Abstract
Background
The hypertensive disorders of pregnancy include pre‐eclampsia, gestational hypertension, chronic hypertension, and undefined hypertension. Pre‐eclampsia is considerably more prevalent in low‐income than in high‐income countries. One possible explanation for this discrepancy is dietary differences, particularly calcium deficiency. Calcium supplementation in the second half of pregnancy reduces the serious consequences of pre‐eclampsia, but has limited effect on the overall risk of pre‐eclampsia. It is important to establish whether calcium supplementation before, and in early pregnancy (before 20 weeks' gestation) has added benefit. Such evidence could count towards justification of population‐level interventions to improve dietary calcium intake, including fortification of staple foods with calcium, especially in contexts where dietary calcium intake is known to be inadequate. This is an update of a review first published in 2017.
Objectives
To determine the effect of calcium supplementation, given before or early in pregnancy and for at least the first half of pregnancy, on pre‐eclampsia and other hypertensive disorders, maternal morbidity and mortality, and fetal and neonatal outcomes.
Search methods
We searched the Cochrane Pregnancy and Childbirth Trials Register (31 July 2018), PubMed (13 July 2018), ClinicalTrials.gov, the WHO International Clinical Trials Registry Platform (ICTRP; 31 July 2018), and reference lists of retrieved studies.
Selection criteria
Eligible studies were randomised controlled trials (RCT) of calcium supplementation, including women not yet pregnant, or women in early pregnancy. Cluster‐RCTs, quasi‐RCTs, and trials published as abstracts were eligible, but we did not identify any.
Data collection and analysis
Two review authors independently assessed trials for inclusion and risk of bias, extracted data, and checked them for accuracy. They assessed the quality of the evidence for key outcomes using the GRADE approach.
Main results
Calcium versus placebo
We included one study (1355 women), which took place across multiple hospital sites in Argentina, South Africa, and Zimbabwe. Most analyses were conducted only on 633 women from this group who were known to have conceived, or on 579 who reached 20 weeks' gestation; the trial was at moderate risk of bias due to high attrition rates pre‐conception. Non‐pregnant women with previous pre‐eclampsia received either calcium 500 mg daily or placebo, from enrolment until 20 weeks' gestation. All participants received calcium 1.5 g daily from 20 weeks until birth.
Primary outcomes: calcium supplementation commencing before conception may make little or no difference to the risk of pre‐eclampsia (69/296 versus 82/283, risk ratio (RR) 0.80, 95% confidence interval (CI) 0.61 to 1.06; low‐quality evidence). For pre‐eclampsia or pregnancy loss or stillbirth (or both) at any gestational age, calcium may slightly reduce the risk of this composite outcome, however the 95% CI met the line of no effect (RR 0.82, 95% CI 0.66 to 1.00; low‐quality evidence). Supplementation may make little or no difference to the severe maternal morbidity and mortality index (RR 0.93, 95% CI 0.68 to 1.26; low‐quality evidence), pregnancy loss or stillbirth at any gestational age (RR 0.83, 95% CI 0.61 to 1,14; low‐quality evidence), or caesarean section (RR 1.11, 95% CI 0.96 to 1,28; low‐quality evidence).
Calcium supplementation may make little or no difference to the following secondary outcomes: birthweight < 2500 g (RR 1.00, 95% CI 0.76 to 1.30; low‐quality evidence), preterm birth < 37 weeks (RR 0.90, 95% CI 0.74 to 1.10), early preterm birth < 32 weeks (RR 0.79, 95% CI 0.56 to 1.12), and pregnancy loss, stillbirth or neonatal death before discharge (RR 0.82, 95% CI 0.61 to 1.10; low‐quality evidence), no conception, gestational hypertension, gestational proteinuria, severe gestational hypertension, severe pre‐eclampsia, severe pre‐eclamptic complications index. There was no clear evidence on whether or not calcium might make a difference to perinatal death, or neonatal intensive care unit admission for > 24h, or both (RR 1.11, 95% CI 0.77 to 1.60; low‐quality evidence).
It is unclear what impact calcium supplementation has on Apgar score < 7 at five minutes (RR 0.43, 95% CI 0.15 to 1.21; very low‐quality evidence), stillbirth, early onset pre‐eclampsia, eclampsia, placental abruption, intensive care unit admission > 24 hours, maternal death, hospital stay > 7 days from birth, and pregnancy loss before 20 weeks' gestation.
Authors' conclusions
The single included study suggested that calcium supplementation before and early in pregnancy may reduce the risk of women experiencing the composite outcome pre‐eclampsia or pregnancy loss at any gestational age, but the results are inconclusive for all other outcomes for women and babies. Therefore, current evidence neither supports nor refutes the routine use of calcium supplementation before conception and in early pregnancy.
To determine the overall benefit of calcium supplementation commenced before or in early pregnancy, the effects found in the study of calcium supplementation limited to the first half of pregnancy need to be added to the known benefits of calcium supplementation in the second half of pregnancy.
Further research is needed to confirm whether initiating calcium supplementation pre‐ or in early pregnancy is associated with a reduction in adverse pregnancy outcomes for mother and baby. Research could also address the acceptability of the intervention to women, which was not covered by this review update.
Plain language summary
Extra calcium in tablets before pregnancy, or in early pregnancy, for preventing high blood pressure complications of pregnancy
What is the issue?
We wanted to know if giving women calcium as a supplement before pregnancy or during early pregnancy would help pregnant women avoid pre‐eclampsia, high blood pressure, and other serious health problems during pregnancy. We wanted to know if these supplements could improve pregnancy, and birth for the baby, as well.
Why is this important?
Women can develop high blood pressure and have protein in their urine after the twentieth week of pregnancy; this condition is known as pre‐eclampsia. Many women, particularly those in low‐income countries, do not have enough calcium in their diets. Giving these women extra calcium during the second half of pregnancy has been shown to reduce their risk of having high blood pressure and protein in the urine, and other related problems, such as convulsions, stroke, blood‐clotting problems, fluid in the lungs, kidney failure, or even death. It is important to know if taking extra calcium before pregnancy and in early pregnancy can reduce the number of women who develop blood pressure problems during pregnancy, and related complications.
We searched for randomised controlled studies that looked at the effect of taking extra calcium before or early in pregnancy on the number of women who developed pre‐eclampsia.
What evidence did we find?
We searched the medical literature in July 2018 and found one relevant clinical trial. This trial included 1355 women who had previously had pre‐eclampsia, who lived in Argentina, South Africa, and Zimbabwe.
The trial compared pregnant women who had daily calcium with women who had placebo (a dummy tablet) until 20 weeks of pregnancy, when all women switched to having daily calcium until birth. We had some concerns about the evidence from this trial, because nearly a quarter of the women who were enrolled were lost to follow‐up, and we do not know whether they went on to become pregnant. Overall, while the results suggested that some women may benefit from calcium supplements, the findings included the possibility that the calcium didn't make a difference. Calcium may have helped some pregnant women avoid either losing the pregnancy or developing blood pressure problems, but we need more studies to be really confident that this effect was due to calcium. Calcium may have made little or no difference to whether pregnant women had other serious health conditions during pregnancy, such as: maternal admission to intensive care, blood pressure problems (pre‐eclampsia, severe pre‐eclampsia, eclampsia), placental separation from the uterus (placental abruption), or death. For babies, calcium may have had little or no impact on whether they were of low birthweight, of poor condition at birth, or required intensive care. The results did not clearly indicate the impact of calcium on whether babies died either before or after the birth, or needed to be admitted to neonatal intensive care for more than 24 hours.
What does this mean?
We need more research to decide whether or not calcium before pregnancy or during early pregnancy helps women avoid high blood‐pressure and other related problems.
Further research is needed to confirm whether initiating calcium supplementation pre‐ or in early pregnancy is associated with a reduction in adverse pregnancy outcomes for mother and baby. Research could also address the acceptability of the intervention to women, which was not covered by this review update.
Summary of findings
Background
Hypertension has been estimated to complicate 3% to 10% of all pregnancies and 11% of first pregnancies (Mol 2016), half of these being associated with pre‐eclampsia, and accounting for up to 30,000 maternal deaths annually worldwide (von Dadelszen 2016). Pre‐eclampsia is defined as high blood pressure and proteinuria occurring after the twentieth week of pregnancy.
In general, pre‐eclampsia is considerably more prevalent in low‐income than in high‐income communities. Two striking exceptions have been identified. More than 50 years ago, a low prevalence of pre‐eclampsia was reported from Ethiopia, where the diet, among other features, contained high levels of calcium (Hamlin 1952). The observation in 1980 that Mayan Indians in Guatemala, who traditionally soaked their corn in lime before cooking, had a low incidence of pre‐eclampsia and eclampsia (Belizan 1980), stimulated interest in the concept that the link between poverty and pre‐eclampsia might be dietary calcium deficiency.
Subsequent epidemiological, clinical, and laboratory studies linking pre‐eclampsia to calcium deficiency have been outlined in another Cochrane Review (Hofmeyr 2018).
Low dietary calcium intake is also associated with hypertension in the general population (Centeno 2009). A systematic review of randomised trials showed a small reduction in systolic and diastolic blood pressure with dietary and non‐dietary calcium supplementation (Griffith 1999). Systolic blood pressure was reduced by ‐1.44 mmHg (95% confidence interval (CI) ‐2.20 to ‐0.68; P < .001) and diastolic blood pressure by ‐0.84 mmHg (95% CI ‐1.44 to ‐0.24; P < .001). Low dietary calcium intake is also considered a risk factor for osteoporosis, renal stones, increased body mass index, insulin resistance and colorectal cancer (Centeno 2009).
The hypothesis that calcium supplementation during pregnancy might reduce the incidence of pre‐eclampsia was tested in several randomised trials commencing in the late 1980s.
The World Health Organization (WHO) conducted a randomised trial of calcium supplementation among low calcium intake pregnant women from 2001 to 2003 (Villar 2006). Results from this trial showed that although 1.5 g calcium/day supplement did not prevent pre‐eclampsia, it reduced its severity, maternal morbidity, and neonatal mortality. Supplementation in this trial was only given during later pregnancy, starting before the twentieth week of pregnancy. This trial was included (along with other randomised trials of calcium supplementation during pregnancy) in another Cochrane Review (Hofmeyr 2018). The results showed that calcium supplementation of at least 1 g daily, commencing around mid‐pregnancy, was associated with a modest reduction in pre‐eclampsia, and notably a reduction in its severe manifestations, particularly among women at increased risk, or with low dietary calcium intake. A review of lower‐dose calcium supplementation (mainly 500 mg/day in the second half of pregnancy), with or without other supplements, including small trials of variable quality, also found a reduction in pre‐eclampsia (9 trials, 2234 women, risk ratio (RR) 0·38, 95% CI 0·28 to 0.52; Hofmeyr 2014; Hofmeyr 2018).
WHO has recommended that in populations where dietary calcium intake is low, pregnant women receive 1.5 g to 2 g elemental calcium daily, particularly those at increased risk of pre‐eclampsia (women with one or more of the following risk factors: obesity, previous pre‐eclampsia, diabetes, chronic hypertension, renal disease, autoimmune disease, nulliparity, advanced maternal age, adolescent pregnancy, and conditions leading to hyperplacentation and large placentas, such as in twin pregnancy) (www.who.int/nutrition/publications/micronutrients/guidelines/calcium_supplementation/en/index.html). Dietary calcium intake is usually estimated by dietary assessment (e.g. 24‐hour dietary recall) in relation to the recommended dietary allowance.
Other related Cochrane Reviews include Buppasiri 2015; De‐Regil 2016; and Hofmeyr 2018.
Description of the condition
The hypertensive disorders of pregnancy include chronic hypertension, gestational hypertension, pre‐eclampsia or eclampsia, and unclassified hypertension (von Dadelszen 2016).
Pre‐eclampsia is defined as high blood pressure and proteinuria occurring for the first time after 20 weeks' gestation. It resolves by three months after delivery (Magee 2014).
Gestational hypertension is defined as diastolic blood pressure > 90 mmHg on two occasions four hours apart, or > 110 mmHg once, or systolic blood pressure > 140 mmHg on two occasions four hours apart, or > 160 mmHg once, after 20 weeks’ gestation (or a combination).
Gestational proteinuria is defined as 2+ or more on a urine dipstix, or > 300 mg/24 hours, or urinary protein/creatinine ratio > 30 g/mol, after 20 weeks’ gestation (von Dadelszen 2016).
Early pregnancy events affecting placentation are thought to contribute to the development of pre‐eclampsia via the following sequence (Lyall 2013; Palei 2013):
failure of cytotrophoblast invasion to remodel uterine spiral arterioles to low‐resistance vessels;
impaired uteroplacental blood flow;
syncytiotrophoblast oxidative stress and oversecretion of anti‐angiogenic and pro‐inflammatory factors from the ischaemic placenta;
widespread maternal endothelial dysfunction with vasoconstriction and renal dysfunction.
This sequence of events has been suggested to be a precursor particularly of early onset pre‐eclampsia (Redman 2014).
Description of the intervention
Previous studies and reviews have focused on calcium supplementation during pregnancy (Hofmeyr 2014; Hofmeyr 2018). Calcium supplementation began at different gestational ages in different trials, although most had begun supplementation by 20 weeks of pregnancy, the rationale being to cover the period during which pre‐eclampsia is manifest. As set out below, 20 weeks' gestation may be too late to interrupt early pregnancy events that are precursors of pre‐eclampsia. This review focuses on interventions that supplement calcium intake in early pregnancy (i.e. before 20 weeks' gestation), including calcium supplementation given to women before or very early in pregnancy and continuing during at least the first half of pregnancy. This review does not include evidence of the effects of food fortification with calcium.
The possibility of harm from calcium supplementation needs to be considered. Calcium supplementation (but not dietary calcium) has been associated with myocardial infarction (heart attack) risk in the Heidelberg study, an observation at risk of confounding (Li 2012); 1.5 g calcium/day during pregnancy may cause rebound postnatal bone demineralisation (an unexpected finding among multiple trial outcomes assessed (Jarjou 2010)); and an earlier review identified an unexpected increase in the syndrome of haemolysis, elevated liver enzymes and low platelets (HELLP) following calcium supplementation (Hofmeyr 2014; Hofmeyr 2018), perhaps through the antihypertensive effect of calcium masking the evolution of mild pre‐eclampsia into HELLP syndrome (Hofmeyr 2007).
Calcium may be administered in the form of carbonate, citrate, lactate, or gluconate, which have good bioavailability. The 19th Expert Committee on the Selection and Use of Essential Medicines recommended the listing of oral solid dosage forms of calcium, providing 500 mg of elemental calcium per dose (www.who.int/medicines/EC19uneditedReport.pdf).
How the intervention might work
An influx of calcium into the smooth muscle cells of the blood vessels causes contraction and increased resistance to blood flow, and therefore, increased blood pressure. The effect of calcium intake on blood pressure may be due to parathormone suppression and reduction in calcium in the vascular smooth muscle cells.
Hofmeyr 2008 conducted a randomised trial, nested within the large WHO trial of calcium supplementation (1.5 g daily from at least 20 weeks’ gestation) in pregnant women with low dietary calcium intake (Villar 2006). The nested trial failed to demonstrate an effect of calcium supplementation on biochemical measures commonly elevated in pre‐eclampsia: serum urate, platelet count, and urine protein/creatinine ratio.
The lack of effect on proteinuria is consistent with the findings of the main WHO trial, in which there was a statistically non‐significant reduction in pre‐eclampsia (RR 0.92, 95% CI 0.75 to 1.13; 8312 women) and severe pre‐eclampsia (RR 0.74, 95% CI 0.48 to 1.15; 8302 women), but no reduction in proteinuria (RR for proteinuria 1.01, 95% CI 0.88 to 1.15; 8312 women; Villar 2006). Proteinuria is a hallmark of pre‐eclampsia, and a predictor of adverse maternal outcome (von Dadelzsen 2004).
To reconcile the evidence from the systematic review for reduced pre‐eclampsia with calcium supplementation (Hofmeyr 2014; Hofmeyr 2018), with the absence of evidence of an effect on proteinuria and other markers for pre‐eclampsia, we proposed the hypothesis that calcium supplementation in the second half of pregnancy reduces blood pressure and thus the diagnosis and severe manifestations of pre‐eclampsia, without a significant effect on the underlying pathology (Hofmeyr 2008).
This hypothesis also serves to explain another anomaly identified in the systematic review: whereas pre‐eclampsia was reduced overall by 22% (RR 0.78, 95% CI 0.68 to 0.89; 12 trials, 15,206 women), and the composite outcome ‘maternal death or severe morbidity’ was reduced by 20% (RR 0.80, 95% CI 0.65 to 0.97; 5 trials, 9734 women), HELLP syndrome was increased 2.7 times with calcium supplementation (RR 2.67, 95% CI 1.05 to 6.82; 2 trials, 12,901 women; Hofmeyr 2014; Hofmeyr 2018). If calcium supplementation in the second half of pregnancy reduces only blood pressure, this would reduce the diagnosis and some of the hypertension‐related complications of pre‐eclampsia, while the effects on other organ systems, such as the endothelium, platelets, and liver might continue for a longer time in the calcium supplementation group in which fewer early deliveries for hypertension would take place.
The second anomaly requiring explanation is the modest effect of calcium supplementation in late pregnancy on pre‐eclampsia, in contrast to the striking epidemiological differences in populations with good and poor dietary calcium. Deficient dietary calcium before and during early pregnancy may place populations at risk for pre‐eclampsia, and the potential to reverse this effect by supplementation in later pregnancy may be limited (Hofmeyr 2008).
Based on the epidemiological association of pre‐eclampsia with low dietary calcium, and the current understanding that pre‐eclampsia has its origins in early pregnancy events, it is hypothesised that calcium supplementation in early pregnancy may reduce the risk of pre‐eclampsia (Hofmeyr 2008).
Why it is important to do this review
The benefits of calcium supplementation in the second half of pregnancy in the prevention of severe morbidity and mortality associated with pre‐eclampsia have been documented in a separate Cochrane Review (Hofmeyr 2018). However, there is no systematic evidence to prove or disprove the potential benefits of pre‐ and early pregnancy calcium supplementation (Hofmeyr 2008). Evidence for such an effect would create the opportunity to have a major impact on pre‐eclampsia at a population level. To our knowledge, there has not been a previous systematic review on this subject. This is an update of a review first published in 2017 (Hofmeyr 2017).
Objectives
To determine the effect of calcium supplementation, given before or early in pregnancy and for at least the first half of pregnancy, on pre‐eclampsia and other hypertensive disorders, maternal morbidity and mortality, and fetal and neonatal outcomes.
Methods
Criteria for considering studies for this review
Types of studies
We included one individually‐randomised trial. In future updates, if identified, cluster‐randomised trials will be included. Quasi‐randomised trials will also be included, with due caution and use of sensitivity analysis. Abstract reports will be included if sufficient information is given to assess trial quality and results. Cross‐over designs are not appropriate for this intervention.
Types of participants
This review is concerned with women of child bearing age but not yet pregnant, and women in the early stages of pregnancy (up to approximately 12 weeks' gestation). Women may be at low or average risk of pre‐eclampsia, or at high risk, as predicted by their previous pregnancies, nulliparity, or being from a high‐risk population.
Types of interventions
We considered interventions including calcium supplementation with or without additional supplements or treatments, compared with placebo, no intervention, or the same additional supplements or treatments, specifically:
calcium supplementation;
calcium plus additional supplements or treatments;
different doses of calcium supplementation.
Types of comparators:
placebo;
no supplementation;
the same additional supplements or treatments as the intervention group.
Comparisons
In future updates, comparisons will include calcium supplementation versus placebo or no supplementation; calcium plus additional supplements or treatments versus the same additional supplements or treatments; and different doses of calcium supplementation versus each other.
Studies of calcium plus other supplements or treatments will be included and subjected to subgroup analysis.
Types of outcome measures
We considered both maternal and fetal outcomes that might be related to the effects of calcium supplementation.
Primary outcomes
Pre‐eclampsia (gestational hypertension and proteinuria, as defined below)
Pre‐elampsia or pregnancy loss or stillbirth (or a combination) at any gestational age
Severe maternal morbidity and mortality index: one or more of secondary outcomes marked # below
Secondary outcomes
Maternal
No conception during study period
Pregnancy loss before 20 weeks' gestational age
Pregnancy loss or stillbirth at any gestational age
Gestational hypertension (diastolic blood pressure > 90 mmHg on two occasions four hours apart, or > 110 mmHg once, or systolic blood pressure > 140 mmHg on two occasions four hours apart, or > 160 mmHg once, appearing after 20 weeks’ gestation, or a combination)
Gestational proteinuria (2+ or more on a urine dipstix, or > 300 mg/24 hours, or > 500 mg/L, or urinary protein/creatinine ratio > 0.034, appearing after 20 weeks’ gestation)
* Severe gestational hypertension (systolic blood pressure > 160 mmHg on two occasions four hours apart, or once followed by antihypertensive therapy, or diastolic blood pressure > 110 mmHg on two occasions four hours apart, or once followed by antihypertensive therapy (or a combination), appearing after 20 weeks' gestation)
* Early onset pre‐eclampsia (< 32 weeks' gestation)
* # Severe pre‐eclampsia (proteinuria plus severe diastolic or systolic hypertension, or both)
Moderately severe thrombocytopenia (< 100 x 109/L or as defined by trial authors)
Uric acid > reference values for gestational age
# Renal failure (serum creatinine > 120 mmol/L or as defined by trial authors)
# Pulmonary oedema
# Cerebrovascular accident
Liver failure (serum aspartate aminotransferase (AST) > 70 U/L or as defined by trial authors)
# Intensive care unit (ICU) admission > 24 hours
* # Eclampsia
* # HELLP (haemolysis, elevated liver enzymes, and low platelets) syndrome (haemolysis (lactate dehydrogenase (LDH) > 600 U/L or bilirubin > 1.2 mg/dL), elevated liver enzymes (AST > 70 U/L), and low platelet count (< 100 x 109/L))
* # Placental abruption
# Maternal death
Mother’s hospital stay seven days or more after birth
Caesarean section
Severe pre‐eclamptic complications index (Villar 2006): one or more of outcomes marked* above
Neonatal
Birthweight < 2500 g
Preterm birth (< 37 weeks' gestation)
Early preterm birth (< 32 weeks’ gestation)
Apgar score less than seven at five minutes
Death or admission to neonatal ICU for 24 hours or more
Stillbirth
Pregnancy loss, stillbirth, or neonatal death before discharge
Neonate small‐for‐gestational age (non‐prespecified)
Search methods for identification of studies
The following methods section of this review is based on a standard template used by Cochrane Pregnancy and Childbirth.
Electronic searches
For this update, we searched Cochrane Pregnancy and Childbirth’s Trials Register by contacting their Information Specialist (31 July 2018).
The Register is a database containing over 25,000 reports of controlled trials in the field of pregnancy and childbirth. It represents over 30 years of searching. For full current search methods used to populate Pregnancy and Childbirth’s Trials Register, including the detailed search strategies for CENTRAL, MEDLINE, Embase, and CINAHL; the list of handsearched journals and conference proceedings; and the list of journals reviewed via the current awareness service, please follow this link
Briefly, Cochrane Pregnancy and Childbirth’s Trials Register is maintained by their Information Specialist and contains trials identified from:
monthly searches of the Cochrane Central Register of Controlled Trials (CENTRAL);
weekly searches of MEDLINE Ovid;
weekly searches of Embase Ovid;
monthly searches of CINAHL EBSCO;
handsearches of 30 journals and the proceedings of major conferences;
weekly current awareness alerts for a further 44 journals, plus monthly BioMed Central email alerts.
Search results are screened by two people, and the full text of all relevant trial reports identified through the searching activities described above is reviewed. Based on the intervention described, each trial report is assigned a number that corresponds to a specific Pregnancy and Childbirth review topic (or topics), and is then added to the Register. The Information Specialist searches the Register for each review using this topic number rather than keywords. This results in a more specific search set that has been fully accounted for in the relevant review sections (Included studies; Excluded studies; Ongoing studies).
In addition, we carried out a supplementary search of PubMed (inception to current) using the strategy given in Appendix 1. Date of last search was 13 July 2018.
We also searched ClinicalTrials.gov and the WHO International Clinical Trials Registry Platform (ICTRP; 31 July 2018) for unpublished, planned, and ongoing trial reports, using the terms given in Appendix 2.
Searching other resources
We searched the reference lists of retrieved papers.
We did not apply any language or date restrictions.
Data collection and analysis
The following methods section of this review is based on a standard template used by Cochrane Pregnancy and Childbirth.
Selection of studies
Two review authors independently assessed for inclusion, all the potential studies identified as a result of the search strategy. We resolved any disagreement through discussion, or if required, we involved the other authors. One study was conducted by two of the authors of this review (Justus Hofmeyr and Sarah Manyame), so it was assessed the two other authors (Nancy Medley and Myfanwy Williams (Hofmeyr 2019)).
Data extraction and management
We designed a form to extract data. Two review authors extracted the data using the agreed form, and resolved discrepancies through discussion, if required, we would have consulted one of the other review authors. Data from one study, conducted by two of the reviewer authors (GJH, SM) was extracted by NM and MJW (Hofmeyr 2019). Data were entered into Review Manager 5 software and checked for accuracy (Review Manager 2014).
When information regarding any of the above was unclear, we had planned to contact authors of the original reports to provide further details.
Assessment of risk of bias in included studies
We independently assessed risk of bias for the study using the criteria outlined in the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2011). Any disagreement was resolved by discussion, if necessary we would have involved a third assessor.
(1) Random sequence generation (checking for possible selection bias)
We described the method used to generate the allocation sequence in sufficient detail to allow an assessment of whether it should produce comparable groups.
We assessed the method as:
low risk of bias (any truly random process, e.g. random number table; computer random number generator);
high risk of bias (any non‐random process, e.g. odd or even date of birth; hospital or clinic record number);
unclear risk of bias.
(2) Allocation concealment (checking for possible selection bias)
We described the method used to conceal allocation to interventions prior to assignment, and assessed whether intervention allocation could have been foreseen in advance of, or during recruitment, or changed after assignment.
We assessed the methods as:
low risk of bias (e.g. telephone or central randomisation; consecutively numbered, sealed, opaque envelopes);
high risk of bias (open random allocation; unsealed or non‐opaque envelopes; alternation; date of birth);
unclear risk of bias.
(3.1) Blinding of participants and personnel (checking for possible performance bias)
We described the methods used, if any, to blind study participants and personnel from knowledge of which intervention a participant received. We considered that studies were at low risk of bias if they were blinded, or if we judged that the lack of blinding was unlikely to affect results. We assessed blinding separately for different outcomes or classes of outcomes.
We assessed the methods as:
low, high, or unclear risk of bias for participants;
low, high, or unclear risk of bias for personnel.
(3.2) Blinding of outcome assessment (checking for possible detection bias)
We described the methods used, if any, to blind outcome assessors from knowledge of which intervention a participant received. We assessed blinding separately for different outcomes or classes of outcomes.
We assessed the methods used to blind outcome assessment as:
low, high, or unclear risk of bias.
(4) Incomplete outcome data (checking for possible attrition bias due to the amount, nature, and handling of incomplete outcome data)
For each outcome or class of outcomes, we described the completeness of data, including attrition and exclusions from the analysis. We stated whether attrition and exclusions were reported, and the numbers included in the analysis at each stage (compared with the total randomised participants), reasons for attrition or exclusion where reported, and whether missing data were balanced across groups or were related to outcomes. Where sufficient information was reported, or could be supplied by the trial authors, we planned to re‐include missing data in the analyses that we undertook.
We assessed methods as:
low risk of bias (e.g. no missing outcome data; missing outcome data balanced across groups);
high risk of bias (e.g. numbers or reasons for missing data imbalanced across groups; ‘as treated’ analysis done with substantial departure of intervention received from that assigned at randomisation);
unclear risk of bias.
(5) Selective reporting (checking for reporting bias)
We described how we investigated the possibility of selective outcome reporting bias and what we found.
We assessed the methods as:
low risk of bias (where it was clear that all of the study’s prespecified outcomes and all expected outcomes of interest to the review were reported);
high risk of bias (where not all the study’s prespecified outcomes were reported; one or more reported primary outcomes were not prespecified; outcomes of interest were reported incompletely and so could not be used; study failed to include results of a key outcome that would have been expected to have been reported);
unclear risk of bias.
(6) Other bias (checking for bias due to problems not covered by (1) to (5) above)
We described any important concerns we had about other possible sources of bias.
(7) Overall risk of bias
We made explicit judgements about whether the study was at high risk of bias, according to the criteria given in the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2011). With reference to (1) to (6) above, we had planned to assess the likely magnitude and direction of the bias and whether we considered it was likely to impact on the findings. In future updates, we will explore the impact of the level of bias by undertaking sensitivity analyses – see Sensitivity analysis.
Assessing the quality of the body of evidence using the GRADE approach
We assessed the quality of the evidence using the GRADE approach, as outlined in the GRADE Handbook, in order to assess the quality of the body of evidence relating to the following outcomes, for the main comparisons (calcium with or without additional supplements or treatments versus placebo, no treatment, or the same additional supplements or treatments (GRADE Handbook)).
Pre‐eclampsia (gestational hypertension and proteinuria, as defined above)
Pre‐eclampsia, pregnancy loss or stillbirth, or a combination, at any gestational age
Severe maternal morbidity and mortality index
Pregnancy loss or stillbirth at any gestational age
Caesarean section
Birthweight < 2500 g
Apgar score less than seven at five minutes
Death or admission to neonatal ICU for 24 hours or more
Pregnancy loss, stillbirth or neonatal death before discharge
We used GRADEpro GDT to import data from Review Manager 5 in order to create a ’Summary of findings’ table (GRADEpro GDT; Review Manager 2014). A summary of the intervention effect and a measure of quality for each of the above outcomes was produced using the GRADE approach. The GRADE approach uses five considerations (study limitations, consistency of effect, imprecision, indirectness, and publication bias) to assess the quality of the body of evidence for each outcome. The evidence can be downgraded from high quality by one level for serious (or by two levels for very serious) limitations, depending on assessments for risk of bias, indirectness of evidence, serious inconsistency, imprecision of effect estimates, or potential publication bias.
Measures of treatment effect
Dichotomous data
For dichotomous data, we presented results as summary risk ratio with 95% confidence intervals.
Continuous data
We did not report any continuous data, however, in future review updates, we will use the mean difference if outcomes are measured in the same way between trials. We will use the standardised mean difference to combine trials that measure the same outcome, but use different methods.
Unit of analysis issues
Cluster‐randomised trials
No cluster‐randomised trials were identified during the search process. We will include cluster‐randomised trials in the analyses along with individually‐randomised trials if they are identified for future updates. We will adjust their standard errors using the methods described in the Cochrane Handbook for Systematic Reviews of Interventions, Section 16.3.4 or 16.3.6, using an estimate of the intracluster correlation co‐efficient (ICC) derived from the trial (if possible), from a similar trial, or from a study of a similar population (Higgins 2011). If we use ICCs from other sources, we will report this, and conduct sensitivity analyses to investigate the effect of variation in the ICC. If we identify both cluster‐randomised trials and individually‐randomised trials, we plan to synthesise the relevant information. We will consider it reasonable to combine the results from both if there is little heterogeneity between the study designs, and the interaction between the effect of intervention and the choice of randomisation unit is considered to be unlikely.
We will also acknowledge heterogeneity in the randomisation unit and perform a sensitivity analysis to investigate the effects of the randomisation unit.
Cross‐over trials
Cross‐over trials are not appropriate for this intervention.
Studies with multiple arms
For multi‐armed studies, in future updates, pairs of arms relevant to the review will be compared. Where one arm appears more than once in a meta‐analysis, the outcomes and denominators will be divided by the number of times it appears to avoid multiple counting.
Dealing with missing data
We noted the level of attrition for the included studies. In future updates, if more eligible studies are identified, the impact of including studies with high levels of missing data in the overall assessment of treatment effect will be explored by using sensitivity analysis.
For all outcomes, we carried out analyses, as far as possible, on an intention‐to‐treat basis, i.e. we attempted to include all participants randomised to each group in the analyses. The denominator for each outcome in the trial was the number randomised minus any participants whose outcomes were known to be missing.
Assessment of heterogeneity
As only one study was included, it was not appropriate to assess statistical heterogeneity. However, if more studies are included in future updates, we will assess heterogeneity in each meta‐analysis using Tau² and I², and Chi² statistics. We will consider heterogeneity as substantial if an I² is greater than 30%, and either Tau² is greater than zero, or there is a low P value (less than 0.10) in the Chi² test for heterogeneity. If we identify substantial heterogeneity (above 30%), we plan to explore it by prespecified subgroup analysis.
Assessment of reporting biases
In future updates, if there are 10 or more studies in the meta‐analysis, we will investigate reporting biases (such as publication bias) using funnel plots. We will assess funnel plot asymmetry visually. If asymmetry is suggested by a visual assessment, we will perform exploratory analyses to investigate it.
Data synthesis
We carried out statistical analysis using Review Manager 5 software (Review Manager 2014). We did not carry out meta‐analysis, because only one study was eligible for inclusion. In future updates, we will use fixed‐effect meta‐analysis for combining data where it is reasonable to assume that studies are estimating the same underlying treatment effect, i.e. where trials are examining the same intervention, and the trials’ populations and methods are judged to be sufficiently similar.
In future updates, if there is clinical heterogeneity sufficient to expect that the underlying treatment effects differs between trials, or if substantial statistical heterogeneity is detected, we will use random‐effects meta‐analysis to produce an overall summary, if an average treatment effect across trials is considered clinically meaningful. The random‐effects summary will be treated as the average range of possible treatment effects, and we will discuss the clinical implications of treatment effects differing between trials. If the average treatment effect is not clinically meaningful, we will not combine trials. If we use random‐effects analyses, the results will be presented as the average treatment effect with 95% confidence intervals, and the estimates of Tau² and I².
Subgroup analysis and investigation of heterogeneity
If we identified substantial heterogeneity, we had planned to investigate it using subgroup analyses and sensitivity analyses, however, we only included one study in this review.
We had planned to carry out the following subgroup analyses.
Women at high risk of pre‐eclampsia versus low risk versus risk unclear or mixed risk
Women with low dietary calcium versus adequate dietary calcium versus dietary calcium unclear or mixed
High‐dose calcium supplementation (1 g daily or more) versus low‐dose supplementation
Calcium alone versus calcium plus other supplements
Calcium commenced before pregnancy versus started in early pregnancy (< 13 weeks)
In future updates of the review, we will conduct subgroup analyses for the outcomes specified in the 'Summary of findings' tables.
We will assess subgroup differences by interaction tests available in Review Manager 5 (Review Manager 2014). We will report the results of subgroup analyses quoting the Chi² statistic and P value, and the interaction test I² value.
Sensitivity analysis
In future updates, we will perform sensitivity analysis by examining the effect on results of excluding:
trials at high risk of bias for allocation concealment;
trials with small sample sizes (less than 200);
trials with no pre‐registered protocols.
We will also carry out a sensitivity analysis to investigate the effect of the randomisation unit (where we analyse cluster‐randomised controlled trial data along with individually‐randomised trials).
Sensitivity analysis will be limited to the outcomes specified in the 'Summary of findings' tables.
Results
Description of studies
Results of the search
We retrieved 162 reports from the 2018 search. See Figure 1 for search details.
1.

Study flow diagram
We excluded one trial that was included in the previous version of this review (Rumiris 2006). In this update, we included one new trial that was ongoing at the time of the previous version of this review (Hofmeyr 2019), and added one new ongoing study (Fawzi 2017).
Included studies
Design
We included one double‐blind, placebo‐controlled trial (Hofmeyr 2019). The study was conducted from 12 July 2011 to 31 October 2017.
Sample sizes
In Hofmeyr 2019, 1355 nonpregnant women were randomised, of whom, 651 were known to have become pregnant during the study; 581/651 progressed to 20 weeks' gestation (although two of these women were lost to follow‐up after 20 weeks' gestation).
Settings
The Hofmeyr 2019 study was conducted in hospitals in South Africa, Zimbabwe, and Argentina.
Participants
Hofmeyr 2019 enrolled non‐pregnant women with previous pre‐eclampsia.
Interventions and comparisons
In Hofmeyr 2019, women received calcium 500 mg or placebo from enrolment until 20 weeks' gestation. After 20 weeks, all women received calcium 1.5 g daily.
See Characteristics of included studies for further details.
Outcomes
Hofmeyr 2019 reported all the prespecified review outcomes: pre‐eclampsia (PE), PE or pregnancy loss at any gestational age (or both), gestational hypertension, gestational proteinuria, pregnancy loss at any gestational age, no pregnancy during study period, severe gestational hypertension, early onset PE, severe PE, moderately severe thrombocytopenia, uric acid > reference values for gestational age, renal failure, liver failure, eclampsia, placental abruption, pulmonary oedema, cerebrovascular accident, ICU admission > 24 hours, haemolysis, elevated liver enzymes, and low platelets (HELLP) syndrome, maternal death, hospital stay ≥ 7 days after childbirth, caesarean section, birthweight < 2500 g, preterm birth, early preterm birth, Apgar score < 7 at five minutes, perinatal death or admission to neonatal ICU for 24 hours or more, stillbirth, pregnancy loss, stillbirth or neonatal death before discharge, pregnancy loss, stillbirth or neonatal death before six weeks, previous WHO calcium trial composites (severe pre‐eclamptic complications index), severe maternal morbidity, and mortality index and compliance outcomes.
Sources of trial funding
Hofmeyr 2019 reported funding from the University of British Columbia, a grantee of the Bill & Melinda Gates Foundation; UNDP/UNFPA/UNICEF/WHO/World Bank Special Programme of Research, Development and Research Training in Human Reproduction, World Health Organization; the Argentina Fund for Horizontal Cooperation of the Argentinean Ministry of Foreign Affairs; and the Centre for Intervention Science in Maternal and Child Health (CISMAC).
Declarations of interest
Hofmeyr 2019 reported: "We declare no competing interests. Midway through the study, the study team was approached by Alternative Discovery & Development, GlaxoSmithKline (GSK) Medicines Research Centre, UK, who partnered with us to collect blood samples from a sub‐group of participants in our trial for an independent, open, innovation pre‐eclampsia biomarker study, following a separate protocol, which was approved by the trial ethics committee. Apart from direct funding to the largest site (Chris Hani Baragwanath Hospital), specifically for the costs of this blood sample collection, GSK provided no funding to the main trial, and did not participate in any aspect of the main trial."
Excluded studies
Although included in the previous version of this review (Hofmeyr 2017), for this update, we excluded Rumiris 2006. It was not possible to determine the effects of calcium supplementation early in pregnancy from this trial, because it compared calcium in combination with a wide range of micronutrients and antioxidants versus placebo.
Risk of bias in included studies
Two of the review authors (GJH and SM) are investigators in the included study of pre‐pregnancy calcium supplementation (Hofmeyr 2019). Assessments for this study were independently conducted by the other two review authors (NM and MJW).
Allocation
We assessed Hofmeyr 2019 at low risk of bias. Computer‐generated random sequence was conducted independently by WHO, independently of the study investigators.
Blinding
We assessed Hofmeyr 2019 at low risk of bias for performance bias as the study used a placebo as a control, which was a sucrose tablet, identical in appearance to the tablet in the treatment group (double‐blind, placebo‐controlled).
Hofmeyr 2019 described the outcomes assessors as being blinded to group allocation, so we deemed this study at low risk of detection bias.
Incomplete outcome data
We assessed Hofmeyr 2019 at high risk of attrition bias. Although there was minimal loss to follow‐up post‐conception (loss to follow‐up = 2, documented in text), a high number of women were lost to follow‐up pre‐conception: 157/678 (23%) in calcium group; and 163/677 (24% in placebo group). Whilst this loss was balanced between the two groups, it is nevertheless possible that outcomes differed between the groups for this substantial proportion of women who were randomised.
Selective reporting
We assessed Hofmeyr 2019 at low risk of bias, because all outcomes specified in the protocol appeared in the published report.
Other potential sources of bias
We assessed Hofmeyr 2019 at unclear risk of bias for other potential sources; see the 'Risk of bias' table in Characteristics of included studies for details.
Effects of interventions
Summary of findings for the main comparison. Calcium supplementation versus placebo (maternal outcomes) commencing before or early in pregnancy, for preventing hypertensive disorders of pregnancy.
| Calcium compared to placebo commencing before or early in pregnancy, to prevent hypertensive disorders of pregnancy (maternal outcomes) | ||||||
| Patient or population: women who are not yet pregnant and women in the early stages of pregnancy Setting: Argentina, South Africa, and Zimbabwe Intervention: calcium Comparison: placebo | ||||||
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effect (95% CI) | № of participants (studies) | Certainty of the evidence (GRADE) | Comments | |
| Risk with placebo (maternal outcomes) | Risk with calcium | |||||
| Pre‐eclampsia | Study population | RR 0.80 (0.61 to 1.06) | 579 (1 RCT) | ⊕⊕⊝⊝ LOW a, b | ||
| 290 per 1000 | 232 per 1000 (177 to 307) | |||||
| Pre‐eclampsia, or pregnancy loss, or stillbirth (or combination) at any gestational age | Study population | RR 0.82 (0.66 to 1.00) | 633 (1 RCT) | ⊕⊕⊝⊝ LOW b, c | ||
| 406 per 1000 | 333 per 1000 (268 to 406) | |||||
| Severe maternal morbidity and mortality index | Study population | RR 0.93 (0.68 to 1.26) | 579 (1 RCT) | ⊕⊕⊝⊝ LOW a, b | ||
| 230 per 1000 | 214 per 1000 (156 to 289) | |||||
| Pregnancy loss or stillbirth at any gestational age | Study population | RR 0.83 (0.61 to 1.14) | 633 (1 RCT) | ⊕⊕⊝⊝ LOW a, b | ||
| 216 per 1000 | 179 per 1000 (132 to 246) | |||||
| Caesarean section | Study population | RR 1.11 (0.96 to 1.28) | 578 (1 RCT) | ⊕⊕⊝⊝ LOW a, b | ||
| 537 per 1000 | 596 per 1000 (516 to 687) | |||||
| *The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: Confidence interval; RR: Risk ratio; OR: Odds ratio; | ||||||
| GRADE Working Group grades of evidence High certainty: We are very confident that the true effect lies close to that of the estimate of the effect Moderate certainty: We are moderately confident in the effect estimate: The true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different Low certainty: Our confidence in the effect estimate is limited: The true effect may be substantially different from the estimate of the effect Very low certainty: We have very little confidence in the effect estimate: The true effect is likely to be substantially different from the estimate of effect | ||||||
a Wide confidence interval crossing the line of no effect; estimate from a single trial (‐1)
b Single included study at high risk of attrition bias (‐1)
c Confidence interval meets the line of no effect; estimate from a single trial (‐1)
Summary of findings 2. Calcium compared to placebo (offspring outcomes) commencing before or early in pregnancy, for preventing hypertensive disorders of pregnancy.
| Calcium compared to placebo commencing before or early in pregnancy, for preventing hypertensive disorders of pregnancy (offspring outcomes) | ||||||
| Patient or population: women who are not yet pregnant, and women in the early stages of pregnancy Setting: Argentina, South Africa, and Zimbabwe Intervention: calcium Comparison: placebo | ||||||
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effect (95% CI) | № of participants (studies) | Certainty of the evidence (GRADE) | Comments | |
| Risk with placebo (offspring outcomes) | Risk with calcium | |||||
| Birthweight < 2500 g | Study population | RR 1.00 (0.76 to 1.30) | 507 (1 RCT) | ⊕⊕⊝⊝ LOW a, b | ||
| 300 per 1000 | 300 per 1000 (228 to 391) | |||||
| Apgar < 7 at 5 minutes | Study population | RR 0.43 (0.15 to 1.21) | 494 (1 RCT) | ⊕⊝⊝⊝ VERY LOWb, c, d | ||
| 46 per 1000 | 20 per 1000 (7 to 56) | |||||
| Perinatal death, or NICU admission for > 24 hours (or both) | Study population | RR 1.11 (0.77 to 1.60) | 508 (1 RCT) | ⊕⊕⊝⊝ LOW a, b | ||
| 177 per 1000 | 196 per 1000 (136 to 283) | |||||
| Pregnancy loss, stillbirth or NND before discharge | Study population | RR 0.82 (0.61 to 1.10) | 632 (1 RCT) | ⊕⊕⊝⊝ LOW 2,3 | ||
| 246 per 1000 | 202 per 1000 (150 to 271) | |||||
| *The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: Confidence interval; RR: Risk ratio; NND: Neonatal death | ||||||
| GRADE Working Group grades of evidence High certainty: We are very confident that the true effect lies close to that of the estimate of the effect Moderate certainty: We are moderately confident in the effect estimate: The true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different Low certainty: Our confidence in the effect estimate is limited: The true effect may be substantially different from the estimate of the effect Very low certainty: We have very little confidence in the effect estimate: The true effect is likely to be substantially different from the estimate of effect | ||||||
a Wide confidence interval crossing the line of no effect whilst also including appreciable harm; estimate from a single trial (‐1)
b Single included study at high risk of attrition bias (‐1)
c Wide confidence interval crossing the line of no effect, including both appreciable benefit and harm; estimate from a single trial (‐1).
d Small number of events (‐1)
We included one study, which addressed one comparison; we did not conduct a meta‐analysis.
1. Calcium versus placebo
We included one study (Hofmeyr 2019). All prespecified review outcomes were reported.
For pregnancy outcomes, the denominators were women who conceived and had data available, except for pre‐eclampsia outcomes, for which the denominators were women with pregnancies beyond 20 weeks' gestation.
Primary outcomes
Pre‐eclampsia
Calcium supplements may make little or now difference in women's risk of pre‐eclampsia (69/296 versus 82/283, risk ratio (RR) 0.80, 95% CI 0.61 to 1.06; 579 women; low‐quality evidence; Analysis 1.1).
1.1. Analysis.
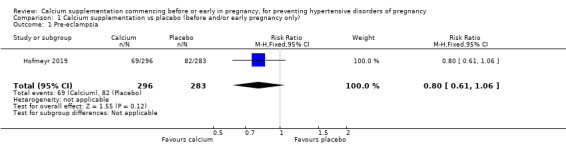
Comparison 1 Calcium supplementation vs placebo (before and/or early pregnancy only), Outcome 1 Pre‐eclampsia.
Pre‐elampsia or pregnancy loss or stillbirth (or a combination) at any gestational age
Calcium supplements may have reduced the risk of pre‐eclampsia and pregnancy loss, though the CI met the line of no effect (RR 0.82, 95% CI 0.66 to 1.00; 633 women; low‐quality evidence; Analysis 1.2).
1.2. Analysis.

Comparison 1 Calcium supplementation vs placebo (before and/or early pregnancy only), Outcome 2 Pre‐eclampsia and/or pregnancy loss/stillbirth at any gestational age.
Severe maternal morbidity and mortality index
Calcium may have made little or no difference to rates of severe maternal morbidity and mortality (RR 0.93, 95% CI 0.68 to 1.26; 579 women; low‐quality evidence; Analysis 1.3).
1.3. Analysis.
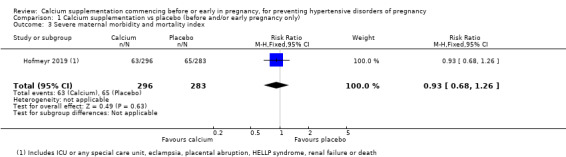
Comparison 1 Calcium supplementation vs placebo (before and/or early pregnancy only), Outcome 3 Severe maternal morbidity and mortality index.
Secondary outcomes
Unless otherwise stated, women assigned to calcium supplementation and to placebo groups had similar results for the following secondary outcomes. We have only included the quality of the evidence for outcomes that are included in the 'Summary of findings' tables. For many outcomes, event rates were low.
Maternal outcomes
No conception (RR 0.97, 95% CI 0.88 to 1.08; 1355 women; Analysis 1.4); all randomised women with data were included in the denominator.
1.4. Analysis.

Comparison 1 Calcium supplementation vs placebo (before and/or early pregnancy only), Outcome 4 No conception.
Pregnancy loss before 20 week's gestation (RR 0.96, 95% CI 0.58 to 1.60; 633 women; Analysis 1.5).
1.5. Analysis.
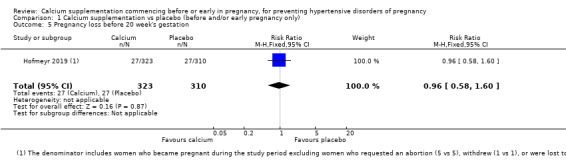
Comparison 1 Calcium supplementation vs placebo (before and/or early pregnancy only), Outcome 5 Pregnancy loss before 20 week's gestation.
Pregnancy loss/stillbirth at any gestational age (RR 0.83, 95% CI 0.61 to 1.14; 633 women; low‐quality; Analysis 1.6).
1.6. Analysis.
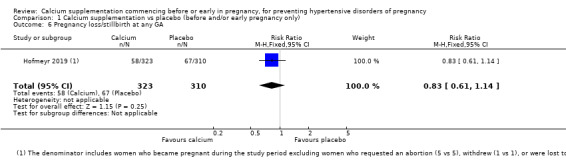
Comparison 1 Calcium supplementation vs placebo (before and/or early pregnancy only), Outcome 6 Pregnancy loss/stillbirth at any GA.
Gestational hypertension (RR 0.94, 95% CI 0.84 to 1.05; 579 women Analysis 1.7).
1.7. Analysis.
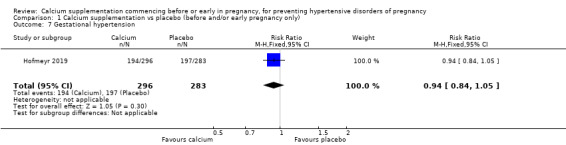
Comparison 1 Calcium supplementation vs placebo (before and/or early pregnancy only), Outcome 7 Gestational hypertension.
Gestational proteinuria (RR 0.83, 95% CI 0.64 to 1.07; 579 women; Analysis 1.8).
1.8. Analysis.
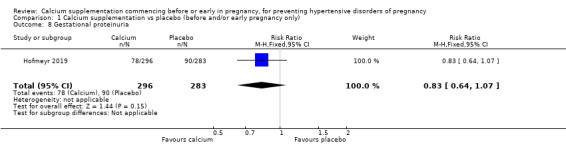
Comparison 1 Calcium supplementation vs placebo (before and/or early pregnancy only), Outcome 8 Gestational proteinuria.
Severe gestational hypertension (RR 0.95, 95% CI 0.75 to 1.20; 579 women; Analysis 1.9).
1.9. Analysis.
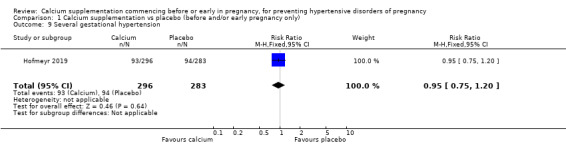
Comparison 1 Calcium supplementation vs placebo (before and/or early pregnancy only), Outcome 9 Several gestational hypertension.
Early onset pre‐eclampsia (RR 0.93, 95% CI 0.61 to 1.42; 579 women; Analysis 1.10).
1.10. Analysis.
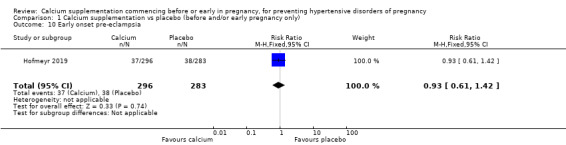
Comparison 1 Calcium supplementation vs placebo (before and/or early pregnancy only), Outcome 10 Early onset pre‐eclampsia.
Severe pre‐eclampsia (RR 0.83, 95% CI 0.59 to 1.16; 579 women; Analysis 1.11).
1.11. Analysis.
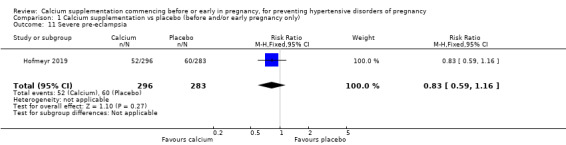
Comparison 1 Calcium supplementation vs placebo (before and/or early pregnancy only), Outcome 11 Severe pre‐eclampsia.
Moderately severe thrombocytopenia (13/63 versus 12/74; 137 women; Analysis 1.12); only women who had pre‐eclampsia, and for whom data were available, were included in the denominator. We have not included effect estimates in the analysis, because the findings relate to so few of the original group of randomised women.
1.12. Analysis.
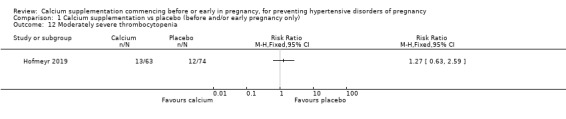
Comparison 1 Calcium supplementation vs placebo (before and/or early pregnancy only), Outcome 12 Moderately severe thrombocytopenia.
Uric acid > reference values for GA (22/27 versus 19/24; 51 women; Analysis 1.13); only women who had pre‐eclampsia, and for whom data were available, were included in the denominator. We have not included effect estimates in the analysis, because the findings relate to so few of the original group of randomised women.
1.13. Analysis.
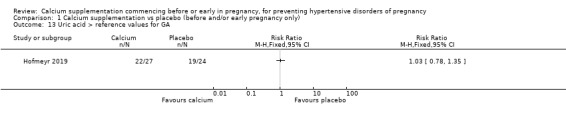
Comparison 1 Calcium supplementation vs placebo (before and/or early pregnancy only), Outcome 13 Uric acid > reference values for GA.
Renal failure (7/58 versus 5/68; 126 women; Analysis 1.14); only women who had pre‐eclampsia, and for whom data were available were included in the denominator. We have not included effect estimates in the analysis, because the findings relate to so few of the original group of randomised women.
1.14. Analysis.
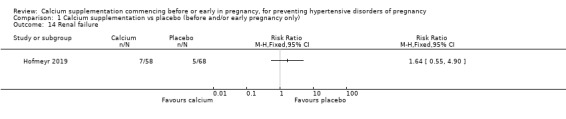
Comparison 1 Calcium supplementation vs placebo (before and/or early pregnancy only), Outcome 14 Renal failure.
Pulmonary oedema (RR 0.32, 95% CI 0.01 to 7.79; 579 women; Analysis 1.15).
1.15. Analysis.
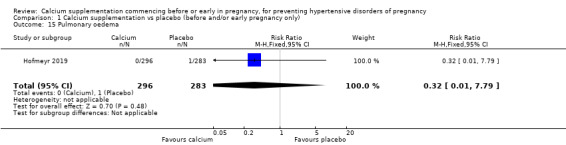
Comparison 1 Calcium supplementation vs placebo (before and/or early pregnancy only), Outcome 15 Pulmonary oedema.
Cerebrovascular accident (RR not estimable due to zero events in both trial arms; 579 women; Analysis 1.16).
1.16. Analysis.
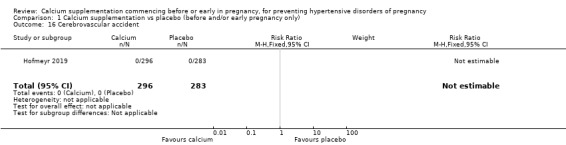
Comparison 1 Calcium supplementation vs placebo (before and/or early pregnancy only), Outcome 16 Cerebrovascular accident.
Liver failure (8/53 versus 7/63; Analysis 1.17); only women who had pre‐eclampsia, and for whom data were available, were included in the denominator. We have not included effect estimates in the analysis, because the findings relate to so few of the original group of randomised women.
1.17. Analysis.
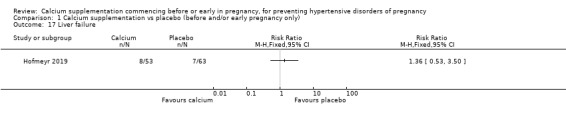
Comparison 1 Calcium supplementation vs placebo (before and/or early pregnancy only), Outcome 17 Liver failure.
ICU admission > 24 hours (RR 0.64, 95% CI 0.11 to 3.79; 579 women; Analysis 1.18).
1.18. Analysis.
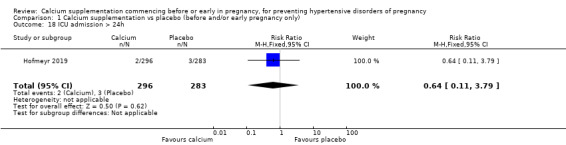
Comparison 1 Calcium supplementation vs placebo (before and/or early pregnancy only), Outcome 18 ICU admission > 24h.
Eclampsia (RR 0.76, 95% CI 0.21 to 2.82; 579 women; Analysis 1.19).
1.19. Analysis.
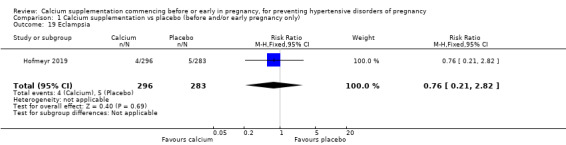
Comparison 1 Calcium supplementation vs placebo (before and/or early pregnancy only), Outcome 19 Eclampsia.
HELLP syndrome (10/69 versus 7/81; 150 women; Analysis 1.20); only women who had pre‐eclampsia, and for whom data were available, were included in the denominator. We have not included effect estimates in the analysis, because the findings relate to so few of the original group of randomised women.
1.20. Analysis.
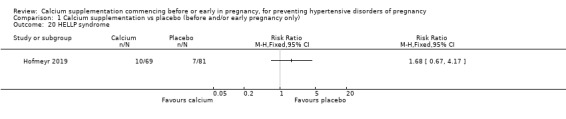
Comparison 1 Calcium supplementation vs placebo (before and/or early pregnancy only), Outcome 20 HELLP syndrome.
Placental abruption (RR 1.73, 95% CI 0.59 to 5.09; 578 women; Analysis 1.21).
1.21. Analysis.

Comparison 1 Calcium supplementation vs placebo (before and/or early pregnancy only), Outcome 21 Placental abruption.
Maternal death (RR 0.96, 95% CI 0.14 to 6.77; 633 women; Analysis 1.22).
1.22. Analysis.

Comparison 1 Calcium supplementation vs placebo (before and/or early pregnancy only), Outcome 22 Maternal death.
Hospital stay > 7 days from birth (RR 1.70, 95% CI 0.88 to 3.30; 575 women; Analysis 1.23).
1.23. Analysis.
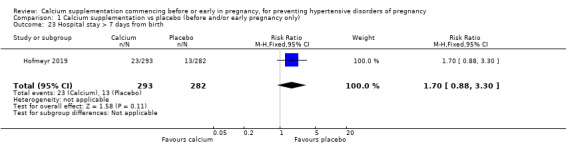
Comparison 1 Calcium supplementation vs placebo (before and/or early pregnancy only), Outcome 23 Hospital stay > 7 days from birth.
Caesarean section (RR 1.11, 95% CI 0.96 to 1.28; 578 women; low‐quality; Analysis 1.24).
1.24. Analysis.
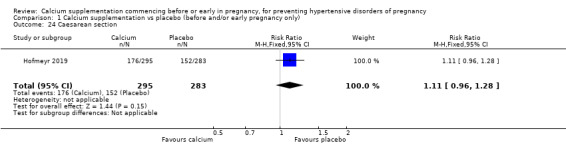
Comparison 1 Calcium supplementation vs placebo (before and/or early pregnancy only), Outcome 24 Caesarean section.
Severe pre‐eclamptic complications index (RR 0.98, 95% CI 0.79 to 1.22; 579 women; Analysis 1.25).
1.25. Analysis.
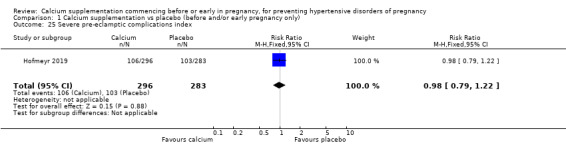
Comparison 1 Calcium supplementation vs placebo (before and/or early pregnancy only), Outcome 25 Severe pre‐eclamptic complications index.
Neonatal outcomes
Birthweight < 2500 g (RR 1.00, 95% CI 0.76 to 1.30; 507 babies; low‐quality; Analysis 1.26).
1.26. Analysis.

Comparison 1 Calcium supplementation vs placebo (before and/or early pregnancy only), Outcome 26 Birthweight < 2500 g.
Preterm birth < 37 weeks (RR 0.90, 95% CI 0.74 to 1.10; 579 women; Analysis 1.27).
1.27. Analysis.

Comparison 1 Calcium supplementation vs placebo (before and/or early pregnancy only), Outcome 27 Preterm birth < 37 weeks.
Early preterm birth < 32 weeks (RR 0.79, 95% CI 0.56 to 1.12; 579 women; Analysis 1.28).
1.28. Analysis.

Comparison 1 Calcium supplementation vs placebo (before and/or early pregnancy only), Outcome 28 Early preterm birth < 32 weeks.
Apgar score < 7 at five minutes (RR 0.43, 95% CI 0.15 to 1.21; 494 babies; very low‐quality; Analysis 1.29).
1.29. Analysis.
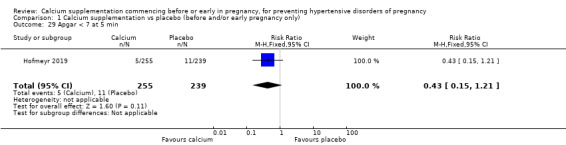
Comparison 1 Calcium supplementation vs placebo (before and/or early pregnancy only), Outcome 29 Apgar < 7 at 5 min.
Perinatal death or NICU admission for > 24 hours, or both (RR 1.11, 95% CI 0.77 to 1.60; 508 babies; low‐quality; Analysis 1.30). Although the point estimate for this composite outcome suggests that calcium may make little or no difference, the 95% CI is quite wide, and fails to exclude the possibility of appreciable harm for the baby.
1.30. Analysis.
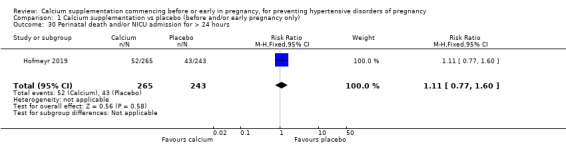
Comparison 1 Calcium supplementation vs placebo (before and/or early pregnancy only), Outcome 30 Perinatal death and/or NICU admission for > 24 hours.
Stillbirth (RR 0.78, 95% CI 0.48 to 1.27; 579 women; Analysis 1.31).
1.31. Analysis.
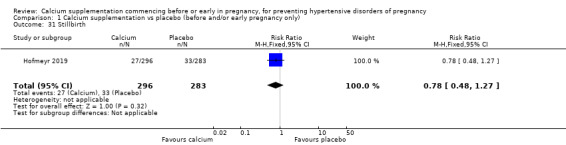
Comparison 1 Calcium supplementation vs placebo (before and/or early pregnancy only), Outcome 31 Stillbirth.
Pregnancy loss, stillbirth, or neonatal death before discharge (RR 0.82, 95% CI 0.61 to 1.10; 632 women; low‐quality; Analysis 1.32).
1.32. Analysis.

Comparison 1 Calcium supplementation vs placebo (before and/or early pregnancy only), Outcome 32 Pregnancy loss, stillbirth or NND before discharge.
Discussion
Summary of main results
Calcium versus placebo
One double‐blind study recruited women with previous pre‐eclampsia before they were pregnant, and continued supplementation with calcium 500 mg or placebo until 20 weeks' gestation (Hofmeyr 2019). All women lived in countries where average dietary calcium in pregnancy is known to be low, and the size of the supplement was intended to bring the women's calcium intake up to the approximate level of that among pregnant women in high‐income countries. After 20 weeks, all women received calcium 1.5 g daily. The study measured the effect of pre‐ and early pregnancy supplementation, over and above the known benefits supplementation commencing later in pregnancy. The point estimates for all of the review's three primary outcomes of pre‐eclampsia, pre‐eclampsia or pregnancy loss or stillbirth (or a combination) at any gestational age, and severe maternal morbidity and mortality index, suggested a posisble reduction with calcium supplementation, but confidence intervals crossed or met the line of no effect for each one. For pregnancy loss or pre‐eclampsia, the upper limit was a risk ratio of 1.00.
Possible reasons for a lack of clear‐cut results include sub‐optimal compliance and inadequate sample size. Hofmeyr 2019 reported on compliance (consumption of > 80% of tablets) in those women who conceived, and were followed through pregnancy up to 20 weeks' gestation. In both the pre‐pregnancy period (100/213 in the calcium group; 100/208 in the control group), and for the course of the pregnancy (145/274 in the calcium group; 149/269 in the control group), only around half of the women in both groups for each of these periods consumed > 80% of their tablets. Better adherence to the intervention may have yielded clearer results.
The study hypothesis was that while calcium supplementation in the second half of pregnancy might reduce pre‐eclampsia by having a direct effect on blood pressure alone, any effect of supplementation seen prior to 20 weeks (before pre‐eclampsia is manifest), might indicate an effect on the underlying placental pathology. The findings were consistent with this hypothesis, but not conclusive. The persistence of significantly reduced diastolic blood pressure at 32 weeks, after 12 weeks of high‐dose calcium to both groups (not an outcome prespecified in this review), suggests an effect on the pathogenesis of pre‐eclampsia.
Overall completeness and applicability of evidence
The evidence comes from only one study of calcium supplementation (Hofmeyr 2019). Although this trial directly addressed the review question, and reached its own prespecified sample size, it is possible that the trial may not have included enough women to yield compelling evidence for the efficacy of the intervention.
The single included trial recruited women who were at increased risk of pre‐eclampsia, on the basis of a history of pre‐eclampsia in the most recent, previous pregnancy. Therefore, this trial did not include primigravid women during their first pregnancy, a group that is also recognised to be at increased risk of pre‐eclampsia. It is possible that calcium may have different (i.e. either more or less beneficial) effects for women who are pregnant for the first time. If so, these could not be identified by this study.
This review did not include evidence on women's acceptability for taking calcium supplements before, and during early pregnancy. The single included trial did include data on adherence to the intervention, and whilst compliance was sub‐optimal, it was similar between the intervention and control groups. Future updates of this review could fruitfully report on outcomes that explicitly address both adherence and women's views about the intervention.
Quality of the evidence
We assessed Hofmeyr 2019 at low risk of selection, performance, and detection bias, and at unclear risk of selective reporting bias. However, we judged it to be at high risk of attrition bias, since nearly one quarter of the women were lost to follow‐up pre‐conception (Figure 2).
2.

'Risk of bias' summary: review authors' judgements about each risk of bias item for each included study
A limitation of this study was that the main outcomes were conditional upon intermediate outcomes. For instance, for pre‐eclampsia, the reported results were conditional upon women reaching the intermediate outcomes of conception, and pregnancy reaching 20 weeks' gestation. This rasies two concerns. First, if calcium supplementation had an effect on conception or early pregnancy loss, this may have introduced bias for the outcome pre‐eclampsia. This suggests that the outcome least susceptible to bias is pre‐eclampsia or early pregnancy loss, since this composite avoids the possible confounding effect of early calcium supplementation on pregnancy loss. Second, it is also possible that there may have been differences between the groups in the women who didn't conceive or who had early pregnancy losses. These differences would then introduce bias into analyses including only women who conceived or reached 20 weeks' gestation. We investigated this possible source of bias by conducting a sensitivity analysis for the maternal outcomes reported in our 'Summary of findings' table, by including all randomised women in the denominators, rather than only those who were known to have conceived, or those who were known to have conceived and reached 20 weeks' gestation. As with our main analysis, the findings were inconclusive (Analysis 2.1; Analysis 2.2; Analysis 2.3; Analysis 2.4; Analysis 2.5). Although there were no major differences, in general, the sensitivity analyses yielded more equivocal results for all of the main maternal outcomes.
2.1. Analysis.
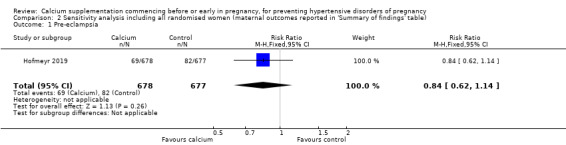
Comparison 2 Sensitivity analysis including all randomised women (maternal outcomes reported in 'Summary of findings' table), Outcome 1 Pre‐eclampsia.
2.2. Analysis.
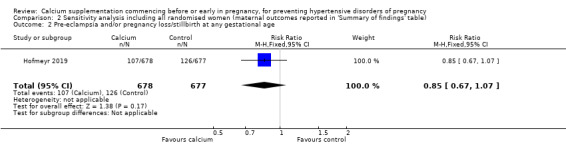
Comparison 2 Sensitivity analysis including all randomised women (maternal outcomes reported in 'Summary of findings' table), Outcome 2 Pre‐eclampsia and/or pregnancy loss/stillbirth at any gestational age.
2.3. Analysis.

Comparison 2 Sensitivity analysis including all randomised women (maternal outcomes reported in 'Summary of findings' table), Outcome 3 Severe maternal morbidity and mortality index.
2.4. Analysis.
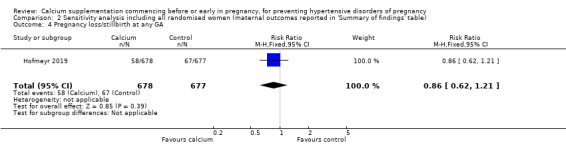
Comparison 2 Sensitivity analysis including all randomised women (maternal outcomes reported in 'Summary of findings' table), Outcome 4 Pregnancy loss/stillbirth at any GA.
2.5. Analysis.

Comparison 2 Sensitivity analysis including all randomised women (maternal outcomes reported in 'Summary of findings' table), Outcome 5 Caesarean section.
It is important to note that interpretation of this sensitivity analysis is limited – just like this review's main analysis – by the high risk of attrition bias pre‐conception. Although this attrition was balanced between groups, a substantial proportion of the women who were initially enrolled withdrew or were lost to follow‐up, to the extent that serious risk of attrition bias cannot be ruled out.
We used GRADEpro GDT software to assess the quality of the outcomes listed in the 'Summary of findings' tables. There was low‐quality evidence for the three primary outcomes: pre‐eclampsia, pre‐eclampsia or pregnancy loss (or both) at any gestational age, and severe maternal morbidity and mortality index, and for the two secondary maternal outcomes: pregnancy loss or stillbirth at any gestational age, and caesarean section.
There was also low‐quality evidence for the secondary baby outcomes: birthweight < 2500g; perinatal death or NICU admission for > 24 hours (or both); and pregnancy loss, stillbirth, or neonatal death before discharge; and very low‐quality evidence for an Apgar score less than seven at five minutes. We downgraded the quality of the evidence because: there were wide confidence intervals (meeting or crossing the line of no effect; including both appreciable harm and appreciable benefit); data were drawn from a single trial; and we had concerns about the high rate of attrition pre‐conception. For low Apgar scores, we also downgraded the quality of the evidence because there were very few events.
Potential biases in the review process
The review authors are investigators in the included study of pre‐pregnancy calcium supplementation. They did not participate in decisions regarding the inclusion, data extraction, or 'Risk of bias' assessment of this study.
We carried out a comprehensive search of the literature and followed standard systematic review methods, including duplication of eligibility assessment and data extraction, to minimise bias.
Agreements and disagreements with other studies or reviews
Hofmeyr 2019 is the first reported trial to assess the effect of calcium supplementation limited to the pre‐and early pregnancy periods.
Authors' conclusions
Implications for practice.
The evidence in this review is drawn from one study of calcium supplementation, given before and in the first half of pregnancy. Calcium supplementation before and early in pregnancy may reduce the risk of women experiencing the composite outcome pre‐eclampsia or pregnancy loss at any gestational age, but the results are inconclusive for all other outcomes for women and babies. Therefore, current evidence neither supports nor refutes the routine use of calcium supplementation before conception and in early pregnancy.
Implications for research.
Further research is needed to confirm whether pre‐ or early‐pregnancy calcium supplementation is associated with a reduction in adverse pregnancy outcomes, such as pre‐eclampsia and pregnancy loss. Such studies should be adequately powered, limited to calcium supplementation, placebo‐controlled, and include the outcomes chosen for this review, with the addition of outcomes that assess acceptability of the intervention to women. However, neither the established benefits of late pregnancy calcium supplementation, nor the possible benefits of pre‐ or early pregnancy supplementation can be realized comprehensively through antenatal supplementation, because many women do not attend antenatal care. Future research could also focus on efforts to improve dietary calcium intake amongst all women.
What's new
| Date | Event | Description |
|---|---|---|
| 13 July 2018 | New citation required and conclusions have changed | New conclusions regarding the effect of pre‐pregnancy and early pregnancy calcium supplementation (new trial) |
| 13 July 2018 | New search has been performed | Review search updated. One trial added (Hofmeyr 2019). One previously included trial has been excluded in this version of the review (Rumiris 2006). |
Acknowledgements
As part of the pre‐publication editorial process, this review has been commented on by three peers (an editor and two referees who are external to the editorial team), a member of Cochrane Pregnancy and Childbirth's international panel of consumers and the Group's Statistical Adviser. The authors are grateful to the following peer reviewers for their time and comments: Rachel Plachcinski, NCT Research Engagement Officer, UK; Dr Vijaya Musini, Assistant Professor, Deptartment of Anesthesiology, Pharmacology and Therapeutics, Manager, Drug Assessment Working Group, Therapeutics Initiative, Editor, Cochrane Hypertension Review Group, University of British Columbia, Vancouver, Canada; and also to the one reviewer who wishes to remain anonymous.
This project was supported by the National Institute for Health Research, via Cochrane Infrastructure funding to Cochrane Pregnancy and Childbirth. The views and opinions expressed therein are those of the authors and do not necessarily reflect those of the Systematic Reviews Programme, NIHR, NHS or the Department of Health.
Appendices
Appendix 1. PubMed search strategy
(calcium) AND ((preeclampsia) OR (eclampsia)) AND ((pregnancy) OR (pregnant) OR (pregnancies)) AND ((random) OR (randomised) OR (randomized)) AND (trial)
Appendix 2. Search terms used in ICTRP and ClinicalTrials.gov
calcium AND pregnancy
Data and analyses
Comparison 1. Calcium supplementation vs placebo (before and/or early pregnancy only).
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Pre‐eclampsia | 1 | 579 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.80 [0.61, 1.06] |
| 2 Pre‐eclampsia and/or pregnancy loss/stillbirth at any gestational age | 1 | 633 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.82 [0.66, 1.00] |
| 3 Severe maternal morbidity and mortality index | 1 | 579 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.93 [0.68, 1.26] |
| 4 No conception | 1 | 1355 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.97 [0.88, 1.08] |
| 5 Pregnancy loss before 20 week's gestation | 1 | 633 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.96 [0.58, 1.60] |
| 6 Pregnancy loss/stillbirth at any GA | 1 | 633 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.83 [0.61, 1.14] |
| 7 Gestational hypertension | 1 | 579 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.94 [0.84, 1.05] |
| 8 Gestational proteinuria | 1 | 579 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.83 [0.64, 1.07] |
| 9 Several gestational hypertension | 1 | 579 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.95 [0.75, 1.20] |
| 10 Early onset pre‐eclampsia | 1 | 579 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.93 [0.61, 1.42] |
| 11 Severe pre‐eclampsia | 1 | 579 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.83 [0.59, 1.16] |
| 12 Moderately severe thrombocytopenia | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 13 Uric acid > reference values for GA | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 14 Renal failure | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 15 Pulmonary oedema | 1 | 579 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.32 [0.01, 7.79] |
| 16 Cerebrovascular accident | 1 | 579 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 17 Liver failure | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 18 ICU admission > 24h | 1 | 579 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.64 [0.11, 3.79] |
| 19 Eclampsia | 1 | 579 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.76 [0.21, 2.82] |
| 20 HELLP syndrome | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 21 Placental abruption | 1 | 578 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.73 [0.59, 5.09] |
| 22 Maternal death | 1 | 633 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.96 [0.14, 6.77] |
| 23 Hospital stay > 7 days from birth | 1 | 575 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.70 [0.88, 3.30] |
| 24 Caesarean section | 1 | 578 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.11 [0.96, 1.28] |
| 25 Severe pre‐eclamptic complications index | 1 | 579 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.98 [0.79, 1.22] |
| 26 Birthweight < 2500 g | 1 | 507 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.00 [0.76, 1.30] |
| 27 Preterm birth < 37 weeks | 1 | 579 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.90 [0.74, 1.10] |
| 28 Early preterm birth < 32 weeks | 1 | 579 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.79 [0.56, 1.12] |
| 29 Apgar < 7 at 5 min | 1 | 494 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.43 [0.15, 1.21] |
| 30 Perinatal death and/or NICU admission for > 24 hours | 1 | 508 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.11 [0.77, 1.60] |
| 31 Stillbirth | 1 | 579 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.78 [0.48, 1.27] |
| 32 Pregnancy loss, stillbirth or NND before discharge | 1 | 632 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.82 [0.61, 1.10] |
Comparison 2. Sensitivity analysis including all randomised women (maternal outcomes reported in 'Summary of findings' table).
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Pre‐eclampsia | 1 | 1355 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.84 [0.62, 1.14] |
| 2 Pre‐eclampsia and/or pregnancy loss/stillbirth at any gestational age | 1 | 1355 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.85 [0.67, 1.07] |
| 3 Severe maternal morbidity and mortality index | 1 | 1355 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.97 [0.70, 1.35] |
| 4 Pregnancy loss/stillbirth at any GA | 1 | 1355 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.86 [0.62, 1.21] |
| 5 Caesarean section | 1 | 1355 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.16 [0.96, 1.40] |
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Hofmeyr 2019.
| Methods | Multicentre, parallel, double‐blind randomised trial; individually randomised in 3 countries: South Africa, Zimbabwe, and Argentina | |
| Participants | Parous women with a history of pre‐eclampsia who were intending to become pregnant Exclusion criteria: younger than 18 yrs old; already pregnant; already taking calcium supplements; women with chronic hypertension with persistent proteinuria; women with symptoms or history of urolithiasis, renal or parathyroid disease; women not in a sexual relationship or using long‐term contraception; women unwilling to consent |
|
| Interventions | Intervention: Calcium (N = 678 randomised; 298 pregnancies beyond 20 weeks) – 500 mg elemental calcium tablet (calcium carbonate) taken daily from pre‐pregnancy randomisation until 20 weeks' gestation; then 1.5 mg calcium daily for duration of pregnancy. Women were asked to take calcium tablets separately from any food or iron supplements they may be taking. Comparator: Placebo (N = 677 randomised; 283 pregnancies beyond 20 weeks) – sucrose tablet identical to the calcium tablet above, taken daily to 20 weeks' gestation; then 1.5 mg calcium table daily for duration of pregnancy |
|
| Outcomes |
Primary outcome: Pre‐eclampsia (PE), defined as gestational hypertension (HT) and gestational proteinuria, as defined below, or diagnosed by the attending clinicians Secondary outcomes:
BP: blood pressure; HT: hypertension; ICU: intensive care unit; HELLP syndrome: haemolysis, elevated liver enzymes and low platelet count † severe pre‐eclamptic complications index: severe PE, early onset PE (< 32 weeks’ gestation) eclampsia, HELLP syndrome, placental abruption, severe gestational HT †† severe maternal morbidity and mortality index: maternal admission to intensive care, eclampsia, severe PE, placental abruption, HELLP syndrome, renal failure, death |
|
| Notes |
Sources of funding: The University of British Columbia, a grantee of the Bill & Melinda Gates Foundation; UNDP/UNFPA/UNICEF/WHO/World Bank Special Programme of Research, Development and Research Training in Human Reproduction, World Health Organization; the Argentina Fund for Horizontal Cooperation of the Argentinean Ministry of Foreign Affairs; and the Centre for Intervention Science in Maternal and Child Health (CISMAC). Declarations of interest: We declare no competing interests. Midway through the study, the study team was approached by Alternative Discovery & Development, GlaxoSmithKline (GSK) Medicines Research Centre, UK, who partnered with us to collect blood samples from a subgroup of participants in our trial for an independent, open‐innovation pre‐eclampsia biomarker study, following a separate protocol, which was approved by the trial ethics committee. Apart from direct funding to the largest site (Chris Hani Baragwanath Hospital) specifically for the costs of this blood sample collection, GSK provided no funding to the main trial, and did not participate in any aspect of the main trial. Trial dates: 12 July 2011 to 31 October 2017 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Computer‐generated sequence in blocks of varied size stratified by site; sequence generated remotely (at WHO in Geneva) |
| Allocation concealment (selection bias) | Low risk | Allocation using an online service administered remotely (hosted by WHO); the system allocated the next available treatment pack number according to the site's individual supply of identical, numbered treatment packs |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Participants and providers were blinded |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Outcomes assessors were blinded to group allocation |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Loss to follow‐up before conception was very high in both groups: 157/678 (23%) in calcium group; 163/677 (24%) in placebo group). The numbers lost to follow‐up were substantial, considering that a similar number of women in each group were known not to have conceived (149/678 in calcium group, 167/677 in placebo group). Although the loss to follow‐up pre‐conception was balanced between groups, this substantial attrition could have introduced bias into the reported results (which were reported only for women who were known to have conceived), because it is possible that conception rates were not similar between the groups for whom outcomes are unknown. Loss to follow‐up after conception was minimal (n = 2) and documented in text |
| Selective reporting (reporting bias) | Low risk | Data published according to published protocol. |
| Other bias | Unclear risk | We note the following:
|
Hb: haemoglobin Cu: copper Fe: iron
Mn: manganese IU: international units mg: milligram mcg: microgram SOD: superoxidedismutase Zn: zinc
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Rumiris 2006 | The intervention was calcium plus antioxidants and a wide range of other micronutrients versus placebo (all women received iron and folic acid). It was not possible to establish the effects of calcium, as opposed to the effects of other elements of the combined intervention, from this comparison. |
Characteristics of ongoing studies [ordered by study ID]
Fawzi 2017.
| Trial name or title | From Clinical trials.gov Non‐inferiority of Lower Dose Calcium Supplementation During Pregnancy Official title: Demonstrating non‐inferiority of lower dose calcium supplementation during pregnancy for reducing pre‐eclampsia and neonatal outcomes From CTRI: Public title: Non‐inferiority of lower dose calcium supplementation during pregnancy Scientific title: A cluster‐randomised trial to demonstrate the equivalency of lower dose calcium supplementation during pregnancy for reducing preeclampsia and preterm birth |
| Methods | The 2 trial registrations report slightly different things. 1 states the trial is a cluster‐randomised trial across at least 2 sites (CTRI); the other registration states there is parallel assignment (Clinical trials.gov). It is possible that they are running 2 trials, 1 individually randomised trial at each site. Quadruple masking (participant, care provider, investigator, outcome assessor) Study locations: India, Tanzania |
| Participants | Inclusion criteria: Nulliparous pregnant women aged 18 to 40 years, attending first ANC visit < 20 weeks' gestation Intending to stay in area until 6 weeks post‐delivery Provide informed consent Exclusion criteria: History, or signs, or symptoms (or a combination) of nephrolithiasis Prior diagnosis of parathyroid disorder or thyroidectomy Diseases that require digoxin, phenytoin, or tetracycline therapy |
| Interventions | Experimental: daily 500 mg calcium. 3 tablets containing 500 mg elemental calcium as calcium carbonate and 2 placebo supplements daily (total of 500 mg daily). The supplements in India will also contain 83.3 IU each of vitamin D3, for a total of 250 IU daily. No vitamin D3 will be given in Tanzania. Active comparator: daily 1500 mg calcium (standard dose). The supplements in India will also contain 83.3 IU each of vitamin D3, for a total of 250 IU daily. No vitamin D3 will be given in Tanzania. |
| Outcomes | 1. Proportion of pregnant women with incident pre‐eclampsia (Time frame: gestational week 20 to delivery) 2. Proportion of preterm birth Time frame: birth) |
| Starting date | 1 July 2018 |
| Contact information | Wafaie W Fawzi, MBBS, DrPH 617‐432‐5299 Harvard School of Public Health, Boston, mina@hsph.harvard.edu Prof Anura V Kurpad, St Johns National Academy of Health Sciences, Bangalore, a.kurpad@sjri.res.in Dr Pratibha Dwarkanath, pratibha@sjri.res.in Andreas Pembe, andreapembe@yahoo.co.uk Honorati Masanja, hmasanja@ihi.or.tz |
| Notes | Trial identification: CTRI/2018/02/012119 ClinicalTrials.gov Identifier: NCT03350516 Funders: Bill and Melinda Gates Foundation, USA Harvard TH Chan School of Public Health |
Differences between protocol and review
There are differences between our published protocol and the full review (Hofmeyr 2014b): please see below for further details.
Scope – the review no longer addresses food fortification with calcium for preventing hypertensive disorders of pregnancy, because we believe that food fortification is best understood as a public health intervention, which raises much wider concerns than an intervention in pregnancy.
Methods – subgroup analysis, and investigation of heterogeneity – we added a further subgroup analysis: Calcium commenced before pregnancy or in early pregnancy (< 13 weeks).
Methods – outcomes – we changed the outcome 'pregnancy loss before 24 weeks' gestational age' to 'pregnancy loss before 20 weeks' gestational age' for consistency with the generally accepted definition of early pregnancy loss. We also added an additional secondary outcome 'Neonate small‐for‐gestational age' (but no data were available for this version of the review).
Methods – types of interventions ‐ we broadened our types of interventions to also include calcium supplementation in combination with other supplements or treatments, compared with the same additional supplements or treatments as in the calcium group
Sensitivity analysis – we were concerned that the reported results for Hofmeyr 2019 could have been biased, because the trial did not include all the women randomised (instead, conclusions were conditional on the knowledge that women reached the intermediate outcome of having conceived and reached 20 weeks' gestation). Therefore, we included a sensitivity analysis (comparison 2) that was not prespecified, in which the maternal outcomes reported in the 'Summary of findings' table were analysed using the total number of women randomised in each group as denominators.
Results – included studies ‐ in the previous published version of this review (Hofmeyr 2017), we included Rumiris 2006. This trial compared calcium plus other micronutrients versus placebo. We have now excluded this trial, because it does not support conclusions on the effects of calcium as distinct from the effects of other micronutrients.
We added an additional search of ClinicalTrials.gov and the WHO International Clinical Trials Registry Platform (ICTRP). We updated our methods to be in line with the standard methods of Cochrane Pregnancy and Childbirth, including the use of GRADE methodology and 'Summary of findings' tables.
Two new review authors (Nancy Medley and Myfanwy Williams) joined the team for this update.
Contributions of authors
GJ Hofmeyr (GJH) conceived the review, revised the protocol, responded to reviewers, and wrote the first draft of the review. S Manyame wrote the first draft of the protocol and revised the final review. Both review authors assessed studies for inclusion, and worked on the current update. Nancy Medley and Myfanwy Williams (MW) both assessed the Hofmeyr 2019 study, and extracted data from this study, as well as undertaking the GRADE assessments for the 'Summary of findings' table. MW also contributed to revisions of the final draft of the review.
Sources of support
Internal sources
-
Eastern Cape Department of Health, South Africa.
Salary support (GJH)
External sources
No sources of support supplied
Declarations of interest
Justus Hofmeyr is author of one study included in the review and did not participate in decisions regarding this study (Hofmeyr 2019). Two of the other review authors, who are not directly involved with the trial, evaluated the trial for inclusion, assessed risk of bias, and carried out data extraction and GRADE assessments. Alternative Discovery & Development, GlaxoSmithKline (GSK) Medicines Research Centre, UK, partnered with the same study to collect blood samples from a sub‐group of participants in the trial for an independent, open‐innovation pre‐eclampsia biomarker study, following a separate protocol, which was approved by the trial ethics committee. Apart from direct funding to the largest site (Chris Hani Baragwanath Hospital) specifically for the costs of this blood sample collection, GSK provided no funding to the main trial, and did not participate in any aspect of the main trial.
Sarah Manyame is an investigator on Hofmeyr 2019; however, she was not involved in any decisions relating to the trial. Two of the other review authors, who are not directly involved with the trial, evaluated the trial for inclusion, assessed risk of bias, and carried out data extraction and GRADE assessments. Alternative Discovery & Development, GlaxoSmithKline (GSK) Medicines Research Centre, UK, partnered with the same study to collect blood samples from a sub‐group of participants in the trial for an independent, open‐innovation pre‐eclampsia biomarker study, following a separate protocol, which was approved by the trial ethics committee. Apart from direct funding to the largest site (Chris Hani Baragwanath Hospital) specifically for the costs of this blood sample collection, GSK provided no funding to the main trial, and did not participate in any aspect of the main trial.
Nancy Medley's contribution to this review was financially supported by the World Health Organization.
Myfanwy J Williams is employed by the University of Liverpool as a Research Associate for Cochrane Pregnancy and Childbirth. Her role is supported by the World Health Organization.
New search for studies and content updated (conclusions changed)
References
References to studies included in this review
Hofmeyr 2019 {published data only}
- Hofmeyr GJ. Pre‐pregnancy and early pregnancy calcium supplementation among women at high risk of pre‐eclampsia: a multicentre, double‐blind randomised, placebo‐controlled trial [personal communication]. email to: F Kellie 11 June 2018. [DOI] [PMC free article] [PubMed]
- Hofmeyr GJ. Protocol 11PRT/4028: Long term calcium supplementation in women at high risk of pre‐eclampsia: a randomised, placebo‐controlled trial (PACTR201105000267371). www.thelancet.com/protocol‐reviews/11PRT‐4028 2011.
- Hofmeyr GJ, Betrán AP, Singata‐Madliki M, Cormick G, Munjanja SP, Fawcus S, et al. Calcium and Pre‐eclampsia Study Group. Pre‐pregnancy and early pregnancy calcium supplementation among women at high risk of pre‐eclampsia: a multicenter, double‐blind, randomised, placebo‐controlled trial. Lancet 2019;393(10169):330‐9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hofmeyr GJ, Seuc AH, Betrán AP, Purnat TD, Ciganda A, Munjanja SP, et al. The effect of calcium supplementation on blood pressure in non‐pregnant women with previous pre‐eclampsia: an exploratory, randomized placebo controlled study. Pregnancy Hypertension 2015;5(4):273‐9. [DOI] [PubMed] [Google Scholar]
- Lawrie TA, Betran AP, Singata‐Madliki M, Ciganda A, Hofmeyr GJ, Belizan JM, et al. Participant recruitment and retention in longitudinal preconception randomized trials: lessons learnt from the calcium and pre‐eclampsia (CAP) trial. Trials 2017;18(1):500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- PACTR201105000267371. WHO randomized trial of calcium supplementation before pregnancy to reduce recurrent pre‐eclampsia. pactr.org/ATMWeb/appmanager/atm/atmregistry?dar=true&tNo=PACTR201105000267371 (first received 6 December 2010).
References to studies excluded from this review
Rumiris 2006 {published data only}
- Rumiris D, Purwosunu Y, Wibowo N, Farina A, Sekizawa A. Lower rate of pre‐eclampsia after antioxidant supplementation in pregnant women with low antioxidant status. Hypertension in Pregnancy 2006;25(3):241‐53. [DOI] [PubMed] [Google Scholar]
References to ongoing studies
Fawzi 2017 {published data only}
- CTRI/2018/02/012119. A cluster randomized trial to demonstrate the equivalency of lower dose calcium supplementation during pregnancy for reducing preeclampsia and preterm birth. ctri.nic.in/Clinicaltrials/pmaindet2.php?trialid=21313 (first received 23 February 2018).
- NCT03350516. Demonstrating non‐inferiority of lower dose calcium supplementation during pregnancy for reducing preeclampsia and neonatal outcomes. clinicaltrials.gov/ct2/show/record/NCT03350516 (first received 22 November 2017).
Additional references
Belizan 1980
- Belizan JM, Villar J. The relationship between calcium intake and edema‐, proteinuria‐, and hypertension‐getosis: an hypothesis. American Journal of Clinical Nutrition 1980;33:2202‐10. [DOI] [PubMed] [Google Scholar]
Buppasiri 2015
- Buppasiri P, Lumbiganon P, Thinkhamrop J, Ngamjarus C, Laopaiboon M, Medley N. Calcium supplementation (other than for preventing or treating hypertension) for improving pregnancy and infant outcomes. Cochrane Database of Systematic Reviews 2015, Issue 2. [DOI: 10.1002/14651858.CD007079.pub3] [DOI] [PMC free article] [PubMed] [Google Scholar]
Centeno 2009
- Centeno V, Barboza GD, Marchionatti A, Rodriguez V, Talamoni NT. Molecular mechanisms triggered by low‐calcium diets. Nutritional Research Reviews 2009;22:163‐74. [DOI] [PubMed] [Google Scholar]
De‐Regil 2016
- De‐Regil LM, Palacios C, Lombardo LK, Peña‐Rosas JP. Vitamin D supplementation for women during pregnancy. Cochrane Database of Systematic Reviews 2016, Issue 1. [DOI: 10.1002/14651858.CD008873.pub3] [DOI] [PubMed] [Google Scholar]
GRADE Handbook
- Schünemann H, Brożek J, Guyatt G, Oxman A, editor(s). Handbook for grading the quality of evidence and the strength of recommendations using the GRADE approach (updated October 2013). GRADE Working Group, 2013. Available from gdt.guidelinedevelopment.org/app/handbook/handbook.html.
GRADEpro GDT [Computer program]
- McMaster University (developed by Evidence Prime). GRADEpro GDT. Version accessed 20 July 2018. Hamilton (ON): McMaster University (developed by Evidence Prime), 2015.
Griffith 1999
- Griffith LE, Guyatt GH, Cook RJ, Bucher HC, Cook DJ. The influence of dietary and non‐dietary calcium supplementation blood pressure: an updated meta‐analysis of randomized controlled trials. American Journal of Hypertension 1999;12(1):84‐92. [DOI] [PubMed] [Google Scholar]
Hamlin 1952
- Hamlin RH. The prevention of eclampsia and pre‐eclampsia. Lancet 1952;1:64‐8. [DOI] [PubMed] [Google Scholar]
Higgins 2011
- Higgins JPT, Green S, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from handbook.cochrane.org.
Hofmeyr 2007
- Hofmeyr G, Duley L, Atallah A. Dietary calcium supplementation for prevention of pre‐eclampsia and related problems: a systematic review and commentary. BJOG: an International Journal of Obstetrics and Gynaecology 2007;114:933‐43. [DOI] [PubMed] [Google Scholar]
Hofmeyr 2008
- Hofmeyr GJ, Mlokoti Z, Nikodem VC, Mangesi L, Ferreira S, Singata M, et al. Calcium supplementation during pregnancy for preventing hypertensive disorders is not associated with changes in platelet count, urate, and urinary protein: a randomized control trial. Hypertension in Pregnancy 2008;27:299‐304. [DOI] [PubMed] [Google Scholar]
Hofmeyr 2014
- Hofmeyr G, Belizán J, Dadelszen P, the Calcium and Pre‐eclampsia (CAP) Study Group. Low‐dose calcium supplementation for preventing pre‐eclampsia: a systematic review and commentary. BJOG: an International Journal of Obstetrics and Gynaecology 2014;121(8):951‐7. [DOI: 10.1111/1471-0528.12613] [DOI] [PMC free article] [PubMed] [Google Scholar]
Hofmeyr 2018
- Hofmeyr GJ, Lawrie TA, Atallah ÁN, Torloni MR. Calcium supplementation during pregnancy for preventing hypertensive disorders and related problems. Cochrane Database of Systematic Reviews 2018, Issue 10. [DOI: 10.1002/14651858.CD001059.pub5] [DOI] [PMC free article] [PubMed] [Google Scholar]
Jarjou 2010
- Jarjou LM, Laskey MA, Sawo Y, Goldberg GR, Cole TJ, Prentice A. Effect of calcium supplementation in pregnancy on maternal bone outcomes in women with a low calcium intake. American Journal of Clinical Nutrition 2010;92(2):450‐7. [DOI] [PMC free article] [PubMed] [Google Scholar]
Li 2012
- Li K, Kaaks R, Linseisen J, Rohrmann S. Associations of dietary calcium intake and calcium supplementation with myocardial infarction and stroke risk and overall cardiovascular mortality in the Heidelberg cohort of the European Prospective Investigation into Cancer and Nutrition study (EPIC‐Heidelberg). Heart 2012;98:920‐5. [DOI] [PubMed] [Google Scholar]
Lyall 2013
- Lyall F, Robson SC, Bulmer JN. Spiral artery remodeling and trophoblast invasion in preeclampsia and fetal growth restriction: relationship to clinical outcome. Hypertension 2013;62(6):1046‐54. [DOI] [PubMed] [Google Scholar]
Magee 2014
- Magee LA, Pels A, Helewa M, Rey E, Dadelszen P, on behalf of the Canadian Hypertensive Disorders of Pregnancy (HDP) Working Group. Diagnosis, evaluation, and management of the hypertensive disorders of pregnancy. Pregnancy Hypertension 2014;4:105–45. [DOI] [PubMed] [Google Scholar]
Mol 2016
- Mol BW, Roberts CT, Thangaratinam S, Magee LA, Groot CJ, Hofmeyr GJ. Pre‐eclampsia. Lancet 2016;387:999‐1011. [DOI] [PubMed] [Google Scholar]
Palei 2013
- Palei AC, Spradley FT, Warrington JP, George EM, Granger JP. Pathophysiology of hypertension in pre‐eclampsia: a lesson in integrative physiology. Acta Physiologica (Oxford, England) 2013;208(3):224‐33. [DOI] [PMC free article] [PubMed] [Google Scholar]
Redman 2014
- Redman CW, Sargent IL, Staff AC. IFPA Senior Award Lecture: making sense of pre‐eclampsia – two placental causes of preeclampsia?. Placenta 2014;35 Suppl:S20‐5. [DOI] [PubMed] [Google Scholar]
Review Manager 2014 [Computer program]
- Nordic Cochrane Centre, The Cochrane Collaboration. Review Manager 5 (RevMan 5). Version 5.3. Copenhagen: Nordic Cochrane Centre, The Cochrane Collaboration, 2014.
Villar 2006
- Villar J, Abdel‐Aleem H, Merialdi M, Mathai M, Ali MM, Zavaleta N, et al. World Health Organization randomized trial of calcium supplementation among low calcium intake pregnant women. American Journal of Obstetrics and Gynecology 2006;194:639‐49. [DOI] [PubMed] [Google Scholar]
von Dadelszen 2016
- Dadelszen P, Magee LA. Preventing deaths due to the hypertensive disorders of pregnancy. Best Practice & Research Clinical Obstetrics & Gynaecology 2016;36:83‐102. [DOI] [PMC free article] [PubMed] [Google Scholar]
von Dadelzsen 2004
- Dadelszen P, Magee LA, Devarakonda RM, Hamilton T, Ainsworth LM, Yin R, et al. The prediction of adverse maternal outcomes in preeclampsia. Journal of Obstetrics and Gynaecology Canada: JOGC 2004;26:871‐9. [DOI] [PubMed] [Google Scholar]
References to other published versions of this review
Hofmeyr 2014b
- Hofmeyr GJ, Manyame S. Calcium supplementation commencing before or early in pregnancy, or food fortification with calcium, for preventing hypertensive disorders of pregnancy. Cochrane Database of Systematic Reviews 2014, Issue 8. [DOI: 10.1002/14651858.CD011192] [DOI] [PMC free article] [PubMed] [Google Scholar]
Hofmeyr 2017
- Hofmeyr GJ, Manyame S. Calcium supplementation commencing before or early in pregnancy, or food fortification with calcium, for preventing hypertensive disorders of pregnancy. Cochrane Database of Systematic Reviews 2017, Issue 9. [DOI: 10.1002/14651858.CD011192.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]


