Abstract
Aim: To evaluate the clinical efficacy of a circulating tumor cell (CTC) test by comparison between healthy volunteers and patients with localized prostate cancer including those under active surveillance. Materials and Methods: CTC counts in peripheral blood were compared between patients with prostate cancer (n=45) and healthy volunteers (n=17). CTCs were identified based on the expression of epithelial cell adhesion molecule (EpCAM) and counted using a SMART BIOPSY™ SYSTEM. Results: The number of EpCAM+ cells was significantly higher in patients with cancer than in healthy volunteers. Among the low-risk patients (n=9), two had up-staging and six had up-grading. Among those up-staged, there was one case which was EpCAM+. Among those cases up-graded, three were EpCAM+. In those with stage T2 tumors, the presence of Gleason pattern 5 was positively correlated with EpCAM positivity (rho=0.59, p< 0.001). Conclusion: CTC counts in localized prostate cancer were associated with Gleason pattern 5. Active treatment should be considered for patients with low-risk disease during active surveillance who are found to have EpCAM+ CTCs because of a risk of up-staging and up-grading.
Keywords: Prostate biopsy, biomarker, EpCAM, PSMA, prostatectomy
Prostate cancer is the second most common cancer in men worldwide, and it is classified according to different cancer phenotypes (1). Active surveillance is recommended as a treatment option for patients with low-risk prostate cancer. The development of prostate-specific antigen (PSA) detection tests has reduced the number of deaths related to prostate cancer and has been associated with an increase in the incidence of prostate cancer (2). Reportedly, an increase in the number of cases that involved low-risk prostate cancer causes overtreatment and related complications (3). The concept of active surveillance was established after the recommendation of the United States Preventive Services Task Force against PSA screening for asymptomatic men because of overdiagnosis (4). Several criteria for active surveillance exist according to different institutes, but most include localized stage, low PSA level, and grade 1 (5). The aim of active surveillance is to reduce overtreatment by choosing those who do not need definitive treatment. In addition, active surveillance offers the chance for curative definitive treatment when appropriate by following additional examinations over time. Many institutions recommend PSA monitoring and repeated biopsies (5). PSA has been used as a representative serum marker of prostate cancer since a long time, but it has limitations. PSA is not cancer-specific, and other conditions affecting the gland can elevate the PSA level, including prostate procedures (6). PSA has both low specificity and low positive predictive value (PPV), particularly in the gray zone of values lower than 10 ng/ml (a common active surveillance criterion) (7). The PSA level also displays a biological variability of approximately 20% (8).
Circulating tumor cells (CTCs) in peripheral blood have been associated with prognosis, and various techniques and products for their detection have been developed with high specificity (9). CTCs are broadly accepted as the cause of metastases (10) but to date, no standard CTC marker exists. Epithelial cell adhesive molecule (EpCAM) is the most commonly used CTC marker (11). Other biomarkers have been evaluated for specific cancer types, such as cytokeratin 7 for lung cancer, prostate-specific membrane antigen (PSMA) for prostate cancer, and caudal type homeobox 2 for colorectal cancer (12). The detection of CTCs as alternatives to biopsy using various markers may reduce the need for invasive repetitive biopsy samplings.
The studies on CTCs in patients with localized prostate cancer are fewer than those with metastatic diseases because of limitations to CTC detection. In this study, we compared CTC counts between patients with localized prostate cancer who underwent radical prostatectomy and healthy volunteers in order to evaluate the clinical efficacy of the test. In addition, we tried to determine the value of CTC counts for patients with low-risk disease who may be candidates for active surveillance.
Materials and Methods
Study design. The Ethical Review Board of our Institution approved the study protocol (approval number: 2017-0192). All participants signed written informed consent forms and agreed to participate by free will. We enrolled 45 patients diagnosed as having prostate adenocarcinoma by needle biopsy and 17 healthy male volunteers between January and March 2018. All patients with prostate cancer underwent bone scans, abdominal and pelvic computed tomography, and prostate magnetic resonance imaging evaluations to detect metastatic disease. Patients were classified as having low-risk category of disease based on criteria including having a biopsy grade 1, PSA < 10 ng/ml, and clinical stage of T2a or lower (13). All patients had undergone radical prostatectomy. A pathologist analyzed resected prostate specimens. Disease in our low-risk patients was categorized as being upgraded (grade ≥2) or upstaged (pathological stage ≥T3 stage) based on the pathological analysis. A clinician interviewed healthy volunteers to evaluate their past medical history and excluded those who had any disease that needed any medication up to 3 months before the interview date. Peripheral blood samples of 15 ml were collected from participants before surgery.
Blood collection and CTC isolation. Blood samples (15 ml) from each participant were collected in acid citrate dextrose solution A tubes (BD Vacutainer®; Franklin Lakes, NJ, USA). The blood samples were processed to enrich CTCs using a CTC isolation kit (Cat# CIKW10; Cytogen, Seoul, Republic of Korea) and SMART BIOPSY™ Cell Isolator (Cat# CIS030; Cytogen).
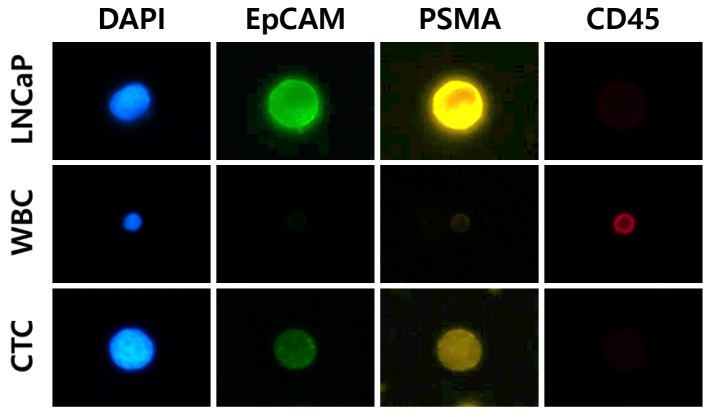
Immunofluorescence staining and CTC counting. For immunofluo-rescence staining, the enriched cells were layered onto a microscope slide, fixed in 4% paraformaldehyde for 5 min, and then washed with phosphate-buffered saline (PBS). The cells were incubated with 0.2% Triton X-100 in PBS for 10 min, then blocked with 1% bovine serum albumin in PBS for 60 min, and incubated with antibodies against CD45 (mouse, dilution 1:100; Agilent, Santa Clara, CA, USA) and PSMA (rabbit, dilution 1:50; Cell Signaling Technology, Danvers, MA, USA, followed by anti-mouse Alexa 647 (dilution 1:1,000; Invitrogen, Carlsbad, CA, USA) and anti-rabbit Alexa 546 (dilution 1:1,000; Invitrogen). In addition, cells were incubated with Alexa 488-conjugated mouse antibody to EpCAM (dilution 1:50; Cell Signaling Technology). The slides were mounted with Fluoroshield™ with 4’,6-diamidino-2-phenylindole (ImmunoBioScience, Mukilteo, WA, USA). In order to identify PSMA-EpCAM+ and CD45− CTCs, LNCaP (PSMA-EpCAM+; Korean Cell Line Bank, Seoul, Republic of Korea) cell line and white blood cells (CD45−), obtained from a patient sample, were used as positive controls (Figure 1). The immunofluorescently stained cells were analyzed for each marker using a SMART BIOPSY™ Cell Image Analyzer (Cat# CIA030; Cytogen), which comes with image analyzing software.
Figure 1. Identification of circulating tumor cells (CTC) by combining staining with 4’,6-diamidino-2-phenylindole (DAPI, blue), and for epithelial cell adhesive molecule (EpCAM, green), prostate-specific membrane antigen (PSMA, yellow), and cluster of differentiation 45 (CD45, red). WBC: white blood cell; LNCap: lymph node carcinoma of the prostate, cell line.
Statistical analysis. The baseline characteristics of participants were expressed as the mean±standard deviation with interquartile ranges, or numbers with percentages. The data were assessed for normality using the Kolmogorov–Smirnov test. Student’s t-test or Mann–Whitney tests were used to analyze differences between continuous variables, and Fisher’s exact test for categorical variables. Spearman correlation analysis was applied to investigate associations between markers and pathological status. All statistical analyses were performed using R version 3.4.3 software (R Project for Statistical Computing; http://www.r-project.org/). All statistical tests were two-tailed, with p< 0.05 considered significant.
Results
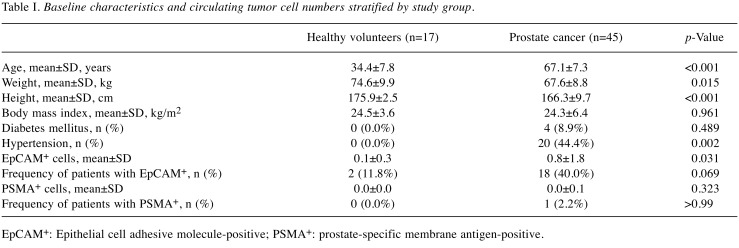
Participant and sample characteristics. Table I displays the baseline characteristics of healthy volunteers (n=17) and patients with prostate cancer (n=45). Healthy volunteers were younger (34.4±7.8 vs. 67.1±7.3, p< 0.001), weighed more on average (74.6±9.9 vs. 67.6±8.8, p=0.015), and were taller (175.9±2.5 vs. 166.3±9.7 cm, p< 0.001) than patients with prostate cancer, but their body mass indices were similar (p=0.961). The number of positive EpCAM cells (EpCAM+) was significantly higher in patients with prostate cancer than that in healthy volunteers (0.8±1.8 vs. 0.1±0.3, p=0.031). Two healthy volunteers each had one EpCAM+ cell, and 40% of patients with prostate cancer showed EpCAM+ cells (1 to 11). Only one patient with prostate cancer displayed one positive PSMA cell (PSMA+).
Table I. Baseline characteristics and circulating tumor cell numbers stratified by study group.
EpCAM+: Epithelial cell adhesive molecule-positive; PSMA+: prostate-specific membrane antigen-positive.
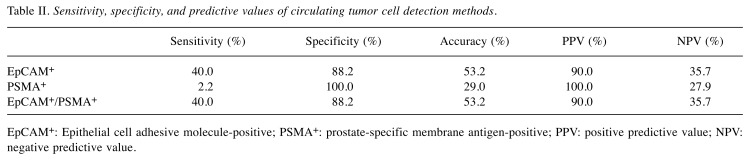
Detection of EpCAM+ cells showed 40.0% sensitivity, 88.2% specificity, 53.2% accuracy, 90.0% PPV, and 35.7% negative predictive value (NPV) (Table II) for prostate cancer. Detection of PSMA+ had 2.2% sensitivity, 100.0% specificity, 29.0% accuracy, 100.0% PPV, and 27.9% NPV for prostate cancer. The combination of EpCAM+ and PSMA+ (EpCAM+/PSAM+) gave the same values as for use EpCAM+ cells.
Table II. Sensitivity, specificity, and predictive values of circulating tumor cell detection methods.
EpCAM+: Epithelial cell adhesive molecule-positive; PSMA+: prostate-specific membrane antigen-positive; PPV: positive predictive value; NPV: negative predictive value.
Tumor characteristics. Table III shows tumor characteristics. Patients with prostate cancer had a median preoperative PSA level of 8.0 ng/ml. Pathology results for prostate cancer specimens showed 44.4% were stage T3 or higher, 2.2% had N1 stage, and 24.4% were grade 4 or higher. The median percentage tumor volume was 12.0%, the median prostate volume was 33.5 g, and the median tumor volume was 3.8 cc. Lymphovascular invasion was confirmed in 14 men (31.1%) and perineural invasion in 34 (75.6%). Only 20% of patients with prostate cancer had a low-risk disease. Among them, disease in two was up-staged and in six was upgraded. Among those with up-staging, there was one with EpCAM+ cells and among those with up-grading, there were 3 with EpCAM+ cells.
Table III. Tumor characteristics of patients who underwent radical prostatectomy.
EpCAM+: Epithelial cell adhesive molecule-positive; IQR: interquartile range; PSA: prostate-specific antigen; PSMA+: prostate-specific membrane antigen-positive; SD: standard deviation.
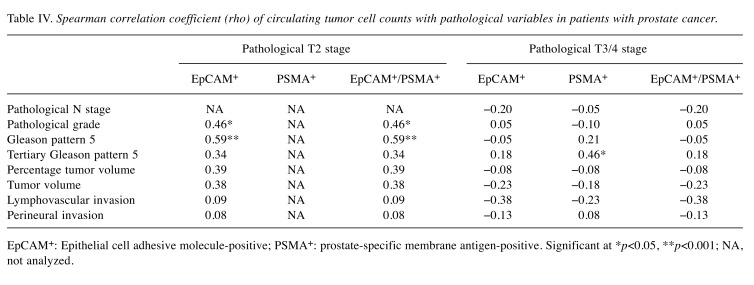
Circulating tumor cells and pathology. Table IV displays the correlation between CTC counts and pathological variables in patients with prostate cancer. In those with stage T2 disease, the grade was significantly positively correlated with EpCAM positivity (rho=0.46, p=0.020) and with EpCAM/ PSMA positivity (rho=0.46, p=0.020). In addition, the presence of Gleason pattern 5 was significantly positively correlated with EpCAM positivity (rho=0.59, p< 0.001) and with EpCAM/ PSMA positivity (rho=0.59, p< 0.001). In those with stages T3/4, the tertiary Gleason pattern 5 was significantly positively correlated with PSMA positivity (rho=0.46, p=0.040).
Table IV. Spearman correlation coefficient (rho) of circulating tumor cell counts with pathological variables in patients with prostate cancer.
EpCAM+: Epithelial cell adhesive molecule-positive; PSMA+: prostate-specific membrane antigen-positive. Significant at *p< 0.05, **p< 0.001; NA, not analyzed.
Discussion
Studies have associated CTC count with survival outcomes after metastatic prostate cancer (11). But studies on CTCs in patients with localized prostate cancer are rare (14). Therefore, we prospectively analyzed the CTC count in patients about to undergo radical prostatectomy. We found the number of EpCAM+ CTCs was significantly higher in the prostate cancer group than it was in the healthy volunteer group, and 40.0% of patients with cancer had at least one circulating EpCAM+ cell. Meyer et al. reported CTC positivity of 11.2% and found no correlation between CTC count and biochemical recurrence (15). Davis et al. also reported that CTCs were present in 21% of patients with localized prostate cancer, and they did not find significant prognostic factors while screening among factors such as PSA, Gleason score, T-stage, and tumor volume (16). Differences in detection rates may come from differences in both detection methods and patient populations. Those two studies were performed using the CellSearch system within 72 hours of sample collection (15,16). We performed our tests within 4 hours from blood collection using a SMART BIOPSY™ SYSTEM. Although a study published on 92 patients with metastatic breast cancer showed no significant difference in CTC numbers within 72 hours, CTC numbers changed after 72 hours, probably due to mean sampling errors or false positivity (17). Therefore, the time restriction may be helpful for increasing sensitivity in those with nonmetastatic disease. CTCs are unstable because they undergo apoptosis after being separated from the tumor and after the blood is removed from the patient (18). In addition, our prostate cancer group had higher pathologic T-stage (44.4% ≥T3) and grade (24.4% ≥4) than the patients in the above two studies (Meyer et al., 37.5% and 4.6%; Davis et al., 20% and 7%, respectively).
We used PSMA as a marker, and had one positive result in the prostate cancer group. PSMA is expressed in epithelial cells of normal prostate tissue and prostate cancer (19). PSMA expression was increased in patients with metastatic castration-resistant prostate cancer and in those with a high grade (20). PSMA may be associated with prostate cancer progression; therefore, it can be used as a CTC marker in prostate cancer. Among the methods for detecting CTCs, including immunoaffinity, biophysical or direct imaging assay, studies have focused on finding the most appropriate technique with high selectivity and sensitivity (21). Using a microfluidic ratchet mechanism, CTCs were found 50% of 50 patients with localized prostate cancer (22). Because no gold-standard for CTC count exists, trials to find the best CTC detection method should continue.
In metastatic disease, CTCs are thought to initiate tumor metastases (23). This idea is supported by the presence of synchronous metastasis without direct invasion through blood circulation, and by recurrences in spite of complete surgical tumor resection (23). CTCs have been found in patients with localized prostate cancer, although the significance of this finding is still not understood. In our study, Gleason pattern 5 and grade were both significantly positively correlated with CTC presence. The association between CTCs and cancer aggressiveness is new and can be explained by the stem-cell-like characteristics of CTCs. Aldehyde dehydrogenase 1, a stem cell marker, has been detected in CTCs of patients with localized prostate cancer (24). Armstrong et al. reported that more than 80% of CTCs co-expressed epithelial (e.g. EpCAM, or cytokeratin) and stem cell markers (e.g. CD133) (25). The Gleason grade is based on the structural features of the prostate gland, and a Gleason pattern of 5 is the worst morphological pattern (26). A mechanism involving the collapse of the prostate gland might be related to the release of CTCs rather than lymphovascular or perineural invasion of tumor in the initial status of localized prostate cancer.
The detection of CTCs in liquid biopsies is a less invasive procedure and is easier to perform than taking repetitive prostate gland biopsies. Our results suggest CTCs may be useful for active surveillance. Half of the patients in the low-risk category with up-staging to T3 and up-grading to grade 2 had EpCAM+ CTCs. In addition, even in cases with localized T2 stage, the CTC count was positively correlated with the Gleason pattern 5. Advances such as droplet digital polymerase chain reaction or next-generation sequencing will improve the CTC detection rate, and the procedural time and cost (27). Until now, the criteria for active surveillance include PSA and repeated biopsies, but in the near future, new biomarkers including CTCs may be introduced.
Our study had some limitations. The number of participants was small, and the patients in the low-risk category were few. However, we did have a control group of healthy volunteers, so we were able to compare the counts for controls to those for the prostate cancer group. The detection rate in patients with localized prostate cancer was not high, as it was in other studies (15,16,22). Therefore, for now, the usage of CTCs for all patients with prostate cancer should be avoided, but our results suggest a use for CTCs in patients with cancer undergoing active surveillance.
Conclusion
CTC positivity in patients with localized prostate cancer may be associated with Gleason pattern 5. The presence of CTCs in low-risk patients undergoing active surveillance may indicate the need for active treatment. Larger-scale trials with more sensitive techniques are needed to confirm our results.
Conflicts of Interest
The Authors declare no conflicts of interest.
Authors’ Contributions
Study conception and design: Se Young Choi, Choung-Soo Kim; Development of methodology: Yoon Soo Kyung, Bumjin Lim; Acquisition of data: Yunlim Kim, Bong Min Kim, Byung Hee Jeon, Jae Chan Park, Young Woong Sohn; Analysis and interpretation of data: Yunlim Kim, Ji-hyun Uh; Manuscript drafting: Se Young Choi; Critical manuscript revisions: Jae Hyuk Lee, Seongsoo Jang; Funding sourcing: Choung-Soo Kim; Supervision: Choung-Soo Kim.
Acknowledgements
This study was supported by a grant from the Asan Institute for Life Sciences, Asan Medical Center (grant number: W17-686) & Korea Health Technology R&D Project, the Korea Health Industry Development Institute (grant number: H16C2193).
References
- 1.Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65(2):87–108. doi: 10.3322/caac.21262. PMID: 25651787. DOI: 10.3322/caac.21262. [DOI] [PubMed] [Google Scholar]
- 2.Etzioni R, Tsodikov A, Mariotto A, Szabo A, Falcon S, Wegelin J, DiTommaso D, Karnofski K, Gulati R, Penson DF, Feuer E. Quantifying the role of PSA screening in the US prostate cancer mortality decline. Cancer Causes Control. 2008;19(2):175–181. doi: 10.1007/s10552-007-9083-8. PMID: 18027095. DOI: 10.1007/s10552-007-9083-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hugosson J, Carlsson S. Overdetection in screening for prostate cancer. Curr Opin Urol. 2014;24(3):256–263. doi: 10.1097/MOU.0000000000000054. PMID: 24670870. DOI: 10.1097/MOU.0000000000000054. [DOI] [PubMed] [Google Scholar]
- 4.Lee DJ, Mallin K, Graves AJ, Chang SS, Penson DF, Resnick MJ, Barocas DA. Recent changes in prostate cancer screening practices and epidemiology. J Urol. 2017;198(6):1230–1240. doi: 10.1016/j.juro.2017.05.074. PMID: 28552708. DOI: 10.1016/j.juro.2017.05.074. [DOI] [PubMed] [Google Scholar]
- 5.Komisarenko M, Martin LJ, Finelli A. Active surveillance review: Contemporary selection criteria, follow-up, compliance and outcomes. Transl Androl Urol. 2018;7(2):243–255. doi: 10.21037/tau.2018.03.02. PMID: 29732283. DOI: 10.21037/tau.2018.03.02. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Stamey TA, Caldwell M, McNeal JE, Nolley R, Hemenez M, Downs J. The prostate-specific antigen era in the United States is over for prostate cancer: What happened in the last 20 years. J Urol. 2004;172(4 Pt 1):1297–1301. doi: 10.1097/01.ju.0000139993.51181.5d. PMID: 15371827. [DOI] [PubMed] [Google Scholar]
- 7.Mistry K, Cable G. Meta-analysis of prostate-specific antigen and digital rectal examination as screening tests for prostate carcinoma. J Am Board Fam Pract. 2003;16(2):95–101. doi: 10.3122/jabfm.16.2.95. PMID: 12665174. [DOI] [PubMed] [Google Scholar]
- 8.Soletormos G, Semjonow A, Sibley PE, Lamerz R, Petersen PH, Albrecht W, Bialk P, Gion M, Junker F, Schmid HP, Van Poppel H. Biological variation of total prostate-specific antigen: A survey of published estimates and consequences for clinical practice. Clin Chem. 2005;51(8):1342–1351. doi: 10.1373/clinchem.2004.046086. PMID: 15961552. DOI: 10.1373/clinchem.2004.046086. [DOI] [PubMed] [Google Scholar]
- 9.Sun YF, Yang XR, Zhou J, Qiu SJ, Fan J, Xu Y. Circulating tumor cells: advances in detection methods, biological issues, and clinical relevance. J Cancer Res Clin Oncol. 2011;137(8):1151–1173. doi: 10.1007/s00432-011-0988-y. PMID: 21681690. DOI: 10.1007/s00432-011-0988-y. [DOI] [PubMed] [Google Scholar]
- 10.Masuda T, Hayashi N, Iguchi T, Ito S, Eguchi H, Mimori K. Clinical and biological significance of circulating tumor cells in cancer. Mol Oncol. 2016;10(3):408–417. doi: 10.1016/j.molonc.2016.01.010. PMID: 26899533. DOI: 10.1016/j.molonc.2016.01.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.de Bono JS, Scher HI, Montgomery RB, Parker C, Miller MC, Tissing H, Doyle GV, Terstappen LW, Pienta KJ, Raghavan D. Circulating tumor cells predict survival benefit from treatment in metastatic castration-resistant prostate cancer. Clin Cancer Res. 2008;14(19):6302–6309. doi: 10.1158/1078-0432.CCR-08-0872. PMID: 18829513. DOI: 10.1158/1078-0432.CCR-08-0872. [DOI] [PubMed] [Google Scholar]
- 12.Lu SH, Tsai WS, Chang YH, Chou TY, Pang ST, Lin PH, Tsai CM, Chang YC. Identifying cancer origin using circulating tumor cells. Cancer Biol Ther. 2016;17(4):430–438. doi: 10.1080/15384047.2016.1141839. PMID: 26828696. DOI: 10.1080/15384047.2016.1141839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Carroll PH, Mohler JL. NCCN guidelines updates: Prostate cancer and prostate cancer early detection. J Natl Compr Canc Netw. 2018;16(5S):620–623. doi: 10.6004/jnccn.2018.0036. PMID: 29784740. DOI: 10.6004/ jnccn.2018.0036. [DOI] [PubMed] [Google Scholar]
- 14.Thalgott M, Rack B, Maurer T, Souvatzoglou M, Eiber M, Kress V, Heck MM, Andergassen U, Nawroth R, Gschwend JE, Retz M. Detection of circulating tumor cells in different stages of prostate cancer. J Cancer Res Clin Oncol. 2013;139(5):755–763. doi: 10.1007/s00432-013-1377-5. PMID: 23358719. DOI: 10.1007/s00432-013-1377-5. [DOI] [PubMed] [Google Scholar]
- 15.Meyer CP, Pantel K, Tennstedt P, Stroelin P, Schlomm T, Heinzer H, Riethdorf S, Steuber T. Limited prognostic value of preoperative circulating tumor cells for early biochemical recurrence in patients with localized prostate cancer. Urol Oncol. 2016;34(5):235 e211–236. doi: 10.1016/j.urolonc.2015.12.003. PMID: 26795608. DOI: 10.1016/ j.urolonc.2015.12.003. [DOI] [PubMed] [Google Scholar]
- 16.Davis JW, Nakanishi H, Kumar VS, Bhadkamkar VA, McCormack R, Fritsche HA, Handy B, Gornet T, Babaian RJ. Circulating tumor cells in peripheral blood samples from patients with increased serum prostate specific antigen: Initial results in early prostate cancer. J Urol. 2008;179(6):2187–2191. doi: 10.1016/j.juro.2008.01.102. PMID: 18423725. DOI: 10.1016/j.juro.2008.01.102. [DOI] [PubMed] [Google Scholar]
- 17.Riethdorf S, Fritsche H, Muller V, Rau T, Schindlbeck C, Rack B, Janni W, Coith C, Beck K, Janicke F, Jackson S, Gornet T, Cristofanilli M, Pantel K. Detection of circulating tumor cells in peripheral blood of patients with metastatic breast cancer: A validation study of the CellSearch system. Clin Cancer Res. 2007;13(3):920–928. doi: 10.1158/1078-0432.CCR-06-1695. PMID: 17289886. DOI: 10.1158/ 1078-0432.CCR-06-1695. [DOI] [PubMed] [Google Scholar]
- 18.Allard WJ, Matera J, Miller MC, Repollet M, Connelly MC, Rao C, Tibbe AG, Uhr JW, Terstappen LW. Tumor cells circulate in the peripheral blood of all major carcinomas but not in healthy subjects or patients with nonmalignant diseases. Clin Cancer Res. 2004;10(20):6897–6904. doi: 10.1158/1078-0432.CCR-04-0378. PMID: 15501967. DOI: 10.1158/1078-0432.CCR-04-0378. [DOI] [PubMed] [Google Scholar]
- 19.Silver DA, Pellicer I, Fair WR, Heston WD, Cordon-Cardo C. Prostate-specific membrane antigen expression in normal and malignant human tissues. Clin Cancer Res. 1997;3(1):81–85. PMID: 9815541. [PubMed] [Google Scholar]
- 20.Bravaccini S, Puccetti M, Bocchini M, Ravaioli S, Celli M, Scarpi E, De Giorgi U, Tumedei MM, Raulli G, Cardinale L, Paganelli G. PSMA expression: A potential ally for the pathologist in prostate cancer diagnosis. Sci Rep. 2018;8(1):4254–4254. doi: 10.1038/s41598-018-22594-1. PMID: 29523813. DOI: 10.1038/s41598-018-22594-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Esmaeilsabzali H, Beischlag TV, Cox ME, Dechev N, Parameswaran AM, Park EJ. An integrated microfluidic chip for immunomagnetic detection and isolation of rare prostate cancer cells from blood. Biomed Microdevices. 2016;18(1):22–22. doi: 10.1007/s10544-016-0041-7. PMID: 26876965. DOI: 10.1007/s10544-016-0041-7. [DOI] [PubMed] [Google Scholar]
- 22.Todenhofer T, Park ES, Duffy S, Deng X, Jin C, Abdi H, Ma H, Black PC. Microfluidic enrichment of circulating tumor cells in patients with clinically localized prostate cancer. Urol Oncol. 2016;34(11):483 e489–483 e416. doi: 10.1016/j.urolonc.2016.06.004. PMID: 27658563. DOI: 10.1016/j.urolonc.2016.06.004. [DOI] [PubMed] [Google Scholar]
- 23.Wan L, Pantel K, Kang Y. Tumor metastasis: Moving new biological insights into the clinic. Nat Med. 2013;19(11):1450–1464. doi: 10.1038/nm.3391. PMID: 24202397. DOI: 10.1038/nm.3391. [DOI] [PubMed] [Google Scholar]
- 24.Russo GI, Bier S, Hennenlotter J, Beger G, Pavlenco L, van de Flierdt J, Hauch S, Maas M, Walz S, Rausch S, Bedke J, Morgia G, Stenzl A, Todenhofer T. Expression of tumour progression-associated genes in circulating tumour cells of patients at different stages of prostate cancer. BJU Int. 2018;122(1):152–159. doi: 10.1111/bju.14200. PMID: 29542849. DOI: 10.1111/bju.14200. [DOI] [PubMed] [Google Scholar]
- 25.Armstrong AJ, Marengo MS, Oltean S, Kemeny G, Bitting RL, Turnbull JD, Herold CI, Marcom PK, George DJ, Garcia-Blanco MA. Circulating tumor cells from patients with advanced prostate and breast cancer display both epithelial and mesenchymal markers. Mol Cancer Res. 2011;9(8):997–1007. doi: 10.1158/1541-7786.MCR-10-0490. PMID: 21665936. DOI: 10.1158/1541-7786.MCR-10-0490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ronen S, Abbott DW, Kravtsov O, Abdelkader A, Xu Y, Banerjee A, Iczkowski KA. PTEN loss and p27 loss differ among morphologic patterns of prostate cancer, including cribriform. Hum Pathol. 2017;65:85–91. doi: 10.1016/j.humpath.2017.04.024. PMID: 28504208. DOI: 10.1016/j.humpath.2017.04.024. [DOI] [PubMed] [Google Scholar]
- 27.Suh YS, Joung JY, Kim SH, Seo HK, Chung J, Lee KH. Establishment and application of prostate cancer circulating tumor cells in the era of precision medicine. Biomed Res Int. 2017;2017:7206307. doi: 10.1155/2017/7206307. PMID: 29230413. DOI: 10.1155/2017/7206307. [DOI] [PMC free article] [PubMed] [Google Scholar]