ABSTRACT
Background
A metabolic adaptation, defined as an increase in energy expenditure (EE) beyond what is expected with weight gain during overfeeding (OF), has been reported but also refuted. Much of the inconsistency stems from the difficulty in conducting large, well-controlled OF studies in humans.
Objectives
The primary aim of this study was to determine whether a metabolic adaptation to OF exists and if so, attenuates weight gain.
Methods
Thirty-five young adults consumed 40% above their baseline energy requirements for 8 wk, and sleeping metabolic rate (SMR) and 24-h sedentary energy expenditure (24h-EE) were measured before and after OF. Subjects were asked to return for a 6-mo post-OF follow-up visit to measure body weight, body composition, and physical activity.
Results
After adjusting for gains in fat-free mass and fat mass, SMR increased by 43 ± 123 kcal/d more than expected (P = 0.05) and 24h-EE by 23 ± 139 kcal/d (P = 0.34), indicating an overall lack of metabolic adaptation during OF despite a wide variability in the response. Among the 30 subjects who returned for the 6-mo follow-up visit, those who had a lower-than-predicted SMR (basal EE) retained more of the fat gained during OF. Likewise, subjects displaying a higher-than-predicted sedentary 24h-EE lost significantly more fat during the 6-mo follow-up.
Conclusions
Metabolic adaptation to OF was on average very small but variable between subjects, revealing “thrifty” or “spendthrift” metabolic phenotypes related to body weight loss 6 mo later. This trial was registered at clinicaltrials.gov as NCT01672632.
Keywords: overfeeding, metabolic adaptation, dietary-induced thermogenesis, energy expenditure, sleeping metabolic rate, weight gain, respiratory chamber, luxuskonsumption, thrifty phenotype, spendthrift phenotype
Introduction
Energy expenditure (EE) increases with weight gain due to the energetic cost of depositing and maintaining new tissues, digesting food, and moving and maintaining a larger body mass. Beyond these obligatory energy costs, an additional increase in EE not explained by changes in body mass has been observed (1) and defined as metabolic adaptation.
A similar metabolic adaptation occurs in response to weight loss following caloric restriction, but in the opposite direction (1–3). Most calorie restriction studies in humans show that metabolic rate declines more than expected based on the reduction in metabolic mass (1, 4, 5). This metabolic slowing—also referred to as adaptive thermogenesis—is thought to result from hormonal, adipokine, and chemokine signals (6) that act to minimize the calorie deficit and protect against further weight loss. One prominent example of adaptive thermogenesis was the Minnesota Semi-Starvation Study where subjects lost only 65% of the weight they were predicted to lose with 50% calorie restriction, which was due to a 35% reduction in metabolic rate independent of changes in lean mass (7). Leibel et al. (1) later quantified adaptive thermogenesis in their classic series of weight perturbation studies and observed a reduction in resting EE by 250–300 kcal/d more than predicted. Our own weight loss studies have shown that EE declines 10–15% more than expected based on the loss of lean mass and fat mass (FM) (2, 3, 8).
The presence of a similar adaptation to dissipate excess energy in times of surplus would offer a considerable advantage to prevent obesity; however, its existence and magnitude are still debated (9–11). If present, such an adaptation—that is, an increase in EE larger than predicted based on weight gain—could explain why some individuals appear to resist weight gain (“spendthrift” phenotype) whereas others gain easily (“thrifty” phenotype) when challenged with caloric abundance (12, 13). In early experiments of protein overfeeding (OF), weight gain was much less than expected based on calories overfed, even after considering body composition and physical activity changes (14, 15). In the Vermont Prisoner Study, nearly 50% more energy intake was necessary to maintain a new higher body weight after OF (16). Subsequent experiments demonstrated that weight gain was less than expected for the caloric excess even after adjusting for the gain in body mass (1, 17) and the increased energy cost of digestion and storage (18). Even in studies in twins, weight gain was highly variable among nonrelated subjects undergoing the same OF regimen but was highly correlated within twin pairs (19), suggesting differences in the metabolic response are likely genetically determined. An adaptive response to OF might also reside in activity-related EE. Levine et al. (20) reported that most of the increase in EE with 1000 kcal/d extra for 8 wk was due to increased nonexercise activity thermogenesis, and the magnitude of increase accounted for 10-fold differences in fat storage. Still, not all findings agree on the presence of a metabolic adaptation to OF (11, 21, 22).
OF studies present many challenges, and the inherent difficulty in conducting these demanding experiments contributes to the mixed conclusions. Indeed, studies should be longer than 2 wk (14), highly controlled, and with sufficient sample sizes to detect meaningful effects. Here, 35 young adults consumed 40% above their baseline energy requirements for 8 wk under supervised conditions. The purpose of our study was to: 1) determine whether a metabolic adaptation to OF exists and assess its intersubject variability; 2) investigate whether this adaptation is associated with weight gain during OF or weight loss after 6 mo of follow-up; and 3) examine changes in circulating hormones and skeletal muscle mitochondrial efficiency that could contribute to a “spendthrift” or “thrifty” phenotype.
Methods
Subjects
Twenty-nine males (19 Caucasian, 10 African-American) and 6 females (2 Caucasian, 4 African-American) participated in the study. Details of the study design and methods have been reported previously (23). The study CONSORT diagram is provided as online supporting material (see Supplemental Figure 1). Participants were aged between 20 and 40 y with a BMI of 22–32 kg/m2 and had no history of chronic disease, obesity, or weight instability (>2.5 kg over the past 6 mo). Prior to inclusion, subjects completed a physical exam, medical history, and biochemical profile assessment to determine health status. Eligible participants were asked to maintain their current level of physical activity (i.e., refrain from starting or stopping an exercise regimen over the course of the study). The study was approved by the Pennington Biomedical Research Center (PBRC) Institutional Review Board, and all volunteers provided written informed consent prior to participation. Individual-level data at baseline and after OF are provided in Supplemental Table 1.
Total daily energy expenditure
Prior to OF, total daily energy expenditure (TDEE) was measured over a 2-wk period using doubly labeled water (DLW). Subjects provided 2 urine samples before being dosed with labeled water (2.0 g of 10% enriched H218O and 0.16 g of 99% enriched 2H2O per kilogram of estimated total body water) (Cambridge Isotopes). Additional urine samples were collected for analysis at 4.5 h and 6 h after dosing and on days 7 and 14. Samples were analyzed for 18O and 2H abundance by isotope ratio mass spectrometry, and the elimination rates were used to determine carbon dioxide production as previously described (24, 25). EE was calculated by multiplying the rate of carbon dioxide production by the energy equivalent of carbon dioxide using the 24-h respiratory quotient measured in a respiratory chamber. TDEE was measured again during the last 2 wk of the 8-wk OF intervention.
Diets
Energy requirements at baseline were determined using the measured EE (by DLW) and 7 d of feeding to energy balance. The average of these 2 values was multiplied by 1.4 to calculate the 40% OF prescription. All meals were prepared by the metabolic kitchen using a validated 5-d rotating menu (26) composed of 41% carbohydrate, 44% fat, and 15% protein. Participants were overfed for 8 wk and consumed all meals (3 meals/d, 7 d/wk) at PBRC under direct supervision but were free-living the remainder of the time. After 8 wk, subjects again consumed to energy balance for 3–5 d prior to post-OF metabolic testing. Following completion of OF, all subjects were offered free nutritional counseling for weight loss by a research dietitian and/or free membership for 6 mo to our on-campus exercise facility. Following 8 wk of OF, subjects were free-living until the 6-mo follow-up visit.
Baseline and post-OF measurements
24-h energy expenditure
Sedentary energy expenditure (24h-EE) was measured over 23 h in a whole-room calorimeter as previously described (27). Subjects were provided with 3 meals and 1 snack, which they consumed in entirety, and EE was calculated from oxygen consumption (VO2), carbon dioxide production (VCO2), and urinary nitrogen excretion (28). Energy expended during sleep (sleeping metabolic rate, SMR) was calculated between 0200 and 0500 when infrared motion sensors were reading near-zero motion. All urine was collected while subjects were in the chamber and was used to measure nitrogen, norepinephrine, and epinephrine excretion.
Physical activity
Activity was determined by combining TDEE (from DLW) and SMR (from the metabolic chamber) to calculate the physical activity level (PAL; defined as TDEE divided by SMR). Steps per day were measured using a validated activity monitor (SenseWear Pro3, BodyMedia, Inc.) at baseline and post-OF (29). Subjects wore the monitor for 7 continuous days (including time spent sleeping) and were asked to remove it only when showering or engaging in other water-related activities.
Mitochondrial efficiency
A combination of phosphorus-31 magnetic resonance spectroscopy (31P-MRS) and optical spectroscopy was used to measure in vivo skeletal muscle mitochondrial function as previously described (30, 31). Briefly, phosphorus spectra were collected on a 3T GE Signa magnet (GE) using a 4- or 6-cm 31P-tuned surface coil positioned over the distal portion of the vastus lateralis. To perturb phosphocreatine concentration [PCr], a pressure cuff was applied to the upper thigh and inflated to 60 mmHg above systolic pressure for 16 min, which blocked blood flow and inhibited oxygen delivery. The rate of PCr breakdown under anoxic conditions represents the basal ATP demand of the cell, that is, the amount that must be supplied by oxidative phosphorylation. The contribution of glycolytic ATP production was determined by the change in pH and PCr signal during ischemia (32) and was <10% of the demand (33). Spectra were analyzed using the Advanced Method for Accurate, Robust, and Efficient Spectral fitting algorithm in the Java-based Magnetic Resonance User Interface software (34). Using this method, repeated measures of muscle ATP flux on the same subject agree to within ±11% (35). In a separate session, optical spectra were obtained by two 10-mm optic bundles positioned over the vastus lateralis that fed into a Horiba Jobin Yvon optical spectrograph (Edison). One fiber illuminated the tissue, whereas the other carried reflected light to a spectrograph-mounted detector. Spectra were obtained for 17 min while a pressure cuff was inflated to 55 mmHg above systolic pressure. Oxygen uptake, representing oxygen demand of the muscle, was determined from the rate of deoxygenation of both oxygenated hemoglobin (HbO2) and oxygenated myoglobin (MbO2) during ischemia as previously described (36, 37). In our hands, repeated measures of oxygen consumption on the same subject agree to within ±13.7%.
Mitochondrial coupling efficiency (P/O ratio) was calculated as the ratio of phosphorylation (ATP demand by MRS) to oxidation (oxygen demand by optical spectroscopy) and was divided by 2 to yield P/O to conform to biochemical convention (31, 38). Repeated measures of P/O in our hands agree to within ±6% (31). These procedures were performed on a subset of subjects; therefore, data are available for 23 (ATP flux), 21 (oxygen uptake), and 20 (P/O ratio) subjects.
6-month follow-up testing
Thirty subjects returned to the PBRC Metabolic Unit 6 mo after completing 8 wk of OF, for measurement of body weight, body composition, and physical activity. No structured intervention was administered to subjects during this 6-mo unsupervised period.
Statistics
Statistical analyses were performed using JMP version 11 software (SAS Institute Inc.). Data are presented as mean ± SD with α level set at 0.05 and statistical tests 2-tailed. Changes in continuous variables from baseline to post-OF were analyzed by paired t test, and associations between variables were analyzed using Pearson correlation coefficients. Predictive equations for 24h-EE and SMR were derived from a reference population taking into account fat-free mass (FFM), FM, age, and race (where 1=African-American, 0=Caucasian) (39):
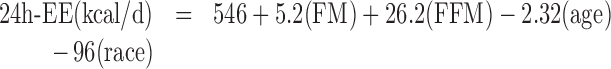 |
(1) |
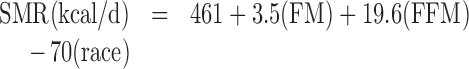 |
(2) |
From these equations, predicted values for 24h-EE and SMR at baseline and after OF were calculated using measured FFM and FM. The differences between actual and predicted values of EE determined the “residual,” that is, an indicator of the individual EE deviation from the reference population at both baseline and post-OF. A metabolic adaptation was considered present if the positive difference between post-OF and baseline residuals was significantly different than zero (40). As described above, PAL was calculated as TDEE/SMR thus eliminating the impact of body weight because both TDEE and SMR are directly proportional to body weight. Energy expended in physical activity (EEPA) was also calculated relative to body weight at baseline and post-OF as EEPA = [(NREE − TEF)/BW] where NREE is nonresting energy expenditure (TDEE − SMR), TEF is the thermic effect of food (estimated to be 10% of caloric intake), and BW is body weight.
Results
OF intervention
Energy requirements at baseline in the 35 subjects averaged 3075 ± 400 kcal/d, and the mean OF level at baseline was 4233 ± 464 kcal/d, or 1158 ± 205 kcal/d over baseline requirements, amounting to 64,848 kcal surplus over the 56 d of intervention. Average BMI at baseline was 25.6 ± 2.3 (range: 22.5–31.4); 2 subjects were obese (BMI 30.7 and 31.4). As previously reported (23), subjects gained an average of 7.5 ± 1.9 kg (range: 2.3–10.7 kg) over 8 wk, or 9.5 ± 2.7% of initial weight, over half of which was fat (4.2 ± 1.4 kg). After OF, average BMI shifted to the overweight category (28.0 ± 2.6) with a range of 24.1–34.7, and 7 participants were categorized as obese. As weight gain progressed, daily positive energy balance declined.
EE and physical activity
SMR, 24h-EE, and TDEE all increased with OF (Table 1). After adjusting for changes in body weight and body composition (increased FFM and FM), SMR was significantly higher than predicted but only by 43 ± 123 kcal/d (P = 0.05), which equated to ∼37% of the absolute increase in SMR. The increase in sedentary 24h-EE was mostly related to the increase in body mass; a marginal trending increase of 23 ± 139 kcal/d (P = 0.34) remained unexplained by weight gain, representing 17% of the total rise in 24h-EE (Figure 1). The weight-independent changes in SMR ranged from −211 to 396 kcal/d among subjects, whereas those in 24h-EE ranged from −220 to 321 kcal/d, indicating a wide variability in the individual EE response to OF. TDEE measured by DLW increased by 280 ± 495 kcal/d and was attributable mostly to the gain in body mass. There was also no change in PAL measured as TDEE/SMR (1.78 ± 0.25 compared with 1.83 ± 0.29; P = 0.44) or the average number of steps taken per day as recorded by activity monitors (9601 ± 3901 compared with 9081 ± 3193 steps/d, baseline and post-OF, respectively; P = 0.79). EEPA averaged 13.0 ± 5.1 kcal/kg/d at baseline and did not change with OF (12.4 ± 5.9 kcal/kg/d; P = 0.60). Together, these data suggest that PAL did not change with OF.
TABLE 1.
Body composition, hormone profile, and energy expenditure before and after overfeeding1
| Before overfeeding | After overfeeding | P value | |
|---|---|---|---|
| Anthropometrics | |||
| BMI, kg/m2 | 25.6 (22.5–31.4) | 28.0 (24.1–34.7) | <0.001 |
| Weight, kg | 79.6 (58.2–100) | 87.1 (65.6–105.8) | <0.001 |
| Fat mass, kg | 17.2 (7.9–25.8) | 21.3 (11.3–30.5) | <0.001 |
| Fat-free mass, kg | 62.4 (38.9–77.8) | 65.5 (40.6–81.2) | <0.001 |
| Energy expenditure (EE) | |||
| Sleeping metabolic rate, kcal/d | 1714 ± 201 | 1828 ± 252 | <0.001 |
| 24h-EE, kcal/d | 2128 ± 250 | 2254 ± 305 | <0.001 |
| TDEE, kcal/d | 3054 ± 567 | 3335 ± 673 | 0.002 |
| Physical activity EE, kcal/d | 1039 ± 442 | 1085 ± 554 | 0.68 |
| Hormone profile | |||
| Triiodothyronine, ng/dL | 99.7 (80.5–138) | 103.5 (76.2–139) | 0.02 |
| Thyroxine, µg/dL | 6.4 (3.4–9.4) | 6.4 (2.7–7.9) | 0.94 |
| Thyroid-stimulating hormone, µIU/mL | 1.8 (0.4–5.0) | 1.9 (0.5–4.4) | 0.41 |
| Reverse triiodothyronine, µg/mL | 0.21 (0.13–0.34) | 0.20 (0.08–0.38) | 0.22 |
| Leptin, ng/mL | 12.2 (1.1–55.6) | 18.4 (1.8–67.9) | <0.001 |
| Urine epinephrine, µg/24 h | 19 (9.4–68) | 18 (9.4–63) | 0.55 |
| Urine norepinephrine, µg/24 h | 97 (27–230) | 112 (36–253) | 0.12 |
Data are presented as mean (range) or ±SD; n = 35 (29 M, 6 F). TDEE, total daily energy expenditure; 24h-EE, 24-h energy expenditure.
FIGURE 1.

“Residuals” in energy expenditure (EE) at baseline and after overfeeding (OF). “Residuals” are calculated as actual EE measurements minus predicted EE measurements, calculated using a previously published dataset (39). (A) Predicted sleeping metabolic rate (SMR, kcal/d) was calculated at baseline and post-OF in 34 adults (28 M, 6 F) as follows: Predicted SMR = 461 + (3.5 × FM) + (19.6 × FFM) − (70*race, where 1=African-American, 0=Caucasian), using measured values of fat mass (FM) and fat-free mass (FFM) at baseline and post-OF. After OF, mean SMR was significantly higher than predicted by 43 ± 123 kcal/d (*P = 0.05) by paired t test. (B) Predicted 24-h sedentary EE (24h-EE, kcal/d) was calculated at baseline and post-OF in all 35 adults (29 M, 6 F) as follows: Predicted 24h-EE = 546 + (5.2 × FM) + (26.2 × FFM) − (2.3 × age) − (96*race, where 1=African-American, 0=Caucasian), using measured values of FM and FFM at baseline and post-OF. Predicted 24h-EE after OF was not significantly different than measured 24h-EE (23 ± 139, P = 0.34) by paired t test, indicating the 24h-EE increased as expected for the increase in body mass.
Circulating hormones
OF induced an increase in circulating triiodothyronine (T3), from 99.7 ± 13.3 to 103.5 ± 15.1 ng/dL (P = 0.02); however, there was no change in thyroxine, thyroid-stimulating hormone, or reverse T3 (Table 1). As expected, leptin concentration rose, from 12.2 ± 14.2 to 18.4 ± 17.7 ng/mL (P < 0.001), in proportion to the increase in FM. Urinary excretion of epinephrine did not change and although excretion of norepinephrine rose from 97 ± 42 to 112 ± 51 µg/24 h, the change was nonsignificant (P = 0.12). There were no direct associations between changes in hormone concentrations and adaptations in EE.
Skeletal muscle mitochondrial efficiency
In a subset of 23 subjects, ATP demand of the resting vastus lateralis did not change with OF (6.2 ± 1.2 to 6.1 ± 1.3 µM ATP/s, baseline and post-OF respectively; P = 0.79) (Figure 2). There was a small but trending decrease in oxygen consumption in resting muscle (2.3 ± 0.6 to 2.0 ± 0.5 µM O2/s; P = 0.09). Together, the magnetic resonance and optical spectrometry data suggested a 13% trend increase in mitochondrial coupling with OF (P/O ratio: 1.35 ± 0.34 to 1.54 ± 0.50; P = 0.12). There were no associations between changes in mitochondrial measures and changes in EE. Changes in EE in this subgroup were not significantly different from the whole sample.
FIGURE 2.
In situ mitochondrial energetics of the vastus lateralis at rest before and after overfeeding. (A) ATP use measured by 31P-magnetic resonance spectroscopy did not change after overfeeding (OF) in 23 adults (19 M, 4 F) (6.2 ± 1.2 vs. 6.1 ± 1.3 µM ATP/s; P = 0.79). (B) There was a trend for oxygen use to decrease after OF in 21 adults (18 M, 3 F) (2.3 ± 0.6 vs. 2.0 ± 0.5 µM O2/s; †P = 0.09) by paired t test. (C) Mitochondrial coupling efficiency (P/O ratio) did not change significantly after OF in 20 adults (18 M, 2 F) (1.35 ± 0.34 vs. 1.54 ± 0.50; P = 0.12) by paired t test.
EE and weight gain
Expectedly, subjects who gained more weight during OF had larger absolute increases in SMR (r = 0.37; P = 0.03) and 24h-EE (r = 0.41; P = 0.01). However, weight gain was not associated with the body-mass–independent change in SMR or 24h-EE (i.e., the metabolic adaptation). Surprisingly, higher body-mass–adjusted 24h-EE at baseline and after OF was associated with larger relative weight gain during OF (baseline: r = 0.42; P = 0.01; and post-OF: r = 0.41; P = 0.02). This latter observation indicates that subjects with the largest metabolic adaptation gain the most weight under OF conditions.
Weight loss at 6-mo follow-up
Average baseline weight (79.8 ± 10.8 kg compared with 78.0 ± 4.4 kg; P = 0.71) and total weight gain during OF (7.3 ± 19 kg compared with 8.7 ± 1.9 kg; P = 0.15) were not different in the 30 participants (25 male, 5 female) who returned for follow-up 6 mo later compared to the 5 subjects who did not return, respectively. Average weight was 82.9 ± 11.6 kg, representing 43 ± 63% retention of the experimentally induced gain. Mean weight loss was 4.3 ± 3.5 kg and consisted of 2.4 ± 1.8 kg of FFM and 2.0 ± 2.7 of FM. To explore the impact of EE responses to OF on post-OF weight regulation, we investigated whether residuals of EE at baseline or after OF were associated with weight loss 6 mo later. Specifically, subjects who had a lower-than-predicted SMR prior to OF lost less weight overall during the follow-up (r = 0.35; P = 0.05) and retained a significantly greater portion of the fat gained during OF (r = −0.38; P = 0.04; Figure 3A). Conversely, a greater-than-predicted 24h-EE after OF was associated with significantly greater loss of FM 6 mo later (r = 0.45; P = 0.01; Figure 3B). These associations were independent of FM at baseline and the amount of fat gained during OF.
FIGURE 3.

Relation between deviations from predicted EE during overfeeding (OF) and body fat changes 6 mo later. (A) A lower-than-predicted sleeping metabolic rate (SMR) at baseline, suggestive of a more “thrifty” metabolic phenotype, was associated with greater retention of the fat mass gained during OF, after 6 mo of follow-up in 30 adults (25 M, 5 F) using Pearson correlation coefficients. (B) A greater-than-predicted sedentary 24-h energy expenditure (24h-EE) after OF, suggestive of a more “spendthrift” phenotype, was associated with significantly greater fat loss after 6 mo of follow-up in 30 adults (25 M, 5 F) using Pearson correlation coefficients. Shaded symbols represent the males and open symbols represent the females. FM, fat mass.
Discussion
To our knowledge, this study is one of the largest and best-controlled OF studies conducted to date. The purpose of this analysis was to determine whether EE increases more than predicted by the increase in body mass (FFM and FM) during OF and if so, minimizes weight and fat gain. The second exploratory objective was to determine whether the variability in metabolic adaptation associates with the amount of weight lost 6 mo after returning to “normal life.” We found that SMR, which most closely resembles true basal metabolic rate, increased significantly more than expected for the increase in FM and FFM, but only by a marginal amount of 43 kcal/d, probably not sufficient to attenuate weight gain. Even more importantly, there was no evidence of a metabolic adaptation in sedentary 24h-EE measured in a metabolic chamber, including not only SMR but also diet-induced thermogenesis and spontaneous physical activity. Together, the data indicate that the unexplained increase in EE (metabolic adaptation) triggered by prolonged OF is rather small and unlikely to confer strong resistance to weight gain similar to the adaptation observed in response to caloric restriction resisting further weight loss. However, the interindividual variability in the measured metabolic adaptation to OF was associated with the individual changes in FM 6 mo after returning to normal life, such that individuals with a lower-than-expected EE retained more of the fat gained, and individuals with a greater-than-expected EE lost more fat. These results suggest that the magnitude of EE increase in response to controlled OF modulates the return toward usual baseline weight after the OF intervention ends.
Although statistically significant, the 43 kcal/d unexplained increase in SMR is of doubtful physiological relevance in limiting weight gain during overfeeding. The present study confirms an earlier OF study by Ravussin et al. (11) in which all the excess calories were accounted for by weight gain and obligatory increases in energy expenditure without inducing "luxuskonsumption." However, it is important to note the interindividual variability in the metabolic adaptation in SMR, ranging from −211 to 396 kcal/d. In comparison, the adaptation in basal metabolism observed during caloric restriction averages around 250 to >300 kcal/d depending on the degree of energy deficit (1, 2). From an etiological perspective, it is reasonable to expect that human metabolism evolved to protect against calorie deprivation, not so much against calorie excess. In times of food scarcity, the ability to reduce metabolic rate and conserve energy would offer considerable advantage in surviving a subsequent famine. A similar metabolic adaptation during times of energy surplus, or feast, would also offer some survival advantage, that is, keeping metabolic rate low relative to the energy excess would promote fat storage, providing an alternative source of energy during the next famine. There would be no conceivable advantage to futilely burning excess energy under harsh environmental conditions. However, in today's environment, a “thrifty metabolism” and the absence of metabolic adaptation to energy surplus could facilitate the deposition of fat, progressively leading to obesity and type 2 diabetes (41).
The concept of metabolic adaptation to OF arose when investigators began to vary their food intake over a wide range yet showed little change in body weight. They hypothesized the presence of an adaptive component in EE working to dissipate excess energy. In one such classic experiment, Neumann (10) varied his caloric intake so that in 3 major periods during the year he was on 1766 calories, 2199 calories, and 2403 calories per day. On such sustained different intake levels, his body weight remained relatively stable. However, the range of caloric intake varied only by about 15% and would not be considered substantial OF in modern experiments. In a later self-study, Gulick (42) varied his intake from 1974 to 4113 kcal/d over the course of 370 d and demonstrated that he could maintain his body weight at calorie levels up to 3500 kcal/d, but not at the extremes of excess. These experiments fueled the notion that both metabolic rate and body weight change very little until extremes of calorie excess and sufficient time (i.e., >2 wk) are present. This was evident in a landmark study among Vermont prisoners and healthy volunteers (43). Lean men were overfed for a 40-wk period with 6000 to 8000 kcal ingested daily. Despite the massive OF and reduction of their physical activity to a minimum, volunteers gained only 6 kg. Months later, they required 5750 kcal/d to maintain the achieved body weight. Together, these classic experiments pointed to some feature of metabolism that must be adequately flexible and responsive to caloric load so as to dissipate the excess energy as heat. Unfortunately, limited methodologies at that time prevented the identification of this metabolic component.
Recent OF studies have carefully controlled energy intake and output, and have more thoroughly investigated different components of EE. Conclusions on whether metabolic adaptation exists vary, with some reports finding that EE increased as expected (i.e., EE rose to meet obligatory demands) (21, 44–48) whereas others found that the increase in EE could not be accounted for by the increase in body weight, the thermic effect of the extra food, or physical activity (19, 49–52). In one study, the increase in activity thermogenesis not due to exercise (i.e., fidgeting, posture, activities of daily life) accounted for a large part of the variability in fat gain (20). Notably, across all studies, there was marked intersubject variability in how EE responded to OF, which makes it difficult to interpret the meaning of average values. The relatively small number of participants usually enrolled in OF studies also makes it difficult to identify spendthrift compared with thrifty phenotypes.
To reveal the spendthrift and thrifty phenotypes, some studies have acutely overfed (100% of energy requirements) and fasted volunteers for 24–48 h while in a metabolic chamber (13, 53). A greater drop in EE during fasting was consistently associated with a lesser increase in EE during OF (thrifty phenotype) and vice versa (spendthrift phenotype). Subjects with a thrifty metabolism gained significantly more weight during a 6-mo follow-up period (13); likewise, those with a spendthrift metabolism lost significantly more weight during 6 wk of controlled 50% caloric restriction (12). In the present study, we found that subjects who displayed a lower-than-predicted SMR at baseline lost significantly less of the fat that was gained during OF 6 months later. Conversely, those who had a greater-than-predicted 24h-EE after OF lost significantly more fat during the follow-up period independently of total weight or fat gain with OF. Schlögl et al. (13) reported that a smaller EE response to caloric excess was associated with future weight gain, especially during low-protein OF.
To our knowledge, our study is one of the first to investigate potential mechanisms underlying the EE changes to OF, particularly changes in skeletal muscle mitochondrial efficiency. We observed no significant changes in ATP demand or P/O ratio and no associations between the mitochondrial measures and weight gain during OF or weight/fat loss after the follow-up period (data not shown). Seyssel et al. (54) reported increased mitochondrial respiration, particularly state 4 respiration, with 56 d of OF leading to a decrease in coupling efficiency. Weight gain was markedly less (average 2.6 kg) than in our study and the OF diet differed substantially, possibly contributing to the contrasting findings. Furthermore, it is questionable whether in vitro mitochondrial respiration accurately represents respiration in intact skeletal muscle (55), further adding to the confusion surrounding the involvement of mitochondrial efficiency in OF- or underfeeding-induced metabolic adaptations.
Hormonal shifts have also been implicated in EE changes, particularly during calorie restriction (56), and the degree of metabolic adaptation has been found to be associated with the decline in leptin concentrations (57). Here, we found that although leptin and T3 increased with OF, there were no direct associations between these hormonal changes and metabolic adaptation.
In summary, although there was an increase in metabolic rate of 43 kcal/d unexplained by weight gain with OF, the clinical relevance of this increase is doubtful, and we conclude that metabolic adaptation during surplus does not occur such as in the metabolic adaptation to caloric restriction. These findings are supported by the lack of change in physiological processes thought to underlie metabolic adaptation, including changes in skeletal muscle mitochondrial efficiency and/or circulating hormones. However, the individual response in EE to caloric excess varied widely but did not protect against weight gain during OF. As a result, we uncovered that a more “thrifty” or “spendthrift” phenotype at the time of OF was associated with fat loss 6 mo after ending the controlled intervention. In support of other recent findings, our results suggest that individual metabolic phenotypes (thrifty compared with spendthrift), although not really associated with the magnitude of weight gain during OF, can still play a role in long-term body weight regulation.
Supplementary Material
Acknowledgments
The authors’ contributions were as follows—DLJ: project design, conducted research, analyzed data, wrote paper; KLM: analyzed data, wrote paper; KEC: project design, conducted research, wrote paper; SRS: project design, conducted research, wrote paper; ER: project design, conducted research, wrote paper, had primary responsibility for final content; and all authors: read and approved the final manuscript.
Author disclosures: DLJ, KLM, KEC, SRS, and ER, no conflicts of interest.
Data described in the article, codebook, and analytic code will be made available upon request.
Notes
This work was supported by a National Institute of Diabetes and Digestive and Kidney Diseases grant (R01DK060412 to ER). This work was also supported by a career development grant from the National Institute of Diabetes and Digestive and Kidney Diseases (K01DK89005 to DLJ), the Ruth L. Kirschstein National Research Service Award Institutional Research Training Grant (T32DK064584 to KLM), and a Nutrition Obesity Research Center (NORC) Center Grant (P30DK72476 to ER).
Supplemental Table 1 and Supplemental Figure 1 are available from the “Supplementary data” link in the online posting of the article and from the same link in the online table of contents at https://academic.oup.com/ajcn/.
Abbreviations used: DLW, doubly labeled water; EE, energy expenditure; EEPA, energy expended in physical activity; FFM, fat-free mass; FM, fat mass; MRS, magnetic resonance spectroscopy; OF, overfeeding; PAL, physical activity level; PCr, phosphocreatine; PRBC, Pennington Biomedical Research Center; SMR, sleeping metabolic rate; T3, triiodothyronine; TDEE, total daily energy expenditure; TEF, thermic effect of food; 24h-EE, 24-h sedentary energy expenditure.
References
- 1. Leibel RL, Rosenbaum M, Hirsch J. Changes in energy expenditure resulting from altered body weight. N Engl J Med. 1995;332:621–8. [DOI] [PubMed] [Google Scholar]
- 2. Johannsen DL, Knuth ND, Huizenga R, Rood JC, Ravussin E, Hall KD. Metabolic slowing with massive weight loss despite preservation of fat-free mass. J Clin Endocrinol Metab. 2012;97:2489–96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Martin CK, Heilbronn LK, De Jonge L, DeLany JP, Volaufova J, Anton SD, Redman LM, Smith SR, Ravussin E. Effect of calorie restriction on resting metabolic rate and spontaneous physical activity. Obesity. 2007;15:2964–73. [DOI] [PubMed] [Google Scholar]
- 4. Sumithran P, Proietto J. The defence of body weight: a physiological basis for weight regain after weight loss. Clin Sci. 2013;124:231–41. [DOI] [PubMed] [Google Scholar]
- 5. Tremblay A, Chaput JP. Short communication: Adaptive reduction in thermogenesis and resistance to lose fat in obese men. Br J Nutr. 2009;102:488–92. [DOI] [PubMed] [Google Scholar]
- 6. MacLean PS, Bergouignan A, Cornier M-A, Jackman MR. Biology's response to dieting: the impetus for weight regain. AJP Regul Integr Comp Physiol. 2011;301:R581–600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Keys A, Brozek J, Henschel A, Mickelson O, Taylor HL. The biology of human starvation. Minneapolis: University of Minnesota Press; 1950. [Google Scholar]
- 8. Redman LM, Heilbronn LK, Martin CK, de Jonge L, Williamson DA, Delany JP, Ravussin E. Metabolic and behavioral compensations in response to caloric restriction: Implications for the maintenance of weight loss. PLoS One. 2009;4:e4377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Westerterp KR. Metabolic adaptations to over- and underfeeding—Still a matter of debate?. Eur J Clin Nutr. 2013;67(5):443–5. [DOI] [PubMed] [Google Scholar]
- 10. Neumann RO. Experimental contributions to the science of human daily nutritional needs with particular regard to the necessary amount of protein (author's experiments). 1902 Obes Res. 1995;3:496–500. [DOI] [PubMed] [Google Scholar]
- 11. Ravussin E, Schutz Y, Acheson KJ, Dusmet M, Bourquin L, Jéquier E. Short-term, mixed-diet overfeeding in man: no evidence for “luxuskonsumption”. Am J Physiol. 1985;249(5 Pt 1):E470–7. [DOI] [PubMed] [Google Scholar]
- 12. Reinhardt M, Thearle MS, Ibrahim M, Hohenadel MG, Bogardus C, Krakoff J, Votruba SB. A human thrifty phenotype associated with less weight loss during caloric restriction. Diabetes. 2015;64:2859–67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Schlögl M, Piaggi P, Pannacciuli N, Bonfiglio SM, Krakoff J, Thearle MS. Energy expenditure responses to fasting and overfeeding identify phenotypes associated with weight change. Diabetes. 2015;64:3680–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Miller DS, Mumford P. Gluttony. 1. An experimental study of overeating low- or high-protein diets. Am J Clin Nutr. 1967;20:1212–22. [DOI] [PubMed] [Google Scholar]
- 15. Miller DS, Mumford P, Stock MJ. Gluttony. 2. Thermogenesis in overeating man. Am J Clin Nutr. 1967;20:1223–9. [DOI] [PubMed] [Google Scholar]
- 16. Sims EAH, Danforth E, Horton ES, Bray GA, Glennon JA, Salans LB. Endocrine and metabolic effects of experimental obesity in man. Proceedings of the 1972 Laurentian Hormone Conference Recent Prog Horm Res. 1973;29:457–96. [DOI] [PubMed] [Google Scholar]
- 17. Joosen AMCP, Bakker AHF, Westerterp KR. Metabolic efficiency and energy expenditure during short-term overfeeding. Physiol Behav. 2005;85:593–7. [DOI] [PubMed] [Google Scholar]
- 18. Ravussin E, Lillioja S, Anderson TE, Christin L, Bogardus C. Determinants of 24-hour energy expenditure in man. Methods and results using a respiratory chamber. J Clin Invest. 1986;78:1568–78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Bouchard C, Tremblay A, Després J-P, Nadeau A, Lupien PJ, Thériault G, Dussault J, Moorjani S, Pinault S, Fournier G. The response to long-term overfeeding in identical twins. N Engl J Med. 1990;322:1477–82. [DOI] [PubMed] [Google Scholar]
- 20. Levine JA, Eberhardt NL, Jensen MD. Role of nonexercise activity thermogenesis in resistance to fat gain in humans. Science. 1999;283:212–4. [DOI] [PubMed] [Google Scholar]
- 21. Diaz EO, Prentice AM, Goldberg GR, Murgatroyd PR, Coward WA. Metabolic response to experimental overfeeding in lean and overweight healthy volunteers. Am J Clin Nutr. 1992;56:641–55. [DOI] [PubMed] [Google Scholar]
- 22. Roberts SB, Fuss P, Heyman MB, Dallal GE, Young VR. Effects of age on energy expenditure and substrate oxidation during experimental underfeeding in healthy men. J Gerontol A Biol Sci Med Sci. 1996;51:B148–57. [DOI] [PubMed] [Google Scholar]
- 23. Johannsen DL, Tchoukalova Y, Tam CS, Covington JD, Xie W, Schwarz JM, Bajpeyi S, Ravussin E. Effect of 8 weeks of overfeeding on ectopic fat deposition and insulin sensitivity: Testing the “adipose tissue expandability” hypothesis. Diabetes Care. 2014;37:2789–97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Schoeller DA. Measurement of energy expenditure in free-living humans by using doubly labeled water. J Nutr. 1988;118:1278–89. [DOI] [PubMed] [Google Scholar]
- 25. Racette SB, Schoeller DA, Luke AH, Shay K, Hnilicka J, Kushner RF. Relative dilution spaces of 2H- and 18O-labeled water in humans. Am J Physiol. 1994;267:E585–90. [DOI] [PubMed] [Google Scholar]
- 26. Bray GA, Smith SR, De Jonge L, Xie H, Rood J, Martin CK, Most M, Brock C, Mancuso S, Redman LM. Effect of dietary protein content on weight gain, energy expenditure, and body composition during overeating: A randomized controlled trial.J Am Med Assoc. 2012;307:47–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Nguyen T, de Longe L, Smith SR, Bray GA. Chamber for indirect calorimetry with accurate measurement and time discrimination of metabolic plateaus of over 20 min. Med Biol Eng Comput. 2003;41:572–8. [DOI] [PubMed] [Google Scholar]
- 28. Acheson KJ, Schutz Y, Bessard T, Flatt JP, Jéquier E. Carbohydrate metabolism and de novo lipogenesis in human obesity. Am J Clin Nutr. 1987;45:78–85. [DOI] [PubMed] [Google Scholar]
- 29. Johannsen DL, Calabro MA, Stewart J, Franke W, Rood JC, Welk GJ. Accuracy of armband monitors for measuring daily energy expenditure in healthy adults. Med Sci Sports Exerc. 2010;42:2134–40. [DOI] [PubMed] [Google Scholar]
- 30. Amara CE, Marcinek DJ, Shankland EG, Schenkman KA, Arakaki LSL, Conley KE. Mitochondrial function in vivo: Spectroscopy provides window on cellular energetics. Methods. 2008;46:312–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Conley KE, Amara CE, Bajpeyi S, Costford SR, Murray K, Jubrias SA, Arakaki L, Marcinek DJ, Smith SR. Higher mitochondrial respiration and uncoupling with reduced electron transport chain content in vivo in muscle of sedentary versus active subjects. J Clin Endocrinol Metab. 2013;98:129–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Conley KE, Blei ML, Richards TL, Kushmerick MJ, Jubrias SA. Activation of glycolysis in human muscle in vivo. Am J Physiol. 1997;273:C306–15. [DOI] [PubMed] [Google Scholar]
- 33. Amara CE, Shankland EG, Jubrias SA, Marcinek DJ, Kushmerick MJ, Conley KE. Mild mitochondrial uncoupling impacts cellular aging in human muscles in vivo. Proc Natl Acad Sci U S A. 2007;104:1057–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Naressi A, Couturier C, Devos JM, Janssen M, Mangeat C, De Beer R, Graveron-Demilly D. Java-based graphical user interface for the MRUI quantitation package. MAGMA. 2001;12:141–52. [DOI] [PubMed] [Google Scholar]
- 35. Blei ML, Conley KE, Odderson IB, Esselman PC, Kushmerick MJ. Individual variation in contractile cost and recovery in a human skeletal muscle. Proc Natl Acad Sci U S A. 1993;90:7396–400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Marcinek DJ, Ciesielski WA, Conley KE, Schenkman KA. Oxygen regulation and limitation to cellular respiration in mouse skeletal muscle in vivo. Am J Physiol Heart Circ Physiol. 2003;285:H1900–8. [DOI] [PubMed] [Google Scholar]
- 37. Marcinek DJ, Schenkman KA, Ciesielski WA, Conley KE. Mitochondrial coupling in vivo in mouse skeletal muscle. Am J Physiol Cell Physiol. 2004;286:C457–63. [DOI] [PubMed] [Google Scholar]
- 38. Sparks LM, Redman LM, Conley KE, Harper ME, Yi F, Hodges A, Eroshkin A, Costford SR, Gabriel ME, Shook C et al.. Effects of 12 months of caloric restriction on muscle mitochondrial function in healthy individuals. J Clin Endocrinol Metab. 2017;102:111–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Lam YY, Redman LM, Smith SR, Bray GA, Greenway FL, Johannsen D, Ravussin E. Determinants of sedentary 24-h energy expenditure: Equations for energy prescription and adjustment in a respiratory chamber. Am J Clin Nutr. 2014;99:834–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Galgani JE, Santos JL. Insights about weight loss-induced metabolic adaptation. Obesity. 2016;24:277–8. [DOI] [PubMed] [Google Scholar]
- 41. Neel JV. Diabetes mellitus: A “thrifty” genotype rendered detrimental by “progress”?. Am J Hum Genet. 1962;14:353–62. [PMC free article] [PubMed] [Google Scholar]
- 42. Gulick A. A study of weight regulation in the adult human body during overnutrition. Obes Res. 1995;3:501–12. [DOI] [PubMed] [Google Scholar]
- 43. Sims EAH. Experimental obesity, dietary-induced thermogenesis, and their clinical implications. Clin Endocrinol Metab. 1976;5:377–95. [DOI] [PubMed] [Google Scholar]
- 44. Forbes GB, Brown MR, Welle SL, Lipinski BA. Deliberate overfeeding in women and men: energy cost and composition of the weight gain. Br J Nutr. 1986;56:1–9. [DOI] [PubMed] [Google Scholar]
- 45. Horton TJ, Drougas H, Brachey A, Reed GW, Peters JC, Hill JO. Fat and carbohydrate overfeeding in humans: Different effects on energy storage. Am J Clin Nutr. 1995;62:19–29. [DOI] [PubMed] [Google Scholar]
- 46. Jebb SA, Prentice AM, Goldberg GR, Murgatroyd PR, Black AE, Coward WA. Changes in macronutrient balance during over- and underfeeding assessed by 12-d continuous whole-body calorimetry. Am J Clin Nutr. 1996;64:259–66. [DOI] [PubMed] [Google Scholar]
- 47. Ravussin E, Burnand B, Schutz Y, Jequier E. Energy expenditure before and during energy restriction in obese patients. Am J Clin Nutr. 1985;41:753–9. [DOI] [PubMed] [Google Scholar]
- 48. Roberts S, Young VR, Fuss P, Fiatarone MA, Richard B, Rasmussen H, Wagner D, Joseph L, Holehouse E, Evans WJ. Energy expenditure and subsequent nutrient intakes in overfed young men. Am J Physiol Regul Integr Comp Physiol. 1990;259:R461–9. [DOI] [PubMed] [Google Scholar]
- 49. Dallosso HM, James WPT. Whole-body calorimetry studies in adult men. 1. The effect of fat over-feeding on 24 h energy expenditure. Br J Nutr. 1984;52:49–64. [DOI] [PubMed] [Google Scholar]
- 50. Norgan NG, Durnin JVGA. The effect of 6 weeks of overfeeding on the body weight, body composition, and energy metabolism of young men. Am J Clin Nutr. 1980;33:978–88. [DOI] [PubMed] [Google Scholar]
- 51. Pasquet P, Brigant L, Froment A, Koppert GA, Bard D, De Garine I, Apfelbaum M. Massive overfeeding and energy balance in men: The Guru Walla model. Am J Clin Nutr. 1992;56:483–90. [DOI] [PubMed] [Google Scholar]
- 52. Webb P, Annis JF. Adaptation to overeating in lean and overweight men and women. Hum Nutr Clin Nutr. 1983;37:117–31. [PubMed] [Google Scholar]
- 53. Weyer C, Vozarova B, Ravussin E, Tataranni PA. Changes in energy metabolism in response to 48 h of overfeeding and fasting in Caucasians and Pima Indians. Int J Obes. 2001;25:593–600. [DOI] [PubMed] [Google Scholar]
- 54. Seyssel K, Alligier M, Meugnier E, Chanseaume E, Loizon E, Canto C, Disse E, Lambert-Porcheron S, Brozek J, Blond E et al.. Regulation of energy metabolism and mitochondrial function in skeletal muscle during lipid overfeeding in healthy men. J Clin Endocrinol Metab. 2014;99:E1254–62. [DOI] [PubMed] [Google Scholar]
- 55. Korzeniewski B. “Idealized” state 4 and state 3 in mitochondria vs. rest and work in skeletal muscle. PLoS One. 2015;10:e0117145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Rosenbaum M, Leibel RL. Adaptive thermogenesis in humans. Int J Obes. 2010;34(Suppl 1):S47–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Knuth ND, Johannsen DL, Tamboli RA, Marks-Shulman PA, Huizenga R, Chen KY, Abumrad NN, Ravussin E, Hall KD. Metabolic adaptation following massive weight loss is related to the degree of energy imbalance and changes in circulating leptin. Obesity. 2014;22: 2563–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.



