Abstract
Marijuana (MJ) is widely used among college students, with peak use between ages 18–22. Research suggests memory dysfunction in adolescent and young adult MJ users, but the neural correlates are unclear. We examined functional magnetic resonance imaging (fMRI) response during a memory task among college students with varying degrees of MJ involvement.
Participants were 64 college students, ages 18–20, who performed a visual encoding and recognition task during fMRI. MJ use was ascertained for 3 months prior to scanning; 27 individuals reported past 3-month MJ use, and 33 individuals did not. fMRI response was modeled during encoding based on whether targets were subsequently recognized (correct encoding), and during recognition based on target identification (hits). fMRI response in left and right inferior frontal gyrus (IFG) and hippocampal regions of interest was examined between MJ users and controls.
There were no group differences between MJ users and controls on fMRI response during encoding, although single sample t-tests revealed that MJ users failed to activate the hippocampus. During recognition, MJ users showed less fMRI response than controls in right hippocampus (Cohen’s d=0.55), left hippocampus (Cohen’s d=0.67) and left IFG (Cohen’s d=0.61). Heavier MJ involvement was associated with lower fMRI response in left hippocampus and left IFG.
This study provides evidence of MJ -related prefrontal and hippocampal dysfunction during recognition memory in college students. These findings may contribute to our previously identified decrements in academic performance in college MJ users and could have substantial implications for academic and occupational functioning.
Keywords: fMRI, Marijuana, Memory, Adolescence
1. Introduction
Marijuana (MJ) is one of the most commonly used psychoactive substances among emerging adults ages 18–22, and this age group shows the highest rates of use and dependence (SAMHSA, 2016). National surveys indicate that MJ use in emerging adults has been increasing over the past 30 years, and most recently, 22% reported use in the past month (Shulenberg et al., 2017). Importantly, the average period of peak MJ use also encompasses the final stages of neuromaturation (Gogtay et al., 2004; Lenroot and Giedd, 2006) and typically coincides with the completion of formal education and the transition into adult roles. MJ use in adolescence and young adulthood has been linked to decrements in subsequent academic, occupational, and social function (Fergusson and Boden, 2008; Macleod et al., 2004; Meda et al., 2017).
Specific neural systems are involved in declarative encoding and retrieval, including prefrontal, posterior parietal, and medial temporal/hippocampal regions (Kim, 2011b, 2013). The hippocampus and frontal lobes contain high densities of cannabinoid receptors (Glass et al., 1997; Herkenham et al., 1990), the primary targets for the psychoactive constituents of MJ, and may be especially vulnerable to effects of MJ use and related memory impairments (Solowij and Battisti, 2008). These regions also continue maturing throughout adolescence and emerging adulthood (Gogtay et al., 2004; Lenroot and Giedd, 2006), and may be at risk for perturbations from intoxicants such as MJ during development.
Memory deficits are one of the most consistently observed neurocognitive abnormalities among adolescent and emerging adult MJ users (Jacobus and Tapert, 2013; Lisdahl et al., 2013), although some research suggests that these effects may be limited to those with recent use (Hooper et al., 2014). Accordingly, blood oxygen level dependent (BOLD) functional magnetic resonance imaging (fMRI) studies have characterized neural activation among adolescent MJ users during long-term encoding, but the results have been somewhat inconsistent (Silveri et al., 2016). One study examined verbal associative learning following 22 days of confirmed MJ abstinence among adolescent users of alcohol, MJ, or both substances, compared to non-using controls. Despite similar task performance between groups, adolescent MJ users showed bilateral prefrontal hyperactivation, while users of both alcohol and MJ showed fMRI response levels intermediate between MJ users and controls (Schweinsburg et al., 2011). Within the hippocampus, users of MJ alone failed to show significant task-related activation, although users of both alcohol and MJ activated similarly as controls. Among young adults, high frequency of MJ use was associated with greater parahippocampal response during face/profession associative learning (Becker et al., 2010). In contrast, others have found no response differences in adolescent male MJ users during a pictorial associative learning task (Jager et al., 2010). Further work is needed in order to clarify the neural sequelae of memory dysfunction in marijuana using young people.
In the current study, we examined the neural correlates of visual encoding and recognition associated with recent MJ use in college students using BOLD fMRI. Participants performed an established figural memory task that ascertains BOLD response during nonverbal visual encoding and subsequent recognition (Beason-Held et al., 2005; Dager et al., 2014b; Jamadar et al., 2013). We focused our analyses on bilateral inferior frontal gyrus (IFG) and hippocampus, as these regions have been most consistently implicated in fMRI memory paradigms in work by our group and others (Blumenfeld and Ranganath, 2007; Harrington et al., 2006; Jamadar et al., 2013; Kim, 2011a, b). Based on previous work examining fMRI response during verbal encoding (Schweinsburg et al., 2010), we predicted that MJ use would be associated with increased response in bilateral IFG, and decreased response in bilateral hippocampus.
2. Method
2.1. Participants
Participants were 60 18–20-year-olds who were recruited as part of a larger study of alcohol and neurocognitive function of first-year college students, the NIAAA-funded Brain and Alcohol Research in College Students (BARCS) study, described in detail elsewhere (Dager et al., 2013; Dager et al., 2014b). The parent BARCS study recruited a pool of approximately 2000 participants by emailing all incoming first-year students in two consecutive years and distributing flyers on campus at two local colleges, resulting in a recruitment rate >95%. From the BARCS study, individuals without MRI contraindications were invited to participate in neuroimaging, using rolling recruitment from BARCS throughout the academic year. Scanning participants were randomly assigned to complete either the figural memory task or a spatial memory task during scanning. Upon enrollment in the BARCS study, participants began completing monthly online substance use surveys (see below); however, not all imaging participants initiated monthly surveys before undergoing imaging (e.g., if imaging was scheduled very soon after enrollment). Thus, the current sample includes a subset of individuals who received the figural memory task and who answered substance use surveys in the 3 months prior to scanning. Participants provided written informed consent, approved by the institutional review boards at Yale University, Hartford Hospital, Trinity College and Central Connecticut State University. Exclusion criteria were history of seizures, head injury with loss of consciousness >10 minutes, figural memory task performance that was invalid (e.g., all false alarms and no correct rejections) or unavailable, excessive motion during scanning, and MRI contraindications.
2.2. Measures
Current use of alcohol, MJ, and other drugs was characterized through secure monthly online self-report surveys as part of the BARCS study (Dager et al., 2014a; Meda et al., 2017). For each monthly survey, participants reported the number of days on which alcohol was consumed, number of drinks per occasion, and number of days of binge alcohol use (≥4 drinks/occasion for females, ≥5 drinks/occasion for males). Peak drinks per month was based on the month with the highest reported drinking, and calculated by multiplying the number of days of alcohol use by the number of drinks per occasion. Participants reported their monthly MJ use in a scaled manner, ranging from 1–6, as follows: 1 = never used in the past 30 days, 2 = used 1–2 times, 3 = used 3–5 times, 4 = used 6–9 times, 5 = used 10–19 times, and 6 = used ≥20 times in the past month. In order to capture sporadic recent use as well as more consistent heavy use, groups in the current study were characterized based on MJ use in the 3 months prior to scanning: 33 individuals did not report MJ use, and 27 individuals reported past 3-month MJ use (see Table 1). Participants also reported monthly use of other substances, as well as cigarette use on the Fagerstrom Test for Nicotine Dependence (Heatherton et al., 1991).
Table 1.
Participant Demographic and Substance Use Characteristics
| MJ Users (n = 27) M (SD) or % [range] |
Controls (n = 33) M (SD) or % [range] |
p value | |
|---|---|---|---|
| Age (range 18 – 20) | 18.3 (0.5) | 18.4 (0.6) | 0.343 |
| Female | 55.6% | 66.7% | 0.530 |
| Caucasian | 77.8% | 69.7% | 0.379 |
| Right Handed | 92.6% | 84.8% | 0.634 |
| Current mood or anxiety disorder | 21.2% | 18.5% | 0.795 |
| Peak MJ use per month, past 3 months | |||
| none | 0% | 100% | |
| 1 – 2 times | 44.4% | 0% | |
| 3 – 5 times | 37.0% | 0% | |
| 6 – 9 times | 14.8% | 0% | |
| 10 – 19 times | 3.7% | 0% | |
| ≥20 times | 0% | 0% | |
| Peak drinks per month, past 3 months | 33.6 (31.0) | 7.5 (10.3) | <.001 |
| Lifetime drinks | 77.7 (97.7) | 38.3 (59.5) | 0.059 |
| Used other drugs, past 3 months | 14.8% | 3.0% | 0.100 |
Diagnoses for current and past DSM-IV-TR psychotic, anxiety, mood, and substance use disorders were ascertained using the Mini International Neuropsychiatric Interview (Sheehan et al., 1998). Lifetime alcohol use was ascertained with an in-house interview at the time of scanning (Dager et al., 2013). At the time of scanning, participants were free of alcohol and drugs other than MJ in the MJ group, as verified by breathalyzer and urine toxicology, and females provided negative pregnancy screens.
2.3. Figural Memory Task
The Figural Memory Task (Beason-Held et al., 2005) is a visual encoding and recognition task designed to minimize verbal encoding of stimuli. The task stimuli (20 targets and 20 distractors) were abstract black line drawings that are designed to be difficult to encode verbally, presented against a white background. Participants performed an encoding phase and a recognition phase during fMRI scanning. The encoding phase presented 20 target stimuli (duration 3 sec, interstimulus (ISI) interval 4 sec), which participants were instructed to silently memorize. For each stimulus, participants pressed a button on a fiber optic response box to confirm that they saw each item. After the encoding phase, there was a 5-minute delay (with no other cognitive task presented during the delay), followed by the recognition phase. During the recognition phase, 20 target and 20 distractor stimuli were presented in a fixed pseudo-random order, each for 3 sec with an ISI of 4 sec. Participants pressed a button with their right index (“yes”) and middle (“no”) fingers to indicate whether they had previously seen each stimulus, and accuracy was emphasized over speed.
2.4. Image Acquisition
Imaging data were collected on a Siemens 3T Allegra high performance head-dedicated system. Structural imaging was acquired with a sagittal T1 MPRAGE protocol using the following parameters: TR = 2500 ms, TE=2.74 ms, flip angle = 8°, FOV=176 × 256 mm, matrix = 256 × 208, voxel size = 1 mm3, 176 slices, total scan time =7:20. Functional images were collected in the axial plane using a T2*-weighted echoplanar image (EPI) gradient-echo pulse sequence covering the whole brain: TR = 1860 ms, TE = 27 ms, flip angle 70°, FOV = 240 mm, matrix=64 × 64, in-plane resolution=3.44 mm × 3.44 mm, slice thickness = 3 mm, gap = 1 mm, 36 slices, total scan time = 12:33.
2.5. Data Analyses
Stimuli from the recognition phase were classified as hits, misses, correct rejections, and false alarms. We used a signal detection approach (Macmillan and Creelman, 2005) to examine the discriminability index, d’, which represents the ability to discriminate targets from distractors. We calculated d’ = z(hit rate) – z(false alarm rate), with a standard correction for false alarm rates of zero. Responses during the recognition phase were compared between groups using repeated measures ANOVA with two within-subjects factors (target vs. distractor and response “yes” vs. “no”) and one between-groups factor (group). We used independent samples t-tests to determine group differences in d’.
Functional images from the task were preprocessed and modeled similarly as in our prior work (Dager et al., 2014b) using SPM5 (http://www.fil.ion.ucl.ac.uk/spm/software/spm5/). The first six volumes were discarded to allow for T1 saturation effects. Images were realigned, spatially normalized to Montreal Neurological Institute (MNI) standardized space, resampled to 3×3×3 mm voxels, and smoothed with a 5mm full-width, half-maximum Gaussian filter. Datasets were inspected for motion, and those with >3mm displacement or >3 degrees rotation were not included in the current study.
BOLD response was modeled based on behavioral performance (items that were subsequent hits and misses), while covarying for the degree of motion and linear baseline trends. Trials from the encoding phase were modeled as correctly encoded if they were subsequently identified as targets during the recognition phase. Targets that were subsequently designated as distractors in the recognition phase were categorized as incorrectly encoded. Events in the recognition phase were coded as hits, misses, false alarms, and correct rejections. BOLD response for each event was modeled using a canonical hemodynamic response function fitted to the onset of the event. The duration of each event was determined by reaction time. BOLD response contrast was determined for correctly encoded vs. incorrectly encoded, hits vs. implicit baseline, misses vs. implicit baseline, and correct rejections vs. implicit baseline. Although false alarm events were modeled at the individual level, we did not examine group differences in BOLD response to false alarms because there were too few to model at the group level (Jamadar et al. 2013).
Regions of interest for bilateral IFG and hippocampus were based on atlas regions delineated by Destrieux and colleagues (Destrieux et al., 2010). The IFG ROI was created by combining frontal opercular, orbital, and triangular gyri, and the hippocampal ROI was defined based solely on the hippocampus. Left and right hemisphere regions were defined separately, for a total of four ROIs. For each ROI, data for each subject were extracted for correct encoding and for recognition hits, and analyzed in SPSS (https://www.ibm.com/products/spss-statistics).
fMRI response was examined between groups using independent samples t-tests. In addition, we conducted exploratory single sample t-tests to determine whether each group showed significant response in each ROI, in order to provide supplementary descriptive information on response patterns in each group. In addition, we conducted exploratory whole-brain analyses (voxel-wise p < .05, uncorrected for multiple comparisons) in order to provide a preliminary characterization of possible group differences in regions outside our a priori-selected ROIs. Follow-up analyses examined the relationship between degree of marijuana use and fMRI response in each ROI using nonparametric correlations. In addition, given the high concordance between marijuana use and alcohol use in college students (Agosti et al., 2002; Meda et al., 2017), we conducted analyses to determine whether peak drinks per month or lifetime drinks influenced the relationship between marijuana use and fMRI response. Given the importance of determining sex differences in research studies, we also conducted exploratory analyses to ascertain group × sex interactions on fMRI response.
3. Results
3.1. Demographic Results
Groups were statistically similar on age, sex, and race (see Table 1). Groups were also similar on rates of current psychiatric disorders, which included only anxiety disorders and depression. MJ users reported more drinks per month (t(58) = −4.54, p < .001). One MJ user reported low nicotine dependence; all other participants were nonsmokers. Five participants reported having used other substances in the 3 months prior to scanning: one control used cough medicine 1–2 times, one MJ user used cocaine 1–2 times and crystal meth 1–2 times, one MJ user used pain medication 1–2 times, one MJ user used hypnotic medication 1–2 times, and one MJ user used hallucinogens other than LSD 1–2 times and stimulant medications 3–5 times. None of these participants had positive urine toxicology screens for these other substances.
3.2. Behavioral Results
On average, participants performed at 67% accuracy on the figural memory task (see Table 2), which is consistent with our previous work with this task in college students (Dager et al., 2014b). There was no group × response type interaction (F(1,58) = 0.37, p = .543), indicating no group differences on hits, misses, correct rejections, or false alarms. MJ users showed somewhat lower d’ values (t(58) = 1.82, p = .074, Cohen’s d = 0.48). Follow-up exploratory analyses suggested that differences in d’ might be related to a trend for more false alarms (t(58) = −1.82, p = .074, Cohen’s d = 0.48) in MJ users, and nonparametric correlations revealed that greater MJ involvement was positively associated with false alarms (Spearman’s rho = 0.28, p = .033).
Table 2.
Figural Memory Task Performance.
| MJ Users (n = 27) M (SD) |
Controls (n = 33) M (SD) |
|
|---|---|---|
| Number of Responses (max=20) | ||
| Hits | 13.2 (3.0) | 13.5 (3.5) |
| Misses | 6.6 (3.2) | 6.2 (3.3) |
| False Alarms | 4.0 (2.5) | 2.9 (2.3) |
| Correct Rejections | 15.8 (2.5) | 16.8 (2.5) |
| d’ | 0.97 (0.44) | 1.20 (0.53) |
3.3. fMRI Results
During the encoding phase, there were no group differences on BOLD response in any ROI: right hippocampus (t(58) = 1.64, p = .107, Cohen’s d = 0.43), left hippocampus (t(58) = 1.62, p = .110, Cohen’s d = 0.43), and left IFG (t(58) = 1.48, p = .143, Cohen’s d = 0.39), right IFG (t(58) = 1.11, p = .272, Cohen’s d = 0.29; see Figure 1). Exploratory single sample t-tests revealed that controls showed significant activation in both right (t(32) = 2.35, p = .025, Cohen’s d = 0.41) and left (t(32) = 2.61, p = .014, Cohen’s d = 0.45) hippocampus during encoding, but MJ users did not (right hippocampus: t(26) = −0.04, p = .97, Cohen’s d = −0.01; left hippocampus: t(26) = −0.08, p = .94, Cohen’s d = −0.02). In the IFG, controls showed significant activation in both right (t(32) = 4.52, p < .001, Cohen’s d = 0.79) and left (t(32) = 4.57, p < .001, Cohen’s d = 0.79) IFG ROIs, and MJ users showed significant activation in the right IFG (t(26) = 2.55, p = .017, Cohen’s d = 0.49) but not the left (t(26) = 1.65, p = .112, Cohen’s d = 0.32). Exploratory whole-brain analyses demonstrated that MJ users showed less BOLD response than controls in additional regions, including cerebellum, left insula, left basal ganglia, left superior frontal gyrus, right precentral gyrus, and bilateral parahippocampal gyri (voxel-wise p < .05, uncorrected, see Figure 3).
Figure 1.
Average BOLD response in each ROI during encoding of stimuli that were subsequently correctly recognized. Error bars represent +/− 1 standard error.
* Single sample t-test shows significant BOLD response (p < .05)
Figure 3.
Whole-brain group differences in BOLD response during correct encoding (voxel-wise p<.05, uncorrected, cluster size 2700μl for display purposes). Blue regions indicate areas where MJ users showed less BOLD response than controls.
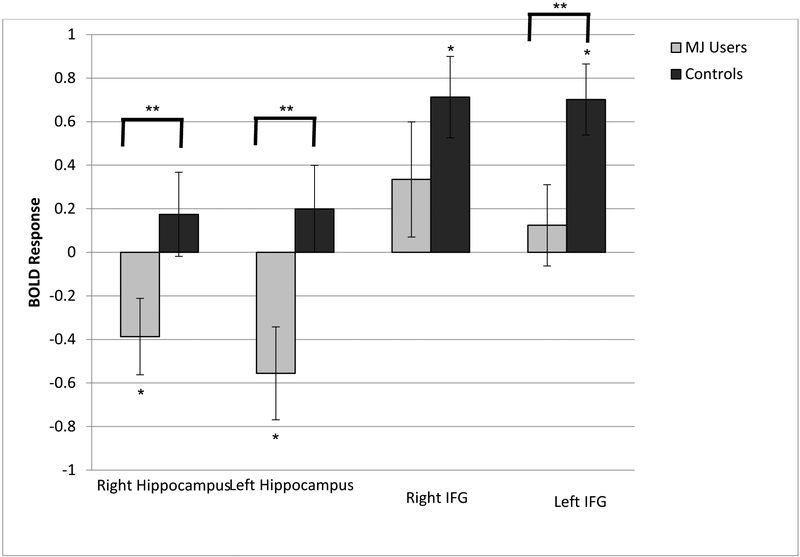
During the recognition phase, MJ users showed significantly less BOLD activation during hits than nonusers in right hippocampus (t(58) = 2.11, p = .039, Cohen’s d = 0.55), left hippocampus (t(58) = 2.57, p = .013, Cohen’s d = 0.67), and left IFG (t(58) = 2.33, p = .023, Cohen’s d = 0.61), but not in the right IFG (t(58) = 1.20, p = .236, Cohen’s d = 0.32; see Figure 2). Follow-up single sample t-tests revealed the nature of these group differences. In the hippocampus, MJ users showed significant deactivation in both right (t(26) = −2.21, p = .036, Cohen’s d = −0.42) and left (t(26) = −2.60, p = .015, Cohen’s d = −0.42) hippocampal ROIs, but controls did not show significant hippocampal response (right hippocampus: t(32) = 0.90, p = .37, Cohen’s d = 0.16; left hippocampus: t(32) = 1.0, p = .33, Cohen’s d = 0.17). In IFG, controls showed significant activation in both right (t(32) = 3.81, p = .001, Cohen’s d = 0.66) and left (t(32) = 4.29, p <.001, Cohen’s d = 0.75) IFG, but MJ users did not show significant response (right IFG: t(26) = 0.90, p = .37, Cohen’s d = 0.24; left IFG: t(26) = 1.0, p = .33, Cohen’s d = 0.13). Nonparametric correlations revealed that higher MJ use was associated with lower activation during recognition in left hippocampus (Spearman’s rho = −0.35, p = .005) and left IFG (Spearman’s rho = −0.28, p = .028), with a trend in right hippocampus (Spearman’s rho = −0.25, p = .060). Exploratory whole-brain analyses demonstrated that MJ users showed less BOLD response than controls in additional regions, including cerebellum, bilateral insula, bilateral basal ganglia, cingulate, and left posterior parietal cortex (voxel-wise p < .05, uncorrected, see Figure 4). Maximum drinks per month was significantly correlated to fMRI response during recognition in left IFG (r = −0.288, p = .025) but not in any other region. After including drinks per month in the model, neither drinks per month (F(1,57) = 1.67, p = .20) nor MJ user group (F(1,57) = 1.84, p = 0.18) was significantly related to left IFG response during recognition. Number of lifetime drinks was significantly correlated to fMRI response during recognition in left hippocampus (r = −0.320, p = .013) and in left IFG (r = −0.290, p = .025). In left hippocampus, after including lifetime drinks in the model, both lifetime drinks (F(1,57) = 4.23, p = .044) and MJ user group (F(1,57) = 4.22, p = .045) were significantly related to fMRI response during recognition. In left IFG, after including lifetime drinks in the model, neither lifetime drinks (F(1,57) = 3.34, p = .073) nor MJ user group (F(1,57) = 3.46, p = .068) were significantly related to fMRI response during recognition.
Figure 2.
Average BOLD response in each ROI during recognition “hits.” Error bars represent +/− 1 standard error.
* Single sample t-test shows significant BOLD response (p < .05)
** Between groups t-test shows significant difference in BOLD response (p < .05)
Figure 4.
Whole-brain group differences in BOLD response during recognition hits (voxel-wise p<.05, uncorrected, cluster size 2700μl for display purposes). Blue regions indicate areas where MJ users showed less BOLD response than controls.
Exploratory analyses of sex differences indicated significant group × sex interactions during recognition in left hippocampus (F(1,56) = 8.87, p = .007, partial η2 = 0.121), right IFG (F(1,56) = 9.63, p = .01, partial η2 = 0.112), and left IFG (F(1,56) = 7.28, p = .004, partial η2 = 0.138). Post-hoc tests showed that in males, MJ users showed less response than controls in each of these regions (left hippocampus: t(21) = 3.47, p = .002, Cohen’s d = 1.51; right IFG: t(21) = 2.68, p = .014, Cohen’s d = 1.17; left IFG: t(21) = 3.41, p = .003, Cohen’s d = 1.49), but females showed no group differences (all ps > .4).
4. Discussion
In this study, we examined the relationship between marijuana use and fMRI response during a visual encoding and recognition task in emerging adult college students. Although no group differences were observed during encoding, MJ users demonstrated less response than non-using controls in bilateral hippocampi and left IFG during recognition, and more MJ involvement was associated with lower fMRI response.
Among controls, we observed significant activation in right and left hippocampus during encoding, as well as activation in both left and right IFG during encoding and during recognition. Together, these findings parallel our previous work with this task (Jamadar et al., 2013) and corroborate the utility of this paradigm in activating these brain regions. Although there were no group differences during the encoding phase of the task, exploratory analyses determined that MJ users showed no significant hippocampal BOLD response during encoding, whereas controls did exhibit significant hippocampal activation. Thus, a group difference might be revealed with larger sample sizes. This is consistent with previous work demonstrating lack of significant hippocampal response during a verbal associative learning task in adolescent MJ users (Schweinsburg et al., 2011). Given that prefrontal-medial temporal relationships are considered a crucial to successful encoding (Kim, 2011b), it is possible that inadequate engagement of these regions by MJ users contributes to poorer encoding.
Importantly, group differences were observed during recognition in bilateral hippocampi and left IFG, and more MJ use was associated with larger deviations in BOLD response. These disparate activation patterns could provide insight into the specific aspects of memory function that are impacted in MJ users. For instance, in previous work examining verbal learning, adolescent MJ users showed intact learning, but poorer delayed recall and more false alarms (Winward et al., 2014). The current results point toward neural mechanisms underlying these cognitive findings. During long-term memory retrieval, the IFG is thought to play a role in selecting goal-relevant information, particularly for single item recognition tasks (Scalici et al., 2017). Therefore, less IFG response during recognition in MJ users could point to poorer engagement while selecting responses. In addition, MJ users showed altered hippocampal response during recognition. Similarly, in an fMRI study spatial learning, adult MJ users did not show differences in medial temporal fMRI response compared to non-users during learning, but showed reduced fMRI response during retrieval (Sneider et al., 2013). Our findings also parallel those of other imaging modalities. For instance, MJ use in adolescents and emerging adults was related to resting state fronto-temporal connectivity (Houck et al., 2013) and poorer hippocampal white matter integrity (Yucel et al., 2010). Together, these studies point to abnormal frontal-hippocampal circuitry associated with MJ use in young people.
There is evidence from rodent models indicating that exposure to cannabinoids interferes with prefrontal and hippocampal plasticity, as well as memory performance, to a greater degree in adolescents compared to adults. For instance, administration of delta-9-tetrahydrocannabinol (THC), the primary psychoactive constituent of MJ, led to greater abnormalities in hippocampal protein expression and memory performance in adolescents compared to adults (Quinn et al., 2008). Similarly, adolescent THC exposure was associated with poorer spatial memory and altered hippocampal glutamate receptor expression (Zamberletti et al., 2016). Moreover, poorer spatial memory performance was related to decreased hippocampal neurogenesis following synthetic cannabinoid exposure in adolescents, but not in adults (Abboussi et al., 2014). In addition, adolescent THC treatment led to impaired endocannabinoid signaling and altered maturation of glutamate receptors in prefrontal cortex, as well as poorer spatial memory (Rubino et al., 2015). Thus, developing adolescent prefrontal-hippocampal circuits appear to be particularly susceptible to MJ-related neuronal damage. It is possible that similar mechanisms underlie memory dysfunction in human adolescents and emerging adults, contributing to the observed results in the current study. Future studies with additional techniques should attempt to identify neurochemical abnormalities underlying these differences in fMRI response.
Interestingly, our exploratory analyses indicated that group differences may be specific to males, though this finding should be interpreted with caution given the relatively small sample size. In contrast to our observations, there have been some studies suggesting that females show greater effects from adolescent MJ exposure. Preclinical evidence indicates that adolescent THC treatment has a larger impact on the cannabinoid system in emotional and memory circuitry in females compared to males (Rubino et al., 2008; Silva et al., 2015). The few extant human studies seem to corroborate the preclinical literature. For example, in young adults, MJ use was associated with greater decrements in episodic memory function in females than in males (Crane et al., 2013). Female adolescent MJ users exhibited larger amygdala volumes compared to female nonusers, but no difference was observed in males (Padula et al., 2007). Similarly, female adolescent MJ users showed larger prefrontal volumes than female controls, whereas male users showed smaller prefrontal volumes than male controls (Medina et al., 2008). It is possible that the discrepant findings between our study and others are related, in part, to interactions between MJ and varying hormone levels in females. For instance, estrogen regulates hippocampal function and enhances memory, and females show changes in memory abilities as levels fluctuate (Frick, 2015). In rats, THC-induced impairments in nonspatial learning and memory were mitigated by estradiol treatment (Daniel et al., 2002). In addition, ovarian hormone levels influence behavioral effects of MJ as well as cannabinoid receptor distribution (Brents, 2016). Together, interactive effects between hormones and MJ may contribute to sex differences in the impact of MJ use on memory. This clearly presents an important avenue for future research.
The current study provides unique insight into the neural mechanisms of memory function in emerging adult MJ users using an established fMRI paradigm. However, there are limitations to this work that should be considered. We only ascertained MJ use in the 3 months prior to scanning, and did not collect information on lifetime use patterns. Our findings are consistent with previous literature indicating memory dysfunction associated with recent heavy MJ use in both adults (Grant et al., 2003) and adolescents (Lisdahl et al., 2014; Schweinsburg et al., 2008). However, it is unclear how our results might be influenced by lifetime patterns of use, which were not assessed. Future studies should characterize the impact of varying patterns of lifetime use, as well as whether these effects would differ with more distal use. In addition, MJ use in the current study was relatively light overall, with most participants only using 5 times per month or less. Thus, our results expand upon existing work by demonstrating observable effects even in low level MJ users. In addition, it is unknown whether the observed fMRI differences might pre-date MJ use. For instance, there are likely pre-existing prefrontal abnormalities in adolescent drinkers that confer risk for developing substance use disorders (Silveri et al., 2016). Similarly, young adults in remission from adolescent cannabis use disorder show altered prefrontal response during a risk-taking task, which could reflect pre-existing vulnerability to MJ use (De Bellis et al., 2013). Importantly, although MJ users in the current study consumed more alcohol than controls in the 3 months before scanning, number of drinks per month was unrelated to hippocampal response in this sample, suggesting that recent alcohol use did not account for the observed effect in MJ users. However, it is unclear whether other unmeasured characteristics, such as lifetime substance use history, age of first MJ use, or personality may have influenced group differences. These relationships should be clarified by carrying out additional work with larger samples, examining varying patterns of lifetime MJ use, and implementing additional imaging techniques.
5. Conclusions
In sum, we identified reduced inferior frontal and hippocampal fMRI response during nonverbal recognition memory among emerging adult MJ users. MJ users failed to show significant hippocampal activation during encoding, but did not significantly differ from controls. Together, these findings suggest poorer engagement of prefrontal-hippocampal circuits during recognition, particularly in relation to response selection. Importantly, these results could contribute to our previously-observed declines in academic performance in college MJ users (Meda et al., 2017), and may have implications for future academic and occupational success.
References
- Abboussi O, Tazi A, Paizanis E, El Ganouni S, 2014. Chronic exposure to WIN55,212–2 affects more potently spatial learning and memory in adolescents than in adult rats via a negative action on dorsal hippocampal neurogenesis. Pharmacol Biochem Behav 120, 95–102. [DOI] [PubMed] [Google Scholar]
- Agosti V, Nunes E, Levin F, 2002. Rates of psychiatric comorbidity among U.S. residents with lifetime cannabis dependence. Am J Drug Alcohol Abuse 28(4), 643–652. [DOI] [PubMed] [Google Scholar]
- Beason-Held LL, Golski S, Kraut MA, Esposito G, Resnick SM, 2005. Brain activation during encoding and recognition of verbal and figural information in older adults. Neurobiol Aging 26(2), 237–250. [DOI] [PubMed] [Google Scholar]
- Becker B, Wagner D, Gouzoulis-Mayfrank E, Spuentrup E, Daumann J, 2010. Altered parahippocampal functioning in cannabis users is related to the frequency of use. Psychopharmacology (Berl) 209(4), 361–374. [DOI] [PubMed] [Google Scholar]
- Blumenfeld RS, Ranganath C, 2007. Prefrontal cortex and long-term memory encoding: an integrative review of findings from neuropsychology and neuroimaging. Neuroscientist 13(3), 280–291. [DOI] [PubMed] [Google Scholar]
- Brents LK, 2016. Marijuana, the Endocannabinoid System and the Female Reproductive System. Yale J Biol Med 89(2), 175–191. [PMC free article] [PubMed] [Google Scholar]
- Crane NA, Schuster RM, Gonzalez R, 2013. Preliminary evidence for a sex-specific relationship between amount of cannabis use and neurocognitive performance in young adult cannabis users. J Int Neuropsychol Soc 19(9), 1009–1015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dager AD, Anderson BM, Rosen R, Khadka S, Sawyer B, Jiantonio-Kelly RE, Austad CS, Raskin SA, Tennen H, Wood RM, Fallahi CR, Pearlson GD, 2014a. Functional magnetic resonance imaging (fMRI) response to alcohol pictures predicts subsequent transition to heavy drinking in college students. Addiction 109(4), 585–595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dager AD, Anderson BM, Stevens MC, Pulido C, Rosen R, Jiantonio-Kelly RE, Sisante J, Raskin SA, Tennen H, Austad CS, Wood RM, Fallahi CR, Pearlson GD, 2013. Influence of alcohol use and family history of alcoholism on neural response to alcohol cues in college drinkers. Alcohol Clin Exp Res 37(Suppl 1), E161 – 171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dager AD, Jamadar S, Stevens MC, Rosen R, Jiantonio-Kelly RE, Sisante J, Raskin SA, Tennen H, Austad CS, Wood RM, Fallahi CR, Pearlson GD, 2014b. fMRI response during figural memory task performance in college drinkers. Psychopharmacology (Berl) 231(1), 167–179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Daniel JM, Winsauer PJ, Brauner IN, Moerschbaecher JM, 2002. Estrogen improves response accuracy and attenuates the disruptive effects of delta9-THC in ovariectomized rats responding under a multiple schedule of repeated acquisition and performance. Behav Neurosci 116(6), 989–998. [PubMed] [Google Scholar]
- De Bellis MD, Wang L, Bergman SR, Yaxley RH, Hooper SR, Huettel SA, 2013. Neural mechanisms of risky decision-making and reward response in adolescent onset cannabis use disorder. Drug Alcohol Depend 133(1), 134–145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Destrieux C, Fischl B, Dale A, Halgren E, 2010. Automatic parcellation of human cortical gyri and sulci using standard anatomical nomenclature. Neuroimage 53(1), 1–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fergusson DM, Boden JM, 2008. Cannabis use and later life outcomes. Addiction 103(6), 969–976; discussion 977–968. [DOI] [PubMed] [Google Scholar]
- Frick KM, 2015. Molecular mechanisms underlying the memory-enhancing effects of estradiol. Horm Behav 74, 4–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Glass M, Dragunow M, Faull RL, 1997. Cannabinoid receptors in the human brain: a detailed anatomical and quantitative autoradiographic study in the fetal, neonatal and adult human brain. Neuroscience 77(2), 299–318. [DOI] [PubMed] [Google Scholar]
- Gogtay N, Giedd JN, Lusk L, Hayashi KM, Greenstein D, Vaituzis AC, Nugent TF 3rd, Herman DH, Clasen LS, Toga AW, Rapoport JL, Thompson PM, 2004. Dynamic mapping of human cortical development during childhood through early adulthood. Proc Natl Acad Sci U S A 101(21), 8174–8179. Epub 2004 May 8117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grant I, Gonzalez R, Carey CL, Natarajan L, Wolfson T, 2003. Non-acute (residual) neurocognitive effects of cannabis use: a meta-analytic study. J Int Neuropsychol Soc 9(5), 679–689. [DOI] [PubMed] [Google Scholar]
- Harrington GS, Tomaszewski Farias S, Buonocore MH, Yonelinas AP, 2006. The intersubject and intrasubject reproducibility of FMRI activation during three encoding tasks: implications for clinical applications. Neuroradiology 48(7), 495–505. [DOI] [PubMed] [Google Scholar]
- Heatherton TF, Kozlowski LT, Frecker RC, Fagerstrom KO, 1991. The Fagerstrom Test for Nicotine Dependence: a revision of the Fagerstrom Tolerance Questionnaire. Br J Addict. 86(9), 1119–1127. [DOI] [PubMed] [Google Scholar]
- Herkenham M, Lynn AB, Little MD, Johnson MR, Melvin LS, de Costa BR, Rice KC, 1990. Cannabinoid receptor localization in brain. Proc Natl Acad Sci U S A 87(5), 1932–1936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hooper SR, Woolley D, De Bellis MD, 2014. Intellectual, neurocognitive, and academic achievement in abstinent adolescents with cannabis use disorder. Psychopharmacology (Berl) 231(8), 1467–1477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Houck JM, Bryan AD, Feldstein Ewing SW, 2013. Functional connectivity and cannabis use in high-risk adolescents. Am J Drug Alcohol Abuse 39(6), 414–423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jacobus J, Tapert SF, 2013. Effects of Cannabis on the Adolescent Brain. Curr Pharm Des. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jager G, Block RI, Luijten M, Ramsey NF, 2010. Cannabis use and memory brain function in adolescent boys: a cross-sectional multicenter functional magnetic resonance imaging study. J Am Acad Child Adolesc Psychiatry 49(6), 561–572, 572 e561–563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jamadar S, Assaf M, Jagannathan K, Anderson K, Pearlson GD, 2013. Figural memory performance and fMRI activity across the adult lifespan. Neurobiol Aging 34(1), 110–127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim H, 2011a. Differential neural activity in the recognition of old versus new events: An Activation Likelihood Estimation Meta-Analysis. Differential neural activity in the recognition of old versus new events: An Activation Likelihood Estimation Meta-Analysis. [DOI] [PMC free article] [PubMed]
- Kim H, 2011b. Neural activity that predicts subsequent memory and forgetting: a meta-analysis of 74 fMRI studies. Neuroimage 54(3), 2446–2461. [DOI] [PubMed] [Google Scholar]
- Kim H, 2013. Differential neural activity in the recognition of old versus new events: an activation likelihood estimation meta-analysis. Hum Brain Mapp 34(4), 814–836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lenroot RK, Giedd JN, 2006. Brain development in children and adolescents: insights from anatomical magnetic resonance imaging. Neurosci Biobehav Rev 30(6), 718–729. [DOI] [PubMed] [Google Scholar]
- Lisdahl KM, Gilbart ER, Wright NE, Shollenbarger S, 2013. Dare to delay? The impacts of adolescent alcohol and marijuana use onset on cognition, brain structure, and function. Front Psychiatry 4, 53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lisdahl KM, Wright NE, Kirchner-Medina C, Maple KE, Shollenbarger S, 2014. Considering Cannabis: The Effects of Regular Cannabis Use on Neurocognition in Adolescents and Young Adults. Curr Addict Rep 1(2), 144–156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Macleod J, Oakes R, Copello A, Crome I, Egger M, Hickman M, Oppenkowski T, Stokes-Lampard H, Davey Smith G, 2004. Psychological and social sequelae of cannabis and other illicit drug use by young people: a systematic review of longitudinal, general population studies. Lancet 363(9421), 1579–1588. [DOI] [PubMed] [Google Scholar]
- Macmillan NA, Creelman CD, 2005. Detection theory: A user’s guide, 2nd ed Erlbaum, Mahwah, NJ. [Google Scholar]
- Meda SA, Gueorguieva RV, Pittman B, Rosen RR, Aslanzadeh F, Tennen H, Leen S, Hawkins K, Raskin S, Wood RM, Austad CS, Dager A, Fallahi C, Pearlson GD, 2017. Longitudinal influence of alcohol and marijuana use on academic performance in college students. PLoS One 12(3), e0172213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Medina KL, McQueeny T, Nagel BJ, Hanson KL, Schweinsburg AD, Tapert SF, 2008. Prefrontal cortex volumes in adolescents with alcohol use disorders: unique gender effects. Alcohol Clin Exp Res 32(3), 386–394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Padula CB, Schweinsburg AD, Tapert SF, 2007. Spatial working memory performance and fMRI activation interaction in abstinent adolescent marijuana users. Psychol Addict Behav 21(4), 478–487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quinn HR, Matsumoto I, Callaghan PD, Long LE, Arnold JC, Gunasekaran N, Thompson MR, Dawson B, Mallet PE, Kashem MA, Matsuda-Matsumoto H, Iwazaki T, McGregor IS, 2008. Adolescent rats find repeated delta(9)-THC less aversive than adult rats but display greater residual cognitive deficits and changes in hippocampal protein expression following exposure. Neuropsychopharmacology 33(5), 1113–1126. [DOI] [PubMed] [Google Scholar]
- Rubino T, Prini P, Piscitelli F, Zamberletti E, Trusel M, Melis M, Sagheddu C, Ligresti A, Tonini R, Di Marzo V, Parolaro D, 2015. Adolescent exposure to THC in female rats disrupts developmental changes in the prefrontal cortex. Neurobiol Dis 73, 60–69. [DOI] [PubMed] [Google Scholar]
- Rubino T, Vigano D, Realini N, Guidali C, Braida D, Capurro V, Castiglioni C, Cherubino F, Romualdi P, Candeletti S, Sala M, Parolaro D, 2008. Chronic delta 9-tetrahydrocannabinol during adolescence provokes sex-dependent changes in the emotional profile in adult rats: behavioral and biochemical correlates. Neuropsychopharmacology 33(11), 2760–2771. [DOI] [PubMed] [Google Scholar]
- SAMHSA, 2016. Key substance use and mental health indicators in the United States: Results from the 2015 National Survey on Drug Use and Health, NSDUH Series H-51 Center for Behavioral Health Statistics and Quality, Rockville, MD. [Google Scholar]
- Scalici F, Caltagirone C, Carlesimo GA, 2017. The contribution of different prefrontal cortex regions to recollection and familiarity. A review of fMRI data. Neurosci Biobehav Rev. [DOI] [PubMed] [Google Scholar]
- Schweinsburg AD, Brown SA, Tapert SF, 2008. The influence of marijuana use on neurocognitive functioning in adolescents. Curr Drug Abuse Rev 1(1), 99–111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schweinsburg AD, McQueeny T, Nagel BJ, Eyler LT, Tapert SF, 2010. A preliminary study of functional magnetic resonance imaging response during verbal encoding among adolescent binge drinkers. Alcohol 44(1), 111–117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schweinsburg AD, Schweinsburg BC, Nagel BJ, Eyler LT, Tapert SF, 2011. Neural correlates of verbal learning in adolescent alcohol and marijuana users. Addiction 106(3), 564–573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sheehan DV, Lecrubier Y, Sheehan KH, Amorim P, Janavs J, Weiller E, Hergueta T, Baker R, Dunbar GC, 1998. The Mini-International Neuropsychiatric Interview (M.I.N.I.): the development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J Clin Psychiatry 59 Suppl 20, 22–33;quiz 34–57. [PubMed] [Google Scholar]
- Shulenberg JE, Johnston LD, O’Malley PM, Bachman JG, Miech RA, Patrick ME, 2017. Monitoring the Future national survey results on drug use, 1975–2016: Volume II, College students and adults ages 19–55 Institute for Social Research, The Univerisity of Michigan, Ann Arbor, Michigan. [Google Scholar]
- Silva L, Harte-Hargrove L, Izenwasser S, Frank A, Wade D, Dow-Edwards D, 2015. Sex-specific alterations in hippocampal cannabinoid 1 receptor expression following adolescent delta-9-tetrahydrocannabinol treatment in the rat. Neurosci Lett 602, 89–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Silveri MM, Dager AD, Cohen-Gilbert JE, Sneider JT, 2016. Neurobiological signatures associated with alcohol and drug use in the human adolescent brain. Neurosci Biobehav Rev 70, 244–259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sneider JT, Gruber SA, Rogowska J, Silveri MM, Yurgelun-Todd DA, 2013. A preliminary study of functional brain activation among marijuana users during performance of a virtual water maze task. J Addict 2013, 461029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Solowij N, Battisti R, 2008. The chronic effects of cannabis on memory in humans: a review. Curr Drug Abuse Rev 1(1), 81–98. [DOI] [PubMed] [Google Scholar]
- Winward JL, Hanson KL, Tapert SF, Brown SA, 2014. Heavy alcohol use, marijuana use, and concomitant use by adolescents are associated with unique and shared cognitive decrements. J Int Neuropsychol Soc 20(8), 784–795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yucel M, Zalesky A, Takagi MJ, Bora E, Fornito A, Ditchfield M, Egan GF, Pantelis C, Lubman DI, 2010. White-matter abnormalities in adolescents with long-term inhalant and cannabis use: a diffusion magnetic resonance imaging study. White-matter abnormalities in adolescents with long-term inhalant and cannabis use: a diffusion magnetic resonance imaging study 35(6), 409–412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zamberletti E, Gabaglio M, Grilli M, Prini P, Catanese A, Pittaluga A, Marchi M, Rubino T, Parolaro D, 2016. Long-term hippocampal glutamate synapse and astrocyte dysfunctions underlying the altered phenotype induced by adolescent THC treatment in male rats. Pharmacol Res 111, 459–470. [DOI] [PubMed] [Google Scholar]