Abstract
Background/Aims
The aim of this study is to report the burden of ocular morbidity following iodine-125 episcleral plaque brachytherapy (EPBT) in the treatment of American Joint Committee on Cancer (AJCC) T4-staged posterior uveal melanoma (PUM).
Methods
Clinical records of patients with T4-staged PUM treated with 125I EPBT were analyzed for incidence of treatment failure and radiation-induced complications.
Results
Cumulative incidence of local treatment failure was 9% (95% CI 5–15%) at 5 years and was associated with decreased tumor height (HR = 0.78; p = 0.01). Cumulative incidence of enucleation at 5 years was 21% and was correlated with worsening baseline visual acuity (HR = 1.42; p = 0.05). Increasing patient age was associated with higher rates of vitreous hemorrhage (HR = 1.03; p = 0.02) and cataract surgery (HR = 1.05; p < 0.001). Increased tumor height was associated with higher rates of neovascular glaucoma (HR = 1.16; p = 0.03) and vitreous hemorrhage (HR = 1.23; p < 0.001).
Conclusion
125I EPBT is an effective treatment for T4-staged PUM and achieves high rates of local control. Treatment failure appears to be more common among minimally elevated tumors. Other causes of ocular morbidity were associated with increasing tumor height, patient age, and baseline visual acuity.
Keywords: Uveal melanoma, Brachytherapy, Local control, Prognostic factor
Introduction
In 2009, the American Joint Committee on Cancer (AJCC) established the first universally accepted staging classification system for uveal melanoma [1]. Based on initial height, basal diameter measurements, and location, tumors are stratified into 4 separate categories (T1 through T4), each conferring a higher risk of metastatic disease and mortality. The previous classification scheme established by the Collaborative Ocular Melanoma Study (COMS) existed primarily to identify which tumors could safely undergo globe-sparing treatment with episcleral plaque brachytherapy (EPBT) with no significant decrease in survival based on the results of a prospective randomized controlled trial [2]. The largest category of tumors in the COMS study (height > 10.0 mm or basal diameter > 16.0 mm) were randomized to either primary enucleation or external beam radiation followed by enucleation. The study was stopped early as there was no observed difference in cohorts [3]. The reported overall mortality at 5 years was 57 and 62% in the enucleation and pre-enucleation radiation treatment arms, respectively [3].
Over the past 2 decades, a growing number of treatment centers have begun to offer conservative therapy for large melanomas which historically would have required enucleation. Multiple case series have demonstrated safety and efficacy for both EPBT and proton beam radiation therapy in treating large tumors [4, 5, 6]. Many clinicians may still hesitate to offer plaque brachytherapy to these patients primarily because complications associated with EPBT occur with greater frequency among large melanomas [7, 8, 9, 10] and likely result in higher enucleation rates [11]. Current recommendations from the American Brachytherapy Society – Ophthalmic Oncology Task Force (ABS-OOTF) indicate that AJCC T4-staged melanomas can be treated with iodine brachytherapy provided there is no evidence of extraocular extension [12]. However, due to limitations in the dose distribution of iodine-125, patients with tumors greater than 12 mm in height or 20 mm in base should be “counseled regarding alternative therapies” according to the ABS-OOTF guidelines. In our experience, many of these patients are reluctant to proceed with enucleation and may still desire conservative therapy.
To our knowledge, only 3 centers have reported long-term clinical outcomes with iodine brachytherapy for “large melanomas” [13, 14, 15], and no publication has specifically addressed conservative therapy for T4-staged posterior uveal melanoma (PUM) exclusively. Our objective for this investigation is to report our single institution experience in treating T4-staged PUM with 125I brachytherapy by quantifying survival as well as the incidence of ocular morbidity following treatment. We hope that our results will further contribute to the existing body of evidence regarding the efficacy and limitations of globe-sparing treatment for advanced tumors.
Methods
Ocular Oncology Database
Following approval by the University of Tennessee Health Science Center's Institutional Review Board, we performed a retrospective chart review of our Health Information Portability and Accountability Act-compliant institutional database for all patients treated between January 1985 and December 2015 for PUM (defined as uveal melanoma involving the choroid or ciliary body). This database contains patient demographic and clinicopathologic data gathered and recorded over the duration of follow-up. All aspects of our investigation were compliant with the Declaration of Helsinki.
Inclusion/Exclusion Criteria for the Current Study
We included all patients with PUM undergoing 125I EPBT whose initial tumor dimensions were consistent with the following AJCC staging criteria for the T4 category: (1) tumor height > 15 mm, or (2) tumor basal diameter > 18 mm, or (3) tumor height > 12 mm and tumor diameter > 15 mm [1]. Patients with blind and painful eyes, extraocular extension, circumpapillary growth, extensive intraocular hemorrhage, or unclear media were considered poor candidates for EPBT and excluded. Patients undergoing primary treatment with any modality other than 125I EPBT were excluded.
Clinical Evaluation
All patients selected for review were diagnosed with PUM between 1985 and 2015 based on clinical examination and diagnostic imaging. Baseline tumor dimensions were measured with B-scan ultrasonography and dilated fundus examination. All patients underwent metastatic screening prior to treatment with chest X-ray, liver imaging (computed tomography or ultrasound), and liver function tests. Patients were offered enucleation or globe-preserving therapy with EPBT. Informed consent was obtained following a detailed discussion of the potential risks and benefits of EPBT with specific emphasis on radiation-related complications. Patients with poor baseline vision were generally advised against conservative therapy but were not categorically excluded.
Surgical Treatment with 125I EPBT
Tumor margins were delineated intraoperatively using transillumination and scleral depression with indirect ophthalmoscopy. Plaque size was selected for a diameter at least 4 mm greater than the maximum basal diameter of the tumor to ensure that the tumor base was fully encompassed by the effective treatment zone. As maximum plaque size is limited to 24 mm, patients with a tumor basal diameter exceeding 20 mm who elected to proceed with EPBT were counseled accordingly regarding potential limitations of therapy. Notched plaques were selected for juxtapapillary melanomas involving less than 180 degrees of the optic nerve head margin. Each plaque was surgically implanted for the duration necessary to achieve a cumulative apex dose of 85 Gy (dosing rate maintained at > 50 cGy per hour) in compliance with the American Association of Physics in Medicine TG-43 recommendations. The targeted tumor apex height was calculated based on ultrasound measurements and adding 1 mm for thickness of the sclera. B-scan ultrasonography was performed on postoperative day 1 to verify that each plaque was well-centered around the tumor base, extending at least 2 mm peripheral to the tumor margin.
Patient Follow-up
Each patient was re-examined every 3–4 months during the first year after treatment and then every 6 months during each subsequent year. The occurrence of the following ocular complications or adverse events was documented prospectively at each appointment:
Enucleation: removal of the eye was indicated for patients with local recurrence, scleromalacia, or blind, painful eyes.
Local recurrence was diagnosed based on definitive tumor growth on ultrasonography or clinical exam.
Cataract removal was indicated for any patient with progressive lens opacification after brachytherapy who was deemed to have significant short-term vision potential. The decision to proceed with surgery was at the discretion of the patient's local ophthalmologist.
Vitreous hemorrhage was diagnosed based on the fundus exam.
Neovascular glaucoma was diagnosed in patients with neovascularization of the iris with intraocular pressure > 21 mm Hg.
Scleromalacia was diagnosed for any patient with a full or partial thickness scleral defect occurring within the plaque treatment zone.
Radiation retinopathy was diagnosed in eyes with exudative changes occurring within the posterior pole, including macular edema, intraretinal hemorrhage, cotton wool spots, or exudates.
Radiation optic neuropathy was diagnosed in eyes with exudative or ischemic changes in the optic nerve.
All patients were concurrently followed by a medical oncologist at 6-month intervals for metastatic disease surveillance, and the occurrence of metastatic relapse was verified by medical record correspondence from the oncologist. Patient mortality was confirmed by family notification or review of public records in the United States Social Security Death Index (SSDI).
Statistical Analysis
Time-to-event calculations were measured from the day plaque brachytherapy was initiated. Kaplan-Meier analysis was carried out using Prism 6 software (GraphPad Software Inc., La Jolla, CA, USA) to estimate overall and metastasis-free survival at 1-, 3-, and 5-year intervals. Relative survival calculations were conducted according to the method proposed by Dickman et al. [16] using the US Census Bureau gender- and age-specific population estimates data with SAS version 9.4 (SAS Inc., Cary, NC, USA). Once age-, gender-, and follow-up-specific relative survival estimates were obtained for the study cohort, the results were summarized as overall and by gender and age ranges as appropriate.
The incidence of ocular complications was then estimated using the cumulative incidence function, which adjusts Kaplan-Meier estimates in the presence of competing events, such as death before the primary event [17]. Due to the high incidence of mortality and enucleation in this cohort, death or loss of the eye were treated as competing events with each complication. This was calculated with SAS version 9.4 (SAS Inc.). Univariate and multivariate Cox proportional hazard regression analysis was performed with SPSS 24 software (IBM Corp., Armonk, NY, USA) to test the association between the time to each ocular complication occurrence and the following patient- or eye-specific covariates: (1) patient age, (2) tumor maximum basal diameter, (3) tumor height, (4) visual acuity at presentation, (5) ciliary body involvement, and (6) posterior tumor margin location. The Pearson correlation coefficient was used to assess for collinearity between different covariates. To reduce the issue of multicollinearity, each covariate was then separated into 1 of 2 multivariate models controlling for the following: (1) patient age, tumor diameter, tumor height, and posterior margin location, or (2) patient age, tumor diameter, visual acuity, and ciliary body involvement. All visual acuity measurements were converted from Snellen units to logarithm of the minimum angle of resolution (logMAR) units for analysis. There were 4 patients who presented with light perception or no light perception vision who were excluded from proportional hazard regression analysis as visual acuity cannot be quantified in logMAR units.
Results
Of 1,521 patients with PUM treated with EPBT, we identified 158 patients (n = 83 males) who met our inclusion criteria with AJCC T4 tumors. Of the 158 tumors, 58 (37.4%) were predominantly ciliary body tumors and 100 arose from the choroid. Median age at diagnosis was 65 years (range 13–88 years). Median logMAR visual acuity at diagnosis was 0.6 (equivalent to approximately 20/80 in Snellen units). Median tumor height at diagnosis was 10.4 mm (range 3.3–19.4 mm) and median maximum basal diameter was 20.0 mm (range 15.1–29.0 mm). Median duration of follow-up for ocular complications was 28 months (range 0–254 months).
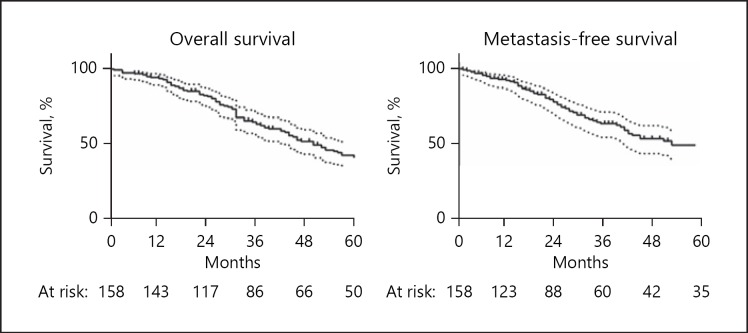
Mortality was confirmed in 103 of 158 patients based on review of public records. Kaplan-Meier estimates for overall survival at 1, 3, and 5 years were 93.6% (95% CI 88.4–96.5), 62.9% (95% CI 54.5–70.2), and 42.0% (95% CI 33.5–50.3), respectively (Fig. 1). Diagnosis of metastatic disease was confirmed in 68 patients with a median time to metastasis of 25 months (0–165 months). Kaplan-Meier estimates for metastasis-free survival at 1, 3, and 5 years were 91.7% (95% CI 85.9–95.2%), 61.6% (95% CI 52.0–69.8%), and 46.8% (95% CI 36.5–56.4%), respectively (Fig. 1). Relative survival analysis was performed to quantify the extent of melanoma-related mortality compared with age- and gender-matched controls. Cumulative relative survival at 1, 3, and 5 years was 97.4, 77.5, and 75.6%, respectively. There was a trend toward increased relative survival among younger patients. Five-year relative survival in patients < 65 years old was 81.3% compared with 64.7% in patients ≥65 years old (Wilcoxon-Mann-Whitney test, p = 0.10). There was no significant difference in 5-year relative survival between males and females at 76.2 and 75.2%, respectively (Wilcoxon-Mann-Whitney test, p = 0.39).
Fig. 1.
Kaplan-Meier survival curves for overall survival and metastasis-free survival in patients with T4-staged posterior uveal melanoma treated with iodine-125 episcleral plaque brachytherapy. Dotted lines indicate 95% confidence intervals.
Local recurrence was diagnosed in 12 patients with a median time to recurrence of 15 months (range 5–33 months). Of those 12 patients, 9 were treated with enucleation, 2 refused subsequent treatment, and 1 underwent transpupillary thermotherapy. Adjusting for mortality and enucleation as competing events, cumulative incidence of local recurrence at 1, 3, and 5 years was 1% (95% CI 0–4%), 9% (95% CI 5–15%), and 9% (95% CI 5–15%), respectively (Table 1). Cox proportional hazards analysis revealed that local recurrence rate was significantly associated with decreased tumor apex height (HR = 0.78; 95% CI 0.64–0.95; p = 0.01) (Table 2). There was a weak association between local recurrence and increased largest basal diameter (HR = 1.08; 95% CI 0.89–1.32; p = 0.43); however, this was not statistically significant. Eight of 12 patients diagnosed with local recurrence eventually died with metastatic disease.
Table 1.
Cumulative incidence of treatment complications at 1, 3, and 5 years
| Cumulative incidence | 1 year, % (95% CI) | 3 years, % (95% CI) | 5 years, % (95% CI) |
|---|---|---|---|
| Enucleation | 4.7 (2.1–9.0) | 19.9 (13.5–27.3) | 21.0 (14.4–28.6) |
| Local recurrence | 1.3 (0.2–4.3) | 9.0 (4 .9–14 .7) | 9.0 (4 .9–14 .7) |
| Cataract surgery | 14.8 (9 .3–21 .5) | 32.8 (24 .4–41 .4) | 45.0 (35.3–54.2) |
| Vitreous hemorrhage | 15.7 (10 .4–21 .9) | 27.1 (20.0–34.6) | 33.7 (25 .6–41 .9) |
| Neovascular glaucoma | 4.8 (2.1–9.1) | 21.2 (14 .7–28 .6) | 24.2 (17 .1–32 .1) |
| Scleromalacia | 6.6 (3 .4–11 .3) | 11.2 (6.7–17.1) | 12.1 (7.3–18.2) |
| Radiation retinopathy | 10.1 (5 .9–15 .6) | 38.8 (30.6–47.0) | 43.7 (34.9–52.0) |
| Radiation optic neuropathy | 5.5 (2 .6–10 .0) | 23.3 (16 .5–30 .9) | 31.0 (23 .0–39 .3) |
Table 2.
Cox proportional hazard regression analysis of treatment complications following iodine-125 brachytherapy
| β (SE) | p value | Hazard ratio (95% CI) | |
|---|---|---|---|
| Enucleation | |||
| Univariate regression | |||
| Age (at diagnosis) | −0.004 (0.01) | 0.76 | 1.00 (0.97–1.02) |
| Tumor LBD | −0.006 (0.07) | 0.94 | 1.00 (0 .87–1 .14) |
| Tumor height | 0.036 (0.07) | 0.60 | 1.04 (0.91–1.18) |
| LogMAR visual acuity | 0.33 (0.18) | 0.06 | 1.39 (0.99–1.96) |
| Ciliary body involvement | 0.19 (0.38) | 0.62 | 1.21 (0.57–2.56) |
| Posterior margin ≤6 DD from fovea | −0.07 (0.41) | 0.86 | 0.93 (0.41–2.09) |
| Multivariate regression model | |||
| Age (at diagnosis) | −0.01 (0.01) | 0.55 | 0.99 (0.97–1.02) |
| Tumor LBD | 0.04 (0.07) | 0.59 | 1.04 (0.91–1.19) |
| logMAR visual acuity | 0.35 (0.18) | 0.05 | 1.42 (1.00–2.01) |
| Ciliary body involvement | 0.11 (0.41) | 0.78 | 1.12 (0.50–2.49) |
| Local recurrence | |||
| Univariate regression | |||
| Age (at diagnosis) | 0.02 (0.02) | 0.26 | 1.02 (0.98–1.07) |
| Tumor LBD | 0.08 (0.10) | 0.43 | 1.08 (0.89–1.32) |
| Tumor height | −0.25 (0.10) | 0.01 | 0.78 (0.64–0.95) |
| LogMAR visual acuity | −0.13 (0.36) | 0.73 | 0.88 (0.43–1.79) |
| Ciliary body involvement | −0.48 (0.67) | 0.47 | 0.62 (0.17–2.29) |
| Posterior margin ≤6 DD from fovea | −0.83 (0.78) | 0.28 | 0.44 (0.10–1.99) |
| Multivariate regression model | |||
| Age (at diagnosis) | 0.03 (0.02) | 0.20 | 1.03 (0.99–1.07) |
| Tumor LBD | 0.04 (0.12) | 0.77 | 1.04 (0.82–1.30) |
| Tumor height | −0.23 (0.10) | 0.02 | 0.79 (0.66–0.96) |
| Posterior margin ≤6 DD from fovea | −0.85 (0.79) | 0.28 | 0.43 (0.09–2.01) |
| Vitreous hemorrhage | |||
| Univariate regression | |||
| Age (at diagnosis) | 0.03 (0.01) | 0.02 | 1.03 (1.00–1.05) |
| Tumor LBD | 0.03 (0.06) | 0.55 | 1.03 (0.93–1.15) |
| Tumor height | 0.21 (0.06) | <0.001 | 1.23 (1.11–1.37) |
| LogMAR visual acuity | 0.41 (0.13) | 0.002 | 1.51 (1.17–1.97) |
| Ciliary body involvement | 0.04 (0.30) | 0.89 | 1.04 (0.58–1.89) |
| Posterior margin ≤6 DD from fovea | 0.002 (0.32) | 0.99 | 1.00 (0 .54–1 .87) |
| Multivariate regression model | |||
| Age (at diagnosis) | 0.03 (0.01) | 0.01 | 1.03 (1.01–1.05) |
| Tumor LBD | 0.04 (0.05) | 0.39 | 1.04 (0.95–1.15) |
| Tumor height | 0.21 (0.05) | <0.001 | 1.24 (1.11–1.37) |
| Posterior margin ≤6 DD from fovea | −0.28 (0.33) | 0.40 | 0.76 (0.40–1.45) |
| Cataract surgery | |||
| Univariate regression | |||
| Age (at diagnosis) | 0.05 (0.01) | <0.001 | 1.05 (1.03–1.08) |
| Tumor LBD | 0.09 (0.05) | 0.10 | 1.09 (0.99–1.21) |
| Tumor height | 0.03 (0.05) | 0.61 | 1.03 (0.93–1.14) |
| LogMAR visual acuity | −0.06 (0.16) | 0.70 | 0.94 (0.69–1.28) |
| Ciliary body involvement | −0.05 (0.30) | 0.87 | 0.95 (0.53–1.71) |
| Posterior margin ≤6 DD from fovea | 0.40 (0.30) | 0.18 | 1.50 (0.83–2.69) |
| Multivariate regression model | |||
| Age (at diagnosis) | 0.05 (0.01) | <0.001 | 1.05 (1.03–1.08) |
| Tumor LBD | 0.05 (0.05) | 0.27 | 1.06 (0.96–1.16) |
| logMAR visual acuity | −0.05 (0.15) | 0.74 | 0.95 (0.70–1.28) |
| Ciliary body involvement | 0.01 (0.30) | 0.98 | 1.01 (0.56–1.83) |
| Neovascular glaucoma | |||
| Univariate regression | |||
| Age (at diagnosis) | −0.01 (0.01) | 0.45 | 0.99 (0.97–1.01) |
| Tumor LBD | 0.09 (0.06) | 0.14 | 1.10 (0 .97–1 .23) |
| Tumor height | 0.15 (0.07) | 0.03 | 1.16 (1.02–1.33) |
| LogMAR visual acuity | 0.08 (0.19) | 0.67 | 1.08 (0.75–1.56) |
| Ciliary body involvement | −0.46 (0.41) | 0.26 | 0.63 (0.28–1.41) |
| Posterior margin ≤6 DD from fovea | −0.47 (0.43) | 0.28 | 0.63 (0.27–1.45) |
| Multivariate regression model | |||
| Age (at diagnosis) | −0.01 (0.01) | 0.51 | 0.99 (0.97–1.02) |
| Tumor LBD | 0.08 (0.06) | 0.14 | 1.09 (0.97–1.22) |
| Tumor height | 0.18 (0.07) | 0.02 | 1.19 (1.03–1.37) |
| Posterior margin ≤6 DD from fovea | −0.49 (0.45) | 0.27 | 0.61 (0.26–1.46) |
| Scleromalacia | |||
| Univariate regression | |||
| Age (at diagnosis) | 0.03 (0.02) | 0.14 | 1.03 (0.99–1.07) |
| Tumor LBD | 0.06 (0.09) | 0.48 | 1.06 (0.90–1.26) |
| Tumor height | 0.11 (0.09) | 0.21 | 1.12 (0.94–1.34) |
| LogMAR visual acuity | −0.30 (0.30) | 0.33 | 0.74 (0.41–1.35) |
| Ciliary body involvement | 1.02 (0.49) | 0.04 | 2.76 (1.05–7.27) |
| Posterior margin ≤6 DD from fovea | 0.66 (0.49) | 0.18 | 1.93 (0.74–5.00) |
| Multivariate regression model | |||
| Age (at diagnosis) | 0.03 (0.02) | 0.11 | 1.03 (0.99–1.07) |
| Tumor LBD | 0.04 (0.09) | 0.66 | 1.04 (0.87–1.25) |
| logMAR visual acuity | −0.33 (0.30) | 0.28 | 0.72 (0.40–1.30) |
| Ciliary body involvement | 1.11 (0.50) | 0.03 | 3.03 (1.13–8.10) |
| Radiation retinopathy | |||
| Univariate regression | |||
| Age (at diagnosis) | −0.02 (0.01) | 0.07 | 0.99 (0.97–1.00) |
| Tumor LBD | 0.002 (0.05) | 0.97 | 1.00 (0.91–1 .10) |
| Tumor height | −0.10 (0.05) | 0.03 | 0.91 (0.83–0.99) |
| LogMAR visual acuity | −0.13 (0.15) | 0.37 | 0.88 (0.66–1.17) |
| Ciliary body involvement | −0.59 (0.30) | 0.05 | 0.56 (0.31–0.99) |
| Posterior margin ≤6 DD from fovea | −0.16 (0.29) | 0.57 | 0.85 (0.48–1.49) |
| Multivariate regression model | |||
| Age (at diagnosis) | −0.02 (0.01) | 0.10 | 0.99 (0.97–1.00) |
| Tumor LBD | −0.001 (0.05) | 0.99 | 0.99 (0.90–1.11) |
| Tumor height | −0.10 (0.05) | 0.04 | 0.91 (0.83–1.00) |
| Posterior margin ≤6 DD from fovea | −0.03 (0.30) | 0.93 | 0.98 (0.55–1.74) |
| Radiation optic neuropathy | |||
| Univariate regression | |||
| Age (at diagnosis) | −0.01 (0.01) | 0.51 | 0.99 (0.97–1.01) |
| Tumor LBD | 0.04 (0.06) | 0.46 | 1.05 (0.93–1.17) |
| Tumor height | −0.03 (0.06) | 0.66 | 0.98 (0.87–1.10) |
| LogMAR visual acuity | 0.70 (0.16) | 0.67 | 1.07 (0.78–1.48) |
| Ciliary body involvement | −0.73 (0.38) | 0.06 | 0.48 (0.23–1.02) |
| Posterior margin ≤6 DD from optic nerve | 0.87 (0.40) | 0.03 | 2.39 (1.09–5.24) |
| Multivariate regression model | |||
| Age (at diagnosis) | −0.01 (0.01) | 0.68 | 0.99 (0.97–1.02) |
| Tumor LBD | 0.04 (0.06) | 0.48 | 1.04 (0.93–1.17) |
| Tumor height | −0.001 (0.06) | 0.99 | 0.99 (0.89–1.12) |
| Posterior margin ≤6 DD from optic nerve | 0.85 (0.41) | 0.04 | 2.33 (1.05–5.16) |
SE, standard error; CI, confidence interval; LBD, largest basal diameter; DD, disc diameters; LogMAR, logarithm of the minimum angle of resolution.
Thirty-two patients ultimately required enucleation following primary treatment with EPBT. Eighteen of these underwent enucleation due to blind, painful eyes, 14 of which had been diagnosed with neovascular glaucoma. Nine required enucleation for local recurrence, and 5 were enucleated for progressive scleromalacia. Adjusting for mortality as a competing event, cumulative incidence of enucleation at 1, 3, and 5 years was 5% (95% CI 2–9%), 20% (95% CI 14–27%), and 21% (95% CI 14–29%), respectively. Cox proportional hazards analysis revealed a significant association between enucleation frequency and poorer baseline logMAR visual acuity in the multivariate model (HR = 1.42; 95% CI 1.00–2.01).
Neovascular glaucoma accounted for the highest number of enucleations and was diagnosed in 33 patients at a median time of 18 months after brachytherapy (range 4–126 months). Cumulative incidence of neovascular glaucoma at 1, 3, and 5 years was 5% (95% CI 2–9%), 21% (95% CI 15–29%), and 24% (95% CI 17–32%), respectively. Development of neovascular glaucoma was significantly associated with increasing tumor height (HR = 1.16; 95% CI 1.02–1.33). Scleromalacia was diagnosed in a total of 17 patients, 5 of whom ultimately required enucleation. Median time from brachytherapy to onset of scleromalacia was 8 months (range 2–38 months). Cumulative incidence was 7% (95% CI 3–11%) at 1 year, 11% (95% CI 7–17%) at 3 years, and 12% (95% CI 7–18%) at 5 years. Scleromalacia frequency was positively associated with ciliary body involvement (HR = 2.76; 95% CI 1.05–7.27).
Vision declined precipitously following treatment. Among the 117 patients who maintained follow-up for at least 1 year, median visual acuity at the most recent exam had declined to logMAR = 3.0 (hand motion). Vision loss was typically attributed to cataract, vitreous hemorrhage, or radiation retinopathy. 135 patients were phakic at the time of brachytherapy, and 51 patients subsequently required cataract surgery. Median time from brachytherapy to cataract surgery was 20 months (range 2–81 months). Cumulative incidence of cataract surgery was 15% (95% CI 9–22%) at 1 year, 33% (95% CI 24–41%) at 3 years, and 45% (95% CI 35–54%) at 5 years. Cataract surgery rates following brachytherapy were significantly associated with increased patient age at diagnosis (HR = 1.05; 95% CI 1.03–1.08). At the most recent follow-up exam, median visual acuity was equivalent among patients who had undergone cataract surgery and those who remained phakic at logMAR 3.0 (hand motion).
Forty-eight patients developed vitreous hemorrhage at a median time of 13.5 months (range 1–82 months) after brachytherapy. Cumulative incidence of vitreous hemorrhage at 1, 3, and 5 years was 16% (95% CI 10–22%), 27% (95% CI 20–35%), and 34% (95% CI 26–42%), respectively. Vitreous hemorrhage rates were positively associated with increased patient age (HR = 1.03; 95% CI 1.00–1.05), increased tumor height (HR = 1.23; 95% CI 1.11–1.37), and poor baseline logMAR visual acuity (HR = 1.51; 95% CI 1.17–1.97).
Radiation retinopathy and optic neuropathy were diagnosed in 59 and 39 patients, respectively. Median time to radiation retinopathy diagnosis was 17 months (range 2–83 months). Median time to optic neuropathy diagnosis was 19 months (range 5–60 months). Cumulative incidence of radiation retinopathy at 1, 3, and 5 years was 10% (95% CI 6–16%), 39% (95% CI 31–47%), and 44% (95% CI 35–52%), respectively. Cumulative incidence of radiation optic neuropathy at 1, 3, and 5 years was 6% (95% CI 3–10%), 23% (95% CI 17–31%), and 31% (95% CI 23–39%), respectively. Frequency of optic neuropathy was positively associated with tumor proximity (≤6 disc diameters) to the optic nerve (HR = 2.39; 95% CI 1.09–5.24).
Discussion
In this study, we investigated clinical outcomes following EPBT for the most advanced category of PUM under the AJCC cancer staging criteria [1]. Previous studies have demonstrated that treatment failure rates increase proportionately with tumor size [7, 9]. Nevertheless, ocular oncology centers are still more apt to treat large PUM with globe-sparing therapies based on our current understanding of uveal melanoma biology and the possibility of subclinical micrometastatic disease [18, 19, 20]. To our knowledge, this is the first large retrospective study investigating EPBT exclusively for T4-staged PUM, and median tumor dimensions were larger in this cohort than in any other previously published case series. Our results may, therefore, lend further insight into the burden of treatment-related morbidity among the largest and most advanced tumors.
As expected, survival in this cohort was poor, with a 5-year metastasis-free survival of 47.1%. This was comparatively lower than that previously reported for T4 melanomas in the AJCC criteria validation study by the OOTF which observed a 5-year disease-specific survival of 61% (95% CI 49–71%) [21]. However, our estimates are within the confidence interval of a similar validation study performed by the European Ocular Oncology Group (EOOG) who reported a 5-year melanoma-specific survival of 53% (95% CI 46–56%) [22]. As cause of death information was not available for all patients in this cohort, relative survival analysis was performed to better estimate melanoma-associated mortality. This is widely regarded as a more rigorous measure of disease-associated mortality and may also be useful for patient counseling [23, 24]. Population-based studies not limited to large tumors have observed 5-year relative survival rates ranging from 77 to 84% [25]. As expected, estimates for this cohort were comparatively lower, particularly among patients > 65 years old where 5-year relative survival was only 65%. This trend between increased age and lower relative survival has been observed at other centers [26] and likely reflects the correlation between increasing age and metastatic disease [27].
Treatment efficacy – as defined by local control of intraocular disease – was remarkably consistent with the findings of previous investigations of 125I EPBT for large melanoma. Cumulative incidence of local recurrence at 5 years was 9% in this study and 6 and 9% in the reports by Puusaari et al. [13] and Shields et al. [14], respectively. In our investigation, the frequency of local recurrence was associated with decreased tumor thickness (HR = 0.78; 95% CI 0.64–0.95) and increased tumor basal diameter (HR = 1.08; 95% CI 0.93–1.15), although the latter finding was not statistically significant (p = 0.43). It has been well established in the literature that increased tumor thickness and basal diameter are significant risk factors for treatment failure after brachytherapy; however, these findings have been limited primarily to medium sized melanomas [7, 11]. Caujolle et al. [8] noted that local recurrence was most common amongst wide and thin melanomas, many of which may exhibit a diffuse-type growth pattern where tumor margins are more difficult to delineate prior to treatment. Our results appear to confirm their findings. In this investigation, there were 99 tumors (63%) which exceeded the maximum diameter or height limitations for 125I EPBT as defined by the ABS-OOTF [12]. Of the 12 tumors which developed local recurrence, only 3 were > 12 mm in height, and 5 were > 20 mm in diameter. Based on our findings, we cannot conclude that there is a significantly increased risk for treatment failure when tumor dimensions exceed the prescribed dose limitations for iodine brachytherapy, despite a weak correlation with increased tumor diameter (HR = 1.08; 95% CI 0.89–1.32). Clinicians should still exercise caution when treating these tumors with conservative therapy.
Cumulative incidence for enucleation was 21% – almost double the rate observed with medium-sized melanomas [11] but comparable to that reported in other studies of large melanoma [4, 14]. Unlike Shields et al. [14], we did not find a significant association between enucleation rates and tumor thickness or location. In our multivariate model, poor baseline logMAR visual acuity was the only significant predictor of enucleation (HR = 1.42; 95% CI 1.00–2.01). Poor visual acuity at baseline is generally associated with pre-existing ocular morbidity, and these eyes may be more likely to become blind and painful after treatment. Of the 32 patients who underwent enucleation during the study period, 16 (50%) presented with logMAR visual acuity ≥1.0 (Snellen 20/200). Patients who desire conservative therapy despite poor baseline vision should be counseled regarding the more guarded prognosis for long-term ocular survival.
Cataract removal was the most common surgical procedure performed after brachytherapy with a cumulative incidence of 45% at 5 years. Cataract surgery rates were significantly associated with increased age at diagnosis, likely reflecting more advanced nuclear sclerosis at baseline resulting in a shorter time to cataract progression. Other studies have reported that cataract is more commonly associated with pre-equatorial tumors given their closer proximity to the lens [28, 29]. We did not observe this association, suggesting that radiation dose distribution for most T4-staged tumors may be too extensive to spare the anterior segment, regardless of tumor location.
Five-year cumulative incidence of neovascular glaucoma was 24%, which was nearly identical to the 21% reported by Shields et al. [14]. Puusaari et al. [13] reported an incidence as high as 60%, but this included all forms of secondary glaucoma. Like Puusaari et al. [13], we observed a strong association between increased tumor height and neovascular complications (HR = 1.16; 95% CI 1.02–1.33). Overall, increased tumor height and increased patient age were the most consistent predictors of ocular morbidity following treatment. This corroborates the findings of Shields et al. [14], who observed that increased tumor height and increased patient age were the most significant risk factors for vision loss following brachytherapy.
There was an unexpected negative association between tumor height and development of radiation retinopathy. We speculate that this may reflect a greater incidence of vitreous hemorrhage associated with thicker lesions, which typically precludes fundus examination for retinopathy. Eyes with unclear media were not censored in this investigation, as this would falsely elevate the observed incidence of some complications without accounting for competing events.
Alternatives to iodine brachytherapy have been reported for large melanomas with successful outcomes. Semenova and Finger [6] observed reduced complication and vision loss rates using 103Pd brachytherapy, which generally results in a lower cumulative radiation dose. This study included only 9 T4-staged melanomas, and direct comparison with our data is difficult. Proton beam teletherapy has been used as an alternative to brachytherapy at some centers. This modality has been associated with higher rates of anterior segment complications with large melanomas, particularly neovascular glaucoma where 5-year estimates range from 35% [30] to over 50% [31]. Our results agree with previous reports demonstrating comparatively lower risk for neovascular glaucoma with brachytherapy [14].
This study is limited by the relatively short duration of patient surveillance, with a median follow-up time of 28 months. This was primarily constrained by the high incidence of mortality in this cohort, as the majority of patients had expired by 5 years. Additionally, because our institutional database does not record visual acuity continuously, we were unable to report visual outcomes in actuarial measures. It is generally accepted that eyes with large melanomas have very poor visual prognosis following conservative therapy [4, 14, 30], and our limited analysis of visual outcomes in this cohort corroborates this.
In this retrospective case series, we have reported our single-institution experience with 125I EPBT in a large cohort with T4-staged PUM. Iodine brachytherapy achieved high rates of local control despite that the majority of tumors exceeded the maximum height and diameter thresholds for 125I EPBT. Decreased tumor thickness was associated with a higher risk of local treatment failure. Ocular morbidity – as defined by the burden of treatment complications and adverse events – appears to be greater with increasing tumor thickness, patient age, and poor visual acuity at presentation. These findings may assist clinicians to better identify those patients with advanced disease who are the most appropriate candidates for conservative therapy.
Statement of Ethics
Informed consent was obtained following a detailed discussion of the potential risks and benefits of EPBT with specific emphasis on radiation-related complications. All aspects of our investigation were compliant with the Declaration of Helsinki.
Disclosure Statement
We wish to confirm that there are no known conflicts of interest associated with this publication and there has been no significant financial or proprietary support for this work that could have influenced its outcome.
References
- 1.Edge SE, Bryd DR, Compton CA, Fritz AG, Greene FL, Troti A, editors. 7th ed. New York (NY): Springer; 2010. AJCC Cancer Staging Manual; pp. pp. 547–59. [Google Scholar]
- 2.Diener-West M, Earle JD, Fine SL, Hawkins BS, Moy CS, Reynolds SM, et al. Collaborative Ocular Melanoma Study Group The COMS randomized trial of iodine 125 brachytherapy for choroidal melanoma, III: initial mortality findings. COMS Report No. 18. Arch Ophthalmol. 2001 Jul;119((7)):969–82. doi: 10.1001/archopht.119.7.969. [DOI] [PubMed] [Google Scholar]
- 3.Collaborative Ocular Melanoma Study Group The Collaborative Ocular Melanoma Study (COMS) randomized trial of pre-enucleation radiation of large choroidal melanoma II: initial mortality findings. COMS report no. 10. Am J Ophthalmol. 1998 Jun;125((6)):779–96. doi: 10.1016/s0002-9394(98)00039-7. [DOI] [PubMed] [Google Scholar]
- 4.Puusaari I, Heikkonen J, Summanen P, Tarkkanen A, Kivelä T. Iodine brachytherapy as an alternative to enucleation for large uveal melanomas. Ophthalmology. 2003 Nov;110((11)):2223–34. doi: 10.1016/S0161-6420(03)00661-4. [DOI] [PubMed] [Google Scholar]
- 5.Mosci C, Lanza FB, Barla A, Mosci S, Hérault J, Anselmi L, et al. Comparison of clinical outcomes for patients with large choroidal melanoma after primary treatment with enucleation or proton beam radiotherapy. Ophthalmologica. 2012;227((4)):190–6. doi: 10.1159/000334401. [DOI] [PubMed] [Google Scholar]
- 6.Semenova E, Finger PT. Palladium-103 plaque radiation therapy for American Joint Committee on cancer T3- and T4-staged choroidal melanomas. JAMA Ophthalmol. 2014 Feb;132((2)):205–13. doi: 10.1001/jamaophthalmol.2013.5677. [DOI] [PubMed] [Google Scholar]
- 7.Wilson MW, Hungerford JL. Comparison of episcleral plaque and proton beam radiation therapy for the treatment of choroidal melanoma. Ophthalmology. 1999 Aug;106((8)):1579–87. doi: 10.1016/S0161-6420(99)90456-6. [DOI] [PubMed] [Google Scholar]
- 8.Caujolle JP, Paoli V, Chamorey E, Maschi C, Baillif S, Herault J, et al. Local recurrence after uveal melanoma proton beam therapy: recurrence types and prognostic consequences. Int J Radiat Oncol Biol Phys. 2013 Apr;85((5)):1218–24. doi: 10.1016/j.ijrobp.2012.10.005. [DOI] [PubMed] [Google Scholar]
- 9.Seibel I, Cordini D, Rehak M, Hager A, Riechardt AI, Böker A, et al. Local Recurrence After Primary Proton Beam Therapy in Uveal Melanoma: Risk Factors, Retreatment Approaches, and Outcome. Am J Ophthalmol. 2015 Oct;160((4)):628–36. doi: 10.1016/j.ajo.2015.06.017. [DOI] [PubMed] [Google Scholar]
- 10.Detorakis ET, Engstrom RE, Jr, Wallace R, Straatsma BR. Iris and anterior chamber angle neovascularization after iodine 125 brachytherapy for uveal melanoma. Ophthalmology. 2005 Mar;112((3)):505–10. doi: 10.1016/j.ophtha.2004.09.028. [DOI] [PubMed] [Google Scholar]
- 11.Jampol LM, Moy CS, Murray TG, Reynolds SM, Albert DM, Schachat AP, et al. Collaborative Ocular Melanoma Study Group (COMS Group) The COMS randomized trial of iodine 125 brachytherapy for choroidal melanoma: IV. Local treatment failure and enucleation in the first 5 years after brachytherapy. COMS report no. 19. Ophthalmology. 2002 Dec;109((12)):2197–206. doi: 10.1016/s0161-6420(02)01277-0. [DOI] [PubMed] [Google Scholar]
- 12.American Brachytherapy Society - Ophthalmic Oncology Task Force The American Brachytherapy Society consensus guidelines for plaque brachytherapy of uveal melanoma and retinoblastoma. Brachytherapy. 2014 Jan-Feb;13((1)):1–14. doi: 10.1016/j.brachy.2013.11.008. [DOI] [PubMed] [Google Scholar]
- 13.Puusaari I, Heikkonen J, Kivelä T. Ocular complications after iodine brachytherapy for large uveal melanomas. Ophthalmology. 2004 Sep;111((9)):1768–77. doi: 10.1016/j.ophtha.2004.03.027. [DOI] [PubMed] [Google Scholar]
- 14.Shields CL, Naseripour M, Cater J, Shields JA, Demirci H, Youseff A, et al. Plaque radiotherapy for large posterior uveal melanomas ([{GT}] or =8-mm thick) in 354 consecutive patients. Ophthalmology. 2002 Oct;109((10)):1838–49. doi: 10.1016/s0161-6420(02)01181-8. [DOI] [PubMed] [Google Scholar]
- 15.Bechrakis NE, Bornfeld N, Zöller I, Foerster MH. Iodine 125 plaque brachytherapy versus transscleral tumor resection in the treatment of large uveal melanomas. Ophthalmology. 2002 Oct;109((10)):1855–61. doi: 10.1016/s0161-6420(02)01273-3. [DOI] [PubMed] [Google Scholar]
- 16.Dickman PW, Sloggett A, Hills M, Hakulinen T. Regression models for relative survival. Stat Med. 2004 Jan;23((1)):51–64. doi: 10.1002/sim.1597. [DOI] [PubMed] [Google Scholar]
- 17.Gooley TA, Leisenring W, Crowley J, Storer BE. Why Kaplan-Meier fails and cumulative incidence succeeds when estimating failure probabilities in the presence of competing risks. In: Crowley J, editor. Handbook of Statistics in Clinical Oncology. Boca Raton (FL): Chapman and Hall/CRC Press; 2001. pp. 513–523. [Google Scholar]
- 18.Grossniklaus HE. Tumor vascularity and hematogenous metastasis in experimental murine intraocular melanoma. Trans Am Ophthalmol Soc. 1998;96:721–52. [PMC free article] [PubMed] [Google Scholar]
- 19.Diaz CE, Rusciano D, Dithmar S, Grossniklaus HE. B16LS9 melanoma cells spread to the liver from the murine ocular posterior compartment (PC) Curr Eye Res. 1999 Feb;18((2)):125–9. doi: 10.1076/ceyr.18.2.125.5380. [DOI] [PubMed] [Google Scholar]
- 20.Eskelin S, Pyrhönen S, Summanen P, Hahka-Kemppinen M, Kivelä T. Tumor doubling times in metastatic malignant melanoma of the uvea: tumor progression before and after treatment. Ophthalmology. 2000 Aug;107((8)):1443–9. doi: 10.1016/s0161-6420(00)00182-2. [DOI] [PubMed] [Google Scholar]
- 21.The AJCC Ophthalmic Oncology Task Force International Validation of the American Joint Committee on Cancer's 7th Edition Classification of Uveal Melanoma. JAMA Ophthalmol. 2015;133((4)):376–383. doi: 10.1001/jamaophthalmol.2014.5395. [DOI] [PubMed] [Google Scholar]
- 22.Kujala E, Damato B, Coupland SE, Desjardins L, Bechrakis NE, Grange JD, et al. Staging of ciliary body and choroidal melanomas based on anatomic extent. J Clin Oncol. 2013 Aug;31((22)):2825–31. doi: 10.1200/JCO.2012.45.2771. [DOI] [PubMed] [Google Scholar]
- 23.Sarfati D, Blakely T, Pearce N. Measuring cancer survival in populations: relative survival vs cancer-specific survival. Int J Epidemiol. 2010 Apr;39((2)):598–610. doi: 10.1093/ije/dyp392. [DOI] [PubMed] [Google Scholar]
- 24.Dickman PW, Adami HO. Interpreting trends in cancer patient survival. J Intern Med. 2006 Aug;260((2)):103–17. doi: 10.1111/j.1365-2796.2006.01677.x. [DOI] [PubMed] [Google Scholar]
- 25.Singh AD, Topham A. Survival rates with uveal melanoma in the United States: 1973-1997. Ophthalmology. 2003 May;110((5)):962–5. doi: 10.1016/S0161-6420(03)00077-0. [DOI] [PubMed] [Google Scholar]
- 26.Caminal JM, Ribes J, Clèries R, Ibáñez N, Arias L, Piulats JM, et al. Relative survival of patients with uveal melanoma managed in a single center. Melanoma Res. 2012 Jun;22((3)):271–7. doi: 10.1097/CMR.0b013e328353ef30. [DOI] [PubMed] [Google Scholar]
- 27.Hawkins BS, Collaborative Ocular Melanoma Study Group The Collaborative Ocular Melanoma Study (COMS) randomized trial of pre-enucleation radiation of large choroidal melanoma: IV. Ten-year mortality findings and prognostic factors. COMS report number 24. Am J Ophthalmol. 2004 Dec;138((6)):936–51. doi: 10.1016/j.ajo.2004.07.006. [DOI] [PubMed] [Google Scholar]
- 28.Finger PT. Radiation therapy for choroidal melanoma. Surv Ophthalmol. 1997 Nov-Dec;42((3)):215–32. doi: 10.1016/s0039-6257(97)00088-x. [DOI] [PubMed] [Google Scholar]
- 29.Fontanesi J, Meyer D, Xu S, Tai D. Treatment of choroidal melanoma with I-125 plaque. Int J Radiat Oncol Biol Phys. 1993 Jul;26((4)):619–23. doi: 10.1016/0360-3016(93)90278-4. [DOI] [PubMed] [Google Scholar]
- 30.Papakostas TD, Lane AM, Morrison M, Gragoudas ES, Kim IK. Long-term Outcomes After Proton Beam Irradiation in Patients With Large Choroidal Melanomas. JAMA Ophthalmol. 2017 Nov;135((11)):1191–6. doi: 10.1001/jamaophthalmol.2017.3805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Foss AJ, Whelehan I, Hungerford JL, Anderson DF, Errington RD, Kacperek A, et al. Predictive factors for the development of rubeosis following proton beam radiotherapy for uveal melanoma. Br J Ophthalmol. 1997 Sep;81((9)):748–54. doi: 10.1136/bjo.81.9.748. [DOI] [PMC free article] [PubMed] [Google Scholar]