Abstract
The basic leucine zipper (bZIP) transcription factor BATF is expressed in multiple Th subsets and cooperates with other factors to regulate gene transcription. BATF activates lineage-specific cytokines in Th subsets, activating IL-9 in Th9 cells and IL-17 in Th17 cells, but not IL-9 or IL-17 in the reciprocal subset. The mechanism for this restricted activity is unclear. In this report we define BATF binding partners that contribute to Th subset-specific functions. Although BATF and IRF4 are expressed in greater amounts in Th9 than Th17, increased expression of both factors is not sufficient to induce IL-9 in Th17 cells. BATF also requires heterodimer formation with Jun family members to bind DNA and induce gene expression. Using primary mouse T cell culture, we observed that JunB and c-Jun, but not JunD, promote IL-9 production in Th9 cells. Ectopic expression of BATF with either JunB or c-Jun generates modest but significant increases in IL-9 production in Th17 cells, suggesting that the low expression of Jun family members is one factor limiting the ability of BATF to induce IL-9 in Th17 cells. We further identified that Bach2 positively regulates IL-9 production by directly binding to the Il9 gene and by increasing transcription factor expression in Th9 cells. Strikingly, co-transduction of Bach2 and BATF significantly induces IL-9 production in both Th9 and Th17 cells. Taken together, our results reveal that JunB, c-Jun and Bach2 cooperate with BATF to contribute to the specificity of BATF-dependent cytokine induction in Th subsets.
Introduction
In response to the signals from antigen presenting cells, naive CD4+ T cells differentiate into distinct T helper subsets, including Th1, Th2, Th17, Treg and Th9 cells (1). T helper cells are critical in the adaptive immune system by producing multiple cytokines and chemokines. In many subsets a “master transcription factor” has been identified that defines the Th lineages, such as T-bet (T-box transcription factor) in Th1 cells, or Foxp3 (forkhead box P3) in Tregs (2). However, emerging data shows that the concept of master transcription factor is not sufficient to define the complexity of the Th cell phenotypes. The transcription factors shared by multiple Th subsets also play a critical role in Th cell differentiation. To further understand the underlying mechanism of Th cell differentiation, it is necessary to define how the transcription factors expressed in multiple lineages lead to the development of specific T helper cells and the interplay among those transcription factors in different Th subsets.
Interleukin-9 (IL-9) is the major cytokine produced by Th9 cells, a recently defined Th subset (3). IL-9 and IL-9-producing cells have been linked to autoimmunity, allergic disease, asthma, the immunity to parasites, and antitumor immunity (4, 5). The development of Th9 cells requires IL-4, TGFβand IL-2. Each of these cytokines activates specific signaling pathways and downstream transcription factors to promote IL-9 expression (6). The development of Th9 cells requires a balance of signaling pathways and a network of transcription factors including STAT6, BATF, IRF4, Smad proteins, PU.1 and STAT5 (7–12).
Basic leucine zipper transcription factor ATF-like (BATF) belongs to activator protein 1 (AP-1) transcription factor family that has a basic DNA-binding region and regularly spaced leucine residues termed a leucine zipper (bZIP). BATF requires heterodimer formation with other transcription factors such as Jun family members to regulate target gene expression, possibly because it lacks a transactivation domain (13). BATF is required for the differentiation of multiple Th subsets. In Th9 cells, BATF cooperates with Interferon regulatory factor 4 (IRF4) to promote IL-9 production in Th9 cells (11, 12). Moreover, BATF and IRF4 cooperate in the induction of IL-17 production in Th17 cells (14, 15). However, the mechanism through which BATF selectively regulates the lineage specific cytokine production in various Th subsets is still not well defined.
Since BATF plays an essential role in driving the differentiation of Th9 and Th17 subsets, we questioned how BATF selectively promoted IL-9 production in Th9 cells, but not in Th17 cells. We found that there is preferential expression of BATF and its binding partners between Th9 and Th17 cells. The low expression of JunB and c-Jun in Th17 cells limits the capacity of BATF to induce IL-9 in Th17 cells. Bach2 (broad complex-tramtrack-bric a brac and Cap’n’collar homology 2) also regulates the activity of AP-1 transcription factors by competing for binding sites on target genes (16, 17). Surprisingly, we find that Bach2 amplified the function of BATF by binding to the Il9 locus and by regulating the expression of transcription factors that induce IL-9 expression. Thus, our study suggests that one factor dictating the specificity of BATF in Th subsets is the availability of BATF binding partners.
Material and Methods
Mice
C57BL/6 mice were purchased from The Jackson Laboratory. BatfΔZ/ΔZ (C57BL/6), Irf4fl/fl mice and Bach2fl/fl mice were previously described (11, 18, 19). All experiments were performed with the approval of the Indiana University Institutional Animal Care and Use Committee.
In vitro mouse T cell differentiation
Naive CD4+CD62L+ T cells were isolated from the spleens and lymph nodes of the mice by using the magnetic separation following the supplier’s protocol (Miltenyi Biotec, Auburn, CA). Cells were cultured in complete RPMI 1640 on plates coated with anti-CD3 and soluble anti-CD28. Cell cultured under Th9 condition were supplemented with human TGF-β1 (2 ng/ml), murine IL-4 (20 ng/ml), human IL-2 (50 U/ml), anti-IL-10R (10 mg/ml) and anti-IFN-γ (10 mg/ml). Th2 cells were cultured with human IL-2 (50 U/ml), murine IL-4 (20 ng/ml) and anti-IFN-γ (10 mg/ml). Th17 cells were cultured with murine IL-6 (100 ng/ml), human TGF-β1 (2 ng/ml), murine IL-1β (10 ng/ml), murine IL-23 (10 ng/ml), anti–IFN-γ (XMG; 20 mg/ml), anti–IL-4 (11B11; 10 mg/ml), and anti–IL-2 (10 mg/ml). After the initial 3 days culture, cells were removed from anti-CD3 coated plates and expanded with 3 volume of fresh media containing the same cytokine concentrations.
In vitro human T cell differentiation
Human naïve CD4+ T cells were isolated from human peripheral blood mononuclear cells (PBMCs) by using magnetic separation (Miltenyi Biotec). The cells were activated with a receptor crosslinking bead, Dynabead human T-activator CD3/CD28 (ThermoFisher Scientific) in complete media to generate Th9 cells (20 ng/ml hIL-4, 2 ng/ml hTGF-β1, 50U/ml hIL-2 and 10 μg/ml anti-IFNγ) and Th17 cells (2 ng/ml hTGF-β1, 20 ng/ml hIL-6, 10 ng/ml hIL-23, 10 ng/ml hIL-1β, 10 μg/ml anti-IL-4 and 10 μg/ml anti-IFNγ). Cells were grown at 37°C under 5% CO2 and were expanded after 3 days with original concentration of cytokines in fresh medium. Cells were harvested on day 5 and day 7 for analysis.
Analysis of gene expression and flow cytometry
Total RNA was isolated from cells cultured for 5 days using Trizol (Life Technologies). RNA was reverse transcribed according to manufacturer’s directions (Quantabio, Beverly, MA). Quantitative Reverse Transcriptase (qRT-PCR) was performed with commercially available primers (Life Technologies) with a 7500 Fast-PCR machine (Life Technologies). Gene expression was normalized to housekeeping gene expression (β2-microglobulin). For cytokine staining, CD4+ T cells were stimulated with Phorbol 12-myristate 13-acetate (PMA, 50 ng/ml, Sigma) and ionomycin (500 ng/ml, Sigma) for 2 hours followed by monensin (2μM, Biolegend) for total 5 hours at 37ºC. After restimulation, cells were washed with FACS buffer (PBS with 0.5% BSA). Cells were then stained with a fixable viability dye (eBioscience) and surface markers (CD4;RM4-4, Biolegend, CD90.1; OX-7, Biolegend) for 30 mins at 4 ºC followed by washing and fixation with 4% formaldehyde for 10 mins at room temperature. Cells were then permeabilized with permeabilization buffer (eBioscience) and stained for cytokines (IL-4; 11B11, Biolegend, IL-9; RM9A4, Biolegend, IL-10; JES5-16E3, Biolegend, IL-17A; eBio17B7, eBioscience,). For transcription factor staining, after surface staining, cells were fixed with Fixation & Permeabilization Buffer (eBioscience) for 2 hours at 4 ºC, and then permeabilized with permeabilization buffer (eBioscience). Transcription factors (BATF, BD Biosciences; IRF4, BioLegend; JunB, Santa Cruz; Bach2, Bioss) were stained for 30 mins at 4 ºC.
Retrovirus transduction
Bicistronic retroviral vectors containing BATF, IRF4, JunB, c-Jun, DN-Jun and Bach2 were used to generate retrovirus as previously described (20). Briefly, after 1 day of culture, activated CD4 T cells were infected with the retrovirus containing the interested gene or their empty vectors as controls in the presence of polybrene (8 μg/ml, Sigma) by centrifugation at 2300 rpm for 90 mins at 32 ºC. After centrifugation, the supernatant was replaced with Th cell polarizing media. Cell were expanded on day 3 and analyzed on day 5.
siRNA transfection
Activated T cells were transfected on day 1 with 1μM SMARTpool siRNA targeted the specific mRNA or the nontarget control (Thermo Scientific Dharmacon®) in the Accell siRNA delivery media with the Th cell condition antibodies and cytokines described as above. Cells were harvested for RT-qPCR analysis on day 3 and cytokine production on day 4 and day 5.
Immunoprecipitation
Whole-cell protein lysates were extracted from Th9 and Th17 cells on day 5. Cell lysates were pre-cleared with Protein A agarose bead slurry (50%, Millipore) for 1 hour at 4°C. Equal amount of protein was incubated with BATF antibody (Cell Signaling Technology) at 4°C overnight with rotation. Immunocomplexes were captured by Protein A agarose for 4 hours at 4°C with rotation and eluted at 100°C for 5 mins in Laemmli’s sample buffer. Immunoprecipitates were separated by 4%−20% SDS-PAGE and immunoblotted with BATF, JunB, c-Jun and Bach2 antibodies (Cell Signaling Technology).
Chromatin Immunoprecipitation (ChIP)
CD4 T cells were cross-linked with 1% formaldehyde for 15 mins at room temperature with rotation. The reaction was quenched with 0.125 M glycine for 5 mins. Cells were lysed by adding the cells lysis buffer for 10 mins on ice and nuclear lysis for 20 mins on ice. Nuclear extracts were fragmented by sonication. After pre-clearing with salmon sperm DNA (ThermoFisher Scientific), bovine serum albumin, and protein A agarose bead slurry (50%, Millipore), the supernatant was incubated with either rabbit polyclonal BATF, JunB, IRF4, Bach2 (Cell Signaling Technology), H3K4me3, H3K27ac (Abcam) antibody or normal rabbit IgG (Millipore) at 4°C overnight with rotation. The immunocomplexes were incubated with protein A agarose bead for 4 hours at 4°C, washed, eluted, and reversed cross-links at 65 °C overnight. DNA was purified by phenol-chloroform extraction and resuspended in 200 μl of nuclease free water and analyzed by qRT-PCR as previously described. The supernatant from IgG samples was kept as input samples. After normalization to the Input DNA, the amount of output DNA of each target protein was calculated by subtracting that of the IgG control. ChIP primers were used as described (20).
Statistical Analysis
All data analysis was done using Prism software version 7 (Graphpad Software) using a Student’s t-test. Post hoc Tukey test was used for multiple comparisons. p≤ 0.05 was considered statistically significant.
Results
BATF and IRF4 are not the sole limiting factors for IL-9 expression in Th17 cells
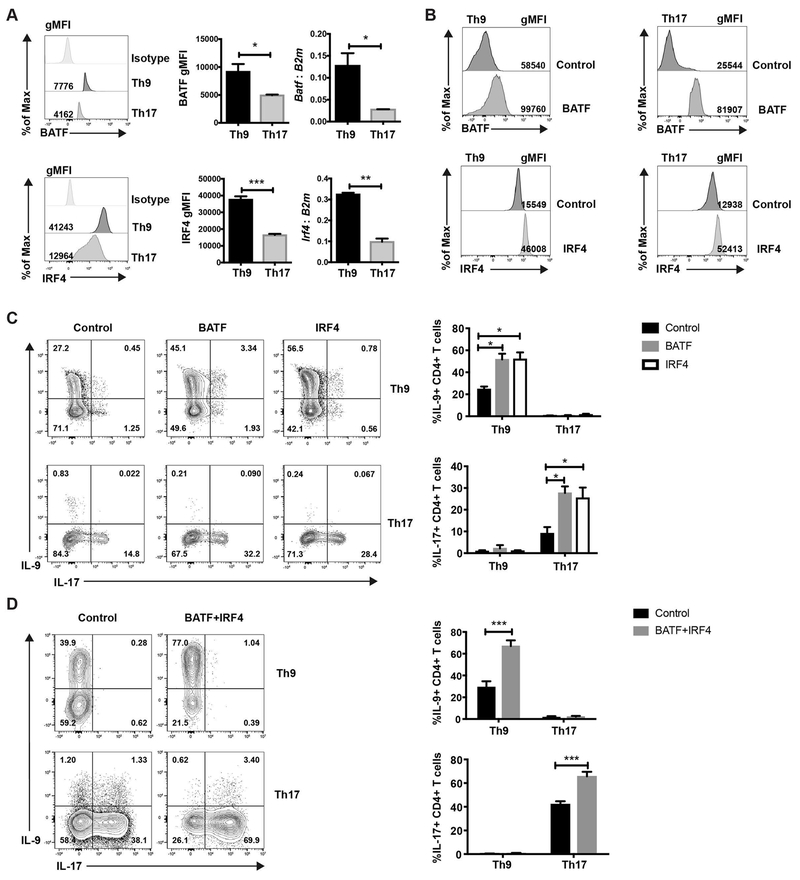
BATF cooperates with IRF4 in driving Th subset differentiation (13), and IL-9 production is diminished in BATF- and IRF4-deficient Th9 cultures (11, 12). Thus, IRF4 might be a key factor that affects the specificity of BATF in Th subsets. To test this, we measured BATF and IRF4 expression in Th9 and Th17 cells. Th9 cells showed greater expression of both BATF and IRF4 than Th17 cells (Fig. 1A). In order to examine whether the low expression of BATF or IRF4 limits IL-9 production in Th17 cells, Th9 and Th17 cells were transduced with BATF- or IRF4-expressing retrovirus separately (Fig. 1B). The transduction significantly increased the protein level of BATF or IRF4 in the cells (Figure 1B). Consistent with previous studies (11, 15), BATF and IRF4 promoted IL-9 production in Th9 cells and IL-17 production in Th17 cells (Fig. 1C). In contrast, BATF or IRF4 transduction failed to enhance IL-9 production in Th17 cells or IL-17 production in Th9 cells (Fig. 1C). The inability to enhance IL-9 production in Th17 cultures might be due to a requirement for high amounts of both BATF and IRF4. Therefore, Th9 and Th17 cells were co-transduced with BATF and IRF4. Similar to what we observed with the single transduction of BATF or IRF4, co-transduction of BATF and IRF4 only promoted IL-9 production in Th9 cells and was still not capable of inducing IL-9 production in Th17 cells (Fig. 1D). BATF and/or IRF4 transduction also failed to increase IL-9 production in Th2 cells (Supplemental Fig. 1). Together, these data indicate that the low expression of BATF and/or IRF4 is not the sole limiting factor for IL-9 production in Th17 cells. Thus, other factors must contribute to the specificity of BATF in regulating IL-9 production.
Fig. 1. BATF promotes lineage specific cytokine production in Th9 and Th17 subsets.
(A-C) Naive CD4 T cells were cultured under Th9 or Th17 conditions. Cells were harvested on day 5. (A) BATF and IRF4 expression was detected by intracellular staining and qRT-PCR. RNA expression data was normalized to B2m expression. (B and C) Cells were transduced with MIEG-GFP, BATF-GFP or IRF4-GFP expressing retrovirus on day 1. Transcription factor expression and cytokine expression was detected on day 5 after PMA/ionomycin stimulation. Dot plots were gated on cells that were live CD4+GFP+ cells. (D) Cells were co-transduced with MSCV-Thy1.1 and MIEG-GFP control or BATF-Thy1.1 and IRF4-GFP expressing retrovirus. Cytokine production was detected on day 5 after PMA/ionomycin stimulation. Dot plots were gated on live CD4+Thy1.1+GFP+ cells. Date are mean ± SEM of 3 mice per experiment and representative of three independent experiments. A two tailed Student’s t-test was used for pairwise comparisons. One-way ANOVA with a post-hoc Tukey test was used to generate p-values for all multiple comparisons. *, p<0.05, **p<0.01, ***p<0.001.
Jun family members are required for IL-9 expression in Th9 cells
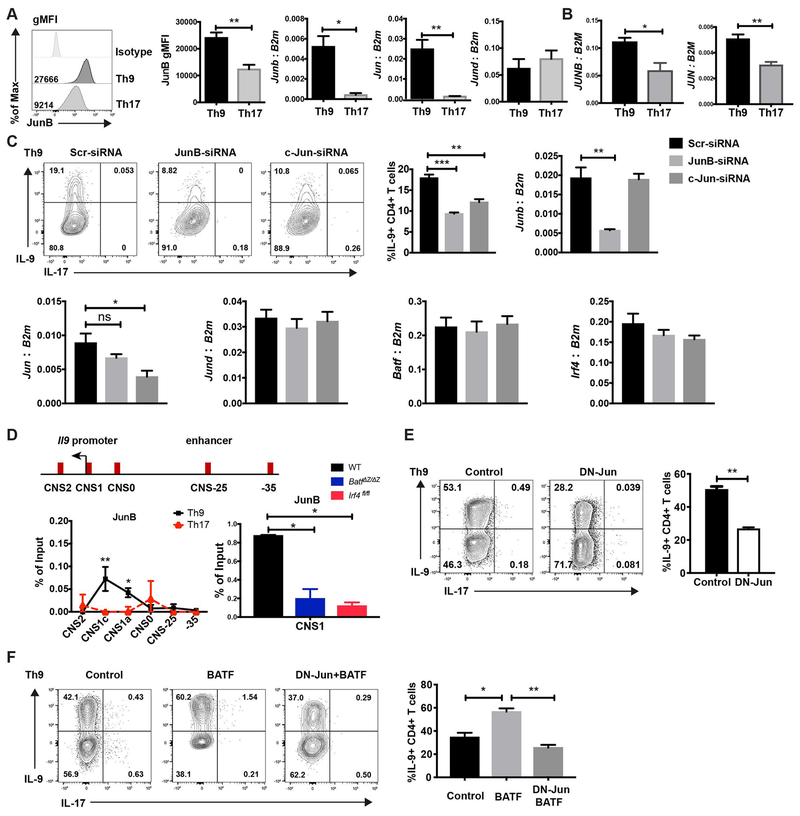
To determine the binding partners of BATF in Th9 and Th17 cells, we performed co-immunoprecipitation and found several factors that interacted with BATF including JunB and c-Jun (Supplemental Fig. 2). Heterodimers of BATF and Jun family members regulate cytokine production in Th2, Th17 and Tfh cells (18, 21–26). Yet, the function of Jun family members in Th9 cells has not been studied. Therefore, we hypothesized that Jun family members were crucial partners of BATF in Th9 cells. Based on our previous microarray data, the expression of Jun family members is higher in Th9 cells than in Th2 and Treg cells (11). Here we compared the expression of Jun family members in mouse Th9 and Th17 cells. Th9 showed greater expression of JunB protein (Fig. 2A). Both Junb and Jun had higher expression in Th9 cells than in Th17 cells, while Jund had similar expression in Th9 and Th17 cells (Fig. 2A). Consistent with the expression pattern in mouse T cells, human Th9 cells also showed higher expression of JUNB and JUN than in Th17 cells (Fig. 2B).
Fig. 2. JunB and c-Jun are required for IL-9 production in Th9 cells.
(A) Naive mouse CD4 T cells were cultured under Th9 and Th17 conditions. JunB expression was detected by intracellular staining. Jun family member expression was detected by qRT-PCR. RNA expression data was normalized to B2m expression. (B) Naïve human CD4 T cells were cultured under Th9 and Th17 conditions and Jun family member expression was detected by qRT-PCR. RNA expression data was normalized to B2M expression. (C) Naive CD4 T cells were cultured under Th9 condition, scrambled siRNA,JunB siRNA or c-Jun siRNA were transfected into the cells on day 1. Cells were collected for mRNA analysis after 48 hours transfection and cytokine production was detected on day 5 after PMA and ionomycin stimulation. Dot plots were gated on live CD4 cells. (D) Schematic of the Il9 locus indicating CNS elements (top). ChIP assay analysis of JunB at the CNS regions of the Il9 gene in WT, BatfΔZ/ΔZ and Irf4fl/fl Th9 and WT Th17 cells (bottom). A non-conserved sequence at −35kb was used as a negative control. Percent input depicted are the JunB ChIP values after subtraction of the control IgG ChIP values. (E) Day1 Th9 cells were transduced with MIEG control or DN-Jun expressing retrovirus on day 1 and cytokine production was detected on day 5 after PMA/ionomycin stimulation for 5 hours. Dot plots were gated on live CD4+ GFP+ cells. (F) Naive CD4 T cells were cultured under Th9 condition and transduced by control retrovirus, BATF expressing retrovirus, or both BATF and DN-Jun expressing retrovirus on day 1. Cytokine production was detected on day 5 after PMA and ionomycin stimulation. Dot plots were gated on live CD4 transduced cells. Data are mean ± SEM of 3 mice per experiment and representative of three independent experiments. A two tailed Student’s t-test was used for pairwise comparisons. One-way ANOVA with a post-hoc Tukey test was used to generate p-values for all multiple comparisons. *p<0.05, **p<0.01.
Next, we investigated if a specific Jun family member was required for IL-9 production in Th9 cells. To approach this, Th9 cells were transfected with JunB-, c-Jun- or JunD-specific siRNA (Fig. 2C and Supplemental Fig. 3). The knockdown efficiency was confirmed 48 hours after the transfection (Fig. 2C and Supplemental Fig. 3). Knocking down Junb or Jun did not interfere with the expression of other Jun family member expression (Fig. 2C). IL-9 expression was diminished when the expression Junb or Jun was decreased (Fig. 2C). However, decreased Jund expression did not affect IL-9 expression (Supplemental Fig. 3). The reduced expression of Junb or Jun did not affect Batf or Irf4 expression in Th9 cells. These results indicate that JunB and c-Jun promote IL-9 production in Th9 cells.
The Il9 gene has been characterized by three conserved non-coding sequences (CNS): the promoter (CNS1), a region 6 kb upstream of the transcriptional start site (CNS0), and a region 5.4 kb downstream of the transcriptional start (CNS2) (6). Our group recently described an enhancer region, 25 kb upstream of the Il9 gene, that promoted Il9 gene expression (Fig. 2D) (20). In our previous study we found that BATF bound to the Il9 promoter and enhancer regions in Th9 cells (11, 20). As a major binding partner of BATF, it is possible that Jun family members directly bound to the Il9 gene and interacted with BATF at the gene locus. As expected, JunB strongly bound at the Il9 promoter in Th9 cells, but not in Th17 cells (Fig. 2D). Binding in Th9 cells was predominantly in the promoter region. The binding of JunB at the Il9 promoter is decreased in both BATF-deficient and IRF4-deficient Th9 cells (Fig. 2D). This result suggests that IRF4 stabilizes a BATF-JunB heterodimer at the Il9 gene in Th9 cells.
To investigate if Jun family members are required by BATF to promote IL-9 production, we utilized a dominate-negative Jun (DN-Jun) retroviral vector (27). The N-terminal activation domain of Jun is truncated, so it dominantly forms a DNA binding complex with BATF predicted to inhibit BATF function. DN-Jun transduction significantly inhibited IL-9 expression in Th9 cells indicating that the transactivation domain of Jun family members is indispensable for IL-9 production in Th9 cells (Fig. 2E). To further confirm the transactivation domain of Jun family members is required by BATF in Th9 cells, we co-transduced BATF and DN-Jun into the Th9 cells. Similar to the result in Figure 1C, ectopic transduction of BATF increased IL-9 production (Fig. 2F). However, the BATF and DN-Jun co-transduced cells expressed significantly less IL-9 than cells transduced with BATF alone (Fig. 2F). These data indicate that DN-Jun inhibits the function of BATF in Th9 cells and further supports that the transactivation domain of JunB and c-Jun family members is required by BATF to promote IL-9 production in Th9 cells.
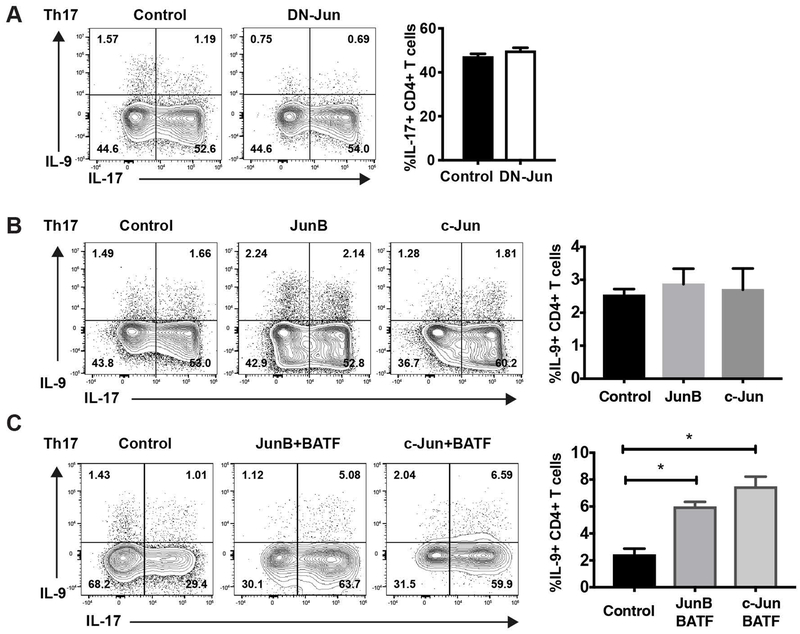
JunB and c-Jun functionally cooperate with BATF in promoting IL-9 production in Th17 cells
Jun family members are required for IL-17 production in Th17 cells, although the structural requirements are not well-defined (22, 23, 28, 29). To test the requirement for Jun family member transactivation domain in IL-17 regulation, Th17 cells were transduced with DN-Jun expressing retrovirus. Surprisingly, IL-17 production was unchanged, suggesting that the Jun transactivation domain is not required for IL-17 production (Fig. 3A). Compared to Th9 cells, Th17 cells showed lower expression of JunB and c-Jun (Fig. 2A). Therefore, the low expression of JunB and c-Jun might restrict the capacity of BATF to induce IL-9 production in Th17 cells. However, the transduction of JunB or c-Jun failed to increase IL-9 production in Th17 cells (Fig. 3B). This might be due to a requirement in Th17 cells for increased expression of JunB/c-Jun, coupled with increased BATF. To test this hypothesis, we co-transduced BATF with JunB or c-Jun into Th17 cells (Fig. 3C). Compared to control vector-transduced cells, co-transduction of BATF and JunB or c-Jun significantly increased IL-9 production, albeit at percentages much less than observed in Th9 cultures (Fig. 3C). In all, these results demonstrate that the low expression of JunB and c-Jun are limiting factors in the ability of BATF to promote IL-9 production. Moreover, JunB and c-Jun functionally cooperate with BATF in promoting IL-9 production and the requirement for the Jun family transactivation domain distinguishes the contribution of family members at specific cytokine loci.
Fig 3. BATF-JunB and BATF-c-Jun complex promotes IL-9 in Th17 cells.
Naive CD4 T cells cultured under Th17 conditions were transduced with DN-Jun-GFP expressing retrovirus alone (A), JunB-GFP or c-Jun-GFP expressing retrovirus alone (B), or BATF-Thy1.1 and JunB-GFP or BATF-Thy1.1 and c-Jun-GFP expressing retrovirus (C) on day 1. Cells were stimulated by PMA/ionomycin for 5 hours on day 5 and cytokine production was analyzed by ICS. Dot plots were gated on live CD4 transduced cells. Data are mean ± SEM of 3 mice per experiment and representative of three independent experiments. A two tailed Student’s t-test was used for pairwise comparisons. One-way ANOVA with a post-hoc Tukey test was used to generate p-values for all multiple comparisons. *p<0.05.
Bach2 promotes IL-9 production in Th9 cells
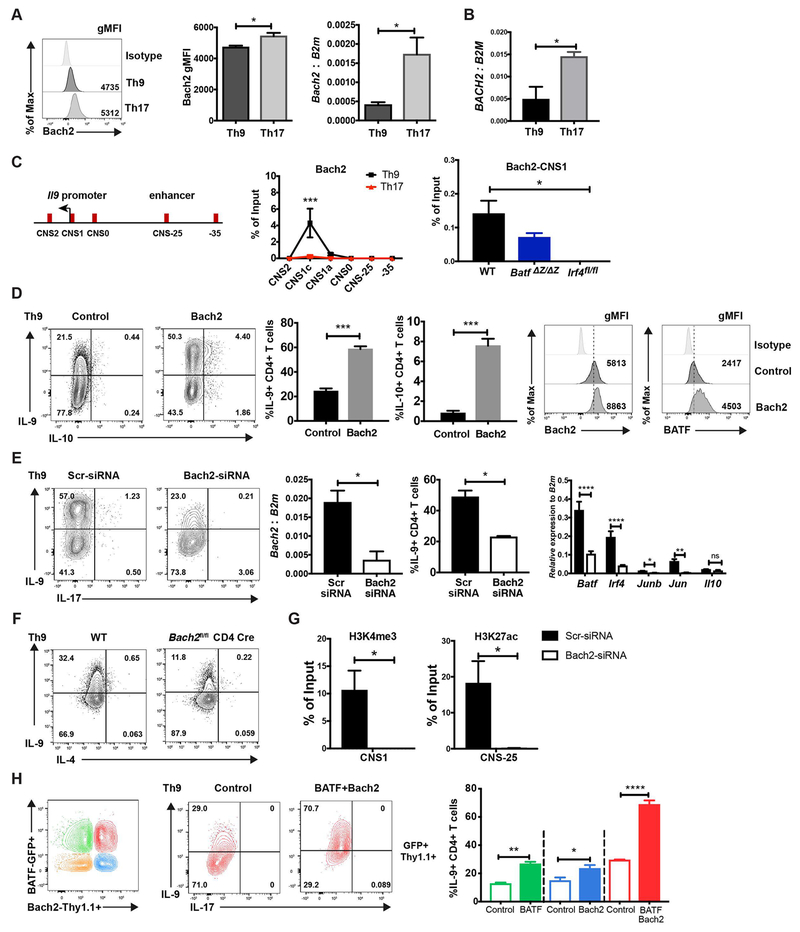
Bach2 represses the production of multiple cytokines in effector CD4 T cells and maintains the stability of Tregs (30). Additionally, Bach2 restrains the ability of BATF to promote IL-4 production in Th2 cells (18). BATF immunoprecipitation showed that Bach2 co-immunoprecipitated with BATF (Supplemental Fig. 2), which suggested that Bach2 might also be a regulator of BATF activity in IL-9 production. Bach2 is expressed in both mouse and human Th9 cells and Th17 cells, with greater expression in Th17 cells (Fig. 4A and 4B). To test the binding of Bach2 to the Il9 gene, ChIP assay was performed in Th9 and Th17 cells. To our surprise, the binding of Bach2 at the Il9 promoter in Th9 cells was much higher than in Th17 cells even though Bach2 showed greater expression in Th17 cells (Fig. 4C). The binding of Bach2 on the Il9 promoter region was modestly decreased in BATF-deficient Th9 cells but was undetectable in IRF4-deficient Th9 cells (Fig. 4C). These results suggested that Bach2 binding at the Il9 promoter was dependent on IRF4.
Fig 4. Bach2 promotes IL-9 production in Th9 cells.
(A) Naive mouse CD4 T cells were cultured under Th9 and Th17 conditions. Bach2 expression was detected by ICS (mouse) and qRT-PCR (mouse and human) on day 5. (B) Naïve human CD4 T cells were cultured under Th9 and Th17 conditions and BACH2 expression was detected by qRT-PCR. RNA expression data was normalized to B2M expression. (C) ChIP assay of Bach2 binding to the Il9 gene locus in WT, BATF- and IRF4-deficient Th9 and WT Th17 cells was performed on day 5. (D) MSCV-Thy1.1 or Bach2-Thy1.1 expressing retrovirus was transduced in Th9 cells on day 1. Cells were collected on day 5 for transcription factor staining. Cytokine production was detected after 5 hours PMA/ionomycin stimulation on day 5. Dot plots were gated on live CD4+ Thy1.1+ cells. (E) WT naïve CD4 cells were cultured under Th9 condition with anti-IL-10R and scrambled siRNA or Bach2-specific siRNA was transfected on day 1. Bach2 mRNA expression was analyzed by RT-qPCR after 48 hours transfection. Additional gene expression was performed on day 5 of culture. Cytokine production was detected after 5 hours PMA/ionomycin stimulation on day 4. Dot plots were gated on live CD4+ cells. (F) Naïve CD4 T cells from wild type and Bach2 conditional mutant mice were cultured under Th9 conditions and analyzed for cytokine production as in (E). (G) Cells generated as in (E) were used for ChIP assay of H3K4me3 and H3K27ac at the Il9 locus, performed on day 5. (H) Naive CD4 T cells were cultured under Th9 conditions. MIEG-GFP and MSCV-Thy1.1 (open box) or BATF-GFP and Bach2-Thy1.1 (closed box) expressing retrovirus was co-transduced into the Th9 cells on day1. Cytokine production was detected after 5 hours PMA/ionomycin stimulation on day 5. Dot plots were gated on live CD4+ transduced cells. Data are mean ± SEM of 3 mice per experiment and representative of three independent experiments. *p<0.05, **p<0.01, ***p<0.001, ****p<0.0001.
Since Th9 cells demonstrated lower Bach2 expression than Th17 cells, we wanted to determine if ectopic expression of Bach2 altered IL-9 production in Th9 cells. To test this, we introduced Bach2 in Th9 cells by retroviral transduction. Interestingly, ectopic Bach2 increased the production of IL-9 (Fig. 4D). Furthermore, Bach2 transduction promoted the expression of BATF (Fig. 4D).
We previously reported that IL-10 was a repressor of IL-9 production in Th9 cells (31), and Bach2 can repress IL-10 (32). Even though we compensate for the repressive effect of IL-10 by incubating Th9 cultures in the presence of anti-IL-10R, we wanted to determine if decreased endogenous IL-10 production could explain the result through an indirect mechanism. However, Bach2 induced IL-10 production in a small percentage of cells (Fig. 4D), suggesting that limiting IL-10 production is not an intermediate step in the ability of Bach2 to regulate IL-9.
To further assess the requirement of Bach2 for IL-9 production, Bach2 expression was inhibited by Bach2 specific siRNA. The knockdown of Bach2 significantly diminished IL-9 production, and other IL-9-inducing transcription factors including Batf, Irf4, Junb and Jun, whereas lower Bach2 did not significantly change Il10 expression (Fig. 4E). Similarly, Bach2-deficient Th9 cells showed decreased IL-9 production (Fig. 4F). However, Bach2 siRNA did not diminish the low amount of IL-9 seen early in Th2 differentiation (Supplemental Fig. 4A) (12). Furthermore, the knockdown of Bach2 in Th9 cells results in decreased H3K4me3 at the promoter and decreased H3K27ac at the Il9 CNS-25 (Fig. 4G). Taken together, these data suggest that Bach2 promotes IL-9 production by directly altering chromatin modifications at the Il9 gene locus and/or indirectly by enhancing IL-9 inducing transcription factors expression in Th9 cells.
To then determine if Bach2 cooperates with BATF in Th9 cells, we co-transduced BATF-IRES-EGFP with BACH2-IRES-Thy1.1 retroviruses into Th9 cells and analyzed IL-9 production on day 5 (Fig. 4H). By gating on GFP+, Thy1.1+ and GFP+Thy1.1+ populations we distinguished cells transduced with BATF alone, Bach2 alone, or both BATF and Bach2, respectively (Fig. 4H). In agreement with the previous results, BATF or Bach2 transduced cells showed increased IL-9 expression. Importantly, cells transduced with both BATF and Bach2 showed significantly increased IL-9 production compared to IL-9 production in BATF- and Bach2-single transduced cells, implying that Bach2 augments the ability of BATF to promote IL-9 production (Fig. 4H and Supplemental Fig. 4E). Thus, Bach2 cooperates with BATF in promoting IL-9 production in Th9 cells.
Bach2 cooperates with BATF in promoting IL-9 production in Th17 cells
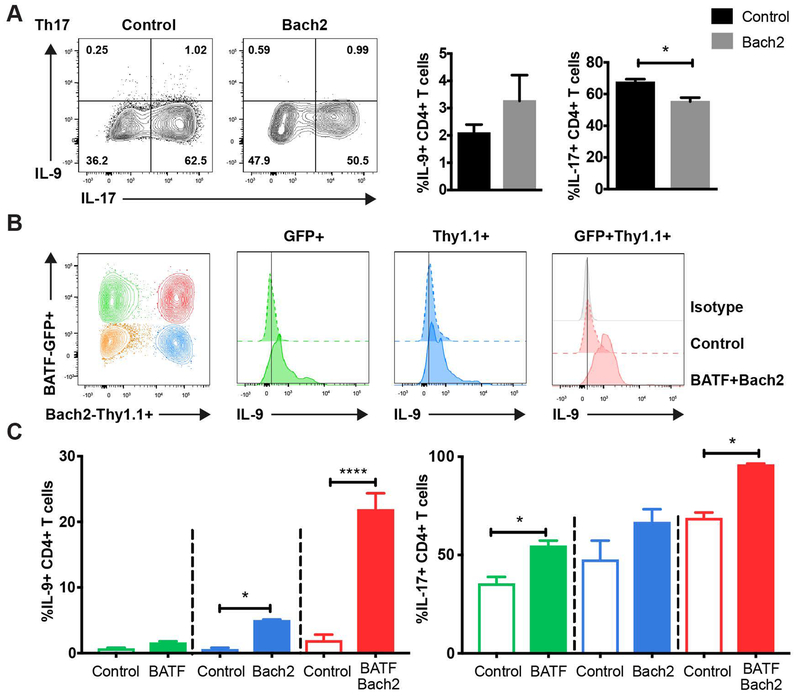
Because Bach2 cooperated with BATF in regulating IL-9 production in Th9 cells, we speculated that Bach2 might also regulate IL-9 production in Th17 cells. To define Bach2 function in Th17 cells, a Bach2-expressing retrovirus was transduced in Th17 cells. Transduction of Bach2 inhibited IL-17 production but did not enhance IL-9 production in Th17 cells (Fig. 5A). Because Bach2 cooperated with BATF in inducing IL-9 production in Th9 cells, we hypothesized that high expression of both BATF and Bach2 is required for IL-9 production in Th17 cells. To test this hypothesis, we co-transduced BATF-GFP and Bach2-Thy1.1 into Th17 cells (Fig. 5B). Compared to the cells transduced with the control vectors, co-transduction of BATF and Bach2 significantly increased the IL-9 production to over 20% in Th17 cells, near the amount of IL-9 production in Th9 cells (Fig. 5C). Taken together, these data indicate that BATF and Bach2 can cooperate to induce IL-9 production in Th17 cells (Fig. 6).
Fig 5. Bach2 functionally cooperates with BATF for IL-9 production in Th17 cells.
Naive cells were cultured under Th17 condition (A-C). (A) MSCV-Thy1.1 control or Bach2-Thy1.1 expressing retrovirus was transduced on day 1. Cytokine production was detected after 5 hours PMA/ionomycin stimulation on day 5. (B-C) MIEG-GFP and MSCV-Thy1.1 control (open box) or BATF-GFP and Bach2-Thy1.1 (closed box) expressing retrovirus were co-transduced on day 1. Cytokine production was detected after 5 hours PMA/ionomycin stimulation on day 5. Contour plots were gated on live CD4+ transduced cells and results are displayed as histograms (B) and bar graphs (C) of gated populations. Data are mean ± SEM of 3 mice per experiment and representative of three independent experiments. A two tailed Student’s t-test was used for pairwise comparisons. *p<0.05, **p<0.01, ***p<0.001, ****p<0.0001.
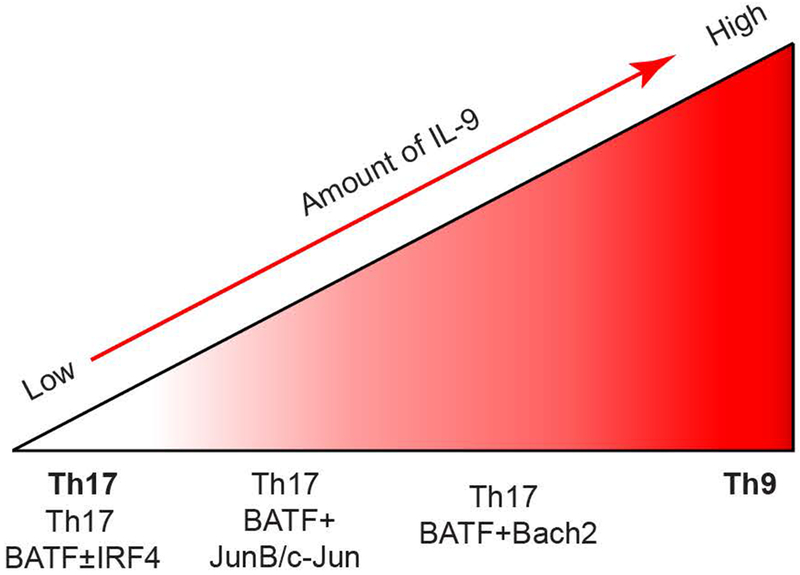
Fig. 6. Conceptual model of the cooperation of bZIP proteins in Il9 regulation.
BATF and/or IRF4 is not sufficient to promote IL-9 expression in Th17 cells. Heterodimer of BATF and JunB/c-Jun modestly activates Il9 in Th17 cells. The BATF-Bach2 complex further promotes IL-9 production in Th17 cells.
Discussion
Some Th subsets express unique transcription factors, such as T-bet in Th1 cells. However, multiple Th subsets share common transcription factors (2, 6). The mechanism that contributes to the specificity of the shared transcription factors in various Th subsets is still not clear. In this study, we find the binding partners affect the specificity of BATF in inducing IL-9 production in Th9 and Th17 cells. We demonstrate JunB and c-Jun are essential for the ability of BATF to promote IL-9 expression in Th9 cells. It was interesting to find that instead of functioning as a transcriptional repressor, Bach2 positively regulates IL-9 production. JunB/c-Jun and Bach2 facilitate the capacity of BATF to promote IL-9 production in Th9 cells.
IRF4 has been shown to interact with BATF-Jun and bind to AP1–IRF composite elements (AICE) on target genes in Th2 and Th17 cells (13, 22, 33). Previously, we demonstrated that BATF cooperated with IRF4 for IL-9 production in Th9 cells. The binding of BATF or IRF4 was diminished in the reciprocal gene-deficient Th9 cells (11). Initially, we thought IRF4 might limit the ability of BATF to promote IL-9 production in Th17 cells. Surprisingly, transduction of BATF and/or IRF4 was not sufficient to induce IL-9 production in Th17 cells,which indicated additional factors interact with BATF alone or BATF and IRF4 to regulate IL-9 production. Importantly, the binding of BATF-containing complexes with JunB or Bach2 are dependent on IRF4 in Th9 cells. As IRF4 also seems to integrate TGFβ signals (34), it is clearly a critical factor in organizing the IL-9-inducing enhanceosome.
Our results do not diminish the importance of distinct Jun family members in other subsets. Both JunB and JunD interact with BATF to regulate IL-4 and IL-13 production in Th2 cells (18, 21). Jun family members also facilitate the DNA binding activity of BATF and IRF4 on target genes in Th17 cells (28). In this report, we demonstrated that JunB and c-Jun functionally cooperated with BATF in promoting IL-9 production. The specificity of BATF in Th9 and Th17 cells is also distinguished by the requirement for the transactivation domain of Jun family members, with results suggesting that the activation domain of Jun family partners can compensate for the absence of the Jun activation domain. In contrast, JunD knockdown did not impair IL-9 production. It was striking to find that JunB or c-Jun knockdown did not affect the expression of other Jun family members in Th9 cells (Fig. 2), which is in contrast with the ability of JunD to functionally compensate for the loss of JunB in Th17 cells (29). This is indicative of JunB and c-Jun having nonredundant function in promoting IL-9 production in Th9 cells. Whether this is entirely through interactions with BATF is not clear. JunB also interacts with Fos-related antigen1 (FRA1) to directly regulate IL-17 production in Th17 cells (35), whereas the c-Jun-c-Fos complex inhibits IL-17 production in Th17 cells (36). Together, these data suggest that Jun family members have distinct functions among the Th subsets and that the balance of function of Fos/Jun and BATF/Jun family members likely dictates gene expression patterns.
Although Bach2 represses IL-4, IL-13, and IFN-γ (16, 30), we observe that Bach2 promotes IL-9 production, suggesting its transcription regulatory function is gene-dependent. Bach2 induces cytokines in other contexts including IL-2 (37)(Supplemental Fig. 4B). Bach2 also has unique effects on the Il10 gene in Th9 cells. Bach2 transduction promotes IL-10 production in Th9 cells, which is different from the inhibitory effect of Bach2 on IL-10 in naive CD4 T cells and iTregs (32, 38). However, Bach2 knockdown did not affect Il10 expression in Th9 cells (Figure 4), suggesting that retroviral expression of Bach2 might have non-physiological effects on the Il10 gene. The mechanism of switching between activating and repressing Bach2 functions is still not clear. Previous studies have shown that Bach2 antagonizes Bcl6 and Blimp1 in the development of B cells (39–41). Both Bcl6 and Blimp1 have repressive effects on IL-9 production (42, 43). Thus, it is possible that Bach2 could promote IL-9 production by inhibiting the repressive effectors of Blimp1 and Bcl6. Bach2 also regulates the expression of IL-9-inducing transcription factors in Th9 cells (Fig. 4), and some of the regulatory effects of Bach2 could be indirect. BATF also cooperates with c-Maf in promoting IL-4 production in Tfh cells (24). Bach2 forms a heterodimer with Maf family members and binds to Maf-recognition elements on target gene locus (16). We previously reported that ectopic c-Maf expression inhibited IL-9 production in Th9 cells (9). Bach2 might compete with c-Maf in repression of the Il9 locus. Bach2 might form an active complex with BATF that inhibits the interaction between BATF and c-Maf, a potential repressive complex for IL-9 production. Modulating c-Maf activity might also explain why Bach2 shows opposite effects in Th2 and Th9 cells. Further investigations are expected to reveal the functions of the complex and context-dependent interactions among these transcription factors.
Our studies have focused on in vitro-derived Th9 and Th17 cultures. The question remains as to what effect Bach2 has during in vivo Th9 development. Previous reports on Bach2 function demonstrated amplified type 2 inflammation (18). Indeed, in that model, there is detectable Il9 expression above the naïve background levels (Supplemental Fig. 4C). Coupled with our in vitro results, these data suggest that Bach2 is an efficiency factor for IL-9 production, and not a critical factor for expression. In agreement with this idea, Th9 cultures with either knockdown or Cre-mediated deletion of Bach2 resulted in only a partial decrease in IL-9 production. The ability of Bach2 to control Th2 cytokines (18), even in Th9 cultures (Supplemental Fig. 4D), obscures our ability to distinguish the effects of Bach2 on Il9. It is possible that a model of Th9 function that is independent of type 2 inflammation would allow isolation of the effects of Bach2 on IL-9 production in vivo, and that will be an important experiment for the future.
BATF binds to the Il9 promoter and CNS-25 enhancer regions in Th9 cells (11, 20). Results from the ChIP assay shows both JunB and Bach2 bind preferentially to the Il9 promoter region. Binding of all three factors is diminished in Th17 cells, compared to Th9 cells. Despite the Il9 CNS-25 enhancer being required for optimal IL-9 production and BATF binding to this region (20), binding of Bach2 and JunB was not significantly detected in this region. While we have shown IRF4 binds to the CNS-25 region in Th9 cells, it is possible that BATF cooperates with other partners at the enhancer region to activate Il9 gene expression. The ability of BATF to integrate site-specific complex formation might be a key feature of its ability to regulate Il9.
While Il9 transcription relies on many transcription factors, it is important to note that overexpression of any one bZIP factor is not sufficient to transdifferentiate Th17 cells into IL-9-producers. Transduction of BATF, Bach2, or Jun family members was not sufficient to induce IL-9 in Th17 cells (Fig. 6). The combination of BATF and Jun family members was sufficient to induce modest IL-9 in Th17, while BATF and Bach2 was more potent at inducing IL-9. However, even the combination of BATF and Bach2 did not induce IL-9 to the amounts observed in Th9 cultures (Fig. 6). This highlights that although ectopic transcription factor expression likely shifts the equilibrium from a closed chromatin state in Th17 cells to a more open configuration similar to Th9 cells by recruiting additional chromatin modifying enzymes to the target loci, that shift might not be sufficient to recapitulate the chromatin signature at the target gene compared to a cell cultured under optimal cytokine-inducing conditions. The chromatin structure at the Il9 locus in Th17 cells is likely another limiting factor in activating expression. Thus, in activating IL-9 production, BATF and Bach2 are not re-writing the network required for Th9 differentiation and not re-creating the optimal Th9 phenotype.
These studies highlight multiple mechanisms at the core of transcription factor specificity in Th subsets. First, there may be differences in the amount of expression. BATF and Jun family members are expressed in all Th subsets, but at varying amounts (11). Second, the requirement for interacting proteins may differ among subsets. BATF appears to require IRF4 for gene induction in multiple subsets (11, 13, 22), but the requirement for specific Jun family members, and Jun functional domains, might be distinct among subsets. Third, there are likely temporal differences in expression among BATF interacting proteins as well as time-dependent and perhaps subset-specific differences in the signals that regulate heterodimer formation. Finally, bZIP-interacting proteins might have distinct functions in each subset. Bach2 represses IL-17 production but induces IL-9 production. Each of these mechanisms likely contributes to the specificity of transcription factor function.
Supplementary Material
Key Points:
Specific Jun family members cooperate with BATF in promoting IL-9 in Th9 cells.
Bach2 cooperates with BATF to promote IL-9 in Th9 cells.
Expression and function of bZIP factors dictate IL-9 activation in Th17 cells.
Acknowledgements
We thank Drs. Baohua Zhou and Alexander Dent for review of this manuscript.
This work was supported by PHS grants from the National Institutes of Health R01 AI057459, and R03 AI135356 to M.H.K. BJU was supported by T32 AI060519 and F30 HL147515. Core facility usage was also supported by IU Simon Cancer Center Support Grant P30 CA082709 and U54 DK106846. Support provided by the Herman B Wells Center was in part from the Riley Children’s Foundation.
Footnotes
Conflicts of Interest: The authors declare no conflicts of interest.
Reference
- 1.Zhu J, Yamane H, and Paul WE. 2010. Differentiation of Effector CD4 T Cell Populations. Annu. Rev. Immunol 28:445–489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Oestreich KJ and Weinmann AS. 2012. Master regulators or lineage-specifying? Changing views on CD4+ T cell transcription factors. Nat. Rev. Immunol 12:799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Goswami R and Kaplan MH. 2011. A Brief History of IL-9. J. Immunol 186:3283–3288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Schmitt E, Klein M, and Bopp T. 2014. Th9 cells, new players in adaptive immunity. Tredns. immunol 35:61–68. [DOI] [PubMed] [Google Scholar]
- 5.Kaplan MH, Hufford MM, and Olson MR. 2015. The development and in vivo function of T helper 9 cells. Nat. Rev. Immunol 15:295–307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kaplan MH 2017. The transcription factor network in Th9 cells. Semin. Immunopathol 39:11–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Elyaman W, Bassil R, Bradshaw Elizabeth M., Orent W, Lahoud Y, Zhu B, Radtke F, Yagita H, and Khoury Samia J.. 2012. Notch Receptors and Smad3 Signaling Cooperate in the Induction of Interleukin-9-Producing T Cells. Immunity. 36:623–634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Yao W, Zhang Y, Jabeen R, Nguyen Evelyn T, Wilkes David S., Tepper Robert S., Kaplan MH, and Zhou B. 2013. Interleukin-9 Is Required for Allergic Airway Inflammation Mediated by the Cytokine TSLP. Immunity. 38:360–372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Goswami R, Jabeen R, Yagi R, Pham D, Zhu J, Goenka S, and Kaplan MH. 2012. STAT6-Dependent Regulation of Th9 Development. J. Immunol 188:968–975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Chang HC, Sehra S, Goswami R, Yao W, Yu Q, Stritesky GL, Jabeen R, McKinley C, Ahyi AN, Han L, Nguyen ET, Robertson MJ, Perumal NB, Tepper RS, Nutt SL, and Kaplan MH. 2010. The transcription factor PU.1 is required for the development of IL-9-producing T cells and allergic inflammation. Nat. Immunol 11:527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Jabeen R, Goswami R, Awe O, Kulkarni A, Nguyen ET, Attenasio A, Walsh D, Olson MR, Kim MH, Tepper RS, Sun J, Kim CH, Taparowsky EJ, Zhou B, and Kaplan MH. 2013. Th9 cell development requires a BATF-regulated transcriptional network. J. Clin. Invest 123:4641–4653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Staudt V, Bothur E, Klein M, Lingnau K, Reuter S, Grebe N, Gerlitzki B, Hoffmann M, Ulges A, Taube C, Dehzad N, Becker M, Stassen M, Steinborn A, Lohoff M, Schild H, Schmitt E, and Bopp T. 2010. Interferon-Regulatory Factor 4 Is Essential for the Developmental Program of T Helper 9 Cells. Immunity. 33:192–202. [DOI] [PubMed] [Google Scholar]
- 13.Murphy TL, Tussiwand R, and Murphy KM. 2013. Specificity through cooperation: BATF–IRF interactions control immune-regulatory networks. Nat. Rev. Immunol 13:499. [DOI] [PubMed] [Google Scholar]
- 14.Schraml BU, Hildner K, Ise W, Lee WL, Smith WAE, Solomon B, Sahota G, Sim J, Mukasa R, Cemerski S, Hatton RD, Stormo GD, Weaver CT, Russell JH, Murphy TL, and Murphy KM. 2009. The AP-1 transcription factor Batf controls TH17 differentiation. Nature. 460:405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ciofani M, Madar A, Galan C, Sellars M, Mace K, Pauli F, Agarwal A, Huang W, Parkurst Christopher N., Muratet M, Newberry Kim M., Meadows S, Greenfield A, Yang Y, Jain P, Kirigin Francis K., Birchmeier C, Wagner Erwin F., Murphy Kenneth M., Myers Richard M., Bonneau R, and Littman Dan R.. 2012. A Validated Regulatory Network for Th17 Cell Specification. Cell. 151:289–303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Richer MJ, Lang ML, and Butler NS. 2016. T Cell Fates Zipped Up: How the Bach2 Basic Leucine Zipper Transcriptional Repressor Directs T Cell Differentiation and Function. J. Immunol 197:1009–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Roychoudhuri R, Clever D, Li P, Wakabayashi Y, Quinn KM, Klebanoff CA, Ji Y, Sukumar M, Eil RL, Yu Z, Spolski R, Palmer DC, Pan JH, Patel SJ, Macallan DC, Fabozzi G, Shih HY, Kanno Y, Muto A, Zhu J, Gattinoni L, O’Shea JJ, Okkenhaug K, Igarashi K, Leonard WJ, and Restifo NP. 2016. BACH2 regulates CD8+ T cell differentiation by controlling access of AP-1 factors to enhancers. Nat. Immunol 17:851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kuwahara M, Ise W, Ochi M, Suzuki J, Kometani K, Maruyama S, Izumoto M, Matsumoto A, Takemori N, Takemori A, Shinoda K, Nakayama T, Ohara O, Yasukawa M, Sawasaki T, Kurosaki T, and Yamashita M. 2016. Bach2–Batf interactions control Th2-type immune response by regulating the IL-4 amplification loop. Nat. Commun 7:12596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kometani K, Nakagawa R, Shinnakasu R, Kaji T, Rybouchkin A, Moriyama S, Furukawa K, Koseki H, Takemori T, and Kurosaki T. 2013. Repression of the Transcription Factor Bach2 Contributes to Predisposition of IgG1 Memory B Cells toward Plasma Cell Differentiation. Immunity. 39:136–147. [DOI] [PubMed] [Google Scholar]
- 20.Koh B, Abdul Qayum A, Srivastava R, Fu Y, Ulrich BJ, Janga SC, and Kaplan MH. 2018. A conserved enhancer regulates Il9 expression in multiple lineages. Nat. Commun 9:4803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Bao K, Carr T, Wu J, Barclay W, Jin J, Ciofani M, and Reinhardt RL. 2016. BATF Modulates the Th2 Locus Control Region and Regulates CD4+ T Cell Fate during Antihelminth Immunity. J. Immunol 197:4371–4381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Li P, Spolski R, Liao W, Wang L, Murphy TL, Murphy KM, and Leonard WJ. 2012. BATF–JUN is critical for IRF4-mediated transcription in T cells. Nature. 490:543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Hasan Z, Koizumi SI, Sasaki D, Yamada H, Arakaki N, Fujihara Y, Okitsu S, Shirahata H, and Ishikawa H. 2017. JunB is essential for IL-23-dependent pathogenicity of Th17 cells. Nat. Commun 8:15628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Sahoo A, Alekseev A, Tanaka K, Obertas L, Lerman B, Haymaker C, Clise-Dwyer K, McMurray JS, and Nurieva R. 2015. Batf is important for IL-4 expression in T follicular helper cells. Nat. Commun 6:7997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Li B, Tournier C, Davis RJ, and Flavell RA. 1999. Regulation of IL-4 expression by the transcription factor JunB during T helper cell differentiation. EMBO. J 18:420–432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hartenstein B, Teurich S, Hess J, Schenkel J, Schorpp-Kistner M, and Angel P. 2002. Th2 cell-specific cytokine expression and allergen-induced airway inflammation depend on JunB. EMBO. J 21:6321–6329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Wang ZY, Sato H, Kusam S, Sehra S, Toney LM, and Dent AL. 2005. Regulation of IL-10 Gene Expression in Th2 Cells by Jun Proteins. J. Immunol 174:2098–2105. [DOI] [PubMed] [Google Scholar]
- 28.Yamazaki S, Tanaka Y, Araki H, Kohda A, Sanematsu F, Arasaki T, Duan X, Miura F, Katagiri T, Shindo R, Nakano H, Ito T, Fukui Y, Endo S, and Sumimoto H. 2017. The AP-1 transcription factor JunB is required for Th17 cell differentiation. Sci. Rep 7:17402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Carr TM, Wheaton JD, Houtz GM, and Ciofani M. 2017. JunB promotes Th17 cell identity and restrains alternative CD4(+) T-cell programs during inflammation. Nat. Commun 8:301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Roychoudhuri R, Hirahara K, Mousavi K, Clever D, Klebanoff CA, Bonelli M, Sciumè G, Zare H, Vahedi G, Dema B, Yu Z, Liu H, Takahashi H, Rao M, Muranski P, Crompton JG, Punkosdy G, Bedognetti D, Wang E, Hoffmann V, Rivera J, Marincola FM, Nakamura A, Sartorelli V, Kanno Y, Gattinoni L, Muto A, Igarashi K, O’Shea JJ, and Restifo NP. 2013. BACH2 represses effector programs to stabilize Treg-mediated immune homeostasis. Nature. 498:506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ulrich BJ, Verdan FF, McKenzie AN, Kaplan MH, and Olson MR. 2017. STAT3 Activation Impairs the Stability of Th9 Cells. J. Immunol 198:2302–2309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Tsukumo SI, Unno M, Muto A, Takeuchi A, Kometani K, Kurosaki T, Igarashi K, and Saito T. 2013. Bach2 maintains T cells in a naive state by suppressing effector memory-related genes. Proc. Natl. Acad. Sci. U. S. A 110:10735–10740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Glasmacher E, Agrawal S, Chang AB, Murphy TL, Zeng W, Vander Lugt B, Khan AA, Ciofani M, Spooner CJ, Rutz S, Hackney J, Nurieva R, Escalante CR, Ouyang W, Littman DR, Murphy KM, and Singh H. 2012. A genomic regulatory element that directs assembly and function of immune-specific AP-1-IRF complexes. Science. 338:975–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Tamiya T, Ichiyama K, Kotani H, Fukaya T, Sekiya T, Shichita T, Honma K, Yui K, Matsuyama T, Nakao T, Fukuyama S, Inoue H, Nomura M, and Yoshimura A. 2013. Smad2/3 and IRF4 play a cooperative role in IL-9-producing T cell induction. J. Immunol 191:2360–71. [DOI] [PubMed] [Google Scholar]
- 35.Moon YM, Lee SY, Kwok SK, Lee SH, Kim D, Kim WK, Her YM, Son HJ, Kim EK, Ryu JG, Seo HB, Kwon JE, Hwang SY, Youn J, Seong RH, Jue DM, Park SH, Kim HY, Ahn SM, and Cho ML. 2017. The Fos-Related Antigen 1-JUNB/Activator Protein 1 Transcription Complex, a Downstream Target of Signal Transducer and Activator of Transcription 3, Induces T Helper 17 Differentiation and Promotes Experimental Autoimmune Arthritis. Front Immunol. 8:1793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Coquet JM, Middendorp S, Van der Horst G, Kind J, Veraar Elise A. M., Xiao Y, Jacobs H, and Borst J. 2013. The CD27 and CD70 Costimulatory Pathway Inhibits Effector Function of T Helper 17 Cells and Attenuates Associated Autoimmunity. Immunity. 38:53–65. [DOI] [PubMed] [Google Scholar]
- 37.Jang E, Lee HR, Lee GH, Oh AR, Cha JY, Igarashi K, and Youn J. 2017. Bach2 represses the AP-1-driven induction of interleukin-2 gene transcription in CD4 T cells. BMB Rep. 50:472–477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Kim EH, Gasper DJ, Lee SH, Plisch EH, Svaren J, and Suresh M. 2014. Bach2 regulates homeostasis of Foxp3+ regulatory T cells and protects against fatal lung disease in mice. J. Immunol 192:985–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Sherr DH and Monti S. 2013. The role of the aryl hydrocarbon receptor in normal and malignant B cell development. Semin. Immunopathol 35:705–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Huang C, Geng H, Boss I, Wang L, and Melnick A. 2014. Cooperative transcriptional repression by BCL6 and BACH2 in germinal center B-cell differentiation. Blood. 123:1012–1020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Swaminathan S, Duy C, and Muschen M. 2014. BACH2-BCL6 balance regulates selection at the pre-B cell receptor checkpoint. Trends Immunol. 35:131–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Bassil R, Orent W, Olah M, Kurdi AT, Frangieh M, Buttrick T, Khoury SJ, and Elyaman W. 2014. BCL6 controls Th9 cell development by repressing Il9 transcription. J. Immunol 193:198–207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Benevides L, Costa RS, Tavares LA, Russo M, Martins GA, da Silva LLP, de Paula Arruda L. Karla, Cunha FQ, Carregaro V, and Silva JS. 2018. B lymphocyte-induced maturation protein 1 controls TH9 cell development, IL-9 production, and allergic inflammation. J. Allergy Clin. Immunol [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.