Abstract
Background
Transmission of Salmonella in veterinary hospitals is typically associated with environmental contamination. Links between isolates recovered from hospitalized large animals and environment suggest animals as the likely source. Therefore, understanding factors influencing shedding is key in control. Shedding in hospitalized animals has been investigated, but many studies focused on subsets of animals limiting generalizability.
Objective
(1) Investigate factors associated with fecal shedding of Salmonella among hospitalized large animals at a veterinary hospital. (2) Compare results obtained using 2 comparison groups for risk factor analysis—large animals with high confidence in negative shedding status and those with potential for misclassification of shedding status.
Animals
Large animals admitted from March 2002 through December 2012.
Methods
A case‐control study was conducted among all hospitalized large animals that were routinely cultured as part of infection control efforts. Animal and hospital factors were evaluated. Data on factors of interest were collected retrospectively from electronic medical records. Multivariable conditional logistic regression was used to evaluate associations between animal factors and fecal shedding of Salmonella enterica.
Results
During the study period, 5.9% (648/11 061) of hospitalized large animals were culture positive for Salmonella, with the majority being cattle (72%; 467/648) and horses (22%; 143/648). Although the odds of shedding varied by comparison group, overall, 69.4% of animal shedding could be attributed to systemic illness (population attributable fraction) in this study.
Conclusions and Clinical Importance
Findings of this study inform our understanding of factors affecting Salmonella shedding in hospitalized large animals, thus improving our ability for implementation of evidence‐based control measures.
Keywords: biosecurity, epidemiology, infection control, infectious disease
Abbreviations
- CI
confidence interval
- CSU
Colorado State University
- HAIs
health care‐associated infections
- NWC
New World camelid
- OR
odds ratio
- VTH
Veterinary Teaching Hospital
1. INTRODUCTION
Salmonella enterica is one of the most commonly reported agents associated with health care‐associated infections (HAIs) in veterinary teaching hospitals (VTHs), and can be associated with high case fatality rate and substantial financial cost.1, 2 Characteristically, these outbreaks are accompanied by widespread environmental contamination, which likely contributes to ongoing transmission among animals.2, 3 Environmental isolates can have the same phenotype (serotype and antimicrobial susceptibility) as animal isolates suggesting animals to be a likely source for environmental contamination.4
Studies inconsistently report many factors that might be associated with animal shedding—some reporting an increased shedding risk associated with abdominal surgery,5, 6, 7, 8 diarrhea,9, 10, 11 colic,12 fever,6, 9 abnormal white count,6, 10 nasogastric intubation,10, 12 antimicrobial treatment,7, 12 a change in diet,6, 9 transportation,10 age,7 and breed6; and others reporting no shedding risk associated with abdominal surgery,9, 10, 12, 13 fever,7, 10, 13 abnormal white count,7, 9, 13 nasogastric intubation,6, 9, 13 antimicrobial treatment,9, 10, 11 change in diet,10 transportation,7, 13 or age, sex, and breed.9, 12, 13 Some of these differences can be attributed to previous reports often targeting specific high‐risk subgroups such as horses with colic8, 9, 10 or diarrhea8, 9, 12, 14), or there use of intermittent sampling5, 15 from a limited time period (eg, 2‐24 months)5, 9, 10, 15 Additionally, the majority of these reports concentrate on horses and do not evaluate the entire large animal hospital population.
Although results of these studies can be useful, it is prudent to exercise caution when extrapolating conclusions to the broader hospital population. If the occurrence of epidemic disease in a hospital population is related to endemic rates of fecal shedding, then evaluating the general hospital population over an extended period of time (eg, 10 years) will allow for the investigation of factors that could be employed in routine prevention of HAIs. At the Colorado State University (CSU)–VTH, routine fecal surveillance of hospitalized large animals has been conducted since 2002, as part of long‐term infection control efforts. This presents a unique opportunity to gain a better understanding of factors associated with endemic shedding among the general large animal hospital population without limiting it to targeted sampling of high‐risk subgroups or particular species, and to do so based on standardized sampling and culture techniques that have been employed in an active surveillance program spanning 10 years.
Choosing the most relevant comparison group in a case‐control study can be challenging. This is especially true when studies are conducted to determine factors associated with shedding at times of epidemic disease as most veterinary hospitals do not test fecal samples for Salmonella in all large animals, especially in the absence of disease.16 Many hospital‐based surveillance programs focus on restricted subpopulations such as large animals presenting for colic, diarrhea, or having clinical signs indicative of salmonellosis, which considerably limits the ability to identify truly negative animals.12 Additionally, the identification of truly negative animals is hampered by limitations in Salmonella detection methods, which are generally considered to have imperfect sensitivity necessitating lengthy culture processes (ie, enriched cultures) and testing of multiple samples per animal. Thus, it can take up to 3 days to realize test results for a single sample, and typically 3‐5 fecal cultures are performed and interpreted in parallel to achieve reasonable sensitivity for the testing strategy. Based on routine surveillance at the CSU‐VTH, culture‐positive large animals (eg, horses, cattle, small ruminants, pigs, and New World camelids [NWCs]) are frequently identified on days 4‐7 of hospitalization (having had at least 2 cultures), whereas culture‐negative large animals are more commonly hospitalized less than 3 days (ie, will typically have had only a single negative culture) and greater than 8 days (ie, will have had ≥3 negative cultures).17 Therein lays the difficulty—which group of culture‐negative animals is the better comparison for this type of research, and what effect will this choice have on overall conclusions?
The objectives of this study were to (1) investigate factors associated with fecal shedding of Salmonella among all hospitalized large animals at a VTH, and (2) compare results obtained using 2 different comparison groups of large animals for risk factor analysis—animals with high confidence in negative shedding status, and those with potential for misclassification of shedding status.
2. MATERIALS AND METHODS
2.1. Study overview
A case‐control study was conducted to detect factors associated with S. enterica fecal shedding among the general large animal hospital population at the CSU‐VTH from 2002 to 2012. Fecal samples were routinely collected at admission and throughout hospitalization, and cultured to detect Salmonella. Cases included all Salmonella‐positive large animals (having ≥1 culture‐positive fecal sample). Controls were enrolled in 2 groups—randomly selected hospitalized large animals with ≥1 negative culture (1NEG controls) and randomly selected large animals with ≥3 negative cultures (3NEG controls). Data were derived from long‐term surveillance conducted as part of the Infection Control Program and electronic medical records at the CSU‐VTH. Conditional logistic regression was used to develop 2 multivariable models to determine factors that might be associated with fecal shedding of Salmonella. All data were derived from in‐hospital animal management data and owner consent was obtained before study initiation as part of the hospital admissions process.
2.2. Population and study subjects
The study population included all hospitalized large animals (ie, had at least 1 night of hospitalization) from the species most commonly admitted (equine, bovine, NWC, and small ruminants [caprine and ovine]) to the CSU‐VTH from April 2002 through December 2012 (n = 11 061; Figure 1). The limited numbers of animals of other species was excluded (eg, bison and elk). Cases were defined as large animals that had ≥1 fecal sample obtained during a single hospitalization that was culture positive (n = 648). Controls were randomly selected from all large animals that were culture negative throughout their hospitalization during the study period (n = 10 413). Two control groups, based on common comparison groups reported in the literature, were randomly selected from these culture‐negative animals: (1) hospitalized large animals with ≥1 negative fecal culture (1NEGcontrols; n = 645), and (2) hospitalized large animals with ≥3 negative fecal cultures (3NEG controls; n = 609). Additionally, cases and controls were matched by the month and year of admission. Each animal could only be enrolled once in the study (ie, once enrolled all other visits were disregarded) and each control could only be enrolled in 1 comparison group.
Figure 1.
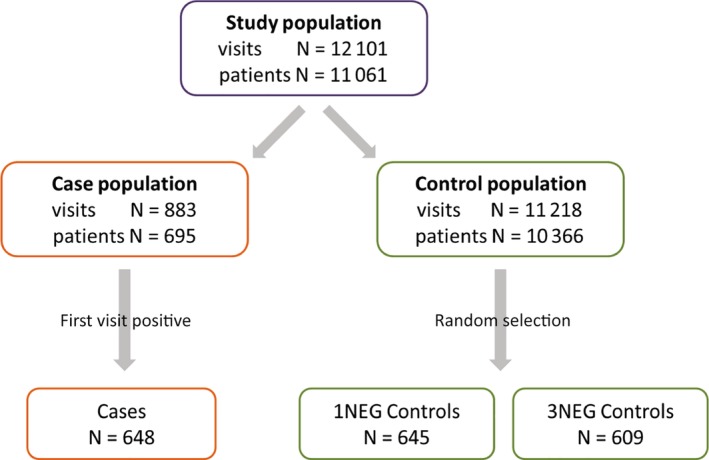
Derivation of study participants. Cases, animals with ≥1 culture‐positive fecal sample; 1NEG controls, randomly selected animals with ≥1 negative culture; 3NEG controls, randomly selected animals with ≥3 negative cultures
2.3. Fecal samples
Per hospital policy (http://csu‐cvmbs.colostate.edu/Documents/biosecurity‐sop.pdf), fecal samples were collected from every large animal at the time of admission and then 3 times weekly from April 2002 to June 2003 or twice weekly from July 2003 to December 2012 as part of ongoing surveillance at the CSU‐VTH. All fecal samples were cultured using standardized enrichment techniques to detect the presence of S. enterica. Briefly, 1 g fecal samples were enriched in 9 mL tetrathionate (TET; Becton Dickinson and Co, Cockeysville, Maryland) broth for 18 hours at 43°C, then streaked for isolation on xylose‐lysine‐tergitol 4 (XLT4; Hardy Diagnostics, Santa Maria, California) agar media and incubated for 18 hours at 43°C. Suspect colonies were subcultured on trypticase soy agar containing 5% sheep blood (TSA; BD Diagnostic Systems, Sparks, Maryland) and incubated 18 hours at 43°C and tested for agglutination using commercial polyvalent and O group‐specific antisera to confirm identification as S. enterica. Serotype determination was performed by the USDA National Veterinary Services Laboratory (NVSL, Ames, Iowa).
2.4. Potential risk factors
Variables considered to be potential risk factors were derived from invoice data (eg, maximum level of care during hospitalization as a marker of disease severity, classes of antimicrobial drug used in treatment) contained within the electronic medical records database, as well as data obtained at the time of fecal sample submission (eg, body system[s] affected, leukopenia, or fever in previous 48 hours). At fecal sample submission, data were recorded by caregivers via a web‐based questionnaire regarding clinical status and treatment history for the 48 hours before sample collection and was contained within the infection control program database. Data was cross‐validated from multiple sources to ensure accuracy.
2.5. Variables related to animal population characteristics
Independent variables representing animal characteristics included species (equine, bovine, NWC, small ruminant [caprine, ovine]), age (<1 year, yearling [1‐2 years], adult [>2 years]), sex (male, female), maximum degree of systemic illness during hospitalization (based on an ordinal rating of healthy to minimal [e.g., minor lameness, arthroscopy, recurrent airway obstruction, reproductive problems, and mares admitted with foals], minor to moderate [e.g., animals with lacerations, recovering fracture repairs, animals ready to go home having recovered from a more severe illness such as colic, left displace abomasum, fever of unknown origin, or mild respiratory infections], or major systemic illness [animals with severe fractures, renal or liver failure, colic, severe pneumonia, pleuritis, colitis/enteritis, or peritonitis]), body system(s) affected (normal [considered to be clinically normal/healthy], musculoskeletal, gastrointestinal, respiratory, renal, hepatic, reproductive, other (note: animals could have more than 1 affected body system), transportation distance to the VTH (≤20 miles, 20‐50 miles, >50 miles), leukopenia (equine WBC < 5000/μL; bovine, NWC, and small ruminant <5500/μL) within 48 hours before fecal sample collection (yes, no, unknown [a CBC was not performed during hospitalization]), fever (equine >101.5°F [38.6°C]; bovine, NWC, and small ruminant >103 °F [39.4°C]) within 48 hours before fecal sample collection (yes/no), diarrhea (defined as loss of formed fecal consistency) within 48 hours before fecal sample collection (yes/no), and reduced feed intake in previous 48 hours (yes/no).
2.6. Variables related to animal management and hospital factors
Independent variables representing animal management factors included route of antimicrobial used within 48 hours before fecal sample collection (none, parenteral, oral, both), received any antimicrobials during hospitalization (yes/no), class of antimicrobial drug administered during hospitalization (aminoglycoside, beta‐lactam, any generation of cephalosporin, florfenicol, fluoroquinolone, macrolide, sulfas, tetracycline), class of gastroprotectant administered during hospitalization (antidiarrheal, H2‐blocker, proton‐pump inhibitor, mucosal protectant), general anesthesia in previous 48 hours (yes/no), maximum level of care during hospitalization (routine care, level 1‐4), and duration of hospitalization. Levels of care were defined as level 1 (eg, no intravenous catheter, awaiting elective surgery or discharge, routine care), level 2 (eg, intravenous catheter, daily treatment, or assessment approximately every 6 hours), level 3 (eg, intravenous catheter, systemically ill or recovering from surgery, daily treatments or assessments approximately every 4 hours), and level 4 (eg, critical care requiring intravenous treatment and daily treatments or assessments approximately every 2 hours or more frequently), as indicated by the medical record.
Independent variables representing hospital factors included stabling location (equine main hospital, equine colic aisle, equine isolation, livestock hospital, calf isolation), service (livestock medicine and surgery, equine medicine, equine surgery, other ancillary services), and hospitalization when within‐hospital horizontal transmission was detected, defined as the identification of at least 2 large animals (not from the same herd or flock) hospitalized within 10 days of each other with S. enterica isolates of the same phenotype (serotype and antimicrobial susceptibility pattern).
2.7. Data analysis
Data were entered in a spreadsheet, validated, and explored using descriptive statistics. Continuous variables were assessed for the assumption of normality in the logit scale; variables not meeting this assumption were categorized based on distributional quartiles or breakpoints with biological relevance. Conditional logistic regression was used to evaluate factors that might be associated with large animals being culture positive for S. enterica (the dependent variable for analyses), while controlling for matching on month and year of admission. All potential risk factors were screened for inclusion in the multivariable model building process using a critical α ≤ .25. The final multivariable model was identified using backwards selection with a critical α ≤ .05 for retention in the model. Confounding was identified by ≥20% change in parameter estimates when previously excluded variables were individually offered back to the multivariable model. When identified, confounding variables were forced into the multivariable models regardless of P‐values. First‐order interaction terms for main effects variables included in final models were also evaluated. Odds ratios (OR) and profile likelihood 95% confidence intervals (CIs) were calculated using the least squares mean estimates. In addition, analyses were stratified by species to assess species influence on parameter estimates (ie, sensitivity analysis). Crude population attributable fractions (PAFs) were estimated using parameter estimates (derived from univariable modeling of 1NEG controls) and population prevalence for variables of interest.18 Pearson residuals and delta‐betas were evaluated to assess model fit—with none indicating a lack‐of‐fit of final multivariable models. All statistical analyses were conducted with SAS v9.3 (SAS Institute, Inc, Cary, North Carolina).
3. RESULTS
Overall Salmonella shedding prevalence among large animals admitted during the study period was 5.9% (648/11 061)—with the highest prevalence occurring in bovids (73.5%; 476/648; Table 1); the majority of which were dairy cattle (98%; including Ayrshire, Brown Swiss, Holstein‐Friesian, and Jersey breeds).
Table 1.
Univariable conditional logistic regression analysis of factors associated with large animal fecal shedding of Salmonella enterica
| Case (N = 648) | Control group 1 (≥1 negative culture) (N = 645) | Control group 2 (≥3 negative cultures) (N = 609) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Variable | N | N | OR | 95% CI | P‐value | N | OR | 95% CI | P‐value | |
| Animal age | <1 year old | 77 | 77 | 0.96 | 0.68‐1.35 | .04 | 58 | 1.23 | 0.85‐1.77 | .01 |
| Yearling | 18 | 37 | 0.48 | 0.03‐0.84 | 38 | 0.45 | 0.25‐0.79 | |||
| Adult | 553 | 531 | Ref | 513 | Ref | |||||
| Sex | Female | 559 | 370 | 4.77 | 3.61‐6.31 | <.001 | 274 | 7.81 | 5.88‐10.39 | <.0001 |
| Male | 89 | 275 | Ref | 335 | Ref | |||||
| Species | Bovine | 476 | 147 | 10.53 | 7.91‐14.01 | <.001 | 25 | 87.82 | 52.63‐146.55 | <.0001 |
| NWC | 23 | 57 | 1.17 | 0.68‐2.00 | 64 | 1.14 | 0.65‐1.98 | |||
| Small ruminant | 6 | 24 | 0.07 | 0.26‐1.73 | 12 | 1.54 | 0.52‐4.56 | |||
| Equine | 143 | 417 | Ref | 508 | Ref | |||||
| Systemic illness | Major | 128 | 83 | 4.85 | 3.32‐7.08 | <.0001 | 158 | 1.67 | 1.16‐2.40 | <.0001 |
| Minor | 432 | 303 | 4.59 | 3.41‐6.18 | 287 | 3.07 | 2.24‐4.19 | |||
| Healthy | 88 | 259 | Ref | 164 | Ref | |||||
| Hospitalization days | ≥15 days | 20 | 6 | 3.69 | 1.47‐9.30 | .001 | 129 | 0.02 | 0.01‐0.03 | <.0001 |
| 8‐14 days | 52 | 29 | 2.10 | 1.27‐3.33 | 229 | 0.09 | 0.06‐0.13 | |||
| 4‐7 days | 157 | 139 | 1.28 | 0.98‐1.67 | 202 | 0.02 | 0.01‐0.04 | |||
| ≤3 days | 419 | 471 | Ref | 49 | Ref | |||||
| Maximum care level during hospitalization | Level 4 | 17 | 10 | 1.55 | 0.69‐3.50 | <.0001 | 33 | 0.25 | 0.13‐0.46 | <.0001 |
| Level 3 | 43 | 52 | 0.69 | 0.45‐1.06 | 91 | 0.20 | 0.14‐0.31 | |||
| Level 2 | 41 | 69 | 0.49 | 0.32‐0.74 | 119 | 0.15 | 0.10‐0.22 | |||
| Level 1 | 32 | 85 | 0.30 | 0.19‐0.46 | 122 | 0.10 | 0.07‐0.16 | |||
| Routine care | 515 | 429 | Ref | 244 | Ref | |||||
| Anesthesia in previous 48 hours | Yes | 131 | 119 | 1.12 | 0.84‐1.49 | .43 | 212 | 0.47 | 0.37‐0.61 | <.0001 |
| No | 517 | 526 | Ref | 397 | Ref | |||||
| Leukopenia in previous 48 hours | Yes | 37 | 18 | 3.47 | 1.91‐6.31 | <.0001 | 63 | 1.79 | 1.14‐2.84 | <.0001 |
| Unknown | 469 | 375 | 2.22 | 1.73‐2.85 | 101 | 15.28 | 11.26‐20.73 | |||
| No | 142 | 252 | Ref | 445 | Ref | |||||
| Diarrhea in previous 48 hours | Yes | 276 | 93 | 4.46 | 3.39‐5.86 | <.0001 | 117 | 3.23 | 2.49‐4.18 | <.0001 |
| No | 372 | 552 | Ref | 492 | Ref | |||||
| Febrile in previous 48 hours | Yes | 173 | 52 | 4.75 | 3.37‐6.69 | <.0001 | 127 | 1.76 | 1.35‐2.31 | <.0001 |
| Unknown | 93 | 59 | 2.26 | 1.57‐3.25 | 1 | 121.75 | 16.82‐881.30 | |||
| No | 382 | 534 | Ref | 481 | Ref | |||||
| Healthy | Yes | 41 | 86 | 0.42 | 0.28‐0.63 | <.0001 | 109 | 0.28 | 0.19‐0.41 | <.0001 |
| No | 607 | 559 | Ref | 500 | Ref | |||||
| Gastrointestinal (GI) disease | Yes | 477 | 244 | 4.81 | 3.77‐6.14 | <.0001 | 207 | 5.74 | 4.45‐7.39 | <.0001 |
| No | 171 | 401 | Ref | 402 | Ref | |||||
| Musculoskeletal disease | Yes | 97 | 213 | 0.35 | 0.27‐0.47 | <.0001 | 345 | 0.14 | 0.11‐0.19 | <.0001 |
| No | 551 | 432 | Ref | 264 | Ref | |||||
| Renal disease | Yes | 9 | 8 | 1.11 | 0.43‐2.87 | .84 | 23 | 0.36 | 0.17‐0.80 | .01 |
| No | 639 | 637 | Ref | 586 | Ref | |||||
| Respiratory disease | Yes | 56 | 33 | 1.75 | 1.12‐2.73 | .01 | yes | 0.78 | 0.54‐1.14 | .20 |
| No | 592 | 612 | Ref | no | Ref | |||||
| Reproductive disease | Yes | 65 | 51 | 1.30 | 0.89‐1.91 | .14 | 33 | 1.99 | 1.28‐3.10 | .002 |
| No | 583 | 594 | Ref | 576 | Ref | |||||
| Disease associated with other body systems | Yes | 68 | 87 | 0.75 | 0.53‐1.05 | .09 | 134 | 0.42 | 0.30‐0.58 | <.0001 |
| No | 580 | 558 | Ref | 475 | Ref | |||||
| GI protectant during hospitalization | Yes | 79 | 62 | 1.31 | 0.92‐1.87 | .01 | 218 | 0.25 | 0.18‐0.33 | <.0001 |
| No | 569 | 583 | Ref | 391 | Ref | |||||
| AMD during hospitalization | Yes | 354 | 321 | 1.22 | 0.98‐1.52 | .08 | 461 | 0.39 | 0.30‐0.50 | <.0001 |
| No | 294 | 324 | Ref | 148 | Ref | |||||
| AMD route of administration | Parenteral | 306 | 260 | 1.30 | 1.04‐1.64 | .004 | 243.00 | 0.67 | 0.51‐0.87 | <.0001 |
| Both | 37 | 31 | 1.30 | 0.79‐2.16 | 173.00 | 0.11 | 0.07‐0.16 | |||
| Oral | 11 | 30 | 0.41 | 0.20‐0.84 | 45.00 | 0.12 | 0.06‐0.24 | |||
| None | 294 | 324 | Ref | 148.00 | Ref | |||||
| Aminoglycoside during hospitalization | Yes | 109 | 146 | 0.69 | 0.52‐0.91 | .01 | 309 | 0.18 | 0.13‐0.23 | <.0001 |
| No | 539 | 499 | Ref | 300 | Ref | |||||
| Beta‐lactam during hospitalization | Yes | 320 | 259 | 1.46 | 1.17‐1.82 | .001 | 395 | 0.54 | 0.43‐0.67 | <.0001 |
| No | 328 | 386 | Ref | 214 | Ref | |||||
| Cephalosporin during hospitalization | Yes | 237 | 182 | 1.46 | 1.16‐1.85 | .002 | 198 | 1.20 | 0.95‐1.51 | .13 |
| No | 411 | 463 | Ref | 411 | Ref | |||||
| Chloramphenicol during hospitalization | Yes | 8 | 9 | 0.86 | 0.33‐2.26 | .76 | 20 | 0.39 | 0.17‐0.89 | .03 |
| No | 640 | 636 | Ref | 589 | Ref | |||||
| Fluoroquinolone during hospitalization | Yes | 9 | 1 | 9.00 | 1.14‐71.04 | .04 | 53 | 0.14 | 0.07‐0.30 | <.0001 |
| No | 639 | 644 | Ref | 556 | Ref | |||||
| Sulfamethoxazole during hospitalizations | Yes | 30 | 48 | 0.61 | 0.38‐0.97 | .04 | 126 | 0.18 | 0.12‐0.27 | <.0001 |
| No | 618 | 597 | Ref | 483 | Ref | |||||
| Tetracycline during hospitalization | Yes | 37 | 44 | 0.82 | 0.52‐1.30 | .39 | 112 | 0.24 | 0.16‐0.36 | <.0001 |
| No | 611 | 601 | Ref | 497 | Ref | |||||
| Transportation distance | ≤20 miles | 471 | 287 | 4.70 | 3.52‐6.29 | <.0001 | 124 | 17.06 | 12.20‐23.85 | <.0001 |
| 20‐50 miles | 95 | 118 | 2.31 | 1.60‐3.34 | 144 | 2.79 | 1.93‐4.04 | |||
| ≥50 miles | 82 | 240 | Ref | 341 | Ref | |||||
| Hospitalized during detected nosocomial transmission | Yes | 247 | 256 | 0.88 | 0.64‐1.19 | .39 | 240 | 0.81 | 0.60‐1.11 | .19 |
| No | 401 | 389 | Ref | 369 | Ref | |||||
Abbreviations: AMD, antimicrobial drug; CI, confidence interval; NWC, New World camelid; OR, odds ratio; Ref, reference.
Among Salmonella‐positive animals, the mean duration of hospitalization until collection of culture‐positive fecal samples was 2 days (median 1, interquartile range [IQR] 1‐3; range 1‐35) with Salmonella detected most commonly in the first fecal sample (median 1; range 1‐9). On average, the maximum number of fecal samples tested for fecal culture‐positive animals was 1.7 (median 1, IQR 1‐2; range 1‐15), for 1NEG controls was 1.3 (median 1, IQR 1‐2; range 1‐3), and for 3NEG controls was 4.4 (median 3, IQR 3‐5; range 3‐25). There was a higher proportion of cattle among fecal culture‐positive animals (73.5%; 476/648) compared with 1NEG controls (22.8%; 147/645) or 3NEG controls (4.1%; 25/609), whereas horses accounted for the majority of 1NEG controls (64.7%; 417/645) and 3NEG controls (83.4%; 508/609).
The duration of hospitalization was similar for cases and 1NEG controls, with a mean of 3.8 days (median 2, IQR 1‐5) and 3.1 days (median 2, IQR 1‐4), respectively, whereas it was notably longer for 3NEG controls with a mean of 11.7 (median 8, IQR 6‐11).
During the study period, there were 81 clusters of within‐hospital horizontal transmission of Salmonella identified retrospectively among hospitalized animals (ie, at least 2 animals from different farms with phenotypically similar isolates hospitalized within 10 days of each other). On average, this involved 3 animals (range 2‐21) and continued for 13 days (range 1‐64). Most commonly they involved bovid, accounting for approximately 52.7% (116/220) of all animals involved (ie, animals shedding phenotypically similar isolates).
3.1. Univariable and multivariable modeling with 1NEG controls
Variables included in multivariable model building included hospitalization days, systemic illness, diarrhea in previous 48 hours, having a fever in previous 48 hours, leukopenia in previous 48 hours, route of antimicrobial drug administration, body system affected (specifically musculoskeletal, gastrointestinal, reproductive, other, or normal), species, receiving a gastroprotectant in previous 48 hours, receiving any antimicrobial drugs in previous 48 hours, receiving aminoglycosides, beta‐lactams, any generation of cephalosporin, or sulfas during hospitalization, age, sex, and transportation distance passed univariable screening (Table 1). The final multivariable model (Table 2) included hospitalization days, species, systemic illness, and fever as main effects and an interaction between aminoglycoside antimicrobial drugs and gastrointestinal system. Diarrhea and being hospitalized during an outbreak were forced into the model, regardless of P‐value, as there was evidence of confounding in the final model.
Table 2.
Final multivariable conditional logistic regression models of factors associated with large animal fecal shedding of Salmonella enterica
| Control group 1 (≥1 negative culture) | Control group 2 (≥3 negative cultures) | ||||||
|---|---|---|---|---|---|---|---|
| Variable | Category | OR | 95% CI | P‐value | OR | 95% CI | P‐value |
| Hospitalization days | ≥15 days | 12.47 | 4.03‐38.60 | <.0001 | 0.17 | 0.08‐0.37 | <.0001 |
| 8‐14 days | 2.51 | 1.37‐4.61 | 0.13 | 0.07‐0.24 | |||
| 4‐7 days | 1.87 | 1.28‐2.72 | 0.30 | 0.18‐0.53 | |||
| ≤3 days | Ref | Ref | |||||
| Systemic illness | Major | 1.89 | 1.10‐3.26 | .04 | Confounder | .61 | |
| Minor | 1.67 | 1.09‐2.54 | |||||
| Healthy | Ref | ||||||
| Species | Bovine | 15.80 | 10.34‐24.14 | <.0001 | 22.32 | 10.15‐49.11 | <.0001 |
| NWC | 1.61 | 0.83‐3.11 | 1.53 | 0.77‐3.03 | |||
| Small ruminant | 0.98 | 0.36‐2.67 | 1.45 | 0.37‐5.70 | |||
| Equine | Ref | Ref | |||||
| Febrile in previous 48 hours | Yes | 1.89 | 1.24‐2.88 | .01 | Confounder | .13 | |
| Unknown | 0.92 | 0.59‐1.45 | |||||
| No | Ref | ||||||
| Gastrointestinal (GI) disease | Yes | Interaction | .07 | 2.10 | 1.21‐3.66 | .01 | |
| No | Ref | ||||||
| Aminoglycoside use during hospitalization | Yes | Interaction | .05 | 1.84 | 1.09‐3.10 | .02 | |
| No | Ref | ||||||
| Diarrhea in previous 48 hours | Yes | Confounder | .14 | Did not pass univariable screening | |||
| No | |||||||
| Hospitalized during detected nosocomial transmission | Yes | Confounder | .95 | Confounder | .56 | ||
| No | |||||||
| Leukopenic in previous 48 hours | Unknown | Did not pass univariable screening | 2.75 | 1.72‐4.41 | .0001 | ||
| Yes | 1.17 | 0.62‐2.20 | |||||
| No | ref | ||||||
| Musculoskeletal disease | Yes | Did not pass univariable screening | Confounder | .10 | |||
| No | |||||||
| Reproductive disease | Yes | Did not pass univariable screening | Confounder | .14 | |||
| No | |||||||
| Animal age | <1 year old | Did not pass univariable screening | Confounder | .52 | |||
| Yearling | |||||||
| Adult | |||||||
| Transportation distance | ≤20 miles | Did not pass univariable screening | 1.58 | 0.96‐2.60 | .05 | ||
| 20‐50 miles | 0.83 | 0.49‐1.42 | |||||
| ≥50 miles | Ref | ||||||
| Sex | Female | Did not pass univariable screening | 1.68 | 1.12‐2.54 | .01 | ||
| Male | |||||||
| GI*aminoglycoside | .01 | No first‐order interactions | |||||
| GI disease | Aminoglycoside | ||||||
| Yes | Yes | 1.94 | 1.31‐2.59 | ||||
| Yes | No | 0.38 | −0.03‐0.78 | ||||
| No | Yes | 0.55 | −0.01‐1.11 | ||||
| No | No | Ref | |||||
Abbreviations: CI, confidence interval; NWC, New World camelid; OR, odds ratio; Ref, reference.
Controlling for the effects of other variables in the model, cattle had a 15 times greater odds of shedding Salmonella as compared to horses (OR 15.8; 95% CI 10.3, 24.1), whereas the odds of shedding among NWC and small ruminants was no different than horses and it was less than that for cattle (Table 2). In general, there was a greater odds of shedding among animals hospitalized for ≥4 days as compared to animals hospitalized ≤3 days (P < .0001), and there was an almost 12 times greater odds among animals hospitalized for ≥15 days (OR 12.47; 95% CI 4.03, 38.60) as compared to animals hospitalized ≤3 days. The odds of shedding among animals with major or minor systemic illness were no different, but almost 2 times greater than that in healthy animals (P = .04), and febrile animals had an almost 2 times greater odds of shedding as compared to afebrile animals (OR 1.9; 95% CI 1.2, 2.9). In addition, the odds of shedding among animals with gastrointestinal disease and receiving aminoglycosides was 2 times greater than for animals without gastrointestinal disease nor receiving aminoglycosides (OR 1.9; 95% CI 1.3, 2.6). In general, analyses stratified by species resulted in stronger measures of association for systemic illness and hospitalization days among horses (data not shown).
3.2. Univariable and multivariable modeling with 3NEG controls
Variables that passed univariable screening and were included in multivariable model building included hospitalization days, systemic illness, diarrhea in previous 48 hours, having a fever in previous 48 hours, leukopenia in previous 48 hours, having general anesthesia in previous 48 hours, route of antimicrobial drug administration, body system affected (specifically musculoskeletal, gastrointestinal, reproductive, respiratory, renal, other or normal), species, receiving a gastroprotectant in previous 48 hours, receiving any antimicrobial drugs in previous 48 hours, receiving aminoglycoside, beta‐lactam, any generation of cephalosporin, tetracycline, fluoroquinolone, florfenicol, or sulfas during hospitalization, age, sex, transportation distance, and being hospitalized during an outbreak (Table 1). The final multivariable model included hospitalization days, species, gastrointestinal disease, receiving aminoglycosides antimicrobial drugs, having leukopenia, sex, and transportation distance. Variables for systemic illness, fever, age, musculoskeletal and reproductive systems, and being hospitalized during an outbreak were forced into the model, regardless of P‐value, as there was evidence of confounding in the final model (Table 2). There were no significant first‐order interactions among main effects in this model.
Controlling for the effects of other variables in the model, cattle had 22 times greater odds of shedding Salmonella compared to horses (22.3; 95% CI 10.2, 49.2), whereas the odds of shedding among NWC and small ruminant were no different from horses but were less than that for cattle. Animals hospitalized for ≥4 days were less likely to shed Salmonella as compared to animals hospitalized for ≤3 days (P < .0001). In addition, there was an increased odds of shedding Salmonella for animals with gastrointestinal disease as compared to healthy animals (P = .01), for animals that received aminoglycoside antimicrobial drugs during hospitalization as compared to animals who did not (P = .01), animals that were female as compared to male (P = .01), and animals transported 20 or fewer miles as compared to animals transported 50 miles or greater (P = .01). Interestingly, animals with “unknown” leukopenia were almost 3 times more likely to shed as compared to animals without leukopenia (OR 2.75; 95% CI 1.72, 4.41), whereas the odds of shedding among animals with leukopenia was no different than for animals without leukopenia. In general, analyses stratified by species resulted in stronger measures of association for hospitalization days among horses (data not shown).
3.2.1. Population attributable fraction
Population attributable fraction is the proportion of disease risk in a population that can be attributed to a particular risk factor or group of factors,18 taking into account both the magnitude of effect and the prevalence of the risk factor in the population. Note that PAF is not dividing total risk among different factors rather it is giving a relative indication as to what proportion of disease might be prevented if that particular factor or constellation of factors were eliminated from the population (assuming the frequencies of all other factors in the population remain unchanged) and that factors can contribute to more than 1 attributable fraction. In this animal population, as compared to animals with at least 1 negative culture, approximately 2/3 of the shedding risk can be attributed to each of systemic illness (PAF = 69.4%; classified as healthy, minor, and major) or gastrointestinal disease (PAF = 59.9%; eg, colic, diarrhea). Only 2.7% of the shedding risk can be attributed to animals demonstrating the classic triad of signs typically associated with Salmonella shedding, diarrhea, fever, and leukopenia.19, 20 When considering each clinical sign alone, the PAF for diarrhea, fever, and leukopenia were 73.8, 30.9, and 57.7%, respectively. Finally, approximately 70% of animal shedding in this population can be attributed to species, specifically cattle (PAF = 68.7%).
4. DISCUSSION
Factors associated with endemic shedding of Salmonella among the general large animal hospital population are similar to those identified with targeted surveillance and during times of epidemic disease6, 9, 10—namely factors related to systemic disease, including fever and leukopenia, gastrointestinal disease, duration of hospitalization, and species. Historically, Salmonella shedding has been assumed to be typified by animals exhibiting the classical triad of signs—diarrhea, fever, and leukopenia.19, 20 Although it is true that all 3 signs can be a specific indicator of clinical salmonellosis, the results reported here indicate that taken together, this triad of signs is not a sensitive indicator of shedding risk (ie, many other large animals shed Salmonella without showing all 3 signs). Because the frequency with which this complex of signs occurs together in a population can be quite low, it results in a poor detection rate upon which to base infection control policy. Alternatively, approximately 70% of animal shedding could be attributed to systemic illness, irrespective of the body system affected. This suggests that infection control efforts aimed at preventing HAIs related to Salmonella might best be directed at animals with substantive signs of systemic illness.
In the study reported here, 2 comparison groups were used to develop 2 different multivariable models: 1 control group consisting of animals with at least 1 negative culture (1NEG controls) and another consisting of animals with at least 3 negative cultures (3NEG controls), based on comparison groups commonly reported in the literature.5, 6, 9 It is important to consider whether these 2 control groups represent different animal populations when they are being used in comparisons with the case population. For example, 1NEG controls generally had a similar duration of hospitalization as cases (mean 3.1 and 3.8 days, respectively), whereas 3NEG controls were hospitalized considerably longer (mean 11.7 days). As a result, the odds of shedding increased with duration of hospitalization when comparing to 1NEG controls, but decreased with duration of hospitalization when comparing to 3NEG controls. These are 2 very different answers—as such, the question becomes which provides the most relevant comparison upon which to base policy decisions?
We contend that the comparison group consisting of 1NEG controls (those with at least 1 negative culture) is the more appropriate comparison group (ie, is more representative of the case population) although some animals might have been misclassified (ie, classified as negative when they are truly shedding) because of the insensitive nature of the Salmonella culture methodology and the limited number of fecal cultures performed on some animals in this study. This misclassification is expected to be differential in this particular study and will likely attenuate the strength of associations (ie, it is a nullifying bias). With this in mind, we can estimate the proportion of Salmonella shedding in this population that can be attributed to systemic illness—approximately 70%—and realize that severity of disease, as defined by systemic illness, is a likely candidate upon which to base infection control policy. If we manage animals with more severe systemic illness differently (ie, manage as a separate cohort in an area with heightened barrier nursing precautions), then we will have effectively managed approximately two‐thirds of animals shedding Salmonella in this population. This will presumably limit the potential for environmental contamination, which is typically seen when animals are shedding, as the lag time of 3‐5 days (depending on testing methodology) to realize results has effectively been eliminated.
Although 3NEG controls might represent a slightly different population as compared to the case population, resultant associations do support our assertion; that animals with more severe disease are more likely to be shedding. Although systemic illness was a confounder in this model, other variables that suggest disease severity, such as gastrointestinal disease, receiving aminoglycoside antimicrobial drugs, and experiencing leukopenia, were associated with animal shedding. Although experiencing leukopenia in the previous 48 hours was associated with animal shedding, a stronger association was noted for animals with an unknown leukocyte count. We suspect this is related to the high proportion of shedding dairy cattle in our population that are hospitalized for less than 3 days and are medically managed without consulting CBCs (typically these animals present with a displaced abomasum). Additional factors associated with shedding in this model were sex (specifically being female) and transportation distance (specifically traveling ≤20 miles). Again, we believe these associations to be related to our animal population, 25% being mostly lactating dairy cattle with a high shedding prevalence, which typically reside within 20 miles of our facility. Although this is specific to our practice area, it highlights the importance of developing an infection control program that is specific to the facility and practice environment.
For facilities managing more than 1 species, consideration should be given to species segregation as a means of shedding risk mitigation, as again approximately 70% of animals shedding can be attributed to species in this study population (PAF = 68.7%). It is important to interpret this in light of the animal population from which it was derived, specifically that the bovine caseload at our facility is predominately dairy breeds and generally has a high shedding prevalence. Although this is likely similar to other academic institutions (ie, managing multiple species), it might not be so with respect to private practices. As the hospital population varies with regard to risk factors of interest, the results of this study become less generalizable. In addition, consideration should be given to differences in facilities and infection control protocols as these will likely impact infectious disease transmission among animals.
Although this was a retrospective study and relied on the quality of data recorded in the medical record, it was based on routine fecal surveillance for an extended period of time. As such, we believe that resultant associations do support our assertion that consideration of severity of disease should be a key component to policy development and will potentially allow for substantial risk mitigation with respect to endemic shedding of Salmonella and ultimately to the prevention of epidemic disease.
CONFLICT OF INTEREST DECLARATION
Authors declare no conflict of interest.
OFF‐LABEL ANTIMICROBIAL DECLARATION
Authors declare no off‐label use of antimicrobials.
INSTITUTIONAL ANIMAL CARE AND USE COMMITTEE (IACUC) OR OTHER APPROVAL DECLARATION
Approval granted by the Colorado State University IACUC.
HUMAN ETHICS APPROVAL DECLARATION
Authors declare human ethics approval was not needed for this study.
ACKNOWLEDGMENTS
Portions of this study were presented at the Annual Meeting of the Conference of Research Workers in Animal Diseases, Chicago, IL; and the 2018 American College of Veterinary Internal Medicine Forum, Seattle, WA. The authors thank students and staff at the Colorado State University JLV‐Veterinary Teaching Hospital (VTH) that assisted with sample collection and entry of data that was used in this study. The authors thank the administration and faculty at the Colorado State University JLV‐VTH for their ongoing support of the VTH Infection Control Program.
Burgess BA, Morley PS. Risk factors for shedding of Salmonella enterica among hospitalized large animals over a 10‐year period in a veterinary teaching hospital. J Vet Intern Med. 2019;33:2239–2248. 10.1111/jvim.15579
Funding information James L. Voss Veterinary Teaching Hospital, Colorado State University, Fort Collins, CO; Storm Cat Career Development Award, Grayson Jockey Club Research Foundation, Lexington, KY
REFERENCES
- 1. Benedict KM, Morley PS, Van Metre DC. Characteristics of biosecurity and infection control programs at veterinary teaching hospitals. J Am Vet Med Assoc. 2008;233(5):767‐773. [DOI] [PubMed] [Google Scholar]
- 2. Dallap Schaer BL, Aceto H, Rankin SC. Outbreak of salmonellosis caused by Salmonella enterica serovar Newport MDR‐AmpC in a large animal veterinary teaching hospital. J Vet Intern Med. 2010;24(5):1138‐1146. [DOI] [PubMed] [Google Scholar]
- 3. Steneroden KK, van Metre DC, Jackson C, Morley PS. Detection and control of a nosocomial outbreak caused by Salmonella newport at a large animal hospital. J Vet Intern Med. 2010;24(3):606‐616. [DOI] [PubMed] [Google Scholar]
- 4. Burgess BA, Morley PS, Hyatt DR. Environmental surveillance for Salmonella enterica in a veterinary teaching hospital. J Am Vet Med Assoc. 2004;225(9):1344‐1348. [DOI] [PubMed] [Google Scholar]
- 5. Smith BP, Reina‐Guerra M, Hardy AJ. Prevalence and epizootiology of equine salmonellosis. J Am Vet Med Assoc. 1978;172(3):353‐356. [PubMed] [Google Scholar]
- 6. Dallap Schaer BL, Aceto H, Caruso MA III, Brace MA. Identification of predictors of Salmonella shedding in adult horses presented for acute colic. J Vet Intern Med. 2012;26(5):1177‐1185. [DOI] [PubMed] [Google Scholar]
- 7. Ernst NS, Hernandez JA, MacKay RJ, et al. Risk factors associated with fecal Salmonella shedding among hospitalized horses with signs of gastrointestinal tract disease. J Am Vet Med Assoc. 2004;225(2):275‐281. [DOI] [PubMed] [Google Scholar]
- 8. Ekiri AB, MacKay RJ, Gaskin JM, et al. Epidemiologic analysis of nosocomial Salmonella infections in hospitalized horses. J Am Vet Med Assoc. 2009;234(1):108‐119. [DOI] [PubMed] [Google Scholar]
- 9. Traub‐Dargatz JL, Salman MD, Jones RL. Epidemiologic study of salmonellae shedding in the feces of horses and potential risk factors for development of the infection in hospitalized horses. J Am Vet Med Assoc. 1990;196(10):1617‐1622. [PubMed] [Google Scholar]
- 10. Kim LM, Morley PS, Traub‐Dargatz JL, Salman MD, Gentry‐Weeks C. Factors associated with Salmonella shedding among equine colic patients at a veterinary teaching hospital. J Am Vet Med Assoc. 2001;218(5):740‐748. [DOI] [PubMed] [Google Scholar]
- 11. Burgess BA, Bauknecht K, Slovis NM, Morley PS. Factors associated with equine shedding of multi‐drug‐resistant Salmonella enterica and its impact on health outcomes. Equine Vet J. 2018;50(5):616‐623. [DOI] [PubMed] [Google Scholar]
- 12. Hird DW, Casebolt DB, Carter JD, Pappaioanou M, Hjerpe CA. Risk factors for salmonellosis in hospitalized horses. J Am Vet Med Assoc. 1986;188(2):173‐177. [PubMed] [Google Scholar]
- 13. Alinovi CA, Ward MP, Couëtil LL, Wu CC. Risk factors for fecal shedding of Salmonella from horses in a veterinary teaching hospital. Prev Vet Med. 2003;60(4):307‐317. [DOI] [PubMed] [Google Scholar]
- 14. Carter JD, Hird DW, Farver TB, Hjerpe CA. Salmonellosis in hospitalized horses: seasonality and case fatality rates. J Am Vet Med Assoc. 1986;188(2):163‐167. [PubMed] [Google Scholar]
- 15. Ward MP. Fecal shedding of Salmonella in horses admitted to a veterinary teaching hospital. J Equine Vet. 2003;23(9):403. [DOI] [PubMed] [Google Scholar]
- 16. Hird DW, Pappaioanou M, Smith BP. Case‐control study of risk factors associated with isolation of Salmonella saintpaul in hospitalized horses. Am J Epidemiol. 1984;120(6):852‐864. [DOI] [PubMed] [Google Scholar]
- 17. Dunowska, M. , Traub‐Dargatz JL, Morley PS. Recent progress in controlling Salmonella in veterinry hospitals. Paper presented at: 50th Annual Convention of the American Association of Equine Practitioners; 2004; Lexington, KY.
- 18. Dohoo I, Martin W, Stryhn H. Population attributable fraction Veterinary Epidemiologic Research. Charlottetown, PEI: VER Inc; 2009:142‐143. [Google Scholar]
- 19. Dorn CR, Coffman JR, Schmidt DA, Garner HE, Addison JB, EL MC. Neutropenia and salmonellosis in hospitalized horses. J Am Vet Med Assoc. 1975;166(1):65‐67. [PubMed] [Google Scholar]
- 20. Owen R, Fullerton JN, Tizard IR, Lumsden JH, Barnum DA. Studies on experimental enteric salmonellosis in ponies. Can J Comp Med. 1979;43(3):247‐254. [PMC free article] [PubMed] [Google Scholar]


