Abstract
Dopaminergic neurotransmission in the prefrontal cortex (PFC) plays an important role in regulating cognitive processes and emotional status. The dopamine D4 receptor, which is highly enriched in the PFC, is one of the principal targets of antipsychotic drugs. To understand the cellular mechanisms and functional implications of D4 receptors, we examined the impact of D4receptors in PFC pyramidal neurons on GABAergic inhibition, a key element in the regulation of “working memory.” Application of the D4 agonistN-(methyl)-4-(2-cyanophenyl)piperazinyl-3-methylbenzamide maleate caused a reversible decrease in postsynaptic GABAA receptor currents; this effect was blocked by the D4 antagonist 3-[(4-[4-chlorophenyl]piperazine-1-yl)methyl]-[1H]-pyrrolo[2,3-b]pyridine but not by the D2 antagonist sulpiride, suggesting mediation by D4 receptors. Application of PD168077 also reduced the GABAA receptor-mediated miniature IPSC amplitude in PFC pyramidal neurons recorded from slices. The D4 modulation of GABAA receptor currents was blocked by protein kinase A (PKA) activation and occluded by PKA inhibition. Inhibiting the catalytic activity of protein phosphatase 1 (PP1) also eliminated the effect of PD168077 on GABAAcurrents. Furthermore, disrupting the association of the PKA/PP1 complex with its scaffold protein Yotiao significantly attenuated the D4 modulation of GABAA currents, suggesting that Yotiao-mediated targeting of PKA/PP1 to the vicinity of GABAA receptors is required for the dopaminergic signaling. Together, our results show that activation of D4 receptors in PFC pyramidal neurons inhibits GABAA channel functions by regulating the PKA/PP1 signaling complex, which could underlie the D4 modulation of PFC neuronal activity and the actions of antipsychotic drugs.
Keywords: dopamine receptors, GABAA receptor channels, protein kinase A, protein phosphatase 1, Yotiao, inhibitor-1
The prefrontal cortex (PFC), a brain region highly associated with cognitive and emotional processes (Goldman-Rakic, 1995; Miller, 1999), receives a major dopaminergic input from the ventral tegmental area (Lewis et al., 1986; Berger et al., 1988). Regional depletion of dopamine in the PFC of monkeys produces impairments in working memory performance (Brozoski et al., 1979), suggesting that PFC dopaminergic transmission plays a key role in cognitive functions. Disorders in dopaminergic signaling are thought to underlie the etiology of many neuropsychiatric disorders, including schizophrenia and depression (Desimone, 1995), and almost all effective antipsychotic drugs target dopamine receptors (Lidow and Goldman-Rakic, 1994; Seeman and Van Tol, 1994). Dopamine can have both inhibitory and excitatory functions in neuronal networks through the coupling of different dopamine receptors to distinct ion channels (for review, seeNicola et al., 2000). These receptors have been classified as either D1-like (D1, D5) or D2-like (D2, D3, D4), based on their sequence homology, pharmacological profiles, and distinct downstream signal transduction pathways. Although considerable evidence suggests that D1 receptors are critically involved in regulating the working memory functions of the PFC (Sawaguchi and Goldman-Rakic, 1991; Williams and Goldman-Rakic, 1995), little is known about the cellular mechanisms and functional consequences of D2-like receptor-mediated signaling in the PFC.
Among the D2-like receptors, D4 receptor is expressed at the highest level in PFC pyramidal principal neurons and GABAergic interneurons (Mrzljak et al., 1996; Wedzony et al., 2000). Given its high affinity for atypical antipsychotic drugs that constitute a major improvement in the treatment of schizophrenia (Van Tol et al., 1991; Kapur and Remington, 2001), D4 receptor has been suggested to play an important role in PFC cognitive functions and therefore to be involved in the pathophysiology of neuropsychiatric disorders (for review, seeOak et al., 2000). In agreement with this, D4receptor antagonists have been found to alleviate stress-induced working memory deficits in monkeys (Murphy et al., 1996) and to ameliorate the cognitive deficits exhibited by monkeys after long-term treatment with the psychotomimetic drug phencyclidine (Jentsch et al., 1997, 1999). Moreover, altered cortical excitability and reduced exploration of novel stimuli have been shown in D4 receptor-deficient mice (Dulawa et al., 1999;Rubinstein et al., 2001), and significantly elevated D4 receptors have been demonstrated in patients with schizophrenia (Seeman et al., 1993). To better understand the functional role of D4 receptors under normal and pathological conditions, it is important to determine cellular substrates of D4 receptors that are involved in the modulation of PFC neuronal activity and cognitive processes.
Recent evidence indicates that GABAergic inhibition in the PFC plays a key role in working memory by sculpting the temporal profile of activation of the neurons during cognitive operations and thereby shaping the temporal flow of information (Constantinidis et al., 2002). Computational models of the dopaminergic modulation of working memory processing predict that dopamine-mediated alterations in GABAA currents in PFC pyramidal neurons are critical for maintaining the specificity and stability of delay-period activity (Durstewitz et al., 2000). Therefore, understanding how the D4 receptor modulates GABAergic inhibition would provide important insights into its role in cognitive functions associated with the PFC.
MATERIALS AND METHODS
Acute-dissociation procedure. PFC neurons from young adult (3–5 weeks postnatal) rats were acutely dissociated using procedures similar to those described previously (Yan and Surmeier, 1996; Feng et al., 2001; Cai et al., 2002). All experiments were performed with the approval of the State University of New York at Buffalo Animal Care Committee. In brief, rats were anesthetized by inhaling 2-bromo-2-chloro-1,1,1-trifluoroe-thane (1 ml/100 gm; Sigma, St. Louis, MO) and decapitated; the brains were quickly removed, iced, and then blocked for slicing. The blocked tissue was cut in 400 μm slices with a Vibratome while bathed in a low Ca2+ (100 μm), HEPES-buffered salt solution containing (in mm): 140 Na isethionate, 2 KCl, 4 MgCl2, 0.1 CaCl2, 23 glucose, 15 HEPES, 1 kynurenic acid, pH 7.4, 300–305 mosm/l. Slices were then incubated for 1–6 hr at room temperature (20–22°C) in an NaHCO3-buffered saline containing (in mm): 126 NaCl, 2.5 KCl, 2 CaCl2, 2 MgCl2, 26 NaHCO3, 1.25 NaH2PO4, 10 glucose, 1 pyruvic acid, 0.05 glutathione, 0.1 NG-nitro-l-arginine, 1 kynurenic acid, pH 7.4, 300–305 mosm/l, and bubbled with 95% O2 and 5% CO2. All reagents were obtained from Sigma.
Slices were then removed into the low Ca2+buffer, and regions of the PFC were dissected and placed in an oxygenated Cell-Stir chamber (Wheaton, Inc., Millville, NJ) containing pronase (1–3 mg/ml) in HEPES-buffered HBSS (Sigma) at 35°C. After 30 min of enzyme digestion, tissue was rinsed three times in the low Ca2+, HEPES-buffered saline and mechanically dissociated with a graded series of fire-polished Pasteur pipettes. The cell suspension was then plated into a 35 mm Lux Petri dish, which was then placed on the stage of a Nikon (Tokyo, Japan) inverted microscope.
Whole-cell recordings. Whole-cell recordings of currents used standard voltage-clamp techniques (Hamill et al., 1981; Yan and Surmeier, 1997). Electrodes were pulled from Corning (Corning, NY) 7052 glass and fire-polished before use. The internal solution (Yan and Surmeier, 1997) consisted of (in mm): 180N-methyl-d-glucamine, 40 HEPES, 4 MgCl2, 0.5 BAPTA, 12 phosphocreatine, 2 Na2ATP, 0.2 Na3GTP, 0.1 leupeptin, pH 7.2–7.3, 265–270 mosm/l. The external solution consisted of (in mm): 135 NaCl, 20 CsCl, 1 MgCl2, 10 HEPES, 5 BaCl2, 10 glucose, 0.001 TTX, pH 7.3–7.4, 300–305 mosm/l.
Recordings were obtained with an Axon Instruments (Union City, CA) 200B patch-clamp amplifier that was controlled and monitored with an IBM personal computer running pClamp (version 8) with a DigiData 1320 series interface. Electrode resistances were typically 2–4 MΩ in the bath. After seal rupture, series resistance (4–10 MΩ) was compensated (70–90%) and periodically monitored. Care was exercised to monitor the constancy of the series resistance; recordings were terminated whenever a significant increase (>20%) occurred. The cell membrane potential was held at 0 mV. The application of GABA (50 μm) evoked a partially desensitizing outward current with the decay rate fitted by a single or double exponential. Peak values were measured for generating the plot as a function of time and drug application. GABA was applied for 2 sec every 30 sec to minimize a desensitization-induced decrease in current amplitude. Drugs were applied with a gravity-fed “sewer pipe” system. The array of application capillaries (∼150 μm inner diameter) was positioned a few hundred micrometers from the cell under study. Solution changes were effected by the SF-77B fast-step solution stimulus delivery device (Warner Instruments, Hamden, CT).
Dopamine receptor ligandsN-(methyl)-4-(2-cyanophenyl)piperazinyl-3-methybenzamide maleate (PD168077) maleate, 3-[(4-[4-chlorophe-nyl]piperazine-1-yl)methyl]-[1H]-pyrrolo[2,3-b]pyridine trihydrochloride (L-745870) (Tocris, Ballwin, MO),R,S-(±)-sulpiride and clozapine (Sigma), as well as the second messenger reagents chlorophenylthio-cAMP (cpt-cAMP), PKC19–36, and PKI[5–24] (Calbiochem, San Diego, CA), microcystin, okadaic acid (OA), and okadaic acid methyl ester (OAE) (Sigma/RBI, Poole, UK) were made up as concentrated stocks in water or DMSO and stored at −20°C. The final DMSO concentration in all applied solutions was <0.1%. No change on GABAA currents has been observed with this concentration of DMSO. Stocks were thawed and diluted immediately before use. The amino acid sequence for the phosphorylated I-1 peptide pThr35I-1[7–39] is PRKIQFTVPLLEPHLDPEAAEQIRRRRP(pT)PATL. The amino acid sequence for the PKA anchoring inhibitory peptide Yotiao[1440–1457] is LEEEVAKVIVSMSIAFAQ. The amino acid sequence for the protein phosphatase 1 (PP1) anchoring inhibitory peptide Gm[63–75] is GRRVSFADNFGFN.
Data analyses were performed with AxoGraph (Axon Instruments), Kaleidagraph (Albeck Software, Reading, PA), Origin 6 (OriginLab Co., Northampton, MA), and Statview (Abacus Concepts, Calabasas, CA). For analysis of statistical significance, Mann–Whitney U tests were performed to compare the current amplitudes in the presence or absence of agonists. ANOVA tests were performed to compare the differential degrees of current modulation between groups subjected to different treatments.
Electrophysiological recordings in slices. To evaluate the regulation of miniature IPSCs (mIPSCs) by D4 receptors in PFC slices, the whole-cell patch technique was used for voltage-clamp recordings using patch electrodes (5–9 MΩ) filled with the following internal solution (in mm): 130 Cs-methanesulfonate, 10 CsCl, 4 NaCl, 10 HEPES, 1 MgCl2, 5 EGTA, 12 phosphocreatine, 5 MgATP, 0.2 Na3GTP, 0.1 leupeptin, pH 7.2–7.3, 265–270 mosm/l. The slice (300 μm) was placed in a perfusion chamber attached to the fixed-stage of an upright microscope (Olympus Optical, Tokyo, Japan) and submerged in continuously flowing oxygenated artificial CSF. Cells were visualized with a 40× water-immersion lens and illuminated with near infrared (IR) light and the image was detected with an IR-sensitive CCD camera (Olympus Optical). A Multiclamp 700A amplifier was used for these recordings (Axon Instruments). Tight seals (2–10 GΩ) from visualized pyramidal neurons were obtained by applying negative pressure. The membrane was disrupted with additional suction and the whole-cell configuration was obtained. The access resistances ranged from 13 to 18 MΩ and were compensated 50–70%. Cells were held at 0 mV for the continuous recording of mIPSCs. The Mini Analysis Program (Synaptosoft, Leonia, NJ) was used to analyze synaptic activity. For each different treatment, mIPSCs of 1 min were used for analysis. Statistical comparisons of the synaptic currents were made using the Kolmogorov–Smirnov (K-S) test.
RESULTS
Activation of D4 receptors reduces GABAAreceptor currents in prefrontal cortical pyramidal neurons
To test the potential impact of D4 dopamine receptors on postsynaptic GABAA receptor channels in PFC, we first examined the effect of PD168077, a potent and highly selective D4 receptor agonist (Glase et al., 1997), on GABAA receptor-mediated currents in dissociated pyramidal neurons located in the intermediate and deep layers (III–VI) of the rat PFC. Acutely isolated PFC pyramidal neurons were readily distinguished from GABAergic interneurons by their distinct morphological features: a pyramidal-shaped soma and a prominent apical dendrite. A representative example is shown in Figure1A. GABA (50 μm) was applied to these neurons, which were voltage-clamped using whole-cell techniques. The application of GABA evoked a partially desensitizing outward current that could be completely blocked by the GABAA receptor antagonist bicuculline (30 μm, data not shown), confirming mediation by the GABAA receptor.
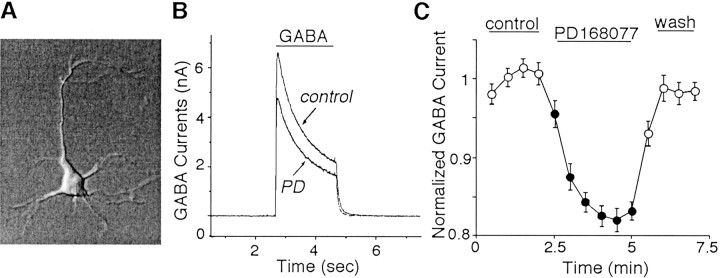
Fig. 1.
Application of the D4agonist PD168077 reversibly reduced GABAA receptor currents in PFC pyramidal neurons. A, Photomicrograph of an acutely isolated PFC pyramidal neuron. B, Currenttraces recorded from the neuron shown inA. The D4 agonist PD168077 (PD; 20 μm) reduced GABA-evoked (50 μm) currents in the cell. C, Plot of peak GABAA current as a function of time and agonist application in a sample of dissociated PFC pyramidal neurons (n= 45).
As shown in Figure 1B, application of PD168077 (20 μm) caused a reduction in the amplitudes of GABAA currents in PFC pyramidal neurons. The modulation was not accompanied by changes in current decay kinetics. The PD168077-induced reduction of GABAA currents was reversible, and it had rapid-onset kinetics, taking 1–2 min to stabilize (Fig. 1C). In a sample of neurons we examined, PD168077 (20 μm) reduced the amplitude of GABA-evoked (50 μm) currents by 15.4 ± 0.6% (n = 86; p < 0.01; Mann–WhitneyU test). After recovery from the first application, a second application of PD168077 resulted in a similar response (93.2 ± 2.4% of first response; n = 18). This PD168077-induced inhibition of GABAA currents did not result from an agonist-independent rundown of the current, because no significant decrease in the current was observed in the absence of PD168077. Similar modulation was observed when different concentrations (25 μm, 100 μm, and 1 mm) of GABA were applied or membrane potentials were held at different levels (−40 mV, −20 mV, and 0 mV) (data not shown). In contrast to the inhibitory effect on GABA-evoked currents, PD168077 (20–50 μm) had no effect on glutamate-evoked (1 mm) currents in dissociated PFC pyramidal neurons tested (n = 10; data not shown).
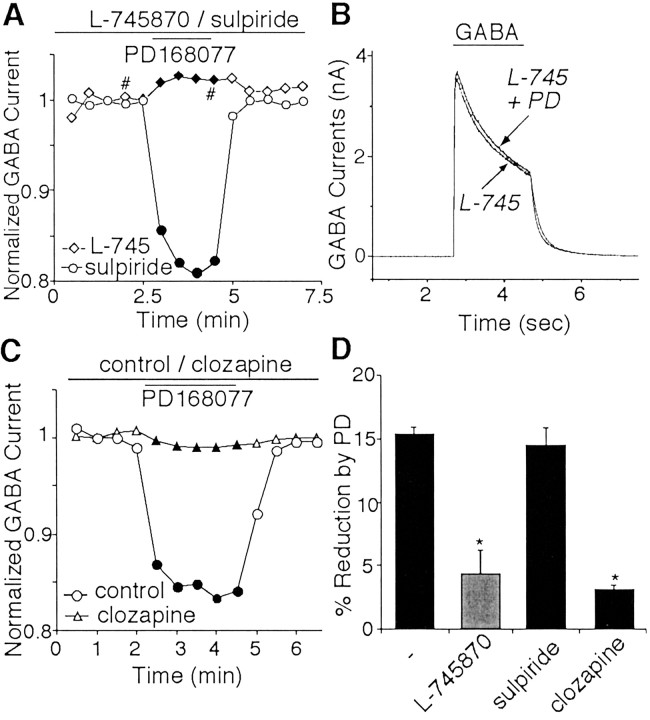
To verify that D4 receptors were mediating the modulation seen with PD168077, we examined the ability of L-745870, a highly selective D4 antagonist (Kulagowski et al., 1996; Patel et al., 1997), to prevent the action of PD168077. Dissociated neurons were treated with L-745870 for 15 min before the examination of the PD168077 effect. As shown in Figure2A and B, in the presence of the D4 antagonist L-745870 (20 μm), PD168077 failed to modulate GABAA currents. In contrast, the PD168077-induced reduction of GABAA currents was still intact in neurons treated with the D2 receptor antagonist sulpiride (20 μm). Because the atypical antipsychotic clozapine has higher affinity to D4receptors compared with D2 receptors (Van Tol et al., 1991), we also examined the effect of clozapine on PD168077 modulation of GABAA currents. As shown in Figure2C, treating neurons with clozapine (20 μm) blocked the PD168077-induced reduction of GABAA currents. The percentage modulation of GABAA currents by PD168077 in the absence or presence of various antagonists is summarized in Figure2D. PD168077 had little effect on GABAA currents in neurons treated with L-745870 (4.2 ± 1.8%; n = 24; p > 0.05; Mann–Whitney U test) or clozapine (3.6 ± 0.4%;n = 10; p > 0.05; Mann–WhitneyU test), which was significantly different from the effect of PD168077 on control cells (15.3 ± 1.1%; n = 36; p < 0.005; ANOVA) or sulpiride-treated neurons (14.8 ± 2.6%; n = 11; p < 0.005; ANOVA). The pharmacological profile of these responses thus identifies D4 as the receptor underlying the PD168077-induced inhibition of GABAAcurrents.
Fig. 2.
The effect of PD168077 on GABAAreceptor currents was mediated by D4 receptors.A, Plot of peak GABAA current as a function of time and agonist application in an L-745870-treated neuron (L-745; diamonds) and a sulpiride-treated neuron (circles). The selective D4antagonist L-745870 (20 μm) but not the D2antagonist sulpiride (20 μm) blocked PD168077-induced reduction of GABAA currents. B, Representative current traces taken from the records used to construct A (at time points denoted by #).C, Plot of peak GABAA current as a function of time and agonist application in a clozapine-treated (20 μm) neuron (triangles) and a nontreated control neuron (circles). D, Cumulative data (means ± SEM) showing the percentage modulation of GABAA currents by PD168077 in the absence (n = 36) or presence of L-745870 (n = 24), sulpiride (n = 11), or clozapine (n = 10). *p < 0.005; ANOVA.
Activation of D4 receptors decreases GABAAreceptor-mediated synaptic transmission in prefrontal cortex
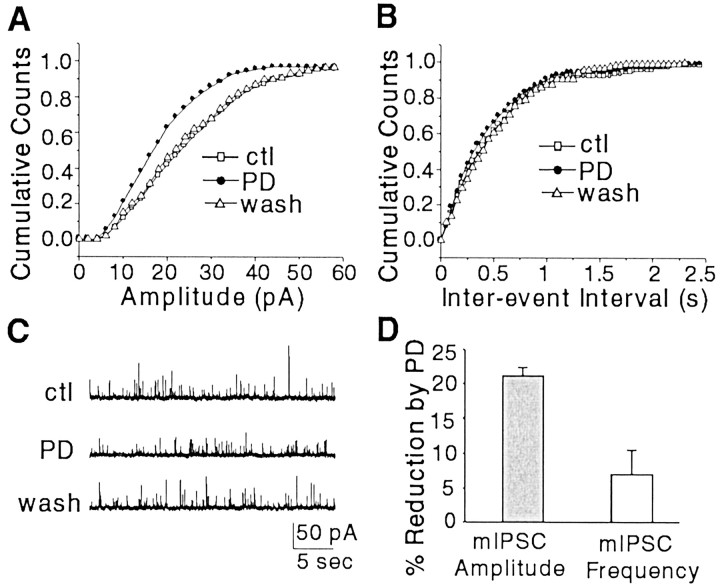
We then examined the effect of PD168077 on GABAA receptor-mediated IPSCs, indicative of the impact of D4 receptors on GABAergic synaptic transmission. PFC slices were exposed to TTX (1 μm), and mIPSCs were recorded in PFC pyramidal neurons to better isolate the postsynaptic effect of D4 receptors. Application of bicuculline (30 μm) blocked the mIPSCs (n = 5), indicating that these synaptic currents are mediated by GABAA receptors. As shown in Figure3A–C, bath application of PD168077 to the PFC slice caused a significant and reversible leftward shift on the distribution of mIPSC amplitudes (p< 0.001; K-S test), but not the distribution of mIPSC frequencies, indicating that PD168077 reduced postsynaptic responses to GABA. In a sample of PFC pyramidal neurons we examined, PD168077 decreased the mean amplitude of mIPSCs by 21.1 ± 1.1% (Fig. 3D) (means ± SEM; n = 27; p < 0.01; Mann–Whitney U test), whereas the frequency of mIPSCs recorded from PFC pyramidal neurons in slices was not significantly changed by PD168077 (Fig. 3D) (6.4 ± 4.1%;n = 27; p > 0.05; Mann–WhitneyU test). These results suggest that activation of D4 receptors could downregulate GABAA receptor functions by a postsynaptic mechanism.
Fig. 3.
Activation of D4 receptors reduced the amplitude of mIPSCs recorded from pyramidal neurons in PFC slices.A, Cumulative plots of the amplitude of mIPSCs in a PFC pyramidal neuron. Note that PD168077 (PD; 20 μm) caused a reversible leftward shift on the distribution of mIPSC amplitudes, indicative of a reduction in the sizes of mIPSCs by PD168077. B, Cumulative plots of the frequency of mIPSCs in the same cell demonstrating that the distribution of mIPSC frequency was not changed by PD168077 (20 μm). C, Representativetraces of mIPSCs recorded from the cell before (control,ctl), during bath application of PD168077, and after washout of the agonist. D, Cumulative data (means ± SEM) showing the percentage modulation of mIPSC amplitude and mIPSC frequency by PD168077 in a sample of PFC pyramidal neurons (n = 27).
D4 modulation of GABAA currents is dependent on the inhibition of protein kinase A
We subsequently examined the signal transduction pathways mediating the modulation of GABAA currents by D4 receptors. GABAAchannels are thought to be heteropentameric structures, composed of different subunits (Macdonald and Olsen, 1994). PKA phosphorylation of GABAA receptor subunits exerts a powerful impact on recombinant and native GABAA channels (Porter et al., 1990; Moss et al., 1992a,b). Activation of D4 receptors can inhibit adenylate cyclase and cAMP formation in transfected cell lines (Chio et al., 1994). This led us to speculate that the D4 reduction of GABAA currents is through the inhibition of PKA. If that is the case, then the effect of D4receptors on GABAA receptor currents should be blocked by stimulating PKA and occluded by inhibiting PKA. To test this, we applied selective PKA activators and inhibitors.
As shown in Figure 4Aand B, application of the membrane-permeable cAMP analog cpt-cAMP (200 μm) caused a small increase in basal GABAA currents. In the presence of cpt-cAMP, PD168077 failed to reduce GABAAcurrents. Removing cpt-cAMP restored the ability of PD168077 to inhibit GABAA currents. To further confirm the involvement of PKA in D4 modulation of GABAA currents, we dialyzed neurons with the specific PKA inhibitory peptide PKI[5–24] (Knighton et al., 1991). The PKC inhibitory peptide PKC19–36 (20 μm) was used as a control. After ∼5 min of dialysis to allow the peptide to enter the cell to inhibit kinase activity, the effect of the subsequent application of PD168077 on GABAA currents was examined. As shown in Figure4C–E, the PD168077-induced reduction of GABAA currents was largely abolished in neurons dialyzed with PKI[5–24] but was almost intact in neurons loaded with PKC19–36. Figure 4F compares the effects of PD168077 in the absence or presence of various kinase activators and inhibitors. PD168077 reduced peak GABAA currents by 4.3 ± 0.8% in the presence of cpt-cAMP (n = 12; p > 0.05; Mann–Whitney U test) and 4.8 ± 0.8% in the presence of PKI[5–24] (n = 8; p > 0.05; Mann–Whitney U test), both of which were significantly smaller than the effect of PD168077 in the absence of these agents (16.2 ± 1.0%; n = 13;p < 0.005; ANOVA) or in the presence of PKC19–36 (14.8 ± 1.6%; n= 14; p < 0.005; ANOVA). These results indicate that reduction of GABAA currents by PD168077 depends on the inhibition of PKA.
Fig. 4.
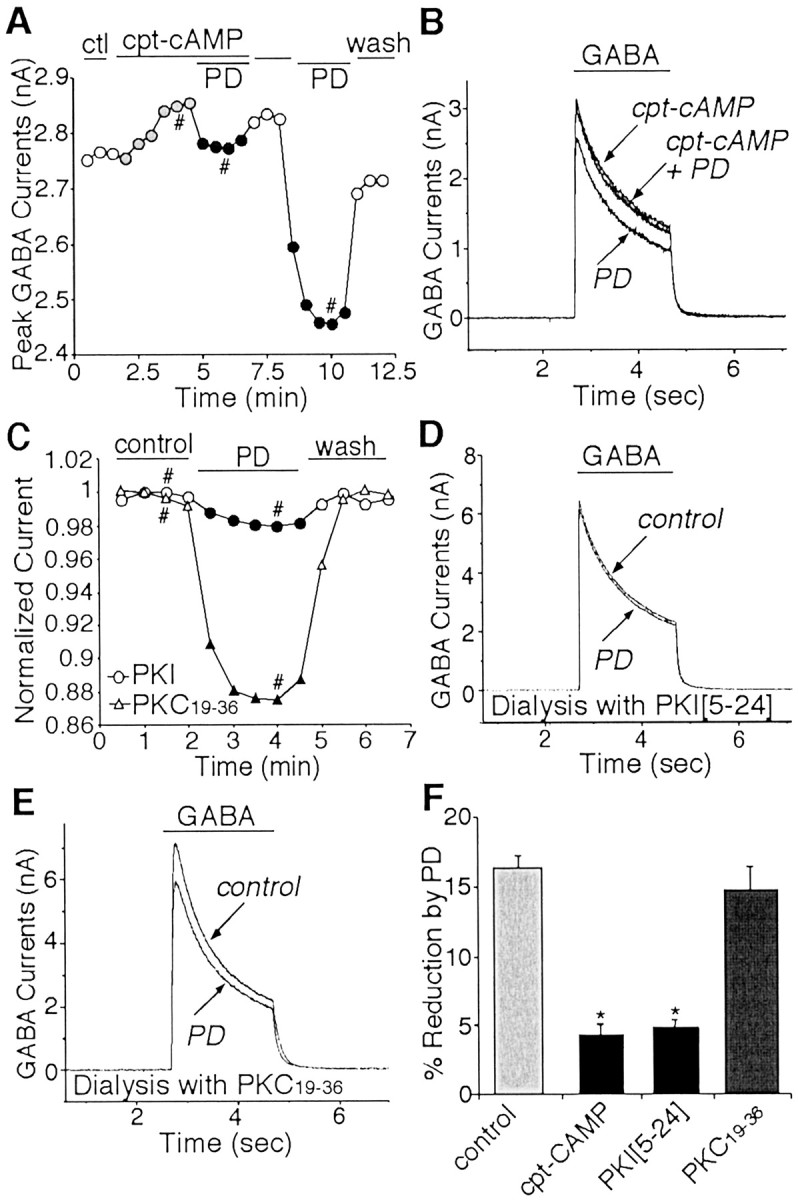
The effect of PD168077 on GABAAcurrents was blocked by PKA activation and occluded by PKA inhibition.A, Plot of peak GABAA currents as a function of time and drug application. In the presence of the membrane-permeable cAMP analog cpt-cAMP (200 μm), PD168077 (PD; 20 μm) failed to reduce GABAA currents. After washing off cpt-cAMP, the effect of PD168077 emerged. B, Representative currenttraces taken from the records used to constructA (at time points denoted by #). C, Plot of peak GABAA currents as a function of time and drug application in neurons dialyzed with PKI[5–24] or PKC19–36. The specific PKA inhibitory peptide PKI[5–24] (20 μm), but not the PKC inhibitory peptide PKC19–36 (20 μm), eliminated PD168077-induced reduction of GABAA currents. D, E, Representative current traces taken from the records used to construct C (at time points denoted by #). F, Cumulative data (means ± SEM) showing the percentage modulation of GABAA currents by PD168077 in the absence (n = 13) or presence of cpt-cAMP (n = 12), PKI[5–24] (n = 8). or PKC19–36 (n = 14). *p < 0.005; ANOVA. ctl, Control.
D4 modulation of GABAA currents requires the activation of protein phosphatase 1
The D4-induced inhibition of PKA could directly modulate GABAA currents through decreased phosphorylation of GABAA receptor β subunits on the PKA sites (Moss et al., 1992a,b; McDonald et al., 1998;Cai et al., 2002). Alternatively, the inhibition of PKA could cause the disinhibition of PP1 via decreased phosphorylation of the inhibitory protein I-1 (Ingebritsen and Cohen, 1983), leading to the increased dephosphorylation of GABAA receptor subunits and downregulation of GABAA currents. To test which is the potential signaling mechanism, we examined the effect of PD168077 on GABAA currents in the presence of phosphatase inhibitors.
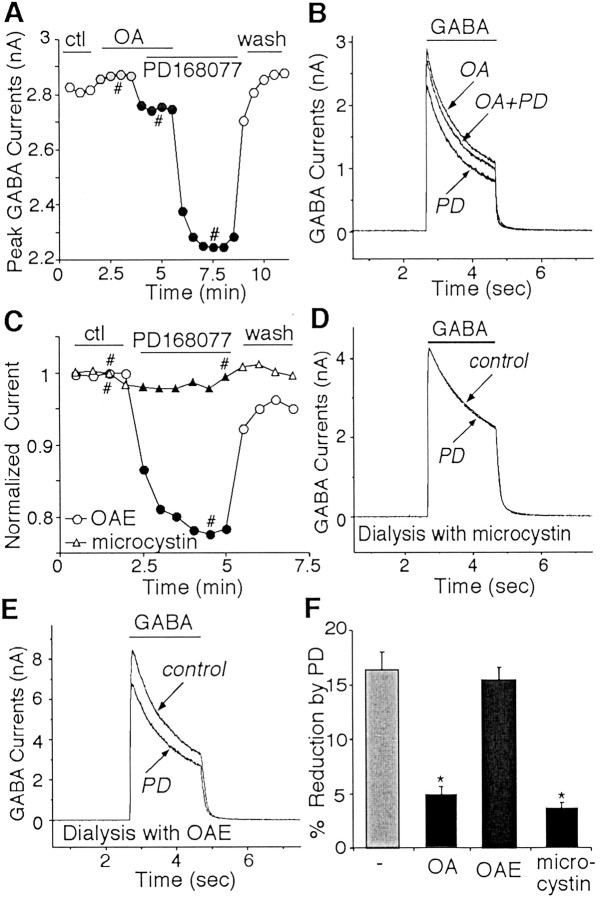
As shown in Figure 5A andB, bath application of the PP1/2A inhibitor OA (0.5 μm) eliminated the ability of PD168077 to inhibit GABAA currents. After washing off OA, the D4 modulation emerged. To further confirm the involvement of PP1/2A in D4 modulation of GABAA currents, we dialyzed cells with microcystin (5 μm), another structurally different and potent PP1/2A inhibitor. In most cells tested, the basal GABAA currents showed a time-dependent increase (9.5 ± 1.7%; n = 7) at the initial dialysis period (∼5 min), probably attributable to the inhibition of constitutively active PP1/2A by microcystin. After the basal GABAA currents became stabilized, subsequent application of PD168077 failed to reduce GABAAcurrents (Fig. 5C,D). On the contrary, injecting with OAE, a compound with a structure similar to OA but lacking the ability to inhibit PP1/2A, did not affect the PD168077-induced inhibition of GABAA currents (Fig. 5C,E). The effect of PD168077 on GABAA currents in the presence of phosphatase inhibitors or their inactive analog is summarized in Figure5F. PD168077 caused little reduction of GABAA currents in the presence of OA (4.8 ± 0.8%; n = 13; p > 0.05; Mann–WhitneyU test) or in the presence of microcystin (3.7 ± 0.5%; n = 9; p > 0.05; Mann–WhitneyU test), both of which were significantly different from the effect of PD168077 in the presence of OAE (15.4 ± 1.1%;n = 10; p < 0.005; ANOVA), suggesting the PP1 or PP2A activation is required in the D4regulation of GABAA receptors.
Fig. 5.
The effect of PD168077 (PD) on GABAA currents was blocked by phosphatase inhibition.A, Plot of peak GABAA currents as a function of time and drug application. In the presence of the membrane-permeable PP1/2A inhibitor OA, PD168077 (20 μm) failed to reduce GABAA currents. Washing off OA led to recovery of the effect of PD168077. B, Representative currenttraces taken from the records used to constructA (at time points denoted by #). C, Plot of peak GABAA currents as a function of time and drug application in neurons dialyzed with microcystin (triangle) or OAE (circles). The PP1/2A inhibitor microcystin (5 μm) but not the inactive agent OAE (1 μm) eliminated the PD168077-induced reduction of GABAA currents. D, E, Representative currenttraces taken from the records used to constructC (at time points denoted by #). F, Cumulative data (means ± SEM) showing the percentage modulation of GABAA currents by PD168077 in the absence (n = 11) or presence of OA (n = 13), OAE (n = 10), or microcystin (n = 9). *p < 0.005; ANOVA.ctl, Control.
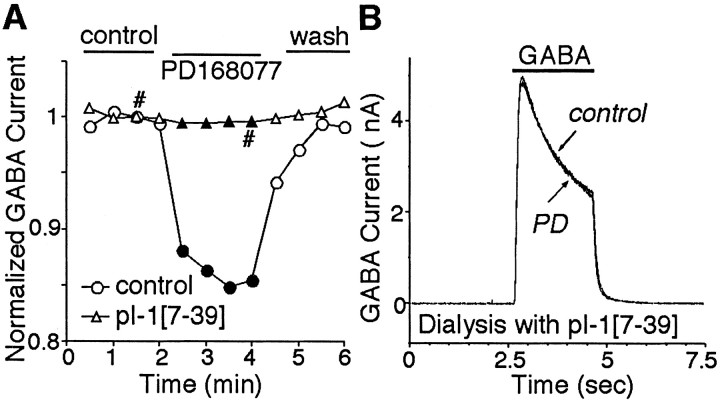
We then tried to determine the identity of the phosphatase involved in the D4 regulation of GABAAcurrents. I-1, once it is phosphorylated by PKA at Thr35, acts as a specific inhibitor of PP1 (Ingebritsen and Cohen, 1983). To test the role of PP1 in D4 modulation of GABAAcurrents, we dialyzed PFC pyramidal neurons with the phosphorylated I-1 peptide pThr35I-1[7–39], derived from the PP1 interaction region. Biochemical analysis demonstrated that the phospho-I-1 peptide pThr35I-1[7–39] potently inhibited PP1 catalytic activity with an IC50 at the nanomolar range, whereas the dephospho-I-1 peptide I-1[7–39], was much less effective (Hemmings et al., 1990; Kwon et al., 1997). As shown in Figure6A and B, dialysis with the active pThr35I-1[7–39] peptide (40 μm) but not the inactive control peptide I-1[7–39] (40 μm) abolished the ability of PD168077 to modulate GABAA currents. In summary, in cells dialyzed with the pThr35I-1[7–39] peptide, PD168077 reduced GABAA currents by 2.3 ± 0.8% (n = 18; p > 0.05; Mann–WhitneyU test), which was significantly smaller than the effect of PD168077 in cells dialyzed with the inactive I-1[7–39] control peptide (13.2 ± 2.5%; n = 7; p< 0.005; ANOVA). These results indicate direct involvement of PP1, which links D4 receptor activation to a reduction of GABAA currents.
Fig. 6.
The effect of PD168077 (PD) on GABAA currents was blocked by I-1 inhibition of PP1 activity. A, Plot of peak GABAA currents as a function of time and drug application in neurons dialyzed with the phosphorylated I-1 peptide pThr35I-1[7–39] or the dephosphorylated I-1[7–39] control peptide. The constitutively active peptide pThr35I-1[7–39] (40 μm) but not the inactive control peptide I-1[7–39] (40 μm) blocked PD168077 modulation of GABAAcurrents. B, Representative currenttraces taken from the records used to constructA (at time points denoted by #).
D4 modulation of GABAA currents requires the Yotiao-mediated anchoring of protein kinase A/ protein phosphatase 1 complex
Emerging evidence has shown that signaling enzymes with broad substrate selectivity, such as PKA and PP1, achieve the efficacy and specificity of signal transduction through anchoring protein-mediated subcellular targeting to their substrates in central neurons (Colledge and Scott, 1999). Previous studies have found that the multivalent scaffold protein Yotiao binds PP1 and PKA, allowing the two enzymes to regulate their substrates dynamically, like NMDA receptors (Westphal et al., 1999). We subsequently examined whether Yotiao is involved in D4 modulation of GABAA channels in PFC pyramidal neurons. If Yotiao is responsible for targeting the PKA/PP1 complex to GABAA receptors and allowing the kinase and phosphatase to regulate the phosphorylation state of these substrates effectively, then disrupting the complex should lead to the removal of PKA/PP1 from GABAA receptors, thereby attenuating the regulation of these channels.
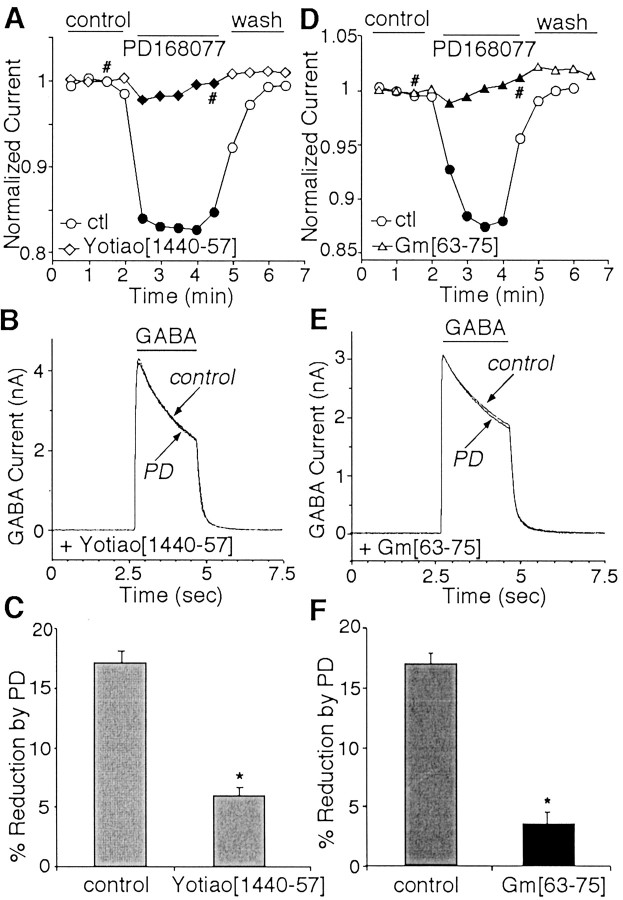
Previous biochemical studies have found that a peptide encompassing residues 1440–1457 of Yotiao blocked PKA binding in vitro(Westphal et al., 1999). Based on this result, we synthesized a peptide, Yotiao[1440–1457], dialyzed neurons with it, and then examined the effects of D4 on GABAA currents. As shown in Figure7A and B, dialysis with Yotiao[1440–1457] (10 μm) significantly attenuated the ability of PD168077 to modulate GABAA currents. In contrast, a control peptide with a scrambled amino acid sequence, sYotiao[1440–1457], had no effect on D4 regulation of GABAA currents. As summarized in Figure7C, in a sample of PFC neurons dialyzed with the peptide Yotiao[1440–1457], PD168077 reduced GABAAcurrents by 5.9 ± 0.6% (n = 12;p > 0.05; Mann–Whitney U test), which was significantly smaller than the effect of PD168077 in neurons dialyzed with the control peptide sYotiao[1440–1457] (17.2 ± 1.1%;n = 7; p < 0.005; ANOVA).
Fig. 7.
The D4 modulation of GABAAreceptor currents required anchoring of the PKA/PP1 complex to the channel by Yotiao. A, Plot of peak GABAAcurrent as a function of time and agonist application with the PKA anchoring inhibitory peptide Yotiao[1440–1457] (10 μm,diamonds) or the control (ctl) peptide sYotiao[1440–1457] (10 μm,circles) in the recording pipette. B, Representative current traces taken from the records used to construct A (at time points denoted by #).C, Cumulative data (means ± SEM) showing the percentage modulation of GABAA currents by PD168077 (PD; 20 μm) in the presence of Yotiao[1440–1457] peptide (n = 12) or the scrambled control peptide sYotiao[1440–1457] (n= 7). *p < 0.005; ANOVA. D, Plot of peak GABAA current as a function of time and agonist application with the PP1 anchoring inhibitory peptide Gm[63–75] (10 μm, triangles) or the control peptide sGm[63–75] (10 μm, circles) in the recording pipette. E, Representative currenttraces taken from the records used to constructD (at time points denoted by #). F, Cumulative data (means ± SEM) showing the percentage modulation of GABAA currents by PD168077 (20 μm) in the presence of Gm[63–75] peptide (n = 14) or the scrambled control peptide sGm[63–75] (n = 8). *p < 0.005; ANOVA.
Because the PD168077-induced reduction of GABAAcurrents requires the activation of PP1, we also examined whether blocking the binding of PP1 to Yotiao affected the D4 modulation of GABAAcurrents. Previous studies have found that the PP1 targeting inhibitor peptide Gm[63–75] (Egloff et al., 1997) could disrupt PP1 binding to Yotiao (Westphal et al., 1999), so we dialyzed PFC pyramidal neurons with the Gm[63–75] peptide. As shown in Figure 7D,E, dialysis with Gm[63–75] (10 μm) almost eliminated the ability of PD168077 to modulate GABAA currents. On the contrary, a control peptide with a scrambled amino acid sequence, sGm[63–75], had no effect on the D4 regulation of GABAA currents. As summarized in Figure7F, in a sample of PFC neurons dialyzed with the peptide Gm[63–75], PD168077 reduced GABAA currents by 4.3 ± 0.7% (n = 14; p > 0.05; Mann–Whitney U test), which was significantly smaller than the effect of PD168077 in neurons dialyzed with the control peptide sGm[63–75] (16.5 ± 1.2%; n = 8;p < 0.005; ANOVA). Collectively, these data suggest that D4 reduction of GABAAcurrents requires the Yotiao-mediated anchoring of the PKA/PP1 complex.
DISCUSSION
Despite the well recognized association of D4 receptors with schizophrenia, attention deficit hyperactivity disorder, and other mental disorders (Oak et al., 2000), the cellular mechanisms by which D4 receptors modulate PFC neuronal functions remain elusive. Anatomical studies have found that D4 receptors are enriched in PFC (Mrzljak et al., 1996; Ariano et al., 1997). Unlike D1 receptors that are concentrated at the dendritic spines of pyramidal neurons (Smiley et al., 1994), D4 receptors are localized predominantly on the periphery of the cell body and dendritic processes (Wedzony et al., 2000). Because GABAA receptors exhibit a compartmentalized distribution on postsynaptic domains of GABAergic synapses on the soma and proximal dendrites (Nusser et al., 1996), it suggests that most D4 receptors may be localized in the vicinity of GABAA receptors in PFC pyramidal neurons.
In this study, we demonstrated that activation of D4 receptors in PFC pyramidal neurons significantly reduced GABAA receptor-mediated currents, indicating that the postsynaptic GABAAreceptor is one of the key cellular substrates of D4 receptors in the PFC. Because GABAergic inhibition in the frontal cortex is critical for controlling the timing of neuronal activities during the thought process, which is fundamental for processing ongoing information and planning appropriate actions at a future time (Constantinidis et al., 2002), the D4 modulation of GABAergic signaling could be one of the mechanisms underlying the involvement of D4 receptors in PFC cognitive functions. It is conceivable that dysregulation of GABAergic inhibition by D4 receptors could contribute to the PFC cognitive deficits associated with schizophrenia. This notion is supported by the discovery that, in addition to elevated D4 receptors (Seeman et al., 1993), selective alterations in GABAA receptors, GABA content, and GABAergic local circuit neurons have been discovered in the PFC of patients with neuropsychiatric disorders (Benes et al., 1996; Dean et al., 1999; Ohnuma et al., 1999; Lewis, 2000). Although it has been suggested that D4 receptors function as an inhibitory modulator of glutamate activity in the PFC (Rubinstein et al., 2001), D4 receptors could exert both excitatory and inhibitory influences on the activity of neurons in the PFC by targeting different channels (Werner et al., 1996; Wilke et al., 1998), similar to the multifunctional feature of D1 receptors (for review, see Nicola et al., 2000).
The effect of dopamine on GABAergic synaptic transmission in the PFC is complex and dependent on the receptors activated. Recent studies have shown that dopamine produces temporally biphasic effects on GABAergic IPSCs, which are mediated by D1 and D2 receptors (Seamans et al., 2001). Changes in the excitability of GABAergic interneurons, the probability of release at GABAergic terminals, as well as the properties of postsynaptic GABAA receptors, have been suggested as the underlying mechanisms for the D1 and D2 modulation of GABAergic transmission (Seamans et al., 2001). The present study has revealed that D4 receptors also produce a significant reduction of the mIPSC amplitude in pyramidal neurons of PFC slices, suggestive of a D4 receptor-mediated downregulation of postsynaptic GABAA receptor sensitivity or conductance. This is consistent with the D4 receptor-mediated reduction of whole-cell GABAA receptor currents found in acutely dissociated PFC pyramidal neurons.
The mechanism underlying the D4 receptor-mediated reduction of GABAA receptor currents has been investigated in this study. Several lines of evidence show that the D4 receptor-mediated suppression of GABAA receptor currents is through a signaling pathway mediated by the inhibition of PKA and subsequent activation of PP1. The activity of PP1 is controlled by PKA through the regulatory protein I-1. I-1, after phosphorylation by PKA at Thr35, becomes a potent inhibitor of PP1 (Ingebritsen and Cohen, 1983). Previous studies have found that GABAA receptor currents are enhanced in response to elevated PKA activation in hippocampal dentate granule cells and neostriatal cholinergic interneurons (Kapur and Macdonald, 1996; Yan and Surmeier, 1997), presumably because of the increased PKA phosphorylation of GABAA receptor β3 subunits (McDonald et al., 1998). In the present study, tonic PKA activity, along with the inhibited PP1, may keep GABAAreceptors in PFC pyramidal neurons at a relatively high phosphorylation state, in which many of the β3 subunits are phosphorylated. The D4 receptor-mediated suppression of PKA activity, along with the disinhibited PP1, switches GABAAreceptors to a lower phosphorylation state, in which β3 subunits are dephosphorylated, therefore leading to the reduction of GABAA receptor currents.
Because both PKA and PP1 have broad substrate selectivity, a crucial issue in channel regulation is to control the specificity of their actions. Subcellular targeting through association with anchoring proteins has emerged as an important mechanism by which signaling enzymes achieve precise substrate recognition and enhanced efficacy of signal transduction (Rosenmund et al., 1994; Pawson and Scott, 1997;Yan et al., 1999; Feng et al., 2001). Yotiao, an NMDA receptor-associated protein (Lin et al., 1998), binds both PKA and PP1 (Feliciello et al., 1999; Westphal et al., 1999). This targeting protein is present in the cortex and is localized at somatodendritic regions (Lin et al., 1998). Recent studies have revealed the key role played by Yotiao in mediating the assembly of a macromolecular signaling complex and the dynamic regulation of NMDA receptor channels and the slow outward potassium channels (Westphal et al., 1999; Marx et al., 2002). To test whether the targeting of activated PKA/PP1 to GABAA receptors via Yotiao may allow these enzymes to regulate the phosphorylation state of GABAA receptors effectively in vivo, we examined the involvement of Yotiao in D4modulation of GABAA currents in PFC neurons. Dialysis with a Yotiao-derived peptide that can specifically disrupt the interaction between Yotiao and PKA (Westphal et al., 1999) significantly attenuated the D4 modulation of GABAA currents. The same was true when a peptide that can disrupt the interaction between Yotiao and PP1 was dialyzed. These data suggest that the D4 regulation of GABAA receptors requires the Yotiao-anchored pool of the PKA/PP1 complex, and that changing the subcellular targeting of these signaling enzymes leads to disruption of this regulation. It remains to be determined whether Yotiao is directly associated with GABAA receptor subunits or acts only to recruit the PKA/PP1 signaling complex to the proximity of GABAA receptors and thus facilitate the compartmentalized regulation of these substrates.
Together, our results show that the activation of D4 receptors decreased the postsynaptic GABAA receptor function in PFC pyramidal neurons via the regulation of the Yotiao-anchored PKA/PP1 signaling complex. Key signaling components engaged in D4 modulation of GABAA receptors provide the potential targets for novel pharmacological agents with greater therapeutic potential and fewer side effects in the treatment of neuropsychiatric disorders in which D4 receptors are highly involved.
Footnotes
This work was supported by National Institutes of Health Grant MH63128 (Z.Y.), National Science Foundation Grant IBN-0117026 (Z.Y.), and Howard Hughes Medical Institute Biomedical Research Support Program Grant 53000261 (State University of New York at Buffalo). We thank Dr. Jian Feng for critically reading this manuscript.
Correspondence should be addressed to Dr. Zhen Yan, Department of Physiology and Biophysics, State University of New York at Buffalo, 124 Sherman Hall, Buffalo, NY 14214. E-mail: zhenyan@buffalo.edu.
REFERENCES
- 1.Ariano MA, Wang J, Noblett KL, Larson ER, Sibley DR. Cellular distribution of the rat D4 dopamine receptor protein in the CNS using anti-receptor antisera. Brain Res. 1997;752:26–34. doi: 10.1016/s0006-8993(96)01422-9. [DOI] [PubMed] [Google Scholar]
- 2.Benes FM, Vincent SL, Marie A, Khan Y. Up-regulation of GABAA receptor binding on neurons of the prefrontal cortex in schizophrenic subjects. Neuroscience. 1996;75:1021–1031. doi: 10.1016/0306-4522(96)00328-4. [DOI] [PubMed] [Google Scholar]
- 3.Berger BS, Trottier C, Verney P, Gaspar P, Alvarez C. Regional and laminar distribution of the dopamine and serotonin innervation in the macaque cerebral cortex: a radioautographic study. J Comp Neurol. 1988;273:99–119. doi: 10.1002/cne.902730109. [DOI] [PubMed] [Google Scholar]
- 4.Brozoski TJ, Brown RM, Rosvold HE, Goldman PS. Cognitive deficit caused by regional depletion of dopamine in prefrontal cortex of rhesus monkey. Science. 1979;205:929–932. doi: 10.1126/science.112679. [DOI] [PubMed] [Google Scholar]
- 5.Cai X, Flores-Hernandez J, Feng J, Yan Z. Activity-dependent bi-directional regulation of GABAA receptor channels by serotonin 5-HT4 receptors in pyramidal neurons of the prefrontal cortex. J Physiol (Lond) 2002;540:743–759. doi: 10.1113/jphysiol.2001.013391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chio CL, Drong RF, Riley DT, Gill GS, Slightom JL, Huff RM. D4 dopamine receptor-mediated signaling events determined in transfected Chinese hamster ovary cells. J Biol Chem. 1994;269:11813–11819. [PubMed] [Google Scholar]
- 7.Colledge M, Scott JD. AKAPs: from structure to function. Trends Cell Biol. 1999;9:216–221. doi: 10.1016/s0962-8924(99)01558-5. [DOI] [PubMed] [Google Scholar]
- 8.Constantinidis C, Williams GV, Goldman-Rakic PS. A role for inhibition in shaping the temporal flow of information in prefrontal cortex. Nat Neurosci. 2002;5:175–180. doi: 10.1038/nn799. [DOI] [PubMed] [Google Scholar]
- 9.Dean B, Hussain T, Hayes W, Scarr E, Kitsoulis S, Hill C, Opeskin K, Copolov DL. Changes in serotonin2A and GABA(A) receptors in schizophrenia: studies on the human dorsolateral prefrontal cortex. J Neurochem. 1999;72:1593–1599. doi: 10.1046/j.1471-4159.1999.721593.x. [DOI] [PubMed] [Google Scholar]
- 10.Desimone R. Neuropsychology: is dopamine a missing link? Nature. 1995;376:549–550. doi: 10.1038/376549a0. [DOI] [PubMed] [Google Scholar]
- 11.Dulawa SC, Grandy DK, Low MJ, Paulus MP, Geyer MA. Dopamine D4 receptor-knock-out mice exhibit reduced exploration of novel stimuli. J Neurosci. 1999;19:9550–9556. doi: 10.1523/JNEUROSCI.19-21-09550.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Durstewitz D, Seamans JK, Sejnowski TJ. Dopamine-mediated stabilization of delay-period activity in a network model of prefrontal cortex. J Neurophysiol. 2000;83:1733–1750. doi: 10.1152/jn.2000.83.3.1733. [DOI] [PubMed] [Google Scholar]
- 13.Egloff MP, Johnson DF, Moorhead G, Cohen PT, Cohen P, Barford D. Structural basis for the recognition of regulatory subunits by the catalytic subunit of protein phosphatase 1. EMBO J. 1997;16:1876–1887. doi: 10.1093/emboj/16.8.1876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Feliciello A, Cardone L, Garbi C, Ginsberg MD, Varrone S, Rubin CS, Avvedimento EV, Gottesman ME. Yotiao protein, a ligand for the NMDA receptor, binds and targets cAMP-dependent protein kinase II(1). FEBS Lett. 1999;464:174–178. doi: 10.1016/s0014-5793(99)01585-9. [DOI] [PubMed] [Google Scholar]
- 15.Feng J, Cai X, Zhao JH, Yan Z. Serotonin receptors modulate GABAA receptor channels through activation of anchored protein kinase C in prefrontal cortical neurons. J Neurosci. 2001;21:6502–6511. doi: 10.1523/JNEUROSCI.21-17-06502.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Glase SA, Akunne HC, Georgic LM, Heffner TG, MacKenzie RG, Manley PJ, Pugsley TA, Wise LD. Substituted [(4-phenylpiperazinyl)-methyl]benzamides: selective dopamine D4 agonists. J Med Chem. 1997;40:1771–1772. doi: 10.1021/jm970021c. [DOI] [PubMed] [Google Scholar]
- 17.Goldman-Rakic PS. Cellular basis of working memory. Neuron. 1995;14:477–485. doi: 10.1016/0896-6273(95)90304-6. [DOI] [PubMed] [Google Scholar]
- 18.Hamill OP, Marty A, Neher E, Sakmann B, Sigworth FJ. Improved patch-clamp techniques for high resolution current recording from cells and cell-free membrane patches. Pflügers Arch. 1981;391:85–100. doi: 10.1007/BF00656997. [DOI] [PubMed] [Google Scholar]
- 19.Hemmings HC, Jr, Nairn AC, Elliott JI, Greengard P. Synthetic peptide analogs of DARPP-32 (Mr 32,000 dopamine- and cAMP-regulated phosphoprotein), an inhibitor of protein phosphatase-1: phosphorylation, dephosphorylation, and inhibitory activity. J Biol Chem. 1990;265:20369–20376. [PubMed] [Google Scholar]
- 20.Ingebritsen TS, Cohen P. Protein phosphatases: properties and role in cellular regulation. Science. 1983;221:331–338. doi: 10.1126/science.6306765. [DOI] [PubMed] [Google Scholar]
- 21.Jentsch JD, Redmond DE, Jr, Elsworth JD, Taylor JR, Youngren KD, Roth RH. Enduring cognitive deficits and cortical dopamine dysfunction in monkeys after long-term administration of phencyclidine. Science. 1997;277:953–955. doi: 10.1126/science.277.5328.953. [DOI] [PubMed] [Google Scholar]
- 22.Jentsch JD, Taylor JR, Redmond DE, Jr, Elsworth JD, Youngren KD, Roth RH. Dopamine D4 receptor antagonist reversal of subchronic phencyclidine-induced object retrieval/detour deficits in monkeys. Psychopharmacology. 1999;142:78–84. doi: 10.1007/s002130050865. [DOI] [PubMed] [Google Scholar]
- 23.Kapur J, Macdonald RL. Cyclic AMP-dependent protein kinase enhances hippocampal dentate granule cell GABAA receptor currents. J Neurophysiol. 1996;76:2626–2634. doi: 10.1152/jn.1996.76.4.2626. [DOI] [PubMed] [Google Scholar]
- 24.Kapur S, Remington G. Atypical antipsychotics: new directions and new challenges in the treatment of schizophrenia. Annu Rev Med. 2001;52:503–517. doi: 10.1146/annurev.med.52.1.503. [DOI] [PubMed] [Google Scholar]
- 25.Knighton DR, Zheng JH, Ten Eyck LF, Xuong NH, Taylor SS, Sowadski JM. Structure of a peptide inhibitor bound to the catalytic subunit of cyclic adenosine monophosphate-dependent protein kinase. Science. 1991;253:414–420. doi: 10.1126/science.1862343. [DOI] [PubMed] [Google Scholar]
- 26.Kulagowski JJ, Broughton HB, Curtis NR, Mawer IM, Ridgill MP, Baker R, Emms F, Freedman SB, Marwood R, Patel S, Patel S, Ragan CI, Leeson PD. 3-((4-(4-chlorophenyl)piperazin-1-yl)-methyl)-1H-pyrrolo-2,3-b-pyridine: an antagonist with high affinity and selectivity for the human dopamine D4 receptor. J Med Chem. 1996;39:1941–1942. doi: 10.1021/jm9600712. [DOI] [PubMed] [Google Scholar]
- 27.Kwon YG, Huang HB, Desdouits F, Girault JA, Greengard P, Nairn AC. Characterization of the interaction between DARPP-32 and protein phosphatase 1 (PP-1): DARPP-32 peptides antagonize the interaction of PP-1 with binding proteins. Proc Natl Acad Sci USA. 1997;94:3536–35341. doi: 10.1073/pnas.94.8.3536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Lewis DA. GABAergic local circuit neurons and prefrontal cortical dysfunction in schizophrenia. Brain Res Brain Res Rev. 2000;31:270–276. doi: 10.1016/s0165-0173(99)00042-9. [DOI] [PubMed] [Google Scholar]
- 29.Lewis DA, Campbell MJ, Foote SL, Morrison JH. The monoaminergic innervation of primate neocortex. Hum Neurobiol. 1986;5:181–186. [PubMed] [Google Scholar]
- 30.Lidow MS, Goldman-Rakic PS. A common action of clozapine, haloperidol, and remoxipride on D1- and D2-dopaminergic receptors in the primate cerebral cortex. Proc Natl Acad Sci USA. 1994;91:4353–4356. doi: 10.1073/pnas.91.10.4353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Lin JW, Wyszynski M, Madhavan R, Sealock R, Kim JU, Sheng M. Yotiao, a novel protein of neuromuscular junction and brain that interacts with specific splice variants of NMDA receptor subunit NR1. J Neurosci. 1998;18:2017–2027. doi: 10.1523/JNEUROSCI.18-06-02017.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Macdonald RL, Olsen RW. GABAA receptor channels. Annu Rev Neurosci. 1994;17:569–602. doi: 10.1146/annurev.ne.17.030194.003033. [DOI] [PubMed] [Google Scholar]
- 33.Marx SO, Kurokawa J, Reiken S, Motoike H, D'Armiento J, Marks AR, Kass RS. Requirement of a macromolecular signaling complex for beta adrenergic receptor modulation of the KCNQ1-KCNE1 potassium channel. Science. 2002;295:496–499. doi: 10.1126/science.1066843. [DOI] [PubMed] [Google Scholar]
- 34.McDonald BJ, Amato A, Connolly CN, Benke D, Moss SJ, Smart TG. Adjacent phosphorylation sites on GABAA receptor beta subunits determine regulation by cAMP-dependent protein kinase. Nat Neurosci. 1998;1:23–28. doi: 10.1038/223. [DOI] [PubMed] [Google Scholar]
- 35.Miller EK. The prefrontal cortex: complex neural properties for complex behavior. Neuron. 1999;22:15–17. doi: 10.1016/s0896-6273(00)80673-x. [DOI] [PubMed] [Google Scholar]
- 36.Moss SJ, Doherty CA, Huganir RL. Identification of the cAMP-dependent protein kinase and protein kinase C phosphorylation sites within the major intracellular domains of the beta 1, gamma 2S, and gamma 2L subunits of the gamma-aminobutyric acid type A receptor. J Biol Chem. 1992a;267:14470–14476. [PubMed] [Google Scholar]
- 37.Moss SJ, Smart TG, Blackstone CD, Huganir RL. Functional modulation of GABAA receptors by cAMP-dependent protein phosphorylation. Science. 1992b;257:661–665. doi: 10.1126/science.1323140. [DOI] [PubMed] [Google Scholar]
- 38.Mrzljak L, Bergson C, Pappy M, Huff R, Levenson R, Goldman-Rakic PS. Localization of dopamine D4 receptors in GABAergic neurons of the primate brain. Nature. 1996;381:245–248. doi: 10.1038/381245a0. [DOI] [PubMed] [Google Scholar]
- 39.Murphy BL, Arnsten AF, Goldman-Rakic PS, Roth RH. Increased dopamine turnover in the prefrontal cortex impairs spatial working memory performance in rats and monkeys. Proc Natl Acad Sci USA. 1996;93:1325–1329. doi: 10.1073/pnas.93.3.1325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Nicola SM, Surmeier J, Malenka RC. Dopaminergic modulation of neuronal excitability in the striatum and nucleus accumbens. Annu Rev Neurosci. 2000;23:185–215. doi: 10.1146/annurev.neuro.23.1.185. [DOI] [PubMed] [Google Scholar]
- 41.Nusser Z, Sieghart W, Benke D, Fritschy JM, Somogyi P. Differential synaptic localization of two major gamma-aminobutyric acid type A receptor alpha subunits on hippocampal pyramidal cells. Proc Natl Acad Sci USA. 1996;93:11939–11944. doi: 10.1073/pnas.93.21.11939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Oak JN, Oldenhof J, Van Tol HH. The dopamine D(4) receptor: one decade of research. Eur J Pharmacol. 2000;405:303–327. doi: 10.1016/s0014-2999(00)00562-8. [DOI] [PubMed] [Google Scholar]
- 43.Ohnuma T, Augood SJ, Arai H, McKenna PJ, Emson PC. Measurement of GABAergic parameters in the prefrontal cortex in schizophrenia: focus on GABA content, GABA(A) receptor alpha-1 subunit messenger RNA and human GABA transporter-1 (HGAT-1) messenger RNA expression. Neuroscience. 1999;93:441–448. doi: 10.1016/s0306-4522(99)00189-x. [DOI] [PubMed] [Google Scholar]
- 44.Patel S, Freedman S, Chapman KL, Emms F, Fletcher AE, Knowles M, Marwood R, Mcallister G, Myers J, Curtis N, Kulagowski JJ, Leeson PD, Ridgill M, Graham M, Matheson S, Rathbone D, Watt AP, Bristow LJ, Rupniak NM, Baskin E. Biological profile of L-745,870, a selective antagonist with high affinity for the dopamine D4 receptor. J Pharmacol Exp Ther. 1997;283:636–647. [PubMed] [Google Scholar]
- 45.Pawson T, Scott JD. Signaling through scaffold, anchoring, and adapter proteins. Science. 1997;278:2075–2080. doi: 10.1126/science.278.5346.2075. [DOI] [PubMed] [Google Scholar]
- 46.Porter NM, Twyman RE, Uhler MD, Macdonald RL. Cyclic AMP-dependent protein kinase decreases GABAA receptor current in mouse spinal neurons. Neuron. 1990;5:789–796. doi: 10.1016/0896-6273(90)90338-g. [DOI] [PubMed] [Google Scholar]
- 47.Rosenmund C, Carr DW, Bergeson SE, Nilaver G, Scott JD, Westbrook GL. Anchoring of protein kinase A is required for modulation of AMPA/kainate receptors on hippocampal neurons. Nature. 1994;368:853–856. doi: 10.1038/368853a0. [DOI] [PubMed] [Google Scholar]
- 48.Rubinstein M, Cepeda C, Hurst RS, Flores-Hernandez J, Ariano MA, Falzone TL, Kozell LB, Meshul CK, Bunzow JR, Low MJ, Levine MS, Grandy DK. Dopamine D4 receptor-deficient mice display cortical hyperexcitability. J Neurosci. 2001;21:3756–3763. doi: 10.1523/JNEUROSCI.21-11-03756.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Sawaguchi T, Goldman-Rakic PS. D1 dopamine receptors in prefrontal cortex: involvement in working memory. Science. 1991;251:947–950. doi: 10.1126/science.1825731. [DOI] [PubMed] [Google Scholar]
- 50.Seamans JK, Gorelova N, Durstewitz D, Yang CR. Bidirectional dopamine modulation of GABAergic inhibition in prefrontal cortical pyramidal neurons. J Neurosci. 2001;21:3628–3638. doi: 10.1523/JNEUROSCI.21-10-03628.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Seeman P, Van Tol HH. Dopamine receptor pharmacology. Trends Pharmacol Sci. 1994;15:264–270. doi: 10.1016/0165-6147(94)90323-9. [DOI] [PubMed] [Google Scholar]
- 52.Seeman P, Guan HC, Van Tol HH. Dopamine D4 receptors elevated in schizophrenia. Nature. 1993;365:441–445. doi: 10.1038/365441a0. [DOI] [PubMed] [Google Scholar]
- 53.Smiley JF, Levey AI, Ciliax BJ, Goldman-Rakic PS. D1 dopamine receptor immunoreactivity in human and monkey cerebral cortex: predominant and extrasynaptic localization in dendritic spines. Proc Natl Acad Sci USA. 1994;91:5720–5724. doi: 10.1073/pnas.91.12.5720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Van Tol HH, Bunzow JR, Guan HC, Sunahara RK, Seeman P, Niznik HB, Civelli O. Cloning of the gene for a human dopamine D4 receptor with high affinity for the antipsychotic clozapine. Nature. 1991;350:610–614. doi: 10.1038/350610a0. [DOI] [PubMed] [Google Scholar]
- 55.Wedzony K, Chocyk A, Mackowiak M, Fijal K, Czyrak A. Cortical localization of dopamine D4 receptors in the rat brain: immunocytochemical study. J Physiol Pharmacol. 2000;51:205–221. [PubMed] [Google Scholar]
- 56.Werner P, Hussy N, Buell G, Jones KA, North RA. D2, D3, and D4 dopamine receptors couple to G protein-regulated potassium channels in Xenopus oocytes. Mol Pharmacol. 1996;49:656–661. [PubMed] [Google Scholar]
- 57.Westphal RS, Tavalin SJ, Lin JW, Alto NM, Fraser ID, Langeberg LK, Sheng M, Scott JD. Regulation of NMDA receptors by an associated phosphatase-kinase signaling complex. Science. 1999;285:93–96. doi: 10.1126/science.285.5424.93. [DOI] [PubMed] [Google Scholar]
- 58.Wilke RA, Hsu SF, Jackson MB. Dopamine D4 receptor mediated inhibition of potassium current in neurohypophysial nerve terminals. J Pharmacol Exp Ther. 1998;284:542–548. [PubMed] [Google Scholar]
- 59.Williams GV, Goldman-Rakic PS. Modulation of memory fields by dopamine D1 receptors in prefrontal cortex. Nature. 1995;376:572–575. doi: 10.1038/376572a0. [DOI] [PubMed] [Google Scholar]
- 60.Yan Z, Surmeier DJ. Muscarinic (m2/m4) receptors reduce N- and P-type Ca2+ currents in rat neostriatal cholinergic interneurons through a fast, membrane-delimited, G-protein pathway. J Neurosci. 1996;16:2592–2604. doi: 10.1523/JNEUROSCI.16-08-02592.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Yan Z, Surmeier DJ. D5 dopamine receptors enhance Zn2+-sensitive GABA(A) currents in striatal cholinergic interneurons through a PKA/PP1 cascade. Neuron. 1997;19:1115–1126. doi: 10.1016/s0896-6273(00)80402-x. [DOI] [PubMed] [Google Scholar]
- 62.Yan Z, Hsieh-Wilson L, Feng J, Tomizawa K, Allen PB, Fienberg AA, Nairn AC, Greengard P. Protein phosphatase 1 modulation of neostriatal AMPA channels: regulation by DARPP-32 and spinophilin. Nat Neurosci. 1999;2:13–17. doi: 10.1038/4516. [DOI] [PubMed] [Google Scholar]