Abstract
This analysis assesses the levels of the carcinogen pulegone in top-marketed brands of e-cigarettes and smokeless tobacco products.
Pulegone, a constituent of oil extracts prepared from mint plants, including peppermint, spearmint and pennyroyal, is a carcinogen that causes hepatic carcinomas, pulmonary metaplasia, and other neoplasms on oral administration in rodents.1 In 2018, the US Food and Drug Administration (FDA) banned synthetic pulegone as a food additive.2 Studies by the Centers for Disease Control and Prevention (CDC) detected substantial amounts of pulegone in mint- and menthol-flavored e-cigarette liquids and smokeless tobacco products marketed in the United States.3,4 The tobacco industry has minimized pulegone levels in cigarette flavorings because of toxicity concerns. Mint- and menthol-flavored e-cigarettes may be exempt from proposed federal regulations; therefore, the health risk associated with pulegone in these products should be considered.
Methods
To assess the risk associated with pulegone, we calculated the margin of exposure (MOE) by dividing the FDA-provided no-observed adverse effect level (NOAEL) of pulegone at which no treatment-related tumors were reported in animal studies (13.39 mg/kg of body weight/d)1,2 by the mean human exposure from mint- and menthol-flavored e-cigarette or smokeless tobacco products analyzed in the CDC studies.3,4 The MOE is the measure used by the FDA and other regulatory agencies for cancer risk assessment of food additives.2 Cancer risk is inversely proportional to the MOE, with values of 10 000 or below requiring mitigation strategies.2 We compared the risk associated with pulegone content in top-marketed brands of combustible menthol cigarettes to mint- and menthol-flavored e-cigarettes (5 e-liquids, 3 brands) and smokeless tobacco (1 brand).3,4,5 Based on daily use, we considered a light user to consume 5 mL of e-liquid, half a pack of cigarettes, or 10 g of smokeless tobacco; a moderate user to consume 10 mL of e-liquid, 1 pack of cigarettes, or 20 g of smokeless tobacco; and a heavy user to consume 20 mL of e-liquid, 2 packs of cigarettes, or 30 g of smokeless tobacco. This study was exempt from Duke University School of Medicine institutional review board approval following tenets of the Principal Investigator Checklists as not involving human participants.
Results
For the e-liquid with the highest pulegone concentration, MOEs were between 1298 and 3084 for 5-mL daily consumption and between 325 and 771 for 20-mL daily consumption (Table and Figure), below the safety threshold of 10 000. Depending on consumption rates, MOEs for the e-liquids ranged between 325 and 6012; for a pulegone-containing smokeless tobacco they ranged between 549 and 1646.
Table. Predicted MOE for Top-Marketed Pulegone-Containing Smokeless Tobacco and e-Cigarette Liquids Used in Vape Tanks and Vape Mod Devices.
| Type and Flavora | Pulegone Concentration (μg/g) | MOE for Daily e-Liquid (mL/d) or Smokeless Tobacco (g/d) Amount Consumedb | Fold Increase in Daily Exposure Compared With Menthol Cigarette Usec | |||
|---|---|---|---|---|---|---|
| 5 | 10 | 20 | 30 | |||
| e-Cigarette | ||||||
| V2 Menthol, range | 50.1-119.0 | 1298-3084 | 649-1542 | 325-771 | NA | 86-1608 |
| V2 Peppermint, range | 78.3-82.7 | 1868-1973 | 934-987 | 467-493 | NA | 136-1118 |
| Premium Menthol | 115.0 | 1344 | 672 | 336 | NA | 198-1554 |
| South Beach Smoke Menthol | 28.2 | 5479 | 2739 | 1370 | NA | 48-282 |
| South Beach Smoke Peppermint | 25.7 | 6012 | 3006 | 1503 | NA | 44-348 |
| Smokeless Tobacco | ||||||
| Skoal Xtra Mint snuff | 48.8 | NA | 1646 | 823 | 549 | 168-1319 (Light users)/126-990 (heavy users)d |
Abbreviation: MOE, margin of exposure.
V2 products were manufactured by VMR Products LLC; Premium products are manufactured by PremiumEStore LLC; South Beach Smoke products are manufactured by southbeachsmoke.com; and Skoal products are produced by the US Smokeless Tobacco Company.
The no observed adverse effect level for pulegone is 13.39 mg/kg of body weight/d.1,2 User body weight was assumed to be 60 kg; e-Liquid specific density equals 1.04 g/cm3.
Menthol cigarettes were estimated to contain 0.037 to 0.290 μg/g.5 Each cigarette was considered to have 1 g of tobacco; 1 pack equals 20 cigarettes.
Based on daily use, a light user is considered to consume 10 g of smokeless tobacco; a moderate user to consume 20 g of smokeless tobacco; and a heavy user to consume 30 g of smokeless tobacco.
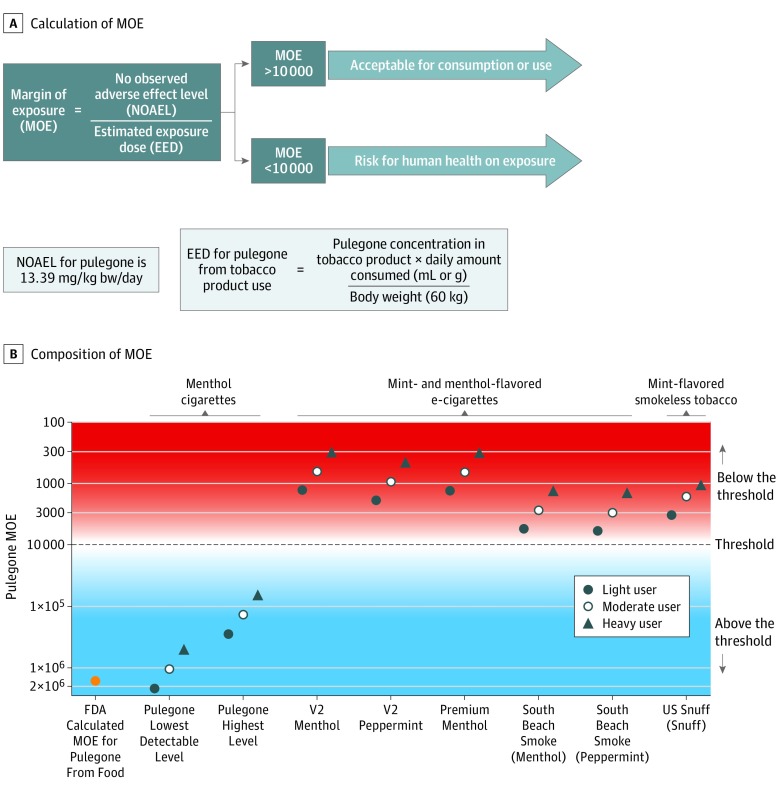
Figure: Margin of Exposure (MOE) Calculation Method and MOE Comparison for Various Tobacco Products.
A, Calculation of MOE for a potential carcinogenic or genotoxic chemical or food additive.1,2 An MOE greater than 10 000 is applied by the US Food and Drug Administration (FDA) and other regulators as threshold for mitigation of potential carcinogenic activity of food additives.2 B, Comparison of the pulegone predicted MOE from tobacco products use (e-cigarette liquids for vape tanks and vape mod devices, smokeless tobacco, and menthol cigarette) to the FDA’s calculated MOE for pulegone from food intake. The MOEs within each tobacco product were also compared based on amount of tobacco product consumed. The MOEs for menthol cigarette use were calculated from the upper and lower level of pulegone range detected in 23 top-marketed brands (estimated to contain 0.037-0.290 μg/g of pulegone).5
Daily pulegone exposure from e-cigarettes compared with menthol cigarette use (estimated to contain 0.037-0.290 μg/g of pulegone)5 was higher across all user groups (ranging from 44-1608 times higher). Compared with menthol cigarettes, estimated pulegone intake from smokeless tobacco was 168 to 1319 times higher in light users and 126 to 990 times higher in heavy users.
Discussion
Our analysis suggests that users of mint- and menthol-flavored e-cigarettes and smokeless tobacco are exposed to pulegone levels higher than the FDA considers unacceptable for intake of synthetic pulegone in food, and higher than in smokers of combustible menthol cigarettes.2,5
Inhalation toxicity data for pulegone are not available. Although carcinogenic or systemic risks associated with pulegone may vary by exposure route (inhalation or ingestion), extrapolation of MOEs derived from oral toxicity studies to inhalation exposure is a common practice among regulatory agencies, with inhalation exposure accepted to increase risk by 2-fold or higher.6 To limit complexity, we did not use extrapolations in this analysis. However, if we had applied the oral-to-inhalation extrapolation factor of 2 to the MOE calculations for pulegone, the MOEs for the e-liquids would have been reduced by half, resulting in exposures even further from the MOE safety threshold of 10 000.
Limitations of this study include consideration of only 5 e-liquids and 1 smokeless tobacco product for which analytical data were published, and following the FDA’s risk assessment procedures, which are based on animal data. Although pulegone is toxic in humans, it is unknown if users of combustible tobacco products, smokeless tobacco, or e-cigarettes absorb and metabolize the quantities associated with production of a carcinogenic effect. Nevertheless, the MOEs for all the products we analyzed are below the accepted MOE threshold of 10 000 for carcinogens. Our findings appear to establish health risks associated with pulegone intake and concerns that the FDA should address before suggesting mint- and menthol-flavored e-cigarettes and smokeless tobacco products as alternatives for people who use combustible tobacco products.
References
- 1.National Toxicology Program Toxicology and carcinogenesis studies of pulegone (CAS No. 89-82-7) in F344/N rats and B6C3F1 mice (gavage studies). Natl Toxicol Program Tech Rep Ser. 2011;(563):1-201. [PubMed] [Google Scholar]
- 2.Synthetic flavoring agents and adjuvants. Fed Regist. 2018;83(195):50490-50503. [Google Scholar]
- 3.Lisko JG, Tran H, Stanfill SB, Blount BC, Watson CH. Chemical composition and evaluation of nicotine, tobacco alkaloids, pH, and selected flavors in e-cigarette cartridges and refill solutions. Nicotine Tob Res. 2015;17(10):1270-1278. doi: 10.1093/ntr/ntu279 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Lisko JG, Stanfill SB, Watson CH. Quantitation of ten flavor compounds in unburned tobacco products. Anal Methods. 2014;6(13):4698-4704. doi: 10.1039/C4AY00271G [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Stanfill SB, Ashley DL. Solid phase microextraction of alkenylbenzenes and other flavor-related compounds from tobacco for analysis by selected ion monitoring gas chromatography-mass spectrometry. J Chromatogr A. 1999;858(1):79-89. doi: 10.1016/S0021-9673(99)00796-7 [DOI] [PubMed] [Google Scholar]
- 6.Schröder K, Escher SE, Hoffmann-Dörr S, Kühne R, Simetska N, Mangelsdorf I. Evaluation of route-to-route extrapolation factors based on assessment of repeated dose toxicity studies compiled in the database RepDose. Toxicol Lett. 2016;261:32-40. doi: 10.1016/j.toxlet.2016.08.013 [DOI] [PubMed] [Google Scholar]