescence (SuperSignal; Pierce, Rockford, IL) by exposure to sheet film (XOMAT; Eastman Kodak, Rochester, NY).
Production of NPY Y4 cDNA clone and RT-PCR. For generation of the rat NPY Y4 cDNA clone, total RNA was collected and PCR was performed as described previously (Brogan et al., 2000). The following primers were used: NPY Y4 forward, 5′-GACTTGCTACCCATCCTCATA-3′; NPY Y4 reverse, 5′-ATCACCACCGCCTCATCTACA-3′. The PCR product was cloned into pGemT (Promega, Madison, WI) and sequenced to confirm its identity. For the qualitative assessment of Y4 mRNA in central and peripheral tissues, RT-PCR was performed on total mRNA using the primers cited above.
Double-label immunofluorescence
To characterize the phenotype of cells expressing the NPY Y4 receptor, double-label immunofluorescence (IF) and confocal microscopic analysis were performed. The polyclonal orexin antibody made in the goat (Santa Cruz Biotechnology, Santa Cruz, CA) was used at a concentration of 1:10,000 in mixture with the Y4 primary antibody used at a concentration of 1:10,000 as described above. In the double-label experiment, Y4-IR signal was visualized using FITC conjugated to streptavidin (1:1000; Jackson ImmunoResearch) after biotinylated tyramide enhancement, and orexin was visualized with a donkey anti-goat antibody conjugated to an Alexa fluorophore (Alexa 546; Molecular Probes, Eugene, OR).
Double-label IF was also used to investigate the phenotype of neurons expressing the NPY Y1 receptor subtype using a previously characterized Y1 antibody (Li et al., 1999, 2000). In these experiments, the polyclonal orexin antibody was used in a mixture with the polyclonal Y1 antibody made in the rabbit (Center for Ulcer Research and Education/Gastroenteric Biology Center, Antibody/RIA Core, University of California Los Angeles, Los Angeles, CA; used at a concentration of 1:7000). The Y1-IR signal was visualized with a donkey anti-rabbit antibody conjugated to FITC after biotinylated tyramide enhancement, and orexin was visualized with a donkey anti-goat antibody conjugated to an Alexa fluorophore (Alexa 546; Molecular Probes).
Confocal laser microscopy
Confocal laser microscopy was used to analyze the double-label IF images as described previously (Campbell et al., 2001). The Leica (Nussloch, Germany) TSC SP confocal system, consisting of a Leica RBE inverted microscope, an argon laser producing light at 488 nm (for visualization of FITC), and a krypton laser producing light at 568 nm (for visualizing rhodamine or Alexa 546), was used to scan the images. Various objectives (25×, numerical aperture 0.75; 40×, numerical aperture 1.25) were used to scan and capture images. For each experiment, fluorophore signals were checked individually for bleed-through to the apposing detector. All bleed-through was eliminated by adjusting laser intensity and detector window width. To assess colocalization of two signals, a series of continuous optical sections, at 0.5 μm intervals along the z-axis of the tissue section, was scanned for each fluorescent signal. The signals were obtained for each fluorophore on one series of optical sections and were stored separately as a series of 512 × 512 pixel images. The stacks of individual optical slices (0.5 μm resolution) were analyzed using the MetaMorph Imaging System (Universal Imaging Corporation, West Chester, PA) to determine colocalization. The confocal images are presented as projections of a stack of optical images. The brightness and contrast of the images were adjusted in Photoshop (Adobe Systems, San Jose, CA) to match microscope visualization.
Double-label immunocytochemistry/in situhybridization for orexin and Y4
For additional confirmation of Y4 receptor expression in orexin neurons, double-labeling of Y4 mRNA and orexin-IR was performed. Initially, staining for orexin-IR was performed on floating sections throughout the hypothalamus. Sections were incubated in the orexin primary antibody made in the goat (Santa Cruz Biotechnology) and used at a 1:5000 concentration with 2 U/ml RNase inhibitor in KPBS plus 0.4% Triton X-100 for 48 hr at 4°C. After washes in KPBS, tissue was incubated for 1 hr in biotinylated donkey anti-rabbit antibody (1:600; Jackson ImmunoResearch) with 10 U/ml RNase inhibitor and then washed and incubated in avidin–biotin solution (Vectastain) for 1 hr. Orexin-IR was visualized with the chromagen 3,3′-diaminobenzidine enhanced with nickel chloride. Sections then were rinsed and fixed in 4% paraformaldehyde, treated with a fresh solution containing 0.25% acetic anhydride in 0.1 mtriethanolamine, pH 8.0, and then rinsed in 2× SSC.
The NPY Y4 cRNA probe was transcribed from a 400 bp cDNA in which 100% of the UTP was 33P labeled. After the rinses above, tissue was incubated in sense or antisense probe (20 million counts per milliliter of labeled Y4 probe) overnight at 37°C. After incubation, tissue was washed in SSC that increased in stringency and RNase A at 37°C, and then 0.1× SSC at 55°C. After washing in 0.9% saline, tissue sections were mounted onto slides and air dried overnight. Slides then were quickly dehydrated through a series of alcohols and exposed to autoradiographic films for 1 d. Slides then were dipped in Kodak NBT2 emulsion diluted 1:1 in 600 mm ammonium acetate and stored in light-tight boxes for 5 d. Slides were developed and counter-stained with cresyl violet, and the distribution of silver grains was analyzed by dark-field microscopy.
Effects of rPP or NPY injections into the lateral hypothalamic area
Cannulation of the lateral hypothalamic area and peptide injections. To provide evidence for a functional interaction between Y4 receptors and orexin neurons, rPP, a Y4 selective ligand, or NPY was injected directly into the lateral hypothalamic area (LHA) using doses that have been shown to induce a maximum stimulation of food intake. c-Fos expression was used to assess whether there was a differential response to the drugs with respect to neuronal populations activated in the LHA, with a specific focus on orexin or melanin-concentrating hormone (MCH) cell populations.
Male rats (weighing 250–275 gm), under isofluorane gas anesthesia, were implanted with permanent, stainless-steel guide cannulas (28 gauge, 8.9 mm long; Plastics One, Wallingford, CT), stereotaxically placed into the LHA according to the atlas of Paxinos and Watson (1998)[stereotaxic coordinates: anteroposterior (AP), −3.3; dorsoventral, −8.9; and lateral, 1.2; relative to bregma]. The incisor bar was set at −5 mm to ensure that the animals' skulls were level. The guide cannula was secured to the skull by acrylic dental cement and anchored with small stainless-steel screws. The guide cannula was plugged with an internal dummy cannula (28 gauge; Plastics One) until the time of injections. Only those rats displaying normal food intake and weight gain after surgery were used.
At 5–6 d after cannulation of the LHA, freely moving rats were remotely injected at 9:00 A.M. with rPP [2 μg (0.45 nmol) per rat in 0.5 μl], NPY [1 μg (0.23 nmol) per rat in 0.5 μl], or vehicle (0.5 μl sterile artificial CSF) using a Hamilton syringe and polyethylene tubing filled with saline (Grove and Smith, 1998). The injector cannula (33 gauge) extended 2 mm beyond the guide cannula. The doses of rPP and NPY were extrapolated from the literature as causing a maximal stimulation of food intake when given intracerebroventricularly (rPP) (Nakajima et al., 1994; Katsuura et al., 2002) or directly into specific brain nuclei (NPY) (Currie and Coscina, 1995; Brown et al., 2000). Immediately after injections, preweighed rat chow and water were placed in cages. The amount of food and water consumed was measured 1 and 3 hr after injection, and behavior was monitored. At 12:00 P.M., 3 hr after injection, animals were killed and their brains were processed for immunocytochemistry.
Immunohistochemistry for c-Fos and LHA neuronal phenotypes.Animals were killed under pentobarbital anesthesia by cardiac infusion with ice-cold PBS followed by ice-cold paraformaldehyde (4%) in NaPO4 buffer, pH 7.4. The brains then were removed, postfixed overnight in 4% paraformaldehyde at 4°C, and saturated in 25% sucrose. Subsequently, brains were frozen on crushed dry ice and stored at −80°C until being sectioned on a microtome (25 μm) in a one-in-three series.
Triple-label immunohistochemistry was used to label c-Fos, orexin, and MCH to quantify c-Fos-positive staining within these two populations of neurons in the LHA. All staining procedures were performed in one experiment, and a complete set of sections was used that represented a one-in-three series encompassing the entire rostrocaudal extent of the LHA. Briefly, floating tissue sections were removed from cryoprotectant and washed in 0.05 m KPBS. Tissue was incubated in 1% H2O2 in methanol for 10 min to eliminate endogenous peroxidase activity, followed by washes in KPBS and incubation in blocking buffer for 20 min (KPBS plus 0.4% Triton X-100 plus 2% normal donkey serum) to reduce background. Sections were then incubated in a mixture of primary antibodies, including the c-Fos antibody made in the rabbit (SC-52; Santa Cruz Biotechnology) (used at concentration of 1:25,000), the orexin antibody made in the goat (Santa Cruz Biotechnology) (used at a concentration of 1:10,000), and the MCH antibody made in the chicken (Bachem, Torrance, CA) (used at a concentration of 1:3000) for 48 hr at 4°C. After washes in KPBS, tissue was incubated for 1 hr in biotinylated donkey anti-rabbit antibody (1:600; Jackson ImmunoResearch) and then washed and incubated in avidin–biotin solution (Vectastain) for 1 hr. c-Fos-IR was visualized with the chromagen 3,3′-diaminobenzidine enhanced with nickel chloride. The sections were then rinsed and incubated with fluorescent secondary antibodies. Orexin was visualized with FITC conjugated to a donkey anti-goat antibody (1:200; Jackson ImmunoResearch), and MCH was visualized with rhodamine conjugated to a donkey anti-chicken antibody (1:200; Jackson ImmunoResearch). Sections were mounted onto subbed slides, cover-slipped with glycerol, and sealed.
Quantification of c-Fos staining in orexin and MCH neurons.Only those animals with proper guide cannula placement in the dorsal extent of the LHA were used in this study, thus placing the injector cannula, which extended 2 mm beyond the guide cannula, near the level of the fornix. Cannula placement was easily identifiable by the cannula tract and area of tissue damage surrounding the cannula (see Fig. 7). Blind to the treatment group, orexin and MCH neurons were counted as positive or negative for c-Fos on both ipsilateral and contralateral sides, relative to the injection site, using a Zeiss(Oberkochen, Germany) Axioskop 2 fluorescence microscope with an adjustable xenon light intensity (AttoArc 2; Zeiss). Orexin- and MCH-positive neurons were counted from the rostral-to-caudal extent of the LHA. On average, 680 orexin and 1226 MCH neurons were counted per rat brain, and there were 8–10 animals per group. Images from selected sections were captured using a Leica TSC SP confocal system.
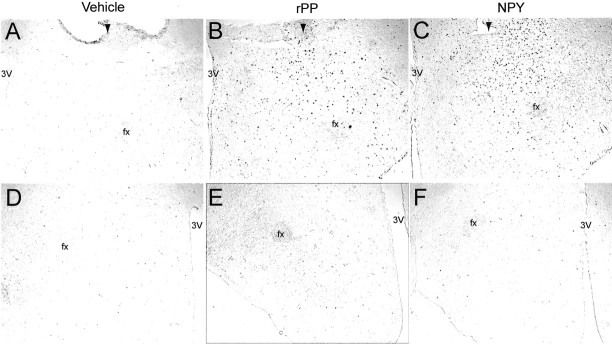
Fig. 7.
Single-label c-Fos expression on ipsilateral and contralateral sides of intra-LHA injections. Representative photomicrographs show c-Fos expression in the LHA on the ipsilateral (A–C) and contralateral (D–F) side of the injection site from animals receiving vehicle (A, D), rPP (B, E), or NPY (C, F). Theblack arrowhead indicates the ventral extent of the guide cannula. The injector cannula extended 2 mm beyond the guide cannula. The images were taken at the approximate AP coordinates −3.5 according to the rat brain atlas of Paxinos and Watson (1998).fx, Fornix; 3V, third ventricle.
Statistical analysis. The food and water intake, cell count, and c-Fos data were analyzed by one-way ANOVA using Prizm software (GraphPad Software, San Diego, CA) followed post hoc by Newman–Keuls multiple-comparison test; p < 0.05 was considered significant.
Results
Y4 distribution
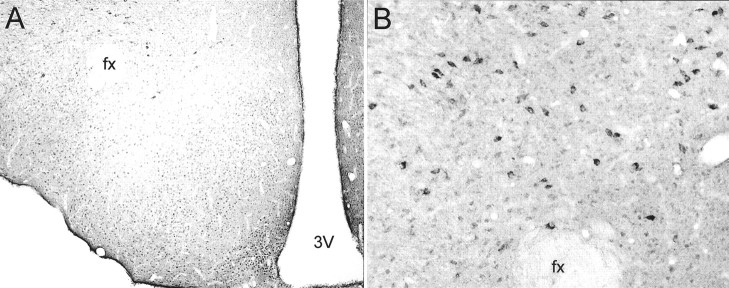
Analysis of the single-label distribution of chromagen Y4-IR demonstrated low levels of staining throughout the hypothalamus (Fig.1A). Similar distribution patterns were observed in male and female rats. The majority of staining appeared to be in small, round cells. Unexpectedly, the most intensely labeled Y4-IR cell bodies were found in the larger cells of the LHA (Fig. 1A,B). Densely labeled Y4-IR cells were apparent throughout the rostral-to-caudal extent of the LHA.
Fig. 1.
Single-label hypothalamic distribution of NPY Y4-IR. A, Low-power photomicrograph example of low levels of Y4-IR expressed in small, round cells throughout the hypothalamus. A greater density of Y4-IR cells can be seen in the arcuate nucleus, but the most intensely labeled cells were found in the LHA (A). B, Higher-power view of Y4-IR cells in the LHA. fx, Fornix; 3v, third ventricle.
To provide additional characterization of the Y4 antibody and Y4 distribution in both central and peripheral structures, we examined the correlation of Y4 mRNA and protein expression using PCR, Western blot analysis, and immunocytochemistry. Centrally, Y4 mRNA was present in both the hypothalamus and the cortex (Fig.2A). In peripheral structures, all of the areas investigated, including the kidney, liver, duodenum, large intestine, colon, stomach, and lung, expressed Y4 mRNA (Fig. 2B). By Western blot analysis, the Y4 antibody identified Y4 protein within membrane extracts of both the hypothalamus and the cortex (Fig. 2C) and all of the peripheral tissues tested (Fig. 2D). Finally, by immunocytochemistry, Y4-IR was observed in both the hypothalamus and the cortex. The Y4 antibody also identified Y4-IR in peripheral tissues. Figure2G shows an example of IF labeling in the duodenum in which Y4-IR was observed within goblet cells and along the basal lamina of intestinal villi. Collectively, these data suggest that the Y4 antibody recognizes the membrane-bound Y4 receptor in both peripheral and central tissues.
Fig. 2.
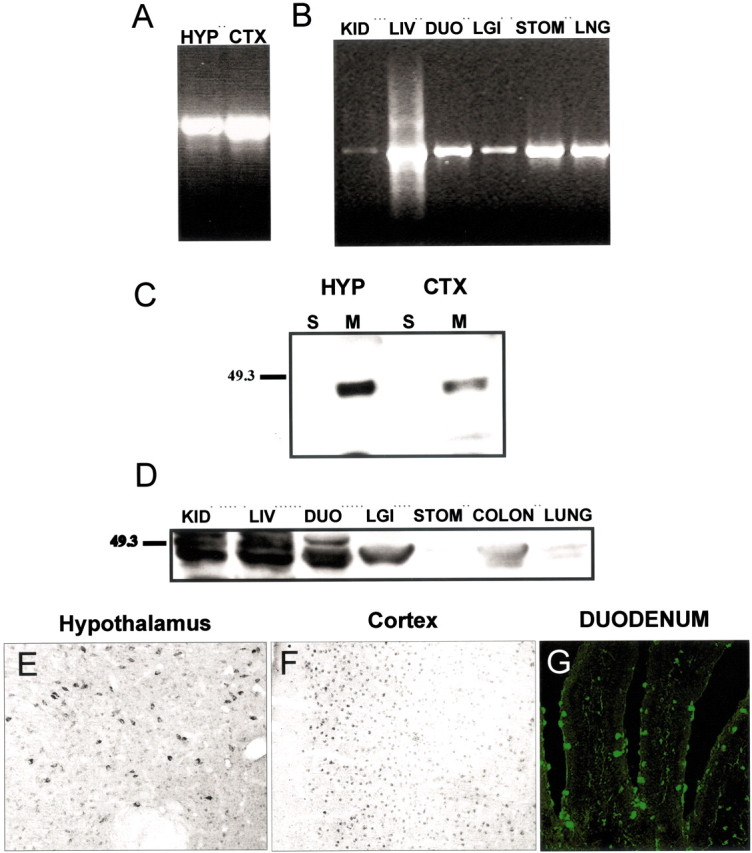
Correlation of Y4 mRNA and protein in central and peripheral tissues. Y4 mRNA was found by RT-PCR in the hypothalamus (HYP) and cortex (CTX) (A) and throughout peripheral structures (B), including kidney (KID), liver (LIV), duodenum (DUO), large intestine (LGI), stomach (STO), and lung (LNG). Western blot analysis shows Y4 protein in membrane (M) fractions of the hypothalamus and cortex (C) and in all peripheral structures (D), but not in the soluble (S) fraction. Y4-IR was identified in cells of the hypothalamus (E), the cortex (F), and throughout peripheral tissues; the duodenum is shown as an example (G).
Y4 expression in orexin neurons
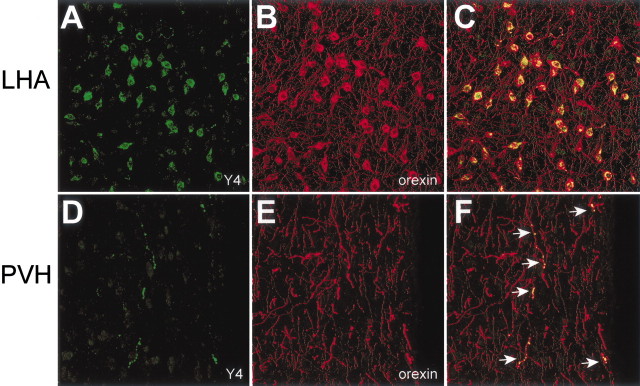
Double-label IF experiments indicated that a majority of neurons displaying Y4-IR observed in the LHA (Fig.3A, green) were colocalized with orexin, whose cell bodies are also entirely restricted to this region (Fig. 3B, red). Although there was dense overlap of these proteins in cell bodies, as shown inyellow, there were only a few orexin fibers that colocalized the Y4 receptor in this region (Fig. 3C). Y4-IR was also observed in sparse fibers throughout the hypothalamus (Fig.3D, green), and all of these fibers were identified as colocalized with orexin (Fig. 3F,yellow). Double-labeling of Y4 mRNA by in situhybridization and orexin by chromagen immunocytochemistry provided additional confirmation for the expression of Y4 receptors on orexin neurons. Silver grain clusters are clearly colocalized with orexin neurons in the LHA (Fig.4A–D). However, there are also a number of non-orexin neurons in the LHA that express Y4 mRNA.
Fig. 3.
NPY Y4-IR colocalizes with orexin in the LHA. Double-label IF for Y4 (A, D, green) and orexin (B, E, red) is shown in LHA cell bodies (A–C) and a representative fiber in the paraventricular nucleus of the hypothalamus (PVH;D–F). Their colocalization is indicated inyellow (C, F). Arrowsin F indicate orexin/Y4 double-label fibers.
Fig. 4.
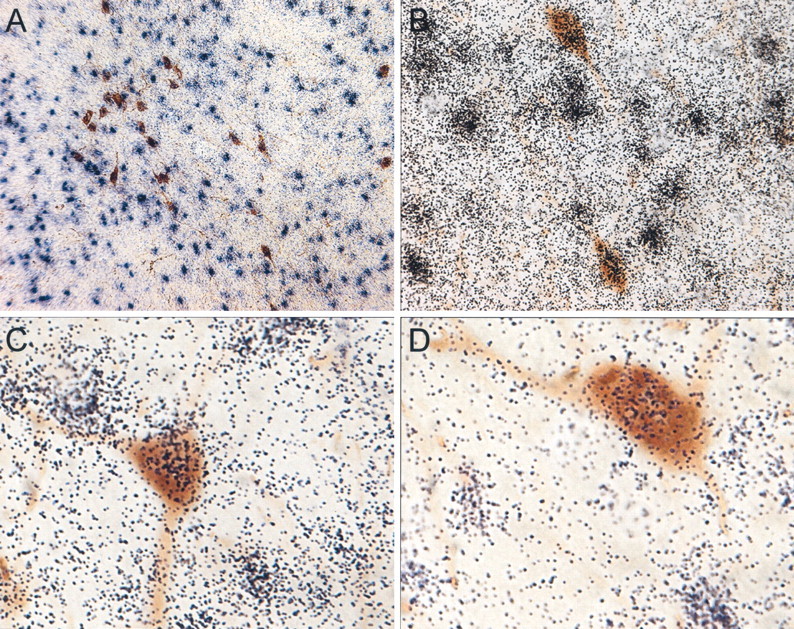
Y4 mRNA is coexpressed in orexin neurons. Representative photomicrographs show Y4 mRNA at low (A) and high (B–D) power identified by silver grain clusters (purple/black) throughout the LHA and double-labeled with orexin neurons (brown).
Y1 receptor subtype expression in the LHA
An additional double-label IF study investigated the expression pattern of the NPY Y1 receptor subtype within the LHA and in relation to orexin neurons. Y1-IR (Fig.5A–D, green) was observed on numerous fibers and some cell bodies (yellow arrows) within the LHA. Many Y1-IR fibers surrounded orexin neurons (red) and appeared to make close contact (yellow), as indicated by white arrows (Fig.5A–D); however, Y1-IR was not colocalized with orexin neurons or fibers.
Fig. 5.
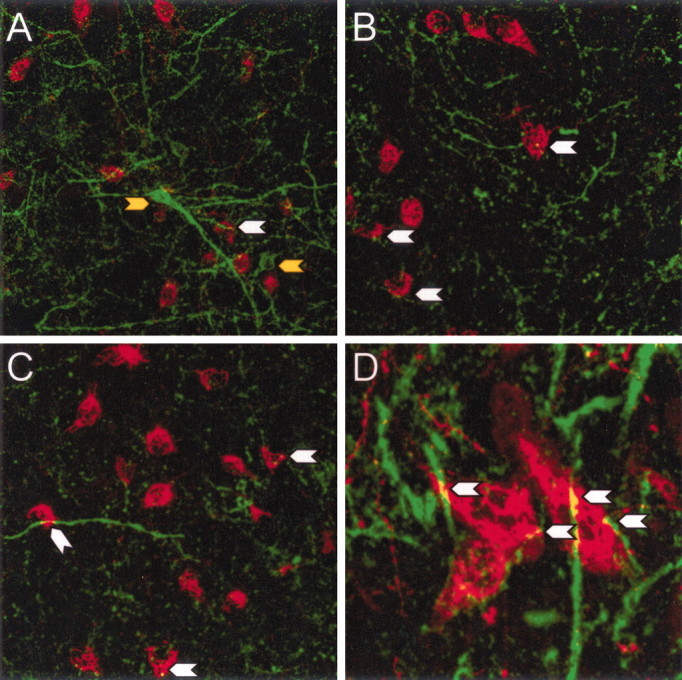
NPY Y1-IR on fibers and cells of an unknown phenotype in the LHA. Double-label IF for Y1 (green) and orexin (red) in the LHA (A–D) shows Y1-IR on non-orexin cells (yellow arrow) and on fibers surrounding orexin neurons. Y1-IR is not colocalized with orexin neurons, but Y1-IR fibers appear to make close contact with orexin neurons (white arrows).
Stimulatory effects of rPP and NPY injected into the LHA
Food and water intake
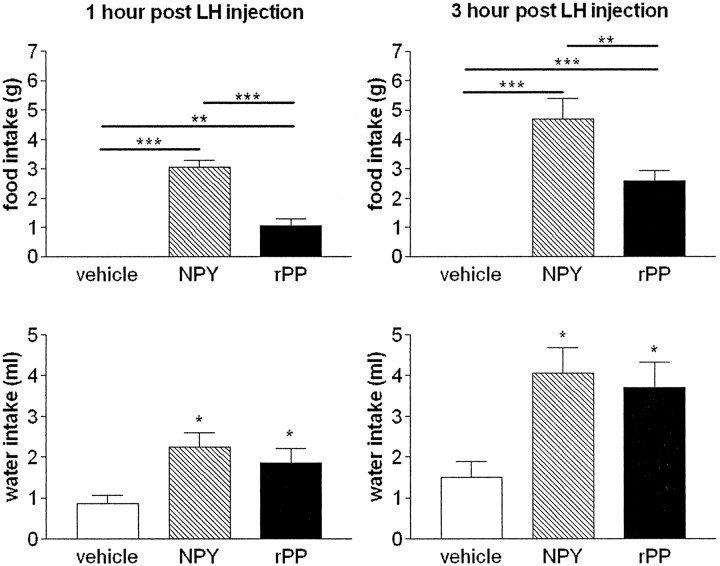
Both NPY and rPP caused a significant increase in food and water intake in male rats (Fig. 6). NPY elicited a robust feeding response, with 3.06 ± 0.23 and 4.69 ± 0.71 gm of cumulative food intake at 1 and 3 hr time points. rPP caused a more moderate food-intake response, with 1.05 ± 0.23 and 2.60 ± 0.33 gm of cumulative food intake at 1 and 3 hr time points. Food intake stimulated by rPP was significantly greater than vehicle (p < 0.01 at 1 hr;p < 0.001 at 3 hr) and significantly less than that stimulated by NPY (p < 0.001 at 1 hr;p < 0.01 at 3 hr) (Fig. 6A,B). Vehicle-injected controls did not consume any food during this period, as would be expected during daylight hours. Although controls consumed a small amount of water, NPY and rPP were equally effective at significantly stimulating water intake at both 1 and 3 hr time points (p < 0.05) (Fig. 6C,D).
Fig. 6.
Intra-LHA administration of NPY and rPP stimulates food and water intake. Mean ± SEM cumulative food intake and water intake 1 and 3 hr after intra-LHA injection of vehicle (open bars), NPY (hatched bars), or rPP (filled bars); n = 10–11 per group. NPY caused a significant increase in food intake compared with both vehicle- and rPP-injected animals (**p < 0.01; ***p < 0.001). rPP significantly increased food intake compared with vehicle (***p < 0.001). Both NPY and rPP significantly increased water intake compared with vehicle (*p < 0.05).
Induction of c-Fos expression
c-Fos patterns were examined 3 hr after injections of either rPP or NPY. It is well established that c-Fos expression remains stable for >3 hr after a stimulus (Hoffman et al., 1992). The results showed that c-Fos expression was specifically induced only on the ipsilateral side to the injection site (Fig.7A–C). Both rPP and NPY stimulated c-Fos expression on the ipsilateral side, whereas vehicle injections caused a low level of c-Fos induction directly adjacent to the injection site (Fig. 7A–C). On the contralateral side to the injection site, extremely low levels of c-Fos were observed in all groups (Fig. 7D–F). The lack of c-Fos activation on the contralateral side provides strong evidence that c-Fos expression on the treatment side was specific to the drugs and not secondary to the induction of physiological responses, such as ingestion of food. Any c-Fos expression resulting from food intake would be expected to occur bilaterally. However, we cannot eliminate the possibility that neurons are being activated by a combination of the stimulus from the peptides and the ingested meal. Furthermore, confinement of c-Fos expression to the treatment side indicates that the peptides were not diffusing to the third ventricle and causing responses via activation of some site remote from the LHA.
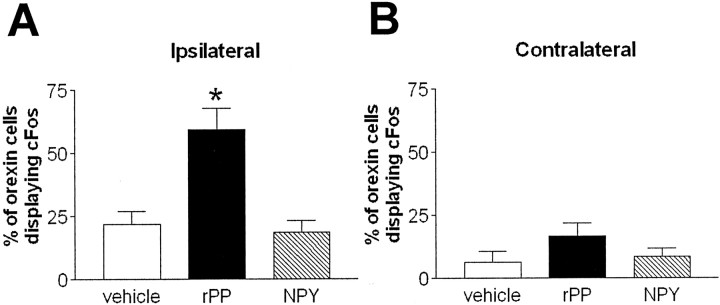
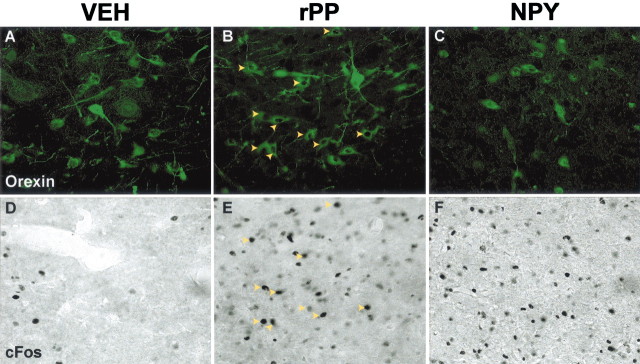
Three hours after rPP injection, there was a significant induction of c-Fos expression in orexin neurons on the ipsilateral side (59.3 ± 8.5%; p < 0.001 compared with vehicle treatment) (Figs. 8,9). In contrast, NPY did not significantly activate c-Fos expression in orexin cells when compared with vehicle-treated animals (18.5 ± 4.7 and 21.7 ± 5.4%, respectively) (Figs. 8, 9), although there were numerous c-Fos-IR cells of unknown phenotype in the area. On the contralateral side, very few orexin neurons displayed c-Fos expression in any of the treatment groups (Fig. 8B).
Fig. 8.
Intra-LHA rPP, but not NPY, induces a robust increase in c-Fos expression in orexin neurons. Mean ± SEM percentage of orexin cells displaying c-Fos 3 hr after intra-LHA injection of vehicle (open bars), NPY (hatched bars), or rPP (filled bars);n = 8–10 per group. rPP injection caused a robust induction of c-Fos expression ipsilateral to the injection site compared with vehicle and NPY (A) (*p < 0.05). Very low levels of c-Fos expression were seen in orexin neurons contralateral to the injection site (B) in all treatment groups.
Fig. 9.
Orexin neurons and c-Fos expression after intra-LHA injections of vehicle, rPP, or NPY. Representative images show orexin-IR (green,A–C) and c-Fos-IR (black,D–F) in identical areas in the LHA from animals injected with vehicle (VEH; A, D), rPP (B, E), or NPY (C, F). Theyellow arrowheads in B represent c-Fos colocalization with orexin-IR neurons and in E represent the same cells showing only c-Fos-IR. There was no c-Fos colocalization in the orexin cells shown in A (vehicle treated) or inC (NPY treated). The images were taken at the approximate AP coordinates −3.6 according the rat brain atlas ofPaxinos and Watson (1998).
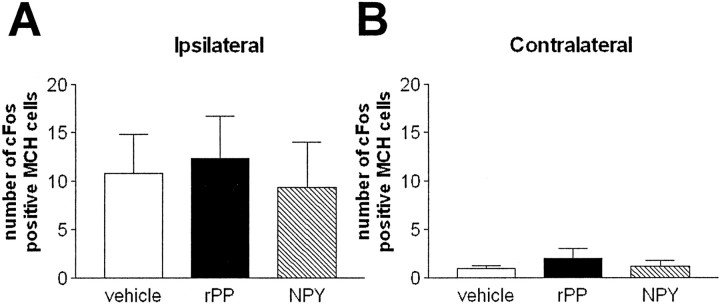
The number of c-Fos-positive MCH neurons was extremely low and was not significantly different among the treatment groups on either the ipsilateral or the contralateral side (Fig.10A,B). The number of ipsilateral MCH cells displaying c-Fos represents only a small percentage (∼1%) of the number of MCH neurons analyzed per animal: 10.80 ± 4.0, 12.29 ± 4.43, and 9.33 ± 4.68 from vehicle-, rPP-, and NPY-injected animals, respectively.
Fig. 10.
MCH neurons do not express c-Fos in response to intra-LHA rPP or NPY. Mean ± SEM number of MCH cells displaying c-Fos 3 hr after intra-LHA injection of vehicle (open bars), NPY (hatched bars), or rPP (filed bars); n = 8–10 per group. Very few MCH neurons on either side of the injection site displayed c-Fos (A, B).
Discussion
The present study demonstrates for the first time that orexin neurons in the LHA coexpress the NPY Y4 receptor subtype and are activated by a Y4-specific ligand, rPP. Using a newly characterized NPY Y4 antibody, a low level of Y4-IR was observed in small, round cells throughout the hypothalamus. However, the most intensely labeled Y4-IR cell bodies in the hypothalamus were observed in the LHA (Fig.1A,B). The overall pattern of Y4-IR was in agreement with the previously reported in situ hybridization pattern of Y4 mRNA (Parker and Herzog, 1999) and the pattern of Y4 receptor binding (Dumont and Quirion, 2000). Additional characterization of the Y4 antibody demonstrated a complete correlation between Y4 mRNA, as detected by RT-PCR, and protein expression, as detected by Western blot analysis and immunocytochemistry, in both central and peripheral regions (Fig. 2A–G), supporting the specificity and sensitivity of this antibody.
Interestingly, immunocytochemistry the most intense Y4-IR observed in the LHA was colocalized with orexin neurons (Fig. 3). The colocalization of Y4 mRNA expression in orexin neurons supports this finding (Fig. 4); however, Y4 mRNA was also expressed in a number of non-orexin-expressing cells. Orexins have been found to regulate arousal states, to influence ingestion (both drinking and feeding), and to play a role in the modulation of neuroendocrine processes, including luteinizing hormone release and thermoregulation (for review, see Beuckmann and Yanagisawa, 2002; Sakurai, 2002; Sutcliffe and de Lecea, 2002). Central injection of orexin A stimulates food intake in satiated rats (Dube et al., 1999; Haynes et al., 1999), and orexin mRNA is upregulated in response to fasting (Sakurai et al., 1998). However, although orexin A increases daytime feeding, overall 24 hr food intake is not increased, and repeated injections have no effect on body weight (Yamanaka et al., 1999). Therefore, orexin may be important in short-term feeding episodes or in the initiation of feeding, but it is not necessarily implicated in obesity. Alternatively, numerous studies have shown that orexin is a key signal in arousal-associated behavior (Sutcliffe et al., 2002). Thus, the orexin system might be responsible for the complex coordination of behavioral and physiological responses to an animal's nutritional needs. Determining the neuroanatomical pathways that influence orexin activity will aid in the understanding of the relative importance of orexin in these physiological roles.
Although the neuroanatomical results presented above strongly support a direct role of the Y4 receptor on orexin neurons, this needed to be directly tested. Injection of rPP, a selective Y4 receptor ligand, directly into the LHA of freely moving rats elicited a robust dipsogenic response and a moderate food-intake response (Fig. 5). These results resemble previously reported responses after orexin administration. Orexin A, administered intracerebroventricularly, increases water intake at a level comparable with angiotensin II, and deprivation of water upregulates orexin mRNA (Kunii et al., 1999). As mentioned above, orexin has been implicated in the regulation of food intake; however, orexin is a much less potent orexigenic agent than NPY (Edwards et al., 1999). Similar to other studies (Stanley et al., 1993), the present study demonstrated a robust increase in both food and water intake after NPY injection into the LHA.
The differences between the abilities of NPY and the rPP to induce food and water intake suggest that these peptides may be activating different neurocircuits in eliciting ingestive behavior. One factor that may contribute to the different responses is the greatly different affinities of NPY or rPP for the various NPY receptors. Whereas NPY has a relatively high affinity for the Y1, Y2, and Y5 receptors, rPP has a relatively low affinity for these receptors but has a 200-fold greater affinity for the Y4 receptor (Bard et al., 1995; Schober et al., 2000). The differences in c-Fos activation of specific neuronal populations within the LHA also support the idea that rPP and NPY may be activating different neurocircuits in the induction of food intake. Although both rPP and NPY elicited significant food intake and stimulated a robust induction of c-Fos expression in the area of the injection site, only rPP significantly induced c-Fos in orexin neurons. Although these findings alone do not demonstrate whether the effects of rPP are direct on orexin neurons, if they are coupled with the neuroanatomical evidence of Y4 protein and mRNA colocalization in orexin neurons, they strongly support this conclusion. However, we cannot eliminate the possibility that the stimulation of food intake by rPP occurs via actions on other populations of neurons in the LHA that then activate orexin neurons. Although rPP did not stimulate c-Fos expression in MCH neurons, it did activate a population of LHA neurons distinct from orexin. Furthermore, whereas only low levels of Y4-IR were observed in non-orexin neurons within the LHA, an abundance of Y4 mRNA was detected in non-orexin cells throughout this area (Fig. 4). These data suggest either that the Y4 antibody may not be sensitive enough to detect low levels of Y4 protein or that Y4 mRNA in these cells may not be translated at a high rate under the present conditions. Additional studies are necessary to identify the phenotypes of this population of LHA neurons, and it remains to be determined whether rPP stimulates food and water intake directly via orexin neurons or via this unidentified population.
Surprisingly, NPY injection into the LHA did not significantly stimulate c-Fos in either orexin or MCH neurons, although c-Fos expression was stimulated in the LHA in a population of neurons with an unknown phenotype. This is in contrast to previous studies showing that NPY injection into the third ventricle causes the induction of c-Fos in a small percentage of orexin neurons (Niimi et al., 2001). This discrepancy is most likely attributable to the mode of administration. Injections into the third ventricle may activate a number of other systems that project to the LHA. Thus, the induction of c-Fos observed may be a second- or third-order event. The lack of significant c-Fos induction in MCH and orexin neurons was particularly unexpected considering the abundance of NPY terminals that are in contact with orexin and MCH neurons (Broberger et al., 1998; Horvath et al., 1999). Reciprocal connections of orexin with NPY and α-melanin-stimulating hormone neurons in the hypothalamic arcuate nucleus also have been described previously (Elias et al., 1998), and it has been suggested that these are key circuits in the maintenance of food-intake signals. However, NPY projections to the LHA may be working through other NPY receptor subtypes, either presynaptically or postsynaptically on different phenotypes of neurons. The lack of c-Fos induction in orexin and MCH neurons is supported by the lack of Y1 receptor expression on orexin (Fig. 4) and MCH neurons (our unpublished observations). Y1-IR was found on fibers and cells of an unknown phenotype dispersed throughout the LHA. Additionally, Y1-IR fibers were found in close contact with orexin and MCH neurons, suggesting presynaptic NPY effects via the Y1 receptor. Future studies will attempt to identify the phenotypes of neurons expressing the Y1 receptor. The Y5 receptor also has been identified in the LHA (Campbell et al., 2001), although the phenotype of cells expressing the Y5 receptor subtype also remains to be determined. In addition to MCH and orexin, the LHA is known to contain large numbers of glucose-receptive neurons (Bernardis and Bellinger, 1996) of unknown phenotypes, as well as the orexigenic neuropeptide galanin (Melander et al., 1986). What is clear from these studies is that orexin and MCH neurons do not express the Y1 receptor and do not express c-Fos in response to NPY administered directly to the LHA.
Although previous reports clearly illustrate the activation of central Y4-like receptors (Jain et al., 1999; Raposinho et al., 2000), and the present study demonstrates physiological and c-Fos responses to the peripheral Y4 agonist rPP, an endogenous ligand for the centrally expressed Y4 receptor has not yet been definitively characterized. PP, the only known endogenous high-affinity ligand for the Y4 receptor, is produced in the islet cells of the endocrine pancreas. Peripherally produced PP, however, does not appear to readily cross the blood–brain barrier. Early studies identified PP-like IR in the brain (Loren et al., 1979; Lundberg et al., 1980; Olschowka et al., 1981; Inui et al., 1985); however, later studies were not able to detect PP-like peptide in the brain (Miyazaki and Funakoshi, 1988), leading to the view that the antibodies used in previous studies were probably cross-reacting with NPY or some other substance. Furthermore, Pieribone et al. (1992) also showed the lack of PP mRNA in the brain, usingin situ hybridization with oligo probes specific for rPP. In contrast, recent preliminary studies in our laboratory have identified rPP mRNA in the hypothalamus and brainstem using RT-PCR (our unpublished observations), raising the possibility that PP or a PP-like ligand for the Y4 receptor may be present in the brain. Additional studies are required to confirm the presence of the Y4 ligand mRNA and protein in the brain as well as the anatomical localization of the PP-expressing cells. Armed with this neuroanatomical information, it then will be possible to study the importance of the central rPP–Y4 system in the regulation of ingestive behavior.
Footnotes
This work was supported by National Institutes of Health Grants HD-14643 and RR-00163. We thank Dr. Anda Cornea for assistance with confocal microscopy.
Correspondence should be addressed to Dr. Kevin L. Grove, Oregon National Primate Research Center, Oregon Health and Science University, Beaverton, OR 97006. E-mail: grovek@ohsu.edu.
References
- 1.Bard JA, Walker MW, Branchek TA, Weinshank RL. Cloning and functional expression of a human Y4 subtype receptor for pancreatic polypeptide, neuropeptide Y, and peptide YY. J Biol Chem. 1995;270:26762–26765. doi: 10.1074/jbc.270.45.26762. [DOI] [PubMed] [Google Scholar]
- 2.Bernardis LL, Bellinger LL. The lateral hypothalamic area revisited: ingestive behavior. Neurosci Biobehav Rev. 1996;20:189–287. doi: 10.1016/0149-7634(95)00015-1. [DOI] [PubMed] [Google Scholar]
- 3.Beuckmann CT, Yanagisawa M. Orexins: from neuropeptides to energy homeostatis and sleep/wake regulation. J Mol Med. 2002;80:329–342. doi: 10.1007/s00109-002-0322-x. [DOI] [PubMed] [Google Scholar]
- 4.Broberger C, De Lecea L, Sutcliffe JG, Hokfelt T. Hypocretin/orexin- and melanin-concentrating hormone-expressing cells form distinct populations in the rodent lateral hypothalamus: relationship to the neuropeptide Y and agouti gene-related protein systems. J Comp Neurol. 1998;402:460–474. [PubMed] [Google Scholar]
- 5.Brogan RS, Grove KL, Smith MS. Differential regulation of leptin receptor but not orexin in the hypothalamus of the lactating rat. J Neuroendocrinol. 2000;12:1077–1086. doi: 10.1046/j.1365-2826.2000.00559.x. [DOI] [PubMed] [Google Scholar]
- 6.Brown CM, Coscina DV, Fletcher PJ. The rewarding properties of neuropeptide Y in perifornical hypothalamus vs. nucleus accumbens. Peptides. 2000;21:1279–1287. doi: 10.1016/s0196-9781(00)00270-9. [DOI] [PubMed] [Google Scholar]
- 7.Cabrele C, Langer M, Bader R, Wieland HA, Doods HN, Zerbe O, Beck-Sickinger AG. The first selective agonist for the neuropeptide YY5 receptor increases food intake in rats. J Biol Chem. 2000;275:36043–36048. doi: 10.1074/jbc.M000626200. [DOI] [PubMed] [Google Scholar]
- 8.Campbell RE, ffrench-Mullen JM, Cowley MA, Smith MS, Grove KL. Hypothalamic circuitry of neuropeptide Y regulation of neuroendocrine function and food intake via the Y5 receptor subtype. Neuroendocrinology. 2001;74:106–119. doi: 10.1159/000054676. [DOI] [PubMed] [Google Scholar]
- 9.Crisioine L, Gigollier P, Batzl-Hartman C, Rueger H, Stricker-Krongrad A, Wyss P, Brunner L, Whitebread S, Yamaguchi Y, Gerald C, Heurich RO, Walker MW, Chiesi M, Schilling W, Hofbauer KG, Levens N. Food intake in free-feeding and energy deprived lean rats is mediated by the neuropeptide Y Y5 receptor. J Clin Invest. 1998;102:2136–2145. doi: 10.1172/JCI4188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Currie PJ, Coscina DV. Dissociated feeding and hypothermia effects of neuropeptide Y in the paraventricular and perifornical hypothalamus. Peptides. 1995;16:599–604. doi: 10.1016/0196-9781(95)00020-k. [DOI] [PubMed] [Google Scholar]
- 11.Dube MG, Kalra SP, Kalra PS. Food intake elicited by central administration of orexins/hypocretins: identification of hypothalamic sites of action. Brain Res. 1999;842:473–477. doi: 10.1016/s0006-8993(99)01824-7. [DOI] [PubMed] [Google Scholar]
- 12.Dumont Y, Quirion R. [(125)I]-GR231118: a high affinity radioligand to investigate neuropeptide Y, Y(1), and Y(4) receptors. Br J Pharmacol. 2000;129:37–46. doi: 10.1038/sj.bjp.0702983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Edwards CM, Abusnana S, Sunter D, Murphy KG, Ghatei MA, Bloom SR. The effect of the orexins on food intake: comparison with neuropeptide Y, melanin-concentrating hormone and galanin. J Endocrinol. 1999;160:7–12. doi: 10.1677/joe.0.160r007. [DOI] [PubMed] [Google Scholar]
- 14.Elias CF, Saper CB, Maratos-Flier E, Tritos NA, Lee C, Kelly J, Tatro JB, Hoffman GE, Ollmann MM, Barsh GS, Sakurai T, Yanagisawa M, Elmquist JK. Chemically defined projections linking the mediobasal hypothalamus and the lateral hypothalamic area. J Comp Neurol. 1998;402:442–459. [PubMed] [Google Scholar]
- 15.Grove KL, Smith MS. Resistance of the hippocampus in the lactating rat to N-methyl-d-aspartate (NMDA)-mediated excitation is not due to a nonfunctional receptor system. Brain Res. 1998;814:157–163. doi: 10.1016/s0006-8993(98)01069-5. [DOI] [PubMed] [Google Scholar]
- 16.Haynes AC, Jackson B, Overend P, Buckingham RE, Wilson S, Tadayyon M, Arch JRS. Effects of single and chronic intracerebroventricular administration of the orexins on feeding in the rat. Peptides. 1999;20:1099–1105. doi: 10.1016/s0196-9781(99)00105-9. [DOI] [PubMed] [Google Scholar]
- 17.Hoffman GE, Smith MS, Fitsimmons MD. Detecting steroidal effects on immediate early gene expression in the hypothalamus. Neuroprotocols. 1992;1:52–66. [Google Scholar]
- 18.Horvath TL, Diano S, van den Pol AN. Synaptic interaction between hypocretin (orexin) and neuropeptide Y cells in the rodent and primate hypothalamus: a novel circuit implicated in metabolic and endocrine regulations. J Neurosci. 1999;19:1072–1087. doi: 10.1523/JNEUROSCI.19-03-01072.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Inui A, Mizuno N, Ooya M, Suenaga K, Morioka H, Ogawa T, Ishida M, Baba S. Cross-reactivities of neuropeptide Y and peptide YY with pancreatic polypeptide antisera: evidence for the existence of pancreatic polypeptide in the brain. Brain Res. 1985;330:386–389. doi: 10.1016/0006-8993(85)90704-8. [DOI] [PubMed] [Google Scholar]
- 20.Jain MR, Pu S, Kalra PS, Kalra SP. Evidence that stimulation of two modalities of pituitary luteinizing hormone release in ovarian steroid-primed ovariectomized rats may involve neuropeptide Y, Y1, and Y4 receptors. Endocrinology. 1999;140:5171–5177. doi: 10.1210/endo.140.11.7107. [DOI] [PubMed] [Google Scholar]
- 21.Kalra SP, Horvath TL. Neuroendocrine interactions between galanin, opioids, and neuropeptide Y in the control of reproduction and appetite. Ann NY Acad Sci. 1998;863:236–240. doi: 10.1111/j.1749-6632.1998.tb10698.x. [DOI] [PubMed] [Google Scholar]
- 22.Kanatani A, Kanno T, Ishihara A, Hata M, Sakuraba A, Tanaka T, Tsuchiya Y, Mase T, Fukuroda T, Fukami T, Ihara M. The novel neuropeptide Y Y(1) receptor antagonist J-104870: a potent feeding suppressant with oral bioavailability. Biochem Biophys Res Commun. 1999;266:88–91. doi: 10.1006/bbrc.1999.1750. [DOI] [PubMed] [Google Scholar]
- 23.Katsuura G, Asakawa A, Inui A. Roles of pancreatic polypeptide in regulation of food intake. Peptides. 2002;23:323–329. doi: 10.1016/s0196-9781(01)00604-0. [DOI] [PubMed] [Google Scholar]
- 24.Kunii K, Yamanaka A, Nambu T, Matsuzaki I, Goto K, Sakurai T. Orexins/hypocretins regulate drinking behaviour. Brain Res. 1999;842:256–261. doi: 10.1016/s0006-8993(99)01884-3. [DOI] [PubMed] [Google Scholar]
- 25.Kushi A, Sasai H, Koizumi H, Takeda N, Yokoyama M, Nakamura M. Obesity and mild hyperinsulinemia found in neuropeptide Y-Y1 receptor-deficient mice. Proc Natl Acad Sci USA. 1998;95:15659–15664. doi: 10.1073/pnas.95.26.15659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Li C, Chen P, Smith MS. Morphological evidence for direct interaction between arcuate nucleus neuropeptide Y (NPY) neurons and gonadotropin-releasing hormone neurons and the possible involvement of NPY Y1 receptors. Endocrinology. 1999;140:5382–5390. doi: 10.1210/endo.140.11.7093. [DOI] [PubMed] [Google Scholar]
- 27.Li C, Chen P, Smith MS. Corticotropin releasing hormone (CRH) neurons in the paraventricular nucleus (PVH) are direct targets for neuropeptide Y (NPY) neurons in the arcuate nucleus (ARH): an anterograde tracing study. Brain Res. 2000;854:122–129. doi: 10.1016/s0006-8993(99)02324-0. [DOI] [PubMed] [Google Scholar]
- 28.Lopez-Valpuesta FJ, Nyce JW, Myers RD. NPY-Y1 receptor antisense injected centrally in rats causes hyperthermia and feeding. NeuroReport. 1996;7:2781–2784. doi: 10.1097/00001756-199611040-00075. [DOI] [PubMed] [Google Scholar]
- 29.Loren I, Alumets J, Hakanson R, Sundler F. Immunoreactive pancreatic polypeptide (PP) occurs in the central and peripheral nervous system: preliminary immunocytochemical observations. Cell Tissue Res. 1979;200:179–186. doi: 10.1007/BF00236410. [DOI] [PubMed] [Google Scholar]
- 30.Lundberg JM, Hokfelt T, Anggard A, Kimmel J, Goldstein M, Markey K. Coexistence of an avian pancreatic polypeptide (APP) immunoreactive substance and catecholamine in some peripheral and central neurons. Acta Physiol Scand. 1980;110:107–109. doi: 10.1111/j.1748-1716.1980.tb06638.x. [DOI] [PubMed] [Google Scholar]
- 31.Marsh DJ, Hollopeter G, Kafer KE, Palmiter RD. Role of the Y5 neuropeptide Y receptor in feeding and obesity. Nat Med. 1998;4:718–721. doi: 10.1038/nm0698-718. [DOI] [PubMed] [Google Scholar]
- 32.Melander T, Hokfelt T, Rokaeus A. Distribution of galaninlike immunoreactivity in the rat central nervous system. J Comp Neurol. 1986;248:475–517. doi: 10.1002/cne.902480404. [DOI] [PubMed] [Google Scholar]
- 33.Miyazaki K, Funakoshi A. A distribution of pancreatic polypeptide-like immunoreactivity in rat tissues. Regul Pept. 1988;21:37–43. doi: 10.1016/0167-0115(88)90089-4. [DOI] [PubMed] [Google Scholar]
- 34.Morton GJ, Schwartz MW. The NPY/AgRP neuron and energy homeostasis. Int J Obes Relat Metab Disord. 2001;5:56–62. doi: 10.1038/sj.ijo.0801915. [DOI] [PubMed] [Google Scholar]
- 35.Nakajima M, Inui A, Teranishi A, Miura M, Hirosue Y, Okita M, Himori N, Baba S, Kasuga M. Effects of pancreatic polypeptide family peptides on feeding and learning behavior in mice. J Pharmacol Exp Ther. 1994;268:1010–1014. [PubMed] [Google Scholar]
- 36.Niimi M, Sato M, Taminato T. Neuropeptide Y in central control of feeding and interactions with orexin and leptin. Endocrine. 2001;14:269–273. doi: 10.1385/ENDO:14:2:269. [DOI] [PubMed] [Google Scholar]
- 37.Olschowka JA, O'Donohue TL, Jacobowitz DM. The distribution of bovine pancreatic polypeptide-like immunoreactive neurons in rat brain. Peptides. 1981;2:309–331. doi: 10.1016/s0196-9781(81)80125-8. [DOI] [PubMed] [Google Scholar]
- 38.Parker RMC, Herzog H. Regional distribution of Y-receptor subtype mRNAs in rat brain. Eur J Neurosci. 1999;11:1431–1448. doi: 10.1046/j.1460-9568.1999.00553.x. [DOI] [PubMed] [Google Scholar]
- 39.Parker SL, Parker MS, Sweatman T, Crowley WF. Characterization of G protein and phospholipase C-coupled agonist binding to the Y1 neuropeptide Y receptor in the rat brain: sensitivity to G protein activators and inhibitors and to inhibitors of phospholipase C. J Pharmacol Exp Ther. 1998;286:382–391. [PubMed] [Google Scholar]
- 40.Paxinos G, Watson C. The rat brain in stereotaxic coordinates. Academic; New York: 1998. [DOI] [PubMed] [Google Scholar]
- 41.Pedrazzini T, Seydoux J, Kunstner P, Aubert J-F, Grouzmann E, Beermann F, Brunner HR. Cardiovascular response, feeding behavior and locomotor activity in mice lacking the NPY Y1 receptor. Nat Med. 1998;4:722–726. doi: 10.1038/nm0698-722. [DOI] [PubMed] [Google Scholar]
- 42.Pieribone VA, Brodin L, Friberg K, Dahlstrand J, Soderberg C, Larhammar D, Hokfelt T. Differential expression of mRNAs for neuropeptide Y-related peptides in rat nervous tissue: possible evolutionary conservation. J Neurosci. 1992;12:3361–3371. doi: 10.1523/JNEUROSCI.12-09-03361.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Raposinho PD, Broqua P, Hayward A, Akinsanya K, Galyean R, Schteingart C, Junien J, Aubert ML. Stimulation of the gonadotropic axis by the neuropeptide Y receptor Y1 antagonist/Y4 agonist 1229U91 in the male rat. Neuroendocrinology. 2000;71:2–7. doi: 10.1159/000054514. [DOI] [PubMed] [Google Scholar]
- 44.Sainsbury A, Schwarzer C, Couzens M, Jenkins A, Oakes SR, Ormandy CJ, Herzog H. Y4 receptor knockout rescues fertility in ob/ob mice. Genes Dev. 2002;16:1077–1088. doi: 10.1101/gad.979102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Sakurai T. Roles of orexin in regulation of feeding and wakefulness. NeuroReport. 2002;13:987–995. doi: 10.1097/00001756-200206120-00001. [DOI] [PubMed] [Google Scholar]
- 46.Sakurai T, Amemiya A, Ishii M, Matsuzaki I, Chemelli RM, Tanaka H, Williams SC, Richardson JA, Kozlowski GP, Wilson S, Arch JR, Buckingham RE, Haynes AC, Carr SA, Annan RS, McNulty DE, Liu WS, Terrett JA, Elshourbagy NA, Bergsma DJ. Orexins and orexin receptors: a family of hypothalamic neuropeptides and G protein-coupled receptors that regulate feeding behavior. Cell. 1998;92:573–585. doi: 10.1016/s0092-8674(00)80949-6. [DOI] [PubMed] [Google Scholar]
- 47.Schaffhauser AO, Stricker-Krongrad A, Brunner L, Cumin F, Gerald C, Whitebread S, Criscione L, Hofbauer KG. Inhibition of food intake by neuropeptide Y Y5 receptor antisense oligodeoxynucleotides. Diabetes. 1997;46:1792–1798. doi: 10.2337/diab.46.11.1792. [DOI] [PubMed] [Google Scholar]
- 48.Schober DA, Gackenheimer SL, Heiman ML, Gehlert DR. Pharmacological characterization of 125I–1229U91 binding to Y1 and Y4 neuropeptide Y/peptide YY receptors. J Pharmacol Exp Ther 2000. 2000;293:275–280. [PubMed] [Google Scholar]
- 49.Selbie LA, Darby K, Schmitz-Peiffer C, Browne CL, Herzog H, Shine J, Biden TJ. Synergistic interaction of Y1-neuropeptide Y and alpha 1β-adrenergic receptors in the regulation of phospholipase C, protein kinase C, and arachidonic acid production. J Biol Chem. 1995;270:11789–11796. doi: 10.1074/jbc.270.20.11789. [DOI] [PubMed] [Google Scholar]
- 50.Smith MS, Grove KL. Integration of the regulation of reproductive function and energy balance: lactation as a model. Front Neuroendocrinol. 2002;23:225–256. doi: 10.1016/s0091-3022(02)00002-x. [DOI] [PubMed] [Google Scholar]
- 51.Stanley BG, Magdalin W, Seirafi A, Thomas WJ, Leibowitz SF. The perifornical area: the major focus of (a) patchily distributed hypothalamic neuropeptide Y-sensitive feeding system(s). Brain Res. 1993;604:304–317. doi: 10.1016/0006-8993(93)90382-w. [DOI] [PubMed] [Google Scholar]
- 52.Sutcliffe JG, de Lecea L. The hypocretins: setting the arousal threshold. Nat Rev Neurosci. 2002;3:339–349. doi: 10.1038/nrn808. [DOI] [PubMed] [Google Scholar]
- 53.Williams G, Bing C, Cai XJ, Harrold JA, King PJ, Liu XH. The hypothalamus and the control of energy homeostasis: different circuits, different purposes. Physiol Behav. 2001;74:683–701. doi: 10.1016/s0031-9384(01)00612-6. [DOI] [PubMed] [Google Scholar]
- 54.Yamanaka A, Sakurai T, Katsumoto T, Yanagisawa M, Goto K. Chronic intracerebroventricular administration of orexin-A to rats increases food intake in daytime, but has no effect on body weight. Brain Res. 1999;849:248–252. doi: 10.1016/s0006-8993(99)01905-8. [DOI] [PubMed] [Google Scholar]