Abstract
The β-adrenergic receptor (βAR) exists in an equilibrium of inactive and active conformational states, which shifts in response to different ligands and results in downstream signaling. In addition to cAMP, βAR signals to hypoxia-inducible factor 1 (HIF-1). We hypothesized that a βAR-active conformation (R**) that leads to HIF-1 is separable from the cAMP-activating conformation (R*) and that pulmonary arterial hypertension (PAH) patients with HIF-biased conformations would not respond to a cAMP agonist. We compared two cAMP agonists, isoproterenol and salbutamol, in vitro. Isoproterenol increased cAMP and HIF-1 activity, while salbutamol increased cAMP and reduced HIF-1. Hypoxia blunted agonist-stimulated cAMP, consistent with receptor equilibrium shifting toward HIF-activating conformations. Similarly, isoproterenol increased HIF-1 and erythropoiesis in mice, while salbutamol decreased erythropoiesis. βAR overexpression in cells increased glycolysis, which was blunted by HIF-1 inhibitors, suggesting increased βAR leads to increased hypoxia-metabolic effects. Because PAH is also characterized by HIF-related glycolytic shift, we dichotomized PAH patients in the Pulmonary Arterial Hypertension Treatment with Carvedilol for Heart Failure trial (NCT01586156) based on right ventricular (RV) glucose uptake to evaluate βAR ligands. Patients with high glucose uptake had more severe disease than those with low uptake. cAMP increased in response to isoproterenol in mononuclear cells from low-uptake patients but not in high-uptake patients’ cells. When patients were treated with carvedilol for 1 wk, the low-uptake group decreased RV systolic pressures and pulmonary vascular resistance, but high-uptake patients had no physiologic responses. The findings expand the paradigm of βAR activation and uncover a novel PAH subtype that might benefit from β-blockers.
Keywords: β-adrenergic receptor, hypoxia, metabolism, pulmonary hypertension
INTRODUCTION
The β-adrenergic receptor (βAR) is a classical G protein-coupled receptor (GPCR) that activates multiple downstream effectors, e.g., the canonical cAMP signaling pathway (11, 43). In its basal state, the βAR is dynamic, shifting between various inactive and active conformations often designated as R and R*, respectively. Spontaneous shifts to R* accounts for constitutive signaling in the absence of a ligand. Receptor ligands affect signaling by modulating the equilibrium of these receptor conformations. For example, full agonists bind and stabilize R*, shifting the equilibrium toward an active state and signaling response. Partial agonists also stabilize R*; however, these ligands only produce a partial cAMP signaling response compared with the full agonist. Inverse agonists have an opposite effect wherein they bind and stabilize R, shifting the equilibrium toward an inactive state. Inverse agonists reduce signaling to levels lower than at basal equilibrium. In contrast, βAR antagonists have no apparent effect on the receptor equilibrium but prevent agonist or inverse agonist binding to either receptor conformations. Other than cAMP, the βAR also regulates other downstream signaling pathways, e.g., the extracellular signal-regulated kinase 1/2 and hypoxia-inducible factor-1 (HIF-1) (5, 11, 43). The various conformations available in the equilibrium paradigm provide a model to understand how some pharmacologic agents serve as a full βAR cAMP agonist yet act as an inverse agonist or antagonist for the other downstream pathways (49, 51).
Previous work shows that the βAR is sufficient for stabilization of HIF-1α in normoxia and necessary for HIF-1α stabilization in hypoxia (5). Interestingly, the βAR is also coordinately regulated by oxygen tension through hydroxylation and phosphorylation (5, 53). HIF-1 is the primary transcription factor that regulates cellular responses to hypoxia. It comprises an α and β subunit, the former of which is degraded in an oxygen-dependent manner. When oxygen concentrations are low, HIF-1α is stabilized and translocates to the nucleus where it dimerizes with HIF-1β and stimulates transcription through binding to hypoxia-response elements (HREs). Despite recent evidence of the reciprocal regulation of HIF and βAR, there is very little understanding of how the pharmacologic ligands may impact βAR signaling in conditions in which the receptor is biased toward a specific conformation. The relationship between βAR and hypoxia signaling is particularly relevant to pulmonary arterial hypertension (PAH), which is characterized by increased pulmonary vascular pressure and progressive right heart failure. β-blockers are a mainstay of left heart failure and recently have been suggested to have benefit in right heart failure. Several clinical trials investigated different β-blockers in PAH with varying and inconclusive results (13, 32, 33, 48), which may be related to phenotypes of PAH that are more or less responsive (6). Mechanistically, βAR and HIF-1 signaling are abnormal in PAH and contribute to disease pathology. The βAR is dysfunctional, and this is associated with worse right ventricular (RV) function (4, 37). Additionally, HIF-1α expression is increased in PAH pulmonary vascular endothelial cells and hearts (27, 47). Metabolic evidence of this is provided in the multiple reports that PAH individuals have higher glucose uptake in the right ventricle, related to a shift toward glycolysis, a well-known HIF-1-driven effect (40, 42, 54). Understanding the linkage between βAR and HIF-1 may provide insights into the variability of responses to β-blockers and identify those individuals in whom β-blockers would be most efficacious.
Here we hypothesize that there is a βAR conformation (R**) that leads to HIF-1 signaling separable from the canonical activated receptor for cAMP (R*) and that biased signaling toward HIF-1 can be identified in patients with PAH who are nonresponders to βAR ligands. We utilized a pharmacological approach to test this. Isoproterenol and salbutamol, both agonists for cAMP, were evaluated for effects on HIF-1 in vitro and in vivo. We also assessed whether or not hypoxia biases the βAR, altering its signaling profile toward HIF and away from cAMP. Finally, using a recent study of carvedilol in patients with PAH in which RV glucose uptake was measured by 2-[18F]fluoro-2-deoxy-d-glucose (FDG) positron emission tomography (PET) scan (8, 40), patients were dichotomized to a high or low RV glucose uptake phenotype as a surrogate of high (or low) HIF-1 activity. Blood mononuclear cells were evaluated in a subgroup of the two PAH phenotypes for response to isoproterenol. Subsequently, all patients received a low dose of carvedilol for 1 wk. Echocardiogram was done to evaluate cardiac responses to carvedilol. Blood responses characteristic of HIF, i.e., red blood cells (RBCs) and endothelin-1 (ET-1), were measured as well. The results identify an expanded model of βAR regulation in which HIF-1 signaling occurs via a receptor state that is independent of the cAMP pathway. This model may help explain the variability in PAH severity and treatment response.
MATERIALS AND METHODS
Compounds.
(−)-Isoproterenol (+)-bitartate salt (Sigma I2760), salbutamol (Sigma S8260 for cells, Sigma 1012600 for mice), and carvedilol (Sigma C3993) were prepared fresh for each experiment.
Cell culture.
Human embryonic kidney cells overexpressing the β2AR (HEK293-β2AR) and nonoverexpressing cells [HEK293-wild type (WT)] were cultured in MEM with 10% fetal bovine serum and 1% penicillin-streptomycin-fungizone. For hypoxia experiments, cells were incubated in a sealed chamber at 37°C with 2% O2, 5% CO2, and balanced with 93% N2.
Luciferase assays.
The luciferase reporter vector was a pGL2 basic vector (Promega E1641) containing three HREs upstream of the firefly luciferase gene (HRE sequence: CACGTC). The luciferase vector was cotransfected with the constitutive control reporter pRL-SV40 Renilla luciferase vector (Promega E2231). HEK293-β2AR cells were plated on 100-mm plates at a density of 10 × 106 cells/plate 24 h before transfection. Cells were cotransfected with HRE-luciferase (1 µg) and pRL Renilla (0.5 µg) plasmids using Lipofectamine 2000 (Invitrogen 11668019) for 6 h in antibiotic-free media. Fourteen hours posttransfection, cells were split to 12-well plates at a density of 1 × 106 cells/well. For baseline HIF-1 comparisons in WT versus overexpressing cells, lysates were collected the next day. For ligand studies, cells were serum starved for 2 h, then treated for 45 min before exposure to hypoxia (2% O2) or normoxia. After 20 h, cells were lysed and processed according to the manufacturer’s instructions in the Dual-Luciferase Reporter Assay System (Promega E1910).
cAMP measurements.
HEK293-β2AR cells were plated on 6-well or 12-well plates at a density of 1.5 × 106 cells/well or 8.0 × 105 cells/well, respectively. The next day, cells were serum starved overnight. Cells were treated for 5 min. For hypoxia experiments, cells were placed in hypoxia (2% O2) then treated with compounds prepared in deoxygenated media for 5 min. Cells were washed with ice-cold PBS. For cAMP measurements, cells were lysed and processed according to the manufacturer’s instructions in the CatchPoint cAMP Fluorescent Assay Kit (Molecular Devices R8089).
Radio-ligand βAR density.
Cell lysates were processed for plasma membrane isolation as previously described (34). The early endosome fraction was recovered after ultracentrifugation of the crude cytosolic fraction for 1 h at 300,000 g. Receptor binding was performed as previously described (34). Briefly, 20 µg of protein from the plasma membrane and early endosomal fraction were incubated with 250 pM βAR ligand [125I]-cyanopindolol (125I-CYP, PerkinElmer, NEX189100UC), and nonspecific binding was determined using 250 pM 125I-CYP plus 100 μM ICI-118,552 (Sigma I127).
Animal model.
Eight-week-old male C57BL/6 mice from Jackson Laboratory were treated with vehicle; 1, 5, or 10 mg/kg salbutamol dissolved in 2% methanol in saline; or 1, 5, or 10 mg/kg isoproterenol dissolved in saline via intraperitoneal injection. Doses were determined based on previously published reports (7, 12, 21, 25, 28, 55). Doses at the low end of the reported range were chosen with a short duration to minimize risk of cardiac abnormalities. Mice were anesthetized with 10% isoflurane, and blood was collected via cardiac puncture. Blood was stored in K2EDTA tubes and centrifuged at 500 g for 5 min to obtain plasma. Kidneys were flash frozen in liquid nitrogen. Bone marrow was collected from hind legs and processed fresh for flow cytometry. All animal experiments were approved by the Cleveland Clinic Institutional Animal Care and Use Committee at the Lerner Research Institute in Cleveland, Ohio.
Western blot.
For HIF-1α Western blot analysis, 200 µg protein from kidney nuclear extracts was loaded. Nuclear extracts were prepared according to manufacturer’s instructions in the Nuclear Extraction Kit (Affymetrix AY2002). HIF-1α and Lamin B1 antibodies (Novus, cat. no. NB100-479, and Santa Cruz, cat. no. sc-374015, respectively) were diluted 1:1,000 in 5% nonfat milk. HIF-1α was detected with anti-rabbit IgG-horseradish peroxidase (GE Healthcare NA9340) diluted 1:5,000 in 5% nonfat milk. Lamin B1 was detected with anti-mouse IgG-horseradish peroxidase (GE Healthcare NA931). Lamin B1 and HIF-1α antibodies were previously validated for specificity (15, 39).
Flow cytometry.
Bone marrow was analyzed fresh on the day of collection. Nonspecific binding sites were blocked with 10% normal goat serum in PBS for 15 min at room temperature. Cells were washed with 1% BSA in PBS and incubated with APC-conjugated anti-CD45 (eBioscience 17-0451-82) at 1:100 (2 μg/mL), phycoerythrin-conjugated TER119 (eBioscience 12-5921-81) at 1:50 (4 μg/mL), and FITC-conjugated anti-CD44 (eBioscience 11-0441-81) at 1:100 (5 μg/mL) for 30 min on ice. All antibodies were diluted in 1% BSA in PBS. Cells were washed twice with 1% BSA in PBS and resuspended in FACS Flow containing the dead cell marker 7-AAD (BD Biosciences 51-68981E) at 1:200. Samples were run on an LSRII flow cytometer (Becton Dickenson), and data were analyzed using FlowJo V10 (Tree Star Inc.). Aggregates, cell debris, and dead cells were gated out. Leukocytes were excluded using CD45. Three populations were defined based on size and CD44 and TER119 levels. Proerythroblasts were defined as CD44hi TER119lo, and basophilic and polychromatic erythroblasts were defined as TER119hi and selected based on decreasing size and CD44 expression. Proerythroblasts, basophilic, and polychromatic erythroblasts were defined such that the percentage of cells in each population fit the expected ratio of 1:2:4:8 (5, 26).
Seahorse XF24 analysis.
HEK293-β2AR or HEK293-WT cells were seeded at a density of 1.5 × 105 cells/well in Seahorse XF24 Cell Culture Microplates (Agilent 100850–001) 1 day before the assay in MEM with 10% fetal bovine serum and 1% penicillin/streptomycin/fungizone. Cells were treated with 100 nM digoxin (Sigma D6003), 30 µM chrysin (Sigma C80105), or vehicle for 4.5 h before analysis. Cells were analyzed on the Seahorse XF24 Analyzer (Agilent) using the Glycolytic Stress Test Kit (Agilent 103020–100) according to the manufacturer’s instructions. Briefly, extracellular acidification rate (ECAR) was measured at baseline, after 10 mM glucose addition, after 1 µM oligomycin treatment, and after 50 mM 2-deoxy-glucose (2-DG) treatment. Estimated glycolysis was calculated as maximum glucose-stimulated ECAR minus baseline ECAR.
Human studies.
The Pulmonary Arterial Hypertension Treatment with Carvedilol for Heart Failure (PAHTCH) clinical trial was approved by the Cleveland Clinic Institutional Review Board (NCT01586156). Thirty PAH subjects enrolled after providing written informed consent. All patients started with a 1 wk, open-label treatment with carvedilol (3.125 mg, 2× day). Study design and participants have been previously described in detail (8). FDG-PET standardized uptake values (SUVs) in the right ventricle versus left ventricle (RV/LV SUV), RV systolic pressure (RVSP), and pulmonary vascular resistance (PVR) were measured as previously described (8). Serum endothelin-1 was measured by the Endothelin-1 Quantikine ELISA kit (R&D Systems, DET100). Urinary creatinine was measured by the Abbott Architect machine according to the manufacturer’s instructions. Mononuclear cells for cAMP analysis were isolated from blood via RBC lysis and centrifugation. Two × 106cells/mL were treated with 300 µM isoproterenol for 10 min at 37°C. The dose was chosen based on previous studies (5). Cells were then lysed and processed according to the CatchPoint cAMP Fluorescent Assay Kit instructions. Mononuclear cells for flow cytometric analysis of βAR density were treated with RBC lysis buffer then fixed with 4% formaldehyde and permeablized with 0.2% Triton-X100. Staining and flow cytometric analysis were performed as previously described (37).
Statistical analysis.
Statistical analysis was performed using JMP Pro 13/14 and GraphPad Prism 5. The P value threshold for significance was 0.05. Data in Figs. 1–3 were normalized to vehicle control, and P values were from the Bonferroni posttest. Data in Fig. 4, A, B, and D–F were analyzed via Student’s t-test. Figure 4C was analyzed via ANOVA. P values in Fig. 5 were derived from Bonferroni posttest. High and low groups in Figs. 6 and 7 were compared via Student’s t-test. Data in Fig. 8 were analyzed via paired t-test.
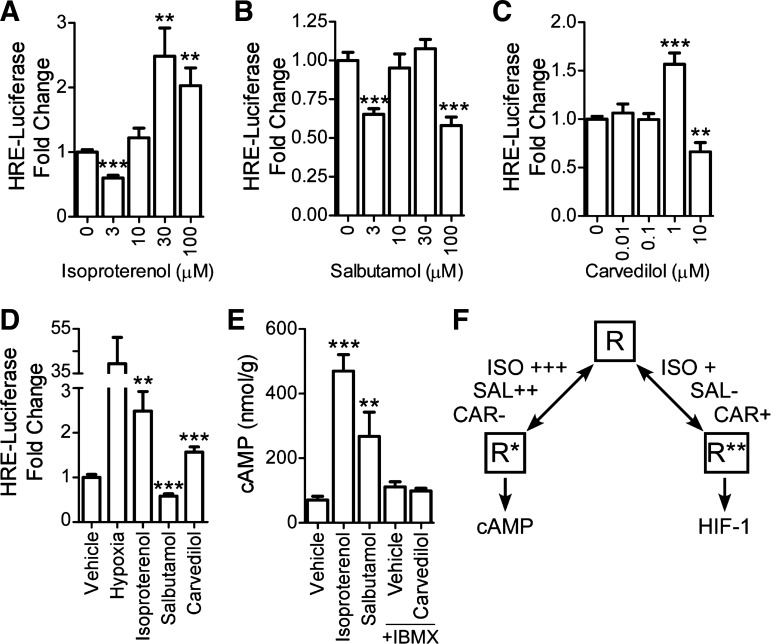
Fig. 1.
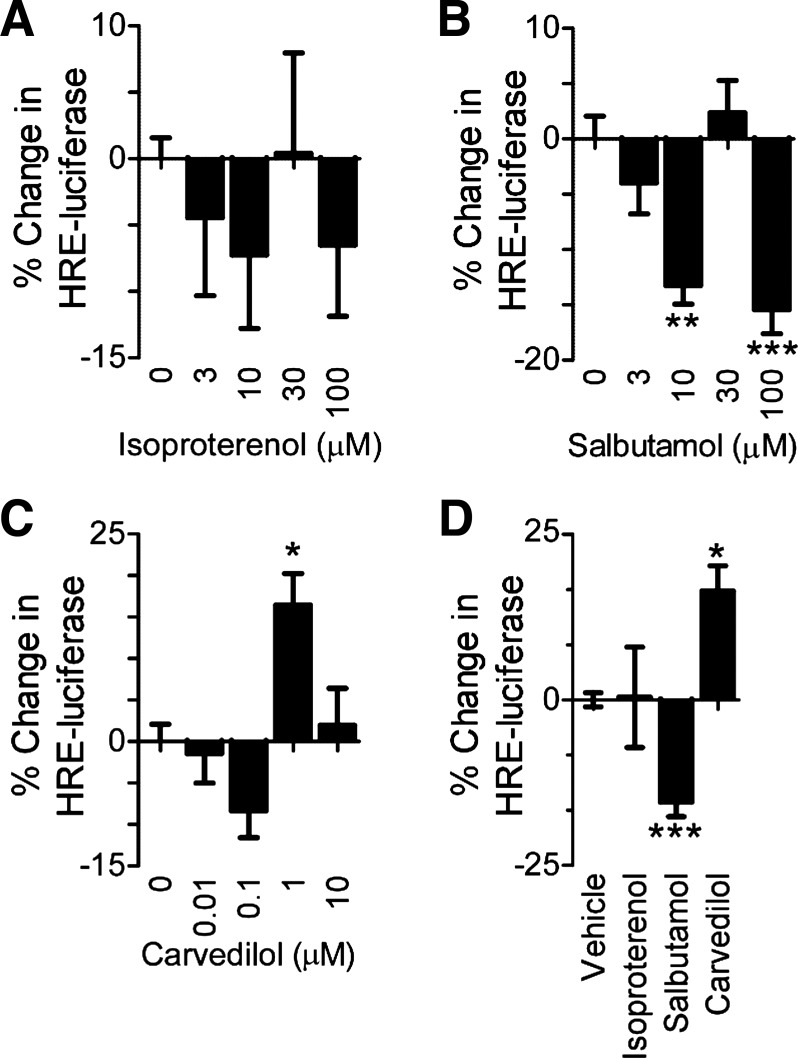
β-Adrenergic receptor (βAR) ligands differentially regulate hypoxia-inducible factor 1 (HIF-1) and cAMP in vitro. A–C: human embryonic kidney cells overexpressing the β2AR (HEK293-β2AR) were treated in normoxic conditions for 20 h with isoproterenol (A), salbutamol (B), or carvedilol (C). HIF-1 activity was measured using a hypoxia-response element (HRE)-luciferase reporter. Error bars represent SE, n = 3–4, in triplicate. **P < 0.01 and ***P < 0.001, relative to vehicle, Bonferroni posttest. D: summary of A–C effects on HRE-luciferase compared with hypoxia-positive control. Isoproterenol (30 µM), 100 µM salbutamol, and 1 µM carvedilol are shown. E: regulation of cAMP in HEK293-β2AR cells treated with isoproterenol (10 µM), salbutamol (10 µM), or carvedilol (10 µM) + IBMX (500 mM) for 5 min. cAMP was measured via fluorescent assay and normalized to total protein. Error bars represent SE; n = 3–4 in duplicate. **P < 0.01 and ***P < 0.001 relative to vehicle (Bonferroni posttest). F: theoretical model of βAR regulation. The receptor status is an equilibrium of multiple conformations that signal through different pathways. Ligands affect signaling by shifting the equilibrium of receptors toward or away from specific conformations. Results indicate that isoproterenol is an agonist for cAMP and a partial agonist for HIF-1, shifting the equilibrium toward both pathways. Salbutamol is an agonist for cAMP and an inverse agonist for HIF-1, shifting the equilibrium away from HIF-1. Carvedilol is an inverse agonist for cAMP and partial agonist for HIF-1, shifting the equilibrium away from cAMP and toward HIF-1. CAR, carvedilol; ISO, isoproterenol; R, inactive conformation; R*, conformation that activates cAMP; R**, conformation that activates HIF-1; SAL, salbutamol.
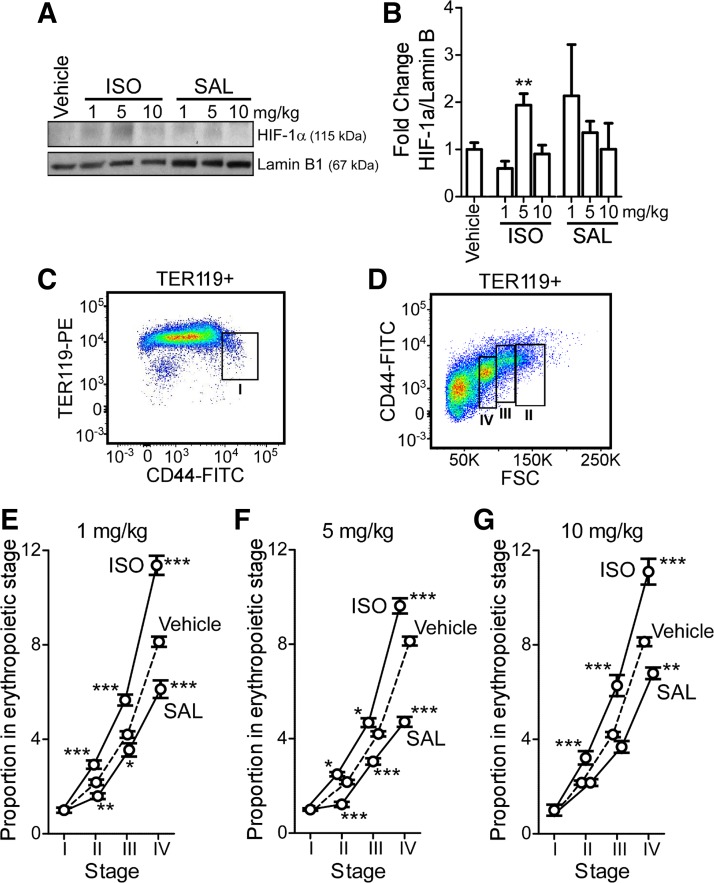
Fig. 3.
Isoproterenol (ISO) and salbutamol (SAL) have opposing effects on erythropoietic response in vivo. Mice received 1, 5, or 10 mg/kg isoproterenol, salbutamol, or vehicle via intraperitoneal injection 2 h before euthanization. n = 5 per group for 1 and 10 mg/kg, and n = 10 for 5 mg/kg doses. A: representative Western blot of kidney hypoxia-inducible factor 1 α (HIF-1α) levels. HIF-1α was normalized to Lamin B1. Samples were run on multiple gels in parallel. Data were normalized to vehicle-treated group. Error bars represent SE. **P < 0.01 (Bonferroni posttest). C and D: gating for erythroid progenitor stages measured via flow cytometry. Proerythroblast (stage I), basophilic erythroblasts (stage II), polychromatic erythroblasts (stage III), and orthochromatic erythroblasts (stage IV) were determined based on their expression of CD44, TER119, and size. E–G: quantification of stages I–IV by dose. Normal erythroid development shows a 1:2:4:8 ratio from stage I to stage IV. Each treatment group was normalized to stage I. Error bars represent SE. *P < 0.05, **P < 0.01, and ***P < 0.001 relative to vehicle (Bonferroni posttest).
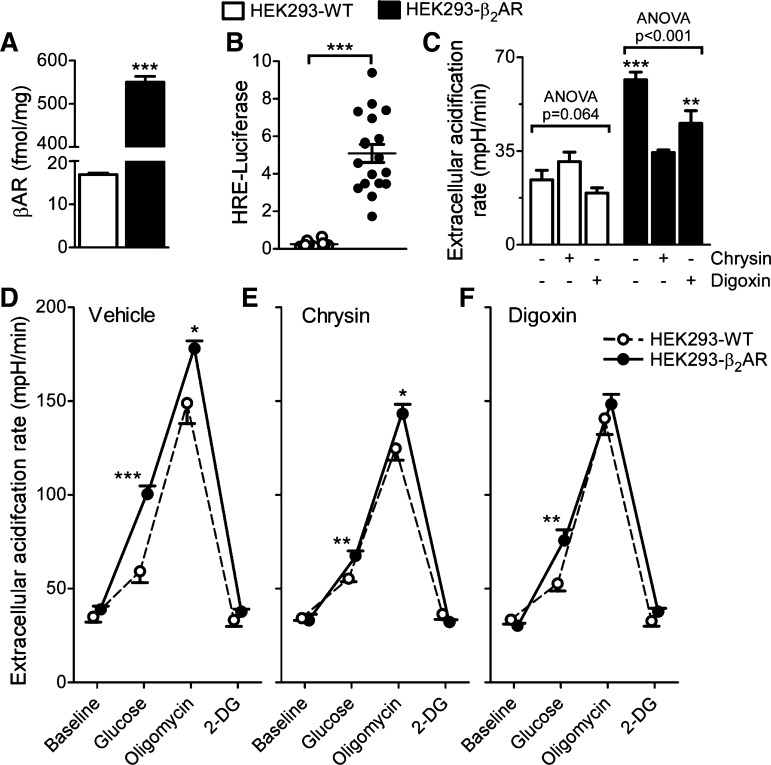
Fig. 4.
Overexpression of β2-adrenergic receptor (β2AR) in human embryonic kidney 293 cells (HEK293) increases basal hypoxia-inducible factor 1 (HIF-1) activity and downstream effects under normoxia. A: β2AR density on the plasma membrane of HEK293-β2AR and HEK293-wild-type (WT) cells was determined via radio-ligand binding. Error bars represent SE; n = 3. ***P < 0.001 (Student’s t-test). B: basal HIF-1 activity was measured via hypoxia-response element (HRE)-luciferase activity normalized to Renilla control plasmid in normoxia. Error bars represent SE; n = 18. ***P < 0.001 (Student’s t-test). C: estimated glycolysis was determined using the Seahorse glycolytic stress test. Glycolysis is calculated as maximal glucose-stimulated extracellular acidification rate (ECAR) minus baseline ECAR. Cells were pretreated with HIF-1 inhibitors digoxin (100 nM) or chrysin (30 µM) or vehicle control for 4.5 h. Error bars represent SE; n = 3 in duplicate. P values determined via ANOVA. **P < 0.01 and *** P < 0.001 compared with WT cells with same treatment (Student’s t-test). D–F: vehicle, chrysin, or digoxin-treated cells were measured at baseline, after 10 mM glucose addition to stimulate glycolysis, after 1 µM oligomycin addition to inhibit oxidative phosphorylation, and after 50 mM 2-deoxy-glucose (2-DG) to inhibit glycolysis. Error bars represent SE; n = 3 in duplicate. *P < 0.05, **P < 0.01, and ***P < 0.001, HEK293-β2AR vs. HEK293-WT (Student’s t-test).
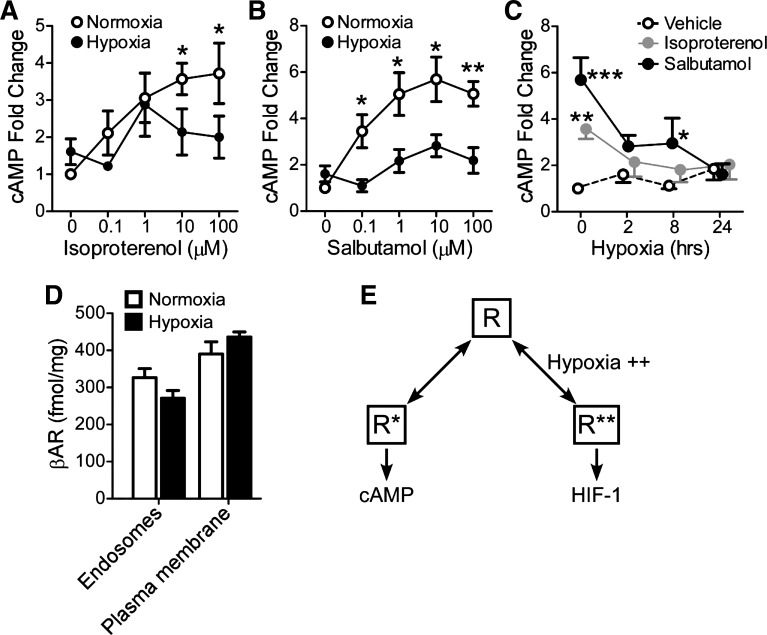
Fig. 5.
Hypoxia blunts cAMP response to isoproterenol and salbutamol in vitro. A and B: human embryonic kidney 293-β2-adrenergic receptor (HEK293-β2AR) cells were exposed to hypoxia (2% O2) or normoxia for 2 h then stimulated with isoproterenol or salbutamol for 5 min. cAMP was measured via fluorescent assay. Data were normalized to vehicle normoxic condition. Error bars represent SE; n = 3–5. *P < 0.05 and **P < 0.01, hypoxia vs. normoxia (paired t-test). C: HEK293-β2AR cells were exposed to hypoxia (2% O2) or normoxia for 2–24 h then stimulated with isoproterenol (10 µM) or salbutamol (10 µM) for 5 min. Data were normalized to vehicle normoxic condition. Error bars represent SE; n = 3–5. *P < 0.05, **P < 0.01 and ***P < 0.001 compared with vehicle (Bonferroni posttest). D: HEK293-β2AR cells were exposed to hypoxia or normoxia for 2 h and then βAR density on plasma membranes and endosomes was measured via [125I]-cyanopindolol binding. Error bars represent SE; n = 7. There were no significant differences between any condition (Student’s t-test). E: theoretical model of βAR regulation based on the data, suggesting hypoxia shifts the equilibrium toward hypoxia-inducible factor 1 (HIF-1) activation, reducing availability of R* and thus blunting cAMP signaling. R, inactive conformational state; R*, cAMP-active conformational state; R**, HIF-1 active conformational state.
Fig. 6.
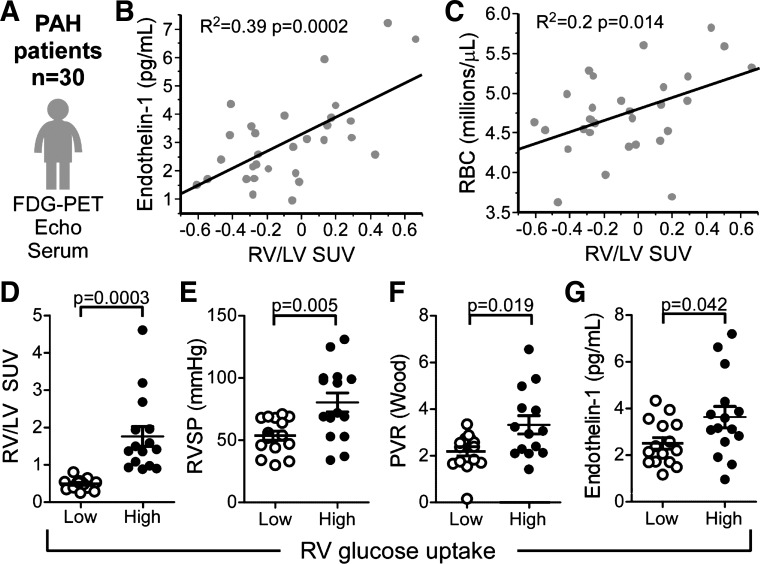
Pulmonary arterial hypertension (PAH) patients with the phenotype of high RV glucose uptake have more severe disease. A: patients with PAH (n = 30) underwent 2-[18F]fluoro-2-deoxy-d-glucose (FDG)-PET scanning to obtain standardized uptake values (SUVs) of glucose in the right ventricle (RV) determined relative to the left ventricle (LV), echocardiogram to estimate cardiac functions and pulmonary vascular resistance (PVR), and blood draw for measure of serum endothelin-1 and red blood cell counts (RBC). B and C: RV/LV SUV correlates with endothelin-1 and RBC. D: patients were dichotomized into high or low RV glucose uptake based on their RV/LV SUV using the log-transformed median as a cutoff for high and low. E–G: PAH patients with high RV glucose uptake had higher right ventricular systolic pressure (RVSP), PVR, and endothelin-1 levels. Error bars represent SE. P values determined from Student’s t-test.
Fig. 7.
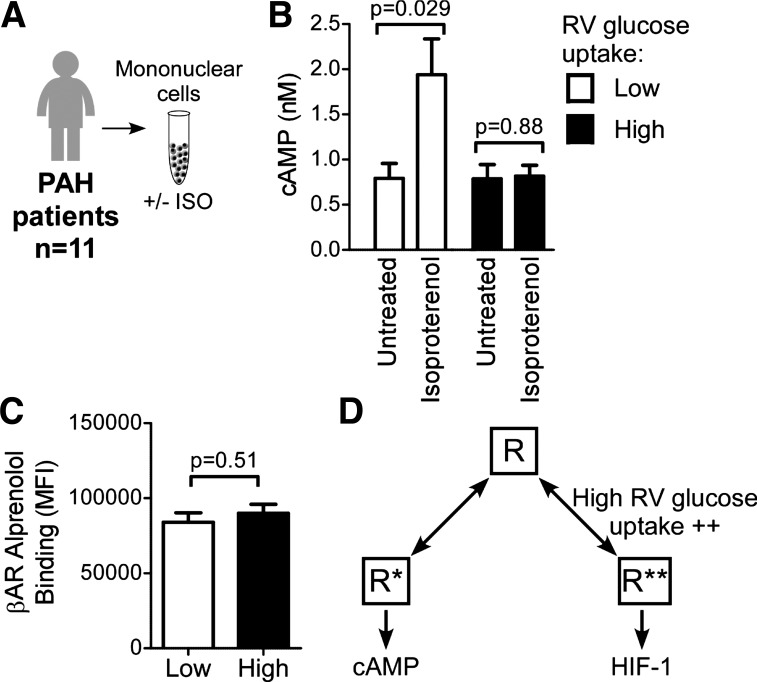
Mononuclear cells from pulmonary arterial hypertension (PAH) patients with the phenotype of high right ventricular (RV) glucose uptake do not produce cAMP in response to isoproterenol (ISO). A: mononuclear cells isolated from PAH patients were treated with 300 µM isoproterenol for 10 min. B: cAMP was measured via fluorescent assay. Error bars represent SE; n = 5–6 per group. P values determined from Student’s t-test. C: β-adrenergic receptor (βAR) density on mononuclear cells from high or low glucose uptake phenotypes of patients is similar. Mononuclear cells were incubated with biotinylated alprenolol, which was detected with phycoerythrin-conjugated streptavidin. Cells were analyzed via flow cytometry to determine relative βAR density based on the median fluorescence intensity (MFI). Error bars represent SE. P values determined from Student’s t-test. D: model of βAR equilibrium in the high glucose uptake PAH phentoype. Patients who have high RV glucose uptake have their βAR shifted toward R** (glycolytic metabolism), and thus less R* availability and inability to activate cAMP signaling. HIF-1, hypoxia-inducible factor 1; R, inactive conformation; R*, conformation that activates cAMP; R**, conformation that activates HIF-1.
Fig. 8.
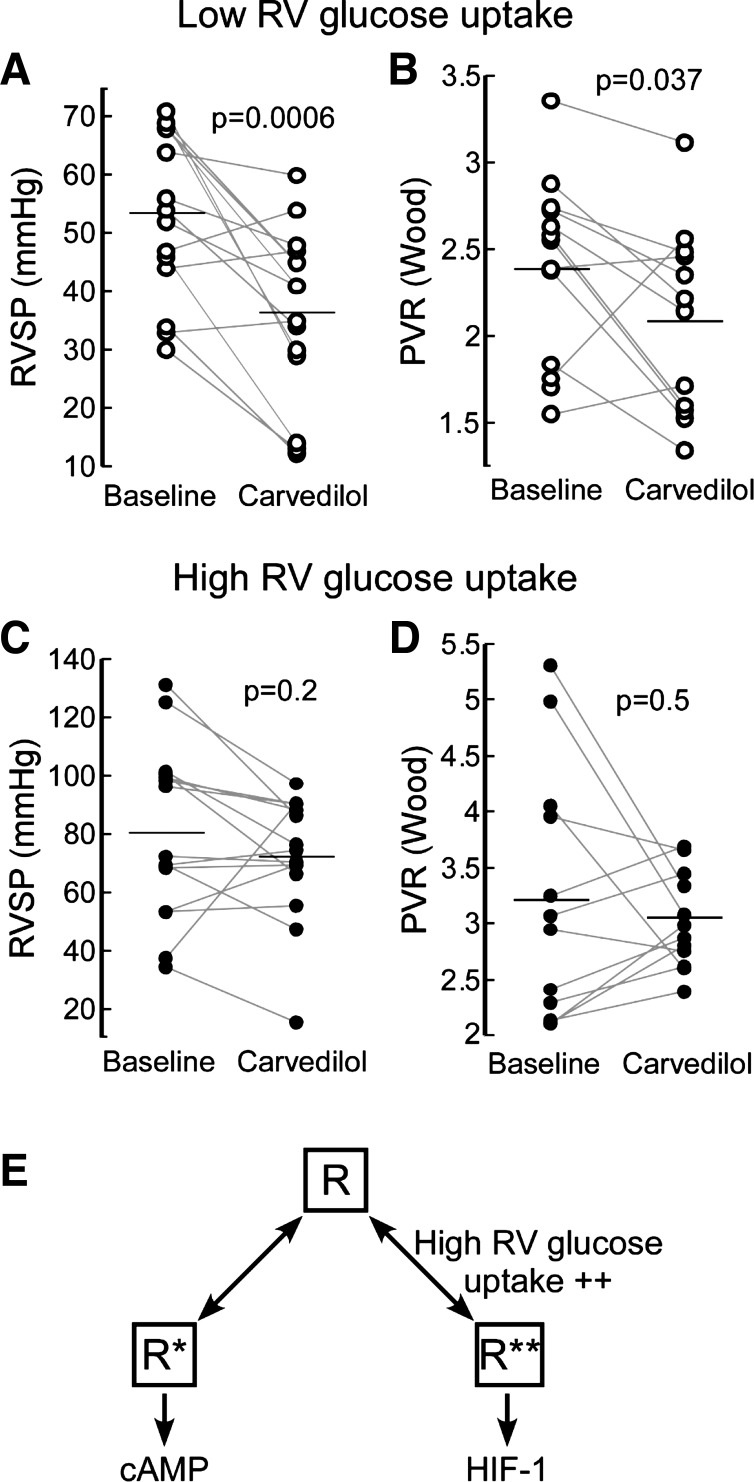
Pulmonary arterial hypertension (PAH) patients with the high right ventricular (RV) glucose uptake phenotype do not respond to carvedilol. PAH patients were all treated with low-dose carvedilol (3.125 mg 2× day) for 1 wk. A–D: right ventricular systolic pressure (RVSP) and pulmonary vascular resistance (PVR) were measured at baseline and after 1-wk treatment in the low (A and B) and high (C and D) RV glucose groups. Bars show means of populations; n = 15 for RVSP and 10 for PVR. P values determined from paired t-test. E: theoretical model of β-adrenergic receptor equilibrium in PAH. Individuals with high RV glucose uptake are shifted toward R** and have less R* available for carvedilol. HIF-1, hypoxia-inducible factor 1; R, inactive conformation; R*, conformation that activates cAMP; R**, conformation that activates HIF-1.
RESULTS
A pharmacological approach was employed to determine how βAR regulation of HIF-1 fits into the overall model of receptor signaling. We utilized HEK293 cells stably transfected with β2AR (HEK293-β2AR), a common model for investigating βAR signaling (1, 43, 51). Cells were treated with isoproterenol and salbutamol, both cAMP agonists, and carvedilol, an inverse agonist, to determine effects on HIF-1 activity. Cells were treated with each ligand or vehicle control in normoxia for 20 h. HIF-1 activity was assessed via an HRE-luciferase reporter transfected into the cells. Vehicle control treatments had no effect on luciferase compared with untreated (luciferase/Renilla mean ± SD: untreated = 2.29 ± 1.03, DMSO = 1.96 ± 2.0, methanol = 2.3 ± 1.4, P = 0.84). All three ligands had dose-dependent effects on HIF-1 activity. Low-dose isoproterenol resulted in a slight decrease in HIF-1 activity, while higher doses increased activity up to 148% above baseline (Fig. 1A). Conversely, salbutamol had a dose-dependent inverse agonist effect, reducing HIF-1 activity by up to 42% (Fig. 1B). Carvedilol acted as partial agonist at 1 µM and an inverse agonist at 10 µM (Fig. 1C).We classified the effect of each drug based on the dose that caused the largest change in HRE-luciferase activity. Thus, isoproterenol and carvedilol act as partial agonists for HIF-1, while salbutamol acts as an inverse agonist. These effects are summarized in Fig. 1D compared with the hypoxia (2% O2) control.
As anticipated, effects of the ligands on cAMP generation confirmed that isoproterenol and salbutamol increased cAMP levels 6.6- and 3.8-fold, respectively, i.e., cAMP agonists (Fig. 1E). Carvedilol has been previously described as an inverse agonist for cAMP (51). This means that carvedilol treatment should decrease cAMP levels below baseline. Since baseline levels of cAMP are at the low end of our detection limit, we tested carvedilol in the presence of IBMX. IBMX is a phosphodiesterase inhibitor that prevents the breakdown of cAMP. This raises the basal cAMP level without affecting cAMP production. This allows us to detect reductions in cAMP production below baseline. Indeed, cAMP levels trended higher in IBMX-treated compared with vehicle-treated subjects (111.3 ± 39.1 vs. 70.8 ± 28.0 nmol/g, P = 0.06). We did not see a significant reduction of cAMP with carvedilol. However, it seems that even with IBMX treatment, cAMP levels were still at the lower detection limit of our assay, so it was not possible to see changes below baseline. Still, the results indicate that βAR regulation of HIF-1 is different from regulation of cAMP. The contrasting effect on HIF-1 between isoproterenol and salbutamol is particularly interesting since both ligands are equivalent agonists for cAMP. This suggests that salbutamol binds the cAMP conformation of the βAR but not the HIF-1 conformation, shifting the equilibrium away from HIF-1 activation (Fig. 1F). Furthermore, carvedilol seems to have a partial agonist effect on HIF-1, despite acting as an inverse agonist for cAMP. This suggests that carvedilol binds the HIF-1 conformation, pulling the equilibrium away from cAMP signaling. Isoproterenol appears to bind and activate both conformations.
These ligands were tested under normoxia. If salbutamol is truly an inverse agonist, we would expect inhibition of HIF-1 under hypoxia as well. To test this we pretreated HEK293-β2AR cells with isoproterenol, salbutamol, or carvedilol for 45 min before 20 h hypoxia (2% O2) exposure. As expected, salbutamol reduced hypoxia-induced HIF-1 activity by ~15% (Fig. 2B). This additional evidence suggests that salbutamol shifts the equilibrium of receptor conformations away from HIF-1 activation. This reduces the availability for HIF-1 activation under hypoxia. Interestingly, though both function as partial agonists under normoxia, only carvedilol increased HIF-1 activity under hypoxia (Fig. 2, A and C). These results are summarized in Fig. 2D.
Fig. 2.
β-Adrenergic receptor (βAR) ligands differentially affect hypoxia-inducible factor 1 activity in vitro. A–C: human embryonic kidney cells overexpressing the β2AR (HEK293-β2AR) were treated for 45 min with isoproterenol (A), salbutamol (B), or carvedilol (C) then exposed to hypoxia (2% O2) for 20 h. Data are represented as % change in hypoxia-response element (HRE)-luciferase activity compared with the vehicle control. D: summary of A–C HRE-luciferase effects with 30 µM isoproterenol, 100 µM salbutamol, and 1 µM carvedilol. Error bars represent SE; n = 3–4 in triplicate. *P < 0.05, **P < 0.01, and ***P < 0.001 compared with vehicle control (Bonferroni posttest).
To assess the physiologic effects of βAR in vivo, we examined the effect of these ligands in mice. In these experiments, isoproterenol and salbutamol were used to discriminate differential effects on HIF-1α based on the in vitro findings in HEK293 cells. Mice were treated with isoproterenol, salbutamol, or vehicle via intraperitoneal injection in normoxic conditions 2 h before euthanization. Isoproterenol had a dose-dependent effect on HIF-1α levels. Only 5 mg/kg isoproterenol increased HIF-1α levels, while the higher or lower doses (1 and 10 mg/kg) had no effect. Salbutamol-treated mice had no significant change in HIF-1α at any dose compared with vehicle controls (Fig. 3, A and B). The group treated with 1 mg/kg salbutamol had greater variability compared with vehicle; however, this was primarily driven by one outlier. No other abnormalities were observed with this mouse, so it was not excluded from analysis. If the outlier was excluded, the mean fold change (± SD) would drop to from 2.1 ± 2.2 to 1.1 ± 1.1.
Classically, HIF-1 stimulates erythropoiesis which can be measured via myeloid erythroid progenitor levels (5, 26). Erythroid progenitor populations in the bone marrow were measured via flow cytometry (26). Proerythroblasts (Stage I), basophilic erythroblasts (Stage II), polychromatic erythroblasts (Stage III), and orthochromatic erythroblasts (Stage IV) were determined based on expression of TER119, CD44, and size (Fig. 3, C and D). Normal erythroid development shows a 1:2:4:8 ratio from Stage I to Stage IV, which is shown in the vehicle-treated mice (Fig. 3, E–G). The isoproterenol-treated mice had increased proportions of erythroid progenitor cells at every dose compared with controls, while the salbutamol-treated mice had decreased proportions of progenitors (Fig. 3, E–G). Overall, isoproterenol induced a HIF response, while we could not detect changes in HIF-1α with salbutamol. Furthermore, erythropoiesis, a sensitive biological marker of HIF-1, was increased with isoproterenol but reduced with salbutamol. Altogether, this data supports our in vitro data, demonstrating opposing effects of these ligands on HIF-1.
If there is an independent receptor conformation that activates HIF-1, we would expect to see basal HIF-1 signaling through this conformation. To test this, we compared HEK293-β2AR cells with native, non-overexpressing cells (HEK293-WT). We reasoned that if HIF-1 signaling occurs via a specific receptor confirmation, increased expression of βAR in cells would increase spontaneous basal signaling via HIF-1. Our HEK293-β2AR cells have ~32-fold increase in β2AR as compared with the WT cells (mean ± SD: 550.1 ± 22.89 vs. 16.86 ± 0.6 fmol/mg, P < 0.001) (Fig. 4A). HRE-luciferase was used to measure basal HIF-1 activity in both cell types. HIF-1 activity was increased in the HEK293-β2AR cells compared with the WT cells (Fig. 4B). To determine the functional consequences of this increased basal signaling, we compared glycolytic rates in both cell types. Hypoxia results in a shift toward increased glycolysis (52), due in part to transcriptional regulation of glycolytic enzymes by HIF-1 (42). Thus, we would expect HEK293-β2AR cells to have increased levels of glycolysis. Glycolysis was determined based on the extracellular acidification rate (ECAR) using a Seahorse XFe24 Analyzer (Agilent). The test measures ECAR at baseline, after glucose stimulation, following inhibition of oxidative phosphorylation with oligomycin, and after 2-DG treatment that inhibits glycolysis. Estimated glycolysis can be calculated by subtracting the baseline ECAR from glucose-stimulated ECAR. Overexpression of the β2AR caused an increase in glycolysis compared with the HEK293-WT cells. (Fig. 4C). To confirm that this effect was due to increased HIF-1 activity, we pretreated cells with two HIF-1 inhibitors. Digoxin inhibits HIF-1α protein synthesis (56), and chrysin increases ubiquitination and degradation of HIF-1α (10). Pretreatment with digoxin (100 nM) or chrysin (30 µM) decreased glycolysis in the HEK293-β2AR but had no effect on the WT cells (Fig. 4C). In addition to increased glucose-stimulated ECAR, HEK293-β2AR also had higher maximal ECAR with oligomycin compared with WT cells (Fig. 4D). Chrysin and digoxin treatments reduced both glucose-stimulated and maximal ECAR in HEK293-β2AR cells (Fig. 4, E and F). The differences were lost after 2-DG treatment, which confirms that the changes in ECAR were due to changes in glycolysis. Overall, these results indicate that β2AR overexpression leads to increased HIF-1 activity and higher levels of glycolysis. This is consistent with increased basal signaling due to increased receptor expression. This supports the theoretical βAR model for HIF-1 regulation via a unique receptor conformation that is capable of spontaneous basal signaling.
Next, the effect of hypoxia on βAR cAMP signal transduction was evaluated. If hypoxia shifts the equilibrium toward a HIF signaling conformation, we would expect a reduction in βAR that is accessible for agonist induction of cAMP signaling. To test this, HEK293-β2AR cells were exposed to hypoxia for 2 h then stimulated with isoproterenol, salbutamol, or vehicle for 5 min. Levels of cAMP were measured via a fluorescent assay. Vehicle control treatment had no significant effect on cAMP compared with untreated (cAMP nmol/g mean ± SD: untreated = 518.7 ± 199.9, methanol = 379.5 ± 166.7, P = 0.25). Under normoxia, isoproterenol and salbutamol treatment resulted dose-dependent increases in cAMP levels (Fig. 5, A and B). However, after 2 h hypoxia exposure, cAMP response to isoproterenol and salbutamol was blunted by up to 46% and 57%, respectively, compared with normoxia (Fig. 5, A and B). The cAMP responses to low doses of isoproterenol (≤1 µM) under hypoxia were similar to normoxia. This may represent subsets of βAR that are in a high-affinity state and are primed for cAMP signaling (44), which appear to be less sensitive to hypoxia. Longer exposure to hypoxia (8–24 h) continued to blunt cAMP response to both agonists (Fig. 5C). After 2, 8, or 24 h hypoxia, isoproterenol induced no significant response compared with the vehicle treatment. There was a slight recovery of response to salbutamol after 8 h hypoxia; however, after 24 h, the response was completely dampened. To determine whether the reduction in cAMP response was due to increased receptor internalization or degradation, we measured the βAR density on plasma membranes and endosomes. βARs were measured after 2 h hypoxia or normoxia via [125I]-cyanopindolol (125I-CYP) binding. The receptor density in either plasma membranes or endosomes was no different under normoxia versus hypoxia (P = 0.48, plasma membrane; P = 0.61, endosomes) (Fig. 5D). This suggests hypoxia leads to a receptor conformation less accessible to ligands for activation of cAMP, i.e., hypoxia shifts the equilibrium of receptor conformations toward a HIF-1 activating state (R**), reducing availability to activate cAMP (Fig. 5E).
To investigate these effects in a pathophysiologic context, patients with PAH were studied in an ancillary study to the clinical trial of Pulmonary Arterial Hypertension Treatment with Carvedilol for Heart Failure (PAHTCH) (NCT01586156). The PAHTCH trial enrolled 30 PAH patients and 12 healthy controls at baseline. PAH patients received 1-wk open-label low-dose carvedilol at 3.125 mg twice daily before randomization to the main double-blind study. Characteristics of the population and results of the main study have been previously published (6, 8). As previously reported, PAH was characterized by greater levels of endothelin-1 (ET-1) and RV glucose standardized uptake values (SUVs) relative to the LV (RV/LV SUV) as measured by 2-[18F]fluoro-2-deoxy-d-glucose (FDG) positron emission tomography (PET) scan (8, 40). Although PAH patients had overall greater RV glucose uptake than controls, there was heterogeneity of uptake among PAH participants. In fact, the RV/LV SUV correlated to ET-1 and the red blood cell count (RBC), suggesting that RV glucose uptake could serve as a surrogate marker of HIF-1 activation (Fig. 6, B and C). Using the median of the RV/LV SUV, there was a clear grouping of PAH patients with high and low glycolytic phenotypes (Fig. 6D). The high RV glucose uptake group had a more severe disease phenotype, with higher right ventricular systolic pressure (RVSP), pulmonary vascular resistance (PVR), ET-1 levels, and lower cardiac index compared with the low group (Fig. 6, E–G) (Table 1).
Table 1.
Characteristics of PAH patients with phenotype of high or low RV glucose uptake
| High RV Glucose Uptake | Low RV Glucose Uptake | t-Test P Value | |
|---|---|---|---|
| n | 15 | 15 | |
| Sex (n, male/female) | 3/12 | 6/9 | |
| Race (n, Caucasian/African American/Asian) | 11/4/0 | 12/2/1 | |
| Age, yr | 42.7 ± 12.9 | 45.9 ± 10.8 | 0.47 |
| Weight, kg | 86.1 ± 20.7 | 78.9 ± 18.6 | 0.33 |
| Heart rate, beats/min | 78.9 ± 7.4 | 74.8 ± 8.4 | 0.16 |
| Systolic blood pressure, mmHg | 112.9 ± 15.0 | 120.7 ± 18.1 | 0.21 |
| Diastolic blood pressure, mmHg | 71.5 ± 12.3 | 71.3 ± 12.0 | 0.96 |
| Oxygen saturation, % | 95.9 ± 2.2 | 96.7 ± 2.7 | 0.38 |
| RVSP, mmHg | 80.3 ± 29.6 | 53.7 ± 14.3 | 0.005 |
| PVR, Wood units | 3.3 ± 1.5 | 2.2 ± 0.8 | 0.019 |
| Cardiac index, L·min−1·m−2 | 2.18 ± 0.67 | 2.76 ± 0.80 | 0.04 |
| 6-Min walk distance, feet | 1,480.7 ± 328.6 | 1,545.3 ± 438.8 | 0.65 |
| RBC, millions/µL | 4.9 ± 0.6 | 4.6 ± 0.4 | 0.18 |
| Hemoglobin, g/dL | 13.7 ± 2.1 | 13.3 ± 1.8 | 0.56 |
| Glucose, mg/dL | 83.7 ± 16.2 | 87.9 ± 16.2 | 0.48 |
| RV/LV SUV ratio | 1.8 ± 1.0 | 0.5 ± 0.1 | 0.0003 |
| Endothelin-1, pg/mL | 3.6 ± 1.8 | 2.5 ± 0.96 | 0.042 |
| Urinary cAMP/creatinine, µmol/g | 1.3 ± 0.5 | 0.9 ± 0.7 | 0.04* |
| β-Adrenergic receptor density (median fluorescence intensity × 103) | 89.9 ± 22.6 | 84.1 ± 24.2 | 0.51 |
| Urinary cGMP/creatinine, nmol/g | 122.5 ± 47.3 | 117.8 ± 103.7 | 0.87 |
Values are means ± SD; P value from Student’s t-test. LV, left ventricular; PAH, pulmonary arterial hypertension; PVR, pulmonary vascular resistance; RBC, red blood cell; RV, right ventricular; RVSP, right ventricular systolic pressure; SUV, standardized uptake value.
P value from Mann-Whitney U test.
To determine whether shift toward HIF-1 activation is associated with diminished availability for cAMP signaling, mononuclear cells were isolated at baseline from a subgroup of the PAH participants (n = 11) and exposed to 300 µM isoproterenol for 10 min (Fig. 7A). The experiment was performed blinded to levels of FDG-PET RV glucose uptake. After unblinding of samples, analyses of results showed that cells from low RV glucose patients had a 2.4-fold increase in cAMP with isoproterenol (n = 5), while cells from high RV glucose uptake patients did not respond to isoproterenol (n = 6) (Fig. 7B). This was consistent with our previous results showing blunted cAMP signaling in HEK293-β2AR cells under hypoxia. The βAR levels were also measured in mononuclear cells from participants. Mononuclear cells were incubated with biotinylated alprenolol, which was detected with phycoerythrin-conjugated streptavidin and measured via flow cytometry. Alprenolol binding was similar among the high and low RV glucose uptake groups, confirming that changes in cAMP response were not due to changes in receptor levels (Fig. 7C). Based on these results and the paradigm for receptor conformations, the βAR in high RV glucose uptake PAH patients is shifted toward a HIF-1- (R**) activating state, reducing levels of R and R* availability to activate cAMP (Fig. 7D). Next, we used the 1-wk, open-label low-dose (3.125 mg) carvedilol intervention of PAHTCH to investigate whether the RV glucose phenotype might predict responders to the βAR inverse agonist carvedilol. Analogous to the ex vivo studies in the mononuclear cells from patients, the low RV glucose uptake group had a significant response to carvedilol, measured as a drop in RVSP and PVR (Fig. 8, A and B), while the high group had no response (Fig. 8, C and D). Furthermore, the low RV glucose uptake group had a significant decrease in systolic blood pressure after 1 wk carvedilol treatment (baseline: 120.7 ± 18.1 mmHg, carvedilol: 113.5 ± 13.9 mmHg, P = 0.033, paired t-test). The high RV glucose uptake group had no change in blood pressure with carvedilol (baseline: 112.9 ± 15.0 mmHg, carvedilol: 110.9 ± 14.2 mmHg, P = 0.48, paired t-test). These results support the proposed model that PAH patients with high RV glucose uptake have a shifted equilibrium of βAR to the hypoxia-responsive state and blunted cAMP response to ligands (Fig. 8E).
DISCUSSION
There is increasing recognition of the complexity in βAR signaling as new pathways continue to be discovered. Regulation of HIF-1 is one of those newly discovered pathways wherein our previous studies (5) showed the necessity of βAR for HIF-1α stabilization. However, the current model of receptor signaling for cAMP and HIF-1 responses was unknown. Here, our results expand the current paradigm of the βAR receptor conformation model. Multiple unique receptor conformations are in equilibrium, leading to low-level basal activation of HIF-1 and cAMP. Bias toward different conformations (via ligands or hypoxia) leads to cAMP and/or HIF-1 signaling by shifting the equilibrium of receptors toward or away from specific pathways.
The two-state model of GPCR signaling proposed that receptors exist in two conformations, an active (R*) and an inactive (R), which are in equilibrium (23). The active conformation can signal in the absence of a ligand, resulting in constitutive baseline signaling. Ligand binding stabilizes a particular receptor state, shifting the equilibrium toward that state. With this model, ligands can be classified based on their relative affinities for R and R*. Agonists have higher affinity for the active conformation, stabilizing R* and shifting the equilibrium toward an active state. Inverse agonists have a higher affinity for R, shifting the equilibrium toward an inactive state and decreasing signaling below baseline. Antagonists have equal affinity for both R and R*, stabilizing both and preventing the action of agonists and inverse agonists. However, the discovery of multiple independent signaling pathways through a single receptor revealed the shortcomings of this prior model. The fact that ligands have different efficacies toward one pathway compared with another supports the idea of multiple active conformations (1, 11, 36). This led to the development of a three-state model of GPCR signaling (24). This model extends the two-state model to include an additional active state (R**) that signals through a different pathway than R*. Ligand bias toward one pathway over another depends on the relative affinity of a ligand for R* versus R**. More recent work suggests the possibility of even more active conformations (49). Consistently, structural studies of the βAR demonstrate that there is considerable heterogeneity in the receptor conformations stabilized by different ligands, even among ligands with similar signaling profiles (17, 29). This indicates that our current understanding of the receptor signaling is incomplete, and as more pathways are discovered, the model may expand to accommodate the increasing complexity of GPCR signaling.
Based on our results, we propose to broaden the model to include an active R** conformation that specifically activates HIF-1. We propose HIF-1 activation occurs via a βAR conformation that is different from the conformation that activates cAMP. This is based on our results, which show isoproterenol and salbutamol, both agonists for cAMP, have differential effects on HIF-1 activity. Only isoproterenol activated HIF-1, while salbutamol reduced HIF-1 activity. This indicates that HIF-1 signaling is not downstream of cAMP but an independent parallel pathway. We theorize that the difference in the isoproterenol and salbutamol effect on HIF-1 is due to differences in their relative affinities for one conformation over another. Although both can bind and activate cAMP signaling, isoproterenol is able to bind and stabilize the HIF-1 conformation, while salbutamol cannot. This is supported by our data, which showed isoproterenol acted as an agonist for both HIF-1 and cAMP, stimulating both under normoxia. Salbutamol stimulated cAMP production but had an inverse agonist effect on HIF-1, which was maintained under hypoxia. This difference was maintained in vivo with isoproterenol increasing HIF-1α levels as well as erythropoiesis in mice under normoxia, while salbutamol decreased erythropoiesis. By binding and stabilizing the cAMP-generating conformation but not the HIF-1 conformation, salbutamol shifts the equilibrium toward cAMP and reduces the number of receptors available to activate HIF-1. The dose-dependent effects of ligands on HIF-1 were quite variable. The varying effects could be due to the known receptor affinity states (i.e., high or low). With agonist signaling, there is often a biphasic or variable response to ligands, with an initial response to low doses, followed by a secondary response to higher doses (14, 44, 45). This could explain the variability in response to different doses, although more work is needed to understand this variability. Based on the model, the conformations are in equilibrium at baseline, leading to low levels of basal signaling. This is supported by the fact that simply overexpressing the βAR leads to increased HIF-1 activity and glycolysis, a HIF-1-driven function. Interestingly, digoxin blunted glycolysis in βAR-overexpressing cells. Digoxin is used clinically to treat left heart failure. Heart failure is often associated with increased HIF-1 and a shift toward glycolysis (41). Based on our results, we speculate that the beneficial effects of digoxin may be due, in part, to inhibition of these pathological, HIF-1-driven metabolic changes.
Oxygen is the main regulator of HIF-1 and likely plays a pivotal role in βAR signaling. Previous studies suggest the βAR is involved in hypoxia sensing. The βAR is hydroxylated in an oxygen-dependent manner which leads to ubiquitination and degradation (53). Additionally, hypoxia induces a unique phosphorylation pattern or “barcode” on the βAR (5). The phosphorylation barcode of the βAR regulates downstream signaling, with different barcodes leading to distinct signaling pathways (35). Here we find that placing cells in hypoxia before agonist stimulation reduced cAMP levels by up to 57%. We confirmed the change in signaling is not due to a change in receptor amount. We found hypoxia exposure had no effect on receptor density in the plasma membrane or endosomes. This means that, under hypoxia, the receptor is still present on the cell surface but cannot activate the cAMP pathway in response to agonist stimulation. Our findings support the idea that hypoxia stabilizes the receptor in a conformation that activates HIF-1 (R**), shifting the equilibrium away from R* and thus reducing the availability for cAMP signaling. Thus, hypoxia mimics the effect of an agonist, stabilizing and activating HIF-1 signaling through βAR. However, hypoxia did not induce receptor internalization or degradation. This is in contrast to cAMP agonists isoproterenol and salbutamol. Acutely, both agonists induce receptor phosphorylation within 1–5 min of treatment, which leads to immediate desensitization of the receptor. This is followed by receptor internalization starting around 5 min and continuing for up to 30 min with treatment (16, 22, 50). Therefore, receptor density at the plasma membrane is reduced. Longer treatment with agonist (3–24 h) results in sustained downregulation of surface receptors. Long-term agonist exposure also decreases receptor protein and mRNA levels (3, 20). Thus, although the effect of hypoxia on the βAR is similar to an agonist, the mechanism of stabilization is likely different.
This expanded model of βAR regulation can help us understand the variable responses to β-blockers in PAH and perhaps in heart failure in general. Knowledge of ligand bias and regulation of signaling pathways of other GPCRs has already led to the development of new drugs that preferentially activate beneficial pathways while simultaneously blocking deleterious pathways (30, 38). Thus, understanding the complexities of the βAR signaling model is imperative to improving and developing new treatments. β-blockers have been investigated for treatment of right heart failure in PAH with variable results. In one study, bisoprolol reduced cardiac index and 6-min walk distance (48). However, another showed that bisoprolol reduced RV dilation and improved tricuspid annular plane systolic excursion (33). Retrospective studies of β1-blockers show no difference in hemodynamic or RV parameters in treated groups (2, 46). Recent studies focusing on third generation β-blockers are promising. A clinical trial showed that carvedilol improved right ventricular ejection fraction (RVEF) and reduced RVSP (13). Another showed that nebivolol lowered pulmonary artery systolic pressure, RV size, and blood endothelin-1 levels (32). Our 6-mo clinical trial investigating carvedilol in PAH demonstrated efficacy of the treatment (8). However, within the cohort of 30 subjects, there was variable response to carvedilol, and subjects could be classified as responders or nonresponders based on cardiac functions and functional improvements (6).
Our new model of βAR regulation may help explain this phenomenon and underlying mechanisms. Here, we classified subjects based on their HIF-1/glycolytic phenotype. We used RV glucose uptake as a marker for HIF-1 signaling activation. Increased glucose uptake suggests a metabolic shift toward glycolysis, a HIF-1-regulated effect (9, 42). While increased glucose uptake has been directly correlated with HIF-1α levels in rat models of PAH and in human tumors (18, 19, 31), we were not able to directly correlate glucose uptake with HIF-1 in our subjects. However, we found that RV glucose uptake was positively correlated with ET-1 and RBC, both HIF-1-regulated effects. The RV glucose uptake defined two phenotypes of PAH, the one with high glucose uptake having much more severe clinical disease and poor response to carvedilol. The low RV glucose uptake group responded well to carvedilol, with lower RVSP and PVR after 1 wk of treatment. It is interesting to consider this differential response in the context of our current and previous studies. In our in vitro studies, carvedilol acted as a partial agonist for HIF-1 activity. Although partial agonists induce low levels of signaling, physiologically this signaling is negligible. In fact, the overall effect of partial agonists is to blunt endogenous signaling responses by blocking receptor activation. This is consistent with the results from our previous study demonstrating that pretreatment with carvedilol blunts HIF-mediated erythropoiesis in mice under hypoxia (5). We speculate that carvedilol holds the receptor in conformations that precludes a robust HIF-1 response. About half of individuals with PAH are shifted toward high glucose uptake (i.e., HIF-1 signaling) phenotype. We theorize that patients with high glucose uptake have a bias toward the βAR conformation that activates HIF-1. In these people, carvedilol is unable to prevent HIF-1 activation, as their receptor conformation is already biased toward HIF-1 signaling. In the low-uptake subjects, carvedilol may serve to inhibit cAMP and blunt HIF-1 activation. This explains the differential response to carvedilol between these two groups. It is interesting to speculate that the high glucose uptake group might respond to HIF-1 inhibitors (e.g., digoxin).
Here we have expanded the model of βAR signaling to include a new parallel pathway through HIF-1. HIF-1 signaling occurs through a unique receptor conformation that is part of the equilibrium spectrum of receptor conformations. Understanding this equilibrium and how it affects βAR signaling is essential for understanding current therapies as well as developing new ones. Specifically, we find that the PAH response to βAR ligands differs depending on how this equilibrium is shifted. This knowledge could help predict who would be a good candidate for β-blocker treatment. Knowledge of βAR regulation may also have more broad implications. Signaling bias, or a receptor’s ability to activate a selective subset of downstream pathways, is a powerful tool for specifically activating beneficial signals while simultaneously blocking deleterious signals. Thus, an in-depth understanding of the βAR signaling model and how ligands affect different pathways could lead to development of new compounds that selectively activate only desired pathways.
GRANTS
This work is supported by National Heart, Lung, and Blood Institute Grants HL115008 and HL060917 and American Heart Association Grant 17PRE33660021. O. R. Stephens is in the Molecular Medicine PhD Program of the Cleveland Clinic Lerner College of Medicine and Case Western Reserve University. S. C. Erzurum is supported in part by the Alfred Lerner Memorial Chair in Innovative Biomedical Research.
DISCLOSURES
No conflicts of interest, financial or otherwise, are declared by the authors.
AUTHOR CONTRIBUTIONS
O.R.S., K.A., S.F., K.B.H., S.V.N.P., and S.C.E. conceived and designed research; O.R.S., K.W., M.F., J.A.R., Y.S., K.B.H., and S.V.N.P. performed experiments; O.R.S., S.F., K.B.H., S.V.N.P., and S.C.E. analyzed data; O.R.S., K.A., S.F., K.B.H., S.V.N.P., and S.C.E. interpreted results of experiments; O.R.S. and S.C.E. prepared figures; O.R.S. and S.C.E. drafted manuscript; O.R.S., K.A., S.F., K.B.H., S.V.N.P., and S.C.E. edited and revised manuscript; O.R.S. and S.C.E. approved final version of manuscript.
ACKNOWLEDGMENTS
We thank Hoi I. Cheong for valuable insights, advice, and assistance in developing this project.
REFERENCES
- 1.Azzi M, Charest PG, Angers S, Rousseau G, Kohout T, Bouvier M, Piñeyro G. β-Arrestin-mediated activation of MAPK by inverse agonists reveals distinct active conformations for G protein-coupled receptors. Proc Natl Acad Sci USA 100: 11406–11411, 2003. doi: 10.1073/pnas.1936664100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bandyopadhyay D, Bajaj NS, Zein J, Minai OA, Dweik RA. Outcomes of β-blocker use in pulmonary arterial hypertension: a propensity-matched analysis. Eur Respir J 46: 750–760, 2015. doi: 10.1183/09031936.00215514. [DOI] [PubMed] [Google Scholar]
- 3.Bouvier M, Collins S, O’Dowd BF, Campbell PT, de Blasi A, Kobilka BK, MacGregor C, Irons GP, Caron MG, Lefkowitz RJ. Two distinct pathways for cAMP-mediated down-regulation of the beta 2-adrenergic receptor. Phosphorylation of the receptor and regulation of its mRNA level. J Biol Chem 264: 16786–16792, 1989. [PubMed] [Google Scholar]
- 4.Bristow MR, Minobe W, Rasmussen R, Larrabee P, Skerl L, Klein JW, Anderson FL, Murray J, Mestroni L, Karwande SV. Beta-adrenergic neuroeffector abnormalities in the failing human heart are produced by local rather than systemic mechanisms. J Clin Invest 89: 803–815, 1992. doi: 10.1172/JCI115659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Cheong HI, Asosingh K, Stephens OR, Queisser KA, Xu W, Willard B, Hu B, Dermawan JKT, Stark GR, Naga Prasad SV, Erzurum SC. Hypoxia sensing through β-adrenergic receptors. JCI Insight 1: e90240, 2016. doi: 10.1172/jci.insight.90240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Cheong HI, Farha S, Park MM, Thomas JD, Saygin D, Comhair SAA, Sharp J, Highland KB, Tang WHW, Erzurum SC. Endothelial phenotype evoked by low dose carvedilol in pulmonary hypertension. Front Cardiovasc Med 5: 180, 2018. doi: 10.3389/fcvm.2018.00180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Dong RQ, Wang ZF, Zhao C, Gu HR, Hu ZW, Xie J, Wu YQ. Toll-like receptor 4 knockout protects against isoproterenol-induced cardiac fibrosis: the role of autophagy. J Cardiovasc Pharmacol Ther 20: 84–92, 2015. doi: 10.1177/1074248414539564. [DOI] [PubMed] [Google Scholar]
- 8.Farha S, Saygin D, Park MM, Cheong HI, Asosingh K, Comhair SA, Stephens OR, Roach EC, Sharp J, Highland KB, DiFilippo FP, Neumann DR, Tang WHW, Erzurum SC. Pulmonary arterial hypertension treatment with carvedilol for heart failure: a randomized controlled trial. JCI Insight 2: 95240, 2017. doi: 10.1172/jci.insight.95240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Fijalkowska I, Xu W, Comhair SA, Janocha AJ, Mavrakis LA, Krishnamachary B, Zhen L, Mao T, Richter A, Erzurum SC, Tuder RM. Hypoxia inducible-factor1α regulates the metabolic shift of pulmonary hypertensive endothelial cells. Am J Pathol 176: 1130–1138, 2010. doi: 10.2353/ajpath.2010.090832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Fu B, Xue J, Li Z, Shi X, Jiang BH, Fang J. Chrysin inhibits expression of hypoxia-inducible factor-1α through reducing hypoxia-inducible factor-1α stability and inhibiting its protein synthesis. Mol Cancer Ther 6: 220–226, 2007. doi: 10.1158/1535-7163.MCT-06-0526. [DOI] [PubMed] [Google Scholar]
- 11.Galandrin S, Bouvier M. Distinct signaling profiles of β1 and β2 adrenergic receptor ligands toward adenylyl cyclase and mitogen-activated protein kinase reveals the pluridimensionality of efficacy. Mol Pharmacol 70: 1575–1584, 2006. doi: 10.1124/mol.106.026716. [DOI] [PubMed] [Google Scholar]
- 12.Gonzalez JP, Ramachandran J, Xie LH, Contreras JE, Fraidenraich D. Selective connexin43 inhibition prevents isoproterenol-induced arrhythmias and lethality in muscular dystrophy mice. Sci Rep 5: 13490, 2015. [Erratum in Sci Rep 5: 15315, 2015.] doi: 10.1038/srep13490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Grinnan D, Bogaard HJ, Grizzard J, Van Tassell B, Abbate A, DeWilde C, Priday A, Voelkel NF. Treatment of group I pulmonary arterial hypertension with carvedilol is safe. Am J Respir Crit Care Med 189: 1562–1564, 2014. doi: 10.1164/rccm.201311-2025LE. [DOI] [PubMed] [Google Scholar]
- 14.Hoffman BB, Lefkowitz RJ. Radioligand binding studies of adrenergic receptors: new insights into molecular and physiological regulation. Annu Rev Pharmacol Toxicol 20: 581–608, 1980. doi: 10.1146/annurev.pa.20.040180.003053. [DOI] [PubMed] [Google Scholar]
- 15.Hu W, Zhang L, Li MX, Shen J, Liu XD, Xiao ZG, Wu DL, Ho IHT, Wu JCY, Cheung CKY, Zhang YC, Lau AHY, Ashktorab H, Smoot DT, Fang EF, Chan MTV, Gin T, Gong W, Wu WKK, Cho CH. Vitamin D3 activates the autolysosomal degradation function against Helicobacter pylori through the PDIA3 receptor in gastric epithelial cells. Autophagy 15: 707–725, 2019. doi: 10.1080/15548627.2018.1557835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.January B, Seibold A, Whaley B, Hipkin RW, Lin D, Schonbrunn A, Barber R, Clark RB. β2-adrenergic receptor desensitization, internalization, and phosphorylation in response to full and partial agonists. J Biol Chem 272: 23871–23879, 1997. doi: 10.1074/jbc.272.38.23871. [DOI] [PubMed] [Google Scholar]
- 17.Kahsai AW, Xiao K, Rajagopal S, Ahn S, Shukla AK, Sun J, Oas TG, Lefkowitz RJ. Multiple ligand-specific conformations of the β2-adrenergic receptor. Nat Chem Biol 7: 692–700, 2011. doi: 10.1038/nchembio.634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kaira K, Okumura T, Ohde Y, Takahashi T, Murakami H, Oriuchi N, Endo M, Kondo H, Nakajima T, Yamamoto N. Correlation between 18F-FDG uptake on PET and molecular biology in metastatic pulmonary tumors. J Nucl Med 52: 705–711, 2011. doi: 10.2967/jnumed.111.087676. [DOI] [PubMed] [Google Scholar]
- 19.Kasahara N, Kaira K, Bao P, Higuchi T, Arisaka Y, Erkhem-Ochir B, Sunaga N, Ohtaki Y, Yajima T, Kosaka T, Oyama T, Yokobori T, Asao T, Nishiyama M, Tsushima Y, Kuwano H, Shimizu K, Mogi A. Correlation of tumor-related immunity with 18F-FDG-PET in pulmonary squamous-cell carcinoma. Lung Cancer 119: 71–77, 2018. doi: 10.1016/j.lungcan.2018.03.001. [DOI] [PubMed] [Google Scholar]
- 20.Kassis S, Fishman PH. Functional alteration of the beta-adrenergic receptor during desensitization of mammalian adenylate cyclase by beta-agonists. Proc Natl Acad Sci USA 81: 6686–6690, 1984. doi: 10.1073/pnas.81.21.6686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Keränen T, Hömmö T, Hämäläinen M, Moilanen E, Korhonen R. Anti-inflammatory effects of β2-receptor agonists salbutamol and terbutaline are mediated by MKP-1. PLoS One 11: e0148144, 2016. doi: 10.1371/journal.pone.0148144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Krasel C, Bünemann M, Lorenz K, Lohse MJ. β-Arrestin binding to the β2-adrenergic receptor requires both receptor phosphorylation and receptor activation. J Biol Chem 280: 9528–9535, 2005. doi: 10.1074/jbc.M413078200. [DOI] [PubMed] [Google Scholar]
- 23.Leff P. The two-state model of receptor activation. Trends Pharmacol Sci 16: 89–97, 1995. doi: 10.1016/S0165-6147(00)88989-0. [DOI] [PubMed] [Google Scholar]
- 24.Leff P, Scaramellini C, Law C, McKechnie K. A three-state receptor model of agonist action. Trends Pharmacol Sci 18: 355–362, 1997. doi: 10.1016/S0165-6147(97)01105-X. [DOI] [PubMed] [Google Scholar]
- 25.Li H, Lu ZZ, Chen C, Song Y, Xiao H, Zhang YY. Echocardiographic assessment of β-adrenoceptor stimulation-induced heart failure with reduced heart rate in mice. Clin Exp Pharmacol Physiol 41: 58–66, 2014. doi: 10.1111/1440-1681.12176. [DOI] [PubMed] [Google Scholar]
- 26.Liu J, Zhang J, Ginzburg Y, Li H, Xue F, De Franceschi L, Chasis JA, Mohandas N, An X. Quantitative analysis of murine terminal erythroid differentiation in vivo: novel method to study normal and disordered erythropoiesis. Blood 121: e43–e49, 2013. doi: 10.1182/blood-2012-09-456079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lundgrin EL, Park MM, Sharp J, Tang WH, Thomas JD, Asosingh K, Comhair SA, DiFilippo FP, Neumann DR, Davis L, Graham BB, Tuder RM, Dostanic I, Erzurum SC. Fasting 2-deoxy-2-[18F]fluoro-d-glucose positron emission tomography to detect metabolic changes in pulmonary arterial hypertension hearts over 1 year. Ann Am Thorac Soc 10: 1–9, 2013. doi: 10.1513/AnnalsATS.201206-029OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Machado MP, Rocha AM, de Oliveira LF, de Cuba MB, de Oliveira Loss I, Castellano LR, Silva MV, Machado JR, Nascentes GA, Paiva LH, Savino W, Junior VR, Brum PC, Prado VF, Prado MA, Silva EL, Montano N, Ramirez LE, Dias da Silva VJ. Autonomic nervous system modulation affects the inflammatory immune response in mice with acute Chagas disease. Exp Physiol 97: 1186–1202, 2012. doi: 10.1113/expphysiol.2012.066431. [DOI] [PubMed] [Google Scholar]
- 29.Manglik A, Kim TH, Masureel M, Altenbach C, Yang Z, Hilger D, Lerch MT, Kobilka TS, Thian FS, Hubbell WL, Prosser RS, Kobilka BK. Structural insights into the dynamic process of β2-adrenergic receptor signaling. Cell 161: 1101–1111, 2015. [Erratum in Cell 162: 1431, 2015.] doi: 10.1016/j.cell.2015.04.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Manglik A, Lin H, Aryal DK, McCorvy JD, Dengler D, Corder G, Levit A, Kling RC, Bernat V, Hübner H, Huang XP, Sassano MF, Giguère PM, Löber S, Da Duan, Scherrer G, Kobilka BK, Gmeiner P, Roth BL, Shoichet BK. Structure-based discovery of opioid analgesics with reduced side effects. Nature 537: 185–190, 2016. doi: 10.1038/nature19112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Marsboom G, Wietholt C, Haney CR, Toth PT, Ryan JJ, Morrow E, Thenappan T, Bache-Wiig P, Piao L, Paul J, Chen CT, Archer SL. Lung 18F-fluorodeoxyglucose positron emission tomography for diagnosis and monitoring of pulmonary arterial hypertension. Am J Respir Crit Care Med 185: 670–679, 2012. doi: 10.1164/rccm.201108-1562OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Martyniuk TV, Konosova ID, Chazova IE. [Use of nebivolol in patients with idiopathic pulmonary hypertension: results of the pilot study]. Ter Arkh 84: 49–53, 2012. [PubMed] [Google Scholar]
- 33.Moretti C, Grosso Marra W, D’Ascenzo F, Omedè P, Cannillo M, Libertucci D, Fusaro E, Meynet I, Giordana F, Salera D, Annone U, Chen SL, Marra S, Gaita F. Beta blocker for patients with pulmonary arterial hypertension: a single center experience. Int J Cardiol 184: 528–532, 2015. doi: 10.1016/j.ijcard.2015.02.033. [DOI] [PubMed] [Google Scholar]
- 34.Naga Prasad SV, Barak LS, Rapacciuolo A, Caron MG, Rockman HA. Agonist-dependent recruitment of phosphoinositide 3-kinase to the membrane by β-adrenergic receptor kinase 1. A role in receptor sequestration. J Biol Chem 276: 18953–18959, 2001. doi: 10.1074/jbc.M102376200. [DOI] [PubMed] [Google Scholar]
- 35.Nobles KN, Xiao K, Ahn S, Shukla AK, Lam CM, Rajagopal S, Strachan RT, Huang TY, Bressler EA, Hara MR, Shenoy SK, Gygi SP, Lefkowitz RJ. Distinct phosphorylation sites on the β2-adrenergic receptor establish a barcode that encodes differential functions of β-arrestin. Sci Signal 4: ra51, 2011. doi: 10.1126/scisignal.2001707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Rajagopal S, Ahn S, Rominger DH, Gowen-MacDonald W, Lam CM, Dewire SM, Violin JD, Lefkowitz RJ. Quantifying ligand bias at seven-transmembrane receptors. Mol Pharmacol 80: 367–377, 2011. doi: 10.1124/mol.111.072801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Rose JA, Wanner N, Cheong HI, Queisser K, Barrett P, Park M, Hite C, Naga Prasad SV, Erzurum S, Asosingh K. Flow cytometric quantification of peripheral blood cell β-adrenergic receptor density and urinary endothelial cell-derived microparticles in pulmonary arterial hypertension. PLoS One 11: e0156940, 2016. doi: 10.1371/journal.pone.0156940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Ryba DM, Li J, Cowan CL, Russell B, Wolska BM, Solaro RJ. Long-term biased β-arrestin signaling improves cardiac structure and function in dilated cardiomyopathy. Circulation 135: 1056–1070, 2017. doi: 10.1161/CIRCULATIONAHA.116.024482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Sato Y, Tsuyama T, Sato C, Karim MF, Yoshizawa T, Inoue M, Yamagata K. Hypoxia reduces HNF4α/MODY1 protein expression in pancreatic β-cells by activating AMP-activated protein kinase. J Biol Chem 292: 8716–8728, 2017. doi: 10.1074/jbc.M116.767574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Saygin D, Highland KB, Farha S, Park M, Sharp J, Roach EC, Tang WHW, Thomas JD, Erzurum SC, Neumann DR, DiFilippo FP. Metabolic and functional evaluation of the heart and lungs in pulmonary hypertension by gated 2-[18F]-fluoro-2-deoxy-d-glucose positron emission tomography. Pulm Circ 7: 428–438, 2017. doi: 10.1177/2045893217701917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Semenza GL. Hypoxia-inducible factor 1 and cardiovascular disease. Annu Rev Physiol 76: 39–56, 2014. doi: 10.1146/annurev-physiol-021113-170322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Semenza GL, Roth PH, Fang HM, Wang GL. Transcriptional regulation of genes encoding glycolytic enzymes by hypoxia-inducible factor 1. J Biol Chem 269: 23757–23763, 1994. [PubMed] [Google Scholar]
- 43.Shenoy SK, Drake MT, Nelson CD, Houtz DA, Xiao K, Madabushi S, Reiter E, Premont RT, Lichtarge O, Lefkowitz RJ. β-Arrestin-dependent, G protein-independent ERK1/2 activation by the β2 adrenergic receptor. J Biol Chem 281: 1261–1273, 2006. doi: 10.1074/jbc.M506576200. [DOI] [PubMed] [Google Scholar]
- 44.Stiles GL, Caron MG, Lefkowitz RJ. β-Adrenergic receptors: biochemical mechanisms of physiological regulation. Physiol Rev 64: 661–743, 1984. doi: 10.1152/physrev.1984.64.2.661. [DOI] [PubMed] [Google Scholar]
- 45.Stiles GL, Lefkowitz RJ. Cardiac adrenergic receptors. Annu Rev Med 35: 149–164, 1984. doi: 10.1146/annurev.me.35.020184.001053. [DOI] [PubMed] [Google Scholar]
- 46.Thenappan T, Roy SS, Duval S, Glassner-Kolmin C, Gomberg-Maitland M. β-Blocker therapy is not associated with adverse outcomes in patients with pulmonary arterial hypertension: a propensity score analysis. Circ Heart Fail 7: 903–910, 2014. doi: 10.1161/CIRCHEARTFAILURE.114.001429. [DOI] [PubMed] [Google Scholar]
- 47.Tuder RM, Chacon M, Alger L, Wang J, Taraseviciene-Stewart L, Kasahara Y, Cool CD, Bishop AE, Geraci M, Semenza GL, Yacoub M, Polak JM, Voelkel NF. Expression of angiogenesis-related molecules in plexiform lesions in severe pulmonary hypertension: evidence for a process of disordered angiogenesis. J Pathol 195: 367–374, 2001. doi: 10.1002/path.953. [DOI] [PubMed] [Google Scholar]
- 48.van Campen JS, de Boer K, van de Veerdonk MC, van der Bruggen CE, Allaart CP, Raijmakers PG, Heymans MW, Marcus JT, Harms HJ, Handoko ML, de Man FS, Vonk Noordegraaf A, Bogaard HJ. Bisoprolol in idiopathic pulmonary arterial hypertension: an explorative study. Eur Respir J 48: 787–796, 2016. doi: 10.1183/13993003.00090-2016. [DOI] [PubMed] [Google Scholar]
- 49.van der Westhuizen ET, Breton B, Christopoulos A, Bouvier M. Quantification of ligand bias for clinically relevant β2-adrenergic receptor ligands: implications for drug taxonomy. Mol Pharmacol 85: 492–509, 2014. doi: 10.1124/mol.113.088880. [DOI] [PubMed] [Google Scholar]
- 50.Violin JD, DiPilato LM, Yildirim N, Elston TC, Zhang J, Lefkowitz RJ. β2-Adrenergic receptor signaling and desensitization elucidated by quantitative modeling of real time cAMP dynamics. J Biol Chem 283: 2949–2961, 2008. doi: 10.1074/jbc.M707009200. [DOI] [PubMed] [Google Scholar]
- 51.Wisler JW, DeWire SM, Whalen EJ, Violin JD, Drake MT, Ahn S, Shenoy SK, Lefkowitz RJ. A unique mechanism of β-blocker action: carvedilol stimulates β-arrestin signaling. Proc Natl Acad Sci USA 104: 16657–16662, 2007. doi: 10.1073/pnas.0707936104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Wölfle D, Jungermann K. Long-term effects of physiological oxygen concentrations on glycolysis and gluconeogenesis in hepatocyte cultures. Eur J Biochem 151: 299–303, 1985. doi: 10.1111/j.1432-1033.1985.tb09100.x. [DOI] [PubMed] [Google Scholar]
- 53.Xie L, Xiao K, Whalen EJ, Forrester MT, Freeman RS, Fong G, Gygi SP, Lefkowitz RJ, Stamler JS. Oxygen-regulated β2-adrenergic receptor hydroxylation by EGLN3 and ubiquitylation by pVHL. Sci Signal 2: ra33, 2009. doi: 10.1126/scisignal.2000444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Xu W, Koeck T, Lara AR, Neumann D, DiFilippo FP, Koo M, Janocha AJ, Masri FA, Arroliga AC, Jennings C, Dweik RA, Tuder RM, Stuehr DJ, Erzurum SC. Alterations of cellular bioenergetics in pulmonary artery endothelial cells. Proc Natl Acad Sci USA 104: 1342–1347, 2007. doi: 10.1073/pnas.0605080104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Zarrindast MR, Ramezani-Tehrani B, Ghadimi M. Effects of adrenoceptor agonists and antagonists on morphine-induced Straub tail in mice. Pharmacol Biochem Behav 72: 203–207, 2002. doi: 10.1016/S0091-3057(01)00749-3. [DOI] [PubMed] [Google Scholar]
- 56.Zhang H, Qian DZ, Tan YS, Lee K, Gao P, Ren YR, Rey S, Hammers H, Chang D, Pili R, Dang CV, Liu JO, Semenza GL. Digoxin and other cardiac glycosides inhibit HIF-1α synthesis and block tumor growth. Proc Natl Acad Sci USA 105: 19579–19586, 2008. doi: 10.1073/pnas.0809763105. [DOI] [PMC free article] [PubMed] [Google Scholar]