Abstract
Background
Glioblastoma (GBM) in children is rare. Pediatric GBM have a distinct molecular profile as compared to adult GBM. There are relatively few studies of pediatric GBMs and no standard of care on adjuvant therapy. We aimed to evaluate the clinical outcome and molecular profile of pediatric GBM.
Methods and Materials
Between 2004 and 2013, 66 consecutive children with histologically proven GBM were identified from our database. The majority of the children underwent maximal safe resection followed by focal radiotherapy with concurrent and adjuvant temozolomide. Immunohistochemical staining was performed for p53, MIB-1 labeling index, MGMT overexpression, and EGFR amplification and isocitrate dehydrogenase (IDH1) R132H point mutation. Survival and impact of possible prognostic factors on outcomes were analyzed.
Result
Median survival was 15 months. The overall survival rate at 1 year was 62%, at 2 years was 30%, and at 3 years was 27%. Patients with thalamic tumors (P < .001), incompletely resected tumors (P < .00001), and tumors with MIB-1 labeling index >25% (P < .002) had poor overall survival rates. p53 was overexpressed in 74% of patients, MGMT promoter methylation was seen in 37% of patients, IDH1 mutation was seen in 4% of patients, and no patients had EGFR amplification. MGMT methylation and p53 overexpression did not impact survival.
Conclusions
Clinical outcome of pediatric GBM is similar to that reported for adult GBM. The frequency of p53 overexpression is higher than in adult GBM, while MGMT methylation, IDH1 mutations and EGFR amplification is lower than in adult GBM. MGMT methylation and p53 expression status do not have any prognostic significance.
Keywords: clinical outcome, molecular characteristics, pediatric glioblastoma, temozolomide
Glioblastoma (GBM) is the most common malignant primary brain tumor in adult patients. In contrast, it accounts for <5% of childhood intracranial neoplasms and is relatively understudied both in terms of clinical outcomes and molecular characteristics.1–3 The emerging biologic, molecular, and genetic data suggest that pediatric high-grade gliomas are biologically distinct lesions from their adult counterparts.4 Despite therapeutic advances, outcomes have remained dismal, with 5-year survival outcomes ranging between 15% to 35%.5,6 Standard management for these patients includes maximal safe resection followed by postoperative focal irradiation plus additional chemotherapy. There is, however, no uniformity in the choice of the chemotherapy regimen. The introduction of temozolomide (TMZ) to treat adult GBM brought about a paradigm shift in treatment approach towards these highly aggressive tumors and several studies have now established TMZ as part of the standard of care for adult GBM.7,8 Given the rarity of pediatric GBM and the relative paucity of clinical data, treatment strategies have largely evolved from adult GBM, although some studies have failed to show benefit of TMZ in pediatric high-grade gliomas.6 This has led to a divided opinion among oncologists about its use in the postoperative adjuvant setting in the pediatric population. The present study attempts to evaluate outcomes and impact of various prognostic factors in pediatric GBM treated with postoperative radiotherapy and concurrent and adjuvant TMZ.
Materials and Methods
We searched our neuro-oncology database retrospectively for newly diagnosed pediatric GBM patients (3–18 years) seen at our institution between 2004 and 2013. Recurrent GBM, radiation-induced glioblastoma, or brainstem gliomas were excluded from analysis. Out of a total of 1440 GBM treated at our institution during this period, we identified 66 (4.5%) children diagnosed with GBM. We documented clinical data including their age, performance status, radiological characteristics, pathological findings, treatment modality, and survival. Paraffin-embedded tissues were available for 49 out of 66 patients and detailed molecular studies were performed wherever possible. A hospital institutional review board approval was taken for this retrospective audit.
Histopathological Examination and Molecular Studies
The hematoxylin and eosin slides were evaluated by an expert neuropathologist and the diagnosis was re-confirmed according to the 2007 WHO classification.2 Only unequivocal GBM were included in this study.
Monoclonal antibodies for p53 (clone D07, 1:50, Dako, Denmark), MIB-1 (1:50, Dako), and isocitrate dehydrogenase (IDH1) R132H point mutation (1:200, Dianova, Germany) were used. The monoclonal antibody H09 (anti-mIDH1R132H) recognizes the mutant-specific epitope of the IDH1R132H mutant protein. The MIB-1 labeling index (LI) was calculated in the highest proliferating area as percentage of labeled nuclei per 1000 cells. IDH1 mutation was evaluated as either positive or negative.
A dual-probe FISH assay was performed, using locus-specific probes for epidermal growth factor receptor (EGFR) paired with centromere probes for chromosome 7 (CEP7) (Vysis, Abbott molecular, Downers Grove, IL, USA). Signals were scored in at least 200 nonoverlapping and intact nuclei. Non-neoplastic cortical tissues obtained from epilepsy surgery specimens were used as positive controls. EGFR amplification was considered when more than 10% of tumor cells exhibited either an EGFR: CEP7 ratio >2 or innumerable tight clusters of signals.
DNA promoter methylation in the MGMT (O-6-methylguanine-DNA methyltransferase) gene was determined by methylation-specific polymerase chain reaction (MS-PCR). Genomic DNA was amplified by a two-step PCR approach to improve the sensitivity.9 Samples were labeled as ‘methylated’ if there was unequivocal band on methylated reaction, ‘unmethylated’ when there was complete absence or weak intensity band on unmethylated reaction, and ‘uninterpretable’ when no bands were seen either on methylated or on unmethylated reactions.
Treatment
Patients were managed by a multi-modality approach comprising maximal safe resection, radiotherapy, and TMZ (concurrent and adjuvant). Levels of resection were defined by postoperative imaging as well as by the surgeon's intraoperative impression. Sixty percent of our patients had a postoperative gadolinium-enhanced MRI scan and all had a postoperative CT scan. We considered >99% resection of the contrast-enhancing tumor as a gross total resection. Subtotal resection was defined as resection of anywhere between 50% and 99% of the enhancing tumor component.
External Beam Radiation Therapy
Patients received external beam radiation therapy (EBRT), using focal conformal techniques with conventional fractionation. Patients were scheduled to start EBRT within 4 weeks of surgery. Patients were immobilized with thermoplastics. Planning CT and MRI scans were taken and fused to define the target volumes and normal structures. For enhancing tumors, an equal matrix fast spoiled gradient echo (FSPGR) MR sequence with 0 mm gap was undertaken while T2-weighted or FLAIR MR sequences were undertaken for nonenhancing tumors. The gross tumor volume (GTV) included the tumor bed, residual tumor, or both, based on enhancing and nonenhancing tumor on the T1-weighted, postgadolinium images; T2-weighted images, and FLAIR MRI. The clinical target volume was defined by GTV, with a 2-cm anatomically constrained margin to encompass the peritumoral edema (microscopic disease) based on the FLAIR MR image. An additional expansion of 0.5 cm in 3 dimensions defined the planning target volume. The EBRT dose prescribed was 59.4 Gy in 33 fractions or 60 Gy in 30 fractions (median dose 59.4 Gy [range 32.4–60 Gy]).
Temozolomide Chemotherapy
Patients received concurrent radiotherapy and TMZ (RT+TMZ) at a dose of 75 mg/m2/day with prophylactic antiemetic and Pneumocystis carinii medication. Patients were monitored weekly. TMZ was withheld when the absolute neutrophil count was lower than 1.5 × 109/L, platelet count lower than 75000/mL, and CTCAE (Common Terminology Criteria for Adverse Events) nonhematological toxicity of grade III or greater.
Adjuvant TMZ was started 28 days after the completion of initial chemoradiotherapy. TMZ was administered at a dose of 150–200 mg/m2 on days 1 through 5, and cycles were repeated every 28 days. Children were monitored for hematological toxicity prior to each cycle. Adjuvant chemotherapy was planned for 6 cycles. Patients underwent a detailed neurological examination and recording of any toxicity profile at each visit during the adjuvant chemotherapy phase.
MRI of the brain was done 4 weeks after completion of initial RT+TMZ and then again after 6 cycles of adjuvant TMZ. Subsequent clinical follow-up was every 3 months for the first two years and then every 6 months. Radiological imaging was done every year or earlier if clinical progression was suspected. Asymptomatic patients showing post-treatment changes on surveillance MRI received further functional imaging (MR spectroscopy and perfusion). The imaging findings were discussed with experienced neuroradiologists at a joint neuro-oncology tumor board.
Statistical Analysis
Kaplan-Meier method was used to estimate the distribution of overall survival. Time was calculated from the date of diagnosis to the event of interest for overall survival and death due to any cause was considered an event of interest. A log-rank test was used to test for differences in the overall survival for dichotomous variables. Factors significant in the univariate analysis were tested by multivariate analysis using the Cox proportional hazard model. All statistical analyses were 2-sided, and the significance level was set at 5%. Statistical analyses were performed using SPSS version 21.0 (SPSS, Inc, USA).
Results
Patient Demographic and Treatment Profile (Table 1)
Table 1.
Patient characteristics and treatment
| Characteristics | Number of Patients | Percentage (%) |
|---|---|---|
| Sex | ||
| Male | 43 | 65 |
| Female | 23 | 35 |
| Age | ||
| Median | 12 years | |
| Range | 3–18 | |
| Location | ||
| Supratentorial | 52 | 79 |
| Frontal | 13 | 20 |
| Parietal | 10 | 15 |
| Temporal | 17 | 26 |
| Occipital | 4 | 6 |
| Thalamic | 8 | 12 |
| Infratentorial | 14 | 21 |
| Cerebellar | 10 | 15 |
| 4th ventricle | 4 | 6 |
| Extent of surgery | ||
| Gross total resection (GTR) | 29 | 44 |
| Supratentorial | 26 | |
| Infratentorial | 3 | |
| Sub-total resection (STR) | 28 | 42 |
| Supratentorial | 19 | |
| Infratentorial | 9 | |
| Biopsy | 9 | 14 |
| Supratentorial | 7 (Thalamic) | |
| Infratentorial | 2 | |
| Radiotherapy dose (Gy) | ||
| Median (Range) | – | 59.4 Gy (32.4–60)a |
| Radiotherapy technique | ||
| Two dimensional (2D) planning | 10 | 15% |
| Three dimensional conformal radiotherapy | 56 | 85% |
| Chemotherapy | ||
| Concurrentb | 60 | 90% |
| Adjuvant | 51 | 78% |
| Median adjuvant Temozolomide (TMZ) cycles | 6 cycles | – |
aTwo patients died before completing radiotherapy due to disease progression.
bTwo patients died during radiotherapy and 4 did not tolerate concurrent TMZ.
Median age at diagnosis was 12 years (range 3–18 years). Male: female ratio was 1.9:1. Tumor location was supratentorial in 52 patients, with temporal lobe being the most common. Infratentorial location was seen in 14 patients. The most common symptoms included headache (in 50% of patients), followed by seizures (36%) and sensory-motor deficits (15%). Patients with supratentorial tumors mainly presented with headache and seizures, while those with infratentorial lesions had headache as their predominant presenting complaint due to raised intracranial pressure. Patients with thalamic tumors presented with predominant sensory deficits. Fifty-seven patients (86%) received initial concurrent chemoradiotherapy and 51 patients (77%) received adjuvant TMZ. Of the 15 patients who did not start adjuvant TMZ, 12 died before adjuvant TMZ was started; 2 died during therapy due to disease progression; and 1 had nonrecovery of cell counts following chemoradiotherapy.
Immunohistochemical and Molecular Markers
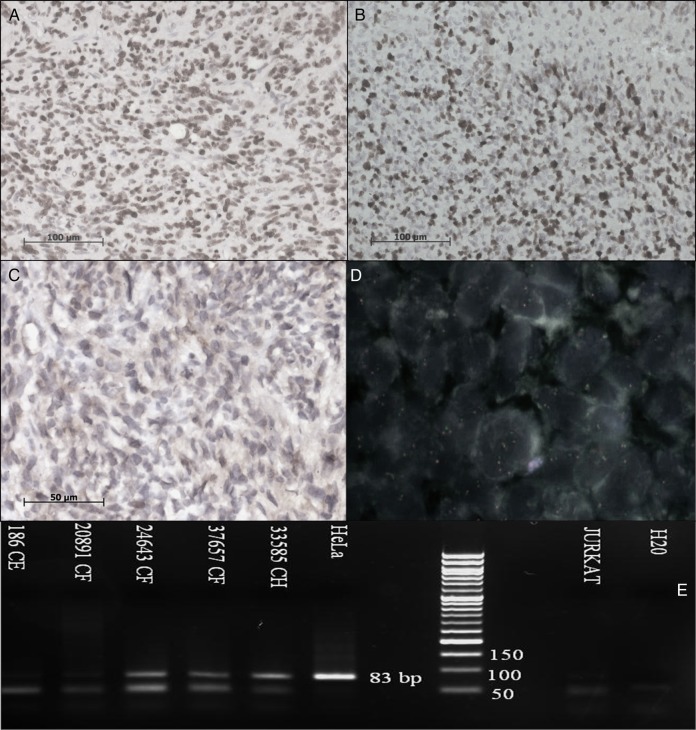
MGMT data were available for 30 of 66 patients (45%). Of the 30 patients, promoter methylation was detected in 11 patients (37%), while it was unmethylated in 11 patients (37%), and un-interpretable in 8 patients (28%). Immunohistochemistry for p53 protein expression was documented in 38 of 66 patients (57.5%). Of the 38 patients, p53 was over expressed in 28 patients (74%), negative in 7 patients (18%), and results were uninterpretable in 3 patients (8%) (Fig. 1A and E). IDH1 mutation and EGFR amplification data were available for 24 of 66 patients (36%). None of the children showed EGFR amplification, while 1 child (4%) showed focal positivity for IDH1 mutation (Fig. 1C and D).
Fig. 1.
Pictomicrograph of (A) Immunohistochemistry (IHC) for P53 showing strong nuclear immunoreactivity, (B) IHC showing high proliferation activity as demonstrated by immunoreactivity to MIB-1, (C) IHC of a representative tumor block showing focal positivity for isocitrate dehydrogenase-1 mutation (IDH-1R132H), (D) FISH showing normal EGFR/CEP7 expression, (E) Methylation specific PCR (MS-PCR) analyses of the MGMT promoter from representative samples (M-Methylated; UM-Unmethylated).
Treatment Outcomes
The median follow-up was 14 months (range: 1–73 months). Survival plots are illustrated in Figure 2. The median overall survival time of the entire cohort was 15 months (95% CI-11.5–18.5 months) and the actuarial survival rates at 1, 2, and 3 years were 62%, 30%, and 27%, respectively. Overall, 45 children (68%) have died, 20 (30%) are alive, and 1 child (2%) has been lost to follow-up at the cut-off date of analysis.
Fig. 2.
Kaplan-Meier estimates of overall survival (OS).
Univariate Survival Analysis
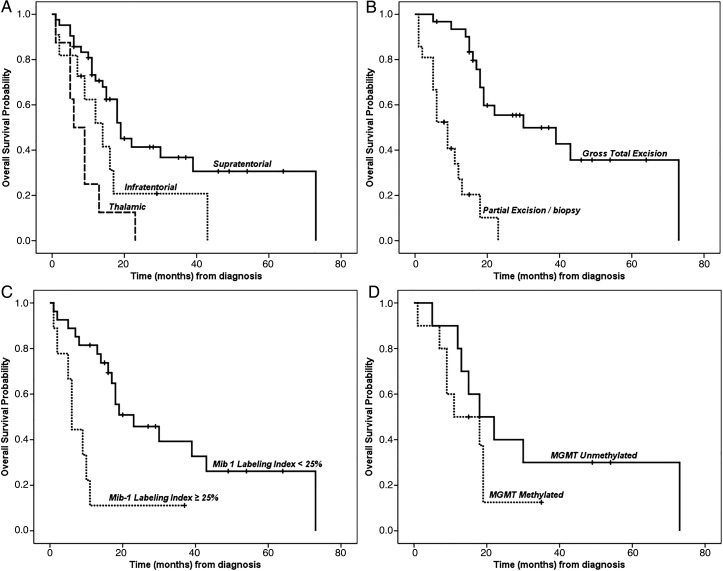
Potential parameters of prognostic significance were tested by univariate analysis. These included age, sex, seizures at presentation, extent of resection, location of tumor (supratentorial vs. infratentorial vs. thalamic), MIB-1 LI (< or > than 25%), MGMT promoter methylation, and p53 overexpression. Patients with supratentorial tumors had more favorable clinical outcomes (median survival: 19 months, 95% CI 13.4–24.6 months) than patients with infratentorial tumors (median survival 14 months; 95%CI 6.6–21.4 months) or thalamic tumors (median survival: 6 months; 95% CI 2.3–9.7 months) (Fig. 3A. P = .001). The actuarial 1-year and 2-year overall survival rates were 71% and 42%, 60% and 21%, and 13% and 0% for supratentorial, infratentorial, and thalamic tumors, respectively (Table 2).
Fig. 3.
Kaplan-Meier estimates comparing OS according to tumor location (A) and extent of resection (B), MIB-1 labeling index (C) and MGMT promoter methylation (D). The median OS was significantly associated with tumor location (P < 0.001), extent of resection (P < 0.00001), MIB-1 labeling index greater or <25% (P < 0.002). MGMT methylation did not affect OS (P = .25).
Table 2.
Univariate and multivariate analysis of overall survival (OS)
| Variables | Univariate Analysis |
aMultivariate Analysis |
||||||
|---|---|---|---|---|---|---|---|---|
| Patients | Median Survival (months) | 1-yr OS (%) | 2-yr OS (%) | P-value | HR | 95% CI | P-value | |
| Age | ||||||||
| >12 years | 32 | 15 | 67.8 | 30.5 | 0.3 | 0.8 | 0.5–1.5 | – |
| <12 years | 34 | 9 | 47.2 | 21 | 1 | |||
| Sex | ||||||||
| Male | 43 | 15 | 66.2 | 31.4 | 0.5 | 0.7 | 0.4–1.2 | – |
| Female | 23 | 16 | 53.8 | 24.6 | 1 | |||
| Location | ||||||||
| Supratentorial | 44 | 19 | 71 | 42 | 0.001 | 0.28 | 0.12–0.62 | 0.63 |
| Infratentorial | 14 | 14 | 60 | 21 | 0.50 | 0.19–1.32 | ||
| Thalamic | 8 | 6 | 13 | 0 | 1 | – | ||
| Extent of resection | ||||||||
| GTR | 29 | 30 | 90 | 56 | 0.00001 | 0.16 | 0.07–0.35 | 0.003 |
| STR/Biopsy | 37 | 9 | 27 | 0 | 1 | – | ||
| Mib1 LI | ||||||||
| <25% | 27 | 23 | 81 | 45.8 | 0.002 | 0.30 | 0.13–0.72 | 0.05 |
| >25% | 9 | 6 | 12 | 0 | 1 | – | ||
| MGMT promoter methylation | ||||||||
| Unmethylated | 11 | 18 | 80 | 40 | 0.2 | 0.6 | 0.20–1.60 | – |
| Methylated | 11 | 11 | 50 | 12.5 | 1 | |||
| P53 over expression | ||||||||
| No | 7 | 23 | 85 | 38 | 0.7 | 0.89 | 0.36–2.23 | – |
| Yes | 28 | 18 | 62 | 36.5 | 1 | – | ||
HR-Hazard Ratio; CI-Confidence Interval; GTR-Gross total resection; STR-Subtotal resection; Mib1 LI-Mib1 Labelling index. MGMT- O-6-methylguanine-DNA methyltransferase.
aFactors that were significant in the univariate analysis (tumor location, extent of surgery and Mib1 LI) were tested for significance by multivariate analysis. P-value ≤ 0.05 was taken as significant.
The median survival of patients who had complete resection was 30 months (95%CI 3.1–56.9 months), compared with children who had subtotal resection or biopsy whose median survival was 9 months (95% CI 5.6–12.4 months, P < .00001; Fig. 3B). The actuarial 1-year and 2-year overall survival rates were 90% and 56% for completely excised tumors. Overall survival at 1 year was only 27% for children who had subtotal resection (Table 2). Age, sex, and the presence or absence of seizures did not have any impact on overall survival.
The MIB-1 LI was an important determinant of survival in pediatric GBM; the median survival was 23 months (95% CI 7.8–38.2 months) for patients with MIB-1 LI ≤ 25% and 6 months (95% CI 4.5–7.5 months) for those with MIB-1 LI > 25% (P < .002, Fig. 3C). p53 overexpression and MGMT promoter methylation (Fig. 3D) did not have any impact on overall survival.
Multivariate Survival Analysis
Multivariate analysis showed that maximal safe resection had a significantly reduced hazard for death as compared with biopsy and subtotal resection (HR 0.16; 95% CI 0.07–0.35; P = .003). MIB-1 LI<25% reduced the hazard of death by 70% when compared with MIB-1 LI >25% (HR = 0.30; 95% CI = 0.13–0.72; P = .05). However, tumor location, which was statistically significant on univariate analysis, fell out of the model, although supratentorial tumors had a reduced hazard of death (HR = 0.28; 95% CI = 0.12–0.62; P = .6) when compared with infratentorial and thalamic tumors (Table 2).
Toxicity Profile
Patients experienced mild to moderate degrees of nausea and vomiting during both concurrent and adjuvant phases of TMZ. Hematological toxicities were seen in 12 patients (18.2%). The most common hematological toxicity was thrombocytopenia. During the concurrent phase, the most common nonhematologic toxicity was vomiting. Skin toxicity was common but not severe; the majority were self-limiting and did not require any active intervention except for topical steroids and emollients. Grade III and IV leucopenia was observed in 6 patients. Grade III thrombocytopenia was seen in 7 patients. No patient died due to TMZ toxicity.
Discussion
The incidence of GBM in children is low and studies evaluating clinical outcome in childhood GBM are scarce. The current study describes the molecular profile and clinical outcome of combined modality therapy (RT+ TMZ) in 66 children diagnosed with GBM. Given the central role of TMZ in adult GBM, we sought to evaluate the clinical outcome of children diagnosed with primary GBM treated with the above regimen.
Although pediatric GBM is considered to be histopathologically indistinguishable from the adult tumors, distinct molecular signatures strongly suggest a separate genetic pathway in their development.10 Given the rarity in children, the paucity of clinical data, and the molecular heterogeneity, there is still no accepted standard of care for postoperative adjuvant chemotherapy. Studies in pediatric patients utilizing a diverse group of biologic and chemotherapeutic agents have not yielded any significant benefit, and no chemotherapy regimen has yet been identified as standard adjuvant therapy for pediatric high-grade gliomas (2-year overall survival ranges from 10% to 30%), highlighting the need for new treatment approaches.11–13 Introduction of TMZ brought about a paradigm shift in the postoperative management of adult GBM. A seminal report from the EORTC/NCIC study demonstrated a 2-year overall survival of 26% for adult patients treated with RT+TMZ,14 which was replicated by many centers throughout the world.15 Based on the adult data, the Children's Oncology Group (COG) undertook a single-arm phase II study of chemoradiotherapy with TMZ followed by adjuvant TMZ in children with high-grade gliomas, but failed to show any improvement in survival.6,16 Our study showed a median survival of 15 months and a 2-year overall survival of 30%, comparable to the EORTC-NCIC study for adult GBM and the Children's Oncology Group study for pediatric high-grade gliomas.6,14 Proponents have advocated the use of TMZ in pediatric GBM due to the compelling adult data, while opponents cite the results of the COG study as lack of evidence for its use in the concomitant and adjuvant settings. Therefore, its utility in pediatric GBM will continue to be debated until robust data emerge from well-designed randomized trials.
The present study also identified supratentorial tumor locations as predictive of better outcomes than infratentorial and thalamic tumor locations. These results may be related to the extent of resection, as supratentorial tumors are more amenable to complete surgical removal, while patients with thalamic tumors generally undergo biopsy only.11
In adults, distinct molecular pathways have been described for the development of these tumors. However, the extent of genetic similarity between malignant gliomas in children and adults is still relatively poorly understood. Large-scale research efforts by the pediatric neuro-oncology community have been directed at studying the molecular biology underlying pediatric GBM, and have triggered an exciting search for clinically relevant molecular biomarkers and therapeutic targets. In the present study, we analyzed the prognostic role of molecular markers in terms of EGFR, p53, IDH1R132H, MGMT and MIB-1 LI. EGFR amplification plays an important role in the tumorigenesis and clinical outcome of GBM.17,18 In adult GBM, various therapeutic strategies including antibody and ligand-mediated therapeutic agents such as immunotoxins and radioimmuno conjugates have been developed to block the EGFR tyrosine kinase and diminish downstream signaling from this receptor. Pediatric GBM is rarely associated with EGFR amplification, whereas it is seen in 35% to 50% of adult GBM patients.19,20 EGFR amplification was not noted in patients in this study, which corroborates with other studies. However, the high degree of EGFR immunopositivity with increasing density in higher grades led to clinical trials with nimotuzumab and cetuximab with variable results.19,21 Antivascular endothelial growth factor therapies have also shown modest outcome in pediatric malignant gliomas.22
Pediatric GBM often demonstrate p53 mutations, similar to secondary adult GBM that evolve from low-grade gliomas.23,24 Overexpression of p53 protein was seen in 74% of children in our study, which is higher than in primary adult GBM, but comparable to secondary adult GBM (Table 3). Our observations are corroborated by Cheng et al and Suri et al, who documented p53 protein accumulation in 75% and 63% of childhood high-grade gliomas.19,24 The CCG-945 study assessed the mutational status of TP53 (in 121 specimens) and performed immunohistochemical analysis of p53 (in 115 specimens) and documented TP53 mutations in 33% and p53 overexpression in 35% of high-grade gliomas in children, which increased to 58% when only GBMs were considered.25 Mutations and alterations in this pathway have been shown to be prognostically relevant in numerous studies.25,26 In the CCG-945 study, the observed rate of progression-free survival at 5 years was 44% in the group of patients whose tumors had low levels of expression of p53 and only 17% in the group of patients whose tumors had overexpression of p53 (P < .001).25 Moreover, the same study showed that overexpression of the p53 protein increases with tumor grade: one fourth of analyzed anaplastic astrocytomas (AA) and half of GBMs overexpressed this protein. In our study, patients with p53 overexpression had a shorter median overall survival (18 months) than those without p53 overexpression (23 months), although this was not statistically significant (P = .7).
Table 3.
Summary of immunohistochemistry (IHC), fluorescence in situ hybridization (FISH) and methylation specific polymerase chain reaction results (MS-PCR) and comparing with adult glioblastoma (primary and secondary)
| Characteristic | Pediatric GBM (present study) | Adult Primary GBM | Adult Secondary GBM |
|---|---|---|---|
| Mean Age [3,4] | 12 years | 62 years | 45 years |
| Median Survival [7,8,24] | 15 months | 14 months | 17 months |
| P53 mutation [23,24] | 74% | 25%–30% | 60%–65% |
| MGMT methylation [31–34] | 37% | 36% | 75% |
| EGFR amplification [17–20] | 0% | 35%–50% | 8% |
| IDH-1 mutation [28–30] | 4% | 10% | 85% |
GBM, Glioblastoma; IDH, Isocitrate dehydrogenase; EGFR, Epidermal growth Factor receptor.
Children with high proliferation index defined as MIB-1 LI > 25% had significantly shorter survival times than those with MIB-1 LI < 25%. Pollack et al reported the impact of proliferation index in a cohort of childhood malignant gliomas accrued in the CCG-945 study17 and observed that MIB-1 LI had a strong impact on progression free survival in GBM (P = .001). The authors reported that tumors with labeling indices >36% conferred a uniformly poor clinical outcome regardless of histology. This is somewhat in contrast to adult GBM where MIB-1 LI has failed to predict overall survival.27
IDH1 mutation is considered to be a strong predictor of favorable outcome in GBM. It is frequently mutated (> 80%) secondary adult GBM28 (Table 3). However, the frequency of IDH1 mutation is rare in pediatric high-grade glioma29 IDH1 mutations are also rare in adult primary GBM (10%). We observed a very low rate of IDH 1 mutations (4%) in our cohort of patients, a fact corroborated by others.30
Over the past decade MGMT promoter methylation has gained profound importance especially in newly diagnosed adult GBM, with a reported methylation frequency ranging from 35% to 45%.31 The MGMT gene encodes a DNA-repair enzyme and its overexpression in tumor tissue can reduce the efficacy of alkylating agents. Promoter hypermethylation of the MGMT gene has been associated with prolonged survival in GBM patients receiving alkylating agents.31–33 Reported literature on MGMT promoter methylation status in childhood GBM has shown similar or lower methylation rates as compared with adult GBM.32–34 In our cohort we observed a methylation frequency of 37%. Although the prognostic significance of MGMT promoter methylation is well established in adult GBM, its significance in childhood high-grade glioma still remains controversial.14,31,35 The CCG-945 study demonstrated a progression-free survival of 42% at 5 years in the 97 patients whose tumors had low levels of MGMT expression vs 8.3% in the 12 patients whose tumors overexpressed MGMT(P = .017).32 Contrary to the studies that affirmed the favorable prognosis associated with methylated tumors, we observed a negative correlation between MGMT promoter methylation and overall survival, although this was not significant (P = .25). One possible reason could be extent of excision acting as a confounding factor. Coincidentally, more patients with unmethylated tumors underwent gross total resection than those with methylated tumors. However, the number of patients in our series was too small to draw any conclusions on its prognostic relevance. Further studies with larger sample sizes and proper follow-up data are needed to confirm its prognostic significance in pediatric GBM.
Although this study highlights the distinct molecular profile of GBM in children and the outcome of patients treated with multimodality approach, the inability to encompass other relevant markers that have gained interest in pediatric GBM viz. H3.3, α-thalassaemia/mental retardation syndrome X-linked (ATRX), death-domain associated protein (DAXX), mismatch repair gene (MLH1,MSH2 and PMS2), ADAM3A, PDGFRA (4q12), and BRAFV600E mutations, form a major limitation of this study.36–39 Recent studies have shown that genes involved in the H3.3-ATRX-DAXX chromatin remodeling pathway are mutated and are exclusively found in pediatric GBM and absent in adult GBM. The H3 histone, family 3A (H3F3A) gene, which encodes the replication-independent histone 3 variant H3.3 is mutated in 30% of high-grade gliomas that are not brainstem tumors and 60% of diffuse infiltrative pontine gliomas. The mutations are usually of a missense type found in 1 or 2 codons encoding lysine 27 (K27) or glycine 34(G34). H3F3A K27 mutation appears to be exclusive to pediatric high-grade gliomas and suggests shorter survival.40 ATRX dimerizes with DAXX and forms a transcription/chromatin remodeling complex required for the incorporation of H3.3 at telomeres and pericentric heterochromatin.36 Tumors with these mutations are associated with alternative lengthening of telomeres.36,41 The observation that a majority of pediatric GBM develops through alterations of the H3F3A/ATRX/DAXX chromatin-remodeling pathway, distinguishes childhood GBM from their adult counterparts, and confirms that these tumors may have a very different biology and should be considered as a separate entity.
Recent evolution in high-throughput technology has led to a discovery of a multitude of molecular and genetic markers, but their actual relevance, especially in an era of targeted therapy, is still a matter of debate.
The clinical outcome in our cohort of children was similar to published literature of adult GBM treated with standard chemoradiotherapy. We expect that future studies utilizing targeted therapy in conjunction with conventional RT and TMZ may increase the probability of long-term survival. In such a scenario there is increasing concern about the impact of treatment on quality of life and long-term neurocognitive effects.42,43 As it was a retrospective study, the inherent difficulty in assessing the quality of life in children limited our ability to study the neurocognitivecognitive effects of treatment.
GBM in children continues to carrt a very poor prognosis even with a multimodal approach. The observed frequency of p53 expression is higher in children than adults. The frequency of MGMT methylation, IDH1 mutation, and EGFR amplification is lower than in adult GBM. This study not only highlighted the distinct molecular pathology of pediatric GBM along with the prognostic importance of clinically relevant markers, but also highlighted the similar clinical outcome of children with GBM treated uniformly with multimodality therapy when compared with adults. Our future studies will be designed with an aim at investigating an array of molecular markers to better understand the genetic alterations that could help in developing newer and efficacious treatment strategies.
Acknowledgments
We thank the Brain Tumour Foundation of India for providing us the financial assistance for conducting the molecular marker research component of the study.
Conflicts of interest statement. None of the authors have any conflict of interest to declare or any financial disclosure.
References
- 1. Pollack IF. Brain tumors in children. N Engl. J. Med. 1994;331(22):1500–1507. [DOI] [PubMed] [Google Scholar]
- 2. Louis DN, Ohgaki H, Wiestler OD et al. The 2007 WHO classification of tumours of the central nervous system. Acta Neuropathol. 2007;114(2):97–109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Arora RS, Alston RD, Eden TO et al. Age-incidence patterns of primary CNS tumors in children, adolescents, and adults in England. Neuro Oncol. 2009;11(4):403–413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Jones C, Perryman L, Hargrave D. Paediatric and adult malignant glioma: close relatives or distant cousins? Nature Reviews. Clinical Oncology. 2012;9(7):400–413. [DOI] [PubMed] [Google Scholar]
- 5. Finlay JL, Zacharoulis S. The treatment of high grade gliomas and diffuse intrinsic pontine tumors of childhood and adolescence: a historical - and futuristic - perspective. J. Neurooncol. 2005;75(3):253–266. [DOI] [PubMed] [Google Scholar]
- 6. Cohen KJ, Pollack IF, Zhou T et al. Temozolomide in the treatment of high-grade gliomas in children: a report from the Children's Oncology Group. Neuro Oncol. 2011;13(3):317–323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Athanassiou H, Synodinou M, Maragoudakis E et al. Randomized phase II study of temozolomide and radiotherapy compared with radiotherapy alone in newly diagnosed glioblastoma multiforme. J Clin Oncol. 2005;23(10):2372–2377. [DOI] [PubMed] [Google Scholar]
- 8. Stupp R, Hegi ME, Mason WP et al. Effects of radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy alone on survival in glioblastoma in a randomised phase III study: 5-year analysis of the EORTC-NCIC trial. Lancet Oncol. 2009;10(5):459–466. [DOI] [PubMed] [Google Scholar]
- 9. McLendon RE, Cleveland L, Pegram C et al. Immunohistochemical detection of the DNA repair enzyme O6-methylguanine-DNA methyltransferase in formalin-fixed, paraffin-embedded astrocytomas. Lab Invest. 1998;78(5):643–644. [PubMed] [Google Scholar]
- 10. Ichimura K, Nishikawa R, Matsutani M. Molecular markers in pediatric neuro-oncology. Neuro Oncol. 2012;14(Suppl 4):iv90–iv99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Finlay JL, Boyett JM, Yates AJ et al. Randomized phase III trial in childhood high-grade astrocytoma comparing vincristine, lomustine, and prednisone with the eight-drugs-in-1-day regimen. Childrens Cancer Group. J Clin Oncol. 1995;13(1):112–123. [DOI] [PubMed] [Google Scholar]
- 12. Broniscer A, Chintagumpala M, Fouladi M et al. Temozolomide after radiotherapy for newly diagnosed high-grade glioma and unfavorable low-grade glioma in children. J. Neurooncol. 2006;76(3):313–319. [DOI] [PubMed] [Google Scholar]
- 13. Narayana A, Kunnakkat S, Chacko-Mathew J et al. Bevacizumab in recurrent high-grade pediatric gliomas. Neuro Oncol. 2010;12(9):985–990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Stupp R, Mason WP, van den Bent MJ et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl. J. Med. 2005;352(10):987–996. [DOI] [PubMed] [Google Scholar]
- 15. Jalali R, Basu A, Gupta T et al. Encouraging experience of concomitant Temozolomide with radiotherapy followed by adjuvant Temozolomide in newly diagnosed glioblastoma multiforme: single institution experience. Br. J. Neurosurg. 2007;21(6):583–587. [DOI] [PubMed] [Google Scholar]
- 16. Geyer JR, Finlay JL, Boyett JM et al. Survival of infants with malignant astrocytomas. A Report from the Childrens Cancer Group. Cancer. 1995;75(4):1045–1050. [DOI] [PubMed] [Google Scholar]
- 17. Pollack IF, Hamilton RL, James CD et al. Rarity of PTEN deletions and EGFR amplification in malignant gliomas of childhood: results from the Children's Cancer Group 945 cohort. J. Neurosurg. 2006;105(5 Suppl):418–424. [DOI] [PubMed] [Google Scholar]
- 18. Zhu H, Acquaviva J, Ramachandran P et al. Oncogenic EGFR signaling cooperates with loss of tumor suppressor gene functions in gliomagenesis. Proc Natl Acad Sci USA. 2009;106(8):2712–2716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Cheng Y, Ng HK, Zhang SF et al. Genetic alterations in pediatric high-grade astrocytomas. Hum. Pathol. 1999;30(11):1284–1290. [DOI] [PubMed] [Google Scholar]
- 20. Sung T, Miller DC, Hayes RL et al. Preferential inactivation of the p53 tumor suppressor pathway and lack of EGFR amplification distinguish de novo high grade pediatric astrocytomas from de novo adult astrocytomas. Brain Pathol. 2000;10(2):249–259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Solomon MT, Miranda N, Jorrin E et al. Nimotuzumab in combination with radiotherapy in high grade glioma patients: a single institution experience. Cancer Biology and Therapy. 2014;15(5):504–509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Gururangan S, Chi SN, Young Poussaint T et al. Lack of efficacy of bevacizumab plus irinotecan in children with recurrent malignant glioma and diffuse brainstem glioma: a Pediatric Brain Tumor Consortium study. J Clin Oncol. 2010;28(18):3069–3075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Parsons DW, Jones S, Zhang X et al. An integrated genomic analysis of human glioblastoma multiforme. Science. 2008;321(5897):1807–1812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Suri V, Das P, Pathak P et al. Pediatric glioblastomas: a histopathological and molecular genetic study. Neuro Oncol. 2009;11(3):274–280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Pollack IF, Finkelstein SD, Woods J et al. Expression of p53 and prognosis in children with malignant gliomas. N Engl. J. Med. 2002;346(6):420–427. [DOI] [PubMed] [Google Scholar]
- 26. Rood BR, MacDonald TJ. Pediatric high-grade glioma: molecular genetic clues for innovative therapeutic approaches. J. Neurooncol. 2005;75(3):267–272. [DOI] [PubMed] [Google Scholar]
- 27. Moskowitz SI, Jin T, Prayson RA. Role of MIB1 in predicting survival in patients with glioblastomas. J. Neurooncol. 2006;76(2):193–200. [DOI] [PubMed] [Google Scholar]
- 28. Nobusawa S, Watanabe T, Kleihues P et al. IDH1 mutations as molecular signature and predictive factor of secondary glioblastomas. Clin Cancer Res. 2009;15(19):6002–6007. [DOI] [PubMed] [Google Scholar]
- 29. Paugh BS, Qu C, Jones C et al. Integrated molecular genetic profiling of pediatric high-grade gliomas reveals key differences with the adult disease. J Clin Oncol. 2010;28(18):3061–3068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Antonelli M, Buttarelli FR, Arcella A et al. Prognostic significance of histological grading, p53 status, YKL-40 expression, and IDH1 mutations in pediatric high-grade gliomas. J. Neurooncol. 2010;99(2):209–215. [DOI] [PubMed] [Google Scholar]
- 31. Hegi ME, Diserens AC, Gorlia T et al. MGMT gene silencing and benefit from temozolomide in glioblastoma. N Engl. J. Med. 2005;352(10):997–1003. [DOI] [PubMed] [Google Scholar]
- 32. Pollack IF, Hamilton RL, Sobol RW et al. O6-methylguanine-DNA methyltransferase expression strongly correlates with outcome in childhood malignant gliomas: results from the CCG-945 Cohort. J Clin Oncol. 2006;24(21):3431–3437. [DOI] [PubMed] [Google Scholar]
- 33. Schlosser S, Wagner S, Muhlisch J et al. MGMT as a potential stratification marker in relapsed high-grade glioma of children: the HIT-GBM experience. Pediatric Blood and Cancer. 2010;54(2):228–237. [DOI] [PubMed] [Google Scholar]
- 34. Srivastava A, Jain A, Jha P et al. MGMT gene promoter methylation in pediatric glioblastomas. Child's Nervous System: ChNS: Official Journal of the International Society for Pediatric Neurosurgery. 2010;26(11):1613–1618. [DOI] [PubMed] [Google Scholar]
- 35. Donson AM, Addo-Yobo SO, Handler MH et al. MGMT promoter methylation correlates with survival benefit and sensitivity to temozolomide in pediatric glioblastoma. Pediatric Blood and Cancer. 2007;48(4):403–407. [DOI] [PubMed] [Google Scholar]
- 36. Schwartzentruber J, Korshunov A, Liu XY et al. Driver mutations in histone H3.3 and chromatin remodelling genes in paediatric glioblastoma. Nature. 2012;482(7384):226–231. [DOI] [PubMed] [Google Scholar]
- 37. Barrow J, Adamowicz-Brice M, Cartmill M et al. Homozygous loss of ADAM3A revealed by genome-wide analysis of pediatric high-grade glioma and diffuse intrinsic pontine gliomas. Neuro Oncol. 2011;13(2):212–222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Puget S, Philippe C, Bax DA et al. Mesenchymal transition and PDGFRA amplification/mutation are key distinct oncogenic events in pediatric diffuse intrinsic pontine gliomas. PloS One. 2012;7(2):e30313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Schindler G, Capper D, Meyer J et al. Analysis of BRAF V600E mutation in 1,320 nervous system tumors reveals high mutation frequencies in pleomorphic xanthoastrocytoma, ganglioglioma and extra-cerebellar pilocytic astrocytoma. Acta Neuropathol. 2011;121(3):397–405. [DOI] [PubMed] [Google Scholar]
- 40. Sturm D, Witt H, Hovestadt V et al. Hotspot mutations in H3F3A and IDH1 define distinct epigenetic and biological subgroups of glioblastoma. Cancer Cell. 2012;22(4):425–437. [DOI] [PubMed] [Google Scholar]
- 41. Heaphy CM, de Wilde RF, Jiao Y et al. Altered telomeres in tumors with ATRX and DAXX mutations. Science. 2011;333(6041):425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Vern-Gross TZ, Schreiber JE, Broniscer A et al. Prospective evaluation of local control and late effects of conformal radiation therapy in children, adolescents, and young adults with high-grade glioma. Neuro Oncol. 2014;1612:1652–1660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Sands SA, Zhou T, O'Neil SH et al. Long-term follow-up of children treated for high-grade gliomas: children's oncology group L991 final study report. J Clin Oncol. 2012;30(9):943–949. [DOI] [PMC free article] [PubMed] [Google Scholar]